Key Points
Question
Is hypoplastic pitted amelogenesis imperfecta a feature of Kindler epidermolysis bullosa?
Findings
In this longitudinal cohort study, deep phenotyping of orofacial features identified enamel structure abnormalities in all 11 patients for whom data were available.
Meaning
These findings suggest that hypoplastic pitted amelogenesis imperfecta is a feature of Kindler epidermolysis bullosa, underscoring the extent and severity of oral manifestations in Kindler epidermolysis bullosa and the need for early and sustained dental care.
This cohort study examines whether hypoplastic pitted amelogenesis imperfecta is a feature of Kindler epidermolysis bullosa, a genetic skin-blistering disease associated with recessive inherited pathogenic variants in FERMT1, which encodes kindlin-1.
Abstract
Importance
Kindler epidermolysis bullosa is a genetic skin-blistering disease associated with recessive inherited pathogenic variants in FERMT1, which encodes kindlin-1. Severe orofacial manifestations of Kindler epidermolysis bullosa, including early oral squamous cell carcinoma, have been reported.
Objective
To determine whether hypoplastic pitted amelogenesis imperfecta is a feature of Kindler epidermolysis bullosa.
Design, Settings, and Participants
This longitudinal, 2-center cohort study was performed from 2003 to 2023 at the Epidermolysis Bullosa Centre, University of Freiburg, Germany, and the Special Care Dentistry Clinic, University of Chile in association with DEBRA Chile. Participants included a convenience sampling of all patients with a diagnosis of Kindler epidermolysis bullosa.
Main Outcomes and Measures
The primary outcomes were the presence of hypoplastic pitted amelogenesis imperfecta, intraoral wounds, gingivitis and periodontal disease, gingival hyperplasia, vestibular obliteration, cheilitis, angular cheilitis, chronic lip wounds, microstomia, and oral squamous cell carcinoma.
Results
The cohort consisted of 36 patients (15 female [42%] and 21 male [58%]; mean age at first examination, 23 years [range, 2 weeks to 70 years]) with Kindler epidermolysis bullosa. The follow-up ranged from 1 to 24 years. The enamel structure was assessed in 11 patients, all of whom presented with enamel structure abnormalities. The severity of hypoplastic pitted amelogenesis imperfecta varied from generalized to localized pitting. Additional orofacial features observed include gingivitis and periodontal disease, which was present in 90% (27 of 30 patients) of those assessed, followed by intraoral lesions (16 of 22 patients [73%]), angular cheilitis (24 of 33 patients [73%]), cheilitis (22 of 34 patients [65%]), gingival overgrowth (17 of 26 patients [65%]), microstomia (14 of 25 patients [56%]), and vestibular obliteration (8 of 16 patients [50%]). Other features included chronic lip ulcers (2 patients) and oral squamous cell carcinoma with lethal outcome (2 patients).
Conclusions and Relevance
These findings suggest that hypoplastic pitted amelogenesis imperfecta is a feature of Kindler epidermolysis bullosa and underscore the extent and severity of oral manifestations in Kindler epidermolysis bullosa and the need for early and sustained dental care.
Introduction
Epidermolysis bullosa (EB) is a group of genetic disorders characterized by skin and mucosal blistering within the basement membrane zone. Kindler epidermolysis bullosa (KEB) is the rarest EB type and is associated with biallelic pathogenic variants in FERMT1, which encodes kindlin-1, an intracellular protein of focal adhesions. The main role of kindlins is to increase integrin affinity for ligands through binding of β-integrin tails.1 Kindlin-mediated integrin activation and integrin-ligand binding promote cell adhesion, spreading, and migration; assembly of the extracellular matrix; and cell survival, proliferation, and differentiation.2 Lack of integrin activation in the gingiva is associated with severe early-onset periodontitis in patients with KEB.3 Here, we describe orofacial features in a cohort of patients with KEB, including for the first time amelogenesis imperfecta as a manifestation of FERMT1 pathogenic variants.
