Abstract
Spinal cord injury is considered one of the most difficult injuries to repair and has one of the worst prognoses for injuries to the nervous system. Following surgery, the poor regenerative capacity of nerve cells and the generation of new scars can make it very difficult for the impaired nervous system to restore its neural functionality. Traditional treatments can only alleviate secondary injuries but cannot fundamentally repair the spinal cord. Consequently, there is a critical need to develop new treatments to promote functional repair after spinal cord injury. Over recent years, there have been several developments in the use of stem cell therapy for the treatment of spinal cord injury. Alongside significant developments in the field of tissue engineering, three-dimensional bioprinting technology has become a hot research topic due to its ability to accurately print complex structures. This led to the loading of three-dimensional bioprinting scaffolds which provided precise cell localization. These three-dimensional bioprinting scaffolds could repair damaged neural circuits and had the potential to repair the damaged spinal cord. In this review, we discuss the mechanisms underlying simple stem cell therapy, the application of different types of stem cells for the treatment of spinal cord injury, and the different manufacturing methods for three-dimensional bioprinting scaffolds. In particular, we focus on the development of three-dimensional bioprinting scaffolds for the treatment of spinal cord injury.
Keywords: biomaterials, embryonic stem cells, induced pluripotent stem cells, mesenchymal stem cells, nerve regeneration, spinal cord injury, stem cell therapy, stem cells, three-dimensional bioprinting
Introduction
Spinal cord injury (SCI) is a traumatic injury that causes impaired mobility, pain, and autonomic dysfunction. Ultimately, SCI can severely impair a patient's quality of life, social status, and independence (Fehlings et al., 2017). SCI is primarily caused by serious trauma such as traffic accidents, extreme sports, and violence. Approximately one million people worldwide are now suffering from paralysis as a direct result of SCI. Although SCI is highly prevalent in young males, some previous reports indicate that the incidence of SCI is rising in the elderly population (Eckert and Martin, 2017).
The mechanism of SCI consists of two phases: primary injury and secondary injury. Usually, a traumatic accident may lead to primary spinal cord damage via contusion, tearing, or compression (Yuan et al., 2022). This is followed by the consequential damage response, including inflammation and apoptosis, vasospasm bleeding, the formation of reactive oxygen species, and lipid peroxidation; this process is referred to as secondary injury (Stenudd et al., 2015). The traditional treatment for SCI consists of medication treatment, surgery, physical therapy, and hyperbaric oxygen therapy. However, to a certain extent, these treatments can only alleviate a patient's symptoms and cannot promote regeneration of the spinal cord (Fehlings et al., 2017). Consequently, it is vital that we develop new techniques to treat SCI.
With the development of technological advances over time, stem cell (SC) therapy for SCI has become a significant focus of research. Due to their own regenerative properties, the transplantation of SCs is considered to represent a promising strategy for regenerating inactive cells or tissues that have been damaged by SCI (Muheremu et al., 2016). SCs possess several key capabilities, including self-renewal, directed differentiation, the establishment of new synapses, and the release of various neurotrophic factors, thus providing a microenvironment that promotes repair following SCI (Lv et al., 2021).
With the emergence of new biomaterials and the development and application of three-dimensional (3D) printing technology in various fields, a new and unique 3D printing technology has emerged; this is referred to as 3D bioprinting. This technology is now being used to create cellular patterns that can exhibit good cell viability and functionality. Considering key factors such as target tissue type, complexity, and tissue function, 3D bioprinting can construct scaffolds that adopt a morphology that is similar to that of living tissues. For complex types of tissues, such as the spinal cord, strategies that utilize the combined application of cells and 3D bioscaffolds are not uncommon (Bishop et al., 2017).
There are still many obstacles to traditional SC therapy; for example, transplanted cells in a damaged region of the spinal cord have a low rate of survival or can be lost in the cerebrospinal fluid (Tahmasebi and Barati, 2022). In contrast, 3D bioprinting scaffolds may promote the survival of SCs, reduce the loss of cells, and even promote neural reattachment. Therefore, a combined therapy consisting of 3D bioprinting scaffolds and SCs represents a highly promising option (Liu et al., 2022). Generally, this combined therapy could serve as a supportive platform to fill gaps in a lesion created by SCI, bridging the broken ends of an injury and promoting axonal regeneration (Ahuja and Fehlings, 2016). Furthermore, as a carrier of seed cells, a scaffold with a 3D porous structure provides a highly suitable platform for the growth and proliferation of cells, thus providing nutrients (such as oxygen and other useful substances) and promoting metabolism; consequently, seeded scaffolds can create a suitable microenvironment for the repair of SCI (Xu et al., 2017; Ahi et al., 2020; Liu et al., 2021a). Research has shown that combined therapy can exert anti-inflammatory effects by inhibiting the activation of microglia or macrophages (Fan et al., 2018).
The main remit of this review is to discuss the combination of SCs and 3D bioprinting scaffolds for the repair of SCI. We discuss the mechanisms underlying simple SC therapy, different types of SCs in the treatment of SCI, and the different methods used to fabricate 3D bioprinting scaffolds. We also consider the existing literature relating to SCI regeneration.
Retrieval Strategy
All the cited articles were written in English, available as full-text articles and were published between 2011 and 2023. The studies cited in this review were mainly conducted in mammals such as rodents and humans. We searched the PubMed database with specific keywords, including (1) SCI, (2) SCs, and (3) 3D bioprinting, but also included other key words. We screened the representative literature related to SCI and clarified various aspects in combination with relevant references.
Stem Cell Therapy
A recent clinical trial has shown that SC therapy can exert beneficial effects and has significant potential for repairing damaged tissue in the spinal cord and promoting the restoration of normal functionality in the nervous system (Yamazaki et al., 2020).
The methods used for cell transplantation, including cell type, dose, method, and time of transplantation, vary widely between studies; consequently, there is no consensus on the most effective treatment strategy. According to their different stages of development, SCs can be divided into adult SCs (ASCs), embryonic SCs (ESCs), and induced pluripotent SCs (iPSCs); ESCs are isolated from blastocyte clusters. ASCs can be divided into mesenchymal SCs (MSCs), hematopoietic SCs (HSCs), and neural SCs (NSCs); these can be isolated from various adult tissues. However, the efficacy and safety of SC therapy in the treatment of SCI remains controversial (Gao et al., 2020; Yamazaki et al., 2020).
Potential options for the treatment of SCI with SCs
Tissue repair and replacement
Translocated SCs can differentiate into neurons and glial cells when stimulated by the local microenvironment and different types of nerve growth factors (Cao et al., 2022; Zhang et al., 2022; Li et al., 2023; Sun et al., 2023), thus initiating the repair and replacement process following SCI (Fan et al., 2022a, b). Previous work demonstrated that after the delayed transplantation NSCs into lesion sites in the spinal cord, NSCs can survive and differentiate into a variety of cell types including neurons, astrocytes, and oligodendrocytes; this strategy reduced injury and significantly improved voluntary motor ability when compared to the control group (Muheremu et al., 2016).
Neurotrophic and regenerative effects
A growing number of findings suggest that transplanted NSCs can release neurotrophic factors that can facilitate the treatment of SCI. Neurotrophicity and neural regeneration are regarded as important factors in the treatment of SCI (Duan et al., 2016). A previous study transplanted SCs into an in vitro model of SCI and then administered valproic acid to improve the harsh microenvironment of fracture and damage. The expression levels of regulatory factors were significantly increased in the NSC-treated group 7 days after injury, including glial fibrillary acidic protein, neurotrophin-3, Bcl-2, and brain-derived neurotrophic factor (BDNF), thus suggesting that NSCs can ameliorate craniocerebral injury by directly releasing neurotrophic factors (Pandamooz et al., 2018). In addition, other substances, such as glial cell-derived neurotrophic factor, have also been shown to be beneficial for tissue repair after SCI (Walker and Xu, 2018).
Promotion of angiogenesis
The reconstruction of nerves is not only based on the regeneration of nerve cells but also depends on the support of the local microenvironment including blood vessels and extracellular matrix; in SCI, this microenvironment is damaged; the formation of new blood vessels in this microenvironment is a significant aspect of spinal cord repair. Primary injury of the spinal cord can rupture local blood vessels and has a significant impact on the process of neurodegeneration. Ischemia leads to the emergence of new blood vessels which generates a new blood supply to the injury site and promotes its functional recovery (Yahata et al., 2016). However, these newly established blood vessels do not always provide a sufficient blood supply for axonal recovery. To achieve optimal conditions in the SCI microenvironment, enhancing angiogenesis and improving functional recovery have become a top priority (Tsivelekas et al., 2022). A previous study showed that the transplantation of NSCs promotes angiogenesis and improves limb motor function by inducing the expression of vascular endothelial growth factor (Li et al., 2014). Furthermore, the continuous release of vascular endothelial growth factor and angiopoietin-1 at the site of SCI has been shown to significantly stimulate angiogenesis, nerve regeneration, and nerve recovery (Yu et al., 2016).
Anti-apoptotic effects
Reducing neuronal apoptosis during the early stages of injury can contribute to the survival of neurons and tissues, thus allowing the recovery of neurological function (Nicola et al., 2017). Previous experimental data showed that pro-apoptotic proteins, such as p53, caspase-9, caspase-3, and Bax, were significantly down-regulated in an SC-treated group while anti-apoptotic proteins, such as Bcl-2, were significantly upregulated (Wang et al., 2016a, b; Han et al., 2017). Other research showed that an SC transplantation group expressed low levels of the pro-apoptotic factor tumor necrosis factor-α (TNF-α) but high levels of the anti-apoptotic factor Bcl-xL (Nicola et al., 2017). A previous study by Chen et al. (2015) confirmed that the extent of apoptosis may increase after SCI and that estrogen may reduce apoptosis after SCI by playing a neuroprotective role through G-protein coupled receptor 30. By further investigating the mechanism underlying this effect, human phosphoinositide-3 kinase/Akt was identified to be one of the anti-apoptotic pathways of estrogen that acts via G-protein coupled receptor 30 (Chen et al., 2015).
