Abstract
Purpose
Most adult patients diagnosed with acute lymphoblastic leukemia (ALL) are below retirement age. The overall survival of patients with ALL has improved with implementation of high intensity pediatric-inspired treatment protocols. However, this treatment comes with a risk of long-term complications, which could affect the ability to work. The aim of this study was to investigate the risk of disability pension (DP) and return to work (RTW) for patients with ALL.
Patients and Methods
Patients aged 18–60 years diagnosed with ALL between 2005 and 2019 were identified in the Danish National Acute Leukemia Registry. Each patient was matched with five comparators from the general population on birth year, sex, and Charlson Comorbidity Index. The Aalen-Johansen estimator was used to calculate the cumulative risk of DP for patients and comparators from index date (defined as 1 year after diagnosis) with competing events (transplantation or relapse, death, retirement pension, or early retirement pension). Differences in cumulative incidences were calculated using Gray’s test. RTW was calculated as proportions one, three, and five years after the index date for patients holding a job before diagnosis.
Results
A total of 154 patients with ALL and 770 matched comparators were included. The 5-year cumulative risk of DP was increased fivefold for patients with ALL compared with the general population. RTW was 41.7%, 65.7%, and 60.7% one, three, and five years after the index date, respectively.
Conclusion
The risk of DP in patients with ALL increased significantly compared with the general population. Five years after the index date, RTW was 60.7% for patients with ALL.
Keywords: acute lymphoblastic leukemia, disability pension, return to work
Introduction
Acute lymphoblastic leukemia (ALL) is an aggressive hematologic malignancy.1 The overall survival of younger adult patients with ALL has increased within the last 15 years, which is linked to the introduction of high intensity pediatric-inspired treatment protocols for adults, and risk-adapted treatment algorithms.2–5 In Denmark, the median age at diagnosis is 56 years for adult patients with ALL.6 Hence, a substantial number of patients with ALL are below the Danish retirement age of currently 67 years.
The Danish welfare system provides economic compensation for Danish citizens who are sick or unable to work. In case of temporary health-related problems, sickness benefit will be granted until it is possible to return to work. People of working age who are permanently unable to work due to health-related issues can be granted a disability pension (DP).
The risk of long-term sick leave and early retirement is higher in cancer patients than in the general population.7,8 Studies furthermore suggest that the risk of DP is higher, and return to work (RTW) is lower in patients with acute leukemia compared with patients with a majority of other hematological malignancies. These studies, however, assessed patients with ALL and acute myeloid leukemia (AML) in combination and did not perform separate sub-group analyses for ALL and AML, respectively.9,10
The ability to work for patients with ALL may be affected by long-term complications due to new, intensified chemotherapy regimens for ALL patients of working age leading to increased risk of DP. Consequently, work disability may reduce patient income and thereby impact the societal economy. To our knowledge, no study has solely examined the risk of DP and RTW for patients with ALL.
We aimed at examining the risk of DP in patients with ALL compared to the general population. Furthermore, we aimed to assess RTW for patients with ALL who were employed before diagnosis.
Materials and Methods
Study Population
Patients with ALL were identified in the Danish National Acute Leukemia Registry. In brief, the registry covers clinicopathological information about patients diagnosed with acute leukemia (AML or ALL) in Denmark (ie, AML since 2000 and ALL since 2005). It was validated in 2013 using variables for AML, and coverage was 99.6%.11 The registry has been described in detail elsewhere.12
Patients aged 18–60 years diagnosed with ALL between January 1, 2005, and May 31, 2019, were included. The cutoff at 60 years was chosen to ensure appropriate time with connection to the labor market after ALL diagnosis and treatment. The index date was defined as 1 year after diagnosis, and patients had to be in complete remission and alive at the index date. Patients were excluded if they received DP, retirement pension, or early retirement pension before the index date. Finally, only patients living in Denmark at time of diagnosis and the index date were included.
Each patient with ALL was matched on sex, birth year, and level of Charlson Comorbidity Index with five comparators from the general population.13–15 The comparators were randomly sampled without replacement from the Danish Civil Registration System.16,17 Each patient’s index date and date of diagnosis were assigned to their matched comparators. Charlson Comorbidity Index was calculated 180 days before the date of diagnosis for patients and the corresponding date for comparators. Included comparators had no history of ALL, DP, retirement pension, or early retirement pension before the index date. Only comparators living in Denmark in the corresponding index date were included.
Data Collection
Several registries were used to access information about patients and comparators. Linkage between registries was possible using the unique civil registration number assigned to all Danish citizens.
The National Register on Social Transfer Payments (DREAM) contains weekly information from 1991 and prospectively about every person who has received welfare benefits.18 The date of DP, retirement pension, and early retirement pension was defined as the first day of the first week a person received the relevant welfare benefit in DREAM. A person was defined as employed during the weeks they were self-supporting (corresponding to weeks without welfare benefit codes in DREAM) or self-supporting with public support (including state education grant, leave of absence, and parental leave). Employment status could be affected by symptoms of ALL before diagnosis was made. Therefore, employment status was calculated between week 6 and week 14 prior to the date of diagnosis and defined as the status that applied in at least five of the nine weeks. To ensure the selected time slot was relevant, a sensitivity analysis was performed calculating the employment status between week 27 and 35 before diagnosis. This time span corresponded to the time of the calculation of the Charlson Comorbidity Index where patients and matched comparators were comparable regarding severe comorbidities.
