Abstract
Background
As the population of people with HIV ages, concerns over managing age-related comorbidities, polypharmacy, immune recovery, and drug-drug interactions while maintaining viral suppression have arisen. We present pooled TANGO and SALSA efficacy and safety results dichotomized by age (< 50 and ≥ 50 years).
Methods
Week 48 data from the open-label phase 3 TANGO and SALSA trials evaluating switch to once-daily dolutegravir/lamivudine (DTG/3TC) fixed-dose combination vs continuing current antiretroviral regimen (CAR) were pooled. Proportions of participants with HIV-1 RNA ≥ 50 and < 50 copies/mL (Snapshot, intention-to-treat exposed) and safety were analyzed by age category. Adjusted mean change from baseline in CD4 + cell count was assessed using mixed-models repeated-measures analysis.
Results
Of 1234 participants, 80% of whom were male, 29% were aged ≥ 50 years. Among those aged ≥ 50 years, 1/177 (< 1%) DTG/3TC participant and 3/187 (2%) CAR participants had HIV-1 RNA ≥ 50 copies/mL at 48 weeks; proportions with HIV-1 RNA < 50 copies/mL were high in both treatment groups (≥ 92%), consistent with overall efficacy and similar to observations in participants aged < 50 years (≥ 93%). Regardless of age category, CD4 + cell count increased or was maintained from baseline with DTG/3TC. Change from baseline in CD4 + /CD8 + ratio was similar across age groups and between treatment groups. One CAR participant aged < 50 years had confirmed virologic withdrawal, but no resistance was detected. In the DTG/3TC group, incidence of adverse events (AEs) was similar across age groups. Proportions of AEs leading to withdrawal were low and comparable between age groups. Although drug-related AEs were generally low, across age groups, drug-related AEs were more frequent in participants who switched to DTG/3TC compared with those who continued CAR. While few serious AEs were observed in both treatment groups, more were reported in participants aged ≥ 50 years vs < 50 years.
Conclusions
Among individuals with HIV-1, switching to DTG/3TC maintained high rates of virologic suppression and demonstrated a favorable safety profile, including in those aged ≥ 50 years despite higher prevalence of concomitant medication use and comorbidities.
Trial registration number: TANGO, NCT03446573 (February 27, 2018); SALSA, NCT04021290 (July 16, 2019).
Supplementary Information
The online version contains supplementary material available at 10.1186/s12981-024-00604-9.
Keywords: Aging, Comorbidity, DTG/3TC, HIV-1, Polypharmacy, Single-tablet regimen, Suppressed switch, ≥ 50 years
Background
The availability of highly effective antiretroviral therapy (ART) has resulted in life expectancy for people with HIV similar to that of the general population [1, 2]. Further, the number of people aged ≥ 50 years acquiring HIV, who are often diagnosed later in the disease course, has increased [2]. As a consequence, globally, the proportion of individuals with HIV aged ≥ 50 years rose from 8% in 2000 to 16% in 2016 and to an estimated 24% in 2022 [3–5]. In 2022, in Western and Central Europe and North America, nearly half of all individuals with HIV were aged ≥ 50 years [5]. Predictive modeling has estimated that by 2030, 70% of people with HIV in the United States will be aged > 50 years [6], and ~ 25% will be aged > 65 years [7]. Similarly, predictive modeling using cohorts from the Netherlands and France has estimated that the population of people with HIV aged ≥ 50 years will reach 73% and > 66%, respectively, by 2030 [8, 9]. However, although individuals aged ≥ 50 years are among the fastest growing segment of the population with HIV [10], they have historically been underrepresented in or excluded from HIV clinical trials [11, 12].
As people with HIV age, treatment requirements to support healthy living extend beyond achieving and maintaining virologic suppression to also managing age-related comorbidities, polypharmacy, and other healthcare priorities. Therefore, addressing the treatment needs of individuals aged ≥ 50 years while maintaining virologic suppression may present additional considerations compared with treatment of those aged < 50 years [13]. Evidence suggests that people with HIV have increased rates and earlier presentation of comorbidities compared with those without HIV, and prevalence of comorbidities increases with age [6, 14, 15]. Additionally, studies have demonstrated more frequent use of concomitant medications as individuals with HIV age [15, 16], which may result in significant drug-drug interactions with ART. Moreover, ART adherence, and consequently viral suppression, have been negatively associated with polypharmacy [17], particularly in individuals aged ≥ 50 years [18, 19]. Therefore, evaluating the efficacy, safety, and tolerability of ART in people with HIV as they age is critical.
Some of the challenges associated with polypharmacy and increased use of concomitant non-ART medications may be alleviated by using single-tablet or 2-drug ART regimens instead of multi-tablet or 3- or 4- drug regimens. Dolutegravir/Lamivudine (DTG/3TC) is a 2-drug, single-tablet regimen that is recommended in international guidelines and has demonstrated durable efficacy, a high barrier to resistance, and good safety and tolerability as first-line ART in treatment-naive participants in the phase 3 GEMINI-1 and GEMINI-2 trials and as a switch option in virologically suppressed participants in the phase 3 TANGO and SALSA studies [7, 20–22]. Subgroup analyses of TANGO and SALSA showed high efficacy and a good safety profile for DTG/3TC in participants aged ≥ 50 years, but sample sizes were small (TANGO, n = 79; SALSA, n = 98) [23, 24]. Here, we present efficacy and safety of DTG/3TC in a larger sample of participants aged ≥ 50 years by pooling the analyses from the TANGO and SALSA studies.
