ABSTRACT
Catheter ablation therapy for persistent atrial fibrillation (PeAF) is both difficult and has limited outcomes. The mechanisms underlying the development and persistence of atrial fibrillation (AF) are not fully understood; therefore, ablation strategies are diverse. A 45-year-old man was referred to our hospital for persistent atrial fibrillation to undergo radiofrequency catheter insertion (RFCA). In the first session we conducted pulmonary vein isolation and additional linear ablation, including that of the roof line and posterior inferior line (posterior box lesion) as the stepwise ablation. However, AF was recurred in six months, therefore he was readmitted for second session ablation preoperative 3D computed tomography (CT) scan for drug-refractory PeAF was performed. The additional isolation of the left superior pulmonary vein and potential drivers of AF by mapping wavefront propagation using multipolar catheters by CARTOFINDER (Biosense Webster, Inc, Diamond Bar, CA, USA) was conducted. However, AF did not terminate. Tomography revealed that the left atrial (LA) diverticulum (LAD) was found uniquely. Electrophysiological findings showed focal firing of the myocardial sleeve and LA diverticulum by an approach for defragmented potentials by re-visiting in interval confidence level (ICL) mode included in the electroanatomical mapping system (CARTO 3, Biosense Webster, Inc, Diamond Bar, CA, USA) and the ablation by encircling this site finally made AF terminate. The AF has not recurred for more than 12 months without the use of antiarrhythmic drugs. This case report suggests that additional ablation around substrates in LAD may be effective for treating refractory AF.
Key Words: radiofrequency ablation, CARTOFINDER, CFAE, persistent atrial fibrillation
INTRODUCTION
Pulmonary vein isolation (PVI) is a well-established strategy that is widely used in the treatment of atrial fibrillation (AF).1 However, catheter ablation therapy for persistent atrial fibrillation (PeAF) owing to non–pulmonary vein (PV) triggers or substrates is both difficult and has limited outcomes. The mechanisms underlying the development and persistence of AF are not fully understood; therefore, ablation strategies are diverse. The 3-dimentional electroanatomical mapping system (CARTO 3, Biosense Webster, Inc, Diamond Bar, CA, USA) is a novel useful system that could not only identify potential drivers of AF by mapping wavefront propagation using multipolar catheters by CARTOFINDER (Biosense Webster, Inc, Diamond Bar, CA, USA), but detect latent substrates by the distribution map of complex fractionated atrial electrograms (CFAE) potential in the interval confidence level (ICL) mode of the CARTO system.2 Here, we report a case of intractable chronic PeAF in which ablation of the LAD was effective.
A left atrial diverticulum (LAD) is the remnant derived from the primitive atrium,3 and LAD were relatively common entities in patients for catheter ablation for AF in the previous study.4 To the best of our knowledge, there have been not a few reported cases of effective ablation of a diverticulum in the interatrial septum during AF by using the CARTO system to detect potential substrates in the area and map them for ablation.5
In this paper, we report a case of intractable chronic PeAF in which ablation of the LAD was effective. The atrial fibrillation has been maintained without recurrence for more than 12 months.
CASE REPORT
This study was conducted out in compliance with the Declaration of Helsinki and was approved by the ethics committees of General Hospital Minamiseikyo Hospital.
A 45-year-old man was diagnosed with long-lasting PeAF on a physical examination and came to our hospital to undergo radiofrequency catheter ablation for PeAF. Three-dimensional multidetector computed tomography revealed a diverticulum on the posteroseptal wall of the LA. In the first session, the standard bilateral encircling PVI was performed using CARTO 3. Due to safety, the ablation was avoided in an area including the diverticulum, which was left outside the ablation line with RF applications in a point-by-point fashion using a ThermoCool SmartTouch™ (Biosense Webster, Inc, Diamond Bar, CA, USA) irrigated-tip contact force-sensing ablation catheter. The electrically successful PVI did not terminate AF. In addition to the standard bilateral encircling pulmonary vein isolation, for the extra-PV LA ablation, we conducted additional linear ablation including that of a roof line, posterior inferior line (posterior box lesion) as the stepwise ablation. AF terminated after cardioversion was performed. A rapid pacing and high-dose administration of isoproterenol did not result in AF.