Cohabitation status was collected from the Danish Civil Registration System the year before index date. Education level was extracted from the Danish Education Registries the year before index date and split into four groups according to the ISCED11 levels (ISCED 0–2, 3, 5–6, 7–8).19,20 ISCED level 4 does not exist in Denmark and was omitted. Data on income was extracted from the Income Statistics Register.21 Equalized income was calculated using the total disposable income of a household and adjusted using the OECD-modified equivalence scale.22 Level of income was then divided into four groups with regard to age and specific year at the time of income. The method for defining equalized income is described in further detail in Maksten et al.23
Treatment was divided into three groups: 1) high-intensity treatment, 2) standard intensity treatment/other, and 3) allogeneic stem cell transplantation. Patients treated with NOPHO ALL-2008 protocol were defined as high-intensity treatment (mainly patients under the age of 45 received this treatment).24 All other curative intended regimens were defined as standard intensity. Information on allogeneic stem cell transplantation was retracted from the Danish National Patient Registry.25,26 Patients having allogeneic stem cell transplantation were assigned the group allogeneic stem cell transplantation regardless of other treatment regimens given. The Danish National Pathology Registry27 was used to supplement the Danish National Acute Leukemia Registry concerning information about relapse.
Data on comorbidities from the Danish National Patient Registry was used to calculate Charlson Comorbidity Index. This was supplemented with information about dementia from the Danish National Psychiatric Central Research Register and information about prescription of diabetes medicine from the Danish National Prescription Registry.28–30
Patients or comparators diagnosed with ALL before 2005 were identified in the Danish Cancer Registry and excluded from this study.31
Statistical Analysis
Median follow-up time was calculated using reverse Kaplan-Meier.32 Patients and comparators were followed from the index date until DP, a competing event, or censoring occurred. Competing events were defined as relapse or transplantation, death, retirement pension, or early retirement pension for both patients and comparators. Patients and comparators were censored if they emigrated, or on the last day of follow-up, December 31, 2021.
The cumulative risk of receiving DP was calculated for patients and comparators using the Aalen-Johansen estimator.33 Gray's test was used to test the difference between cumulative risk differences.34 A sub-analysis was performed to test the difference between cumulative risk for transplanted patients with ALL and their matched comparators.
Risk differences in percentage points (%p) were calculated at 5 years and confidence intervals (95% CI) were calculated by pooling standard errors and assuming normality.
RTW was calculated as proportions and assessed only for patients and their matched comparators employed before diagnosis (or corresponding date of diagnosis). Employment status one, three and five years after the index date was calculated over 9 weeks, from 4 weeks before and 4 weeks after the anniversary week of diagnosis. Patients were excluded if they did not have at least one employed matched comparator. RTW was further assessed using Work Participation Score (WPS) with the number of weeks as self-supporting divided by the total number of weeks of follow-up.35 WPS was calculated during 52 weeks from the first to the second year after index. To enable this calculation, patients diagnosed after February 28, 2019, were excluded. Patients and comparators who received any kind of retirement pension or had a flexible job were also excluded. Finally, censored, emigrated, relapsed, or dead patients and comparators were excluded from both sub-analyses (RTW proportions and WPS).
SAS version 9.4 (SAS Institute Inc, Gary, North Carolina, USA) and R version 4.0.3 (R Foundation for Statistical Computing, Vienna, Austria) were used for data handling and statistical analysis. Data handling and statistical analysis were performed on a secured remote server at Statistics Denmark.36 Danish legislation on register-based research only requires approval from the register owners and registration in the regional research registry. The relevant approvals were obtained, and the study was registered in the research registry of the North Denmark Region (F2022-100).
Results
Patients and Baseline Characteristics
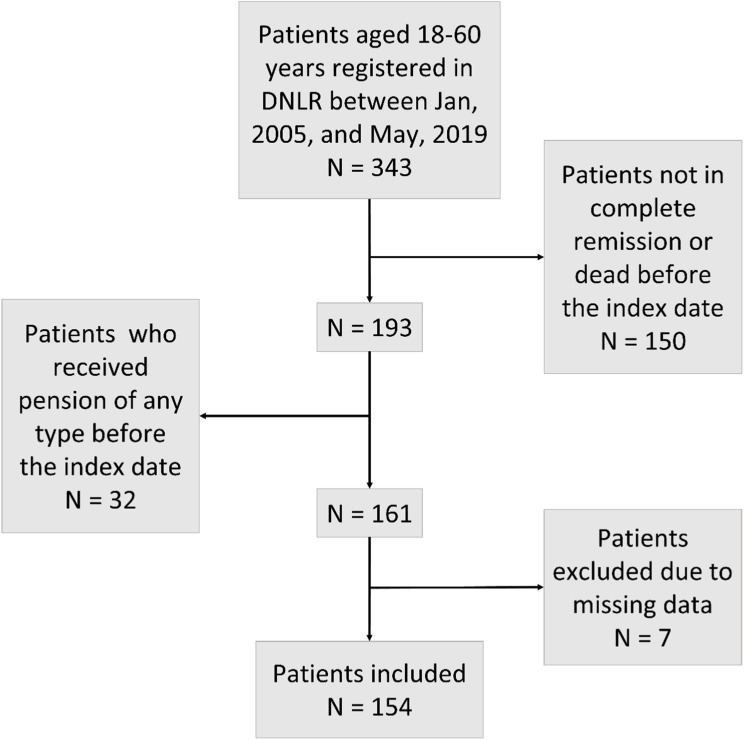
In total, 154 patients from the Danish National Acute Leukemia Registry (Figure 1) and 770 matched comparators from the general population were included. The median age was 33 years, and 64.9% were males. Employment status among ALL patients six to fourteen weeks prior to diagnosis showed that 85.7% were employed, whereas 5.2% were on sick leave. In comparison, 87.4% of comparators were employed, and 1.9% were on sick leave (Table 1). A sensitivity analysis estimating the employment status 27–35 weeks before diagnosis showed comparable results (for ALL 87.7% were employed, and 4.5% were on sick leave versus 86.8% and 2.1% for comparators). Patients with ALL were also more prone to have a minimum of four consecutive weeks of sick leave in the year before diagnosis (15.6%) compared to comparators (4.4%). The median follow-up time was 7.0 years for patients with ALL and 7.4 years for comparators.
