Abstract
An advisory board meeting was held with experts in Vietnam (Hanoi, August 2022), to review the evidence on invasive meningococcal disease (IMD) epidemiology, clinical management, and meningococcal vaccines to reach a consensus on recommendations for meningococcal vaccination in Vietnam. IMD is a severe disease, with the highest burden in infants and children. IMD presents as meningitis and/or meningococcemia and can progress extremely rapidly. Almost 90% of deaths in children occur within the first 24 h, and disabling sequelae (e.g., limb amputations and neurological damage) occur in up to 20% of survivors. IMD patients are often hospitalized late, due to mild and nonspecific early symptoms and misdiagnosis. Difficulties related to diagnosis and antibiotic misuse mean that the number of reported IMD cases in Vietnam is likely to be underestimated. Serogroup B IMD is predominant in many regions of the world, including Vietnam, where 82% of IMD cases were due to serogroup B (surveillance data from 2012 to 2021). Four component meningococcal B vaccine (4CMenB) is used in many countries (and is part of the pediatric National Immunization Program in 13 countries), with infant vaccination starting from two months of age, and a 2 + 1 dosing schedule. Experts recommend 4CMenB vaccination as soon as possible in Vietnam, starting from two months of age, with a 2 + 1 dosing schedule, and at least completing one dose before 6 months of age.
Supplementary Information
The online version contains supplementary material available at 10.1007/s40121-023-00905-y.
Keywords: Invasive meningococcal disease, Vaccination, Vietnam, 4CMenB, Infant
Key Summary Points
| Invasive meningococcal disease (IMD) is severe, primarily affects infants and young children, and has a high risk of morbidity and mortality. IMD is difficult to diagnose at an early stage, especially in young children. |
| The incidence is likely to be underreported in Vietnam. Most cases today are caused by serogroup B infection. |
| The four-component meningococcal B vaccine (4CMenB) is recommended in many countries for infants (from 2 months of age) and has demonstrated real-world effectiveness. |
| Experts in Vietnam recommend 4CMenB approval for infants in Vietnam, in a 2 + 1 dosing schedule (a possible schedule could be at 2.5, 3.5, and 12 months of age and given separately from other routine vaccinations in the early phase of implementation. After gaining experience with 4CMenB, it can be coadministered with routine vaccines) with parental counseling. |
Introduction
Invasive meningococcal disease (IMD), caused by Neisseria meningitidis, is an acute infection and contagious respiratory illness. The disease is severe, often progresses extremely rapidly, and, if not treated promptly, has a high risk of mortality (4–20% within 48 h) and of sequelae in 10–20% of survivors (e.g., amputation of limbs, hearing loss, and learning difficulties) [1].
In recent years, IMD epidemiology around the world, including in Vietnam, is experiencing shifts in the predominant disease-causing serogroups. There is, however, no clear consensus in Vietnam regarding the current epidemiology, or meningococcal B vaccine recommendations.
On 22 August 2022, an advisory board meeting was held with ten leading experts from Preventive Medicine, Infectious Diseases and Pediatrics from Vietnam and five vaccine and medical experts from GSK, aiming to review the existing evidence and provide recommendations regarding four-component meningococcal B (4CMenB) vaccination to prevent serogroup B IMD. The outcomes of the meeting are presented here.
As IMD evidence from Asia can be limited, a broader outlook was taken, first presenting data from North America and Europe, followed by Asia and Vietnam, specifically.
This article is based on previously conducted studies and does not contain any new studies, with human participants or animals, performed by any of the authors.
Epidemiology of IMD in the World, Asia, and Vietnam
IMD is a Global Disease, with the Highest Burden in Infants and Children
IMD is a global disease, which is endemic in almost all regions, from the Americas to Europe, Africa and Asia [2]. Health policies to control the disease and health system responses differ by country, resulting in different incidence rates [2]. Despite country-specific differences, most countries tend to find that incidence and case fatality rates are highest in infants and young children.
In the United States of America (USA), IMD incidence declined from 0.10 to 0.07 cases per 100,000 population, from 2018 to 2020 [probably due to coronavirus disease 2019 (COVID-19) measures], respectively, with the highest rates in infants (0.83–0.43) and children aged 1–4 years (0.18–0.12). The case fatality rate went from 12.0% to 10.4% for IMD overall, peaking in infants in 2020 (28.6% versus 12.9% in infants in 2018) [3, 4].
