Key Points
Question
Does the increase in risk of transition to psychosis associated with baseline antipsychotic dose in individuals at clinical high risk of psychosis (CHR-P) follow a dose-effect pattern?
Findings
In this systematic review and meta-analysis of 290 individuals at CHR-P from 8 studies, those who were exposed to higher antipsychotic doses at baseline had an increased likelihood of transitioning to psychosis compared to those exposed to lower doses.
Meaning
The findings suggest that evaluating baseline antipsychotic dosage could enhance current CHR-P criteria-based risk stratification and improve the precision of outcome prediction models, leading to a more personalized approach to treatment management.
This systematic review and meta-analysis evaluates the association of baseline antipsychotic dose and transition to psychosis among individuals at high risk for psychosis.
Abstract
Importance
Emerging meta-analytical evidence indicates that baseline exposure to antipsychotics is associated with an increased risk of transitioning to psychosis in individuals at clinical high-risk for psychosis (CHR-P) and that such effect is not a result of pretest risk enrichment. However, to maximize its translational utility for prognostic stratification in clinical practice, testing for the potential presence of a dose-response association is crucial.
Objective
To test whether the negative prognostic effect of baseline antipsychotic exposure in individuals at CHR-P follows a dose-effect pattern, as indicated by mean chlorpromazine equivalent doses (CPZ-ED).
Data Sources
MEDLINE and Cochrane Library, performed up to August 31, 2023, searching for English-language studies on individuals at CHR-P reporting data on exposure to antipsychotics at baseline and detailed information on dosage by transition status.
Study Selection
Studies that provided information on antipsychotic exposure at baseline and included detailed dosage data categorized by transition status.
Data Extraction and Synthesis
Eligible studies were identified following PRISMA guidelines and evaluated independently by 2 reviewers with the Newcastle-Ottawa Scale for assessing the quality of nonrandomized studies in meta-analyses.
Main Outcomes and Measures
The primary outcome was transition to psychosis in individuals at CHR-P who were receiving antipsychotic treatment at baseline, measured by baseline mean CPZ-ED in individuals at CHR-P who transitioned to psychosis compared to those who did not.
Results
Eight studies were included in the systematic review and meta-analysis. Among 290 individuals at CHR-P (mean [SD] age, 19.4 [2.6] years) who were exposed to antipsychotics at baseline and remained in contact up to the completion of the study, 66 converted to psychosis and 224 did not. The mean CPZ-ED ranged 60 to 395 mg/d in those who converted and 13 to 224 mg/d in those who did not. Those who converted to psychosis had higher CPZ-ED than those who did not in both the common-effects model (Hedges g, 0.41; 95% CI, 0.12-0.70; z, 2.78; P = .005) and in the random-effects model (Hedges g, 0.41; 95% CI, 0.15-0.67; z, 3.69; P = .008; τ2, 0.0). There was no relevant heterogeneity (Cochran Q, 3.99; df, 7; P = .78; I2, 0.0%; 95% CI, 0.0-68.0). The radial plot indicated a good fit of the model.
Conclusions and Relevance
In individuals at CHR-P who were exposed to antipsychotics at baseline, those receiving higher antipsychotic doses demonstrated an increased likelihood of transitioning to psychosis. This meta-analytic evidence of putative dose-effect association confirms that baseline antipsychotic exposure and the corresponding dosage carry salient prognostic information that could improve current CHR-P criteria-based risk stratification at inception.
Introduction
Recent meta-analyses have found that baseline exposure to antipsychotics is associated with an increase in risk of transition to psychosis in individuals at clinical high risk for psychosis (CHR-P) compared to individuals at CHR-P not exposed to antipsychotics at baseline (29% vs 16%),1,2 independent of pretest risk enrichment3 and further corroborated in CHR-P cohorts.4,5 While additional research is necessary to identify possible underlying causal factors, the clinical implications of such meta-analytic effect for risk stratification, prognostic precision, and personalized treatment are important.1,2,3 Indeed, individuals at CHR-P already receiving antipsychotic treatment at baseline constitute a readily identifiable subgroup at higher imminent risk of progressing to psychosis, which, although not formally captured by current CHR-P categories,6 is empirically signaled by the treating clinicians’ decision to prescribe antipsychotics. Nonetheless, it remains uncertain whether antipsychotic prescription is a risk factor in itself or whether antipsychotic dosage may also retain tangible prognostic value.