After half a year of the first session, he had a recurrence of atrial fibrillation on his ECG. CARTO 3 also showed that the previous cauterization site was located by a 1-mm multielectrode-mapping catheter (PentaRay catheter, Biosense Webster, Inc, Diamond Bar, CA, USA) (Figure 1B and 1C). The mean AF cycle length from 10 beats at the diverticulum site and its surrounding areas was manually calculated. The mean cycle length of the diverticulum and its surrounding area were 130 ms (n=3) and 147.6 ms (n=4), respectively (p<0.01 by using Student’s t test for unmatched pairs). The isolation of the left superior pulmonary vein was additionally isolated electrically. However, the AF did not terminate after cardioversion in addition to this procedure. The LA was mapped with CARTOFINDER using a PentaRay catheter (Figure 2A and 2B). The CARTOFINDER module map revealed AF drivers with a focal repetitive activation pattern in the LA posterior wall outside the posterior inferior line (Figure 2C). The PentaRay catheter was stabilized at each site for 30 seconds and 30-second unipolar and bipolar electrograms were stored in CARTO3 and the unipolar electrogram on the PentaRay catheter (Biosense Webster, Inc, Diamond Bar, CA, USA) recorded posteroinferior and posterolateral sites showed a QS pattern (Figure 2D). However, AF did not terminate after the ablations of 2 potential sources with a focal repetitive activation.
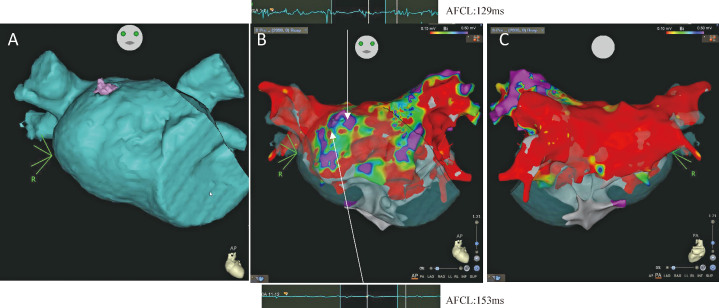
Fig. 1.
High density left atrial voltage maps
Fig. 1A: Preoperative 3D-CT that detects a diverticulum on the posteroseptal wall of the LA. High density left atrial voltage maps created during AF ablation are shown using CARTO (Biosense Webster, Inc, Diamond Bar, CA, USA) recorded by a PentaRay catheter (Biosense Webster, Inc, Diamond Bar, CA, USA). Purple color represents areas greater than the threshold of 0.5 mV.
Fig. 1B, 1C:Anterior-posterior (AP) view (B) and posterior-anterior (PA) view (C). They show the voltage map after the first session ablation procedure with isolation of PVI and post wall isolation. PV reconnection was also identified in the left anterior PV and the roof with atrial fibrillation recurrence after undergoing the pulmonary vein isolation (PVI) procedure. Figure 1B illustrates representative electrograms and cycle lengths (CL) for left atrial diverticulum (LAD) and the surrounding area.
AFCL: atrial fibrillation cycle length
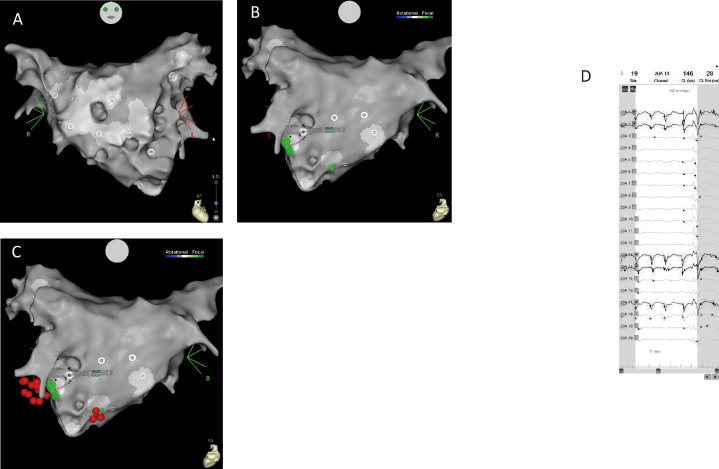
Fig. 2.
CARTOFINDER (Biosense Webster, Inc, Diamond Bar, CA, USA) module maps and electrograms
Fig. 2A, 2B: Anterior-posterior (AP) view (A) and posterior-anterior (PA) view (B). They show the CARTOFINDER module map around the diverticulum after pulmonary vein isolation and box isolation. Drivers with a focal repetitive activation pattern (labeled in green) were identified at the posteroinferior and posterolateral sites (B).
