Abstract
The existence of both neuroendocrine and non-neuroendocrine histology in variable proportion in a lesion has been described by the World Health Organisation (WHO) as mixed neuroendocrine and non-neuroendocrine neoplasm (MiNEN). The pathogenesis of this tumour remains controversial but molecular studies point towards a common monoclonal origin. Tumours are classified as functioning and nonfunctioning based on substances secreted. The nonfunctioning tumours may be discovered due to its local effect. Presented is a 66-year-old male with an intra-abdominal mass, underwent laparotomy and excision biopsy with transient right lower limb lymphoedema. Histology confirmed retroperitoneal MiNEN with no evidence of tumour recurrence 12 months following surgery. MiNENs should be considered as a differential diagnosis in patients with intra-abdominal mass. Surgical resection is recommended as this may offer the best treatment option.
Keywords: mixed neuroendocrine non-neuroendocrine neoplasm, mixed adenoneuroendocrine carcinoma, WHO classifications, retroperitoneum
Introduction
The coexistence of both neuroendocrine and non-neuroendocrine histology of epithelial neoplasms in a variable proportion have been recently described as mixed neuroendocrine non-neuroendocrine neoplasms (MiNENs). 1 The World Health Organisation (WHO) in 2010 classified Neuroendocrine Tumours (NETs) based on Ki67 into (i). NET G1, G2, (ii). NEC G3, (iii). Mixed adenoneuroendocrine carcinoma (MANEC), (iv). Hyperplastic and paraneoplastic lesions. 2 The first case of MANEC was described in 1924 followed by a few reported cases in Medical literature. 3 Furthermore, in 2010 the WHO Classified MANEC as a separate entity containing a neuroendocrine and an exocrine component with each as separate entities present in at least 30% of the tumour mass and being malignant.1,3 In 2017, WHO renamed MANEC as MiNENs. 4
It is found in almost all organs and a rare disease. 5 It was earlier reported as 0.227 cases per 1,000,000, but by 2019, it had increased by approximately 4.4 times to 1.018 cases per 1,000,000. The prevalence of MiNENs was slightly higher in males and elderly patients, consistent with previous studies. 6 The pathogenesis of this tumour remains controversial but molecular studies point towards a common monoclonal origin. 1 Tumours are classified as functioning (based on substances secreted) and nonfunctioning, the later tumours may be discovered due to its local effect. 7 Surgery is the mainstay of treatment. 1 Tumour grade, tumour size, distant metastasis, lymph node metastasis and surgery are independent risk factors for the prognosis. Treatment should be based on the most aggressive component of neoplasia, determined reliably by analysis of a resected specimen, even if it is less than 30% of the neoplasm. 8 We present a 66 year old patient with an intra-abdominal mass, underwent laparotomy with excision biopsy, histology revealed retroperitoneal MiNEN with no evidence of tumour recurrence 12 months following surgery to highlight the favorable outcome for a tumour located in the retroperitoneum.
Case report
A 66-year-old male retired civil servant presented with recurrent abdominal pain and abdominal mass of a year duration. The pain was located at the right iliac fossa characterized by gradual onset, dull but occasionally burning in character, intermittent, non-radiating without known aggravating factors but relieved by over-the-counter analgesics. Pain was moderate in intensity. The associated abdominal mass was also noticed at the right lower abdomen, with an initial size less than the patient’s thumb but gradually increased to about the size of a tennis ball. There was no anorexia, nausea, vomiting, change in bowel habit, bloody stool, fever or history of trauma. However, he had weight loss and easy fatigability.
Because of the above, he presented to a Secondary Health Facility where abdominal ultrasound and CT scans were done with findings that suggested an intra-abdominal mass and was referred to our facility for expert care. He is a known hypertensive patient on control medications: he was also diagnosed of benign prostatic hyperplasia (BPH), not on prescribed medications. He has no previous hospital admission and no previous surgeries.
Examination findings revealed an elderly man in no painful or respiratory distress, afebrile, not pale, not dehydrated and with no pedal oedema. The abdomen was full and moved with respiration, with mild right lower quadrant tenderness. There was an irregular right iliac fossa mass that measured about 8 cm by 6 cm, mildly tender, firm, nil differential warmth, slightly mobile. The liver, spleen and kidneys were not enlarged. There was no demonstrable ascites and bowel sounds were normoactive. Rectal examination findings were in keeping with BPH.