Methods
Study design
This analysis included pooled Week 48 data from the open-label phase 3 TANGO and SALSA trials evaluating switch to once-daily DTG/3TC fixed-dose combination vs continuing current antiretroviral regimen (CAR). Detailed methodology for the TANGO (ClinicalTrials.gov, NCT03446573) and SALSA (ClinicalTrials.gov, NCT04021290) studies has previously been published [20, 21]. In brief, eligibility criteria included no prior virologic failure, no documented nucleoside reverse transcriptase inhibitor (NRTI) or integrase strand transfer inhibitor (INSTI) resistance, no hepatitis B virus infection or need for hepatitis C virus therapy, and documented virologic suppression (HIV-1 RNA < 50 copies/mL for > 6 months). Exclusion criteria included plasma HIV-1 RNA ≥ 50 copies/mL within 6 months of screening or ≥ 2 measurements ≥ 50 copies/mL or any measurement > 200 copies/mL within 6 and 12 months of screening. Pooled data were dichotomized by age categories (< 50 and ≥ 50 years). Data were also summarized in a third category (≥ 65 years), but due to the low numbers of participants, no statistical analyses for this category were conducted.
Both studies were conducted in accordance with the International Conference on Harmonization Good Clinical Practice and followed the principles of the Declaration of Helsinki; all participants provided written informed consent before study initiation.
Procedures
Detailed procedures have been previously published [20, 21]. Briefly, eligible participants were randomized 1:1 to switch to once-daily DTG/3TC fixed-dose combination or continue their current ART regimen. In TANGO, participants switched from tenofovir alafenamide/emtricitabine (TAF/FTC) plus a protease inhibitor (PI), INSTI, or non-nucleoside reverse transcriptase inhibitor (NNRTI). Participants with initial tenofovir disoproxil fumarate (TDF) treatment who switched to TAF ≥ 3 months before screening, with no changes to other drugs in their regimen, were also eligible. In SALSA, participants on uninterrupted ART for ≥ 3 months switched from regimens composed of 2 NRTIs plus a PI, INSTI, or NNRTI. No regimen modifications were allowed on study, except switching between ritonavir and cobicistat (TANGO, SALSA) or 3TC and FTC (SALSA) in the CAR group. Study visits were planned at baseline (Day 1) and Weeks 4, 8 (TANGO only), 12, 24, 36, and 48. Plasma for HIV-1 RNA quantification was collected and safety outcomes were assessed at each visit.
Outcomes
The primary endpoint was the proportion of participants with HIV-1 RNA ≥ 50 copies/mL at Week 48 using the US Food and Drug Administration Snapshot algorithm in the intention-to-treat–exposed (ITT-E) population. Key secondary endpoints included proportion of participants with HIV-1 RNA < 50 copies/mL (Snapshot, ITT-E) and proportion of participants meeting confirmed virologic withdrawal (CVW) criteria, defined as HIV-1 RNA ≥ 50 copies/mL followed by a second consecutive on-treatment HIV-1 RNA ≥ 200 copies/mL. Other secondary endpoints included change from baseline in CD4 + cell count and CD4 + /CD8 + ratio. Safety assessments included incidence and severity of adverse events (AEs) as well as change from baseline in weight, lipid parameters, and renal and bone biomarkers.
Statistical analysis
All randomized participants who received ≥ 1 dose of study treatment were included in the ITT-E population, which was used for efficacy and safety analyses. Proportions of participants with HIV-1 RNA ≥ 50 copies/mL and < 50 copies/mL (Snapshot, ITT-E) at Week 48 were analyzed using a Cochran-Mantel–Haenszel test adjusting for baseline third agent class. Mixed-models repeated-measures analysis was used for adjusted mean change from baseline in CD4 + cell count, CD4 + /CD8 + ratio, weight, lipid parameters, and renal and bone biomarkers in the 2 age groups (< 50 and ≥ 50 years). Adjustment terms included treatment, visit, age, sex, race, baseline value, baseline third agent class, baseline CD4 + cell count, treatment-by-visit interaction, baseline value-by-visit interaction, and study, with visit as the repeated factor; subgroup analyses by age were also adjusted for visit-by-age, treatment-by-age, and treatment-by-visit-by-age interactions. Baseline body mass index (BMI) was an additional adjustment term for CD4 + cell count and CD4 + /CD8 + ratio. Baseline TAF and TDF use were additional adjustment terms for weight. Baseline BMI, diabetes, and hypertension were additional adjustment terms for renal biomarkers. Baseline BMI, smoking history, and vitamin D use were additional adjustment terms for bone biomarkers. If logistic regression models did not converge due to either small sample size or low event rates, a Firth penalized maximum likelihood regression model generating product-limit CIs was used instead [25]. To determine treatment regimen–adjusted likelihood of ≥ 10% weight gain at Week 48, a logistic Firth regression model analysis was used. Adjustment terms were treatment, sex, age, race, baseline third agent class, baseline CD4 + cell count, baseline weight, and study. Incidence and severity of AEs were summarized descriptively.