Methods
This longitudinal cohort study was performed at the EB Centre, University of Freiburg, Freiburg, Germany (2003-2023), and the Special Care Dentistry Clinic, University of Chile, Santiago, Chile, in association with DEBRA Chile (2005-2023). A retrospective analysis of medical records was performed by 3 calibrated clinicians (S.K., A.L.H., and Y.W.; interexaminer and intraexaminer κ values ranged from 0.9 to 1.0). Discrepancies were discussed during a consensus meeting. Ethical approval was obtained from the University of Freiburg and the University of Chile. Patients gave written consent for publication of their clinical pictures and oral consent for clinical assessment. The report follows the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guideline for cohort studies.4 We used Excel software version 2021 (Microsoft) to manage the data.
Results
Generalized pitted hypoplastic amelogenesis imperfecta was first diagnosed in patient 26 in his thirties (Table and Figure 1). Because reports of FERMT1-associated amelogenesis imperfecta were lacking,5 we proceeded in validating this finding.
Table. Orofacial Features in Patients With Kindler Epidermolysis Bullosaa.
| Patient No. | Age at examination, y | Sex | FERMT1 pathogenic variants c.DNA | Kindlin-1 protein | Pitted enamel hypoplasia | Caries/tooth loss | Intraoral erosions/ulcers | Chronic lip ulcers | Cheilitis | Angular cheilitis | Oral squamous cell carcinoma | Gingivitis or periodontal disease | Gingival hyperplasia (overgrowth) | Microstomia | Vestibular obliteration | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| First | Last | |||||||||||||||
| 1 | <1 | 1-5 | Male | 676dupC | Q226Pfs*16 | NA | NA | Absent | Absent | Absent | Absent | Absent | Absent | Absent | Absent | NA |
| 2 | 1-5 | 1-5 | Male | 676delC;IVS9 + 740G>A | Q226Sfs*25;SS | NA | NAb | Absent | Absent | Absent | Absent | Absent | Absent | Absent | Absent | NA |
| 3 | 1-5 | 1-5 | Male | 1140-2A>T | SS | NA | NA | Absent | Absent | Absent | Absent | Absent | Present | Absent | Absent | Absent |
| 4 | 6-12 | 6-12 | Female | 1140-2A>T | SS | NA | Present | Absent | Absent | Absent | Absent | Absent | Present | Present | Absent | Absent |
| 5 | 6-12 | 6-12 | Male | 676dupC;877T>G | Q226Pfs*16;Y293D | Present | NA | NA | NA | Absent | Present | Absent | Present | NA | NA | NA |
| 6 | 6-12 | 6-12 | Male | 190G>T | E64* | NA | NA | NA | NA | Absent | Present | Absent | Absent | Absent | NA | NA |
| 7 | 6-12 | 6-12 | Male | 907G>T | E303* | NA | NA | NA | NA | Present | Absent | Absent | NA | NA | NA | NA |
| 8 | 1-5 | 6-12 | Male | IVS9 + 740G>A | SS | NA | Present | NA | NA | Present | Absent | Absent | Present | Present | NA | NA |
| 9 | 1-5 | 13-19 | Female | 385 + 2T>C | SS | Present | Absent | Presentc | Absent | Present | Present | Absent | Present | Present | Absent | Absent |
| 10 | 1-5 | 13-19 | Female | 676dupC | Q226Pfs*16 | Present | Present | NA | Absent | Absent | Present | Absent | Present | Absent | Absent | Present |
| 11 | 1-5 | 13-19 | Male | IVS13_15del | Large deletion | Present | Present | Present | Present | Present | Present | Absent | Present | Present | Present | Present |
| 12 | 6-12 | 13-19 | Male | 676dupC;1677G>A | Q226Pfs*16;W559* | NA | NA | Present | Present | Present | Present | Absent | Present | Absent | Present | NA |
| 13 | 6-12 | 20s | Male | IVS13_15del | Large deletion | Present | Present | Present | Absent | Present | Present | Absent | Present | Present | Present | Absent |
