Abstract
Scedosporium apiospermum species complex are widely distributed fungi that can be found in a variety of polluted environments, including soil, sewage, and decaying vegetation. Those opportunistic pathogens with strong potential of invasion commonly affect immunosuppressed populations However, few cases of scedosporiosis are reported in immunocompetent individuals, who might be misdiagnosed, leading to a high mortality rate. Here, we reported an immunocompetent case of systemtic infection involved in lung, brain and spine, caused by S. apiospermum species complex (S. apiospermum and S. boydii). The patient was an elderly male with persistent fever and systemtic infection after near-drowning. In the two tertiary hospitals he visited, definite diagnosis was extremely difficult. After being admitted to our hospital, he was misdiagnosed as tuberculosis infection, before diagnosis of S. apiospermum species complex infection by the metagenomic next-generation sequencing. His symptoms were alleviated after voriconazole treatment. In the present case, the details associated with its course were reported and published studies on Scedosporium spp. infection were also reviewed, for a better understanding of this disease and reducing the misdiagnosis rate.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12879-023-08279-9.
Keywords: Scedosporium apiospermum species complex, Near-drowning, Systemtic infection, Metagenomic next-generation sequencing
Introduction
Scedosporium spp. are widely distributed in soils of temperate climates, rather than tropical climates [1]. Scedosporium boydii (Pseudallescheria boydii) was once considered to be the sexual state of Scedosporium apiospermum. However, due to little difference in phylogenetic and clinical significance, both S. apiospermum and S. boydii can be described as “Scedosporium apiospermum species complex” [2].
At least 5 species of Scedosporium (S. apiospermum, S. boydii, S. aurantiacum, S. dehoogii, and S. minutisporum) can cause human infections [3], while S. apiospermum and S. boydii are fonud to be the two most common pathogens [4, 5]. They can cause systemic infections in immunosuppressed individuals, such as organ transplant recipients, and patients with hematological malignancies or receiving long-term glucocorticoid therapy [5]. In immunocompetent individuals, certain conditions, such as near-drowning or injuries may let Scedosporium cause therapy-refractory and life-threatening infections in the central nervous system (CNS) or lung, including respiratory symptoms, superficial infections, and severe invasive localized or disseminated mycoses [6, 7].
Scedosporium spp. are resistant to 5-flucytosine and amphotericin B, as well as to the first generation triazole drugs, fluconazole and itraconazole [5]. This species also shows a reduced susceptibility to echinocandins (particularly caspofungin and anidulafungin) and the triazole drug, isavuconazole. According to a global guideline for the diagnosis and management of rare mould infections, voriconazole represents the first-line treatment of Scedosporium infections [8].
The clinical manifestations of Scedosporium infection are complex, resulting in misdiagnosis. Here, we present the case of an immunocompetent patient with a systemic infection, which was found to be caused by S. apiospermum and S. boydii using metagenomic next-generation sequencing (mNGS). We also summarized literature reviews on Scedosporium infection in immunocompetent individuals.
Case report
The patient was a 70-year-old man who born and bred in a suburb of Wuhan, China, and both agriculture and industry exist in his surroundings. He had undergone lumbar disc surgery, rheumatoid arthritis, hypertension, and lacunar infarction. On April 26, 2021, he accidently fell into a pond (standing water) in a field (Figure S1). Aspiration and choking occurred spontaneously, and then fever, cough and expectoration started to appear. The highest body temperature reached 39.6 ℃, and the patient suffered from paroxysmal cough with a small amount of white sticky sputum, accompanied by shortness of breath, dizziness and transient loss of consciousness, without any symptoms of chest pain, hemoptysis, abdominal pain, hematemesis, or limb dysfunction. He was quickly admitted to a local hospital and was diagnosed with pulmonary infection. As Klebsiella pneumoniae and Candida albicans were successively cultured from deep sputum and the patient kept fever, multiple antibiotics were adopted and switched according to his clinical manifestation. With a drug combination of imipenem/cilastatin (500 mg/500 mg q.8.h. i.v.d), amikacin (400 mg q.12.h. i.v.d), and voriconazole (200 mg q.12.h. i.v.d) was adopted and lasted for 1 week, patient’s clinical manifestation was partially improved. He was discharged from the hospital on May 18, and did not adhere to the medication regimen since then. However, ten days after discharge, the patient developed fever again, with a body temperature of up to 39 ℃, accompanied by feeling of tightness in the chest and shortness of breath, without obvious cough or expectoration. Then, he visited Wuhan's Tongji Hospital where anti-infectives and oral antipyretic drugs were given in an emergency observation ward. Although the body temperature returned to normal, recurrent fever persisted, which led to a second hospitalization on June 1, 2021. Examination results showed increases in the values of various infection indicators, multiple nodules in both lungs from a computed tomography (CT) image (Figs. 1 and 2). Patient was empirically treated with cefoperazone/sulbactam (1,500 mg/1,500 mg q.12.h. i.v.d) and micafungin (100 mg q.d. i.v.d). Three days later, Aspergillus fumigatus was detected in bronchoalveolar lavage (BAL) fluid using mNGS (number of sequences: 15), and culture of BAL fluid by blood plate and China blue agar plat yielded K. pneumoniae again. However, the patient's blood culture (aerobic and anaerobic, up to 2 weeks), the (1,3)—β -D dextran test (G test), and the galactomannan test (GM test) were all negative. Therefore, micafungin was replaced by voriconazole (200 mg q.12.h. i.v.d), and cefoperazone and sulbactam was replaced by piperacillin/tazobactam (3,000 mg/375 mg q.6.h. i.v.d). Besides, amikacin (280 mg q.8.h. i.v.d) was added into the treatment prescription. However, the patient's symptoms was not significantly improved. On July 3, culture of BAL fluid was performed again and only yielded C. tropicalis and C. glabrata. The antifungal regimen was changed to posaconazole (400 mg q.12.h. p.o) combined with micafungin (100 mg q.d. i.v.d). Given his continuous high fever, piperacillin/tazobactam was replaced by imipenem/cilastatin (500 mg/500 mg q.8.h. i.v.d) combined with teicoplanin (400 mg q.d. i.v.d). After treatment, the symptoms of dizziness and shortness of breath disappeared, and the frequency of cough and expectoration was reduced.
