Abstract
Background and Objectives
Social isolation is a risk factor for cognitive decline and dementia. We conducted a randomized controlled clinical trial (RCT) of enhanced social interactions, hypothesizing that conversational interactions can stimulate brain functions among socially isolated older adults without dementia. We report topline results of this multisite RCT (Internet-based conversational engagement clinical trial [I-CONECT]; NCT02871921).
Research Design and Methods
The experimental group received cognitively stimulating semistructured conversations with trained interviewers via internet/webcam 4 times per week for 6 months (induction) and twice per week for an additional 6 months (maintenance). The experimental and control groups both received weekly 10 minutes telephone check-ins. Protocol modifications were required due to the coronavirus disease 2019 pandemic.
Results
A total of 186 participants were randomized. After the induction period, the experimental group had higher global cognitive test scores (Montreal Cognitive Assessment [primary outcome]; 1.75 points [p = .03]) compared with the control group. After induction, experimental group participants with normal cognition had higher language-based executive function (semantic fluency test [secondary outcome]; 2.56 points [p = .03]). At the end of the maintenance period, the experimental group of mild cognitive impairment subjects had higher encoding function (Craft Story immediate recall test [secondary outcome]; 2.19 points [p = .04]). Measure of emotional well-being improved in both control and experimental groups. Resting-state functional magnetic resonance imaging showed that the experimental group had increased connectivity within the dorsal attention network relative to the control group (p = .02), but the sample size was limited.
Discussion and Implications
Providing frequent stimulating conversational interactions via the internet could be an effective home-based dementia risk-reduction strategy against social isolation and cognitive decline.
Clinical Trials Registration Number
Keywords: Alzheimer’s disease and related dementias, Behavioral intervention, Cognitive reserve, Semistructured conversations, Social interactions
Epidemiological evidence indicates that social isolation (small social networks, lack of social contact, and support) is a risk factor for cognitive decline and dementia (Evans et al., 2019; Kallianpur et al., 2023; National Academies of Sciences, Engineering, and Medicine, 2020; Penninkilampi et al., 2018; Poey et al., 2017; Shen et al., 2022). The Lancet Commission on Dementia Prevention suggested that 4% of incident dementia could be prevented by reducing social isolation, higher than 2% and 1% reductions estimated, respectively, for reducing physical inactivity and diabetes (Livingston et al., 2020). There may be a significant public health impact in developing interventions that mitigate the negative impacts of social isolation on cognitive functions. These interventions need to be readily scalable for efficient community health care systems implementation (National Academies of Sciences, Engineering, and Medicine, 2020). Developing such interventions, however, is challenging, partly because social isolation involves multiple components potentially affecting cognition. Another obstacle to developing effecting interventions is that the biological mechanisms underlying the association between social isolation and declining cognition are poorly understood.
In epidemiological studies, for example, examination of social activities as potential protective factors against incident dementia or Alzheimer’s disease (Bennett et al., 2006; Evans et al., 2019; Fratiglioni et al., 2004; Penninkilampi et al., 2018; Poey et al., 2017) was mostly based on social engagement without standardized definition of social activities. Some activities not typically categorized as social activities, such as running and walking (generally considered as physical activities, Dodge et al., 2008) or playing games (considered as cognitive activities, Klaming et al., 2017; Park et al., 2007), may be social activities. The umbrella use of social engagement in observational studies makes it difficult to pinpoint crucial aspects of social engagement that might mitigate cognitive decline.
Additionally, social isolation (or social connectedness) might affect cognition in several ways. Larger social networks might provide tangible support (e.g., care and transportation for medical treatments) and/or intangible support (e.g., emotional support with resulting diminished anxiety; information and encouragement for healthy lifestyles; Amieva et al., 2010; Ashida & Heaney, 2008; Kelly et al., 2017). Perry et al. (2021) differentiated “social bonding” and “social bridging” to characterize two types of social connectedness. The former represents family or close friends’ networks with a sense of belonging and emotional bonding within small social units, whereas the latter represents a larger and broader network with peripheral, irregular, or heterogeneous ties not necessarily generating emotional bonding. Perry et al. (2021) showed that the latter is associated with higher cognitive function, implying that cognitive stimulation stemming from interactions with various social groups is more important for maintaining cognitive function than emotional bonding. This conceptually important study was based on cross-sectional data, potentially confounded by reverse causation. Whether increasing peripheral social interactions that enhance social bridging can improve cognitive functions is unexamined.
Multidomain interventions aiming to examine potentially synergistic effects of modifying multiple lifestyle factors gained popularity in recent years (e.g., Andrieu et al., 2017; Kivipelto et al., 2020). However, focusing on single elements of social engagement is critical to clarifying mechanisms of protective effects of social connectedness on cognitive functions. Single-component approaches may also provide improved scalability and ease of implementation.
We conducted a series of randomized controlled trials (RCTs), hypothesizing that increasing social interactions, specifically conversational interactions—a single crucial component of social interactions—can enhance cognitive functions. Following encouraging results from a pilot study (Cerino et al., 2020; Dodge et al., 2015), we conducted a Phase II study (Internet-Based Conversational Engagement Clinical Trial [I-CONECT]; www.i-conect.org; ClinicalTrials.gov: NCT02871921). The study protocol was published previously (Yu et al., 2021).
The primary aim of the I-CONECT was to examine whether frequent and stimulating conversational interactions could lead to improved cognitive functions and whether the effect might be mediated by improved emotional well-being. We hypothesized enhanced compensatory neural activities, that is, improved cognitive reserve or scaffolding (Park & Bischof, 2013; Stern, 2012), as an underlying biological mechanism for the efficacy. The secondary aim was to assess whether the improved cognitive test scores could be translated into improved functional outcomes.
Here, we report the topline results of I-CONECT, presenting the results for the primary aim. The study outcomes presented include changes from baseline to the month 6 (M6) and 12 (M12) assessments in:
(1) Primary outcome: global cognitive function measured by the Montreal Cognitive Assessment (MoCA; Nasreddine et al., 2005).
(2) Secondary outcomes: episodic memory measured by the Craft Story Immediate and Delayed Recall tests (Craft et al., 1996), and language-based executive cognitive functions measured by the semantic fluency test (Category Fluency Animals; Lezak et al., 2012).
(3) Variables for a potential mediator and biological mechanism: emotional well-being measured by the NIH Toolbox Emotion Battery (NIHTB-EB) (Babakhanyan et al., 2018; a mediator) and resting-state functional MRI (rsfMRI)-derived connectivity of four large cognitive networks (a biological mechanism) including dorsal attention network (DAN), default mode network (DMN), salience network (SN), and executive control network (ECN).
Analyses were stratified by cognitive status (mild cognitive impairment [MCI] vs normal) based on differential efficacy found in the prior pilot study (Dodge et al., 2015).
I-CONECT was designed with several features to isolate the impact of social interactions from other potential components. First, to increase interaction novelty and to reduce the confounding effects of emotional bonding, we rotated conversation partners assigned to each participant. Second, we deployed user-friendly devices allowing participants without any internet/webcam experience to rapidly engage in video-based conversations. This eliminated cognitive stimulation secondary to learning device use. Third, we used semistructured conversations with standardized daily themes and picture prompts, facilitating participants’ organizing and articulating their thoughts to conversation partners (Radecki et al., 2019; Spreng et al., 2020).
We targeted this Phase II RCT to the socially isolated older old because (a) they have a higher risk of cognitive decline yet are seldom included in clinical trials; (2) we hypothesized that subjects with social isolation are more likely to benefit from this intervention than those without isolation as these activities are novel to them (Park & Bischof, 2013). To make the RCT feasible for older-old subjects, we used conversations that can be initiated with little motivation, are likely to have high adherence, are sustainable, and can reach the homebound through internet/webcam.
Method
Participants
I-CONECT is a multisite Phase II intervention study. Participants with normal cognition (NC) or MCI were recruited from Portland, OR, and Detroit, MI. Participants were required to provide written informed consent. Study approval was obtained from the Institutional Review Board at Oregon Health & Science University (IRB STUDY00015937) using a single IRB process. This report adheres to CONSORT standards.
Inclusion/exclusion criteria
Key inclusion criteria included: (1) age 75 or older, (2) consent to receive magnetic resonance imaging (MRI; if safely and comfortably able to receive MRI), (3) socially isolated, defined by at least one of the following: (i) score ≤12 on the six-item Lubben Social Network Scale (LSNS-6; Lubben et al., 2006), (ii) engages in conversations lasting 30 min or longer no more than twice per week, per subject self-report, (iii) answers “Often” to at least one question on the Three-Item UCLA Loneliness Scale (Hughes et al., 2004). Key exclusion criteria included (1) dementia and (2) moderate-to-severe depressive symptoms, operationally defined as a 15-item Geriatric Depression Scale (GDS-15) score >7 (Yesavage et al., 1982). Additional criteria and recruitment sources are listed in Supplementary Table 1 (Yu et al., 2021).
Cognitive assessment and clinical diagnosis of normal versus MCI
A thorough cognitive assessment with validated neuropsychological tests was administered at baseline, M6 (endpoint of induction) and M12 (endpoint of maintenance), using the National Alzheimer’s Coordinating Center (NACC; https://naccdata.org/) Uniform Data Set version 3 (UDS V3; Dodge et al., 2020; Sachs et al., 2020; Weintraub et al., 2018). The cognitive test battery, detailed screening, and diagnostic procedure were described previously (Yu et al., 2021) and included in Supplementary Table 1. A consensus meeting was held among the study neurologists and neuropsychologists to clinically determine each potential participant’s cognitive status grouping (NC, MCI, or dementia) based on a standardized approach used in National Institute of Health (NIH)-funded Alzheimer’s Disease Centers across the United States (Albert et al., 2011; Jack et al., 2011; McKhann et al., 2011). Those determined to have dementia were excluded.
