Dear Editor,
In thin melanoma — specifically, melanomas with a thickness of 1.0 mm or less — only 1–3% of cases advance to fatality.1,2 Precisely pinpointing high-risk lesions in such cases is a difficult clinical task. Several prognostic indicators, such as mitotic rate (MR), tumor ulceration, anatomical location, lymphovascular invasion, Breslow thickness (BT), and micrometastatic sentinel lymph nodes (SLN), have been suggested.3 Following approval from our Institutional Review Board, we sought to evaluate these risk indicators by selecting a subset of 196 thin melanoma patients from a larger cohort previously published.4 These patients received their diagnosis and treatment at the Mayo Clinic between the years 2004 and 2018 and underwent SLN biopsies. Inclusion in this study required patients to fulfill at least one of the following criteria: a BT ranging from 0.75 to 0.99 mm paired with a minimum of one risk factor or a BT between 0.50 and 0.74 mm accompanied by at least two risk factors. These risk factors, derived from the Mayo Clinic’s institutional practice guidelines, encompassed tumor ulceration, an MR exceeding zero, and an age below 40 at the time of diagnosis.
To assess the prognostic accuracy of the highlighted risk factors, we specifically focused on a five-year relapse-free survival (RFS) as a marker for clinical aggression in thin melanoma. Out of 196 patients with thin melanoma (Table 1), 10% exhibited a positive SLN. 13/20 of these positive SLN instances (65%) displayed minimal tumor burden, identified by cell clusters < 0.1 mm or individual tumor cells. The median duration of follow-up was 7.1 years (IQR: 4.8–10.2 years). In the five years post-SLN biopsy, 11 relapses were recorded: two local, one in-transit, four regional, and four distant. Factors such as MR (≥ 2/mm2 vs. < 2/mm2), the presence of tumor ulceration, anatomic location (head/neck vs. other locations), and BT (≥ 0.9/mm vs. < 0.9/mm) were not found to significantly impact the five-year RFS by Cox proportional hazards model. However, the presence of micrometastatic SLN (as opposed to nonmetastatic SLN) was associated with a reduced five year RFS (hazard ratio [HR]: 4.2; 95% confidence intervals [CI] = 1.1–15.7; p = 0.035, Wald test). To elaborate, patients with a negative SLN showed a five-year RFS rate of 95%, in contrast to an 80% rate in patients with a positive SLN (p = 0.022, log-rank test; Fig. 1). This notable correlation between SLN metastasis and RFS diminished over time as more SLN-negative patients reported relapses. By the end of 7 years, the association between SLN metastasis and reduced RFS was no longer statistically significant (HR: 2.7; 95% CI = 0.8–9.6; p = 0.12, Wald test). Results were constrained by a limited sample size.
Table 1.
Demographic and clinicopathologic characteristics of the study cohort
| SLNB Results |
P valuea | |||
|---|---|---|---|---|
| Patient Characteristic | All patient (n = 196) | Negative (n = 176) | Positive (n = 20) | |
|
| ||||
| Sex, No. (%) | 0.41 | |||
| Female | 81 (41.3%) | 71 (40.3%) | 10 (50.0%) | |
| Male | 115 (58.7%) | 105 (59.6%) | 10 (50.0%) | |
| Age at diagnosis, years, median (IQR) | 59 (45–69) | 60 (48–69) | 48 (36–63) | 0.035 |
| Breslow thickness, mm, median (IQR) | 0.87 (0.80–0.95) | 0.86 (0.80–0.95) | 0.90 (0.85–0.96) | 0.22 |
| Histologic type, No. (%) | - | |||
| Superficial spreading | 146 (74.5%) | 129 (73.3%) | 17 (85.0%) | |
| Nodular | 6 (20.2%) | 6 (3.4%) | 0 | |
| Desmoplastic | 3 (1.5%) | 3 (1.7%) | 0 | |
| Lentigo maligna | 11 (5.6%) | 11 (6.3%) | 0 | |
| Acral lentiginous | 3 (1.5%) | 2 (1.1%) | 1 (5.0%) | |
| Other | 27 (13.8%) | 25 (14.2%) | 2 (10.0%) | |
| T-Category (AJCC 8), No. (%) | 0.09 | |||
| T1a | 38 (19.4%) | 37 (21.0%) | 1 (5.0%) | |
| T1b | 158 (80.6%) | 139 (79.0%) | 19 (95.0%) | |
Comparisons between SLN-negative and SLN-positive patients were conducted using the chi-square test for categorical variables (gender and T-category) and the two-sample t-test for continuous variables (patient age and Breslow thickness).
Fig 1. Five-year relapse-free survival: T1 melanoma.