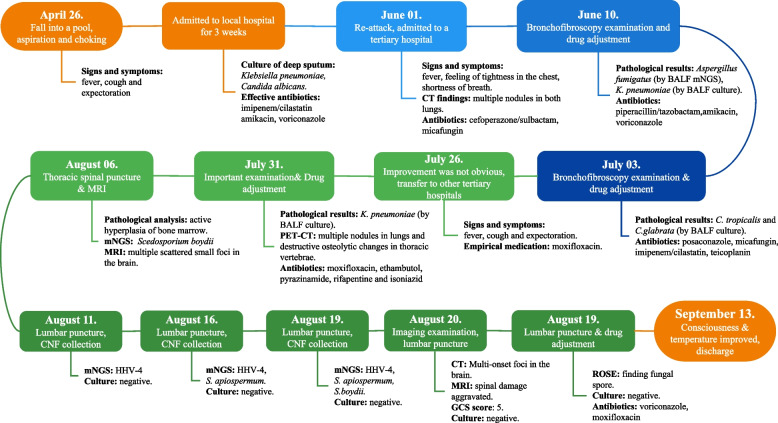
Fig. 1.
Timeline of patient journey. CT: Computed Tomography; mNGS: metagenomic next-generation sequencing; BALF: bronchoalveolar lavage fluid; PET: positron emission tomography; MRI: magnetic resonance imaging; GCS: Glasgow Coma Scale; ROSE: rapid on-site evaluation
Fig. 2.
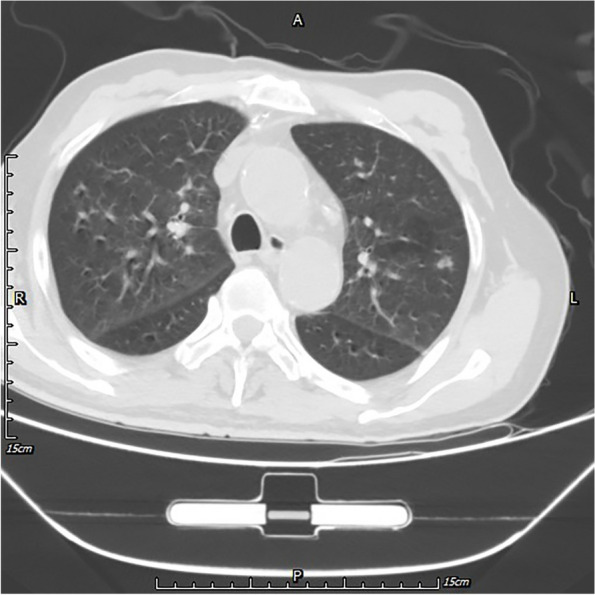
A lung CT (Computed Tomography) image showing multiple nodules in both lungs
Unfortunately, the patient still experienced recurrent episodes of fever, and had a significantly reduced ability to perform daily activities with fatigue leaving him bedridden. Due to worsening symptoms of fever, cough and expectoration lasting for 3 days, the patient was eventually transferred to our hospital on July 26, 2021. Antipyretic drugs were given to temporarily control the body temperature which had been up to 39 °C (sustained fever), but the symptom of cough with a small amount of white sticky sputum remained. Meanwhile, symptoms of chest pain, night sweats, hemoptysis, palpitation, wheezing, abdominal pain, and diarrhea were not observed. BAL fluid cultured yielded K. pneumoniae again, but by Sabouraud dextrose agar (SAD) plat yielded negative. Thus, moxifloxacin (400 mg q.d. i.v.d) was empirically applied to treat infections before diagnosis of the specific pathogen. On the 3rd day of admission, positron emission tomography (PET)/CT imaging (Fig. 3) revealed multiple nodules and streaks in both lungs and destructive osteolytic changes between T6 and T7, implying infectious lesions. Based on the consultation results from the Tuberculosis Department, the empirical anti-tuberculosis treatment was introduced, consisting of ethambutol, pyrazinamide, rifapentine and isoniazid. Seven days after admission, a thoracic spinal puncture at T6-T7 was performed with tissues sampled for examination. The pathological analysis of bone tissues (Fig. 4) showed excessive bone marrow tissues in trabecular bones, indicating active hyperplasia of bone marrow, and S. boydii (number of sequence: 1) was detected by mNGS. As the number of sequences was very low, S. boydii was not considered as a pathogen, but as a pollutant. On the 10th day of admission, contrast-enhanced magnetic resonance imaging (MRI) was carried out for brain examination and result indicated multiple scattered small foci of signal abnormality with enhancement in the brain, multiple lacunar infarcts and ischemic foci (Fig. 5), according to which the patient was initially diagnosed with infectious disease, tuberculous meningitis in particular. This hypothesis was further supported by persistent fever and systemtic dysfunction. For further confirmation of Mycobacterium tuberculosis infection, a series of tests were