Abstract
Background:
Neuromuscular deficits exist following anterior cruciate ligament (ACL) injury. To observe these deficits, we examined nonlinear characteristics of vastus medialis electromyography (EMG) signals during submaximal isometric knee extensor contractions. Our purpose was to examine if determinism and entropy in EMG signals reflected neuromuscular control deficits in individuals with ACL-deficient limbs.
Methods:
24 participants (12 male, 12 female, mean age = 18.8 ± 3.1 years) with unilaterally injured ACLs and 25 age-similar healthy controls (11 male, 14 female, mean age = 18.8 ± 3.1 years) volunteered. Isometric knee extensions were tested at 10%, 25%, 35%, and 50% maximum voluntary contractions. Surface electrodes adhered over the vastus medialis captured EMG signals. EMG data were processed with recurrence quantification analyses. Specifically, determinism (an index of system predictability) and entropy (an index of system disorder) were calculated from recurrence plots.
Findings:
Determinism and entropy in EMG signals were lower in the injured than uninjured limb, and lower than that from healthy controls (P < .05).
Interpretation:
Vastus medialis EMG signals from the injured limb were less predictable and less complex than those from healthy limbs. The findings reflect impaired neuromuscular control in the injured limb’s quadriceps and are consistent with a ‘loss of complexity’ hypothesis in physiologic signals emanating from pathologic states. Determinism and entropy in EMG signals may represent biomarkers of one’s neuromuscular control system.
Keywords: Electromyography, Anterior cruciate ligament, Quadriceps muscle, Isometric contraction, Entropy
1.0. INTRODUCTION
Individuals with anterior cruciate ligament (ACL) injuries exhibit neuromuscular control deficits. Commonly, those deficits occur as an arthrogenic muscle inhibition characterized by diminished quadriceps activation, defined as the proportion of volitional knee extension torque to that produced with superimposed electrical stimulation (Hart, Pietrosimone, Hertel, & Ingersoll, 2010). During initial post-injury stages, that inhibition may offer protection by minimizing anterior tibial translation produced by knee extensor forces. Longstanding inhibition, however, impairs voluntary knee extensor strength (Snyder-Mackler, De Luca, Williams, Eastlack, & Bartolozzi, 1994), alters gait biomechanics (Pietrosimone et al., 2014), impedes rehabilitation (Hart et al., 2010; Hopkins & Ingersoll, 2000), and may elevate risk of reinjury (Kyritsis, Bahr, Landreau, Miladi, & Witvrouw, 2016). Understanding mechanisms contributing to neuromuscular control deficits is therefore important.
Neuromuscular control refers to the process by which the brain communicates with muscles to produce desired outcomes or movements (van Leeuwen, 1999). Muscle strength and joint stability are dependent in part on neuromuscular control, as is the ability to elicit precise submaximal forces in goal-directed tasks. Whereas activation deficits summarized by Hart et al. (2010) represent neuromuscular control impairments during maximal contractions, examining targeted submaximal contractions provides information about one’s capacity to precisely gradate forces (Tracy, Maluf, Stephenson, Hunter, & Enoka, 2005), as is encountered in most functional activities. Investigators examining force steadiness of the first dorsal interosseus muscle, for example, have associated increased fluctuations at low to moderate forces with aging and poor manual dexterity (Kornatz, Christou, & Enoka, 2005; Tracy et al., 2005). Studies examining force steadiness during submaximal isometric contractions in ACL-deficient individuals have identified deficits in quadriceps motor unit rate coding (Schilaty, McPherson, Nagai, & Bates, 2022), as well as altered fractal scaling exponents from knee extensor force signals (Hollman, Nagai, Bates, McPherson, & Schilaty, 2021). Such findings illustrate impaired neuromuscular control associated with the characteristic arthrogenic muscle inhibition that occurs following ACL injury (Hopkins, Ingersoll, Edwards, & Klootwyk, 2002; Hopkins & Ingersoll, 2000; Norte, Rush, & Sherman, 2021).
Quantifying neuromuscular control is multifaceted and can be assessed at molecular (e.g., actin-myosin interactions), cellular (e.g., propagation of action potentials), organ (e.g., muscle force output), and organismic levels (e.g., whole body dynamics). Insight into neuromuscular control at the level of descending inputs into individual muscles is provided by assessing complexity in motor unit recruitment (Schuermans, Danneels, Van Tiggelen, Palmans, & Witvrouw, 2017; van Leeuwen, 1999), measured through electromyography (EMG). Complex signals from physiologic systems reflect chaotic, typically non-homeostatic, outputs that emerge from interactions among multiple structural units and feedback loops operating over a range of temporal and spatial scales (Goldberger, Peng, & Lipsitz, 2002). In the framework of our study, neuromuscular control deficits would potentially be evidenced by altered complexity in knee extensor EMG signals. Complexity in EMG signals reflects the neuromuscular system’s ability to adjust its output in response to variable demands (Vaillancourt & Newell, 2003), and is analyzed using nonlinear processing methods such as recurrence quantification analysis (RQA) (Filligoi & Felici, 1999). While several measures can be extracted from RQA, determinism and entropy in EMG signals provide context for assessing neuromuscular control. Determinism characterizes predictability of recurring patterns in EMG signals (Webber Jr, Schmidt, & Walsh, 1995; Webber Jr & Zbilut, 1994), and captures information about motor unit synchronization (Del Santo, Gelli, Mazzocchio, & Rossi, 2007; Del Santo et al., 2006; Farina, Fattorini, Felici, & Filligoi, 2002; Fattorini, Felici, Filligoi, Traballesi, & Farina, 2005). Entropy characterizes the magnitude of disorder—lack of structure—as a measure of complexity in EMG signals (Kaufman, Zurcher, & Sung, 2007). Davi and colleagues (Davi, Woxholdt, Rush, Lepley, & Lepley, 2021), for example, reported not only activation deficits but also reduced entropy in EMG signals from the vastus medialis—though not the vastus lateralis—during maximum isometric contractions in individuals following ACL reconstruction. Entropy in the vastus medialis EMG signal accounted for 44% of variance in quadriceps activation magnitudes. Whether a similar phenomenon occurs in ACL-deficient individuals prior to reconstructive surgery, or in submaximal isometric contractions more typically used to examine motor control (Enoka & Farina, 2021; Pethick, Taylor, & Harridge, 2022), is unknown.