| 14 | 20s | 20sd | Female | 676dupC | Q226Pfs*16 | NA | All teeth extracted at age 13 y | Absent | NA | Present | Present | Absent | NA | Present | Present | Present |
| 15 | 13-19 | 20sd | Female | IVS9 + 740G>A; IVS2_5del | SS; Large deletion | Present | Present | Present | NA | NA | NA | Absent | Present | Present | Absent | Present |
| 16 | 13-19 | 20s | Male | 910G>T | E304* | Present | NA | NA | NA | Present | Present | Absent | Present | NA | NA | NA |
| 17 | 20s | 20s | Male | 1729delA | S577Afs*14 | NA | All incisors crowned at age 22y | NA | NA | Present | Present | Absent | Present | NA | Absent | NA |
| 18 | 13-19 | 20s | Male | 1209C>G | Y403* | NA | Present | NA | Present | Present | Absent | Absent | NA | NA | NA | NA |
| 19 | 20s | 20s | Male | 170C>A | S57* | NA | NA | NA | NA | Present | Present | Absent | Present | NA | Present | NA |
| 20 | 20s | 20s | Female | IVS9_11del | Large deletion | NA | NA | Present | NA | Present | NA | Absent | Present | NA | Present | NA |
| 21 | 13-19 | 20s | Female | IVS9 + 740G>A | SS | Present | NA | Present | NA | NA | Present | Absent | Present | Present | Absent | Absent |
| 22 | 20s | 20s | Male | 1718 + 1G>A | SS | Present | NA | Present | Absent | Present | Present | Absent | Present | Absent | Absent | Absent |
| 23 | 20s | 20s | Female | 676dupC | Q226Pfs*16 | NA | All teeth crowned in mid 20s | Present | Absent | Present | Present | Absent | Present | Present | Present | Present |
| 24 | 20s | 20s | Female | IVS9 + 740G>A | SS | NA | NA | NA | NA | Absent | Present | Absent | NA | NA | NA | NA |
| 25 | 13-19 | 30sd | Male | 456dupA | D153Rfs*3 | NA | All teeth crowned at age 27 y | Present | Absent | Present | Present | At age ≥30 y | Present | Present | Present | NA |
| 26 | 30s | 30s | Male | IVS6_9del | Large deletion | Present | Present | Present | NA | Present | Present | Absent | Present | Absent | Present | Present |
| 27 | 30s | 30s | Female | IVS9 + 740G>A | SS | NA | All teeth extracted at age 28 y | Absent | NA | Absent | Present | Absent | Present | Present | Present | Present |
| 28 | 30s | 40s | Female | 676dupC | Q226Pfs*16 | NA | Present | Present | NA | Present | NA | Absent | Present | Present | NA | NA |
| 29 | 30s | 40sd | Male | 910G>T | E304* | NA | All teeth crowned at age 42 y | Present | Absent | Present | Present | Absent | Present | Present | Absent | NA |
| 30 | 20s | 50s | Female | IVS13_15del | Large deletion | Present | Present | Present | Present | Absent | Absent | Absent | Present | Present | Present | Absent |
| 31 | 30s | 50s | Male | IVS13_15del | Large deletion | NA | All teeth extracted at age 45 y | Present | Present | Present | Present | Absent | Present | Present | Present | Absent |
| 32 | 50s | 60sd | Female | IVS13_15del | Large deletion | NA | Present | Present | Absent | Present | Present | At age ≥53 y | Present | Present | NA | Present |
| 33 | 60s | 60s | Male | 328C>T | R110* | NA | NA | NA | Absent | Present | Present | Absent | Present | Present | Present | NA |
| 34 | 60s | 60s | Female | 1761T>A | R587* | NA | NA | NA | NA | Absent | Present | Absent | NA | NA | NA | NA |
| 35 | 60s | 60s | Male | IVS9 + 740G>A | SS | NA | Present | NA | NA | Present | Present | Absent | NA | NA | Present | NA |
| 36 | 70s | 70s | Female | 328C>T | R110* | NA | NA | NA | NA | Absent | Absent | Absent | Present | Absent | NA | NA |
Abbreviations: IVS, intron; NA, not available; SS, splice site variant.
If 1 variant indicated, then it is homozygous.
Refers to dyspigmentation of teeth.
Refers to recurrent mucocele.
Patient is deceased.