Randomization
Randomization was conducted by an independent data manager. Participants were randomized in a 1:1 ratio to the experimental or control groups balancing age, sex, years of formal education, and cognitive status (MCI/NC) and MoCA score. A minimization algorithm was used for randomization (Schouten, 1995).
Masking
Experienced, trained research associates conducted baseline, M6, and M12 follow-up assessments. Assessors were not allowed to discuss any issues with participants revealing whether the subject was in an experimental or control group.
Intervention Approach
The experimental group engaged in 30-min semistructured conversations with trained staff 4 days/week for 6 months (induction), followed by twice per week for an additional 6 months (maintenance). Both intervention and control groups received a phone call once per week (approximately 10-min duration) to assess changes in health and social activities. This weekly phone call also served to retain control group participation. Conversation sessions (audio and video) and weekly calls (audio) were recorded and stored in a HIPAA-compliant archive. Recruitment was conducted between July 2018 and December 2020.
Standardization of interviewer skills
Several standardization strategies were used and reported earlier including monitoring word counts generated by the interviewer vs. participant, and periodic assessments of changes in negative and positive effects of participants using the International Positive and Negative Affect Schedule Short Form (PANAS) (Crawford & Henry, 2004; Watson et al., 1988), immediately before and after the 30-minute conversation across interviewers.. ( Supplementary Table 1).
Conversational materials
Conversations were semistructured with prespecified topics and crafted questions to promote natural conversations. Over 150 themes were developed, with topics including historical events, philosophical ideas, social issues, leisure activities, and travels. For every session, conversational staff presented three subtopics under one theme. For example, under a theme “Defining moments of the 20th century,” participants could select from three topics, such as the Great Depression, the JFK Assassination, and the Vietnam War (see Supplementary Appendix A for sample PowerPoint slides of the picture and prompts). By allowing participants to select a topic, we aimed to enhance a sense of autonomy and increase motivation to participate in conversations.
Outcomes
Primary outcome
Change in global cognitive function measured by the MoCA from baseline to M6.
Secondary outcomes
Changes in language-based executive function measured by semantic fluency (Category Fluency Animals test), and episodic memory measured by Craft Story immediate (learning/encoding) and delayed recall (episodic memory) scores from the NACC UDS V3 (see Supplementary Table 1).
Potential mediator variable
Changes in psychological well-being
This is measured by the NIH-Toolbox emotion battery (NIHTB-EB; Babakhanyan et al., 2018). NIHTB-EB includes four general domains: negative affect (anger, fear, and sadness), psychological well-being (positive affect, general life satisfaction, and meaning and purpose), stress and self-efficacy (perceived stress and self-efficacy), and social relationships (social support, companionship, and social distress). Three domain scores, negative affect, social satisfaction, and psychological well-being, were calculated with 17 subscale scores weighted by factor loading (Babakhanyan et al., 2018; Yu et al., 2022).
Biological mechanism
Resting-state fMRI
To explore underlying mechanisms of trial efficacy, we examined pre- and post-trial changes in brain network intrinsic functional connectivity (iFC) measured by rsfMRI. We specifically focused on four large-scale brain networks supporting a broad array of cognitive processes including social function (Feng et al., 2021): DAN, DMN, SN, and ECN. See Supplementary Table 2 for the fMRI protocol.
Post-COVID Protocol Modification
Recruitment sites issued the first stay-at-home order for the COVID-19 pandemic in March 2020. Our internet interventions and video chats/phone check-ins were able to continue without interruption during the pandemic. However, as with other clinical trials, new recruitment and post-trial assessments, including MRI, were halted after the order. Due to the potential high risk of COVID-19 infection among our participants (aged 75+), protocol modifications were necessary.
Prepandemic cohort analyses versus ITT analyses
We modified our assessment protocol from in-person to telephone cognitive testing (T-COG) after the adoption of T-COG by NACC. T-COG includes the modified MoCA without visual items. This modality change was expected to affect cognitive test scores. Additionally, we were concerned that the COVID-19 pandemic might amplify trial efficacy due to heightened social isolation experienced by the control group, limiting the generalizability of study results. Because of these concerns, the research group and the Data Safety Monitoring Board (DSMB) agreed to limit analysis of the primary outcome to participants completing the M6 assessment before March 18, 2020, the date of the stay-home orders issue. We call this analysis “prepandemic cohort analysis.”
To control for pandemic-related effects on the outcomes, the DSMB suggested that for secondary and exploratory outcomes, those who enrolled after March 18, 2020, or those whose assessments used different modalities across baseline, M6, and M12 assessments, analysis be conducted by including a covariate indicating pre- versus post-COVID-19 pandemic times. This latter analysis was limited to secondary and exploratory outcomes as MoCA (primary outcome) differed in total scores between in-person and telephone MoCA, while Semantic Fluency and Craft Story tests (secondary outcomes) can be reasonably assumed to be similar between the two modalities.
Additional modifications after March 18, 2020
These included (1) MRI assessments were discontinued. (2) NIHTB-EB was collected by mailing hard copies with prepaid return envelopes. (3) Due to staff retention costs during the pandemic, when no new recruitment and endpoint assessments occurred, and difficulty in conducting in-home assessments even after the full research hiatus ended, it was necessary to prioritize completing M6 assessments for all existing participants, but not possible to complete M12 assessments scheduled after June 15, 2020. (4) Subjects were required to provide their own internet connection to participate in the trial during the pandemic, as we could not safely have vendors enter homes to install new connections. An internet stipend was provided. (5) Sample size: we aimed to conduct the analyses separately by ancestry, aiming to randomize 160 White and 160 Black participants. With a 10% drop-out between each assessment, we projected 144 and 128 completers at M6 and M12, respectively. With 50% MCI (targeted MCI/NC: 72/72 at M6, 64/64 at M12), these sample sizes gave 80% power to detect Cohen’s d of 0.67 at M6 and 0.71 at M12 at α = 0.05 (two-tailed) within each ancestral group. Due to the low participation of Black participants (discussed later), we were unable to conduct ancestry-stratified analyses.
Statistical Analyses
For the primary outcome, we conducted a modified intention-to-treat (ITT) analysis using the prepandemic cohort, including those with MoCA assessed in-person at their homes at baseline and at M6 assessments. A separate linear regression model was run by cognitive status (MCI/NC) with M6 MoCA scores as the dependent variable, a randomization group (control vs experimental) being a dependent variable, controlling for MoCA baseline scores. For the larger analyses, where all randomized subjects were included for the secondary outcomes, we conducted ITT analyses combining in-home and remote-assessed cognitive tests. We used a mixed-effects model repeated measure (MMRM). The pre- versus post-COVID-19 pandemic indicator was included as a control variable. R and SAS9.4 were used for the analyses.
fMRI analyses (exploratory outcome)
To assess differences between experimental and control groups in network iFC, we tested separate ANCOVA models for each of four networks using JASP (JASP Team, 2021), with network connectivity at M6 as the dependent variable. We controlled for age, in-scanner head motion, and network connectivity at baseline. As network iFC is an exploratory outcome measure, we used an alpha-threshold of 0.05, without correcting for the number of models.
Results
Baseline Sample Characteristics
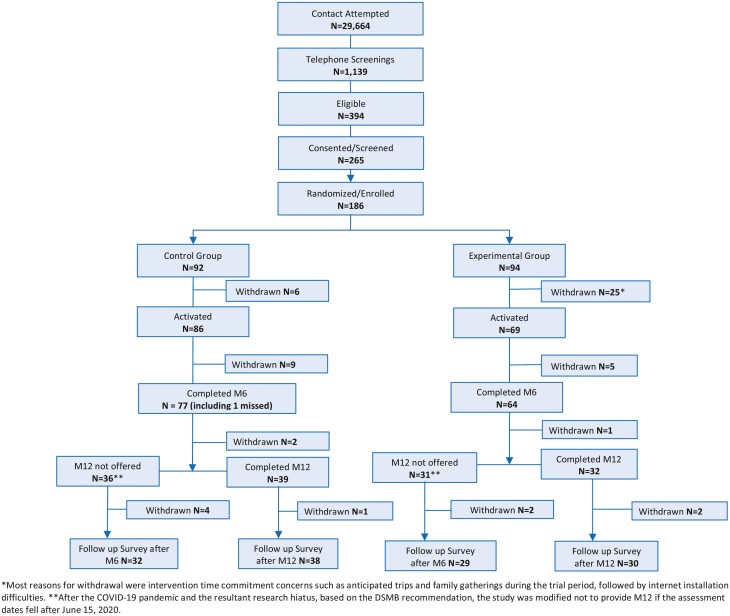
One hundred eighty-six participants (86 NC and 100 (52.8%) MCI) were randomized into the experimental (n = 94) or the control group (n = 92; Figure 1). Although we achieved 93% of the target sample size (149 out of 160 targeted) for White participants despite the pandemic, we could recruit only 23% of the target (37 out of 160) for Black participants. The majority of dropouts occurred before the start of the weekly call and intervention; 31 participants withdrew (25 experimental; 6 control) mainly due to unanticipated conflict in time commitments during the 1-year trial period. After starting the weekly call and intervention, an additional 14 participants withdrew (5 experimental; 9 control). In total, 141 participants remained in the M6 follow-up (9.9% withdrawal between baseline and M6). One participant skipped the M6 assessment, but remained in the study and participated in the M12 assessment. Between M6 and M12 follow-up, 3 participants withdrew (1 experimental; 2 control). Sixty-seven participants (31 experimental; 36 control), with M12 assessments scheduled after June 15, 2020, were not invited for the M12 assessments and were informed of protocol modifications (see earlier). Those completing M6 assessments and noncompleters did not differ in baseline characteristics, including cognitive test scores. Demographic characteristics and cognitive test scores at M6 did not differ between participants whose M12 assessments were discontinued after the pandemic and those completing M12 assessments.