Results
Participants
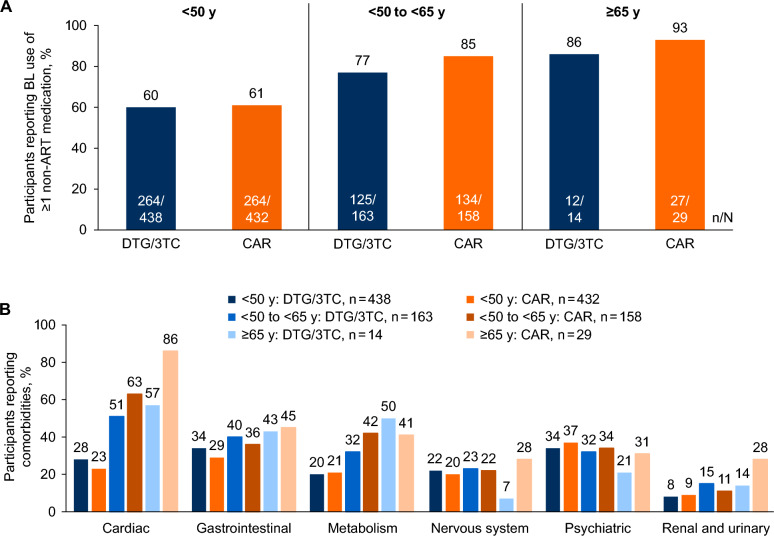
Of 1234 participants in the pooled TANGO and SALSA ITT-E population (DTG/3TC, n = 615; CAR, n = 619), 71% (n = 870) were aged < 50 years and 29% (n = 364) were aged ≥ 50 years (Table 1). Of note, 3% (n = 43) were aged ≥ 65 years. Overall, baseline characteristics were similar between the DTG/3TC and CAR groups. Baseline characteristics between the < 50 and ≥ 50 years age groups were generally similar across treatment groups. However, participants aged ≥ 50 years had greater concomitant medication use, more comorbidities, and longer prior ART duration compared with participants aged < 50 years. In both treatment groups, the proportions of participants with baseline non-ART medication use and comorbidities increased with age (Fig. 1). Cardiac, gastrointestinal, and metabolism disorders were the most prevalent comorbidities in participants aged ≥ 50 years. Across all participants, 131 (11%) had polypharmacy (use of > 5 medications) at baseline. The most commonly used (> 5% of the overall population) concomitant medications were cholecalciferol (9%), vitamins (not otherwise specified; 8%), and ibuprofen (6%).
Table 1.
Demographics and Baseline Characteristics Overall and by Age: TANGO and SALSA Pooled ITT-E Population
| Parameter | Overall | < 50 y | 50 to < 65 y | ≥ 65 y | ||||
|---|---|---|---|---|---|---|---|---|
| DTG/3TC (N = 615) |
CAR (N = 619) |
DTG/3TC (N = 438) |
CAR (N = 432) |
DTG/3TC (N = 163) |
CAR (N = 158) |
DTG/3TC (N = 14) |
CAR (N = 29) |
|
| Sex, female, n (%) | 133 (22) | 117 (19) | 68 (16) | 71 (16) | 59 (36) | 38 (24) | 6 (43) | 8 (28) |
| Age, median (range), y | 42 (20–74) | 42 (18–83) | 37 (20–49) | 36 (18–49) | 56 (50–64) | 55 (50–64) | 67 (65–74) | 69 (65–83) |
| Weight, median (range), kg | 77 (43–154) | 78 (36–160) | 78 (43–154) | 77 (48–160) | 75 (44–128) | 80 (36–127) | 72 (59–106) | 75 (49–116) |
| BMI, median (range), kg/m2 | 25 (17–51) | 26 (14–69) | 25 (17–51) | 25 (17–69) | 25 (18–43) | 27 (14–45) | 26 (18–32) | 27 (19–44) |
| Baseline use of ≥ 1 non-ART medication, n (%) | 401 (65) | 425 (69) | 264 (60) | 264 (61) | 125 (77) | 134 (85) | 12 (86) | 27 (93) |
| Number of baseline non-ART medications, median (range) | 1 (0–13) | 1 (0–20) | 1 (0–12) | 1 (0–16) | 2 (0–13) | 3 (0–20) | 2 (0–6) | 4 (0–11) |
| Baseline polypharmacy (> 5 medications), n (%)a | 55 (9) | 76 (12) | — | — | — | — | — | — |
| Baseline comorbidities, n (%) | 457 (74) | 474 (77) | 311 (71) | 307 (71) | 134 (82) | 138 (87) | 12 (86) | 29 (100) |
| Baseline CD4 + cell count, median (range), cells/mm3 |
680 (133–2089) |
684 (94–1954) |
685 (154–1904) |
686 (94–1954) |
653 (133–2089) |
677 (119–1530) |
638 (228–1199) |
657 (122–1133) |
| Baseline CD4 + /CD8 + ratio, mean (SD) | 1.1 (0.54) | 1.1 (0.50) | 1.0 (0.51) | 1.0 (0.44) | 1.1 (0.61) | 1.1 (0.65) | 1.2 (0.58) | 1.0 (0.46) |
| Duration of ART before Day 1, median (range), mo | 41 (4–240) | 45 (7–253) | 37 (4–188) | 41 (7–206) | 61 (7–240) | 57 (9–253) | 58 (8–201) | 74 (20–214) |
| Baseline NRTI, n/N (%)b | ||||||||
| TDF | 109/605 (18) | 110/606 (18) | 70/431 (16) | 73/422 (17) | 34/160 (21) | 31/155 (20) | 5/14 (36) | 6/29 (21) |
| TAF | 451/605 (75) | 462/606 (76) | 340/431 (79) | 333/422 (79) | 104/160 (65) | 109/155 (70) | 7/14 (50) | 20/29 (69) |
| ABC | 45/605 (7) | 34/606 (6) | 21/431 (5) | 16/422 (4) | 22/160 (14) | 15/155 (10) | 2/14 (14) | 3/29 (10) |
| Baseline third agent, n (%) | ||||||||
| INSTI | 387 (63) | 394 (64) | 295 (67) | 285 (66) | 83 (51) | 97 (61) | 9 (64) | 12 (41) |
| NNRTI | 174 (28) | 172 (28) | 109 (25) | 110 (25) | 61 (37) | 47 (30) | 4 (29) | 15 (52) |
| PI | 54 (9) | 53 (9) | 34 (8) | 37 (9) | 19 (12) | 14 (9) | 1 (7) | 2 (7) |
ABC abacavir, ART antiretroviral therapy, BMI body mass index, CAR current antiretroviral regimen, DTG dolutegravir, INSTI integrase strand transfer inhibitor, ITT-E intention-to-treat exposed, NNRTI non-nucleoside reverse transcriptase inhibitor, NRTI nucleoside reverse transcriptase inhibitor, PI protease inhibitor, TAF tenofovir alafenamide, 3TC lamivudine, TDF tenofovir disoproxil fumarate
aData available for overall treatment groups only. bOther NRTI backbone regimens were included in SALSA (zidovudine, tenofovir disoproxil succinate, biovir [not otherwise specified])
Fig. 1.
