Abstract
Objectives
Position- and posture-dependent deformation of the vascular system is a relatively unexplored field. The goal of this scoping review was to create an overview of existing vascular imaging modalities in different body positions and postures and address the subsequent changes in vascular anatomy.
Methods
Scopus, Medline, and Cochrane were searched for literature published between January 1, 2000, and June 30, 2022, incorporating the following categories: image modality, anatomy, orientation, and outcomes.
Results
Out of 2446 screened articles, we included 108. The majority of papers used ultrasound (US, n = 74) in different body positions and postures with diameter and cross-sectional area (CSA) as outcome measures. Magnetic resonance imaging (n = 22) and computed tomography (n = 8) were less frequently used but allowed for investigation of other geometrical measures such as vessel curvature and length. The venous system proved more sensitive to postural changes than the arterial system, which was seen as increasing diameters of veins below the level of the heart when going from supine to prone to standing positions, and vice versa.
Conclusions
The influence of body positions and postures on vasculature was predominantly explored with US for vessel diameter and CSA. Posture-induced deformation and additional geometrical features that may be of interest for the (endovascular) treatment of vascular pathologies have been limitedly reported, such as length and curvature of an atherosclerotic popliteal artery during bending of the knee after stent placement. The most important clinical implications of positional changes are found in diagnosis, surgical planning, and follow-up after stent placement.
Clinical relevance statement
This scoping review presents the current state and opportunities of position- and posture-dependent imaging of vascular structures using various imaging modalities that are relevant in the fields of clinical diagnosis, surgical planning, and follow-up after stent placement.
Key Points
• The influence of body positions and postures on the vasculature was predominantly investigated with US for vessel diameter and cross-sectional area.
• Research into geometrical deformation, such as vessel length and curvature adaptation, that may be of interest for the (endovascular) treatment of vascular pathologies is limited in different positions and postures.
• The most important clinical implications of postural changes are found in diagnosis, surgical planning, and follow-up after stent placement.
Supplementary information
The online version contains supplementary material available at 10.1007/s00330-023-10154-9.
Keywords: Diagnostic imaging, Posture, Blood vessels, Humans
Introduction
Medical imaging modalities have been used for decades to visualize human vasculature. The most commonly used modalities are digital subtraction angiography (DSA), computed tomography angiography (CTA), magnetic resonance angiography (MRA), and ultrasound (US) [1, 2]. Vascular imaging is used to provide insights in anatomy, geometry, patency of blood vessels, and various vascular pathologies. Each modality has benefits and drawbacks in terms of spatial resolution, imaging speed, tissue contrast, field of view, reproducibility, cost, and safety.
Most often, the patient’s vascular anatomy is visualized in supine position with extended limbs. In some cases, however, clinical symptoms may be position or posture dependent. Therefore, the underlying vascular pathology will not always be visualized appropriately in supine position. The geometry (diameter, cross-sectional area, curvature, torsion, etc.) of vessels in the human body may differ in the supine, prone, and standing position, but also in flexed and/or rotated head and limb postures, which may be associated with clinical outcomes such as stenosis development [3]. When only visualizing the vascular structures in supine, neutral position, features predisposing vascular pathologies may be missed [4].
Some imaging modalities have already been clinically applied to visualize vasculature in body positions and postures other than a neutral supine position. An example is US in a posture with abducted arms, where the elevated limbs induce compression of arterial and/or venous structures, also referred to as thoracic outlet syndrome (TOS) [5]. Opportunities to visualize vasculature in an upright fashion have emerged in CT and MRI, but are often only applied in research settings and not commonly applied in clinical practice yet [6–8]. Still, postural and positional information can aid in several clinical situations, such as diagnosing posture-triggered atrial fibrillation or Bow Hunter’s syndrome [9–11], screw placement in prone body position during spine surgery without aortic injury [12], or in quantifying iliac artery deformation by musculoskeletal motion for the purpose of decreasing stent-graft-related complications [13]. Therefore, the potential and added value of vascular imaging in different body positions and posture remain largely unexplored. The goal of this scoping review was to obtain an overview of existing vascular imaging modalities in different body positions and postures and to address subsequent geometrical changes in vascular anatomy.
Methods
This literature review complied with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement standards [14]. The review protocol was prospectively registered in the PROSPERO database (identifier CRD42021264322).
Search strategy
A search in the databases Scopus, Medline, and Cochrane for literature published between January 1, 2000, and June 30, 2022, was performed. The search incorporated the following categories: (1) image modality, (2) anatomy, (3) orientation, and (4) outcomes. The exact search terms can be found in Table 1. To accommodate the different body positions and postures, subcategories were made for the orientation category: 3a represents body positions such as upright position; the changes in body postures such as flexion and extension (3b) of different body parts such as the elbow and knee (3c). The search terms within the categories were separated by Boolean OR, and the categories were separated by Boolean AND. Incorporation of the different subcategories for (3) orientation is presented in the last row of Table 1. An asterisk (*) was used to indicate a conjugation of the search term. No language restrictions were applied. After the elimination of double hits, two authors ((J.v.Z. and J.S.)) independently screened the search results on title and abstract to adhere to the inclusion criteria:
Table 1.
The categories and search terms used. The block diagram below the table shows the composition between categories with special attention to the orientation category that consisted of body position and body posture terms
1. Medical imaging was applied.
2. The study involved adult human subjects.
3. The anatomical focus was in the vascular domain.
4. The different position/posture was performed during imaging.
5. The different position/posture was compared to a standard reference position/posture.
6. Changes in outcome were stated as objective measure (measurable unit).
7. It was an original research article.
Data extraction and quality assessment
For each study, the authors, publication year, subjects, vascular system, anatomical location, imaging modality, body posture/position, and study outcome measures were collected. Subsequently, each article was classified by the anatomical location: (1) head and neck, (2) thoracic, (3) abdominal, and (4) peripheral (arms and legs). Then, the data was sorted per vascular system that was being researched: (1) arterial, (2) venous, or (3) arterial and venous (AV). Additionally, the found studies were described while taking into account the different imaging modalities: (1) US, (2) CT, (3) MRI, (4) DSA, and (5) a combination of two or more (multiple) imaging modalities (MMOD). In case a large number of articles (≥ 20) was found on a specific disease or condition, these articles were excluded on eligibility at the authors’ discretion with the justification that a specialized review on that topic would be more appropriate.
To standardize the subject populations, we distinguished healthy volunteers from patients, where the type of patients or pathology was specified. To obtain consistency in position and posture descriptions, the following positional terms were used: supine, prone, lateral (also including lateral decubitus, recumbent, or park bench position), Trendelenburg (TB, also including head-down tilt), and reverse Trendelenburg (R-TB, also including head-up tilt). To focus on how blood vessels are affected by different positions and postures, the outcomes of the studies were reported in a generic way describing the observed trends without specific details (e.g., exact values, percentage changes, significance levels) for all studies and without replacing the original outcome term (e.g., diameter, CSA).
Results
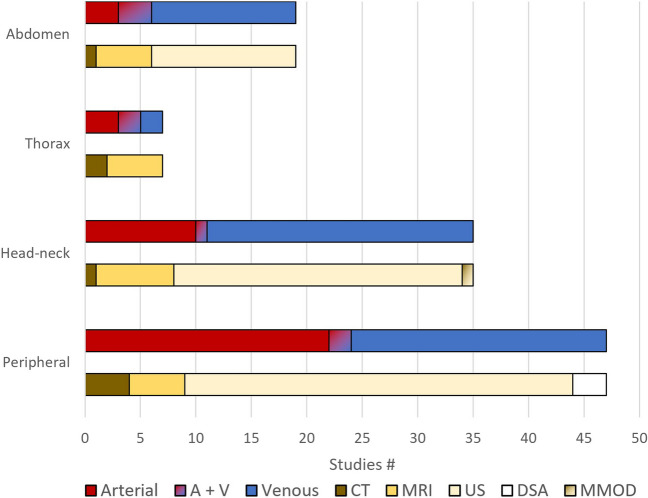
A total of 2446 articles were screened for adherence to the inclusion criteria, resulting in 280 articles that were evaluated for eligibility. A large part of these articles focused on the Bow Hunter’s syndrome (n = 93) and vessel dimensions in the scope of cannulation (n = 48). Because these are specific and widely investigated subjects, the authors consider them stand-alone topics that may overshadow other interesting findings of the present review and the corresponding articles were therefore deemed not eligible. Figure 1 illustrates the flowchart of the study selection. Characteristics of the included articles are shown in Table 2, categorized by anatomical location and imaging modality. The distribution of included articles over the different anatomical locations is shown in Fig. 2.
Fig. 1.
Flow chart of the studies included in the review
Table 2.