Figure 1.
Flowchart. Inclusion of patients with ALL.
Abbreviation: DNLR, The Danish National Acute Leukemia Registry.
Table 1.
Baseline and Clinical Characteristics of Patients with ALL and Matched Comparators
| ALL (n=154) | Comparators (n=770) | |
|---|---|---|
| Age group, n (%) | ||
| 18–30 | 67 (43.5) | 335 (43.5) |
| 31–40 | 26 (16.9) | 130 (16.9) |
| 41–50 | 32 (20.8) | 160 (20.8) |
| 51–60 | 29 (18.8) | 145 (18.8) |
| Sex, n (%) | ||
| Male | 100 (64.9) | 500 (64.9) |
| Female | 54 (35.1) | 270 (35.1) |
| Cohabiting status, n (%) | ||
| Living alone | 53 (34.4) | 257 (33.4) |
| Living with partner | 101 (65.6) | 502 (65.2) |
| Unknown | 0 (0.0) | 11 (1.4) |
| Education level (ISCED), n (%) | ||
| ISCED 0–2 | 44 (28.6) | 218 (28.3) |
| ISCED 3 | 74 (48.1) | 321 (41.7) |
| ISCED 5–6 | 24 (15.6) | 142 (18.4) |
| ISCED 7–8 | 12 (7.8) | 67 (8.7) |
| Unknown | 0 (0.0) | 22 (2.9) |
| Equalized income, quartiles, n (%) | ||
| Lowest | 37 (24.0) | 190 (24.7) |
| Second lowest | 40 (26.0) | 169 (21.9) |
| Second highest | 39 (25.3) | 200 (26.0) |
| Highest | 38 (24.7) | 196 (25.5) |
| Unknown | 0 (0.0) | 15 (1.9) |
| Employment status, n (%) | ||
| Working* | 132 (85.7) | 673 (87.4) |
| Unemployed/Flexible job | 14 (9.1) | 70 (9.1) |
| Sick leave | 8 (5.2) | 15 (1.9) |
| Unknown | 0 (0.0) | 12 (1.6) |
| Long-term sickness, n (%)** | ||
| No | 130 (84.4) | 736 (95.6) |
| Yes | 24 (15.6) | 34 (4.4) |
| CCI prior to diagnosis, n (%) | ||
| 0 | 122 (79.2) | 610 (79.2) |
| ≥1 | 32 (20.8) | 160 (20.8) |
| ECOG Performance status, n (%) | ||
| 0 | 84 (54.5) | - |
| ≥1 | 70 (45.5) | - |
| ALL immunophenotype, n (%) | ||
| T-ALL | 40 (26.0) | - |
| B-ALL | 114 (74.0) | - |
| Treatment***, n (%) | ||
| High-intensity | 55 (35.7) | - |
| Standard intensity/Other | 44 (28.6) | - |
| Transplantation | 55 (35.7) | - |
| Year of diagnosis, n (%) | ||
| 2005–2009 | 38 (24.7) | - |
| 2010–2014 | 53 (34.4) | - |
| 2015–2019 | 63 (40.9) | - |
Notes: *Working group including receipt of state education grant, leave of absence, and parental leave. **4 or more consecutive weeks of sick leave within the last year before diagnosis (patients) or corresponding date (comparators). ***High-intensity: NOPHO ALL-2008 protocol, no transplantation. Standard intensity: All other curative intended regimens, no transplantation. Transplantation: All transplanted patients regardless of other treatment regimens.
Abbreviations: ISCED, International Standard Classification of Education; CCI, Charlson Comorbidity Index; ECOG, Eastern Cooperative Oncology Group.