Thanks to the growing awareness of the issue of baseline antipsychotic prescription,1,2,3,4,5,6 recent CHR-P studies have adopted more transparent reporting habits, enabling the search of meta-analytic signals of dose-effect associations within the overall negative prognostic effect of baseline antipsychotic exposure. In this systematic review and meta-analysis, we tested if higher antipsychotic baseline doses among individuals at CHR-P were associated with a higher likelihood of transition to psychosis.
Methods
The current study followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) reporting guideline.7 Pairwise meta-analysis was applied to the differences in chlorpromazine equivalent doses (CPZ-ED) between individuals at CHR-P who were exposed to antipsychotics at baseline and converted to psychosis and control individuals (individuals at CHR-P who were exposed to antipsychotics at baseline and did not convert). The effect size was expressed as the bias-corrected standardized mean score (Hedges g) with 95% CIs. The threshold for statistically significant results was set at 2-sided P < .05; heterogeneity was assessed with Cochran Q and I2 statistics, and publication bias was evaluated by the trim-and-fill procedure. The radial plot was used to assess model adequacy, with large standardized residuals (above 2, as a rule of thumb) suggesting that the study does not fit the assumed model. See the eMethods in Supplement 1 for further details.
Results
Our search yielded 2008 articles, 425 of which were assessed by full text and 8 of which met the inclusion criteria (eFigure 1 in Supplement 1). The Table synthetizes the main characteristics of the included studies,4,8,9,10,11,12,13,14 whereas their general characteristics are reported in the eResults in Supplement 1.
Table. Included Studies Reporting Mean Chlorpromazine Equivalent Doses (CPZ-ED) of Antipsychotics at Baseline in Relation to Transition to Psychosis.
| Source | Location | Baseline CHR-P sample, No. | Follow-up, mo | Follow-up sample, No. | Raw transition, No. | UHR instrument | Baseline antipsychotic exposure, No./total No. (%) | Age, mean (SD), y | Female, No./total No. (%) | AP baseline CV, No. | AP baseline NCV, No. | CPZ-ED in CV, mean (SD), mg/d | CPZ-ED in NCV, mean (SD), mg/d |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bang et al8 | Korea | 77 | 24 | 77 | 16 | SIPS | 24/77 (31.2) | 19.9 (3.4) | 31/77 (40.3) | 4 | 20 | 170.8 (170.3) | 122.5 (84.2) |
| Bourgin et al9 | France | 46 | 22 | 27 | 11 | CAARMS | 11/27 (40.7) | 17.6 (3.7) | 4/27 (14.8) | 5 | 6 | 178 (115) | 119 (158) |
| Nägele et al10 | Germany | 24 | 15 | 24 | 8 | SIPS | 8/24 (33.3) | 21.3 (3.5) | 12/24 (50.0) | 3 | 5 | 150 (132.29) | 180 (172.16) |
| Yoviene Sykes et al11 | US | 764 | 12 | 431 | 33 | SIPS | 88/431 (20.4) | 19.1 (4.4) | 181/431 (41.8) | 21 | 67 | 395 (348) | 224 (170) |
| Kristensen et al12 | Denmark | 110 | 12 | 110 | 10 | CAARMS | 37/110 (33.6) | 24 (4) | 58/110 (52.7) | 4 | 33 | 60 (106) | 71 (175) |
| Tateno et al13 | Japan | 39 | 24 | 39 | 11 | CAARMS | 9/39 (23.1) | 18.5 (4.2) | 14/39 (35.9) | 5 | 4 | 95.4 (135.1) | 13.5 (39.4) |
| Fortea et al14 | Spain | 54 | 18 | 49 | 13 | SIPS | 22/49 (44.8) | 15 (1.8) | 36/49 (73.46) | 9 | 13 | 62.8 (75.2) | 40 (76.6) |
| Pelizza et al4 | Italy | 178 | 12 | 159 | 22 | CAARMS | 91/178 (51.1) | 19.54 (3.35) | 88/178 (49.5) | 15 | 76 | 180 (134.4) | 156 (112.2) |
| Summary | Europe, 5; Asia, 2; US, 1 | Mean (SD), 161 (248) | Range, 12-24 | Mean (SD), 114 (136) | Mean (SD), 20.2% (9.5%) | SIPS, 4; CAARMS, 4 | Mean (SD), 34.8% (10.4%) | Mean (SD), 19.4 (2.6) | Mean (SD), 45% (16%) | Mean (SD), 8 (6) | Mean (SD), 28 (28) | Mean (SD), 161.5 (106.7) | Mean (SD), 115.7 (71.4) |
Abbreviations: AP, antipsychotics; CAARMS, Comprehensive Assessment At Risk Mental States; CHR-P, clinical high risk for psychosis; CV, individuals who converted to psychosis; NCV, individuals who did not convert to psychosis; UHR, ultra–high risk; SIPS, Structured Interview for Prodromal Symptoms.