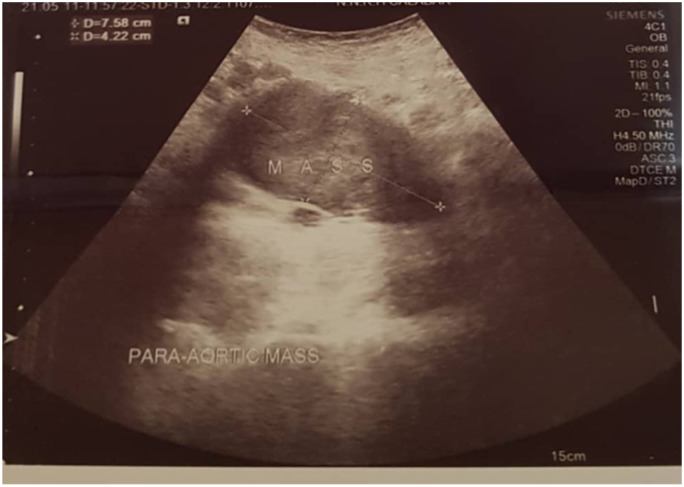
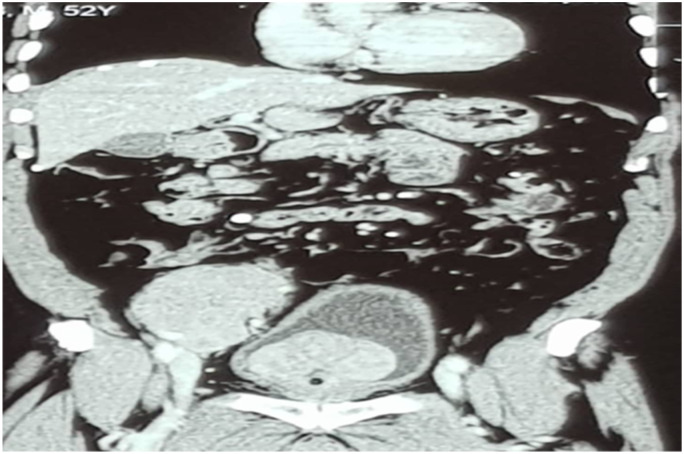
Abdomino-pelvic ultrasound scan done (Figure 1) showed a huge hypodense mass lying at the para-aortic region measuring 9.58 cm by 4.54 cm with an enlarged prostate. Also, Abdomino-pelvic CT scan showed a heterogeneously hypodense multilobulated mass measuring 9.1 cm by 9.1 cm by 5.9 cm in the right iliac region anterior to the right psoas muscle and right iliac vessels (Figure 2). Other investigations, full blood count, urinalysis, ECG and renal function tests (E/U/Cr) were unremarkable. A preoperative diagnosis of Intra-abdominal mass (likely lymphoma) was made.
Figure 1.
USS showing an intra abdominal mass.
Figure 2.
CT scan showing hypodense multi-lobulated mass anterior to the right psoas muscle.
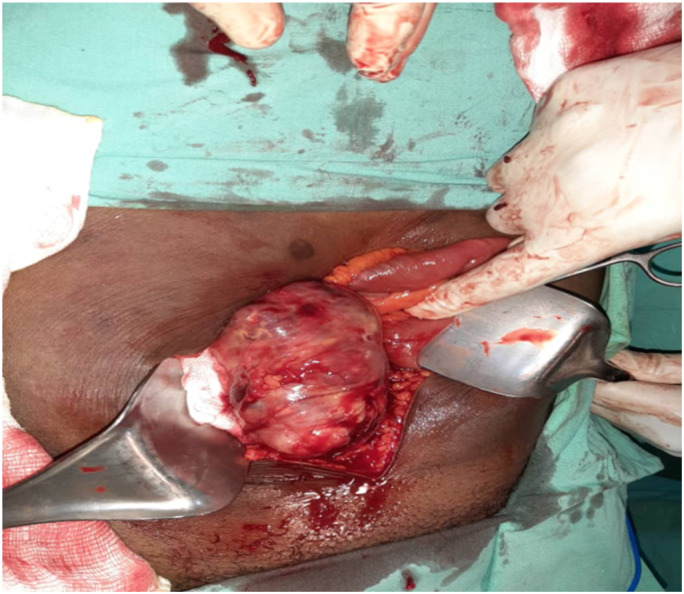
He was admitted and worked up for surgery. Had exploratory laparotomy with excision biopsy of the mass under general anaesthesia and endotracheal intubation. At laparotomy, access was hindered by multiple adhesions between part of the terminal ileum and the anterior surface of the tumour. The adhesions were divided between haemostats, transfixed with vicryl 2/0 and haemostasis secured using diathermy. The mass, (Figure 3) was separated from the tumour bed at the right side of the retroperitoneum by blunt and sharp dissection. Haemorrhage was controlled by ties and diathermy. Estimated blood loss was about 1.2 L. Intraoperative findings were: Clear peritoneal fluid, right iliac fossa retroperitoneal mass, oval and measuring 9 cm by 6 cm; numerous blood vessels on the tumour bed feeding the tumour; a lymph node that measured about 2 cm in diameter located at the supero-posterior aspect of the urinary bladder. Further exploration revealed no evidence of metastasis. The lymph node was excised and thereafter tagged. An abdominal drain was left in-situ directed at the tumour bed. The tumour and lymph node were sent for histology.
