Summary
Background
The SARS-CoV-2 virus can bind to angiotensin-converting enzyme 2 receptors on host renal cells and may cause acute kidney injury (AKI). The comparative risks of AKI in patients severely ill with COVID-19 and influenza A have not been examined.
Methods
This is a retrospective cohort study including patients with positive PCR results for SARS-CoV-2 or influenza A virus admitted to the intensive care units (ICUs) of 15 public hospitals in Hong Kong between 1st January 2013 and 30th April 2023. Patients who were already on chronic dialysis or had missing values in the confounder model were excluded. Data were retrieved from Hong Kong Hospital Authority's electronic healthcare records. The primary outcome was incident AKI during ICU stay. Secondary outcomes included acute kidney disease (AKD) and hospital mortality. All analyses were examined in multivariable regression adjusting for potential confounders (age, sex, baseline eGFR, PaO2/FiO2 ratio, baseline comorbidities, APACHE IV predicted risk of death, Charlson Comorbidity Index, emergent hospital admission, admission from elderly home, reason for ICU admission, presence of bacterial co-infections, use of medications [including vasopressors, antiviral medications, steroids and nephrotoxic antibiotics], as well as anaemia and leucocytosis). Patients were matched in a 1:1 ratio using a propensity score generated based on the full confounder model. The analyses were repeated using inverse probability weighting and in propensity-score matched cohorts.
Findings
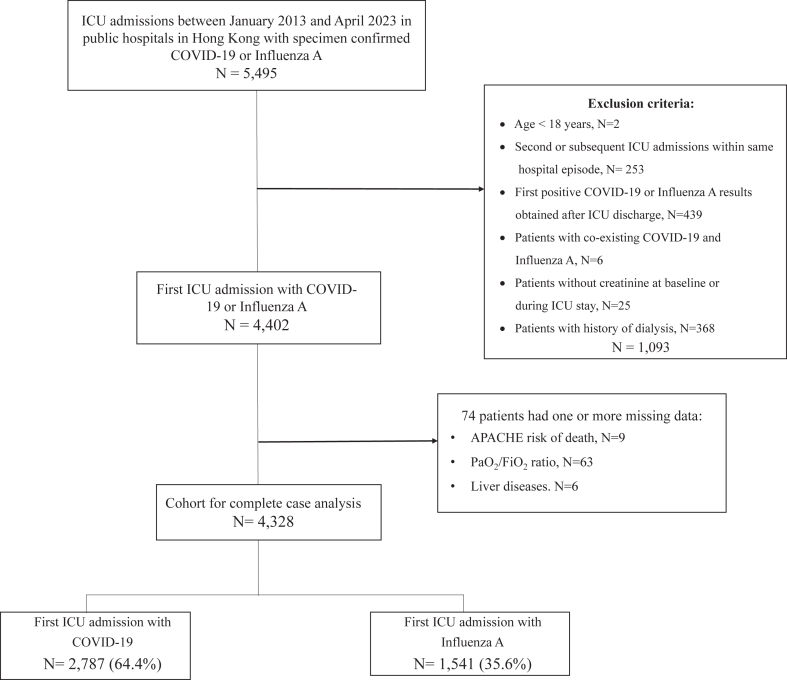
A total of 5495 ICU patients were identified. After excluding 1093 (19.9%) patients who met the exclusion criteria and 74 (1.3%) patients who had one or more missing values in the logistic regression model, a total of 4328 patients were included in the final analysis, with 2787 (64.4%) patients who tested positive for SARS-CoV-2 reverse transcription (RT)-PCR and 1541 (35.6%) patients who tested positive for influenza A virus RT-PCR. The comorbidity burden was greater in patients with COVID-19 (Charlson Comorbidity Index 3 [2–4] vs. 3 [1–4]), but the median APACHE IV predicted risk of death was significantly lower (0.19 [0.08–0.38] vs. 0.25 [0.11–0.52]). A total of 1053 (37.8%) patients with COVID-19 and 828 (53.7%) patients with influenza A developed AKI of any stage during ICU stay. In adjusted analysis, the risk of AKI was significantly lower in patients with COVID-19 compared with influenza A (adjusted odds ratio 0.51, 95% confidence interval 0.42–0.61, P < 0.0001]. The risk of stage 3 AKI and AKD were also significantly lower in patients with COVID-19. These results remained robust in multiple pre-planned sensitivity analyses including inverse probability weighting and propensity score matching.
Interpretation
Our results suggest that the risk of AKI in patients severely ill with COVID-19 was lower than in patients with influenza A. The burden of concurrent organ failure complicating respiratory viral infections, such as the higher disease-attributable risk of AKI associated with influenza, should be clarified.
Funding
An unrestricted philanthropic donation from Mr and Mrs Laurence Tse, The Wai Im Charitable Foundation, Chan Sui Kau Family Benefits and Charitable Foundation, So Ka Wing and Lee Sau Ying Charitable Foundation, Mr & Mrs Tam Wing Fun Edmund Renal Research Fund, the Theme-Based Research Scheme of the Research Grants Council, Hong Kong Special Administrative Region, The Government of the Hong Kong Special Administrative Region; Programme of Enhancing Laboratory Surveillance and Investigation of Emerging Infectious Diseases and Antimicrobial Resistance for the Department of Health of the Hong Kong Special Administrative Region Government; Emergency COVID-19 Project, Major Projects on Public Security, National Key Research and Development Program; Emergency Collaborative Project of Guangzhou Laboratory; the National Key Research and Development Program of China; Sanming Project of Medicine in Shenzhen China; and the High Level-Hospital Program, Health Commission of Guangdong Province, China.
Keywords: SARS-CoV-2, COVID-19, Influenza, Acute kidney injury, Intensive care unit, Organ failure
Research in context.
Evidence before this study
A literature review was conducted on PubMed and MEDLINE using the search terms “influenza”, “COVID-19” and “acute kidney injury (AKI)” on June 09, 2023. After screening 32 articles, there was only one study that examined the effects of COVID-19 and influenza A on AKI. The study which was conducted during the first wave of the COVID-19 pandemic reported that the incidence of AKI was higher in patients with COVID-19 compared with influenza (40.9% vs. 29.4%) and was also more severe. However, whether the risk of AKI persisted during the subsequent waves of the COVID-19 pandemic and in the context of vaccination and anti-viral therapy have not been explored.
Added value of this study
This retrospective cohort study included 4328 patients who were admitted to the intensive care units in public hospitals over the course of 10 years. A total of 2787 patients tested positive for COVID-19, while 1541 tested positive for Influenza A. After adjusting for differences in comorbidities and acute disease severity, patients with COVID-19 had greater than 40% reduction in risk of developing AKI compared with seasonal influenza. Furthermore, the risk of stage 3 AKI and acute kidney disease (AKD) were significantly lower in patients with COVID-19 when compared to patients with influenza A. These data highlight the importance of examining extra-respiratory complications associated with viral respiratory illnesses.
Implications of all the available evidence
Our results suggest that patients with COVID-19 who are critically ill are associated with lower risks of developing AKI and AKD compared with patients with influenza. Whether this under-recognized complication of seasonal influenza could be modified by a higher uptake of influenza vaccination and antiviral therapy should be studied.
Introduction
Influenza and more recently coronavirus disease 2019 (COVID-19) are the two most common respiratory viral infections among intensive care unit (ICU) patients. Patients who become critically ill after respiratory viral infections pose a huge resource burden to the society, particularly if they develop consequential multiple organ failure necessitating intensive or long-term organ support.