Overview of studies included in this review
| Year | First author | Modality | System | Vessel(s) | Subjects | Position/posture | Outcome measures | |
| Abdomen (n = 13+6) | 2019 | Hensley | US | VEN | IVC | 10 pat. (surgery) | Supine vs prone | Diameter |
| 2011 | Ho | US | VEN | IVC | 24 vol. | Upright vs prone | Diameter | |
| 2015 | Kundra | US | VEN | IVC | 32 pat. (term parturients) | Supine vs R-TB | Diameter | |
| 2014 | Panebianco | US | VEN | IVC | 45 pat. (dialysis) | Supine vs R-TB (45°) | Diameter | |
| 2001 | Rist | US | VEN | CIV | 10 pat. (surgery) | Supine vs TB (20°) vs R-TB (20°) | Diameter | |
| 2010 | Asbeutah | US | ART | CeA | 30 vol. | Supine vs upright | Diameter | |
| 2019 | Krzanowski | US | VEN | CIV + LRV | 41 pat. (w/ pelvic venous disorder) | Supine vs LL vs upright | CSA | |
| 2020 | Ebubedike | US | VEN | SV | 82 pat. (w/ varicocele) | Supine vs upright | Diameter | |
| 2021 | Engelhorn | US | VEN | Left CIV | 50 vol. | Prone vs upright | Diameter | |
| 2016 | Ukere | US | VEN | Right HV + PV | 20 pat. (liver resection) | Supine vs R-TB (20°) | Diameter | |
| 2021 | Arshad | US | VEN | SV | 35 pat. (w/ varicocele) | Supine vs upright | Diameter | |
| 2011 | Mookadam | US | VEN | IVC | 43 pat. (w/ echocardiography) | Supine vs LL | Diameter | |
| 2016 | Najari | US | VEN | SV | 114 pat. (w/ varicocele) | Supine vs upright | Diameter | |
| 2020 | Jinzaki | CT | A+V | VC + aorta | 32 vol. | Supine vs upright | CSA | |
| 2019 | Kadoya | MRI | VEN | PV | 12 vol. | Supine vs upright | CSA | |
| 2021 | Kadoya | MRI | A+V | IVC + AA | 12 vol. | Supine vs upright | CSA | |
| 2016 | Saravanakumar | MRI | A+V | IVC + AA | 6 pat. (obese and pregnant) | RL; LL; supine with pelvic tilt; R-TB (5°, 10° and 15°) | CSA | |
| 2012 | Ganesan | MRI | ART | CIA | 7 pat. (MRI) | Supine vs prone | Vessel-spine distance | |
| 2009 | Choi | MRI | ART | Aorta + CIA | 7 vol. | Supine vs fetal (hip flexion) | Curvature, length, angle, distance | |
| Thorax (n = 7) | 2019 | Riccio | MRI | A+V | IVC + AA | 20 vol. | Supine vs prone | Aorta-spine distance |
| 2015 | Qian | MRI | ART | Aorta | 22 pat. (w/ ankylosing spondylitis) | Supine vs prone | Aorta-spine distance | |
| 2019 | Plataniotis | CT | ART | Aorta | 200 pat. (w/ lower back pain) | Supine vs prone | Aorta-spine distance | |
| 2012 | Jiang | MRI | ART | Aorta | 26 pat. (w/ idiopathic scoliosis) | Supine vs prone | Aorta-spine distance | |
| 2008 | Yamaji | CT | VEN | PuV | 116 pat. (w/ atrial fibrillation) | Supine vs prone | Diameter | |
| 2019 | Wieslander | MRI | A+V | PuV + PuA | 10 vol. | Supine vs prone vs LL vs RL | CSA | |
| 2021 | Gottlieb | MRI | VEN | PuV | 20 vol. | Supine vs LL | Diameter | |
| Head-neck (n = 25 + 10) | 2009 | Morimoto | US | VEN | IJV + EJV + VV | 22 vol. | Supine vs upright vs. TB (90°) | Diameter |
| 2016 | Montero | US | ART | ICA + VA | 10 vol. | TB (30°) vs supine vs R-TB (30°) | Diameter | |
| 2012 | Kantarci | US | VEN | IJV + VV | 62 pat. (w/ multiple sclerosis) & 21 vol. | Supine vs upright | CSA | |
| 2009 | Saeed | US | ART | CCA | 20 vol. | Supine vs upright (sitting) | Diameter | |
| 2016 | Yeoh | US | VEN | IJV | 27 vol. | Supine vs prone vs RL | CSA | |
| 2014 | Lee | US | VEN | IJV | 58 pat. (w/ ASA I-II) | Supine vs TB (10°) | Diameter, CSA | |
| 2013 | Jo | US | VEN | IJV | 40 pat. (w/ ASA I-II) | Supine vs TB (10°) | CSA | |
| 2019 | Chen | US | VEN | IJV | 82 pat. (prostatectomy) | Supine vs TB | CSA | |
| 2004 | Gisolf | US | VEN | IJV | 6 vol. | Supine vs upright | CSA | |
| 2017 | Holmlund | US | VEN | IJV | 17 vol. | Supine vs upright (0°, 16°, 71°) | CSA | |
| 2019 | Wang | US | VEN | IJV | 20 vol. | Supine vs. upright (0°, with increment 10° till 90°) | CSA | |
| 2008 | Kim | US | VEN | IJV + SCVs | 20 vol. | Supine vs. TB (15°) vs. R-TB (15°) vs. leg elevation (50°) | CSA | |
| 2018 | Judickas | US | VEN | IJV | 63 vol. |
30° R-TB, 45° passive leg raise, and 10° TB. All vs. Supine and with and without 30° head rotation |
CSA | |
| 2017 | Watkins | US | VEN | IJV | 15 vol. | Supine vs 15° head down tilt vs. sitting | CSA | |
| 2021 | Marshall-Goebel | US | VEN | IJV | 10 vol. | Supine vs upright (sitting) | CSA | |
| 2011 | Mayer | US | VEN | IJV + VV | 20 pat. (w/ multiple sclerosis) & 20 vol. | Supine vs upright (sitting) | CSA | |
| 2019 | Okamura | US | VEN | IJV | 82 pat. (surgery) | Supine vs TB (10°) | CSA | |
| 2019 | Westlund | US | VEN | IJV | 17 vol. | Supine vs upright (sitting, 0°, 16° and 71°) | CSA | |
| 2019 | Boschert | US | VEN | IJV + EJV | 11 vol. | Supine vs TB (12°) | CSA | |
| 2017 | Holmlund | US | VEN | IJV | 11 vol. | Supine vs. upright (8°, 16°, 24°, 32°, 40°, 69°) | CSA | |
| 2004 | Glor | US | ART | CCA (right) | 9 vol. | Supine vs. supine with head in max. left turn | CSA | |
| 2001 | Muhammad | US | VEN | IJV | 25 pat. (surgery) | Semi-prone (10° tilt) head positions: neutral vs lateral rotation (sideways) vs medial rotation (towards body) | Diameter | |
| 2018 | Park | US | ART | CCA | 18 vol. | supine-head neutral vs supine-45° head rotation vs supine-80° head rotation vs LL vs RL | Distance from the C6 anterior tubercle to the carotid artery | |
| 2021 | Yu | US | ART | ICA | 28 pat. (w/ ASA 1–11) | Supine vs TB | Diameter | |
| 2011 | Doepp | US | VEN | IJV + AV + VV | 40 pat. (w/ multiple sclerosis) | Supine vs upright | CSA | |
| 2013 | Papaharilaou | MRI | ART | ICA + ECA + CCA | 2 vol. | Supine vs. prone with up to 80° left and right head rotation | Angles between the ICA-CCA, ICA-ECA, and ECA-CCA | |
| 2009 | Aristokleous | MRI | ART | ICA + ECA + CCA | 5 vol. | Supine vs. supine with up to 80° left and right head rotation | Angles between the ICA-CCA, the ICA-ECA, and ECA-CCA | |
| 2011 | Niggemann | MRI | VEN | IJV + EJV + NV + VP | 15 vol. | Supine vs. upright (sitting) | Diameter | |
| 2020 | Kosugi | CT | A+V | IJV + EJV + ACV + ICA | 20 vol. | Supine vs. upright | CSA | |
| 2016 | Holtackers | US + MRI | ART | CCA | 12 vol. | Supine neutral vs. supine ~50° left head rotation + tilt back | Diameter, vessel length | |
| 2017 | Ishida | MRI | A+V | IJV + ICA + VA | 22 vol. | Supine vs. –6° HDT vs. –12° HDT | CSA | |
| 2022 | van Zandwijk | MRI | VEN | IJV | 15 vol. | Supine vs. 21°, 45°, 69° and 90° upright | Diameter | |
| 2003 | Vos | MRI | ART | CCA + ICA | 7 pat. (carotid stenting) | 5 different head positions (neutral, turned left and right, and bent forward and backward) | Artery angulation | |
| 2017 | Qureshi | DSA | A+V | IJV | 3 pat. (angiography) | Supine vs. 60° upright | Diameter | |
| 2012 | Aristokleous | MRI | ART | CCA + ICA | 2 vol. | Supine neutral vs. prone with 80° left head rotation vs. prone with 80° right rotation | Bifurcation angle, artery angle, CSA | |
| Peripheral (n = 34 + 13) | 2021 | Mestre | US | VEN | SSV + DCV | 57 pat. (w/ CVD) & 54 vol. | Supine vs upright (standing) | CSA |
| 2002 | Lurie | US | VEN | SFV | 10 pat. (w/ chronic venous insuff.) | Standing vs R-TB (15°) | CSA | |
| 2012 | DeMuth | US | VEN | GSV | 28 pat (w/ venous insuff.) | Supine vs R-TB | Diameter | |
| 2005 | Groothuis | US | ART | CFA | 11 pat (w/ spinal cord injury) & 10 vol. | Supine vs R-TB | Diameter | |
| 2002 | Hoballah | US | VEN | GSV | 20 vol. | Supine vs R-TB (15°) | Diameter | |
| 2003 | Nguyen | US | VEN | FV | 30 pat. (w/ gastric bypass) | Supine vs R-TB (30°) | CSA | |
| 2013 | Villar | US | ART | PoA | 11 vol. | Supine vs TB (35°) vs R-TB (45°) | Diameter | |
| 2003 | Limpus | US | VEN | CFV | 20 vol. | Sitting vs knee flexed (90°) vs LL vs supine | Diameter | |
| 2005 | Dix | US | VEN | PoV (and all deep veins) | 29 pat. (w/ leg ulcers) & 10 vol. | Standing, sitting, horizontal, elevated leg with increment 5° till 45° | CSA | |