Anti-inflammatory effects
SC transplantation can act synergistically with other related mechanisms to exert an anti-inflammatory role and promote the repair of tissue function after SCI (Cheng et al., 2017; Ross et al., 2017). Indeed, SCI can activate innate immune cells and infiltrate white blood cells, triggering a full-blown inflammatory cascade. Along with the release of pro-inflammatory cytokines, chemokines, excitotoxic amino acids, free radicals, and nitric oxide, these neurotoxins can cause damage to axons and neurons (Lv et al., 2021). Some authors have reported a significant reduction in the number of neutrophils at the site of injury following NSC transplantation (Cheng et al., 2016). These data imply that by inhibiting the activation of M1 macrophages or oligodendrocytes and neutrophil infiltration, the inflammatory response induced by SCI can be regulated to improve nerve function after SCI (Cheng et al., 2016; Fan et al., 2018; Figure 1).
Figure 1.
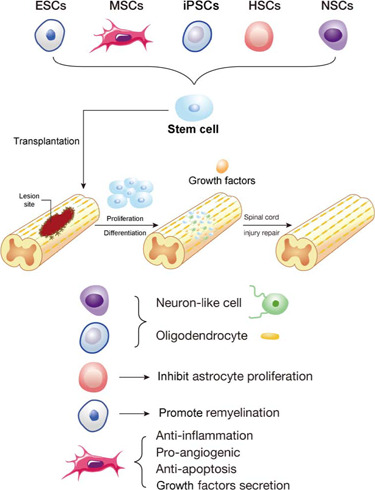
Different stem cells play different primary roles when transplanted to a lesion site in the spinal cord.
NSCs and iPSCs can differentiate into neuron-like cells and oligodendrocytes while HSCs mainly inhibit the proliferation of astrocytes. ESCs have the strongest ability to promote remyelination while MSCs can secrete growth factors and exhibit anti-inflammation, pro-angiogenic and anti-apoptosis properties. Created using Adobe illustrator (version 2017 cc). ESCs: Embryonic stem cells; HSCs: hematopoietic stem cells; iPSCs: induced pluripotent stem cells; MSCs: mesenchymal stem cells; NSCs: neural stem cells.
Different types of SCs are used for the treatment of SCI
ESCs
Several studies have shown that ESC transplantation is effective in the recovery from SCI. Being pluripotent SCs, ESCs can differentiate into new cell types in vivo. For example, NSCs from ESCs can differentiate into neurons and glial cells to replace non-functional cells and tissues (Cui et al., 2011; Lee, 2013). In a previous study, Rossi et al. (2010) transplanted highly purified human motor neuron progenitor cells from ESCs into the lesion sites of SCI and then recorded the survival of neurons, nerve branches, disease reduction, and functional recovery. However, due to the considerable proliferative capacity of ESCs, their pluripotent nature may lead to tumor formation and genetic changes may occur during cell culture. Overall, there is a critical need to optimize protocols for differentiation to reduce tumorigenesis and to control cell populations to adapt to the requirements of different patients with SCI (Marichal et al., 2012).
MSCs
MSCs are widely derived from almost all tissues (including fetuses and adults), including blood, bone marrow, umbilical cord, and placenta. Mesenchymal SCs derived from pluripotent SCs have also become prominent in recent research, including human embryonic SCs and inducible pluripotent SC-derived mesenchymal SCs (Matsukura et al., 2014). In addition, recent studies have investigated the role of exosome-derived mesenchymal SCs as an innovative treatment option for patients with SCI (Schepici et al., 2023). MSCs can secrete growth factors, such as BDNF, vascular endothelial growth factor, and glial cell-derived neurotrophic factor, and these growth factors may help to repair SCI (Forostyak et al., 2013). Research has shown that nerves need to be protected by these growth factors. For example, the neuroprotective effect of BDNF on various neuronal populations after injury is most likely attributable to the downstream effects of TrkB receptor signaling; this form of communication is inhibited by TrkB signaling and involves the peptidase inhibitor 3 kinase/Akt pathways, thus damaged cells to survive. BDNF has also been shown to reduce glutamate-induced apoptosis. In addition to neuronal protection, BDNF can also enhance the regeneration of injured axons and increase the formation of myelin around injured axons. These observations were based on the fact that BDNF induces the upregulation of growth-related genes such as growth associated protein 43 and T-alpha-1-tubulin in neurons. Upon injury, TrkB is highly upregulated in spinal neurons; this makes BDNF the most suitable molecule for spinal neuroprotection. However, high levels of the TrkB receptor can exert negative effects, as off-target effects, such as pain or spasticity, are sometimes associated with BDNF treatment; however, modulation or brief dosing may address this potential problem (Keefe et al., 2017). On the other hand, MSCs can also play a role in promoting the production of anti-inflammatory factors, such as interleukin-1β and TNF-a, thus reducing the inflammatory response (Zhukareva et al., 2010; Wright et al., 2011). A previous study showed that MSCs restored function via neurotrophic and pro-angiogenic effects in thoracic SCI after transplantation (Quertainmont et al., 2012). MSC transplantation is considered a safe approach because MSCs may not have the potential to form teratomas when compared to iPSCs or ESCs. On the other hand, however, MSCs are associated with lower reproducibility. Moreover, other tumor formation and chromosomal aberrations are known to occur in MSCs that are cultured for long periods of time; this emphasizes the importance of identifying an optimal culture time for the transplantation of MSCs (Cantinieaux et al., 2013).
HSCs
The application of HSCs in SCI therapy has become a hot topic over recent years (Bryukhovetskiy and Bryukhovetskiy, 2015). In a previous study, Xiong et al. found that HSCs could form oligodendrocytes and 5-hydroxytryptamine-positive fibers in SCI by injecting HSCs into rat models (Xiong et al., 2017); these HSCs also inhibited the proliferation of astrocytes, thus promoting neurological recovery in experimental rats. Another study used HSCs to treat 20 patients with chronic SCI and intermittant C4–C8 injuries; this was accompanied by the application of somatosensory evoked potentials and motor evoked potentials. The authors found that HSCs spread from the lumbar region to the cervical region to create a neurorestorative effect (Frolov and Bryukhovetskiy, 2012). In another study, the repeated transplantation of HSCs and hematopoietic progenitors led to the recovery of neurological deficits; these effects were stable and significant in 57.4% of cases (Bryukhovetskiy and Bryukhovetskiy, 2015).
NSCs
NSC transplantation uses an effective replacement method and is often used to induce differentiation in damaged cells such as neurons or astrocytes. Moreover, neurotrophic factors, such as growth factors, are secreted to promote axonal growth and form synaptic integration by binding with host neurons in the spinal cord (Lee et al., 2014; Bonner and Steward, 2015; Lane et al., 2017). In a study in which NSCs were transplanted into a hemisected model of SCI, the upregulation of neurotrophic factors was observed along with functional recovery; in addition, there was no tumor formation or other complications (Xiong et al., 2018). Many previous studies have shown that NSCs exert positive effects on neuronal regeneration from many different aspects. Several clinical trials have been carried out recently; however, none of these have generated satisfactory results (Åkesson and Sundström, 2016). In one study, most of the transplanted NSCs differentiated into astrocytes without differentiating into neurons or oligodendrocytes under the influence of the SCI microenvironment; these results question the feasibility of NSC transplantation. Therefore, there is a lack of effective strategies to fully utilize NSCs to treat severe SCI (Kameda et al., 2018).
iPSCs
iPSCs also have pluripotent properties, including proliferation and teratoma formation; they are also amenable to gene labeling and expression studies. The use of iPSCs avoids the problem of the origin of ESCs, such as ethical issues, because iPSCs are generated from cells that have been reprogrammed and isolated from accessible tissues such as autologous skin, thus allowing autologous cell transplantation without rejection. Similar to some other types of SCs, iPSC-derived stem/progenitor cells are known to inhibit demyelination, promote synaptic formation, and secrete neurotrophic factors (Salewski et al., 2013, 2015; Kawabata et al., 2016), thus improving functional recovery after SCI without the formation of tumors (Kobayashi et al., 2012). Contradictory results have also shown that NPCs derived from human iPSCs do not facilitate the treatment of SCI. One study also reported limitations relating to transplantation time (Nutt et al., 2013). In addition, the tumorigenicity and high cost of developing iPSCs have also restricted the development of this strategy for clinical scenarios (López-Serrano et al., 2016; Fischer et al., 2020).
The advantages and disadvantages of different types of SCs for SCI therapy are shown in Table 1.
Table 1.
The advantages and disadvantages of different types of stem cells in spinal cord injury therapy
| Stem cells | Advantages | Disadvantages |
|---|---|---|
| ESCs | Self-renewal | Ethical issues |
| Multipotency | Risk of teratoma and tumor formation | |
| Risk of genetic changes | ||
| Immune rejection | ||
| MSCs | Wide variety of sources | Low reproducibility |
| Low risk of teratoma formation | Risk of other tumor formation | |
| Anti-inflammatory effect | Biological differences of different sources | |
| Pro-angiogenic effect | ||
| Growth factors secretion | ||
| HSCs | Inhibit astrocyte proliferation | |
| Promote the formation of 5-hydroxytryptamine-positive fibers | ||
| Promote oligodendrocytes formation | ||
| NSCs | Self-renewal | Low applicability |
| Secrete neurotrophic factors | High possibility of differentiating into astrocytes | |
| Combine and repair damaged neurons | ||
| Anti-inflammatory effect | ||
| Low risk of tumor formation | ||
| iPSCs | Self-renewal | Risk of teratoma or other tumors formation |
| No ethical issues | High cost | |
| Multipotency | Limitation on transplantation time | |
| No immune rejection | ||
| Allow autologous cell transplantation |
ESCs: Embryonic stem cells; HSCs: hematopoietic stem cells; iPSCs: induced pluripotent stem cells; MSCs: mesenchymal stem cells; NSCs: neural stem cells.