Acute kidney injury (AKI) is a common complication in critically ill patients and is a well-recognized sequelae in patients with respiratory infections caused by coronaviruses or seasonal influenza viruses. For example, it has been estimated that as much as 40–50% of patients with Middle East respiratory syndrome coronavirus (MERS-CoV) infection may develop acute renal failure.1 Previous studies using data from the National Inpatient Sample showed that hospitalizations for patients with AKI were $11,016 costlier, and 3.9 days longer than those without AKI.2 Early data from the first wave of the COVID-19 pandemic reported that the incidence of AKI was higher compared with influenza (40.9% vs. 29.4%) and was also more severe.3 Mechanistically, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) can cause AKI via direct virological invasion of renal cells and endothelial cell dysfunction, or via indirect mechanisms such as immune-mediated injuries (e.g., acute interstitial nephritis or glomerulonephritis) or as a part of the systemic manifestations of sepsis.4, 5, 6, 7, 8, 9, 10, 11 Likewise, influenza virus can cause various glomerular diseases, tubulointerstitial pathologies, as well as sepsis in critically ill patients, thereby leading to AKI.12, 13, 14, 15, 16
The objective of this study was to compare the incidence and disease pattern of AKI in critically ill patients with COVID-19 and influenza A. We hypothesized that patients with COVID-19 would have higher rates of AKI than patients with influenza A. We compared the baseline disease severity, incidence of AKI including transition to chronic kidney disease (CKD), and its impact on mortality in patients with COVID-19 and influenza A.
Methods
Study design and population
This was a retrospective cohort study using data from a territory-wide electronic health record system. Patients admitted to the ICU in all 15 public hospitals and 230 ICU beds between 1st January 2013 and 30th April 2023 were reviewed. At the time of the study, all patients with COVID-19 who required admission to the ICU in Hong Kong were managed in public hospitals. This study complied with the Declaration of Helsinki 1975 and its later amendments, and was approved by the Institutional Review Board of the University of Hong Kong/Hospital Authority Hong Kong West Cluster (HKU/HA HKW IRB) with a waiver of signed informed consent (IRB Reference Number: UW 20-572, updated on 11th July 2023). This study adhered to the STROBE reporting guidelines.
All patients who were ≥18 years at the time of ICU admission and had a positive polymerase chain reaction (PCR) result for either SARS-CoV-2 or influenza A virus were included. Exclusion criteria included patients who had hospital-acquired SARS-CoV-2 or influenza after discharge from the ICU, patients with concurrent SARS-CoV-2 and influenza infections, patients without creatinine level at baseline or during ICU stay, and patients already on chronic dialysis. Patients with one or more missing data in the values used in the confounder model were also excluded. For patients with recurrent ICU admissions during the same hospital stay, only the first ICU admission was included in analysis.
Definitions of study outcomes
The primary outcome was incident AKI during ICU stay, defined according to the KDIGO consensus criteria.17 The baseline creatinine level was defined as the most recent creatinine level 7 days before the current hospital admission. AKI was defined if the patient fulfilled the magnitude of increase in creatinine (increase by 50% within 7 days or increase by 0.3 mg/dL within 2 days). The urine output criteria was not used (Table S1).
Secondary outcomes included stage 3 AKI, acute kidney disease (AKD) defined if patients met the criteria of AKI, or had glomerular filtration rate (GFR) < 60 mL/min/1.73 m2, or decrease in GFR by ≥ 35%, or increase in creatinine by >50% within 3 months. Progression of AKI to CKD was defined if GFR remained <60 mL/min/1.73 m2 beyond 3 months after ICU admission (Table S1).18 In patients with pre-existing CKD, if they had worsening of serum creatinine or estimated glomerular filtration rate (eGFR) meeting the AKI, AKD, and CKD definitions of KDIGO, they would also be considered as having a new outcome event.
Other secondary outcomes included need for renal replacement therapy (RRT) during hospital stay, hospital mortality, and 90-day mortality.
All outcome events were recorded until the date of death, or the data cut-off date of 30th June 2023, whichever occurred first. Detailed ICD-9-CM codes for clinical outcomes are listed in Table S2.
Definitions of other variables
Patient's baseline characteristics including age, sex, and comorbidities were collected. Chronic diseases were recorded according to the Acute Physiology and Chronic Health Evaluation (APACHE) IV form,19 which is available for all patients admitted to the ICU. The revised Charlson Comorbidity Index was calculated as a measure of baseline comorbidity.20 The reason for ICU admission were classified into sepsis, respiratory, metabolic, renal and other causes using the disease category on the APACHE IV form. Bacterial co-infection was defined as the identification of positive bacterial culture in specimens from the respiratory tract, including bronchial aspirate, endotracheal tube aspirate, nasal swab, nasopharyngeal aspirate, pleural fluid, and sputum, identified between hospital admission and ICU discharge. Antiviral medications included oseltamivir, zanamivir and peramivir for patients with influenza A, and nirmatrelvir/ritonavir (Paxlovid), remdesivir and molnupiravir for patients with COVID-19. Vasopressor medications included epinephrine, norepinephrine, dobutamine, dopamine, phenylephrine, and vasopressin. Nephrotoxic antibiotics included amikacin, colistin sulphomethate, gentamicin and vancomycin. Anaemia was defined if the first haemoglobin level after hospital admission <12 g/dL in females or <13 g/dL in males. Leucocytosis was defined if the first white cell count after hospital admission >11 × 109/L. eGFR was estimated using the MDRD GFR formula.21
Statistical analysis
Categorical variables were described as frequencies with percentages (%), and continuous variables as mean with standard deviation or median with interquartile range (IQR). Categorical variables were compared with Fisher-exact test or Pearson's chi-squared test, as appropriate. Continuous variables were compared using student's t-test or Mann–Whitney U test. Multivariable logistic regression was used to examine the association between exposure groups and AKI, adjusting for potential confounders. All confounders were selected a priori based on biological plausibility. Three models of confounder control were used: Model 1 included age and sex; Model 2 included age, sex, baseline eGFR and PaO2/FiO2 ratio; and Model 3 additionally included baseline comorbidities including hypertension, congestive heart failure, CKD, malignancies, diabetes mellitus, myocardial infarction or coronary artery disease, and liver diseases, APACHE IV predicted risk of death, Charlson Comorbidity Index, emergent hospital admission, admission from elderly home, reason for ICU admission, presence of bacterial co-infections, use of vasopressor medications, use of antiviral medications, use of steroids, use of nephrotoxic antibiotics, anaemia, and leucocytosis at baseline.
In order to achieve stringent control for confounders, planned primary analyses also included examining the association between exposure groups and AKI using propensity score adjustment in the outcome model, inverse probability weighting (using stabilized weights with truncation, Supplemental Section S1), and 1:1 propensity score matching. The propensity score for the exposure group was constructed using a logistic regression model based on the same set of confounders. Propensity score matching was performed using a 1:1 matching ratio with nearest neighbour and sampling without replacement with a caliper of 0.10. Standardized mean differences of matching variables for the inverse probability weighted and the propensity score matched cohort were examined.
Subgroup analyses
The possibility of effect modification of the exposure-outcome association was examined by introducing interactive terms to the regression model. These included age ≥65 years, sex, baseline eGFR <60 mL/min/1.73 m2, Charlson Comorbidity Index >4, APACHE IV predicted risk of death >0.5, and history of diabetes. The homogeneity of effect estimates across these subgroups were compared.