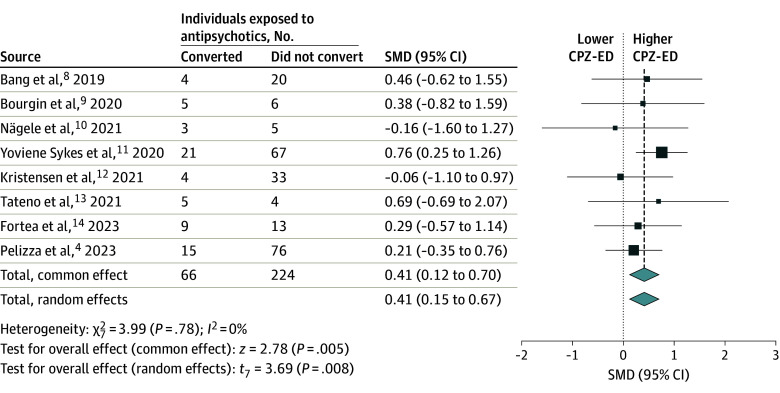
In the merged sample of CHR-P undergoing baseline antipsychotic treatment, 290 individuals (mean [SD] age, 19.4 [2.6] years) were followed up to the completion of the source studies; 66 of them transitioned to psychosis and 224 did not. Information on mean doses of antipsychotics at baseline concerned this sample. The mean (SD) CPZ-ED received by individuals at CHR-P who transitioned to psychosis was 161.5 (106.7) mg/d (range, 60-395 mg/d) compared to 115.7 (71.4) mg/d (range, 13-224 mg/d) in those who did not convert. In 5 studies, the mean CPZ-ED was greater than 150 mg/d in those who converted vs 3 studies in those who did not convert. Among those exposed to antipsychotics at baseline, individuals at CHR-P who converted to psychosis had higher CPZ-ED than those who did not convert to psychosis in both the common-effects model (Hedges g, 0.41; 95% CI, 0.12-0.70; z, 2.78; P = .005) and in the random-effects model (Hedges g, 0.41; 95% CI, 0.15-0.67; z, 3.69; P = .008; τ2, 0.0) (Figure). A daily dose of 150 to 200 mg is considered the minimum CPZ-ED for antipsychotic effectiveness in schizophrenia (eTable in Supplement 1). There was no relevant heterogeneity (Cochran Q, 3.99; df, 7; P = .78; I2, 0.0%; 95% CI, 0.0-68.0). The radial plot indicated a good fit of the model (eFigure 2 in Supplement 1). There was modest asymmetry in the funnel plot, and the trim-and-fill method suggested that 2 other studies, both on the side of a greater impact of antipsychotic dosage on the risk of transition, should be added to increase symmetry (eFigure 3 in Supplement 1).
Figure. Chlorpromazine-Equivalent Antipsychotic Dose (CPZ-ED) Differences Between Individuals Who Converted to Psychosis and Those Who Did Not.
Shown are differences between individuals at clinical high risk of psychosis who were exposed to antipsychotics at baseline and converted to psychosis or did not convert to psychosis. Effect sizes were calculated as Hedges g. Squares are proportional to the weight assigned to the study in the analysis.
Discussion
The findings in this systematic review and meta-analysis confirmed that among individuals at CHR-P who were exposed to antipsychotics at baseline (ie, a specific CHR-P subgroup with even higher documented higher risk of transition to psychosis1,2,3,4), those who converted to psychosis were prescribed higher antipsychotic doses compared to those who did not convert. The results were congruent between the common-effects model and the random-effects models, with an effect size approaching a moderate level. No heterogeneity was detected, and the radial plot suggested a good fit of the model. Data were extrapolated from primary studies that were not originally intended to test the 8 prognostic effects of baseline antipsychotics; therefore, we did not expect relevant publication bias in this respect. The trim-and-fill method suggested that, if there is publication bias, it is in favor of future studies with a larger difference between those who converted to psychosis and those who did not in terms of CPZ-ED. Quality of the studies was reasonably good, with 3 studies having a poor quality (eMethods and eResults in Supplement 1).
