Abstract
Recent advances in percutaneous image-guided techniques have empowered interventional radiologists with diverse treatment options for the management of musculoskeletal lesions. Of note, there is growing utility for cementoplasty procedures, with indications ranging from stabilization of bone metastases to treatment of painful vertebral compression fractures. Likewise, cryoablation has emerged as a viable adjunct in the treatment of both primary and secondary bone and soft tissue neoplasms. These treatment options have been progressively incorporated into the multidisciplinary approach to holistic care of patients, alongside conventional radiotherapy, systemic therapy, surgery, and analgesia. This review article serves to outline the indications, technical considerations, latest developments, and evidence for the burgeoning role of cementoplasty and cryoablation in the musculoskeletal system, with an emphasis on pain palliation and tumour control.
Keywords: musculoskeletal, interventional radiology, interventional oncology, cementoplasty, cryoablation
Introduction
Over the last few decades, various percutaneous image-guided ablation and stabilization techniques, including cryoablation and cementoplasty, have emerged in the management of musculoskeletal lesions. There is growing utility of these procedures especially in the context of malignancy, where bone pain is one of the most common types of pain, and the skeleton is the third most frequent site of metastases.1 This is compounded by the prevalence of bone metastases, with the annual age-adjusted incidence rate of newly diagnosed bone metastasis approximately 18.8 per 100 000 in the United States.2
Currently, radiotherapy is considered the preferred treatment option for palliation for uncomplicated metastatic bone pain. However, radiotherapy alone does not contribute to bone stability.3,4 Other limitations of radiotherapy include radio-resistance of some tumours, risk of radiation-related fracture especially to weight-bearing bones, osteitis, osteonecrosis, as well as radiation dose limits for 1 body site.5,6 Additionally, radiotherapy has a delayed pain relief onset, typically 4 weeks and occasionally up to 15 weeks.7
Surgery is only indicated as first-line treatment for selected metastatic bone lesions, such as slow-growing tumours and patients with relatively good prognosis.8 Surgical decompression and instrumentation have a primary role in the management of acute spinal instability and spinal cord compression with neurological deficits.4 While opioids are recommended for management of cancer-related bone pain, they are associated with multiple common side effects and sometimes suboptimal pain relief.9,10
Given the limitations of traditional strategies, this review article serves to provide an update on the increasing role of cementoplasty and cryoablation in the musculoskeletal system, with an emphasis on pain palliation and tumour control.
Cementoplasty
Percutaneous cementoplasty is a minimally invasive technique comprising of cannula placement within a bone lesion with subsequent cement injection.11 The cement, usually polymethylmethacrylate, hardens in consistency after about 10-20 min. Cement polymerization and hardening involve an exothermic reaction, with temperature peaks of up to 75°C, and play an accessory analgesic role through the destruction of adjacent nociceptors.12
Indication
When considering cementoplasty for bone metastases, a multidisciplinary team including interventional radiologists, radiation oncologists, medical oncologists, and orthopaedic surgeons should be engaged. Cementoplasty is typically considered in the following clinical scenarios: persistent pain or imaging evidence of tumour progression despite maximized radiation therapy, contraindications to radiation therapy, lack of patient desire for radiation therapy, and/or inadequate treatment response to systemic therapies and analgesia,13 as well as prophylactic stabilization for impending pathological fractures in the axial skeleton.14
Preprocedural cross-sectional imaging is essential to determine the number and site of lesions, needle size, and approach, as well as measurements for ablation zone (if performed). MRI imaging features may also be useful to guide patient and vertebral level selection; levels with marrow oedema correlating with pain should be targeted for optimal pain relief.15 Careful review of prior imaging, with a low threshold for repeating the patient’s cross-sectional imaging, is advised, especially if new or significantly worsening symptoms are reported since the latest imaging was performed.16 In the authors’ experience, preprocedural imaging is usually current, preferably within a few days of procedure, and at most within a month from procedure.
Severity of the spinal instability is an important consideration and can be evaluated via various scoring systems, including the Spinal Instability Neoplastic Score (SINS).17 Surgical consultation for potential tumour resection or debulking with stabilization is suggested for patients with a SINS of 7 or higher. Spinal metastases resulting in central canal stenosis are typically managed with surgical intervention.18
As regards the spine, suitable patients for vertebroplasty include those with chronic pathological fractures refractory to radiotherapy or other conservative measures, acute vertebral fractures with less than 6 weeks of symptoms, and painful pathological vertebral fractures without spinal cord compression or significant neurological deficit.19 The most appropriate timeframe for substantial pain relief may be 2-4 weeks from onset of injury, as suggested by a meta-analysis.20 However, treatment in the initial 4 weeks of vertebral fractures may lead to a higher risk of complications, including cement leakage.21,22 In the authors’ experience, vertebroplasty can be performed before or after radiotherapy and readily coordinated during multidisciplinary discussions.
Cementoplasty is contraindicated in patients with coagulopathy, unstable spinal lesions, presence of local or systemic infections, allergy to bone cement, and asymptomatic vertebral compression.23,24 Vertebral plana with 90% or greater height loss and extensive osteolytic destruction especially at the posterior cortex of the target vertebral body are relative contraindications.25
Technique
In the authors’ experience, cementoplasty is usually tolerated well under conscious sedation with local anaesthesia; general anaesthesia can also be considered for certain patients. Patients are positioned according to the site of target lesion; in conventional vertebroplasty, this usually means a prone position with padding under the thorax and ankles. Local anaesthesia is administered along the puncture site and expected cannula trajectory, infiltrating the subcutaneous tissues, muscles, and pain-sensitive periosteum.
For vertebroplasty of a lumbar segment, a transpedicular approach is preferred where the upper outer aspect of the pedicle is accessed. The cannula is directed medially through the pedicle and into the anterior third of the vertebral body to the midline inferiorly. This optimizes cement distribution to ideally include most of the vertebral body with cement crossing the midline (Figure 1).19 A costotransverse approach for the thoracic vertebra is also feasible due to smaller pedicle sizes. Bipedicular access may be considered in some scenarios including suboptimal positioning of a single cannula, or severe central vertebral height loss preventing cannula placement along the midline. Other novel access routes, including transoral access of C2 and a trans-discal approach, have been reported.26,27
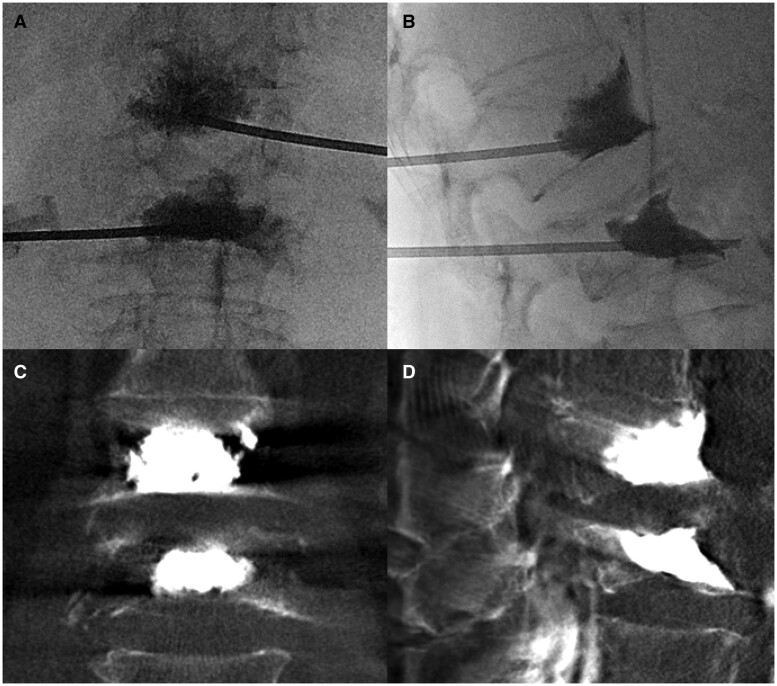
Figure 1.
Two-level vertebroplasty for osteoporotic lumbar compression fractures. Intra-procedural fluoroscopic images obtained in frontal (A) and lateral (B) planes to visualize cement deposition in the vertebral bodies. Postprocedural cone-beam CT images in coronal (C) and sagittal planes (D) demonstrated satisfactory cement fill with no leakage.
Aside from vertebroplasty, other percutaneous vertebral augmentation procedures include balloon kyphoplasty and vertebral stenting. During kyphoplasty, a balloon-like device is inflated inside the vertebral body, and its expansion restores vertebral body height, creating a cavity into which cement is then injected.24 Patient positioning and vertebral body access are similar to conventional vertebroplasty. A reamer is inserted over the access cannula, through which the stenting device or inflatable balloon is inserted. Once deployed, an intravertebral cavity is created with partial restoration of vertebral height.
Cementoplasty has also been performed at extraspinal positions, notably the sacrum, acetabulum, and long bones. For percutaneous sacroplasty (Figure 2), both a short-axis or long-axis cannula placement technique can be adopted depending on the size and extent of the fracture or lesions. The short-axis technique is better suited under CT guidance, with the cannula advanced perpendicular to the dorsal surface of the sacral ala. However, the volume of injected cement at each cannula may be limited, necessitating the placement of additional cannulas or adopting a more oblique approach.28
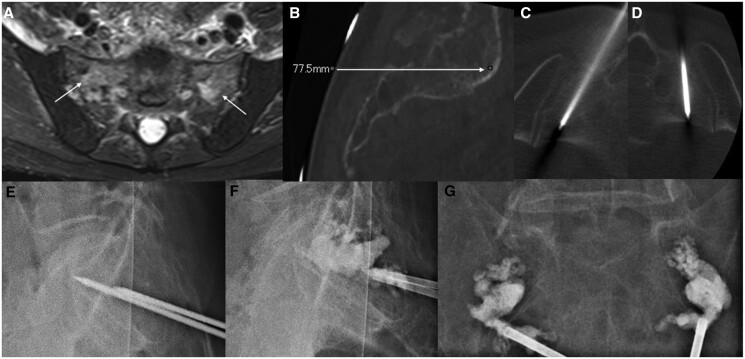
Figure 2.
Percutaneous sacroplasty. (A) MRI axial T2W with fat suppression image revealed marrow oedema in the sacral ala bilaterally (arrows) due to insufficiency fractures. (B) An oblique approach of cannula placement was planned on CT (arrow). Under CT guidance, bone cannulas were advanced into the sacral ala bilaterally (C and D), lateral to the sacral foramina. (E) Fluoroscopic control image in the sagittal plane was obtained prior to cement injection. Postprocedural images in the lateral (F) and frontal (G) projections demonstrated satisfactory cement fill with no cement leakage.
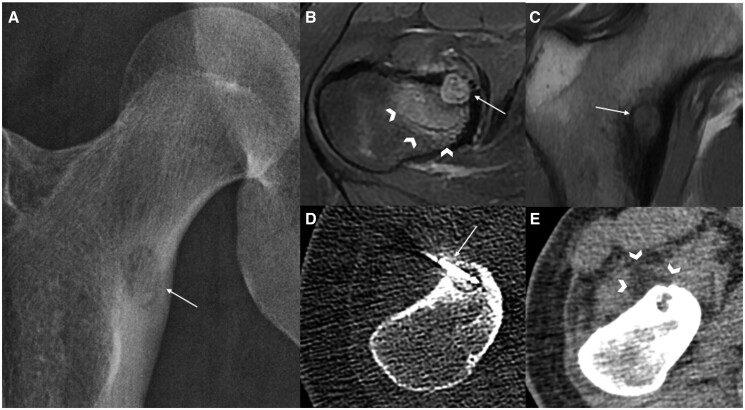
For percutaneous acetabuloplasty (Figure 3), either a posterior or anterior approach can be adopted. Cement is deposited along the acetabular roof and posterior column, as they form part of the load-transmission axis. Displaced fractures with acetabular protrusion are contraindications due to the risk of intra-articular cement leakage.
