Abstract
Purpose
The objective of this systematic review and meta-analysis was to evaluate the survival rate of implants installed in bone type IV (Lekholm and Zarb, 1995) compared to that of implants installed in bone types I, II, and III.
Material and methods
This review followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines (PRISMA) and was registered in the PROSPERO International Database of Systematic Reviews (CRD42021229775). The PubMed/MEDLINE, Scopus, and Cochrane databases were searched through July 2021. The PICO question was: “Dental implants installed in type IV bone have a lower success rate when compared to implants installed in type I bone, II and III?”. The established inclusion criteria were: 1) controlled and randomized clinical trials (RCT), 2) prospective and retrospective studies with at least 10 participants with dental implants, and 3) patients with dental implants installed in bone tissue types I, II, III, and IV (Lekholm and Zarb, 1985). The minimum followup duration was 1 year.
Results
After searching the identified databases, 117 articles were selected for full reading and 68 were excluded. Thus, 49 studies were included for qualitative and quantitative analyses. The total number of participants included was 12,056, with a mean age of 41.56 years and 29,905 implants installed. Bone types I, II, and III exhibit a lower implant failure rate when compared to bone type IV.
Conclusion
Dental implants installed in bone types I, II, and III showed significantly higher survival rates than those installed in type IV. The bone type I success rate was not significantly different than that of type II; however, the success rate of bone type I and II was higher than that of type III.
Keywords: Dental implants, Bone type, Bone quality, Survival rate, Systematic review
1. Introduction
Dental implants are a commonly recommended option for rehabilitating lost teeth (Singh et al., 2020), with a survival rate of 98.8 % over 10 years of function documented in the literature (Buser et al., 2012). The success of this treatment depends on factors related to the patient and the surgical procedure (Beer et al., 2003). In a clinical study carried out by Branemark research group, shows that some requirements are necessary for successful osseointegration, are included: controlled surgical technique, macroscopic and microscopic characteristics of the implants, individualization of the process depending on the quantity and quality of bone and adequate application of chewing load (Albrektsson et al., 1981, Thomková et al., 2023) Among patient-related factors, bone quantity and quality play important roles (Singh et al., 2020) and are critical prerequisites for achieving primary stability of dental implants (Eskandarloo et al., 2019).
Many types of classifications for bone quality have been suggested, but the most common is the Lekholm-Zarb classification (Sargolzaie et al., 2019). According to Lekholm and Zarb (1985), bone quality can be classified into four types depending on the relationship between the quantity and quality of cortical and spongy bone. Their classifications include type I, homogeneous compact bone with high density; type II, high-density spongy bone surrounded by thick cortical bone; type III, high-density spongy bone surrounded by thin cortical bone; and type IV, low-density spongy bone surrounded by thin cortical bone. In addition to these types, in 1992 Bahat proposed a modification to the Lekholm-Zarb classification with the addition of bone type V, a bone without the upper and lower cortical bone.
The posterior maxilla bone changes significantly after tooth extraction due to resorption of the alveolar and maxillary sinus pneumatization, resulting in reduced width and height of the alveolar bone. Further, type IV bone is present in the posterior maxilla, on account of this poor quality, making site preparation difficult as it shown thin cortical bone and interfere with primary stability (King EM, Schofield J, 2023).
Reports show that implants inserted in areas of poor bone quality are more likely to fail. A systematic review by Goiato et al. (2014) reported a reduction in the survival rate of implants inserted into bone types IV and V, with the type V without cortical bone resulting from grafting. However, these estimates are controversial (Eskandarloo et al., 2019). This is mainly due to the development of technologies to improve the performance of implants in low-density bone, such as the use of self-threaded implants and applied surface treatments (Orestein et al., 2000). Therefore, this systematic review aimed to evaluate the success rate of implants installed in bone type IV compared to that in types I, II, and III, according to the classification of Lekholm and Zarb (1995).
2. Materials and methods
2.1. Registration protocol
This systematic review adhered to the guidelines delineated by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) and was duly registered on the global platform dedicated to the registration of systematic reviews, PROSPERO (CRD42021229775).
2.2. Eligibility criteria
The PICO question (patient, intervention, comparison, outcome) was: “Do dental implants installed in type IV bone have a lower success rate when compared to implants installed in type I, II and III bone? The “Population” included patients submitted to the installation of dental implants; “Intervention”, implants installed in type IV bone tissue; “Comparison”, implants installed in type I, II and III bone, according to the Lekholm-Zarb classification; “Outcome”, the success rate of implants installed in bone type IV, was the primary outcome, as a secondary outcome, success rates were evaluated between bone types I, II and III and to evaluate the influence of surface treatment on the success of implants. implants installed in type IV bone.
The established inclusion criteria were: controlled and randomized clinical trials (RCT), as it is considered the most reliable source of evidence on the effectiveness of interventions (Higgins and Green, 2011) and prospective and retrospective studies, as many times the amount of RCT is insufficient and well-conducted cohort studies improve the strength of the comparison and the result (Ramamoorthi, Narvekar and Esfandiari, 2017). Studies that evaluated at least 10 participants, with patients presenting dental implants installed in type IV bone tissue, as well as in type I, II and III bone tissues (Lekholm and Zarb, 1985), and a minimum follow-up time of 1 year.
Exclusion criteria were: studies with patients younger than 18 years, studies that use a classification other than that of Lekholm and Zarb, 1985, studies that did not present bone types I, II, III, and IV in the same study, studies that did not present the number of implants installed and the number of implants that failed for a minimum follow-up period of 1 year, studies that included patients undergoing radiotherapy, chemotherapy, use of bisphosphonates and osteoporosis patients.
2.3. Search strategy
Two investigators (CDDRDR and CAAL) performed the search independently in the following databases: PubMed/MEDLINE, Scopus and Cochrane. Surveying the studies published until July 2021, according to the eligibility criteria.
The search strategy was based on the combination of the following terms: “(dental implants) AND (bone quality OR bone type)”. Likewise, a manual search was performed in high-impact journals in implant dentistry, such as Clinical Implant Dentistry and Related Research, Clinical Oral Implants Research, European Journal of Oral Implantology, Implant Dentistry, International Journal of Oral and Maxillofacial Implants, International Journal of Oral and Maxillofacial Surgery. In addition, a manual search of the references of the included articles was performed.
2.4. Data analysis
Two reviewers (CDDRDR and CAAL) independently reviewed the search results and identified potentially relevant studies based on title and abstract. Relevant studies were selected for reading in full and included in the systematic review according to the eligibility criteria. One author (CDDRDR) collected data from the included studies and a second author (CAAL) verified the information. If there was any disagreement between the reviewers, a third reviewer (EPP) was consulted. Qualitative data collected were study author and year, type of study, number of patients, mean age of study participants, implant systems, implant surface treatment, diameter and length, follow-up time in years, number of implants installed and flawed.
2.5. Data synthesis
The performed meta-analysis was based on the Mantel – Haenszel (MH) and Inverse Variance (IV) methods. Failure rates were assessed by hazard ratio (RR) RR values were considered significant when the P value was < 0.05. For statistically significant heterogeneity (P < 0.10), a random effects model was used to assess the significance of treatment effects. When no statistically significant heterogeneity was found, the analysis was performed using a fixed effects model. Reviewer Manager 5 software (Cochrane Group) was used for meta-analyses.
2.6. Risk of bias
Two authors (VAAB, JPJOL) performed risk of bias analysis on the included RCTs using the Cochrane risk of bias tool; this tool checks for selection bias (random sequence generation and allocation), performance bias (blinding of participants and staff), detection bias (blinding of outcome evaluation), attrition bias (result of incomplete data), reporting (selective reporting) and other biases (other sources of bias). The Newcastle-Ottawa (NOS) scale was used for non-randomized studies. NOS verifies three elements: selection, comparability and outcome for cohort studies. The scale classifies studies with a maximum of 9 stars; > 6 stars indicates a low risk of bias and scores ≤ 5 stars indicate a high risk of bias.
3. Results
A total of 9429 studies were found in the previously selected databases, 4721 in PubMed, 4167 in Scopus, 519 in Cochrane and 22 articles in the manual search. With the removal of duplicate references, 7727 articles were analyzed by title and abstract, of which 117 were chosen for full reading. Sixty-eight references were excluded, the causes are; Patients undergoing radiotherapy and/or chemotherapy; Patients using bisphosphonates (Alsaadi et al., 2008, Alsaadi et al., 2008, Chrcanovic et al., 2016; Tovalino et al., 2019; Friberg and Jemt, 2008, Friberg et al., 2005); Absence of any bone type or absence of separation of bone types (Anitua et al., 2016, Bahat, 1992, Becker et al., 1998, Demiralp et al., 2015, Duminil et al., 2008, Glauser et al., 2003, Glauser, 2013, Göçmen et al., 2016, Hingsammer et al., 2017, Jaffin and Berman, 1991, Jeong et al., 2015, Kumar et al., 2017, Rocci et al., 2003, Romanos et al., 2016; Sonal et al., 2017; Tawil and Mawla, 2001, Trbakovic et al., 2020, Wang et al., 2016, Zinser et al., 2013, Burdurlu et al., 2021, Kim et al., 2021, Staedt et al., 2020; Becker et al., 2015; Turner and Nentwig, 2014, Maiorana et al., 2015, Salimov et al., 2014, Mangano et al., 2014, Lai et al., 2013, Montero et al., 2012, Guarnieri et al., 2021, Boboeva et al., 2021, Bielemann et al., 2018, Waechter et al., 2017, Polizzi et al., 2000, Khang et al., 2001, Feldman et al., 2004); Presence of patients under 18 years of age (Balshi et al., 2015, Ottoni et al., 2005, Becker et al., 2013, Bianco et al., 2000, Widmark et al., 2003); Absence of number failed implants (Collaert and De Bruyn, 2008, Ganeles et al., 2008, Simmons et al., 2016, Zumstein et al., 2019, Raikar et al., 2017, Blume et al., 2020, Triches et al., 2019, Doan et al., 2014, Shivu et al., 2021, Castellanos-Cosano et al., 2019, Blus et al., 2010); Use of another classification for bone type (Degidi and Piattelli, 2005, Grandi et al., 2012, Al-Nawas et al., 2013); Follow-up less than 1 year (Fu et al., 2017, Nappo et al., 2019, Sargolzaie et al., 2019, Shin et al., 2014, Toia et al., 2017, Mohajerani et al., 2017). Thus, 49 studies were part of the qualitative and quantitative analysis. Details of the search strategy are illustrated in Fig. 1.
Fig. 1.
Search strategy.
3.1. Characteristics of the selected studies
Detailed data from the forty-nine selected studies are listed in Table 1. The total number of patients included was 12056, with a mean age of 41.56 years, 29,905 implants were analyzed, of which 2570 were installed in type I bone, 12,434 in bone type II, 12,240 in bone type III and 2661 in bone type IV. The follow-up period ranged from 1 to 19 years. The diameter of the implants ranged from 3.3 to 6.0 mm and the length ranged from 7 to 20 mm. Most studies used implants of the Brånemark System type.