Sensitivity analyses
Sensitivity analysis was performed in patients without bacterial co-infections using a multivariable logistic regression model adjusting for all the variables in the full model (Model 3). The analysis was repeated using propensity score adjustment, inverse probability weighting, and propensity score matching.
A subcohort analysis was performed with detailed patient physiological data from one of the hospitals, by including age, baseline eGFR, use of vasopressors, use of steroids, use of nephrotoxic antibiotics, mechanical ventilation, and daily average fluid balance as potential confounders in a multivariable logistic regression model for the primary outcome.
Exploratory analyses
The time to dialysis for the two groups were examined in a time-to-event analysis, accounting for the competing risks of death or lost to follow up. Kaplan–Meier curves were constructed and compared using the log-rank test.
The association between exposure groups and change in eGFR was examined with a linear mixed effects model with random effects to account for repeated measurements within individuals, and adjusting for all confounders in the primary regression model. Serial eGFR for patients from hospital admission to the worst level during their stay in ICU were included.
In order to examine the effect of secular trends on the incidence of AKI in the ICU, the rate of AKI in patients with influenza A was examined for significant trends across years.
Data management were done with Stata MP, version 16.1. All analyses were performed with two-tailed tests and a P-value <0.05 was considered statistically significant.
Role of the funding source
The funder of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report. PYN, AKN, AI, and DYY had access to dataset and had final responsibility for the decision to submit for publication.
Results
Study population and patient characteristics
A total of 5495 patients were admitted to the ICUs in public hospitals in Hong Kong between 1st January 2013 and 30th April 2023. After excluding 1093 (19.9%) patients who met the exclusion criteria and 74 (1.3%) patients who had one or more missing values in the logistic regression model, a total of 4328 patients were included in the final analysis (Fig. 1). There were 2787 (64.4%) patients who had positive SARS-CoV-2 RT-PCR results and 1541 (35.6%) patients who had positive influenza A RT-PCR results. Microbiological investigation for bacterial co-infections was performed in 4086 (94.4%) patients. Patients with COVID-19 admitted to the ICU were significantly older (median 69.5 [IQR 61.1–76.9] vs. 63.6 [54.3–74.6] years, P < 0.0001) with a higher median Charlson Comorbidity Index (3 [2–4] vs. 3 [1–4], P < 0.0001). Fewer patients with COVID-19 compared with influenza A were admitted to the hospital emergently (2393 [85.9%] vs. 1449 [94.0%], P < 0.0001). The median APACHE IV predicted risk of death was significantly lower in patients with COVID-19 (0.19 [0.08–0.38] vs. 0.25 [0.11–0.52], P < 0.0001), but there was a greater use of vasopressors compared with patients with influenza A (1332 [47.8%] vs. 614 [39.8%], P < 0.0001). The number of patients admitted to the ICU for metabolic or renal causes was significantly higher in the COVID-19 group compared to the influenza A group (metabolic cause: 462 [16.6%] vs. 71 [4.6%]; and renal cause: 107 [3.8%] vs. 21 [1.4%], both P < 0.0001), while the number of patients admitted to the ICU for sepsis was significantly lower in the COVID-19 group (663 [23.8%] vs. 739 [48.0%], P < 0.0001). ICU admission for respiratory causes was similar in the 2 groups. The baseline characteristics are shown in Table 1. Patient characteristics and standardized mean differences after inverse probability weighting and 1:1 propensity score matching are presented in Tables S3 and S4, respectively.
Fig. 1.
Study flow. A total of 5495 patients were admitted to the ICUs during the study period. After excluding 1093 (19.9%) patients who met the exclusion criteria and 74 (1.3%) patients who had missing data, a total of 4328 patients were included in the final analysis. Abbreviations: APACHE–Acute Physiology and Chronic Health Evaluation; COVID-19–coronavirus disease 2019; ICU, intensive care unit.
Table 1.
Baseline characteristics of study population.
| Characteristics | COVID-19 |
Influenza A |
P-value |
|---|---|---|---|
| N = 2787 | N = 1541 | ||
| Age, years | 69.5 61.1–76.9 |
63.6 54.3–74.6 |
<0.0001 |
| Sex, female | 1027 (36.8%) | 648 (42.1%) | 0.0008 |
| Sex, male | 1760 (63.2%) | 893 (57.9%) | 0.0008 |
| Emergent hospital admission | 2393 (85.9%) | 1449 (94.0%) | <0.0001 |
| Admission from elderly home | 63 (2.3%) | 27 (1.8%) | 0.26 |
| APACHE-IV predicted risk of death | 0.19 0.08–0.38 |
0.25 0.11–0.52 |
<0.0001 |
| Bacterial co-infections during ICU stay | 808 (29.0%) | 665 (43.2%) | <0.0001 |
| Use of vasopressors | 1332 (47.8%) | 614 (39.8%) | <0.0001 |
| Use of antiviral drugsa | 1511 (54.2%) | 1475 (95.7%) | <0.0001 |
| Use of steroids | 1780 (63.9%) | 634 (41.1%) | <0.0001 |
| Use of nephrotoxic antibiotics b | 270 (9.7%) | 233 (15.1%) | <0.0001 |
| Reason for ICU admission | |||
| Metabolic | 462 (16.6%) | 71 (4.6%) | <0.0001 |
| Renal | 107 (3.8%) | 21 (1.4%) | <0.0001 |
| Respiratory | 865 (31.0%) | 506 (32.8%) | 0.22 |
| Sepsis | 663 (23.8%) | 739 (48.0%) | <0.0001 |
| Othersc | 690 (24.8%) | 204 (13.2%) | <0.0001 |
| Comorbidities | |||
| Charlson Comorbidity Index | 3 (2–4) | 3 (1–4) | <0.0001 |
| Chronic kidney diseases | 182 (6.5%) | 53 (3.4%) | <0.0001 |
| Congestive heart failure | 167 (6.0%) | 153 (9.9%) | <0.0001 |
| Diabetes mellitus | 909 (32.6%) | 312 (20.2%) | <0.0001 |
| Hypertension | 1352 (48.5%) | 413 (26.8%) | <0.0001 |
| Malignancies | 310 (11.1%) | 149 (9.7%) | 0.14 |
| Myocardial infarction or Coronary artery diseases | 299 (10.7%) | 184 (11.9%) | 0.23 |
| Laboratory results | |||
| Baseline eGFR, mL/min/1.73 m2 | 73.5 54.1–93.9 |
73.9 51.5–93.8 |
0.67 |
| PaO2/FiO2 ratio | 224.8 130.0–365.0 |
138.2 76.9–252.6 |
<0.0001 |
| Liver diseasesd | 545 (19.6%) | 414 (26.9%) | <0.0001 |
| Anaemia e | 1487 (53.4%) | 771 (50.0%) | 0.036 |
| Leucocytosisf | 1043 (37.4%) | 580 (37.6%) | 0.89 |
Abbreviations: APACHE-IV, acute physiology and chronic health evaluation IV; COVID-19–coronavirus disease 2019; eGFR, estimated glomerular filtration rate; ICU, intensive care unit.
Data for continuous variables are presented as frequency (percentage) or median (interquartile range (IQR)) unless specified.
Between groups comparisons are performed using Chi-squared test for categorical variables, and Mann–Whitney U test for continuous variables. The significance level was set at P < 0.05.