Figure 3.
Acetabular cementoplasty for a patient with metastatic lung cancer with persistent left hip pain. (A) Preprocedural CT coronal image revealed lytic lesions involving the left acetabular roof (arrows), while axial CT image (B) revealed sclerotic components in the iliac bone (arrowheads), suspicious for bony metastases. Under CT guidance with needle guidance software (XperGuide, Philips, Netherlands), acetabular cementoplasty was performed with 16 mL of cement deposited along the acetabular roof, preferentially at the posterior column as confirmed on axial (C), sagittal (D), and coronal (E) reformats.
Other novel techniques described include the usage of curved or directional needles for cementoplasty of the acetabulum and vertebral bodies.29,30
Imaging guidance
In the authors’ experience, cannula placement is usually performed under fluoroscopy, cone-beam CT, or conventional CT, sometimes with the assistance of advanced navigation software and real-time multiplanar reformats, depending on resource availability and operator preference. CT is preferred for cervical and upper thoracic spinal access due to the excellent spatial resolution of adjacent soft tissue structures. CT gantry angulation or needle guidance software are useful adjuncts in difficult-to-access lesions. Ultrasound is useful in certain situations such as visualizing critical neurovascular structures close to the cannula trajectory. Fluoroscopic guidance for cement injection is preferred to visualize real-time cement flow and distribution.
As musculoskeletal interventional oncology procedures become increasingly complex, many procedures are performed under a combination of CT and fluoroscopic guidance. This allows the acquisition of excellent spatial resolution on the axial plane with CT and real-time visualization of cement injection or embolization under fluoroscopy. The newest cone-beam CT units have incorporated artificial intelligence capabilities including deep learning algorithms that allow guidance of bone access cannulas without requiring fluoroscopy, markedly reducing radiation dose.31 MRI guidance has also emerged, allowing target identification in soft tissue lesions while avoiding ionizing radiation,32,33 although its usage is limited by costs and availability.
Effectiveness
Vertebrae
Previous trials such as VERTOS I and II trials demonstrated the benefit of vertebroplasty over conservative management for osteoporotic vertebral fractures.34,35 However, 2 large prospective multicentre studies and a subsequent Cochrane review failed to show a difference in pain improvement compared to a sham procedure, which involved injection of local anaesthetics but without cement deposition.36–39 The VERTOS IV trial replicated this conclusion in acute fractures up to 9 weeks.39 Major criticisms of these trials include a heterogeneous and less than ideal patient selection, heterogeneous and methodological flaws in the calculation of response to pain, underpowered study, and varying and suboptimal cement volume injected. In addition, the sham procedure of periosteal local anaesthesia at the dorsal pedicle was considered active treatment with significant effects on medial branch and sinuvertebral nerve blockade.40 Subsequently, the VAPOUR trial showed benefit over placebos in patients with acute fractures and symptom duration of less than 6 weeks.41
A follow-up of patients recruited for the VERTOS II and IV trials revealed statistically significantly more patients had a high pain score at 12-month follow-up in the sham and conservative group, as compared with the vertebroplasty group.42 Patients with moderate fracture deformity were also less likely to have high pain scores if they were treated with vertebroplasties.42
Pathological vertebral compression fractures are the second most common indication for vertebroplasty. A retrospective observation study of patients with metastatic compression fractures who underwent vertebroplasty reported improvement of visual analog scale (VAS) pain score from 5.8 to 2.7; patients with posterior column involvement and paravertebral extension reported greater pain relief.43 The efficacy of vertebroplasty on pain reduction and disability improvement is also well documented in multiple myeloma patients, with sustained results on long-term follow-up (5 years).44 Pain relief can be substantial; a study documented median VAS scores decreasing from 9 to 1 postvertebroplasty.45 Vertebroplasty is also a potentially effective percutaneous technique to treat symptomatic intraosseous haemangiomas, with lasting results.46
Currently, the overall consensus is that vertebral augmentation is appropriate for painful vertebral compression fractures refractory to conservative therapy, as well as for select neoplastic vertebral lesions.47 The authors are concordant with these recommendations. In the authors’ experience, vertebral augmentation for symptomatic neoplastic vertebral lesions with multidisciplinary input tends to yield favourable results. In addition, there are potentially additional benefits of vertebroplasty aside from pain relief and mortality which require further research.48
Sacrum
Percutaneous sacroplasty is established to be a safe procedure and provides good short-term and long-term outcomes for patients with painful insufficiency sacral fractures or pathological sacral lesions.28,49 A multicentre study of 243 patients reported mean VAS scores improving from 9.2 to 1.9 for sacral insufficiency fractures and 9.0 to 2.6 for sacral lesions, at 1-year follow-up interval posttreatment.28 However, patients with displaced or unstable sacral fragility fractures would benefit more from surgical fixation.50
Pelvis
In extraspinal bone metastases, cementoplasty is commonly performed to reinforce the acetabular roof and restore load transmission across the acetabulum. There is growing evidence demonstrating percutaneous cementoplasty as a safe and effective choice for patients with painful osteolytic pelvic bone metastases, significantly reducing pain and disability while preserving gait function.51–53 Most recently, Park et al54 evaluated percutaneous cement injection in 178 patients with pelvic bone lesions, which achieved significant pain reduction with mean numerical pain scores decreasing from 6.1 (pretreatment) to 2.1 (1 month posttreatment), while maintaining gait function in 68% of recruited patients.
Long bones
The role of cementoplasty for metastases in long weight-bearing bones is still debated, due to the presence of complex loading forces (tensile and torsion) in addition to compression. Initial studies reported a 1-year pathological fracture rate of 40.6% after cementoplasty of proximal femoral metastases.55 A systematic review by Cazzato et al53 reported that standalone cementoplasty in the long bones is effective to achieve pain alleviation and to improve mechanical function, but fracture is the most frequent complication.
More recently, a systematic review compared the efficacy between cementoplasty alone versus cementoplasty augmented with fixation devices for impending pathologic proximal femoral fractures. Both appeared effective for pain relief and had a similar postintervention fracture rate (5% for cementoplasty alone versus 7% for augmented cementoplasty), with no statistically significant differences.56 Given current evidence, cementation of the long bones can be considered for palliation of immobile patients who are unsuitable surgical candidates.
Kyphoplasty
Kyphoplasty is purported to combine the benefit of analgesia with the restoration of vertebral body height.24,57 By restoring vertebral body height and improving spinal sagittal balance, kyphoplasty prevents increased loading of the spinal anterior column and reduces the risk of additional compression fractures.58 Both kyphoplasty and vertebroplasty conferred a significant mortality benefit over conservative management, with the adjusted number needed to treat to save 1 life at up to 5 years estimated at 11.9 and 23.8, for conservative management versus kyphoplasty and vertebroplasty, respectively.59
For spinal metastases, both vertebroplasty and kyphoplasty significantly improved pain, disability, and health-related quality of life, with no technique substantially superior to the other58 nor had significant difference in pain relief.60 However, balloon kyphoplasty may also decrease cement interdigitation into the surrounding trabecular matrix, potentially compromising anchorage and bone consolidation.61 In addition, balloon kyphoplasty is postulated to cause further tumour dissemination due to the balloon inflation.62 It is also costlier than vertebroplasty.
Vertebral augmentation with implants
Other techniques of vertebral augmentation include devices such as vertebral body stenting, SpineJack system (Figure 4),59 and Osseofix, Vertelift, and KIVA system.63 A prospective randomized study comparing implantable titanium vertebral augmentation devices versus balloon kyphoplasty for painful osteoporotic vertebral compression fractures reported greater pain relief, improved height restoration, and lower incidence of adjacent fractures of the former, with similar results for adverse events and degree of functional improvement.64
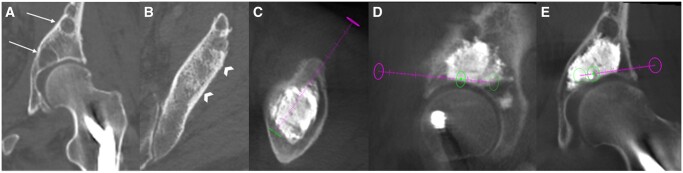
Figure 4.
Vertebral augmentation with stenting. (A) Radiograph in lateral view demonstrated an osteoporotic L1 compression fracture with severe height loss and kyphotic deformity (arrows). (B) SpineJack vertebral stenting devices (Stryker, United States) were advanced into the vertebral body via a transpedicular approach, and deployed under fluoroscopy with height restoration of the vertebral body (arrows in C). (D) Fluoroscopic frontal view confirmed satisfactory position. Cement injection subsequently commenced with adequate fill under lateral (E) and frontal (F) projections. Images courtesy of Dr Mark Wang and Dr Alexander Tan (Singapore General Hospital).
Osteosynthesis
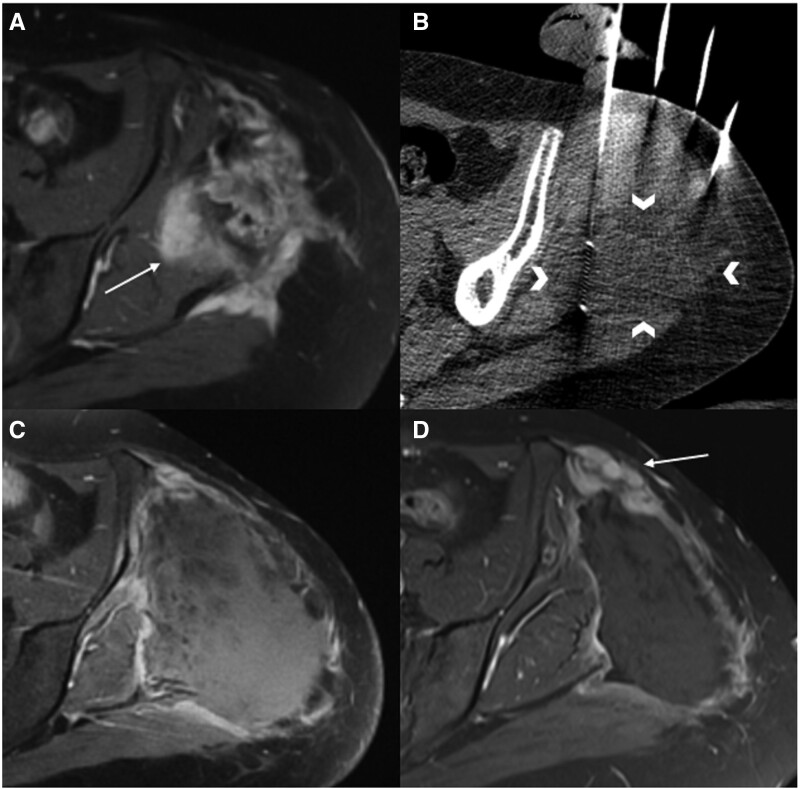
Unlike cement, screw fixation is its resistance to tensile, torsion, and shearing forces.65 Percutaneous image-guided screw fixation (Figure 5) has been recently introduced for stabilization and consolidation of minimally displaced or impending pathologic fractures (Mirel Score 8 or greater) predominantly in the pelvis and proximal femur and can be combined with cement augmentation (percutaneous internal cemented screws).66–69 These studies reported significant improvement in pain and functional scores, with Pusceddu et al68 reporting a decrease in mean VAS scores in 27 patients from 7.1 (pretreatment) to 1.4 (6 months posttreatment). Few major complications were reported; Deschamps et al66 reported 2 cases of secondary proximal femur fractures. A study using cone-beam CT guidance for placement of 75 percutaneous screws in pelvic bone metastases including steep angulations reported no adverse events.70
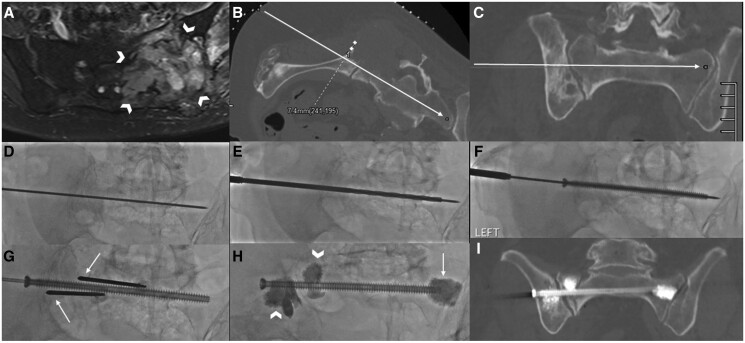
Figure 5.
