Abstract

Here, we present a covalent nanolayer system that consists of a conductive and biorepulsive base layer topped by a layer carrying biorecognition sites. The layers are built up by electropolymerization of pyrrole derivatives that either carry polyglycerol brushes (for biorepulsivity) or glycoside moieties (as biorecognition sites). The polypyrrole backbone makes the resulting nanolayer systems conductive, opening the opportunity for constructing an electrochemistry-based sensor system. The basic concept of the sensor exploits the highly selective binding of carbohydrates by certain harmful bacteria, as bacterial adhesion and infection are a major threat to human health, and thus, a sensitive and selective detection of the respective bacteria by portable devices is highly desirable. To demonstrate the selectivity, two strains of Escherichia coli were selected. The first strain carries type 1 fimbriae, terminated by a lectin called FimH, which recognizes α-d-mannopyranosides, which is a carbohydrate that is commonly found on endothelial cells. The otherE. coli strain was of a strain that lacked this particular lectin. It could be demonstrated that hybrid nanolayer systems containing a very thin carbohydrate top layer (2 nm) show the highest discrimination (factor 80) between the different strains. Using electrochemical impedance spectroscopy, it was possible to quantify in vivo the type 1-fimbriated E. coli down to an optical density of OD600 = 0.0004 with a theoretical limit of 0.00005. Surprisingly, the selectivity and sensitivity of the sensing remained the same even in the presence of a large excess of nonbinding bacteria, making the system useful for the rapid and selective detection of pathogens in complex matrices. As the presented covalent nanolayer system is modularly built, it opens the opportunity to develop a broad band of mobile sensing devices suitable for various field applications such as bedside diagnostics or monitoring for bacterial contamination, e.g., in bioreactors.
Keywords: electrochemical sensor, biorepulsive, distinction of bacteria, glycosides, polypyrrole, nanolayer polymerization
1. Introduction
The detection and quantification of pathogens such as bacteria is one of the most important tasks in health care, as bacterial infections are a major cause of illness and death all over the world. These include, inter alia, diagnostics,1,2 biotechnology,3,4 and quality control for food.5,6 Traditional culture tests for the identification of bacteria are time-consuming and require highly trained personnel, thus producing immense costs and temporal delays.7 In addition, the differentiation between very similar bacteria of one genus typically requires additional analyses, such as microscopy or antibody tests.8 A notorious species of bacteria are Escherichia coli (E. coli), which are ubiquitous and highly differentiated, with some of the strains being essential for human digestion, while other strains are highly pathogenic.9 The latter have caused several major outbreaks and food recalls in the U.S. according to the Centers for Disease Control,10 which annually cost companies multimillion-dollar lawsuits on top of the reimbursement for and the destruction of the infected food products.11 Hence, rapid, selective, and sensitive sensors that can detect and identify such pathogens in complex matrices are in high demand.12,13 Modern detection systems, such as quantitative polymerase chain reaction (qPCR) or enzyme-linked immunosorbent assays (ELISA), are highly selective and sensitive but often relatively large (not portable) and expensive in acquisition, operation, and maintenance.14,15 Driven by the need to reduce analysis time and cost while enabling portability, electrochemical sensors have entered the field of biosensing.16−18 From the variety of electrochemical methods, for instance, cyclic voltammetry (CV),19 amperometry,20 and differential pulse voltammetry (DPV),21 electrochemical impedance spectroscopy (EIS)22 has shown various advantages, including low experimental setup costs, high stability and reliability under various environmental conditions, and a high sensitivity toward surface phenomena such as the adsorption of bacteria.23,24 In these experimental setups, the sensor probes the adsorption of bacterial cells by measuring the change in electrical properties of the sensor due to the insulating properties of the cell membrane (ca. 10–7 S/m).25,26 With an increasing amount of adhered cells, the effective electrode area gets reduced, hence the electrical impedance increases due to the decelerated interfacial electron transfer kinetics.27 The advantage of this approach is the immensely decreased detection time, as the bacteria do not need to be cultivated but have to become attached to the surface. Thus, impedance-based biosensors are highly advantageous, as they can detect bacteria in a label-free manner, hence without any additional reagents apart from a conductive buffer solution. The method is inexpensive and can be easily miniaturized as only two electrodes are needed.28 Furthermore, due to the relatively low applied alternating voltage, this method is nondestructive for the sample.26 However, many methods in the literature show high sensitivities but no real selectivity as the measurements are performed predominantly in pure single-strain bacterial suspensions29 and not in complex matrixes, such as mixtures of bacteria.
To be able to identify and quantify a single target, a biosensor must bind the target compound specifically and selectively.30,31 Therefore, in the last two decades, much effort was not only put into the development of better detection methods but also into the modification of the sensor surfaces to tailor them for highest selectivities.32,33 The two most frequently used recognition systems are antibodies and DNA aptamers, respectively, as they can be custom-designed for many targets with very high selectivity.34,35 Nevertheless, these systems are not free of drawbacks as they become easily decomposed enzymatically in physiological environments, such as bacterial cultures,36 or they denature irreversibly if not stored under optimal conditions.37 In addition, many antibody- and DNA-based sensors are dependent on cell digestion/disintegration to access the respective analyte (cell proteins and complementary DNA strands) inside, adding another step during analysis.
A different approach is to leverage the mechanisms operating in cell–cell recognition. Many essential recognition events involve the glycocalyx of a cell, which consists of highly diverse glycoproteins and glycolipids comprising specific carbohydrate epitopes, which are characteristic of the cell.38 Thus, it is an interesting approach to decorate sensor surfaces with carbohydrates or glycoconjugates, respectively, in order to bind the desired pathogens in vivo without any need for processing or labeling the bacteria.39 The high complexity of the glycocalyx is often modeled by various multivalent glycoarchitectures such as glycosylated polymers,40 which are typically stable against denaturation or enzymatic degradation.41 Synthetic glycoconjugates have been investigated by several research groups42,43 and used for various biorecognition-based systems in order to detect analytes such as infectious agents, bacteria,44 or even eukaryotic cells.45,46
Regardless of the exact recognition mechanism in a sensor setup, nonspecific adsorption has to be minimized in order to uphold high levels of selectivity, as uncontrolled adhesion generates false signals and/or decreases the signal-to-noise ratio47 deteriorating the overall sensitivity of the sensor.48 Many approaches are described in the literature to prevent the nonspecific adsorption of bioentities (proteins, viruses, cells, organisms) on surfaces. Most of these biorepulsive molecular architectures are based on hydrogel films due to the entropy gain during their hydration.49−52 Polyglycerol (PG), a statistically hyperbranched, highly hydroxylated polyether has been demonstrated to be extraordinarily biorepulsive, nonimmunogenic, and very stable against oxidation, e.g., in electrochemical systems.53−57 Unfortunately, PG is inherently not conductive to electrons, a property that all previously reported biorepulsive materials share. At best, the transport of ions within the formed hydrogel is observed,58 hampering its application in electrochemical sensors. We therefore recently developed a hybrid material consisting of PG groups (Mn = 1.6 kDa) covalently grafted to polypyrrole (PPy) chains, which shows both, good electronic conductivity and excellent biorepulsivity, with a sweet spot at about 50 nm thickness.59 The material (poly(N-polyglycerylpyrrole), PPG) can be deposited by oxidative electropolymerization of the respective pyrrole derivative (PyPG, see Figure 1, top). It could be demonstrated by in situ electrochemical quartz-micro balance analysis that the polymerization process can be classified as “living”, as it may be paused by simply shutting off the current and then be continued by turning on the current again even after minutes of interruption.59 We figured that this should allow for the formation of diversified covalent nanolayer systems by simply exchanging the electroactive monomer during the polymerization process. The different layers will still be connected by the conductive PPy backbone but will be decorated with variable functional groups at each layer (Figure 1).