Fig. 2C: Posterior-anterior (PA) view after ablation. Red tags represent ablation lesions.
Fig. 2D: The unipolar electrogram on the PentaRay catheter (Biosense Webster, Inc, Diamond Bar, CA, USA). This figure illustrates the unipolar electrogram on the PentaRay catheter (Biosense Webster, Inc, Diamond Bar, CA, USA) recorded posteroinferior and posterolateral sites showing a QS pattern (20A1, 2, 13, 14, and17) (cycle length 146 ms).
Finally, we decided to use the mapping module of CARTO system to evaluate the fragmented potentials in the ICL mode. The CFAE map expressed the distribution of fragmented potentials in the LA by the PentaRay catheter (Figure 3A and 3B). In the CFAE map, the “Threshold values” were set to 0.03 mv and 1.5 mv, and “Duration values” were set to 3 ms and 150 ms.2 The color bar is configured to display 60% of the maximum ICL value. The electrogram (Figure 3C) revealed electrical activity in the diverticulum at the posteroseptal wall. Therefore, diverticulum isolation was performed with a ThermoCool SmartTouch with RF energy at a power setting of 40 W and an ablation index of 420 adjacent to the LA diverticulum carefully to enclose it not to apply RF directly to the substrates inside the diverticulum to avoid perforation, and the diverticulum was electrically isolated with reference to local electrograms by the encircling RF applications to terminate AF (Figure 3C). Subsequently, rapid pacing and high-dose administration of isoproterenol made the AF noninducible. Currently, the atrial fibrillation has not recurred for more than 12 months with no symptoms without the use of antiarrhythmic drugs.
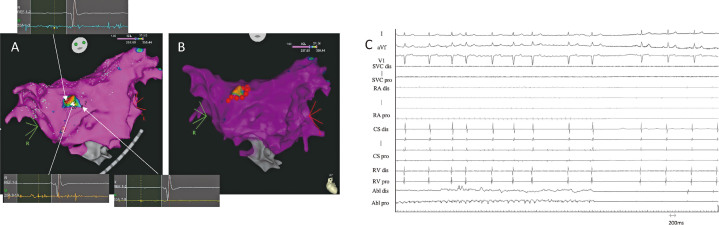
Fig. 3.
The distribution of CFAE potential (CFAE map), the degree of fragmented potentials (CFAE potential) by module of CARTO system, and electrogram under ablation of AF
Fig. 3A: Complex fractionated atrial electrograms (CFAE) map by interval confidence level (ICL) mode of the CARTO system during AF before CFAE ablation (anterior-posterior [AP] view) and representative electrograms for LAD and the surrounding area. The areas with the highest number of ICLs are shown in red, while those with a low number of ICLs are displayed in purple. The color bar is configured to display 60% of the maximum ICL value.
Fig. 3B: CFAE map after atrial fibrillation (AF) termination. Red tags represent the ablation lesions for CFAE.
Fig. 3C: Intracardiac electrograms. These electrograms are shown to be wider and have more fragmented deflections in the left atrial diverticulum (LAD) than in the right atrium (RA) and coronary sinus (CS). Atrial fibrillation terminated into sinus rhythm during ablation encircling a diverticulum source at the posteroseptal left atrium.
SVC dis: distal superior vena cava
SVC pro: proximal superior vena cava
RA dis: distal right atrium
RA pro: proximal right atrium
CS dis: distal coronary sinus
CS pro: sproximal coronary sinus
RV dis: distal right ventricular
RV pro: proximal right ventricular
Abl dis: distal bipole of the ablation catheter
Abl pro: proximal bipole of the ablation catheter
DISCUSSION
We encountered a challenging case in which atrial fibrillation recurred six months after successful ablation of box isolation. CARTOFINDER mapping for PeAF revealed repetitive local activation in the LA posterior wall. Although ablation was performed, AF did not stop. CFAE was detected in the LAD, and atrial fibrillation was subsequently terminated by ablation of local activation in the area.
