Abstract
Background
The efficacy of pharmacological and nutritional interventions in individuals at clinical high risk for psychosis (CHR-P) remains elusive. This study aims to investigate the efficacy of pharmacological and nutritional interventions in CHR-P and whether these interventions can enhance the efficacy of psychological treatments.
Methods
We systematically reviewed data from 5 databases until July 24, 2021: PubMed, Web of Science, EMBASE, China National Knowledge Infrastructure, and WanFang Data. The primary outcome was the transition to psychosis. Network meta-analyses were conducted at 3 time points (6, 12, and ≥24 months) considering both pharmacological/nutritional interventions alone and its combination with psychotherapy.
Results
Out of 11 417 identified references, 21 studies were included, comprising 1983 participants. CHR-P participants receiving omega-3 polyunsaturated fatty acids treatment were associated with a lower probability of transition compared with placebo/control at 6 months (odds ratio [OR] = 0.07, 95% confidence interval [CI] = .01 to .054), 12 months (OR = 0.14, 95% CI = .03 to .66), and ≥24 months (OR = 0.16, 95% CI = .05 to .54). Moreover, risperidone plus psychotherapy was associated with a lower likelihood of transition at 6 months compared with placebo/control plus psychotherapy, but this result was not sustained over longer durations.
Conclusion
Omega-3 polyunsaturated fatty acids helped in preventing transitions to psychosis compared with controls.
PROSPERO registration number
CRD42021256209.
Keywords: Clinical high risk of psychosis, omega-3 polyunsaturated fatty acids, network meta-analysis
Significance Statement.
Preventing the transition to psychosis in individuals at clinical high risk for psychosis (CHR-P) is vitally important. The efficacy of pharmacological and nutritional interventions in CHR-P remains elusive. We performed a network meta-analysis to investigate the efficacy of these interventions in CHR-P and whether they can enhance the efficacy of psychological treatments. Our research indicates that omega-3 polyunsaturated fatty acids may have potential in preventing the transition to psychosis in CHR-P individuals. However, omega-3 polyunsaturated fatty acids do not appear to enhance the efficacy of psychological treatments. Our study, which integrates a diverse range of interventions, may have implications for clinical practice.
INTRODUCTION
The individuals at clinical high risk for psychosis (CHR-P) manifest early indicators of emerging psychosis, including negative and cognitive symptoms, attenuated psychotic symptoms, and brief remitting psychotic episodes, but do not meet the severity or duration necessary for a definitive diagnosis of a psychotic disorder according to established DSM/ICD criteria (Fusar-Poli, 2017; De Pablo et al., 2020; Solmi et al., 2023). Recent research shows that over 20% of CHR patients transition to psychosis (De Pablo et al., 2021a, 2021b; Solmi et al., 2023).
In light of the considerable risk of these individuals transitioning to psychosis, the past quarter-century has seen a growing effort to delay or even prevent this transition in CHR-P individuals (National Institute for Health and Care Excellence, 2014; Schmidt et al., 2015). Present treatment guidelines for CHR-P predominantly endorse cognitive behavioral therapy (CBT), family-focused therapy, and other integrated psychological interventions (National Institute for Health and Care Excellence, 2014; Schmidt et al., 2015; Addington et al., 2017; Barnes et al., 2019). However, the financial and temporal demands of psychotherapies often make them inaccessible to economically disadvantaged individuals.
Given the economic challenges associated with psychotherapies, pharmacological and nutritional interventions often become the primary preventive measure for these individuals. Yet the impact of pharmacological and nutritional interventions on the transition to psychosis in CHR-P is fraught with uncertainties. Take, for instance, the use of antipsychotics: the National Institute for Health and Care Excellence guidelines clearly advise against using them as a preventative measure (National Institute for Health and Care Excellence, 2014). On the other hand, the European Psychiatric Association and the British Association for Psychopharmacology offer a detailed perspective in their guidelines, recommending their use in cases with severe and worsening symptoms (Schmidt et al., 2015; Barnes et al., 2019). Moreover, The European Psychiatric Association does not offer information on the recommended use of nutritional supplements, while the Australian clinical guidelines emphasize the potential benefits of specific supplements, such as omega-3 polyunsaturated fatty acids (omega-3 PUFAs), in preventing the onset of psychosis in CHR-P individuals (Early Psychosis Guidelines Writing Group and EPPIC National Support Program, 2016). These discrepancies in guidelines underscore the complexities and uncertainties surrounding the best preventive strategies for CHR-P populations. With such varied recommendations, clinicians are often left navigating a challenging decision-making landscape.
Furthermore, another critical question arises: can pharmacological or nutritional interventions enhance the effects of psychological interventions to produce better therapeutic outcomes? A meta-analysis indicated an absence of evidence supporting any specific preventive interventions can significantly enhance the effects of psychological interventions (Davies et al., 2018). The combination of pharmacological/nutritional interventions with psychotherapy did not prove more effective than psychotherapy alone for CHR-P individuals (Davies et al., 2018). While a randomized controlled trial proffered tantalizing hints, positing potential benefits of amalgamating risperidone with psychological therapy (McGorry et al., 2002), the overall consensus remains nebulous.
Therefore, this article aimed to address 2 primary questions: (1) what is the efficacy of pharmacological and nutritional interventions in preventing psychosis in the CHR-P population; and (2) Can these interventions enhance the effectiveness of psychological interventions for CHR-P patients?
METHODS
We conducted a meta-analysis following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement (Moher et al., 2009). Before commencing our search, we registered our protocol detailing the search strategy and analytic methods on the PROSPERO database (registration no. CRD42021256209).
Search Strategy and Selection
We systematically searched 5 electronic databases—PubMed, Web of Science, EMBASE, China National Knowledge Infrastructure, and WanFang Data—from inception until July 24, 2021, using the keywords: “pharmacological” OR “medicine” AND “psychosis” AND “at risk” OR “high clinical risk” OR “prodromal.” We also screened the reference lists of pertinent reviews for additional studies.
After removing duplicate articles, 2 reviewers (M.Z. and Y.D.) independently screened the articles by their titles and abstracts using the following criteria:
Population: patients older than 12 who have been identified as being at clinical high risk of psychosis, as determined by validated assessments, including the Structured Interview for Psychosis-risk Syndromes (SIPS), and the Comprehensive Assessment of At-Risk Mental States (CAARMS).
Study design: studies were published in either English or Chinese, and the types of studies included were randomized controlled trials, case-control studies, and cohort studies.
Any disagreements or uncertainties were reviewed and discussed by the 2 initial reviewers. If consensus could not be reached, another reviewer (B.Z.) was consulted. The full texts of the selected articles were then independently assessed by Y.D. and W.B. using the following inclusion criteria: (1) intervention and comparator—studies investigating pharmacological or nutritional interventions with a parallel control; and (2) outcomes—availability of data for extraction.
Exclusion criteria were as follows: (1) the presence of organic conditions or neurological disorders (such as encephalitis and acute intoxication), some neurodevelopmental disorders (autism and intellectual disabilities), and concurrent substance use disorders; (2) studies with fewer than 10 participants; (3) studies with overlapping samples; and (4) studies in which mixed medications were used within a group compromised the clear classification of specific interventions.