Figure 3.
Intraabdominal tumour in situ.
Postoperatively he was managed in the ward with intravenous fluids, antibiotics (Ceftriaxone and Metronidazole), and analgesics. The immediate postoperative period was uneventful. On postoperative day 2, the abdominal drain was removed: he also commenced graded oral intake. On postoperative day 3, he complained of painless right lower limb swelling and exanimation revealed postoperative right lower limb lymphoedema. He was immediately commenced on Deep vein thrombosis prophylaxis including compression stockings in addition to elevation of the limb. The lymphoedema decreased gradually and regressed, sutures were removed on the postoperative day 9 and was discharged.
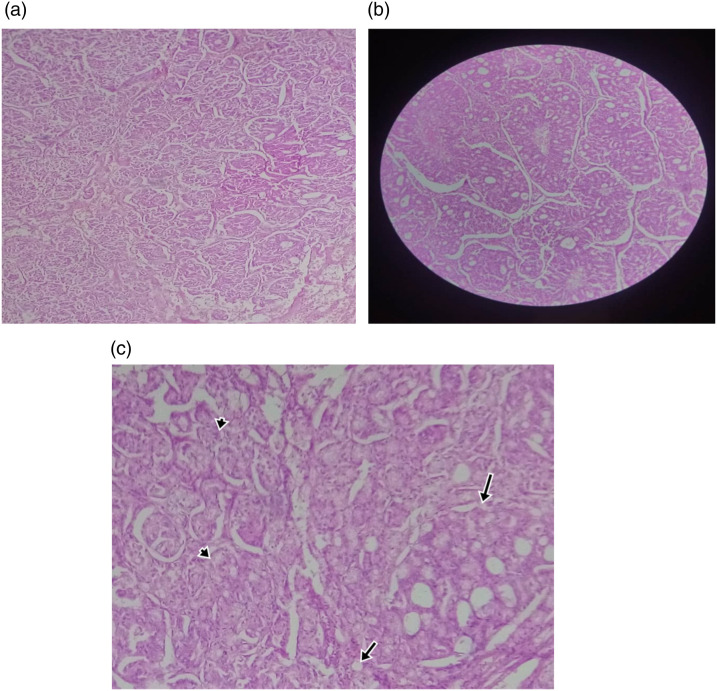
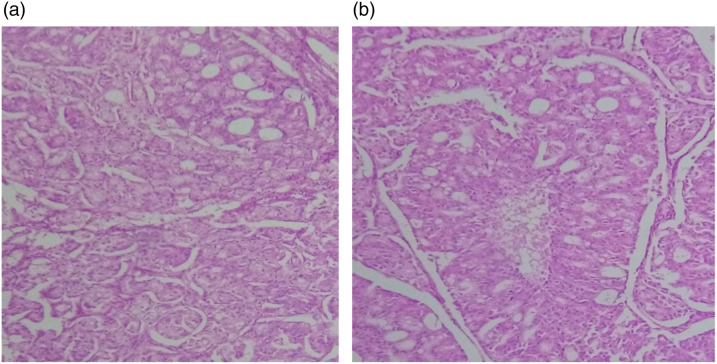
He presented to the surgical outpatient department after 2 weeks for follow up alongside with the histology report: Macroscopy, the tumour and the lymph node measured 9 × 7 × 4cm and 3 × 1 × 0.5 cm respectively. On microscopy, the tumour composed of irregularly distributed tubular structure in a desmoplastic stroma with areas of complex cribriform architecture and intraluminal “dirty” necrosis. Elsewhere was seen uniform nests of cells with small hyperchromatic nuclei and pale eosinophilic cytoplasm and granular chromatin pattern consistent with mixed neuroendocrine non-endocrine neoplasm- low grade, (Figures 4 and 5 showing different sections and magnification of the tumour), a similar lesion was seen in the lymph node. The definitive diagnosis was mixed neuroendocrine non-neuroendocrine neoplasm, low grade. Follow up 12 months post-surgery revealed no evidence of tumour recurrence.