Table 1.
Details of included studies.
| Author | study type | Patients | mean age | implant system | Surface | Diameter and Length | follow-up | Placed/Failed implants | |||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Type I | Type II | Type III | Type IV | ||||||||
| De Bruyn & Collaert, 1994 | Retrospective | 117 | 53.0 | Brånemark System | Machined | D = 3.75–4 mm L = 7; 10; 13; 15 mm |
3 years | 1/38 | 6/84 | 3/51 | 71/0 |
| Becker et al., 1995 | Retrospective | 24 | 58.6 | NR | NR | 10 x 3.75 mm 13 x 3.75 mm 15 x 3.75 mm 10x4mm 6x5mm 8x5mm |
1 year | 3/0 | 6/0 | 9/0 | 6/0 |
| Bergendal & Engquist, 1998 | Prospective | 49 | 66 | Brånemark System | Machined | D = 3.75 mm L = 7;10;13;15;18;20 mm |
7 years | 4/0 | 38/0 | 55/2 | 11/18 |
| Becker et al., 1999 | Prospective | 212 | 55.0 | Branemark System | Machined | D = 3.75;4.0;5.0 mm L = 8.5;10;11.5;13;15;18 mm |
6 years | 77/4 | 151/4 | 11/49 | 5/0 |
| Lekholm et al., 1999 | Prospective | 125 | 18–70 | Brånemark System | Machined | D = 3.75–4 mm L = 7;10;13;15;18; 20 mm |
10 years | 6/0 | 70/6 | 320/22 | 65/6 |
| Balshi et al., 1999 | Retrospective | 189 | 60.0 | Brånemark System | Machined | D = 3.75;4.0;5.0 mm L = 7;8.5;10;11.5;13;15;18;20 mm |
4.68 years | 2/0 | 2/26 | 131/16 | 217/24 |
| Friberg et al., 1999 | Prospective | 105 | 64.0 | Brånemark System MK-II | Machined | D = 3.75;4.0;5.0 mm L = 6;7;8.5;10;11.5;12;13;15;18;20 mm | 3 ye ars | 7/0 | 236/11 | 105/1 | 72/2 |
| Truhlar et al., 2000 | Prospective | NR | 20–80 | Spectra-System (CoreVent Corporation, Las Vegas, NV) | Acid-etched HA-coated and uncoated | D = 3.5;4.5 mm L = 8;10;10.5;13;16 mm | 3 years | 242/16 | 1294/93 | 1007/93 | 231/22 |
| Orenstein et al., 2000 | Retrospective | 800 | NR | Spectra system (micro -vent, Screw Vent, Bio Vent, Core-Vent) | Acid-etched HA-coated and uncoated | NR | 3 years | 238/14 | 1260/77 | 1026/83 | 238/19 |
| Glauser et al., 2001 | prospective | 41 | 52 | Brånemark System IV | Machined | D = 4.0;5.0 mm L = 7;8.5;10;11.5;13;15;18 mm | 1 year | 8/1 | 4/43 | 6/38 | 11/38 |
| Morris et al., 2001 | prospective | 31–90 | ankylos implants | NR | D = 3.5;4.5;5.5 mm L = 8;9.5;11;14;17 mm | 18 months | 163/1 | 601/21 | 543/22 | 95/5 | |
| Cochran et al., 2002 | Prospective | 110 | 54.5 | ITI SLA solid screw | Sandblasted and acid-etched | D = 4.1 mm L = 8;10;12;14 mm |
1 year | 30/0 | 163/1 | 133/2 | 57/0 |
| Engquist et al., 2002 | Prospective | 82 | 64.9 | Brånemark System | Machined | L = 10;13;15;18;21 mm | 1 year | 4/16 | 2/128 | 11/152 | 24/0 |
| Cal andriello et al., 2003 | Prospective | 26 | 52.3 | Brånemark System (standard fixture, Mk II, Mk III, and Mk IV) | Machined | 3.75;4.0 x 13 mm 5 x 10 mm |
1 year | 5/0 | 17/0 | 24/0 | 4/0 |
| Raghoebar et al., 2003 | Prospective | 40 | 18–70 | Brånemark System | Machined | 3.75 x 10;13;15;18 mm | 3 years | 13/0 | 93/4 | 3/57 | 7/4 |
| Tawil & Younan, 2003 | Prospective | 111 | 53.6 | Brånemark System | Machined | D = 3.3; 3.75; 4.5 mm L = 6;7;8;8.5;10 mm |
12–92 months | 3/0 | 79/2 | 8/160 | 2/27 |
| Friberg et al., 2005 | Prospective | 187 | 53.0 | Brånemark System MK-III, MK-IV | oxidized | D = 3.75 mm L = 7;8.5;10;11.5;13;15;18 mm |
1 year | 12/0 | 150/1 | 230/1 | 10/0 |
| Herrmann et al., 2005 | Retrospective | 487 | 51.3 | Brånemark System; Nobel Biocare, Goteborg, Sweden | Machined | 3.75 x 7 mm 3.75 x 10 mm |
5 yea lol | 2/18 | 7/155 | 265/15 | 12/49 |
| Noguerol et al., 2006 | Retrospective | 316 | NR | Brånemark System | Machined | D = 3.3; 3.75; 4.0; 5 mm L = 7, 8, 8.5, 10, 11.5, 12, 13, 15 and 18 mm |
10 years | 4/58 | 449/13 | 493/31 | 79/7 |
| Romeo et al., 2006 | Prospective | 120 | 55.8 | ITIs (Institute Straumann) | plasma-sprayed titanium | D = 3.3;4.1 mm L = 10;12 mm |
3–6 years | 1/42 | 122/3 | 115/6 | 51/5 |
| Becktor et al., 2007 | Prospective | 77 | 64.5 | Brånemark System | Machined | Standard and MKII – 8.5;10;11.5;13;15;18 mm | 36 months | 1/38 | 12/227 | 8/122 | 1/17 |
| Siddiqui et al., 2008 | Prospective | 65 | 41 | Tapered Screw-Vent MTX, Zimmer Dental | Sandblasted and HA-coated | D = 3.7;4.7;6.0 mm L = 10;13;16 mm | 1 years | 10/0 | 32/0 | 19/0 | 8/1 |
| Fischer et al., 2009 | Prospective | 32 | 54.0 | Replace Select TiUnite | oxidized | D = 4.3;5.0 mm L = 10;13;16 mm |
1 year | 1/0 | 29/0 | 20/0 | 3/1 |
| Ostman et al., 2010 | prospective | 35 | >18 | PREVAIL®, BIOMET 3i | Acid-etched and CaP-coated | D = 4.0;5.0 mm L = 8.5;10;11.5;13;15 mm | 1 years | 10/0 | 21/0 | 52/0 | 1/19 |
| Agliardi et al., 2009 | Prospective | 20 | 57 | Brånemark System MK IV and NobelSpeedy Groovy | oxidized | 4 x 11 mm 4 x 13 mm 4 x 15 mm |
27 months | 11/0 | 73/0 | 32/0 | 4/0 |
| Gallucci et al., 2009 | Prospective | 45 | 59.5 | Straumann AG | Sandblasted and acid-etched | L = 8;10;12;14;16 mm | 5 years | 43/0 | 126/0 | 38/0 | 25/0 |
| Johansson et al., 2009 | Prospective | 52 | 72 | Brånemark System MK- III TiUnite | oxidized | D = 3.75–4.0 L = 10;11.5;13;15 mm |
1 year | 126/1 | 134/1 | 49/0 | 3/0 |
| Sennerby et al., 2012 | Prospective | 90 | 50.6 | Neoss Implant System | Sandblasted and acid-etched | D = 3.5;4.0;4.5 mm L = 7;9;11;13;15 mm | 2 years | 12/0 | 2/142 | 44/0 | 1/20 |
| Jang et al., 2011 | Retrospective | 3755 | 65.0 | paragon TSV, Camlog, Biohorizon, Astra, Replace, GS | Machined, Oxidized, acid-etched, titanium plasma sprayed, sandblasted/acid-etched | D=<3.75; 3.75–4.5, >4.5 L=<10: 10–15; > 15 mm | 10 years | 271/1 | 2690/45 | 3312/59 | 112/3 |
