Abstract
Older adults with Type II Diabetes (T2D) are at risk of developing nerve disorders that result in functional impairment. Most work in proprioceptive dysfunction in older adults with T2D has focused on functional deficits of the lower limb. The purpose of this study was to examine proprioceptive effects of T2D on the upper limb in older adults. Kinematic performance of a reach-to-pinch action toward a virtual target was assessed in a T2D group (60+ years old with T2D) and a healthy age- and sex-matched control group. Tactile and vibratory thresholds did not differ between T2D and controls. Task accuracy via mean pinch location was significantly worse for persons with T2D (pwT2D) with differences in wrist extension/flexion (ex/fl), wrist abduction/adduction (ab/ad), 1st carpometacarpal (CMC) ab/ad, 2nd metacarpophalangeal (MCP2) ex/fl, MCP2 ab/ad, and digit 2 and hand transport trajectories. Group differences persisted with consideration of body mass index; sex differences in task accuracy emerged. Findings indicate that proprioception of the upper extremity is altered in pwT2D such that they exhibit a unique aperture position and aiming strategy during a reach-to-pinch action. These findings characterize functional sensorimotor impairment of the upper limb in pwT2D with respect to workspaces without visual or tactile feedback.
Keywords: aging processes, kinematics, virtual environments, tactile, motor control
Introduction
As of 2014, the World Health Organization estimated 422 million people with diabetes, of which, approximately 90% are classified as having Type II Diabetes (T2D) (WHO, 2017). One in every four older adults has T2D (Centers for Disease Control and Prevention, 2020). Health consequences of T2D can include cardiovascular disease (CVD), peripheral neuropathy (PN), amputation, blindness, and kidney failure. It is well-known T2D is associated with diabetic PN, resulting in sensorimotor dysfunction of the hands and feet—most notably in older adults (Mold et al., 2004). Previous studies have suggested that motor deficits associated with T2D can be attributed to tactile impairment caused by PN (Orlando et al., 2016); however, motor impairments may also be due to sensorimotor dysfunction not specific to PN (Ochoa et al., 2016). Sensorimotor deficits caused by T2D may also affect proprioception and function of the limbs during routine tasks (Ochoa et al., 2016).
Postural stability studies of older adults with T2D and PN have found these individuals to have impaired balance and increased fall risk attributable to T2D (Hong et al., 1997; Lord et al., 1993; Simmons et al., 1997; Uccioli, 1995). The negative impact of T2D on postural performance is not strictly due to T2D alone but also by the negative effects of age on the sensorimotor system (Gorniak, Lu, et al., 2019; Hewston & Deshpande, 2016; Lord et al., 1993; Mustapa et al., 2016; Pai et al., 1997; Shaffer & Harrison, 2007; Simoneau et al., 1996; R. W. M. van Deursen & Simoneau, 1999). Proprioception, a key component of the sensorimotor system, plays a crucial role in postural stability. Because the loss of tactile sensation is more easily measured during upright stance than the functioning of proprioceptive receptors, many postural stability studies are not able to assess the effects of T2D directly on proprioception. Instead, a common method to evaluate proprioception is by using joint position perception and matching tasks in which visual feedback is not provided. Such studies have found impaired proprioception in the lower limb associated with aging, T2D, and related PN. Specifically, ankle movement perception has been reported to be significantly worse in persons with T2D (pwT2D) (Lord et al., 1993; Simoneau et al., 1996; R. W. van Deursen et al., 1998). Muscle spindles–key proprioception receptors–produce a kinesthetic illusion of joint movement and muscle lengthening when vibration is applied to the muscle tendon. In pwT2D, this kinesthetic illusion is reduced (Simoneau et al., 1996; R. W. van Deursen et al., 1998; R. W. M. van Deursen et al., 2001). The findings from these studies suggest that both age and T2D degrade proprioceptive ability in the lower limbs.
While sensorimotor dysfunction associated with T2D in the lower extremities has been well documented, few studies have investigated the effects of both T2D and age on the upper extremity. We know that the upper extremity is somewhat affected in T2D. Studies have reported impaired tactile sensation, decreased grip, and reduced hand strength associated with T2D (Cetinus et al., 2005; Gorniak et al., 2014; Ochoa et al., 2016; Ochoa & Gorniak, 2014; Ramji et al., 2007; Sayer et al., 2005)—all of which impact functional hand use. Tactile impairment due to T2D was previously assumed to be a main contributor to impairment in maximal force production in pwT2D. Recently, it was revealed that tactile dysfunction alone is not responsible for T2D-associated functional declines in the upper extremity (Ochoa et al., 2016). In Ochoa et al., 2016, evidence of impaired proprioception was also reported in pwT2D; however, it was not further explored. In order to investigate this further, our team sought to utilize a task to test proprioception of the upper extremity in a task that was more linked to functions of daily living (e.g., reaching and grasping a virtual target) instead of simple single joint matching. Given the systemic nature of T2D, it is likely that proprioception deficits co-occur at more than one joint during activities of daily living, contributing together to functional deficits reported in motor function in pwT2D (Gorniak et al., 2014; Gorniak, Ray, et al., 2019; Gorniak, Wagner, et al., 2020; Ochoa & Gorniak, 2014). A protocol to evaluate proprioception in this manner in patient populations was pioneered by Nataraj and colleagues (Nataraj et al., 2014).
The goal of the current study was to evaluate proprioceptive changes in the upper limb in older adult pwT2D through kinematic analysis of a reach-to-pinch action to a visually presented virtual target. In such a task, only proprioceptive feedback guides the action. Evidence from prior observations in pwT2D provide support for impaired proprioception contributing to functional motor deficits in the upper extremity in T2D. In the current study, pwT2D and age- and sex-matched controls performed a reach-to-pinch task relying on only proprioceptive feedback. Consistent with observations in (Ochoa et al., 2016), we expected to see the T2D group exhibit increased movement variability of the digits, hand, and arm during the reach-to-pinch task when compared to healthy age- and sex-matched controls (Hypothesis 1). Additionally, we expected to see reductions in accuracy and precision relative to the target location in pwT2D as compared to controls (Hypothesis 2). No specific hypotheses regarding changes in proprioception with disease state were developed a priori, as multiple mechanistic pathways have been suggested within the evidence base (e.g., high glycated hemoglobin (HbA1c), hypertension, etc.).