Shortcomings of SC therapy
Although the potential application of SCs for the treatment of SCI has been studied for many years, there are still significant disadvantages, as follows: (i) immune responses to allogeneic or xenologous cells may promote cell rejection, thus jeopardizing the success of treatment; (ii) it will be difficult to colonize the injury site with implanted SCs due to the flow of cerebrospinal fluid, thus leading to poor effects, and (iii) the death of transplanted SCs resulting from ischemia and the inflammatory microenvironment at the injury site. Therefore, to make up for the defects associated with using SCs alone, this method could be combined with 3D bioprinted scaffolds such that SCs can colonize the injury site and encourage the directional regeneration of axons.
Three-dimensional Bioprinted Scaffolds
In recent years, advances in nanotechnology and tissue engineering have promoted the clinical development and treatment of several neurological diseases. The focus of nerve tissue engineering is to improve the local microenvironment at the injury site; this strategy is more conducive to the recovery of nerve tissue function. 3D scaffolds, in turn, provide the physical and chemical support for successful nerve regeneration. Recently, attention has been drawn to 3D bioprinting, a technology that allows the easy adjustment of the shape and size of 3D tissue scaffolds while retaining the ability to produce scaffolds containing cells (Cunha et al., 2011; Joung et al., 2020).
Compared to traditional scaffold manufacturing methods, 3D bioprinting can be seen as a combination of both 3D printing and bioprinting technologies; however, it also features the advantages of two relevant technologies. First, 3D printing allows for a “personalized” manufacturing solution, which allows the shape of the scaffold to be tailored to the specific characteristics of each SCI site, thus achieving maximum natural fidelity, and allowing the printed tissue structure to adapt to the damaged microenvironment. More importantly, 3D printing could provide direction for axonal regeneration, thus guiding the reconstruction of neural pathways in the spinal cord, and ultimately facilitating the formation of neural networks (Kadoya et al., 2016). In addition, bioprinted samples can be formed faster than traditional tissue engineering. This form of in vitro modeling allows for more effective “preoperative simulation” of neurological tissue reconstruction, thus allowing for more accurate injury repair (Johnson et al., 2016). Thus, by combining these two technologies, the 3D bioprinting technique creates the potential for personalized tissue fabrication, thus allowing each SCI patient to have a scaffold that matches his or her own individual injury characteristics.
(i) An ideal neural tissue engineering scaffold should meet the following criteria: (i) biocompatible; encourages cell adhesion, proliferation, and differentiation in the absence of immune and cytotoxic responses; (ii) biodegradable; degrading at a rate that matches the rate of new tissue formation and eventually being removed from the system; (iii) conductive; supporting neural protrusion, growth and nerve regeneration; (iv) similar mechanical properties; a biological scaffold should be designed to conform as closely as possible to the mechanical properties of the spinal cord, ideally at pressure of 10 kPa (Joung et al., 2020) to ensure that it does not increase the stress applied to the area of injury or collapse through regular motion; (v) porous interconnections; imitating the extracellular matrix of natural tissues, encouraging vascularization, cell dispersal and the exchange of nutrients and waste (Subramanian et al., 2009; Cunha et al., 2011), and (vi) appropriate channel diameter; channel diameters greater than 450 μm are known to result in slower nerve regeneration (Joung et al., 2020).
Methods of 3D bioprinting
To create the ideal scaffold, developing appropriate 3D bioprinting technologies are indispensable. There have been several recent developments, including inkjet bioprinting, fused deposition modeling (FDM), stereolithography (SLA), micro-extrusion bioprinting, and 3D bioplotting (Figure 2).
Figure 2.
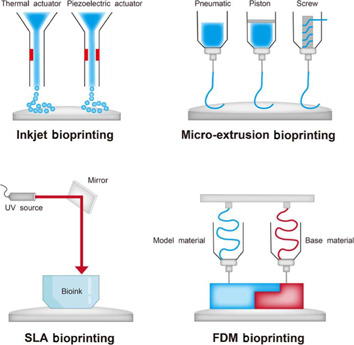
The most prevalent 3D bioprinting methods used for tissue engineering applications.
(A) Inkjet bioprinting; (B) micro-extrusion bioprinting; (C) SLA bioprinting; and (D) FDM bioprinting. Created using Adobe illustrator (version 2017 cc). FDM: Fused deposition modelling; SLA: stereolithography.
Inkjet bioprinting
Inkjet bioprinting is a cost-effective technique that allows for the good dispersion of bioink; this permits the simultaneous contact-free deposition of cells at a micron resolution in several directions (Zheng et al., 2011). Due to the small size of the ink nozzle, the resulting small cumulative volume per drop means that a high concentration of cells must be used to ensure that each bioink drop must contain at least one cell. The main disadvantage of this technique is poor mechanical performance, because the low-viscosity bioink cannot resist external pressure to maintain its original shape after being inserted into the broken end of the spinal cord (O'Brien et al., 2015).
There are now two main types of inkjet printers. The first type is the thermal inkjet printer, which creates pulses of air pressure by heating the printer head; this causes droplets to be ejected from the nozzle. Although this method is fast and has a resolution ranging from 20 μm to 100 μm, it is rarely used in tissue engineering because of the high temperatures involved as these can inactivate macromolecules (Chang et al., 2011; Zheng et al., 2011). The second method is piezoelectric inkjet printing, in which an applied voltage excites a piezoelectric crystal located in the printer head, thus prompting the formation of droplets. Due to this voltage, deformation is both rapid and reversed (Murphy and Atala, 2014). The main advantage of the latter is that the bioink can be dispersed in a controlled manner, independent of pressure and heat; this promotes high levels of cell viability (Cui et al., 2010).
FDM
FDM technology uses fibers made from thermoplastic polymers to build 3D scaffolds. A semi-liquid printed form is obtained by heating the fibers at the nozzle and extruding them onto a platform. This is an easy method to use thermoplastic polymer fiber filaments during the printing process and can be cured at room temperature after the printing is complete (Abas et al., 2022). Moreover, since the FDM method does not require any solvents, it offers convenience in terms of material fabrication and handling. This technique enables continuous and rapid production without changing raw materials. In addition, because of its low processing cost, simple and rapid process, FDM has been used to manufacture biological scaffolds to help repair nerve tissue (O'Brien et al., 2015). However, for SCI, considering the small amount of thermoplastic polymer, poor mechanical and surface properties, this method is not suitable for direct cell printing due to the use of high temperatures; furthermore, it is more appropriate to print the cells before culturing them with the scaffold (Cui et al., 2017).
SLA
Stereolithography methods are performed by focusing an ultraviolet laser on a hydrogel or resin containing a photo-initiator. Since the energy provided by the laser forms covalent bonds between two macromolecular chains, it causes the liquid to solidify (Billiet et al., 2012). SLA printing takes into account a range of different factors, including laser power, time, spot size, and wavelength value; therefore, this method is considered a high resolution form of printing (Melchels et al., 2010). After special steps, including preheating or photocuring, it is possible to achieve many desirable mechanical properties (Zhu et al., 2013). The advantages of SLA include high speed, high resolution, high cell concentration, and no isses related to nozzle clogging. However, there are still some drawbacks that need to be improved, such as high cost and the wide-ranging requirements of the materials used in 3D printing applications (Ozbolat and Hospodiuk, 2016). In addition, the thickness of the material in each layer is determined by the energy generated by the light source and the exposure time of the material; this means that this technology is associated with complex dynamics and reaction processes (Skoog et al., 2014).
Micro-extrusion bioprinting
Micro-extrusion-based bioprinting provides excellent structural integrity to the technology by extruding a continuous bioink. This method allows cells and biomaterials to be combined and the bioink to be distributed through a printer nozzle or needle (Panwar and Tan, 2016). The micro-extrusion head uses instructions from a computer-aided design and manufacturing program to fabricate complex 3D structures, that cause the material to accumulate in the x-y direction in a substrate; the extrusion head then moves along the z-axis (Peltola et al., 2008). The advantage of this method is the wide range of bioinks available; this is due to the use of different micro-nozzle sizes that can accommodate high viscosity bioinks (O'Brien et al., 2015). This technique also includes a fast production speed (Bishop et al., 2017). In addition, the cell survival rate in the biostructures formed by micro-extrusion is reported to be higher than 90% (Seol et al., 2014). In a previous study, Liu et al. (2021b) used a 3D printing system to construct 3D scaffolds at 20 kPa of air pressure by applying micro-extrusion technology. However, this technology is limited by the fact that the pressure applied can affect the feasibility of pressure-based systems. Currently, micro-extrusion technology has been developed and applied to a wide range of tissue engineering structures, including the cardiovascular system and tumor therapy (Skardal et al., 2010; Hsu et al., 2017).
3D bioplotting
In 3D bio-extrusion, a viscous liquid medium is squeezed out of a nozzle drop-by-drop to form a distribution of micro-chains. The longer the exposure time, the higher the degree of curing in the hydrogel material (Gómez-Lizárraga et al., 2017). A 3D bioplotter can print co-cultured scaffolds and tissue junctions (O'Brien et al., 2015). The core technology is the 3D dispersion of viscous printing materials into liquid media with similar densities. The materials used in this technology come from a wide range of sources. In particular, the extrusion and hardening process can ensure that the surface of the support is smooth. Therefore, it can be necessary to modify the roughness to improve the adhesion of cells in the structure of biomarkers (Huang et al., 2016). The advantages and disadvantages associated with different methods of 3D bioprinting are shown in Table 2.