Pelvic osteosynthesis for a 72-year-old patient with renal cell carcinoma bony metastases. (A) MRI axial T2W fat-suppressed image revealed an infiltrative T2W hyperintense lesion (arrowheads) in the left sacral ala and posterior iliac bone in keeping with metastases. (B) Patient was positioned prone with left side wedged up, and planning CT was performed to establish screw trajectory in the axial (arrow), with due consideration for screw diameter (dotted line). (C) Coronal trajectory planning was also acquired (arrow in C), with care to avoid traversing the neural foramina. (D) Under fluoroscopic guidance, a K-wire was advanced along the planned trajectory, with subsequent (E) predrilling and (F) screw insertion. (G) Additional bone cannulas were placed into the lytic sacral ala components (arrows). (H) Under fluoroscopic guidance, cement augmentation of the screw tip was performed, as well as cement deposition into the lytic components (arrowheads). (I) Completion CT demonstrated satisfactory screw placement and cement filling. Images courtesy of Dr Mark Wang and Dr Zhuang Kun Da (Singapore General Hospital).
The decision for percutaneous synthesis is multifactorial, taking into account factors such as the patient’s premorbid status, expected life expectancy, and lesion location. Percutaneous osteosynthesis is generally reserved for nonsurgical patients, for timely pain palliation and mobilization.66,71 In addition, more research is required to establish the long-term efficacy and safety profile.
Complications
Complications can be broadly divided into needle access-related and cement-related complications. Needle manipulation or errant placement may result in traumatic injury to critical structures including arteries, nerves, or tendons.72,73 Careful consideration and an appropriate imaging modality are key to optimize the visualization of critical structures along the needle trajectory. In the event of muscle or nerve injury, the preferred treatment option is nonsteroidal anti-inflammatory agents or steroids. Vascular injury may require a vascular surgery consult and surgical repair.74
As with all invasive procedures, infection is a potential complication and a routine aseptic technique with a preprocedural single dose of intravenous antibiotics that cover skin flora is usually adequate. Tumour seeding associated with needle manipulation is rare, with a few case reports published.75,76
Cement leakage occurs more frequently during the treatment of pathological vertebral compression fractures compared to osteoporotic fractures.77 Cement leakage into the peri-vertebral veins, paravertebral soft tissues, or intervertebral discs is commonly encountered, occurring in approximately 70% of cases, and is usually asymptomatic.78 Posterior cement leakage into the spinal canal is rare but can lead to spinal cord compression.79 Intra-foraminal leakage may be responsible for radiculopathy. In the hip joint, chondrolysis and osteonecrosis may result from intra-articular cement leakage, potentially requiring arthroplasty.80
Cement intravasation via the periosteal venous plexus has been reported with an incidence of up to 25%,73 while intravasation to the azygous vein portends a higher risk of pulmonary emboli.81 Similarly, cement injection may result in the displacement of bone marrow into the venous plexus, with potential fat embolism.82 It has been recommended to limit cement injection volume to less than 30 mL or 6 vertebral segments per session to minimize symptomatic fat embolism.83
Cryoablation
Cryoablation is a percutaneous thermal ablation technique where tumour tissue is cooled to extremely low temperatures achieved via placement of probes filled with liquid nitrogen or a compressed gas (usually argon) into the target lesion.84 Alternating cycles of rapid freezing and gradual thawing result in tumour cell death, via a complex mechanism involving the formation of intracellular ice crystals, endothelial damage to local tumoural blood supply, inducing ischaemia and devascularization.85 A temperature of lower than −20°C results in complete cell death, while about 80% of cells are destroyed in the 0-20°C zone.86
Indication
Cryoablation has an expanding role in the management of primary and secondary bone tumours. For primary bone tumours, cryoablation has been included in the European Society of Medical Oncology (ESMO) guidelines as a local adjuvant to curettage for atypical cartilaginous tumours and giant cell tumours and palliative treatment for recurrent extracranial chordomas.87 Cryoablation has also been reported as a viable alternative to radiofrequency ablation for osteoid osteoma (Figure 6)88 and the current preferred treatment modality,89 including a recent retrospective study of 50 patients which achieved a 96% overall clinical success rate.90
Figure 6.
Cryoablation for osteoid osteoma in a 20-year-old patient. (A) Anteroposterior hip radiograph revealed a small lucent lesion with a narrow zone of transition in the basicervical region of the right femoral neck (arrow). (B) MRI axial T2W fat-suppressed image revealed a well-circumscribed cortical-based T2W hyperintense lesion (arrow) and adjacent marrow oedema (arrowheads). (C) MRI coronal T1W image revealed a well-circumscribed lesion isointense to muscle (arrow). Findings are consistent with an osteoid osteoma and decision was made to treat via cryoablation. (D) Under CT guidance, an IceSphere ablation probe (Boston Scientific, USA) was advanced into the lesion (arrow). (E) Intra-procedural CT in soft tissue window confirmed ice formation (arrowheads) encompassing the lesion.
For bony metastases, the United States National Comprehensive Cancer Network guidelines have included percutaneous thermal ablation as a palliative treatment option for metastatic bone pain.91 The updated American College of Radiology appropriateness criteria includes percutaneous thermal ablation as a viable initial treatment option for symptomatic pathological vertebral compression fractures.92
While there is no current evidence favouring the use of 1 ablation technique over another for the palliation of metastatic bone disease,93 cryoablation has certain advantages. Compared to other ablation techniques, cryoablation has an intrinsic analgesic effect, with less pain for the patient during and immediately after treatment.94,95 Cryoablation can be repeated if indicated, and operators can use multiple cryoprobes simultaneously to customize the size and shape of the ablation zone.96 In addition, a radiofrequency-unsafe cardiac pacemaker is not a contra-indication to cryoablation.
While there is no clear consensus presently, oligometastatic and oligoprogressing disease with bone metastases with a size of <2 cm and no cortical erosion have been associated with a better local tumour control after ablation.97,98 Lesions to be considered for ablation should also be clearly associated with focal pain of at least moderate intensity (pain score above 4), with insufficient response to treatment options including analgesics or radiotherapy.99
Lesions that are less than 1 cm to important neural structures or organs are usually excluded. Large vessels often have a “heat sink” property which protects the vessels but also lead to an irregular and unpredictable ablation zone.100 Osteoblastic lesions are usually not amenable to ablation because of difficulty in probe deployment but may be occasionally possible if deployed adjacent to the lesion with no adjacent critical structures, such as the iliac wings.19
A myriad of applications of image-guided cryoneurolysis have been reported, from the head and neck region, intercostal, coeliac plexus, spine, inguinodynia, pudendal neuralgia, Morton’s neuroma, and other peripheral nerves.101,102
Contraindications to cryoneurolysis are similar to peripheral nerve blocks, including infection (both local and systemic), anti-coagulation, bleeding disorders, cold urticaria, and Raynaud's syndrome.103 Cryoneurolysis is also contraindicated when the target nerve contributes an important motor function, for example, the femoral nerve which may cause quadriceps muscle weakness and limits ambulation.103 Nevertheless, the risk of motor compromise may be acceptable in terminal illness and severe symptoms and necessitates an individualized approach.
Technique
Similar to other percutaneous ablation techniques, cross-sectional images are scrutinized to plan an appropriate needle trajectory, as well as number and type of cryoprobes necessary to mould the ablation zone. Cryoablation is usually well tolerated under conscious sedation.
Reported protocols for cryotherapy vary widely, ranging from a single cycle of 120 s to repeated cycles of 10 minutes, with intervening active or passive thawing.102 Since the margin of the ice ball indicates 0°C, the boundary of the ice ball should extend beyond the lesion itself by at least 5-8 mm to assure complete tumour ablation.95,104,105 To achieve the best outcome in pain reduction, the interface between normal bone and tumour should be targeted.106
Some cell damage also occurs beyond the 0°C ablation zone and should be accounted for during planning and probe positioning. Various thermal protection techniques can also be employed, including hydrodissection, pneumo-dissection, or usage of neuromonitoring devices.
For cryoneurolysis, it is critical to induce Sunderland 2 peripheral nerve injury (myelinolysis and axonolysis) to achieve clinical success and predictable outcomes. Cryoneurolysis provides longer pain relief compared with that of local injections or temporary catheter infusions, and a lower risk of formation of neuromas compared with that of heat-mediated ablation or surgical transection.102 In addition, as axons regenerate at a rate of 1-2 mm per day, the duration of analgesia is related to the distance between the cryoneurolysis site and source of pain.107 A diagnostic injection of long-acting local anaesthetic, with or without accompanying steroid, may provide critical diagnostic information prior to the actual procedure.108
Imaging guidance
CT and MRI are preferred as techniques of guidance because it is possible to visualize the ablation zone (commonly referred to as “ice ball”), ensuring complete coverage of the lesion while reducing complications to adjacent critical structures.109,110
Ultrasound can also guide the targeting of superficial structures and visualization of ice ball formation, with the added benefit of real-time Doppler assessment of vascular structures. However, the ice ball is sono-opaque and obscures visualization of deep structures, and the utility of ultrasound is limited in deep-seated lesions or in patients with large body habitus.102
Effectiveness
Bone tumours
There is mounting evidence of cryoablation for bone metastases both for palliation purposes (Figure 7), as well as curative aim in oligometastatic disease,97,111–113 which is defined as 1-5 metastases, where all metastatic sites are considered safely treatable.114 Most recently, the MOTION trial reported rapid and durable pain relief for patients with metastatic bone disease, as well as improved quality of life and maintained functional status over 6 months.112 Percutaneous cryoablation as an alternative or adjunct therapy for selected patients with plasmacytomas has also been reported.115
Figure 7.
CT-guided percutaneous cryoablation of a T10 vertebral metastatic lesion. (A) MRI T1W sagittal image revealed a hypointense lesion involving the T10 vertebral body and posterior elements (arrow). It demonstrated hyperintense signal on (B) sagittal STIR image (arrow in B) with avid enhancement on (C) axial postcontrast T1W with fat saturation (arrow in C), encroaching into the spinal canal (arrowhead in C). (D) Under CT guidance, 2 IceRod cryoablation probes (Boston Scientific, USA) were placed within the T10 vertebral lesion (arrow in D) via a posterior approach, with the more cranial probe inserted coaxially through a 13G Arrow needle (arrowheads in D). (E) Two freeze-active thaw cycles were performed, with the first freeze cycle of 3.5-min duration and the second freeze cycle of 2-min duration, with iceball visualization on CT soft tissue window (arrow in E).
