Abstract
The putative envelope glycoproteins of hepatitis C virus (HCV) likely play an important role in the initiation of viral infection. Available information suggests that the genomic regions encoding the putative envelope glycoproteins, when expressed as recombinant proteins in mammalian cells, largely accumulate in the endoplasmic reticulum. In this study, genomic regions which include the putative ectodomain of the E1 (amino acids 174 to 359) and E2 (amino acids 371 to 742) glycoproteins were appended to the transmembrane domain and cytoplasmic tail of vesicular stomatitis virus (VSV) G protein. This provided a membrane anchor signal and the VSV incorporation signal at the carboxy termini of the E1 and E2 glycoproteins. The chimeric gene constructs exhibited expression of the recombinant proteins on the cell surface in a transient expression assay. When infected with a temperature-sensitive VSV mutant (ts045) and grown at the nonpermissive temperature (40.5°C), cells transiently expressing the E1 or E2 chimeric glycoprotein generated VSV/HCV pseudotyped virus. The resulting pseudotyped virus generated from E1 or E2 surprisingly exhibited the ability to infect mammalian cells and sera derived from chimpanzees immunized with the homologous HCV envelope glycoproteins neutralized pseudotyped virus infectivity. Results from this study suggested a potential functional role for both the E1 and E2 glycoproteins in the infectivity of VSV/HCV pseudotyped virus in mammalian cells. These observations further suggest the importance of using both viral glycoproteins in a candidate subunit vaccine and the potential for using a VSV/HCV pseudotyped virus to determine HCV neutralizing antibodies.
Hepatitis C virus (HCV) is a major causative agent of parenterally transmitted hepatitis (1, 4). HCV accounts for most cases of acute and chronic liver disease, with serious consequences which may lead to the development of hepatocellular carcinoma (49). HCV is classified in the family Flaviviridae, in a separate and as yet unnamed genus. The virus genome contains a linear, positive-stranded RNA molecule of ∼9,500 nucleotides, encoding a polyprotein precursor of ∼3,000 amino acids (aa) (4). This polyprotein is cleaved by both host and viral proteases (18, 19, 53) to generate at least nine distinct polypeptides: the putative structural proteins (C, E1, and E2) and several nonstructural proteins (NS2, NS3, NS4A, NS4B, NS5A, and NS5B). Processing of the viral proteins examined by in vitro translation (19) and by transient expression (18) suggests that the putative structural proteins are located in the N-terminal one-fourth of the polyprotein. The core protein (∼21 kDa) is followed by two putative envelope proteins, E1 (∼31 kDa) and E2 (∼70 kDa), both of which are heavily modified by N-linked glycosylation. The remainder of the polyprotein is believed to encode the nonstructural proteins. The biosynthesis of the E1 and E2 glycoproteins has been studied by using a cDNA template and shown to be produced by common specific cleavage from the precursor protein at approximately amino acid positions 191 and 383 (19). The glycoproteins are presumed to be typical type 1 membrane-associated proteins with anchorage through the carboxyl-terminal portion. The majority of E1 and E2 expressed as recombinant proteins are localized intracellularly and appear to form a complex, as evidenced by coimmunoprecipitation with antibodies to E1 or E2 (13, 45, 57). The predominant heterodimer complex of the E1 and E2 glycoproteins is probably stabilized by noncovalent interactions, with a minor fraction of heterogenous disulfide-linked aggregates, representing misfolded E1-E2 complexes.
Biosynthesis and processing of the E2 glycoprotein has been studied extensively in the past few years, and available information suggests that posttranslational processing occurs. The existence of three E2 species with distinct C termini has been suggested to be the result of complex processing of the HCV proteins and by protein-protein interactions (53). Amino acid sequences upstream of the cleavage site of E2 are well conserved among all HCV isolates and are similar to signal sequences. However, the efficiency of the cleavage of this newly identified site is lower than that apparent between aa 809 and 810. Inefficient cleavage at the newly identified site suggests the presence of at least two E2 products with various lengths of peptide backbones in their C-terminal moieties. When the entire region of E2 is expressed by an in vitro transcription-translation system and analyzed to determine the size of the peptide backbone after treatment with endoglycosidase F, two proteins of 40 and 37 kDa are observed. Lin et al. (33) have identified a protein, called p7, by expression of a series of C-terminally truncated polyproteins which has been mapped between E2 and NS2. The presence of potential signal/anchor hydrophobic sequences preceding the E2/p7 and p7/NS2 cleavage sites and the results of cell-free translation analyses indicate that host signal peptidase may catalyze both of these cleavages. However, cleavage at the E2/p7 site is incomplete, leading to the production of two stable E2-specific proteins with different C termini, E2 and E2-p7.
There is no clear evidence which may define the mechanism of HCV interaction with mammalian cells. The lack of a convenient in vitro cell culture system (2, 32, 40, 54, 55, 64) to analyze the neutralization of HCV infectivity makes it difficult to understand the role of the individual glycoproteins in the initiation of viral infection. Phenotypic mixing of virus envelope glycoproteins (6, 36, 63) and the incorporation of proteins into vesicular stomatitis virus (VSV) have been used to facilitate the study of the biological properties of a number of envelope glycoproteins (42, 51). VSV infects and efficiently replicates in a large range of mammalian cells. The VSV pseudotypes resist neutralization by anti-VSV antibodies but are sensitive to neutralizing antibodies specific to the envelope antigens of the donor virus. The host range for viral attachment and penetration of pseudotypes is restricted to cells bearing receptors for the donor virus. Following penetration and uncoating, however, the VSV genome contained in the pseudotyped particle replicates at the permissive temperature to produce nonpseudotyped progeny. Thus, a cytopathic plaque assay of VSV pseudotypes may be used to determine receptor expression and receptor interference and to measure neutralizing antibodies specific to the donor virus encoding the envelope glycoproteins (7).
In this study, we have investigated the role of the HCV glycoproteins in the viral infection of mammalian cells. Chimeric genes comprised of the putative ectodomain of E1 and E2 from the genotype HCV 1a strain (HCV-1a) and the transmembrane and cytoplasmic tail of VSV G glycoprotein were constructed to express the chimeric HCV glycoproteins on the surface of mammalian cells, thereby facilitating their incorporation into the pseudotyped virus envelope. Pseudotyped virus was generated by transfection of chimeric genes and infection with a temperature-sensitive mutant of VSV (ts045). A single amino acid substitution in the ectodomain leads to misfolding and aggregation of the ts045 G protein at 40°C. This results in the retention of the G protein within the endoplasmic reticulum, and infectious virus is not produced (17, 52, 61). This system was used to ensure the recovery of progeny VSV/HCV pseudotyped virus while decreasing the nonpseudotyped background VSV which would be present with use of wild-type virus. Infectivity of the pseudotyped virus and neutralization by HCV glycoprotein-specific antisera suggested separate roles for the E1 and E2 glycoproteins in the interaction with host cells and virus entry.
MATERIALS AND METHODS
Cells.
Human cervical carcinoma cells (HeLa), baby hamster kidney cells (BHK-21), a human T-cell lymphoma line (MOLT4), a human hepatoma cell line (HepG2), and a human embryonic lung (L-132) cell line were obtained from the American Type Culture Collection (Rockville, Md.). HeLa, BHK-21, and HepG2 cells were grown in Dulbecco’s medium supplemented with 10% fetal calf serum, penicillin (100 U/ml), and streptomycin (100 μg/ml). MOLT4 cells were grown in RPMI 1640 supplemented with 15% fetal calf serum and antibiotics.
HCV-specific antibodies.