Materials and Methods
Participants
Thirteen (13) older adults (7M, 6F) with a history of established T2D and 12 age- and sex-matched control participants (Control) (6M, 6F) were recruited for this study, see Table 1 for demographics. Inclusion criteria for both groups was: age 60 years and above, history of moderate range blood pressure (90/60 – 160/100 mmHg), and right-handed (laterality quotient (LQ) > 40, assessed with the Edinburgh Handedness Inventory). Exclusion criteria for both groups included: diagnosis of Type 1 Diabetes, diagnosis of T2D prior to age 18, history of uncontrolled hypertension, history of limb amputation, chemotherapy, or neurological diseases (Alzheimer’ Disease, Dementia, Huntington’s Disease, Traumatic Brain Injury, Multiple Sclerosis, Parkinson’s Disease, Paraproteinemic Demyelinating Neuropathy (PDN), Muscular Dystrophy, Carpal Tunnel Syndrome, Charcot-Marie-Tooth Disorder, and other hereditary or compressional neuropathies), and pain in the extremities that limits activities of daily living. This study was approved by the Institutional Review Board (IRB) at the University of Houston in accordance with the Declaration of Helsinki. All subjects provided written informed consent.
Table 1:
Values are mean ± SD or count. BMI = body mass index, DIA = diastole, HbA1c = glycated hemoglobin, HDL = high-density lipoprotein, LDL = low-density lipoprotein, LQ = laterality quotient, PN = peripheral neuropathy, SYS = systole, TC = total cholesterol
| Study participant characteristics and health data | ||
|---|---|---|
|
| ||
| pwT2D | Controls | |
| N (Males, Females) | 13 (6, 7) | 12 (6, 6) |
| Age (y) | 69.6 ± 6.8 | 68.1 ± 4.5 |
| Height (m) | 1.655 ± 0.061 | 1.717 ± 0.099 |
| Mass (kg) | 92.3 ± 16.5 | 89.4 ± 28.7 |
| BMI (kg/m2) | 33.7 ± 7.2 | 30.3 ± 11.3 |
| HbA1c (%) | 6.4 ± 0.7 | 5.6 ± 0.5 |
| LQ | 88 ± 12 | 92 ± 12 |
| SYS (mmHg) | 133 ± 22 | 139 ± 15 |
| DIA (mmHg) | 70 ± 11 | 78 ± 11 |
| PN | 4 | 0 |
| Disease duration (months) | 104 ± 85 | - |
| TC (mg/dL) | 164 ± 27 | 194 ± 50 |
| HDL (mg/dL) | 55 ± 14 | 59 ± 9 |
| LDL (mg/dL) | 83 ± 18 | 105 ± 39 |
Health Status Information
Blood pressure, cholesterol, and HbA1c values were assessed for all study participants. Cholesterol and HbA1c were assessed using commercially available point of care evaluation kits (Cardiocheck+ and A1c Now+, PTS Diagnostics, Indianapolis, IN, USA). Blood pressure was measured using a commercially available wrist-worn device (Omron Intellisense 10 series Blood Pressure Monitor, Model BP785, Bannockburn, IL, USA). History of prediabetes diagnosis was determined via self-report. Peripheral neuropathy (PN) status was determined by either clinical examination or nerve conduction testing (per physician).
Tactile Evaluation
Tactile sensation of the dominant (right) hand was evaluated using the Semmes-Weinstein monofilament test. The Semmes-Weinstein monofilament test is an assessment of tactile function that uses monofilaments of various thickness and diameter to detect sensory loss of the distal extremities. Tactile sensation was tested at the tip of digit 1, digit 2, and digit 5, the hypothenar eminence, and the dorsum thenar of the right hand. Additional details regarding this procedure can be found in Gorniak, Ochoa, et al., 2020. Vibrotactile threshold of the dominant hand was evaluated using a biothesiometer device (BioMedical, Newbury, Ohio, USA). Vibrotactile threshold was evaluated at the same five sites used for monofilament testing.
Reach and Pinch Task
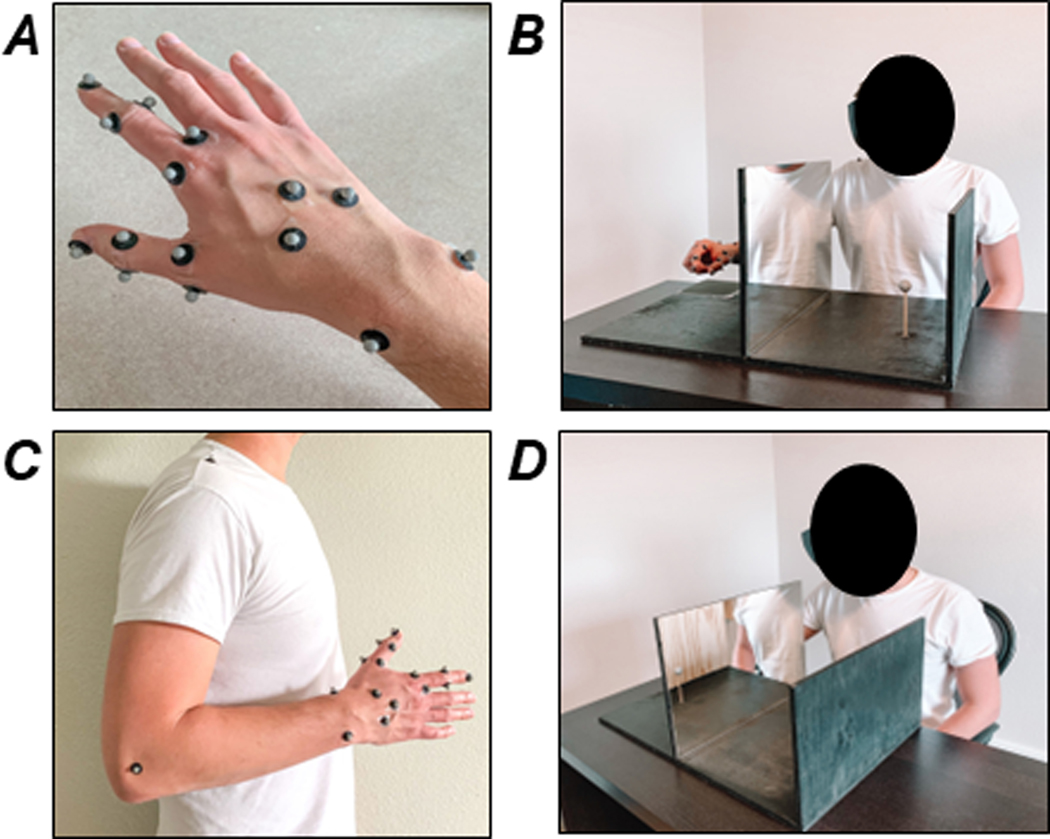
Retro-reflective markers were placed on the surface of the right hand and arm to acquire digit 1, digit 2, wrist, and elbow kinematic data. Marker placement was inspired by Nataraj et al. (2014; 2015). One marker each was placed on the very distal tip of the distal phalanx of digit 1 and the very distal tip of the distal phalanx of digit 2. Two markers were placed on each side of the 1st carpometacarpal (CMC), 1st metacarpophalangeal (MCP1), 1st interphalangeal (IP1), 2nd metacarpophalangeal (MCP2), 2nd proximal interphalangeal (PIP2), and 2nd distal interphalangeal (DIP2) joints. A marker cluster was placed on the second metacarpal as a local hand reference coordinate system (Wu et al., 2005). Markers were also placed on the right acromioclavicular joint, the lateral epicondyle, and the radial and ulnar styloid processes. See Fig. 1A and 1C for marker placement.