Recent studies have shown that LAD are a common anatomic variant in patients referred for CT (Figure 1A) coronary angiography. LAD is reported to be common, with a prevalence of 36.0% in patients with AF.6 However, previous studies have reported to detect no significant difference in the prevalence of LAD between patients with sinus rhythm and atrial fibrillation.7 CFAEs are localized to specific regions of the atria during PeAF and serve as substrate sites for the arrhythmia, making them an important target for ablation in AF. By ablating the areas where CFAEs are persistently recorded, atrial fibrillation can be eliminated, typically resulting in non-inducibility of AF. It has been reported that fragmented signals are more frequently recorded in areas near the diverticulum and its orifice, which is consistent with the present case. CFAEs are known to occur frequently within the diverticulum, and Pachon et al8 attributed the presence of inhomogeneous muscle within the diverticulum, which they referred to as fibromuscular myocardium, to the appearance of complex potentials on endocardial electrograms in the LAD. Alternatively, it has been suggested that the appearance of CFAEs within the diverticulum may simply reflect the collision of wavefronts within the diverticulum rather than being indicative of an arrhythmogenic substrate because of the composition of many cell groups that form wavefronts in different phases and appear as polymorphic signals in the endocardial potential. Therefore, CFAEs observed within LAD is less likely to serve as ablation targets. If the diverticulum is derived from the pulmonary veins, it could serve as a substrate. There is also a report of congenital diverticula containing myocardial fibers intermingling within the fibrous tissue of the diverticular wall.9 However, the pathophysiology of LAD is not yet fully understood. CFAE is characterized by a low atrial voltage and a very short cycle length, and the sites that exhibit CFAE substrates have the shortest cycle lengths compared with other atrial sites.10 In the present case, the potential within LAD was significantly faster than the surrounding myocardial potentials (136 ms vs 147.6 ms, respectively), theoretically making it a valid substrate for atrial fibrillation. However, the effect of additional ablation appears to be negative and limited, and the causal relationship between LAD and arrhythmia development has not been identified except for case reports. In cases where left atrial remodeling like this case, progresses and intractable AF is observed, ablation of LA in the atrium could be effective. However, on the other hand, it has been suggested that the activity that causes atrial fibrillation arises from the myocardial wall and sleeves adjacent to the LAD,4 and that there is a high risk of perforation of the myocardial wall due to local tissue overheating when high-frequency electric current is applied within LAD. Furthermore, considering that the expert consensus statement classifies CFAE ablation for PeAF as Class IIb,1 it was considered reasonable to ablate the area surrounding LAD in such cases as this.
Although LAD is thought to be theoretically valid as a substrate of AF, there are few reports that have found a causal relationship between LAD and arrhythmogenesis, except for a few case reports.10 In patients with progressive left atrial remodeling, as in this case, cauterization to the diverticulum could be effective.
The CARTOFINDER mapping system uses unipolar potentials to analyze the spread of AF and identifies repeated local QS patterns in the electrogram as focal activations (Figure 2C), which may indicate the origin or termination of electrical conduction on the atrial surface. Ablation guided by CARTOFINDER has been successful in terminating AF in some patients with persistent AF. However, in this case, although CARTOFINDER detected repetitive focal activation in the LA posterior wall, ablation of these focal activations did not terminate AF. In this case, the CARTOFINDER map was performed after box isolation, not before ablation; therefore, the AF recorded in the posterolateral wall rotor may not have affected the entire atrial AF process from the beginning.
Although there have been several previous reports of epicardial bypass tracts between the LA and vena cava,11 and LADs are not uncommon,3 little is known about the relationship between epicardial bypass tracts and atrial diverticula. As seen in a report of macro-reentrant atrial tachycardia using the epicardial connection of the LAD,12 the epicardial bypass tract attached to the diverticulum is a potentially arrhythmogenic structure. In the present case, the ostium of the diverticulum was encircled and terminated; however, if non-PV triggering was induced, additional ablation to the RA insertion site might have been necessary for its disconnection.
There may be clinically a certain number of cases in which ablation of the diverticulum is effective in patients with advanced left atrial remodeling, as in the present case. However, this procedure should not be performed routinely because the procedure and ablation time are longer, leading to increased complications and the development of other arrhythmias such as atrial tachycardia. Performing additional and unnecessary ablations may increase the procedure time and the risk of incidence of serious adverse events such as tamponade and stroke.13
In conclusion, we experienced a case in which the ablation of the activity with complex fractionated electrogram around a LAD was effective in a patient with atrial fibrillation after box isolation. This study highlights that LAD could sometimes be involved in the onset and maintenance of AF. Further studies are needed to confirm our observations and to identify the optimal approach for ablation therapy in such cases.