Figure 1.
Study recruitment and retention flow chart. COVID-19 = coronavirus disease 2019; DSMB = Data Safety Monitoring Board; M6 = Month 6 Assessment; M12 = Month 12 Assessment.
Table 1 presents baseline sample characteristics of randomized participants by cognitive status (MCI/NC) and group assignment (experimental/control groups). No demographic variables or cognitive test scores differed by randomization group, consistent with successful randomization.
Table 1.
Baseline NC/MCI & Experiment/Control (n = 186)
| Characteristics [n (%)] | NC (N = 86) | t-Statistics/ χ2-statistics/ Fisher’s test (df) | p Value | MCI (N = 100) | t-Statistics/ χ2-statistics/ Fisher’s test (df) | p Value | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Experiment | Control | Experiment | Control | |||||||||
| 43 | (50.0) | 43 | (50.0) | 51 | (51.0) | 49 | (49.0) | |||||
| Age [mean (SD)] | 79.6 | (4.0) | 80.0 | (3.7) | t (84) = −0.46 | .65 | 81.9 | (4.9) | 82.6 | (4.9) | t (98) = −0.70 | .49 |
| Gender (female) [n (%)] | 30 | (69.8) | 35 | (81.4) | χ2(1) = 1.58 | .21 | 34 | (66.7) | 31 | (63.3) | χ2(1) = 0.13 | .72 |
| Race (White) [n (%)] | 37 | (86.1) | 36 | (83.7) | χ2(1) = 0.09 | .76 | 37 | (72.6) | 39 | (79.6) | χ2(1) = 0.68 | .41 |
| Years of education [mean (SD)] | 15.5 | (2.6) | 15.1 | (2.2) | t (84) = 0.63 | .53 | 15.5 | (2.5) | 14.8 | (1.9) | t (98) = 1.52 | .13 |
| Lubben’s isolation score [mean (SD)] | 14.0 | (5.4) | 13.6 | (5.7) | t (84) = 0.33 | .74 | 12.8 | (4.8) | 12.6 | (4.4) | t (98) = 0.28 | .78 |
| Cognitive test [mean (SD)] | ||||||||||||
| MoCA (n = 146)* | 25.1 | (3.0) | 25.7 | (2.8) | t (62) = −0.82 | .41 | 22.3 | (3.1) | 21.8 | (3.6) | t (80) = 0.69 | .49 |
| Blind MoCA (n = 40)* | 19.6 | (1.7) | 19.4 | (1.6) | t (20) = 0.38 | .71 | 17.2 | (1.8) | 15.8 | (1.8) | t (16) = 1.71 | .11 |
| Category fluency animals | 20.3 | (4.8) | 19.7 | (3.8) | t (84) = 0.74 | .46 | 17.5 | (4.3) | 16.8 | (4.4) | t (98) = 0.77 | .44 |
| Paraphrase composite | 16.9 | (3.3) | 16.7 | (2.8) | t (84) = 0.19 | .85 | 13.2 | (3.9) | 13.0 | (4.2) | t (98) = 0.23 | .82 |
| Craft Story immediate recall (Verbatim) | 23.1 | (6.3) | 22.2 | (5.5) | t (84) = 0.70 | .49 | 18.9 | (5.9) | 17.7 | (6.7) | t (98) = 0.93 | .36 |
| Craft Story immediate recall (Paraphrase) | 17.3 | (3.2) | 17.1 | (3.0) | t (84) = 0.31 | .76 | 14.1 | (4.0) | 13.8 | (4.2) | t (98) = 0.34 | .73 |
| Immediate recall composite | 20.2 | (4.6) | 19.7 | (4.1) | t (84) = 0.58 | .56 | 16.5 | (4.8) | 15.7 | (5.3) | t (98) = 0.71 | .48 |
| Craft Story Delayed recall (Verbatim) | 20.1 | (5.8) | 18.8 | (4.8) | t (84) = 1.10 | .27 | 14.5 | (5.7) | 13.6 | (6.3) | t (98) = 0.76 | .45 |
| Craft Story Delayed recall (Paraphrase) | 16.4 | (3.6) | 16.4 | (2.9) | t (84) = 0.07 | .95 | 12.3 | (4.2) | 12.2 | (4.5) | t (98) = 0.11 | .92 |
| Delayed recall composite | 18.3 | (4.5) | 17.6 | (3.6) | t (84) = 0.74 | .46 | 13.4 | (4.7) | 12.9 | (5.3) | t (98) = 0.50 | .61 |
| Copy of Benson figure (n = 146)a | 16.0 | (0.7) | 15.6 | (0.9) | t (84) = 1.88 | .06 | 15.3 | (1.3) | 15.1 | (1.5) | t (98) = 0.69 | .49 |
| Number span forward Correct | 8.0 | (2.3) | 8.2 | (2.4) | t (84) = −0.41 | .68 | 6.7 | (2.0) | 7.2 | (2.0) | t (98) = −1.24 | .22 |
| Number span forward longest span | 6.5 | (1.2) | 6.7 | (1.4) | t (84) = −0.50 | .62 | 5.9 | (1.2) | 6.1 | (1.0) | t (98) = −0.99 | .32 |
| Number span backward Correct | 7.1 | (2.1) | 7.1 | (2.1) | t (84) = 0.00 | 1.00 | 5.9 | (1.9) | 6.1 | (1.9) | t (98) = −0.69 | .49 |
| Number span backward longest span | 5.0 | (1.1) | 5.1 | (1.1) | t (84) = −0.29 | .77 | 4.4 | (1.1) | 4.4 | (1.1) | t (98) = −0.35 | .73 |
| Category fluency vegetables | 15.2 | (4.2) | 14.9 | (3.0) | t (84) = 0.47 | .64 | 12.5 | (3.9) | 10.7 | (3.7) | t (98) = 2.29 | .02 |
| Delayed drawing of Benson figure | 11.0 | (2.8) | 11.2 | (2.4) | t (62) = −0.34 | .74 | 8.7 | (2.6) | 8.9 | (3.4) | t (80) = −0.35 | .73 |
| Number of correct F words | 14.8 | (4.1) | 15.3 | (4.1) | t (84) = −0.66 | .51 | 11.4 | (3.9) | 10.6 | (3.9) | t (98) = 0.96 | .34 |
| Number of correct L words | 13.9 | (3.7) | 14.3 | (3.8) | t (84) = −0.52 | .60 | 10.5 | (4.0) | 10.4 | (3.8) | t (98) = −0.19 | .85 |
| Multilingual naming test (n = 146)a | 30.4 | (1.4) | 30.3 | (1.7) | t (62) = 0.24 | .81 | 29.1 | (2.2) | 28.7 | (2.8) | t (80) = 0.79 | .43 |
| Trail Making A (lines per second; n = 146)a | 0.72 | (0.2) | 0.71 | (0.2) | t (62) = 0.12 | .91 | 0.5 | (0.2) | 0.5 | (0.2) | t (80) = −0.50 | .62 |
| Trial Making B (lines per second; n = 146)a | 0.29 | (0.1) | 0.27 | (0.1) | t (61) = 0.80 | .42 | 0.2 | (0.1) | 0.2 | (0.1) | t (78) = 0.13 | .90 |
Notes: COVID-19 = coronavirus disease 2019; df = degree of freedom; MCI = participants with mild cognitive impairment; MoCA = Montreal Cognitive Assessment. MoCA was replaced with Blind MoCA. NC = participants with normal cognition; SD = standard deviation.
aAfter COVID-19 pandemic, in-home assessment was replaced with telephone assessment and tests which require visional input were dropped from the battery.
Intervention Efficacy
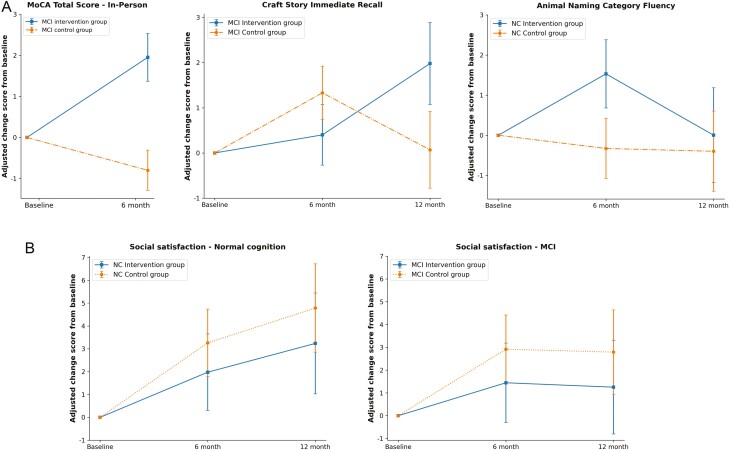
The primary outcome analysis was based on the pre-COVID cohort. Among those with MCI (n = 31), the intervention group’s mean MoCA score was 1.75 points higher than that of the control group at M6 (p = .03, Cohen’s d = 0.73). To assess the clinically meaningful implication of this difference, we ran a linear regression model with all randomized participants at baseline and examined the association between age and MoCA total scores, controlling for MCI status. The coefficient for age was −0.16 (standard error [SE] = 0.056, p = .005) and MCI was −2.91 (SE = 0.531, p < .001), implying that 1.75 points are approximately equivalent to 10 years of age difference (i.e., 1.75/0.16 = 10.93) in MoCA scores in this cohort. No group difference was found in those with NC (n = 25; Table 2A).
Table 2.