conducted, including the purified protein derivative (PPD) test, contrast-enhanced spinal MRI, and genetic testing, but all results turned out to be negative. Subsequently, lumbar puncture was performed on the 14th day of admission and cerebrospinal fluid (CSF) was then collected for examination. CSF examination showed a significant increases of total cell count (360 × 106/L), white blood cell counts (189 × 106/L), and trace protein (2.16 g/L), while concentrations of chlorine and glucose were at normal levels; Human herpes virus infection indicated by mNGS (Table 1); culture yielded negative. As a result, the patient was maintained with moxifloxacin treatment and quadruple therapy for tuberculosis. Twenty days after admission, lumbar puncture and CSF examination were repeated. It’s cytologic and biochemical results showed no big change than before, however, only 1 sequence of S. apiospermum was detected at this time by mNGS. CSF culture still yielded negative. Subsequently, the patient had his third lumbar puncture and CSF examination on the 23rd day of admission, with the reports showed the total cell count was nearly 11 times increased compared to base line, but the WBC count and trace protein decreased. mNGS porformed again and Human herpes virus-4 (number of sequences: 5), S. boydii (number of sequences: 2) and S. apiospermum (number of sequence: 1) were determined. On the 24th day of admission, the patient was found unresponsive to speech, no eye-opening and no speech during examination, but responses to painful stimuli with a Glasgow Coma Scale (GCS) score of 5. From a new cranial CT scan, multiple low-density lesions, mild hydrocephalus, and multiple lacunar infarcts in the brain were observed. Furthermore, contrast-enhanced MRI of the thoracic vertebrae demonstrated abnormal signals unevenly enhanced at T6-T7 accompanied by slight patchy enhancement of the swelling soft tissues in the vicinity, and hyperintense signals at the intervertebral disc between T9 and T10 (Fig. 5). Then, lumbar puncture and CSF examinations were carried out for the fourth time, and it’s result showed a decreased number of total cell count (621 × 106/L) but an increased number of WBC count (594 × 106/L); the biochemical results and number of pathogen sequence obtained using mNGS Microbiologic rapid on-site evaluation of CSF revealed a fungal spore, but culture still yielded negative.
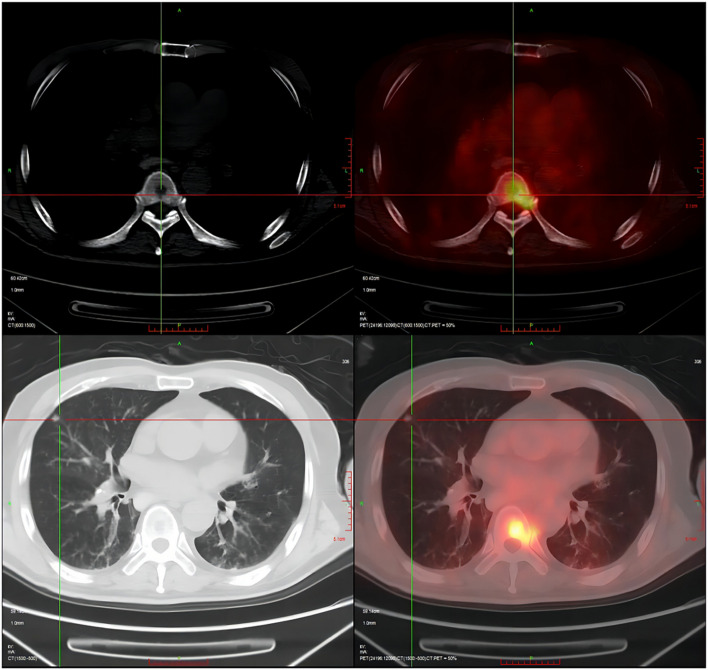
Fig. 3.
PET/CT (Positron emission tomography computed tomography) imaging revealed multiple nodules and streaks in both lungs and destructive osteolytic changes between T6 and T7, implying infectious lesions. Large bullae and calcification nodules were found in the upper lobe of the left lung
Fig. 4.