Figure 1.
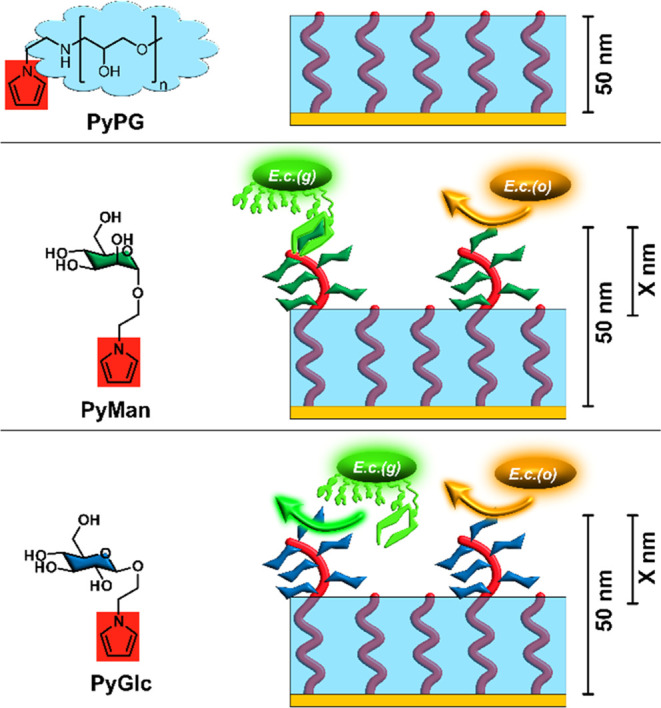
Schematic overview of the investigated nanolayer systems. Top: The basic nanolayer (PPG), which provides the biorepulsive background in the sensing setup, is formed by electropolymerization of the monomer PyPG. PPG is conductive due to the PPy backbone (red) and at the same time biorepulsive due to the PG polymer (light blue) surrounding the PPy strands. By exchange of the electroactive monomer (from PyPG to PyMan or PyGlc, respectively), it is possible to vary the functionalization of the PPy backbone with either α-d-mannopyranosyl (Man, dark green, center) or β-d-glucopyranosyl (Glc, dark blue, bottom) residues to generate selective recognition sites for bacterial adhesion. Nomenclature: with a total thickness of 50 nm, the identity and thickness (X nm) of the glycosylated part of the layer system are described as ManX and GlcX, respectively. E. coli strain, which carries type 1 fimbriae and thus selectively adheres to mannosylated surfaces (green, E.c.(g)), will bind to ManX layers, while other E. coli strains (orange, E.c.(o)) lacking mannose-specific fimbriae and lectins will not recognize any of the surfaces.
In this work, we used this idea to graft biorecognition sites onto the conductive, biorepulsive nanolayer system to allow the selective detection of a specific E. coli strain. As a model analyte, we selected a type 1-fimbriated E. coli strain, representative of the group of E. coli bacteria able to infect the urogenital tract. For this, the fimbriae are terminated by the lectin FimH, which recognizes the α-d-mannopyranoside groups exposed at the surface of endothelial cells.60 This strain should not only be detected by our sensor system but also quantified in the presence of other E. coli strains lacking the FimH lectin. The idea was to synthetically attach the mannose moiety to a pyrrole unit to attain a pyrrole mannoside, PyMan, which then can be electropolymerized to form a top layer on a previously formed PPG nanolayer. Based on the previous experiences,59 a total thickness of 50 nm was selected for the nanolayer system. For comparison, similar systems with β-d-glucosylated top layers (PyGlc), which should not be recognized by any of the used E. coli strains, were employed. The nanolayer systems are labeled in reference to the chosen glycoside and the thickness of the glycosylated layer, e.g., a layer system consisting of 10 nm of polymerized PyMan (Figure 1) on top of 40 nm PPG is named Man10. In these nanolayer systems, while nonspecific adsorption should be prevented by the PPG layer, the electrical signal should still be transmitted through the PPy backbone, thus allowing for a selective recognition of type 1-fimbriated E. coli in the presence of other E. coli strains not binding to the nanolayers.
2. Experimental Section
2.1. Chemicals
1-(2-(1-Pyrrolyl)ethylamino)-polyglycerol (PyPG) was synthesized as described in previous work (Mn = 1.6 kDa).59 The syntheses and characterization of the pyrrole glycosides PyMan and PyGlc are described in Section S2 in the Supporting Information. Ethanol (99% with 1% petroleum ether) was freshly distilled before use. Water was purified using a Millipore Milli-Q lab water system (Merck) with a conductivity of 18.2 MΩ·cm at 25 °C. A 10-fold phosphate-buffered saline concentrate (PBS (10×)) was prepared as stock solution by adding NaCl (80 g), KCl (2 g), Na2HPO4 (14.4 g), and KH2PO4 (2.4 g) to a total volume of 1 L of purified water, before the pH value was adjusted with HCl to 7.4. Before use, this solution was diluted by a factor of 10 with purified water to obtain standard PBS.
2.2. Preparation of Gold Substrates
Gold substrates were prepared by physical vapor deposition of an adhesion layer of titanium (5 nm) followed by gold (200 nm) onto polished single-crystal silicon wafers with a (100) orientation (Wacker Siltronic AG, Germany). The gold substrates were cleaved into small pieces (3 × 1 cm2), rinsed with water and ethanol (p.a.), and dried in a stream of dry nitrogen. Afterward, they were cleaned in a H2 plasma using a Harrick PDC-32G plasma cleaner (high level, pressure of 0.5 mbar, 2 min).61
2.3. Formation of the Nanolayer Systems by Sequential Electropolymerization of the Pyrrole Monomers (PyPG, PyMan, PyGlc)
The electropolymerization was carried out with a Vertex potentiostat (Ivium Technologies) in a three-electrode electrochemical cell setup (described in the Supporting Information). Data acquisition and evaluation were performed using the IviumSoft (ver. 4.952) program package. As the working electrode, the gold substrate (Si(100)/Ti/Au) was immersed 2 cm deep into the solution, while the upper 1 cm of the substrate was used for attaching the clamp. The polymerizations were performed at room temperature with a spirally coiled platinum wire (thickness: 0.5 mm, see Figure S10 in the Supporting Information) as counter electrode and an Ag/AgCl wire (stored in 3 M KCl, diameter: 0.5 mm) as reference. Parallel to the working electrode, the counter electrode was placed at a distance of 1 cm, while the Ag/AgCl wire was placed equidistantly to the other two electrodes. All polymerization solutions contained NaClO4 (0.3 M) as supporting electrolyte as well as HClO4 (0.2 M) as dopant in water. All electropolymerizations were conducted galvanostatically with a current density of 5 mA/cm2 and a concentration of 5 mM for all electroactive monomers. To obtain the different thicknesses, the reaction times were varied, as shown in Figure 2. All copolymerizations were carried out successively: The substrate was removed from the first monomer solution after the galvanostatic treatment, washed with water, dried in a stream of dry nitrogen and then immersed into the second monomer solution, and again treated galvanostatically. Afterward, the substrate was removed from the reaction solution, rinsed with water, and dried in a stream of dry nitrogen.
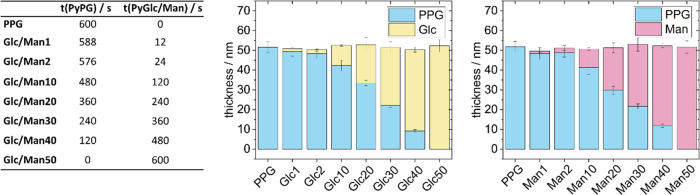
Figure 2.