Soft tissue tumour
Soft tissue sarcomas are rare tumours with multiple subtypes, the most common of which are liposarcomas and leiomyosarcomas.116 There is emerging evidence of cryoablation as a feasible and safe treatment option for these tumours, especially in recurrent or metastatic disease.117,118 Other tumour subtypes treated by cryoablation include abdominal wall endometriosis,119 neurofibromas,120 and gastrointestinal stromal tumours.121
This is especially so for extra-abdominal desmoid fibromatosis (Figure 8).111 Previously, treatment consists of a combination of surgery and radiotherapy.122 The current first-line treatment for symptomatic desmoid tumours is based on medical therapies, including tyrosine kinase inhibitors and chemotherapy (including methotrexate),123 with respective systemic side effects.
Figure 8.
Percutaneous cryoablation for desmoid fibromatosis in a 32-year-old patient. (A) MRI axial postcontrast T1W with fat saturation image revealed a heterogeneously enhancing infiltrative predominantly intramuscular mass in the left glutaeal region (arrow), representing desmoid fibromatosis. Under CT guidance, multiple cryoablation probes were deployed to customize a large ablation zone as visualized by ice formation (arrowheads). Posttreatment MRI axial postcontrast T1W with fat saturation image performed at 1 month (C) and 6 months (D) showed reduced enhancing tissue at ablation site with residual enhancement (arrow in D). The patient proceeded to undergo further cryoablation sessions at residual and recurrent sites.
Recent studies have demonstrated cryotherapy as a safe and effective treatment modality for extra-abdominal desmoid tumours, with efficacy similar to those managed via conventional approaches in the short to medium term.124,125
Cryoneurolysis/cryorhizotomy
Percutaneous image-guided neurolysis (Figures 9-12) is a safe, efficient, and relatively cost-effective means of managing refractory pain, which may arise from direct tumour invasion or iatrogenic to surgical and radiation therapy.101 Unlike heat-mediated ablation, cryoablation does not disrupt the acellular epineurium or perineurium, reducing risk of neuroma formation and potentially allowing nerve regeneration.126 Cryoneurolysis is also not associated with systemic toxicity occasionally encountered with chemical nerve ablation.127
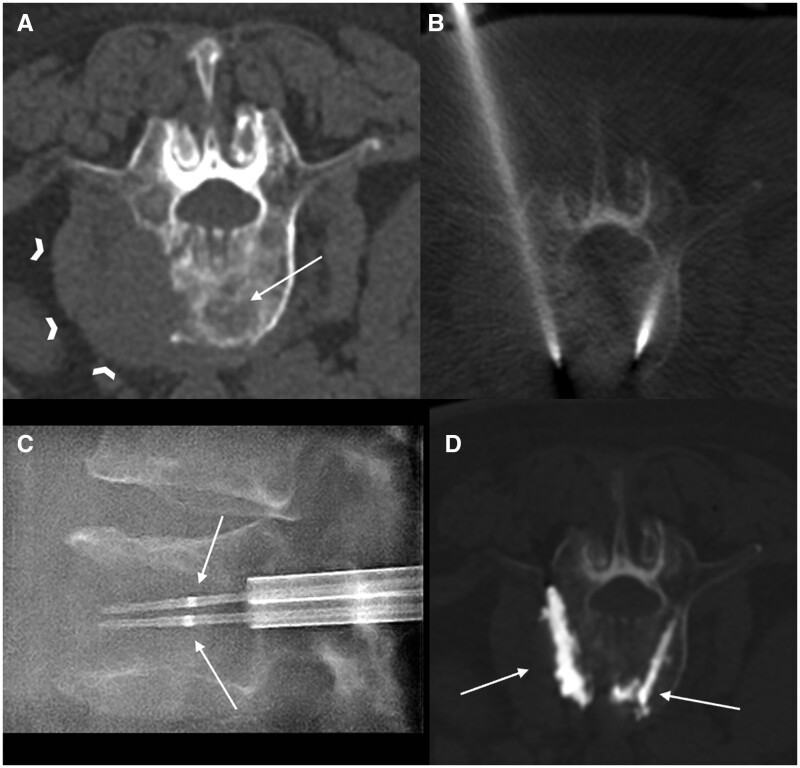
Figure 9.
Cryoablation for Morton metatarsalgia. (A) MRI long-axis T2W fat-suppressed image of the forefoot and (B) MRI short-axis T1W sagittal image at the level of the metatarsal heads revealed a T2w-hypointense (arrowheads in A) and T1w hypointense lesions in the second and third web spaces (arrows in B), in keeping with Morton neuromas. The third web space lesion was treated with cryoablation. (C) Preprocedural CT revealed soft tissue density (arrow) corresponding with MRI images. (D) An IceSeed cryoablation probe (Boston Scientific, USA) was advanced into the lesion (arrow) with adjacent iceball formation (arrowheads).
Figure 12.
Cryoablation of neurogenic tumour (40-year-old male). (A) MRI axial T2W fat-suppressed image and (B) axial T1W image revealed a small well-circumscribed T2W hyperintense (arrow in A), T1W hypointense (arrow in B) lesion closely related to the posterior left iliac bone, probably a neurogenic tumour with no overt aggressive features. (A) Under CT guidance, an IceSeed cryoablation probe (Boston Scientific, USA) was advanced into the lesion (arrow), with cryoablation performed and iceball visualized (arrowheads).
Figure 10.
Intercostal nerve cryoneurolysis. (A) CT axial bone window image revealed extensive lytic bony metastases from lung cancer, involving the left seventh rib (arrow), eighth rib (not shown), and vertebral bodies (arrowheads). (B) An IceRod cryoablation probe (Boston Scientific, USA) was advanced into the intercostal groove of the seventh rib close to the costotransverse junction. (C) CT sagittal reformats confirmed appropriate position of cryoablation probes at the intercostal grooves (arrows), with soft tissue window (D) demonstrating iceball formation (arrows). Images courtesy of Dr Alfred Tan and Dr Too Chow Wei (Singapore General Hospital).
Figure 11.
L5 cryorhizotomy for metastatic colorectal cancer in a 65-year-old male. Preprocedural CT images in (A) axial bone window, (B) axial soft tissue window, and (C) sagittal bone window revealed an expansile aggressive sclerotic lesion in the L5 vertebral body and right posterior elements (arrowheads in A) in keeping with bony metastases, invading the L5-S1 neural foramen (arrows in B and C). (D) Under CT guidance, an IceForce cryoablation probe (Boston Scientific, USA) was advanced into the lesion (arrow) and commenced with CT soft tissue window demonstrating iceball formation (arrowheads).
Vascular malformation
Cryotherapy has also been evaluated as a treatment option for vascular malformations, in particular venous malformations (Figure 13) and fibroadipose vascular anomalies, either as a first-line treatment128 or second-line treatment after sclerotherapy.129 A systematic review of cryoablation in the treatment of venous malformations (55 lesions) reported promising results in terms of lesion size decrease and symptom improvement, with weighted mean postprocedural decrease in lesion size of 92.0%, weighted mean reduction in pain score of 77%, and complete resolution of symptoms (35/55) (63.6%).130
Figure 13.
Cryoablation for a vascular lesion (52-year-old female). Pretreatment MRI elbow with (A) axial T1W, (B) axial T2W, (C) coronal STIR, and (D) axial postcontrast T1W with fat saturation images. There was a lobulated T1W-mildly hyperintense mass-like lesion at the supracondylar region of the humerus (arrow in A), T2W-hyperintense with thin internal septation (arrows in B and C) and mild enhancement postgadolinium administration (arrow in D). (E) Under CT guidance, an IceRod cryoablation probe (Boston Scientific, USA) was advanced into the lesion (arrow); with cryoablation performed and iceball visualized in soft tissue window (arrowheads).
Complications
Bone tumour cryoablation is safe, with a reported 2.5% rate of major complications, most commonly secondary fracture (1.2%). Major complications are associated with age greater than 70 years and use of more than 3 cryoprobes.131
The most important potential complication of thermal ablation of skeletal metastases remains nontarget thermal injury to the spinal cord or adjacent neural structures. These are usually transient and typically managed with local injection of steroids and long-acting anaesthetic agents.13 Knowledge of neuroanatomy is crucial and can prevent inadvertent thermal injury.132 Thermal injury to the vital organs and skin should be minimized by implementing thermal protection strategies such as hydrodissection or pneumo-dissection.
Other complications vary depending on the site of ablation. When treating lesions near the skin, skin necrosis is a consideration. Caution should be taken when treating lesions near joint to avoid joint effusion, synovitis, and osteonecrosis.
Other potential side effects of cryoneurolysis include bleeding, bruising, and rarely infection.103 If the target nerve is superficial, skin and hair in the adjacent region may be affected, including hyperpigmentation, depigmentation, and alopecia, particularly near the eyebrow when treating the supraorbital nerve.133
Combined
Indications
Percutaneous cementoplasty and thermal ablation have synergistic properties. Given a potential complication of pathological fracture due to cryoablation-induced necrosis, adjunct cementoplasty has successfully been performed and demonstrated durable pain relief and stabilization (Figure 14).134–136 Cementoplasty has little to no antitumoural effect and has been reported to transiently increase the level of circulating cancer cells in the minutes following injection.137 Therefore, ablation prior to cement injection may be indicated in cases requiring local tumour control or tumour debulking to prevent complications, such as a growing lesion near a nerve, improve the quality of consolidation and decrease tumour seeding.
Figure 14.
Combined cryoablation and cementoplasty for hepatocellular carcinoma metastases to the left iliac bone. (A) MRI axial T2W with fat suppression image revealed an expansile hyperintense lesion in the left iliac crest (arrow) with extraosseous component at the glutaeal muscles (arrowheads) suspicious for metastases, and (B) MRI coronal T1W image demonstrated craniocaudal extent of the lesion adjacent to the acetabular roof (arrow). (B) Under CT guidance, 2 bone access cannulas were advanced into the lesion, with position confirmed on oblique sagittal reformats (arrows). (D) Cryoablation probes were advanced into the lesion coaxially (arrow). (E) Cryoablation was performed with iceball formation visualized on CT axial soft tissue window (arrow). (F) Cementoplasty was subsequently performed under fluoroscopic guidance (arrow). (G) Postprocedural CT revealed satisfactory cement fill (arrow) with no leakage or immediate complication.
Effectiveness
Other ablation techniques such as radiofrequency ablation and microwave ablation with concurrent stabilization have also yielded effective results.138–142 In the authors’ experience, temperature-controlled radiofrequency ablation (“coablation”) of vertebral metastases provided satisfactory results in selected patients who have limited pain relief despite prior radiotherapy and opioid titration (Figure 15); similar results have been reported for pelvic and acetabular metastases.143
Figure 15.
Combined ablation and vertebroplasty for L3 vertebral body breast cancer metastases in a 62-year-old female. (A) Preprocedural CT images revealed lucent lesions in the vertebral body anteriorly (arrow in A), as well as a sizeable paravertebral extraosseous component (arrowheads in A). (B) Under CT guidance, a bilateral transpedicular approach was performed, with bone access cannulas in place. (C) Fluoroscopy lateral image confirmed satisfactory placement of the Osteocool (Medtronic, Ireland) radiofrequency ablation probes within the vertebral body, with posterior margins of the ablation zones indicated by the radio-opaque markers (arrows in C). (D) Postprocedural images revealed satisfactory cement fill of the metastatic lesions (arrows in D), with no cement leak.