| 2000 | Delis | US | ART | PoA | 36 pat. (w/ intermittent claudication) & 29 vol. | Recumbency, sitting, return to recumbency | Diameter | |
| 2008 | Newcomer | US | ART | BA + SFA | 21 vol. | Supine vs seated vs standing | Diameter | |
| 2019 | Tauraginskii | US | VEN | GSV | 61 pat. (w/ GSV incompetency) | Supine vs upright (w/ stretched legs) vs vertical | Diameter | |
| 2019 | Ciufo | US | VEN | PoV | 13 vol. | Standing vs knee flexed | Diameter | |
| 2006 | Pemble | US | VEN | Superficial + deep leg veins | 39 pat. (primigravida women) | Supine vs upright | CSA | |
| 2013 | Villar | US | ART | PoA | 15 vol. | TB (35°) vs R-TB (45°) | Diameter | |
| 2004 | Delis | US | VEN | PoV + FV + CFV | 13 vol. | Sitting vs standing | Diameter | |
| 2006 | Morita | US | VEN | PoV | 21 vol. | Prone vs sitting | CSA | |
| 2013 | Warwick | US | VEN | SFV | 10 vol. | Supine vs leg elevated vs upright | CSA | |
| 2008 | van Rij | US | VEN | FV | 934 pat. (venous disease) | Standing vs sitting vs lying vs ambulating | Diameter | |
| 2021 | Reb | US | VEN | PoV | 24 vol. | Upright straight leg vs knee flex. (crutch position, 90°) | Diameter | |
| 2016 | Lattimer | US | VEN | FV | 11 pat. (obstruction group) & 11 pat. (reflux group) & 11 vol. | R-TB (70°) vs R-TB (45°) vs TB (40°) | Diameter | |
| 2013 | Villar | US | ART | PoA | 15 vol. | Prone vs TB (35°) vs R-TB (45°) | Diameter | |
| 2017 | Becker | US | ART | ATA | 18 vol. | Supine vs R-TB (15°, 6°) vs TB (6°, 15°) | Diameter | |
| 2019 | Yang | US | VEN | GSV + SSV | 9 vol. | Supine or prone vs standing | Diameter | |
| 2017 | Villar | US | ART | PoA | 10 vol. | Supine vs TB (35°) vs R-TB (45°) | Diameter | |
| 2006 | Cirovic | US | VEN | ATV + PTV + PeV + GSV + SSV | 12 vol. | Supine, sitting legs horizontal, sitting one leg suspended, supine one leg raised | CSA | |
| 2019 | Claydon | US | ART | BA | 16 vol. | Supine vs R-TB (60°) | Diameter | |
| 2019 | Brown | US | ART | PoA | 45 pat. (15 w/ symptomatic lower extremity, 30 asymp.) | Ankle neutral vs maximally plantar flex. | Diameter | |
| 2011 | Levine | US | VEN | PoV | 16 vol. | Supine vs knee flexed or hyperextended | Diameter | |
| 2011 | Pannucci | US | VEN | CFV | 12 vol. | Supine w/ knee flex. (90°) & hip flex. (0°–30°–60°–90°) | Diameter | |
| 2016 | Sadek | US | VEN | SuV | 49 vol. | Neutral vs arm abd. (90°) | CSA | |
| 2009 | Stapleton | US | ART | SuA | 31 vol. | Arm in 12 positions (degrees of horizontal flex./ext., abd. and er.) | Diameter | |
| 2007 | Stapleton | US | ART | AA | 26 vol. | Glenohumeral glide position | Diameter | |
| 2006 | Demondion | US | ART | SCA | 28 pat. (w/ arterial TOS) & 44 vol. | Neutral vs abd. (90°, 130°, 17°) | CSA | |
| 2013 | Park | CT | ART | SCA | 1 vol. | Supine vs a full-draw position in archery | CSA | |
| 2020 | Turin | MRI | VEN | GV (sup. + inf.) | 16 vol. | Supine, prone, prone with a bump (jack-knife), lateral positions | Diameter, vessel location with respect to bony landmarks | |
| 2021 | Fujii | MRI | VEN | SSV + GSV | 56 vol. | Supine vs sitting vs standing | CSA | |
| 2021 | Breen | MRI | ART | PoA | 10 vol. | Supine w/ wedge again plantar side of feet vs supine w/ active plantar flex. | Diameter | |
| 2010 | Cheng | MRI | ART | SFA | 7 vol. | Supine vs LL | Vessel length, axial twist, curvature | |
| 2004 | Diaz | DSA | ART | PoA | 57 pat. (angiography) | Leg flex. (100°) vs extension | Distance between the popliteal hinch point and the medial supracondylar tubercle of the femur | |
| 2013 | Gökgöl | CT | ART | PoA | 5 pat. (w/ symptomatic PAD) | Leg ext. vs flex. (70°, 20°) in the knee/hip | Vessel length, axial twist, curvature | |
| 2017 | Gökgöl | DSA | ART | SFA + PoA | 35 pat. (w/ PAD) | Leg ext. vs flex. (70°, 20°) in the knee/hip | Vessel length, curvature | |
| 2009 | Klein | DSA | ART | SFA + PoA | 9 pat. (w/ PAD) | Leg straight vs flex. | Vessel length, curvature, torsion | |
| 2011 | LaBan | CT | A+V | SCA + SCV | 17 pat. (w/ TOS) | Arm neutral vs abd. (90°) w/ er. | Diameter | |
| 2017 | Rabtsun | CT + US | VEN | SFA + PoA | 10 pat. (w/ SFA occlusion) | Leg straight vs flex. (110° hip, 20° knee) | Vessel length | |
| 2019 | Spinella | CT | ART | PoA + SFA | 7 pat. (w/ popliteal aneurysm) | Straight leg vs 90° knee flex, | Vessel length, tortuosity index, curvature, diameter | |
| 2004 | Charon | MRI | A+V | SCA + SCV | 51 pat. (w/ suspected TOS) | Arm abd. vs adducted | Diameter |
Acronyms: Modalities: CT = computed tomography; DSA = digital subtraction angiography; MMOD = multiple modalities; MRI = magnetic resonance imaging; US = ultrasound. System: ART = arterial; VEN = venous; A+V = arterial and venous. Subjects: CVD = chronic venous disease; insuf. = insufficiency; pat. = patients. vol. = healthy volunteers. PAD = peripheral arterial disease; TOS = thoracic outlet syndrome. Vessels: AA = abdominal aorta; ACV = anterior condylar vein; ATA = anterior tibial artery; ATV = anterior tibial vein; AV = azygos vein; BA = Brachial artery; CCA = common carotid artery; CeA = celiac artery; CFA = common femoral artery; CFV = common femoral vein; CIA = common iliac artery; CIV = common iliac vein; DCV = deep calf vein; EJV = external jugular vein; FV = femoral vein; GSV = great saphenous vein; GV = gluteal veins; HV = hepatic vein; ICA = internal carotid artery; IJV = internal jugular vein; inf. = inferior; IVC = inferior vena cava; LRV = left renal vein; NV = nuchal veins; PeV = peroneal vein; PoA = popliteal artery; PoV = popliteal vein; PTV = posterior tibial vein; PuA = pulmonary artery; PuV = pulmonary vein; PV = portal vein; SCA = subclavian artery; SCV = subclavian vein; SFA = superficial femoral artery; SFV = superficial femoral vein; SSV = small saphenous vein; SuA = subclavian artery; sup. = superior; SuV = subclavian vein; SV = spermatic vein (also; varicocele vein / testicular vein); VA = vertebral artery; VC = vena cava; VP = veins of the vertebral plexus; VV = vertebral vein. Positions: abd. = abduction; ABER = abduction and external rotation; CL = contralateral; er. = external rotation; ex. = extension; flex. = flexion; HUT = head-up tilting; IL = ipsilateral; LDP = lateral decubitus position; LL = left lateral; R-TB = reverse Trendelenburg; RL = right lateral; TB = Trendelenburg; WB = weight bearing. Outcomes: CSA = cross-sectional area
Fig. 2.
Included studies categorized by location and divided by the vascular system and modality and imaging modality. A+V = arterial and venous; CT = computed tomography; DSA = digital subtraction angiography; MMOD = multiple imaging modalities; MRI = magnetic resonance imaging; US = ultrasound
Abdomen
In the abdominal region, US was the most frequently reported imaging modality in different body postures and positions with 13 out of 19 articles [15–27]. Of these thirteen studies, the majority focused on the inferior vena cava (IVC) diameter that increased when going from supine to prone position by approximately 0.2 cm [15] and increased further with 0.2 cm from prone to standing position [27]. A 45° R-TB position did not influence the IVC diameter compared to supine position [16]. Interestingly, a left lateral position increased IVC diameters in a healthy population [17] and decreased diameters in elective cesarean section women [18]. Furthermore, the spermatic vein in patients with (suspected) varicocele increased in diameter in upright relative to supine position [19–21]. The common iliac vein diameter increased in R-TB [22] and upright position [23, 24]. For the right hepatic vein and portal vein dimensions, no correlation was found between supine and TB positions [26]. In the arterial system, only the celiac artery diameter was investigated for positional changes using US, which increased in standing position compared to supine [25].