Formation and composition of the nanolayer systems. Left: reaction conditions for the deposition of the nanolayer systems by sequential electropolymerization of PyPG followed by PyGlc and PyMan, respectively. The digit behind the glycoside acronym (Glc/Man) describes the thickness of the glycoside layer on top of the PPG layer. All polymer films were fabricated to be 50 nm thick. The polymerizations were conducted under constant-current conditions with a current density of 5 mA/cm2 and a concentration of 5 mM for all electroactive monomers. All reactions were conducted in water with NaClO4 (0.3 M) as the supporting electrolyte as well as HClO4 (0.2 M) as dopant. Right: Ellipsometric data for the different nanolayer systems: the thickness of the PPG layer is indicated in blue, the glycosylated ones in yellow (Glc) and red (Man).
2.4. Ellipsometry
Ellipsometric measurements of all substrates were conducted with a Sentech SE 400 ellipsometer equipped with a He/Ne laser (wavelength: 632.8 nm; beam diameter 1–2 mm) after the substrates were dried in a stream of dry nitrogen. The incidence angle was 70° with respect to the sample surface normal. The complex refractive indices of the unmodified substrates were determined after hydrogen plasma treatment for 2 min at 0.5 mbar. The extinction coefficient (k) of the refractive index and the real part (n) of the films were determined as described in previous work.59 For each sample, three substrates on which three points each were measured were averaged. The data were plotted with an Origin 2019 (OriginLab).
2.5. Measurement of the Conductivity by Electrochemical Impedance Spectroscopy (EIS)
For EIS measurements, a Vertex potentiostat (Ivium Technologies) with the same electrode setup as that for the electropolymerization was used at room temperature. The coated Au substrates were used as working electrode and all electrodes were placed 2 cm deep and 1 cm apart in a monomer-free 0.3 M NaClO4 solution. A sinusoidal potential modulation of an ±10 mV amplitude was superimposed on a fixed potential (Estart: 0.10 V) and the impedances were measured for 181 frequencies logarithmically distributed between 0.1 Hz and 100 kHz. EIS data were analyzed using the IviumSoft (Version 4.952) program package and were approximated using appropriate electronic equivalent circuits and model parameters. All data were plotted with Origin 2019 (OriginLab).
2.6. Protein Adsorption Tests
To determine the amount of protein adsorbed onto the polymer films, each substrate was incubated in PBS solution (pH 7.4) at room temperature for 2 h, rinsed with water, dried in a stream of dry nitrogen, and afterward, the thickness was measured via ellipsometry to account for swelling effects of the hydrogel (PG) from the PBS solution. Then the substrates were immersed in a solution of the respective protein (1 mg/mL) in PBS under the same conditions and with the same treatment afterward. All data were plotted with Origin 2019 (OriginLab).
2.7. Bacterial Adhesion Tests
The strain E. coli PKL1162 (SAR1862 pPKL17463) was originally provided by the laboratory of Per Klemm (Technical University of Denmark, Kongens Lyngby). The plasmid pPKL174 constitutively expresses the complete fim gene cluster required for the control and assembly of type 1 fimbriae and was transferred into E. coli SAR18, expressing the gfpmut3 gene under control of a constitutive promoter, resulting in a green fluorescence. In the following, this strain is abbreviated as E.c.(g).
E. coli S17–1 pMRE-Tn5–134 was purchased from Addgene (Teddington, UK). The plasmid pMRE-Tn5–134 was a gift from Mitja Remus-Emsermann64 (Addgene plasmid # 118520; http://n2t.net/addgene:118520; RRID:Addgene_118520) and expresses the mOrange2 protein. Because of the resulting orange fluorescence, this strain will be referred to as E.c.(o).
For the bacterial adhesion experiments, suspensions of the two different E. coli strains in CASO broth with an optical density (OD) at 600 nm of 0.5 each (beginning of the exponential growth phase) were used. All used materials and solutions were sterilized prior to use (Tuttnauer autoclave-steam sterilizer, 2540EL), and for the solutions, the pH value was set to 7.4. For the growth of E.c.(g), ampicillin (100 μg/mL) and chloramphenicol (25 μg/mL) were added to the medium after sterilization to select for the bacteria carrying the gfpmut3 gene, which also carried resistance against these antibiotics. Similarly, ampicillin (100 μg/mL) and chloramphenicol (15 μg/mL) were added to the medium after sterilization to select for the E.c.(o) bacteria carrying the mOrange2 gene.
Adsorption tests were carried out at 37.5 °C using 250 mL of bacterial suspension for 10 substrates. For each modified substrate, a clean Au substrate was used as a reference in the same solution. The substrates were placed in a poly(tetrafluoroethylene) (PTFE) carrier, completely covered with the bacterial suspension, and shaken (60 rpm) for 2 h in the dark. Afterward, the PTFE carriers were dipped three times in a PBS solution, and then the single substrates were placed in Falcon tubes (20 mL of PBS) and shaken for 5 min (100 rpm), dipped in a 20 mL beaker with PBS and put on a microscope slide. Still wet and prepared with a coverslip, they were analyzed using an Olympus BX51 microscope equipped with a metal halide lamp. For E.c.(g), a 365 nm band filter for excitation and a 450 nm edge filter for detection was used while for E.c.(o), a 525 nm band filter for excitation and a 595 nm edge filter for detection was used. For the mixed adsorption experiments, both strains were used with identical optical densities (OD600 = 0.5) at the beginning of the exponential growth phase, diluted with CASO broth, and mixed in the respective ratios before exposing the substrates in the same fashion as described before for the single adhesion experiments. For the determination of the areal number density of bacteria on the surface, for each sample three substrates with 10 randomized spots each were measured with the software “infinity analyze—DataStation” provided by Horiba Jobin Yvon (DynaMyc). An ImageJ macro was used for counting to provide a consistent routine and a large sample range (average of 30 images in total per sample). All data were plotted with Origin 2019 (OriginLab).
2.8. Bacterial Detection Tests
For the detection tests, the same two E. coli strains, PKL1162 and S17–1 pMRE-Tn5–134, were used after being cultured for 18 h at 37.5 °C in CASO broth. Afterward, the optical density was determined, and the respective dilutions were prepared with PBS. For detection by EIS, the substrates were immersed for 30 min at room temperature in the respective solution for the adhesion of the bacteria, after which the substrate was measured. For this, the same parameters were used as described in Section 2.5. All data were plotted with Origin 2019 (OriginLab).
3. Results and Discussion
3.1. Synthesis of PyMan and PyGlc and Preparation of the Covalent Nanolayer System
To build a highly selective recognition system, a covalently bound nanolayer stack should be developed, which consists of a bottom PPG layer that prevents nonspecific adsorption and a second layer that introduces specific recognition sites to bind the targeted cells. As model system, the type 1-fimbriated E. coli strain PKL1162 was selected, the fimbriae of which are terminated with the FimH lectin binding selectively to α-d-mannopyranosyl units. As competing strain E. coli S17–1 pMRE-Tn5–134 was employed, which carries no fimbriae. To distinguish between these two strains by fluorescence microscopy, the first one was modified to express the green fluorescent protein (GFP, acronym for the strain in this project: E.c.(g)), while the second one was transformed to carry the gene for expression of the mOrange2 fluorescence label (strain acronym: E.c.(o)).