Table 2.
Advantages and disadvantages of different methods of 3D bioprinting
| Methods of 3D bioprinting | Advantages | Disadvantages |
|---|---|---|
| Inkjet bioprinting | High print speed | Low cell viscosity and density |
| High resolution | Relatively low cell viability | |
| Cost-effective technique | Poor mechanical properties | |
| Cell printing ability | Only create simple structures | |
| FDM | No solvents | Poor mechanical properties |
| Low cost | Layer-by-layer appearance | |
| High print speed | Poor surface properties | |
| Simple and continuous production | Limited thermoplastic polymer | |
| Clean material and less pollution | High temperature damage cell | |
| Poor cell printing ability | ||
| SLA | Create highly complex geometrical features | Limitation on bioink |
| No limitation on cell viscosity | Damage to cells during photocuring | |
| No nozzle clogging problems | High cost | |
| High concentration of cells | Low print speed | |
| High speed and resolution | ||
| High surface quality | ||
| Micro-extrusion bioprinting | No limitation on bioink | Poor pressure-based systems |
| High cell viability | ||
| High print speed | ||
| Print very high cell density models | ||
| 3D bioplotting | High accuracy | Low cell adhesion |
3D: Three-dimensional; FDM: fused deposition modelling; SLA: stereolithography.
Materials for manufacturing 3D bioprinting scaffolds
In addition to the manufacturing process, it is necessary to carefully consider the manufacturing materials to achieve the ideal outcome. First, it is necessary to identify soft materials to prevent mechanical mismatches. The most widely used materials are hydrogels, as these are associated with good biocompatibility and predictable biodegradability (Huang et al., 2017). Furthermore, most natural hydrogels have specific cell binding sites that provide the necessary conditions for cell attachment, promote growth, proliferation, and differentiation, and have the ability to support axonal growth and ultimately fill the cavity in the spinal cord (Gungor-Ozkerim et al., 2018). The specific structure of hydrogels can also facilitate the exchange of gases and various nutrients. Furthermore, hydrogels are water-swollen cross-linked polymer networks that can mimic the mechanical structure of the soft spinal cord tissue, at least to some extent (Zhu and Marchant, 2011). In addition, due to its high water content and non-toxicity, hydrogels are capable of forming an analog of extracellular matrix and has been a preferred choice for 3D bioprinting scaffolds over recent years (Hölzl et al., 2016).
Natural hydrogels are widely used because of their good biocompatibility and include chitosan, agarose, and sodium alginate (Hospodiuk et al., 2017). However, these natural hydrogels are soft and it is difficult to achieve the necessary mechanical properties for a scaffold; this limitation can result in collapse of the structure after implantation, thus resulting in a failure to bridge the broken end of the spinal cord. In contrast, synthetic hydrogels can enhance the mechanical strength of scaffolds by adding other ingredients and chemical modifications. One comparative experiment showed that neurons cultured with softer hydrogels were more likely to induce neurite development in different strength hydrogels (Gungor-Ozkerim et al., 2018). However, astrocytes are an exception as they have different adhesion properties than neurons and can grow efficiently on a hard medium. For example, hydrogel-gelatin methacrylate (GelMA) is synthesized by adding methyl propenyl to gelatin; furthermore, the mechanical strength of GelMA can be altered by changing the exposure time to ultraviolet light. In addition, the mechanical strength and degradability of hydrogels can be regulated by the concentration of methyl propenyl. In addition, modified synthetic hydrogels can carry a variety of bioactive factors and successfully control the differentiation of SCs in an effective manner (Gungor-Ozkerim et al., 2018).
Scaffolds made of multiple materials are more effective for SCI than scaffolds made from a single material. In a previous study, Chen et al. used a 3D printing technique to fabricate a heparan sulfate/collagen scaffold (Chen et al., 2017); the internal structure of the 3D bioprinted scaffold was regular and porous. Moreover, the collagen/heparin sulfate complex significantly improved the compression modulus and strength of the scaffold when compared to a collagen scaffold and showed good biocompatibility when co-cultured with NSCs. Months after stent implantation in the rat spinal cord, collagen/heparan sulfate treatment further improved the pathological process, and a significant increase in the number of neurofilament-positive cells was observed (Chen et al., 2017). The advantages and disadvantages of different bioinks are shown in Table 3. In addition, it is possible to add cells to 3D bioprinted scaffolds during, thus allowing the production of a specific scaffold that features cells, thus creating a special scaffold that features functional cells (Liu et al., 2021b). In the next section, we discuss the combination of 3D bioprinted scaffolds with different types of SCs for the treatment of SCI.
Table 3.
Advantages and disadvantages of different bioinks
| Bioinks | Advantages | Disadvantages |
|---|---|---|
| Fibrin-based bioinks | Support cell growth and proliferation | Rapid degradation rate |
| Polysaccharide-based bioinks | Favorable biocompatibility | Require mild cross-linking |
| Limited impact on cells’ properties | Markedly fragile | |
| Low mechanical properties | ||
| Hyaluronic acid-based bioinks | Natural component | Fast degradation rate |
| No immunogenicity | Poor compression resistance | |
| Good biocompatibility | ||
| Bioinks based on synthetic components | Tunable mechanical properties | Complicated process |
| Better biocompatibility |
The Combination of Three-Dimensional Bioprinting Scaffolds and Stem Cells for the Treatment of Spinal Cord Injury
Combined transplantation is the most effective treatment for SCI. Biological 3D printing technology has made the combination of 3D scaffolds and cells more accurate and effective in the treatment of SCI (Fu et al., 2017). This combination provides a powerful and versatile method to align large numbers of uniformly and densely distributed functional cells to create precisely controlled neural tissue structures. Overall, the significant advantage of cell/activator-loaded 3D bioprinted scaffolds is that they can construct a “spinal cord-like” scaffold featuringa an abundance of cells that are precisely spatially aligned for optimal axonal reattachment (Figure 3).
Figure 3.
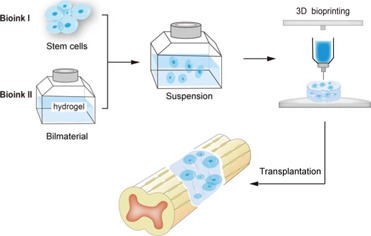
The combined application of stem cells and three-dimensional bioprinting scaffolds in the repair of spinal cord injury.
Created with Adobe illustrator (version 2017 cc).
One material and one SC
Although SCs therapy has been studied for many years, due to the easy loss of stem cells with the CSF and the limited viability of stem cells at the injury site, an increasing number of studies are attempting to optimize the efficacy of SCs by using scaffolds. One previous study combined 3D methacrylate gelatin (GelMA) hydrogels with induced neural SCs for transplantation. In vitro observations revealed that these composite scaffolds survived and differentiated well and exhibited strong ability to promote axonal growth. In vivo, there was a significant reduction in cavity area and collagen deposition in the graft group. In addition, GelMA and induced neural SC co-grafts reduced inflammation by inhibiting activated macrophages/microglia, inhibiting glial fibrillary acidic protein-positive cells and glial scar formation (Gong et al., 2023), and improving the microenvironment. There is no doubt that the combination of a 3D hydrogel with SCs is a promising therapeutic method for the repair of SCI (Fan et al., 2018).
Multiple materials and one SC
Over recent years, research has gradually begun to shift from scaffolds composed of a single material to scaffolds composed of multiple materials. Furthermore, SCs can be combined with scaffolds to create new therapies for SCI. In a previous study, Jiang et al. created a composite scaffold of 3D collagen/filamentous protein and NSCs and observed that this combination therapy was the best option for continuity of the spinal cord and filling the injury cavity when compared to other controls, as determined by magnetic resonance imaging and diffusion tensor imaging (Jiang et al., 2020). In addition, these authors reported that there were an abundance of axons and only minimal scarring in this group, thus confirming that combined implantation can promote the treatment of SCI (Jiang et al., 2020).
In another study, Liu et al. (2022) constructed sodium alginate/gelatin scaffolds loaded with NSCs and oligodendrocytes and transplanted them into spinal cords from rats that had been completely transected. Hematoxylin-eosin staining was performed after transplantation and no significant cavities were observed microscopically. Furthermore, immunofluorescence showed that the combined transplantation group had the greatest numbers of myelin basic protein-, 5-hydroxytryptamine- and neurofilament-positive cells. These results showed that combined transplantation was more effective than scaffold and SC transplantation alone (Liu et al., 2022).
One material and multiple SCs
Some studies have also attempted to apply a single material in combination with multiple cells to the injured spinal cord. One study prepared a polycaprolactone scaffold and loaded it with activated Schwann cells and iPSC-derived NSCs (Zhou et al., 2018). The polycaprolactone scaffold was approximately 0.5 mm thick and was biocompatible and biodegradable. Microscopic results showed that ASCs and iPSC-derived NSCs grew well on the polycaprolactone scaffold. Furthermore, the graft reduced the volume of the injury cavity and improved motor recovery in experimental rats (Zhou et al., 2018). Experimental progress relating to the combination therapy of SCs with 3D bioprinting scaffolds over recent years is shown in Table 4.
Table 4.