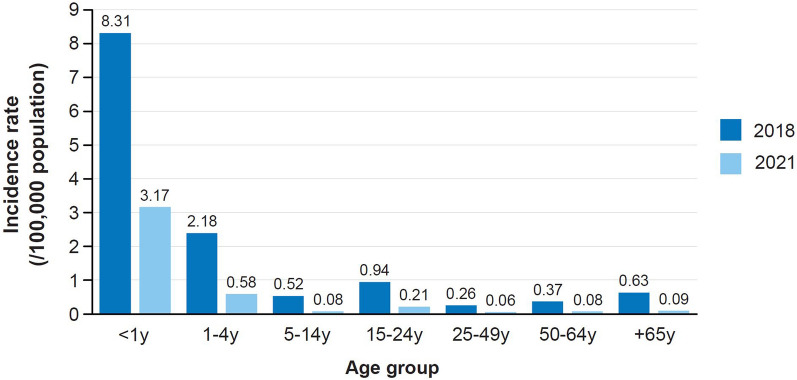
In 2018, the European Centre for Disease Prevention and Control (ECDC) reported 0.62 IMD cases per 100,000 population, with the highest incidence rates in the youngest age groups (i.e., 8.31 in infants aged < 1 year and 2.38 in children aged 1–4 years) (Fig. 1). Country-specific incidence rates in infants peaked at 22.4. The case fatality rate was 12% [5]. As was also the case with other infectious diseases, there was a reduction in cases during the COVID-19 pandemic, probably due to social distancing measures [6]. The overall incidence decreased from 0.57 in 2019 and 0.26 in 2020 to 0.14 in 2021; however, infants still had the highest incidence (7.25 in 2019, 4.80 in 2020, and 3.17 in 2021 [7]) (Fig. 1).
Fig. 1.
IMD incidence by age group in Europe [European Centre for Disease Prevention and Control (ECDC) 2018 and 2021] [5, 7]. The incidence of IMD per 100,000 population by age group in 2018 and 2021 is shown. y year
Data from Türkiye (2015–2018) also show IMD incidence in children peaks among infants followed by children < 5 years old (Fig. 2) [8].
Fig. 2.
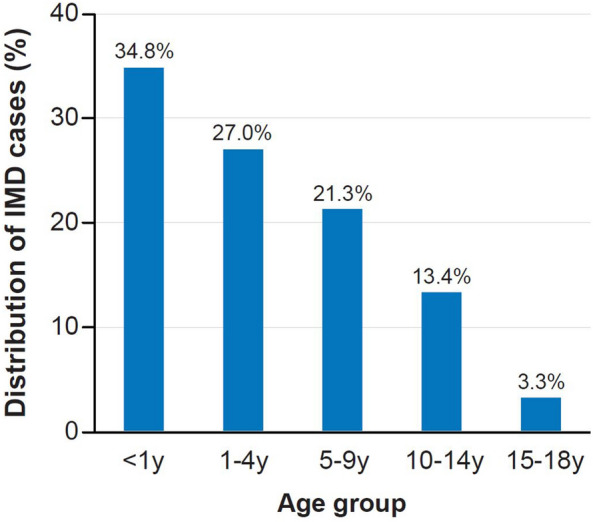
Distribution of IMD cases in children by age group in Türkiye (2015–2018) [8]. The distribution of IMD cases by age group in under 18 s is shown. IMD invasive meningococcal disease; y year
For Asian countries in the Asia–Pacific region, IMD incidence ranged from 0.02 (the Philippines) to 0.2 (Singapore) cases per 100,000 population, with higher incidence rates reported in some subpopulations, e.g., in infants, children, and adolescents [9]. In the Philippines, surveillance data from 2019 reported that around half of all cases occurred in children < 5 years, and around 25% in infants < 1 year [10]. Surveillance data in Thailand reported IMD incidence rates of < 0.10 from 2010 to 2019, with the highest incidence in children < 5 years [10]. Case fatality rates were considerably higher in some countries e.g., around 40–50% in Thailand and the Philippines compared with 10–16% in China and Japan [9].