Antiviral drugs included oseltamivir, zanamivir and peramivir for patients with influenza A; nirmatrelvir/ritonavir (Paxlovid), remdesivir and molnupiravir for patients with COVID-19.
Nephrotoxic antibiotics included amikacin, colistin sulphomethate, gentamicin and vancomycin.
Other reasons for ICU admission included cardiovascular, gastrointestinal, gynaecological, haematological, musculoskeletal, neurological and trauma.
Liver diseases were defined using 2 times upper normal limit of total bilirubin or alanine transaminase, or international normalized ratio ≥1.5.
Anaemia was defined if the first haemoglobin level after hospital admission <12 g/dL in female patients or <13 g/dL in male patients.
Leucocytosis was defined if the first white cell count after hospital admission >11 × 109/L.
Acute kidney injury
A total of 1053 (37.8%) patients with COVID-19 and 828 (53.7%) patients with influenza A developed AKI of any stage during their ICU stay (P < 0.0001). After adjusting for potential confounders, the risk of AKI was significantly lower in patients with COVID-19 (adjusted odds ratio [aOR] 0.51, 95% confidence interval [CI] 0.42–0.61, P < 0.0001]. Detailed risk estimates and the full confounder model are presented in Table S5. Results were consistent across different regression models, in an outcomes model adjusting for propensity score, using inverse probability weighting, and in the 1:1 propensity score matched cohort (aOR 0.48 to 0.61, P < 0.0001 for all). Detailed results of the primary analysis using all confounder models are presented in Table 2.
Table 2.
Primary outcome—acute kidney injury.
| Acute kidney injury | Events/Patients (%) |
P-valuea | Odds Ratio | 95% confidence interval | P-valueb | ||
|---|---|---|---|---|---|---|---|
| COVID-19 | Influenza A | ||||||
| Logistic regression | 1053/2787 (37.8%) | 828/1541 (53.7%) | <0.0001 | Model 1 | 0.48 | 0.42–0.55 | <0.0001 |
| Model 2 | 0.60 | 0.52–0.69 | <0.0001 | ||||
| Model 3 | 0.51 | 0.42–0.61 | <0.0001 | ||||
| Propensity score adjustment | Model 1 | 0.48 | 0.42–0.55 | <0.0001 | |||
| Model 2 | 0.61 | 0.54–0.70 | <0.0001 | ||||
| Model 3 | 0.57 | 0.48–0.68 | <0.0001 | ||||
| Inverse probability weighting | 1061/2819 (37.6%) | 842/1561 (53.9%) | <0.0001 | Model 1 | 0.52 | 0.45–0.58 | <0.0001 |
| 1115/2922 (38.2%) | 917/1767 (51.9%) | <0.0001 | Model 2 | 0.57 | 0.51–0.64 | <0.0001 | |
| 1174/3116 (37.7%) | 634/1227 (51.7%) | <0.0001 | Model 3 | 0.57 | 0.49–0.65 | <0.0001 | |
| Propensity score matching | 619/1540 (40.2%) | 827/1540 (53.7%) | <0.0001 | Model 1 | 0.58 | 0.50–0.67 | <0.0001 |
| 585/1530 (38.2%) | 824/1530 (53.9%) | <0.0001 | Model 2 | 0.53 | 0.46–0.61 | <0.0001 | |
| 356/858 (41.5%) | 472/858 (55.0%) | <0.0001 | Model 3 | 0.58 | 0.48–0.70 | <0.0001 | |
Model 1 was adjusted for age and sex.
Model 2 was adjusted for age, sex, baseline eGFR and PaO2/FiO2 ratio.
Model 3 was adjusted for the covariates listed in Table 1.
Abbreviations: COVID-19–coronavirus disease 2019.
Chi-squared test was used for the unadjusted analyses. The significance level was set at P < 0.05.
Multivariable logistical regression was used in the logistic regression model and the model adjusting for propensity score. Univariable logistic regression was used in the inverse probability weighted cohort and the propensity score matched cohort. The significance level was set at P < 0.05.
Secondary outcomes
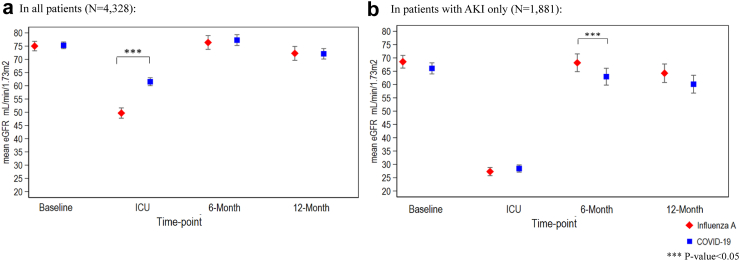
The mean level of eGFR at 4 time-points, including baseline, worst reading during ICU stay, 6 months and 12 months from ICU admission, in patients with COVID-19 and patients with influenza A are presented in a time-series plot (Fig. 2). The mean eGFR at 6 months for patients with COVID-19 and influenza A was 77.3 ± 42.2 mL/min/1.73 m2 and 76.4 ± 38.2 mL/min/1.73 m2, respectively (P = 0.60). The mean eGFR at 12 months was 72.1 ± 34.5 mL/min/1.73 m2 for patients with COVID-19 and 72.3 ± 33.6 mL/min/1.73 m2 for patients with influenza A (P = 0.92).
Fig. 2.
eGFR levels from baseline to 12 months after ICU admission. The mean level and 95% confidence interval of eGFR at 4 time-points, including baseline, worst during ICU stay, 6 months and 12 months from ICU admission, in patients with COVID-19 (blue dots) and patients with influenza A (red dots) are presented for all patients (Panel A, N = 4328) and patients with AKI only (Panel B, N = 1881). Abbreviations: AKI, acute kidney injury; COVID-19, coronavirus disease 2019; eGFR, estimated glomerular filtration rate; ICU, intensive care unit.
A total of 432 (15.5%) patients with COVID-19 and 375 (24.3%) patients with influenza A developed stage 3 AKI (P < 0.0001). In adjusted analysis, COVID-19 was associated with lower odds of stage 3 AKI compared with influenza A (aOR 0.57, 95% CI 0.45–0.72, P < 0.0001). There were also fewer patients with COVID-19 who required RRT compared with patients with influenza (316/2787 [11.3%] vs. 320/1541 [20.8%], aOR 0.52, 95% CI 0.40–0.67, P < 0.0001). Patients with COVID-19 had significantly lower risks of AKD compared to patients with influenza A (1844/2787 [66.2%] vs. 1177/1541 [76.4%], aOR 0.66, 95% CI 0.53–0.82, P = 0.0002).
Among the 1881 patients who had AKI, the risk of developing CKD in patients with COVID-19 was similar to patients with influenza A (252/1053 [23.9%] vs. 190/828 [23.0%], aOR 0.80, 95% CI 0.58–1.09, P = 0.16). Additionally, a total of 112 (6.0%) patients developed end-stage kidney disease, defined as eGFR <15 mL/min/1.73 m2 or the need for RRT beyond 3 months, including 64 (6.1%) patients with COVID-19 and 48 (5.8%) patients with influenza.