Rabbit antisera WU122 and WU105 (18), raised against bacterially expressed HCV E1 and E2 proteins, respectively, were kindly provided by Charles M. Rice (Washington University, St. Louis, Mo.). Murine monoclonal antibodies (MAbs) to E1 (3D5/C3) and E2 (3E5-1) and a polyclonal mouse antiserum (2m) to the recombinant E2 glycoprotein were kindly provided by Michael Houghton (Chiron Corporation, Emeryville, Calif.). These antibodies and the polyclonal antisera generated to two different peptides representing B-cell epitopes of E1 (46) were used to characterize the chimeric glycoproteins or to test for specific neutralization of HCV/VSV pseudotyped virus infectivity. Sera from chimpanzees immunized with the recombinant E1 and E2 coexpressed in and purified from HeLa cells, or with a combination of yeast-expressed E1 (aa 190 to 330) and baculovirus-expressed E2 (aa 404 to 661) proteins (5, 48), kindly provided by Michael Houghton, were also used to test the neutralization of pseudotyped virus infectivity. Hyperimmune rabbit sera to VSV (42) and vaccinia virus (VV) (43) were used to neutralize any potential VSV or VV generated in the production of pseudotyped virus.
Chimeric gene construction.
Cloned DNA encoding the complete G protein of VSV (pSVGLI; kindly provided by John K. Rose, Yale University, New Haven, Conn.) was used to obtain the transmembrane domain and cytoplasmic tail of the G protein. The VSV G insert was excised from pSVGLI by digestion with EcoRI and inserted into the mammalian expression vector pcDNA3 (Invitrogen, San Diego, Calif.). Subsequently, an oligonucleotide (5′ TTC AGT AGT gGt Acc AGC TCC ATT 3′) containing the KpnI site (underlined) and an SP6 primer were used to synthesize the genomic region encoding the entire transmembrane domain and cytoplasmic tail (39 amino acid residues from the carboxyl terminus) of the VSV G protein along with the 3′ flanking vector sequences by PCR. (The nucleotides in lowercase show changes made in the synthetic oligonucleotide primers to create suitable restriction enzyme sites to facilitate cloning into the plasmid vector or for in-frame ligation with the VSV G sequences.) The amplified product was digested with KpnI and EcoRI for unidirectional cloning into the pcDNA3 expression vector for use in chimeric gene constructs.
Based on the available information, the amino acids positions of the E1 and E2 molecule were chosen to represent their respective putative ectodomains for the generation of the chimeric proteins. The HCV E1 (aa 174 to 359) genomic region was amplified from a partial cDNA clone (Blue4/C5 p-1) of HCV-1a containing the 5′ untranslated region, C, E1, E2, and a portion of NS2 (kindly provided by Michael Houghton). Both sense (5′ CTT CCT GGT acC atg TTC TCT ATC 3′, nucleotide positions 504 to 528, containing the underlined KpnI site) and antisense (3′ GAA ATA CGC ggT aCC CGC CAG 5′, nucleotide positions 1665 to 1686, containing the underlined KpnI site) primers were used for PCR amplification of the genomic region as described earlier (25). Amplified DNA was digested with KpnI for in-frame ligation of its carboxyl terminus with the transmembrane and the cytoplasmic domain of VSV G, already cloned into the pcDNA3 vector, as described.
We selected E2 genomic sequences up to aa 742 to cover a known E2 product and to retain those sequences which have the potential to interact with E1. The HCV E2 (aa 371 to 742) genomic region was similarly amplified by using a sense (5′ ATG GTG GGt AcC TGG GCG ATG GTC 3′, nucleotide positions 1092 to 1113, containing the underlined KpnI site) and antisense (5′ CTC CGC TTG GGt acc GAG GAT 3′, nucleotide positions 2214 to 2235, containing the underlined KpnI restriction site) primers and ligated in frame through the carboxyl terminus to the transmembrane domain of VSV G protein. A stop codon was present at the end of the VSV cytoplasmic tail in all of the chimeric gene constructs. In the chimeric E1 gene construct, aa 359 was changed from isoleucine to threonine. In the E2 gene construct, minor amino acid changes (Leu379 to Ile, Ilu741 to Gly, and Ser742 to Thr) were made to facilitate in-frame ligation with the VSV G transmembrane domain.
In vitro translation.
Chimeric gene constructs were used for in vitro translation using the TNT coupled reticulocyte lysate system with T7 RNA polymerase in the presence of canine pancreatic microsomal membrane (Promega Corporation, Madison, Wis.) as described earlier (47). As a negative control, pcDNA3 vector alone was included. The reaction was carried out in the presence of [35S]methionine (Amersham Corporation), and the translation products were analyzed by immunoprecipitation with specific antibodies.
Immunofluorescence.
Protein expression from the chimeric gene constructs was tested by transient expression (16) using recombinant VV expressing the bacteriophage T7 polymerase (vvT7). HeLa-T4 cells, which are relatively resistant to the cytopathic effect of VV, were infected with vvT7 and transfected with plasmids containing the chimeric genes by using Lipofectamine (Bethesda Research Laboratories, Gaithersburg, Md.) as described earlier (20) and incubated for expression of the viral proteins. Cells were tested after 20 h of transfection for surface expression of the viral glycoproteins by indirect immunofluorescence. MAbs to the E1 (3D5/C3) and E2 (3E5-1) glycoproteins were added as the primary antibodies and incubated for 30 min at room temperature. After washing with phosphate-buffered saline (PBS), a fluorochrome-conjugated second antibody to mouse immunoglobulin (Zymed Laboratories Inc., San Francisco, Calif.) was added and incubated for an additional 30 min. After subsequent washings with PBS, the coverslips were mounted on microscope slides with a solution of glycerol in PBS and viewed with a fluorescence microscope (Nikon Optiphot-2) to observe the cell surface expression of HCV glycoproteins.
Generation of VSV/HCV pseudotyped virus.
BHK cells were infected with vvT7 (multiplicity of infection ∼5) and VSV ts045 (multiplicity of infection of ∼5). After a 1-h adsorption of the virus, cells were rinsed and transfected individually with the E1G or E2G plasmid DNA (experimental), wild-type VSV G (VSV Gwt) cloned into pcDNA3 under the control of T7 promoter (Gwt/VSV; positive control), or pcDNA3 vector DNA (negative control) by using Lipofectamine (Bethesda Research Laboratories) and incubated at 40.5°C for 5 h. The transfection mix was removed, serum-free medium was added, and incubation continued for another 16 h. Cell culture medium was collected and clarified by centrifugation at 3,000 × g for 15 min. The supernatant was filtered through a sterile MILLEX-V 0.1-μm-pore-size filter unit (Millipore Products, Bedford, Mass.) to aid in the removal of VV (50) or treated with high-titer antiserum to VV at the appropriate time. The culture fluid was flash frozen and stored at −70°C in aliquots for future use, with each aliquot thawed for a single time prior to use. Pseudotyped virus preparations were tested for reverted ts045 or VV by neutralization with a high-titer anti-VSV or anti-VV serum, respectively. Additionally, the pseudotyped virus was assayed at 40°C, and absence of plaque formation further indicated its temperature-sensitive phenotype.
Analysis for infectivity of pseudotyped virus.
The virus titer of the supernatant was determined after incubation with neutralizing antiserum to VSV and/or VV by plaque assay using HepG2, BHK, and MOLT4 cell lines. To test with MOLT4 cells, the pseudotyped virus was incubated with the cells at 33°C, washed, and dispersed on top of BHK indicator cells to detect progeny VSV release from MOLT4 cells (9). Cells were overlaid with 0.8% agarose containing Dulbecco’s medium and incubated at 33°C. Alternatively, virus titer was determined by direct infection of the HepG2 or BHK cell monolayer with an agar overlay. Infectivity of the pseudotyped virus depends on the attachment and interactions of the HCV glycoproteins present on the pseudotyped virus with the target cells. Once the genome of VSV enters into cells, normal replication of VSV occurs and plaque formation is detected. Cells were stained with an additional overlay of agar containing 0.005% neutral red 44 h after infection to facilitate plaque counting. Fuzzy plaques ∼2 mm in diameter were visible and counted after 6 h of staining.