Primary and Secondary Outcomes
| Outcomes | NC | MCI | ||||
|---|---|---|---|---|---|---|
| A.Primary outcome—treatment effect at 6 months using those who completed in-home assessment before COVID-19 pandemic (prepandemic cohort) based on linear regression model under modified ITTa | NC (n = 25) Con/Exp = 15/10 |
MCI (n = 31) Con/Exp = 18/13 |
||||
| Estimate | SE | p Value | Estimate | SE | p Value | |
| Total MoCA score (reference: control group). | 0.14 | 0.86 | .87 | 1.75 | .76 | 0.03 |
| Sensitivity analysis based on multiple imputations | NC (N = 66) Con/Exp = 35/31 |
MCI (N = 74) Con/Exp = 41/33 |
||||
|---|---|---|---|---|---|---|
| Total MoCA Score (imputed missed items in telephone MoCA) (reference: control group) | 0.70 | 0.47 | .14 | 1.17 | .56 | 0.03 |
| B.Secondary outcomes—ITT using all randomized participants, based on MMRMb | NC (n = 86) Con/Exp = 43/43 |
MCI (n = 100) Con/Exp = 49/51 |
||||
|---|---|---|---|---|---|---|
| Estimate | SE | p Value | Estimate | SE | p Value | |
| Category fluency (Animal naming) | ||||||
| COVID-19 | 0.15 | 0.71 | .83 | 0.19 | 0.63 | .44 |
| Group × 6 months | 2.56 | 1.19 | .03 | 0.69 | 0.93 | .46 |
| Group × 12 months | 1.10 | 1.29 | .40 | 1.14 | 1.31 | .39 |
| Craft Story (Immediate recall) (Paraphrase scoring) | ||||||
| COVID-19 | -0.47 | 0.50 | .36 | -1.05 | 0.54 | .05 |
| Group × 6 months | -0.63 | 0.82 | .45 | -0.66 | 0.80 | .41 |
| Group × 12 months | 0.62 | 1.02 | .55 | 2.19 | 1.05 | .04 |
| Craft Story (Delayed recall) (Paraphrase scoring) | ||||||
| COVID-19 | -0.84 | 0.52 | .11 | -1.07 | 0.55 | .06 |
| Group × 6 months | -0.26 | 0.77 | .74 | -0.10 | 0.79 | .90 |
| Group × 12 months | 0.10 | 1.00 | .92 | 1.64 | 1.10 | .14 |
Notes: Con = Control Group; Exp = Experimental Group; COVID = coronavirus disease; COVID-19 = coronavirus disease 2019; MCI = participants with mild cognitive impairment; NC = participants with normal cognition; MoCA = Montreal Cognitive Assessment; SE = standard error. The results with p-value ≤ 0.05 were bolded.
aITT: intention to treat. Each outcome was modeled separately. The full model included baseline cognitive test score and the group allocation.
bMMRM: Mixed Model for Repeated Measures. Each outcome was modeled separately. Group: experimental group (control group as a reference). The full model included cognitive status (MCI vs normal), a COVID pandemic indicator (pre- vs post-COVID outbreak), group, time, and group by time interaction. For secondary outcomes (B), multiple comparison adjusted p value is .016.
Sensitivity analysis
We conducted a sensitivity analysis with multiple imputations in which we imputed missing items in the Blind MoCA (visuospatial items, total of 8 points) based on the associations between the visuospatial items and other cognitive and demographic variables observed among those with the full MoCA. We applied the same regression models used in the prepandemic cohort analyses to all participants with M6 assessments (N = 140). Among MCI participants, the MoCA at M6 was 1.17 points higher among the experimental group, compared with the control group (p = .03, Cohen’s d = 0.70; Table 2A). There was no significant difference among those with NC. The detailed multiple imputation approach is in Supplementary Table 4.
Secondary outcome analyses were performed with all available data for 186 participants (ITT cohort) at baseline, M6, and M12 using MMRM. The MMRM models included baseline cognitive measure, a pre- versus post-COVID-19 pandemic indicator, group assignment, assessment time points (M6 and M12), and group × time interaction term. For participants with MCI (N = 100), the intervention group had higher Craft Story immediate recall scores by 2.19 points than the control group at M12 (β = 2.19, p = .04). Among those with NC (N = 86), the intervention group had better semantic fluency score than the control group at M6 by over 2 points (β = 2.56, p = .03; Table 2B). The post-COVID pandemic indicator showed a negative effect on Craft Story immediate recall (β = −1.05, p = .05) among the MCI, but not among those with NC.
NIHTB-EB Outcomes
The analysis included 134 participants with Toolbox emotional battery data at both baseline and M6 (64 NC and 70 MCI). The sample size was lower than the number of randomized participants because some did not return the mailed hard copy battery after in-person assessments were halted. Those completing the battery and those with missing data did not differ in age, sex, and education at baseline.
Both intervention and control groups had increased social satisfaction scores at M6 and M12 (Table 3 and Figure 2B), but there was no difference between groups (experimental/control). The COVID-19 pandemic negatively affected the social satisfaction of both groups among MCI (β = −4.88, p = .03), but did not affect participants with NC.
Table 3.
Mixed Model for Repeated Measures (MMRM) Results for NIH-Toolbox Emotional Battery (NIHTB-EB)
| Variables | NC, N = 64 Con/Exp = 34/30 |
MCI, N = 70 Con/Exp = 39/31 |
||||
|---|---|---|---|---|---|---|
| Coef. | SE | p Value | Coef. | SE | p Value | |
| Negative affect | ||||||
| Time—ref: baseline | ||||||
| 6 months | −2.627 | 1.409 | .062 | −0.835 | 1.080 | .440 |
| 12 months | 0.009 | 1.715 | .996 | 1.440 | 1.301 | .268 |
| Group—ref: NC | −3.618 | 2.271 | .111 | −2.210 | 2.221 | .320 |
| Time × Group | ||||||
| Group × 6 months | 1.804 | 2.135 | .398 | 0.338 | 1.668 | .839 |
| Group × 12 months | 0.445 | 2.626 | .865 | −0.707 | 1.880 | .707 |
| Post-COVID indicator | −0.200 | 1.984 | .920 | 2.973 | 2.199 | .176 |
| Social Satisfaction | ||||||
| Time—ref: baseline | ||||||
| 6 months | 3.262** | 1.136 | .004 | 2.918** | 1.065 | .006 |
| 12 months | 4.785*** | 1.088 | .000 | 2.790* | 1.281 | .029 |
| Group—ref: NC | 1.969 | 2.569 | .444 | 0.764 | 2.386 | .749 |
| Time × Group | ||||||
| Group × 6 months | −1.286 | 1.686 | .446 | −1.474 | 1.644 | .370 |
| Group × 6 months | −1.548 | 1.647 | .347 | −1.538 | 1.891 | .416 |
| Post-COVID indicator | 2.896 | 2.236 | .195 | -4.879* | 2.356 | .038 |
| Psychological well-being | ||||||
| Time—ref: baseline | ||||||
| 6 months | 1.788 | 1.298 | .168 | −0.166 | 0.964 | .863 |
| 12 months | 0.802 | 0.876 | .360 | −0.250 | 1.300 | .847 |
| Group—ref: NC | 3.347 | 2.270 | .140 | −1.203 | 2.284 | .598 |
| Time X Group | ||||||
| Group × 6 months | −0.027 | 1.996 | .989 | 0.201 | 1.460 | .890 |
| Group × 6 months | 0.820 | 1.304 | .529 | 0.499 | 1.936 | .797 |
| Post-COVID indicator | 1.795 | 2.200 | .415 | 0.083 | 2.186 | .970 |
Notes: Coef = coefficient; Con = control group; COVID = coronavirus disease; Exp = experimental group; Group = experimental group (control group as a reference); MCI = participants with mild cognitive impairment; NC = participants with normal cognition; SE = standard error. All models controlled for age, sex, year of education, and a COVID pandemic indicator (pre- vs post- COVID outbreak). P-value ≤ 0.05 was bolded. The coefficients for age, sex and education were not shown here. Full model information available upon request.
*p < .05. **p < .01. *** p < .001.
Figure 2.
Changes in primary and secondary outcome measures. MCI: mild cognitive impairment. NC: normal cognition. Solid? line: intervention group. Dotted line: control group. (A) Cognitive outcomes. (B) Emotional outcomes (NIH-toolbox emotion batteries): Social satisfaction. Both intervention and control groups improved social satisfaction at M6 and M12 compared with the baseline (p < .01).
fMRI Results
Among those with MRI at baseline (n = 50), 15 subjects completed M6 MRIs and were included in this analysis. Due to the small sample size (less than 10 participants for each group), we combined both cognitive groups (NC = 6, MCI = 9) for this exploratory outcome. Participants in the experimental group (n = 6) showed increased connectivity within the DAN at M6 relative to the control group (F = 7.28, p = .02), controlling for age, in-scanner head motion, and DAN connectivity at baseline. We did not find differences in network connectivity between the randomization groups in DMN (F = 0.005, p = .945), ECN (F < 0.001, p = .977), or SN (F = 0.300, p = .597).
Project-Related Adverse Events and Acceptability of the Intervention
Tiredness was reported by one participant during the screening assessment and by one participant after one of the video chat sessions. One of the exploratory outcomes examined whether the intervention showed efficacy on medication adherence (results not reported here). Participants were provided with low-dose (250 mg) vitamin C tablets to be taken daily. Two participants experienced stomach pain after taking the vitamin C pill, assessed as project-related adverse events. No other intervention-related adverse events were observed.