Risk of Disability Pension
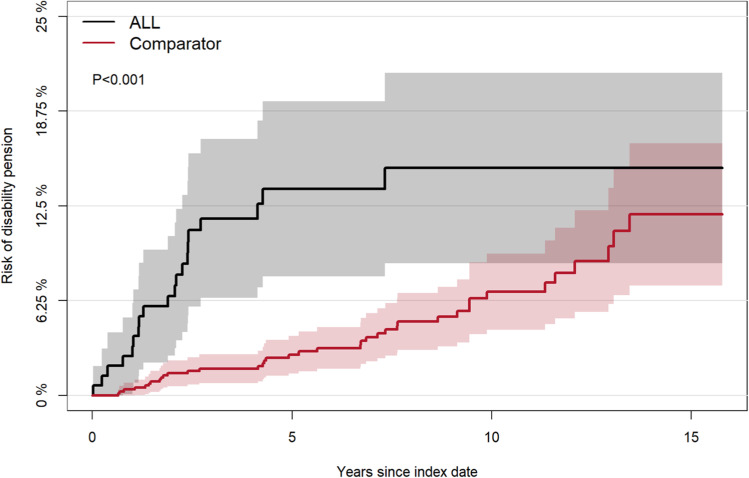
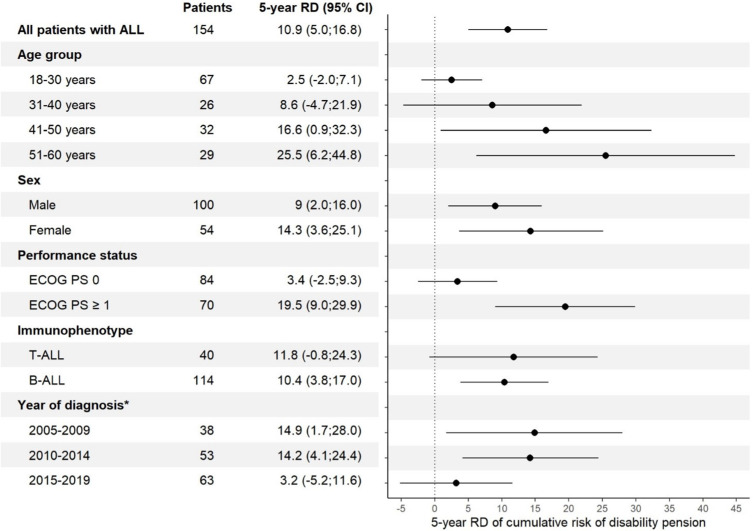
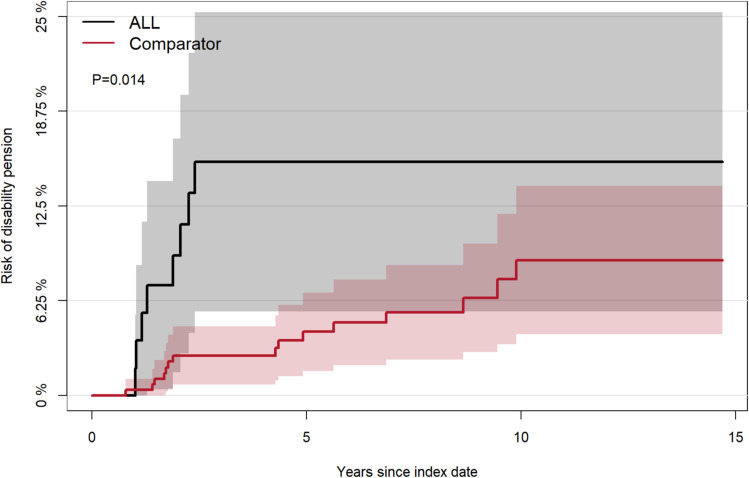
The risk of DP for patients with ALL was significantly higher (p<0.001) than in the general population (Figure 2). The 5-year cumulative risk of DP for patients with ALL was fivefold higher than for comparators. The 5-year cumulative risk for patients was 13.6% (95% CI: 7.9;19.4), and the 10-year cumulative risk was 15.0% (95% CI: 8.7;21.3). The comparators’ 5-year cumulative risk was 2.7% (95% CI: 1.5;3.9), and the 10-year cumulative risk was 6.9% (95% CI: 4.3;9.4). The 5-year risk difference of DP was significantly higher (10.9%p (95% CI: 5.0;16.8%p)) for all patients with ALL compared to comparators. An increase in risk difference was seen with increasing age (Figure 3). The cumulative risk of DP in the sub-analysis of transplanted patients with ALL was also higher than for their matched comparators (Figure 4). The cumulative risk of disability pension was increased in both males and females (Supplementary Figure 1).
Figure 2.
Cumulative risk of disability pension for patients with ALL and their matched comparators.
Figure 3.
The 5-year risk difference (RD) in percentage points of disability pension in subgroups of patients with ALL compared with comparators from the general population. *The Danish legislation on disability pension was changed in 2013 resulting in a decrease in number of disability pensions granted.
Abbreviations: RD, Risk difference; ECOG, Eastern Cooperative Oncology Group; PS, Performance score; T-ALL, T-cell ALL; B-ALL, B-cell ALL.
Figure 4.
Cumulative risk of disability pension for transplanted patients with ALL and their matched comparators.
