ABSTRACT
Raw area on the breast, especially when it is lactating, can lead to complications, including hyperprolactinemia and development of milk fistulae. A 25-year-old female presented with raw area over the left breast after 2 months of childbirth. She had history suggestive of necrotizing disease, which had primarily been managed elsewhere with debridement and dressings. We excised the raw area and applied split thickness skin grafts with minimal meshing. Bulky dressing prevented breastfeeding. On postoperative day 3, there were blebs containing milk underneath the graft. The blebs were drained and oral cabergoline was administered for 3 months. The skin graft healed well. If expression of breast milk is not possible then suppression of lactation should be considered before definitive cover of the raw area of breast.
KEY WORDS: Cabergoline, galactorrhea, hyperprolactinemia, infant, lactation, pain
Introduction
The breast is a specialized structure consisting of glandular tissue, ductules, the nipple, and a skin envelope. A raw area on this specialized structure, especially when it is lactating, can result in complications such as hyperprolactinemia, milk fistulae, and glandular loss.[1] Skin grafting procedure is usually performed for covering such raw area. We present this case where we encountered a vexing problem after skin grafting.
Case Report
A 25-year-old female presented with raw area over the lower part left breast after 2 months of childbirth. The patient had developed pain and diffuse, painful swelling in the left breast 1 month ago, for which she was prescribed antibiotics (no documentation present). The pain reduced in intensity, but the skin over the affected area of breast started turning dark and hard, like eschar. The patient underwent serial debridement and daily dressings at another hospital and presented to us with well-granulated raw area [Figure 1]. The patient had continued to breastfeed her child from the affected breast.
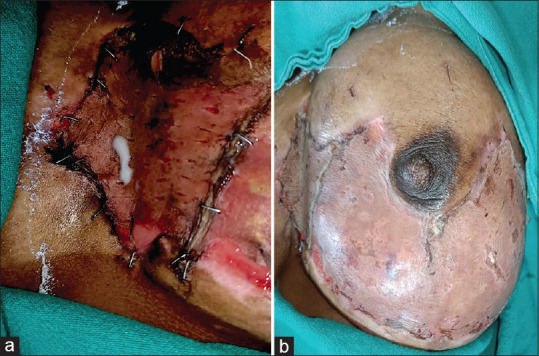
Figure 1.

(a) raw area on left breast and chest wall; (b) the skin graft with areas of milk collected under it on postoperative day 3
We took written informed consent for surgery, photography, and publication of photographs. After finding the routine investigations to be normal, we took the patient for surgery under general anesthesia. We did not perform bacterial cultures as it is not routinely advocated in healthy granulating wounds. We excised the raw area and applied a split-thickness skin graft with minimal meshing. A bulky bolster dressing was done, due to which the patient could not feed from the affected breast. On postoperative day 3, the dressing was removed for inspection of the graft. The skin graft was “taking” well, but there were areas of collected milk under the graft [Figure 2]. We punctured the graft in those areas and drained the collected milk. The use of dopamine antagonists such as antipsychotics, antihypertensives, antidepressants, and metoclopramide was excluded. Renal and thyroid function tests were repeated and found normal. We sought obstetrician’s opinion for this conundrum, and the patient was prescribed cabergoline. Tests for prolactin levels were not done. Further dressings were uneventful. The patient was discharged with full graft take and kept on oral cabergoline for a further 3 months. The baby was fed cow milk and was gaining weight at the last visit.
Figure 2.
(a) on incising over the “bleb,” clear milk was seen oozing out on postoperative day 3; (b) the skin graft was healing well on postoperative day 10
Discussion
Puerperal mastitis affects 1.4%–8.9% of nursing mothers.[2] When mastitis is treated within 24 h of onset, it is cured in few days. When treatment is delayed, it may progress to form breast abscess and extensive necrotizing disease.[2] In all cases of mastitis, the woman should be advised to nurse as frequently as possible from the affected breast as nursing dilates the mammary blood vessels, bringing in antibodies and antibiotics, and the flow of milk helps clear the lactiferous ducts from infective organisms.[3] If breastfeeding is not feasible, expression of milk by hand (if not very painful) or pump (mechanical or electrical) can be attempted. Frequent breastfeeding coupled with milk expression, as per case, is the best way to prevent progression of the disease. Milk stasis or blockage of lactiferous ducts by epithelial slough may lead to development of mammillary fistula.[1]
In addition to the existing hyperprolactinemia in a lactating mother, breast or chest injury can cause a 300%–400% increase in prolactin levels.[1] This hyperprolactinemia may be due to stimulation of nerves of thoracic wall, leading to response by anterior pituitary gland. This pathway has been reported to result in lactation, even in previously nonlactating breasts.[4] Transection or irritation of thoracic nerves following surgery may also induce milk production.[5] This theory is further supported by the success of intercostal blocks in cessation of post-surgical lactation.