Adherence was high. Ninety-one percent of the experimental group completed over 80% of the total video chat sessions scheduled to occur between baseline and M6; out of a total potential of 96 sessions per participant for the first 6 months of the intervention, the mean number of completed sessions was 83.1 (standard deviation [SD] = 10.3). Eighty-four percent of the experimental group also completed over 80% of the total video chat sessions scheduled for the second half of the intervention (M6–M12); Out of a total potential of 46 M6–12 sessions for the experimental group, the mean of completed sessions was 43.7 (SD = 4.6). Within the control group, 91% and 95% completed over 80% of 24 weekly calls between baseline and M6 (mean 22.4; SD = 1.0) and M6 and M12 (mean 23.0; SD = 1.8), respectively.
At the end of the intervention, many of the participants requested continued conversational interactions with the interviewers. We created a nonprofit organization providing conversational engagements using the same materials used in the trial, supported by volunteer interviewers (https://www.iconnectfoundation.org/). Participation in this service is free of charge and open to any individuals aged 60 and older desiring conversational engagement.
Discussion
I-CONECT provided up to 1 year of conversational interactions via a user-friendly video chat platform. Despite a reduced sample size partly due to the COVID-19 pandemic, we found evidence of efficacy. Among participants with MCI, global cognition as measured by MoCA was significantly higher at M6 by 1.75 points (Cohen’s d = 0.73) in the experimental group compared with the control group. Sensitivity analyses with imputed full MoCA scores showed similar outcomes. 1.75 points is approximately the difference produced by a decade of age difference in MoCA scores in this cohort.
For the secondary outcomes, semantic fluency (language-based executive functions measured by category fluency test) and story immediate and delayed recall (learning/encoding and episodic memory), we combined in-home and telephone-assessed cognitive test scores due to the similarity of the assessment modalities (telephone vs in person) for these tests. We used all randomized subjects under ITT, controlling for pandemic impact. This post-COVID DSMB-specified analysis showed higher semantic fluency scores at M6 among those with normal cognition, and story immediate recall scores at M12 among those with MCI. The magnitude of effects among those with normal cognition in semantic fluency scores is similar to the results of our Phase I study (Dodge et al., 2015), confirming the reproducibility of the earlier result. For emotional well-being, both experimental and control groups had similar improvements in social satisfaction. Exploring the underlying biological mechanisms of the intervention, resting-state fMRI showed that DAN connectivity increased among the experimental group in comparison with the control group. This result needs to be interpreted with caution because fMRI analyses were limited in sample size due to the pandemic-related research hiatus.
Targeting Socially Isolated Older Old
It has been hypothesized that cognitive reserve would be more enhanced when the activities are new to subjects (Park & Bischof, 2013). We targeted socially isolated participants, hypothesizing that they would most likely benefit from this intervention because the activity is novel. As both social isolation and age are risk factors for cognitive decline, targeting this underserved group has high public health importance. The older old is the fastest-growing population in the world, particularly in high-income nations, with higher likelihood of social isolation due to health conditions and limited mobility (Kannan & Veazie, 2022). Delaying dementia incidence even for a year could have a significant impact on dementia prevalence (Brookmeyer et al., 1998). To enhance feasibility of this intervention for older-old subjects, we used conversations that can be initiated with little motivation. This intervention can be tested among other age groups and extended to other risk factors for cognitive decline, such as depression, closely tied to social isolation.
Social bridging Versus Social bonding
Both experimental and control groups improved to similar extents in emotional well-being in the domain of social satisfaction, but only the intervention group exhibited improved cognitive functions. Thus, improvements in cognitive functions observed in the experimental group are not explained or mediated by improved emotional well-being. This result partly coincides with the theoretical framework and empirical cross-sectional results of Perry et al. (2021) suggesting that social bridging and not social bonding is critical for sustaining cognitive functions. It is possible, however, that undetected effects of weak social bonding via video chats might have influenced cognition among the experimental group. Assessments detecting subtle emotional changes secondary to social bonding might be used in the future studies to address this question.
Brain reserve, Cognitive Reserve, and Underlying Biological Mechanisms
In cross-sectional studies, social isolation or perceived isolation is associated with brain structure abnormalities (volumes and thickness), including lower total brain volumes, gray matter volumes, and greater white matter lesion volumes (Lewis et al., 2011; Salinas et al., 2022; Shen et al., 2022; van der Velpen et al., 2022; Kanai et al., 2012). Social isolation is also associated with abnormal connectivity (Spreng et al., 2020). These cross-sectional results suggest that those with social isolation have lower brain reserve, likely to make them susceptible to accelerated clinical manifestations from pathological insults.
Cross-sectional associations cannot identify causal directions; it is possible that underlying age- or disease-associated brain changes lead to impaired management of larger social networks or to reduced motivation for social interactions. Some longer-term social stimulation studies showed potentially beneficial structural changes. In a well-known behavioral intervention study aimed at enhancing intergenerational interactions (Carlson et al., 2015), cortical and hippocampal volumes showed intervention-specific increases reaching significance in men (p’s ≤ .04) after 2 years of intervention. Another study conducted in Japan showed reduced atrophy in hippocampal volume among an intervention group 6 years after trial completion (Sakurai et al., 2018).
Although we did not hypothesize that short-term cognitive stimulation modifies brain structures or pathological progressions, we hypothesized that it might enhance compensatory neural activities and aid in maintaining cognitive functions against age- and pathology-related insults. This concept was theoretically framed by Park and Bischof (2013) as “scaffolding,” similar to the closely related cognitive reserve theory proposed by Stern (2012). We examined rsfMRI network connectivity as a potential neurobiological mechanism and/or indicator of cognitive reserve. The network iFC analyses identified the DAN as a candidate for further exploration. The DAN facilitates voluntary direction and maintenance of visuospatial attention and was likely engaged by our intervention, leading to the improvement in global cognition and encoding indicated by story immediate recall scores among the MCI. In the context of cognitive aging, prior work observed disrupted DAN function in older adults with cognitive impairment or dementia (Li et al., 2012; Wang et al., 2019; Wu et al., 2022; Zhang et al., 2015), suggesting that intervention-related increases in DAN connectivity may bring these individuals more in line with their cognitively normal peers. In a recent dietary intervention, MCI participants receiving a ketogenic drink, which targeted enhanced brain metabolic processes through absorption of ketone bodies, demonstrated increased iFC in DAN at a 6-month follow-up relative to placebo, with increased iFC associated with improvements in attention-related cognition (Roy et al., 2022). Our results suggest the DAN as a neurobiological system modifiable through intervention in older adults, meriting further investigation as a potential mediator of the effects of social engagement on cognitive functions.
Similar to our pilot feasibility study (Dodge et al., 2015), in which most participants had normal cognition, we found improvements in the language-based executive function measured by the semantic fluency test among those with normal cognition. In Alzheimer’s disease, semantic fluency is disproportionately impaired, with phonemic fluency ability less impaired (Cerhan et al., 2002; Henry et al., 2004). Also, a validated test battery developed to detect preclinical subjects (i.e., those without clinical symptoms with Alzheimer’s disease pathology) improved its detection ability by adding a semantic fluency test (Papp et al., 2017). Therefore, semantic fluency tests may be sensitive to subtle changes in cognitive function and able to capture intervention efficacy more effectively among those with normal cognition.
Limitations and Future Directions
This study has limitations. Sample size for the primary outcome was substantially reduced as we were only able to use those with completed M6 assessments prior to the pandemic. However, sensitivity analysis with imputed MoCA scores supported our primary analysis result. For the secondary outcomes, we used a conventional Type I error rate without adjusting for multiple comparisons. These results should be interpreted cautiously. Our original goal was to examine the sustainability of efficacy after maintenance stimulation, but with the research hiatus during the pandemic and resultant uncertainty in time and budget, we discontinued M12 assessments for half of the participants. We believe that since this sample selection was based on timing of participant enrollment, not drop-out, selection bias is minimal. We lack racial and ethnic diversity in our participants. During the pandemic, we had to limit participants to those with existing internet services. These factors limit the generalizability of our findings.
This RCT provided evidence that social interactions, specifically stimulating conversations, could enhance cognitive function in later life and suggests a novel approach for home-based behavioral interventions. We hope to use our conversational materials and related platform as a practical treatment for social isolation and mitigation of associated cognitive decline. Reproducibility, feasibility, and efficacy of this intervention platform in health care systems need to be confirmed. Additionally, even though our study showed high adherence once the intervention started, the frequency and the amount of interactions can be perceived as burdensome, especially as we expand this intervention to more vulnerable patient populations in health care systems. We need to monitor adherence in future studies carefully.
How social interaction affects cognition is yet to be identified. For future studies, we plan baseline characterization of brain structures, as well as personality, to determine optimal beneficiaries from this intervention. Responder analysis might provide additional information on the underlying biological mechanisms and aid in developing personalized interventions. As suggested in a recent editorial (Breitner et al., 2022), behavioral interventions that affect symptomatic features to a significant extent (even without modifying pathology) are as important as pharmacological interventions targeting pathological changes, and may shed light on disease mechanisms affecting neural plasticity.
Supplementary Material
Acknowledgments
We are grateful for the I-CONECT participants and the study team. Special thanks go to our study coordinators and conversational staff: Elena Alfaro, Mattie McDonald, Jacob Lindsey, Colton Scavone, Nita Sheridan, Khoa Nguyen, Alexis Ellis, Louis Weisberg, Deborah Moore, Brandy Peacock, Brennan Heller, Carissa Thornall, Sandy Ruhf, Mary Ziemer-McGinn, Farah Custodio, Diane Farrell, Aimee Lynch, Dillon Banker, Gracia Reyes, Kathryn Rabe, Laura Seeton, Jessica Pedroza, David Forystek, Nicolas May, Jonathan Reader, and Arijit K. Bhaumik.
Contributor Information
Hiroko H Dodge, Department of Neurology, Massachusetts General Hospital, Harvard Medical School, Boston, Massachusetts, USA; Oregon Center for Aging and Technology (ORCATECH), Oregon Health & Science University, Portland, Oregon, USA.