| Calandriello and Tomatis, 2011 | Retrospective | 33 | 52 | Brånemark System TiUniteWide Platform MK | oxidized | Wide – L = 8.5;10;11.5;13;18 mm | 5 years | 3/1 | 21/0 | 1/15 | 1/0 |
| Galindo-moreno et al., 2012 | Prospective | 69 | 18–72 | Astra Tech OsseoSpeed™ TX 3.0S | TiO2-blasted fluoride -modified | D = 3.0 mm L = 11;13;15 mm |
1 years | 1/0 | 3/61 | 1/32 | 3/0 |
| Barewal et al., 2012 | RCT | 40 | 51.0 | OsseoSpeed, Astra Tech | TiO2-blasted fluoride -modified | 4 x 11 mm 4 x 13 mm |
3 years | 1/0 | 12/0 | 19/0 | 8/1 |
| Ostman et al., 2012 | Prospective | 42 | >18 | NanoTite™ Tapered Certain | Acid-etched and CaP-coated | D = 4.1;5.0 mm L = 8.5;10;11.5;13;15 mm |
1 year | 3/0 | 46/0 | 58/1 | 32/0 |
| Finne et al., 2012 | Prospective | 56 | 47.0 | NobelDirect and NobelPerfect | oxidized | D = 3.5;4.3;5.0 mm L = 10;13;16 mm |
3 years | 5/0 | 54/1 | 22/0 | 1/0 |
| Maló et al., 2003 | Prospective | 116 | 41 | Brånemark System | Machined | L = 10;11.5;13;15;18 mm | 1 year | 1/0 | 3/49 | 2/57 | 6/0 |
| Mangano et al., 2014 | Prospective | 642 | 51.0 | Leone Implant System | Sandblasted and acid-etched | D = 3.3, 4.1, 4.8 mm L = 8.0, 10.0, 12.0, 14.0 mm |
10 years | 84/1 | 292/2 | 727/7 | 9/391 |
| Dahlin et al., 2013 | Prospective | 177 | Majority > 50 | NEOSS implants | Sandblasted and acid-etched | D = 3.5;4;4.5;5.5 mm L = 7;9;11;13;15;17 mm | 1 year | 12/0 | 237/5 | 312/4 | 4/29 |
| Vasak et al., 2014 | Prospective | 30 | 31–80 | NobelReplace® | oxidized | D = 3.5;4.3;5.0 mm L = 8;10;13;16 mm |
1 year | 7/0 | 84/0 | 66/1 | 6/1 |
| Mangano et al., 2014 | Prospective | 279 | 25–73 | Leone implant system | Sandblasted and acid-etched | D = 3.3 mm L = 8;10;12;14 mm |
10 years | 26/0 | 75/0 | 154/3 | 69/1 |
| Glibert et al., 2016 | Prospective | 48 | >18 | Osseotitis 2 Certain | dual-acid etched | D = 4.0;5.0 mm L = 8.5;10;11.5;13;15 mm | 1 year | 4/0 | 30/0 | 61/0 | 20/0 |
| Han et al., 2014 | Retrospective | 879 | NR | Nobel Biocare, Straumann, Biolok International | Machined, Oxidized, acid-etched, titanium plasma sprayed, sandblasted/acid-etched | D=<3.75, 3.75–4.5, >4.5 – L=<10, 10–15, > 15 mm | 19 years | 73/0 | 1197/19 | 388/5 | 88/5 |
| Muelas-Jiménez et al., 2017 | Retrospective | 52 | 47.5 | SwissPlus | MTX surface | D = 3.7;4.1;4.8 mm L = 10;12;14 mm |
5 years | 17/0 | 70/3 | 9/61 | 6/16 |
| Olmedo-Gaya et al., 2016 | Retrospective | 142 | 48.49 | BTI implants | Bioactive surface | D = 2.5;3.0;3.3 mm L = 7;7.5;8.5;10;11.5;13;15 mm | 5 years | 1/21 | 8/83 | 133/3 | 4/23 |
| Díaz-Sánchez et al., 2017 | Prospective | 27 | 18–65 | ELEMENT INICELL, Thommen Medical AG, Waldenburg, Germany | Sandblasted and acid-etched | D = 3.5;4.0;4.2;5.0 mm L = 8;9.5;11;12.25 mm |
1 years | 42/0 | 16/0 | 8/0 | 1/0 |
| Andersson et al., 2019 | Retrospective | 334 | 52.0 | neoss implant | Sandblasted and acid-etched | D = 3.5–5.5 mm L = 7–17 mm |
5 years | 37/0 | 392/11 | 263/8 | 50/1 |
| Eskandarloo et al., 2019 | Retrospective | 22 | 42.6 | NR | NR | NR | 30 months | 3/0 | 35/0 | 44/0 | 01/18 |
| Singh et al., 2020 | Retrospective | 826 | NR | NR | NR | D=< 3.75 mm; 3.75–4.5 mm; >4.5 mm x L= < 10 mm; 10–11.5 mm; >11.5 mm |
10 years | 648/52 | 412/38 | 210/24 | 150/31 |
| Thome et al., 2020 | Retrospective | 101 | 59.17 | Helix Acqua GM, Neodent | Sandblasted and acid-etched surfaces with hydrophilic properties | D = 3.5;3.75;4.0;4.3;5.0;6.0 mm L = 8;10;11.5;13;16;18 |
2 years | 49/0 | 98/1 | 116/0 | 25/0 |
| Wang et al., 2021 | Retrospective | 827 | 44.11 | Straumann® Dental Implant System | Sandblasted and acid-e tched |
D = 3.3; 4.1; 4.8 mm L = 8;10 mm |
9 years | 2/26 | 531/20 | 843/14 | 149/4 |
3.2. Risk of bias
For randomized clinical trials, the Cochrane scale was used only one study was assessed by this scale (Barewal et al., 2012), the risk of bias was considered low for the following outcomes: random sequence generation, blinding of participants and raters, incomplete outcomes, report of selective outcome and other sources of bias, the outcome allocation concealment was assessed with uncertain risk of bias. All prospective and retrospective studies evaluated using the NOS scale demonstrated a low risk of bias, as they obtained more than six stars, as specified in Table 2.
Table 2.
Detailed Newcastle-Ottawa Scale of each included cohort study.
3.3. Meta-analysis
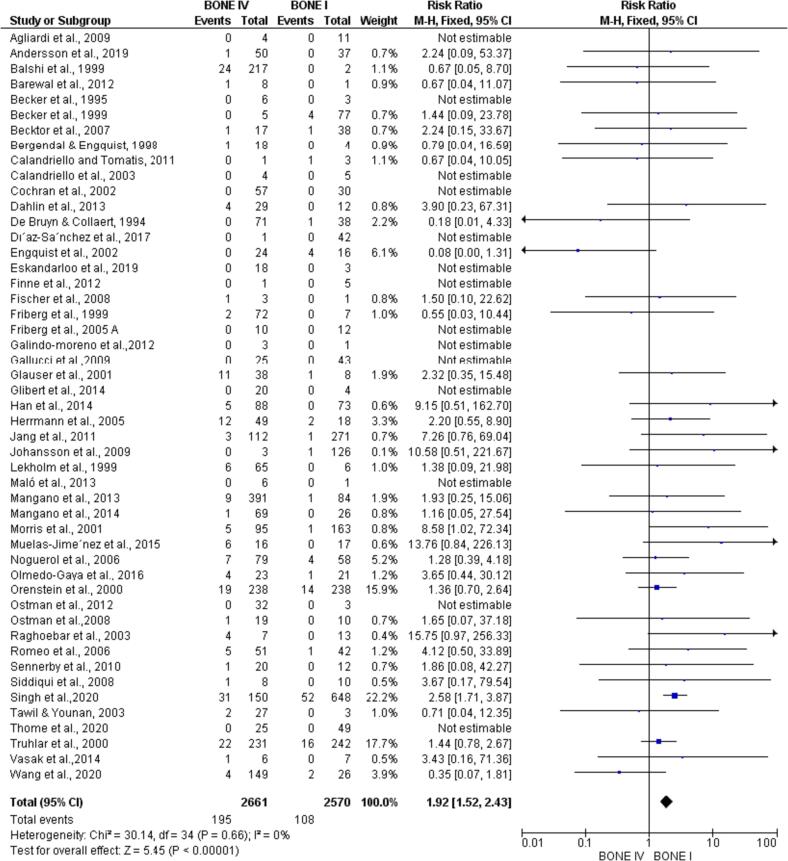
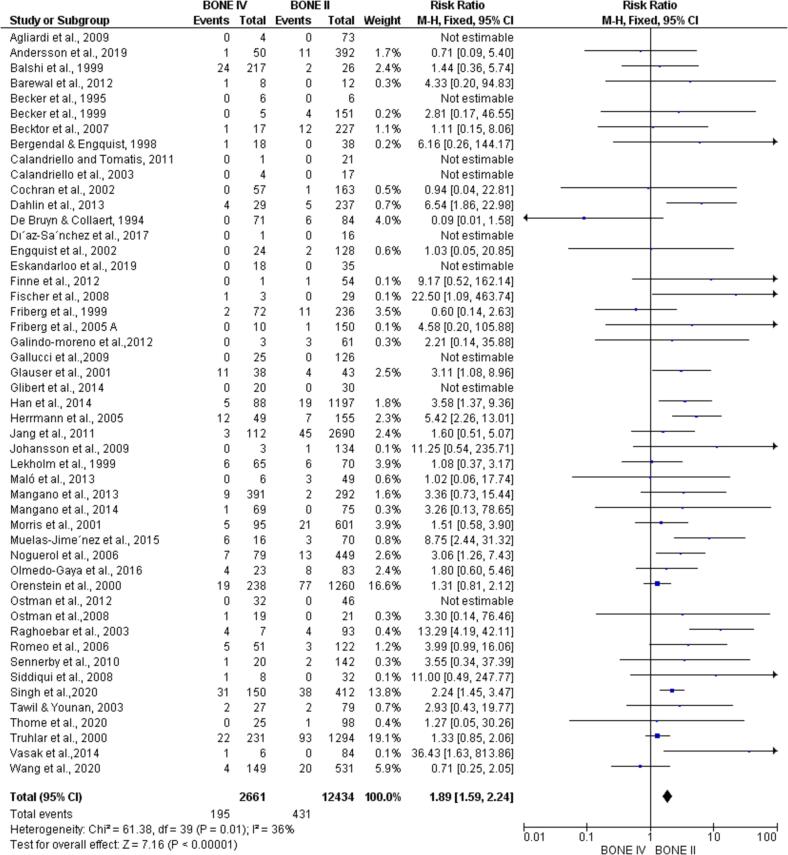
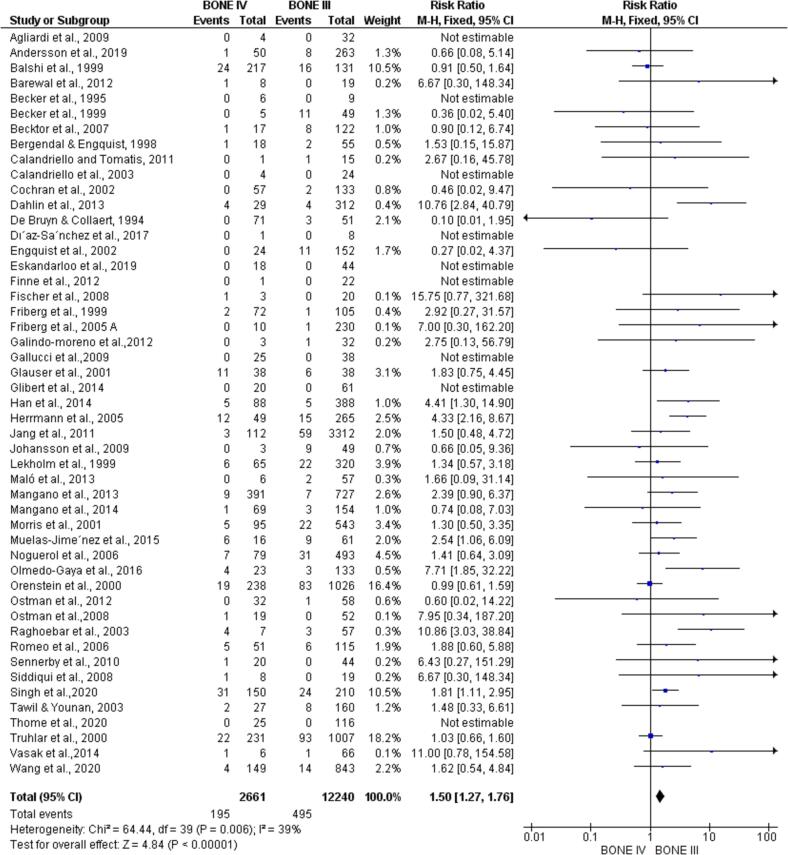
All 49 articles could be included in the meta-analysis. When comparing the number of installed/failed implants between type I and type IV bone, there was a statistically significant difference in favor of type I bone (p < 0.00001; RR: 1.92; 95 %CI: 1.52–2.43) (Fig. 2) and when compared bone types II and IV, being significantly favorable to bone type II (p < 0.00001; RR: 1.89; 95 %CI: 1.59–2.24) (Fig. 3). Furthermore, there was a statistically significant difference when comparing type III and IV bone, favorable to type III bone (p < 0.00001; RR: 1.50; 95 %CI: 1.27–1.76) (Fig. 4).
Fig. 2.
Meta-analysis comparing the failure rate of implants installed in type I x IV bone. By meta-analysis, implants installed in bone type I had a significantly higher survival rate than implants installed in bone type IV.
Fig. 3.