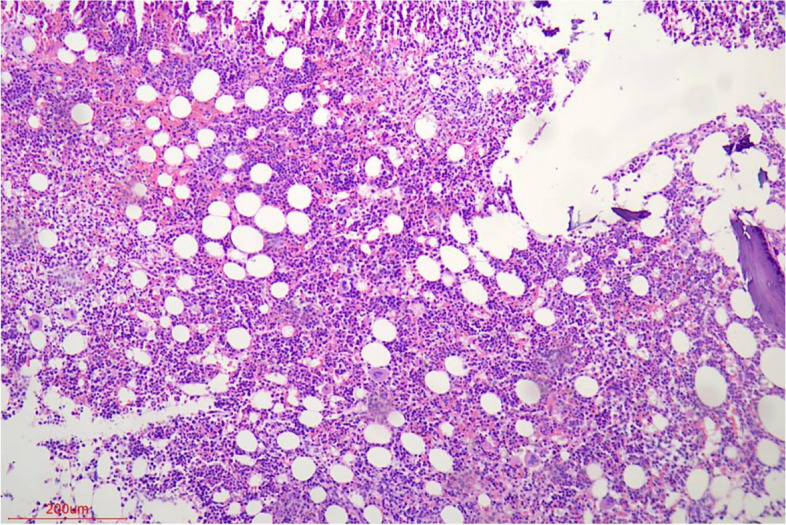
The pathological analysis of bone tissues showed excessive bone marrow tissues in trabecular bones and implied active hyperplasia of bone marrow
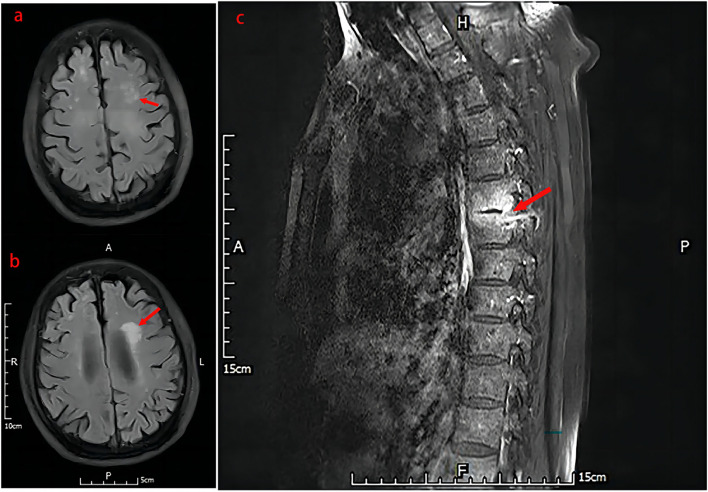
Fig. 5.
Contrast-enhanced MRI: The left temporal lobe (a) and lateral paraventricular (b) demonstrated abnormal signal and enhancement, considering infectious lesions. The thoracic vertebrae (c) demonstrated abnormal signals unevenly enhanced at T6-T7 accompanied by slight patchy enhancement of the swelling soft tissues in the vicinity
Table 1.
Results of patient’s CSF examinations in admission
| Date | Total cells count (× 106/L) | WBC count (× 106/L) | Chlorine (mmol/L) | Glucose (mmol/L) | Trace protein (g/L) | Adenosine deaminase (U/L) | Lactate dehydrogenase (U/L) | Number of sequence |
|---|---|---|---|---|---|---|---|---|
| 11/8 | 360 | 189 | 108.91 | 1.48 | 2.16 | - | - | HHV-4: 437 |
| 16/8 | 360 | 189 | 120.55 | 2.25 | 1.82 | 3.3 | 81 | S. apiospermum: 1 |
| 19/8 | 4100 | 110 | 124.54 | 3.54 | 1.36 | 1.8 | 57 | HHV-4: 5; S. apiospermum: 1; S.boydii: 2 |
| 20/8 | 621 | 594 | 114.06 | 2.05 | 1.68 | 2.1 | 55 | - |
| 23/8 | 3900 | 300 | 125.54 | 2.98 | 1.76 | 2.1 | 79 | Negative |
| 27/8 | 117 | 45 | 127.67 | 3.72 | 0.97 | 2.0 | 51 | Negative |
| 13/9 | 22 | 22 | 117.95 | 7.46 | 1.06 | 1.4 | 33 | Negative |
Character of 7 CSF samples were clear and colorless and Tuberculosis related examinations were always negative
S. apiospermum Scedosporium apiospermum, S.boydii Scedosporium boydii, HHV Human herpes virus
Due to the history of near-drowning, it was suspected that this case of systemtic infection (lung, brain and spine) was caused by S. apiospermum species complex, and antifungal therapy with voriconazole (200 mg q.12.h. i.v.d) was thereafter included to the existing regimen. One week after voriconazole treatment, the body temperature returned to normal, but the overall cognitive ability was still poor. Though the patient was able to remember his own name, he could not perform simple calculations and had no memory of being admitted to the hospital. With the treatment continued, the patient's state of consciousness was gradually improved, as evidenced by the ability to perform addition and subtraction within 10 and recognize some of his families after half a month of treatment. The patient's cognitive ability almost returned to normal 3 weeks later when the patient could recognize his families, remember his home address, perform multiplications and get out of bed. After discharge, voriconazole was changed to oral administration and the entire treatment of scedosporiosis continued for 6 months. Follow-up at 8 months after discharge, CNF mNGS detected no microorganisms. The patient had sequelae of slow response and no other special discomfort.
Review and discussion
The incidence rate of scedosporiosis has been increasing in recent years. As an emerging fungal pathogen, Scedosporium spp. is receiving an increasing attention. We searched PubMed to find articles on cases with Scedosporium (including “Pseudallescheria/Scedosporium complex”, except “Lomentospora prolificans”) deep infection in immunocompetent individuals, which were published between 1982 and 2022 (Table 2).
Table 2.