Kexin Yu, Oregon Center for Aging and Technology (ORCATECH), Oregon Health & Science University, Portland, Oregon, USA; Layton Aging and Alzheimer’s Disease Center, Department of Neurology, Oregon Health & Science University, Portland, Oregon, USA.
Chao-Yi Wu, Department of Neurology, Massachusetts General Hospital, Harvard Medical School, Boston, Massachusetts, USA; Oregon Center for Aging and Technology (ORCATECH), Oregon Health & Science University, Portland, Oregon, USA.
Patrick J Pruitt, Department of Neurology, Massachusetts General Hospital, Harvard Medical School, Boston, Massachusetts, USA.
Meysam Asgari, Oregon Center for Aging and Technology (ORCATECH), Oregon Health & Science University, Portland, Oregon, USA; Department of Pediatrics, Oregon Health & Science University, Portland, Oregon, USA.
Jeffrey A Kaye, Oregon Center for Aging and Technology (ORCATECH), Oregon Health & Science University, Portland, Oregon, USA; Layton Aging and Alzheimer’s Disease Center, Department of Neurology, Oregon Health & Science University, Portland, Oregon, USA.
Benjamin M Hampstead, Michigan Alzheimer’s Disease Center, Department of Neurology, University of Michigan, Ann Arbor, Michigan, USA; Research Program on Cognition and Neuromodulation Based Interventions, Department of Psychiatry, University of Michigan, Ann Arbor, Michigan, USA.
Laura Struble, Department of Systems, Populations and Leadership, University of Michigan School of Nursing, Ann Arbor, Michigan, USA.
Kathleen Potempa, Department of Systems, Populations and Leadership, University of Michigan School of Nursing, Ann Arbor, Michigan, USA.
Peter Lichtenberg, Institute of Gerontology and Department of Psychology, Wayne State University, Detroit, Michigan, USA.
Raina Croff, Oregon Center for Aging and Technology (ORCATECH), Oregon Health & Science University, Portland, Oregon, USA; Layton Aging and Alzheimer’s Disease Center, Department of Neurology, Oregon Health & Science University, Portland, Oregon, USA.
Roger L Albin, Michigan Alzheimer’s Disease Center, Department of Neurology, University of Michigan, Ann Arbor, Michigan, USA; GRECC & Neurology Service, VA Ann Arbor Healthcare System, Ann Arbor, Michigan, USA.
Lisa C Silbert, Oregon Center for Aging and Technology (ORCATECH), Oregon Health & Science University, Portland, Oregon, USA; VA Portland Healthcare System, Portland, Oregon, USA.
the I-CONECT Team:
Elena Alfaro, Mattie McDonald, Jacob Lindsey, Colton Scavone, Nita Sheridan, Khoa Nguyen, Alexis Ellis, Louis Weisberg, Deborah Moore, Brandy Peacock, Brennan Heller, Carissa Thornall, Sandy Ruhf, Mary Ziemer-McGinn, Farah Custodio, Diane Farrell, Aimee Lynch, Dillon Banker, Gracia Reyes, Kathryn Rabe, Laura Seeton, Jessica Pedroza, David Forystek, Nicolas May, Jonathan Reader, and Arijit K Bhaumik
Funding
National Institute on Aging grants: R01AG051628, R01AG056102, R01AG056712, P30AG066518, P30AG024978, P30AG053760, F99AG068492, K00AG068492.
Conflict of Interest
None.
Data Availability
The data, analytic methods, and materials used in this report are available to other researchers for replication purposes. The data request can be made at: https://www.i-conect.org/request-data. The studies reported in the manuscript were preregistered at clinicaltrials.gov (NCT02871921).
Author Contributions
Hiroko Dodge (Conceptualization [Lead], Data curation [Lead], Formal analysis [Equal], Funding acquisition [Lead], Investigation [Lead], Methodology [Lead], Project administration [Lead], Resources [Lead], Supervision [Lead], Validation [Lead], Visualization [Lead], Writing – original draft [Lead], Writing – review & editing [Lead]), Kexin Yu (Conceptualization [Supporting], Formal analysis [Supporting], Visualization [Equal], Writing – original draft [Supporting], Writing – review & editing [Supporting]), Chao-Yi Wu (Conceptualization [Supporting], Formal analysis [Equal], Investigation [Equal], Visualization [Equal], Writing – review & editing [Equal]), Patrick Pruitt (Conceptualization [Supporting], Formal analysis [Equal], Investigation [Supporting], Methodology [Supporting], Software [Equal], Visualization [Equal], Writing – review & editing [Equal]), Meysam Asgari (Conceptualization [Supporting], Formal analysis [Supporting], Investigation [Supporting], Methodology [Supporting], Resources [Supporting], Software [Supporting], Writing – review & editing [Equal]), Jeffrey Kaye(Conceptualization [Supporting], Funding acquisition [Supporting], Investigation [Supporting], Resources [Supporting], Writing – review & editing [Equal]), Benjamin Hampstead (Conceptualization [Supporting], Funding acquisition [Supporting], Investigation [Supporting], Methodology [Supporting], Resources [Supporting], Writing – review & editing [Equal]), Laura Struble (Conceptualization [Supporting], Funding acquisition [Supporting], Investigation [Supporting], Methodology [Supporting], Writing – review & editing [Equal]), Kathleen Potempa (Conceptualization [Supporting], Funding acquisition [Supporting], Investigation [Supporting], Methodology [Supporting], Writing – review & editing [Equal]), Peter Lichtenberg (Conceptualization [Supporting], Funding acquisition [Supporting], Investigation [Supporting], Methodology [Supporting], Writing – review & editing [Equal]), Raina Croff (Conceptualization [Supporting], Investigation [Supporting], Methodology [Supporting], Writing – review & editing [Equal]), Roger Albin (Conceptualization [Supporting], Investigation [Supporting], Methodology [Supporting], Validation [Supporting], Writing – review & editing [Equal]), Lisa Silbert (Conceptualization [Supporting], Funding acquisition [Supporting], Investigation [Supporting], Methodology [Equal], Project administration [Supporting], Writing – review & editing [Equal])
Ethics Statement
Study procedures were approved by the Institutional Review Board at the Oregon Health & Science University (OHSU) IRB STUDY00015937) using a single IRB process.
References
- Albert, M. S., DeKosky, S. T., Dickson, D., Dubois, B., Feldman, H. H., Fox, N. C., Gamst, A., Holtzman, D. M., Jagust, W. J., Petersen, R. C., Snyder, P. J., Carrillo, M. C., Thies, B., & Phelps, C. H. (2011). The diagnosis of mild cognitive impairment due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer's & Dementia, 7(3), 270–279. 10.1016/j.jalz.2011.03.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amieva, H., Stoykova, R., Matharan, F., Helmer, C., Antonucci, T. C., & Dartigues, J. -F. (2010). What aspects of social network are protective for dementia? Not the quantity but the quality of social interactions is protective up to 15 years later. Psychosomatic Medicine, 72(9), 905–911. 10.1097/PSY.0b013e3181f5e121 [DOI] [PubMed] [Google Scholar]
- Andrieu, S., Guyonnet, S., Coley, N., Cantet, C., Bonnefoy, M., Bordes, S., Bories, L., Cufi, M. -N., Dantoine, T., Dartigues, J. -F., Desclaux, F., Gabelle, A., Gasnier, Y., Pesce, A., Sudres, K., Touchon, J., Robert, P., Rouaud, O., Legrand, P., MAPT Study Group. (2017). Effect of long-term omega 3 polyunsaturated fatty acid supplementation with or without multidomain intervention on cognitive function in elderly adults with memory complaints (MAPT): A randomised, placebo-controlled trial. The Lancet. Neurology, 16(5), 377–389. 10.1016/S1474-4422(17)30040-6 [DOI] [PubMed] [Google Scholar]
- Ashida, S., & Heaney, C. A. (2008). Differential associations of social support and social connectedness with structural features of social networks and the health status of older adults. Journal of Aging and Health, 20(7), 872–893. 10.1177/0898264308324626 [DOI] [PubMed] [Google Scholar]
- Babakhanyan, I., McKenna, B. S., Casaletto, K. B., Nowinski, C. J., & Heaton, R. K. (2018). National Institutes of Health Toolbox Emotion Battery for English- and Spanish-speaking adults: Normative data and factor-based summary scores. Patient Related Outcome Measures, 9, 115–127. 10.2147/PROM.S151658 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bennett, D. A., Schneider, J. A., Tang, Y., Arnold, S. E., & Wilson, R. S. (2006). The effect of social networks on the relation between Alzheimer’s disease pathology and level of cognitive function in old people: A longitudinal cohort study. Lancet Neurology, 5(5), 406–412. 10.1016/S1474-4422(06)70417-3 [DOI] [PubMed] [Google Scholar]
- Breitner, J. C. S., Dodge, H. H., Khachaturian, Z. S., & Khachaturian, A. S. (2022). “Exceptions that prove the rule”—Why have clinical trials failed to show efficacy of risk factor interventions suggested by observational studies of the dementia-Alzheimer’s disease syndrome? Alzheimer’s & Dementia, 18(3), 389–392. 10.1002/alz.12633 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brookmeyer, R., Gray, S., & Kawas, C. (1998). Projections of Alzheimer’s disease in the United States and the public health impact of delaying disease onset. American Journal of Public Health, 88(9), 1337–1342. 10.2105/ajph.88.9.1337 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carlson, M. C., Kuo, J. H., Chuang, Y.-F., Varma, V. R., Harris, G., Albert, M. S., Erickson, K. I., Kramer, A. F., Parisi, J. M., Xue, Q.-L., Tan, E. J., Tanner, E. K., Gross, A. L., Seeman, T. E., Gruenewald, T. L., McGill, S., Rebok, G. W., & Fried, L. P. (2015). Impact of the Baltimore Experience Corps Trial on cortical and hippocampal volumes. Alzheimer's & Dementia, 11(11), 1340–1348. 10.1016/j.jalz.2014.12.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cerhan, J. H., Ivnik, R. J., Smith, G. E., Tangalos, E. C., Petersen, R. C., & Boeve, B. F. (2002). Diagnostic utility of letter fluency, category fluency, and fluency difference scores in Alzheimer’s disease. Clinical Neuropsychologist, 16(1), 35–42. 10.1076/clin.16.1.35.8326 [DOI] [PubMed] [Google Scholar]