To date, there are limited data comparing the effectiveness of combined cryoablation and cementoplasty procedures versus other combined ablation-stabilization techniques, partly due to the complexity of conducting randomized trials regarding individualized therapy for palliative procedures.144
It is also difficult to evaluate the exact benefit of combined ablation and cementoplasty versus cementoplasty alone, especially for bony lesions without extraosseous component.140,145 In a retrospective study of 35 patients, Wang et al146 reported a better analgesic effect, increased cement injected, and lower cement leakage rates from combined RFA and vertebroplasty, compared to vertebroplasty alone.
Conclusion
There have been substantial advancements and mounting evidence of safety and efficacy in percutaneous image-guided procedures for the musculoskeletal system, including cementoplasty and cryoablation techniques. These enable the interventional radiologist to offer a diverse repertoire of options in a multidisciplinary approach to patient management, especially cancer patients with bony metastases.
Acknowledgements
The authors would like to thank the following from the Department of Interventional Radiology, Singapore General Hospital, for their contributions towards the figures in the article: Dr Alexander Tan, Dr Mark Wang, Dr Zhuang Kun Da, Dr Alfred Tan, and Dr Too Chow Wei. This review article is an invited submission as per correspondence with Ms Miranda Ashby-Wood, Professor Peter Munk, and Dr Yet Yen Yan.
Contributor Information
Jin Rong Tan, Department of Diagnostic Radiology, Singapore General Hospital, Singapore Health Service (SingHealth), Singapore 169608, Singapore.
Yet Yen Yan, Department of Radiology, Changi General Hospital, Singapore Health Service (SingHealth), Singapore 529889, Singapore.
Adnan Sheikh, Musculoskeletal Radiology Division, Vancouver General Hospital, BC, V5Z 1M9, Canada.
Hugue Ouellette, Musculoskeletal Radiology Division, Vancouver General Hospital, BC, V5Z 1M9, Canada.
Paul Mallinson, Musculoskeletal Radiology Division, Vancouver General Hospital, BC, V5Z 1M9, Canada.
Peter L Munk, Musculoskeletal Radiology Division, Vancouver General Hospital, BC, V5Z 1M9, Canada.
Funding
The authors declare that there is no funding source in the publication of this article.
Conflicts of interest
The authors declare that there is no conflict of interest in the publication of this article.
References
- 1. Zajączkowska R, Kocot-Kępska M, Leppert W, Wordliczek J.. Bone pain in cancer patients: mechanisms and current treatment. Int J Mol Sci. 2019;20(23). 10.3390/IJMS20236047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Ryan C, Stoltzfus KC, Horn S, et al. Epidemiology of bone metastases. Bone. 2022;158:115783. 10.1016/J.BONE.2020.115783 [DOI] [PubMed] [Google Scholar]
- 3. Saravana-Bawan S, David E, Sahgal A, Chow E.. Palliation of bone metastases—exploring options beyond radiotherap. Ann Palliat Med. 2019;8(2):168-177. 10.21037/APM.2018.12.04 [DOI] [PubMed] [Google Scholar]
- 4. Coleman RE, Croucher PI, Padhani AR, et al. Bone metastases. Nat Rev Dis Primers. 2020;6(1):83. 10.1038/S41572-020-00216-3 [DOI] [PubMed] [Google Scholar]
- 5. Nguyen TK, Sahgal A, Dagan R, et al. Stereotactic body radiation therapy for nonspine bone metastases: international practice patterns to guide treatment planning. Pract Radiat Oncol. 2020;10(6):e452-e460. 10.1016/J.PRRO.2020.02.011 [DOI] [PubMed] [Google Scholar]
- 6. Kougioumtzopoulou A, Zygogianni A, Liakouli Z, Kypraiou E, Kouloulias V.. The role of radiotherapy in bone metastases: a critical review of current literature. Eur J Cancer Care (Engl). 2017;26(6). 10.1111/ECC.12724 [DOI] [PubMed] [Google Scholar]
- 7. van der Velden JM, van der Linden YM, Versteeg AL, et al. Evaluation of effectiveness of palliative radiotherapy for bone metastases: a prospective cohort study. J Radiat Oncol. 2018;7(4):325-333. 10.1007/S13566-018-0363-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Errani C, Bazzocchi A, Spinnato P, et al. What’s new in management of bone metastases? Eur J Orthop Surg Traumatol. 2019;29(7):1367-1375. 10.1007/S00590-019-02446-Y [DOI] [PubMed] [Google Scholar]
- 9. Paice JA. Cancer pain management and the opioid crisis in America: how to preserve hard‐earned gains in improving the quality of cancer pain management. Cancer. 2018;124(12):2491-2497. 10.1002/cncr.31303 [DOI] [PubMed] [Google Scholar]
- 10. World Health Organization. WHO Guidelines for the Pharmacological and Radiotherapeutic Management of Cancer Pain in Adults and Adolescents. Geneva; 2018. Licence: CC BY-NC-SA 3.0 IGO. [PubMed] [Google Scholar]
- 11. Mastier C, Gjorgjievska A, Thivolet A, et al. Musculoskeletal metastases management: the interventional radiologist’s toolbox. Semin Intervent Radiol. 2018;35(4):281-289. 10.1055/S-0038-1673420 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Chen-Xu S, Martel-Villagrán J, Bueno-Horcajadas Á.. Percutaneous management of bone metastases: state of the art. Radiologia (Engl Ed). 2021;63(4):345-357. 10.1016/J.RX.2021.02.006 [DOI] [PubMed] [Google Scholar]
- 13. Tomasian A, Jennings JW.. Bone metastases: state of the art in minimally invasive interventional oncology. Radiographics. 2021;41(5):1475-1492. 10.1148/rg.2021210007 [DOI] [PubMed] [Google Scholar]
- 14. Delpla A, Tselikas L, De Baere T, et al. Correction to: preventive vertebroplasty for long-term consolidation of vertebral metastases. Cardiovasc Intervent Radiol. 2020;43(5):807. 10.1007/S00270-020-02445-1 [DOI] [PubMed] [Google Scholar]
- 15. Xu W, Wang S, Chen C, et al. Correlation analysis between the magnetic resonance imaging characteristics of osteoporotic vertebral compression fractures and the efficacy of percutaneous vertebroplasty: a prospective cohort study. BMC Musculoskelet Disord. 2018;19(1):114. 10.1186/S12891-018-2040-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Mallinson PI, Munk PL.. Musculoskeletal interventional techniques pitfalls. In: Peh W.C.G., eds. Pitfalls in Musculoskeletal Radiology, Cham, Switzerland: Springer; 2017:139-148. 10.1007/978-3-319-53496-1_8/COVER [DOI] [Google Scholar]
- 17. Fisher CG, DiPaola CP, Ryken TC, et al. A novel classification system for spinal instability in neoplastic disease: an evidence-based approach and expert consensus from the spine oncology study group. Spine (Phila Pa 1976). 2010;35(22):E1221-E1229. 10.1097/BRS.0B013E3181E16AE2 [DOI] [PubMed] [Google Scholar]
- 18. Patchell RA, Tibbs PA, Regine WF, et al. Direct decompressive surgical resection in the treatment of spinal cord compression caused by metastatic cancer: a randomised trial. Lancet. 2005;366(9486):643-648. 10.1016/S0140-6736(05)66954-1 [DOI] [PubMed] [Google Scholar]
- 19. Jayaram PR, Yan YY, Mallinson PI, Ouellette HA, Munk PL.. Interventional radiologic techniques in the management of bone tumors. In: Heymann D, ed., Bone Cancer: Bone Sarcomas and Bone Metastases—From Bench to Bedside, London, UK: Academic Press; 2022:807-825. 10.1016/B978-0-12-821666-8.00024-4 [DOI] [Google Scholar]
- 20. Seah SJ, Yeo MH, Tan JH, Hey HWD.. Early cement augmentation may be a good treatment option for pain relief for osteoporotic compression fractures: a systematic review and meta-analysis. Eur Spine J. 2023;32(5):1751-1762. 10.1007/S00586-023-07658-9 [DOI] [PubMed] [Google Scholar]
- 21. Yu SW, Lee PC, Ma CH, Chuang TY, Chen YJ.. Vertebroplasty for the treatment of osteoporotic compression spinal fracture: comparison of remedial action at different stages of injury. J Trauma. 2004;56(3):629-632. 10.1097/01.TA.0000053471.73514.2E [DOI] [PubMed] [Google Scholar]
- 22. Diamond TH, Hartwell T, Clarke W, Manoharan A.. Percutaneous vertebroplasty for acute vertebral body fracture and deformity in multiple myeloma: a short report. Br J Haematol. 2004;124(4):485-487. 10.1111/J.1365-2141.2004.04809.X [DOI] [PubMed] [Google Scholar]
- 23. Saliou G, Kocheida EM, Lehmann P, et al. Percutaneous vertebroplasty for pain management in malignant fractures of the spine with epidural involvement. Radiology. 2010;254(3):882-890. 10.1148/RADIOL.09081698 [DOI] [PubMed] [Google Scholar]
- 24. Kassamali RH, Ganeshan A, Hoey ETD, Crowe PM, Douis H, Henderson J.. Pain management in spinal metastases: the role of percutaneous vertebral augmentation. Ann Oncol. 2011;22(4):782-786. 10.1093/ANNONC/MDQ605 [DOI] [PubMed] [Google Scholar]
- 25. Corcos G, Dbjay J, Mastier C, et al. Cement leakage in percutaneous vertebroplasty for spinal metastases: a retrospective evaluation of incidence and risk factors. Spine (Phila Pa 1976). 2014;39(5):E332-E338. 10.1097/BRS.0000000000000134 [DOI] [PubMed] [Google Scholar]
- 26. Mehdizade A, Payer M, Somon T, et al. Percutaneous vertebroplasty through a transdiscal access route after lumbar transpedicular instrumentation. Spine J. 2004;4(4):475-479. 10.1016/j.spinee.2003.08.032 [DOI] [PubMed] [Google Scholar]
- 27. Martin J-B, Gailloud P, Dietrich P-Y, et al. Direct transoral approach to C2 for percutaneous vertebroplasty. Cardiovasc Intervent Radiol. 2002;25(6):517-519. 10.1007/S00270-001-0122-7/METRICS [DOI] [PubMed] [Google Scholar]
- 28. Kortman K, Ortiz O, Miller T, et al. Multicenter study to assess the efficacy and safety of sacroplasty in patients with osteoporotic sacral insufficiency fractures or pathologic sacral lesions. J Neurointerv Surg. 2013;5(5):461-466. 10.1136/NEURINTSURG-2012-010347 [DOI] [PubMed] [Google Scholar]
- 29. Soon WC, Mathew RK, Timothy J.. Comparison of vertebroplasty using directional versus straight needle. Acta Radiol Open. 2015;4(3):2047981615569268. 10.1177/2047981615569268 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Murphy DT, Korzan JR, Ouellette HA, Liu DM, Clarkson PW, Munk PL.. Driven around the bend: novel use of a curved steerable needle. Cardiovasc Intervent Radiol. 2013;36(2):531-535. 10.1007/S00270-012-0482-1 [DOI] [PubMed] [Google Scholar]