MRI was used to study abdominal vasculature in five studies, mainly comparing upright and prone positions. The portal vein CSA decreased from supine to upright position [7]. Vena cava collapsibility was relatively high in upright position, whereas aorta CSA did not differ between supine and upright position at multiple aortic levels on MRI [28] and CT [6]. In obese pregnant women, R-TB position increased the aorta and IVC CSA by reducing vessel compression [29]. Two studies investigated common iliac artery diameters of which one (n = 97) did not find relevant differences in anatomical vascular orientation between prone and supine position [30], while the other (n = 7) observed shortening, higher curvature, and superior translation of the common iliac arteries in a hip flexion position [13].
Thorax
Seven studies reported on position-dependent imaging of thoracic vasculature on either CT or MRI, of which five compared prone and supine position. The distance between the IVC and spine was smaller in prone position than in supine position [12]. The aorta-spine distances in patients with spine deformities were described to decrease by approx. 3 mm (n = 20, n = 200, and n = 26) [12, 31, 32] or stay equal (n = 22) [33] in prone position. The pulmonary veins showed increased CSA in prone position (patients with atrial fibrillation) and in lateral position (volunteers) [9, 34, 35], but CSA of pulmonary arteries was not affected by body position [34].
Head-neck
The internal jugular vein (IJV) was extensively investigated using US in supine and upright positions, and its diameter and CSA were found to be around 0.90 cm2 in supine position and decreased or completely collapsed in upright position [36–46], which appeared to be more pronounced for the right IJV than the left IJV [37]. Similar results were found on the other imaging modalities MRI, CT, and DSA [47–49]. Other papers found that the IJV diameter and CSA increased up to 1.85 cm2 in TB compared to supine (n = 360) [41, 50–55], although this was not seen by Boschert et al [56] for both IJV and external jugular vein (n = 11). Additionally, the IJV CSA increased in prone position [57], with leg elevation [54] and with flexion of the head [58], while the IJV CSA decreased in R-TB position [54] and by rotating the head to the ipsilateral side [58], all compared to a neutral supine position. The external jugular vein and vertebral vein showed a diameter and CSA decrease in upright position compared to supine and TB positions [36, 37].
In the arterial system, the internal carotid artery (ICA) underwent no significant changes between supine and upright position on MRI [48]. According to Montero et al [59], the ICA and vertebral artery diameter increased in TB on US (n = 10), but this increase was not found by Yu et al (n = 28) [60]. No change in ICA or vertebral artery diameter was seen in R-TB position compared to supine [59]. For the common carotid artery, an upright position resulted in a diameter decrease from 6.7 to 6.5 mm relative to a supine position [61]. Maximal head rotation to the left had no effect on the right common carotid artery CSA compared to a neutral head position when investigated with US [62], while other MRI studies reported significant diameter change with head rotation, although without a specific tendency towards CSA increase or decrease [3, 63–66]. Holtackers et al [64] revealed that the difference between systolic and diastolic carotid artery diameters decreased when the volunteers’ heads were extended and rotated to the left. Ishida et al [67] investigated the influence of R-TB and TB on IJV, ICA, and vertebral artery CSA with MRI, where only the IJV diameter was found to significantly increase in TB compared to a supine position. Park et al [68] found that the distance between the common carotid artery and anterior C6 tubercle increased with increasing contralateral neck rotation.
Peripheral
There were 34 articles in the peripheral category that used US as main imaging modality. Twenty-nine of these focused on lower-extremity vasculature in neutral supine position versus upright (standing), various degrees of TB and R-TB, hip flexion, knee flexion, elevated legs with multiple increments, sitting, and ankle plantar flexion [69–97].
The femoral vein diameter and CSA increased (up to higher than 50 mm2) in R-TB, knee flexion (sitting position), upright position, and hip flexion up to 60° compared to a supine position (approx. 28 mm2) [69–77]. The great saphenous vein, tibial vein, and small saphenous vein diameter and CSA increased in upright and R-TB positions compared to supine and prone positions [77–82, 88]. Only Yang et al [80] did not find measurable differences in tibial vein and small saphenous vein diameters in upright position compared to prone and supine positions (n = 9). Popliteal vein diameter and CSA were larger in upright or sitting positions than in supine and prone position, and in postures with leg elevation [72, 83, 84] or leg flexion [85], but did not differ between standing and kneeling positions [86, 87].
The anterior tibial artery diameter was not significantly different between supine and TB or R-TB positions [95], similar to the femoral artery diameter which did not differ between supine, seated, standing, and R-TB positions [96, 97]. Also, the popliteal artery did not show significant differences in diameter in TB or R-TB positions compared to supine or prone [89–93], but popliteal artery diameter did decrease with plantar ankle flexion because of calf muscle compression [94, 98]. Aside from US, seven studies investigating popliteal artery or superficial femoral artery using CT and angiography found that leg flexion shortens artery length up to 12% and increases curvature up to 100% [99–104]. On MRI, the superficial femoral artery shortened in the lateral position with leg flexion [105], and inferior and superior gluteal vein diameters decreased in lateral position on the contralateral side [106]. Furthermore, Fujii et al [107] found on MRI that the small saphenous vein CSA was significantly larger in the sitting and standing positions than in supine position.
In the upper extremity, six studies investigated the brachial artery [96, 108], axillary artery [109], subclavian artery [5, 110], and subclavian vein [111]. Different postures had no significant effect on brachial artery diameter [96, 108] or subclavian artery diameter and CSA in volunteers [110]. However, in subjects with (suspected) TOS, the subclavian artery CSA decreased or even occluded in a posture of combined head rotation with shoulder abduction and in abduction and exorotation investigated with CT and MRI [5, 112–114]. Furthermore, the axillary artery diameter decreased in a combined abduction, horizontal flexion, and external rotated posture [109].
Discussion
This scoping review provided an overview of the currently available imaging modalities for visualization and characterization of human vascular anatomy in different body positions and postures. In this section, we will focus solely on the overall concept of different postures and positions rather than zooming in on specific body positions as the included literature covers a broad spectrum. Different postures (flexion, rotation, abduction, etc.) only make up 16% of the found papers, while different body positions (prone, upright, TB, etc.) were fairly well investigated (84%). The majority of the papers used US as imaging modality in different body positions and postures. Of these, a large portion investigated peripheral vasculature and found that the dimensions of lower-extremity veins decrease in positions with elevation of the feet. Similar observations of decreasing vein diameters were made for US examinations of the head and neck in head elevating positions. Diameter and CSA were the most commonly investigated outcome measures, which is related to a limited field of view in US that allows only for in-plane assessment of such measures [115]. Other modalities also allow assessment of geometric characteristics like vessel length, angulation, and distances between anatomical structures. This provides additional relevant information for clinical assessment that can be relevant in, for example, (positional) surgery planning [12].
Diameters and CSAs in the venous system appeared to be more sensitive to positional changes like upright, prone, R-TB/TB, and lateral positions than in the arterial. The difference between the venous and arterial positional deformation can be attributed to the structure of the vessel wall, which is more proliferated and thick for arteries than it is for veins. In our opinion, however, curvature and other geometrical parameters of the arterial and venous systems are similarly affected by postural changes such as flexion and rotation. With the increasing number of endovascular treatments of peripheral arterial occlusive disease and aortic aneurysms, it would be of interest to quantify the geometrical deformation of the target vessels so this can be taken into account in treatment planning and stent manufacturing. Geometrical deformation by positional and/or postural changes leads to different forces, torsion, or shear stresses along a vessel and the in situ stents [116]. This would especially be of interest in arteries (and veins) in body parts that allow for more movement, for example, the femoral and popliteal arteries during leg and hip flexion, but also the carotid, vertebral, and axillary arteries during head rotation. Characterizing the deformation will aid in optimization of stent placement and identification, and thereby reduce the risk for complications such as stent kinking and fractures [13, 65, 75]. Moreover, the deformation of these structures in different postures could be used as input for patient-specific computational fluid dynamics analysis to identify deviating flow patterns that could lead to adverse events [117]. Position-dependent imaging could provide essential information in treatment procedures such as spine surgery [12, 31–33] or pulmonary vein isolation therapy [9, 34, 35].
The search results in this review also held papers discussing several syndromes that relate to an aberrant vascular anatomy due to body positions and postures, such as TOS and Bow Hunter’s syndrome. Since most of these syndromes are fairly rare and presented as case reports and/or without proper outcome measures, they may need independent consideration. Bow Hunter’s syndrome studies were excluded from the present review considering the large number of papers available regarding this syndrome and already available reviews [10, 11]. In this syndrome, neck rotation or extension causes mechanical compression of the vertebral artery, most commonly caused by an osteophyte. All imaging modalities that were discussed in this review are widely used in the diagnosis and evaluation of this syndrome, with digital subtraction angiography considered as the gold standard. Nonetheless, both TOS and Bow Hunter’s syndrome are important examples of how different postures can affect vessel geometry.
Subjects in the included studies were predominantly healthy volunteers or patients scheduled for diagnostic imaging or (elective) surgery that was not related to any vascular pathology. Even though the results of these studies give a proper indication of the vessel deformation in different positions and postures, it should be taken into account that the behavior of diseased vessels may differ when the treatment plan is determined. Furthermore, based on the articles that were evaluated in this review, a wide and heterogeneous range of clinical purposes was observed that relate the vessel deformation to diagnosis or treatment outcome. Physicians should be aware that the orientation in which vessels are being imaged in the preprocedural phase is not representative of the dynamic forces during everyday movement. The vessels undergo ever-changing deformation due to the different positions and postures a person adopts during the day.
