Abstract
Purpose
Varicoceles can be a source of elevated seminal oxidative stress (OS) and sperm DNA fragmentation (SDF). However, it remains unclear whether varicocele repair (VR) could reduce these parameters. This systematic review and meta-analysis (SRMA) aims to investigate the impact of VR on SDF and seminal malondialdehyde (MDA).
Materials and Methods
A literature search was performed in Scopus, PubMed, Ovid, Embase, and Cochrane databases. This SRMA included randomized controlled trials and observational studies reporting the pre- and postoperative levels of SDF and seminal OS in infertile men with clinical varicocele that underwent VR. Subgroup analyses included techniques of VR and SDF testing. The effect size was expressed as standardized mean difference (SMD).
Results
Out of 1,632 abstracts assessed for eligibility, 29 studies with 1,491 infertile men were included. The analysis showed a significant reduction in SDF after VR, compared to preoperative values (SMD −1.125, 95% confidence interval [CI] −1.410, −0.840; p<0.0001) with high inter-study heterogeneity (I2=90.965%). Reduction in SDF was evident with microsurgical technique and non-microsurgical inguinal approaches (SMD −1.014, 95% CI −1.263, −0.765; p<0.0001, and SMD −1.495, 95% CI −2.116, −0.873; p<0.0001), respectively. Reduction in SDF was significant irrespective of testing was done by sperm chromatin dispersion (SMD −2.197, 95% CI −3.187, −1.207; p<0.0001), sperm chromatin structure assay (SMD −0.857, 95% CI −1.156, −0.559; p<0.0001) or TUNEL (SMD −1.599, 95% CI −2.478, −0.719; p<0.0001). A significant decrease in seminal MDA levels was observed following VR (SMD −2.450, 95% CI −3.903 to −0.997, p=0.001) with high inter-study heterogeneity (I2=93.7%).
Conclusions
Using pre- and post-intervention data, this SRMA indicates a significant reduction in SDF and seminal MDA levels in infertile men with clinical varicocele treated with VR. These findings may have important implications for the future management of this selected group of infertile patients.
Keywords: DNA fragmentation; Infertility, male; Malondialdehyde; Varicocele
INTRODUCTION
Infertility is defined as the inability of a couple to conceive a child after one year of regular, unprotected sexual intercourse [1]. It has been recognized as a universal public health issue by the World Health Organization (WHO), with current evidence indicating 9% prevalence and 56% of couples seeking medical care [2]. An estimated 48.5 million couples that have unprotected sexual intercourse suffer from infertility worldwide [3], with a male factor solely contributing to approximately 2.5% to 12% of couple infertility around the world [4].
Although semen analysis represents a crucial investigation in the infertile male, it remains sub-optimal in the evaluation and management of male infertility, with serious concerns regarding its validity in discerning between fertile and infertile men [5,6]. Sperm DNA fragmentation (SDF) has emerged as a useful tool in offering a more functional assessment of male fertility [7,8]. SDF is a marker of damaged chromatin in spermatozoa. Chromatin damage may include a broad spectrum of DNA defects such as single or double DNA strand breaks, base deletions or modifications, inter/intra-strand DNA cross-linkage, or protamine deficiency [9]. The rationale for assessing DNA integrity resides in the crucial role of chromatin compaction and structural stability in producing a fully functional sperm cell [10,11,12,13]. The recent WHO Laboratory Manual for the Examination and Processing of Human Semen 6th edition has subsequently dedicated an extensive narrative highlighting the importance of SDF assessment in male infertility management [14].
Several studies have demonstrated the association between elevated SDF and failure of natural pregnancy [13,15,16,17]. In a systematic review and meta-analysis (SRMA) on males from couples suffering from recurrent idiopathic miscarriage, SDF levels were reported to be higher compared to that of fertile controls (mean difference [MD]=11.98%, 95% confidence interval [CI] 6.64-17.32 p<0.001) [18]. Additionally, in the context of assisted reproductive technology (ART), SDF has been recognized as a factor that correlates with failed intra-uterine insemination (IUI) [19,20,21].
SDF is considered an indirect marker of oxidative stress (OS). OS is defined as the imbalance between the generation of reactive oxygen species (ROS) and the scavenger capacity. While low levels of ROS are needed for sperm development and for acquiring their fertilizing capacity, high levels or impairment of sperm antioxidant systems result in OS and infertility [22]. Spermatozoa are particularly vulnerable to OS, as ROS can disrupt the sperm cell membrane by lipoperoxidation and can lead to sperm protein and DNA damage. Seminal malondialdehyde (MDA) is a direct biomarker of OS, which reflects the degree of sperm cell lipoperoxidation by ROS and is paralleled with an increase of SDF and impairment of sperm motility [23]. Interestingly, sperm lipid peroxidation has been questioned as possibly involved in recurrent pregnancy loss (RPL). A study on 16 RPL cases indeed reported significantly higher levels of MDA in the patient group compared to fertile healthy controls [24], thus configuring a possible future role of this test in the diagnosis of male infertility. Varicocele is one of the most common correctable causes of male infertility, which is found in around 15% of healthy men, 35%–44% of men with primary infertility, and up to 45%–80% of men with secondary infertility [25,26,27]. Its prevalence rises with age and most varicocele develops during puberty [28,29]. Varicocele can cause male infertility through numerous mechanisms. Impairment of venous drainage may increase OS, increase scrotal temperature, induce testicular hypoxia, and cause abnormal reflux of metabolites [30]. Varicocele can cause up to 2.6 ℃ temperature elevation in the scrotum [31]. Increasing scrotal temperature has a detrimental effect on spermatogenesis, including increased SDF and impaired sperm fertilization capability [32].
Varicoceles are associated with increased OS which can lead to DNA damage in sperm [33]. Approximately 50% of men with clinically palpable varicocele have elevated SDF, and varicocele repair (VR) is associated with a decrease in SDF, especially in higher-grade varicoceles [34]. The reduction in SDF after VR may result in higher clinical pregnancy rates [35]. Despite evidence linking elevated SDF with varicoceles and improvement in SDF after VR, there is a paucity of recommendations in the major male infertility guidelines regarding VR in patients with a palpable varicocele and elevated SDF, as well as scenarios on when to obtain SDF testing.
Whether VR can influence seminal OS markers—particularly seminal MDA—is unknown. A better understanding of the impact of VR on SDF and OS—beyond the bulk semen parameters—can help guide the treatment of infertile men with varicoceles. The aim of this study is to conduct a SRMA to examine the impact of VR on SDF values as well as seminal MDA levels in infertile men.
MATERIALS AND METHODS
1. Search strategy
This meta-analysis was performed following the Meta-analysis Of Observational Studies in Epidemiology (MOOSE) guidelines [36] and the Preferred Reporting Items for SRMA Protocols (PRISMA-P) guidelines [37]. The data were extracted through extensive search in the Scopus, PubMed, Embase and Cochrane databases considering publications from the 1970s to December 2022. The search strategy included the combination of the following Medical Subjects Headings (MeSH) terms and keywords: varicoc*, management, embolization, mbolization, microsurg*, varicocelectomy, repair, correction, treatment, ligation, surg*, operation, radiolog*, sperm*, semen, seminal, ejaculate, asperm*, azoo*, azoosperm*, oligo*, oligosperm*, oligozoosperm*, astheno*, asthenosperm*, asthenozoosperm*, terato*, teratosperm*, teratozoosperm*, and necro*, necrosperm*, necrozoosperm*, sperm DNA fragmentation, SDF, DNA fragmentation index, DFI. The search was restricted to human studies and no language limitations were applied. The studies were initially assessed for inclusion by reading their titles and abstracts. When the abstract did not immediately reveal whether the study contained data relevant to our meta-analysis, the full text was read carefully. The evaluation for inclusion of the studies was performed by a team of 49 researchers selected after a training period, to allow an objective approach. In detail, each article was independently assessed for eligibility by two different researchers. Any disagreement was resolved by a third author. The protocol of this SRMA has been registered in the PROSPERO database (CRD42022329848).
2. Selection criteria
This review included all studies on infertile males with clinical varicocele until December 2022 that measured SDF, and included data on seminal MDA levels when available, in varicocele treated patients, whose pre-treatment values are used as control. All the eligible studies were chosen following the Population, Intervention, Comparison/Comparator, Outcomes, and Study type (PICOS) model (Supplement Table 1) [38]. Studies including adolescents, studies of subclinical varicocele, studies with no VR, in vitro studies, animal studies, case reports, case series, communications, and studies in which the control group received any treatment were excluded.
3. Data extraction
Data were extracted from the selected studies by 49 trained researchers distributed among four teams [39]. They worked in pairs, and a third senior author resolved disagreements. The following information was obtained from each study: authors, year of publication, study design, number of varicocele patients, assays used to assess sperm DNA integrity, type of VR, follow-up period, mean SDF value before treatment, mean SDF value after treatment, mean MDA seminal level before treatment and mean MDA seminal level after treatment. When all researchers had completed their extractions, we generated a combined database, and discrepancies were resolved by consensus among extractors under the control of the leader of each team.