This review covers pharmacological and nutritional interventions such as antipsychotics (AP), antidepressants (AD), mood stabilizers (MS), and benzodiazepines (BEN), as well as nutritional supplements like omega-3 PUFAs and N-methyl-D-aspartate (NMDA) modulators, notably D-serine. We exclude psychoactive substances that are associated with an increased risk of conversion in CHR-P patients, including marijuana and alcohol. Given that CHR-P patients in clinical trials are mostly help-seeking adolescents and young adults, it is ethically inappropriate to allocate them to a nontreatment group. Interventions tailored to specific patient needs, or those labeled as “treatment as usual” before the study and retained in the experiment, are referred to as need-based interventions (NBI) (McGorry et al., 2002; Davies et al., 2018).
Outcome Measures and Data Extraction
The primary outcome was the transition to psychosis, which was operationalized as defined by CHR-P instruments, including the SIPS, Positive and Negative Syndrome Scale, Brief Psychiatric Rating Scale, and CAARMS, or according to any version of DSM/ICD (De Pablo et al., 2021b). We analyzed 3 conversion rates (6 months, 12 months, and ≥24 months) at 2 levels: pharmacological/nutritional interventions alone and pharmacological/nutritional interventions combined with psychotherapy. To prevent artificial inflation of transition risk, sample sizes were determined based on the number of participants randomized, and those who withdrew after randomization were counted as nontransitions.
Data extraction was carried out by 1 reviewer (C.C.) and verified by a second (K.X.). Extracted data included study authorship, publication year, country of origin, intervention method, study design, sample size, transition count, mean age, gender distribution, diagnostic tools for CHR-P, and outcome data time points. In instances where data were only presented graphically, numerical data were digitally extracted from Kaplan-Meier plots using WebPlotDigitizer version 4.6. (https://apps.automeris.io/wpd/index.zh_CN.html).
Quality of the Evidence
Risk of Bias
To evaluate the risk of bias in the included randomized controlled trials (RCTs), we employed the RoB 1 tool (Sterne et al., 2016). Seven domains were appraised: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, and selective reporting. Each domain received one of the following assessments: “low risk of bias,” “unclear risk of bias,” or “high risk of bias.” The overall risk of bias for each study was determined based on its lowest-performing domain (Higgins et al., 2019).
For case-control and cohort studies, we used the Newcastle-Ottawa Scale (https://www.ohri.ca/programs/clinical_epidemiology/oxford.asp). This scale employs a “star system” to assess studies across 3 pivotal perspectives: the selection of study groups, the comparability of groups, and ascertainment of exposure (in case-control studies) or outcome (in cohort studies).
Grade
The quality of evidence for all outcomes was gauged using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach. Five domains were considered: study limitations, indirectness, inconsistency, imprecision, and publication bias. Detailed information on these domains can be found in the supplementary Methods section.
Statistical Analysis
We employed network meta-analyses within a frequentist framework using the netmeta package in R (version 4.3.1). A network plot was generated for each analysis group to ensure adequate node connections. We then executed a network meta-analysis that assumed consistency and a common heterogeneity across all comparisons in the network. The odds ratio (OR) was chosen as the effect size for dichotomous outcomes due to its favorable mathematical attributes in meta-analysis (Bakbergenuly et al., 2019). We computed a treatment effect (OR) for each pairwise treatment comparison by amalgamating both direct and indirect evidence. ORs with 95% confidence intervals (CIs) for each treatment pairing were presented in forest and league tables, and interventions were ranked by P scores. The closer the P score is to 100%, the higher the likelihood that a given intervention ranks at the top.
We evaluated potential inconsistencies using both the node-splitting method and the design-by-treatment model. And to appraise the influence of study quality, we conducted sensitivity analyses limited to high-quality studies. We also reran analyses focusing solely on RCTs and amalgamated the nodes of placebo/control plus NBI and placebo/control plus NBI plus psychotherapy due to a scarcity of RCTs. We evaluated the presence of small-trial effects, potentially indicative of publication bias, using comparison-adjusted funnel plots (Chaimani et al., 2013). Furthermore, the Harbord test was used to determine if there was significant publication bias (Rodolico et al., 2022).
RESULTS
Included Studies and Sample Characteristics
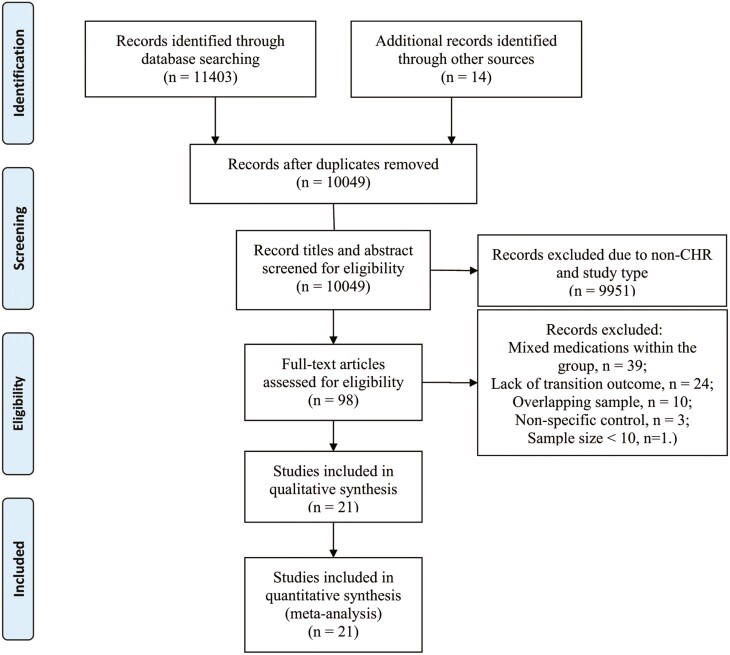
From an initial screening of 11 417 references, 98 studies were identified as potentially eligible (Figure 1). After a detailed full-text evaluation, 21 studies met the criteria for inclusion, which comprised 9 RCTs and 12 observational studies (Table 1). For time-point analyses, there were 8, 10, and 11 studies included for the 6, 12, and ≥24 months, respectively. Notably, all studies included in the analyses at 6 months, as well as those in the pharmacological/nutritional interventions combined with psychotherapy group, were RCTs. Only the analyses of pharmacological/nutritional interventions at 12 and ≥24 months incorporated observational studies.
Figure 1.
PRISMA flow chart of the study selection process.
Table 1.