To determine the neutralizing antibody response, pseudotyped virus (approximately 100 PFU) was incubated with anti-VSV and anti-VV neutralizing antiserum (42, 43), with or without HCV-specific test antibodies. HCV-specific ascites fluid or antisera were heat inactivated at 56°C for 30 min and adsorbed with kaolin to remove potential viral inhibitors as described earlier (10). The predetermined number of plaque-forming virus particles with (experimental) or without (control) antibody was incubated at 33°C for 45 min, added to HepG2, BHK, or phytohemagglutinin-stimulated MOLT4 cells, and incubated at 33°C for 1 h with intermittent tilting. HepG2 or BHK cells were washed two times with Dulbecco’s medium, and an overlay with 0.8% agarose containing Dulbecco’s medium adjusted to 1× was added onto the cell layer. Similarly, MOLT4 cells were washed and added on top of BHK cells. Cells were overlaid with a minimum volume of Dulbecco’s medium (∼0.2 ml), and a similar overlay of 0.8% agarose was added 15 min after the addition of cells. Cells were stained with an additional overlay of agar containing 0.005% neutral red and counted after 6 h of incubation as described above. A reduction of ≥50% in the number of pseudotype PFU compared to the control was considered the neutralization titer of the experimental antibody.
RESULTS
Chimeric gene constructs of HCV E1 and E2 glycoproteins.
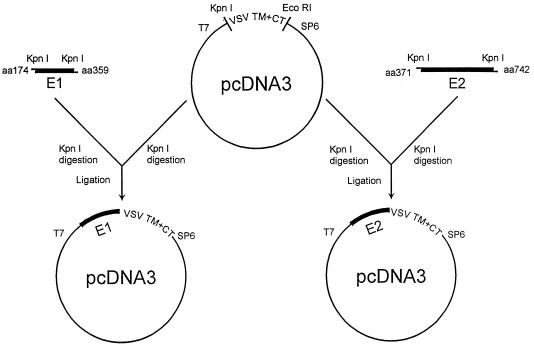
The carboxyl-terminally truncated E1 (aa 174 to 359) gene construct from HCV-1a allows for the secretion of the HCV glycoprotein in mammalian cells (53). On the other hand, the major contributing factor for the oligomerization of E1 and E2 was mapped to the vicinity of hypervariable region 2 (HVR2) of E2. The amino acid sequence WHY in this region plays a crucial role in the association between E1 and E2, and the N-terminal part of E1 is important for binding to E2 sequences (62). Furthermore, a major hydrophobic region is present at the amino terminus of E2. Based on this information, the amino acid positions of the E1 (aa 174 to 359) and E2 (aa 371 to 742) glycoproteins were chosen for these chimeric gene constructs. To this region, the transmembrane domain and cytoplasmic tail of VSV G were appended at the C terminus to provide a membrane anchor signal. Chimeric gene constructs (E1G and E2G) composed of the putative ectodomain of the E1 and E2 glycoproteins and the transmembrane and cytoplasmic tail of VSV G are shown in Fig. 1. The gene constructs contained the signal peptide and cleavage site from the amino terminus of the E1 or E2 protein. Orientation of the gene constructs were tested by restriction enzyme digestion and by nucleotide sequencing of the junction regions.
FIG. 1.
Schematic presentation of the chimeric gene constructs used for expression of the HCV glycoproteins on the mammalian cell surface.
Analysis of protein products from chimeric gene constructs.
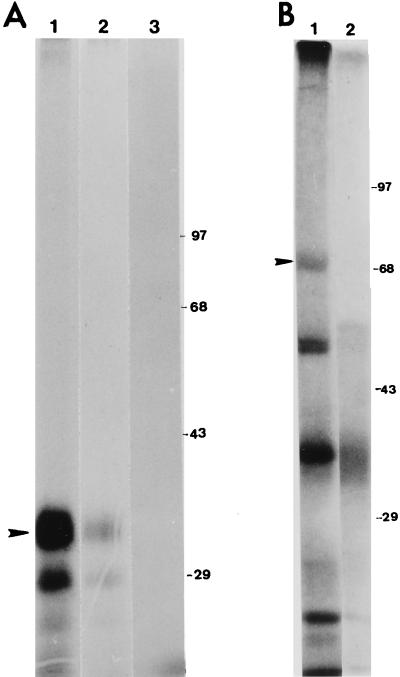
To characterize the polypeptides encoded by the chimeric gene constructs, an in vitro translation assay was performed in the presence of microsomal membrane. Protein expression from the chimeric gene constructs was also analyzed in transfected mammalian cells by using vvT7 (16) and immunoprecipitation. In vitro-translated products were immunoprecipitated by HCV- or VSV-specific antibodies and analyzed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) (Fig. 2). Two major polypeptide bands of ∼32 and ∼28 kDa appeared as translated protein products from the E1G chimeric gene construct. The polypeptides probably represent different glycosylated forms of the E1 protein, as they were immunoprecipitated by a MAb to the E1 glycoprotein or an anti-VSV serum (Fig. 2A). Similarly, antibodies immunoprecipitated the polypeptides from transient expression assay (data not shown). Analysis of the in vitro-translated E2G chimeric gene construct suggests synthesis of the authentic E2 polypeptide of ∼70 kDa, immunoprecipitated by anti-VSV serum (Fig. 2B). The other immunoprecipitated low-molecular-size polypeptides may represent precursor or partial glycosylated forms of E2. However, use of the available antibodies to E2 failed to convincingly immunoprecipitate E2 glycoprotein either from in vitro-translated product or from transiently transfected mammalian cells. The reason for lack of reactivity of the antibodies to the E2 glycoprotein is not clear at this time. Since VSV/HCV proteins are exported to the cell surface rather than retained in the endoplasmic reticulum, they may have different glycosylation or conformation patterns, which may alter the antigenicity of the chimeric proteins.
FIG. 2.
In vitro translation and immunoprecipitation of E1 and E2 chimeric glycoproteins with VSV G by specific antibodies. (A) [35S]methionine-labeled in vitro-translated E1 chimeric proteins in the presence of microsomal membrane were immunoprecipitated separately with a MAb to E1 (lane 1) and a polyclonal antiserum to VSV (lane 2) and analyzed by SDS-PAGE (10% gel). A negative control was similarly run with pcDNA3 vector and the polyclonal antiserum to VSV (lane 3). (B) An in vitro-translated E2 chimeric glycoprotein was tested by using a polyclonal antibody to VSV (lane 1) with vector DNA as the negative control (lane 2). Immunoprecipitates were analyzed by SDS-PAGE (7% gel). Arrowheads on the left indicate locations of the mature E1 or E2 chimeric proteins; sizes on the right are expressed in kilodaltons.
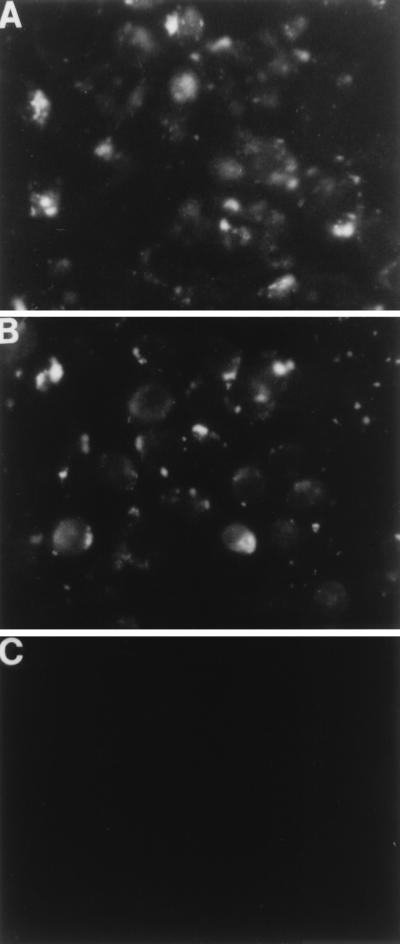
Cell surface expression of the HCV chimeric glycoproteins.