Meta-analysis comparing the failure rate of implants placed in type II x IV bone. By meta-analysis, implants installed in bone type II had a significantly higher survival rate than implants installed in bone type IV.
Fig. 4.
Meta-analysis comparing the failure rate of implants installed in type III x IV bone. By meta-analysis, implants installed in bone type III had a significantly higher survival rate than implants installed in bone type IV.
4. Discussion
Although the survival rate of dental implants over a 10-year observation period was reported as greater than 90 % (Alghamdi, 2018), implants do fail in some patients. In this study, the null hypothesis that implants placed in bone type IV have the same success rates as those in types I, II, and III was rejected, since according to the meta-analysis, a significant difference in the survival rate was observed when other bone types were compared with type IV.
This systematic review presents results in agreement with those of Goiato et al. (2014), who showed that implants inserted in bone type IV had a lower success rate than that of other types, and that implants with surface treatments showed a higher success rate than that of machined implants. However, the current review adds improvements such as the use of a meta-analysis to acquire data, the inclusion of a greater number of new articles, and the assessment of the risk of bias. In addition, new data were acquired in the meta-analysis showing equal performance of implants installed in bone types I and II.
Preoperative bone evaluation is critical for implant treatment planning as it improves the accurate determination of prognosis and increases the implant success rate (Eskandarloo et al., 2019). Various factors are important for long-term implant success, including good bone quality around the implant and a solid bone/implant interface (Linetskiy et al., 2017), implant design and shape (Thomé et al., 2020), and local and systemic patient-related factors (Mohajerani et al., 2017).
Bone quality was suggested by Díaz-Sánchez et al. (2017) as one of the main factors related to the success of implant therapy. Their study showed that implants installed in bone types III and IV were more likely to fail, whereas the highest survival rates were achieved in bone types I and II, which have a higher density (Muelas-Jiménez et al., 2015) and superior primary stability. A high primary stability is a prerequisite for implant survival (Glibert et al., 2016). Dense and spongy compact bones are favorable because they hold the implant firmly in the bone. In contrast, porous and trabecular bones decrease implant stability (Kate et al., 2016) leading to micromovements at the bone-implant interface during healing, which in turn stimulates soft tissue formation fibrosis rather than bone repair (Andersson et al., 2019). In their 5-year analysis, Jaffin and Berman (1991) reported a 35 % failure of implants installed in bone type IV, with 36 failures in a total of 102 installed implants due to thin cortical bone. In the same study, 952 implants were placed in bone types I, II, and III, of which 29 failed (3 %). Andersson et al. (2019) reported a lower survival rate of implants with low stability than that of implants with greater stability.
A decrease in the cortical bone leads to an increase in local bone stress, which may increase the micromovement of the peri-implant bone, impairing primary stability. Primary stability is critical for improving the success rate of dental implants (Chou et al., 2014). The studies by Andersson et al., 2019, Barewal et al., 2012, and Sennerby et al. (2012) reported implant stability quotient (ISQ) values acquired using the Osstell device immediately after implant installation in different bone types. Bone type IV revealed the lowest mean primary stability values, i.e., 58 (Barewal et al., 2012), 63.8 (Andersson et al., 2019), and 60–65 (Sennerby et al., 2012). The bone type I values were 72 (Barewal et al., 2012), 76.5 (Andersson et al., 2019), and 75–80 (Sennerby et al., 2012), the bone type II values were 72 (Barewal et al., 2012), 75 (Sennerby et al., 2012), and 75.2 (Andersson et al., 2019), and finally, the type III bone values, were 70 (Barewal et al., 2012), 70–75 (Sennerby et al., 2012), and 72 (Andersson et al., 2019). In addition, despite being identified as an important factor in achieving final osseointegration, introducing implants with high installation torque resulted in bone resorption due to impairment of the vascularized bone caused by increased tension in the bone walls (Galindo-Moreno et al., 2012).
DeBruyn and Collaert, 1994, Friberg et al., 1999, Tawil and Younan, 2003, and Jang et al. (2011) reported higher failure rates in the maxilla than in the mandible. According to Andersson et al. (2019), an analysis of resonance frequency (RFA) showed that the stability of maxillary implants was lower than that of mandibular implants. In addition, the implant stability quotient (ISQ) values were significantly higher in the mandible, and the author speculated that this finding was due to the bones in the maxilla commonly being of lower density than those in the mandible. In addition to bone quality, the main factors that influenced primary stability are the implant design and surgical procedure (Huang et al., 2014).
Tapered dental implants demonstrate superior mechanical stability compared to cylindrical dental implants (Romanos et al., 2014). In addition, primary stability is influenced by implant dimensions (length and diameter), surface characteristics (rough or smooth), number of threads on the implant surface (Romanos et al., 2020), and pitch, shape, and thread depth (Alghamdi, 2018), as the presence of threads increases the surface area for osseointegration (Sykaras et al., 2000).
Various implantation techniques have been proposed to promote a high degree of implant stability without removing additional bone, particularly in situations where bone density is limited (Alghamdi, 2018). Studies have shown good results in less-dense bones, which may be the result of modified surgical techniques, such as the use of self-threading implants, conical implants, and final drills with a smaller diameter than the diameter of the implant (Fischer et al., 2009). Lateral compression of the recipient bed has been successfully used in implant dentistry (Tabassum et al., 2013). Lateral compression is achieved by placing an implant in a bone cavity that is considerably smaller in diameter than the diameter of the implant itself, resulting in greater insertion torque, which is an indicator of high primary stability (Tabassum et al., 2013). Although the posterior regions, especially the maxilla, are considered more challenging because of the presence of less-dense bone, stability can be achieved using undermilling techniques (Gehrke et al., 2018, Östman et al., 2010).
Recent advances in dentistry have revolutionized the use of dental implants. Thomé et al. (2020) reported survival rates ranging from 99 % to 100 % for different bone types (I–IV) with a 2-year followup. This result may be explained by the conical design of the evaluated hybrid implants using the protocol of a suitable drill, allowing compaction of the trabecular bone in the middle and cervical portions, and promoting better results regardless of the type of bone site.
Osseointegration may be impaired in patients with diabetes mellitus (Mombelli and Cionca, 2006) and osteoporosis (Giro et al., 2015), and in those treated with radiotherapy (Koudougou et al., 2020). Jang et al. (2011) reported that the failure rate tends to increase with patient age due to decreasing bone density. This is because over time, the rate of bone resorption is greater than that of bone production. Thus, the cortical bone was thinner and the porosity of the cancellous bone increased, highlighting the need for surface modifications that accelerated osseointegration after implant insertion (Sekar et al., 2019).
The surface of the implants and speed of bone apposition around the implantation site are key factors in bone-implant contact (Mohajerani et al., 2017). In a previous report, surface microtopography significantly influenced the osseointegration of titanium implants due to an increase in surface roughness leading to greater bone apposition and decreased healing time (Ding et al., 2020). Topographically modified titanium implant surfaces, such as sandblasted, acid-etched (SLA), and chemically modified hydrophilic SLA surfaces, have shown promising results when compared to polished titanium surfaces (Chakravorty et al., 2017). Studies revealed that implants with modified surfaces showed a higher success rate than that of superficial implants machined in compromised bone situations (Del Fabbro et al., 2006, Del Fabbro et al., 2008, Smeets et al., 2016).
The heterogeneity identified in meta-analyses comparing the number of installed vs. failed implants among different bone types is considered low (Higgins and Green, 2011). A limitation of the present meta-analysis is that the studies did not determine the length and width of each implant installed in each bone type, nor did they determine the type of connection used. These factors may interfere with the biomechanics of implant-supported prostheses, and consequently, with the success of rehabilitation. Therefore, conducting new randomized clinical trials including standardization of implant length and width, type of connection, and surface treatment is warranted.
In order to achieve higher success rates for implants placed in bone type IV, the clinician must take into account various features including systemic factors related to the patient, the diameter and length of the implant, properties related to the thread of the implant (pitch, shape, and depth), surface treatment, and the use of surgical techniques that preserve the bone in the recipient site. In addition, a less traumatic surgical procedure, two-stage approach, and adequate healing time are important (Gehrke et al., 2018, Jang et al., 2011).
5. Conclusion
Dental implants installed in bone types I, II, and III had a significantly higher survival rate than implants installed in bone type IV. To reduce implant failure rates, it is clinically important, when selecting an implant, to consider bone density, as it is a critical factor for achieving high insertion torque. Therefore, it is advisable to adapt drilling protocols according to the bone density of the rehabilitated area. In situations involving type IV bone, the use of additional drills, such as thread male drills and cortical drills, is strongly discouraged, as such practices may compromise the success of the treatment entirely.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Footnotes
Peer review under responsibility of King Saud University. Production and hosting by Elsevier.
Contributor Information
Davi Del Rei Daltro, Email: cleberdavi2@hotmail.com.
Augusto Alves, Email: vtrbento97@gmail.com.
Dantas, Email: duarte-nathalia@hotmail.com.
Mateus Cavalaro, Email: jm.sayeg@unesp.br.
Justo, Email: thawan.justo@unesp.br.
Piza, Email: ed.pl@uol.com.br.