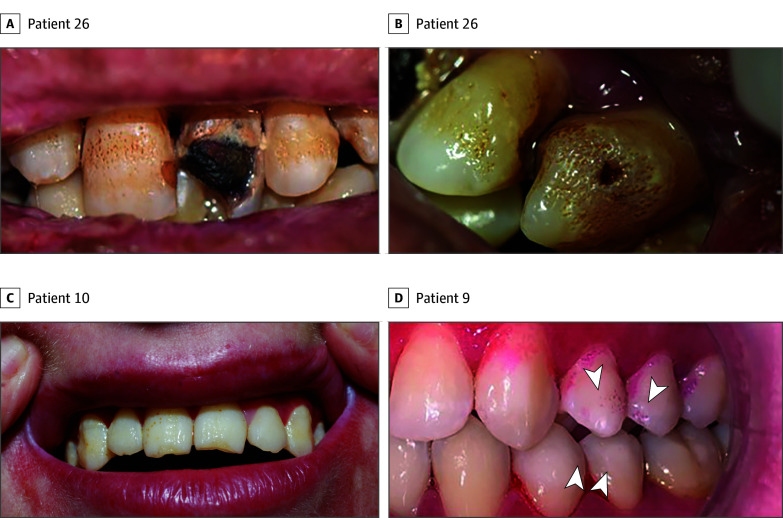
Figure 1. Syndromic Amelogenesis Imperfecta in Kindler Epidermolysis Bullosa.
A and B, Hypoplastic pitted syndromic amelogenesis imperfecta with generalized pits in patient 26. C, Generalized pitting in patient 10. D, Localized pits on upper and lower premolars in patient 9.
Validation of Amelogenesis Imperfecta in KEB
Clinical records of 65 patients with KEB were reviewed. Information on orofacial features was available for 36 patients (15 female [42%] and 21 male [58%]; mean age at first examination, 23 years [range, 2 weeks to 70 years]). Follow-up ranged from 1 to 24 years.
Clinical records of 11 patients included information on enamel structure. All had pitted enamel anomalies, but the severity of the pits was variable; for example, patients 10, 21, 22, and 26 showed generalized pitted enamel of most teeth (Figure 1A-1C), whereas patient 9 presented with localized pits on premolars only (Figure 1D). Among the patients whose enamel could not be analyzed, 3 had all teeth crowned in their twenties, suggesting possible generalized enamel defects, and 2 patients had all teeth extracted in their second or third decade of life, indicating severe periodontal disease (Table).
Additional Orofacial Anomalies and Their Management
The most common orofacial feature was periodontal disease, found in 75% of the cohort (27 of 36 patients), including 90% (27 of 30 patients) of those examined, followed by intraoral lesions (16 of 22 patients [73%]), angular cheilitis (24 of 33 patients [73%]), and cheilitis (22 of 34 patients [65%]) (Table). Two patients presented with chronic lip ulcers in their second decade of life (Figure 2A). On histopathologic examination, patient 12’s lesion was diagnosed as granuloma pyogenicum, whereas patient 18’s lesion was diagnosed as plasma cell cheilitis. Both lesions healed under topical clobetasol propionate, 0.05% twice daily (eFigure 1 in Supplement 1).
Figure 2. Orofacial Anomalies in Kindler Epidermolysis Bullosa.
A, Chronic wound on the lip of patient 12. B, Angular cheilitis in patient 25. C, The same patient 1 year later when oral squamous cell carcinoma was diagnosed. D, Gingival overgrowth. E, Severe limited mouth opening (microstomia) in patient 26.
Cheilitis angularis was a highly prevalent, but probably neglected, feature. Patient 25 presented with chronic angular cheilitis with Candida albicans and Staphylococcus aureus that did not heal over 19 years of follow-up. In his thirties, a verrucous squamous cell carcinoma (SCC) was diagnosed at that site (Figure 2B and 2C; eFigure 2 in Supplement 1). Despite surgery and anti–epidermal growth factor receptor therapy, the tumor relapsed and extended to the oral cavity, tongue, tonsilla, and lymph nodes, causing the patient’s death 4.5 years later. Patient 32 also developed SCC with lethal outcome at a site of chronic lesions on the lower lip.6
Gingivitis and periodontal disease represented a major clinical issue, even at young ages. Gingival overgrowth was diagnosed in 17 of the 26 patients (65%) assessed. It was present as early as the age of 3 years (patient 8). Histopathologic analysis of excised lesions of patient 23 showed chronic inflammation, granulation tissue, ulceration, and fibrosis with a plasma cell–rich inflammation. In patients 9, 23, 25, and 27, gingival overgrowth required surgical removal, with high relapse rate.