Limitations
The present review focused on anatomical and geometrical deformation of vasculature in different body positions or postures and did not evaluate functional parameters such as blood flow measurements with duplex US. In clinical practice, evaluation of such functional parameters may be relevant as well. A separate review on functional vascular imaging in different body positions and postures may be advised.
Conclusion
Vascular geometry in different body positions and postures was predominately evaluated with the outcome measures vessel diameter and CSA using US as imaging modality. Positional changes were more often evaluated than postural changes. Venous diameters and CSA were generally more sensitive to positional changes like upright, prone, R-TB/TB, and lateral positions than the arterial equivalents. However, curvature and other geometrical parameters of the arterial and venous systems are equally affected by postural changes (e.g., flexion, abduction, rotation), which was often evaluated on CT or MRI, rather than US. The most important clinical implications of positional changes are found in diagnosis, surgical planning, and in the stent placement and follow-up. However, the knowledge of the influence of body positions and postures on the vasculature and how these may affect treatment of vascular pathologies remains limited, such as the influence of bending of the knee on stent geometry in atherosclerotic popliteal arteries.
Supplementary information
(XML 3 kb)
Acknowledgements
The authors thank Judith Brands, PhD (information specialist faculty Science & Technology, University of Twente), for her advice on the review structure and search strategy.
Abbreviations
- A+V
Arterial and venous
- AA
Abdominal aorta
- abd.
Abduction
- ABER
Abduction and external rotation
- ACV
Anterior condylar vein
- ART
Arterial
- ASA
American Society of Anesthesiology classification score
- ATA
Anterior tibial artery
- ATV
Anterior tibial vein
- AV
Azygos vein
- BA
Brachial artery
- CCA
Common carotid artery
- CeA
Celiac artery
- CFA
Common femoral artery
- CFV
Common femoral vein
- CIA
Common iliac artery
- CIV
Common iliac vein
- CL
Contralateral
- CSA
Cross-sectional area
- CT
Computed tomography
- CTA
Computed tomography angiography
- CVD
Chronic venous disease
- DCV
Deep calf vein
- DSA
Digital subtraction angiography
- EJV
External jugular vein
- er.
External rotation
- ex.
Extension
- flex.
Flexion
- FV
Femoral vein
- GSV
Great saphenous vein
- GV
Gluteal veins
- HUT
Head-up tilting
- HV
Hepatic vein
- ICA
Internal carotid artery
- IJV
Internal jugular vein
- IL
Ipsilateral
- inf.
Inferior
- insuf.
Insufficiency
- IVC
Inferior vena cava
- LDP
Lateral decubitus position
- LL
Left lateral
- LRV
Left renal vein
- MMOD
Multiple modalities
- MRA
Magnetic resonance angiography
- MRI
Magnetic resonance imaging
- NV
Nuchal veins
- PAD
Peripheral arterial disease
- pat.
Patients
- PeV
Peroneal vein
- PoA
Popliteal artery
- PoV
Popliteal vein
- PTV
Posterior tibial vein
- PuA
Pulmonary artery
- PuV
Pulmonary vein
- PV
Portal vein
- RL
Right lateral
- R-TB
Reverse Trendelenburg
- SCA
Subclavian artery
- SCV
Subclavian vein
- SFV
Superficial femoral vein
- SSV
Small saphenous vein
- SuA
Subclavian artery
- sup.
Superior
- SuV
Subclavian vein
- SV
Spermatic vein (also varicocele vein/testicular vein)
- TB
Trendelenburg
- TOS
Thoracic outlet syndrome
- US
Ultrasound
- VAa
Vertebral artery
- VC
Vena cava
- VEN
Venous
- vol.
Healthy volunteers
- VP
Veins of the vertebral plexus
- VV
Vertebral vein
- WB
Weight bearing
Funding
The authors state that this work has not received any funding.
Declarations
Guarantor
The scientific guarantor of this publication is Prof. MD Robert H. Geelkerken.
Conflict of interest
The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was not required for this study because this study is based on a review of publicly available data and does not involve human or animal subjects
Ethical approval
Institutional Review Board approval was not required because this study is based on a review of publicly available data and does not involve human or animal subjects.
Study subjects or cohorts overlap
All study subjects and cohorts have been previously reported since this is a review.
Methodology
Performed at one institution
Footnotes
Jordy K. van Zandwijk and Jaimy A. Simmering are shared first authors on this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Hansen KL, Carlsen JF. New trends in vascular imaging. Diagnostics. 2021;11:4–7. doi: 10.3390/diagnostics11010112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chan P, Spudich S (2021) Investigating vascular diseases in people living with HIV by nuclear imaging. J Nucl Cardiol. 10.1007/s12350-021-02613-x [DOI] [PubMed]
- 3.Aristokleous N, Seimenis I, Papaharilaou Y, et al. Effect of posture change on the geometric features of the healthy carotid bifurcation. IEEE Trans Inf Technol Biomed. 2011;15:148–154. doi: 10.1109/TITB.2010.2091417. [DOI] [PubMed] [Google Scholar]
- 4.Cunningham KS, Gotlieb AI. The role of shear stress in the pathogenesis of atherosclerosis. Lab Investig. 2005;85:9–23. doi: 10.1038/labinvest.3700215. [DOI] [PubMed] [Google Scholar]
- 5.Demondion X, Vidal C, Herbinet P, et al. Ultrasonographic assessment of arterial cross-sectional area in the thoracic outlet on postural maneuvers measured with power Doppler ultrasonography in both asymptomatic and symptomatic populations. J Ultrasound Med. 2006;25:217–224. doi: 10.7863/jum.2006.25.2.217. [DOI] [PubMed] [Google Scholar]
- 6.Jinzaki M, Yamada Y, Nagura T, et al. Development of upright computed tomography with area detector for whole-body scans: phantom study, efficacy on workflow, effect of gravity on human body, and potential clinical impact. Invest Radiol. 2020;55:73–83. doi: 10.1097/RLI.0000000000000603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kadoya Y, Miyati T, Kobayashi S, et al. Effect of gravity on portal venous flow: Evaluation using multiposture MRI. J Magn Reson Imaging. 2019;50:83–87. doi: 10.1002/jmri.26626. [DOI] [PubMed] [Google Scholar]
- 8.Alperin N, Lee SH, Sivaramakrishnan A, Hushek SG. Quantifying the effect of posture on intracranial physiology in humans by MRI flow studies. J Magn Reson Imaging. 2005;22:591–596. doi: 10.1002/jmri.20427. [DOI] [PubMed] [Google Scholar]
- 9.Gottlieb LA, El Hamrani D, Naulin J, et al. A left lateral body position increases pulmonary vein stress in healthy humans. Physiol Rep. 2021;9:1–10. doi: 10.14814/phy2.15022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Duan G, Xu J, Shi J, Cao Y. Advances in the pathogenesis, diagnosis and treatment of Bow Hunter’s syndrome: a comprehensive review of the literature. Interv Neurol. 2016;5:29–38. doi: 10.1159/000444306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rastogi V, Rawls A, Moore O, et al. Rare etiology of Bow Hunter’s syndrome and systematic review of literature. J Vasc Interv Neurol. 2015;8:7–16. [PMC free article] [PubMed] [Google Scholar]
- 12.Riccio AR, Bishop A, Carl AL. Great vessel excursion: prone versus supine position. Int J Spine Surg. 2019;13:158–161. doi: 10.14444/6021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Choi G, Shin LK, Taylor CA, Cheng CP. In vivo deformation of the human abdominal aorta and common iliac arteries with hip and knee flexion: implications for the design of stent-grafts. J Endovasc Ther. 2009;16:531–538. doi: 10.1583/09-2806.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Page MJ, McKenzie JE, Bossuyt PM, et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372: 10.1136/bmj.n71 [DOI] [PMC free article] [PubMed]
- 15.Hensley J, Wang H (2019) Assessment of volume status during prone spine surgery via a novel point-of-care ultrasound technique. Cureus. 10.7759/cureus.4601 [DOI] [PMC free article] [PubMed]
- 16.Panebianco NL, Shofer F, Cheng A, et al. The effect of supine versus upright patient positioning on inferior vena cava metrics. Am J Emerg Med. 2014;32:1326–1329. doi: 10.1016/j.ajem.2014.07.036. [DOI] [PubMed] [Google Scholar]
- 17.Mookadam F, Warsame TA, Yang HS, et al. Effect of positional changes on inferior vena cava size. Eur J Echocardiogr. 2011;12:322–325. doi: 10.1093/ejechocard/jer018. [DOI] [PubMed] [Google Scholar]