The experimental progress of the combination therapy of stem cells with 3D bioprinting scaffolds in recent years
| Methods of print | Stem cells | Materials | Results |
|---|---|---|---|
| Micro-scale continuous projection printing | NPCs | PEG-GelMA | The injured axons regenerate and synapse onto NPCs, NPCs extend axons into the spinal cord below the injury and restore synaptic transmission |
| Micro-extrusion | NSCs | chitosan, hyaluronic acid derivatives, and Matrigel | The axon regeneration and decreased glial scar deposition, significant locomotor recovery of the SCI rats |
| SLA | iNSCs | GelMA | The cavity areas were significantly reduced, less collagen was deposited, striking therapeutic effects of inhibiting glial fibrillary acidic protein-positive cells and glial scar formation while simultaneously promoting axonal regeneration |
| FDM | SCs + (iPSC)-NSCs | Polycaprolactone | The ASCs and iPSC-NSCs grew well, reduced the volume of lesion cavity, and improved the locomotor recovery of rats |
| Micro-extrusion | NSCs | Collagen-heparin sulfate | Improved locomotor function was observed |
ASCs: Adult stem cells; ESCs: embryonic stem cells; FDM: fused deposition modelling; GelMA: gelatin methacrylate; HSCs: hematopoietic stem cells; iPSCs: induced pluripotent stem cells; MSCs: mesenchymal stem cells; NPCs: neural progenitor cells; NSCs: neural stem cells; PEG-GelMA: polyethylene glycol-gelatin methacrylate; SCI: spinal cord injury; SLA: stereolithography.
It is highly evident that research is no longer limited to the few bioprinting methods described above, and more methods have emerged that allow the construction of 3D bioscaffolds. In one study, the survival, density and delivery of paracrine factors from human bone marrow MSCs (hMSCs) were optimized by using a new lyophilized scaffold loading procedure (the soaked up effect) that combined an agarose/carbohydrate based hydrogel with human bone marrow MSCs, thus reducing mechanical stress on human bone marrow MSCs being encapsulated into the hydrogel (Caron et al., 2016). These results demonstrated that the modified combination of human bone marrow MSCs and bionic hydrogels led to significant immunomodulation of the pro-inflammatory environment, an increase in the number of M2 macrophages, and the promotion of the in situ pro-regenerative environment in a mouse model of SCI (Caron et al., 2016).
Interaction factors in combination therapy
Instead of traditional single-layer and two-dimensional culture, there is now a greater tendency to use “3D culture” to simulate the important characteristics of target tissues and the extracellular microenvironment. 3D biomaterials have the potential to guide cell fate and function in ways that were not previously possible.
Biological materials largely determine the survival rate and efficiency of SCs. In a previous study, Gu et al. demonstrated the effectiveness of a printing platform to generate 3D tissue from human SCs in an experiment using a 3D bioprinting scaffold as a carrier to grow SCs (Gu et al., 2017). As long as the bioink has the right consistency and viscosity, 3D bioprinting can produce scaffolds of the right size and shape to fit the site of injury and contains an even distribution of highly active cells. A proportion of cell death is known to occur after surgery, including cross-linking and folding; however, these losses are negligible. In addition, even iPSCs, which are notoriously difficult to culture, were observed to proliferate upon printing, peaking on day 9 with normal differentiation. The high porosity of this material ensures the permeability of the bioactive factors while the hardness ensures lasting biocompatibility. In addition, some materials are known to have other beneficial properties, such as high moisture retention, low inflammation, toxicity, and antibacterial responses, and can also promote cell adhesion, migration and proliferation (Gu et al., 2017). Furthermore, SCs can be fine-tuned by changing the properties of the bioink, the printing parameters, and the printing process. However, specific adjustments to this methodology will depend on the type of cell required and will also involve a trade-off between iPSC support and differentiated cell and tissue support.
The microenvironment is another important factor that affects SC seeding, survival, or death. Local bleeding leads to the flow of inflammatory cells, vasoactive peptides and cytokines into the spinal cord, activation of apoptotic signaling pathways, and changes in cellular permeability that lead to the loss and demyelination of many functional neurons, thus completely destroying the spinal microenvironment. It is clear from the literature that local hemostasis, anti-inflammatory therapy, vascular remodeling, and the production and release of growth factors and anti-apoptotic factors all support SC transplantation, survival, and directional axonal regeneration, at least to some extent (Fan et al., 2022a).
Current limitations influencing the efficacy of combination therapy
The biological preparation of scaffolds combined with SCs is still a relatively new research field, and various schemes are still in their infancy (Yuan et al., 2022). Clinical applications are still a long way off. The main reasons for the limited use of this technology thus far relate to three main issues. First, the 3D bioprinting method. The current printing speed is slow, and the printing accuracy needs to be improved further. Moreover, the process of bioprinting is complicated and the mechanical stability of the structure is insufficient. Therefore, optimization of the printing method is critical if we are to achieve clinical transformation. Second, the selection of biological materials. Biological ink carriers should not only have the best biocompatibility, degradability and mechanical properties, they must also be suitable for mass production. Such biological materials are still being identified and developed. Third, SC acquisition and survival. The origin of some SCs is associated with ethical problems. Therefore, it is worth considering further options to isolate high-quality SCs in sufficient numbers. There are also fundamental issues related tocell survival and viability. Therefore, the development of specialized and advanced extraction and culture techniques is also a priority.
Conclusion and Future Perspectives
The use of 3D bioprinted scaffolds loaded with SCs is being actively investigated as a potential therapeutic approach for SCI. Each type of SC has certain advantages, although there is still a need to determine which cell type is the most effective. Different printing methods for 3D bioprinted scaffolds can overcome the limitations of other methods such as cost, complexity, and risk of immune rejection and activation. Of course, this method also has its limitations, such as the low possibility of large-scale manufacturing, the tedious process of bioprinting, the long fabrication time, the low mechanical stability, and the low cell survival rate. In addition, preservation and transportation methods also need significant improvement. Therefore, a significant body of research is needed to identify the best bioink and methods to combine 3D bioprinted scaffolds with SCs during manufacture and clinical trials aimed to improve the quality-of-life of patients with SCI. Thus far, no randomized clinical trial has demonstrated the efficacy of a treatment to promote functional recovery in patients with SCI.
It is important to emphasize that due to the complex pathophysiology of SCI, a single approach is unlikely to overcome the multifaceted barriers. In addition to biological interventions, the addition of other engineering strategies should be considered, including electrical neuromodulation and activity-based rehabilitation; such combined therapies are likely to achieve better outcomes than single therapies. It is evident that exosomes are being increasingly used in the treatment of SCI. We hypothesize that the combination of exosomes with four-dimensional printed bioscaffolds might represent a promising approach for the treatment of SCI in the future.
Funding Statement
Funding: This work was supported by the National Natural Science Foundation of China, No. 82171380 (to CD); and Jiangsu Students' Platform for Innovation and Entrepreneurship Training Program, No. 202110304098Y (to DJ).
Footnotes
Conflicts of interest: The authors declare no conflicts of interest.
Data availability statement: Not applicable.
C-Editor: Zhao M; S-Editors: Yu J, Li CH; L-Editors: Yu J, Song LP; T-Editor: Jia Y
References
- Abas M, Habib T, Noor S, Salah B, Zimon D. Parametric investigation and optimization to study the effect of process parameters on the dimensional deviation of fused deposition modeling of 3D printed parts. Polymers (Basel) 2022;14:3667. doi: 10.3390/polym14173667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahi ZB, Assunção-Silva RC, Salgado AJ, Tuzlakoglu K. A combinatorial approach for spinal cord injury repair using multifunctional collagen-based matrices: development, characterization and impact on cell adhesion and axonal growth. Biomed Mater. 2020;15:055024. doi: 10.1088/1748-605X/ab924d. [DOI] [PubMed] [Google Scholar]