Demographic and Clinical Characteristics of Included Studies
| Study (author, y) | Study arms (N) | Treatment | Network inclusion (group: time points) | Follow-up time points (mo) | % Male | Mean age (y) | CHR-P criteria | Study design | Country | Quality |
|---|---|---|---|---|---|---|---|---|---|---|
| (Kantrowitz et al., 2015) | D-serine+NBI (20) | D-serine+NBI | Pharm/Nutr: 6 | 4 | 34 | 19.5 | SIPS | DB-RCT | USA | High |
| Plcb/Ctrl+NBI (24) | Plcb+NBI | |||||||||
| (McGlashan et al., 2006) | OLA+NBI (31) | OLA+NBI | Pharm/Nutr: 6, 12 |
6, 12 | 65 | 17.7 | SIPS | DB-RCT | USA, Canada | Moderate |
| Plcb/Ctrl+NBI (29) | Plcb+NBI | |||||||||
| (Amminger et al., 2010) | Omega-3+NBI (41) | EPA+DHA+VE +NBIa |
Pharm/Nutr: 6, 12 |
6, 12 | 33.3 | 16.4 | PANSS | DB-RCT | Austria | High |
| Plcb/Ctrl+NBI (40) | Coconut oil+VE +NBIa |
|||||||||
| (Glenthøj et al., 2015)b | AP+NBI (37) | ARI/AMI/OLA/PAL/ QUE/RIS+NBI |
Pharm/Nutr: 12 | 12 | 47.3 | 24 | CAARMS | Observational study | Denmark | Moderate |
| AD+NBI (29) | SSRIs/SNRIs /NaSSA+NBIc |
|||||||||
| (Nieman et al., 2009) | AP+NBI (15) | AP+NBIc | Pharm/Nutr: 12 | 12.8 | 65.3 | 19.4 | SIPS | Observational study | Netherlands | High |
| Plcb/Ctrl+NBI (42) | No medication+NBI | |||||||||
| (Yoviene Sykes et al., 2020) | AP+NBI (88) | AP+NBIc | Pharm/Nutr: 12 | 12 | 58.2 | 19.1 | SIPS | Observational study | USA | High |
| AD+NBI (128) | AD+NBIc | |||||||||
| MS+NBI (15) | MS+NBIc | |||||||||
| BEN+NBI (35) | BEN+NBIc | |||||||||
| (Grent-‘t-Jong et al., 2021) | AD+NBI (41) | AD+NBIc | Pharm/Nutr: 12 | 18 | 29.3 | 22 | CAARMS | Observational study | Scotland | Moderate |
| Plcb/Ctrl+NBI (59) | No medication+NBI | |||||||||
| (Amminger et al., 2015) | Omega-3+NBI (41) | EPA+DHA+VE +NBIa |
Pharm/Nutr: ≥24 | 84 | 33.3 | 16.4 | PANSS | DB-RCT | Austria | High |
| Plcb/Ctrl+NBI (40) | Coconut oil+VE +NBIa |
|||||||||
| (Bang et al., 2019) | AP+NBI (24) | Atypical AP+NBIc | Pharm/Nutr: ≥24 | 25.8 | 59.7 | 19.9 | SIPS | Observational study | Korea | High |
| Plcb/Ctrl+NBI (53) | No medication+NBI | |||||||||
| (Bearden et al., 2011) | AP+NBI (10) | Atypical AP+NBIc | Pharm/Nutr: ≥24 | 24 | 70.4 | 17.1 | SIPS | Observational study | USA | Moderate |
| AD+NBI (12) | SSRIs+NBIc | |||||||||
| (Brucato et al., 2017) | AP+NBI (11) | AP+NBIc | Pharm/Nutr: ≥24 | 24 | 73 | 20 | SIPS | Observational study | USA | Moderate |
| AD+NBI (25) | AD+NBIc | |||||||||
| AD+AP+NBI (20) | AD+AP+NBIc | |||||||||
| Plcb/Ctrl+NBI (144) | No medication+NBI | |||||||||
| (DeVylder et al., 2014) | AP+NBI (14) | AP+NBIc | Pharm/Nutr: ≥24 | 30 | 76 | 20.1 | SIPS | Observational study | USA | High |
| AD+NBI (22) | AD+NBIc | |||||||||
| (Francesconi et al., 2017) | AD+NBI (76) | AD+NBIc | Pharm/Nutr: ≥24 | 36 | 51.7 | 24.3 | CAARMS | Observational study | Italy | High |
| Anxiolytics+NBI (57) | Anxiolytics+NBIc | |||||||||
| MS+NBI (26) | MS+NBIc | |||||||||
| Plcb/Ctrl+NBI (11) | No medication+NBI | |||||||||
| (Pawełczyk et al., 2021) | AP+NBI (10) | AP+NBIc | Pharm/Nutr: ≥24 | 62 | 42.5 | 19.2 | CAARMS | Observational study | Poland | High |
| AD+NBI (30) | SSRI+NBIc | |||||||||
| (Schultze-Lutter et al., 2014) | AP+NBI (34) | AP+NBIc | Pharm/Nutr: ≥24 | 24 | 63 | 24.9 | SIPS | Observational study | Germany | Moderate |
| AD+NBI (32) | AD+NBIc | |||||||||
| (van Tricht et al., 2010) | AP+NBI (16) | AP+NBIc | Pharm/Nutr: ≥24 | 36 | 68.9 | 19.6 | SIPS | Observational study | Netherlands | High |
| Plcb/Ctrl+NBI (30) | No medication+NBI | |||||||||
| (Woods et al., 2017) | ZIP+NBI+PT (24) | ZIP+NBI+SIT | Pharm/Nutr +PT: 6 | 6 | 64 | 22.3 | SIPS | DB-RCT | USA | Moderate |
| Plcb/Ctrl+NBI+PT (27) | Plcb+NBI+SIT | |||||||||
| (McGorry et al., 2013) | RIS+NBI+PT (43) | RIS+NBI+CT | Pharm/Nutr +PT: 6, 12 | 6, 12 | 36.8 | 17.8 | CAARMS | DB-RCT | Australia | High |
| Plcb/Ctrl+NBI+PT (44) | Plcb+NBI+CT | |||||||||
| (Bechdolf et al., 2011)d | ARI+NBI+PT (96) | ARI+NBI+PE | Pharm/Nutr +PT: 6, 12 | 6, 12 | 68.9 | 24.4 | SIPS | DB-RCT | Germany | High |
| Plcb/Ctrl+NBI+PT (55) | Plcb+NBI+PE | |||||||||
| (McGorry et al., 2002) | RIS+NBI+PT (31) | RIS+NBI+CBT | Pharm/Nutr +PT: 6, 12, ≥24 | 6, 12, 36 | 58 | 20 | BPRS | SB-RCT | Australia | Low |
| Plcb/Ctrl+NBI+PT (28) | NBI+ST | |||||||||
| (Nelson et al., 2018) | Omega-3+NBI+PT (153) | EPA+DHA+NBI+CBCMe | Pharm/Nutr +PT: 6, 12, ≥24 | 6, 12, 40 | 45.7 | 19.2 | CAARMS | DB-RCT | Multi-national | High |
| Plcb/Ctrl+NBI+PT (151) | Paraffin oil +NBI+CBCM |
aEfficacy of omega-3 PUFAs may be influenced by EPA/DHA ratios, so we have provided detailed information on the omega-3 PUFAs used: 700 mg EPA + 480 mg DHA + 7.6 mg VE; the placebo consisted of coconut oil + 7.6 mg VE.
b(Glenthøj et al., 2015) presents the study protocol, and the results of the study are provided in (Kristensen et al., 2021).
cTreatment descriptions in some studies were not specific to any particular medication; they employed dichotomous variables, simply indicating “yes” or “no” for the usage of AD, AP, MS, BEN, or Anxiolytics.
d(Bechdolf et al., 2011) details the rationale and baseline characteristics of the study, and the results of the study are provided in (Bechdolf et al., 2023).
eThe omega-3 PUFAs consisted of 840 mg of EPA and 560 mg of DHA.
Abbreviations: AD, antidepressants; AMI, amilsulpride; AP, antipsychotics; ARI, aripiprazole; BEN, benzodiazepine; BPRS, Brief Psychiatric Rating Scale; CAARMS, Comprehensive Assessment of At-Risk Mental States; CBCM, cognitive-behavioral case management; CT, cognitive therapy; DB-RCT, double-blind randomized controlled trial; DHA, docosahexaenoic acid; Dser, D-serine; EPA, eicosapentaenoic acid; MS, mood stabilizers; NaSSA, noradrenergic and specific serotonergic antidepressants; NBI, needs-based interventions; OLA, olanzapine; Omega-3, omega-3 fatty acids; PAL, paliperidone; PANSS, Positive and Negative Syndrome Scale; PE, psychoeducation; Pharm/Nutr, pharmacological/nutritional interventions; Plcb/Ctrl, placebo/control; PT, psychotherapy; QUE, quetiapine; RIS, risperidone; SB-RCT, single-blind randomized controlled trial; SIPS, Structured Interview for Psychosis-risk Syndromes; SIT, supportive interpersonal therapy; SNRIs, serotonin and norepinephrine reuptake inhibitors; SSRIs, selective serotonin reuptake inhibitors; ST, supportive therapy; VE, vitamin E; ZIP, ziprasidone.