These findings are congruent with the hypothesis that the dosage of baseline antipsychotic prescription in individuals at CHR-P carries substantial prognostic information. Crucially, the minimum CPZ-ED effective for acute schizophrenia was approached more often in those who converted to psychosis than in those who did not, and some of those who converted were plausibly exposed to CPZ-EDs that are deemed effective in controlling psychotic symptoms (eTable in Supplement 1). This aligns with the previously formulated hypothesis of a pharmacologically attenuated first-episode psychosis6 to explain the higher chance of transition in individuals at CHR-P who are exposed to antipsychotics at baseline (ie, ongoing antipsychotic treatment at baseline could clinically mask already-developed psychosis, which might only later be fully detected at a psychometric level). Furthermore, it confirms that a fraction of individuals at CHR-P, despite belonging to stage 1b of the clinical staging model of psychosis, are being treated as though they are at stage 2 (ie, first episode psychosis). Their conflation in the global CHR-P group may deteriorate precision in outcome prediction as well as in treatment.
Dropout rates in the analyzed samples were, on average, 15% (eResults in Supplement 1), yet the studies did not detail information on dropout according to the transition to psychosis status. However, 3 of the included studies reported that individuals at CHR-P who were exposed to antipsychotics were less likely to drop out from treatment than those who were not exposed. When investigated in a sample of 288 individuals at CHR-P (follow-up = 24 months), no clinical, functional, or demographic variable was identified as related to the likelihood of dropping out.15 Overall, the higher retention in treatment of individuals at CHR-P who were exposed to antipsychotics and the lack of predictive variables for dropping out, including the severity of psychopathology, suggest that the data reviewed in this meta-analysis are plausibly representative of the impact of antipsychotic dosage on transition to psychosis.
Data were extrapolated from primary studies that were not originally intended to test the prognostic effect of baseline antipsychotics; therefore, we did not expect relevant publication bias in this respect.
Limitations
The numerosity of the studies precluded sensitivity analyses with potential confounders via meta-regression. Also, included original studies did not systematically detail baseline psychopathological information for those who converted to psychosis and those who did not. Similarly, they did not systematically specify whether antipsychotic prescription was continued or suspended at follow-up. Among the few studies specifying it, 100% of individuals at CHR-P who converted to psychosis were receiving antipsychotic medication at follow-up in 1 study, and about two-thirds in 3 other studies.
Conclusions
The results in this study confirm that baseline antipsychotic dose matters for negative prognostic effect in terms of increased risk of transition to psychosis. This coheres with the accumulating evidence that baseline psychopharmacological treatment is a widely neglected yet relevant prognostic modulator1,6 that might mask the potential effectiveness of some of the treatments currently prescribed to individuals at CHR-P who are seeking treatment. Furthermore, the current study indicates the salience of baseline antipsychotic exposure and respective dosage for the initial prognostic staging of individuals at CHR-P.
In other words, these parameters should be included in the risk stratification of individuals at CHR-P to enhance prognostic precision in predictive models. Besides contributing to the further refinement of the clinical staging model of psychosis, this would enable a more tailored and personalized treatment management in individuals at CHR-P.
eMethods. Supplementary details
eResults. General characteristics of the studies
eTable. Current evidence on chlorpromazine equivalent doses (CPZ-ED) and efficacy of antipsychotics in acute schizophrenia
eFigure 1. PRISMA flowchart of studies reporting data on chlorpromazine-equivalent antipsychotic dose in CHR-P exposed at baseline to antipsychotics
eFigure 2. (Galbraith) Radial plot of the standardized effect (in z-scores, on the vertical axis) against the inverse of standard error (on the horizontal axis) in studies comparing CHR-P exposed to antipsychotic at baseline who converted to psychosis and those who did not
eFigure 3. Trim-and-Fill funnel plot, reporting studies included in the meta-analysis (squares), and studies added to make the funnel plot more symmetrical (triangles)
Data sharing statement
References
- 1.Raballo A, Poletti M, Preti A. Meta-analyzing the prevalence and prognostic effect of antipsychotic exposure in clinical high-risk (CHR): when things are not what they seem. Psychol Med. 2020;50(16):2673-2681. doi: 10.1017/S0033291720004237 [DOI] [PubMed] [Google Scholar]