The chimeric gene constructs were tested in a transient expression assay to determine the localization of the recombinant glycoproteins by surface immunofluorescence. Both gene constructs showed expression of E1 and E2 on the plasma membrane, as detected by specific MAbs in unfixed cells (Fig. 3A and B). Parallel mock-transfected negative control cells did not show any reactivity with the antibodies in a similar assay (Fig. 3C). Results from this experiment indicated that the HCV chimeric glycoproteins were processed from the transfected gene constructs and transported to the mammalian cell surface. Although the MAb to the E2 glycoprotein reacted with the chimeric E2 in an immunofluorescence assay, the same antibody could not immunoprecipitate E2 either from in vitro-translated product or from transiently transfected mammalian cells.
FIG. 3.
Surface immunofluorescence of cells transiently expressing chimeric glycoproteins from the E1G and E2G gene constructs. HeLa cells were infected with recombinant vvT7 and transfected with E1G (A), E2G (B), or pcDNA3 vector DNA (C) and reacted with a MAb to E1 or E2 after 18 h of incubation. Cells in panel C were tested with a mixture of the MAbs to both E1 and E2.
Infectivity of the VSV/HCV pseudotyped virus.
Culture medium from BHK cells transfected with the chimeric gene constructs and infected with a temperature-sensitive mutant of VSV (ts045) was examined for the generation of VSV/HCV pseudotypes (Table 1). The virus showed titers in the order of >104 PFU/ml, and these levels were not altered following treatment with high-titer anti-VSV or anti-VV serum, suggesting the absence of detectable revertant VSV or VV leakage in the pseudotyped virus preparation. Furthermore, plaques were not observed in culture fluid derived from mock-transfected controls under identical assay conditions. The titer in the culture fluid of the pseudotype Gwt/VSV (positive control), generated by transfection with VSV Gwt cloned into pcDNA3 under the control of a T7 promoter, was greater than 105 PFU/ml (Table 1). Plaques were not observed with Gwt/VSV when assayed at 40°C, indicating that the rescued virus was still temperature sensitive and showing that rescue of ts045 did not result from reversion of the temperature-sensitive phenotype. Infectivity of the pseudotyped virus generated from cells expressing either the E1 or E2 glycoprotein in the presence of anti-VSV and anti-VV serum suggested that the individual glycoproteins of HCV may have a functional role associated with or leading to virus entry into host cells. A similar plaque assay of the E1 or E2 pseudotype virus in MOLT4 cells on top of BHK indicator cells showed distinct plaque formation. Discrete small plaques in HepG2 cells were also noted when pseudotyped virus stock was similarly titrated. However, the number of distinct plaques was visibly lower, especially with the HepG2 cells, over a similar incubation period. Plaque size also differed with cell type: BHK cells presented the more distinct large plaques, an intermediate size was seen in MOLT4 cells, and the smallest plaques were seen in HepG2 cells. On the other hand, a similar plaque assay of the HCV/VSV pseudotyped virus failed to show detectable plaque formation on HeLa or L-132 cells. However, Gwt/VSV (positive control) showed distinct plaque formation on these two cell types. This result suggested the lack of interaction of the chimeric E1 or E2 glycoprotein with HeLa and L-132 cells or lack of entry of the pseudotyped virus by pinocytosis as a possible nonspecific entry mechanism. A similar chimeric protein between human immunodeficiency virus Env and VSV G (Env-G665) was examined earlier, using the ts045 rescue system (42a). This chimeric protein was incorporated into pseudotyped virions but did not rescue infectivity. If pinocytosis is sufficient to confer infectivity of pseudotyped virus, this chimeric protein would also be expected to be infectious. Since the pseudotyped virus causes infection in BHK cells, it appears that the receptors for HCV glycoproteins are also present in nonhepatic and nonhuman cells, but it is not proven that this cell line supports HCV replication. Limited virus replication has previously been observed in porcine kidney (PK-15) and Vero cells (1a). Efficient long-term viral replication, however, has not been described for any system. It is possible that HCV has receptors on a number of cell types. However, the virus is probably stringently hepatotropic, being restricted to replicate and release only in differentiated hepatocytes which may have specific functions to support viral growth. Since plaquing efficiency of the pseudotype virus was highest on BHK cells, the remainder of the study was performed with this cell line.
TABLE 1.
Infectivity of BHK cells by the pseudotyped virus
| Pseudotyped virusa | Antibody testedb | No. of plaques/mlc |
|---|---|---|
| HCV E1/VSV | None | 1.2 × 104–1.8 × 104 |
| Anti-VSV | 1.1 × 104–1.7 × 104 | |
| Anti-VV | 1.3 × 104–1.6 × 104 | |
| HCV E2/VSV | None | 8.0 × 103–1.4 × 104 |
| Anti-VSV | 8.6 × 103–1.3 × 104 | |
| Anti-VV | 7.1 × 103–1.3 × 104 | |
| Gwt/VSV (positive control) | None | 1.2 × 105–2.1 × 105 |
| Anti-VSV | <101 | |
| None (mock transfected [pcDNA3/VSV negative control]) | None | <101 |
Pseudotyped virus were generated by coinfection of cells with vvT7 and VSV ts045, followed by transfection with the HCV E1/E2 or VSV Gwt gene under the control of the T7 promoter.
Rabbit antisera to VSV and VV were heat inactivated at 56°C for 30 min and separately titrated for neutralization of the indicated viruses prior to use in this analysis. Both antisera had neutralization titers of >105 at 1:1,000 dilution.
Range of virus titers at 32°C from three experiments. Viruses did not form detectable plaques when grown at 40°C.
Role of antibodies in the neutralization of HCV/VSV pseudotyped virus infectivity.