References
- Agliardi E.L., Francetti L., Romeo D., Del Fabbro M. Immediate rehabilitation of the edentulous maxilla: preliminary results of a single-cohort prospective study. Int. J. Oral Maxillofac. Implants. 2009;24(5) [PubMed] [Google Scholar]
- Albrektsson T., Brånemark P.I., Hansson H.A., Lindström J. Osseointegrated titanium implants: requirements for ensuring a long-lasting, direct bone-to-implant anchorage in man. Acta Orthop. Scand. 1981;52(2):155–170. doi: 10.3109/17453678108991776. [DOI] [PubMed] [Google Scholar]
- Alghamdi H.S. Methods to improve osseointegration of dental implants in low quality (type-IV) bone: an overview. J. Funct. Biomater. 2018;9(1):7. doi: 10.3390/jfb9010007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Al-Nawas B., Krummenauer F., Büchter A., Kleinheinz J., Neukam F., Petrin G., Wagner W. Multicenter randomized clinical trial: early loading of implants in maxillary bone. Clin. Implant Dent. Relat. Res. 2013;15(5):625–636. doi: 10.1111/j.1708-8208.2011.00427.x. [DOI] [PubMed] [Google Scholar]
- Alsaadi G., Quirynen M., Komárek A., Van Steenberghe D. Impact of local and systemic factors on the incidence of late oral implant loss. Clin. Oral Implant Res. 2008;19(7):670–676. doi: 10.1111/j.1600-0501.2008.01534.x. [DOI] [PubMed] [Google Scholar]
- Alsaadi G., Quirynen M., Michiles K., Teughels W., Komárek A., Van Steenberghe D. Impact of local and systemic factors on the incidence of failures up to abutment connection with modified surface oral implants. J. Clin. Periodontol. 2008;35(1):51–57. doi: 10.1111/j.1600-051X.2007.01165.x. [DOI] [PubMed] [Google Scholar]
- Andersson, P., Pagliani, L., Verrocchi, D., Volpe, S., Sahlin, H., & Sennerby, L. (2019). Factors influencing resonance frequency analysis (RFA) measurements and 5-year survival of Neoss dental implants. International journal of dentistry, 2019. [DOI] [PMC free article] [PubMed]
- Anitua E., Flores J., Flores C., Alkhraisat M.H. Long-term outcomes of immediate loading of short implants: A controlled retrospective cohort study. Int. J. Oral Maxillofac. Implants. 2016;31(6) doi: 10.11607/jomi.5330. [DOI] [PubMed] [Google Scholar]
- Bahat O. Osseointegrated implants in the maxillary tuberosity: report on 45 consecutive patients. Int. J. Oral Maxillofac. Implants. 1992;7(4) [PubMed] [Google Scholar]
- Balshi T.J., Wolfinger G.J., Balshi S.F. Analysis of 356 pterygomaxillary implants in edentulous arches for fixed prosthesis anchorage. Int. J. Oral Maxillofac. Implants. 1999;14(3):398–406. [PubMed] [Google Scholar]
- Balshi T.J., Wolfinger G.J., Stein B.E., Balshi S.F. A long-term retrospective analysis of survival rates of implants in the mandible. Int. J. Oral Maxillofac. Implants. 2015;30(6):1348–1354. doi: 10.11607/jomi.3910. [DOI] [PubMed] [Google Scholar]
- Barewal R.M., Stanford C., Weesner T.C. A randomized controlled clinical trial comparing the effects of three loading protocols on dental implant stability. Int. J. Oral Maxillofac. Implants. 2012;27(4) [PubMed] [Google Scholar]
- Becker W., Becker B.E. Replacement of maxillary and mandibular molars with single endosseous implant restorations: a retrospective study. J. Prosthet. Dent. 1995;74(1):51–55. doi: 10.1016/s0022-3913(05)80229-x. [DOI] [PubMed] [Google Scholar]
- Becker B.E., Becker W., Ricci A., Geurs N. A prospective clinical trial of endosseous screw-shaped implants placed at the time of tooth extraction without augmentation. J. Periodontol. 1998;69(8):920–926. doi: 10.1902/jop.1998.69.8.920. [DOI] [PubMed] [Google Scholar]
- Becker W., Becker B.E., Alsuwyed A., Al-Mubarak S. Long-term evaluation of 282 implants in maxillary and mandibular molar positions: A prospective study. J. Periodontol. 1999;70(8):896–901. doi: 10.1902/jop.1999.70.8.896. [DOI] [PubMed] [Google Scholar]
- Becker W., Hujoel P., Becker B.E., Wohrle P. Survival rates and bone level changes around porous oxide-coated implants (T i U nite™) Clin. Implant Dent. Relat. Res. 2013;15(5):654–660. doi: 10.1111/cid.12106. [DOI] [PubMed] [Google Scholar]
- Becktor J.P., Isaksson S., Billström C. A prospective multicenter study using two different surgical approaches in the mandible with turned Brånemark implants: conventional loading using fixed prostheses. Clin. Implant Dent. Relat. Res. 2007;9(4):179–185. doi: 10.1111/j.1708-8208.2007.00041.x. [DOI] [PubMed] [Google Scholar]
- Beer A., Gahleitner A., Holm A., Tschabitscher M., Homolka P. Correlation of insertion torques with bone mineral density from dental quantitative CT in the mandible. Clin. Oral Implants Res. 2003;14:616–620. doi: 10.1034/j.1600-0501.2003.00932.x. [DOI] [PubMed] [Google Scholar]
- Bergendal T., Engquist B. Implant-supported overdentures: a longitudinal prospective study. Int. J. Oral Maxillofac. Implants. 1998;13(2) [PubMed] [Google Scholar]
- Bianco G., Di Raimondo R., Luongo G., Paoleschi C., Piccoli P., Piccoli C., Rangert B. Osseointegrated implant for single-tooth replacement: a retrospective multicenter study on routine use in private practice. Clin. Implant Dent. Relat. Res. 2000;2(3):152–158. doi: 10.1111/j.1708-8208.2000.tb00006.x. [DOI] [PubMed] [Google Scholar]
- Bielemann A.M., Marcello-Machado R.M., Leite F.R.M., Martinho F.C., Chagas-Júnior O.L., Cury A.A.D.B., Faot F. Comparison between inflammation-related markers in peri-implant crevicular fluid and clinical parameters during osseointegration in edentulous jaws. Clin. Oral Invest. 2018;22(1):531–543. doi: 10.1007/s00784-017-2169-0. [DOI] [PubMed] [Google Scholar]
- Blume O., Schnödt E.M., Back M., Wildenhof J.I., Probst F.A., Otto S. Long-term efficacy of variable-thread tapered implants—A retrospective, clinical and radiological evaluation. Medicine. 2020;56(11):564. doi: 10.3390/medicina56110564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blus C., Szmukler-Moncler S., Vozza I., Rispoli L., Polastri C. Split-crest and immediate implant placement with ultrasonic bone surgery (piezosurgery): 3-year follow-up of 180 treated implant sites. Quintessence Int. 2010;41(6) [PubMed] [Google Scholar]
- Boboeva O., Kwon T.G., Kim J.W., Lee S.T., Choi S.Y. Comparing factors affecting dental-implant loss between age groups: A retrospective cohort study. Clin. Implant Dent. Relat. Res. 2021;23(2):208–215. doi: 10.1111/cid.12967. [DOI] [PubMed] [Google Scholar]
- Burdurlu, M. Ç., Dagasan, V. Ç., Tunç, O., & Güler, N. (2021). Retrograde peri-implantitis: evaluation and treatment protocols of a rare lesion. Quintessence international (Berlin, Germany: 1985), 52(2), 112-121. [DOI] [PubMed]
- Buser D., Janner S.F., Wittneben J.G., Brägger U., Ramseier C.A., Salvi G.E. 10-year survival and success rates of 511 titanium implants with a sandblasted and acid-etched surface: a retrospective study in 303 partially edentulous patients. Clin. Implant Dent. Report Res. 2012 Dec;14(6):839–851. doi: 10.1111/j.1708-8208.2012.00456.x. PMID: 22897683. [DOI] [PubMed] [Google Scholar]
- Calandriello R., Tomatis M. Immediate occlusal loading of single lower molars using brånemark system® wide platform tiunite™ implants: A 5-year follow-up report of a prospective clinical multicenter study. Clin. Implant Dent. Relat. Res. 2011;13(4):311–318. doi: 10.1111/j.1708-8208.2009.00214.x. [DOI] [PubMed] [Google Scholar]
- Calandriello R., Tomatis M., Vallone R., Rangert B., Gottlow J. Immediate occlusal loading of single lower molars using Brånemark System® wide-platform TiUnite™ implants: an interim report of a prospective open-ended clinical multicenter study. Clin. Implant Dent. Relat. Res. 2003;5:74–80. doi: 10.1111/j.1708-8208.2003.tb00018.x. [DOI] [PubMed] [Google Scholar]
- Castellanos-Cosano L., Rodriguez-Perez A., Spinato S., Wainwright M., Machuca-Portillo G., Serrera-Figallo M.A., Torres-Lagares D. Descriptive retrospective study analyzing relevant factors related to dental implant failure. Oral Med. Oral Pathol. Oral Surg. 2019;24(6):e726. doi: 10.4317/medoral.23082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chakravorty N., Jaiprakash A., Ivanovski S., Xiao Y. Implant Surface Modifications and Osseointegration. Springer; Berlin, Heidelberg: 2017. pp. 107–131. [Google Scholar]
- I.-C. Chou S.-Y. Lee C.-P. Jiang Effects of implant neck design on primary stability and overload in a type IV mandibular bone International Journal for Numerical Methods in Biomedical Engineering 30 11 2014 1223 1237 10.1002 /cnm.2653. [DOI] [PubMed]
- Chrcanovic B.R., Kisch J., Albrektsson T., Wennerberg A. Bruxism and dental implant failures: a multilevel mixed effects parametric survival analysis approach. J. Oral Rehabil. 2016;43(11):813–823. doi: 10.1111/joor.12431. [DOI] [PubMed] [Google Scholar]
- Cochran D.L., Buser D., Ten Bruggenkate C.M., Weingart D., Taylor T.M., Bernard J.P., Simpson J.P. The use of reduced healing times on ITI® implants with a sandblasted and acid-etched (SLA) surface: Early results from clinical trials on ITI® SLA implants. Clin. Oral Implant Res. 2002;13(2):144–153. doi: 10.1034/j.1600-0501.2002.130204.x. [DOI] [PubMed] [Google Scholar]
- Collaert B., De Bruyn H. Immediate functional loading of TiOblast dental implants in full-arch edentulous maxillae: a 3-year prospective study. Clin. Oral Implant Res. 2008;19(12):1254–1260. doi: 10.1111/j.1600-0501.2008.01586.x. [DOI] [PubMed] [Google Scholar]
- Dahlin C., Widmark G., Bergkvist G., Fürst B., Widbom T., Kashani H. One-year results of a clinical and radiological prospective multicenter study on NEOSS® dental implants. Clin. Implant Dent. Relat. Res. 2013;15(2):303–308. doi: 10.1111/j.1708-8208.2011.00356.x. [DOI] [PubMed] [Google Scholar]
- DeBruyn H., Collaert B. The effect of smoking on early implant failure. Clin. Oral Implant Res. 1994;5(4):260–264. doi: 10.1034/j.1600-0501.1994.050410.x. [DOI] [PubMed] [Google Scholar]
- Degidi M., Piattelli A. A 7-year follow-up of 93 immediately loaded titanium dental implants. J. Oral Implantol. 2005;31(1):25–31. doi: 10.1563/0-730.1. [DOI] [PubMed] [Google Scholar]
- Del Fabbro M., Testori T., Francetti L., Taschieri S., Weinstein R. Systematic review of survival rates for immediately loaded dental implants. Int. J. Periodont. Restor. Dent. 2006;26:249–263. [PubMed] [Google Scholar]
- Del Fabbro M., Rosano G., Taschieri S. Implant survival rates after maxillary sinus augmentation. Eur. J. Oral Sci. 2008;116(6):497–506. doi: 10.1111/j.1600-0722.2008.00571.x. [DOI] [PubMed] [Google Scholar]
- Demiralp, K. Ö., Akbulut, N., Kursun, S., Argun, D., Bagis, N., & Orhan, K. (2015). Survival rate of short, locking taper implants with a plateau design: a 5-year retrospective study. BioMed Research International, 2015. [DOI] [PMC free article] [PubMed]