4. Quality assessment
The quality of evidence (QoE) assessment aims to evaluate whether each article reports high-quality data. The QoE assessment of the articles was done by the Cambridge Quality Checklists [40]. In-depth, three checklists are devised for recognizing robust studies pertaining to associations, risk factors, and causal risk factors. The correlation checklist assesses sample size and outcome measurement quality. The risk factor checklist awards high scores to studies with suitable chronological data. The causal risk factor checklist evaluates study design. Strong inferences necessitate elevated scores across corresponding checklists: for associations, risk factors, and causal risk factors. Researchers double-checked the QoE assessment of all included articles. For each article, a screener made the initial assessment of QoE scores, a verifier checked the screener’s assessment, and a third expert affirmed the final agreed score.
5. Statistical analysis
1) Quantitative data analysis
For the meta-analysis, the Comprehensive Meta-Analysis software (version-2; Biostat Inc) was used. Both the standard mean difference (SMD) and the MD were used as the measure of effect size for statistical comparison between preoperative and postoperative values. To check the heterogeneity across pooled studies, the heterogeneity index (I2) was used. I2 is the representation of heterogeneity and it generally lies between 0 and 100%, where <50% represents low-to-moderate heterogeneity and >50% suggests high heterogeneity. Both the fixed effect and random effects models were used to calculate the pooled effect size. In the case of low heterogeneity, the fixed effect model was used to draw the statistical inference. In the case of >50% heterogeneity, the random effects model was acquired. For generating high-resolution forest plots, the SMD and CI were considered for both the “all studies” and the pooled data [41]. Additionally, we performed a meta-regression analysis to evaluate the effect of preoperative SDF rates on SDF differences before and after VR.
2) Sensitivity analysis
The method of one study removal at a time was used for sensitivity analysis. One study was excluded at a time and their pooled effect size and CI were calculated to determine if that study was sensitive. If the exclusion of a particular study resulted in a change of the inference, it was labeled as a ‘sensitive study’.
3) Publication bias analysis
Asymmetrical funnel plots were taken to qualitatively suggest the presence of publication bias. The presence of publication bias represents the missing studies from one side of the graph. We further used Egger’s intercept test, for representing publication bias quantitatively.
RESULTS
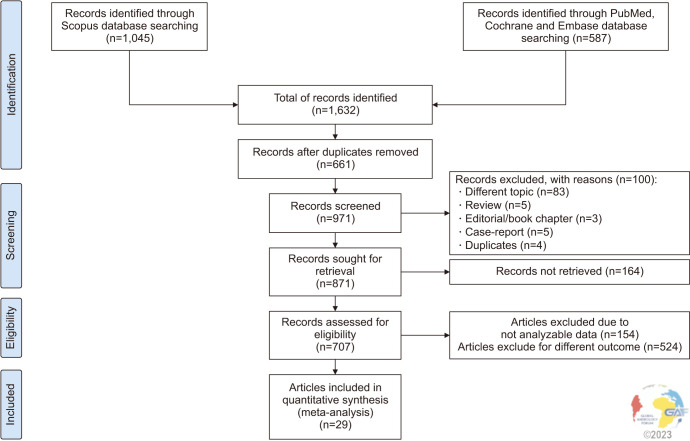
Using the aforementioned search strategy, 1,632 abstracts were extracted. After the elimination of 665 duplicates, 967 abstracts were assessed. Of these, 96 articles were identified by title and abstract as review articles, case reports, book chapters, papers on different topics, and were deemed not eligible. Of the remaining 871 articles, 164 full-texts were not found, and after reading the full-text, an additional 678 papers were excluded because they contained non-extractable data, and 524 for reporting different outcomes. Finally, 29 studies assessing the impact of VR on SDF and MDA were included in the present analysis (Fig. 1) [34,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69].
Fig. 1. Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA-P) flow-chart for inclusion/exclusion of studies.
Among the 29 included studies, 23 reported data on SDF, 5 reported MDA levels and one study reported both SDF and MDA levels before and after VR (Table 1). In three studies [50,53,55], the results were subdivided by grade of varicocele, in one study, by laterality of VR [51], in one study by different time to evaluation [64], and in one study the patients were subclassified after VR as responders or non-responders [34]. The main characteristics of the included studies are shown in Table 1. The QoE assessment is shown in Table 2.
Table 1. Main characteristics of the included studies.
| First author | Year | Study design | Varicocele | Varicocele repair | Time to evaluation (mo) | Patients (n) | SDF (%) | MDA (nmol/mL) | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Side | Grade | Type | Side | Before | After | Before | After | |||||
| Kavoussi [42] | 2021 | Observational | - | - | MS | - | 3 | 121 | 35.3±11.6 | 19.6±5.3 | ||
| Fathi [43] | 2021 | Observational | - | - | MS | - | 6 | 45 | 34.93±5.56 | 25.75±5.15 | ||
| Abbasi [44] | 2020 | Observational | - | - | MS | - | 3 | 22 | 13.65±11.44 | 10.38±4.64 | ||
| Kamal [45] | 2020 | Observational | - | - | NMI | - | 6 | 34 | 1.9±0.6 | 1.1±0.3 | ||
| Omar [46] | 2020 | Observational | - | - | MS | - | 3 | 100 | 7.7±1.1 | 2.2±0.6 | ||
| Mostafa [47] | 2020 | Observational | - | - | NMI | - | 3 | 30 | 3.4±1.3 | 1.9±0.7 | ||
| Camargo [48] | 2019 | Observational | - | - | MS | - | 12 | 25 | 43.4±22.62 | 32.9±8.87 | ||
| Afsin [49] | 2018 | Observational | - | - | - | - | 12 | 40 | 20.57±4.6 | 15.3±3.63 | ||
| Vahidi [50] | 2018 | Observational | Left | - | MS | - | 3 | 30 | 15.93±4.96 | 10.86±4.44 | ||
| 1 | MS | - | 3 | 5 | 15.4±9.42 | 5.2±1.09 | ||||||
| 2 | MS | - | 3 | 13 | 16±3.39 | 12.07±4.71 | ||||||
| 3 | MS | - | 3 | 12 | 16.08±4.46 | 11.91±3.14 | ||||||
| Sun [51] | 2018 | Observational | Bilateral | - | MS | Bilateral | 12 | 179 | 21.6±7.1 | 11.8±6 | ||
| Left | 12 | 179 | 23±8.1 | 12.1±6.8 | ||||||||
| Lu [52] | 2018 | Observational | Left | - | MS | Left | 6 | 27 | 1.75±0.22 | 0.58±0.06 | ||
| Zaazaa [53] | 2018 | RCT | - | - | MS | - | 3 | 40 | 34.6±4.1 | 28.3±5.2 | ||
| 2 | MS | - | 3 | 14 | 32.7±1.6 | 28±2.3 | ||||||
| 3 | MS | - | 3 | 26 | 35.5±4.7 | 28.5±6.3 | ||||||
| Abdelbaki [54] | 2017 | Observational | - | - | NMI | - | 6 | 55 | 29.49±8.58 | 18.78±7.23 | ||
| Ni [55] | 2016 | Observational | - | 1 | MS | - | 6 | 19 | 23.56±7.55 | 19.54±5.48 | 12.18±4.86 | 8.76±2.73 |
| 2 | MS | - | 6 | 18 | 27.75±9.05 | 22.38±4.54 | 14.12±5.42 | 9.71±2.83 | ||||
| 3 | MS | - | 6 | 14 | 30.03±8.27 | 21.82±5.95 | 15.86±6.78 | 9.5±3.28 | ||||
| Telli [56] | 2015 | Observational | - | - | NMI | - | 3 | 72 | 34.5±3.3 | 28.2±3.5 | ||
| Tavalaee [57] | 2015 | Observational | 1 | - | - | Left | 3 | 23 | 15.9±1.2 | 10.8±1.1 | ||
| Mohammed [58] | 2015 | Observational | - | - | NMI | - | 6 | 75 | 32.4±7.4 | 20±4.1 | ||
| Smit [59] | 2013 | Observational | - | - | - | - | 3 | 49 | 35.2±13.1 | 30.2±14.7 | ||
| Li [60] | 2012 | Observational | - | - | MS | - | 3 | 19 | 28.4±15.6 | 22.4±12.9 | ||
| Gabriel [61] | 2012 | Observational | - | - | MS | - | 4 | 14 | 22.4±11.9 | 12.6±6 | ||
| La Vignera [62] | 2012 | Observational | Left | 3 | MS | Left | 4 | 30 | 5±3 | 2.1±0.4 | ||
| Sadek [63] | 2011 | Observational | Bilateral | - | NMI | - | 3 | 72 | 65.15±20.97 | 51.9±15.2 | ||
| Zini [64] | 2011 | Observational | - | - | MS | - | 4–6 | 25 | 18±11 | 11±6 | ||
| 4 | 19 | 18±11 | 10±5 | |||||||||
| 6 | 19 | 18±11 | 7±3 | |||||||||
| Ghazi [65] | 2011 | Observational | Bilateral | - | MS | Bilateral | 6 | 82 | 21.5±11.2 | 13±14.3 | ||
| Dada [66] | 2010 | Observational | - | - | - | - | 6 | 11 | 60.82±7.12 | 32.58±6.12 | ||
| Nasr Esfahani [67] | 2010 | Observational | - | 3 | - | - | 6 | 70 | 45.69±2.43 | 35.9±2.4 | ||
| Smit [34] | 2010 | Observational | - | - | - | - | 3 | 31a | 35.3±14.3 | 28.6±14.7 | ||
| 18b | 35±11.2 | 33±14.8 | ||||||||||
| Zini [68] | 2005 | Observational | - | - | MS | - | 6 | 37 | 27.7±17.64 | 24.6±16.42 | ||
| Yeşilli [69] | 2005 | Observational | - | - | MS | - | 6 | 26 | 0.61±0.09 | 0.58±0.06 | ||
Values are presented as mean±standard deviation.