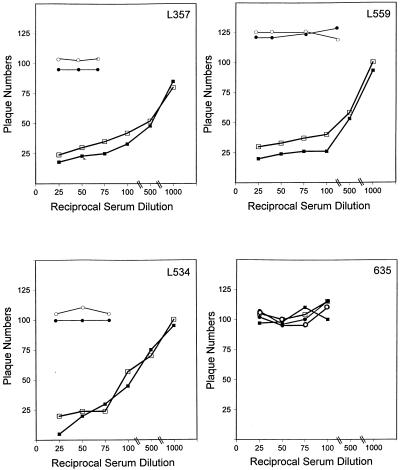
We included a number of antibodies and blinded sera from chimpanzees immunized with the viral glycoproteins to test for neutralization of pseudotyped virus infectivity. Antibodies that were positive for reactivity with the E1 or E2 glycoprotein in a binding or immunoprecipitation assay and antibodies known not to react with the viral antigens were tested for the ability to neutralize VSV/HCV infection of BHK cells by incubating the pseudotyped virus with antibody prior to addition onto the cells. Results clearly demonstrated that the sera from chimpanzees immunized with HCV recombinant glycoprotein preparations which exhibited protection from challenge infection produced significant reductions (≥50%) in the number of pseudotype PFU at a dilution of 1/100 to 1/500 (Fig. 4). In contrast, chimpanzee 635, which received a vaccine derived from yeast and insect cells (48) rather than the mammalian cell-derived vaccine that chimpanzees L357, L534, and L559 received, failed to show neutralization of pseudotyped virus infectivity. The neutralization of pseudotyped virus infectivity was tested in at least three separate experiments using different stocks of pseudotyped virus preparations. The neutralizing chimpanzee sera specific for the HCV glycoproteins, when tested for reactivity with Gwt/VSV at 1:50 and 1:100 dilutions, did not display neutralization of virus plaque formation. The results further suggested that antibodies specifically prevent pseudotyped virus infection generated from the HCV E1 or E2 glycoprotein. Chimpanzees L357, L534, and L559 were completely protected against homologous challenge with 10 infectious doses of HCV-1 (5). Chimpanzee 635 became infected upon challenge with HCV and is a carrier. Results derived from immunized chimpanzees suggest the importance of subunit vaccination using recombinant glycoproteins produced in mammalian cells and correlates with the induction of neutralizing antibodies to the VSV/HCV pseudotyped virus. On the other hand, a MAb or mouse antisera to two B-cell epitopes of the E1 glycoprotein, when tested similarly (Table 2), displayed a weak pseudotype neutralization titer (1/20). We also tested monospecific rabbit antisera to unglycosylated (bacterially expressed) E1 or E2 proteins and a polyclonal mouse antiserum (2m) to the recombinant E2 glycoprotein of HCV for pseudotyped virus neutralization. The rabbit antiserum to the E1 glycoprotein showed a weak neutralization titer (1/20), whereas anti-E2 serum had a neutralizing titer of <1/10 against the relevant pseudotyped virus. Further, the mouse antiserum (2m) to the recombinant E2 glycoprotein showed neutralization (titer of 1/100) specifically to the E2 pseudotyped virus.
FIG. 4.
Serial dilutions of sera from chimpanzees L357, L559, L534, and 635 vaccinated with recombinant envelope glycoproteins of HCV were tested for neutralizing antibodies to VSV/HCV pseudotyped virus. Activities at different reciprocal dilutions of immunized sera to the E1 (□) and E2 (▪) glycoproteins are expressed against pseudotype plaque numbers. Preimmune chimpanzee sera were similarly tested for reactivity to the E1 (○) and E2 (•) glycoproteins as negative controls. Plaque numbers varied within ±5 in each independent experiment.
TABLE 2.
Neutralization of pseudotyped virus infectivity on BHK cells
| Antibody testeda | Neutralization withb:
|
|
|---|---|---|
| HCV E1/VSV | HCV E2/VSV | |
| Mouse antiserum to P1 peptide | 1/20 | ND |
| Mouse antiserum to P2 peptide | 1/20 | ND |
| MAb to E1 | 1/20 | ND |
| Rabbit antiserum to E1 (WU122) | 1/20 | <1/10 |
| Rabbit antiserum to E2 (WU105) | ND | <1/10 |
| Mouse antiserum to E2 (2m) | <10 | 1/100 |
| MAb to E2 | ND | <1/10 |
HCV-specific antibodies were tested at 1/10 and higher dilutions.
The indicated pseudotyped viruses were treated with hyperimmune rabbit antisera to VSV (1:50) and VV (1:50) as an additional precautionary step in case of reversion of VSV or leakage of VV during testing for neutralization with HCV-specific antibodies. ND, not determined.
DISCUSSION
We have used an artificial HCV/VSV pseudotyped virus as an alternative to an in vitro infection system for HCV to study the binding and entry of HCV into target cells. Results from this study suggested an independent functional role for both the E1 and E2 chimeric glycoproteins of HCV in pseudotyped virus infectivity of mammalian cells, provided that the functions of the chimeric glycoproteins used in this assay reflect those seen in natural infection. We do not know at this time whether the E1 and E2 glycoproteins of HCV display a fusion activity with the host cell membrane or if the parenteral VSV genome of the pseudotyped virus enters into cells by receptor-mediated endocytosis at or around the neutral pH used under our experimental conditions. The antibodies generated as a result of immunization of the recombinant E1 and E2 glycoproteins expressed in mammalian cells induced pseudotyped virus neutralizing antibodies in chimpanzees, and the immunized chimpanzees also showed protection or ameliorated disease following challenge infection with a homologous HCV strain (5).
Immunity in HCV infection appears to be weak, but the reasons for this are not clear (44). Both E1 and E2 possess an N-terminal hypervariable domain (3, 22, 23). Although the immune response to the E1 glycoprotein has not been analyzed, some important observations have been made regarding the E2 glycoprotein of HCV. Amino acid sequence variation in the E2 hypervariable domain is comparable to that of the human immunodeficiency virus type 1 gp120 V3 domain (60), and changes in HCV amino acid coding are reported to be associated with a number of mutations, clustered at the 5′ end of the E2 coding region (41). Two hypervariable regions, HVR1 (27 amino acids) and HVR2 (7 amino acids), in the putative E2 envelope glycoprotein have been identified (14, 23). HVR1 contains a sequence-specific immunological epitope which induces the production of antibodies restricted to the specific viral isolate (23, 60). E2 variants can coexist simultaneously within a single individual, or a particular variant may predominate during different episodes of disease, contributing to a quasispecies nature of HCV (23, 34, 35, 60). HVR1 is probably the major site of HCV genetic drift, with amino acid substitutions in the two overlapping B-cell epitopes of HVR1 leading to escape from recognition by preexisting anti-HVR1 antibodies, and qualitative changes in antibody accompany HVR1 epitope shifts during the clinical course of hepatitis (24). Antibodies to the hypervariable region of the E2 glycoprotein have been shown to be protective and contribute to the selective replication of HCV in chimpanzees (26). The early appearance of antibodies to the N terminus of HVR1 is suggested with acute self-limiting infections of HCV (65). Binding of HCV to cells measured by reverse transcription-PCR for monitoring viral infection parallels in vitro infectivity of HPB-Ma cells and neutralization of HCV mediated, in part, by an isolate-specific antibody recognizing the HVR1 region (54, 56). In vitro neutralization of HCV by patient sera (14) and by the hyperimmune serum to the HVR1 of E2 (15) showed similar results in in vitro binding studies (56). Zibert et al. (64) suggested that although a majority of neutralizing antibodies are directed against the HVR1 of E2, the existence of high titers of HVR1-specific antibodies may not predict neutralization and is not sufficient to block the binding of virus to human fibroblast cells. In a different study (48), the E2 glycoprotein was shown to bind human cells with high affinity, and the ability to neutralize the binding of E2 derived from the HCV-1 genotype was equally distributed among sera from patients infected with HCV genotypes 1, 2, and 3, demonstrating that binding of E2 is partly independent of E2 hypervariable regions.
Viral envelope proteins play roles in several aspects of the viral life cycle such as receptor binding, penetration of host cells, and virus morphogenesis at budding (reviewed in reference 30). Enveloped viruses often contain one or more types of membrane proteins which may constitute a higher-order oligomer. Single-stranded enveloped RNA viruses such as influenza viruses, alphaviruses, rhabdoviruses, and flaviviruses are internalized into cells by receptor-mediated endocytosis. Other enveloped viruses, including paramyxoviruses, coronaviruses, and retroviruses such as human immunodeficiency virus, have glycoproteins which mediate fusion at neutral pH. While these viruses can cause fusion of the plasma membrane, it remains to be determined conclusively if successful entry leading to viral replication occurs via entry at the plasma membrane or after internalization and with pH-independent fusion occurring in endosomes. In addition, the roles of host and viral membrane lipid composition in conjunction with the nature of the cellular cytoskeletal architecture have to be considered and may contribute to initiate fusion.