The primary purpose of this study was to compare nonlinear characteristics of vastus medialis EMG signals during submaximal isometric contractions in injured and uninjured limbs from ACL-deficient individuals to those of healthy control subjects. Assuming similar findings to those of Davi, et al. (2021), we hypothesized EMG signals from the injured limb would demonstrate reduced predictability (determinism) and complexity (entropy) compared to the uninjured limb and to healthy controls. Secondarily, examining submaximal isometric contractions (10%, 25%, 35%, and 50% of max) provides capacity to examine if the magnitude of targeted forces differentially influences that relationship. Motor unit synchrony increases at higher isometric forces (Schmied & Descarreaux, 2010), which should be reflected by increased determinism in EMG signals. Our secondary purpose, therefore, was to compare complexity and entropy in EMG signals at varying levels of isometric force generation.
2.0. METHODS
2.1. Study design and participants
This was a cross-sectional study with two groups: ACL-deficient participants who had not yet undergone reconstructive surgery, and age-similar healthy controls without a history of lower extremity injury. Care plans for ACL-deficient participants were managed by an orthopaedic surgeon, and each had at least one pre-surgical visit with a physical therapist. Prehabilitation plans focused on reducing swelling and inflammation, increasing knee range of motion and quadriceps/hamstring strength prior to reconstructive surgery, and education on post-surgical rehabilitation expectations. Dynamic activities (e.g., running, jumping, cutting, pivoting) were not included in prehabilitation care plans.
The study was approved by the Mayo Clinic Institutional Review Board and utilized data collected from 24 ACL-deficient participants and 25 age-similar control participants, as described elsewhere (Hollman et al., 2021; Schilaty et al., 2022). Participants were healthy, active individuals between the ages of 14 and 25. Exclusion criteria included other lower extremity injuries (besides the ACL), surgery in 6 months prior to testing, neurological disorders, paralysis, cardiovascular disease, exercise-induced injury, asthma, and pregnancy. Written informed consent and parent assent, when appropriate (participants under age 18), were obtained prior to participation.
2.3. Instrumentation
EMG signals were recorded with 5-pin differential surface electrodes and a Delsys 16 amplifier (Delsys, Inc., Natick, MA, USA). Pins were 0.5 mm in diameter, placed in a 5 × 5-mm square configuration. Input impedance of the EMG system was 1 MOhm; each channel was 15 kOhm. The common mode rejection ratio was (6/10hz) −92 dB. Estimated noise in the EMG system was less than 1.2 μV RMS. The analog to digital converter was a 16-bit NI USB-6351 (National Instruments Corp, Austin, TX, USA).
Isometric knee extension contractions were performed from a seated position in a dynamometer (HumacNORM; CSMi, Stoughton, MA, USA), with a custom load cell apparatus (MLP-300; Transducer Techniques, Temecula, CA, USA) affixed to the dynamometer torque arm to measure force production.
2.4. Procedures
Prior to electrode application, skin was shaved to remove excess hair and cleansed with alcohol to minimize skin impedance and optimize electrode contact. Electrodes were placed over the vastus medialis, according to surface EMG for a non-invasive assessment of muscles (SENIAM) standards (Hermens, Freriks, Disselhorst-Klug, & Rau, 2000). The vastus medialis muscle was chosen specifically because it is more susceptible to inhibition than other quadriceps muscles (Spencer, Hayes, & Alexander, 1984).
Participants performed isometric knee extension tests from a seated position at 80° knee flexion in the dynamometer while secured with trunk stabilization straps. Three maximum voluntary isometric contractions (MVIC) were performed. Participants were verbally encouraged to contract as hard as possible for 3-s and permitted to view a monitor for visual feedback and motivation. The highest force produced from the trials was used to determine submaximal effort levels for subsequent tests. After the MVIC trials, participants performed sets of isometric knee extension contractions at 10%, 25%, 35%, and 50% MVIC, in random order. The reference force was visible to participants on a monitor, and they were asked to match the target force as closely as possible. For each test, participants followed a trapezoidal contraction pattern including a 3-s ramp up, 10-s sustained contraction at the designated effort level, and 3-s ramp down (Fig. 1A). This procedure was completed for both limbs in a randomized order.
Fig. 1.
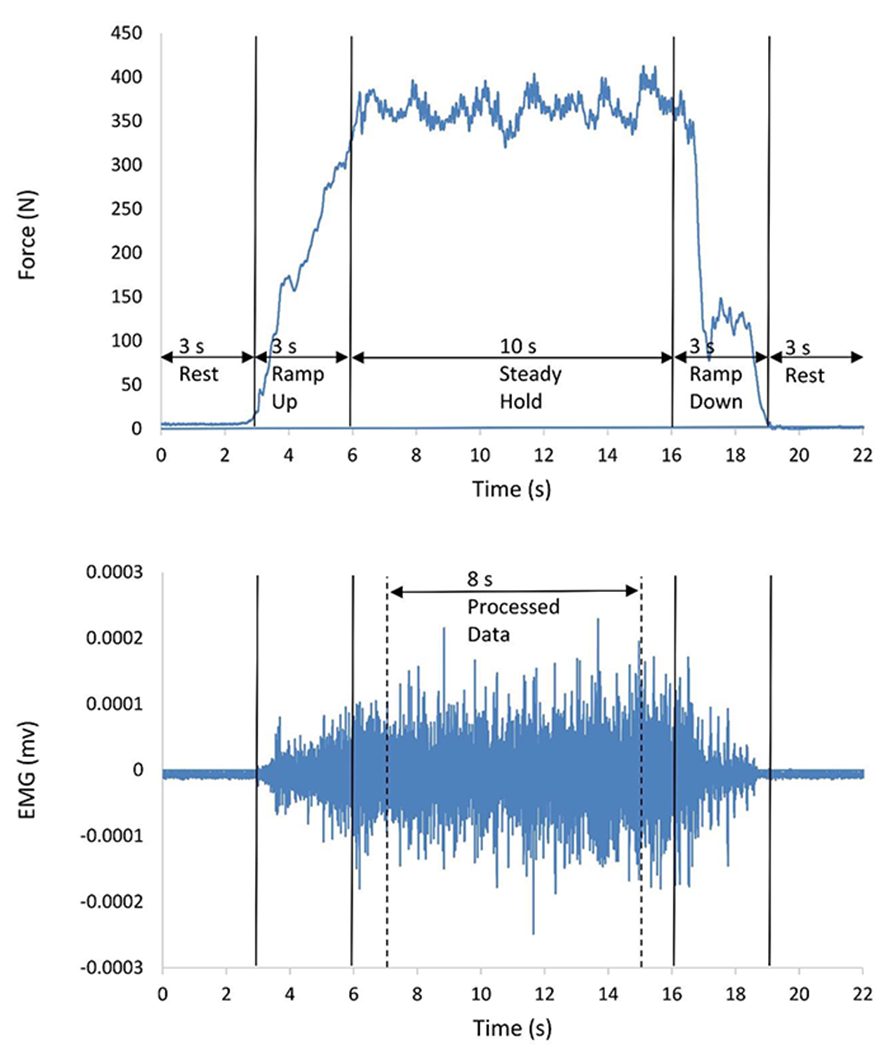
Representative plots of (A) knee extensor force output during a participant’s submaximal contraction at 50% effort, and (B) the same participant’s corresponding EMG signal from the vastus medialis muscle during the trial. EMG data from the middle 8 seconds of the 10 second isometric contraction were extracted for processing and analysis.