The 21 included studies encompassed data from 1983 participants, with a mean age of 20.2 ± 2.59 years and a mean male percentage of 54.5% ± 14.9%. Table 1 delineates the details of the included studies. The average sample size was 94, with individual studies ranging between 10 and 153 participants. The baseline age spanned from 16.4 to 24.9 years, and the male percentage varied from 29.3% to 76%. Most of the trials originated from America and Europe. The CAARMS and the SIPS emerged as the predominant diagnostic instruments for CHR-P, used in 6 and 12 studies, respectively.
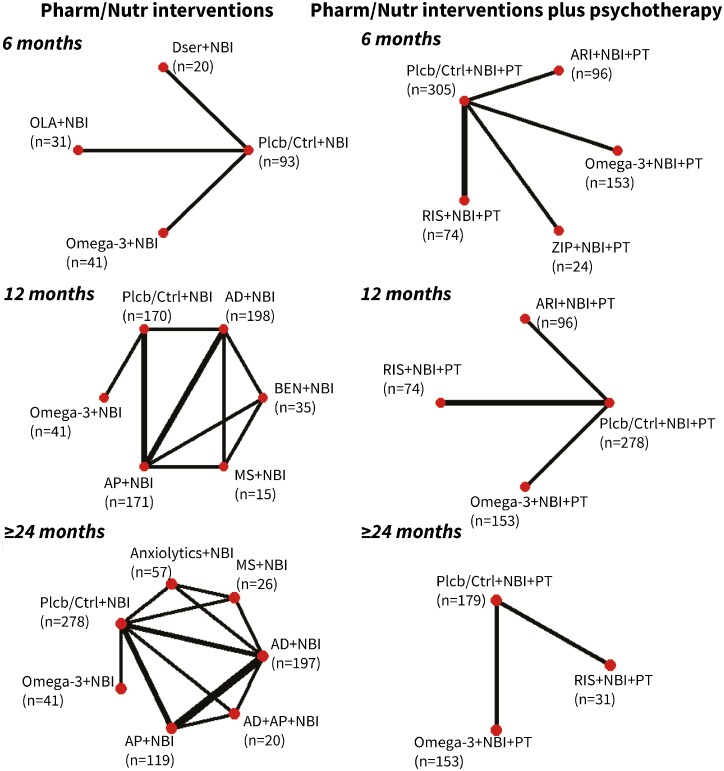
In the 6-month analysis, 8 studies provided data on 8 direct comparisons; of these, 3 were between 4 different pharmacological/nutritional interventions nodes, and 5 were between 5 different nodes combining pharmacological/nutritional interventions and psychotherapy (Figure 2). Of the 8 studies used for the 6-month analysis, 6 also provided 12-month follow-up data, resulting in the 12-month analysis comprising 10 studies. These studies provided data on 15 direct comparisons; of these, 11 were between 6 different pharmacological/nutritional interventions nodes, and 4 were between 4 different nodes combining pharmacological/nutritional interventions and psychotherapy (Figure 2). Additionally, the ≥24-month analysis included 11 studies that offered data on 21 direct comparisons; of these, 19 were between 7 different pharmacological/nutritional interventions nodes, and 2 were between 3 different nodes combining pharmacological/nutritional interventions and psychotherapy (Figure 2).
Figure 2.
Network plots of direct comparisons in the network meta-analysis for transition outcome at 6, 12, and ≥24 months in 2 levels: pharmacological/nutritional interventions group (on the left) and pharmacological/nutritional interventions plus psychotherapy group (on the right). The width of the lines is proportional to the number of trials comparing each pair of treatments. AD, antidepressants; AP, antipsychotics; ARI, aripiprazole; BEN, benzodiazepine; Dser, D-serine; MS, mood stabilizers; NBI, needs-based interventions; OLA, olanzapine; Omega-3, omega-3 fatty acids; Pharm/Nutr interventions, pharmacological/nutritional interventions; Plcb/Ctrl, placebo/control; PT, psychotherapy; RIS, risperidone; ZIP, ziprasidone.
Among the 10 RCTs, 70% were assessed as having a low risk of bias, 20% were assessed as having an unclear risk of bias, and 10% were assessed as having a high risk of bias. Among the 12 case-controlled studies, 58.3% were assessed as high quality and 41.7% as moderate quality.
Transition for Pharmacological / Nutritional: Interventions Group
Network meta-analysis for pharmacological/nutritional interventions incorporated D-serine, omega-3 PUFAs, AD, AP, MS, BEN, anxiolytics, and placebo/control. Results indicated that omega-3 PUFAs were significantly associated with a lower risk of transition to psychosis compared with placebo/control plus NBI at the 6-month, 12-month, and ≥24-month time points (Figure 3). The mean OR and 95% CI were 0.07 (0.01, 0.054) at 6 months, 0.14 (0.03,0.66) at 12 months, and 0.16 (0.05, 0.54) at ≥24 months.
Figure 3.
Forest plot of comparison in the risk ratio of conversion to psychosis in the network meta-analysis for transition outcome at 6, 12, and ≥24 months in 2 levels: pharmacological/nutritional interventions group (on the left) and pharmacological/nutritional interventions plus psychotherapy group (on the right). *Bold letters indicate statistical significance. AD, antidepressants; AP, antipsychotics; ARI, aripiprazole; BEN, benzodiazepine; CI, confidence interval; Dser, D-serine; MS, mood stabilizers; NBI, needs-based interventions; OLA, olanzapine; Omega-3, omega-3 fatty acids; OR, odds ratio; Pharm/Nutr interventions, pharmacological/nutritional interventions; Plcb/Ctrl, placebo/control; PT, psychotherapy; RIS, risperidone; ZIP, ziprasidone.
Other pharmacological/nutritional interventions, including D-serine, AP, AD, BEN, MS, and anxiolytics, demonstrated no clear evidence of superior efficacy in preventing transition when compared with placebo/control plus NBI (Figure 3).
Omega-3 PUFAs were also significantly more effective than other treatments, including AP (at 12 months, OR = 0.13, 95% CI = .02 to .75; at ≥24 months, OR = 0.12, 95% CI = .03 to .48), MS (at 12 months, OR = 0.09, 95% CI = .01 to .77; at ≥24 months, OR = 0.16, 95% CI = .03 to .90), and AD (at ≥24 months, OR = 0.25, 95% CI = .06 to .99; but nonsignificant at 12 months with OR = 0.30, 95% CI = .05 to 1.81). Furthermore, AD proved more effective than AP with a mean OR and 95% CI of 0.44 (0.23 to 0.85) at 12 months and 0.50 (0.27 to 0.90) at ≥24 months. For additional details, please refer to supplementary Tables 1–3 and supplementary Figures 1–3.
No inconsistency was found to be statistically significant at 12 months and ≥24 months (supplementary Tables 4–5). The design-by-treatment interaction test also revealed no significant inconsistency for 12 months (Q = 5.18, P = .075) or for ≥24 months (Q = 0.49, P = .92). For the 6-month analysis, there were insufficient articles to test inconsistency (supplementary Table 6).