Figure 1:
A) Close up view of hand marker placement with 6.4 mm spherical reflective markers. One marker each was located at the very tip of the distal phalanx of digit 1 and the very tip of the distal phalanx of digit 2. Two markers were placed on each side of the CMC, MCP1, IP1, MCP2. PIP2, and DIP2 joints. A “T” shaped marker cluster on the second metacarpal was used as a local hand reference coordinate system. B) Front view of set up of the reach-to-pinch task. Subjects viewed a virtual target in a mirror while performing a reach-to-pinch movement with their hand. The maneuver is performed without visual feedback of the moving arm and hand. C) Marker placement of the right arm and hand. In addition to hand markers, markers are located at right acromioclavicular joint, the lateral epicondyle, and the radial and ulnar styloid processes. D) View of virtual target and mirror setup.
A VICON motion capture system was used to collect the three-dimensional positions of the retro-reflective markers at 100 Hz (VICON, Oxford Metrics, Oxford, UK). Joint angles trajectories were collected for: CMC, MCP1, IP1, MCP2, PIP2, DIP2, flexion/extension and abduction/adduction of the wrist, and flexion/extension of the elbow.
Subjects were asked to perform a reach-to-pinch task with digit 1 and digit 2 of their dominant (right) hand without visual feedback of the reaching hand but allowing for visualization of a spherical virtual target, as developed by Nataraj et al. (2014; 2015). While seated at a table, subjects sat to the left side of a high-resolution vertical mirror inserted into a custom made set up (Fig. 1B and 1D), as described in Nataraj et al., 2014. The mirror blocked subjects’ view of the right side of the table. On the left side of the center of the mirror was a target placed on a stand (2 cm diameter, 10 cm from the surface of the table, 20 cm horizontally from the mirror, 20 cm from the edge of the table). From their seated position, the subject was able to view the target in the mirror, creating a “virtual target” in the open space behind the mirror. Subjects were asked to treat the virtual target in the mirror as if it were a real target that he or she could grasp with their right hand. The subject’s reaching hand was located behind the mirror (the right side of the table set up), which prevented visual feedback. Additionally, the subject wore a right-side blinder to further prevent visual movement feedback of the upper arm during reaching.
An auditory metronome was used to control for movement speed across subjects. Subjects heard an auditory stimulus (tone) at a beat frequency of 1 Hz throughout the experiment protocol. For each trial, they were asked to begin movement on a tone and finish the maneuver by the following tone. Each trial lasted for approximately 2 seconds, with a few seconds rest between trials. Subjects performed a total of 30 consecutive reach-to-pinch trials. Digit thickness was measured using calipers for both digits 1 and 2. This thickness was then used as the radius for a spherical model of the digit-pad and aperture. Pinch contact between digits 1 and 2 was assumed to occur when inter-pad distance was below the threshold of 1 mm (Nataraj et al., 2014; Nataraj & Li, 2015). Mean endpoint pinch aperture and location for individual and mean T2D and Control subjects with respect to the virtual target location was calculated using a sphere, as in Nataraj et al., 2014. The center of the pinch sphere denotes the endpoint pinch location and the radius denotes the pinch precision (± standard deviation) about the endpoint.
Statistical Analysis
SPSS version 26.0 (SPSS IBM, New York, NY, USA) and Minitab 17 (Minitab LLC, State College, PA, USA) were used to perform statistical analyses. For each variable of interest, automatic linear modeling (ALM) was used to select significant covariates from health status data using forward stepwise selection in SPSS (Gorniak, Wagner, et al., 2020). ALM was utilized to reduce the potential for expectation biases that may occur when hand-selecting potential statistical models, as all covariates measured in this study could influence study outcomes (Gorniak et al., 2023; Gorniak, Ochoa, et al., 2020; Gorniak, Wagner, et al., 2020; Nataraj et al., 2014). Follow-up correlation analyses were performed for all ALM-identified significant covariates. Potential covariates included: HbA1c, blood pressure (systole and diastole), cholesterol (Total, HDL, LDL), duration of diagnosis (in months), PN status (via indicator variable), prediabetes status (via indicator variable), body mass index (BMI), age, LQ, and digit 1 and digit 2 finger size. For tactile evaluation, monofilament data was log-transformed due to non-linearity of the data, consistent with the approach taken in Gorniak, Ochoa, et al., 2020. Two-way analyses of covariance (ANCOVAs) were used to compare tactile thresholds and vibration perception thresholds of the hand between Groups (T2D and Control). Nerve (median, radial, ulnar nerves) was a within-subjects factor.
Similarly, kinematic data was analyzed using two-way ANCOVAs to compare between Groups. Data included mean (average) trajectory values for each joint of interest in extension/flexion and abduction/adduction, trajectory path length of the hand, digit 1, digit 2, mean aperture, pinch contact location, and precision as described in Nataraj et al., 2014. Variability of each trajectory was considered via the coefficient of variability (CV) computed for each participant. CV was chosen as movement variability generally increases proportionally with range of movement (Nataraj et al., 2014). Digit path lengths were normalized by subject palm width, also as described in Nataraj et al., 2014.
Two participants from the Control Group were excluded from kinematic trajectory, aperture, and accuracy analyses as they did not complete the pinching task of the virtual object correctly despite practice beforehand and instructions throughout the task. These two participants reached for the mirror instead of reaching for a virtual target located on the right side of the experimental setup.
Results
Tactile & Vibration Perception Thresholds
ALM did not indicate any significant covariates for log transformed monofilament (tactile perception) data. Neither Group (p = 0.306) nor Nerve (p = 0.660) showed significant differences in log transformed tactile perception threshold between the T2D group and controls. ALM also did not identify significant covariates for hand vibration perception threshold amplitude. No significant differences in Group (p = 0.751) or Nerve (p = 0.441) were found in hand vibration perception threshold.