Case reports with Scedosporium in immunocompetent individuals between 1982 and 2022
| No | Reference | Patient (Age/Sex) | Country | Initial event | Risk factors | Signs and Symptoms | Period after the initial event | Affected parts | Identification of Scedosporium | Method of identification | Antifungal treatment | Treatment Duration | Outcome |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Hung LH et al., 1993 [9] | 32/M | USA | Trauma | None | Pain and swelling in the left knee | 6 years | Knee and proximal tibia | S. boydii | Culture | KETO | Not mentioned | Improved |
| 2 | Rüchel R et al., 1995 [10] | 21/M | Germany | Near-drowning | None | Fever, drowsiness, spastic paralysis | c. 1 week | Brain, heart | S. apiospermum | Culture and Microscopy | Flucytosine + FLU | 26 days | Death |
| 3 | Khurshid A et al., 1999 [11] | 61/F | USA | None | Bullous emphysema | Weight loss, night sweats, cough, fever, and hemoptysis | Not applicable | Lung,heart, liver, spleen, kidney, pancreas, and brain | S. boydii | Culture | AmB | Unknown | Death |
| 4 | Poza G et al., 2000 [12] | 24/M | Spain | Surgical operation | None | Headache, back pain | c. 1 month | CNS | S. boydii | Culture | VCZ | 1 year | Improved |
| 5 | Tirado-Miranda R et al., 2001 [13] | 32/M | Spain | Traffic accident | None | Fever, swelling and pain in the knee | 53 days | Right knee | S. aurantiacum | Culture | 1)AmB 2)ITRA | Not mentioned | Improved |
| 6 | Kiraz N et al., 2001 [14] | 24/F | Turkey | None | None | Enlarged cervical lymph nodes | Not applicable | Lymph nodes | S. apiospermum | Culture and Microscopy | ITRA | 1 year | Improved |
| 7 | Levine NB et al., 2002 [15] | 52/M | USA | None | None | Thoracolumbar spinal pain, hemoptysis | Not applicable | Spinal column | S. apiospermum | Culture | ITRA | Not mentioned | Death |
| 8 | Farina C et al., 2002 [16] | 23/M | Italy | Traffic accident | None | Purulent secretions | 3 days | Skull | S. apiospermum | Culture | AmB | 21 days | Improved |
| 9 | Chakraborty A et al., 2005 [17] | 1.5/M | UK | Near-drowning | None | Low-grade fever, unconsciousness, grand mal seizures | 3 months | Brain | S. apiospermum | Culture | 1)ITRA 2)AmB 3)VCZ | c. 18 months | Improved |
| 10 | Kooijman CM et al., 2007 [18] | 36/M | The Netherlands | Trauma | None | Fistula and abscess | c. 11 weeks | Femur | S. aurantiacum | Culture | VCZ | 8 weeks | Improved |
| 11 | Leechawengwongs M et al., 2007 [19] | 21/M | Thailand | Traffic accident, near-drowning | None | Headache, unconsciousness | 14 days | Brain | S. apiospermum | Culture | 1)AmB + CASPO 2)VCZ | 23 months | Improved |
| 12 | Stripeli F et al., 2009 [20] | 10/F | Greece | Trauma | None | Knee swelling and pain | c. 2 months | Left knee | S. apiospermum | Culture and Sequencing | 1)AmB 2)VCZ | 6 moths | Improved |
| 13 | Al-Jehani H et al., 2010 [21] | 33/M | Canada | Extra-Corporeal Membrane Oxygenation | Neutropenia | Fever, decreased movement, partial seizure | c. 14 days | Brain | S. apiospermum | Culture | VCZ | 1 week | Death |
| 14 | Hell M et al., 2011 [22] | 16/M | Austria | Trauma | None | Soft tissue healing defect, persisting fistula | c. 3 weeks | Bone, muscle | S. apiospermum | Culture | VCZ | c. 6 months | Improved |
| 15 | Cumbo-Nacheli G et al., 2012 [23] | 62/F | USA | None | Mycobacterium avium complex infection history | Fever, dyspnea, cough, and worsening pulmonary nodules | Not applicable | Lung | S. boydii | Pathological examination | VCZ | Not mentioned | Improved |
| 16 | Angelini A, Drago G et al., 2013 [24] | 27/F | Italy | Suffered tsunami | None | Pain in the knee | 2 years | Left knee | S. apiospermum | Culture and Sequencing | 1)VCZ 2)POSA + TERB | > 1 year | Improved |
| 17 | Wilson HL et al., 2013 [25] | 69/M | Australia | None | Silicosis, COPD | Headache, fever, lethargy, nausea and vomiting | Not applicable | Brain | S. apiospermum | Culture, Microscopy and Sequencing | 1)ABLC 2)VCZ 3)CASPO | c. 2 months | Death |
| 18 | Cruysmans C et al., 2015 [26] | 7/M | UK | Trauma | None | low-grade fever, lethargy, weakness of lower limb | 5 moths | Endorachis | S. apiospermum | Culture | VCZ | 1 year | Improved |
| 19 | Rahman FU et al., 2016 [27] | 40/M | Pakistan | None | Pulmonary TB history | Cough, hemopty | Not applicable | Lung | S. apiospermum | Culture and Microscopy | VCZ | 4 moths | Improved |
| 20 | Dinh A et al., 2018 [28] | 57/M | France | None | None | Iterative fractures | Not applicable | Tibial pseudarthrosis | S. apiospermum | Culture and MALDI-TOF | VCZ | 6 moths | Improved |
| 21 | Tan SYL et al., 2020 [29] | 39/M | China | Percutaneous driveline tugged | Left ventricular assist devices equipping | Fever, local pustular lesion | c. 2 months | Blood stream | S. apiospermum | Culture and Sequencing | Not able to take | Not applicable | Death |
| 22 | Jabr R et al., 2020 [30] | 72/M | USA | Peripherally inserted central catheter placement | Pulmonary arterial hypertension | Fever, intermittent hemoptysis, worsening shortness of breath | c. 1 month | Blood stream | S. apiospermum | Culture | 1)VCZ 2)ABLC 3)TERB | 7 months | Improved |
| 23 | Liu W et al., 2020 [31] | 44/M | China | None | None | Hemoptysis | Not applicable | Lung | S. apiospermum | Culture and Microscopy | VCZ | c. 11 months | Improved |