CONFLICT OF INTEREST
The author declares no conflict of interest for this article.
Abbreviations
- PeAF
persistent atrial fibrillation
- ICL
interval confidence level
- LAD
left atrial diverticulum
- CFAE
complex fractionated atrial electrograms
REFERENCES
- 1.Calkins H, Hindricks G, Cappato R, et al. 2017 HRS/EHRA/ECAS/APHRS/SOLAECE expert consensus statement on catheter and surgical ablation of atrial fibrillation. Heart Rhythm. 2017;14(10):e275-e444. doi: 10.1016/j.hrthm.2017.05.012. [DOI] [PMC free article] [PubMed]
- 2.Ohe M, Haraguchi G, Kumanomido J, et al. New tailored approach using a revised assessment of fragmented potentials for persistent atrial fibrillation: Early area defragmentation by modified CFAE module. J Cardiovasc Electrophysiol. 2019;30(6):844–853. doi: 10.1111/jce.13888. [DOI] [PubMed]
- 3.Igawa O, Miake J, Adachi M. The small diverticulum in the right anterior wall of the left atrium. Europace. 2008;10(1):120. doi: 10.1093/europace/eum275. [DOI] [PubMed]
- 4.Peng LQ, Yu JQ, Yang ZG, et al. Left atrial diverticula in patients referred for radiofrequency ablation of atrial fibrillation: assessment of prevalence and morphologic characteristics by dual-source computed tomography. Circ Arrhythm Electrophysiol. 2012;5(2):345–350. doi: 10.1161/CIRCEP.111.965665. [DOI] [PubMed]
- 5.Killeen RP, O’Connor SA, Keane D, Dodd JD. Ectopic focus in an accessory left atrial appendage: radiofrequency ablation of refractory atrial fibrillation. Circulation. 2009;120(8):e60-e62. doi: 10.1161/CIRCULATIONAHA.109.855569. [DOI] [PubMed]
- 6.Abbara S, Mundo-Sagardia JA, Hoffmann U, Cury RC. Cardiac CT assessment of left atrial accessory appendages and diverticula. AJR Am J Roentgenol. 2009;193(3):807–812. doi: 10.2214/AJR.08.2229. [DOI] [PubMed]
- 7.De Ponti R, Lumia D, Marazzi R, et al. Left atrial diverticula in patients undergoing atrial fibrillation ablation: morphologic analysis and clinical impact. J Cardiovasc Electrophysiol. 2013;24(11):1232–1239. doi: 10.1111/jce.12213. [DOI] [PubMed]
- 8.Pachon M JC, Pachon M EI, Pachon M JC, et al. A new treatment for atrial fibrillation based on spectral analysis to guide the catheter RF-ablation. Europace. 2004;6(6):590–601. doi: 10.1016/j.eupc.2004.08.005. [DOI] [PubMed]
- 9.Pitts RM, Potts W. Congenital diverticulum of the left atrium. Arch Surg. 1962;84(3):334–336. doi: 10.1001/archsurg.1962.01300210068014. [DOI] [PubMed]
- 10.Nademanee K, Lockwood E, Oketani N, Gidney B. Catheter ablation of atrial fibrillation guided by complex fractionated atrial electrogram mapping of atrial fibrillation substrate. J Cardiol. 2010;55(1):1–12. doi: 10.1016/j.jjcc.2009.11.002. [DOI] [PubMed]
- 11.Yoshida K, Baba M, Shinoda Y, et al. Epicardial connection between the right-sided pulmonary venous carina and the right atrium in patients with atrial fibrillation: A possible mechanism for preclusion of pulmonary vein isolation without carina ablation. Heart Rhythm. 2019;16(5):671–678. doi: 10.1016/j.hrthm.2018.11.017. [DOI] [PubMed]
- 12.Maeda S, Kawabata M, Tsuda Y, Hirao K. Epicardial macroreentrant atrial tachycardia involving a large left atrial roof diverticulum: insights using high-resolution coherent mapping. Eur Heart J Case Rep. 2020;4(5):1–2. doi: 10.1093/ehjcr/ytaa268. [DOI] [PMC free article] [PubMed]
- 13.Verma A, Jiang CY, Betts TR, et al. Approaches to catheter ablation for persistent atrial fibrillation. N Engl J Med. 2015;372(19):1812–1822. doi: 10.1056/NEJMoa1408288. [DOI] [PubMed]