In Vietnam (Fig. 3), from population-based surveillance (2000–2002) for bacterial meningitis, incidence rates in children aged < 5 years were estimated to be 7.4 per 100,000 population (95% confidence interval [CI] 3.6–15.3) compared with 6.8 (95% CI 3.5–13.5) in Korea and 2.1 (95% CI 0.7–6.2) in China. Among children < 5 years, higher incidence rates were observed for age groups < 6 months old in China (14.6 [95% CI 4.5–47.2]) and Korea (27.2 [95% CI 9.9–74.2]), respectively, and for the age group 7–11 months old in Vietnam (29.1 [95% CI 8.0–106.0]). The highest rates were observed in infants < 1 month old in all three countries: 36.2 (95% CI 3.8–346.0) in Vietnam versus 96 (95% CI 29.7–309.4) in China and 81.5 (95% CI 18.8–352.2) in Korea [11].
Fig. 3.
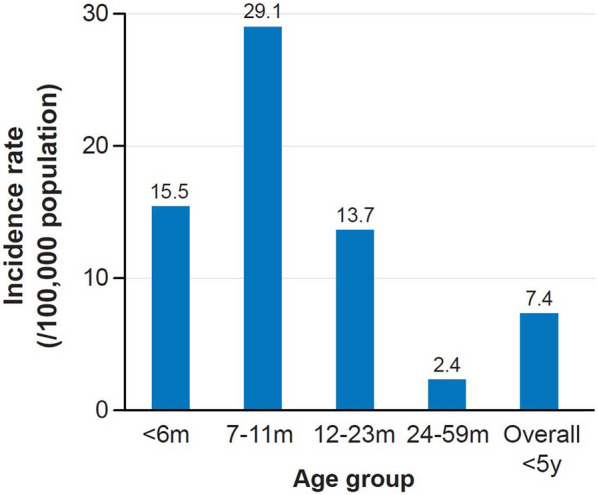
IMD incidence by age group in Vietnamese children (prospective surveillance 2000–2002) [11]. The incidence per 100,000 population of IMD in children under 5 years is shown. IMD invasive meningococcal disease; m month; y year
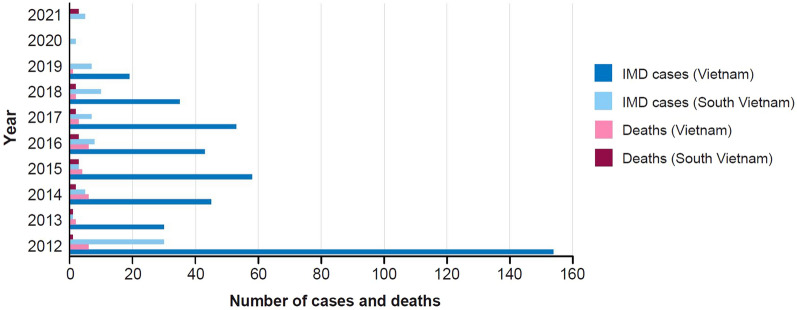
Data from the Routine National Surveillance System in Vietnam reported 184 IMD cases from 2016 to 2022, of which most were in adults: 13.5% (25 cases) were in infants < 12 months old and 10.3% (19 cases) were in babies < 6 months old (personal communication, Pham Quang Thai). Based on these data, the overall incidence of IMD in 2016 was 2.3 per 100,000 population (personal communication, Nguyen Van Kinh), with a decreasing trend in recent years in Southern Vietnam (Fig. 4) (Laboratory information system, Vo Thi Trang Dai).
Fig. 4.
Number of yearly IMD cases and deaths (2012–2021) for Vietnam and South Vietnam, reported by the National Surveillance System and Communicable Diseases Surveillance System in South Vietnam. The annual number of IMD cases and deaths in Vietnam and South Vietnam from 2012 to 2021 is shown. IMD invasive meningococcal disease
Serogroup B IMD Appears to be Predominant in Many Regions (USA, Europe, and Southeast Asia), Including Vietnam, and in Children
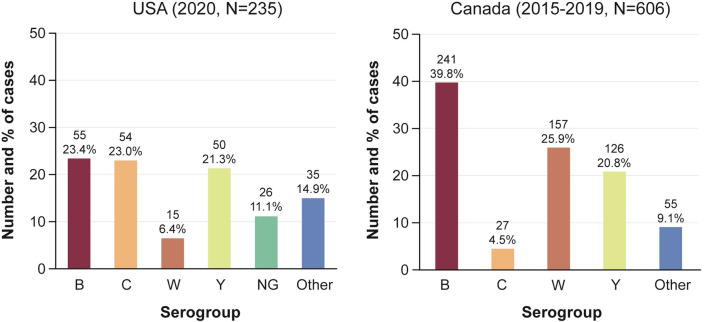
In the USA (2020) [3] and Canada (2015–2019) [12], serogroup B was predominant (Fig. 5), with a case fatality rate of 10.9% (serogroup B IMD) [3] and 7.8% (all IMD) [12], respectively. The highest incidence rate was for serogroup B IMD in infants (0.19 in the USA and 1.9 in Canada per 100,000 population) [3, 12].