Joint Kinematics
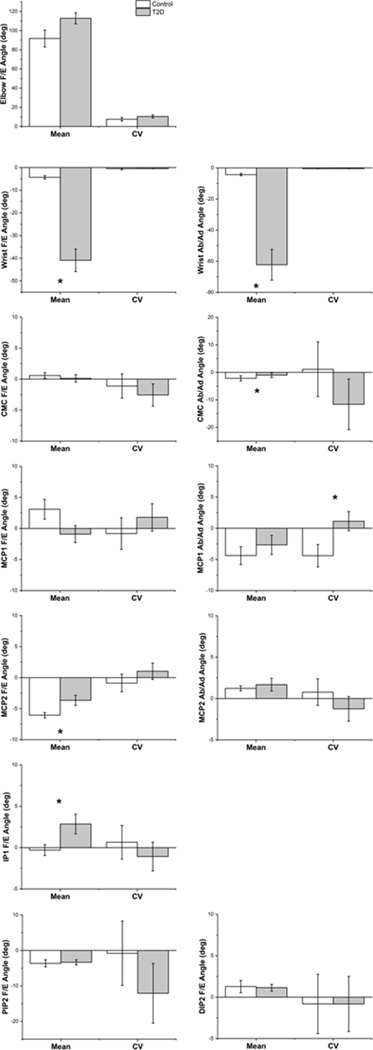
Covariates for mean joint angles identified through ALM are listed in Table 2. Significant health state covariates appeared in many of the models; the most common significant health state markers included: systole, BMI, disease duration, and presence of PN. For mean joint angles, the Group effects remained significant despite the addition of BMI within the statistical models. Digit 2 size was also found to significantly impact joint angles distal to the wrist. Mean ± SD joint angles are shown in Figure 2. Data are presented as: flexion (+), extension (−), adduction (+), and abduction (−).
Table 2:
Joint angle ALM results, listed proximal to distal; (Joint angle coefficient of variation (CV) ALM results), t- values listed BMI = body mass index, DD = Disease Duration, DIA = diastole, Digit 1 = digit 1 thickness, Digit 2 = digit 2 thickness, HbA1c = glycated hemoglobin, HDL = high-density lipoprotein, LDL = low-density lipoprotein, LQ = laterality quotient, PD = Prediabetes status, PN = peripheral neuropathy, SYS = systole, TC = total cholesterol.
| Age | BMI | DD | DIA | Digit 1 | Digit 2 | HbA1c | HDL | LDL | LQ | PD | PN | Sex | SYS | TC | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Elb | 12.58* | 3.46* | −5.62* | ||||||||||||
| W ex/fl | −4.85* | −2.74* | |||||||||||||
| W ab/ad | 2.34* | (2.44)* | 4.93* | ||||||||||||
| CMC ex/fl | 2.36* (2.75)* |
(3.32)* | (3.05)* | ||||||||||||
| CMC ab/ad | 4.80* | 3.82* | −2.60* | (2.23)* | |||||||||||
| MCP1 ex/fl | −3.20* | (2.37)* | |||||||||||||
| MCP1 ab/ad | (3.04)* | 6.17* | |||||||||||||
| IP1 | 8.55* (3.98)* |
8.49* | 5.45* | ||||||||||||
| MCP2 ex/fl | 2.94* | (2.44)* | −3.90* | ||||||||||||
| MCP2 ab/ad | (3.05)* | (3.19)* | (2.31)* | (2.45)* | −3.23* | 2.82* (4.68)* |
|||||||||
| PIP2 | −5.48* | −2.53* | −3.22* | 7.08* | (2.31)* | ||||||||||
| DIP2 | (4.61)* | 6.42* | (2.58)* |
denotes significance
Figure 2:
Comparison of mean ± SD of joint angles and coefficient of variation (CV) of mean joint angles between T2D and Control groups. Data are presented as: flexion (+), extension (−), adduction (+), and abduction (−). Significance at a minimum of p < 0.05 is denoted by *.
Between Group joint kinematic analyses indicated that the T2D Group exhibited: greater wrist extension and abduction, less CMC abduction, less extension of MCP2, and more flexion at IP1 as compared to controls (Fig. 2). Significant differences (indicated by * in Fig. 2) in mean joint angle were confirmed between Groups for wrist flexion/extension (W ex/fl: F1,16 = 25.73, p < 0.001), wrist ab/adduction (W ab/ad: F1,17 = 64.33, p < 0.001), CMC ab/adduction (CMC ab/ad: F1,15 = 17.42, p < 0.001), and MCP2 flexion/extension (F1,16 = 23.01, p < 0.001). MCP1 flexion/extension was nearly significant (p = 0.066). Mean IP1 joint angle was significantly different between Groups when covariates were not included in the statistical analysis (F1,23 = 5.15, p < 0.05) but did not persist with the addition of covariates.
Covariates for CV of joint angles were identified through ALM are listed in parentheses in Table 2. Digit 1 size was found to significantly impact CV of joint angles distal to the wrist. Between Group joint kinematic variability analyses indicated that the T2D Group exhibited greater variability in MCP1 ab/adduction (F1,16 = 4.71, p < 0.05), indicated by * in Fig. 2.
Endpoint Trajectories
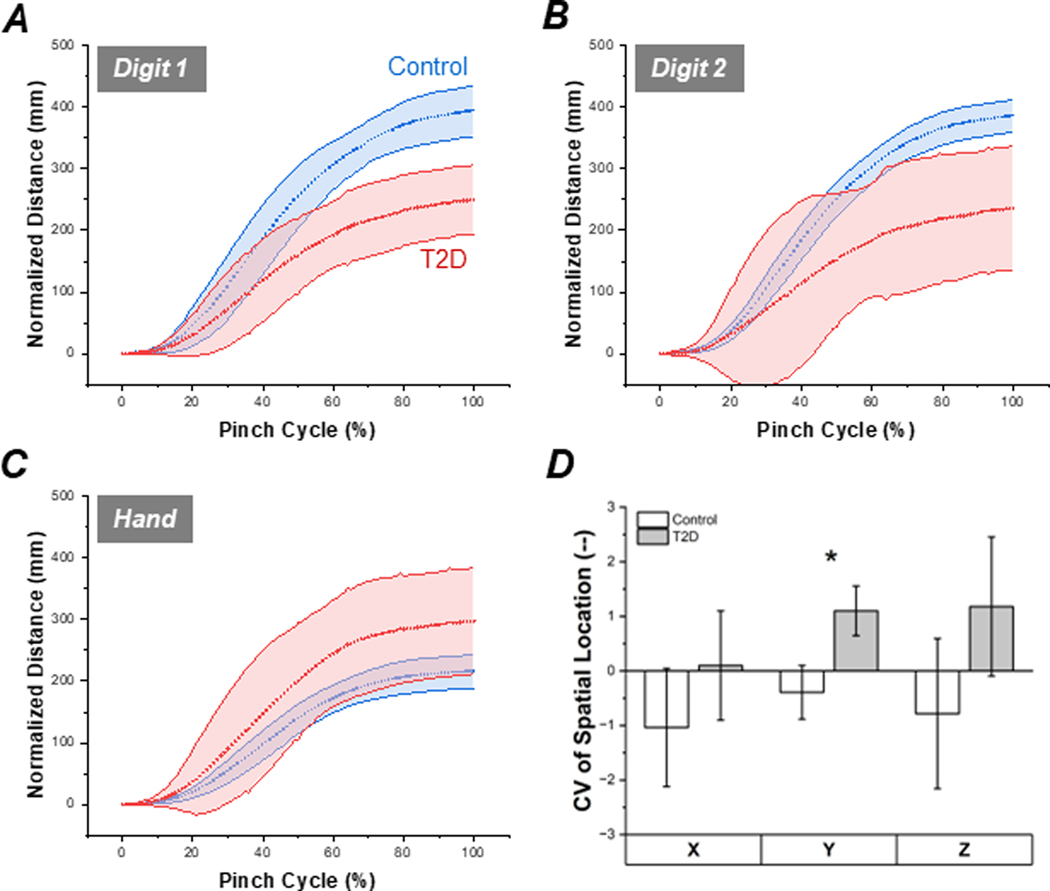
ALM covariates of kinematic variables are listed in Table 3. Regarding endpoint trajectories, T2D participants had significantly shorter distance traveled by digit 1 than controls (Group: F1,19 = 115.19, p < 0.001), which can be seen in Fig. 3A. Digit 1 path trajectory CV was not significantly different between Group; however, it was significantly impacted by blood pressure measures (systole & diastole). A significant difference in digit 2 trajectory also emerged between Groups (F1,19 = 72.27, p < 0.001; Fig. 3B), such that digit 2 path length was shorter in the T2D Group as compared to controls. Digit 2 trajectory CV was not significantly different between Groups.