- 31. Auloge P, Cazzato RL, Ramamurthy N, et al. Augmented reality and artificial intelligence-based navigation during percutaneous vertebroplasty: a pilot randomised clinical trial. Eur Spine J. 2020;29(7):1580-1589. 10.1007/S00586-019-06054-6/METRICS [DOI] [PubMed] [Google Scholar]
- 32. Rauch A, Cazzato RL, Garnon J, et al. MRI-guided cryoablation of in-transit metastases from cutaneous melanoma: a brief report on a preliminary experience. Cardiovasc Intervent Radiol. 2017;40(8):1285-1289. 10.1007/S00270-017-1645-X/METRICS [DOI] [PubMed] [Google Scholar]
- 33. Autrusseau P-A, Cazzato RL, De Marini P, et al. Percutaneous MR-guided cryoablation of low-flow vascular malformation: technical feasibility, safety and clinical efficacy. Cardiovasc Intervent Radiol. 2020;43(6):858-865. 10.1007/S00270-020-02455-Z [DOI] [PubMed] [Google Scholar]
- 34. Voormolen MHJ, Lohle PN, Lampmann LE, et al. Prospective clinical follow-up after percutaneous vertebroplasty in patients with painful osteoporotic vertebral compression fractures. J Vasc Interv Radiol. 2006;17(8):1313-1320. 10.1097/01.RVI.0000231952.75209.4A [DOI] [PubMed] [Google Scholar]
- 35. Klazen CAH, Lohle PNM, de Vries J, et al. Vertebroplasty versus conservative treatment in acute osteoporotic vertebral compression fractures (Vertos II): an open-label randomised trial. Lancet. 2010;376(9746):1085-1092. 10.1016/S0140-6736(10)60954-3 [DOI] [PubMed] [Google Scholar]
- 36. Buchbinder R, Osborne RH, Ebeling PR, et al. A randomized trial of vertebroplasty for painful osteoporotic vertebral fractures. N Engl J Med. 2009;361(6):557-568. 10.1056/NEJMOA0900429/SUPPL_FILE/NEJM_BUCHBINDER_557SA1.PDF [DOI] [PubMed] [Google Scholar]
- 37. Kallmes DF, Comstock BA, Heagerty PJ, et al. A randomized controlled trial of vertebroplasty for osteoporotic spine fractures. N Engl J Med. 2009;361(6):569-579. 10.1056/NEJMOA0900563 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Buchbinder R, Johnston RV, Rischin KJ, et al. ; Cochrane Musculoskeletal Group. Percutaneous vertebroplasty for osteoporotic vertebral compression fracture. Cochrane Database Syst Rev. 2018;2018(11). 10.1002/14651858.CD006349.PUB4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Firanescu CE, de Vries J, Lodder P, et al. Vertebroplasty versus sham procedure for painful acute osteoporotic vertebral compression fractures (VERTOS IV): randomised sham controlled clinical trial. BMJ. 2018;361:k1551. 10.1136/BMJ.K1551 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. De Leacy R, Chandra RV, Barr JD, et al. The evidentiary basis of vertebral augmentation: a 2019 update. J Neurointerv Surg. 2020;12(5):442-447. 10.1136/NEURINTSURG-2019-015026 [DOI] [PubMed] [Google Scholar]
- 41. Clark W, Bird P, Gonski P, et al. Safety and efficacy of vertebroplasty for acute painful osteoporotic fractures (VAPOUR): a multicentre, randomised, double-blind, placebo-controlled trial. Lancet. 2016;388(10052):1408-1416. 10.1016/S0140-6736(16)31341-1 [DOI] [PubMed] [Google Scholar]
- 42. Firanescu CE, Venmans A, de Vries J, et al. Predictive factors for sustained pain after (sub)acute osteoporotic vertebral fractures. combined results from the VERTOS II and VERTOS IV trial. Cardiovasc Intervent Radiol. 2022;45(9):1314-1321. 10.1007/S00270-022-03170-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Bae JW, Gwak H-S, Kim S, et al. Percutaneous vertebroplasty for patients with metastatic compression fractures of the thoracolumbar spine: clinical and radiological factors affecting functional outcomes. Spine J. 2016;16(3):355-364. 10.1016/j.spinee.2015.11.033 [DOI] [PubMed] [Google Scholar]
- 44. Onggo JR, Maingard JT, Nambiar M, Buckland A, Chandra RV, Hirsch JA.. Role of vertebroplasty and balloon kyphoplasty in pathological fracture in myeloma: a narrative review. Eur Spine J. 2021;30(10):2825-2838. 10.1007/S00586-021-06955-5/METRICS [DOI] [PubMed] [Google Scholar]
- 45. Anselmetti GC, Manca A, Montemurro F, et al. Percutaneous vertebroplasty in multiple myeloma: prospective long-term follow-up in 106 consecutive patients. Cardiovasc Intervent Radiol. 2012;35(1):139-145. 10.1007/S00270-011-0111-4/METRICS [DOI] [PubMed] [Google Scholar]
- 46. Hao J, Hu Z.. Percutaneous cement vertebroplasty in the treatment of symptomatic vertebral hemangiomas. Pain Physician. 2012;15(1):43-49. [PubMed] [Google Scholar]
- 47. Barr JD, Jensen ME, Hirsch JA, et al. ; Society of Neurointerventional Surgery. Position statement on percutaneous vertebral augmentation: a consensus statement developed by the Society of Interventional Radiology (SIR), American Association of Neurological Surgeons (AANS) and the Congress of Neurological Surgeons (CNS), American College of Radiology (ACR), American Society of Neuroradiology (ASNR), American Society of Spine Radiology (ASSR), Canadian Interventional Radiology Association (CIRA), and the Society of NeuroInterventional Surgery (SNIS). J Vasc Interv Radiol. 2014;25(2):171-181. 10.1016/J.JVIR.2013.10.001 [DOI] [PubMed] [Google Scholar]
- 48. Hirsch JA, Zini C, Anselmetti GC, et al. Vertebral augmentation: is it time to get past the pain? A consensus statement from the sardinia spine and stroke congress. Medicina (Lithuania). 2022;58(10):1431. 10.3390/medicina58101431 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Lee J, Lee E, Lee JW, Kang Y, Ahn JM, Kang HS.. Percutaneous sacroplasty: effectiveness and long-term outcome predictors. J Korean Neurosurg Soc. 2020;63(6):747-756. 10.3340/JKNS.2020.0014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Andresen JR, Radmer S, Andresen R, et al. Comparative outcome of different treatment options for fragility fractures of the sacrum. BMC Musculoskelet Disord. 2022;23(1):1106. 10.1186/S12891-022-06039-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Liu HF, Wu CG, Tian QH, Wang T, Yi F.. Application of percutaneous osteoplasty in treating pelvic bone metastases: efficacy and safety. Cardiovasc Intervent Radiol. 2019;42(12):1738-1744. 10.1007/S00270-019-02320-8 [DOI] [PubMed] [Google Scholar]
- 52. Moser TP, Onate M, Achour K, Freire V.. Cementoplasty of pelvic bone metastases: systematic assessment of lesion filling and other factors that could affect the clinical outcomes. Skeletal Radiol. 2019;48(9):1345-1355. 10.1007/S00256-019-3156-0/METRICS [DOI] [PubMed] [Google Scholar]
- 53. Cazzato RL, Palussière J, Buy X, et al. Percutaneous long bone cementoplasty for palliation of malignant lesions of the limbs: a systematic review. Cardiovasc Intervent Radiol. 2015;38(6):1563-1572. 10.1007/S00270-015-1082-7 [DOI] [PubMed] [Google Scholar]
- 54. Park JW, Lim H-j, Kang HG, Kim JH, Kim HS.. Percutaneous cementoplasty for the pelvis in bone metastasis: 12-year experience. Ann Surg Oncol. 2022;29(2):1413-1422. 10.1245/S10434-021-10640-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Deschamps F, Farouil G, Hakime A, et al. Cementoplasty of metastases of the proximal femur: is it a safe palliative option? J Vasc Interv Radiol. 2012;23(10):1311-1316. 10.1016/J.JVIR.2012.06.027 [DOI] [PubMed] [Google Scholar]
- 56. Kitridis D, Saccomanno MF, Maccauro G, Givissis P, Chalidis B.. Augmented versus non-augmented percutaneous cementoplasty for the treatment of metastatic impending fractures of proximal femur: a systematic review. Injury. 2020;51(Suppl 3):S66-S72. 10.1016/J.INJURY.2020.02.045 [DOI] [PubMed] [Google Scholar]
- 57. Barral M, Auperin A, Hakime A, et al. Percutaneous thermal ablation of breast cancer metastases in oligometastatic patients. Cardiovasc Intervent Radiol. 2016;39(6):885-893. 10.1007/S00270-016-1301-X [DOI] [PubMed] [Google Scholar]
- 58. Sadeghi-Naini M, Aarabi S, Shokraneh F, Janani L, Vaccaro AR, Rahimi-Movaghar V.. Vertebroplasty and kyphoplasty for metastatic spinal lesions. Clin Spine Surg. 2018;31(5):203-210. 10.1097/BSD.0000000000000601 [DOI] [PubMed] [Google Scholar]
- 59. Hirsch JA, Chandra RV, Carter NS, Beall D, Frohbergh M, Ong K.. Number needed to treat with vertebral augmentation to save a life. AJNR Am J Neuroradiol. 2020;41(1):178-182. 10.3174/AJNR.A6367 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Bae JW, Gwak H-S, Kim S, et al. Percutaneous vertebroplasty for patients with metastatic compression fractures of the thoracolumbar spine: clinical and radiological factors affecting functional outcomes. Spine J. 2016;16(3):355-364. 10.1016/j.spinee.2015.11.033 [DOI] [PubMed] [Google Scholar]
- 61. Dalton BE, Kohm AC, Miller LE, Block JE, Poser RD.. Radiofrequency-targeted vertebral augmentation versus traditional balloon kyphoplasty: radiographic and morphologic outcomes of an ex vivo biomechanical pilot study. Clin Interv Aging. 2012;7:525-531. 10.2147/CIA.S37025 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Zhang H-r, Xu M-y, Yang X-g, Qiao R-q, Li J-k, Hu Y-c.. Percutaneous vertebral augmentation procedures in the management of spinal metastases. Cancer Lett. 2020;475:136-142. 10.1016/J.CANLET.2020.01.038 [DOI] [PubMed] [Google Scholar]
- 63. Luo Y, Yang DM, Yang HM, Wu D, Xie FY.. Innovative minimally invasive implants for osteoporosis vertebral compression fractures. Front Med (Lausanne). 2023;10:1161174. 10.3389/fmed.2023.1161174 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Noriega D, Marcia S, Theumann N, et al. A prospective, international, randomized, noninferiority study comparing an implantable titanium vertebral augmentation device versus balloon kyphoplasty in the reduction of vertebral compression fractures (SAKOS study). Spine J. 2019;19(11):1782-1795. 10.1016/J.SPINEE.2019.07.009 [DOI] [PubMed] [Google Scholar]
- 65. Garnon J, Jennings JW, Meylheuc L, et al. Biomechanics of the osseous pelvis and its implication for consolidative treatments in interventional oncology. Cardiovasc Intervent Radiol. 2020;43(11):1589-1599. 10.1007/S00270-020-02624-0/METRICS [DOI] [PubMed] [Google Scholar]