For the preparation of the mannosylated layer, the α-d-mannopyranoside PyMan, functionalized with a pyrrole aglycon, was used (cf. Figure 1). To further distinguish between specific and nonspecific binding events at the nanolayer system, the analogous pyrrole β-d-glucopyranoside PyGlc was employed. Synthetic details for both monomers, PyMan and PyGlc, can be found in the Supporting Information. In contrast to α-d-mannoside, β-d-glucoside should not be recognized by either of the two tested bacterial strains. Both pyrrole glycosides could be electropolymerized under the very same conditions as the PyPG derivative (galvanostatic conditions with j = 5 mA/cm2, monomer concentration 5 mM). Somewhat surprisingly, the very same layer growth kinetics was found for all three pyrrole derivates, although their molecular weight significantly differs. This observation facilitated the synthesis of the nanolayer systems with different thicknesses, as only the electropolymerization times had to be controlled regardless of the substitution at the pyrrole rings. As the system must meet three conditions for a sensing application (electrical conductivity, prevention of nonspecific adsorption, and specific adsorption of the target bacteria), we investigated the influence of the glycoside layer thicknesses. For this, we manufactured different kinds of glycoside/PPG covalent nanolayer systems including the ones consisting of the pure glycoside polymers (Glc50, Man50) and the nonfunctionalized PPG films for comparison. The respective ellipsometric data are presented in Figure 2, showing the predictable and reproducible formation of all nanolayer systems. We found that the morphology of the layers is basically independent of the exact composition with a root-mean-square (RMS) roughness of about 2 nm. It thus resembles the structures of the pure PPG layers as discussed in ref (59).
3.2. Investigation of the Electric Conductivity as a Function of Composition
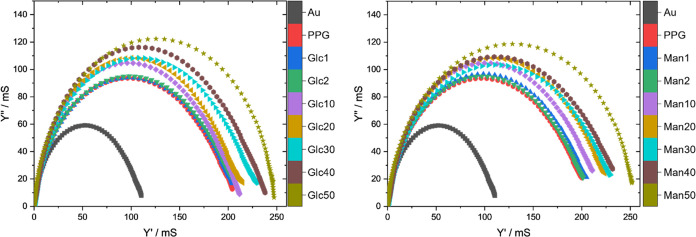
To investigate the influence of the effect of the glycoside moieties on the conductivity of the overall system, electrochemical impedance measurements were conducted in monomer-free, aqueous NaClO4 solution (0.3 M) to avoid the contribution of charge transfer onto external species and to prevent further film growth. As the admittance plot depicted in Figure 3 suggests, the conductivity increases with increasing relative and absolute thickness of the glycoside layers. The quantitative evaluation by using a suitable equivalent circuit is described in Section S4 in the Supporting Information. The numerical data (Tables S1 and S2) verify the increase of the conductivity with higher PyMan/PyGlc content, which might be explained by the decreased structural deformation of the PPy backbone, which should be the highest for the hyperbranched and sterically demanding PyPG (average molecular mass of 1600 Da59) and the lowest for the rather small glycoside moieties. Therefore, with respect to the sensing capabilities, a layer system purely composed of carbohydrate monomers would promise the highest sensitivity. As prevention of nonspecific adsorption is another important requirement for a functioning system in addition to its sensing capabilities, the biorepulsivity of the nanolayers had to be evaluated next.
Figure 3.
Admittance plot (inverse Nyquist plot) of all nanolayers (left: GlcX, right: ManX) and Au as a reference in 0.3 M NaClO4 solution. All measurements were conducted at Estart: 0.1 V (vs Ag/AgCl) with an amplitude of 0.01 V, and the impedances were measured for 181 frequencies logarithmically distributed between 0.1 and 100 kHz.
3.3. Investigation of the Nonspecific and Specific Adsorption
As both kinds of moieties, PG as well as glycosides, provide a high density of hydroxyl groups, the formation of biorepulsive hydrogels could be expected in either case.65 Surface IR spectroscopy (spectra are shown in Figure S12) of the mixed layer systems revealed no distinctive changes compared to the pure PPG system: in all spectra, broad bands (due to the polymeric nature of the films) and strong signals of hydroxyl groups were found. Such highly hydroxylated surfaces should show excellent wettability, for which we analyzed the water contact angles (WCA) of all of the nanolayer systems. Starting from a WCA of ca. 33° (neat PPG) going to about 45° for neat glycoside films (Man50, Glc50) a continuous increase with increasing glycoside layer thickness can be observed (see Figure S13). The latter values are not unexpected as glycoside-terminated surfaces typically show WCAs in the range of 40–50°. As WCA is usually a good indicator for the biorepulsive properties of a surface, we next investigated the influence of the glycoside top layer on the nonspecific adsorption. For this, we employed a standard procedure with two proteins of different molecular weights: bovine serum albumin (BSA, 66 kDa, pI 4.8) and γ-globulin (120 kDa, pI 5.8).66 For the tests, the substrates were immersed in solutions of the different proteins for 2 h, and the resulting increase in layer thickness was determined by ellipsometry. As references, bare Au surfaces and the PPG layers were employed (see Figure S14). While each protein formed a thick adlayer on the bare gold surface, protein adsorption was neglectable at the PPG film, as reported in the literature.59 For all hybrid nanolayers, a very reduced adsorption could be observed for both proteins, with a trend toward lower protein adlayers for lower glycoside film thicknesses, indicating that the glycoside films were less biorepulsive against protein adsorption than pure PPG layers, as already suggested by the WCA data.
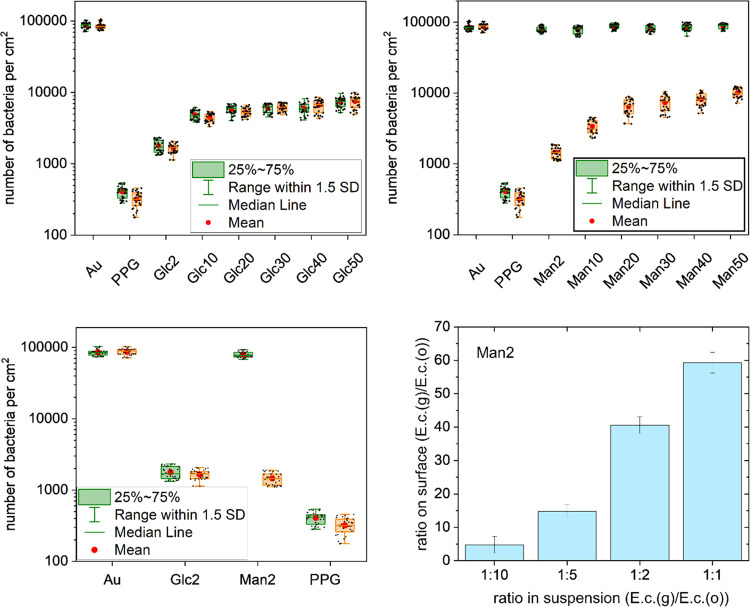
While the prevention of protein adsorption is rather straightforward, the prevention of bacterial adhesion is much more complex and challenging. Hence, we examined the adsorption of bacteria, for which we used E.c.(g) and E.c.(o) (see Section 3.1) as model bacteria. The area number density of the adhered bacteria was determined by fluorescence microscopy, with representative images shown in Figure S15 in the Supporting Information. As shown in Figure 4, top left, the GlcX nanolayers do not discriminate between the different E. coli strains, but there is a general tendency for increased nonspecific adsorption with increasing thickness of the glycoside layer part. At the GlcX layer with X = 10 to 50, almost 20 times as many bacteria (of any kind) adhere as at the PPG layer, with the situation being somewhat better for the Glc2 nanolayer (factor 8). For the Man system (see Figure 4, top, right), the adhesion of the E.c.(g) and E.c.(o) strains is strongly discriminated (factor 70). While the amount of E.c.(g) bacteria per cm2 is basically independent of the thickness of the Man top layer, for E.c.(o) a very similar trend is observed as in the GlcX case, hinting again at nonspecific adhesion to the glycoside layer, although this nonspecific adhesion is at least 1 order of magnitude lower than on bare gold surfaces. The biggest difference in adhesion was achieved in the Man2 nanolayer system, in which a 48 nm thick, biorepulsive layer of PPG is covered by a mannosylated layer of only 2 nm thickness. To demonstrate that indeed discrimination between the bacterial strains can take place, competition-based experiments were performed in which the bacterial strains were mixed in the same suspension at predetermined number ratios. Their distinction at the surface is made possible by their autofluorescence at different wavelengths. As can be seen in Figure 4, bottom right, basically the same numerical discrimination (about 80) can be seen for the exposure to the mixtures as for the exposure to suspensions of the pure strains (Figure 4, bottom left). A similar behavior was found for the Man10 nanolayer system, where the discrimination is lower in both cases (factors of about 20, see Figure S16). These data indicate that the differentiation in bacterial adhesion might be optimized by lowering the thickness of the glycoside layer even further; however, we refrained from these experiments as the attained error bars for the ellipsometric data of the Man1 systems were on the same order of magnitude as the glycoside layer thickness (see Figure 2), preventing reliable readouts.