Table 3:
Trajectory ALM results, t-values listed. BMI = body mass index, CV = coefficient of variation, DIA = diastole, Digit 1 = digit 1 thickness, Digit 2 = digit 2 thickness, DD = disease duration, HbA1c = glycated hemoglobin, HDL = high-density lipoprotein, LDL = low-density lipoprotein, LQ = laterality quotient, PN = peripheral neuropathy, SYS = systole, TC = total cholesterol.
| Hand Trajectory | CV Hand | Digit 1 Trajectory | CV Digit 1 | Digit 2 Trajectory | CV Digit 2 | Accuracy | |||
|---|---|---|---|---|---|---|---|---|---|
| x | y | z | |||||||
| BMI | 3.53* | 4.01* | 3.28* | 5.86* | |||||
| DD | |||||||||
| DIA | −3.21* | 2.22* | |||||||
| Digit 1 | 4.30* | 2.29* | |||||||
| Digit 2 | −3.67* | −3.32* | |||||||
| HbA1c | |||||||||
| HDL | |||||||||
| LDL | |||||||||
| LQ | −2.44* | −2.17* | |||||||
| PN | |||||||||
| Sex | |||||||||
| SYS | 3.17* | ||||||||
| TC | 4.15* | ||||||||
denotes significance at a minimum of p < 0.05.
Figure 3:
Digit and hand path length accumulated over the pinch cycle; distances self-normalized by subject palm with, as per Nataraj et al., 2014. A) Mean digit 1 trajectory ± 1 SD. Blue lines indicate control data; red indicate T2D. B) Mean digit 2 trajectory ± 1 SD. C) Mean hand trajectory ± 1 SD. D) Coefficient of variation (CV) of end-point spatial location in the x-, y-, and z-directions for Control and T2D groups.
A significant Group difference (F1,16 = 9.28, p < 0.01; Fig. 3C) in hand trajectory was found. Overall, pwT2D hand traveled a significantly greater distance during the reach-to-pinch trial compared to controls. No significant difference in CV of hand trajectory between Groups was found.
Accuracy and Aperture
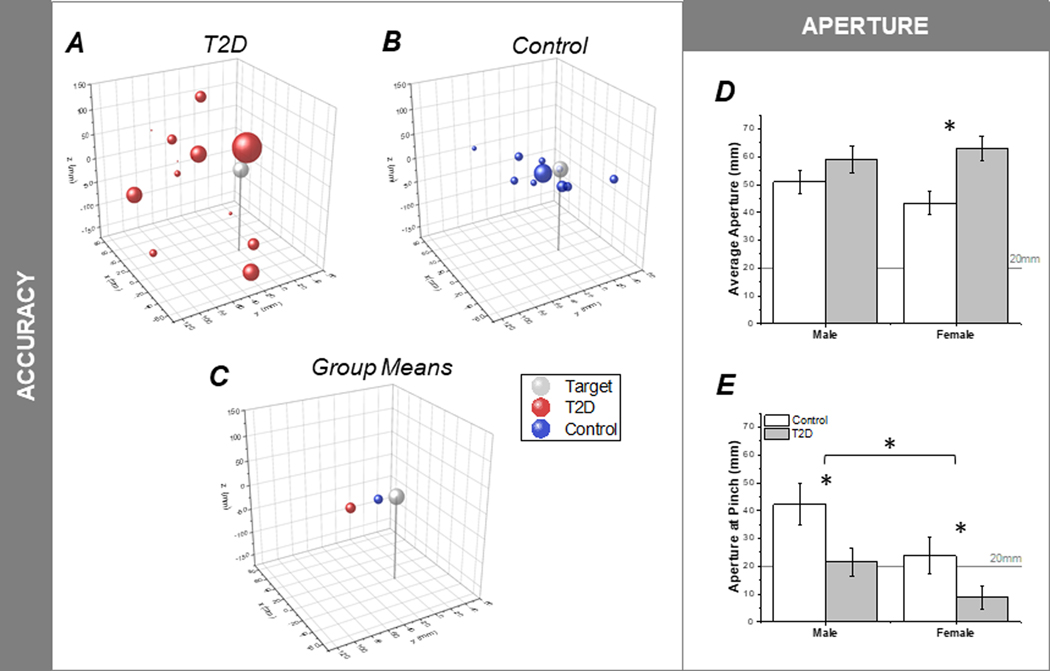
Mean pinch location (accuracy) for both Groups was generally further away from the trunk (positive x-coordinate) and to the left (positive y-coordinate) of the target. However, the T2D group was below (negative z-coordinate) while controls were slightly above (positive z-coordinate) the target.
Covariates identified via ALM for accuracy variables were as follows: Accuracy in x-direction – Digit 2 thickness, BMI, and TC; no covariates were found for accuracy in the y- and z-directions. In addition, ALM identified BMI as a covariate of overall accuracy; however, the significant Group effect remained despite the addition of BMI within the statistical model. CV data for accuracy showed a Group difference only in the y-direction (F1,16 = 4.98, p < 0.05), such that the T2D group had larger CV values in the y-direction.
Controls were significantly more accurate overall compared to target location compared to the T2D group (Overall Accuracy Group: F1,16 = 20.90, p < 0.001); values can be found in Table 4 and visualized in Fig. 4A-C. Pinch precision (variation about the target, ±1 SD about mean pinch error) did not differ significantly between groups.