Hospital mortality and 90-day mortality were higher in patients with COVID-19 compared with influenza A only in adjusted logistic regression (aOR 1.29, 95% CI 1.01–1.65, P = 0.038, and aOR 1.37, 95% CI 1.08–1.72, P = 0.0080, respectively). However, the association was not significant after propensity score adjustment, or in the inverse probability weighted or propensity score matched analyses. Detailed results of secondary outcomes are presented in Table 3.
Table 3.
Secondary outcomes.
| Outcomes | Events/Patients (%) |
P-valuea | Odds Ratio | 95% confidence interval | P-valueb | |
|---|---|---|---|---|---|---|
| COVID-19 | Influenza A | |||||
| Stage 3 AKI | ||||||
| Logistic regression | 432/2787 (15.5%) | 375/1541 (24.3%) | <0.0001 | 0.57 | 0.45–0.72 | <0.0001 |
| Propensity score adjustment | 432/2787 (15.5%) | 375/1541 (24.3%) | <0.0001 | 0.70 | 0.57–0.87 | 0.0013 |
| Inverse probability weighting | 494/3116 (15.9%) | 256/1227 (20.9%) | <0.0001 | 0.71 | 0.60–0.85 | 0.0001 |
| Propensity score matching | 145/858 (16.9%) | 194/858 (22.6%) | 0.0030 | 0.70 | 0.55–0.88 | 0.0031 |
| Hospital mortality | ||||||
| Logistic regression | 562/2787 (20.2%) | 349/1541 (22.7%) | 0.055 | 1.29 | 1.01–1.65 | 0.038 |
| Propensity score adjustment | 562/2787 (20.2%) | 349/1541 (22.7%) | 0.055 | 1.16 | 0.94–1.43 | 0.17 |
| Inverse probability weighting | 671/3116 (21.5%) | 251/1227 (20.5%) | 0.43 | 1.07 | 0.91–1.26 | 0.43 |
| Propensity score matching | 205/858 (23.9%) | 200/858 (23.3%) | 0.78 | 1.03 | 0.83–1.29 | 0.78 |
| 90-day mortality | ||||||
| Logistic regression | 631/2787 (22.6%) | 385/1541 (25.0%) | 0.082 | 1.37 | 1.08–1.72 | 0.0080 |
| Propensity score adjustment | 631/2787 (22.6%) | 385/1541 (25.0%) | 0.082 | 1.21 | 0.99–1.48 | 0.066 |
| Inverse probability weighting | 756/3116 (24.3%) | 284/1227 (23.2%) | 0.44 | 1.06 | 0.91–1.24 | 0.44 |
| Propensity score matching | 232/858 (27.0%) | 218/858 (25.4%) | 0.44 | 1.09 | 0.88–1.35 | 0.44 |
| AKD | ||||||
| Logistic regression | 1844/2787 (66.2%) | 1177/1541 (76.4%) | <0.0001 | 0.66 | 0.53–0.82 | 0.0002 |
| Propensity score adjustment | 1844/2787 (66.2%) | 1177/1541 (76.4%) | <0.0001 | 0.69 | 0.57–0.84 | 0.0002 |
| Inverse probability weighting | 2079/3116 (66.7%) | 920/1227 (75.0%) | <0.0001 | 0.67 | 0.58–0.78 | <0.0001 |
| Propensity score matching | 601/858 (70.1%) | 670/858 (78.1%) | 0.0001 | 0.66 | 0.53–0.82 | 0.0002 |
| CKD | ||||||
| Logistic regression | 252/1053 (23.9%) | 190/828 (23.0%) | 0.62 | 0.80 | 0.58–1.09 | 0.16 |
| Propensity score adjustment | 252/1053 (23.9%) | 190/828 (23.0%) | 0.62 | 0.91 | 0.68–1.21 | 0.50 |
| Inverse probability weighting | 268/1174 (22.8%) | 164/634 (25.9%) | 0.15 | 0.85 | 0.68–1.06 | 0.15 |
| Propensity score matching | 85/356 (23.9%) | 106/472 (22.5%) | 0.63 | 1.08 | 0.78–1.50 | 0.63 |
| Renal replacement therapy | ||||||
| Logistic regression | 316/2787 (11.3%) | 320/1541 (20.8%) | <0.0001 | 0.52 | 0.40–0.67 | <0.0001 |
| Propensity score adjustment | 316/2787 (11.3%) | 320/1541 (20.8%) | <0.0001 | 0.65 | 0.51–0.82 | 0.0004 |
| Inverse probability weighting | 356/3116 (11.4%) | 194/1227 (15.8%) | <0.0001 | 0.69 | 0.57–0.83 | 0.0001 |
| Propensity score matching | 104/858 (12.1%) | 154/858 (18.0%) | 0.0007 | 0.63 | 0.48–0.83 | 0.0008 |
Only analyses using Model 3 were included in this table.
Abbreviations: AKD, acute kidney disease; AKI, acute kidney injury; CKD, chronic kidney disease; COVID-19–coronavirus disease 2019; KDIGO, Kidney Disease: Improving Global Outcomes.
Chi-squared test was used for the unadjusted analyses. The significance level was set at P < 0.05.
Multivariable logistical regression was used in the logistic regression model and the model adjusting for propensity score. Univariable logistic regression was used in the inverse probability weighted cohort and the propensity score matched cohort. The significance level was set at P < 0.05.
Patients with pre-existing CKD
Within the study cohort, there were 1391 (32.1%) patients who had pre-existing CKD or a baseline eGFR below 60 mL/min/1.73 m2 prior to ICU admission, including 868 (31.1%) patients with COVID-19 and 523 (33.9%) patients with influenza. A total of 465 (53.6%) patients with COVID-19 and 343 (65.6%) patients with influenza developed new AKI. Patients with COVID-19 had a lower risk of new AKI compared to patients with influenza (aOR 0.61, 95% CI 0.45–0.83, P = 0.0020). In addition, there were 123 (26.5%) patients with COVID-19 and 88 (25.7%) patients with influenza who developed worsening CKD after AKI.
In addition, a total of 566 (65.2%) patients with COVID-19 and 393 (75.1%) patients with influenza developed new AKD. Patients with COVID-19 had a lower risk of new AKD compared to patients with influenza (aOR 0.61, 95% CI 0.43–0.86, P = 0.0042).
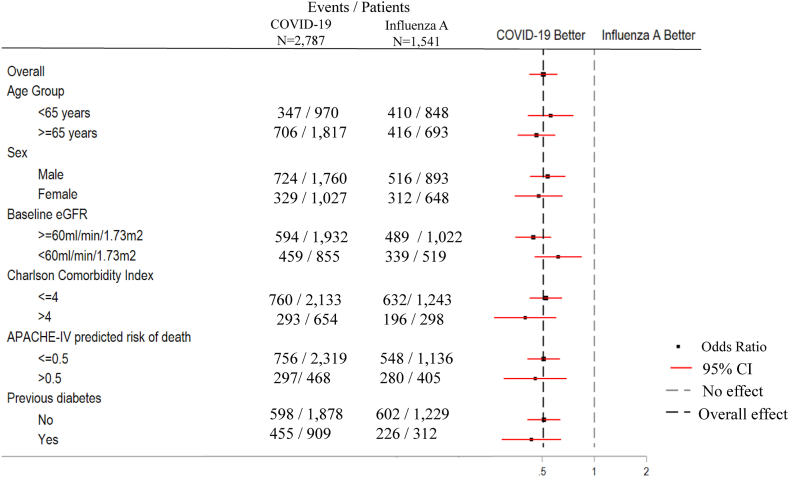
Subgroup analyses
There was no significant effect modification of the lower risks of AKI in patients with COVID-19 by factors including age, sex, baseline eGFR, Charlson Comorbidity Index, APACHE IV predicted risk of death, and history of diabetes. Details of subgroup analyses are presented in Table 4 and Fig. 3.