Analog EMG signals were band-pass filtered with 20-1750 Hz cut-off frequencies (De Luca, Donald Gilmore, Kuznetsov, & Roy, 2010; De Luca, Kline, & Contessa, 2014). Signals were sampled at 20,000 Hz, then down sampled to 2,000 Hz for subsequent processing.
2.5. Data Processing
Pairwise subtractions of voltages from the 5-pin electrodes were used to derive multi-channel signals (Nawab, Chang, & De Luca, 2010). A single channel’s raw EMG signal from the middle 8-s of the 10-s sustained isometric contraction was extracted (Fig. 1B). EMG signals from the injured and uninjured limbs of ACL-deficient participants, and from randomly selected dominant (56%) and non-dominant (44%) limbs among control participants, were processed with RQA. Detailed RQA methods are described elsewhere (Webber Jr & Zbilut, 1994), and described briefly here.
First, based on Takens’ theorem (Takens, 1981), a raw signal (Fig. 2A) is reconstructed in a phase space (Fig. 2B) with time-delayed copies, via
where X(t) is the dE-dimensional state vector, x(t) represents raw data at time t, T represents a time delay between reconstructed data points, and dE represents an embedding dimension. The time delay T = 10 samples (5 ms) was determined with average mutual information functions (Fraser & Swinney, 1986). The embedding dimension dE = 4 was determined with global false nearest neighbors analyses (Kennel, Brown, & Abarbanel, 1992). These parameters were maintained consistently across participants to ensure any differences with inferential statistical testing were not confounded by parameter selection.
Fig. 2.
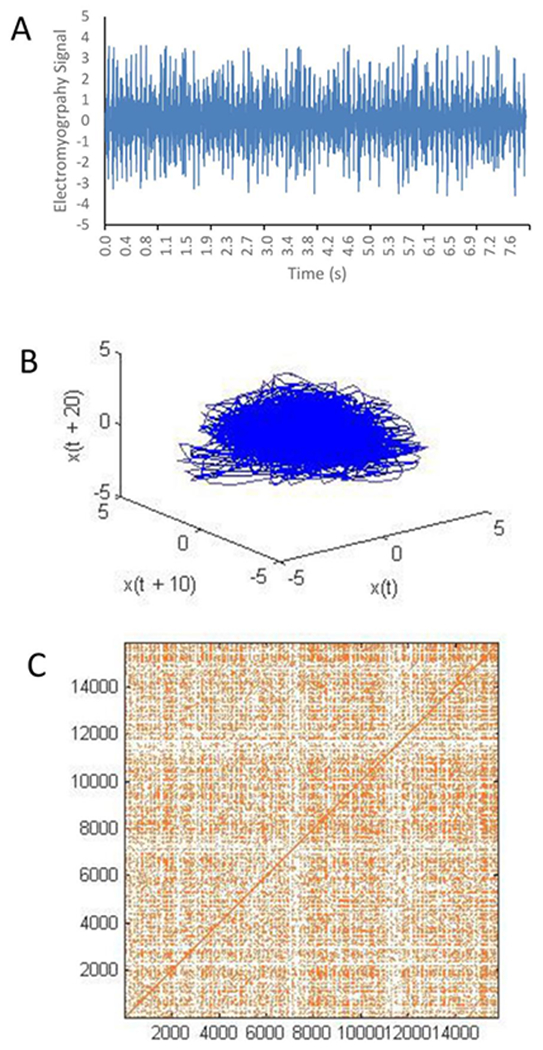
A raw EMG signal (A) is plotted in a phase space reconstruction with a time delay of 10 data points (B), from which a recurrence plot is composed (C), defined with a radius of 10% of a standard deviation.
After phase space reconstruction, a 2-dimensional recurrence plot was formed (Fig. 2C). Marks on the recurrence plot at position (i,j) represent a phase space vector j which falls closely enough to a given phase space vector i to be considered a recurrence. Any reconstructed phase space vector occurring beyond a defined r = 10% SD threshold from the original vector created empty (white) space in the recurrence plot. The 10% radius yielded recurrence rates of approximately 1% without oversaturating individual recurrence plots (Riley, Balasubramaniam, & Turvey, 1999).
While several measures can be extracted from recurrence plots with RQA, we focused specifically on determinism and entropy as described by Marwan, et al. (Marwan, Romano, Thiel, & Kurths, 2007). Determinism has been characterized as the most important RQA parameter in EMG analyses (Filligoi & Felici, 1999). Determinism quantifies the magnitude of signal correlations in higher dimensional space; hence it characterizes rule-obeying predictability of dynamical systems. Whereas random, uncorrelated white noise yields recurrence plots with many single dots and few diagonal lines, deterministic systems yield recurrence plots with few single dots but many diagonal lines. Specifically, determinism (%DET) quantifies the proportion of recurrence points falling on diagonal lines, with a minimum length lmin = 2. %DET is calculated as
where P(l) is the frequency distribution of diagonal lines of length l.