MDA: malondialdehyde, MS: microsurgical varicocele repair, NMI: non-microsurgical inguinal varicocele repair, RCT: randomized controlled trial, SDF: sperm DNA fragmentation.
aResponders to varicocele repair (improvement >50% in sperm concentration), bNon-responders to varicocele repair.
Table 2. Quality of evidence assessment (results of the Cambridge Quality Checklist [40]).
| First author | Year | Cambridge Quality Checklist | Total Quality score (3–15) | ||
|---|---|---|---|---|---|
| Checklist for correlates (1–5) | Checklist for risk factors (1–3) | Checklist for causal risk factor (1–7) | |||
| Kavoussi [42] | 2021 | 1 | 2 | 4 | 7 |
| Fathi [43] | 2021 | 2 | 3 | 4 | 9 |
| Abbasi [44] | 2020 | 1 | 2 | 3 | 6 |
| Kamal [45] | 2020 | 2 | 3 | 6 | 11 |
| Omar [46] | 2020 | 1 | 3 | 3 | 7 |
| Mostafa [47] | 2020 | 2 | 3 | 3 | 8 |
| Camargo [48] | 2019 | 1 | 3 | 3 | 7 |
| Afsin [49] | 2018 | 3 | 3 | 3 | 9 |
| Vahidi [50] | 2018 | 2 | 3 | 3 | 8 |
| Sun [51] | 2018 | 2 | 3 | 3 | 8 |
| Zaazaa [53] | 2018 | 2 | 3 | 7 | 12 |
| Lu [52] | 2018 | 2 | 3 | 3 | 8 |
| Abdelbaki [54] | 2017 | 2 | 3 | 4 | 9 |
| Ni [55] | 2016 | 3 | 3 | 4 | 10 |
| Telli [56] | 2015 | 3 | 3 | 3 | 9 |
| Tavalaee [57] | 2015 | 2 | 3 | 3 | 8 |
| Mohammed [58] | 2015 | 3 | 3 | 3 | 9 |
| Smit [59] | 2013 | 3 | 3 | 3 | 9 |
| Li [60] | 2012 | 1 | 3 | 3 | 7 |
| Gabriel [61] | 2012 | 3 | 3 | 3 | 9 |
| La Vignera [62] | 2012 | 3 | 3 | 3 | 9 |
| Sadek [63] | 2011 | 3 | 3 | 4 | 10 |
| Zini [64] | 2011 | 3 | 3 | 3 | 9 |
| Ghazi [65] | 2011 | 2 | 3 | 3 | 8 |
| Dada [66] | 2010 | 0 | 3 | 5 | 8 |
| Nasr Esfahani [67] | 2010 | 2 | 3 | 6 | 11 |
| Smit [34] | 2010 | 3 | 3 | 3 | 9 |
| Zini [68] | 2005 | 4 | 2 | 3 | 9 |
| Yeşilli [69] | 2005 | 1 | 3 | 4 | 8 |
1. Sperm DNA fragmentation
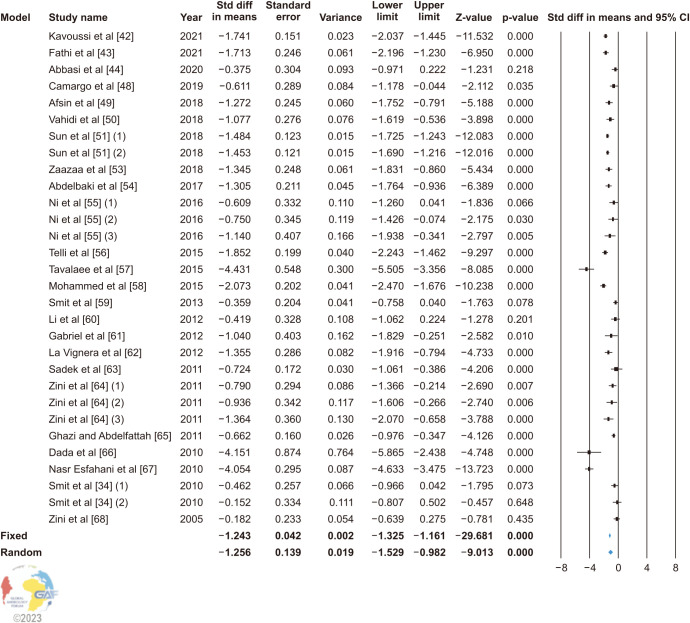
A total of 23 studies reported the SDF differences between before and after VR. Twenty-three studies showed that there was a significant change in SDF in varicocele patients after treatment. Overall, the random effect model (heterogeneity: I2=90.965%) revealed that the SDF was significantly reduced after VR (SMD: −1.256; 95% CI −1.529, −0.982; p<0.0001; MD: −6.924; 95% CI −8.438, −5.410; p<0.001) (Fig. 2).
Fig. 2. Forest plot of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair. 95% CI: 95% confidence interval.
Sensitivity analysis was performed, but no study was sensitive enough to change the conclusion that VR significantly decreases the SDF in the random model (Supplement Fig. 1). Publication bias was not apparent for all studies in the meta-analyses of SDF based on funnel plot and Egger’s test (p=0.298) (Supplement Fig. 2).
1) Subgroup analysis: technique of varicocele repair
Subgroup analysis was performed based on the type of VR (including microsurgical, non-microsurgical inguinal, high retroperitoneal, laparoscopic, embolization, and others).
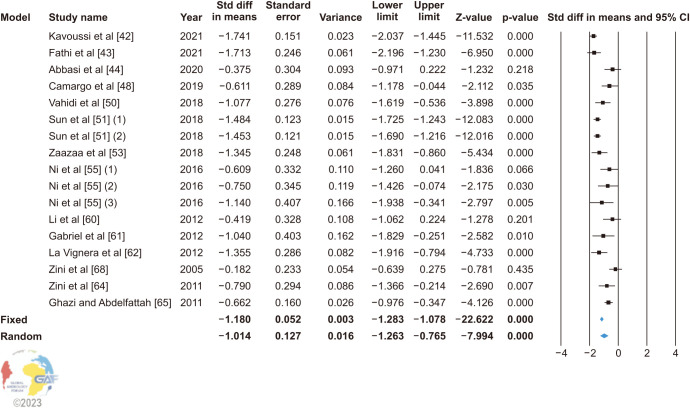
In 13 studies using microsurgical treatment, the random effect model (heterogeneity: I2=80.517%) showed that VR significantly decreases SDF (SMD: −1.014; 95% CI: −1.263, −0.765; p<0.0001) (Fig. 3).
Fig. 3. Forest plot of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair using microsurgical treatment. 95% CI: 95% confidence interval.
Sensitivity analysis was performed but no study was sensitive enough to change the conclusion that microsurgical repair of varicocele significantly decreases SDF (Supplement Fig. 3). Publication bias was apparent in those studies according to the funnel plot and Egger’s test (p=0.028). By bias correction, the adjusted effect value indicated that SDF in varicocele men was robustly reduced after microsurgical repair (Supplement Fig. 4).
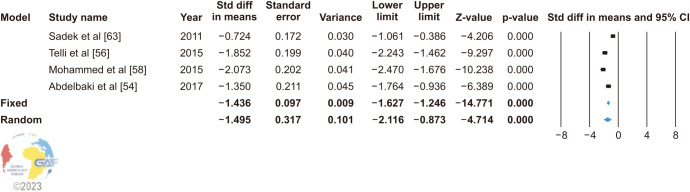
In four studies using the non-microsurgical inguinal method, the random effect model (heterogeneity: 90%–50%) showed that VR significantly decreases SDF (SMD: −1.495; 95% CI: −2.116, −0.873; p<0.0001) (Fig. 4).
Fig. 4. Forest plot of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair using non-microsurgical inguinal treatment. 95% CI: 95% confidence interval.
2) Subgroup analysis: technique of sperm DNA fragmentation analysis
Additionally, subgroup analysis was checked according to the techniques used in SDF estimation (including sperm terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate-nick end labeling [TUNEL], sperm chromatin structure assay [SCSA], chromatin dispersion assay [SCD] and others).