Table 4.
Subgroup analyses for the outcome of acute kidney injury.
| Subgroups | Events/patients |
Adjusted odds ratio | 95% confidence interval | P-value for interaction | |
|---|---|---|---|---|---|
| COVID-19 N = 2787 | Influenza A N = 1541 |
||||
| Age group | 0.058 | ||||
| Age <65 | 347/970 | 410/848 | 0.56 | 0.41–0.76 | |
| Age ≥65 | 706/1817 | 418/693 | 0.46 | 0.36–0.59 | |
| Sex | 0.80 | ||||
| Male | 724/1760 | 516/893 | 0.51 | 0.42–0.61 | |
| Female | 329/1027 | 312/648 | 0.51 | 0.42–0.61 | |
| Baseline creatinine clearance | 0.18 | ||||
| eGFR ≥60 mL/min/1.73 m2 |
594/1932 | 489/1022 | 0.44 | 0.35–0.56 | |
| eGFR <60 mL/min/1.73 m2 |
459/855 | 339/519 | 0.62 | 0.45–0.85 | |
| Charlson comorbidity index | 0.54 | ||||
| ≤4 | 760/2133 | 632/1243 | 0.52 | 0.42–0.65 | |
| >4 | 293/654 | 196/298 | 0.40 | 0.26–0.60 | |
| APACHE-IV predicted risk of death | 0.48 | ||||
| ≤0.5 | 756/2319 | 548/1136 | 0.51 | 0.41–0.63 | |
| >0.5 | 297/468 | 280/405 | 0.46 | 0.30–0.69 | |
| Previous diabetes | 0.19 | ||||
| No | 598/1878 | 602/1229 | 0.51 | 0.41–0.64 | |
| Yes | 455/909 | 226/312 | 0.43 | 0.29–0.64 | |
This table refers to Model 3, which includes the full confounder model.
Abbreviations: APACHE-IV, Acute physiology and chronic health evaluation IV; COVID-19, coronavirus disease 2019; eGFR, estimated glomerular filtration rate.
Fig. 3.
Forest plot of subgroup analyses. The subgroup analyses was adjusted using the full confounder model (Model 3). There were no significant effect modification of the lower risks of AKI in patients with COVID-19 by factors including age, sex, baseline eGFR, Charlson Comorbidity Index, APACHE IV predicted risk of death, and history of diabetes. Abbreviations: APACHE-IV, Acute physiology and chronic health evaluation IV; CI, confidence interval; COVID-19–coronavirus disease 2019; eGFR, estimated glomerular filtration rate.
Sensitivity analyses
A total of 2855 (66.0%) patients did not have bacterial co-infections, including 1979 patients with COVID-19 and 876 patients with influenza A. In adjusted analysis performed in this subgroup, patients with COVID-19 had a lower risk of AKI compared with patients with influenza A (aOR 0.54, 95% CI 0.43–0.69, P < 0.0001). The results were consistent with using propensity score adjustment, inverse probability weighting and propensity score matching with all P-values <0.0001 (Table S7).
A subcohort analysis was conducted using data from 312 patients from a single hospital, including 205 (65.7%) patients with COVID-19 and 107 (34.3%) patients with influenza. Mechanical ventilation and daily fluid management were recorded for all 312 patients during their ICU stay. Within the subcohort, 100 (48.8%) patients with COVID-19 and 56 (52.3%) patients with influenza developed AKI. Patients with COVID-19 had a lower risk of AKI compared to patients with influenza (aOR 0.42, 95% CI 0.22–0.79, P = 0.0076) (Table S8).
Exploratory analyses
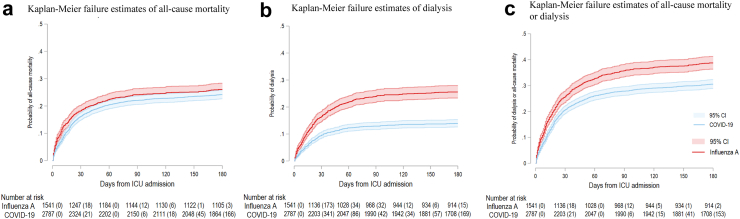
The time-to-event curves for the outcome of dialysis and all-cause mortality from ICU admission to 180 days are shown in Fig. 4. COVID-19 was associated with lower risks of the individual and composite outcomes (P for all log-rank tests <0.05).
Fig. 4.
Time-to-dialysis curves of patients with COVID-19 and influenza A. Time-to-event curves for the outcomes of all-cause mortality (Panel a), dialysis (Panel b), and combined outcome (Panel c) from ICU admission to 180 days. COVID-19 (blue line) compared with influenza A (red line) was associated with lower risks of the individual and composite outcomes (P for all log-rank tests <0.05). The number of censoring cases was presented in brackets. Abbreviation: COVID-19, coronavirus disease 2019; ICU, intensive care unit.
The rate of decline in eGFR was significantly slower in patients with COVID-19 compared with influenza A (adjusted mean difference in daily eGFR decline: 0.37, 95% CI 0.30–0.45 mL/min/1.73 m2, P < 0.0001; Table S9).
The rate of AKI in patients with influenza A during the entire study period ranged from 50.0% to 57.6% and did not differ significantly across years (P for trend = 0.29, Figure S1).
Other exploratory analyses are detailed in the Supplement.
Discussion
In our territory-wide analyses including all patients who have severe respiratory viral infections, the data suggested that SARS-CoV-2 is associated with more than 40% lower risk of AKI compared with seasonal influenza amongst critically ill patients requiring ICU care. Physicians need to be aware of the significant burden of common respiratory viral infections on adverse renal outcomes, including the resource implications of patients requiring dialysis. The higher disease-attributable risk of AKI associated with influenza calls for more research into mitigating this significant complication.
Our data showed that patients with COVID-19 had a 49% lower risk of AKI than those with influenza, and similarly 43% lower risk of stage 3 AKI and 48% less requirement for acute dialysis. These findings are in contrast to the early results from a previous study by Birkelo et al., which reported a higher risk of AKI and more severe renal impairment with SARS-CoV-2 infection.3 One major difference between our study and the study by Birkelo et al. is the time period of the patients with COVID-19. The prior study included primarily patients with COVID-19 during the earlier wave of the pandemic up to May 2020 when vaccines and antivirals for COVID-19 were not readily available, but the current cohort included patients up to April 2023. Importantly, most of our patients (84.5%) were infected during or after the fifth wave of COVID-19 in 2022.22 Globally, there have been major changes in treatment and vaccination strategies since June 2020. First, the use of dexamethasone, which reduced mortality related to severe COVID-19 disease, was only used routinely after the preliminary results of the RECOVERY Study were released in July 2020.23 In our study, all patients admitted to the ICU with COVID-19 received dexamethasone treatment. Second, antivirals for COVID-19 were not used routinely before May 2020. The results of the first widely-used antiviral, remdesivir, was published in May 2020.24 The use of oral antivirals nirmatrelvir/ritonavir and molnupiravir were approved in late 2021. Third, COVID-19 vaccination became available since late 2020. Hence, the patients in the study by Birkelo et al. received vastly different management compared to our cohort.