The interaction between E1 and E2 and their role as a heterodimeric functional subunit during HCV infection has been suggested earlier (11, 13, 18, 31, 45). A recent study with pestivirus, an enveloped positive-stranded RNA virus, suggested that infection is accomplished by the interaction of at least two structural proteins, Erns and E2, which interact with different cell surface components (20a). The alphavirus Semliki Forest virus (SFV), another positive-stranded RNA virus, enters cells by receptor-mediated endocytosis and undergoes fusion at low pH. The SFV spikes consist of heterotrimers of noncovalently associated subunits, two transmembrane proteins E1 and E2, and a small peripherally associated subunit E3. The E2 and E3 polypeptides of SFV are derived from a precursor protein, cleavage of which is required for efficient fusion. However, it is generally believed that a hydrophobic domain contained within E1 is the fusion peptide (30). In Sindbis virus, the highly conserved E1 glycoprotein is involved in cell attachment, membrane fusion, and entry (reviewed in reference 59). On the other hand, the E2 glycoprotein also helps in cell attachment and contains the most potent epitopes for eliciting neutralizing antibodies. The binding of herpes simplex virus types 1 and 2 (HSV-1 and HSV-2), DNA viruses, is mediated by the interaction of viral envelope glycoproteins B and C (gB and gC), and fusion is mediated by gB, gD, and gH-gL hetero-oligomers (58). In HSV syncytial mutants, even though the gK is not localized on the cell surface, it influences fusion of infected cells (21). Our results regarding VSV/HCV pseudotype infectivity suggest that the HCV E1 and E2 glycoproteins may be involved either independently or in concert as a high-order oligomer for virus entry into cells. If HCV entry occurs through a hetero-oligomeric complex of the envelope glycoproteins, the question remains as to the specific role of E1 and/or E2 in this active conformation. Since the pseudotyped virus expressed individual monomers, their conformation may be altered in the heterodimer of E1 and E2 on the native virus particle. Hyperimmune serum to HVR1 of E2 is known to neutralize HCV infectivity (15). Even though the individual E1 and E2 glycoproteins may have a role in the heterodimeric form in virus particle, antibodies to either of them may perturb the active conformation for receptor binding and virus entry into a susceptible form. Future studies should further clarify the biological role of the envelope glycoproteins in the normal life cycle of HCV.
ACKNOWLEDGMENTS
We gratefully acknowledge the interest and constructive criticisms of Robert B. Belshe during the entire course of this study. We thank Mark Buller, Michael Houghton, Charles M. Rice, and John K. Rose for providing research materials, Ranga V. Srinivas and Michael A. Whitt for helpful suggestions, and SuzAnn Price for preparation of the manuscript.
This research was supported by internal funding from Saint Louis University and AI-45250 from the NIAID.
REFERENCES
- 1.Alter H J, Purcell R H, Shih J W, Melpolder J C, Houghton M, Choo Q L, Kuo G. Detection of antibody to hepatitis C virus in prospectively followed transfusion recipients with acute and chronic non-A, non-B hepatitis. N Engl J Med. 1989;321:1494–1500. doi: 10.1056/NEJM198911303212202. [DOI] [PubMed] [Google Scholar]
- 1a.Beach, M. (Centers for Disease Control and Prevention, Atlanta, Ga.). Personal communication.
- 2.Bertolini L, Iacovacci S, Ponzetto A, Battaglia M, Carloni G. The human bone-marrow-derived B-cell line CE, susceptible to hepatitis C virus infection. Res Virol. 1993;144:281–285. doi: 10.1016/s0923-2516(06)80041-5. [DOI] [PubMed] [Google Scholar]
- 3.Bukh J, Purcell R H, Miller R H. At least 12 genotypes of hepatitis C virus predicted by sequence analysis of the putative E1 gene of isolates collected worldwide. Proc Natl Acad Sci USA. 1993;90:8234–8238. doi: 10.1073/pnas.90.17.8234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Choo Q L, Kuo G, Weiner A J, Overby L R, Bradley D W, Houghton M. Isolation of a cDNA clone derived from a blood-borne non-A, non-B viral hepatitis genome. Science. 1989;244:359–362. doi: 10.1126/science.2523562. [DOI] [PubMed] [Google Scholar]
- 5.Choo Q L, Kuo G, Ralston R, Weiner A, Chien D, Van Nest G, Han J, Berger K, Thudium K, Kuo C. Vaccination of chimpanzees against infection by the hepatitis C virus. Proc Natl Acad Sci USA. 1994;91:1294–1298. doi: 10.1073/pnas.91.4.1294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Choppin P W, Compans R W. Phenotypic mixing of envelope proteins of the parainfluenza virus SV5 and vesicular stomatitis virus. J Virol. 1970;5:609–616. doi: 10.1128/jvi.5.5.609-616.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Clapham P, Nagy K, Weiss R A. Pseudotypes of human T-cell leukemia virus types 1 and 2: neutralization by patients’ sera. Proc Natl Acad Sci USA. 1984;81:2886–2889. doi: 10.1073/pnas.81.9.2886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Clemens J M, Taskar S, Chau K, Vallari D, Shih J W, Alter H J, Schleicher J B, Mimms L T. IgM antibody response in acute hepatitis C viral infection. Blood. 1992;79:169–172. [PubMed] [Google Scholar]
- 9.Dalgleish A G, Beverley P C L, Clapham P R, Crawford D H, Greaves M F, Weiss R A. The CD4 (T4) antigen is an essential component of the receptor for the AIDS retrovirus. Nature. 1984;312:763–767. doi: 10.1038/312763a0. [DOI] [PubMed] [Google Scholar]
- 10.Darcel C L Q, Kozub G C. The effect of kaolin adsorption of serum of the virus neutralization and enzyme-linked immunosorbent assays of antibody to bovine herpes virus 1. J Biol Stand. 1984;12:87–92. doi: 10.1016/s0092-1157(84)80024-4. [DOI] [PubMed] [Google Scholar]
- 11.Deleersnyder V, Pillez A, Wychowski C, Blight K, Xu J, Hahn Y S, Rice C M, Dubuisson J. Formation of native hepatitis C virus glycoprotein complexes. J Virol. 1997;71:697–704. doi: 10.1128/jvi.71.1.697-704.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Doms R W, Keller D S, Helenius A, Balch W E. Role for adenosine triphosphate in regulating the assembly and transport of vesicular stomatitis virus G protein trimers. J Cell Biol. 1987;105:1957–1969. doi: 10.1083/jcb.105.5.1957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dubuisson J, Hsu H H, Cheung R C, Greenberg H B, Russell D G, Rice C M. Formation and intracellular localization of hepatitis C virus envelope glycoprotein complexes expressed by recombinant vaccinia and Sindbis viruses. J Virol. 1994;68:6147–6160. doi: 10.1128/jvi.68.10.6147-6160.