Table 4:
Accuracy and Aperture Data. Group means ± SE reported. With respect to target locations, +x indicates movement away from the trunk, + y indicates leftward movement, and +z indicates upward movement relative to virtual target.
| T2D | Controls | |
|---|---|---|
| Accuracy in x-direction (mm) | 12.75 ± 10.36 | 2.32 ± 12.43 |
| Accuracy in y-direction (mm) | 49.65 ± 11.42 | 21.69 ± 10.68 |
| Accuracy in z-direction (mm) | −13.64 ± 28.49 | 1.37 ± 3.39 |
| Overall accuracy (mean pinch error, mm) | 118.78 ± 10.39* | 48.16 ± 8.70* |
| Pinch precision (mm) | 6.33 ± 4.56 | 5.59 ± 2.31 |
| Aperture at pinch (mm) | 15.60 ± 3.76* | 32.94 ± 5.60* |
| Average aperture (mm) | 60.83 ± 3.22* | 47.05 ± 3.10* |
denotes significance at a minimum of p < 0.05.
Figure 4:
Accuracy and aperture data. A-C) Mean endpoint pinch aperture and location for individual and mean T2D and Control subjects with respect to the virtual target location (grey). Center of each sphere indicates pinch contact location while the radius indicates pinch precision (+1 SD) about pinch contact location. + x forward, + y leftward, +z upward, relative to virtual target. D) Average aperture across the task, separated by males and females. The gray 20mm line indicates the target size. E) Aperture at pinch location, separated by males and females. The gray 20mm line indicates the target size. Significance at a minimum of p < 0.05 is denoted by *.
Average aperture differed significantly between Groups (F1,22 = 9.06, p < 0.01), such that the T2D group exhibited larger overall apertures throughout the movement, Fig. 4D. Aperture at pinch location (at target) also differed between Groups (F1,19 = 9.33, p < 0.005) and with Sex (F1,19 = 7.13, p < 0.05) without a Group x Sex interaction, such that aperture at pinch location (at target) was significantly smaller in the T2D Group as compared to controls, with female participants generally having small apertures at pinch location, Fig. 4E.
Discussion
The purpose of this study was to examine upper limb proprioception in older adult pwT2D as compared to healthy age- and sex-matched controls during a reaching action involving a virtual target. In Hypothesis 1, we expected pwT2D to exhibit increased movement variability of the digits, hand, and arm when compared to the control group. This hypothesis was partially supported. The T2D group exhibited significantly increased variability in transport trajectory of digit 1 when compared to controls. However, when examining joint angle trajectories, there were conflicting findings. Group differences in joint angle kinematics and variability were present in multiple joints across the upper extremity; however, these differences were not clear cut. In Hypothesis 2, we expected pwT2D to be less accurate and less precise relative to the virtual target location when compared to the control group. Hypothesis 2 was partially supported such pwT2D had significantly reduced accuracy; however, precision was not affected.
Vibrotactile Function of the Upper Extremity in T2D
Vibrotactile function of the upper extremity was assessed using the Semmes-Weinstein Monofilament Test and Biothesiometer vibration device. Contrary to most prior findings in the T2D population, neither tactile nor vibration sensation differed between groups. Previous findings in our lab have reported conflicting differences in tactile sensitivity between pwT2D and healthy controls (Gorniak et al., 2014; Gorniak, Ochoa, et al., 2020; Gorniak, Wagner, et al., 2020; Ochoa et al., 2016; Ochoa & Gorniak, 2014). The lack of tactile and vibration sensitivity differences between pwT2D and healthy age- and sex-matched controls suggests that in our study cohort, mechanoreceptors responsible for light touch (Merkel’s disks, located in the base of the epidermis, in the skin on the palms, fingers, and soles of the feet) and vibration (Meissner’s corpuscles located in the upper dermis primarily in the fingertips and deeper Ruffini endings) were intact. These findings challenge our understanding of vibrotactile dysfunction and how it relates to the development, progression, and manifestation of T2D-associated functional deficits of the upper extremity.
Proprioception & Motor Function in Older Adults with T2D
Proprioception and motor function of the upper limb was assessed using a reach-to-pinch task. Specifically, effects of T2D on kinematics of reaching and grasping a visually presented virtual target object were examined in older adults. A similar study on proprioception performed in middle-aged patients with carpal tunnel syndrome (CTS) reported greater variability about the mean angular trajectory in all joint degrees of freedom in addition to significant differences in mean trajectory values in a majority of joint degrees of freedom in persons with CTS (Nataraj et al., 2014).
Similar to the CTS study (Nataraj et al., 2014), significant differences in the mean kinematic trajectory and some indices of kinematic variability were found in pwT2D versus controls in the current study. Throughout the reach and pinch movement, pwT2D exhibited greater wrist extension and adduction, CMC abduction, IP1 flexion, and MCP2 extension and abduction compared to health age-and sex-matched controls. Additionally, pwT2D exhibited greater variability in MCP1 abduction/adduction and in y-direction (mediolateral) endpoint aiming. Total distance traveled throughout the entire movement was reduced for digits 1 and 2 in pwT2D as compared to controls, while whole hand distance traveled was increased in pwT2D. Despite moving digits 1 and 2 less during the task, the T2D group exhibited greater variability in digit 1 and hand transport when compared to controls. This suggests that throughout the reach and pinch movement in the virtual workspace, pwT2D were moving their digits 1 and 2 less, while using joints of the hand to adjust and aim for the target. In terms of functional performance outcomes, pwT2D exhibited worse mean accuracy than controls while no differences in precision were associated with T2D.
Our results are supported by findings in a recent study examining knee proprioception in pwT2D. PwT2D demonstrated 46% greater knee joint position sense inaccuracy but no differences in precision (Ettinger et al., 2018). Other lower extremity proprioception studies in pwT2D report impaired ankle joint movement perception and response to tendon vibration (Simoneau et al., 1996; R. W. van Deursen et al., 1998; R. W. M. van Deursen & Simoneau, 1999). Our results indicate that despite an absence of tactile impairment typically associated with T2D, proprioception of the upper extremity is altered in pwT2D. These changes are beyond the well-documented age-related changes in somatosensation (Shaffer & Harrison, 2007), suggesting significant systemic impacts of T2D to the human body as it ages. One potential reason that this change in upper extremity motion may occur is due to subclinical joint changes concurrent with T2D. PwT2D are more likely to experience slow-progressing structural changes to the joints, specifically to the periarticular structures, that may limit motion, including the upper extremity (Colberg et al., 2010; Francia et al., 2018).These systemic impacts may manifest as subtle functional impairment in sensorimotor function that may have significant influence on use of virtual and robotic devices to perform remote tasks.
In addition to these proprioceptive changes, altered upper extremity kinematics may also point to upstream changes in motor control strategies by the central nervous system in T2D, including altered motor cortex and cerebellar contributions that may impact spatiotemporal coordination (Geed et al., 2017; Gorniak, Wagner, et al., 2020). The altered limb kinematics may be indicative of altered aiming strategies in pwT2D (using a wrist or whole hand aiming strategy) versus modulation of digit 1 and 2 position in controls. Neither the proprioceptive changes nor the altered aiming strategies found in the T2D group are due to increased BMI associated with T2D, as group effects persisted when BMI was accounted for, consistent with (Gorniak, Lu, et al., 2019). Such a change in upper extremity kinematics could also be the result of a compensatory change of the neuromuscular system to achieve a hand posture more likely to intercept the intended target, similar to what is seen in other cardiovascular conditions such as stroke (Levin et al., 2009). Overall, these data suggest that use of a virtual reach task could be used as a potential movement biomarker to assess onset and progression of proprioceptive and motor function deficits in T2D with BMI considered. Additionally, these data also indicate long-term functional deficits in hand function that may have an impact on functional hand use when vision is not available to guide movement.