Baseline A use of ≥ 1 non-ART medication and B comorbidities by age. TANGO and SALSA pooled ITT-E population. BL baseline, CAR current antiretroviral regimen, DTG dolutegravir, ITT-E intention-to-treat exposed, 3TC lamivudine
Efficacy
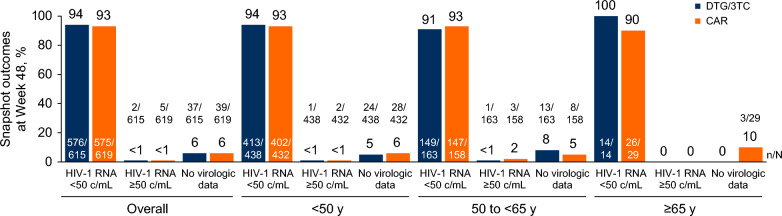
Proportions of participants with HIV-1 RNA ≥ 50 copies/mL (Snapshot, ITT-E) at Week 48 were similar among all participants regardless of age and across treatment groups (DTG/3TC vs CAR: < 50 years, < 1% [1/438] vs < 1% [2/432]; ≥ 50 years, < 1% [1/177] vs 2% [3/187]), which was consistent with results from the overall study population (DTG/3TC, < 1% [2/615]; CAR, < 1% [5/619]; Fig. 2). Notably, no participants aged ≥ 65 years had HIV-1 RNA ≥ 50 copies/mL in either treatment group. Similarly, proportions of participants with HIV-1 RNA < 50 copies/mL were high across all age and treatment groups (DTG/3TC vs CAR: < 50 years, 94% [413/438] vs 93% [402/432]; 50 to < 65 years, 91% [149/163] vs 93% [147/158]; ≥ 65 years, 100% [14/14] vs 90% [26/29]) and consistent with overall study results (DTG/3TC, 94% [576/615]; CAR, 93% [575/619]). Regardless of age category, no participants in the DTG/3TC group met CVW criteria, while 1 participant aged < 50 years in the CAR group met CVW criteria with no resistance detected.
Fig. 2.
Snapshot virologic outcomes at Week 48 overall and by age. TANGO and SALSA pooled ITT-E population. CAR current antiretroviral regimen, c/mL copies/mL, DTG dolutegravir, ITT-E intention-to-treat exposed, 3TC lamivudine
Across both age and treatment groups, adjusted mean change (SE) in CD4 + cell count from baseline to Week 48 was similar (DTG/3TC vs CAR: < 50 years, 29.0 [8.5] vs 7.6 [8.2] cells/mm3; ≥ 50 years, 6.3 [13.6] vs − 24.7 [12.5] cells/mm3), which reflected results from the overall study population (DTG/3TC, 22.4 [7.2] vs CAR, − 2.0 [6.9] cells/mm3; Additional file 1). Similar results were also observed for adjusted mean change (SE) from baseline to Week 48 in CD4 + /CD8 + ratio (DTG/3TC vs CAR: < 50 years, 0.039 [0.010] vs 0.048 [0.010]; ≥ 50 years, 0.032 [0.016] vs 0.062 [0.016]; overall, 0.037 [0.008] vs 0.052 [0.009]).
Safety
Adverse events
Incidence of AEs was comparable across age and treatment groups (Table 2). Adverse events leading to withdrawal and drug-related AEs were low and comparable between age groups. While serious AEs (SAEs) were generally low, more SAEs were observed in participants aged ≥ 50 years in both treatment groups (DTG/3TC vs CAR: < 50 years, 4% [18/438] vs 4% [16/432]; 50 to < 65 years, 5% [8/163] vs 8% [12/157]; ≥ 65 years, 14% [2/14] vs 14% [4/29]). In all age categories, AE incidence was comparable between the DTG/3TC and CAR groups, although drug-related AEs were more frequent in participants who switched to DTG/3TC compared with those who continued CAR (DTG/3TC vs CAR: < 50 years, 15% [65/438] vs 4% [18/432]; 50 to < 65 years, 15% [25/163] vs 2% [3/157]; ≥ 65 years, 21% [3/14] vs 0% [0/29]), which was consistent with the overall population (DTG/3TC, 15% [93/615]; CAR, 3% [21/618]).
Table 2.