- 66. Deschamps F, de Baere T, Hakime A, et al. Percutaneous osteosynthesis in the pelvis in cancer patients. Eur Radiol. 2016;26(6):1631-1639. 10.1007/S00330-015-3971-1 [DOI] [PubMed] [Google Scholar]
- 67. Lee FY, Latich I, Toombs C, et al. Minimally invasive image-guided Ablation, Osteoplasty, Reinforcement, and Internal Fixation (AORIF) for osteolytic lesions in the pelvis and periarticular regions of weight-bearing bones. J Vasc Interv Radiol. 2020;31(4):649-658.e1. 10.1016/J.JVIR.2019.11.029 [DOI] [PubMed] [Google Scholar]
- 68. Pusceddu C, Fancellu A, Ballicu N, Fele RM, Sotgia B, Melis L.. CT-guided percutaneous screw fixation plus cementoplasty in the treatment of painful bone metastases with fractures or a high risk of pathological fracture. Skeletal Radiol. 2017;46(4):539-545. 10.1007/S00256-017-2584-Y [DOI] [PubMed] [Google Scholar]
- 69. Roux C, Tselikas L, Delpla A, et al. Percutaneous fixation of impending fracture of the hip. Tech Vasc Interv Radiol. 2022;25(1):100802. 10.1016/j.tvir.2022.100802 [DOI] [PubMed] [Google Scholar]
- 70. Cornelis FH, Razakamanantsoa L, Ammar MB, et al. Percutaneous screw fixation of pelvic bone metastases using cone-beam computed tomography navigation. Diagn Interv Imaging. 2022;103(7-8):367-374. 10.1016/j.diii.2022.01.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Kastler A, Cornelis FH, Kastler B.. Patient’s selection and evaluation for bone stabilization. Tech Vasc Interv Radiol. 2022;25(1):100797. 10.1016/J.TVIR.2022.100797 [DOI] [PubMed] [Google Scholar]
- 72. Trumm CG, Pahl A, Helmberger TK, et al. CT fluoroscopy-guided percutaneous vertebroplasty in spinal malignancy: technical results, PMMA leakages, and complications in 202 patients. Skeletal Radiol. 2012;41(11):1391-1400. 10.1007/S00256-012-1365-X/METRICS [DOI] [PubMed] [Google Scholar]
- 73. Pitton MB, Herber S, Koch U, Oberholzer K, Drees P, Düber C.. CT-guided vertebroplasty: analysis of technical results, extraosseous cement leakages, and complications in 500 procedures. Eur Radiol. 2008;18(11):2568-2578. 10.1007/S00330-008-1020-Z/METRICS [DOI] [PubMed] [Google Scholar]
- 74. Yevich S, Tselikas L, Gravel G, De Baère T, Deschamps F.. Percutaneous cement injection for the palliative treatment of osseous metastases: a technical review. Semin Intervent Radiol. 2018;35(4):268-280. 10.1055/s-0038-1673418 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Chen YJ, Chang GC, Chen WH, Hsu HC, Lee TS.. Local metastases along the tract of needle: a rare complication of vertebroplasty in treating spinal metastases. Spine (Phila Pa 1976). 2007;32(21):E615-E618. 10.1097/BRS.0B013E318154C5E8 [DOI] [PubMed] [Google Scholar]
- 76. Lee CH, Lee JW, Hyun SJ, Kim KJ, Jahng TA, Kim HJ.. Needle-tract seeding after percutaneous vertebroplasty: a case report. Spine (Phila Pa 1976). 2014;39(12):E752-E756. 10.1097/BRS.0000000000000329 [DOI] [PubMed] [Google Scholar]
- 77. Barragán-Campos HM, Vallée J-N, Lo D, et al. Percutaneous vertebroplasty for spinal metastases: complications. Radiology. 2006;238(1):354-362. 10.1148/RADIOL.2381040841 [DOI] [PubMed] [Google Scholar]
- 78. Shi X, Cui Y, Pan Y, Wang B, Lei M.. Epidemiology and detection of cement leakage in patients with spine metastases treated with percutaneous vertebroplasty: a 10-year observational study. J Bone Oncol. 2021;28:100365. 10.1016/j.jbo.2021.100365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Laredo JD, Chiras J, Kemel S, Taihi L, Hamze B.. Vertebroplasty and interventional radiology procedures for bone metastases. Joint Bone Spine. 2018;85(2):191-199. 10.1016/J.JBSPIN.2017.05.005 [DOI] [PubMed] [Google Scholar]
- 80. Friedman MV, Hillen TJ, Wessell DE, Hildebolt CF, Jennings JW.. Hip chondrolysis and femoral head osteonecrosis: a complication of periacetabular cryoablation. J Vasc Interv Radiol. 2014;25(10):1580-1588. 10.1016/J.JVIR.2014.06.016 [DOI] [PubMed] [Google Scholar]
- 81. Choe DH, Marom EM, Ahrar K, Truong MT, Madewell JE.. Pulmonary embolism of polymethyl methacrylate during percutaneous vertebroplasty and kyphoplasty. AJR Am J Roentgenol. 2012;183(4):1097-1102. 10.2214/AJR.183.4.1831097 [DOI] [PubMed] [Google Scholar]
- 82. Syed MI, Jan S, Patel NA, Shaikh A, Marsh RA, Stewart RV.. Fatal fat embolism after vertebroplasty: identification of the high-risk patient. AJNR Am J Neuroradiol. 2006;27(2):343. [PMC free article] [PubMed] [Google Scholar]
- 83. Heini PF, Orler R.. Vertebroplastik bei hochgradiger osteoporose: technik und erfahrung mit plurisegmentalen injektionen. Orthopade. 2004;33(1):22-30. 10.1007/S00132-003-0574-3/METRICS [DOI] [PubMed] [Google Scholar]
- 84. Tomasian A, Jennings JW.. Percutaneous minimally invasive thermal ablation for management of osseous metastases: recent advances. Int J Hyperthermia. 2019;36(2):3-12. 10.1080/02656736.2019.1613573 [DOI] [PubMed] [Google Scholar]
- 85. Arrigoni F, Bianchi G, Formiconi F, et al. CT-guided cryoablation for management of bone metastases: a single center experience and review of the literature. Radiol Med. 2022;127(2):199-205. 10.1007/S11547-021-01437-6 [DOI] [PubMed] [Google Scholar]
- 86. Erinjeri JP, Clark TWI.. Cryoablation: mechanism of action and devices. J Vasc Interv Radiol. 2010;21(Suppl 8):S187-S191. 10.1016/j.jvir.2009.12.403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Casali PG, Bielack S, Abecassis N, et al. ; ESMO Guidelines Committee, PaedCan and ERN EURACAN. Bone sarcomas: ESMO–PaedCan–EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2018;29(Suppl 4):iv79-iv95. 10.1093/ANNONC/MDY310 [DOI] [PubMed] [Google Scholar]
- 88. Liu DM, Kee ST, Loh CT, et al. Cryoablation of osteoid osteoma: two case reports. J Vasc Interv Radiol. 2010;21(4):586-589. 10.1016/j.jvir.2009.12.389 [DOI] [PubMed] [Google Scholar]
- 89. De Filippo M, Russo U, Papapietro VR, et al. Radiofrequency ablation of osteoid osteoma. Acta Biomed. 2018;89(1-S):175-185. 10.23750/ABM.V89I1-S.7021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Le Corroller T, Vives T, Mattei J-C, et al. Osteoid osteoma: percutaneous CT-guided cryoablation is a safe, effective, and durable treatment option in adults. Radiology. 2022;302(2):392-399. 10.1148/RADIOL.2021211100/ASSET/IMAGES/LARGE/RADIOL.2021211100FIG6.JPEG [DOI] [PubMed] [Google Scholar]
- 91. NCCN Guidelines: Adult Cancer Pain; 2023. Accessed June 02, 2023. https://www.nccn.org/guidelines/category_3
- 92. Khan MA, Jennings JW, Baker JC, et al. ; Expert Panels on Neurological Imaging, Interventional Radiology, and Musculoskeletal Imaging. ACR Appropriateness Criteria® management of vertebral compression fractures: 2022 update. J Am Coll Radiol. 2023;20(5S):S102-S124. 10.1016/J.JACR.2023.02.015 [DOI] [PubMed] [Google Scholar]
- 93. Gennaro N, Sconfienza LM, Ambrogi F, Boveri S, Lanza E.. Thermal ablation to relieve pain from metastatic bone disease: a systematic review. Skeletal Radiol. 2019;48(8):1161-1169. 10.1007/S00256-018-3140-0/METRICS [DOI] [PubMed] [Google Scholar]
- 94. Rose PS, Morris JM.. Cryosurgery/cryoablation in musculoskeletal neoplasms: history and state of the art. Curr Rev Musculoskelet Med. 2015;8(4):353-360. 10.1007/S12178-015-9307-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Masala S, et al. Percutaneous ablative treatment of metastatic bone tumours: visual analogue scale scores in a short-term series. Singapore Med J. 2011;52(3):182-189. Accessed May 30, 2023. https://europepmc.org/article/med/21451927 [PubMed] [Google Scholar]
- 96. Callstrom MR, Kurup AN.. Percutaneous ablation for bone and soft tissue metastases-why cryoablation? Skeletal Radiol. 2009;38(9):835-839. 10.1007/S00256-009-0736-4 [DOI] [PubMed] [Google Scholar]
- 97. Deschamps F, Farouil G, Ternes N, et al. Thermal ablation techniques: a curative treatment of bone metastases in selected patients? Eur Radiol. 2014;24(8):1971-1980. 10.1007/S00330-014-3202-1 [DOI] [PubMed] [Google Scholar]
- 98. Luigi Cazzato R, Auloge P, De Marini P, et al. Percutaneous image-guided ablation of bone metastases: local tumor control in oligometastatic patients. Int J Hyperthermia. 2018;35(1):493-499. 10.1080/02656736.2018.1508760 [DOI] [PubMed] [Google Scholar]
- 99. Callstrom MR, Charboneau JW.. Image-guided palliation of painful metastases using percutaneous ablation. Tech Vasc Interv Radiol. 2007;10(2):120-131. 10.1053/J.TVIR.2007.09.003 [DOI] [PubMed] [Google Scholar]
- 100. Rosenthal DI. Radiofrequency treatment. Orthop Clin North Am. 2006;37(3):475-484, viii. 10.1016/j.ocl.2006.05.004 [DOI] [PubMed] [Google Scholar]
- 101. Bittman RW, Peters GL, Newsome JM, et al. Percutaneous image-guided cryoneurolysis. AJR Am J Roentgenol. 2018;210(2):454-465. 10.2214/AJR.17.18452 [DOI] [PubMed] [Google Scholar]
- 102. Sag AA, Bittman R, Prologo F, et al. Percutaneous image-guided cryoneurolysis: applications and techniques. Radiographics. 2022;42(6):1776-1794. 10.1148/rg.220082 [DOI] [PubMed] [Google Scholar]
- 103. Ilfeld BM, Gabriel RA, Trescot AM.. Ultrasound-guided percutaneous cryoneurolysis for treatment of acute pain: could cryoanalgesia replace continuous peripheral nerve blocks? Br J Anaesth. 2017;119(4):703-706. 10.1093/BJA/AEX142 [DOI] [PubMed] [Google Scholar]
- 104. Callstrom MR, Dupuy DE, Solomon SB, et al. Percutaneous image-guided cryoablation of painful metastases involving bone: multicenter trial. Cancer. 2013;119(5):1033-1041. 10.1002/CNCR.27793 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Choi J, Raghavan M.. Diagnostic imaging and image-guided therapy of skeletal metastases. Cancer Control. 2012;19(2):102-112. 10.1177/107327481201900204 [DOI] [PubMed] [Google Scholar]
- 106. Munk PL, Legiehn GM.. Musculoskeletal interventional radiology: applications to oncology. Semin Roentgenol. 2007;42(3):164-174. 10.1053/J.RO.2007.04.004 [DOI] [PubMed] [Google Scholar]