The second measure extracted with RQA, entropy (ENT) (Shannon, 1948), reflects complexity—or disorder—of a dynamical system’s deterministic structure. Quantifying the negative natural logarithm of the conditional probability p(l) that a diagonal line in the recurrence plot has exactly length l, ENT is calculated as
Higher entropy in a dynamical system signifies higher complexity, whereas lower values from systems with uncorrelated noise denote less complexity.
2.6. Statistical Analysis
Descriptive statistics, including means (SDs) for continuous variables and frequencies (%) for categorical variables, were calculated. Differences in determinism (%DET) and Shannon entropy (ENT) from the EMG signals were compared between limbs (injured and uninjured from ACL-deficient participants, and randomly selected from controls) and across effort levels (10%, 25%, 35%, and 50% MVIC) with generalized linear model (GLM) repeated measures procedures. Post hoc comparisons with Bonferroni corrections were used to examine statistically significant main effects or limb × effort level interaction effects. All analyses were conducted with IBM SPSS Statistics 28 software (IBM Corp, Armonk, NY, USA) at α = .05.
3.0. RESULTS
3.1. Participant demographics
Participants included 12 males and 12 females with ACL injuries, and 11 male and 14 female controls. Of ACL-deficient participants, 79.2% sustained non-contact and 20.8% contact injuries. Activity levels varied from sedentary (4.2%) to recreational (20.8%) to competitive (75%), while those in the control group were recreational (44%) to competitive (56%) athletes. ACL-deficient participants were 18.8 ± 3.1 years old with body mass indices (BMI) of 25.0 ± 2.7 kg/m2. The mean duration from injury was 54.9 ± 66.1 days (range 10-320 days). Participants in the control group were 18.8 ± 3.1 years old with BMIs of 22.2 ± 3.3 kg/m2.
3.2. Determinism
When comparing %DET in EMG signals (Fig. 3), the limb (Wald χ2 = 14.603, P < .001) and effort level (Wald χ2 = 38.370, P < .001) main effects were statistically significant, though the limb × effort level interaction was not (Wald χ2 = 1.089, P = .982). Collapsed across effort levels, mean magnitudes of determinism in EMG signals were lower from the injured (mean = 7.342% DET, 95% CI = 5.117 to 9.568% DET) than uninjured limb (mean = 11.576% DET, 95% CI = 9.415 to 13.737% DET; mean difference = 4.234% DET, 95% CI = 0.444 to 8.023% DET, P = .022), and lower than in controls (mean = 13.094% DET, 95% CI = 11.050 to 15.140% DET; mean difference = 5.752% DET, 95% CI = 2.061 to 9.443% DET, P = .001). The difference between ACL-deficient participants’ uninjured limbs and controls, however, was not statistically significant (mean difference = 1.518% DET, 95% CI = −2.116 to 5.151% DET, P = .952).
Fig. 3.
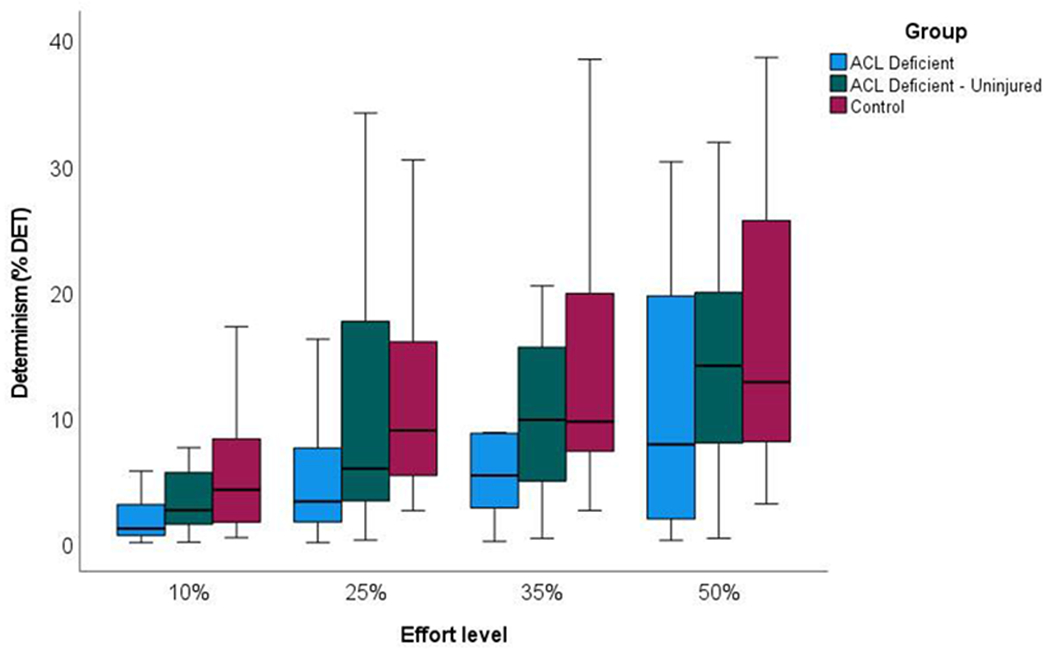
Box plots of determinism in the EMG signal from the vastus medialis at four volitional force levels during submaximal, isometric knee extensor contractions.
Across limbs, %DET in the EMG signal increased as effort levels increased from 10% to 50% MVIC (Fig. 3; mean determinism = 5.227, 9.525, 11.869, and 16.062% DET, respectively). Based on post hoc pairwise comparisons, determinism in EMG signals was lower at 10% than at 35% and 50% MVIC (P = .001 and P < .001, respectively), and lower at 25% than at 50% MVIC (P = .001). Since the limb × effort level interaction was not statistically significant, the stepwise increase in EMG signal determinism from 10% to 50% MVIC (Fig. 3) was similar across limbs.