Figure 4.
Top row: Number of adhered E. coli bacteria (green: E. coli PKL1162 = E.c.(g), orange: E. coli S17–1 pMRE-Tn5–134 = E.c.(o)) on the different functionalized surfaces, with bare Au as reference. SD = standard deviation. All data sets were tested positively for normal distribution (α = 0.05) with the Kolmogorov–Smirnov and the Shapiro–Wilk test, and a significance level of p = 0.001 was confirmed for all data in reference to the Au data set. Bottom left: Highlighted comparison of the biorepulsive performance (pure strains) of Glc2 and Man2 compared to that of Au and PPG. Bottom right: Results of competitive adhesion on a Man2 surface using mixtures of the E.c.(g) and E.c.(o) strains.
3.4. Application of the Nanolayers in a Selective Sensor Setup
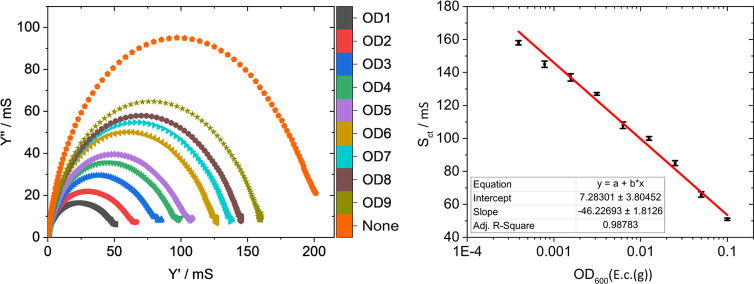
The adsorption of bacteria onto the presented conductive hydrogel nanolayers should influence their electrochemical properties and thus give the foundation for the development of a sensing system. In order to quantify these effects, Man10 layers were exposed to suspensions of E.c.(g) at different ODs. With decreasing bacterial densities, the number of adhering bacteria to the surfaces declines, leading to a higher charge transfer rate at the interface (see Section S11 in the Supporting Information). Quantitative analysis of the EIS data shows a linear correlation when the charge transfer conductivity Sct was plotted against the logarithm of the applied OD within the whole experimental range (OD600 = 3.9 × 10–4 to 0.1, see Figure S16 in the Supporting Information). Extrapolation of the graph to the Sct of the native Man10 nanolayer (Sct = 210 mS) suggests a detection limit of roughly 5 × 10–5 OD600, which corresponds to about 104 bacteria per cm3.67
Motivated by these promising results and the capacity for selection found in particular for the nanolayer systems with the thinner glycoside layers, Man2 surfaces were exposed to mixtures of bacteria and characterized by EIS (see Figure 5). The mixed suspensions contained variable densities (OD600 = 3.9 × 10–4 to 0.1) of the type 1-fimbriated strain E.c.(g), while the density of the E.c.(o) strain was kept constant at an OD600 = 0.01. Again, a linear relationship (R2 = 0.988) for Sct against the logarithm of the OD of E.c.(g) was found, with the data being almost the same as those for the pure E.c.(g) suspensions (see Section S12 in the Supporting Information). This supports the previous observation that almost no E.c.(o) bacteria adhere to the electrode surface. As before, extrapolation of the linear trend toward the Sct of the native Man2 nanolayer results in a detection limit of 5 × 10–5 OD600, so the influence of the E.c.(o) bacteria is basically negligible even at large excess in the suspension. The demonstrated biorepulsive characteristics of the sensing system in combination with the selective recognition of a targeted bacterial strain within a simplified substrate mixture containing a secondary noninteracting strain, imply that a functional framework for field application with samples containing pathogenic bacteria is provided.
Figure 5.
Left: Admittance plot of Man2 in suspensions of E.c.(g) (E. coli PKL1162) in PBS/CASO broth of different optical densities at 600 nm (OD1–9: OD1 = 0.1, OD2 = 0.05, OD3 = 0.025, OD4 = 0.0125, OD5 = 0.00625, OD6 = 0.00313, OD7 = 0.00156, OD8 = 0.00081, and OD9 = 0.00039). E.c.(o) was present at a constant density of OD = 0.01 to demonstrate selectivity during the sensing process. All measurements were reproducibly conducted under the same conditions as the ones described in Figure 3. The data of Man2 without any E.c.(g) ("None") are presented as reference. Right: Logarithmic plot of the OD of the E.c.(g) bacteria against the measured charge transfer conductance (Sct). The error bars indicate the standard deviation.
4. Conclusions
In this work, it could be shown that the use of polypyrrole backbones (PPy) does not only permit the construction of electrically conductive nanolayer systems but that a suitable functionalization permits to attain surfaces that can very selectively bind bioentities, which then can be detected by electrochemical means. As a model system, mixtures of E. coli strains were used, as these bacteria are not only ubiquitous but can be either beneficial or pathogenic due to their genetic variability. A simple differentiation by a sensing system thus was considered highly desirable. In contrast to many other sensing systems, we used the recognition capabilities of the infectious strain itself by designing the nanolayer system to resemble parts of the glycocalyx of the natural target cells. The respective glycosylation pattern was attained by forming the terminal, exposed layer using a respective pyrrole α-d-mannopyranoside derivative as the building block. Comparative adhesion experiments with the selected type 1-fimbriated E. coli strain (E.c.(g)) and another, nonbinding one (E.c.(o)) showed that nanolayers consisting only of the mannosylated PPy do not effectively suppress the nonspecific adhesion of the latter strain so that an underlying, biorepulsive PG layer had to be introduced. The selective adhesion properties were validated using a second system, carrying the respective β-d-glucopyranoside instead of α-d-mannopyranoside groups in which no selective binding event for either strain can occur. Apparently, it is highly advantageous for increased selectivity to keep the recognition layer as thin as possible on top of a thicker, purely biorepulsive nanolayer. The control of the relative thicknesses of the layers is easily possible as the layer thickness is only dependent on the time interval for which the current is applied.
Electrochemical characterization of the nanolayer systems in the presence of the bacterial strains shows a very good linearity of the charge transfer conductivity Sct as a function of binding E.c.(g) bacteria even at very low bacterial densities (OD600 4 × 10–4, possibly even 5 × 10–5). With the Man2 system, which showed the highest discrimination between the bacterial strains, the E.c.(g) serotype could be detected and quantified even in the presence of an excess of the E.c.(o) strain. This selectivity can be explained only partly by the discriminative adhesion at the nanolayer surface. We suggest that an additional effect results from the binding sites of the respective bacteria: while the selective binding of the E.c.(g) strain occurs at the end of the conductive PPy “wires”, the nonselective adsorption may take place anywhere at the polymeric system. Thus, the influence of the specifically recognized bacteria on charge transfer conductivity Sct should be much more pronounced, additionally enhancing the sensing effect.