| 24 | Mir WAY et al., 2021 [32] | 83/F | USA | None | Chronic atrial fibrillation, COPD | Shortness of breath, cough with blood-tinged sputum, fatigue | Not applicable | Lung | S. apiospermum | Culture | VCZ | 6 moths | Improved |
| 25 | Ghasemian R et al., 2021 [33] | 67/F | Iran | Near-drowning | None | Fever, respiratory distress | 7 days | Lung | S. aurantiacum | Culture and Sequencing | 1)VCZ 2)ABLC | 6 days | Death |
| 26 | Song Y et al., 2022 [34] | 56/M | China | Inhalation of Biogas | None | Nausea, vomiting, haemoptysis, fever | 10 days | Lung | S. apiospermum | Culture and MALDI-TOF | 1)VCZ 2)ABLC | 230 days | Improved |
| 27 | Shi XW et al., 2022 [35] | 60/M | China | None | None | Lumbosacral pain, stooped back, restricted walking | Not applicable | Lumbar vertebra | S. apiospermum | Culture | VCZ | 6 moths | Improved |
| 28 | This study | 70/M | China | Near-drowning | None | Fever, chest tightness, shortness of breath, dizziness | c. 1 month | Lung, brain and spine | S. apiospermum, S. boydii | mNGS | VCZ | 6 months | Improved |
S. apiospermum: Scedosporium apiospermum, S. aurantiacum: Scedosporium aurantiacum, S.boydii: Scedosporium boydii
M Male, F Female, ABLC Amphotericin B (Lipid Complex), AmB Amphotericin B deoxycholate, CASPO Caspofungin, FLU Fluconazole, ITRA Itraconazole, KETO Ketoconazole, POSA Posaconazole, TERB Terbinafine, VCZ Voriconazole
Among the 28 published studies, there were 21 males and 7 females infected by Scedosporium apiospermum species complex. Age of those patients ranged from 1.5 to 83 years old. Besides, 15 patients (53.6%) suffered trauma or near-drowing, and 3 patients (10.7%) received invasive medical treatment, while any initial events of 10 patients (35.7%) were not reported. The interval of 18 cases from the initial event to onset ranged from 3 days to 6 years.
The most common infection sites were bone, muscle, and joint (11 cases, 39.3%) followed by CNS (include brain and endorachis, 9 cases, 32.1%) and lung (8 cases, 28.6%). Most importantly, 4 out of 9 cases with CNS infection had a history of near-drowing. Fever was the most common systemic symptom (13 cases, 46.4%), which was often associated with infection dissemination. Clinical manifestation of focal infections mainly included local pain, swelling, and dysfuncion, while fever was found in few cases.
S. apiospermum was found to be the causative pathogen in most of patients (21 cases, 75%), follow by S. boydii (5 cases, 17.9%) and S. aurantiacum (3 cases, 10.7%). Among 27 patients receiving anti-fungal drugs, voriconazole treatment was performed on 20 cases (71.4%) and most of them (17 out of 20, 85%) had improved outcome, while more than half of patients (4 out of 7, 57.1%) without voriconazole treatment had poor prognosis.
Among 18 cured cases, treatment duration ranged from 21 days to 23 months. The length of the treatment duration was related to infection sites. Patients with CNS infection need the longest treatment duration (428 days ± 174 days), followed by infections of cardiovascular and lymph nodes (313 days ± 73 days), pulmonary infection (215 days ± 77 days), and infections of bone, muscle, and joint (166 days ± 102 days). The clinical outcome of disseminated or CNS infection is dismal. Previous studies have shown that the mortality rate can reach up to 65%-100%, once Scedosporium disseminates systematically or invades the brain [36].
Culture has always been gold standard of fungal infection diagnosis, and microscopy and molecular biology methods such as sequencing or Matrix-Assisted Laser Desorption/ Ionization Time of Flight Mass Spectrometry (MALDI-TOF–MS) are often adopted after successful cultivation. However, due to the low load of fungi in the CNS, culture is insensitive diagnostic tool. In our case, blood and CSF samples were used to isolate pathogens many times. However, we cannot successfully isolate any fungai. Fortunately, using mNGS, we successfully detected Scedosporium from bone marrowand CSF specimens, which was diagnosed as pathogen.
NGS is an emerging microbiological sequencing diagnostic approach which has advantages of culture-independent, short turnaround time, and high efficiency in cataloging and recognizing pathogens [37]. In this case, although the number of reads detected was low, mNGS results of samples from multiple sites were positive, according to which doctors made a preliminary clinical diagnosis of scedosporiosis. Fortunately, the patient had a good response to the subsequent treatment with voriconazole, which also confirmed the correctness of the clinical diagnosis. However, our case report has limitations, including lack of classical evidence of fungal culture, drug susceptibility testing, and precise species identification. Future study should focus on howto improve the accuracy and specificity of mNGS in fungal pathogen detection to provide more information on fungal drug resistance.
Conclusion
Collectively, scedosporiosis is a rare and challenging illness to diagnose. The pathogens should be confirmed as soon as feasible for a patient who has risk factors, such as being close to drowning, by microbiological analysis and histological inspection of specimens taken from the damaged tissues, along with clinical manifestations and imaging data. Appropriate treatment should be provided quickly so as to reduce the mortality rate.