- Díaz-Sánchez R.M., Delgado-Muñoz J.M., Hita-Iglesias P., Pullen K.T., Serrera-Figallo M.Á., Torres-Lagares D. Improvement in the initial implant stability quotient through use of a modified surgical technique. J. Oral Implantol. 2017;43(3):186–193. doi: 10.1563/aaid-joi-D-16-00159. [DOI] [PubMed] [Google Scholar]
- Q. Ding R. Zhang L. Zhang Y. Sun Q. Xie Effects of Different Microstructured Surfaces on the Osseointegration of CAD/CAM Zirconia Dental Implants: An Experimental Study in Rabbits The International Journal of Oral & Maxillofacial Implants 35 6 2020 1113 1121 10.11607 /jomi.8207. [DOI] [PubMed]
- Doan N.V.T., Du Z., Reher P., Xiao Y. Flapless dental implant surgery: a retrospective study of 1,241 consecutive implants. Int. J. Oral Maxillofac. Implants. 2014;29(3) doi: 10.11607/jomi.3195. [DOI] [PubMed] [Google Scholar]
- Duminil G., Muller-Bolla M., Brun J.P., Leclercq P., Bernard J.P., Dohan Ehrenfest D.M. Success rate of the EVL evolution implants (SERF): a five-year longitudinal multicenter study. J. Oral Implantol. 2008;34(5):282–289. doi: 10.1563/1548-1336(2008)34[283:SROTEE]2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- Engquist B., Åstrand P., Anzén B., Dahlgren S., Engquist E., Feldmann H., Svärdström P. Simplified methods of implant treatment in the edentulous lower jaw. A controlled prospective study. Part I: one-stage versus two-stage surgery. Clin. Implant Dent. Relat. Res. 2002;4(2):93–103. doi: 10.1111/j.1708-8208.2002.tb00158.x. [DOI] [PubMed] [Google Scholar]
- Eskandarloo A., Arabi R., Bidgoli M., Yousefi F., Poorolajal J. Association between marginal bone loss and bone quality at dental implant sites based on evidence from cone beam computed tomography and periapical radiographs. Contemp. Clin. Dent. 2019;10(1):36. doi: 10.4103/ccd.ccd_185_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feldman S., Boitel N., Weng D., Kohles S.S., Stach R.M. Five-Year Survival Distributions of Short-Length (10 mm or less) Machined-Surfaced and Osseotite® Implants. Clin. Implant Dent. Relat. Res. 2004;6(1):16–23. doi: 10.1111/j.1708-8208.2004.tb00023.x. [DOI] [PubMed] [Google Scholar]
- Finne K., Rompen E., Toljanic J. Three-year prospective multicenter study evaluating marginal bone levels and soft tissue health around a one-piece implant system. Int. J. Oral Maxillofac. Implants. 2012;27(2) [PubMed] [Google Scholar]
- Fischer K., Backström M., Sennerby L. Immediate and early loading of oxidized tapered implants in the partially edentulous maxilla: a 1-year prospective clinical, radiographic, and resonance frequency analysis study. Clin. Implant Dent. Relat. Res. 2009;11(2):69–80. doi: 10.1111/j.1708-8208.2008.00096.x. [DOI] [PubMed] [Google Scholar]
- Friberg B., Sennerby L., Gröndahl K., Bergström C., Back T., Lekholm U. On cutting torque measurements during implant placement: a 3-year clinical prospective study. Clin. Implant Dent. Relat. Res. 1999;1(2):75–83. doi: 10.1111/j.1708-8208.1999.tb00095.x. [DOI] [PubMed] [Google Scholar]
- Friberg B., Dahlin C., Widmark G., Östman P.O., Billström C. One-year results of a prospective multicenter study on Brånemark System® implants with a TiUnite™ surface. Clin. Implant Dent. Relat. Res. 2005;7:s70–s75. doi: 10.1111/j.1708-8208.2005.tb00077.x. [DOI] [PubMed] [Google Scholar]
- Friberg B., Henningsson C., Jemt T. Rehabilitation of edentulous mandibles by means of turned Brånemark System® implants after one-stage surgery: a 1-year retrospective study of 152 patients. Clin. Implant Dent. Relat. Res. 2005;7(1):1–9. doi: 10.1111/j.1708-8208.2005.tb00040.x. [DOI] [PubMed] [Google Scholar]
- Friberg B., Jemt T. Rehabilitation of edentulous mandibles by means of five TiUnite™ implants after one-stage surgery: A 1-year retrospective study of 90 patients. Clin. Implant Dent. Relat. Res. 2008;10(1):47–54. doi: 10.1111/j.1708-8208.2007.00060.x. [DOI] [PubMed] [Google Scholar]
- Fu M.W., Fu E., Lin F.G., Chang W.J., Hsieh Y.D., Shen E.C. Correlation between resonance frequency analysis and bone quality assessments at dental implant recipient sites. Int. J. Oral Maxillofac. Implants. 2017;32(1) doi: 10.11607/jomi.4684. [DOI] [PubMed] [Google Scholar]
- Galindo-Moreno P., Nilsson P., King P., Becktor J., Speroni S., Schramm A., Maiorana C. Clinical and radiographic evaluation of early loaded narrow diameter implants–1-year follow-up. Clin. Oral Implant Res. 2012;23(5):609–616. doi: 10.1111/j.1600-0501.2011.02254.x. [DOI] [PubMed] [Google Scholar]
- Gallucci G.O., Doughtie C.B., Hwang J.W., Fiorellini J.P., Weber H.P. Five-year results of fixed implant-supported rehabilitations with distal cantilevers for the edentulous mandible. Clin. Oral Implant Res. 2009;20(6):601–607. doi: 10.1111/j.1600-0501.2008.01699.x. [DOI] [PubMed] [Google Scholar]
- Ganeles J., Zöllner A., Jackowski J., Ten Bruggenkate C., Beagle J., Guerra F. Immediate and early loading of Straumann implants with a chemically modified surface (SLActive) in the posterior mandible and maxilla: 1-year results from a prospective multicenter study. Clin. Oral Implant Res. 2008;19(11):1119–1128. doi: 10.1111/j.1600-0501.2008.01626.x. [DOI] [PubMed] [Google Scholar]
- Gehrke S.A., Guirado J.L.C., Bettach R., Fabbro M.D., Martinez C.P.A., Shibli J.A. Evaluation of the insertion torque, implant stability quotient and drilled hole quality for different drill design: an in vitro investigation. Clin. Oral Implant Res. 2018;29(6):656–662. doi: 10.1111/clr.12808. [DOI] [PubMed] [Google Scholar]
- Giro G., Chambrone L., Goldstein A., Rodrigues J.A., Zenóbio E., Feres M., Shibli J.A. Impact of osteoporosis in dental implants: a systematic review. World J. Orthop. 2015;6(2):311. doi: 10.5312/wjo.v6.i2.311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glauser R. Implants with an oxidized surface placed predominantly in soft bone quality and subjected to immediate occlusal loading: results from a 7-year clinical follow-up. Clin. Implant Dent. Relat. Res. 2013;15(3):322–331. doi: 10.1111/j.1708-8208.2011.00352.x. [DOI] [PubMed] [Google Scholar]
- Glauser R., Ree A., Lundgren A., Gottlow J., Hammerle C.H., Scharer P. Immediate occlusal loading of Brånemark implants applied in various jawbone regions: a prospective, 1-year clinical study. Clin. Implant Dent. Relat. Res. 2001;3(4):204–213. doi: 10.1111/j.1708-8208.2001.tb00142.x. [DOI] [PubMed] [Google Scholar]
- Glauser R., Lundgren A., Gottlow J., Sennerby L., Portmann M., Ruhstaller P., Hammerle C.H. Immediate occlusal loading of Brånemark TiUnite™ implants placed predominantly in soft bone: 1-year results of a prospective clinical study. Clin. Implant Dent. Relat. Res. 2003;5:47–56. doi: 10.1111/j.1708-8208.2003.tb00015.x. [DOI] [PubMed] [Google Scholar]
- Glibert M., Vervaeke S., De Bruyn H., Östman P.O. Clinical and radiographic comparison between platform-shifted and non-platform-shifted implant: a one-year prospective study. Clin. Implant Dent. Relat. Res. 2016;18(1):129–137. doi: 10.1111/cid.12269. [DOI] [PubMed] [Google Scholar]
- Göçmen G., Atalı O., Aktop S., Sipahi A., Gönül O. Hyaluronic acid versus ultrasonic resorbable pin fixation for space maintenance in non-grafted sinus lifting. J. Oral Maxillofac. Surg. 2016;74(3):497–504. doi: 10.1016/j.joms.2015.10.024. [DOI] [PubMed] [Google Scholar]
- Goiato M.C., Dos Santos D.M., Santiago J.J., Moreno A., Pellizzer E.P. Longevity of dental implants in type IV bone: a systematic review. Int. J. Oral Maxillofac. Surg. 2014;43(9):1108–1116. doi: 10.1016/j.ijom.2014.02.016. [DOI] [PubMed] [Google Scholar]
- Grandi T., Garuti G., Samarani R., Guazzi P., Forabosco A. Immediate loading of single post-extractive implants in the anterior maxilla: 12-month results from a multicenter clinical study. J. Oral Implantol. 2012;38(S1):477–484. doi: 10.1563/AAID-JOI-D-11-00058. [DOI] [PubMed] [Google Scholar]
- Guarnieri R., Di Nardo D., Di Giorgio G., Miccoli G., Testarelli L. Evaluation of peri-implant tissues condition after 10–15 years of loading in treated chronic periodontitis patients attending a private practice setting: A retrospective study. Clin. Oral Implant Res. 2021;32(4):422–436. doi: 10.1111/clr.13712. [DOI] [PubMed] [Google Scholar]
- Han H.J., Kim S., Han D.H. Multifactorial evaluation of implant failure: a 19-year retrospective study. Int. J. Oral Maxillofac. Implants. 2014;29(2) doi: 10.11607/jomi.2869. [DOI] [PubMed] [Google Scholar]
- Herrmann I., Lekholm U., Holm S., Kultje C. Evaluation of patient and implant characteristics as potential prognostic factors for oral implant failures. Int. J. Oral Maxillofac. Implants. 2005;20(2) [PubMed] [Google Scholar]
- Higgins D.G., Altman P.C., Gøtzsche P., Jüni D., Moher A.D., Oxman J., Savovic K.F., Schulz L., Weeks J.A. Sterne, Cochrane Bias Methods Group, Cochrane Statistical Methods Group, The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343 doi: 10.1136/bmj.d5928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hingsammer L., Watzek G., Pommer B. The influence of crown-to-implant ratio on marginal bone levels around splinted short dental implants: A radiological and clinical short term analysis. Clin. Implant Dent. Relat. Res. 2017;19(6):1090–1098. doi: 10.1111/cid.12546. [DOI] [PubMed] [Google Scholar]
- Huang Y.M., Chou I., Jiang C.P., Wu Y.S., Lee S.Y. Finite element analysis of dental implant neck effects on primary stability and osseointegration in a type IV bone mandible. Biomed. Mater. Eng. 2014;24(1):1407–1415. doi: 10.3233/BME-130945. [DOI] [PubMed] [Google Scholar]
- Jaffin R.A., Berman C.L. The excessive loss of Branemark fixtures in type IV bone: A 5-year analysis. J Periodontology. 1991;62:2–4. doi: 10.1902/jop.1991.62.1.2. [DOI] [PubMed] [Google Scholar]
- Jang H.W., Kang J.K., Lee K., Lee Y.S., Park P.K. A retrospective study on related factors affecting the survival rate of dental implants. J. Adv. Prosthodont. 2011;3(4):204–215. doi: 10.4047/jap.2011.3.4.204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeong M.A., Jung M.K., Kim S.G., Oh J.S. Implant stability measurements in the long-term follow-up of dentis implants: a retrospective study with periotest. Implant Dent. 2015;24(3):263–266. doi: 10.1097/ID.0000000000000239. [DOI] [PubMed] [Google Scholar]
- Johansson B., Friberg B., Nilson H. Digitally planned, immediately loaded dental implants with prefabricated prostheses in the reconstruction of edentulous maxillae: a 1-year prospective, multicenter study. Clin. Implant Dent. Relat. Res. 2009;11(3):194–200. doi: 10.1111/j.1708-8208.2008.00111.x. [DOI] [PubMed] [Google Scholar]
- M.A. Kate S. Palaskar P. Kapoor Implant failure: A dentist's nightmare. 6 2016 [cited 2021 Jan 6]; 51 –6 J Dent Implant [serial online]Available from: https://www.jdionline.org/text.asp?2016/6/2/51/202154.