Scarring complications of the oral cavity were also common. Microstomia with variable severity was diagnosed in 14 of 25 patients assessed (56%). Patient 26 had a maximal mouth opening of 6 mm in his thirties (Figure 2E and eFigure 3 in Supplement 1). This limited his oral intake, affecting his general condition. After 2 oral maxillofacial surgical procedures, his mouth opening improved, but scarring relapsed 1 year later, despite mouth stretching exercises (eFigure 3D-3H in the Supplement 1). Obliteration of the oral vestibule was present in 8 of 16 assessed patients (50%), causing functional problems. Patients 15 and 26 required oral surgery to deepen the oral vestibule.
Discussion
The findings of this cohort study underscore the extent and severity of oral manifestations in KEB and the need for early and sustained dental care.7 We found enamel defects in the form of pitted amelogenesis imperfecta in all patients for whom information was available. Amelogenesis imperfecta is a heterogeneous group of rare genetic diseases presenting with developmental enamel defects (eg, hypoplasia, hypomaturation, and hypomineralization) that can exist in isolation or as part of a syndrome.5 Amelogenesis imperfecta can be associated with tooth hypersensitivity, tooth eruption disturbances, crown resorption, and posteruptive breakdown, as well as periodontal conditions such as gingival hyperplasia and periodontitis.8 Early diagnosis is important to establish interceptive strategies.7
Pathogenic variants in at least 100 genes have been reported to cause amelogenesis imperfecta.5 The genes encode proteins with a role in amelogenesis, acting as enamel matrix proteins or as proteases that degrade them, cell adhesion molecules, or regulators of calcium homeostasis.5 Pitted and generalized hypoplastic amelogenesis imperfecta has been associated with junctional EB caused by variants in the genes encoding laminin 332, α6β4 integrin, and type XVII collagen. Enamel abnormalities have been explained by the role of these proteins in cell adhesion within the inner dental epithelium, whose cells are giving rise after terminal differentiation to postmitotic highly specialized ameloblasts, producing and maturing dental enamel.7
Kindlin-1 is expressed during tooth development at the E14.5 cap stage both in molars and incisors before ameloblast differentiation.9,10 FERMT1 is expressed in mouse and fish dental epithelium at placode, bud, and/or cap stages,10 and also in differentiated secretory ameloblasts.11 As in the case of junctional EB, the absence of kindlin-1 may weaken the anchorage of secretory ameloblasts to the underlying enamel matrix.12 Enamel hypoplasia was once reported in siblings with KEB, but was considered as an independent item.13
Periodontal disease, the main concern in KEB owing to its associated bone and tooth loss,7 was found in most of our patients. Signs of generalized gingival inflammation were observed as early as the age of 2 years, and severe periodontal disease resulted in complete loss of teeth as early as the age of 13 years. In contrast, patient 9, who performed dental follow-ups every 6 months, had marginal gingivitis and inflammation of some interdental papilla; however, periodontal probing was uneventful, and radiography revealed no alveolar bone disease at the age of 15 years.
In patients with KEB, oral SCC with poor prognosis may occur at a young age at sites of chronic inflammation. A pathogenic chain including mucosal fragility, tissue damage, inflammation, and bacterial colonization may build the ground for carcinogenesis.14 DNA repair defects associated with nuclear functions of kindlin-1 and oxidative stress may contribute to early accumulation of oncogenic mutations.15 Activation of fibroblasts and increased activity of transforming growth factor–β and interleukin-6 represent mechanisms that generate a cancer-prone environment in KEB.1
Limitations
This study has limitations that should be mentioned. In particular, the number of patients is low. In addition, this is a retrospective analysis.
Conclusions
The findings of this cohort study suggest that hypoplastic pitted amelogenesis imperfecta is a feature associated with KEB with pathogenic FERMT1 variants and underscore the extent and severity of oral manifestations in KEB. In patients with KEB, oral care is mandatory.