Return to Work
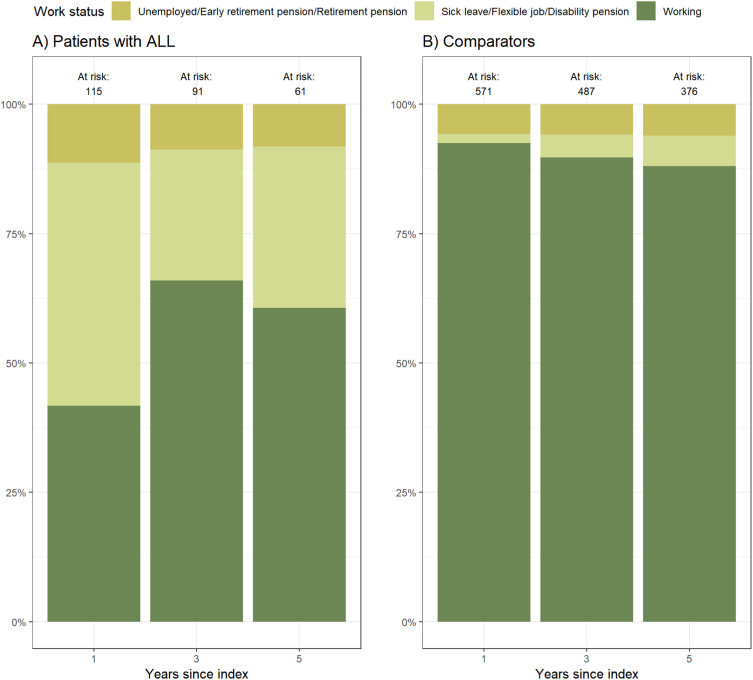
In total, 132 (85.7%) patients with ALL were employed before diagnosis. One year after the index date, 41.7% of the 132 patients had returned to work, while 47% had a flexible job, were on sick leave or disability pension. Three years after the index date, 65.9% had returned to work and 5 years after the index date, 60.7% had returned to work (Figure 5A and Supplementary Table 1). In comparison, 92.5%, 89.7%, and 88.0% of the matched comparators who held a job before the corresponding date of diagnosis were employed one, three, and five years after the index date, respectively (Figure 5B and Supplementary Table 1).
Figure 5.
Employment status one, three, and five years after index date for (A) Patients and (B) Comparators who worked before diagnosis.
In total, 95 patients and 404 comparators were included in the WPS analysis. Between the first and the second year after index, patients supported themselves for an average of 29 weeks and comparators supported themselves for an average of 48 weeks corresponding to a WPS of 55.1% and 91.9%, respectively.
Discussion
This study investigated the risk of DP and RTW in patients with ALL between 2005 and 2021. We found a fivefold higher 5-year risk of DP among ALL survivors compared with the comparators from the general population. To our knowledge, this is the first study to investigate the risk of DP and RTW for patients with ALL in a nationwide study.
Horsboel et al investigated the risk of DP in subgroups of hematological malignancies diagnosed between 2000 and 2007 and compared it with a reference cohort. They found a fourfold increased cumulative risk of DP for patients with AML or ALL 6 years after diagnosis.10 Meanwhile, our study found a fivefold increased 5-year cumulative risk of DP for patients with ALL after the index date (ie, 6 years after diagnosis). This difference may be explained by an improvement in overall survival. Nowadays, more patients survive ALL, but possibly with complications that prevent them from working. Moreover, Horsboel et al included patients aged 19–55 years, while we included adult patients aged up to 60 years. As increasing age heightens the risk of DP, this could also explain the difference.37 Carlsen et al found the overall risk of DP was threefold higher for patients with leukemia regardless of subtype.38 However, they included patients diagnosed between 1981 and 2000, and the overall survival has since improved. Finally, both Horsboel et al and Carlsen et al investigated the risk of DP in a mixed group of patients with both ALL and AML, and it is not known how a diagnosis with AML affects the risk of DP.
In our study, the risk of DP for transplanted patients with ALL was also higher than for their matched comparators (Figure 4). To our knowledge, there are no other data on risk of DP in transplanted patients with ALL specifically. However, Tichelli et al found that 37% of long-term survivors with various hematological malignancies who received allogeneic stem cell transplantation had received DP at follow-up 12 years after transplantation.39 Meanwhile, only 3.17% of the working general population received DP.
In 2013, a reform regarding DP was introduced by Danish law to keep more people in the workforce and reduce the number of granted DP, especially for people under the age of 40. Pedersen et al investigated the change in risk of DP in cancer patients and comparators before and after the reform. They concluded that the reform had reduced the cumulative incidence proportion of DP in cancer patients.40 However, the risk of DP for cancer patients was still higher than for the general public. In our study, the relative 5-year risk difference of receiving DP was significantly higher for patients diagnosed before 2014 compared to their matched comparators, whereas we did not find a significant difference for those diagnosed from 2015 to 2019. However, this is probably caused by short follow-up for these patients as only patients diagnosed before 2016 would have attained 5 years of follow-up. Furthermore, we found that the relative 5-year risk of DP was significantly higher for patients above the age of 40 at diagnosis compared to comparators. Age is a known risk factor,41 but the difference may also be explained by changes in treatment modalities, as the use of NOPHO-2008 protocol24 increased, especially for the youngest patients, during the study period. This could have affected both survival and treatment toxicity, which again could have affected the risk of DP.