Sex Differences in T2D
Recent evidence suggests differential presentation of T2D and its complications between the sexes−particularly with increased age (Campesi et al., 2017; Centers for Disease Control and Prevention, 2020; Seghieri et al., 2017). In addition to potential proprioceptive and aiming changes in pwT2D, potential sex-based differences in sensorimotor function with advanced age emerged in this study in the functional performance outcome (aperture). It was found that female pwT2D exhibited large apertures during transport but very small apertures at the endpoint of motion. This is consistent with reports of altered proprioceptive function in females as compared to males (Cheng et al., 2007; Ko et al., 2015). Given the age of study participants, it is possible that reduced testosterone and its anti-inflammatory effects in combination with the absence of neuroprotective estrogen in postmenopausal females may negatively impact proprioception and muscle hemodynamic function in women living with T2D (Gorniak, Ochoa, et al., 2020; Pollonini et al., 2020). Further work is needed in this area to determine the role of testosterone, estrogen, estrogen signaling, and other androgens in disambiguating potential muscle hemodynamic impacts versus impaired proprioceptive function in women with T2D as they approach and navigate the menopausal transition.
Conclusion
This study examined the proprioceptive effects of T2D on the upper limb in older adults via kinematic performance of a reach-to-pinch action toward a visually-presented virtual target. Findings indicate that proprioception is altered this population such that they exhibit a unique aperture position and aiming strategy with respect to workspaces without visual feedback. Physiologic characteristics such as BMI, sex, and cardiovascular health measures were found to impact motor outcomes.
Funding:
This project was funded by R01 CA200263 via subcontract to SLG and R56 AG080816 to SLG.
Footnotes
Declarations: The authors have no relevant financial or non-financial interests to disclose. The authors have no competing interests to declare that are relevant to the content of this article.
Data Availability:
De-identified data analyzed in this project can be provided upon reasonable request.
References
- Campesi I, Franconi F, Seghieri G, & Meloni M. (2017). Sex-gender-related therapeutic approaches for cardiovascular complications associated with diabetes. Pharmacological Research, 119, 195–207. 10.1016/j.phrs.2017.01.023 [DOI] [PubMed] [Google Scholar]
- Centers for Disease Control and Prevention. (2020). National Diabetes Statistics Report. 32. https://www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf
- Cetinus E, Buyukbese MA, Uzel M, Ekerbicer H, & Karaoguz A. (2005). Hand grip strength in patients with type 2 diabetes mellitus. Diabetes Research and Clinical Practice, 70(3), 278–286. 10.1016/j.diabres.2005.03.028 [DOI] [PubMed] [Google Scholar]
- Cheng Y, Decety J, Lin C-P, Hsieh J-C, Hung D, & Tzeng OJL (2007). Sex differences in spinal excitability during observation of bipedal locomotion. NeuroReport, 18(9), 887. 10.1097/WNR.0b013e3280ebb486 [DOI] [PubMed] [Google Scholar]
- Colberg SR, Sigal RJ, Fernhall B, Regensteiner JG, Blissmer BJ, Rubin RR, Chasan-Taber L, Albright AL, & Braun B. (2010). Exercise and type 2 diabetes: The American College of Sports Medicine and the American Diabetes Association: joint position statement. Diabetes Care, 33(12), e147–167. 10.2337/dc10-9990 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ettinger LR, Boucher A, & Simonovich E. (2018). Patients with type 2 diabetes demonstrate proprioceptive deficit in the knee. World Journal of Diabetes, 9(3), 59–65. 10.4239/wjd.v9.i3.59 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Francia P, Anichini R, Seghieri G, Bellis AD, & Gulisano M. (2018). History, Prevalence and Assessment of Limited Joint Mobility, from Stiff Hand Syndrome to Diabetic Foot Ulcer Prevention: A Narrative Review of the Literature. Current Diabetes Reviews, 14(5), 411. 10.2174/1573399813666170816142731 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geed S, McCurdy ML, & van Kan PLE (2017). Neuronal Correlates of Functional Coupling between Reach- and Grasp-Related Components of Muscle Activity. Frontiers in Neural Circuits, 11. https://www.frontiersin.org/articles/10.3389/fncir.2017.00007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gorniak SL, Khan A, Ochoa N, Sharma MD, & Phan CL (2014). Detecting subtle fingertip sensory and motor dysfunction in adults with type II diabetes. Experimental Brain Research, 232(4), 1283–1291. 10.1007/s00221-014-3844-x [DOI] [PubMed] [Google Scholar]
- Gorniak SL, Lu FY, Lee BC, Massman PJ, & Wang J. (2019). Cognitive impairment and postural control deficit in adults with Type 2 diabetes. Diabetes/Metabolism Research and Reviews, 35(2), e3089. 10.1002/dmrr.3089 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gorniak SL, Ochoa N, Cox LIG, Khan A, Ansari S, Thames B, Ray H, Lu YF, Hibino H, Watson N, & Dougherty PM (2020). Sex-based differences and aging in tactile function loss in persons with type 2 diabetes. PloS One, 15(11), e0242199. 10.1371/journal.pone.0242199 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gorniak SL, Ray H, Lee B-C, & Wang J. (2019). Cognitive–Motor Impairment in Manual Tasks in Adults With Type 2 Diabetes: OTJR: Occupation, Participation and Health, 40(2), 113–121. 10.1177/1539449219880536 [DOI] [PubMed] [Google Scholar]
- Gorniak SL, Wagner VE, Vaughn K, Perry J, Cox LG, Hernandez AE, & Pollonini L. (2020). Functional neuroimaging of sensorimotor cortices in postmenopausal women with type II diabetes. Neurophotonics, 7(03), 1–17. 10.1117/1.nph.7.3.035007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gorniak SL, Wagner VE, Vaughn K, Perry J, Gulley Cox LI, Hernandez AE, & Pollonini L. (2023). Cortical activation deficits and memory loss in postmenopausal women with Type II Diabetes. Experimental Brain Research, (in press). 10.1007/s00221-023-06581-1 [DOI] [PMC free article] [PubMed]