In our case, because the mother was expressing her milk during the preoperative period, we reckoned that there was no milk stasis in the lobules and ductules. Intraoperatively, during excision of wound bed for graft application, some ductules must have gotten inadvertently injured. This, added to the fact that bulky dressing over the graft did not allow expression of milk, led to development of milk fistulae. Timely intervention, in the form of drainage of milk and administration of cabergoline, rescued the graft. van Heereden and Mabuza reported loss of skin graft, which in retrospect was determined to be due to galactorrhea from the wound bed.[6]
Cabergoline is a long-acting dopamine agonist drug that suppresses prolactin secretion.[7] Giele et al. allowed complete cessation of lactation before doing tangential excision and grafting in a case of burn of breast.[1] Post-surgical lactation usually begins within days of surgery and, without treatment, may last for weeks or months.[4] There is no literature as to the duration of time for which to suppress lactation. A review by Sharma and Basu[8] does not provide any insight to our dilemma. Rothkopf et al.[4] prescribed bromocriptine for 4 months as treatment of post-augmentation galactorrhea. We continued cabergoline for 3 months, assuming 3 months to be sufficient time for healing of the injured ductules by fibrosis. We figured that the development of immature scar, the inadvertent itching over the scar by the patient, and the act of massage over the scar may prove to be stimulus-inducing recurrent galactorrhea, and in 3 months’ time, all these would be fairly settled.
We realize the importance of expression of breastmilk before doing a definitive cover of the raw area of breast. If that is not possible, cessation of breastmilk is the last option. Not only is the risk of development of mamillary fistulae reduced but also the breast (and hence the raw area) reduces in size.
Conclusion
We report a case of collection of milk under the skin graft over lactating breast. Prompt recognition and therapy were critical in the management of our patient. This case presents a surgical and anatomical perspective related to milk-secreting ducts. Mastitis is a preventable complication among breastfeeding mothers. Surgical debridement needs to be done carefully. Expression of milk must be encouraged. Suppression of lactation should be the last resort.
Declaration of patient consent
The authors certify that appropriate patient consent was obtained.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- 1.Giele HP, Nguy?n H, Wood F, Crocker AD. Management of full thickness burns to lactating breasts. Burns. 1999;20:278–80. doi: 10.1016/0305-4179(94)90203-8. [DOI] [PubMed] [Google Scholar]
- 2.Cantlie HB. Treatment of acute puerperal mastitis and breast abscess. Can Fam Physician. 1988;34:2221–7. [PMC free article] [PubMed] [Google Scholar]
- 3.Ezrati JB, Gordon H. Puerperal mastitis: Causes, prevention and management. J Nurse Midwifery. 1979;24:3–8. doi: 10.1016/0091-2182(79)90027-2. [DOI] [PubMed] [Google Scholar]
- 4.Rothkopf DM, Rosen HM. Lactation as a complication of aesthetic breast surgery successfully treated with Bromocriptine. Br J Plast Surg. 1990;43:373–5. doi: 10.1016/0007-1226(90)90095-h. [DOI] [PubMed] [Google Scholar]
- 5.Berger RL, Joison J, Braverman LE. Lactation after incision on the thoracic cage. N Engl J Med. 1966;274:1493–5. doi: 10.1056/NEJM196606302742609. [DOI] [PubMed] [Google Scholar]
- 6.van Heereden M, Mabuza D. Galactorrhoea and hyperprolactinaemia in a non-pregnant female with burns. EJIFCC. 2020;31:354–61. [PMC free article] [PubMed] [Google Scholar]
- 7.Webster J, Piscitelli G, Polli A, Ferrari CI, Ismail I, Scanlon MF. A comparison of cabergoline and bromocriptine in the treatment of hyperprolactinemic amenorrhoea. N Engl J Med. 1994;331:904–9. doi: 10.1056/NEJM199410063311403. [DOI] [PubMed] [Google Scholar]
- 8.Sharma S, Basu NN. Galactorrhea/galactocele after breast augmentation: A systematic review. Ann Plast Surg. 2021;86:115–20. doi: 10.1097/SAP.0000000000002290. [DOI] [PubMed] [Google Scholar]