- Ahuja CS, Fehlings M. Concise review: Bridging the gap: novel neuroregenerative and neuroprotective strategies in spinal cord injury. Stem Cells Transl Med. 2016;5:914–924. doi: 10.5966/sctm.2015-0381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Åkesson E, Sundström E. Human neural progenitor cells in central nervous system lesions. Best Pract Res Clin Obstet Gynaecol. 2016;31:69–81. doi: 10.1016/j.bpobgyn.2015.11.020. [DOI] [PubMed] [Google Scholar]
- Billiet T, Vandenhaute M, Schelfhout J, Van Vlierberghe S, Dubruel P. A review of trends and limitations in hydrogel-rapid prototyping for tissue engineering. Biomaterials. 2012;33:6020–6041. doi: 10.1016/j.biomaterials.2012.04.050. [DOI] [PubMed] [Google Scholar]
- Bishop ES, Mostafa S, Pakvasa M, Luu HH, Lee MJ, Wolf JM, Ameer GA, He TC, Reid RR. 3-D bioprinting technologies in tissue engineering and regenerative medicine: current and future trends. Genes Dis. 2017;4:185–195. doi: 10.1016/j.gendis.2017.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonner JF, Steward O. Repair of spinal cord injury with neuronal relays: From fetal grafts to neural stem cells. Brain Res. 2015;1619:115–123. doi: 10.1016/j.brainres.2015.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bryukhovetskiy AS, Bryukhovetskiy IS. Effectiveness of repeated transplantations of hematopoietic stem cells in spinal cord injury. World J Transplant. 2015;5:110–128. doi: 10.5500/wjt.v5.i3.110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cantinieaux D, Quertainmont R, Blacher S, Rossi L, Wanet T, Noel A, Brook G, Schoenen J, Franzen R. Conditioned medium from bone marrow-derived mesenchymal stem cells improves recovery after spinal cord injury in rats: an original strategy to avoid cell transplantation. PLoS One. 2013;8:e69515. doi: 10.1371/journal.pone.0069515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao TT, Chen H, Pang M, Xu SS, Wen HQ, Liu B, Rong LM, Li MM. Dose optimization of intrathecal administration of human umbilical cord mesenchymal stem cells for the treatment of subacute incomplete spinal cord injury. Neural Regen Res. 2022;17:1785–1794. doi: 10.4103/1673-5374.332151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caron I, Rossi F, Papa S, Aloe R, Sculco M, Mauri E, Sacchetti A, Erba E, Panini N, Parazzi V, Barilani M, Forloni G, Perale G, Lazzari L, Veglianese P. A new three dimensional biomimetic hydrogel to deliver factors secreted by human mesenchymal stem cells in spinal cord injury. Biomaterials. 2016;75:135–147. doi: 10.1016/j.biomaterials.2015.10.024. [DOI] [PubMed] [Google Scholar]
- Chang CC, Boland ED, Williams SK, Hoying JB. Direct-write bioprinting three-dimensional biohybrid systems for future regenerative therapies. J Biomed Mater Res B Appl Biomater. 2011;98:160–170. doi: 10.1002/jbm.b.31831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen C, Zhao ML, Zhang RK, Lu G, Zhao CY, Fu F, Sun HT, Zhang S, Tu Y, Li XH. Collagen/heparin sulfate scaffolds fabricated by a 3D bioprinter improved mechanical properties and neurological function after spinal cord injury in rats. J Biomed Mater Res A. 2017;105:1324–1332. doi: 10.1002/jbm.a.36011. [DOI] [PubMed] [Google Scholar]
- Chen J, Hu R, Ge H, Duanmu W, Li Y, Xue X, Hu S, Feng H. G-protein-coupled receptor 30-mediated antiapoptotic effect of estrogen on spinal motor neurons following injury and its underlying mechanisms. Mol Med Rep. 2015;12:1733–1740. doi: 10.3892/mmr.2015.3601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng Z, Zhu W, Cao K, Wu F, Li J, Wang G, Li H, Lu M, Ren Y, He X. Anti-inflammatory mechanism of neural stem cell transplantation in spinal cord injury. Int J Mol Sci. 2016;17:1380. doi: 10.3390/ijms17091380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng Z, Bosco DB, Sun L, Chen X, Xu Y, Tai W, Didier R, Li J, Fan J, He X, Ren Y. Neural stem cell-conditioned medium suppresses inflammation and promotes spinal cord injury recovery. Cell Transplant. 2017;26:469–482. doi: 10.3727/096368916X693473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cui H, Nowicki M, Fisher JP, Zhang LG. 3D bioprinting for organ regeneration. Adv Healthc Mater. 2017;6 doi: 10.1002/adhm.201601118. 10.1002/adhm.201601118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cui X, Dean D, Ruggeri ZM, Boland T. Cell damage evaluation of thermal inkjet printed Chinese hamster ovary cells. Biotechnol Bioeng. 2010;106:963–969. doi: 10.1002/bit.22762. [DOI] [PubMed] [Google Scholar]
- Cui YF, Xu JC, Hargus G, Jakovcevski I, Schachner M, Bernreuther C. Embryonic stem cell-derived L1 overexpressing neural aggregates enhance recovery after spinal cord injury in mice. PLoS One. 2011;6:e17126. doi: 10.1371/journal.pone.0017126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cunha C, Panseri S, Antonini S. Emerging nanotechnology approaches in tissue engineering for peripheral nerve regeneration. Nanomedicine. 2011;7:50–59. doi: 10.1016/j.nano.2010.07.004. [DOI] [PubMed] [Google Scholar]
- Duan H, Song W, Zhao W, Gao Y, Yang Z, Li X. Endogenous neurogenesis in adult mammals after spinal cord injury. Sci China Life Sci. 2016;59:1313–1318. doi: 10.1007/s11427-016-0205-2. [DOI] [PubMed] [Google Scholar]
- Eckert MJ, Martin MJ. Trauma: spinal cord injury. Surg Clin North Am. 2017;97:1031–1045. doi: 10.1016/j.suc.2017.06.008. [DOI] [PubMed] [Google Scholar]
- Fan B, Wei Z, Feng S. Progression in translational research on spinal cord injury based on microenvironment imbalance. Bone Res. 2022a;10:35. doi: 10.1038/s41413-022-00199-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fan L, Liu C, Chen X, Zou Y, Zhou Z, Lin C, Tan G, Zhou L, Ning C, Wang Q. Directing induced pluripotent stem cell derived neural stem cell fate with a three-dimensional biomimetic hydrogel for spinal cord injury repair. ACS Appl Mater Interfaces. 2018;10:17742–17755. doi: 10.1021/acsami.8b05293. [DOI] [PubMed] [Google Scholar]
- Fan L, Liu C, Chen X, Zheng L, Zou Y, Wen H, Guan P, Lu F, Luo Y, Tan G, Yu P, Chen D, Deng C, Sun Y, Zhou L, Ning C. Exosomes-loaded electroconductive hydrogel synergistically promotes tissue repair after spinal cord injury via immunoregulation and enhancement of myelinated axon growth. Adv Sci (Weinh) 2022b;9:e2105586. doi: 10.1002/advs.202105586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fehlings MG, Tetreault LA, Wilson JR, Kwon BK, Burns AS, Martin AR, Hawryluk G, Harrop JS. A clinical practice guideline for the management of acute spinal cord injury: introduction, rationale, and scope. Global Spine J. 2017;7:84s–94s. doi: 10.1177/2192568217703387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fischer I, Dulin JN, Lane MA. Transplanting neural progenitor cells to restore connectivity after spinal cord injury. Nat Rev Neurosci. 2020;21:366–383. doi: 10.1038/s41583-020-0314-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forostyak S, Jendelova P, Sykova E. The role of mesenchymal stromal cells in spinal cord injury, regenerative medicine and possible clinical applications. Biochimie. 2013;95:2257–2270. doi: 10.1016/j.biochi.2013.08.004. [DOI] [PubMed] [Google Scholar]
- Frolov AA, Bryukhovetskiy AS. Effects of hematopoietic autologous stem cell transplantation to the chronically injured human spinal cord evaluated by motor and somatosensory evoked potentials methods. Cell Transplant 21 Suppl. 2012;1:S49–55. doi: 10.3727/096368912x633761. [DOI] [PubMed] [Google Scholar]
- Fu F, Qin Z, Xu C, Chen XY, Li RX, Wang LN, Peng DW, Sun HT, Tu Y, Chen C, Zhang S, Zhao ML, Li XH. Magnetic resonance imaging-three-dimensional printing technology fabricates customized scaffolds for brain tissue engineering. Neural Regen Res. 2017;12:614–622. doi: 10.4103/1673-5374.205101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao L, Peng Y, Xu W, He P, Li T, Lu X, Chen G. Progress in stem cell therapy for spinal cord injury. Stem Cells Int. 2020;2020:2853650. doi: 10.1155/2020/2853650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gómez-Lizárraga KK, Flores-Morales C, Del Prado-Audelo ML, Álvarez-Pérez MA, Piña-Barba MC, Escobedo C. Polycaprolactone- and polycaprolactone/ceramic-based 3D-bioplotted porous scaffolds for bone regeneration: A comparative study. Mater Sci Eng C Mater Biol Appl. 2017;79:326–335. doi: 10.1016/j.msec.2017.05.003. [DOI] [PubMed] [Google Scholar]
- Gong L, Gu Y, Han X, Luan C, Liu C, Wang X, Sun Y, Zheng M, Fang M, Yang S, Xu L, Sun H, Yu B, Gu X, Zhou S. Spatiotemporal dynamics of the molecular expression pattern and intercellular interactions in the glial scar response to spinal cord injury. Neurosci Bull. 2023;39:213–244. doi: 10.1007/s12264-022-00897-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gu Q, Tomaskovic-Crook E, Wallace GG, Crook JM. 3D bioprinting human induced pluripotent stem cell constructs for in situ cell proliferation and successive multilineage differentiation. Adv Healthc Mater. 2017;6:1700175. doi: 10.1002/adhm.201700175. [DOI] [PubMed] [Google Scholar]
- Gungor-Ozkerim PS, Inci I, Zhang YS, Khademhosseini A, Dokmeci MR. Bioinks for 3D bioprinting: an overview. Biomater Sci. 2018;6:915–946. doi: 10.1039/c7bm00765e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han D, Chen S, Fang S, Liu S, Jin M, Guo Z, Yuan Y, Wang Y, Liu C, Mei X. The neuroprotective effects of muscle-derived stem cells via brain-derived neurotrophic factor in spinal cord injury model. Biomed Res Int. 2017;2017:1972608. doi: 10.1155/2017/1972608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hölzl K, Lin S, Tytgat L, Van Vlierberghe S, Gu L, Ovsianikov A. Bioink properties before, during and after 3D bioprinting. Biofabrication. 2016;8:032002. doi: 10.1088/1758-5090/8/3/032002. [DOI] [PubMed] [Google Scholar]