Horsboel et al investigated RTW for patients with hematological malignancies aged 19–55 years and diagnosed between 2000 and 2007 in a national registry-based study.9 During the study period, RTW was 77% for patients with acute leukemia (ALL or AML). In comparison, we found that 60.7% of patients with ALL had returned to work 5 years after the index date. Horsboel et al investigated the cumulative incidence of RTW. Therefore, patients who initially returned to work but were granted DP or returned to sick leave afterwards were defined as returned to work. Combined with the 1-year longer follow-up, this may explain why RTW was higher in the study by Horsboel et al. On the contrary, our study was performed as a cross-sectional analysis. Therefore, patients would be defined exactly as the employment status they had at the time of assessment, and they could not be defined as returned to work after receiving DP. Furthermore, RTW is affected by many factors, eg, late toxicities and Quality-of-Life. Information on these factors was not available either in the present study or in the study by Horsboel et al. Differences in these factors between the two studies could also affect the outcome.
This study found WPS for patients with ALL to be 55.1% between the first and second years after index. No other study has investigated WPS in patients with ALL. In a study by Maksten et al, WPS for patients with lymphoma was 81.4% between the first and second years after diagnosis,23 while Pedersen et al found WPS 3 years after diagnosis to be 81.1% and 77.2% for colon cancer and rectal cancer, respectively.42 Differences in treatment length, method, and intensity across diagnoses may explain the difference in WPS. For instance, patients with ALL receive chemotherapy for a longer period, whereas some patients with colon or rectal cancer may have been cured with a less time-consuming treatment.24
This study has several strengths. It is a nationwide registry-based study with a high degree of completeness thanks to the Danish National Acute Leukemia Registry. Selection bias is low since registration in the Danish National Acute Leukemia Registry is mandatory for all hematological departments in Denmark. Other registries utilized have high coverage and registries such as the Danish Civil Registration System and DREAM are updated frequently,16,18 which secures improved follow-up. Finally, recall bias is reduced since the collected data is reported by health-care professionals or registered automatically from the public registries used for, eg, payment of social benefits. Another strength is the study design with matching as this reduces confounding; however, residual confounding cannot be ruled out.
The study also has limitations. We excluded patients who died or were not in complete remission a year after diagnosis to make it relevant to investigate work disability and RTW. Therefore, tight exclusion criteria were applied and only 154 patients with ALL were included in total.
Short-term sick leave may have been underestimated. In Denmark, the employer pays the first part of sick leave, which increased from lasting 14 to 30 days during the study period. Short sick leaves paid by the employer are only registered in DREAM if the sick leave extends beyond the period the employer pays. In that case, the whole sick leave period is registered in DREAM.
In our study, 5.1% of patients with ALL were on sick leave when employment status before diagnosis was calculated, while only 1.9% comparators were on sick leave. Since the sample size is small, few additional patients on sick leave quickly create a difference compared to comparators. However, we cannot rule out that patients with ALL were sicker than the comparators even though they were matched on Charlson Comorbidity Index. This is further made probable by the difference in long-term sickness (minimum four consecutive weeks) in the year before diagnosis as 15.4% of patients with ALL had a period of long-term sickness compared to only 4.4% of comparators. This higher proportion of sick leave could reflect a diagnostic delay and may have been driven the results toward a higher risk of DP for patients with ALL since receipt of welfare payments is a risk factor for DP. Lastly, DREAM contains information about received welfare benefits, but it does not register reasons for receiving them. Therefore, sick leave, flexible job, or grant of DP may be unrelated to ALL in some cases.
Conclusion
This study found that the 5-year cumulative risk of receiving disability pension for patients with ALL was fivefold higher than in the general population and that ALL survivors have a considerably reduced work ability. Furthermore, the study found that the return to work 5 years after the index date was 60.7% for patients with ALL compared to 88% for comparators.
Acknowledgments
The authors want to thank the doctors at the hematological departments in Denmark, who have provided data for the Danish National Acute Leukemia Registry.
Funding Statement
This study is supported by Dagmar Marshalls Foundation (E.F.M).
Disclosure
Eva Futtrup Maksten, Rasmus Rask Kragh Jørgensen, Kirsten Fonager, Ingolf Mølle, Andreas Due Ørskov, Ulrik Malthe Overgaard, and Marianne Tang Severinsen have no conflicts of interest to report in this work. Ms Mathilde Pedersen reports grants from Aalborg University, during the conduct of the study. Mrs Rie Sander Bech reports non-financial support from Pfizer, outside the submitted work. Mrs Claudia Schöllkopf reports personal fees from BMS, Incyte; virtual congress fee from Novartis, outside the submitted work. Tarec C. El-Galaly is a former employee of Roche. Gunhild Nynke Thomsen reports educational fee (podcast development) from Pfizer.