Patients consent
Patients provided informed consent for the publication of the cases.
Supplementary Information
Additional file 1: Figure S1. The pond the patient accidentally fell into.
Acknowledgements
None declared.
Disclosure
No potential conflict of interest was reported by the authors.
Authors’ contributions
Peng Yan, Haodi Wang wrote the main manuscript text and Junfeng Chen, Qi Jia prepared Figures S1, 2–5 and Table 1. All authors reviewed the manuscript. The author(s) read and approved the final manuscript.
Funding
None declared.
Availability of data and materials
Sequencing data were deposited to the National Genomics Data Center under accession numbers PRJCA014995 and CRA009845. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
The case report was approved by the Institutional Review Board (Research Review Committee and Ethical Review Committee) of the Ethics Committee of PLA General Hospital; the ethics code is S2020-141–01. And the patient provided informed consent.
Consent for publication
Patient provided informed consent for the publication of the case.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Jungang Xie, Email: xiejjgg@hotmail.com.
Guoxin Mo, Email: ted_momo@aliyun.com.
References
- 1.Cortez KJ, et al. Infections caused by Scedosporium spp. Clin Microbiol Rev. 2008;21(1):157–197. doi: 10.1128/CMR.00039-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chen M, et al. The ‘species complex’ issue in clinically relevant fungi: a case study in Scedosporium apiospermum. Fungal Biol. 2016;120(2):137–146. doi: 10.1016/j.funbio.2015.09.003. [DOI] [PubMed] [Google Scholar]
- 3.Rougeron A, et al. Ecology of Scedosporium species: present knowledge and future research. Mycopathologia. 2018;183(1):185–200. doi: 10.1007/s11046-017-0200-2. [DOI] [PubMed] [Google Scholar]
- 4.Luplertlop N. Pseudallescheria/Scedosporium complex species: From saprobic to pathogenic fungus. J Mycol Med. 2018;28(2):249–256. doi: 10.1016/j.mycmed.2018.02.015. [DOI] [PubMed] [Google Scholar]
- 5.Ramirez-Garcia A, et al. Scedosporium and Lomentospora: an updated overview of underrated opportunists. Med Mycol. 2018;56(1):102–125. doi: 10.1093/mmy/myx113. [DOI] [PubMed] [Google Scholar]
- 6.Guarro J, et al. Scedosporium apiospermum: changing clinical spectrum of a therapy-refractory opportunist. Med Mycol. 2006;44(4):295–327. doi: 10.1080/13693780600752507. [DOI] [PubMed] [Google Scholar]
- 7.Katragkou A, et al. Scedosporium apiospermum infection after near-drowning. Mycoses. 2007;50(5):412–421. doi: 10.1111/j.1439-0507.2007.01388.x. [DOI] [PubMed] [Google Scholar]
- 8.Hoenigl M, et al. Global guideline for the diagnosis and management of rare mould infections: an initiative of the European Confederation of Medical Mycology in cooperation with the International Society for Human and Animal Mycology and the American Society for Microbiology. Lancet Infect Dis. 2021;21(8):e246–e257. doi: 10.1016/S1473-3099(20)30784-2. [DOI] [PubMed] [Google Scholar]
- 9.Hung LH, Norwood LA. Osteomyelitis due to Pseudallescheria boydii. South Med J. 1993;86(2):231–234. doi: 10.1097/00007611-199302000-00020. [DOI] [PubMed] [Google Scholar]
- 10.Rüchel R, Wilichowski E. Cerebral Pseudallescheria mycosis after near-drowning. Mycoses. 1995;38(11–12):473–475. doi: 10.1111/j.1439-0507.1995.tb00022.x. [DOI] [PubMed] [Google Scholar]
- 11.Khurshid A, et al. Disseminated Pseudallescheria boydii infection in a nonimmunocompromised host. Chest. 1999;116(2):572–574. doi: 10.1378/chest.116.2.572. [DOI] [PubMed] [Google Scholar]
- 12.Poza G, et al. Meningitis caused by Pseudallescheria boydii treated with voriconazole. Clin Infect Dis. 2000;30(6):981–982. doi: 10.1086/313817. [DOI] [PubMed] [Google Scholar]
- 13.Tirado-Miranda R, et al. Septic arthritis due to Scedosporium apiospermum: case report and review. J Infect. 2001;43(3):210–212. doi: 10.1053/jinf.2001.0866. [DOI] [PubMed] [Google Scholar]
- 14.Kiratli H, et al. Scedosporium apiospermum chorioretinitis. Acta Ophthalmol Scand. 2001;79(5):540–542. doi: 10.1034/j.1600-0420.2001.790524.x. [DOI] [PubMed] [Google Scholar]
- 15.Levine NB, et al. An immunocompetent patient with primary Scedosporium apiospermum vertebral osteomyelitis. J Spinal Disord Tech. 2002;15(5):425–430. doi: 10.1097/00024720-200210000-00014. [DOI] [PubMed] [Google Scholar]
- 16.Farina C, et al. Scedosporium apiospermum post-traumatic cranial infection. Brain Inj. 2002;16(7):627–631. doi: 10.1080/02699050110119808. [DOI] [PubMed] [Google Scholar]
- 17.Chakraborty A, Workman MR, Bullock PR. Scedosporium apiospermum brain abscess treated with surgery and voriconazole. Case report. J Neurosurg. 2005;103(1 Suppl):83–87. doi: 10.3171/ped.2005.103.1.0083. [DOI] [PubMed] [Google Scholar]