- Hewston P, & Deshpande N. (2016). Falls and Balance Impairments in Older Adults with Type 2 Diabetes: Thinking Beyond Diabetic Peripheral Neuropathy. Canadian Journal of Diabetes, 40(1), 6–9. 10.1016/j.jcjd.2015.08.005 [DOI] [PubMed] [Google Scholar]
- Hong C, Chia S, & Ling S. (1997). Postural stability in non-insulin depedent diabetics. Annals of the Academy of Medicine, Singapore, 26(6), 736–741. [PubMed] [Google Scholar]
- Ko S-U, Simonsick E, Deshpande N, & Ferrucci L. (2015). Sex-specific age associations of ankle proprioception test performance in older adults: Results from the Baltimore Longitudinal Study of Aging. Age and Ageing, 44(3), 485–490. 10.1093/ageing/afv005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin MF, Kleim JA, & Wolf SL (2009). What Do Motor “Recovery” and “Compensation” Mean in Patients Following Stroke? Neurorehabilitation and Neural Repair, 23(4), 313–319. 10.1177/1545968308328727 [DOI] [PubMed] [Google Scholar]
- Lord S, Caplan G, Colagiuri R, Colagiuri S, & Ward J. (1993). Sensori-motor function in older persons with diabetes. Diabetic Medicine, 10(7), 614–618. [DOI] [PubMed] [Google Scholar]
- Mold JW, Vesely SK, Keyl BA, Schenk JB, & Roberts M. (2004). The Prevalence, Predictors, and Consequences of Peripheral Sensory Neuropathy in Older Patients. The Journal of the American Board of Family Practice, 17(5), 309–318. 10.3122/jabfm.17.5.309 [DOI] [PubMed] [Google Scholar]
- Mustapa A, Justine M, Mohd Mustafah N, Jamil N, & Manaf H. (2016). Postural Control and Gait Performance in the Diabetic Peripheral Neuropathy: A Systematic Review. BioMed Research International, 2016. 10.1155/2016/9305025 [DOI] [PMC free article] [PubMed]
- Nataraj R, Evans PJ, Seitz WH, & Li ZM (2014). Effects of carpal tunnel syndrome on reach-to-pinch performance. PLoS ONE, 9(3), 1–9. 10.1371/journal.pone.0092063 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nataraj R, & Li Z-M (2015). Integration of marker and force data to compute three-dimensional joint moments of the thumb and index finger digits during pinch. Comput Methods Biomech Biomed Engin, 18(6), 592–606. 10.1080/10255842.2013.820722 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ochoa N, Gogola GR, & Gorniak SL (2016). Contribution of tactile dysfunction to manual motor dysfunction in type II diabetes. Muscle & Nerve, 54(5), 895–902. 10.1002/mus.25137 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ochoa N, & Gorniak SL (2014). Changes in sensory function and force production in adults with type II diabetes. Muscle & Nerve, 50(6), 984–990. 10.1002/mus.24261 [DOI] [PubMed] [Google Scholar]
- Orlando G, Balducci S, Bazzucchi I, Pugliese G, & Sacchetti M. (2016). Physical Exercise as therapy for type II diabetes. Diabetes/Metabolism Research and Reviews, 32(30), 40–50. 10.1002/dmrr [DOI] [PubMed] [Google Scholar]
- Pai Y-C, Rymer WZ, Chang RW, & Sharma L. (1997). Effect of age and osteoarthritis on knee proprioception. Arthritis & Rheumatism, 40(12), 2260–2265. 10.1002/art.1780401223 [DOI] [PubMed] [Google Scholar]
- Pollonini L, Gulley Cox L, & Gorniak SL (2020). Hemodynamic Function of Forearm Muscle in Postmenopausal Women With Type 2 Diabetes. Journal of Aging and Physical Activity, 1–8. 10.1123/japa.2019-0221 [DOI] [PMC free article] [PubMed]
- Ramji N, Toth C, Kennedy J, & Zochodne DW (2007). Does diabetes mellitus target motor neurons? Neurobiology of Disease, 26(2), 301–311. 10.1016/j.nbd.2006.11.016 [DOI] [PubMed] [Google Scholar]
- Sayer AA, Dennison E, Syddall H, Gilbody H, Phillips D, & Cooper C. (2005). Type 2 Diabetes, Muscle Strength, and Impaired Physical Function. Diabetes Care, 28(10), 2541–2542. 10.2337/diacare.28.10.2541 [DOI] [PubMed] [Google Scholar]
- Seghieri G, Policardo L, Anichini R, Franconi F, Campesi I, Cherchi S, & Tonolo G. (2017). The Effect of Sex and Gender on Diabetic Complications. Current Diabetes Reviews, 13(2), 148–160. 10.2174/1573399812666160517115756 [DOI] [PubMed] [Google Scholar]
- Shaffer SW, & Harrison AL (2007). Aging of the Somatosensory System: A Translational Perspective. Physical Therapy, 87(2), 193–207. 10.2522/ptj.20060083 [DOI] [PubMed] [Google Scholar]
- Simmons RW, Richardson C, & Pozos R. (1997). Postural stability of diabetic patients with and without cutaneous sensory deficit in the foot. Diabetes Research and Clinical Practice, 36(3), 153–160. 10.1016/S0168-8227(97)00044-2 [DOI] [PubMed] [Google Scholar]
- Simoneau GG, Derr JA, Ulbrecht JS, Becker MB, & Cavanagh PR (1996). Diabetic sensory neuropathy effect on ankle joint movement perception. Archives of Physical Medicine and Rehabilitation, 77(5), 453–460. 10.1016/S0003-9993(96)90033-7 [DOI] [PubMed] [Google Scholar]
- Uccioli L. (1995). Body Sway in Diabeteic Neuropathy. Diabetes Care, 18(3), 339–344. [DOI] [PubMed] [Google Scholar]
- van Deursen RWM, Sanchez MM, Derr JA, Becker MB, Ulbrecht JS, & Cavanagh PR (2001). Vibration perception threshold testing in patients with diabetic neuropathy: Ceiling effects and reliability. Diabetic Medicine, 18(6), 469–475. [DOI] [PubMed] [Google Scholar]
- van Deursen RWM, & Simoneau GG (1999). Foot and Ankle Sensory Neuropathy, Proprioception, and Postural Stability. Journal of Orthopaedic & Sports Physical Therapy, 29(12), 718–726. 10.2519/jospt.1999.29.12.718 [DOI] [PubMed] [Google Scholar]
- van Deursen RW, Sanchez MM, Ulbrecht JS, & Cavanagh PR (1998). The role of muscle spindles in ankle movement perception in human subjects with diabetic neuropathy. Experimental Brain Research, 120(1), 1–8. 10.1007/s002210050371 [DOI] [PubMed] [Google Scholar]
- WHO. (2017). Diabetes. http://www.who.int/mediacentre/factsheets/fs312/en/
- Wu G, van der Helm F, Veeger H, Makhsous M, Van Roy P, Anglin C, Nagels J, Karduna A, McQuade K, Wang X, Werner F, & Buchholz B. (2005). ISB recommendation on definitions of joint coordinate systems of various joints for the reporting of human joint motion—Part II: shoulder, elbow, wrist and hand. J Biomech, 38(5), 981–992. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
De-identified data analyzed in this project can be provided upon reasonable request.