Adverse Events Through Week 48 Overall and by Age: TANGO and SALSA Pooled Safety Populationa
| Parameter, n (%) | Overall | < 50 y | 50 to < 65 y | ≥ 65 y | ||||
|---|---|---|---|---|---|---|---|---|
| DTG/3TC (N = 615) |
CAR (N = 618) |
DTG/3TC (N = 438) |
CAR (N = 432) |
DTG/3TC (N = 163) |
CAR (N = 157) |
DTG/3TC (N = 14) |
CAR (N = 29) |
|
| Any AE | 475 (77) | 464 (75) | 333 (76) | 330 (76) | 133 (82) | 113 (72) | 9 (64) | 21 (72) |
| AEs leading to withdrawal | 18 (3) | 5 (< 1) | 11 (3) | 2 (< 1) | 7 (4) | 2 (1) | 0 | 1 (3) |
| Grade 2–5 AEs | 281 (46) | 303 (49) | 189 (43) | 206 (48) | 85 (52) | 82 (52) | 7 (50) | 15 (52) |
| Drug-related AEs | 93 (15)b | 21 (3)c | 65 (15) | 18 (4) | 25 (15) | 3 (2) | 3 (21)d | 0 |
| Serious AEse | 28 (5)f | 32 (5) | 18 (4)f | 16 (4) | 8 (5) | 12 (8) | 2 (14) | 4 (14) |
AE adverse event, CAR current antiretroviral regimen, DTG dolutegravir, 3TC lamivudine, TDF tenofovir disoproxil fumarate
aIn TANGO, 1 participant was found to be taking a TDF-based regimen and was excluded from the safety population. bThe most common (≥ 0.5%) grade 2–5 drug-related AEs reported were grade 2 insomnia (7/615 [1.1%]) and weight increase (4/615 [0.7%]); there were no grade ≥ 3 drug-related AEs. cThe most common grade 2–5 drug-related AE was grade 2 gastroesophageal reflux disease (2/618 [0.3%]); 2 grade 3 drug-related AEs were reported (hypertriglyceridemia (1/618 [0.2%]) and hypercholesterolemia (1/618 [0.2%]). d4 drug-related AEs were reported (flatulence, vertigo, abnormal dreams, and renal impairment). eThere were no drug-related serious AEs. f2 non–drug-related fatal serious AEs were reported (gunshot wound [homicide] and unknown cause)
Weight
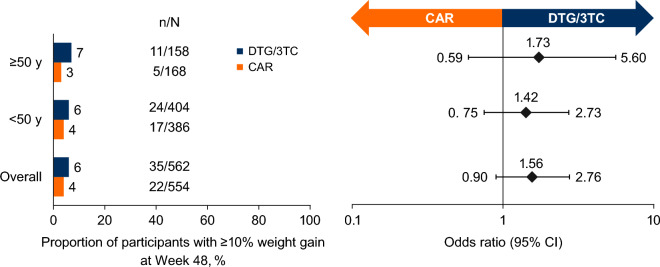
Regardless of age category, the DTG/3TC group experienced greater weight gain from baseline at Week 48 than the CAR group, although changes in weight were small. The difference between treatment groups was not significant in participants aged < 50 years (adjusted mean change [SE] for DTG/3TC vs CAR: 1.28 [0.23] vs 1.02 [0.20] kg; treatment difference [95% CI]: 0.27 [− 0.32, 0.86]) but was significant in those aged ≥ 50 years (adjusted mean change [SE] for DTG/3TC vs CAR: 1.28 [0.36] vs 0.03 [0.30] kg; treatment difference [95% CI]: 1.24 [0.33–2.16]). These results were consistent with the overall study results (adjusted mean change [SE]: DTG/3TC, 1.28 [0.19] vs CAR, 0.71 [0.17] kg). Weight gain observed with DTG/3TC was similar between participants aged < 50 vs ≥ 50 years (1.28 kg each), while greater weight increases were observed with CAR in participants aged < 50 vs ≥ 50 years (1.02 vs 0.03 kg, respectively). Among participants aged ≥ 50 years, the difference in weight observed with DTG/3TC vs CAR was significant in female participants (adjusted mean change [SE]: 1.86 [0.61] vs − 0.21 [0.62] kg; treatment difference [95% CI]: 2.08 [0.40–3.75]) but was not significant in male participants (adjusted mean change [SE]: 0.96 [0.45] vs 0.11 [0.35] kg; treatment difference [95% CI]: 0.85 [− 0.26, 1.96]). In the overall pooled population, greater weight gain observed in the DTG/3TC group was mostly driven by outcomes from the SALSA study (adjusted mean change [SE] for DTG/3TC vs CAR: SALSA, 2.01 [0.33] vs 0.64 [0.25] kg; TANGO, 0.82 [0.23] vs 0.77 [0.22] kg; treatment difference [95% CI]: SALSA, 1.37 [0.55–2.19]; TANGO, 0.05 [− 0.57, 0.67]). Within treatment groups, the proportion of participants with ≥ 10% weight gain was low and similar across age groups (DTG/3TC vs CAR: < 50 years, 24/404 [6%] vs 17/386 [4%]; odds ratio [95% CI], 1.42 [0.75–2.73]; ≥ 50 years, 11/158 [7%] vs 5/168 [3%]; odds ratio [95% CI], 1.73 [0.59–5.60]) and consistent with the overall study results (DTG/3TC, 35/562 [6%] vs CAR, 22/554 [4%]; odds ratio [95% CI], 1.56 [0.90–2.76]; Fig. 3).
Fig. 3.
Proportion of participants with ≥ 10% weight gain at Week 48 overall and by age. TANGO and SALSA pooled safety population. CAR current antiretroviral regimen, DTG dolutegravir, 3TC lamivudine
Metabolic parameters
Across both age categories, changes in lipids from baseline to Week 48 were small and generally favored DTG/3TC compared with CAR (Additional file 2). Changes from baseline to Week 48 in plasma/serum renal biomarkers were generally small in both treatment groups within each age group (Additional file 2). A greater decrease from baseline in estimated glomerular filtration rate (eGFR) based on serum cystatin C was observed in participants aged ≥ 50 years in both treatment groups compared with those aged < 50 years (adjusted mean [SE] change from baseline for DTG/3TC vs CAR: < 50 years, 0.812 [0.56] vs 0.003 [0.56] mL/min/1.73 m2; ≥ 50 years, − 3.79 [0.93] vs − 4.55 [0.89] mL/min/1.73 m2). Regardless of age category, changes in bone biomarkers from baseline to Week 48 were generally small and comparable between treatment groups (Additional file 2).
Discussion
Overall, pooled findings from the TANGO and SALSA trials demonstrated that DTG/3TC maintained high rates of virologic suppression 1 year after treatment switch with no reported resistance and small and similar changes in CD4 + cell count and CD4 + /CD8 + ratio vs continuing CAR among participants aged ≥ 50 years compared with those aged < 50 years.