- 18.Kundra P, Arunsekar G, Vasudevan A, et al. Effect of postural changes on inferior vena cava dimensions and its influence on haemodynamics during caesarean section under spinal anaesthesia. J Obstet Gynaecol. 2015;35:667–671. doi: 10.3109/01443615.2014.991291. [DOI] [PubMed] [Google Scholar]
- 19.Ebubedike U, Enukegwu S, Nwofor A. Ultrasound evaluation of testicular vein diameter in suspected cases of varicocele: comparison of measurements in supine and upright positions. Niger J Clin Pract. 2020;23:1004–1007. doi: 10.4103/njcp.njcp_579_19. [DOI] [PubMed] [Google Scholar]
- 20.Arshad MA, Siddiqui AA, Rehman KU. Subinguinal versus peritesticular diameters of varicocele veins in the supine and standing posture. J Pak Med Assoc. 2021;71:2766–2769. doi: 10.47391/JPMA.199. [DOI] [PubMed] [Google Scholar]
- 21.Najari BB, Katz MJ, Schulster ML, et al. Increased body mass index in men with varicocele is associated with larger spermatic vein diameters when supine. Urology. 2016;89:40–44. doi: 10.1016/j.urology.2015.11.033. [DOI] [PubMed] [Google Scholar]
- 22.Rist M, Hemmerling TM, Rauh R, et al. Influence of pneumoperitoneum and patient positioning on preload and splanchnic blood volume in laparoscopic surgery of the lower abdomen. J Clin Anesth. 2001;13:244–249. doi: 10.1016/S0952-8180(01)00242-2. [DOI] [PubMed] [Google Scholar]
- 23.Krzanowski M, Partyka L, Drelicharz L, et al. Posture commonly and considerably modifies stenosis of left common iliac and left renal veins in women diagnosed with pelvic venous disorder. J Vasc Surg Venous Lymphat Disord. 2019;7:845–852.e2. doi: 10.1016/j.jvsv.2019.05.009. [DOI] [PubMed] [Google Scholar]
- 24.Engelhorn ALDV, Lima LDB, Werka MJS, et al. Left common iliac vein compression identified by vascular ultrasonography in asymptomatic women: does standing position influence diagnosis? J Vasc Bras. 2021;20:1–10. doi: 10.1590/1677-5449.200188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Asbeutah AM, Al-Hussaini AJ, Al-Otaibi JA, et al. Patient position and phase of respiration affect the doppler waveform in the celiac artery. J Vasc Ultrasound. 2010;34:21–26. doi: 10.1177/154431671003400105. [DOI] [Google Scholar]
- 26.Ukere A, Meisner S, Greiwe G, et al. The influence of PEEP and positioning on central venous pressure and venous hepatic hemodynamics in patients undergoing liver resection. J Clin Monit Comput. 2017;31:1221–1228. doi: 10.1007/s10877-016-9970-1. [DOI] [PubMed] [Google Scholar]
- 27.Ho JD, Dawes DM, Moore JC, et al. Effect of position and weight force on inferior vena cava diameter - implications for arrest-related death. Forensic Sci Int. 2011;212:256–259. doi: 10.1016/j.forsciint.2011.07.001. [DOI] [PubMed] [Google Scholar]
- 28.Kadoya Y, Miyati T, Kobayashi S, et al. Evaluation of gravity effect on inferior vena cava and abdominal aortic flow using multi-posture MRI. Acta Radiol. 2021;62:1122–1128. doi: 10.1177/0284185120950112. [DOI] [PubMed] [Google Scholar]
- 29.Saravanakumar K, Hendrie M, Smith F, Danielian P. Influence of reverse Trendelenburg position on aortocaval compression in obese pregnant women. Int J Obstet Anesth. 2016;26:15–18. doi: 10.1016/j.ijoa.2015.09.007. [DOI] [PubMed] [Google Scholar]
- 30.Ganesan C, Petrus L, Ross IB. Regarding the possibility of anterior vascular injury from the posterior approach to the lumbar disc space: an anatomical study. Spine (Phila Pa 1976) 2012;37:1371–1375. doi: 10.1097/BRS.0b013e318267fb36. [DOI] [PubMed] [Google Scholar]
- 31.Plataniotis N, Evangelopoulos DS, Katzouraki G, Pneumaticos S. The effect of patient positioning on the relative position of the aorta to the thoracic spine. Eur Spine J. 2019;28:477–483. doi: 10.1007/s00586-018-5812-9. [DOI] [PubMed] [Google Scholar]
- 32.Jiang H, Qiu X, Wang W et al (2012) The position of the aorta changes with altered body position in single right thoracic adolescent idiopathic scoliosis: a magnetic resonance imaging study. Spine (Phila Pa 1976):37. 10.1097/BRS.0b013e3182600a7d [DOI] [PubMed]
- 33.Qian B-P, Qu Z, Ji M, et al. Does the position of the aorta change with the altered body position in ankylosing spondylitis patients with thoracolumbar kyphosis? A magnetic resonance imaging investigation. Glob. Spine J. 2015;5:s-0035-1554245–s-0035-1554245. doi: 10.1055/s-0035-1554245. [DOI] [PubMed] [Google Scholar]
- 34.Wieslander B, Ramos JG, Ax M, et al. Supine, prone, right and left gravitational effects on human pulmonary circulation. J Cardiovasc Magn Reson. 2019;21:1–15. doi: 10.1186/s12968-019-0577-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yamaji H, Hina K, Kawamura H, et al. Prone position is essential for detection of pulmonary vein pseudostenosis by enhanced multidetector computed tomography in patients who undergo pulmonary vein isolation. Circ J. 2008;72:1460–1464. doi: 10.1253/circj.CJ-08-0055. [DOI] [PubMed] [Google Scholar]
- 36.Morimoto A, Takase I, Shimizu Y, Nishi K. Assessment of cervical venous blood flow and the craniocervical venus valve using ultrasound sonography. Leg Med. 2009;11:10–17. doi: 10.1016/j.legalmed.2008.07.006. [DOI] [PubMed] [Google Scholar]
- 37.Kantarci F, Albayram S, Demirci NO, et al. Chronic cerebrospinal venous insufficiency: does ultrasound really distinguish multiple sclerosis subjects from healthy controls? Eur Radiol. 2012;22:970–979. doi: 10.1007/s00330-011-2338-5. [DOI] [PubMed] [Google Scholar]
- 38.Gisolf J, van Lieshout JJ, van Heusden K, et al. Human cerebral venous outflow pathway depends on posture and central venous pressure. J Physiol. 2004;560:317–327. doi: 10.1113/jphysiol.2004.070409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Holmlund P, Johansson E, Qvarlander S, et al. Human jugular vein collapse in the upright posture: implications for postural intracranial pressure regulation. Fluids Barriers CNS. 2017;14:17. doi: 10.1186/s12987-017-0065-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wang W, Liao X, Chen ECS, et al. The effects of positioning on the volume/location of the internal jugular vein using 2-dimensional tracked ultrasound. J Cardiothorac Vasc Anesth. 2020;34:920–925. doi: 10.1053/j.jvca.2019.08.049. [DOI] [PubMed] [Google Scholar]
- 41.Watkins W, Hargens AR, Seidl S, et al. Lower-body negative pressure decreases noninvasively measured intracranial pressure and internal jugular vein cross-sectional area during head-down tilt. J Appl Physiol (1985) 2017;123:136–146. doi: 10.1152/JAPPLPHYSIOL.00091.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Marshall-Goebel K, Macias BR, Laurie SS, et al. Mechanical countermeasures to headward fluid shifts. J Appl Physiol (1985) 2021;130:1766–1777. doi: 10.1152/japplphysiol.00863.2020. [DOI] [PubMed] [Google Scholar]
- 43.Westlund A, Holmlund P, Johansson E et al (2019) Semi-automatic method for segmentation of the internal jugular vein in ultrasound movies evaluated at different body postures. Biomed Phys Eng Express 5. 10.1088/2057-1976/ab285e
- 44.Holmlund P, Eklund A, Koskinen L-OD, et al. Venous collapse regulates intracranial pressure in upright body positions. Am J Physiol Integr Comp Physiol. 2017;314:R377–R385. doi: 10.1152/ajpregu.00291.2017. [DOI] [PubMed] [Google Scholar]
- 45.Mayer CA, Pfeilschifter W, Lorenz MW, et al. The perfect crime? CCSVI not leaving a trace in MS. J Neurol Neurosurg Psychiatry. 2011;82:436–440. doi: 10.1136/jnnp.2010.231613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Doepp F, Würfel JT, Pfueller CF, et al. Venous drainage in multiple sclerosis: a combined MRI and ultrasound study. Neurology. 2011;77:1745–1751. doi: 10.1212/WNL.0b013e318236f0ea. [DOI] [PubMed] [Google Scholar]
- 47.van Zandwijk JK, Kuijer KM, Stassen CM et al (2022) Internal jugular vein geometry under multiple inclination angles with 3D low-field MRI in healthy volunteers. J Magn Reson Imaging 1–7. 10.1002/jmri.28182 [DOI] [PMC free article] [PubMed]
- 48.Kosugi K, Yamada Y, Yamada M, et al. Posture-induced changes in the vessels of the head and neck: evaluation using conventional supine CT and upright CT. Sci Rep. 2020;10:1–12. doi: 10.1038/s41598-020-73658-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Qureshi AI, Saleem MA. Upright catheter-based cerebral angiography. J Vasc Interv Neurol. 2017;9:14–19. [PMC free article] [PubMed] [Google Scholar]