The results of the sensitivity analysis were consistent with the main analysis. Omega-3 PUFAs demonstrated a lower probability of transition to psychosis compared with placebo/control plus NBI at 6, 12, and ≥24 months (supplementary Figures 4–6). Repeated analyses of RCTs were only conducted at 12 and ≥24 months. These analyses also highlighted that omega-3 PUFAs was associated with a lower risk of transition to psychosis compared with placebo/control/PT plus NBI at 12 and ≥24 months (supplementary Figures 7–8). For the pharmacological/nutritional interventions group, 2 out of 3 studies were of high quality at 6 months, 3 out of 6 were of high quality at 12 months, and 6 out of 9 were of high quality at ≥24 months. No studies were of low quality in the pharmacotherapy analysis at any of the 3 time points (supplementary Tables 7–8). No significant publication bias was detected (supplementary Figures 9–11). The certainty of evidence, as assessed by the GRADE system, indicated that outcomes at 6 months and 12 months were “low” to “very low,” whereas those for ≥24 months ranged from “moderate” to “very low.” Detailed information can be found in supplementary Tables 9–11.
Transition for Pharmacological / Nutritional Interventions Plus Psychotherapy Group
Network meta-analysis for pharmacological/nutritional interventions plus psychotherapy included studies on psychotherapy paired with omega-3 PUFAs, aripiprazole, risperidone, ziprasidone, or placebo/control. Results indicate that risperidone combined with psychotherapy had a lower likelihood of psychosis transitions compared with placebo/control plus NBI and psychotherapy at 6 months. However, this effect was not significant at 12 months or ≥24 months. The mean OR and 95% CI were 0.28 (0.09,0.83) at 6 months, 0.67 (0.30, 1.52) at 12 months, and 0.44 (0.15, 1.28) at ≥24 months. Psychotherapy combined with other pharmacological/nutritional interventions, including ziprasidone, omega-3 PUFAs, and aripiprazole, did not show significant superior efficacy in preventing transitions compared with placebo/control plus NBI and psychotherapy (Figure 3; supplementary Tables 12-14). The ranking of pharmacological/nutritional interventions combined with psychotherapy can be found in supplementary Figures 12–14. And there were insufficient articles to test inconsistency (supplementary Tables 15–17).
Sensitivity analysis of high-quality studies was only conducted at 6 and 12 months because there was only 1 high-quality study at ≥24 months. No treatment demonstrated a lower likelihood of psychosis transitions compared with placebo/control plus NBI and psychotherapy at any time point (supplementary Figures 15–16).
Three out of 5 studies were of high quality at 6 months, 3 out of 4 were of high quality at 12 months, and 1 out of 2 was of high quality at ≥24 months. Notably, one of the studies focusing on risperidone plus psychotherapy was of low quality and is the primary outcome in the pharmacological/nutritional interventions plus psychotherapy group at both 6 and 12 months (supplementary Table 7). No significant publication bias was detected (supplementary Figures 17–19). GRADE was also applied to evaluate outcomes for pharmacological/nutritional interventions combined with psychotherapy interventions. The certainty of evidence was “low” to “very low” for 6 months, and “low” for both 12 months and ≥24 months (supplementary Tables 18–20).
DISCUSSION
In our comprehensive review of 21 studies involving 1983 participants, we found that omega-3 PUFAs had a lower probability of transition to psychosis compared with placebo/control at 6 months, 12 months, and ≥24 months. However, other pharmacological or nutritional interventions, such as D-serine, AP, and AD, did not exhibit similar benefits. Although omega-3 PUFAs showed a lower risk of transition to psychosis, it did not enhance the efficacy of psychotherapy in CHR-P patients. Additionally, although the combination of risperidone and psychotherapy had a lower likelihood of transition to psychosis at 6 months, this effect was not observed at longer durations.
Our current study has observed that omega-3 PUFAs were correlated with a lower risk of transition to psychosis compared with placebo, AP, MS, and AD. This finding is congruent with a meta-analysis exploring the different efficacy profiles of omega-3 PUFAs across various stages of schizophrenia (Chen et al., 2015). Specifically, it was determined that, during the prodromal phase of schizophrenia, omega-3 PUFA supplementation notably diminished psychotic symptom severity and reduced the conversion rates to first-episode psychosis (Chen et al., 2015). Furthermore, some research has highlighted the safety and cost-effectiveness of omega-3 PUFAs in addition to their potential in preventing the transition to psychosis, positioning them as a favorable first-line treatment choice (Nelson et al., 2018; Erzin and Gülöksüz, 2021). However, it is important to acknowledge that our result, indicating a lower risk of transitioning to psychosis with omega-3 PUFAs compared with a placebo, is derived from 2 articles with varying time points and a small sample of only 41 participants.
The efficacy of omega-3 PUFAs in CHR-P is potentially attributable to its roles in membrane phospholipid modulation and anti-inflammatory effects. The human brain, a lipid-dense organ, sources 60% of its membrane from phospholipids, with over one-half of these lipids being PUFAs (Horrobin, 1998; Benatti et al., 2004). Numerous preclinical research studies highlight that PUFAs play pivotal roles in ensuring membrane fluidity, safeguarding cell membrane integrity, influencing the gut microbiome, and directing synaptic pruning processes in the brain (Stillwell et al., 2005; Knochel et al., 2015; Robertson et al., 2017; Madore et al., 2020). More recent studies have underscored the potential anti-inflammatory impacts of omega-3 PUFAs on cytokine synthesis and complement and coagulation proteins (Groeger et al., 2010; Susai et al., 2022).
Despite the evidence supporting the effectiveness of omega-3 PUFAs in CHR-P, they do not appear to enhance the efficacy of psychotherapy. This observed phenomenon might be attributed to the robust protective effects conferred by psychotherapy, especially therapies like CBT. The therapeutic modalities, which emphasize cognitive restructuring, resilience building, and stress management, may provide such profound protective effects that they overshadow or negate any supplemental benefits from omega-3 PUFAs (Nelson et al., 2018). This observation underscores the strength and long-term protective potential of therapies like CBT.
Our study found that individuals on AD showed a significantly lower likelihood of transition to psychosis than those on AP. Notably, the transition probability from AD was comparable with that of placebo/control, aligning with existing literature, such as the meta-analysis on AD-exposed CHR-P individuals(Raballo et al., 2023) and findings by clinical audits (Cornblatt et al., 2007; Fusar-Poli et al., 2007). Moreover, our analysis did not reveal a significant benefit of AP treatment in preventing the transition from CHR-P to psychosis, which aligns with findings from earlier studies (Davies et al., 2018; Devoe et al., 2018). It is crucial to consider the variable states of CHR-P patients, especially in terms of the severe psychotic symptoms and the frequency of psychotic experiences. These factors could potentially confound the interpretation of the effect of AP on the transition to psychosis. A study has found that in mild CHR-P cases, antipsychotic treatment is more likely to be associated with conversion to psychosis compared with the no-antipsychotics group (Zhang et al., 2020). Furthermore, CHR-P individuals with severe positive and general symptoms but mild negative symptoms may benefit from antipsychotic treatment (Zhang et al., 2020). We also found that D-serine, an NMDAR modulator, did not show a notable effect in preventing the transition in CHR-P. This result aligns with prior meta-analyses on CHR-P (Davies et al., 2018). It is vital to approach these results with caution. The results from clinical audits could be affected by various factors, such as short follow-ups and biases. This highlights the need for better-designed RCTs to truly understand the efficacy of pharmacological and nutritional interventions for CHR-P.