Consistent with other studies [14–16], we observed a higher number of concomitant medications used and greater prevalence of comorbidities in participants aged ≥ 50 years. However, this did not impact efficacy and safety outcomes for participants aged ≥ 50 years who switched to DTG/3TC, demonstrating the robust efficacy and safety profile of DTG/3TC regardless of age. In individuals with HIV aged ≥ 50 years, who often have greater concomitant medication use and a higher risk of polypharmacy compared with those aged < 50 years, treatment with a 2-drug vs 3-drug regimen may have the added benefit of limiting the number of medications used, with potentially fewer drug-drug interactions. Few significant drug-drug interaction considerations are noted for DTG/3TC, and it may be an appropriate ART regimen for individuals taking multiple comedications for comorbid conditions as they age [26].
Regardless of age, we observed few AEs leading to withdrawal and SAEs, with comparable incidences between treatment groups. The incidence of SAEs was slightly higher in participants aged ≥ 50 years in both treatment groups, which may be associated with the increased prevalence of age-associated comorbidities observed in this population. As is expected in stable-switch studies such as TANGO and SALSA, drug-related AEs were more frequent in the DTG/3TC group compared with the CAR group across both age groups. Overall AE frequency and intensity were similar across age categories in both treatment groups.
We observed greater weight gain among participants receiving DTG/3TC compared with CAR in both age categories, although the difference was small. In this pooled analysis, this outcome was mostly driven by results from the SALSA study, which may be partly explained by some participants switching from potentially weight-suppressive agents, such as TDF and efavirenz as opposed to TANGO, in which all participants switched from a TAF-based regimen [27–29]. Despite similar absolute weight gain observed with DTG/3TC across age categories, the difference in weight gain between the DTG/3TC vs CAR groups was more pronounced in the ≥ 50 years age group due to lesser weight gain among participants aged ≥ 50 years compared with those aged < 50 years continuing CAR. The difference observed in the CAR group could potentially be due to participants aged ≥ 50 years being more likely to be taking an efavirenz- or TDF-based regimen than those aged < 50 years at study entry as well as having a longer duration of ART use.
Across both age categories, we observed no relevant changes from baseline to Week 48 in renal or bone biomarkers between treatment groups, and directionality of change was similar between age categories. As is expected in a population that is aging [30], decreases in eGFR were slightly greater in the ≥ 50 years age group in both treatment groups. Small changes in lipids generally favored DTG/3TC in both age groups. These results were consistent with 48-week results from the individual TANGO and SALSA studies as well as with a previous study in ART-naive participants [20–22].
There were some limitations to this study, including the relatively small sample of participants aged ≥ 50 and ≥ 65 years. Although the proportion of participants aged ≥ 50 years in TANGO and SALSA (29%) was consistent with the proportion aged ≥ 50 years in the global population of people with HIV (an estimated 24% in 2022) [5], participants aged ≥ 65 years are likely underrepresented (3%). Both studies had a predominantly White and male population, which is not representative of the global population of individuals living and aging with HIV and may limit the generalizability of the results. Furthermore, the heterogeneity of the CAR population in the SALSA study, using a variety of ART regimens, should be considered when interpreting results as switches from different agents may have differential effects.
Conclusions
Overall, switching to DTG/3TC maintained high rates of virologic suppression and baseline immunologic status among participants aged < 50 and ≥ 50 years, including those aged ≥ 65 years, with no confirmed virologic withdrawals. With a higher prevalence of non-ART medication use and comorbidities associated with age, a well-tolerated 2-drug, single-tablet regimen with robust efficacy such as DTG/3TC may help support the clinical management of individuals with HIV-1 aged ≥ 50 years, particularly those at risk of polypharmacy-driven drug-drug interactions due to evolving health needs and comorbidities.
Supplementary Information
Additional file 1. Adjusted Mean Change From Baseline to Week 48 in CD4 + Cell Count and CD4 + /CD8 + Ratio Overall and by Age: TANGO and SALSA Pooled ITT-E Population. Table showing change from baseline to Week 48 in CD4 + cell count and CD4 + /CD8 + ratio overall and by age
Additional file 2. Change from baseline in A fasting lipids, B plasma/serum renal biomarkers, and C bone biomarkers at Week 48 by age: TANGO and SALSA pooled safety population. Figure showing change from baseline to Week 48 in fasting lipids, renal biomarkers, and bone biomarkers by age
Acknowledgements
The authors thank the study participants and their families and caregivers, the investigators and site staff who participated in the TANGO and SALSA studies, and the ViiV Healthcare and GSK study team members. Editorial assistance was provided under the direction of the authors by Deborah Lew, PhD, and Jennifer Rossi, MA, ELS, MedThink SciCom, and was funded by ViiV Healthcare.
Abbreviations
- AE
Adverse event
- ART
Antiretroviral therapy
- BMI
Body mass index
- CAR
Current antiretroviral regimen
- CVW
Confirmed virologic withdrawal
- DTG/3TC
Dolutegravir/lamivudine
- eGFR
Estimated glomerular filtration rate
- FDC
Fixed-dose combination
- FTC
Emtricitabine
- INSTI
Integrase strand transfer inhibitor
- ITT-E
Intention-to-treat exposed
- NNRTI
Non-nucleoside reverse transcriptase inhibitor
- NRTI
Nucleoside reverse transcriptase inhibitor
- PI
Protease inhibitor
- SAE
Serious adverse event
- TAF
Tenofovir alafenamide
- TDF
Tenofovir disoproxil fumarate
Author contributions
BRW, RG, and MA-K contributed to the conception of the study. BRW, RG, GB, RM, AC, and MA-K contributed to the design of the study. SW, DES, MG, PEC, TL, KL, and PNK contributed to the acquisition of data. BRW, RG, and MA-K contributed to the analysis of data. RG, MK, and MA-K contributed to drafting the manuscript. All authors contributed to the interpretation of data, critically revising the manuscript for important intellectual content, and approved the manuscript for publication.
Funding
These studies were funded by ViiV Healthcare. The funder had a role in the conceptualization, design, data collection, analysis, and preparation of the manuscript. The decision to publish was made by the authors.