Figure 4.
(a) Mixed neuroendocrine (b) non-neuroendocrine neoplasm, H & E × 40 (c) mixed neuroendocrine(arrow heads), non-neuroendocrine(arrows), H & E × 100.
Figure 5.
(a) Mixed neuroendocrine, (b) non-neuroendocrine neoplasm, H & E × 100.
Discussion
The incidence of MiNENs is underestimated due to the limitations in method of diagnosis, complicated by heterogenicity in pathology, poor awareness worsened by assignment of different definitions of the lesion overtime (mixed adenoneuroendocrine carcinoma-MANEC now renamed MiNEN). 1 Since the first case of MANEC was described in 1924, 3 few cases have been reported in literature due to above stated reasons. This is the first case reported from our facility. Literature search didn’t reveal reports from Nigeria and Africa.
The tumour usually presents in the 6th and 7th decades 9 in keeping with ours in the 7th decade.
The mixed neoplasms demonstrate variable morphological features: Composite tumours with juxtaposed areas of the tumour, Collision tumour with separate juxtaposed area of tumour mass. May be the neuroendocrine and non-neuroendocrine coexist at a cellular level- Amphicrine tumour. 1 The pathogenesis of the tumour is still a matter of debate with three major proposed theories: First postulate suggests that the neuroendocrine and non-neuroendocrine parts arise independently from distinct precursor cells and merge thereafter. The second suggests that both components come from a common pluripotential stem cell that acquires dual phenotypic differentiation during carcinogenesis. The third postulate assumes a common monoclonal origin of both components but that the neuroendocrine differentiation develops from an initial non-neuroendocrine phenotype from progressive accumulation of molecular/genetic aberrations and not vice versa- Amphicrine tumour. 1
Pathogenesis of this tumour remains controversial, however molecular studies point towards a common monoclonal origin of the two components an area of further research. The molecular landscape of MiNENs though not well understood will in future identify potential targets for novel therapeutic approaches. 1
Clinical presentation depends on the neuroendocrine cell secretion following which the tumours are classified as functioning and nonfunctioning. 2 Functioning tumours are carcinoids based on the substances secreted. Nonfunctioning tumours may be discovered due to local effect as seen in our patient. Rarely they can present as acute abdomen. 10
Pathological assessment of Haematoxylin and Eosin (H&E) stained sections are essential for detecting the neuroendocrine and nonneuroendocrine components for the diagnosis of MiNEN. 11 Their clinical relevance has increased since the use of immunohistochemistry (IHC) was introduced in the work-up of tumours, facilitating the possibility of their identification with a consequent increase in diagnosis. 5 IHC markers used in detecting and grading the endocrine component should be accompanied by markers appropriate for the type of nonneuroendocrine component. Chromogranin A (CgA) and synaptophysin are currently considered the most specific IHC markers for neuroendocrine neoplasms. However, the findings of IHC alone are not sufficient for the diagnosis of MiNEN, the histopathological findings should be present in each morphological component. 11 Similarly, the possibility of the negative influence of a small component of high-grade NEN on tumour behaviour should not be discountenanced hence there is need for the 30% cut-off value to be reaffirmed by systematic studies. 11 The importance of morphological evaluation is emphasized as the current therapeutic approach depends on the grade of MiNEN with each component evaluated and graded separately. 11
The WHO in 2017 renamed MANEC from pancreas as MiNEN retaining the 30% threshold for each component, however the term exocrine and carcinoma was replaced by the term “neoplasm” since both components could be low grade malignancy 4 in keeping with the report of the index patient. Recently the WHO extended the use of the term to all neoplasms meeting the diagnostic criteria for MiNENs arising from any site in 2019. 10
Following the low incidence of these tumours, treatment plan can be challenging. MiNENs are usually treated similarly to their pure NEC counterpart. Alternatively, when the exocrine component is preponderant, some clinicians choose to apply the standard of care for adenocarcinoma from same site of origin. Standard of care is not well developed but a few reports have shown successful outcomes with Surgery as in our index patient. 12 A previous study with 80 patients showed that 3 years overall survival in patients with adenocarcinoma dominant tumour was higher with lower recurrence rates compared to neuroendocrine dominant tumours. 13 The median overall survival for localized MiNENs, including regional lymph node metastases (Stages I-III), is 39 months. For advanced disease with distant metastasis (Stage IV) median overall survival is 11 months. 14 Prognosis in our patient is guarded as he had metastasis to the adjoining lymph node. The tumours are graded into low, intermediate and high grade, 5 our patient had a low grade tumour perhaps in keeping with the absence of recognizable recurrence 12 months post-surgery.