- Khang W., Feldman S., Hawley C.E., Gunsolley J. A multi-center study comparing dual acid-etched and machined-surfaced implants in various bone qualities. J. Periodontol. 2001;72(10):1384–1390. doi: 10.1902/jop.2001.72.10.1384. [DOI] [PubMed] [Google Scholar]
- Kim J.H., Lim Y.J., Kim B., Lee J. How do parameters of implant primary stability correspond with CT-Evaluated Bone quality in the posterior maxilla? The correlation analysis. Materials. 2021;14(2):270. doi: 10.3390/ma14020270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- M. King, E., & Schofield, J. (2023). Restoratively driven planning for implants in the posterior maxilla-Part 1: alveolar bone healing, bone assessment and clinical classifications. British Dental Journal, 235(8), 585-592. [DOI] [PMC free article] [PubMed]
- Koudougou C., Bertin H., Lecaplain B., Badran Z., Longis J., Corre P., Hoornaert A. Post-implantation radiotherapy in patients with head and neck cancer: Literature review. Head Neck. 2020;42(4):794–802. doi: 10.1002/hed.26065. [DOI] [PubMed] [Google Scholar]
- Kumar V.V., Kumar U., Pillai V., Ponnusamy V., Al-Nawas B., Kuriakose M.A. Implant stability and bone characteristics in free fibula flaps used for jaw reconstruction: A prospective cohort study. Int. J. Oral Maxillofac. Implants. 2017;32(5) doi: 10.11607/jomi.5359. [DOI] [PubMed] [Google Scholar]
- Lai H.C., Si M.S., Zhuang L.F., Shen H., Liu Y.L., Wismeijer D. Long-term outcomes of short dental implants supporting single crowns in posterior region: a clinical retrospective study of 5–10 years. Clin. Oral Implant Res. 2013;24(2):230–237. doi: 10.1111/j.1600-0501.2012.02452.x. [DOI] [PubMed] [Google Scholar]
- Lekholm U.L.F., Gunne J., Henry P., Higuchi K., Lindén U., Bergström C., Van Steenberghe D. Survival of the Brånemark implant in partially edentulous jaws: a 10-year prospective multicenter study. Int. J. Oral Maxillofac. Implants. 1999;14(5):639–645. [PubMed] [Google Scholar]
- Lekholm, U. (1985). Tissue-integrated prostheses. Osseointegration in clinical dentistry.
- Linetskiy I., Demenko V., Linetska L., Yefremov O. Impact of annual bone loss and different bone quality on dental implant success–a finite element study. Comput. Biol. Med. 2017;91:318–325. doi: 10.1016/j.compbiomed.2017.09.016. [DOI] [PubMed] [Google Scholar]
- Maiorana C., Farronato D., Pieroni S., Cicciu M., Andreoni D., Santoro F. A four-year survival rate multicenter prospective clinical study on 377 implants: correlations between implant insertion torque, diameter, and bone quality. J. Oral Implantol. 2015;41(3):e60–e65. doi: 10.1563/AAID-JOI-D-13-00206. [DOI] [PubMed] [Google Scholar]
- Maló P., Friberg B., Polizzi G., Gualini F., Vighagen T., Rangert B. Immediate and early function of Brånemark System® implants placed in the esthetic zone: a 1-year prospective clinical multicenter study. Clin. Implant Dent. Relat. Res. 2003;5:37–46. doi: 10.1111/j.1708-8208.2003.tb00014.x. [DOI] [PubMed] [Google Scholar]
- Mangano F., Macchi A., Caprioglio A., Sammons R.L., Piattelli A., Mangano C. Survival and complication rates of fixed restorations supported by locking-taper implants: a prospective study with 1 to 10 years of follow-up. J. Prosthodont. 2014;23(6):434–444. doi: 10.1111/jopr.12152. [DOI] [PubMed] [Google Scholar]
- Mangano F., Shibli J.A., Sammons R.L., Veronesi G., Piattelli A., Mangano C. Clinical outcome of narrow-diameter (3.3-mm) locking-taper implants: a prospective study with 1 to 10 years of follow-up. Int. J. Oral Maxillofac. Implants. 2014;29(2) doi: 10.11607/jomi.3327. [DOI] [PubMed] [Google Scholar]
- Mangano F.G., Shibli J.A., Sammons R.L., Iaculli F., Piattelli A., Mangano C. Short (8-mm) locking-taper implants supporting single crowns in posterior region: a prospective clinical study with 1-to 10-years of follow-up. Clin. Oral Implant Res. 2014;25(8):933–940. doi: 10.1111/clr.12181. [DOI] [PubMed] [Google Scholar]
- Mohajerani H., Roozbayani R., Taherian S., Tabrizi R. The risk factors in early failure of dental implants: a retrospective study. J. Dent. 2017;18(4):298. [PMC free article] [PubMed] [Google Scholar]
- Mombelli A., Cionca N. Systemic diseases that affect osseointegration therapy. Clin. Oral Implant Res. 2006;17(S2):97–103. doi: 10.1111/j.1600-0501.2006.01354.x. [DOI] [PubMed] [Google Scholar]
- Montero J., López-Valverde A., de Diego R.G. A retrospective study of the risk factors for ridge expansion with self-tapping osteotomes in dental implant surgery. Int. J. Oral Maxillofac. Implants. 2012;27(1) [PubMed] [Google Scholar]
- Morris H.F., Winkler S., Ochi S., Kanaan A. A new implant designed to maximize contact with trabecular bone: Survival to 18 months. J. Oral Implantol. 2001;27(4):164–173. doi: 10.1563/1548-1336(2001)027<0164:ANIDTM>2.3.CO;2. [DOI] [PubMed] [Google Scholar]
- Muelas-Jiménez M.I., Olmedo-Gaya M.V., Manzano-Moreno F.J., Reyes-Botella C., Vallecillo-Capilla M. Long-term survival of dental implants with different prosthetic loading times in healthy patients: a 5-year retrospective clinical study. J. Prosthodont. 2017;26(2):99–106. doi: 10.1111/jopr.12371. [DOI] [PubMed] [Google Scholar]
- Nappo A., Rengo C., Pantaleo G., Spagnuolo G., Ferrari M. Influence of implant dimensions and position on implant stability: A prospective clinical study in maxilla using resonance frequency analysis. Appl. Sci. 2019;9(5):860. [Google Scholar]
- Noguerol B., Muñoz R., Mesa F., de Dios Luna J., O'Valle F. Early implant failure. Prognostic capacity of Periotest®: retrospective study of a large sample. Clin. Oral Implant Res. 2006;17(4):459–464. doi: 10.1111/j.1600-0501.2006.01250.x. [DOI] [PubMed] [Google Scholar]
- Olmedo-Gaya, MV, Manzano-Moreno, FJ, Cañaveral-Cavero, E., de Dios Luna-del Castillo, J., & Vallecillo-Capilla, M. (2016). Risk factors associated with early implant failure: A 5-year retrospective clinical study. The Journal of prosthetic dentistry, 115(2), 150-155. [DOI] [PubMed]
- Orenstein I.H., Tarnow D.P., Morris H.F., Ochi S. Three-year post-placement survival of mobile implants at placement. Ann. Periodontol. 2000;5(1):32–41. doi: 10.1902/annals.2000.5.1.32. [DOI] [PubMed] [Google Scholar]
- Östman P.O., Wennerberg A., Albrektsson T. Immediate occlusal loading of NanoTite™ PREVAIL® implants: a prospective 1-year clinical and radiographic study. Clin. Implant Dent. Relat. Res. 2010;12(1):39–47. doi: 10.1111/j.1708-8208.2008.00128.x. [DOI] [PubMed] [Google Scholar]
- Östman P.O., Wennerberg A., Ekestubbe A., Albrektsson T. Immediate occlusal loading of NanoTite™ tapered implants: a prospective 1-year clinical and radiographic study. Clin. Implant Dent. Relat. Res. 2012;15(6):809–818. doi: 10.1111/j.1708-8208.2011.00437.x. [DOI] [PubMed] [Google Scholar]
- Ottoni J.M.P., Oliveira Z.F.L., Mansini R., Cabral A.M. Correlation between placement torque and survival of single-tooth implants. Int. J. Oral Maxillofac. Implants. 2005;20(5) [PubMed] [Google Scholar]
- Polizzi G., Grunder U., Goené R., Hatano N., Henry P., Jackson W.J., Lithner B. Immediate and delayed implant placement into extraction sockets: a 5-year report. Clin. Implant Dent. Relat. Res. 2000;2(2):93–99. doi: 10.1111/j.1708-8208.2000.tb00111.x. [DOI] [PubMed] [Google Scholar]
- Raghoebar G.M., Friberg B., Grunert I., Hobkirk J.A., Tepper G., Wendelhag I. 3-year prospective multicenter study on one-stage implant surgery and early loading in the edentulous mandible. Clin. Implant Dent. Relat. Res. 2003;5(1):39–46. doi: 10.1111/j.1708-8208.2003.tb00180.x. [DOI] [PubMed] [Google Scholar]
- Raikar S, Talukdar P, Kumari S, Panda SK, Oommen VM, Prasad A. Factors Affecting the Survival Rate of Dental Implants: A Retrospective Study. J Int Soc Prev Community Dent. 2017 Nov-Dec ;7 (6):351-355. doi : 10.4103/jispcd.JISPCD_380_17. Epub 2017 Dec 29. PMID: 29387619; PMCID: PMC5774056. [DOI] [PMC free article] [PubMed]
- Ramamoorthi M., Narvekar A., Esfandiari S. A meta-analysis of retention systems for implant-supported prostheses in partially edentulous jaws. J. Prosthet. Dent. 2017;118(5):587–595. doi: 10.1016/j.prosdent.2016.11.019. [DOI] [PubMed] [Google Scholar]
- Rocci A., Martignoni M., Gottlow J. Immediate loading of Brånemark System® TiUnite™ and machined-surface implants in the posterior mandible: a randomized open-ended clinical trial. Clin. Implant Dent. Relat. Res. 2003;5:57–63. doi: 10.1111/j.1708-8208.2003.tb00016.x. [DOI] [PubMed] [Google Scholar]
- Romanos G.E., Ciornei G., Jucan A., Malmstrom H., Gupta B. In vitro assessment of primary stability of Straumann® implant designs. Clin. Implant Dent. Relat. Res. 2014;16(1):89–95. doi: 10.1111/j.1708-8208.2012.00464.x. [DOI] [PubMed] [Google Scholar]
- Romanos G., Grizas E., Laukart E., Nentwig G.H. Effects of early moderate loading on implant stability: a retrospective investigation of 634 implants with platform switching and Morse-tapered connections. Clin. Implant Dent. Relat. Res. 2016;18(2):301–309. doi: 10.1111/cid.12314. [DOI] [PubMed] [Google Scholar]