Availability of data and materials
Anonymized individual participant data and study documents can be requested for further research from www.clinicalstudydatarequest.com.
Declarations
Ethics approval and consent to participate
Both studies were conducted in accordance with the International Conference on Harmonization Good Clinical Practice and followed the principles of the Declaration of Helsinki; all participants provided written informed consent before study initiation.
Consent for publication
Not applicable.
Competing interests
SW has received investigator-initiated grants from Gilead Sciences, Merck, and ViiV Healthcare and has participated in advisory boards for Merck and ViiV Healthcare. DES has served as a consultant for Gilead Sciences and ViiV Healthcare. MG has received grants from Janssen and ViiV Healthcare and consulting fees and honoraria from Janssen, Gilead Sciences, and ViiV Healthcare. PEC has received consulting fees from Gilead Sciences, MSD, and ViiV Healthcare and honoraria from Gilead Sciences and ViiV Healthcare, and has participated in data safety monitoring or advisory boards for Moderna. TL has received funding from GSK, paid to his institution, and grants from Charité Berlin, DAGNÄ e.V., Gilead Sciences, Heidelberg ImmunoTherapeutics GmbH, Leberstiftungs-GmbH, Moderna, and MSD, paid to his institution. KL has received honoraria and travel support from Gilead Sciences, MSD, and ViiV Healthcare. PNK has received grants from Gilead Sciences, GSK, Merck, TheraTechnologies, and ViiV Healthcare, paid to her institution; has received consulting fees from Gilead Sciences, GSK, Merck, and ViiV Healthcare; has participated in data safety monitoring or advisory boards for Gilead Sciences, GSK, Merck, and ViiV Healthcare; and holds stock or stock options in Gilead Sciences, GSK, Johnson & Johnson, Merck, Moderna, and Pfizer. BW, RG, GB, RM, CO, MK, BJ, AC, and MA-K are employees of ViiV Healthcare or GSK and may own stock in GSK.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Sematlane NP, Knight L, Masquillier C, Wouters E. Adapting to, integrating and self-managing HIV as a chronic illness: a scoping review protocol. BMJ Open. 2021;11(6):e047870. doi: 10.1136/bmjopen-2020-047870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.UNAIDS. The gap report 2014: people aged 50 years and older. 2014. https://www.unaids.org/sites/default/files/media_asset/12_Peopleaged50yearsandolder.pdf. Accessed on 23 Jun 2023.
- 3.Autenrieth CS, Beck EJ, Stelzle D, Mallouris C, Mahy M, Ghys P. Global and regional trends of people living with HIV aged 50 and over: estimates and projections for 2000–2020. PLoS ONE. 2018;13(11):e0207005. doi: 10.1371/journal.pone.0207005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Longevity The Lancet Healthy. Ageing with HIV. Lancet Healthy Longev. 2022;3(3):e119. doi: 10.1016/S2666-7568(22)00041-1. [DOI] [PubMed] [Google Scholar]
- 5.UNAIDS. The path that ends AIDS: UNAIDS Global AIDS Update 2023. 2023. https://www.unaids.org/en/resources/documents/2023/global-aids-update-2023. Accessed 6 Feb 2024.
- 6.Wing EJ. HIV and aging. Int J Infect Dis. 2016;53:61–68. doi: 10.1016/j.ijid.2016.10.004. [DOI] [PubMed] [Google Scholar]
- 7.Gandhi RT, Bedimo R, Hoy JF, Landovitz RJ, Smith DM, Eaton EF, et al. Antiretroviral drugs for treatment and prevention of HIV infection in adults: 2022 recommendations of the International Antiviral Society-USA Panel. JAMA. 2023;329(1):63–84. doi: 10.1001/jama.2022.22246. [DOI] [PubMed] [Google Scholar]
- 8.Smit M, Brinkman K, Geerlings S, Smit C, Thyagarajan K, van Sighem A, et al. Future challenges for clinical care of an ageing population infected with HIV: a modelling study. Lancet Infect Dis. 2015;15(7):810–818. doi: 10.1016/S1473-3099(15)00056-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Marty L, Diawara Y, Rachas A, Grabar S, Costagliola D, Supervie V. Projection of age of individuals living with HIV and time since ART initiation in 2030: estimates for France. J Int AIDS Soc. 2022;25(Suppl 4):e25986. doi: 10.1002/jia2.25986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.National Institute on Aging. HIV, AIDS, and older adults. 2021. https://www.nia.nih.gov/health/hiv-aids-and-older-adults. Accessed 17 Jan 2023.