Strengths and Limitations
The strength of our study lies in its comprehensive approach, which encompasses not only an evaluation of the efficacy of various pharmacological and nutritional interventions in the CHR-P population but also an exploration of whether these interventions augment the effectiveness of psychological treatments. Our work integrates a diverse range of interventions and might provide implications for clinical practice.
This study has some limitations. First, we analyzed a set of 21 papers, which point to the paucity of existing research in the CHR-P domain. Such a scarcity emphasizes the pressing need for broader studies to comprehensively evaluate the safety and efficacy of the medications in question. Second, including observational studies in our analysis presents a potential limitation because they might contribute to increased bias. Nevertheless, even when we narrowed down our examination to only RCTs, the core results were in line with our initial findings. Third, based on the GRADE framework, numerous comparisons were rated as being low or very low quality. Notwithstanding this challenge, our analysis strived to extract significant conclusions from these evaluations. The certainty of the evidence was integrated into our primary findings to aid in clinical decision-making. Fourth, some of the included studies did not provide detailed information on the specific medicines used.
CONCLUSIONS
Our study explored the clinical effects of pharmacological and nutritional interventions on the prevention of transition to psychosis and the augmentation of psychological interventions in the CHR-P population. We found that omega-3 PUFAs showed a lower likelihood of transition from CHR-P to psychosis compared with controls. However, it did not notably enhance the effectiveness of psychological interventions for CHR-P. Future high-quality RCTs are essential to further elucidate its effects.
Supplementary Material
Contributor Information
Chengfeng Chen, Department of Psychiatry, The Affiliated Brain Hospital of Guangzhou Medical University, Guangzhou, China; Department of Psychiatry, Guangzhou Medical University, Guangzhou, China.
Yongyan Deng, Peking University Sixth Hospital, Peking University Institute of Mental Health, Beijing, China; NHC Key Laboratory of Mental Health (Peking University), National Clinical Research Center for Mental Disorders (Peking University Sixth Hospital), Beijing, China.
Yuling Li, Department of Psychiatry, The Affiliated Brain Hospital of Guangzhou Medical University, Guangzhou, China; Department of Psychiatry, Guangzhou Medical University, Guangzhou, China.
Meiting Zhang, Department of Psychiatry, The Affiliated Brain Hospital of Guangzhou Medical University, Guangzhou, China; Department of Psychiatry, Guangzhou Medical University, Guangzhou, China.
Tong Yu, Department of Psychiatry, The Affiliated Brain Hospital of Guangzhou Medical University, Guangzhou, China; Department of Psychiatry, Guangzhou Medical University, Guangzhou, China.
Kun Xie, Department of Psychiatry, The Affiliated Brain Hospital of Guangzhou Medical University, Guangzhou, China; Department of Psychiatry, Guangzhou Medical University, Guangzhou, China.
Wuyou Bao, Institute of Mental Health, Tianjin Anding Hospital, Tianjin Medical University, Tianjin, China.
Peiying Li, Institute of Mental Health, Tianjin Anding Hospital, Tianjin Medical University, Tianjin, China.
Ling Sun, Institute of Mental Health, Tianjin Anding Hospital, Tianjin Medical University, Tianjin, China.
Tianhong Zhang, Shanghai Key Laboratory of Psychotic Disorders, Shanghai Mental Health Center, Shanghai Jiao Tong University School of Medicine, Shanghai, China.
Yikang Zhu, Shanghai Key Laboratory of Psychotic Disorders, Shanghai Mental Health Center, Shanghai Jiao Tong University School of Medicine, Shanghai, China.
Bin Zhang, Institute of Mental Health, Tianjin Anding Hospital, Tianjin Medical University, Tianjin, China.
Funding
This study did not receive any grants from funding agencies.
Interest Statement
None.
Data Availability
All the data extracted from included studies are publicly available in PubMed, Web of Science, EMBASE, China National Knowledge Infrastructure, and WanFang Data.
Author Contributions
Chengfeng Chen (Conceptualization [Equal], Data curation [Equal], Formal analysis [Lead], Investigation [Lead], Methodology [Lead], Project administration [Lead], Resources [Equal], Software [Lead], Supervision [Equal], Validation [Equal], Visualization [Lead], Writing—original draft [Lead], Writing—review and editing [Lead]), Yongyan Deng (Conceptualization [Equal], Data curation [Equal], Investigation [Equal], Methodology [Supporting], Resources [Equal], Validation [Equal], Writing—review and editing [Supporting]), Yuling Li (Conceptualization [Equal], Data curation [Equal], Formal analysis [Supporting], Methodology [Supporting], Resources [Equal], Software [Supporting], Supervision [Supporting], Validation [Equal], Writing—original draft [Supporting], Writing—review and editing [Supporting]), Meiting Zhang (Conceptualization [Equal], Data curation [Equal], Investigation [Supporting], Methodology [Supporting], Resources [Equal], Writing—review and editing [Supporting]), Tong Yu (Data curation [Equal], Methodology [Supporting], Resources [Supporting], Supervision [Supporting], Validation [Supporting], Writing—review and editing [Supporting]), Kun Xie (Data curation [Supporting], Methodology [Supporting], Resources [Supporting], Validation [Supporting], Writing—review and editing [Supporting]), Wuyou Bao (Data curation [Supporting], Methodology [Supporting], Resources [Supporting], Validation [Supporting], Writing—review and editing [Supporting]), Peiying Li (Conceptualization [Equal], Methodology [Supporting], Supervision [Supporting], Writing—review and editing [Supporting]), Ling Sun (Conceptualization [Equal], Methodology [Supporting], Supervision [Equal], Writing—review and editing [Equal]), Tianhong Zhang (Conceptualization [Equal], Methodology [Supporting], Supervision [Supporting], Writing—review and editing [Equal]), Yikang Zhu (Conceptualization [Equal], Methodology [Supporting], Supervision [Supporting], Writing—review and editing [Equal]), and Bin Zhang (Conceptualization [Equal], Data curation [Equal], Formal analysis [Supporting], Funding acquisition [Lead], Investigation [Supporting], Methodology [Supporting], Project administration [Lead], Resources [Equal], Software [Supporting], Supervision [Lead], Validation [Equal], Writing—review and editing [Equal]).