- Romanos G.E., Lau J., Delgado-Ruiz R., Javed F. Primary stability of narrow-diameter dental implants with a multiple condensing thread design placed in bone with and without osteotomes: An in vitro study. Clin. Implant Dent. Relat. Res. 2020 doi: 10.1111/cid.12908. [DOI] [PubMed] [Google Scholar]
- Romeo E., Lops D., Amorfini L., Chiapasco M., Ghisolfi M., Vogel G. Clinical and radiographic evaluation of small-diameter (3.3-mm) implants followed for 1–7 years: a longitudinal study. Clin. Oral Implant Res. 2006;17(2):139–148. doi: 10.1111/j.1600-0501.2005.01191.x. [DOI] [PubMed] [Google Scholar]
- Salimov F., Tatli U., Kürkçü M., Akoğlan M., Öztunç H., Kurtoğlu C. Evaluation of relationship between preoperative bone density values derived from cone beam computed tomography and implant stability parameters: a clinical study. Clin. Oral Implant Res. 2014;25(9):1016–1021. doi: 10.1111/clr.12219. [DOI] [PubMed] [Google Scholar]
- Sargolzaie N., Samizade S., Arab H., Ghanbari H., Khodadadifard L., Khajavi A. The evaluation of implant stability measured by resonance frequency analysis in different bone types. J. Korean Assoc. Oral Maxillofac. Surg. 2019;45(1):29–33. doi: 10.5125/jkaoms.2019.45.1.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sekar V., et al. Implant considerations and surface modifications - A literature review. Drug Invention Today. 2019;11:1. [Google Scholar]
- Sennerby L., Andersson P., Verrocchi D., Viinamäki R. One-year outcomes of Neoss bimodal implants. A prospective clinical, radiographic, and RFA study. Clin. Implant Dent. Relat. Res. 2012;14(3):313–320. doi: 10.1111/j.1708-8208.2010.00273.x. [DOI] [PubMed] [Google Scholar]
- Shin S.I., Yun J.H., Kim S.G., Park B., Herr Y., Chung J.H. Survival of 352 titanium implants placed in 181 patients: a 4-year multicenter field study. J. Periodont. Implant Sci. 2014;44(1):8–12. doi: 10.5051/jpis.2014.44.1.8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shivu M.E., Reddy N., Swetha Vempalli A.M., Kaur A., Jagadeesh K.N. Evaluation of risk factors for dental implants failure. Int. J. Cur. Res. Rev. 2021;13(05):39. [Google Scholar]
- Siddiqui A.A., O'Neal R., Nummikoski P., Pituch D., Ochs M., Huber H., Wang I.C. Immediate loading of single-tooth restorations: one-year prospective results. J. Oral Implantol. 2008;34(4):208–218. doi: 10.1563/0.926.1. [DOI] [PubMed] [Google Scholar]
- Simmons D.E., Palaiologou A., Teitelbaum A.G., Billiot S., Popat L.J., Maney P. Immediate and early loading of hydrothermally treated, hydroxyapatite-coated dental implants: 2-year results from a prospective clinical study. J. Oral Implantol. 2016;42(1):17–25. doi: 10.1563/aaid-joi-D-14-00283. [DOI] [PubMed] [Google Scholar]
- Singh R., Parihar A.S., Vaibhav V., Kumar K., Singh R., Jerry J.J. A 10 years retrospective study of assessment of prevalence and risk factors of dental implant failures. J. Family Med. Primary Care. 2020;9(3):1617. doi: 10.4103/jfmpc.jfmpc_1171_19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- R. Smeets, B. Stadlinger, F Schwarz et al., “Impact of dental implant surface modifications on osseointegration,” Biomed Res Int, vol. 2016, Article ID 6285620, 16 pages, 2016. [DOI] [PMC free article] [PubMed]
- Staedt H., Rossa M., Lehmann K.M., Al-Nawas B., Kämmerer P.W., Heimes D. Potential risk factors for early and late dental implant failure: a retrospective clinical study on 9080 implants. Int. J. Implant Dent. 2020;6(1):1–10. doi: 10.1186/s40729-020-00276-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sykaras N., Iacopino A.M., Marker V.A., Triplett R.G., Woody R.D. Implant materials, designs, and surface topographies: Their effect on osseointegration. A literature review. Int. J. Oral Maxillofac. Implants. 2000;15:675–690. [PubMed] [Google Scholar]
- A. Tabassum G.J. Meijer X.F. Walboomers J.A. Jansen Evaluation of primary and secondary stability of titanium implants using different surgical techniques Clinical Oral Implants Research 25 4 2013 487 492 10.1111 /clr.12180. [DOI] [PubMed]
- Tawil G., Mawla M. Sinus floor elevation using a bovine bone mineral (Bio-Oss) with or without the concomitant use of a bilayered collagen barrier (Bio-Gide): a clinical report of immediate and delayed implant placement. Int. J. Oral Maxillofac. Implants. 2001;16(5) [PubMed] [Google Scholar]
- Tawil G., Younan R. Clinical evaluation of short, machined-surface implants followed for 12 to 92 months. Int. J. Oral Maxillofac. Implants. 2003;18(6) [PubMed] [Google Scholar]
- Thomé G., Cartelli C.A., Vianna C.P., Trojan L.C. Retrospective clinical study of 453 novel tapered implants placed in all bone types: Survival rate analysis up to 2 years of follow-up. Int. J. Oral Maxillofac. Implants. 2020;35(4) doi: 10.11607/jomi.8083. [DOI] [PubMed] [Google Scholar]
- Thomková, B., Marcián, P., Borák, L., Joukal, M., & Wolff, J. (2023). Biomechanical performance of dental implants inserted in different mandible locations and at different angles: A finite element study. The Journal of Prosthetic Dentistry. [DOI] [PubMed]
- Toia M., Stocchero M., Cecchinato F., Corrà E., Jimbo R., Cecchinato D. Clinical considerations of adapted drilling protocol by bone quality perception. Int. J. Oral Maxillofac. Implants. 2017;32(6) doi: 10.11607/jomi.5881. [DOI] [PubMed] [Google Scholar]
- Trbakovic A., Toljanic J.A., Kumar V.V., Thor A. Eight to eleven-year follow-up of immediately loaded implants placed in edentulous maxillae with compromised bone volume and poor bone quality: A prospective cohort study. Clin. Implant Dent. Relat. Res. 2020;22(1):69–76. doi: 10.1111/cid.12874. [DOI] [PubMed] [Google Scholar]
- Triches D.F., Alonso F.R., Mezzomo L.A., Schneider D.R., Villarinho E.A., Rockenbach M.I., Shinkai R.S. Relation between insertion torque and tactile, visual, and rescaled gray value measures of bone quality: a cross-sectional clinical study with short implants. Int. J. Implant Dent. 2019;5(1):1–8. doi: 10.1186/s40729-019-0158-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Truhlar R.S., Morris H.F., Ochi S. Implant surface coating and bone quality-related survival outcomes through 36 months post-placement of root-form endosseous dental implants. Ann. Periodontol. 2000;5(1):109–118. doi: 10.1902/annals.2000.5.1.109. [DOI] [PubMed] [Google Scholar]
- Turner P.S., Nentwig G.H. Evaluation of the value of bone training (progressive bone loading) by using the Periotest: A clinical study. Contemp. Clin. Dent. 2014;5(4):461. doi: 10.4103/0976-237X.142811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vasak C., Kohal R.J., Lettner S., Rohner D., Zechner W. Clinical and radiological evaluation of a template-guided (N obelGuide™) treatment concept. Clin. Oral Implant Res. 2014;25(1):116–123. doi: 10.1111/clr.12038. [DOI] [PubMed] [Google Scholar]
- Waechter J., Madruga M.D.M., Carmo Filho L.C.D., Leite F.R.M., Schinestsck A.R., Faot F. Comparison between tapered and cylindrical implants in the posterior regions of the mandible: A prospective, randomized, split-mouth clinical trial focusing on implant stability changes during early healing. Clin. Implant Dent. Relat. Res. 2017;19(4):733–741. doi: 10.1111/cid.12501. [DOI] [PubMed] [Google Scholar]
- Wang Y., Fan Y., Lin Z., Song Z., Shu R., Xie Y. Survival rate and potential risk indicators of implant loss in non-smokers and systemically healthy periodontitis patients: An up to 9-year retrospective study. J. Periodont. Res. 2021 doi: 10.1111/jre.12854. [DOI] [PubMed] [Google Scholar]
- Wang F., Monje A., Huang W., Zhang Z., Wang G., Wu Y. Maxillary Four Implant-retained Overdentures via Locator® Attachment: Intermediate-term Results from a Retrospective Study. Clin. Implant Dent. Relat. Res. 2016;18(3):571–579. doi: 10.1111/cid.12335. [DOI] [PubMed] [Google Scholar]
- Widmark G., Friberg B., Johansson B., Sindet-Pedersen S., Taylor Å. Mk III: A third generation of the self-tapping Brånemark System® implant, including the new stargrip internal grip design. A 1-year prospective four-center study. Clin. Implant Dent. Relat. Res. 2003;5(4):273–279. doi: 10.1111/j.1708-8208.2003.tb00211.x. [DOI] [PubMed] [Google Scholar]
- Zinser M.J., Randelzhofer P., Kuiper L., Zöller J.E., De Lange G.L. The predictors of implant failure after maxillary sinus floor augmentation and reconstruction: a retrospective study of 1045 consecutive implants. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2013;115(5):571–582. doi: 10.1016/j.oooo.2012.06.015. [DOI] [PubMed] [Google Scholar]
- Zumstein T., Schütz S., Sahlin H., Sennerby L. Factors influencing marginal bone loss at a hydrophilic implant design placed with or without GBR procedures: A 5-year retrospective study. Clin. Implant Dent. Relat. Res. 2019;21(5):817–826. doi: 10.1111/cid.12826. [DOI] [PubMed] [Google Scholar]