- 18.Kooijman CM, et al. Successful treatment of Scedosporium aurantiacum osteomyelitis in an immunocompetent patient. Surg Infect (Larchmt) 2007;8(6):605–610. doi: 10.1089/sur.2006.038. [DOI] [PubMed] [Google Scholar]
- 19.Leechawengwongs M, et al. Multiple Scedosporium apiospermum brain abscesses after near-drowning successfully treated with surgery and long-term voriconazole: a case report. Mycoses. 2007;50(6):512–516. doi: 10.1111/j.1439-0507.2007.01410.x. [DOI] [PubMed] [Google Scholar]
- 20.Stripeli F, et al. Scedosporium apiospermum skeletal infection in an immunocompetent child. Med Mycol. 2009;47(4):441–444. doi: 10.1080/13693780802695470. [DOI] [PubMed] [Google Scholar]
- 21.Al-Jehani H, et al. Scedosporium cerebral abscesses after extra-corporeal membrane oxygenation. Can J Neurol Sci. 2010;37(5):671–676. doi: 10.1017/S0317167100010878. [DOI] [PubMed] [Google Scholar]
- 22.Hell M, et al. Post-traumatic Pseudallescheria apiosperma osteomyelitis: positive outcome of a young immunocompetent male patient due to surgical intervention and voriconazole therapy. Mycoses. 2011;54(Suppl 3):43–47. doi: 10.1111/j.1439-0507.2011.02106.x. [DOI] [PubMed] [Google Scholar]
- 23.Cumbo-Nacheli G, de Sanctis J, Holden D. Pseudallescheria Boydii pneumonia in an immunocompetent host. Am J Case Rep. 2012;13:163–165. doi: 10.12659/AJCR.883276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Angelini A, Drago G, Ruggieri P. Post-tsunami primary Scedosporium apiospermum osteomyelitis of the knee in an immunocompetent patient. Int J Infect Dis. 2013;17(8):e646–e649. doi: 10.1016/j.ijid.2013.02.011. [DOI] [PubMed] [Google Scholar]
- 25.Wilson HL, Kennedy KJ. Scedosporium apiospermum brain abscesses in an immunocompetent man with silicosis. Med Mycol Case Rep. 2013;2:75–78. doi: 10.1016/j.mmcr.2013.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Cruysmans C, et al. Epidural abscess caused by Scedosporium apiospermum in an immunocompetent child. Pediatr Infect Dis J. 2015;34(11):1277–1278. doi: 10.1097/INF.0000000000000864. [DOI] [PubMed] [Google Scholar]
- 27.Rahman FU, et al. Pulmonary scedosporiosis mimicking aspergilloma in an immunocompetent host: a case report and review of the literature. Infection. 2016;44(1):127–132. doi: 10.1007/s15010-015-0840-4. [DOI] [PubMed] [Google Scholar]
- 28.Dinh A, et al. Case of femoral pseudarthrosis due to Scedosporium apiospermum in an immunocompetent patient with successful conservative treatment and review of literature. Mycoses. 2018;61(6):400–409. doi: 10.1111/myc.12739. [DOI] [PubMed] [Google Scholar]
- 29.Tan S, et al. An unusual case of Scedosporium apiospermum fungaemia in an immunocompetent patient with a left ventricular assist device and an implantable cardiac device. Access Microbiol. 2020;2(9):acmi000148. doi: 10.1099/acmi.0.000148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Jabr R, Hammoud K. Scedosporium apiospermum fungemia successfully treated with voriconazole and terbinafine. IDCases. 2020;22:e00928. doi: 10.1016/j.idcr.2020.e00928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Liu W, Feng R, Jiang H. Management of pulmonary Scedosporium apiospermum infection by thoracoscopic surgery in an immunocompetent woman. J Int Med Res. 2020;48(7):300060520931620. doi: 10.1177/0300060520931620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mir W, et al. Scedosporium apiospermum Pneumonia in an Immunocompetent Host. Cureus. 2021;13(8):e16891. doi: 10.7759/cureus.16891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ghasemian R, et al. Fatal pulmonary Scedosporium aurantiacum infection in a patient after near-drowning: a case report. Curr Med Mycol. 2021;7(4):38–42. doi: 10.18502/cmm.7.4.8410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Song Y, et al. Scedosporium apiospermum and Lichtheimia corymbifera co-infection due to inhalation of biogas in immunocompetent patients: a case series. Infect Drug Resist. 2022;15:6423–6430. doi: 10.2147/IDR.S388166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Shi XW, et al. Scedosporium apiospermum infection of the lumbar vertebrae: a case report. World J Clin Cases. 2022;10(10):3251–3260. doi: 10.12998/wjcc.v10.i10.3251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Troke P, et al. Treatment of scedosporiosis with voriconazole: clinical experience with 107 patients. Antimicrob Agents Chemother. 2008;52(5):1743–1750. doi: 10.1128/AAC.01388-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chiu CY, Miller SA. Clinical metagenomics. Nat Rev Genet. 2019;20(6):341–355. doi: 10.1038/s41576-019-0113-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Figure S1. The pond the patient accidentally fell into.
Data Availability Statement
Sequencing data were deposited to the National Genomics Data Center under accession numbers PRJCA014995 and CRA009845. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.