In conclusion, MiNENs should be considered as a differential diagnosis in a patient with intra-abdominal mass. It may attain a large size prior to diagnosis when it originates from the retroperitoneum. Despite the huge size, outcome following resection may be good especially with localized, low grade tumours.
Acknowledgments
We acknowledge the patient and all the participating author for their contribution to the paper.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
Contributorship: ALL authors contributed in different aspect of the report.
Ethical statement
Ethical approval
(include full name of committee approving the research and if available mention reference number of that approval): The requirement for ethics approval was waived because this study was a case report of a patient managed by our team, not an interventional study (i.e., no intervention or experimentation was carried out on the patient for the purpose of the study).
Informed consent
The patient provided both verbal and written informed consent for the reporting of this case and all accompanying images.
ORCID iDs
John Adi Ashindoitiang https://orcid.org/0000-0003-0763-9707
Alfred Ottoh Eyong https://orcid.org/0000-0001-6351-877X
References
- 1.Frizziero M, Chakrabarty B, Nagy B, et al. Mixed neuroendocrine non-neuroendocrine neoplasms: a systematic review of a controversial and underestimated diagnosis. J Clin Med 2020; 9(273): 1–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Johnson PR. Gastroenteropancreatic neuroendocrine (carcinoid) tumors in children. Semin Pediatr Surg 2014; 23: 91–95. [DOI] [PubMed] [Google Scholar]
- 3.Bosman FT, Carneiro F, Hruban RH, et al. WHO classification of tumours of the digestive system. 4th ed. Lyon France: International Agency for Research on Cancer(IARC), 2010. [Google Scholar]
- 4.Lloyd RV, Osamma RY, Kloppel G, et al. WHO Classification of Tumours of endocrine organs. 4th ed. Lyon France: International Agency on Research for Cancer(IARC), 2017. [Google Scholar]
- 5.La Rosa S, Sessa F, Uccella S. Mixed neuroendocrine-nonneuroendocrine neoplasms (MiNENs): unifying the concept of a heterogeneous group of neoplasms. Endocr Pathol 2016; 27: 284–311. [DOI] [PubMed] [Google Scholar]
- 6.Xu B, Zhang F, Wu R, et al. Incidence, survival and prognostic factors for patients with gastrointestinal mixed neuroendocrine and non-neuroendocrine neoplasms: a SEER population-based study. Cancer Res Clin Oncol. 2003; 3: 64. DOI: 10.1009/s00432-023-05356-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Shah R, Nalamati GP. Neuroendocrine tumours of the colon and rectum. Semin Colon Rectal Surg 2016; 26: 60–63. [Google Scholar]
- 8.Barkaoui ZE, Najih M, Mehdi AE, et al. Mixed neuroendocrine-non-neuroendocrine neoplasms of the right colon: a case report. Pan Afr Med J 2021; 40: 243. DOI: 10.11604/pamj.2021.40.243.30627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yeo S, Primary mixedadeno-neuroendocrine carcinoma(MANEC) of the appendix presenting with small bowel obstruction and requiring emergency laparotomy. Clin Surg 2018; 1: 204–207. [Google Scholar]
- 10.WHO classification of tumour editorial board, digestive system tumours. 5th ed. Lyon, France: International Agency for Research on Cancer(IARC), 2019. [Google Scholar]
- 11.Elpek GO. Mixed neuroendocrine- nonneuroendocrine neoplasms of the gastrointestinal system: an update. World J Gastroenterol 2022; 28(8): 794–810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Millet C, Farokhian A, Mekheal N, et al. Massive mixed adenoneuroendocrine carcinoma: a case report. Cureus 2021; 13(6): e15928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Richards D, Davis D, Yan P, et al. Unusual case of small cell gastric carcinoma. Case report and literature review. Dis Sci 2011; 56: 951–957. [DOI] [PubMed] [Google Scholar]
- 14.Toor D, Loree JM, Gao ZH, et al. Mixed neuroendocrine-non-neuroendocrine neoplasms of the digestive system: a mini-review. World J Gastroenterol 2022; 28(19): 2076–2087. DOI: 10.3748/wjg.v28.i19.2076. [DOI] [PMC free article] [PubMed] [Google Scholar]