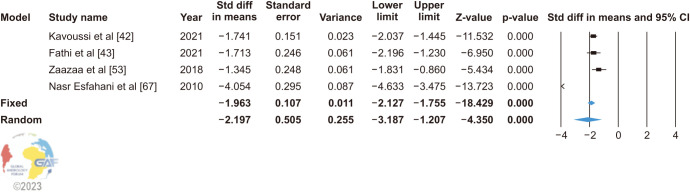
In four studies using SCD to measure SDF, the random effect model (heterogeneity: I2=94.958%), showed that VR significantly decreases the SDF (SMD: −2.197; 95% CI: −3.187, −1.207; p<0.0001) (Fig. 5). Sensitivity analysis was performed but no study was sensitive enough to change the main result (Supplement Fig. 5). Funnel plot and Egger’s test (p=0.487) found that there was no publication bias in the four papers (Supplement Fig. 6).
Fig. 5. Forest plot of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair using SCD. 95% CI: 95% confidence interval.
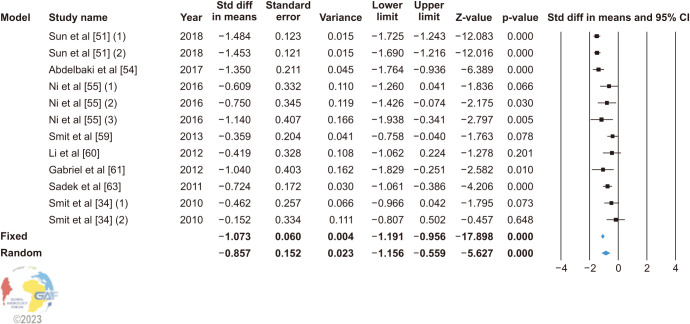
In 6 studies using SCSA to analyze SDF, the random effect model (heterogeneity: I2=81.446%) showed there was a significant decrease in SDF (SMD: −0.857; 95% CI: −1.156, −0.559; p<0.0001) (Fig. 6).
Fig. 6. Forest plot of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair using SCSA. 95% CI: 95% confidence interval.
Sensitivity analysis was performed but did not reveal a change in the main meta-analysis result (Supplement Fig. 7). Publication bias was apparent in those studies according to the funnel plot and Egger’s test (p=0.022). By bias adjustment, the corrected effect value indicated that SDF in varicocele men was robustly reduced after surgical repair when using SCSA testing (Supplement Fig. 8).
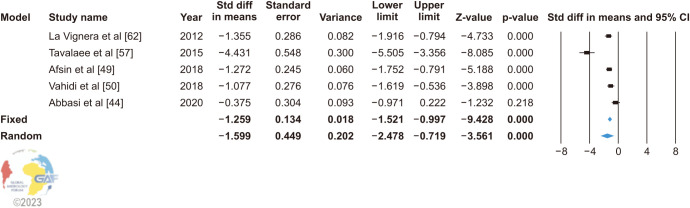
In five studies using TUNEL to measure SDF, the random effect model (heterogeneity: I2=90.588%) showed that VR significantly decreases SDF (SMD: −1.599; 95% CI: −2.478, −0.719; p<0.0001) (Fig. 7). Sensitivity analysis was performed but no study was sensitive enough to change the conclusion (Supplement Fig. 9). The funnel plot and Egger’s test (p=0.116) found that there was no publication bias in the five papers (Supplement Fig. 10).
Fig. 7. Forest plot of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair using TUNEL. 95% CI: 95% confidence interval.
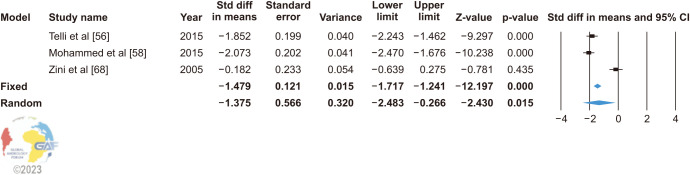
In the three studies which did not describe the specific testing methods for SDF, the random effect model (heterogeneity: I2=95.361%) showed that VR significantly improved the SDF (SMD: −1.375; 95% CI: −2.483, −0.266; p=0.015) (Fig. 8). Sensitivity analysis was performed but no study was sensitive enough to change the conclusion (Supplement Fig. 11). The funnel plot and Egger’s test (p=0.137) found that there was no publication bias in the three papers (Supplement Fig. 12).
Fig. 8. Forest plot of the sperm DNA fragmentation (SDF) in infertile patients after varicocele repair compared to before varicocele repair using a not specified methods for analysis of SDF. 95% CI: 95% confidence interval.
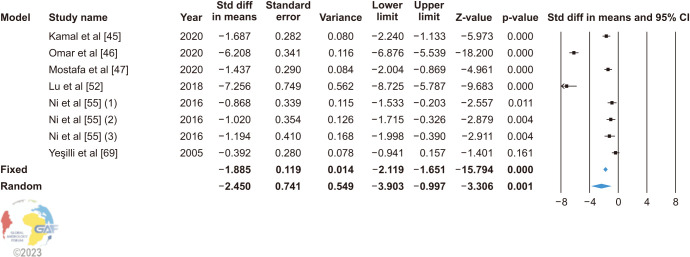
2. Malondialdehyde
A total of 6 studies analyzed seminal MDA as an outcome before and after VR in infertile men. The random effect model (heterogeneity: I2=97.3%) showed that VR significantly reduced seminal MDA levels (SMD: −2.450; 95% CI: −3.903, −0.997; p=0.001; MD: −2.394; 95% CI −2.524, −1.275; p<0.001) (Fig. 9). As an overall, there appeared to be no study that was sensitive enough to alter the findings that VR lowers seminal MDA levels (Supplement Fig. 13). Based on the symmetry of the standard error and precision Funnel plots respectively (Supplement Fig. 14), as well as the Egger’s test (p>0.05), no publication bias was apparent.
Fig. 9. Forest plot of seminal malondialdehyde level in infertile patients after varicocele repair compared to before varicocele repair. 95% CI: 95% confidence interval.
3. Meta-regression analysis
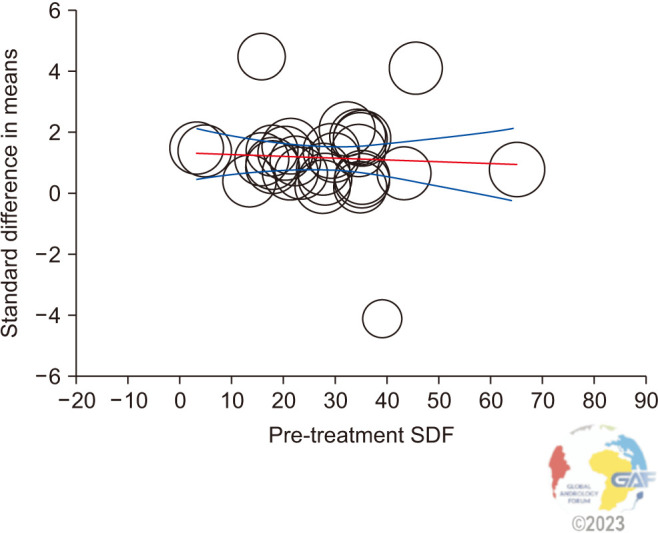
The meta-regression analysis revealed that the SDF reduction after VR was independent of the preoperative SDF levels. In the present analysis, the magnitude of the effect size did not increase as a function of preoperative SDF levels (coefficient: −0.01; 95% CI: −0.03, 0.02; p=0.64) (Fig. 10).
Fig. 10. Metaregression showing the influence of pre-treatment sperm DNA fragmentation (SDF) levels (moderator) on the standard difference of the mean of SDF after varicocelectomy. The size of the circles indicates the studies’ sample sizes. The red line represents linear predictions for the standard mean difference of SDF change after varicocelectomy as a function of the mean absolute increase in preoperative SDF levels. The curved lines indicate the 95% confidence interval lines around the regression line.
DISCUSSION
Varicocele is linked with DNA damage of spermatozoa due to elevated OS and decreased antioxidant capacity. In this meta-analysis, we strove to evaluate the effect of VR on SDF and seminal MDA levels in infertile patients with clinical varicocele. For SDF, a total of 23 studies were found to investigate the SDF differences before and after VR and a significant reduction was seen in SDF levels after VR. The improvement in SDF levels after VR emerged regardless of the surgical technique and methods that were used in SDF estimation. For the MDA, a total of six studies were included in the analysis, finding a significant decrease after VR.
The American Urological Association/American Society for Reproductive Medicine (AUA/ASRM) guideline on male infertility suggests performing VR in the setting of clinically palpable varicocele, infertility, and abnormal sperm parameters on semen analysis [70]. This guideline does not specifically address the role of VR in men with abnormal SDF. The European Association of Urology (EAU) male infertility guideline states that varicoceles can be a source of elevated SDF, and VR can be considered in men with increased SDF with abnormal semen parameters [71]. It is unclear whether VR should be offered to men who have elevated SDF in the setting of normal semen parameters, unexplained infertility, or those who have suffered from failed ART or RPL [71].