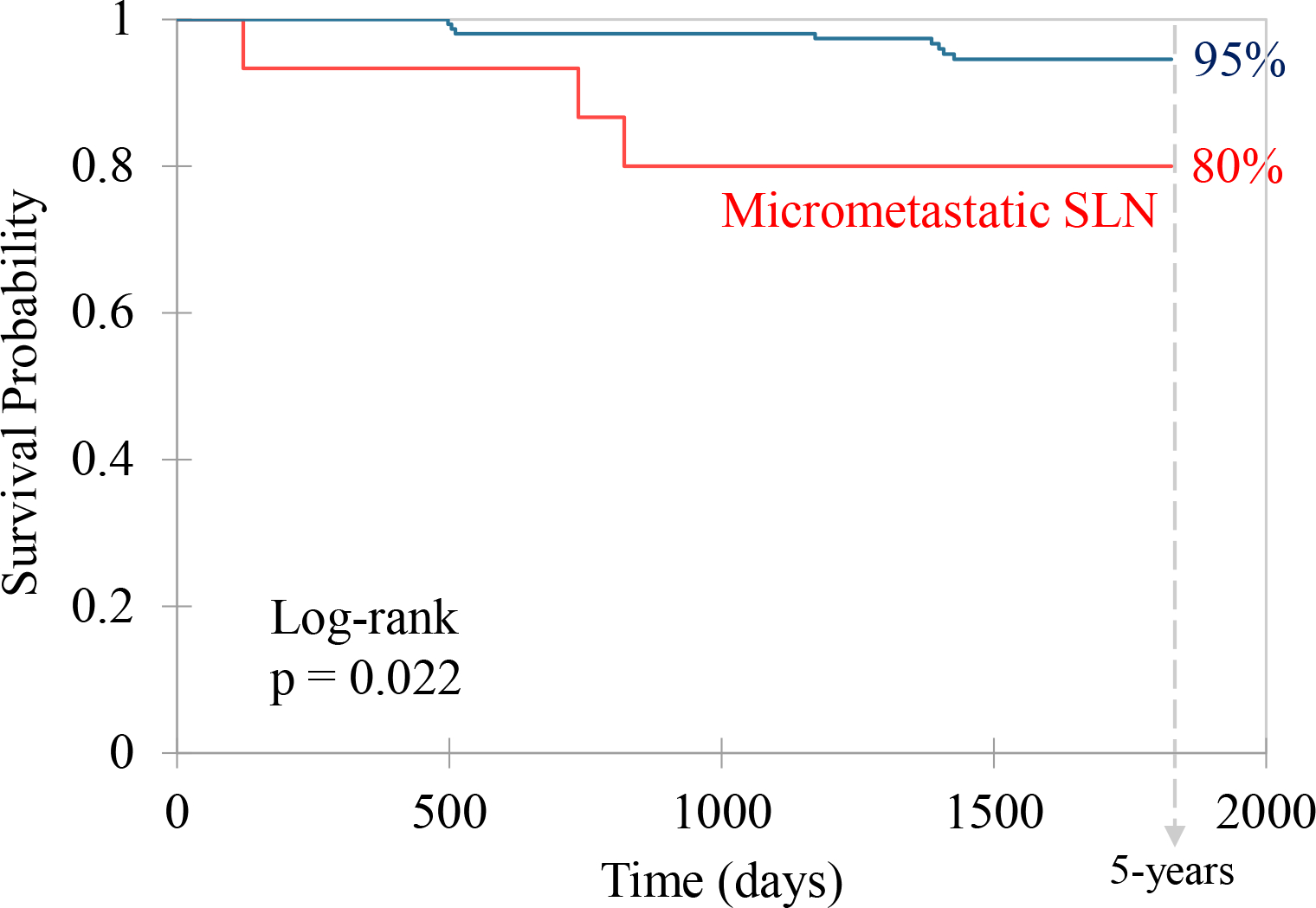
Kaplan-Meier analysis of a cohort of 196 T1 melanoma patients, stratified by SLN biopsy status: micrometastatic (red) vs. non-metastatic (blue). Survival endpoint was relapse-free survival (RFS).
Leading surgical oncologists, including Boland and Gershenwald, have underscored the pivotal role of SLN status as a prognostic indicator for these patients.5 The significance given to a positive SLN in melanoma risk stratification aligns with prevailing clinical guidelines. Our data suggests that the SLN status identifies thin melanoma patients with an elevated risk of relapse within five years. Given these findings, patients with positive SLN may consider systemic adjuvant therapy to mitigate recurrence risks. As such, SLN biopsy offers therapeutic efficacy for individuals with thin melanoma, and it is prudent — in accordance with clinical guidelines — to discuss and consider SLN biopsy for these patients. However, the difficulty in predicting thin melanoma recurrences beyond five years post-diagnosis, even when using sophisticated methods such as SLN biopsy, deserves attention. Consistent surveillance is essential for patients with thin melanoma and a negative SLN result, extending beyond the initial five years post-diagnosis to detect any potential relapses. We speculate that in the future, the capacity to frequently test for recurrences using circulating tumor DNA in plasma may diminish the reliance on prognostic tools predicting long-term outcomes.
Footnotes
Conflict of interest: None
References
- 1.Quattrocchi E, Meves ES, Meves A. CP-GEP (Merlin) gene expression profiling: can my melanoma patient forgo sentinel lymph node biopsy? Ital J Dermatol Venerol 2023; 158: 292–301. [DOI] [PubMed] [Google Scholar]
- 2.Whiteman DC, Baade PD, Olsen CM. More People Die from Thin Melanomas (≤1 mm) than from Thick Melanomas (>4 mm) in Queensland, Australia. J Invest Dermatol 2015; 135: 1190–1193. [DOI] [PubMed] [Google Scholar]
- 3.Sadurní MB, Meves A. Breslow thickness 2.0: Why gene expression profiling is a step toward better patient selection for sentinel lymph node biopsies. Mod Pathol 2022; 35: 1509–1514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bellomo D, Arias-Mejias SM, Ramana C, et al. Model Combining Tumor Molecular and Clinicopathologic Risk Factors Predicts Sentinel Lymph Node Metastasis in Primary Cutaneous Melanoma. JCO Precis Oncol 2020; 4: 319–334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Boland G, Caudle A, Warneke C, et al. Predictors of survival in contemporary era patients with thin melanoma who underwent sentinel node biopsy (Abstract 53). Ann Surg Oncol 2014; 21: S23–S24. [Google Scholar]