3.3. Shannon entropy
Shannon entropy data from EMG signals (Fig. 4) carried similar differences to that of the determinism data. The limb (Wald χ2 = 29.956, P < .001) and effort level (Wald χ2 = 80.735, P < .001) main effects were statistically significant, whereas the limb × effort level interaction was not (Wald χ2 = 1.642, P = .950). Mean entropy in EMG signals, collapsed across effort levels, were lower in the injured (mean = 0.436 bits/bin, 95% CI = 0.353 to 0.520 bits/bin) than uninjured limb (mean = 0.632 bits/bin, 95% CI = 0.551 to 0.713 bits/bin; mean difference = 0.196 bits/bin, 95% CI = 0.054 to 0.338 bits/bin, P = .033), and lower than in controls (mean = 0.751 bits/bin, 95% CI = 0.675 to 0.828; mean difference = 0.315 bits/bin, 95% CI = 0.177 to 0.453 bits/bin, P < .001). Like comparisons from the determinism data, mean EMG signal entropy values did not differ between uninjured limbs from ACL-deficient and control participants (mean difference = 0.119 bits/bin, 95% CI = −0.017 to 0.255 bits/bin, P = .109).
Fig. 4.
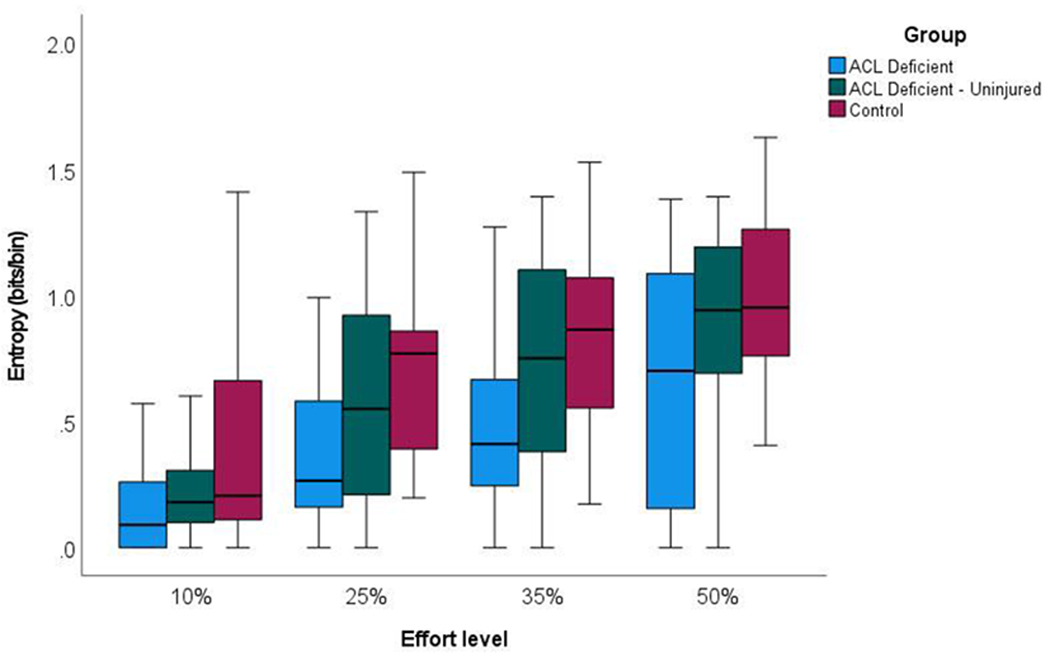
Box plots of entropy in the EMG signal from the vastus medialis at four volitional force levels during submaximal, isometric knee extensor contractions.
Moreover, when comparing entropy across effort levels, EMG signal entropy from both limbs in ACL-deficient participants as well as limbs in control participants increased as effort increased from 10% to 50% MVIC (mean = 0.279, 0.588, 0.698, and 0.861 bits/bin, respectively). Based on post hoc comparisons, entropy in the EMG signal was lower at 10% than at 25%, 35%, and 50% MVIC (P < .001, P = .002, and P < .001, respectively); and was lower at 25% than 50% MVIC (P < .001).
4.0. DISCUSSION
We examined nonlinear characteristics of vastus medialis EMG signals during submaximal, isometric knee extension contractions from individuals with ACL injuries. Specifically, determinism in EMG signals as a measure of neuromuscular system predictability, and entropy as a measure of complexity, were examined. Key findings indicated determinism and entropy in vastus medialis EMG signals were lower in the injured limb in ACL-deficient participants. Secondarily, determinism and entropy increased among all participants—healthy controls as well as from injured and uninjured limbs in ACL-deficient participants—as contraction forces increased from 10% to 50% MVIC.
EMG signals from the injured limb’s vastus medialis were less deterministic than from the uninjured limb and from healthy controls. Previous studies have examined EMG amplitudes, EMG on/offset, latencies, co-contraction ratios, and asymmetry indices as neuromuscular control measures in ACL-deficient knees (Blasimann, Koenig, Baert, Baur, & Vissers, 2021). While related literature suggests reduced determinism in EMG signals is associated with fatigue resistance (Liu, Kankaanpää, Zbilut, & Webber, 2004), to the best of our knowledge, this is the first study to examine determinism as a measure for neuromuscular control in the context of ACL injury. Determinism in EMG signals provides information about motor unit synchronization (Del Santo et al., 2007; Del Santo et al., 2006; Farina et al., 2002; Fattorini et al., 2005). Increased determinism in EMG signals occurs as motor units recruited for a given task become more synchronized. Increased motor unit synchronization represents a neuromuscular control strategy that either enhances rapid force development or enhances muscle synergies (Semmler, 2002), both of which are observed in strength-trained individuals during submaximal isometric contractions (Milner-Brown & Lee, 1975; Semmler & Nordstrom, 1998). In contrast, almost no motor unit synchronization is observed from muscles during submaximal isometric contractions in individuals with corticospinal pathway pathology (Schmied, Pouget, & Vedel, 1999), whereas low but detectable motor unit synchronization is observed in the first dorsal interosseus muscle during submaximal isometric contractions in musicians (Semmler & Nordstrom, 1998). We postulate that certain magnitudes of motor unit synchronization—not too much synchronization and not too little—may reflect optimal neuromuscular control in a muscle. While those optimal levels are not yet determined, the arthrogenic muscle inhibition associated with ACL injuries (Schilaty et al., 2022), according to our findings, is characterized in part by a loss of deterministic structure in the EMG signal and reduced motor unit synchronization.