References
- Addington J, Addington D, Abidi S, Raedler T, Remington G (2017) Canadian treatment guidelines for individuals at clinical high risk of psychosis. Can J Psychiatry 62:656–661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amminger GP, Schäfer MR, Papageorgiou K, Klier CM, Cotton SM, Harrigan SM, Mackinnon A, McGorry PD, Berger GE (2010) Long-chain ω-3 fatty acids for indicated prevention of psychotic disorders: a randomized placebo-controlled trial. Arch Gen Psychiatry 67:146–154. [DOI] [PubMed] [Google Scholar]
- Amminger GP, Schäfer MR, Schlögelhofer M, Klier CM, McGorry PD (2015) Longer-term outcome in the prevention of psychotic disorders by the Vienna omega-3 study. Nat Commun 6:7934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bakbergenuly I, Hoaglin DC, Kulinskaya E (2019) Pitfalls of using the risk ratio in meta-analysis. Res Synth Methods 10:398–419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bang M, Park JY, Kim KR, Lee SY, Song YY, Kang JI, Lee E, An SK (2019) Psychotic conversion of individuals at ultra‐high risk for psychosis: the potential roles of schizotypy and basic symptoms. Early Interv Psychia 13:546–554. [DOI] [PubMed] [Google Scholar]
- Barnes TRE, et al. (2019) Evidence-based guidelines for the pharmacological treatment of schizophrenia: updated recommendations from the British Association for Psychopharmacology. J Psychopharmacol 34:3–78. [DOI] [PubMed] [Google Scholar]
- Bearden CE, Wu KN, Caplan R, Cannon TD (2011) Thought disorder and communication deviance as predictors of outcome in youth at clinical high risk for psychosis. J Am Acad Child Adolesc Psychiatry 50:669–680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bechdolf A, Müller H, Stützer H, Wagner M, Maier W, Lautenschlager M, Heinz A, de Millas W, Janssen B, Gaebel W, Michel TM, Schneider F, Lambert M, Naber D, Brüne M, Krüger-Özgürdal S, Wobrock T, Riedel M, Klosterkötter J; PREVENT study group (2011) Rationale and baseline characteristics of PREVENT: a second-generation intervention trial in subjects at-risk (prodromal) of developing first-episode psychosis evaluating cognitive behavior therapy aripiprazole and placebo for the prevention of psychosis. Schizophr Bull 37:S111–S121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bechdolf A, et al. (2023) Prevention of first-episode psychosis in people at clinical high risk: a randomized controlled multicentre trial comparing cognitive-behavioral therapy and clinical management plus low-dose aripiprazole or placebo (PREVENT). Schizophr Bull 49:1055–1066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benatti P, Peluso G, Nicolai R, Calvani M (2004) Polyunsaturated fatty acids: biochemical nutritional and epigenetic properties. J Am Coll Nutr 23:281–302. [DOI] [PubMed] [Google Scholar]
- Brucato G, Masucci MD, Arndt LY, Ben-David S, Colibazzi T, Corcoran CM, Crumbley AH, Crump FM, Gill KE, Kimhy D, Lister A, Schobel SA, Yang LH, Lieberman JA, Girgis RR (2017) Baseline demographics clinical features and predictors of conversion among 200 individuals in a longitudinal prospective psychosis-risk cohort. Psychol Med 47:1923–1935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chaimani A, Higgins JP, Mavridis D, Spyridonos P, Salanti G (2013) Graphical tools for network meta-analysis in STATA. PLoS One 8:e76654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen AT, Chibnall JT, Nasrallah HA (2015) A meta-analysis of placebo-controlled trials of omega-3 fatty acid augmentation in schizophrenia: possible stage-specific effects. Ann Clin Psychiatry 27:289–296. [PubMed] [Google Scholar]
- Cornblatt BA, Lencz T, Smith CW, Olsen R, Auther AM, Nakayama E, Lesser ML, Tai JY, Shah MR, Foley CA, Kane JM, Correll CU (2007) Can antidepressants be used to treat the schizophrenia prodrome? Results of a prospective naturalistic treatment study of adolescents. J Clin Psychiatry 68:546–557. [DOI] [PubMed] [Google Scholar]
- Davies C, Cipriani A, Ioannidis JP, Radua J, Stahl D, Provenzani U, McGuire P, Fusar‐Poli P (2018) Lack of evidence to favor specific preventive interventions in psychosis: a network meta‐analysis. World Psychiatry 17:196–209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Pablo GS, Catalan A, Fusar-Poli P (2020) Clinical validity of DSM-5 attenuated psychosis syndrome: advances in diagnosis prognosis and treatment. Jama Psychiat 77:311–320. [DOI] [PubMed] [Google Scholar]
- De Pablo GS, Davies C, De Diego H, Solmi M, Shin JI, Carvalho AF, Radua J, Fusar-Poli P (2021a) Transition to psychosis in randomized clinical trials of individuals at clinical high risk of psychosis compared to observational cohorts: a systematic review and meta-analysis. Eur Psychiat 64:e51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Pablo GS, Radua J, Pereira J, Bonoldi I, Arienti V, Besana F, Soardo L, Cabras A, Fortea L, Catalan A (2021b) Probability of transition to psychosis in individuals at clinical high risk: an updated meta-analysis. Jama Psychiat 78:970–978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devoe DJ, Peterson A, Addington J (2018) Negative symptom interventions in youth at risk of psychosis: a systematic review and network meta-analysis. Schizophr Bull 44:807–823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeVylder JE, Muchomba FM, Gill KE, Ben-David S, Walder DJ, Malaspina D, Corcoran CM (2014) Symptom trajectories and psychosis onset in a clinical high-risk cohort: the relevance of subthreshold thought disorder. Schizophr Res 159:278–283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Early Psychosis Guidelines Writing Group and EPPIC National Support Program. (2016) Australian Clinical Guidelines for Early Psychosis. 2nd edition update. Melbourne, Australia: Orygen the National Centre of Excellence in Youth Mental Health Melbourne. [Google Scholar]
- Erzin G, Gülöksüz S (2021) Early interventions for clinical high-risk state for psychosis. Noro Psikiyatri Arsivi 58:S7–S11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Francesconi M, Minichino A, Carrión RE, Delle Chiaie R, Bevilacqua A, Parisi M, Rullo S, Bersani FS, Biondi M, Cadenhead K (2017) Psychosis prediction in secondary mental health services. A broad, comprehensive approach to the “at risk mental state” syndrome. Eur Psychiat 40:96–104. [DOI] [PubMed] [Google Scholar]
- Fusar-Poli P (2017) The clinical high-risk state for psychosis (CHR-P), version II. Schizophr Bull 43:44–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fusar-Poli P, Valmaggia L, McGuire P (2007) Can antidepressants prevent psychosis? Lancet 370:1746–1748. [DOI] [PubMed] [Google Scholar]
- Glenthøj LB, Fagerlund B, Randers L, Hjorthøj CR, Wenneberg C, Krakauer K, Vosgerau A, Gluud C, Medalia A, Roberts DL, Nordentoft M (2015) The FOCUS trial: cognitive remediation plus standard treatment versus standard treatment for patients at ultra-high risk for psychosis: study protocol for a randomised controlled trial. Trials 16:1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grent-‘t-Jong T, Gajwani R, Gross J, Gumley AI, Krishnadas R, Lawrie SM, Schwannauer M, Schultze-Lutter F, Uhlhaas PJ (2021) 40-Hz auditory steady-state responses characterize circuit dysfunctions and predict clinical outcomes in clinical high-risk for psychosis participants: a magnetoencephalography study. Biol Psychiatry 90:419–429. [DOI] [PubMed] [Google Scholar]
- Groeger AL, Cipollina C, Cole MP, Woodcock SR, Bonacci G, Rudolph TK, Rudolph V, Freeman BA, Schopfer FJ (2010) Cyclooxygenase-2 generates anti-inflammatory mediators from omega-3 fatty acids. Nat Chem Biol 6:433–441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Higgins JP, Savović J, Page MJ, Elbers RG, Sterne JA (2019) Assessing risk of bias in a randomized trial. In: Cochrane Handbook for Systematic Reviews of Intervention, (Cochrane.org), 205–228. Online Cochrane Training. https://training.cochrane.org/handbook/current/chapter-08. Accessed on August 31, 2023.