- Cerino, E. S., Hooker, K., Goodrich, E., & Dodge, H. H. (2020). Personality moderates intervention effects on cognitive function: A 6-week conversation-based intervention. Gerontologist, 60(5), 958–967. 10.1093/geront/gnz063 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Craft, S., Newcomer, J., Kanne, S., Dagogo-Jack, S., Cryer, P., Sheline, Y., Luby, J., Dagogo-Jack, A., & Alderson, A. (1996). Memory improvement following induced hyperinsulinemia in Alzheimer’s disease. Neurobiology of Aging, 17(1), 123–130. 10.1016/0197-4580(95)02002-0 [DOI] [PubMed] [Google Scholar]
- Crawford, J. R., & Henry, J. D. (2004). The positive and negative affect schedule (PANAS): Construct validity, measurement properties and normative data in a large non-clinical sample. British Journal of Clinical Psychology, 43(Pt 3), 245–265. 10.1348/0144665031752934 [DOI] [PubMed] [Google Scholar]
- Dodge, H. H., Goldstein, F. C., Wakim, N. I., Gefen, T., Teylan, M., Chan, K. C. G., Kukull, W. A., Barnes, L. L., Giordani, B., Hughes, T. M., Kramer, J. H., Loewenstein, D. A., Marson, D. C., Mungas, D. M., Mattek, N., Sachs, B. C., Salmon, D. P., Willis‐Parker, M., & Welsh‐Bohmer, K. A.; National Alzheimer’s Coordinating Center (NACC). (2020). Differentiating among stages of cognitive impairment in aging: Version 3 of the Uniform Data Set (UDS) neuropsychological test battery and MoCA index scores. Alzheimer’s & Dementia: Translational Research & Clinical Interventions, 6(1):e12103. 10.1002/trc2.12103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dodge, H. H., Kita, Y., Takechi, H., Hayakawa, T., Ganguli, M., & Ueshima, H. (2008). Healthy cognitive aging and leisure activities among the oldest old in Japan: Takashima Study. Journals of Gerontology, Series A: Biological Sciences and Medical Sciences, 63(11), 1193–1200. 10.1093/gerona/63.11.1193 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dodge, H. H., Zhu, J., Mattek, N. C., Bowman, M., Ybarra, O., Wild, K. V., Loewenstein, D. A., & Kaye, J. A. (2015). Web-enabled conversational interactions as a method to improve cognitive functions: Results of a 6-week randomized controlled trial. Alzheimer’s & Dementia: Translational Research & Clinical Interventions, 1(1), 1–12. 10.1016/j.trci.2015.01.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans, I. E. M., Martyr, A., Collins, R., Brayne, C., & Clare, L. (2019). Social isolation and cognitive function in later life: A systematic review and meta-analysis. Journal of Alzheimer's Disease, 70(Suppl 1), S119–S144. 10.3233/JAD-180501 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng, C., Eickhoff, S. B., Li, T., Wang, L., Becker, B., Camilleri, J. A., Hétu, S., & Luo, Y. (2021). Common brain networks underlying human social interactions: Evidence from large-scale neuroimaging meta-analysis. Neuroscience & Biobehavioral Reviews, 126, 289–303. 10.1016/j.neubiorev.2021.03.025 [DOI] [PubMed] [Google Scholar]
- Fratiglioni, L., Paillard-Borg, S., & Winblad, B. (2004). An active and socially integrated lifestyle in late life might protect against dementia. Lancet Neurology, 3(6), 343–353. 10.1016/S1474-4422(04)00767-7 [DOI] [PubMed] [Google Scholar]
- Henry, J. D., Crawford, J. R., & Phillips, L. H. (2004). Verbal fluency performance in dementia of the Alzheimer’s type: A meta-analysis. Neuropsychologia 42(9):1212–1222. 10.1016/j.neuropsychologia.2004.02.001 [DOI] [PubMed] [Google Scholar]
- Hughes, M. E., Waite, L. J., Hawkley, L. C., & Cacioppo, J. T. (2004). A short scale for measuring loneliness in large surveys. Research on Aging, 26(6), 655–672. 10.1177/0164027504268574 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jack, C. R., Albert, M. S., Knopman, D. S., McKhann, G. M., Sperling, R. A., Carrillo, M. C., Thies, B., & Phelps, C. H. (2011). Introduction to the recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer's & Dementia, 7(3), 257–262. 10.1016/j.jalz.2011.03.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kallianpur, K. J., Masaki, K. H., Chen, R., Willcox, B. J., Allsopp, R. C., Davy, P., & Dodge, H. H. (2023). Weak social networks in late life predict incident Alzheimer’s disease: The Kuakini Honolulu-Asia Aging Study. Journals of Gerontology, Series A: Biological Sciences and Medical Sciences, 78(4), 663–672. 10.1093/gerona/glac215 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanai, R., Bahrami, B., Duchaine, B., Janik, A., Banissy, M. J., & Rees, G. (2012). Brain structure links loneliness to social perception. Current Biology, 22(20), 1975–1979. 10.1016/j.cub.2012.08.045 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kannan, V. D., & Veazie, P. J. (2022). US trends in social isolation, social engagement, and companionship—Nationally and by age, sex, race/ethnicity, family income, and work hours, 2003–2020. SSM—Population Health, 21, 101331. 10.1016/j.ssmph.2022.101331 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelly, M. E., Duff, H., Kelly, S., McHugh Power, J. E., Brennan, S., Lawlor, B. A., & Loughrey, D. G. (2017). The impact of social activities, social networks, social support and social relationships on the cognitive functioning of healthy older adults: A systematic review. Systematic Reviews, 6(1), 259. 10.1186/s13643-017-0632-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kivipelto, M., Mangialasche, F., Snyder, H. M., Allegri, R., Andrieu, S., Arai, H., Baker, L., Belleville, S., Brodaty, H., Brucki, S. M., Calandri, I., Caramelli, P., Chen, C., Chertkow, H., Chew, E., Choi, S. H., Chowdhary, N., Crivelli, L., Torre, R. D. L., Carrillo, M. C. (2020). World-Wide FINGERS Network: A global approach to risk reduction and prevention of dementia. Alzheimer’s & Dementia, 16(7), 1078–1094. 10.1002/alz.12123 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klaming, R., Annese, J., Veltman, D. J., & Comijs, H. C. (2017). Episodic memory function is affected by lifestyle factors: A 14-year follow-up study in an elderly population. Aging, Neuropsychology, and Cognition, 24(5), 528–542. 10.1080/13825585.2016.1226746 [DOI] [PubMed] [Google Scholar]
- Lewis, P. A., Rezaie, R., Brown, R., Roberts, N., & Dunbar, R. I. M. (2011). Ventromedial prefrontal volume predicts understanding of others and social network size. Neuroimage, 57(4), 1624–1629. 10.1016/j.neuroimage.2011.05.030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lezak, M. D., Howieson, D. B., Bigler, E. D., & Tranel, D. (2012). Neuropsychological assessment (5th ed.). Oxford University Press. [Google Scholar]
- Li, R., Wu, X., Fleisher, A. S., Reiman, E. M., Chen, K., & Yao, L. (2012). Attention-related networks in Alzheimer’s disease: A resting functional MRI study. Human Brain Mapping, 33(5), 1076–1088. 10.1002/hbm.21269 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Livingston, G., Huntley, J., Sommerlad, A., Ames, D., Ballard, C., Banerjee, S., Brayne, C., Burns, A., Cohen-Mansfield, J., Cooper, C., Costafreda, S. G., Dias, A., Fox, N., Gitlin, L. N., Howard, R., Kales, H. C., Kivimäki, M., Larson, E. B., Ogunniyi, A., … Mukadam, N. (2020). Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. Lancet (London, England), 396(10248), 413–446. 10.1016/S0140-6736(20)30367-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lubben, J., Blozik, E., Gillmann, G., Iliffe, S., von Renteln Kruse, W., Beck, J. C., & Stuck, A. E. (2006). Performance of an abbreviated version of the Lubben Social Network Scale among three European community-dwelling older adult populations. Gerontologist, 46(4), 503–513. 10.1093/geront/46.4.503 [DOI] [PubMed] [Google Scholar]
- McKhann, G. M., Knopman, D. S., Chertkow, H., Hyman, B. T., Jack, C. R., Kawas, C. H., Klunk, W. E., Koroshetz, W. J., Manly, J. J., Mayeux, R., Mohs, R. C., Morris, J. C., Rossor, M. N., Scheltens, P., Carrillo, M. C., Thies, B., Weintraub, S., & Phelps, C. H. (2011). The diagnosis of dementia due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer's & Dementia, 7(3), 263–269. 10.1016/j.jalz.2011.03.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nasreddine, Z. S., Phillips, N. A., Bédirian, V., Charbonneau, S., Whitehead, V., Collin, I., Cummings, J. L., & Chertkow, H. (2005). The Montreal Cognitive Assessment, MoCA: A brief screening tool for mild cognitive impairment. Journal of the American Geriatrics Society, 53(4), 695–699. 10.1111/j.1532-5415.2005.53221.x [DOI] [PubMed] [Google Scholar]