As EIS-based sensors can be miniaturized easily,68 we envision that the present system is ideally suited for the development of mobile, rapid, electrochemical sensing devices that can be used in various field applications, e.g., to monitor food contamination. As the cost of one sensor chip with this system is fairly low, we expect the device to be suitable for single-use applications. Due to the modularity of the system, it can be easily adapted for the selective binding of other bioentities, such as other bacteria but also proteins, viruses, and eukaryotic cells, opening the opportunity to generate highly selective, electrochemical sensors at very low cost. Currently, utmost priority is laid on procuring data using infected samples from real-life substrates to advance the project toward medical application. The shared mechanistic principles between carbohydrate-lectin interactions make the recognition of pathogenic bacteria uniquely suited for the presented sensing system. This offers opportunities to apply the method to targets of special interest with regard to public health. Examples include but are not limited to enterohemorrhagic E. coli (EHEC), or bacteria of the species Salmonella or Mycobacterium tuberculosis.
Acknowledgments
The authors acknowledge the donation of the bacterial strains/plasmids by Per Klemm (Technical University of Denmark, Kongens Lyngby, Denmark) and Mitja Remus-Emsermann (FU Berlin, Berlin, Germany).
Data Availability Statement
The raw/processed data required to reproduce these findings will be available from the corresponding authors upon request.
Supporting Information Available
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsami.3c14387.
Syntheses, additional experimental details, fluorescence counting data, materials, and methods (PDF)
Author Contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.
Financial support by the “Konrad-Adenauer-Stiftung” (S.B.) in the form of a PhD grant is gratefully acknowledged.
The authors declare no competing financial interest.
Supplementary Material
References
- Riglar D. T.; Silver P. A. Engineering Bacteria for Diagnostic and Therapeutic Applications. Nat. Rev. Microbiol. 2018, 16, 214–225. 10.1038/nrmicro.2017.172. [DOI] [PubMed] [Google Scholar]
- Yoo S. M.; Lee S. Y. Optical Biosensors for the Detection of Pathogenic Microorganisms. Trends Biotechnol. 2016, 34, 7–25. 10.1016/j.tibtech.2015.09.012. [DOI] [PubMed] [Google Scholar]
- Yagi K. Applications of Whole-Cell Bacterial Sensors in Biotechnology and Environmental Science. Appl. Microbiol. Biotechnol. 2007, 73, 1251–1258. 10.1007/s00253-006-0718-6. [DOI] [PubMed] [Google Scholar]
- Molina F.; López-Acedo E.; Tabla R.; Roa I.; Gómez A.; Rebollo J. E. Improved Detection of Escherichia Coli and Coliform Bacteria by Multiplex PCR. BMC Biotechnol. 2015, 15, 48. 10.1186/s12896-015-0168-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Panwar S.; Duggirala K. S.; Yadav P.; Debnath N.; Yadav A. K.; Kumar A. Advanced Diagnostic Methods for Identification of Bacterial Foodborne Pathogens: Contemporary and Upcoming Challenges. Crit. Rev. Biotechnol. 2022, 18, 1–19. 10.1080/07388551.2022.2095253. [DOI] [PubMed] [Google Scholar]
- Nnachi R. C.; Sui N.; Ke B.; Luo Z.; Bhalla N.; He D.; Yang Z. Recent Progress on Biosensors for Rapid Detection of Bacterial Pathogens in Water, Food and Environment. Environ. Int. 2022, 166, 107357 10.1016/j.envint.2022.107357. [DOI] [PubMed] [Google Scholar]
- Rajapaksha P.; Elbourne A.; Gangadoo S.; Brown R.; Cozzolino D.; Chapman J. A Review of Methods for the Detection of Pathogenic Microorganisms. Analyst 2019, 144, 396–411. 10.1039/C8AN01488D. [DOI] [PubMed] [Google Scholar]
- Rompré A.; Servais P.; Baudart J.; De-Roubin M. R.; Laurent P. Detection and Enumeration of Coliforms in Drinking Water: Current Methods and Emerging Approaches. J. Microbiol. Methods 2002, 49, 31–54. 10.1016/S0167-7012(01)00351-7. [DOI] [PubMed] [Google Scholar]
- Kaper J. B.; Nataro J. P.; Mobley H. L. Pathogenic Escherichia Coli. Nat. Rev. Microbiol. 2004, 2, 123–140. 10.1038/nrmicro818. [DOI] [PubMed] [Google Scholar]
- Food Safety and Foodborne Illness. World Health Organization Fact Sheet No. 237, 2007.
- Hoffmann S.; Scallan-Walter E. (2020). Acute Complications and Sequelae from Foodborne Infections: Informing Priorities for Cost of Foodborne Illness Estimates. Foodborne Pathog. Dis. 2020, 17, 172–177. 10.1089/fpd.2019.2664. [DOI] [PubMed] [Google Scholar]
- Chakraborty P.; Krishnani K. K. Emerging Bioanalytical Sensors for Rapid and Close-to-Real-Time Detection of Priority Abiotic and Biotic Stressors in Aquaculture and Culture-Based Fisheries. Sci. Total Environ. 2022, 838, 156128 10.1016/j.scitotenv.2022.156128. [DOI] [PubMed] [Google Scholar]
- Andrei C. C.; Moraillon A.; Lau S.; Felidj N.; Yamakawa N.; Bouckaert J.; Larquet E.; Boukherroub R.; Ozanam F.; Szunerits S.; Gouget-Laemmel A. C. Rapid and Sensitive Identification of Uropathogenic Escherichia Coli Using a Surface-Enhanced-Raman-Scattering-Based Biochip. Talanta 2020, 219, 121174 10.1016/j.talanta.2020.121174. [DOI] [PubMed] [Google Scholar]
- Castle L. M.; Schuh D. A.; Reynolds E. E.; Furst A. L. Electrochemical Sensors to Detect Bacterial Foodborne Pathogens. ACS Sens. 2021, 6, 1717–1730. 10.1021/acssensors.1c00481. [DOI] [PubMed] [Google Scholar]
- Baranwal J.; Barse B.; Gatto G.; Broncova G.; Kumar A. Electrochemical Sensors and their Applications: A Review. Chemosensors 2022, 10, 363. 10.3390/chemosensors10090363. [DOI] [Google Scholar]
- Kundu M.; Krishnan P.; Kotnala R. K.; Sumana G. Recent Developments in Biosensors to Combat Agricultural Challenges and their Future Prospects. Trends Food Sci. Technol. 2019, 88, 157–178. 10.1016/j.tifs.2019.03.024. [DOI] [Google Scholar]
- Chadha U.; Bhardwaj P.; Agarwal R.; Rawat P.; Agarwal R.; Gupta I.; Panjwani M.; Singh S.; Ahuja C.; Selvaraj S. K.; Banavoth M.; Sonar P.; Badoni B.; Chakravorty A. Recent Progress and Growth in Biosensors Technology: A Critical Review. J. Ind. Eng. Chem. 2022, 109, 21–51. 10.1016/j.jiec.2022.02.010. [DOI] [Google Scholar]