- 50.Lee JG, Bin PH, Shin HY, et al. Effect of trendelenburg position on right and left internal jugular vein cross-sectional area. Korean J Anesthesiol. 2014;67:305–309. doi: 10.4097/kjae.2014.67.5.305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Jo YY, Kim HS, Lee MG, et al. Effects of incrementally increasing tidal volume on the right internal jugular vein in pediatric patients. Korean J Anesthesiol. 2013;65:312–316. doi: 10.5301/jva.5000362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Chen K, Wang L, Wang Q et al (2019) Effects of pneumoperitoneum and steep Trendelenburg position on cerebral hemodynamics during robotic-assisted laparoscopic radical prostatectomy: a randomized controlled study. Medicine (Baltimore) 98:e15794 10.1097/MD.0000000000015794 [DOI] [PMC free article] [PubMed]
- 53.Kim JT, Kim HS, Lim YJ, et al. The influence of passive leg elevation on the cross-sectional area of the internal jugular vein and the subclavian vein in awake adults. Anaesth Intensive Care. 2008;36:65–68. doi: 10.1177/0310057x0803600111. [DOI] [PubMed] [Google Scholar]
- 54.Judickas Š, Gineitytė D, Kezytė G, et al. Is the Trendelenburg position the only way to better visualize internal jugular veins? Acta Med Litu. 2019;25:125–131. doi: 10.6001/actamedica.v25i3.3859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Okamura K, Nomura T, Mizuno Y, et al. Pre-anesthetic ultrasonographic assessment of the internal jugular vein for prediction of hypotension during the induction of general anesthesia. J Anesth. 2019;33:612–619. doi: 10.1007/s00540-019-02675-9. [DOI] [PubMed] [Google Scholar]
- 56.Boschert AL, Elmenhorst D, Gauger P, et al. Sleep is compromised in −12° head down tilt position. Front Physiol. 2019;10:1–10. doi: 10.3389/fphys.2019.00397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Yeoh TY, Tan A, Manninen P, et al. Effect of different surgical positions on the cerebral venous drainage: a pilot study using healthy volunteers. Anaesthesia. 2016;71:806–813. doi: 10.1111/anae.13494. [DOI] [PubMed] [Google Scholar]
- 58.Muhammad JK, Pugh ND, Boden L, et al. The effect of head rotation on the diameter of the internal jugular vein: Implications for free tissue transfer. J Craniomaxillofacial Surg. 2001;29:214–218. doi: 10.1054/jcms.2001.0225. [DOI] [PubMed] [Google Scholar]
- 59.Montero D, Rauber S. Brain perfusion and arterial blood flow velocity during prolonged body tilting. Aerosp Med Hum Perform. 2016;87:682–687. doi: 10.3357/AMHP.4546.2016. [DOI] [PubMed] [Google Scholar]
- 60.Yu J, Park JY, Hong JH, et al. Effect of pneumoperitoneum and Trendelenburg position on internal carotid artery blood flow measured by ultrasound during robotic prostatectomy. Clin Physiol Funct Imaging. 2022;42:139–145. doi: 10.1111/cpf.12742. [DOI] [PubMed] [Google Scholar]
- 61.Saeed NP, Reneman RS, Hoeks APG. Contribution of vascular and neural segments to baroreflex sensitivity in response to postural stress. J Vasc Res. 2009;46:469–477. doi: 10.1159/000200962. [DOI] [PubMed] [Google Scholar]
- 62.Glor FP, Ariff B, Hughes AD et al (2004) Influence of head position on carotid hemodynamics in young adults. Am J Physiol Hear Circ Physiol 287. 10.1152/ajpheart.01186.2003 [DOI] [PubMed]
- 63.Papaharilaou Y, Aristokleous N, Seimenis I, et al. Effect of head posture on the healthy human carotid bifurcation hemodynamics. Med Biol Eng Comput. 2013;51:207–218. doi: 10.1007/s11517-012-0985-6. [DOI] [PubMed] [Google Scholar]
- 64.Holtackers RJ, Spronck B, Heusinkveld MHG, et al. Head orientation should be considered in ultrasound studies on carotid artery distensibility. J Hypertens. 2016;34:1551–1555. doi: 10.1097/HJH.0000000000000985. [DOI] [PubMed] [Google Scholar]
- 65.Vos AWF, Linsen MAM, Marcus JT, et al. Carotid artery dynamics during head movements: a reason for concern with regard to carotid stenting? J Endovasc Ther. 2003;10:862–869. doi: 10.1583/1545-1550(2003)010<0862:CADDHM>2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- 66.Aristokleous N, Seimenis I, Papaharilaou Y, et al (2012) Head posture influences the geometric and hemodynamic features on the healthy human carotid bifurcation. IEEE 12th Int Conf Bioinforma Bioeng BIBE 2012 727–731. 10.1109/BIBE.2012.6399757
- 67.Ishida S, Miyati T, Ohno N, et al. MRI-based assessment of acute effect of head-down tilt position on intracranial hemodynamics and hydrodynamics. J Magn Reson Imaging. 2018;47:565–571. doi: 10.1002/jmri.25781. [DOI] [PubMed] [Google Scholar]
- 68.Park DY, Kang S, Kang HJ, et al. Impact of neck position on the probability of common carotid artery puncture during ultrasound-guided stellate ganglion block. PM R. 2019;11:463–469. doi: 10.1016/j.pmrj.2018.08.376. [DOI] [PubMed] [Google Scholar]
- 69.Lurie F, Ogawa T, Kistner RL, Eklof B. Changes in venous lumen size and shape do not affect the accuracy of volume flow measurements in healthy volunteers and patients with primary chronic venous insufficiency. J Vasc Surg. 2002;35:522–526. doi: 10.1067/mva.2002.121565. [DOI] [PubMed] [Google Scholar]
- 70.Nguyen NT, Cronan M, Braley S, et al. Duplex ultrasound assessment of femoral venous flow during laparoscopic and open gastric bypass. Surg Endosc Other Interv Tech. 2003;17:285–290. doi: 10.1007/s00464-002-8812-z. [DOI] [PubMed] [Google Scholar]
- 71.Limpus A, Chaboyer WP, Purcell C, et al. Effect of body position and graduated compression stocking length on femoral venous blood flow velocity. Phlebology. 2003;18:198–202. doi: 10.1258/026835503322598027. [DOI] [Google Scholar]
- 72.Delis KT, Knaggs AL, Sonecha TN, et al. Lower limb venous haemodynamic impairment on dependency: quantification and implications for the “economy class” position. Thromb Haemost. 2004;91:941–950. doi: 10.1160/th03-12-0754. [DOI] [PubMed] [Google Scholar]
- 73.Warwick DJ, Shaikh A, Gadola S, et al. Neuromuscular electrostimulation via the common peroneal nerve promotes lower limb blood flow in a below-knee cast. Bone Joint Res. 2013;2:179–185. doi: 10.1302/2046-3758.29.2000176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.van Rij AM, De Alwis CS, Jiang P, et al. Obesity and impaired venous function. Eur J Vasc Endovasc Surg. 2008;35:739–744. doi: 10.1016/j.ejvs.2008.01.006. [DOI] [PubMed] [Google Scholar]
- 75.Lattimer CR, Mendoza E. Reappraisal of the utility of the tilt-table in the investigation of venous disease†. Eur J Vasc Endovasc Surg. 2016;52:854–861. doi: 10.1016/j.ejvs.2016.09.012. [DOI] [PubMed] [Google Scholar]
- 76.Pannucci CJ, Henke PK, Cederna PS, et al. The effect of increased hip flexion using stirrups on lower-extremity venous flow: a prospective observational study. Am J Surg. 2011;202:427–432. doi: 10.1016/j.amjsurg.2011.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Pemble L (2006) Lower extremity venous cross-sectional area changes associated with pregnancy. J Vasc Ultrasound 30(2):75–80. 10.1177/154431670603000203
- 78.DeMuth RP, Caylor K, Walton T, et al. Clinical significance of standing versus reversed trendelenburg position for the diagnosis of lower-extremity venous reflux in the great saphenous vein. J Vasc Ultrasound. 2012;36:19–22. doi: 10.1177/154431671203600103. [DOI] [Google Scholar]
- 79.Hoballah JJ, Corry DC, Rossley N, et al. Duplex saphenous vein mapping: venous occlusion and dependent position facilitate imaging. Vasc Endovascular Surg. 2002;36:377–380. doi: 10.1177/153857440203600507. [DOI] [PubMed] [Google Scholar]
- 80.Yang A, Yasaman B, Parsi Y, et al. Two layers of graduated compression stockings can reduce healthy saphenous vein diameters in the standing position. Phlebology. 2019;34:559–565. doi: 10.1177/0268355519828871. [DOI] [PubMed] [Google Scholar]
- 81.Cirovic S, Walsh C, Fraser WD, Gulino A. Venous filling and elastance in the calf positioned above and below heart level. Aviat Sp Environ Med. 2006;77:1009–1014. [PubMed] [Google Scholar]
- 82.Tauraginskii RA, Lurie F, Simakov S, et al. Gravity force is not a sole explanation of reflux flow in incompetent great saphenous vein. J Vasc Surg Venous Lymphat Disord. 2019;7:693–698. doi: 10.1016/j.jvsv.2019.04.012. [DOI] [PubMed] [Google Scholar]
- 83.Dix FP, Reilly B, David MC, et al. Effect of leg elevation on healing, venous velocity and ambulatory venous pressure in venous ulceration. Phlebology. 2005;20:87–94. doi: 10.1258/0268355054069179. [DOI] [Google Scholar]
- 84.Morita H, Abe C, Tanaka K, et al. Neuromuscular electrical stimulation and an Ottoman-type seat effectively improve popliteal venous flow in a sitting position. J Physiol Sci. 2006;56:183–186. doi: 10.2170/physiolsci.SC002006. [DOI] [PubMed] [Google Scholar]