- 2.Raballo A, Poletti M, Preti A. The temporal dynamics of transition to psychosis in individuals at clinical high-risk (CHR-P) shows negative prognostic effects of baseline antipsychotic exposure: a meta-analysis. Transl Psychiatry. 2023;13(1):112. doi: 10.1038/s41398-023-02405-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Raballo A, Poletti M, Preti A. Negative prognostic effect of baseline antipsychotic exposure in clinical high risk for psychosis (CHR-P): is pre-test risk enrichment the hidden culprit? Int J Neuropsychopharmacol. 2021;24(9):710-720. doi: 10.1093/ijnp/pyab030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Pelizza L, Leuci E, Quattrone E, et al. Baseline antipsychotic prescription and short-term outcome indicators in individuals at clinical high-risk for psychosis: findings from the Parma At-Risk Mental States (PARMS) program. Early Interv Psychiatry. 2023; Advance online publication. doi: 10.1111/eip.13434 [DOI] [PubMed] [Google Scholar]
- 5.Zeng J, Raballo A, Gan R, et al. Antipsychotic exposure in clinical high risk of psychosis: empirical insights from a large cohort study. J Clin Psychiatry. 2022;83(3):21m14092. doi: 10.4088/JCP.21m14092 [DOI] [PubMed] [Google Scholar]
- 6.Raballo A, Poletti M, Preti A. Attenuated psychosis syndrome or pharmacologically attenuated first-episode psychosis? an undesirably widespread confounder. JAMA Psychiatry. 2020;77(12):1213-1214. doi: 10.1001/jamapsychiatry.2020.1634 [DOI] [PubMed] [Google Scholar]
- 7.Rethlefsen ML, Kirtley S, Waffenschmidt S, et al. ; PRISMA-S Group . PRISMA-S: an extension to the PRISMA Statement for Reporting Literature Searches in Systematic Reviews. Syst Rev. 2021;10(1):39. doi: 10.1186/s13643-020-01542-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bang M, Park JY, Kim KR, et al. Psychotic conversion of individuals at ultra-high risk for psychosis: the potential roles of schizotypy and basic symptoms. Early Interv Psychiatry. 2019;13(3):546-554. doi: 10.1111/eip.12518 [DOI] [PubMed] [Google Scholar]
- 9.Bourgin J, Duchesnay E, Magaud E, Gaillard R, Kazes M, Krebs MO. Predicting the individual risk of psychosis conversion in at-risk mental state (ARMS): a multivariate model reveals the influence of nonpsychotic prodromal symptoms. Eur Child Adolesc Psychiatry. 2020;29(11):1525-1535. doi: 10.1007/s00787-019-01461-y [DOI] [PubMed] [Google Scholar]
- 10.Nägele FL, Pasternak O, Bitzan LV, et al. Cellular and extracellular white matter alterations indicate conversion to psychosis among individuals at clinical high-risk for psychosis. World J Biol Psychiatry. 2021;22(3):214-227. doi: 10.1080/15622975.2020.1775890 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yoviene Sykes LA, Ferrara M, Addington J, et al. Predictive validity of conversion from the clinical high risk syndrome to frank psychosis. Schizophr Res. 2020;216:184-191. doi: 10.1016/j.schres.2019.12.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kristensen TD, Glenthøj LB, Ambrosen K, et al. Global fractional anisotropy predicts transition to psychosis after 12 months in individuals at ultra-high risk for psychosis. Acta Psychiatr Scand. 2021;144(5):448-463. doi: 10.1111/acps.13355 [DOI] [PubMed] [Google Scholar]
- 13.Tateno T, Higuchi Y, Nakajima S, et al. Features of duration mismatch negativity around the onset of overt psychotic disorders: a longitudinal study. Cereb Cortex. 2021;31(5):2416-2424. doi: 10.1093/cercor/bhaa364 [DOI] [PubMed] [Google Scholar]
- 14.Fortea A, van Eijndhoven P, Calvet-Mirabent A, et al. Age-related change in cortical thickness in adolescents at clinical high risk for psychosis: a longitudinal study. Eur Child Adolesc Psychiatry. Published online August 29, 2023. doi: 10.1007/s00787-023-02278-6 [DOI] [PubMed] [Google Scholar]
- 15.Stowkowy J, Liu L, Cadenhead KS, et al. Exploration of clinical high-risk dropouts. Schizophr Res. 2018;195:579-580. doi: 10.1016/j.schres.2017.09.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
eMethods. Supplementary details
eResults. General characteristics of the studies
eTable. Current evidence on chlorpromazine equivalent doses (CPZ-ED) and efficacy of antipsychotics in acute schizophrenia
eFigure 1. PRISMA flowchart of studies reporting data on chlorpromazine-equivalent antipsychotic dose in CHR-P exposed at baseline to antipsychotics
eFigure 2. (Galbraith) Radial plot of the standardized effect (in z-scores, on the vertical axis) against the inverse of standard error (on the horizontal axis) in studies comparing CHR-P exposed to antipsychotic at baseline who converted to psychosis and those who did not
eFigure 3. Trim-and-Fill funnel plot, reporting studies included in the meta-analysis (squares), and studies added to make the funnel plot more symmetrical (triangles)
Data sharing statement