- Horrobin DF (1998) The membrane phospholipid hypothesis as a biochemical basis for the neurodevelopmental concept of schizophrenia. Schizophr Res 30:193–208. [DOI] [PubMed] [Google Scholar]
- Kantrowitz JT, Woods SW, Petkova E, Cornblatt B, Corcoran CM, Chen H, Silipo G, Javitt DC (2015) D-serine for the treatment of negative symptoms in individuals at clinical high risk of schizophrenia: a pilot double-blind placebo-controlled randomised parallel group mechanistic proof-of-concept trial. Lancet Psychiatry 2:403–412. [DOI] [PubMed] [Google Scholar]
- Knochel C, Voss M, Gruter F, Alves GS, Matura S, Sepanski B, Stablein M, Wenzler S, Prvulovic D, Carvalho AF (2015) Omega 3 fatty acids: novel neurotherapeutic targets for cognitive dysfunction in mood disorders and schizophrenia? Curr Neuropharmacol 13:663–680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kristensen TD, Glenthøj LB, Ambrosen K, Syeda W, Raghava JM, Krakauer K, Wenneberg C, Fagerlund B, Pantelis C, Glenthøj BY, Nordentoft M, Ebdrup BH (2021) Global fractional anisotropy predicts transition to psychosis after 12 months in individuals at ultra-high risk for psychosis. Acta Psychiat Scand 144:448–463. [DOI] [PubMed] [Google Scholar]
- Madore C, Leyrolle Q, Morel L, Rossitto M, Greenhalgh A, Delpech J, Martinat M, Bosch-Bouju C, Bourel J, Rani B (2020) Essential omega-3 fatty acids tune microglial phagocytosis of synaptic elements in the mouse developing brain. Nat Commun 11:6133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGlashan TH, Zipursky RB, Perkins D, Addington J, Miller T, Woods SW, AHawkins K, EHoffman R, Preda A, Epstein I (2006) Randomized double-blind trial of olanzapine versus placebo in patients prodromally symptomatic for psychosis. Am J Psychiat 163:790–799. [DOI] [PubMed] [Google Scholar]
- McGorry PD, Yung AR, Phillips LJ, Yuen HP, Francey S, Cosgrave EM, Germano D, Bravin J, McDonald T, Blair A, Adlard S, Jackson H (2002) Randomized controlled trial of interventions designed to reduce the risk of progression to first-episode psychosis in a clinical sample with subthreshold symptoms. Arch Gen Psychiatry 59:921–928. [DOI] [PubMed] [Google Scholar]
- McGorry PD, Nelson B, Phillips LJ, Yuen HP, Francey SM, Thampi A, Berger GE, Amminger P, Simmons MB, Kelly D (2013) Randomized controlled trial of interventions for young people at ultra-high risk of psychosis: twelve-month outcome. J Clin Psychiatry 74:10030. [DOI] [PubMed] [Google Scholar]
- Moher D, Liberati A, Tetzlaff J, Altman DG, Group P (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med 151:264–269. [DOI] [PubMed] [Google Scholar]
- National Institute for Health and Care Excellence (2014) Psychosis and schizophrenia in adults: prevention and management. Guideline CG178. London: National Institute for Health and Care Excellence. [Google Scholar]
- Nelson B, et al. (2018) NEURAPRO: a multi-centre RCT of omega-3 polyunsaturated fatty acids versus placebo in young people at ultra-high risk of psychotic disorders—medium-term follow-up and clinical course. NPJ Schizophr 4:11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nieman DH, Rike WH, Becker HE, Dingemans PM, van Amelsvoort TA, de Haan L, van der Gaag M, Denys DAJP, Linszen DH (2009) Prescription of antipsychotic medication to patients at ultra high risk of developing psychosis. Int Clin Psychopharmacol 24:223–228. [DOI] [PubMed] [Google Scholar]
- Pawełczyk A, Łojek E, Żurner N, Kotlicka-Antczak M, Pawełczyk T (2021) Higher order language impairments can predict the transition of ultrahigh risk state to psychosis—An empirical study. Early Interv Psychiatry 15:314–327. [DOI] [PubMed] [Google Scholar]
- Raballo A, Poletti M, Preti A (2023) Do antidepressants prevent transition to psychosis in individuals at clinical high-risk (CHR-P)? Systematic review and meta-analysis. Psychol Med 53:4550–4560. [DOI] [PubMed] [Google Scholar]
- Robertson RC, Oriach CS, Murphy K, Moloney GM, Cryan JF, Dinan TG, Ross RP, Stanton C (2017) Omega-3 polyunsaturated fatty acids critically regulate behaviour and gut microbiota development in adolescence and adulthood. Brain Behav Immun 59:21–37. [DOI] [PubMed] [Google Scholar]
- Rodolico A, Bighelli I, Avanzato C, Concerto C, Cutrufelli P, Mineo L, Schneider-Thoma J, Siafis S, Signorelli MS, Wu H, Wang D, Furukawa TA, Pitschel-Walz G, Aguglia E, Leucht S (2022) Family interventions for relapse prevention in schizophrenia: a systematic review and network meta-analysis. Lancet Psychiatry, 9:211–221. [DOI] [PubMed] [Google Scholar]
- Schmidt SJ, Schultze-Lutter F, Schimmelmann BG, Maric NP, Salokangas RKR, Riecher-Rössler A, van der Gaag M, Meneghelli A, Nordentoft M, Marshall M, Morrison A, Raballo A, Klosterkötter J, Ruhrmann S (2015) EPA guidance on the early intervention in clinical high risk states of psychoses. European Psychiatry 30:388–404. [DOI] [PubMed] [Google Scholar]
- Schultze-Lutter F, Klosterkötter J, Ruhrmann S (2014) Improving the clinical prediction of psychosis by combining ultra-high risk criteria and cognitive basic symptoms. Schizophr Res 154:100–106. [DOI] [PubMed] [Google Scholar]
- Solmi M, Soardo L, Kaur S, Azis M, Cabras A, Censori M, Fausti L, Besana F, Salazar de Pablo G, Fusar-Poli P (2023) Meta-analytic prevalence of comorbid mental disorders in individuals at clinical high risk of psychosis: the case for transdiagnostic assessment. Mol Psychiatry 28:2291–2300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sterne JA, et al. (2016) ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 355:i4919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stillwell W, Shaikh SR, Zerouga M, Siddiqui R, Wassall SR (2005) Docosahexaenoic acid affects cell signaling by altering lipid rafts. Reprod Nutr Dev 45:559–579. [DOI] [PubMed] [Google Scholar]
- Susai SR, et al. (2022) Evidence that complement and coagulation proteins are mediating the clinical response to omega-3 fatty acids: a mass spectrometry-based investigation in subjects at clinical high-risk for psychosis. Transl Psychiatry 12:454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Tricht MJ, Nieman DH, Koelman JHTM, van der Meer JN, Bour LJ, de Haan L, Linszen DH (2010) Reduced parietal P300 amplitude is associated with an increased risk for a first psychotic episode. Biol Psychiatry 68:642–648. [DOI] [PubMed] [Google Scholar]
- Woods S, Saksa J, Compton M, Daley M, Rajarethinam R, Graham K, Breitborde N, Cahill J, Srihari V, Perkins D, Bearden C, Cannon T, Walker E, McGlashan T (2017) Effects of ziprasidone versus placebo in patients at clinical high risk for psychosis. Schizophr Bull 43:S58–S58. [Google Scholar]
- Yoviene Sykes LA, et al. (2020) Predictive validity of conversion from the clinical high risk syndrome to frank psychosis. Schizophr Res 216:184–191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang T, Xu L, Tang X, Wei Y, Hu Q, Hu Y, Cui H, Tang Y, Hui L, Li C, Cao LP, Lu Z, Wang JJ (2020) Real-world effectiveness of antipsychotic treatment in psychosis prevention in a 3-year cohort of 517 individuals at clinical high risk from the SHARP (ShangHai At Risk for Psychosis). Aust N Z J Psychiatry 54:696–706. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All the data extracted from included studies are publicly available in PubMed, Web of Science, EMBASE, China National Knowledge Infrastructure, and WanFang Data.