The lack of specific guidelines concerning the benefits and indications of VR for male infertility may raise doubts as to the implementation of this therapy in daily clinical practice [71]. Several studies have shown that there was an increase in SDF in varicocele cases and that VR could reduce these SDF levels [34]. Furthermore, increased levels of MDA can also be used as an indicator of high OS which is also one of the proposed effects of varicocele. Additionally, significant improvement was seen in MDA levels after VR, indicating a reduction in OS.
Of the 6 studies examined in the SRMA on MDA, all were found to show a significant reduction after VR. Previous studies have shown that MDA levels are increased in men with varicocele, and it has been shown that VR reduces MDA levels [45]. When calculated cumulatively with this SRMA, the increase in MDA levels in varicocele patients and the decrease after VR support the concept that VR decreases the levels of sperm lipoperoxidation, which can concur with loss of pregnancy [24].
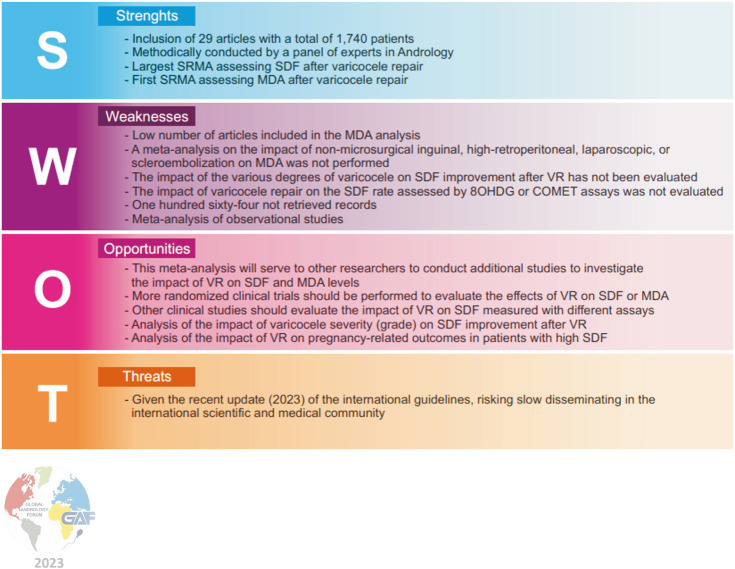
We examined the strengths of our MA compared to previously published SRMA (Table 3 and Fig. 11). Baazeem et al [72] in their meta-analysis took into account only 3 prospective studies about sperm DNA damage. In those studies, all the investigators recorded pre- and post-VR sperm DFI (DNA fragmentation index). Two out of 3 studies found a statistically significant decrease in DFI postoperatively, however, in the remaining study, only 10 out of 25 patients completed the follow-up so the improvement in DNA fragmentation was not significant. In our opinion, the strength of this meta-analysis is that all the articles involved used the same method (SCSA) to measure the DFI for better comparison. Another recent meta-analysis involved 11 prospective studies with various methods of DNA damage measurement [73]. In the present meta-analysis, patients served as their controls, as preoperative parameters were used as control values. On average, the DFI % of clinical varicocele patients decreased by 5.79 (95% CI, −7.39 to −4.19) after VR. Furthermore, in the fixed effect model for 10 studies (number of participants=364, heterogeneity chi-squared=15.15, df=9, p=0.087, I2=40.6%), DFI% decreased by 6.14 (95% CI, −6.90 to −5.37) on average.
Table 3. Comparison with previous meta-analyses.
| Study (publication year) | No. of studies/patients (total) | Study designs included in SRMA | The measure of sperm DNA damage | Outcomes |
|---|---|---|---|---|
| Present study | 29/1,523 | Prospective and retrospective and unspecified | SCSA, SCD, TUNEL, and others | Varicocele repair reduces sperm DNA damage and MDA levels. |
| 6/268a | 8OHDG: Didn’t qualify for meta-analysis | |||
| Comet: Didn’t qualify for meta-analysis | ||||
| Lira Neto et al (2021) [76] | 19/1,070 | Prospective and retrospective | SCSA, TUNEL, SCD, Comet | Varicocele repair reduces sperm DNA damage. |
| Birowo et al (2020) [75] | 7/289 | Prospective | SCSA, TUNEL | Varicocele repair reduces sperm DNA damage. |
| Qiu et al (2021) [73] | 11/394 | Prospective | SCSA, TUNEL, SCD, Comet, AOT | Varicocele repair reduces sperm DNA damage. |
| Wang et al (2012) [74] | 6/177 | Prospective, retrospective, and unspecified | SCSA, TUNEL, Comet | Varicocele repair reduces sperm DNA damage. |
| Baazeem et al (2011) [72] | 3/84 | Prospective | SCSA | Varicocele repair reduces sperm DNA damage. |
SRMA: systematic review and meta-analysis, SCSA: sperm chromatin structure assay, SCD: sperm chromatin dispersion, TUNEL: terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate-nick end labeling, 8OHDG: 8-hydroxydeoxyguanosine, AOT: acridine orange test.
aThis number represents MDA studies only.
Fig. 11. Strengths, Weaknesses, Opportunities, and Threats (SWOT) Analysis. SRMA: systematic review and meta-analysis, SDF: sperm DNA fragmentation, MDA: malondialdehyde, VR: varicocele repair, 8OHDG: 8-hydroxydeoxyguanosine.
In their 2012 MA, Wang and colleagues reported that patients with varicoceles had significantly higher sperm DNA damage than controls, with a MD of 9.84% (95% CI 9.19–10.49; p<0.00001). Authors stated that a VR performed with either microsurgical or an unspecified method can improve sperm DNA integrity, with a MD of 3.37% (95% CI −4.09 to −2.65; p<0.00001) [74].
In another recent meta-analysis [75], the sperm DFI decreased by 6.86% after VR (MD: −6.86; 95% CI: −10.04, −3.69; p<0.00001). SCSA was the preferred method in most of the included studies, with the exception of two studies that used the TUNEL assay.
The most recent and largest MA on this topic was reported by Lira Neto et al [76] in 2021. VR was associated with postoperative decreased SDF rates (weighted mean difference −7.23%; 95% CI: −8.86 to −5.59; I2=91%). The pooled results were consistent for studies using SCSA, TUNEL, SCD, Comet, and microsurgical VR which in our opinion is a strength of this SRMA. Subgroup analyses in the latter study demonstrated a more pronounced treatment effect in men with elevated preoperative SDF levels. Indeed, their meta-regression analysis showed that SDF decreased after VR as a function of preoperative SDF levels (coefficient: 0.23; 95% CI 0.07–0.39). Surprisingly, the present study found that SDF reduction after VR was independent of the preoperative SDF levels. In other words, this indicates that the extent of SDF improvement after VR is similar in both patients with either high and low SDF. The reasons for the finding of opposing results are not immediately clear. The methods used in this study are very similar to those used in the study by Lira Neto et al [76]. However, having been conducted more recently, it could be supposed that the increase in the size of the population examined in the present study (which is the largest examined so far) could represent one of the main factors responsible for the results.
Till now, five meta-analyses were conducted with a total of 2,014 patients investigating the effect of VR on SDF. Four meta-analyses included prospective studies and one meta-analysis included both prospective and retrospective studies. All meta-analyses indicate that sperm DNA damage is reduced after VR. Our current meta-analysis carried out on 1,523 patients (considering only studies on SDF), confirms and supports the current literature, adding 453 patients.
Up until now, there was no meta-analysis in the literature investigating the effect of VR on seminal levels of MDA in infertile patients with clinical varicocele. An interesting aspect of our meta-analysis is that it reports, for the first time, a reduction in MDA after VR.
Before concluding we would like to underline the strength and limitations of this study using a SWOT (strengths, weaknesses, opportunities, and threats) analysis (Fig. 11). The strengths of the present study were the inclusion of 29 articles that collected a total of 1491 patients. Additionally, we conducted subgroup analyses to assess heterogeneity and conducted the sensitivity analysis. Finally, our SRMA is the first one to have meta-analyzed the effects of VR on MDA. One limitation was the fact that our meta-analysis did not compare the impact of one VR technique to another. Furthermore, we did not evaluate the impact of different degrees of varicocele on SDF improvement after VR. Finally, we were unable to assess the impact of VR on the SDF rate as evaluated by the 8-hydroxydeoxyguanosine or Comet assays. The results of this SRMA should be a guide for professional organizations connected with the field of male infertility to further guide on the need for VR in men with not only quantitative indicators of male infertility in the conventional semen analysis but qualitative indicators of sperm dysfunction via SDF and MDA.
CONCLUSIONS
By analyzing the largest sample so far, this SRMA provides additional evidence that VR significantly improves SDF. Reductions of SDF were observed independent of the surgical technique, type of SDF testing assay, or preoperative SDF values. To the best of our knowledge, the present study is the first SRMA evaluating the effect of VR on seminal MDA levels and demonstrating significant decrease of this OS biomarker following repair. Our findings further strengthen currently available evidence and could potentially serve to upgrade the professional societies’ practice recommendations favoring VR to improve SDF and to ameliorate seminal OS in infertile men.
Footnotes
Conflict of Interest: The authors have nothing to disclose.
Funding: None.
- Conceptualization: RC, AA, R Shah.
- Data curation: R Singh, PS.