- Kaya H. O.; Cetin A. E.; Azimzadeh M.; Topkaya S. N. Pathogen Detection with Electrochemical Biosensors: Advantages, Challenges and Future Perspectives. J. Electroanal. Chem. 2021, 882, 114989 10.1016/j.jelechem.2021.114989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alatraktchi F. A. A.; Breum Andersen S.; Krogh Johansen H.; Molin S.; Svendsen W. E. Fast Selective Detection of Pyocyanin Using Cyclic Voltammetry. Sensors 2016, 16, 408. 10.3390/s16030408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gau J. J.; Lan E. H.; Dunn B.; Ho C. M.; Woo J. C. A MEMS Based Amperometric Detector for E. Coli Bacteria Using Self-Assembled Monolayers. Biosens. Bioelectron. 2001, 16, 745–755. 10.1016/S0956-5663(01)00216-0. [DOI] [PubMed] [Google Scholar]
- Xu J.; Zhao C.; Chau Y.; Lee Y. K. The Synergy of Chemical Immobilization and Electrical Orientation of T4 Bacteriophage on a Micro Electrochemical Sensor for Low-Level Viable Bacteria Detection via Differential Pulse Voltammetry. Biosens. Bioelectron. 2020, 151, 111914 10.1016/j.bios.2019.111914. [DOI] [PubMed] [Google Scholar]
- Yang L.; Bashir R. Electrical/Electrochemical Impedance for Rapid Detection of Foodborne Pathogenic Bacteria. Biotechnol. Adv. 2008, 26, 135–150. 10.1016/j.biotechadv.2007.10.003. [DOI] [PubMed] [Google Scholar]
- Magar H. S.; Hassan R. Y.; Mulchandani A. Electrochemical Impedance Spectroscopy (EIS): Principles, Construction, and Biosensing Applications. Sensors 2021, 21, 6578. 10.3390/s21196578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brett C. M. A. Electrochemical Impedance Spectroscopy in the Characterisation and Application of Modified Electrodes for Electrochemical Sensors and Biosensors. Molecules 2022, 27, 1497. 10.3390/molecules27051497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furst A. L.; Francis M. B. Impedance-Based Detection of Bacteria. Chem. Rev. 2019, 119, 700–726. 10.1021/acs.chemrev.8b00381. [DOI] [PubMed] [Google Scholar]
- Ramírez N.; Regueiro A.; Arias O.; Contreras R. Electrochemical Impedance Spectroscopy: An Effective Tool for a Fast Microbiological Diagnosis. Biotecnol. Apl. 2009, 1, 72–78. [Google Scholar]
- Dheilly A.; Linossier I.; Darchen A.; Hadjiev D.; Corbel C.; Alonso V. Monitoring of Microbial Adhesion and Biofilm Growth Using Electrochemical Impedancemetry. Appl. Microbiol. Biotechnol. 2008, 79, 157–164. 10.1007/s00253-008-1404-7. [DOI] [PubMed] [Google Scholar]
- Lisdat F.; Schafer D. The Use of Electrochemical Impedance Spectroscopy for Biosensing. Anal. Bioanal. Chem. 2008, 391, 1555–1567. 10.1007/s00216-008-1970-7. [DOI] [PubMed] [Google Scholar]
- Amiri M.; Bezaatpour A.; Jafari H.; Boukherroub R.; Szunerits S. Electrochemical Methodologies for the Detection of Pathogens. ACS Sens. 2018, 3, 1069–1086. 10.1021/acssensors.8b00239. [DOI] [PubMed] [Google Scholar]
- Rogers K. R. Principles of Affinity-Based Biosensors. Mol. Biotechnol. 2000, 14, 109–129. 10.1385/mb:14:2:109. [DOI] [PubMed] [Google Scholar]
- Wang Y.; Ye Z.; Ying Y. New Trends in Impedimetric Biosensors for the Detection of Foodborne Pathogenic Bacteria. Sensors 2012, 12, 3449–3471. 10.3390/s120303449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mirmoghtadaie L.; Ensafi A. A.; Kadivar M.; Norouzi P. Highly Selective Electrochemical Biosensor for the Determination of Folic Acid Based on DNA Modified-Pencil Graphite Electrode Using Response Surface Methodology. Mater. Sci. Eng., C 2013, 33, 1753–1758. 10.1016/j.msec.2012.12.090. [DOI] [PubMed] [Google Scholar]
- Yüce M.; Kurt H. How to Make Nanobiosensors: Surface Modification and Characterization of Nanomaterials for Biosensing Applications. RSC Adv. 2017, 78, 49386–49403. 10.1039/c7ra10479k. [DOI] [Google Scholar]
- Skottrup P. D.; Nicolaisen M.; Justesen A. F. Towards On-Site Pathogen Detection Using Antibody-Based Sensors. Biosens. Bioelectron. 2008, 24, 339–348. 10.1016/j.bios.2008.06.045. [DOI] [PubMed] [Google Scholar]
- Byrne B.; Stack E.; Gilmartin N.; O’Kennedy R. Antibody-Based Sensors: Principles, Problems and Potential for Detection of Pathogens and Associated Toxins. Sensors 2009, 9, 4407–4445. 10.3390/s90604407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindahl T. Instability and Decay of the Primary Structure of DNA. Nature 1993, 362, 709–715. 10.1038/362709a0. [DOI] [PubMed] [Google Scholar]
- Wang W.; Singh S.; Zeng D. L.; King K.; Nema S. Antibody Structure, Instability, and Formulation. J. Pharm. Sci. 2007, 96, 1–26. 10.1002/jps.20727. [DOI] [PubMed] [Google Scholar]
- Möckl L. The Emerging Role of the Mammalian Glycocalyx in Functional Membrane Organization and Immune System Regulation. Front. Cell Develop. Biol. 2020, 8, 253. 10.3389/fcell.2020.00253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jelinek R.; Kolusheva S. Carbohydrate Biosensors. Chem. Rev. 2004, 104, 5987–6016. 10.1021/cr0300284. [DOI] [PubMed] [Google Scholar]
- Wang Q.; Dordick J. S.; Linhardt R. J. Synthesis and Application of Carbohydrate-Containing Polymers. Chem. Mater. 2002, 14, 3232–3244. 10.1021/cm0200137. [DOI] [Google Scholar]
- Zeng X.; Andrade C. A.; Oliveira M. D.; Sun X. L. Carbohydrate–Protein Interactions and their Biosensing Applications. Anal. Bioanal. Chem. 2012, 402, 3161–3176. 10.1007/s00216-011-5594-y. [DOI] [PubMed] [Google Scholar]
- Müller C.; Despras G.; Lindhorst T. K. Organizing Multivalency in Carbohydrate Recognition. Chem. Soc. Rev. 2016, 45, 3275–3302. 10.1039/C6CS00165C. [DOI] [PubMed] [Google Scholar]
- Hartmann M.; Lindhorst T. K. The Bacterial Lectin FimH, a Target for Drug Discovery–Carbohydrate Inhibitors of Type 1 Fimbriae-Mediated Bacterial Adhesion. Eur. J. Org. Chem. 2011, 2011, 3583–3609. 10.1002/ejoc.201100407. [DOI] [Google Scholar]
- Weber T.; Chandrasekaran V.; Stamer I.; Thygesen M. B.; Terfort A.; Lindhorst T. K. Switching of Bacterial Adhesion to a Glycosylated Surface by Reversible Reorientation of the Carbohydrate Ligand. Angew. Chem., Int. Ed. 2014, 53, 14583–14586. 10.1002/anie.201409808. [DOI] [PubMed] [Google Scholar]