eFigure 1. Plasma Cell Cheilitis in Kindler Epidermolysis Bullosa
eFigure 2. Angular Cheilitis and Squamous Cell Carcinoma in Kindler Epidermolysis Bullosa
eFigure 3. Microstomia in Kindler Epidermolysis Bullosa
Data Sharing Statement
References
- 1.Rognoni E, Widmaier M, Jakobson M, et al. Kindlin-1 controls Wnt and TGF-β availability to regulate cutaneous stem cell proliferation. Nat Med. 2014;20(4):350-359. doi: 10.1038/nm.3490 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Rognoni E, Ruppert R, Fässler R. The kindlin family: functions, signaling properties and implications for human disease. J Cell Sci. 2016;129(1):17-27. doi: 10.1242/jcs.161190 [DOI] [PubMed] [Google Scholar]
- 3.Larjava H, Koivisto L, Heino J, Häkkinen L. Integrins in periodontal disease. Exp Cell Res. 2014;325(2):104-110. doi: 10.1016/j.yexcr.2014.03.010 [DOI] [PubMed] [Google Scholar]
- 4.von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP; STROBE Initiative . The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. J Clin Epidemiol. 2008;61(4):344-349. doi: 10.1016/j.jclinepi.2007.11.008 [DOI] [PubMed] [Google Scholar]
- 5.Bloch-Zupan A, Rey T, Jimenez-Armijo A, et al. ; O-Rare Consortium; ERN Cranio Consortium; International Consortium . Amelogenesis imperfecta: next-generation sequencing sheds light on Witkop’s classification. Front Physiol. 2023;14:1130175. doi: 10.3389/fphys.2023.1130175 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Saleva M, Has C, He Y, Vassileva S, Balabanova M, Miteva L. Natural history of Kindler syndrome and propensity for skin cancer: case report and literature review. J Dtsch Dermatol Ges. 2018;16(3):338-341. doi: 10.1111/ddg.13435 [DOI] [PubMed] [Google Scholar]
- 7.Krämer S, Lucas J, Gamboa F, et al. Clinical practice guidelines: oral health care for children and adults living with epidermolysis bullosa. Spec Care Dentist. 2020;40(suppl 1):3-81. doi: 10.1111/scd.12511 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Poulsen S, Gjørup H, Haubek D, et al. Amelogenesis imperfecta: a systematic literature review of associated dental and oro-facial abnormalities and their impact on patients. Acta Odontol Scand. 2008;66(4):193-199. doi: 10.1080/00016350802192071 [DOI] [PubMed] [Google Scholar]
- 9.Laugel-Haushalter V, Paschaki M, Thibault-Carpentier C, Dembelé D, Dollé P, Bloch-Zupan A. Molars and incisors: show your microarray IDs. BMC Res Notes. 2013;6(1):113. doi: 10.1186/1756-0500-6-113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Rostampour N, Appelt CM, Abid A, Boughner JC. Expression of new genes in vertebrate tooth development and p63 signaling. Dev Dyn. 2019;248(8):744-755. doi: 10.1002/dvdy.26 [DOI] [PubMed] [Google Scholar]
- 11.Sharir A, Marangoni P, Zilionis R, et al. A large pool of actively cycling progenitors orchestrates self-renewal and injury repair of an ectodermal appendage. Nat Cell Biol. 2019;21(9):1102-1112. doi: 10.1038/s41556-019-0378-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Simmer JP, Hu JCC, Hu Y, et al. A genetic model for the secretory stage of dental enamel formation. J Struct Biol. 2021;213(4):107805. doi: 10.1016/j.jsb.2021.107805 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Blanchet I, Tardieu C, Casazza E. Oral care in Kindler syndrome: 7-year follow-up of 2 brothers. J Clin Pediatr Dent. 2021;45(1):41-47. doi: 10.17796/1053-4625-45.1.8 [DOI] [PubMed] [Google Scholar]
- 14.Chacón-Solano E, León C, Díaz F, et al. Fibroblast activation and abnormal extracellular matrix remodelling as common hallmarks in three cancer-prone genodermatoses. Br J Dermatol. 2019;181(3):512-522. doi: 10.1111/bjd.17698 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zapatero-Solana E, García-Giménez JL, Guerrero-Aspizua S, et al. Oxidative stress and mitochondrial dysfunction in Kindler syndrome. Orphanet J Rare Dis. 2014;9:211. doi: 10.1186/s13023-014-0211-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
eFigure 1. Plasma Cell Cheilitis in Kindler Epidermolysis Bullosa
eFigure 2. Angular Cheilitis and Squamous Cell Carcinoma in Kindler Epidermolysis Bullosa
eFigure 3. Microstomia in Kindler Epidermolysis Bullosa
Data Sharing Statement