References
- 1.Paul S, Kantarjian H, Jabbour EJ. Adult acute lymphoblastic leukemia. Mayo Clin Proc. 2016;91(11):1645–1666. doi: 10.1016/j.mayocp.2016.09.010 [DOI] [PubMed] [Google Scholar]
- 2.Lennmyr E, Karlsson K, Ahlberg L, et al. Survival in adult acute lymphoblastic leukaemia (ALL): a report from the Swedish ALL registry. Eur J Haematol. 2019;103(2):88–98. doi: 10.1111/ejh.13247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dinmohamed AG, Szabó A, van der Mark M, et al. Improved survival in adult patients with acute lymphoblastic leukemia in the Netherlands: a population-based study on treatment, trial participation and survival. Leukemia. 2016;30(2):310–317. doi: 10.1038/leu.2015.230 [DOI] [PubMed] [Google Scholar]
- 4.DeAngelo DJ, Stevenson KE, Dahlberg SE, et al. Long-term outcome of a pediatric-inspired regimen used for adults aged 18–50 years with newly diagnosed acute lymphoblastic leukemia. Leukemia. 2015;29(3):526–534. doi: 10.1038/leu.2014.229 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Huguet F, Leguay T, Raffoux E, et al. Pediatric-inspired therapy in adults with Philadelphia chromosome–negative acute lymphoblastic leukemia: the GRAALL-2003 Study. JCO. 2009;27(6):911–918. doi: 10.1200/JCO.2008.18.6916 [DOI] [PubMed] [Google Scholar]
- 6.Marcher C, Friis LS, Ommen HB, Theilgaard-Mönch K, Holm MS, Andersen MK. The Danish National Acute Leukemia Registry. The Danish Clinical Quality Program – National Clinical Registries; 2021.
- 7.Torp S, Nielsen RA, Gudbergsson SB, Fosså SD, Dahl AA. Sick leave patterns among 5-year cancer survivors: a registry-based retrospective cohort study. J Cancer Surviv. 2012;6(3):315–323. doi: 10.1007/s11764-012-0228-8 [DOI] [PubMed] [Google Scholar]
- 8.Mehnert A. Employment and work-related issues in cancer survivors. Crit Rev Oncol/Hematol. 2011;77(2):109–130. doi: 10.1016/j.critrevonc.2010.01.004 [DOI] [PubMed] [Google Scholar]
- 9.Horsboel TA, Nielsen CV, Nielsen B, Jensen C, Andersen NT, de Thurah A. Type of hematological malignancy is crucial for the return to work prognosis: a register-based cohort study. J Cancer Survivorship. 2013;7(4):614–623. doi: 10.1007/s11764-013-0300-z [DOI] [PubMed] [Google Scholar]
- 10.Horsboel TA, Nielsen CV, Andersen NT, Nielsen B, de Thurah A. Risk of disability pension for patients diagnosed with haematological malignancies: a register-based cohort study. Acta Oncologica. 2014;53(6):724–734. doi: 10.3109/0284186X.2013.875625 [DOI] [PubMed] [Google Scholar]
- 11.Østgård LSG, Nørgaard M, Nørgaard JM, et al. Data quality in the Danish national acute leukemia registry: a hematological data resource. CLEP. 2013:335. doi: 10.2147/CLEP.S48411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ostgard L, Nørgaard JM, Raaschou-Jensen K, et al. The Danish national acute leukemia registry. CLEP. 2016;8:553–560. doi: 10.2147/CLEP.S99460 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–383. doi: 10.1016/0021-9681(87)90171-8 [DOI] [PubMed] [Google Scholar]
- 14.Sundararajan V, Quan H, Halfon P, et al. Cross-national comparative performance of three versions of the ICD-10 Charlson index. Med Care. 2007;45(12):1210–1215. doi: 10.1097/MLR.0b013e3181484347 [DOI] [PubMed] [Google Scholar]
- 15.Quan H, Sundararajan V, Halfon P, et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care. 2005;43(11):1130–1139. doi: 10.1097/01.mlr.0000182534.19832.83 [DOI] [PubMed] [Google Scholar]
- 16.Pedersen CB. The Danish civil registration system. Scand J Public Health. 2011;39(7_suppl):22–25. doi: 10.1177/1403494810387965 [DOI] [PubMed] [Google Scholar]
- 17.Pedersen CB, Gøtzsche H, Møller JO, Mortensen PB. The Danish civil registration system. A cohort of eight million persons. Dan Med Bull. 2006;53(4):441–449. [PubMed] [Google Scholar]
- 18.Hjollund NH, Larsen FB, Andersen JH. Register-based follow-up of social benefits and other transfer payments: accuracy and degree of completeness in a Danish interdepartmental administrative database compared with a population-based survey. Scand J Public Health. 2007;35(5):497–502. doi: 10.1080/14034940701271882 [DOI] [PubMed] [Google Scholar]
- 19.Jensen VM, Rasmussen AW. Danish education registers. Scand J Public Health. 2011;39(7_suppl):91–94. doi: 10.1177/1403494810394715 [DOI] [PubMed] [Google Scholar]
- 20.UNESCO Institute for Statistics. International Standard Classification of Education, ISCED 2011; 2012.
- 21.Baadsgaard M, Quitzau J. Danish registers on personal income and transfer payments. Scand J Public Health. 2011;39(7_suppl):103–105. doi: 10.1177/1403494811405098 [DOI] [PubMed] [Google Scholar]
- 22.OECD. What are equivalence scales? OECD Proj Income Distrib Poverty. 2011:1–2.