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Farci P, Alter H J, Wong D C, Miller R H, Govindarajan S, Engle R, Shapiro M, Purcell R H. Prevention of hepatitis C virus infection in chimpanzees after antibody-mediated in vitro neutralization. Proc Natl Acad Sci USA. 1994;91:7792–7796. doi: 10.1073/pnas.91.16.7792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Farci P, Shimoda A, Wong D, Cabezon T, DeGioannis D, Strazzera A, Shimizu Y, Shapiro M, Alter H J, Purcell R H. Prevention of hepatitis C virus infection in chimpanzees by hyperimmune serum against the hypervariable region 1 of the envelope 2 protein. Proc Natl Acad Sci USA. 1996;93:15394–15399. doi: 10.1073/pnas.93.26.15394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Fuerst T R, Niles E G, Studier F W, Moss B. Eukaryotic transient-expression system based on recombinant vaccinia virus that synthesized bacteriophage T7 RNA polymerase. Proc Natl Acad Sci USA. 1986;83:8122–8126. doi: 10.1073/pnas.83.21.8122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gallione C J, Rose J K. A single amino acid substitution in a hydrophobic domain causes temperature-sensitive cell-surface transport of a mutant viral glycoprotein. J Virol. 1985;54:374–382. doi: 10.1128/jvi.54.2.374-382.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Grakoui A, Wychowski C, Lin C, Feinstone S M, Rice C M. Expression and identification of hepatitis C virus polyprotein cleavage products. J Virol. 1993;67:1385–1395. doi: 10.1128/jvi.67.3.1385-1395.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hijikata M, Kato N, Ootsuyama Y, Nakagawa M, Shimotohno K. Gene mapping of the putative structural region of the hepatitis C virus genome by in vitro processing analysis. Proc Natl Acad Sci USA. 1991;88:5547–5551. doi: 10.1073/pnas.88.13.5547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hu X H, Ray R, Compans R W. Functional interactions between the fusion protein and hemagglutinin-neuraminidase of human parainfluenza viruses. J Virol. 1992;66:1528–1534. doi: 10.1128/jvi.66.3.1528-1534.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20a.Hulst M M, Moormann R J M. Inhibition of pestivirus infection in cell culture by envelope proteins Erns and E2 of classical swine fever virus: Erns and E2 interact with different receptors. J Gen Virol. 1997;78:2779–2787. doi: 10.1099/0022-1317-78-11-2779. [DOI] [PubMed] [Google Scholar]
- 21.Hutchinson L, Roop-Beauchamp C, Johnson D C. Herpes simplex virus glycoprotein K is known to influence fusion of infected cells, yet is not on the cell surface. J Virol. 1995;69:4556–4563. doi: 10.1128/jvi.69.7.4556-4563.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kato N, Ootsuyama Y, Ohkoshi S, Nakazawa T, Sekiya H, Hijikata M, Shimotohno K. Characterization of hypervariable regions in the putative envelope protein of hepatitis C virus. Biochem Biophys Res Commun. 1992;189:119–127. doi: 10.1016/0006-291x(92)91533-v. [DOI] [PubMed] [Google Scholar]
- 23.Kato N, Sekiya H, Ootsuyama Y, Nakazawa T, Hijikata M, Ohkoshi S, Shimotohno K. Humoral immune response to hypervariable region 1 of the putative envelope glycoprotein (gp70) of hepatitis C virus. J Virol. 1993;67:3923–3930. doi: 10.1128/jvi.67.7.3923-3930.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kato N, Ootsuyama Y, Sekiya H, Ohkeshi S, Nakazawa T, Hijikata M. Genetic drift in hypervariable region 1 of the viral genome in persistent hepatitis C virus infection. J Virol. 1994;68:4776–4784. doi: 10.1128/jvi.68.8.4776-4784.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Khanna A, Ray R. Hepatitis C virus core protein: synthesis, affinity purification, and immunoreactivity with infected human sera. Gene. 1995;153:185–189. doi: 10.1016/0378-1119(94)00782-n. [DOI] [PubMed] [Google Scholar]
- 26.Kojima M, Osuga T, Tsuda F, Tanaka T, Okamoto H. Influence of antibodies to the hypervariable region E2/NS1 glycoprotein on the selective replication of hepatitis C virus in chimpanzees. Virology. 1994;204:665–672. doi: 10.1006/viro.1994.1582. [DOI] [PubMed] [Google Scholar]
- 27.Kolykhalov, A. A., E. V. Agapov, and C. M. Rice. Specificity of the hepatitis C virus NS3 serine proteinase: effects of substitution at the 3/4A, 4A/4B, 4B/5A, and 5A/5B cleavage sizes on polyprotein processing. Submitted for publication. [DOI] [PMC free article] [PubMed]
- 28.Koziel M J, Dudley D, Afdhal N, Choo Q L, Houghton M, Ralston R, Walker B D. Hepatitis C virus (HCV)-specific cytotoxic T lymphocytes recognize epitopes in the core and envelope proteins of HCV. J Virol. 1993;67:7522–7532. doi: 10.1128/jvi.67.12.7522-7532.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Koziel M J, Dudley D, Wong J T, Dienstag J, Houghton M, Ralston R, Walker B D. Intrahepatic cytotoxic T lymphocytes specific for hepatitis C virus in persons with chronic hepatitis. J Immunol. 1992;149:3339–3344. [PubMed] [Google Scholar]
- 30.Lamb R A. Paramyxovirus fusion: a hypothesis for changes. Virology. 1993;197:1–11. doi: 10.1006/viro.1993.1561. [DOI] [PubMed] [Google Scholar]
- 31.Lanford R E, Notvall L, Chavez D, White R, Frenzel G, Simonsen C, Kim J. Analysis of hepatitis C virus capsid, E1, and E2/NS1 proteins expressed in insect cells. Virology. 1993;197:225–235. doi: 10.1006/viro.1993.1583. [DOI] [PubMed] [Google Scholar]
- 32.Lanford R E, Sureau C, Jacob J R, White R, Fuerst T R. Demonstration of in vitro infection of chimpanzee hepatocytes with hepatitis C virus using strand-specific RT/PCR. Virology. 1994;202:606–614. doi: 10.1006/viro.1994.1381. [DOI] [PubMed] [Google Scholar]
- 33.Lin C, Lindenbach B D, Pragai B M, McCourt D W, Rice C M. Processing in the hepatitis C virus E2-NS2 region: identification of p7 and two distinct E2-specific products with different C termini. J Virol. 1994;68:5063–5073. doi: 10.1128/jvi.68.8.5063-5073.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Martell M, Esteban J I, Quer J, Gensca J, Weiner A, Esteban R, Guardia J, Gomez J. Hepatitis C virus (HCV) circulates as a population of different but closely related genomes: quasispecies nature of HCV genome distribution. J Virol. 1992;66:3225–3229. doi: 10.1128/jvi.66.5.3225-3229.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Martell M, Esteban J I, Quer J, Vargas V, Esteban R, Guardia J, Gomez J. Dynamic behavior of hepatitis C virus quasispecies in patients undergoing orthotopic liver transplantation. J Virol. 1994;68:3425–3436. doi: 10.1128/jvi.68.5.3425-3436.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.McSharry J J, Compans R W, Choppin P W. Proteins of vesicular stomatitis virus and of phenotypically mixed vesicular stomatitis virus-simian virus 5 virions. J Virol. 1971;8:722–729. doi: 10.1128/jvi.8.5.722-729.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Miyamura T, McHutchinson J, Houghton M. Evidence for immune selection of hepatitis C virus (HCV) putative envelope glycoprotein variants: potential role in chronic HCV infections. Proc Natl Acad Sci USA. 1992;89:3468–3472. doi: 10.1073/pnas.89.8.3468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Mizushima H, Hijikata M, Asabe S-I, Hirota M, Kimura K, Shimotohno K. Two hepatitis C virus glycoprotein E2 products with different C termini. J Virol. 1994;68:6215–6222. doi: 10.1128/jvi.68.10.6215-6222.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Nelson D R, Marousis C G, Davis G L, Rice C M, Wong J, Houghton M, Lau J Y. The role of hepatitis C virus-specific cytotoxic-lymphocytes in chronic hepatitis C. J Immunol. 1997;158:1473–1481. [PubMed] [Google Scholar]
- 40.Nissen E, Hohne M, Schreier E. In vitro replication of hepatitis C virus in a human lymphoid cell line (H9) J Hepatol. 1993;20:437. doi: 10.1016/s0168-8278(94)80023-5. [DOI] [PubMed] [Google Scholar]