- 11.Johnston RE, Heitzeg MM. Sex, age, race and intervention type in clinical studies of HIV cure: a systematic review. AIDS Res Hum Retroviruses. 2015;31(1):85–97. doi: 10.1089/aid.2014.0205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Spinelli F, Prakash M, Slater J, van der Kolk M, Bassani N, Grove R, et al. Dolutegravir-based regimens in treatment-naive and treatment-experienced aging populations: analyses of 6 phase III clinical trials. HIV Res Clin Pract. 2021;22(2):46–54. doi: 10.1080/25787489.2021.1941672. [DOI] [PubMed] [Google Scholar]
- 13.Cahill S, Valadéz R. Growing older with HIV/AIDS: new public health challenges. Am J Public Health. 2013;103(3):e7–e15. doi: 10.2105/AJPH.2012.301161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Naumann U, Moll A, Schleehauf D, Lutz T, Schmidt W, Jaeger H, et al. Similar efficacy and tolerability of raltegravir-based antiretroviral therapy in HIV-infected patients, irrespective of age group, burden of comorbidities and concomitant medication: real-life analysis of the German 'WIP' cohort. Int J STD AIDS. 2017;28(9):893–901. doi: 10.1177/0956462416679550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Funke B, Spinner CD, Wolf E, Heiken H, Christensen S, Stellbrink HJ, et al. High prevalence of comorbidities and use of concomitant medication in treated people living with HIV in Germany—results of the BESIDE study. Int J STD AIDS. 2021;32(2):152–161. doi: 10.1177/0956462420942020. [DOI] [PubMed] [Google Scholar]
- 16.Calcagno A, de Nicolò A, Pizzi C, Trunfio M, Tettoni C, Ferrara M, et al. Medication burden and clustering in people living with HIV undergoing therapeutic drug monitoring. Br J Clin Pharmacol. 2021;87(11):4432–4438. doi: 10.1111/bcp.14869. [DOI] [PubMed] [Google Scholar]
- 17.Krentz HB, Gill MJ. The impact of non-antiretroviral polypharmacy on the continuity of antiretroviral therapy (ART) among HIV patients. AIDS Patient Care STDS. 2016;30(1):11–17. doi: 10.1089/apc.2015.0199. [DOI] [PubMed] [Google Scholar]
- 18.Zheng C, Meng J, Xiao X, Xie Y, Zhao D, Wang H. Polypharmacy, medication-related burden and antiretroviral therapy adherence in people living with HIV aged 50 and above: a cross-sectional study in Hunan, China. Patient Prefer Adherence. 2022;16:41–49. doi: 10.2147/PPA.S340621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Murray MM, Lin J, Buros Stein A, Wilcox ML, Cottreau J, Postelnick M, et al. Relationship of polypharmacy to HIV RNA suppression in people aged ≥ 50 years living with HIV. HIV Med. 2021;22(8):742–749. doi: 10.1111/hiv.13122. [DOI] [PubMed] [Google Scholar]
- 20.Osiyemi O, De Wit S, Ajana F, Bisshop F, Portilla J, Routy JP, et al. Efficacy and safety of switching to dolutegravir/lamivudine versus continuing a tenofovir alafenamide-based 3- or 4-drug regimen for maintenance of virologic suppression in adults living with human immunodeficiency virus type 1: results through week 144 from the phase 3, noninferiority TANGO randomized trial. Clin Infect Dis. 2022;75(6):975–986. doi: 10.1093/cid/ciac036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Llibre JM, Brites C, Cheng C-Y, Osiyemi O, Galera C, Hocqueloux L, et al. Efficacy and safety of switching to the 2-drug regimen dolutegravir/lamivudine versus continuing a 3- or 4-drug regimen for maintaining virologic suppression in adults living with human immunodeficiency virus 1 (HIV-1): week 48 results from the phase 3, noninferiority SALSA randomized trial. Clin Infect Dis. 2023;76(4):720–729. doi: 10.1093/cid/ciac130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cahn P, Sierra Madero J, Arribas JR, Antinori A, Ortiz R, Clarke AE, et al. Three-year durable efficacy of dolutegravir plus lamivudine in antiretroviral therapy–naive adults with HIV-1 infection. AIDS. 2022;36(1):39–48. doi: 10.1097/QAD.0000000000003070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ait-Khaled M, Nascimento M-C, Pappa KA, Wang R, Wright J, Tenorio AR, et al. Switching to DTG/3TC fixed-dose combination (FDC) is non-inferior to continuing a TAF-based regimen through 48 weeks: subgroup analyses from the TANGO study. Presented at: 17th European AIDS Conference; November 6–9, 2019; Basel, Switzerland.
- 24.Taylor S, Andrade-Villanueva JF, Kaplan R, Nahass R, Hocqueloux L, Scholten S, et al. Switching to DTG/3TC is non-inferior to continuing current antiretroviral regimen at week 48: SALSA subgroup analyses. Presented at: 18th European AIDS Conference; October 27–30, 2021; London, UK.
- 25.Karabon P. Rare events or non-convergence with a binary outcome? The power of Firth regression in PROC LOGISTIC. Presented at: SAS® Global Forum 2020; June 16, 2020; Virtual.
- 26.Dovato [prescribing information]. Durham, NC: ViiV Healthcare; 2023.
- 27.Shah S, Pilkington V, Hill A. Is tenofovir disoproxil fumarate associated with weight loss? AIDS. 2021;35(Suppl 2):S189–S195. doi: 10.1097/QAD.0000000000003083. [DOI] [PubMed] [Google Scholar]
- 28.Erlandson KM, Carter CC, Melbourne K, Brown TT, Cohen C, Das M, et al. Weight change following antiretroviral therapy switch in people with viral suppression: pooled data from randomized clinical trials. Clin Infect Dis. 2021;73(8):1440–1451. doi: 10.1093/cid/ciab444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Haggins D, Mussini C, Zhang F, Kumar PN, Hocqueloux L, Espinosa N, et al. Week 48 metabolic health after switch to DTG/3TC vs CAR by baseline regimen (SALSA). Presented at: Conference on Retroviruses and Opportunistic Infections; February 12–16, 2022; Virtual.
- 30.Waas T, Schulz A, Lotz J, Rossmann H, Pfeiffer N, Beutel ME, et al. Distribution of estimated glomerular filtration rate and determinants of its age dependent loss in a German population-based study. Sci Rep. 2021;11(1):10165. doi: 10.1038/s41598-021-89442-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1. Adjusted Mean Change From Baseline to Week 48 in CD4 + Cell Count and CD4 + /CD8 + Ratio Overall and by Age: TANGO and SALSA Pooled ITT-E Population. Table showing change from baseline to Week 48 in CD4 + cell count and CD4 + /CD8 + ratio overall and by age
Additional file 2. Change from baseline in A fasting lipids, B plasma/serum renal biomarkers, and C bone biomarkers at Week 48 by age: TANGO and SALSA pooled safety population. Figure showing change from baseline to Week 48 in fasting lipids, renal biomarkers, and bone biomarkers by age
Data Availability Statement
Anonymized individual participant data and study documents can be requested for further research from www.clinicalstudydatarequest.com.