Apart from the differences in participants with ACL-deficient limbs, determinism in the vastus medialis EMG signal increased as participants volitionally increased force output from 10% to 50% MVIC (Fig. 3), both in ACL-deficient participants and in healthy control subjects. This finding was expected. Schmied & Descarreaux (2010), among others (Datta & Stephens, 1990; Fling, Christie, & Kamen, 2009), have shown that motor unit synchrony is enhanced as isometric forces increase. As more motor units are recruited and as motor unit rate coding accelerates at greater force levels, pre-synaptic inputs generate greater synchronization (Schmied & Descarreaux, 2010). That synchronization is interpreted as a more efficient way for the central nervous system to coordinate motor unit activity and optimize motor performance (Santello & Fuglevand, 2004). Since elevated levels of determinism in the EMG signal reflect greater motor unit synchronization (Del Santo et al., 2007; Del Santo et al., 2006; Farina et al., 2002; Fattorini et al., 2005), we expected the deterministic structure of variability in EMG signals to increase at higher levels of volitional force output.
Further supporting our hypotheses, EMG signals from the injured limb of ACL-deficient participants had lower entropy than the uninjured limb and healthy controls (Fig. 4). This finding is consistent with the “loss of complexity” hypothesis posed by Lipsitz & Goldberger (1992) suggesting that, when compared with pathologic systems, physiologic signals from healthier biological systems exhibit greater entropy. Our findings share similarities with those of Davi, et al. (2021), who reported lower entropy levels in the vastus medialis from participants with ACL-reconstructed knees during maximal isometric contractions. They concluded loss of complexity in the EMG signal correlates with diminished muscle activation following reconstructive surgery, suggesting a reduction in neuromuscular system adaptability that may hinder quadriceps firing. Other studies, with different patient populations, consistently have demonstrated decreased entropy in physiologic signals is associated with pathologic states (Afsar, Tirnakli, & Marwan, 2018; Lipsitz & Goldberger, 1992). Our findings are consistent with this literature. Participants with an ACL deficiency present with decreased entropy in vastus medialis EMG signals, suggesting less complexity and reduced neuromuscular system volitional control, indicating its association to a pathologic state.
Additionally, as volitional force increased, entropy in the EMG signal increased concomitantly (Fig. 4). As with determinism data, this finding was expected. Similar relationships between volitional force and entropy have been reported elsewhere, albeit with other muscles than the vastus medialis (Cashaback, Cluff, & Potvin, 2013; Troiano et al., 2008; Zambalde, Germer, & Elias, 2021). As volitional force increases, more motor units are recruited and firing rates of individual motor units accelerate, thereby producing a more stochastic EMG signal (Troiano et al., 2008). The stochastic signal is characterized by higher levels of entropy as a measure of neuromuscular system complexity.
Limitations in the study are acknowledged. This study was cross-sectional rather than longitudinal in design. Whether entropy and determinism in EMG signals change as individuals recover over time, with rehabilitation, or following reconstructive surgery, is unknown. Longitudinal analyses are required to answer those questions. Second, we did not have a sufficient sample to rigorously examine data in EMG signals as a function of chronicity (e.g., participants with acute vs. chronic ACL injuries); therefore, we do not know if injury chronicity influences findings. However, a cursory analysis of our data suggests time from injury is positively correlated with both %DET (partial r = .419, p < .001) and ENT (partial r = .227, p = .043), when controlling for contraction effort levels. These correlations imply that, in individuals with ACL-deficient limbs, determinism and entropy in the EMG signal from the vastus medialis during submaximal isometric contractions may return toward normal magnitudes as time-from-injury advances, reflecting compensatory improvements in neuromuscular control following an ACL injury. Third, we do not know whether leg dominance may influence findings, as we had 15 participants with right-sided and 9 with left-sided ACL deficiency, and 22 of the 24 participants were right leg dominant. This limitation can be overcome by comparing data of individuals with dominant versus nondominant limb injuries in future studies. Despite these limitations, we still can establish that determinism and entropy in the vastus medialis EMG signal during submaximal isometric knee extension contractions are lower in individuals with ACL-deficient limbs, indicating diminished neuromuscular control.
5.0. CONCLUSION
Neuromuscular control of the vastus medialis was examined through nonlinear analyses of submaximal isometric EMG signals from individuals with an ACL-deficient knee. Determinism and entropy in EMG signals were lower in the injured than uninjured limb, and lower than that from healthy controls, suggesting diminished neuromuscular control in the injured limb’s vastus medialis muscle. The findings are consistent with a “loss of complexity” hypothesis in pathologic states (Afsar et al., 2018; Davi et al., 2021; Lipsitz & Goldberger, 1992). Determinism and entropy in EMG signals may represent biomarkers of the state of one’s neuromuscular control system at the muscle level.
ACKNOWLEDGEMENTS:
This work was supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development (Grant K12HD065987), National Center for Advancing Translational Sciences (Grant UL1TR002377), National Institute of Arthritis and Musculoskeletal and Skin Diseases (Grant L30AR070273, R01AR056259), the Mayo Clinic Department of Physical Medicine & Rehabilitation, and the University of South Florida Center for Neuromusculoskeletal Research. The study sponsors were not involved in the study design; in the collection, analysis or interpretation of data; in the writing of the manuscript; nor in the decision to submit the manuscript for publication.
Footnotes
CONFLICT OF INTEREST: None Declared.