Apart from differences in treatment and vaccination, the ancestral SARS-CoV-2 strain in early 2020 was also quite different from the Omicron variant which replaced all previous lineages. While the exact strain of SARS-CoV-2 was not examined in all patient samples and therefore not considered in this analysis, the Omicron variant was probably predominant in our cohort—it was the locally circulating strain in 2022 during which 84.5% patients of our cohort were infected. Studies have found that patients infected with the Omicron variant exhibited milder illness when compared to those with prior lineages.25,26 We have previously demonstrated that mice and hamsters infected with the Omicron variant had much reduced disease severity when compared to those infected with the ancestral strain.27,28 More data are needed to examine whether the risks of systemic disease and AKI are heterogeneous amongst different strains of the SARS-CoV-2 and influenza viruses.
The true incidence of kidney injury associated with respiratory viral infection is difficult to ascertain, as most patients with mild presentation remain untested and undiagnosed in the community. Even among hospitalised patients, the baseline comorbidity burden and severity of acute illness render studying disease associations susceptible to the effects of confounding. By restricting our cohort to patients with severe infections necessitating admission to the ICU and adjusting for differences in severity of acute illness and the patients’ presentation, we showed a consistently lower incidence of adverse renal outcomes with COVID-19 across all pre-specified sensitivity analyses, suggesting that this may be a virus-specific characteristic. In addition, we were able to adjust for the effects of daily fluid management. Subgroup analyses revealed that the lower risks were observed across strata of baseline comorbidities and acute illness severity. Notably, the differences persisted beyond the initial disease course, and the risk of AKD up to 3 months following ICU admission remained lower in patients with COVID-19. These data should be considered during resource allocation to dialysis facilities, which are often overloaded in healthcare systems, but overlooked as part of the response to outbreaks of viral respiratory infections.29, 30, 31, 32
It was not possible to include the vaccination status in the main analysis, due to lack of accurate data where influenza or COVID-19 vaccination could have been received in private primary care settings and not recorded in the public hospital electronic patient records. Based on population statistics, the local COVID-19 vaccination rate was >70% for adults aged between 20 and 59 years and >60% for adults aged between 60 and 69 years old, in contrast to about 10–20% for persons aged 50–64 years and <50% for persons aged 65 years or above for annual influenza vaccination.33 It remains to be examined whether the higher vaccination uptake during the COVID-19 pandemic moderated a lower risk of systemic organ injury.
One important limitation of this study was its retrospective nature, with biases and heterogeneity in patient characteristics and management. It was possible that the triage criteria for ICU admission, and hence disease severity of patients, evolved during the course of a pandemic. Nevertheless, the use of quantitative outcomes such as creatinine-based definitions of AKI, and the rigorous attempts to adjust for confounding render our findings reliable and representative of the real-world situation. Our results remained robust in multiple pre-planned sensitivity analyses including inverse probability weighting and propensity score matching. Second, due to the unavailability of detailed data, the urine output criteria was not included in the definition of AKI, and potential confounders such as fluid balance and the initiation of mechanical ventilation before AKI were not accounted for. Third, the inclusion of patients from 2013 to 2023 poses risks of secular changes in ICU care. However, we showed that amongst patients with influenza, the proportion of AKI in the earlier pre-COVID-19 period were not significantly different form the post-COVID-19 period. Lastly, renal biopsy was not routinely performed for critically ill patients with AKI related to influenza or SARS-CoV-2 infection, and hence conclusions about pathophysiological mechanisms could not be drawn.
In conclusion, the risk of AKI in critically ill patients with COVID-19 was significantly lower than patients with influenza A. Whether this under-recognized complication of seasonal influenza could be modified by a higher uptake of seasonal influenza vaccination and antiviral therapy should be studied.
Contributors
PYN, AI, AKN, SWS, DYY and KKT were responsible for the conception and design of the study. PYN, AKN, AI, and DYY have access to and verified the underlying study data. PYN, AI and AKN analyzed the data. PYN, AI, AKN, DYY and KKT interpreted the data. PYN, AI and AKN drafted the manuscript. All authors revised and approved the final manuscript, and are accountable for the accuracy and integrity of the work.
Data sharing statement
Due to concerns regarding patient privacy, data sharing is restricted solely to academic research purposes, and only aggregated study data will be available to researchers. Upon publication, researchers may request access to the aggregated data by submitting a reasonable request with proposed analysis plans to the corresponding author via email. Data requestors will be required to sign a data access agreement.
Declaration of interests
All authors declare no competing interests.
Acknowledgements
PYN is supported by an unrestricted philanthropic donation from Mr and Mrs Laurence Tse. DYY received research donations from the Wai Im Charitable Foundation, Chan Sui Kau Family Benefits and Charitable Foundation, So Ka Wing and Lee Sau Ying Charitable Foundation, and Mr & Mrs Tam Wing Fun Edmund Renal Research Fund. JFC and KKT received funding from the Theme-Based Research Scheme of the Research Grants Council [T11-709/21-N], Hong Kong Special Administrative Region, The Government of the Hong Kong Special Administrative Region, Programme of Enhancing Laboratory Surveillance and Investigation of Emerging Infectious Diseases and Antimicrobial Resistance for the Department of Health of the Hong Kong Special Administrative Region Government; Emergency COVID-19 Project, Major Projects on Public Security, National Key Research and Development Program (2021YFC0866100); Emergency Collaborative Project of Guangzhou Laboratory (EKPG22-01); the National Key Research and Development Program of China (projects 2021YFC0866100 and 2023YFC3041600); Sanming Project of Medicine in Shenzhen China (SZSM201911014); and the High Level-Hospital Program, Health Commission of Guangdong Province, China.
Footnotes
Supplementary data related to this article can be found at https://doi.org/10.1016/j.eclinm.2024.102535.
Contributor Information
Kelvin Kai-Wang To, Email: kelvinto@hku.hk.
Desmond Yat-Hin Yap, Email: desmondy@hku.hk.