- Formal analysis: R Singh, PS.
- Investigation: RC, R Shah.
- Methodology: RC, GS, F Boitrelle.
- Project administration: AA, R Shah.
- Software: R Singh.
- Visualization: all authors.
- Writing – original draft: all authors.
- Writing – review & editing: all authors.
Acknowledgements
The authors express gratitude to Gianmaria Salvio, MD (Catania, Italy) for his support in graphics and artwork, and to Damayanthi Durairajanayagam, PhD (Selangor, Malaysia) for assistance with scientific editing of this manuscript.
Supplementary Materials
Supplementary materials can be found via https://doi.org/10.5534/wjmh.230235.
Selection criteria in included studies (PICOS)
Forest plot for sensitivity analysis of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair. 95% CI: 95% confidence interval.
Funnel plot of (A) standard error and (B) precision of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair.
Forest plot for sensitive analysis of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair using microsurgical treatment. 95% CI: 95% confidence interval.
Funnel plot of (A) standard error and (B) precision of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair using microsurgical treatment.
Forest plot for sensitive analysis of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair using SCD. 95% CI: 95% confidence interval.
Funnel plot of (A) standard error and (B) precision of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair using sperm chromatin dispersion.
Forest plot for sensitive analysis of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair using sperm chromatin structure assay. 95% CI: 95% confidence interval.
Funnel plot of (A) standard error and (B) precision of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair using sperm chromatin structure assay.
Forest plot for sensitive analysis of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair using TUNEL. 95% CI: 95% confidence interval.
Funnel plot of (A) standard error and (B) precision of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair using TUNEL.
Forest plot for sensitive analysis of the sperm DNA fragmentation (SDF) in infertile patients after varicocele repair compared to before varicocele repair using a not specified methods for analysis of SDF. 95% CI: 95% confidence interval.
Funnel plot of (A) standard error and (B) precision of the sperm DNA fragmentation (SDF) in infertile patients after varicocele repair compared to before varicocele repair using a not specified methods for analysis of SDF.
Forest plot for sensitive analysis of seminal malondialdehyde level in infertile patients after varicocele repair compared to before varicocele repair. 95% CI: 95% confidence interval.
Funnel plot of (A) standard error and (B) precision of malondialdehyde level in infertile patients after varicocele repair compared to before varicocele repair.
References
- 1.Centers for Disease Control and Prevention (CDC) National public health action plan for the detection, prevention, and management of infertility. CDC; 2014. [Google Scholar]
- 2.Boivin J, Bunting L, Collins JA, Nygren KG. International estimates of infertility prevalence and treatment-seeking: potential need and demand for infertility medical care. Hum Reprod. 2007;22:1506–1512. doi: 10.1093/humrep/dem046. Erratum in: Hum Reprod 2007;22:2800. [DOI] [PubMed] [Google Scholar]
- 3.Martinez G, Daniels K, Chandra A. Fertility of men and women aged 15-44 years in the United States: national survey of family growth, 2006-2010. Natl Health Stat Report. 2012;51:1–28. [PubMed] [Google Scholar]
- 4.Agarwal A, Mulgund A, Hamada A, Chyatte MR. A unique view on male infertility around the globe. Reprod Biol Endocrinol. 2015;13:37. doi: 10.1186/s12958-015-0032-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Snow-Lisy D, Sabanegh E., Jr What does the clinician need from an andrology laboratory? Front Biosci (Elite Ed) 2013;5:289–304. doi: 10.2741/e616. [DOI] [PubMed] [Google Scholar]
- 6.Barbăroșie C, Agarwal A, Henkel R. Diagnostic value of advanced semen analysis in evaluation of male infertility. Andrologia. 2021;53:e13625. doi: 10.1111/and.13625. [DOI] [PubMed] [Google Scholar]
- 7.Santi D, Spaggiari G, Simoni M. Sperm DNA fragmentation index as a promising predictive tool for male infertility diagnosis and treatment management - meta-analyses. Reprod Biomed Online. 2018;37:315–326. doi: 10.1016/j.rbmo.2018.06.023. [DOI] [PubMed] [Google Scholar]
- 8.Lewis SE, Agbaje I, Alvarez J. Sperm DNA tests as useful adjuncts to semen analysis. Syst Biol Reprod Med. 2008;54:111–125. doi: 10.1080/19396360801957739. [DOI] [PubMed] [Google Scholar]
- 9.Esteves SC, Sharma RK, Gosálvez J, Agarwal A. A translational medicine appraisal of specialized andrology testing in unexplained male infertility. Int Urol Nephrol. 2014;46:1037–1052. doi: 10.1007/s11255-014-0715-0. [DOI] [PubMed] [Google Scholar]
- 10.García-Peiró A, Martínez-Heredia J, Oliver-Bonet M, Abad C, Amengual MJ, Navarro J, et al. Protamine 1 to protamine 2 ratio correlates with dynamic aspects of DNA fragmentation in human sperm. Fertil Steril. 2011;95:105–109. doi: 10.1016/j.fertnstert.2010.06.053. [DOI] [PubMed] [Google Scholar]
- 11.Rogenhofer N, Dansranjavin T, Schorsch M, Spiess A, Wang H, von Schönfeldt V, et al. The sperm protamine mRNA ratio as a clinical parameter to estimate the fertilizing potential of men taking part in an ART programme. Hum Reprod. 2013;28:969–978. doi: 10.1093/humrep/des471. [DOI] [PubMed] [Google Scholar]
- 12.Ni K, Spiess AN, Schuppe HC, Steger K. The impact of sperm protamine deficiency and sperm DNA damage on human male fertility: a systematic review and meta-analysis. Andrology. 2016;4:789–799. doi: 10.1111/andr.12216. [DOI] [PubMed] [Google Scholar]
- 13.Spanò M, Bonde JP, Hjøllund HI, Kolstad HA, Cordelli E, Leter G. Sperm chromatin damage impairs human fertility. The Danish First Pregnancy Planner Study Team. Fertil Steril. 2000;73:43–50. doi: 10.1016/s0015-0282(99)00462-8. [DOI] [PubMed] [Google Scholar]
- 14.World Health Organization (WHO) WHO laboratory manual for the examination and processing of human semen. 6th ed. WHO; 2021. [Google Scholar]
- 15.Evenson DP, Jost LK, Marshall D, Zinaman MJ, Clegg E, Purvis K, et al. Utility of the sperm chromatin structure assay as a diagnostic and prognostic tool in the human fertility clinic. Hum Reprod. 1999;14:1039–1049. doi: 10.1093/humrep/14.4.1039. [DOI] [PubMed] [Google Scholar]
- 16.Simon L, Zini A, Dyachenko A, Ciampi A, Carrell DT. A systematic review and meta-analysis to determine the effect of sperm DNA damage on in vitro fertilization and intracytoplasmic sperm injection outcome. Asian J Androl. 2017;19:80–90. doi: 10.4103/1008-682X.182822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Siddhartha N, Reddy NS, Pandurangi M, Muthusamy T, Vembu R, Kasinathan K. The effect of sperm DNA fragmentation index on the outcome of intrauterine insemination and intracytoplasmic sperm injection. J Hum Reprod Sci. 2019;12:189–198. doi: 10.4103/jhrs.JHRS_22_19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tan J, Taskin O, Albert A, Bedaiwy MA. Association between sperm DNA fragmentation and idiopathic recurrent pregnancy loss: a systematic review and meta-analysis. Reprod Biomed Online. 2019;38:951–960. doi: 10.1016/j.rbmo.2018.12.029. [DOI] [PubMed] [Google Scholar]
- 19.Bungum M, Humaidan P, Axmon A, Spano M, Bungum L, Erenpreiss J, et al. Sperm DNA integrity assessment in prediction of assisted reproduction technology outcome. Hum Reprod. 2007;22:174–179. doi: 10.1093/humrep/del326. [DOI] [PubMed] [Google Scholar]
- 20.Duran EH, Morshedi M, Taylor S, Oehninger S. Sperm DNA quality predicts intrauterine insemination outcome: a prospective cohort study. Hum Reprod. 2002;17:3122–3128. doi: 10.1093/humrep/17.12.3122. [DOI] [PubMed] [Google Scholar]
- 21.Rilcheva VS, Ayvazova NP, Ilieva LO, Ivanova SP, Konova EI. Sperm DNA integrity test and assisted reproductive technology (Art) outcome. J Biomed Clin Res. 2016;9:21–29. [Google Scholar]
- 22.Agarwal A, Parekh N, Panner Selvam MK, Henkel R, Shah R, Homa ST, et al. Male oxidative stress infertility (MOSI): proposed terminology and clinical practice guidelines for management of idiopathic male infertility. World J Mens Health. 2019;37:296–312. doi: 10.5534/wjmh.190055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chen CS, Chao HT, Pan RL, Wei YH. Hydroxyl radical-induced decline in motility and increase in lipid peroxidation and DNA modification in human sperm. Biochem Mol Biol Int. 1997;43:291–303. doi: 10.1080/15216549700204071. [DOI] [PubMed] [Google Scholar]