- Hospodiuk M, Dey M, Sosnoski D, Ozbolat IT. The bioink: a comprehensive review on bioprintable materials. Biotechnol Adv. 2017;35:217–239. doi: 10.1016/j.biotechadv.2016.12.006. [DOI] [PubMed] [Google Scholar]
- Hsu SH, Chang WC, Yen CT. Novel flexible nerve conduits made of water-based biodegradable polyurethane for peripheral nerve regeneration. J Biomed Mater Res A. 2017;105:1383–1392. doi: 10.1002/jbm.a.36022. [DOI] [PubMed] [Google Scholar]
- Huang CY, Hu KH, Wei ZH. Comparison of cell behavior on pva/pva-gelatin electrospun nanofibers with random and aligned configuration. Sci Rep. 2016;6:37960. doi: 10.1038/srep37960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang Q, Zou Y, Arno MC, Chen S, Wang T, Gao J, Dove AP, Du J. Hydrogel scaffolds for differentiation of adipose-derived stem cells. Chem Soc Rev. 2017;46:6255–6275. doi: 10.1039/c6cs00052e. [DOI] [PubMed] [Google Scholar]
- Jiang JP, Liu XY, Zhao F, Zhu X, Li XY, Niu XG, Yao ZT, Dai C, Xu HY, Ma K, Chen XY, Zhang S. Three-dimensional bioprinting collagen/silk fibroin scaffold combined with neural stem cells promotes nerve regeneration after spinal cord injury. Neural Regen Res. 2020;15:959–968. doi: 10.4103/1673-5374.268974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson BN, Lancaster KZ, Hogue IB, Meng F, Kong YL, Enquist LW, McAlpine MC. 3D printed nervous system on a chip. Lab Chip. 2016;16:1393–1400. doi: 10.1039/c5lc01270h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joung D, Lavoie NS, Guo SZ, Park SH, Parr AM, McAlpine MC. 3D printed neural regeneration devices. Adv Funct Mater. 2020;30 doi: 10.1002/adfm.201906237. 10.1002/adfm.201906237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kadoya K, Lu P, Nguyen K, Lee-Kubli C, Kumamaru H, Yao L, Knackert J, Poplawski G, Dulin JN, Strobl H, Takashima Y, Biane J, Conner J, Zhang SC, Tuszynski MH. Spinal cord reconstitution with homologous neural grafts enables robust corticospinal regeneration. Nat Med. 2016;22:479–487. doi: 10.1038/nm.4066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kameda T, Imamura T, Nakashima K. Epigenetic regulation of neural stem cell differentiation towards spinal cord regeneration. Cell Tissue Res. 2018;371:189–199. doi: 10.1007/s00441-017-2656-2. [DOI] [PubMed] [Google Scholar]
- Kawabata S, Takano M, Numasawa-Kuroiwa Y, Itakura G, Kobayashi Y, Nishiyama Y, Sugai K, Nishimura S, Iwai H, Isoda M, Shibata S, Kohyama J, Iwanami A, Toyama Y, Matsumoto M, Nakamura M, Okano H. Grafted human iPS cell-derived oligodendrocyte precursor cells contribute to robust remyelination of demyelinated axons after spinal cord injury. Stem Cell Reports. 2016;6:1–8. doi: 10.1016/j.stemcr.2015.11.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keefe KM, Sheikh IS, Smith GM. Targeting neurotrophins to specific populations of neurons: NGF, BDNF, and NT-3 and their relevance for treatment of spinal cord injury. Int J Mol Sci. 2017;18:548. doi: 10.3390/ijms18030548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi Y, Okada Y, Itakura G, Iwai H, Nishimura S, Yasuda A, Nori S, Hikishima K, Konomi T, Fujiyoshi K, Tsuji O, Toyama Y, Yamanaka S, Nakamura M, Okano H. Pre-evaluated safe human iPSC-derived neural stem cells promote functional recovery after spinal cord injury in common marmoset without tumorigenicity. PLoS One. 2012;7:e52787. doi: 10.1371/journal.pone.0052787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lane MA, Lepore AC, Fischer I. Improving the therapeutic efficacy of neural progenitor cell transplantation following spinal cord injury. Expert Rev Neurother. 2017;17:433–440. doi: 10.1080/14737175.2017.1270206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee KZ, Lane MA, Dougherty BJ, Mercier LM, Sandhu MS, Sanchez JC, Reier PJ, Fuller DD. Intraspinal transplantation and modulation of donor neuron electrophysiological activity. Exp Neurol. 2014;251:47–57. doi: 10.1016/j.expneurol.2013.10.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee TH. Functional effect of mouse embryonic stem cell implantation after spinal cord injury. J Exerc Rehabil. 2013;9:230–233. doi: 10.12965/jer.130004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Z, Guo GH, Wang GS, Guan CX, Yue L. Influence of neural stem cell transplantation on angiogenesis in rats with spinal cord injury. Genet Mol Res. 2014;13:6083–6092. doi: 10.4238/2014.August.7.23. [DOI] [PubMed] [Google Scholar]
- Li Z, Zhao T, Ding J, Gu H, Wang Q, Wang Y, Zhang D, Gao C. A reactive oxygen species-responsive hydrogel encapsulated with bone marrow derived stem cells promotes repair and regeneration of spinal cord injury. Bioact Mater. 2023;19:550–568. doi: 10.1016/j.bioactmat.2022.04.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu S, Xie YY, Wang LD, Tai CX, Chen D, Mu D, Cui YY, Wang B. A multi-channel collagen scaffold loaded with neural stem cells for the repair of spinal cord injury. Neural Regen Res. 2021a;16:2284–2292. doi: 10.4103/1673-5374.310698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu S, Yang H, Chen D, Xie Y, Tai C, Wang L, Wang P, Wang B. Three-dimensional bioprinting sodium alginate/gelatin scaffold combined with neural stem cells and oligodendrocytes markedly promoting nerve regeneration after spinal cord injury. Regen Biomater. 2022;9:rbac038. doi: 10.1093/rb/rbac038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu X, Hao M, Chen Z, Zhang T, Huang J, Dai J, Zhang Z. 3D bioprinted neural tissue constructs for spinal cord injury repair. Biomaterials. 2021b;272:120771. doi: 10.1016/j.biomaterials.2021.120771. [DOI] [PubMed] [Google Scholar]
- López-Serrano C, Torres-Espín A, Hernández J, Alvarez-Palomo AB, Requena J, Gasull X, Edel MJ, Navarro X. Effects of the post-spinal cord injury microenvironment on the differentiation capacity of human neural stem cells derived from induced pluripotent stem cells. Cell Transplant. 2016;25:1833–1852. doi: 10.3727/096368916X691312. [DOI] [PubMed] [Google Scholar]
- Lv B, Zhang X, Yuan J, Chen Y, Ding H, Cao X, Huang A. Biomaterial-supported MSC transplantation enhances cell-cell communication for spinal cord injury. Stem Cell Res Ther. 2021;12:36. doi: 10.1186/s13287-020-02090-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marichal N, García G, Radmilovich M, Trujillo-Cenóz O, Russo RE. Spatial domains of progenitor-like cells and functional complexity of a stem cell niche in the neonatal rat spinal cord. Stem Cells. 2012;30:2020–2031. doi: 10.1002/stem.1175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsukura Y, Muneta T, Tsuji K, Koga H, Sekiya I. Mesenchymal stem cells in synovial fluid increase after meniscus injury. Clin Orthop Relat Res. 2014;472:1357–1364. doi: 10.1007/s11999-013-3418-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melchels FP, Feijen J, Grijpma DW. A review on stereolithography and its applications in biomedical engineering. Biomaterials. 2010;31:6121–6130. doi: 10.1016/j.biomaterials.2010.04.050. [DOI] [PubMed] [Google Scholar]
- Muheremu A, Peng J, Ao Q. Stem cell based therapies for spinal cord injury. Tissue Cell. 2016;48:328–333. doi: 10.1016/j.tice.2016.05.008. [DOI] [PubMed] [Google Scholar]
- Murphy SV, Atala A. 3D bioprinting of tissues and organs. Nat Biotechnol. 2014;32:773–785. doi: 10.1038/nbt.2958. [DOI] [PubMed] [Google Scholar]
- Nicola FDC, Marques MR, Odorcyk F, Arcego DM, Petenuzzo L, Aristimunha D, Vizuete A, Sanches EF, Pereira DP, Maurmann N, Dalmaz C, Pranke P, Netto CA. Neuroprotector effect of stem cells from human exfoliated deciduous teeth transplanted after traumatic spinal cord injury involves inhibition of early neuronal apoptosis. Brain Res. 2017;1663:95–105. doi: 10.1016/j.brainres.2017.03.015. [DOI] [PubMed] [Google Scholar]
- Nutt SE, Chang EA, Suhr ST, Schlosser LO, Mondello SE, Moritz CT, Cibelli JB, Horner PJ. Caudalized human iPSC-derived neural progenitor cells produce neurons and glia but fail to restore function in an early chronic spinal cord injury model. Exp Neurol. 2013;248:491–503. doi: 10.1016/j.expneurol.2013.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Brien CM, Holmes B, Faucett S, Zhang LG. Three-dimensional printing of nanomaterial scaffolds for complex tissue regeneration. Tissue Eng Part B Rev. 2015;21:103–114. doi: 10.1089/ten.teb.2014.0168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Reilly RC, McClelland JL. Hippocampal conjunctive encoding, storage, and recall: avoiding a trade-off. Hippocampus. 1994;4:661–682. doi: 10.1002/hipo.450040605. [DOI] [PubMed] [Google Scholar]
- Ozbolat IT, Hospodiuk M. Current advances and future perspectives in extrusion-based bioprinting. Biomaterials. 2016;76:321–343. doi: 10.1016/j.biomaterials.2015.10.076. [DOI] [PubMed] [Google Scholar]
- Pandamooz S, Salehi MS, Zibaii MI, Ahmadiani A, Nabiuni M, Dargahi L. Epidermal neural crest stem cell-derived glia enhance neurotrophic elements in an ex vivo model of spinal cord injury. J Cell Biochem. 2018;119:3486–3496. doi: 10.1002/jcb.26520. [DOI] [PubMed] [Google Scholar]