REFERENCES
- Afsar O, Tirnakli U, & Marwan N (2018). Recurrence quantification analysis at work: Quasi-periodicity based interpretation of gait force profiles for patients with Parkinson disease. Sci Rep, 8(1), 1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blasimann A, Koenig I, Baert I, Baur H, & Vissers D (2021). Which assessments are used to analyze neuromuscular control by electromyography after an anterior cruciate ligament injury to determine readiness to return to sports? A systematic review. BMC Sports Sci Med Rehabil, 13(1), 1–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cashaback JGA, Cluff T, & Potvin JR (2013). Muscle fatigue and contraction intensity modulates the complexity of surface electromyography. J Electromyogr Kinesiol, 23(1), 78–83. doi: 10.1016/j.jelekin.2012.08.004 [DOI] [PubMed] [Google Scholar]
- Datta A, & Stephens J (1990). Synchronization of motor unit activity during voluntary contraction in man. J Physiol, 422(1), 397–419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davi SM, Woxholdt CK, Rush JL, Lepley AS, & Lepley LK (2021). Alterations in quadriceps neurologic complexity after anterior cruciate ligament reconstruction. J Sport Rehabil, 30(5), 731–736. doi: 10.1123/jsr.2020-0307 [DOI] [PubMed] [Google Scholar]
- De Luca CJ, Donald Gilmore L, Kuznetsov M, & Roy SH (2010). Filtering the surface EMG signal: Movement artifact and baseline noise contamination. J Biomech, 43(8), 1573–1579. doi: 10.1016/j.jbiomech.2010.01.027 [DOI] [PubMed] [Google Scholar]
- De Luca CJ, Kline JC, & Contessa P (2014). Transposed firing activation of motor units. J Neurophysiol, 112(4), 962–970. doi: 10.1152/jn.00619.2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Del Santo F, Gelli F, Mazzocchio R, & Rossi A (2007). Recurrence quantification analysis of surface EMG detects changes in motor unit synchronization induced by recurrent inhibition. Exp Brain Res, 178(3), 308–315. [DOI] [PubMed] [Google Scholar]
- Del Santo F, Gelli F, Schmied A, Vedel J-P, Rossi A, & Mazzocchio R (2006). Motor unit synchronous firing as revealed by determinism of surface myoelectric signal. J Neurosci Methods, 155(1), 116–121. [DOI] [PubMed] [Google Scholar]
- Enoka RM, & Farina D (2021). Force steadiness: from motor units to voluntary actions. Physiol, 36(2), 114–130. [DOI] [PubMed] [Google Scholar]
- Farina D, Fattorini L, Felici F, & Filligoi G (2002). Nonlinear surface EMG analysis to detect changes of motor unit conduction velocity and synchronization. J Appl Physiol, 93(5), 1753–1763. [DOI] [PubMed] [Google Scholar]
- Fattorini L, Felici F, Filligoi GC, Traballesi M, & Farina D (2005). Influence of high motor unit synchronization levels on non-linear and spectral variables of the surface EMG. J Neurosci Methods, 143(2), 133–139. doi: 10.1016/j.jneumeth.2004.09.018 [DOI] [PubMed] [Google Scholar]
- Filligoi G, & Felici F (1999). Detection of hidden rhythms in surface EMG signals with a non-linear time-series tool. Med Eng Phys, 21(6–7), 439–448. [DOI] [PubMed] [Google Scholar]
- Fling BW, Christie A, & Kamen G (2009). Motor unit synchronization in FDI and biceps brachii muscles of strength-trained males. J Electromyogr Kinesiol, 19(5), 800–809. [DOI] [PubMed] [Google Scholar]
- Fraser AM, & Swinney HL (1986). Independent coordinates for strange attractors from mutual information. Phys Rev A, 33(2), 1134. [DOI] [PubMed] [Google Scholar]
- Goldberger AL, Peng C-K, & Lipsitz LA (2002). What is physiologic complexity and how does it change with aging and disease? Neurobiol Aging, 23(1), 23–26. [DOI] [PubMed] [Google Scholar]
- Hart JM, Pietrosimone B, Hertel J, & Ingersoll CD (2010). Quadriceps activation following knee injuries: a systematic review. J Athl Train, 45(1), 87–97. doi: 10.4085/1062-6050-45.1.87 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hermens HJ, Freriks B, Disselhorst-Klug C, & Rau G (2000). Development of recommendations for SEMG sensors and sensor placement procedures. J Electromyogr Kinesiol, 10(5), 361–374. [DOI] [PubMed] [Google Scholar]
- Hollman JH, Nagai T, Bates NA, McPherson AL, & Schilaty ND (2021). Diminished neuromuscular system adaptability following anterior cruciate ligament injury: Examination of knee muscle force variability and complexity. Clin Biomech, 90, 105513. doi: 10.1016/j.clinbiomech.2021.105513 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hopkins J, Ingersoll CD, Edwards J, & Klootwyk TE (2002). Cryotherapy and transcutaneous electric neuromuscular stimulation decrease arthrogenic muscle inhibition of the vastus medialis after knee joint effusion. J Athl Train, 37(1), 25–31. [PMC free article] [PubMed] [Google Scholar]
- Hopkins JT, & Ingersoll CD (2000). Arthrogenic muscle inhibition: A limiting factor in joint rehabilitation. J Sport Rehabil, 9(2), 135–159. doi: 10.1123/jsr.9.2.135 [DOI] [Google Scholar]
- Kaufman M, Zurcher U, & Sung PS (2007). Entropy of electromyography time series. Physica A: Statistical Mechanics and its Applications, 386(2), 698–707. doi: 10.1016/j.physa.2007.07.045 [DOI] [Google Scholar]
- Kennel MB, Brown R, & Abarbanel HD (1992). Determining embedding dimension for phase-space reconstruction using a geometrical construction. Phys Rev A, 45(6), 3403. [DOI] [PubMed] [Google Scholar]
- Kornatz KW, Christou EA, & Enoka RM (2005). Practice reduces motor unit discharge variability in a hand muscle and improves manual dexterity in old adults. J Appl Physiol, 98(6), 2072–2080. doi: 10.1152/japplphysiol.01149.2004 [DOI] [PubMed] [Google Scholar]
- Kyritsis P, Bahr R, Landreau P, Miladi R, & Witvrouw E (2016). Likelihood of ACL graft rupture: Not meeting six clinical discharge criteria before return to sport is associated with a four times greater risk of rupture. Br J Sports Med, 50(15), 946–951. [DOI] [PubMed] [Google Scholar]
- Lipsitz LA, & Goldberger AL (1992). Loss of ‘complexity’ and aging: Potential applications of fractals and chaos theory to senescence. JAMA, 267(13), 1806–1809. [PubMed] [Google Scholar]
- Liu Y, Kankaanpää M, Zbilut JP, & Webber CL (2004). EMG recurrence quantifications in dynamic exercise. Biol Cybern, 90(5), 337–348. [DOI] [PubMed] [Google Scholar]