- 107. Finneran IV JJ, Ilfeld BM.. Percutaneous cryoneurolysis for acute pain management: current status and future prospects. Expert Rev Med Devices. 2021;18(6):533-543. 10.1080/17434440.2021.1927705 [DOI] [PubMed] [Google Scholar]
- 108. Prologo JD, Lin RC, Williams R, Corn D.. Percutaneous CT-guided cryoablation for the treatment of refractory pudendal neuralgia. Skeletal Radiol. 2015;44(5):709-714. 10.1007/S00256-014-2075-3/METRICS [DOI] [PubMed] [Google Scholar]
- 109. Cazzato RL, Garnon J, Ramamurthy N, et al. Percutaneous image-guided cryoablation: current applications and results in the oncologic field. Med Oncol. 2016;33(12). 10.1007/S12032-016-0848-3 [DOI] [PubMed] [Google Scholar]
- 110. Gangi A, Buy X.. Percutaneous bone tumor management. Semin Intervent Radiol. 2010;27(2):124-136. 10.1055/S-0030-1253511 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Ferrer-Mileo L, Luque Blanco AI, González-Barboteo J.. Efficacy of cryoablation to control cancer pain: a systematic review. Pain Pract. 2018;18(8):1083-1098. 10.1111/PAPR.12707 [DOI] [PubMed] [Google Scholar]
- 112. Jennings JW, Prologo JD, Garnon J, et al. Cryoablation for palliation of painful bone metastases: the motion multicenter study. Radiol Imaging Cancer. 2021;3(2):e200101. 10.1148/RYCAN.2021200101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113. Hegg RM, Kurup AN, Schmit GD, et al. Cryoablation of sternal metastases for pain palliation and local tumor control. J Vasc Interv Radiol. 2014;25(11):1665-1670. 10.1016/J.JVIR.2014.08.011 [DOI] [PubMed] [Google Scholar]
- 114. Lievens Y, Guckenberger M, Gomez D, et al. Defining oligometastatic disease from a radiation oncology perspective: an ESTRO-ASTRO consensus document. Radiother Oncol. 2020;148:157-166. 10.1016/J.RADONC.2020.04.003 [DOI] [PubMed] [Google Scholar]
- 115. Schmit GD, Kurup AN, Morris JM, et al. Percutaneous cryoablation of plasmacytomas: oncologic effectiveness and adverse events. Journal of Vascular and Interventional Radiology. 2023;34(8):1303-1310. 10.1016/J.JVIR.2023.04.013 [DOI] [PubMed] [Google Scholar]
- 116. Gronchi A, Miah AB, Dei Tos AP, et al. ; ESMO Guidelines Committee, EURACAN and GENTURIS. Soft tissue and visceral sarcomas: ESMO–EURACAN–GENTURIS Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2021;32(11):1348-1365. 10.1016/J.ANNONC.2021.07.006/ATTACHMENT/A9170587-4D5D-4FB1-A159-D8A0A7B53BF9/MMC1.PDF [DOI] [PubMed] [Google Scholar]
- 117. Doshi A, Zhou M, Bui N, Wang DS, Ganjoo K, Hwang GL.. Safety and feasibility of cryoablation during immunotherapy in patients with metastatic soft tissue sarcoma. J Vasc Interv Radiol. 2021;32(12):1688-1694. 10.1016/j.jvir.2021.08.017 [DOI] [PubMed] [Google Scholar]
- 118. Susa M, Kikuta K, Nakayama R, et al. CT guided cryoablation for locally recurrent or metastatic bone and soft tissue tumor: initial experience. BMC Cancer. 2016;16(1):798. 10.1186/S12885-016-2852-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119. Welch BT, Ehman EC, VanBuren WM, et al. Percutaneous cryoablation of abdominal wall endometriosis: the Mayo Clinic approach. Abdom Radiol (NY). 2020;45(6):1813-1817. 10.1007/S00261-019-02379-4/METRICS [DOI] [PubMed] [Google Scholar]
- 120. Marjanska A, Galazka P, Wysocki M, Styczynski J.. New frontiers in therapy of peripheral nerve sheath tumors in patients with neurofibromatosis type 1: latest evidence and clinical implications. Anticancer Res. 2020;40(4):1817-1831. 10.21873/ANTICANRES.14136 [DOI] [PubMed] [Google Scholar]
- 121. Mai D, Hashimoto R, Yu A, et al. Successful curative cryoablation of an esophageal gastrointestinal stromal tumor. ACG Case Rep J. 2019;6(6):e00076. 10.14309/CRJ.0000000000000076 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122. Mullen JT, Delaney TF, Kobayashi WK, et al. Desmoid tumor: analysis of prognostic factors and outcomes in a surgical series. Ann Surg Oncol. 2012;19(13):4028-4035. 10.1245/S10434-012-2638-2/METRICS [DOI] [PubMed] [Google Scholar]
- 123. Riedel RF, Agulnik M.. Evolving strategies for management of desmoid tumor. Cancer. 2022;128(16):3027-3040. 10.1002/CNCR.34332 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. Vora BMK, Munk PL, Somasundaram N, et al. Cryotherapy in extra-abdominal desmoid tumors: a systematic review and meta-analysis. PLoS One. 2021;16(12):e0261657. 10.1371/JOURNAL.PONE.0261657 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125. Yan YY, Walsh JP, Munk PL, et al. A single-center 10-year retrospective analysis of cryoablation for the management of desmoid tumors. J Vasc Interv Radiol. 2021;32(9):1277-1287. 10.1016/j.jvir.2021.05.025 [DOI] [PubMed] [Google Scholar]
- 126. Hsu M, Stevenson FF.. Wallerian degeneration and recovery of motor nerves after multiple focused cold therapies. Muscle Nerve. 2015;51(2):268-275. 10.1002/MUS.24306 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127. Jones WB, Jordan P, Pudi M.. Pain management of pancreatic head adenocarcinomas that are unresectable: celiac plexus neurolysis and splanchnicectomy. J Gastrointest Oncol. 2015;6(4):445-451. 10.3978/J.ISSN.2078-6891.2015.052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128. Ramaswamy RS, Tiwari T, Darcy MD, et al. Cryoablation of low-flow vascular malformations. Diagn Interv Radiol. 2019;25(3):225-230. 10.5152/DIR.2019.18278 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129. Cornelis FH, Marin F, Labrèze C, et al. Percutaneous cryoablation of symptomatic venous malformations as a second-line therapeutic option: a five-year single institution experience. Eur Radiol. 2017;27(12):5015-5023. 10.1007/S00330-017-4892-Y [DOI] [PubMed] [Google Scholar]
- 130. Fish A, Moushey A, Chan SM, Staib L, Marino A, Schlachter T.. Cryoablation of venous malformations: a systematic review. J Vasc Interv Radiol. 2022;33(8):993-1000. 10.1016/j.jvir.2022.04.010 [DOI] [PubMed] [Google Scholar]
- 131. Auloge P, Cazzato RL, Rousseau C, et al. Complications of percutaneous bone tumor cryoablation: a 10-year experience. Radiology. 2019;291(2):521-528. 10.1148/RADIOL.2019181262/ASSET/IMAGES/LARGE/RADIOL.2019181262.TBL4.JPEG [DOI] [PubMed] [Google Scholar]
- 132. Kurup AN, Morris JM, Schmit GD, et al. Neuroanatomic considerations in percutaneous tumor ablation. Radiographics. 2013;33(4):1195-1215. 10.1148/RG.334125141 [DOI] [PubMed] [Google Scholar]
- 133. Trescot AM. Cryoanalgesia in interventional pain management. Pain Physician. 2003;6(3):345-360. 10.36076/PPJ.2003/6/345 [DOI] [PubMed] [Google Scholar]
- 134. Coupal TM. The hopeless case? Palliative cryoablation and cementoplasty procedures for palliation of large pelvic bone metastases. Pain Physician. 2017;20(7):E1053-E1061. 10.36076/PPJ/2017.7.E1053 [DOI] [PubMed] [Google Scholar]
- 135. Castañeda Rodriguez WR, Callstrom MR.. Effective pain palliation and prevention of fracture for axial-loading skeletal metastases using combined cryoablation and cementoplasty. Tech Vasc Interv Radiol. 2011;14(3):160-169. 10.1053/J.TVIR.2011.02.008 [DOI] [PubMed] [Google Scholar]
- 136. Kurup AN, Schmit GD, Atwell TD, et al. Palliative percutaneous cryoablation and cementoplasty of acetabular metastases: factors affecting pain control and fracture risk. Cardiovasc Intervent Radiol. 2018;41(11):1735-1742. 10.1007/S00270-018-1998-9 [DOI] [PubMed] [Google Scholar]
- 137. Mohme M, Riethdorf S, Dreimann M, et al. Circulating tumour cell release after cement augmentation of vertebral metastases. Sci Rep. 2017;7(1):7196-7199. 10.1038/s41598-017-07649-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138. Levy J, Hopkins T, Morris J, et al. Radiofrequency ablation for the palliative treatment of bone metastases: outcomes from the multicenter OsteoCool Tumor Ablation Post-Market Study (OPuS One Study) in 100 patients. J Vasc Interv Radiol. 2020;31(11):1745-1752. 10.1016/J.JVIR.2020.07.014 [DOI] [PubMed] [Google Scholar]
- 139. Jiao D, Yao Y, Li Z, Ren J, Han X.. Simultaneous C-arm computed tomography-guided microwave ablation and cementoplasty in patients with painful osteolytic bone metastases: a single-center experience. Acad Radiol. 2022;29(1):42-50. 10.1016/J.ACRA.2020.09.026 [DOI] [PubMed] [Google Scholar]
- 140. Cazzato RL, Garnon J, Caudrelier J, Rao PP, Koch G, Gangi A.. Percutaneous radiofrequency ablation of painful spinal metastasis: a systematic literature assessment of analgesia and safety. Int J Hyperthermia. 2018;34(8):1272-1281. 10.1080/02656736.2018.1425918 [DOI] [PubMed] [Google Scholar]
- 141. Munk PL, Rashid F, Heran MK, et al. Combined cementoplasty and radiofrequency ablation in the treatment of painful neoplastic lesions of bone. J Vasc Interv Radiol. 2009;20(7):903-911. 10.1016/J.JVIR.2009.03.035 [DOI] [PubMed] [Google Scholar]
- 142. Lane MD, Le HBQ, Lee S, et al. Combination radiofrequency ablation and cementoplasty for palliative treatment of painful neoplastic bone metastasis: experience with 53 treated lesions in 36 patients. Skeletal Radiol. 2011;40(1):25-32. 10.1007/S00256-010-1010-5 [DOI] [PubMed] [Google Scholar]
- 143. Jakanani GC, Jaiveer S, Ashford R, Rennie W.. Computed tomography-guided coblation and cementoplasty of a painful acetabular metastasis: an effective palliative treatment. J Palliat Med. 2010;13(1):83-85. 10.1089/JPM.2009.0176 [DOI] [PubMed] [Google Scholar]
- 144. Munk PL. The accrual of knowledge and experience in MSK ablation. J Vasc Interv Radiol. 2020;31(11):1763-1764. 10.1016/J.JVIR.2020.08.032 [DOI] [PubMed] [Google Scholar]
- 145. Kam NM, Maingard J, Kok HK, et al. Combined vertebral augmentation and radiofrequency ablation in the management of spinal metastases: an update. Curr Treat Options Oncol. 2017;18(12):74. 10.1007/S11864-017-0516-7 [DOI] [PubMed] [Google Scholar]
- 146. Wang F, Gu J, Xu C, Li G, Lv P.. The combination of radiofrequency ablation and vertebroplasty shows advantages over single vertebroplasty in treating vertebral neoplastic lesions. Skeletal Radiol. 2022;51(3):565-571. 10.1007/S00256-021-03788-7 [DOI] [PubMed] [Google Scholar]