- Templier V.; Roux A.; Roupioz Y.; Livache T. Ligands for Label-Free Detection of Whole Bacteria on Biosensors: A Review. TrAC Trends Analyt. Chem. 2016, 79, 71–79. 10.1016/j.trac.2015.10.015. [DOI] [Google Scholar]
- García I.; Mosquera J.; Plou J.; Liz-Marzán L. M. Plasmonic Detection of Carbohydrate-Mediated Biological Events. Adv. Opt. Mater. 2018, 23, 1800680 10.1002/adom.201800680. [DOI] [Google Scholar]
- Chen A.; Kozak D.; Battersby B. J.; Forrest R. M.; Scholler N.; Urban N.; Trau M. Antifouling Surface Layers for Improved Signal-to-Noise of Particle-Based Immunoassays. Langmuir 2009, 25, 13510–13515. 10.1021/la903148n. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kozak D.; Surawski P.; Thoren K. M.; Lu C. Y.; Marcon L.; Trau M. Improving the Signal-to-Noise Performance of Molecular Diagnostics with PEG-Lysine Copolymer Dendrons. Biomacromolecules 2009, 10, 360–365. 10.1021/bm8011314. [DOI] [PubMed] [Google Scholar]
- Balser S.; Zhao Z.; Zharnikov M.; Terfort A. Effect of the Crosslinking Agent on the Biorepulsive and Mechanical Properties of Polyglycerol Membranes. Colloids Surf., B 2023, 225, 113271 10.1016/j.colsurfb.2023.113271. [DOI] [PubMed] [Google Scholar]
- Murosaki T.; Ahmed N.; Gong J. P. Antifouling Properties of Hydrogels. Sci. Technol. Adv. Mater. 2012, 6, 064706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su D.; Bai X.; He X. Research Progress on Hydrogel Materials and their Antifouling Properties. Eur. Polym. J. 2022, 181, 111665 10.1016/j.eurpolymj.2022.111665. [DOI] [Google Scholar]
- Fu M.; Liang Y.; Lv X.; Li C.; Yang Y. Y.; Yuan P.; Ding X. Recent Advances in Hydrogel-Based Anti-Infective Coatings. J. Mater. Sci. Technol. 2021, 85, 169–183. 10.1016/j.jmst.2020.12.070. [DOI] [Google Scholar]
- Frey H.; Haag R. Dendritic Polyglycerol: A Versatile Biocompatible Material. Mol. Biotechnol. 2002, 90, 257–267. 10.1016/S1389-0352(01)00063-0. [DOI] [PubMed] [Google Scholar]
- Lange M.; Braune S.; Luetzow K.; Richau K.; Scharnagl N.; Weinhart M.; Neffe A. T.; Jung F.; Haag R.; Lendlein A. Surface Functionalization of Poly(ether imide) Membranes with Linear, Methylated Oligoglycerols for Reducing Thrombogenicity. Macromol. Rapid Commun. 2012, 33, 1487–1492. 10.1002/marc.201200426. [DOI] [PubMed] [Google Scholar]
- Ngo B. K. D.; Grunlan M. A. Protein Resistant Polymeric Biomaterials. ACS Macro Lett. 2017, 6, 992–1000. 10.1021/acsmacrolett.7b00448. [DOI] [PubMed] [Google Scholar]
- Wanka R.; Aldred N.; Finlay J. A.; Amuthalingam A.; Clarke J. L.; Clare A. S.; Rosenhahn A. Antifouling Properties of Dendritic Polyglycerols Against Marine Macrofouling Organisms. Langmuir 2019, 35, 16568–16575. 10.1021/acs.langmuir.9b02720. [DOI] [PubMed] [Google Scholar]
- Dey P.; Adamovski M.; Friebe S.; Badalyan A.; Mutihac R. C.; Paulus F.; Leimkühler S.; Wollenberger U.; Haag R. Dendritic Polyglycerol–Poly (Ethylene Glycol)-Based Polymer Networks for Biosensing Application. ACS Appl. Mater. Interfaces 2014, 6, 8937–8941. 10.1021/am502018x. [DOI] [PubMed] [Google Scholar]
- Ishikawa K.; Sugihara T.; Oshima Y.; Kato T.; Imai A. Li Ion Conductive Polymer Electrolyte Based on Polyglycerine. Solid State Ionics 1990, 40–41, 612–615. 10.1016/0167-2738(90)90080-B. [DOI] [Google Scholar]
- Balser S.; Bernd J.; Fritsche L.; Terfort A. Preparation and Characterization of Highly Conductive and Biorepulsive Polypyrrole/Polyglycerol Surface Films. ACS Appl. Polym. Mater. 2022, 4, 8344–8356. 10.1021/acsapm.2c01302. [DOI] [Google Scholar]
- Poole J.; Day C. J.; v Itzstein M.; Paton J. C.; Jennings M. P. Glycointeractions in bacterial pathogenesis. Nat. Rev. Microbiol. 2018, 16, 440–452. 10.1038/s41579-018-0007-2. [DOI] [PubMed] [Google Scholar]
- Raiber K.; Terfort A.; Benndorf C.; Krings N.; Strehblow H.-H. Removal of Self-Assembled Monolayers of Alkanethiolates on Gold by Plasma Cleaning. Surf. Sci. 2005, 595, 56–63. 10.1016/j.susc.2005.07.038. [DOI] [Google Scholar]
- Reisner A.; Haagensen J. A. J.; Schembri M. A.; Zechner E. L.; Molin S. Development and Maturation of Escherichia Coli K-12 Biofilms. Mol. Microbiol. 2003, 48, 933–946. 10.1046/j.1365-2958.2003.03490.x. [DOI] [PubMed] [Google Scholar]
- Hartmann M.; Horst A. K.; Klemm P.; Lindhorst T. K. A Kit for the Investigation of Live Escherichia Coli Cell Adhesion to Glycosylated Surfaces. Chem. Commun. 2010, 46, 330–332. 10.1039/B922525K. [DOI] [PubMed] [Google Scholar]
- Schlechter R. O.; Jun H.; Bernach M.; Oso S.; Boyd E.; Muñoz-Lintz D. A.; Dobson R. C. J.; Remus D. M.; Remus-Emsermann M. N. P. Chromatic Bacteria - A Broad Host-Range Plasmid and Chromosomal Insertion Toolbox for Fluorescent Protein Expression in Bacteria. Front Microbiol. 2018, 9, 3052. 10.3389/fmicb.2018.03052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lopez A. I.; Kumar A.; Planas M. R.; Li Y.; Nguyen T. V.; Cai C. Biofunctionalization of Silicone Polymers Using Poly (Amidoamine) Dendrimers and a Mannose Derivative for Prolonged Interference Against Pathogen Colonization. Biomaterials 2011, 32, 4336–4346. 10.1016/j.biomaterials.2011.02.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber T.; Bechthold M.; Winkler T.; Dauselt J.; Terfort A. Direct Grafting of Anti-Fouling Polyglycerol Layers to Steel and Other Technically Relevant Materials. Colloids Surf., B 2013, 111, 360–366. 10.1016/j.colsurfb.2013.05.026. [DOI] [PubMed] [Google Scholar]
- Beal J.; Farny N. G.; Haddock-Angelli T.; Selvarajah V.; Baldwin G. S.; Buckley-Taylor R.; Gershater M.; Kiga D.; Marken J.; Sanchania V.; Sison A.; Workman C. T.; et al. Robust Estimation of Bacterial Cell Count From Optical Density. Commun. Biol. 2020, 3, 512. 10.1038/s42003-020-01127-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang W.; Wang R.; Luo F.; Wang P.; Lin Z. Miniaturized Electrochemical Sensors and Their Point-Of-Care Applications. Chin. Chem. Lett. 2020, 31, 589–600. 10.1016/j.cclet.2019.09.022. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The raw/processed data required to reproduce these findings will be available from the corresponding authors upon request.