- Mantashloo Z, Letafatkar A, & Moradi M (2020). Vertical ground reaction force and knee muscle activation asymmetries in patients with ACL reconstruction compared to healthy individuals. Knee Surg Sports Traumatol Arthrosc, 28(6), 2009–2014. [DOI] [PubMed] [Google Scholar]
- Marwan N, Romano MC, Thiel M, & Kurths J (2007). Recurrence plots for the analysis of complex systems. Phys Rep, 438(5-6), 237–329. [Google Scholar]
- Milner-Brown HS, & Lee RG (1975). Synchronization of human motor units: Possible roles of exercise and supraspinal reflexes. Electroencephalogr Clin Neurophysiol, 38(3), 245–254. doi: 10.1016/0013-4694(75)90245-X [DOI] [PubMed] [Google Scholar]
- Nawab SH, Chang S-S, & De Luca CJ (2010). High-yield decomposition of surface EMG signals. Clin Neurophysiol, 121(10), 1602–1615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Norte G, Rush J, & Sherman D (2021). Arthrogenic muscle inhibition: Best evidence, mechanisms, and theory for treating the unseen in clinical rehabilitation. J Sport Rehabil, 1–19. doi: 10.1123/jsr.2021-0139 [DOI] [PubMed] [Google Scholar]
- Pethick J, Taylor MJD, & Harridge SDR (2022). Aging and skeletal muscle force control: Current perspectives and future directions. Scand J Med Sci Sports, 32(10), 1430–1443. doi: 10.1111/sms.14207 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pietrosimone B, Lepley AS, Murray AM, Thomas AC, Bahhur NO, & Schwartz TA (2014). Changes in voluntary quadriceps activation predict changes in muscle strength and gait biomechanics following knee joint effusion. Clin Biomech, 29(8), 923–929. doi: 10.1016/j.clinbiomech.2014.06.014 [DOI] [PubMed] [Google Scholar]
- Riley M, Balasubramaniam R, & Turvey M (1999). Recurrence quantification analysis of postural fluctuations. Gait Posture, 9(1), 65–78. [DOI] [PubMed] [Google Scholar]
- Santello M, & Fuglevand AJ (2004). Role of across-muscle motor unit synchrony for the coordination of forces. Exp Brain Res, 159(4), 501–508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schilaty ND, McPherson AL, Nagai T, & Bates NA (2022). Arthrogenic muscle inhibition manifests in thigh musculature motor unit characteristics after anterior cruciate ligament injury. Eur J Sport Sci, 1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmied A, & Descarreaux M (2010). Influence of contraction strength on single motor unit synchronous activity. Clin Neurophysiol, 121(10), 1624–1632. [DOI] [PubMed] [Google Scholar]
- Schmied A, Pouget J, & Vedel J-P (1999). Electromechanical coupling and synchronous firing of single wrist extensor motor units in sporadic amyotrophic lateral sclerosis. Clin Neurophysiol, 110(5), 960–974. [DOI] [PubMed] [Google Scholar]
- Schuermans J, Danneels L, Van Tiggelen D, Palmans T, & Witvrouw E (2017). Proximal neuromuscular control protects against hamstring injuries in male soccer players: A prospective study with electromyography time-series analysis during maximal sprinting. Am J Sports Med, 45(6), 1315–1325. doi: 10.1177/0363546516687750 [DOI] [PubMed] [Google Scholar]
- Semmler J, & Nordstrom M (1998). Motor unit discharge and force tremor in skill-and strength-trained individuals. Exp Brain Res, 119(1), 27–38. [DOI] [PubMed] [Google Scholar]
- Semmler JG (2002). Motor unit synchronization and neuromuscular performance. Exerc Sport Sci Rev, 30(1), 8–14. [DOI] [PubMed] [Google Scholar]
- Shanbehzadeh S, Mohseni Bandpei MA, & Ehsani F (2017). Knee muscle activity during gait in patients with anterior cruciate ligament injury: a systematic review of electromyographic studies. Knee Surg Sports Traumatol Arthrosc, 25(5), 1432–1442. [DOI] [PubMed] [Google Scholar]
- Shannon CE (1948). A mathematical theory of communication. The Bell System Technical Journal, 27(3), 379–423. [Google Scholar]
- Snyder-Mackler L, De Luca PF, Williams PR, Eastlack ME, & Bartolozzi AR 3rd. (1994). Reflex inhibition of the quadriceps femoris muscle after injury or reconstruction of the anterior cruciate ligament. J Bone Joint Surg, 76(4), 555–560. [DOI] [PubMed] [Google Scholar]
- Spencer JD, Hayes K, & Alexander IJ (1984). Knee joint effusion and quadriceps reflex inhibition in man. Arch Phys Med Rehabil, 65(4), 171–177. [PubMed] [Google Scholar]
- Takens F (1981). Detecting strange attractors in turbulence. In Rand DA & Young LS (Eds.), Dynamical Systems and Turbulence, Lecture Notes in Mathematics (Vol. 898, pp. 366–381). Berlin/Heidelberg: Springer-Verlag. [Google Scholar]
- Tracy BL, Maluf KS, Stephenson JL, Hunter SK, & Enoka RM (2005). Variability of motor unit discharge and force fluctuations across a range of muscle forces in older adults. Muscle Nerve, 32(4), 533–540. doi: 10.1002/mus.20392 [DOI] [PubMed] [Google Scholar]
- Troiano A, Naddeo F, Sosso E, Camarota G, Merletti R, & Mesin L (2008). Assessment of force and fatigue in isometric contractions of the upper trapezius muscle by surface EMG signal and perceived exertion scale. Gait Posture, 28(2), 179–186. [DOI] [PubMed] [Google Scholar]
- Vaillancourt DE, & Newell KM (2003). Aging and the time and frequency structure of force output variability. J Appl Physiol, 94(3), 903–912. [DOI] [PubMed] [Google Scholar]
- van Leeuwen JL (1999). Neuromuscular control: introduction and overview. Philos Trans R Soc Lond B Biol Sci, 354(1385), 841–847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Webber CL Jr, Schmidt M, & Walsh JM (1995). Influence of isometric loading on biceps EMG dynamics as assessed by linear and nonlinear tools. J Appl Physiol, 78(3), 814–822. [DOI] [PubMed] [Google Scholar]
- Webber CL Jr, & Zbilut JP (1994). Dynamical assessment of physiological systems and states using recurrence plot strategies. J Appl Physiol, 76(2), 965–973. [DOI] [PubMed] [Google Scholar]
- Zambalde E, Germer C, & Elias L (2021). Entropy of motor unit activity and surface electromyogram in an isometric force task. Proceedings SPIE 12088, 17th International Symposium on Medical Information Processing and Analysis, 1208802. doi: 10.1117/12.2606261 [DOI] [Google Scholar]