- 85.Levine A, Huber J, Huber D. Changes in popliteal vein diameter and flow velocity with knee flexion and hyperextension. Phlebology. 2011;26:307–310. doi: 10.1258/phleb.2010.010035. [DOI] [PubMed] [Google Scholar]
- 86.Ciufo DJ, Anderson MR, Baumhauer JF. Impact of knee scooter flexion position on venous flow rate. Foot Ankle Int. 2019;40:80–84. doi: 10.1177/1071100718794966. [DOI] [PubMed] [Google Scholar]
- 87.Reb CW, Haupt ET, Vander Griend RA, Berlet GC (2021) Pedal musculovenous pump activation effectively counteracts negative impact of knee flexion on human popliteal venous flow. Foot Ankle Spec. 10.1177/1938640021997275 [DOI] [PubMed]
- 88.Mestre S, Triboulet J, Demattei C, et al. Acute effects of graduated and progressive compression stockings on leg vein cross-sectional area and viscoelasticity in patients with chronic venous disease. J Vasc Surg Venous Lymphat Disord. 2022;10:186–195.e25. doi: 10.1016/j.jvsv.2021.03.021. [DOI] [PubMed] [Google Scholar]
- 89.Villar R, Hughson RL. Effect of altered arterial perfusion pressure on vascular conductance and muscle blood flow dynamic response during exercise in humans. J Appl Physiol (1985) 2013;114:620–627. doi: 10.1152/japplphysiol.01094.2012. [DOI] [PubMed] [Google Scholar]
- 90.Villar R, Hughson RL. Lower limb vascular conductance and resting popliteal blood flow during head-up and head-down postural challenges. Clin Physiol Funct Imaging. 2013;33:186–191. doi: 10.1111/cpf.12008. [DOI] [PubMed] [Google Scholar]
- 91.Villar R, Hughson RL. Repeatability of popliteal blood flow and lower limb vascular conductance at rest and exercise during body tilt using Doppler ultrasound. Physiol Meas. 2013;34:291–306. doi: 10.1088/0967-3334/34/3/291. [DOI] [PubMed] [Google Scholar]
- 92.Villar R, Hughson RL. Vascular conductance and muscle blood flow during exercise are altered by inspired oxygen fraction and arterial perfusion pressure. Physiol Rep. 2017;5:1–11. doi: 10.14814/phy2.13144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Delis KT, Nicolaides AN, Stansby G. Effect of posture on popliteal artery hemodynamics. Arch Surg. 2000;135:265–269. doi: 10.1001/archsurg.135.3.265. [DOI] [PubMed] [Google Scholar]
- 94.Brown CD, Muniz M, Kauvar DS. Response of the popliteal artery to treadmill exercise and stress positioning in patients with and without exertional lower extremity symptoms. J Vasc Surg. 2019;69:1545–1551. doi: 10.1016/j.jvs.2018.08.171. [DOI] [PubMed] [Google Scholar]
- 95.Becker RL, Siamwala JH, Macias BR, Hargens AR. Tibia bone microvascular flow dynamics as compared to anterior tibial artery flow during body tilt. Aerosp Med Hum Perform. 2018;89:357–364. doi: 10.3357/AMHP.4928.2018. [DOI] [PubMed] [Google Scholar]
- 96.Newcomer SC, Sauder CL, Kuipers NT et al (2008) Effects of posture on shear rates in human brachial and superficial femoral arteries. Am J Physiol Hear Circ Physiol 294. 10.1152/ajpheart.01108.2007 [DOI] [PMC free article] [PubMed]
- 97.Groothuis JT, Boot CRL, Houtman S, et al. Does peripheral nerve degeneration affect circulatory responses to head-up tilt in spinal cord-injured individuals? Clin Auton Res. 2005;15:99–106. doi: 10.1007/s10286-005-0248-9. [DOI] [PubMed] [Google Scholar]
- 98.Breen MA, Hassan MM, Johnston P, et al. Quantification of popliteal artery narrowing with QISS MRA during active ankle plantarflexion in healthy, asymptomatic volunteers and its potential application in the diagnosis of popliteal artery entrapment syndrome (PAES) Skeletal Radiol. 2021;50:2091–2102. doi: 10.1007/s00256-021-03751-6. [DOI] [PubMed] [Google Scholar]
- 99.Diaz JA, Villegas M, Tamashiro G, et al. Flexions of the popliteal artery: dynamic angiography712. J Invasive Cardiol. 2004;16:712–715. [PubMed] [Google Scholar]
- 100.Gökgöl C, Diehm N, Kara L, Büchler P. Quantification of popliteal artery deformation during leg flexion in subjects with peripheral artery disease: a pilot study. J Endovasc Ther. 2013;20:828–835. doi: 10.1583/13-4332MR.1. [DOI] [PubMed] [Google Scholar]
- 101.Gökgöl C, Schumann S, Diehm N, et al. In vivo quantification of the deformations of the femoropopliteal segment. J Endovasc Ther. 2017;24:27–34. doi: 10.1177/1526602816677530. [DOI] [PubMed] [Google Scholar]
- 102.Klein AJ, Chen SJ, Messenger JC, et al. Quantitative assessment of the conformational change in the femoropopliteal artery with leg movement. Catheter Cardiovasc Interv. 2009;74:787–798. doi: 10.1002/ccd.22124. [DOI] [PubMed] [Google Scholar]
- 103.Rabtsun A, Karpenko A, Zoloev DG, et al. Remote endarterectomy and lamina vastoadductoria dissection improves superficial femoral artery biomechanical behavior during limb flexion. Ann Vasc Surg. 2018;50:112–118. doi: 10.1016/j.avsg.2017.12.007. [DOI] [PubMed] [Google Scholar]
- 104.Spinella G, Finotello A, Pane B, et al. In vivo morphological changes of the femoropopliteal arteries due to knee flexion after endovascular treatment of popliteal aneurysm. J Endovasc Ther. 2019;26:496–504. doi: 10.1177/1526602819855441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Cheng CP, Choi G, Herfkens RJ, Taylor CA. The effect of aging on deformations of the superficial femoral artery resulting from hip and knee flexion: potential clinical implications. J Vasc Interv Radiol. 2010;21:195–202. doi: 10.1016/j.jvir.2009.08.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Turin SY, Fracol M, Keller E, et al. Gluteal vein anatomy: location, caliber, impact of patient positioning, and implications for fat grafting. Aesthetic Surg J. 2020;40:642–649. doi: 10.1093/asj/sjz260. [DOI] [PubMed] [Google Scholar]
- 107.Fujii T, Ohno N, Sawazaki T, et al. Gravity magnetic resonance imaging measurement of muscle pump change accompanied by aging and posture. Japan J Nurs Sci. 2021;18:1–8. doi: 10.1111/jjns.12407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Claydon VE, Moore JP, Greene ER, et al. Evaluation of forearm vascular resistance during orthostatic stress: velocity is proportional to flow and size doesn’t matter. PLoS One. 2019;14:1–11. doi: 10.1371/journal.pone.0224872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Stapleton CH, Lee H, George K. Anterior translation at the glenohumeral joint: a cause of axillary artery compression? Am J Sports Med. 2008;36:539–544. doi: 10.1177/0363546507309314. [DOI] [PubMed] [Google Scholar]
- 110.Stapleton C, Herrington L, George K. Sonographic evaluation of the subclavian artery during thoracic outlet syndrome shoulder manoeuvres. Man Ther. 2009;14:19–27. doi: 10.1016/j.math.2007.07.010. [DOI] [PubMed] [Google Scholar]
- 111.Sadek M, Roger C, Bastide S, et al. The influence of arm positioning on ultrasonic visualization of the subclavian vein: an anatomical ultrasound study in healthy volunteers. Anesth Analg. 2016;123:129–132. doi: 10.1213/ANE.0000000000001327. [DOI] [PubMed] [Google Scholar]
- 112.Park JY, Oh KS, Yoo HY, Lee JG. Case report: Thoracic outlet syndrome in an elite archer in full-draw position. Clin Orthop Relat Res. 2013;471:3056–3060. doi: 10.1007/s11999-013-2865-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.LaBan MM, Zierenberg AT, Yadavalli S, Zaidan S. Clavicle-induced narrowing of the thoracic outlet during shoulder abduction as imaged by computed tomographic angiography and enhanced by three-dimensional reformation. Am J Phys Med Rehabil. 2011;90:572–578. doi: 10.1097/PHM.0b013e31821a70ff. [DOI] [PubMed] [Google Scholar]
- 114.Charon JPM, Milne W, Sheppard DG, Houston JG. Evaluation of MR angiographic technique in the assessment of thoracic outlet syndrome. Clin Radiol. 2004;59:588–595. doi: 10.1016/j.crad.2003.11.020. [DOI] [PubMed] [Google Scholar]
- 115.Li X, Cokkinos D, Gadani S, et al. Advanced ultrasound techniques in arterial diseases. Int J Cardiovasc Imaging. 2022;38:1711–1721. doi: 10.1007/s10554-022-02558-3. [DOI] [PubMed] [Google Scholar]
- 116.Desyatova A, Poulson W, Deegan P et al (2017) Limb flexion-induced twist and associated intramural stresses in the human femoropopliteal artery. J R Soc Interface 14. 10.1098/rsif.2017.0025 [DOI] [PMC free article] [PubMed]
- 117.Zhong L, Zhang JM, Su B et al (2018) Application of patient-specific computational fluid dynamics in coronary and intra-cardiac flow simulations: challenges and opportunities. Front Physiol 9. 10.3389/fphys.2018.00742 [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(XML 3 kb)