- 24.Mohanty G, Swain N, Goswami C, Kar S, Samanta L. Histone retention, protein carbonylation, and lipid peroxidation in spermatozoa: possible role in recurrent pregnancy loss. Syst Biol Reprod Med. 2016;62:201–212. doi: 10.3109/19396368.2016.1148798. [DOI] [PubMed] [Google Scholar]
- 25.Clarke BG. Incidence of varicocele in normal men and among men of different ages. JAMA. 1966;198:1121–1122. [PubMed] [Google Scholar]
- 26.Gorelick JI, Goldstein M. Loss of fertility in men with varicocele. Fertil Steril. 1993;59:613–616. [PubMed] [Google Scholar]
- 27.Witt MA, Lipshultz LI. Varicocele: a progressive or static lesion? Urology. 1993;42:541–543. doi: 10.1016/0090-4295(93)90268-f. [DOI] [PubMed] [Google Scholar]
- 28.Oster J. Varicocele in children and adolescents. An investigation of the incidence among Danish school children. Scand J Urol Nephrol. 1971;5:27–32. doi: 10.3109/00365597109133569. [DOI] [PubMed] [Google Scholar]
- 29.Levinger U, Gornish M, Gat Y, Bachar GN. Is varicocele prevalence increasing with age? Andrologia. 2007;39:77–80. doi: 10.1111/j.1439-0272.2007.00766.x. [DOI] [PubMed] [Google Scholar]
- 30.Su JS, Farber NJ, Vij SC. Pathophysiology and treatment options of varicocele: an overview. Andrologia. 2021;53:e13576. doi: 10.1111/and.13576. [DOI] [PubMed] [Google Scholar]
- 31.Garolla A, Torino M, Miola P, Caretta N, Pizzol D, Menegazzo M, et al. Twenty-four-hour monitoring of scrotal temperature in obese men and men with a varicocele as a mirror of spermatogenic function. Hum Reprod. 2015;30:1006–1013. doi: 10.1093/humrep/dev057. [DOI] [PubMed] [Google Scholar]
- 32.Agarwal A, Sharma RK, Desai NR, Prabakaran S, Tavares A, Sabanegh E. Role of oxidative stress in pathogenesis of varicocele and infertility. Urology. 2009;73:461–469. doi: 10.1016/j.urology.2008.07.053. [DOI] [PubMed] [Google Scholar]
- 33.Agarwal A, Hamada A, Esteves SC. Insight into oxidative stress in varicocele-associated male infertility: part 1. Nat Rev Urol. 2012;9:678–690. doi: 10.1038/nrurol.2012.197. [DOI] [PubMed] [Google Scholar]
- 34.Smit M, Romijn JC, Wildhagen MF, Veldhoven JL, Weber RF, Dohle GR. Decreased sperm DNA fragmentation after surgical varicocelectomy is associated with increased pregnancy rate. J Urol. 2010;183:270–274. doi: 10.1016/j.juro.2009.08.161. [DOI] [PubMed] [Google Scholar]
- 35.Ni K, Steger K, Yang H, Wang H, Hu K, Chen B. Sperm protamine mRNA ratio and DNA fragmentation index represent reliable clinical biomarkers for men with varicocele after microsurgical varicocele ligation. J Urol. 2014;192:170–176. doi: 10.1016/j.juro.2014.02.046. [DOI] [PubMed] [Google Scholar]
- 36.Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, et al. Meta-analysis of Observational Studies in Epidemiology (MOOSE) Group. Meta-analysis of observational studies in epidemiology: a proposal for reporting. JAMA. 2000;283:2008–2012. doi: 10.1001/jama.283.15.2008. [DOI] [PubMed] [Google Scholar]
- 37.Page MJ, Moher D, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. PRISMA 2020 explanation and elaboration: updated guidance and exemplars for reporting systematic reviews. BMJ. 2021;372:n160. doi: 10.1136/bmj.n160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Methley AM, Campbell S, Chew-Graham C, McNally R, Cheraghi-Sohi S. PICO, PICOS and SPIDER: a comparison study of specificity and sensitivity in three search tools for qualitative systematic reviews. BMC Health Serv Res. 2014;14:579. doi: 10.1186/s12913-014-0579-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Cannarella R, Shah R, Boitrelle F, Saleh R, Durairajanayagam D, Harraz AM, et al. Need for training in research methodology prior to conducting systematic reviews and meta-analyses, and the effectiveness of an online training program: the Global Andrology Forum model. World J Mens Health. 2023;41:342–353. doi: 10.5534/wjmh.220128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Murray J, Farrington DP, Eisner MP. Drawing conclusions about causes from systematic reviews of risk factors: the Cambridge quality checklists. J Exp Criminol. 2009;5:1–23. [Google Scholar]
- 41.Andrade C. Understanding the difference between standard deviation and standard error of the mean, and knowing when to use which. Indian J Psychol Med. 2020;42:409–410. doi: 10.1177/0253717620933419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kavoussi PK, Abdullah N, Gilkey MS, Hunn C, Machen GL, Chen SH, et al. The impact of ipsilateral testicular atrophy on semen quality and sperm DNA fragmentation response to varicocele repair. Asian J Androl. 2021;23:146–149. doi: 10.4103/aja.aja_50_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Fathi A, Mohamed O, Mahmoud O, Alsagheer GA, Reyad AM, Abolyosr A, et al. The impact of varicocelectomy on sperm DNA fragmentation and pregnancy rate in subfertile men with normal semen parameters: a pilot study. Arab J Urol. 2021;19:186–190. doi: 10.1080/2090598X.2021.1889746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Abbasi B, Molavi N, Tavalaee M, Abbasi H, Nasr-Esfahani MH. Alpha-lipoic acid improves sperm motility in infertile men after varicocelectomy: a triple-blind randomized controlled trial. Reprod Biomed Online. 2020;41:1084–1091. doi: 10.1016/j.rbmo.2020.08.013. [DOI] [PubMed] [Google Scholar]
- 45.Kamal HM, El-Fallah AA, Abdelbaki SA, Khalil MM, Kamal MM, Behiry EG. Association between seminal granulysin and malondialdehyde in infertile men with varicocele and the potential effect of varicocelectomy. Andrologia. 2020;52:e13579. doi: 10.1111/and.13579. [DOI] [PubMed] [Google Scholar]
- 46.Omar SS, Mahfouz W, Dawood W, Abo El-Wafa RAH, Ghazala RA, Zahran AM. Relation of nitric oxide synthase gene (NOS3) polymorphisms to varicocele risk and post-varicocelectomy seminal oxidative stress reduction. Andrologia. 2020;52:e13525. doi: 10.1111/and.13525. [DOI] [PubMed] [Google Scholar]
- 47.Mostafa T, Nabil N, Rashed L, Abo-Sief AF, Eissa HH. Seminal SIRT1-oxidative stress relationship in infertile oligoasthenoteratozoospermic men with varicocele after its surgical repair. Andrologia. 2020;52:e13456. doi: 10.1111/and.13456. [DOI] [PubMed] [Google Scholar]
- 48.Camargo M, Intasqui P, Belardin LB, Antoniassi MP, Cardozo KHM, Carvalho VM, et al. Molecular pathways of varicocele and its repair - a paired labelled shotgun proteomics approach. J Proteomics. 2019;196:22–32. doi: 10.1016/j.jprot.2019.01.019. [DOI] [PubMed] [Google Scholar]
- 49.Afsin M, Otludil B, Dede O, Akkus M. An examination on composition of spermatozoa obtained from pre-operative and post-operative varicocele patients. Reprod Biol. 2018;18:361–367. doi: 10.1016/j.repbio.2018.10.005. [DOI] [PubMed] [Google Scholar]
- 50.Vahidi S, Moein M, Nabi A, Narimani N. Effects of microsurgical varicocelectomy on semen analysis and sperm function tests in patients with different grades of varicocele: role of sperm functional tests in evaluation of treatments outcome. Andrologia. 2018;50:e13069. doi: 10.1111/and.13069. [DOI] [PubMed] [Google Scholar]
- 51.Sun XL, Wang JL, Peng YP, Gao QQ, Song T, Yu W, et al. Bilateral is superior to unilateral varicocelectomy in infertile males with left clinical and right subclinical varicocele: a prospective randomized controlled study. Int Urol Nephrol. 2018;50:205–210. doi: 10.1007/s11255-017-1749-x. [DOI] [PubMed] [Google Scholar]
- 52.Lu XL, Liu JJ, Li JT, Yang QA, Zhang JM. Melatonin therapy adds extra benefit to varicecelectomy in terms of sperm parameters, hormonal profile and total antioxidant capacity: a placebo-controlled, double-blind trial. Andrologia. 2018;50:e13033. doi: 10.1111/and.13033. [DOI] [PubMed] [Google Scholar]
- 53.Zaazaa A, Adel A, Fahmy I, Elkhiat Y, Awaad AA, Mostafa T. Effect of varicocelectomy and/or mast cells stabilizer on sperm DNA fragmentation in infertile patients with varicocele. Andrology. 2018;6:146–150. doi: 10.1111/andr.12445. [DOI] [PubMed] [Google Scholar]