- Panwar A, Tan LP. Current status of bioinks for micro-extrusion-based 3D bioprinting. Molecules. 2016;21:685. doi: 10.3390/molecules21060685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peltola SM, Melchels FP, Grijpma DW, Kellomäki M. A review of rapid prototyping techniques for tissue engineering purposes. Ann Med. 2008;40:268–280. doi: 10.1080/07853890701881788. [DOI] [PubMed] [Google Scholar]
- Quertainmont R, Cantinieaux D, Botman O, Sid S, Schoenen J, Franzen R. Mesenchymal stem cell graft improves recovery after spinal cord injury in adult rats through neurotrophic and pro-angiogenic actions. PLoS One. 2012;7:e39500. doi: 10.1371/journal.pone.0039500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ross CL, Syed I, Smith TL, Harrison BS. The regenerative effects of electromagnetic field on spinal cord injury. Electromagn Biol Med. 2017;36:74–87. doi: 10.3109/15368378.2016.1160408. [DOI] [PubMed] [Google Scholar]
- Rossi SL, Nistor G, Wyatt T, Yin HZ, Poole AJ, Weiss JH, Gardener MJ, Dijkstra S, Fischer DF, Keirstead HS. Histological and functional benefit following transplantation of motor neuron progenitors to the injured rat spinal cord. PLoS One. 2010;5:e11852. doi: 10.1371/journal.pone.0011852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salewski RP, Buttigieg J, Mitchell RA, van der Kooy D, Nagy A, Fehlings MG. The generation of definitive neural stem cells from PiggyBac transposon-induced pluripotent stem cells can be enhanced by induction of the NOTCH signaling pathway. Stem Cells Dev. 2013;22:383–396. doi: 10.1089/scd.2012.0218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salewski RP, Mitchell RA, Li L, Shen C, Milekovskaia M, Nagy A, Fehlings MG. Transplantation of induced pluripotent stem cell-derived neural stem cells mediate functional recovery following thoracic spinal cord injury through remyelination of axons. Stem Cells Transl Med. 2015;4:743–754. doi: 10.5966/sctm.2014-0236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schepici G, Silvestro S, Mazzon E. Regenerative effects of exosomes-derived MSCs: an overview on spinal cord injury experimental studies. Biomedicines. 2023;11:201. doi: 10.3390/biomedicines11010201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seol YJ, Kang HW, Lee SJ, Atala A, Yoo JJ. Bioprinting technology and its applications. Eur J Cardiothorac Surg. 2014;46:342–348. doi: 10.1093/ejcts/ezu148. [DOI] [PubMed] [Google Scholar]
- Skardal A, Zhang J, Prestwich GD. Bioprinting vessel-like constructs using hyaluronan hydrogels crosslinked with tetrahedral polyethylene glycol tetracrylates. Biomaterials. 2010;31:6173–6181. doi: 10.1016/j.biomaterials.2010.04.045. [DOI] [PubMed] [Google Scholar]
- Skoog SA, Goering PL, Narayan RJ. Stereolithography in tissue engineering. J Mater Sci Mater Med. 2014;25:845–856. doi: 10.1007/s10856-013-5107-y. [DOI] [PubMed] [Google Scholar]
- Stenudd M, Sabelström H, Frisén J. Role of endogenous neural stem cells in spinal cord injury and repair. JAMA Neurol. 2015;72:235–237. doi: 10.1001/jamaneurol.2014.2927. [DOI] [PubMed] [Google Scholar]
- Subramanian A, Krishnan UM, Sethuraman S. Development of biomaterial scaffold for nerve tissue engineering: Biomaterial mediated neural regeneration. J Biomed Sci. 2009;16:108. doi: 10.1186/1423-0127-16-108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun X, Huang LY, Pan HX, Li LJ, Wang L, Pei GQ, Wang Y, Zhang Q, Cheng HX, He CQ, Wei Q. Bone marrow mesenchymal stem cells and exercise restore motor function following spinal cord injury by activating PI3K/AKT/mTOR pathway. Neural Regen Res. 2023;18:1067–1075. doi: 10.4103/1673-5374.355762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tahmasebi F, Barati S. Effects of mesenchymal stem cell transplantation on spinal cord injury patients. Cell Tissue Res. 2022;389:373–384. doi: 10.1007/s00441-022-03648-3. [DOI] [PubMed] [Google Scholar]
- Tsivelekas K, Evangelopoulos DS, Pallis D, Benetos IS, Papadakis SA, Vlamis J, Pneumaticos SG. Angiogenesis in spinal cord injury: progress and treatment. Cureus. 2022;14:e25475. doi: 10.7759/cureus.25475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walker MJ, Xu XM. History of glial cell line-derived neurotrophic factor (GDNF) and its use for spinal cord injury repair. Brain Sci. 2018;8:109. doi: 10.3390/brainsci8060109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang C, Shi D, Song X, Chen Y, Wang L, Zhang X. Calpain inhibitor attenuates ER stress-induced apoptosis in injured spinal cord after bone mesenchymal stem cells transplantation. Neurochem Int. 2016a;97:15–25. doi: 10.1016/j.neuint.2016.04.015. [DOI] [PubMed] [Google Scholar]
- Wang Y, Liu H, Ma H. Intrathecally transplanting mesenchymal stem cells (MSCs) activates ERK1/2 in spinal cords of ischemia-reperfusion injury rats and improves nerve function. Med Sci Monit. 2016b;22:1472–1479. doi: 10.12659/MSM.896503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wright KT, El Masri W, Osman A, Chowdhury J, Johnson WE. Concise review: Bone marrow for the treatment of spinal cord injury: mechanisms and clinical applications. Stem Cells. 2011;29:169–178. doi: 10.1002/stem.570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiong LL, Zou Y, Shi Y, Zhang P, Zhang RP, Dai XJ, Liu B, Wang TH. Tree shrew neural stem cell transplantation promotes functional recovery of tree shrews with a hemi-sectioned spinal cord injury by upregulating nerve growth factor expression. Int J Mol Med. 2018;41:3267–3277. doi: 10.3892/ijmm.2018.3553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiong LL, Liu F, Deng SK, Liu J, Dan QQ, Zhang P, Zou Y, Xia QJ, Wang TH. Transplantation of hematopoietic stem cells promotes functional improvement associated with NT-3-MEK-1 activation in spinal cord-transected rats. Front Cell Neurosci. 2017;11:213. doi: 10.3389/fncel.2017.00213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu B, Zhao Y, Xiao Z, Wang B, Liang H, Li X, Fang Y, Han S, Li X, Fan C, Dai J. A dual functional scaffold tethered with EGFR antibody promotes neural stem cell retention and neuronal differentiation for spinal cord injury repair. Adv Healthc Mater. 2017;6:1601279. doi: 10.1002/adhm.201601279. [DOI] [PubMed] [Google Scholar]
- Yahata K, Kanno H, Ozawa H, Yamaya S, Tateda S, Ito K, Shimokawa H, Itoi E. Low-energy extracorporeal shock wave therapy for promotion of vascular endothelial growth factor expression and angiogenesis and improvement of locomotor and sensory functions after spinal cord injury. J Neurosurg Spine. 2016;25:745–755. doi: 10.3171/2016.4.SPINE15923. [DOI] [PubMed] [Google Scholar]
- Yamazaki K, Kawabori M, Seki T, Houkin K. Clinical trials of stem cell treatment for spinal cord injury. Int J Mol Sci. 2020;21:3994. doi: 10.3390/ijms21113994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu S, Yao S, Wen Y, Wang Y, Wang H, Xu Q. Angiogenic microspheres promote neural regeneration and motor function recovery after spinal cord injury in rats. Sci Rep. 2016;6:33428. doi: 10.1038/srep33428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yuan TY, Zhang J, Yu T, Wu JP, Liu QY. 3D bioprinting for spinal cord injury repair. Front Bioeng Biotechnol. 2022;10:847344. doi: 10.3389/fbioe.2022.847344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang K, Lu WC, Zhang M, Zhang Q, Xian PP, Liu FF, Chen ZY, Kim CS, Wu SX, Tao HR, Wang YZ. Reducing host aldose reductase activity promotes neuronal differentiation of transplanted neural stem cells at spinal cord injury sites and facilitates locomotion recovery. Neural Regen Res. 2022;17:1814–1820. doi: 10.4103/1673-5374.330624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng M, Guo LJ, Xu XL, Hu HZ, Zong XG. ZD7288 inhibits the synaptic transmission in the pathway from perforant pathway fibers to CA3 region in rat hippocampus. Yao Xue Xue Bao. 2006;41:565–571. [PubMed] [Google Scholar]
- Zheng Q, Lu J, Chen H, Huang L, Cai J, Xu Z. Application of inkjet printing technique for biological material delivery and antimicrobial assays. Anal Biochem. 2011;410:171–176. doi: 10.1016/j.ab.2010.10.024. [DOI] [PubMed] [Google Scholar]
- Zhou X, Shi G, Fan B, Cheng X, Zhang X, Wang X, Liu S, Hao Y, Wei Z, Wang L, Feng S. Polycaprolactone electrospun fiber scaffold loaded with iPSCs-NSCs and ASCs as a novel tissue engineering scaffold for the treatment of spinal cord injury. Int J Nanomedicine. 2018;13:6265–6277. doi: 10.2147/IJN.S175914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu J, Marchant RE. Design properties of hydrogel tissue-engineering scaffolds. Expert Rev Med Devices. 2011;8:607–626. doi: 10.1586/erd.11.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu XY, Li P, Yang YB, Liu ML. Xuezhikang, extract of red yeast rice, improved abnormal hemorheology, suppressed Caveolin-1 and increased eNOS expression in atherosclerotic rats. PLoS One. 2013;8:e62731. doi: 10.1371/journal.pone.0062731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhukareva V, Obrocka M, Houle JD, Fischer I, Neuhuber B. Secretion profile of human bone marrow stromal cells: donor variability and response to inflammatory stimuli. Cytokine. 2010;50:317–321. doi: 10.1016/j.cyto.2010.01.004. [DOI] [PubMed] [Google Scholar]