- 41.Okamoto H, Kojima M, Okada S, Yoshizawa H, Iizuka H, Tanaka T, Muchmore E E, Peterson D A, Ito Y, Mishiro S. Genetic drift of hepatitis C virus during an 8.2-year infection in a chimpanzee: variability and stability. Virology. 1992;190:894–899. doi: 10.1016/0042-6822(92)90933-g. [DOI] [PubMed] [Google Scholar]
- 42.Owens R J, Rose J K. Cytoplasmic domain requirement for incorporation of a foreign envelope protein into vesicular stomatitis virus. J Virol. 1993;67:360–365. doi: 10.1128/jvi.67.1.360-365.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42a.Owens R J, Burke C, Rose J K. Mutations in the membrane-spanning domain of the human immunodeficiency virus envelope glycoprotein that affect fusion activity. J Virol. 1994;68:570–574. doi: 10.1128/jvi.68.1.570-574.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Palumbo G J, Pickup D, Fredrickson T N, McIntyre L J, Buller R M L. Inhibition of an inflammatory response is mediated by a 38-kDa protein of cowpox virus. Virology. 1989;172:262–273. doi: 10.1016/0042-6822(89)90128-1. [DOI] [PubMed] [Google Scholar]
- 44.Prince A M, Brotman B, Huima T, Pascual D, Jaffery M, Inchauspe G. Immunity in hepatitis C infection. J Infect Dis. 1992;165:438–443. doi: 10.1093/infdis/165.3.438. [DOI] [PubMed] [Google Scholar]
- 45.Ralston R, Thudium K, Berger K, Kuo C, Gervase B, Hall J, Selby M, Kuo G, Houghton M, Choo Q. Characterization of hepatitis C virus envelope glycoprotein complexes expressed by recombinant vaccinia viruses. J Virol. 1993;67:6753–6761. doi: 10.1128/jvi.67.11.6753-6761.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ray R, Khanna A, Lagging L M, Meyer K, Choo Q L, Ralston R, Houghton M, Becherer P R. Peptide immunogen mimicry of putative E1 glycoprotein specific epitopes in hepatitis C virus. J Virol. 1994;68:4420–4426. doi: 10.1128/jvi.68.7.4420-4426.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ray R B, Lagging L M, Meyer K, Steele R, Ray R. Transcriptional regulation of cellular and viral promoters by the hepatitis C virus core protein. Virus Res. 1995;27:209–220. doi: 10.1016/0168-1702(95)00034-n. [DOI] [PubMed] [Google Scholar]
- 48.Rosa D, Capagnoli S, Moretto C, Guenzi E, Cousens L, Chin M, Dong C, Weiner A J, Lau J Y N, Choo Q-L, Chien D, Pileri P, Houghton M, Abrignani S. A quantitative test to estimate neutralizing antibodies to the hepatitis C virus: cytofluorimetric assessment of envelope glycoprotein 2 binding to target cells. Proc Natl Acad Sci USA. 1996;93:1759–1763. doi: 10.1073/pnas.93.5.1759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Saito I, Miyamura T, Ohbayashi A, Harada H, Katayama T, Kikuchi S, Watanabe Y, Koi S, Onji M, Choo Q-L, Houghton M, Kuo G. Hepatitis C virus infection associated with the development of hepatocellular carcinoma. Proc Natl Acad Sci USA. 1990;87:6547–6549. doi: 10.1073/pnas.87.17.6547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Schnell M J, Mebatison T, Conzelmann K. Infectious rabies viruses from clone cDNA. EMBO J. 1994;13:4195–4203. doi: 10.1002/j.1460-2075.1994.tb06739.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Schnell M J, Buonocore L, Kretzschmar E, Johnson E, Rose J K. Foreign glycoproteins expressed from recombinant vesicular stomatitis viruses are incorporated efficiently into virus particles. Proc Natl Acad Sci USA. 1996;93:11359–11365. doi: 10.1073/pnas.93.21.11359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Schnitzer T J, Dickson C, Weiss R A. Morphological and biochemical characterization of viral particles produced by the tsO45 mutant of vesicular stomatitis virus at restrictive temperature. J Virol. 1979;29:185–195. doi: 10.1128/jvi.29.1.185-195.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Selby M J, Glazer E, Masiarz F, Houghton M. Complex processing and protein: protein interactions in the E2:NS2 region of HCV. Virology. 1994;204:114–122. doi: 10.1006/viro.1994.1515. [DOI] [PubMed] [Google Scholar]
- 54.Shimizu Y, Purcell R H, Yoshikura H. Correlation between the infectivity of hepatitis C virus in vivo and its infectivity in vitro. Proc Natl Acad Sci USA. 1993;90:6037–6041. doi: 10.1073/pnas.90.13.6037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Shimizu Y K, Iwamoto A, Minako H, Purcell R H, Yoshikura H. Evidence for in vitro replication of hepatitis C virus genome in a human T-cell line. Proc Natl Acad Sci USA. 1992;89:5477–5481. doi: 10.1073/pnas.89.12.5477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Shimuzu Y K, Igarashi H, Kiyohara T, Cabezon T, Farci P, Purcell R H, Yoshikura H. A hyperimmune serum against a synthetic peptide corresponding to the hypervariable region 1 of hepatitis C virus can prevent viral infection in cell cultures. Virology. 1996;223:409–412. doi: 10.1006/viro.1996.0497. [DOI] [PubMed] [Google Scholar]
- 57.Spaete R R, Alexander D, Rugroden M E, Choo Q L, Berger K, Crawford K, Kuo C, Leng S, Lee C, Ralston R, Thudium K, Tung J W, Kuo G, Houghton M. Characterization of the hepatitis C virus E2/NS1 gene product expressed in mammalian cells. Virology. 1992;188:819–830. doi: 10.1016/0042-6822(92)90537-y. [DOI] [PubMed] [Google Scholar]
- 58.Spear P G. Entry of alphaherpesviruses into cells. Semin Virol. 1993;4:167–180. [Google Scholar]
- 59.Strauss J H, Strauss E G. The alphaviruses: gene expression, replication, and evolution. Microbiol Rev. 1994;58:491–562. doi: 10.1128/mr.58.3.491-562.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Weiner A J, Geysen H M, Christopherson C, Hall J E, Mason T J, Saracco G, Bonino F, Crawford K, Marion C D, Crawford K A, Brunetto M, Barr P J, Miyamura T, McHutchinson J, Houghton M. Evidence for immune selection of hepatitis C virus (HCV) putative envelope glycoprotein variants: potential role in chronic HCV infections. Proc Natl Acad Sci USA. 1992;89:3468–3472. doi: 10.1073/pnas.89.8.3468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Whitt M A, Chong L, Rose J K. Glycoprotein cytoplasmic domain sequences required for rescue of a vesicular stomatitis virus glycoprotein mutant. J Virol. 1989;63:3569–3578. doi: 10.1128/jvi.63.9.3569-3578.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Yi M, Nakamoto Y, Kaneko S, Yamashita T, Murakami S. Delineation of regions important for heteromeric association of hepatitis C virus E1 and E2. Virology. 1997;231:119–129. doi: 10.1006/viro.1997.8516. [DOI] [PubMed] [Google Scholar]
- 63.Zavada J. The pseudotypic paradox. J Gen Virol. 1982;63:15–24. doi: 10.1099/0022-1317-63-1-15. [DOI] [PubMed] [Google Scholar]
- 64.Zibert A, Eckart S, Roggendorf M. Antibodies in human sera specific to hypervariable region 1 of hepatitis C virus can block viral attachment. Virology. 1995;208:653–661. doi: 10.1006/viro.1995.1196. [DOI] [PubMed] [Google Scholar]
- 65.Zibert A, Kraas W, Meisel H, Jung G, Roggendorf M. Epitope mapping of antibodies directed against hypervariable region 1 in acute self-limiting and chronic infections due to hepatitis C virus. J Virol. 1997;71:4123–4127. doi: 10.1128/jvi.71.5.4123-4127.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]