- National Academies of Sciences, Engineering, and Medicine. (2020). Social isolation and loneliness in older adults: Opportunities for the health care system (p. 25663). National Academies Press. 10.17226/25663 [DOI] [PubMed] [Google Scholar]
- Papp, K. V., Rentz, D. M., Orlovsky, I., Sperling, R. A., & Mormino, E. C. (2017). Optimizing the preclinical Alzheimer's cognitive composite with semantic processing: The PACC5. Alzheimer’s & Dementia: Translational Research & Clinical Interventions 10;3(4):668–677. 10.1016/j.trci.2017.10.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park, D. C., & Bischof, G. N. (2013). The aging mind: Neuroplasticity in response to cognitive training. Dialogues in Clinical Neuroscience, 15(1), 109–119. 10.31887/DCNS.2013.15.1/dpark [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park, D. C., Gutchess, A. H., Meade, M. L., & Stine-Morrow, E. A. L. (2007). Improving cognitive function in older adults: Nontraditional approaches. Journals of Gerontology, Series B: Psychological Sciences and Social Sciences, 62, 45–52. 10.1093/geronb/62.special_issue_1.45 [DOI] [PubMed] [Google Scholar]
- Penninkilampi, R., Casey, A.-N., Singh, M. F., & Brodaty, H. (2018). The association between social engagement, loneliness, and risk of dementia: A systematic review and meta-analysis. Journal of Alzheimer's Disease, 66(4), 1619–1633. 10.3233/JAD-180439 [DOI] [PubMed] [Google Scholar]
- Perry, B. L., McConnell, W. R., Peng, S., Roth, A. R., Coleman, M., Manchella, M., Roessler, M., Francis, H., Sheean, H., & Apostolova, L. A. (2021). Social networks and cognitive function: An evaluation of social bridging and bonding mechanisms. The Gerontologist, 62, 865–875. 10.1093/geront/gnab112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poey, J. L., Burr, J. A., & Roberts, J. S. (2017). Social connectedness, perceived isolation, and dementia: Does the social environment moderate the relationship between genetic risk and cognitive well-being? Gerontologist, 57(6), 1031–1040. 10.1093/geront/gnw154 [DOI] [PubMed] [Google Scholar]
- Radecki, M. A., Cox, S. R., & MacPherson, S. E. (2019). Theory of mind and psychosocial characteristics in older men. Psychology and Aging, 34(1), 145. 10.1037/pag0000324 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roy, M., Edde, M., Fortier, M., Croteau, E., Castellano, C. -A., St-Pierre, V., Vandenberghe, C., Rheault, F., Dadar, M., Duchesne, S., Bocti, C., Fulop, T., Cunnane, S. C., & Descoteaux, M. (2022). A ketogenic intervention improves dorsal attention network functional and structural connectivity in mild cognitive impairment. Neurobiology of Aging, 115, 77–87. 10.1016/j.neurobiolaging.2022.04.005 [DOI] [PubMed] [Google Scholar]
- Sachs, B. C., Steenland, K., Zhao, L., Hughes, T. M., Weintraub, S., Dodge, H. H., Barnes, L. L., Craft, S., Parker, M. L., & Goldstein, F. C. (2020). Expanded demographic norms for version 3 of the Alzheimer disease centers’ neuropsychological test battery in the uniform data set. Alzheimer Disease & Associated Disorders, 34(3), 191–197. 10.1097/WAD.0000000000000388 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sakurai, R., Ishii, K., Sakuma, N., Yasunaga, M., Suzuki, H., Murayama, Y., Nishi, M., Uchida, H., Shinkai, S., & Fujiwara, Y. (2018). Preventive effects of an intergenerational program on age‐ related hippocampal atrophy in older adults: The REPRINTS study. International Journal of Geriatric Psychiatry, 2018;33:e264–e272. 10.1002/gps.4785 [DOI] [PubMed] [Google Scholar]
- Salinas, J., Beiser, A. S., Samra, J. K., O’Donnell, A., DeCarli, C. S., Gonzales, M. M., Aparicio, H. J., & Seshadri, S. (2022). Association of loneliness with 10-year dementia risk and early markers of vulnerability for neurocognitive decline. Neurology, 98(13), e1337–e1348. 10.1212/WNL.0000000000200039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schouten, H. J. A. (1995). Adaptive biased urn randomization in small strata when blinding is impossible. Biometrics, 51(4), 1529. 10.2307/2533283 [DOI] [PubMed] [Google Scholar]
- Shen, C., Rolls, E. T., Cheng, W., Kang, J., Dong, G., Xie, C., Zhao, X. -M., Sahakian, B. J., & Feng, J. (2022). Associations of social isolation and loneliness with later dementia. Neurology, 99(2), e164–e175. 10.1212/WNL.0000000000200583 [DOI] [PubMed] [Google Scholar]
- Spreng, R. N., Dimas, E., Mwilambwe-Tshilobo, L., Dagher, A., Koellinger, P., Nave, G., Ong, A., Kernbach, J. M., Wiecki, T. V., Ge, T., Li, Y., Holmes, A. J., Yeo, B. T. T., Turner, G. R., Dunbar, R. I. M., & Bzdok, D. (2020). The default network of the human brain is associated with perceived social isolation. Nature Communications, 11(1), 6393. 10.1038/s41467-020-20039-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spreng, R. N., Dimas, E., Mwilambwe-Tshilobo, L., Dagher, A., Koellinger, P., Nave, G., Ong, A., Kernbach, J. M., Wiecki, T. V., Ge, T., Li, Y., Holmes, A. J., Yeo, B. T. T., Turner, G. R., Dunbar, R. I. M., & Bzdok, D. (2020). The default network of the human brain is associated with perceived social isolation. Nature Communications, 11(1), 6393. 10.1038/s41467-020-20039-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stern, Y. (2012). Cognitive reserve in ageing and Alzheimer’s disease. The Lancet. Neurology, 11(11), 1006–1012. 10.1016/S1474-4422(12)70191-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Velpen, I. F., Melis, R. J. F., Perry, M., Vernooij-Dassen, M. J. F., Ikram, M. A., & Vernooij, M. W. (2022). Social health is associated with structural brain changes in older adults: The Rotterdam Study. Biological Psychiatry: Cognitive Neuroscience and Neuroimaging, 7(7), 659–668. 10.1016/j.bpsc.2021.01.009 [DOI] [PubMed] [Google Scholar]
- Wang, J., Liu, J., Wang, Z., Sun, P., Li, K., & Liang, P. (2019). Dysfunctional interactions between the default mode network and the dorsal attention network in subtypes of amnestic mild cognitive impairment. Aging, 11(20), 9147–9166. 10.18632/aging.102380 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watson, D., Clark, L. A., & Tellegen, A. (1988). Development and validation of brief measures of positive and negative affect: The PANAS scales. Journal of Personality and Social Psychology, 54(6), 1063–1070. 10.1037//0022-3514.54.6.1063 [DOI] [PubMed] [Google Scholar]
- Weintraub, S., Besser, L., Dodge, H. H., Teylan, M., Ferris, S., Goldstein, F. C., Giordani, B., Kramer, J., Loewenstein, D., Marson, D., Mungas, D., Salmon, D., Welsh-Bohmer, K., Zhou, X. -H., Shirk, S. D., Atri, A., Kukull, W. A., Phelps, C., & Morris, J. C. (2018). Version 3 of the Alzheimer disease centers’ neuropsychological test battery in the uniform data set (UDS). Alzheimer Disease and Associated Disorders, 32(1), 10–17. 10.1097/wad.0000000000000223 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu, H., Song, Y., Chen, S., Ge, H., Yan, Z., Qi, W., Yuan, Q., Liang, X., Lin, X., & Chen, J. (2022). An activation likelihood estimation meta-analysis of specific functional alterations in dorsal attention network in mild cognitive impairment. Frontiers in Neuroscience, 16:876568. 10.3389/fnins.2022.876568 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yesavage, J. A., Brink, T. L., Rose, T. L., Lum, O., Huang, V., Adey, M., & Leirer, V. O. (1982). Development and validation of a geriatric depression screening scale: A preliminary report. Journal of Psychiatric Research, 17(1), 37–49. 10.1016/0022-3956(82)90033-4 [DOI] [PubMed] [Google Scholar]
- Yu, K., Wild, K., Dowling, N. M., Kaye, J. A., Silbert, L. C., & Dodge, H. H. (2022). Emotional characteristics of socially isolated older adults with MCI using tablet administered NIH toolbox: I-CONECT study. Alzheimer's & Dementia (Amsterdam, Netherlands), 14(1), e12372. 10.1002/dad2.12372 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu, K., Wild, K., Potempa, K., Hampstead, B. M., Lichtenberg, P. A., Struble, L. M., Pruitt, P., Alfaro, E. L., Lindsley, J., MacDonald, M., Kaye, J. A., Silbert, L. C., & Dodge, H. H. (2021). The Internet-Based Conversational Engagement Clinical Trial (I-CONECT) in socially isolated adults 75+ years old: Randomized controlled trial protocol and COVID-19 related study modifications. Frontiers in Digital Health, 3, 714813. 10.3389/fdgth.2021.714813 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang, Z., Zheng, H., Liang, K., Wang, H., Kong, S., Hu, J., Wu, F., & Sun, G. (2015). Functional degeneration in dorsal and ventral attention systems in amnestic mild cognitive impairment and Alzheimer’s disease: An fMRI study. Neuroscience Letters, 585, 160–165. 10.1016/j.neulet.2014.11.050 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data, analytic methods, and materials used in this report are available to other researchers for replication purposes. The data request can be made at: https://www.i-conect.org/request-data. The studies reported in the manuscript were preregistered at clinicaltrials.gov (NCT02871921).