- 54.Abdelbaki SA, Sabry JH, Al-Adl AM, Sabry HH. The impact of coexisting sperm DNA fragmentation and seminal oxidative stress on the outcome of varicocelectomy in infertile patients: a prospective controlled study. Arab J Urol. 2017;15:131–139. doi: 10.1016/j.aju.2017.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ni K, Steger K, Yang H, Wang H, Hu K, Zhang T, et al. A comprehensive investigation of sperm DNA damage and oxidative stress injury in infertile patients with subclinical, normozoospermic, and astheno/oligozoospermic clinical varicocoele. Andrology. 2016;4:816–824. doi: 10.1111/andr.12210. [DOI] [PubMed] [Google Scholar]
- 56.Telli O, Sarici H, Kabar M, Ozgur BC, Resorlu B, Bozkurt S. Does varicocelectomy affect DNA fragmentation in infertile patients? Indian J Urol. 2015;31:116–119. doi: 10.4103/0970-1591.152811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Tavalaee M, Bahreinian M, Barekat F, Abbasi H, Nasr-Esfahani MH. Effect of varicocelectomy on sperm functional characteristics and DNA methylation. Andrologia. 2015;47:904–909. doi: 10.1111/and.12345. [DOI] [PubMed] [Google Scholar]
- 58.Mohammed EE, Mosad E, Zahran AM, Hameed DA, Taha EA, Mohamed MA. Acridine orange and flow cytometry: which is better to measure the effect of varicocele on sperm DNA integrity? Adv Urol. 2015;2015:814150. doi: 10.1155/2015/814150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Smit M, Romijn JC, Wildhagen MF, Veldhoven JL, Weber RF, Dohle GR. Decreased sperm DNA fragmentation after surgical varicocelectomy is associated with increased pregnancy rate. J Urol. 2013;189(1 Suppl):S146–S150. doi: 10.1016/j.juro.2012.11.024. [DOI] [PubMed] [Google Scholar]
- 60.Li F, Yamaguchi K, Okada K, Matsushita K, Ando M, Chiba K, et al. Significant improvement of sperm DNA quality after microsurgical repair of varicocele. Syst Biol Reprod Med. 2012;58:274–277. doi: 10.3109/19396368.2012.692431. [DOI] [PubMed] [Google Scholar]
- 61.Gabriel MS, Chan SW, Alhathal N, Chen JZ, Zini A. Influence of microsurgical varicocelectomy on human sperm mitochondrial DNA copy number: a pilot study. J Assist Reprod Genet. 2012;29:759–764. doi: 10.1007/s10815-012-9785-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.La Vignera S, Condorelli R, Vicari E, D'Agata R, Calogero AE. Effects of varicocelectomy on sperm DNA fragmentation, mitochondrial function, chromatin condensation, and apoptosis. J Androl. 2012;33:389–396. doi: 10.2164/jandrol.111.013433. [DOI] [PubMed] [Google Scholar]
- 63.Sadek A, Almohamdy AS, Zaki A, Aref M, Ibrahim SM, Mostafa T. Sperm chromatin condensation in infertile men with varicocele before and after surgical repair. Fertil Steril. 2011;95:1705–1708. doi: 10.1016/j.fertnstert.2011.01.008. [DOI] [PubMed] [Google Scholar]
- 64.Zini A, Azhar R, Baazeem A, Gabriel MS. Effect of microsurgical varicocelectomy on human sperm chromatin and DNA integrity: a prospective trial. Int J Androl. 2011;34:14–19. doi: 10.1111/j.1365-2605.2009.01048.x. [DOI] [PubMed] [Google Scholar]
- 65.Ghazi S, Abdelfattah H. Effect of microsurgical varicocelectomy on sperm DNA fragmentation in infertile men. Middle East Fertil Soc J. 2011;16:149–153. [Google Scholar]
- 66.Dada R, Shamsi MB, Venkatesh S, Gupta NP, Kumar R. Attenuation of oxidative stress & DNA damage in varicocelectomy: implications in infertility management. Indian J Med Res. 2010;132:728–730. [PMC free article] [PubMed] [Google Scholar]
- 67.Nasr Esfahani MH, Abbasi H, Mirhosseini Z, Ghasemi N, Razavi S, Tavalaee M, et al. Can altered expression of HSPA2 in varicocele patients lead to abnormal spermatogenesis? Int J Fertil Steril. 2010;4:104–113. [Google Scholar]
- 68.Zini A, Blumenfeld A, Libman J, Willis J. Beneficial effect of microsurgical varicocelectomy on human sperm DNA integrity. Hum Reprod. 2005;20:1018–1021. doi: 10.1093/humrep/deh701. [DOI] [PubMed] [Google Scholar]
- 69.Yeşilli C, Mungan G, Seçkiner I, Akduman B, Açikgöz S, Altan K, et al. Effect of varicocelectomy on sperm creatine kinase, HspA2 chaperone protein (creatine kinase-M type), LDH, LDH-X, and lipid peroxidation product levels in infertile men with varicocele. Urology. 2005;66:610–615. doi: 10.1016/j.urology.2005.03.078. [DOI] [PubMed] [Google Scholar]
- 70.Schlegel PN, Sigman M, Collura B, De Jonge CJ, Eisenberg ML, Lamb DJ, et al. Diagnosis and treatment of infertility in men: AUA/ASRM guideline part I. Fertil Steril. 2021;115:54–61. doi: 10.1016/j.fertnstert.2020.11.015. [DOI] [PubMed] [Google Scholar]
- 71.Minhas S, Bettocchi C, Boeri L, Capogrosso P, Carvalho J, Cilesiz NC, et al. EAU Working Group on Male Sexual and Reproductive Health. European Association of Urology guidelines on male sexual and reproductive health: 2021 update on male infertility. Eur Urol. 2021;80:603–620. doi: 10.1016/j.eururo.2021.08.014. [DOI] [PubMed] [Google Scholar]
- 72.Baazeem A, Belzile E, Ciampi A, Dohle G, Jarvi K, Salonia A, et al. Varicocele and male factor infertility treatment: a new meta-analysis and review of the role of varicocele repair. Eur Urol. 2011;60:796–808. doi: 10.1016/j.eururo.2011.06.018. [DOI] [PubMed] [Google Scholar]
- 73.Qiu D, Shi Q, Pan L. Efficacy of varicocelectomy for sperm DNA integrity improvement: a meta-analysis. Andrologia. 2021;53:e13885. doi: 10.1111/and.13885. [DOI] [PubMed] [Google Scholar]
- 74.Wang YJ, Zhang RQ, Lin YJ, Zhang RG, Zhang WL. Relationship between varicocele and sperm DNA damage and the effect of varicocele repair: a meta-analysis. Reprod Biomed Online. 2012;25:307–314. doi: 10.1016/j.rbmo.2012.05.002. [DOI] [PubMed] [Google Scholar]
- 75.Birowo P, Rahendra Wijaya J, Atmoko W, Rasyid N. The effects of varicocelectomy on the DNA fragmentation index and other sperm parameters: a meta-analysis. Basic Clin Androl. 2020;30:15. doi: 10.1186/s12610-020-00112-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Lira Neto FT, Roque M, Esteves SC. Effect of varicocelectomy on sperm deoxyribonucleic acid fragmentation rates in infertile men with clinical varicocele: a systematic review and meta-analysis. Fertil Steril. 2021;116:696–712. doi: 10.1016/j.fertnstert.2021.04.003. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Selection criteria in included studies (PICOS)
Forest plot for sensitivity analysis of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair. 95% CI: 95% confidence interval.
Funnel plot of (A) standard error and (B) precision of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair.
Forest plot for sensitive analysis of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair using microsurgical treatment. 95% CI: 95% confidence interval.
Funnel plot of (A) standard error and (B) precision of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair using microsurgical treatment.
Forest plot for sensitive analysis of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair using SCD. 95% CI: 95% confidence interval.
Funnel plot of (A) standard error and (B) precision of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair using sperm chromatin dispersion.
Forest plot for sensitive analysis of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair using sperm chromatin structure assay. 95% CI: 95% confidence interval.
Funnel plot of (A) standard error and (B) precision of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair using sperm chromatin structure assay.
Forest plot for sensitive analysis of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair using TUNEL. 95% CI: 95% confidence interval.
Funnel plot of (A) standard error and (B) precision of the sperm DNA fragmentation in infertile patients after varicocele repair compared to before varicocele repair using TUNEL.
Forest plot for sensitive analysis of the sperm DNA fragmentation (SDF) in infertile patients after varicocele repair compared to before varicocele repair using a not specified methods for analysis of SDF. 95% CI: 95% confidence interval.
Funnel plot of (A) standard error and (B) precision of the sperm DNA fragmentation (SDF) in infertile patients after varicocele repair compared to before varicocele repair using a not specified methods for analysis of SDF.
Forest plot for sensitive analysis of seminal malondialdehyde level in infertile patients after varicocele repair compared to before varicocele repair. 95% CI: 95% confidence interval.
Funnel plot of (A) standard error and (B) precision of malondialdehyde level in infertile patients after varicocele repair compared to before varicocele repair.