- 23.Maksten EF, Jakobsen LH, Kragholm KH, et al. Work disability and return to work after lymphoma: a Danish Nationwide Cohort Study. CLEP. 2023;15:337–348. doi: 10.2147/CLEP.S399488 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Overgaard U, Dimitrijevic A, Schöllkopf C, Bech RS Acute lymphoblastic leukemia - clinical guidelines. The Danish multidisciplinary cancer groups (www.dmsg.dk) and the Danish Clinical Quality Program - National Clinical Registries. 2022. [Google Scholar]
- 25.Lynge E, Sandegaard JL, Rebolj M. The Danish national patient register. Scand J Public Health. 2011;39(7_suppl):30–33. doi: 10.1177/1403494811401482 [DOI] [PubMed] [Google Scholar]
- 26.Schmidt M, Schmidt SAJ, Sandegaard JL, Ehrenstein V, Pedersen L, Sørensen HT. The Danish national patient registry: a review of content, data quality, and research potential. CLEP. 2015;449. doi: 10.2147/CLEP.S91125 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Erichsen R, Lash TL,Hamilton-Dutoit SJ, et al. Existing data sources for clinical epidemiology: the Danish national pathology registry and data bank. CLEP. 2010;51. doi: 10.2147/CLEP.S9908 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mors O, Perto GP, Mortensen PB. The Danish psychiatric central research register. Scand J Public Health. 2011;39(7_suppl):54–57. doi: 10.1177/1403494810395825 [DOI] [PubMed] [Google Scholar]
- 29.Wallach Kildemoes H, Toft Sørensen H, Hallas J. The Danish national prescription registry. Scand J Public Health. 2011;39(7_suppl):38–41. doi: 10.1177/1403494810394717 [DOI] [PubMed] [Google Scholar]
- 30.Pottegård A, Schmidt SAJ, Wallach-Kildemoes H, Sørensen HT, Hallas J, Schmidt M. Data resource profile: the Danish national prescription registry. Int J Epidemiol. 2016:dyw213. doi: 10.1093/ije/dyw213 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gjerstorff ML. The Danish cancer registry. Scand J Public Health. 2011;39(7_suppl):42–45. doi: 10.1177/1403494810393562 [DOI] [PubMed] [Google Scholar]
- 32.Schemper M, Smith TL. A note on quantifying follow-up in studies of failure time. Controlled Clin Trials. 1996;17(4):343–346. doi: 10.1016/0197-2456(96)00075-X [DOI] [PubMed] [Google Scholar]
- 33.Putter H, Fiocco M, Geskus RB. Tutorial in biostatistics: competing risks and multi-state models. Statist Med. 2007;26(11):2389–2430. doi: 10.1002/sim.2712 [DOI] [PubMed] [Google Scholar]
- 34.Gray RJ. A class of K-sample tests for comparing the cumulative incidence of a competing risk. Ann Statist. 1988;16(3). doi: 10.1214/aos/1176350951 [DOI] [Google Scholar]
- 35.Biering K, Hjøllund NH, Lund T. Methods in measuring return to work: a comparison of measures of return to work following treatment of coronary heart disease. J Occup Rehabil. 2013;23(3):400–405. doi: 10.1007/s10926-012-9405-x [DOI] [PubMed] [Google Scholar]
- 36.Thygesen LC, Daasnes C, Thaulow I, Brønnum-Hansen H. Introduction to Danish (nationwide) registers on health and social issues: structure, access, legislation, and archiving. Scand J Public Health. 2011;39(7_suppl):12–16. doi: 10.1177/1403494811399956 [DOI] [PubMed] [Google Scholar]
- 37.Karlsson N, Borg K, Carstensen J, Hensing G, Alexanderson K. Risk of disability pension in relation to gender and age in a Swedish county; a 12-year population based, prospective cohort study. Work. 2006;27(2):173–179. [PubMed] [Google Scholar]
- 38.Carlsen K, Oksbjerg Dalton S, Frederiksen K, Diderichsen F, Johansen C. Cancer and the risk for taking early retirement pension: a Danish cohort study. Scand J Public Health. 2008;36(2):117–125. doi: 10.1177/1403494807085192 [DOI] [PubMed] [Google Scholar]
- 39.Tichelli A, Gerull S, Holbro A, et al. Inability to work and need for disability pension among long-term survivors of hematopoietic stem cell transplantation. Bone Marrow Transplant. 2017;52(10):1436–1442. doi: 10.1038/bmt.2017.115 [DOI] [PubMed] [Google Scholar]
- 40.Pedersen P, Aagesen M, Tang LH, Bruun NH, Zwisler AD, Stapelfeldt CM. Risk of being granted disability pension among incident cancer patients before and after a structural pension reform: a Danish population-based, matched cohort study. Scand J Work Environ Health. 2020;46(4):382–391. doi: 10.5271/sjweh.3883 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Bruusgaard D, Smeby L, Claussen B. Education and disability pension: a stronger association than previously found. Scand J Public Health. 2010;38(7):686–690. doi: 10.1177/1403494810378916 [DOI] [PubMed] [Google Scholar]
- 42.Pedersen P, Laurberg S, Andersen NT, et al. Differences in work participation between incident colon and rectal cancer patients—a 10-year follow-up study with matched controls. J Cancer Surviv. 2022;16(1):73–85. doi: 10.1007/s11764-021-01005-x [DOI] [PubMed] [Google Scholar]