Appendix A. Supplementary data
References
- 1.Chan J.F., Lau S.K., To K.K., Cheng V.C., Woo P.C., Yuen K.Y. Middle East respiratory syndrome coronavirus: another zoonotic betacoronavirus causing SARS-like disease. Clin Microbiol Rev. 2015;28:465–522. doi: 10.1128/CMR.00102-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Silver S.A., Long J., Zheng Y., Chertow G.M. Cost of acute kidney injury in hospitalized patients. J Hosp Med. 2017;12:70–76. doi: 10.12788/jhm.2683. [DOI] [PubMed] [Google Scholar]
- 3.Birkelo B.C., Parr S.K., Perkins A.M., et al. Comparison of COVID-19 versus influenza on the incidence, features, and recovery from acute kidney injury in hospitalized United States Veterans. Kidney Int. 2021;100:894–905. doi: 10.1016/j.kint.2021.05.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wichmann D., Sperhake J.P., Lütgehetmann M., et al. Autopsy findings and venous thromboembolism in patients with COVID-19: a prospective cohort study. Ann Intern Med. 2020;173:268–277. doi: 10.7326/M20-2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Schurink B., Roos E., Radonic T., et al. Viral presence and immunopathology in patients with lethal COVID-19: a prospective autopsy cohort study. Lancet Microbe. 2020;1:e290–e299. doi: 10.1016/S2666-5247(20)30144-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ahmadian E., Hosseiniyan Khatibi S.M., Razi Soofiyani S., et al. Covid-19 and kidney injury: pathophysiology and molecular mechanisms. Rev Med Virol. 2021;31 doi: 10.1002/rmv.2176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Paranjpe I., Jayaraman P., Su C.Y., et al. Proteomic characterization of acute kidney injury in patients hospitalized with SARS-CoV2 infection. Commun Med. 2023;3:81. doi: 10.1038/s43856-023-00307-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Santoriello D., Khairallah P., Bomback A.S., et al. Postmortem kidney pathology findings in patients with COVID-19. J Am Soc Nephrol. 2020;31:2158–2167. doi: 10.1681/ASN.2020050744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Su H., Yang M., Wan C., et al. Renal histopathological analysis of 26 postmortem findings of patients with COVID-19 in China. Kidney Int. 2020;98:219–227. doi: 10.1016/j.kint.2020.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ferlicot S., Jamme M., Gaillard F., et al. The spectrum of kidney biopsies in hospitalized patients with COVID-19, acute kidney injury, and/or proteinuria. Nephrol Dial Transplant. 2021 doi: 10.1093/ndt/gfab042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Legrand M., Bell S., Forni L., et al. Pathophysiology of COVID-19-associated acute kidney injury. Nat Rev Nephrol. 2021;17:751–764. doi: 10.1038/s41581-021-00452-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sood M.M., Rigatto C., Zarychanski R., et al. Acute kidney injury in critically ill patients infected with 2009 pandemic influenza A(H1N1): report from a Canadian Province. Am J Kidney Dis. 2010;55:848–855. doi: 10.1053/j.ajkd.2010.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jouvet P., Hutchison J., Pinto R., et al. Critical illness in children with influenza A/pH1N1 2009 infection in Canada. Pediatr Crit Care Med. 2010;11:603–609. doi: 10.1097/PCC.0b013e3181d9c80b. [DOI] [PubMed] [Google Scholar]
- 14.Keckler S.J., Laituri C.A., Ostlie D.J., St Peter S.D. A review of venovenous and venoarterial extracorporeal membrane oxygenation in neonates and children. Eur J Pediatr Surg. 2010;20:1–4. doi: 10.1055/s-0029-1231053. [DOI] [PubMed] [Google Scholar]
- 15.Carmona F., Carlotti A.P., Ramalho L.N., Costa R.S., Ramalho F.S. Evidence of renal infection in fatal cases of 2009 pandemic influenza A (H1N1) Am J Clin Pathol. 2011;136:416–423. doi: 10.1309/AJCP1Y6LLHWSKYHW. [DOI] [PubMed] [Google Scholar]
- 16.Watanabe T. Renal complications of seasonal and pandemic influenza A virus infections. Eur J Pediatr. 2013;172:15–22. doi: 10.1007/s00431-012-1854-x. [DOI] [PubMed] [Google Scholar]
- 17.Lameire N.H., Levin A., Kellum J.A., et al. Harmonizing acute and chronic kidney disease definition and classification: report of a kidney disease: improving global outcomes (KDIGO) consensus conference. Kidney Int. 2021;100:516–526. doi: 10.1016/j.kint.2021.06.028. [DOI] [PubMed] [Google Scholar]
- 18.Bellomo R., Ronco C., Kellum J.A., Mehta R.L., Palevsky P. Acute renal failure–definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care. 2004;8:R204–R212. doi: 10.1186/cc2872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zimmerman J.E., Kramer A.A., McNair D.S., Malila F.M. Acute Physiology and Chronic Health Evaluation (APACHE) IV: hospital mortality assessment for today's critically ill patients. Crit Care Med. 2006;34:1297–1310. doi: 10.1097/01.CCM.0000215112.84523.F0. [DOI] [PubMed] [Google Scholar]
- 20.Quan H., Li B., Couris C.M., et al. Updating and validating the Charlson comorbidity index and score for risk adjustment in hospital discharge abstracts using data from 6 countries. Am J Epidemiol. 2011;173:676–682. doi: 10.1093/aje/kwq433. [DOI] [PubMed] [Google Scholar]
- 21.Levey A.S., Coresh J., Greene T., et al. Using standardized serum creatinine values in the modification of diet in renal disease study equation for estimating glomerular filtration rate. Ann Intern Med. 2006;145:247–254. doi: 10.7326/0003-4819-145-4-200608150-00004. [DOI] [PubMed] [Google Scholar]
- 22.Chen L.L., Abdullah S.M.U., Chan W.M., et al. Contribution of low population immunity to the severe Omicron BA.2 outbreak in Hong Kong. Nat Commun. 2022;13:3618. doi: 10.1038/s41467-022-31395-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Horby P., Lim W.S., Emberson J.R., et al. Dexamethasone in hospitalized patients with covid-19. N Engl J Med. 2021;384:693–704. doi: 10.1056/NEJMoa2021436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Beigel J.H., Tomashek K.M., Dodd L.E., et al. Remdesivir for the treatment of covid-19–final report. N Engl J Med. 2020;383:1813–1826. doi: 10.1056/NEJMoa2007764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wolter N., Jassat W., Walaza S., et al. Early assessment of the clinical severity of the SARS-CoV-2 omicron variant in South Africa: a data linkage study. Lancet. 2022;399:437–446. doi: 10.1016/S0140-6736(22)00017-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Menni C., Valdes A.M., Polidori L., et al. Symptom prevalence, duration, and risk of hospital admission in individuals infected with SARS-CoV-2 during periods of omicron and delta variant dominance: a prospective observational study from the ZOE COVID Study. Lancet. 2022;399:1618–1624. doi: 10.1016/S0140-6736(22)00327-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Shuai H., Chan J.F., Hu B., et al. Attenuated replication and pathogenicity of SARS-CoV-2 B.1.1.529 Omicron. Nature. 2022;603:693–699. doi: 10.1038/s41586-022-04442-5. [DOI] [PubMed] [Google Scholar]
- 28.Yuan S., Ye Z.W., Liang R., et al. Pathogenicity, transmissibility, and fitness of SARS-CoV-2 Omicron in Syrian hamsters. Science. 2022;377:428–433. doi: 10.1126/science.abn8939. [DOI] [PubMed] [Google Scholar]
- 29.Liyanage T., Ninomiya T., Jha V., et al. Worldwide access to treatment for end-stage kidney disease: a systematic review. Lancet. 2015;385:1975–1982. doi: 10.1016/S0140-6736(14)61601-9. [DOI] [PubMed] [Google Scholar]
- 30.Bello A.K., Levin A., Lunney M., et al. Status of care for end stage kidney disease in countries and regions worldwide: international cross sectional survey. BMJ. 2019;367:l5873. doi: 10.1136/bmj.l5873. [DOI] [PubMed] [Google Scholar]
- 31.Li P.K., Chan G.C., Chen J., et al. Tackling dialysis burden around the world: a global challenge. Kidney Dis. 2021;7:167–175. doi: 10.1159/000515541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Htay H., Bello A.K., Levin A., et al. Hemodialysis use and practice patterns: an international survey study. Am J Kidney Dis. 2021;77:326–335.e1. doi: 10.1053/j.ajkd.2020.05.030. [DOI] [PubMed] [Google Scholar]
- 33.Centre for Health Protection of the HKSAR government Statistics on vaccination programmes in the past 3 years. https://www.chp.gov.hk/en/features/102226.html# accessed 19th August, 2023.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.