Fig. 5.
IMD cases in the USA (2020) [3] and Canada (2015–2019) [12] by serogroup. The serogroup distribution of IMD cases in the USA (2020) and Canada (2015–2019) is shown. IMD invasive meningococcal disease; NG nongroupable; USA United States of America; N number; Other includes other or unknown serogroup cases, including 1 serogroup E case in the USA
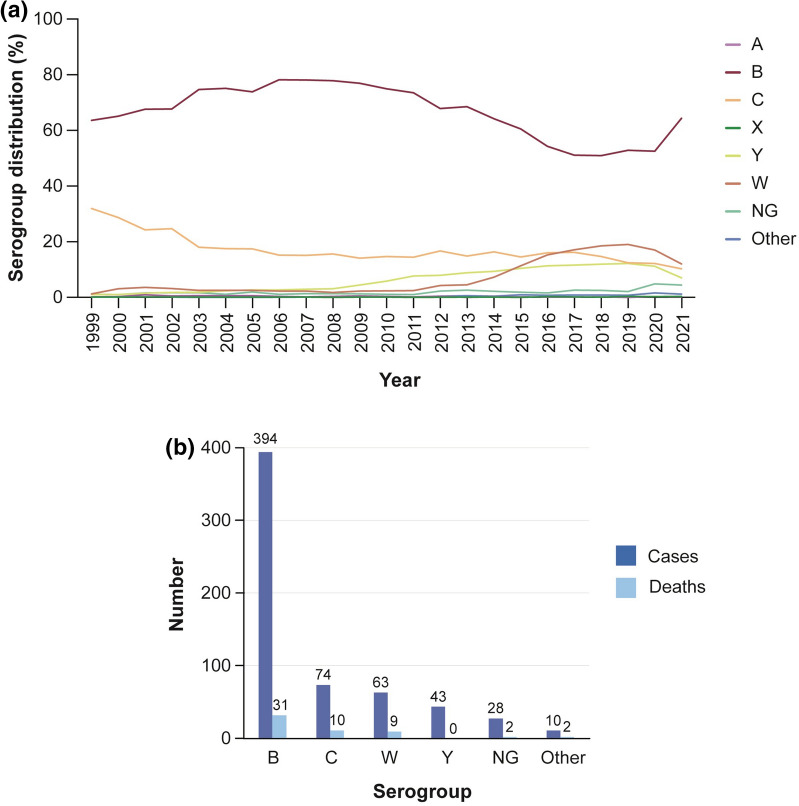
Serogroup B was the major cause of IMD in Europe from 2000 to 2021 (Fig. 6a), responsible for 64.4% of cases overall and 56.3% of IMD deaths in 2021 (Fig. 6b) [7]. Although overall IMD incidence declined during the COVID-19 pandemic, a sharp increase in serogroup B cases was noted between 2020 and 2021 (Fig. 6a) [7].
Fig. 6.
a Serogroup distribution of IMD cases in Europe (ECDC 1999–2021) [7]; b IMD cases and deaths by serogroup in Europe (ECDC 2021) [7]. ECDC European Centre for Disease Prevention and Control; IMD invasive meningococcal disease; NG nongroupable
Between 2006 and 2010 in England and Wales, serogroup B IMD was responsible for 87% of cases, with the highest incidence in infants (36.2 per 100,000 population). Most infant cases occurred in those < 6 months old, peaking at 5 months old, highlighting the importance of vaccination before the age of 6 months. The case fatality rate for serogroup B IMD was 5.2% (all ages), with the largest number of deaths in children < 5 years [13].
In Asian countries, serogroup B IMD was reported to be predominant in Singapore [14], in the Philippines (with 68% of cases due to serogroup B from 2017 to 2018 surveillance data), and in Thailand (with 50–80% of cases due to serogroup B from 1994 to 1999) [10].
In Vietnam, the Meningitis Sentinel Surveillance System (run by the National Institute of Hygiene and Epidemiology) has collected data from Children’s Hospital 1 and 2 (in the South) and the Vietnam Children hospital (in the North) since 2012. Most IMD cases from the Northern and Southern regions during this period were serotype B IMD (Fig. 7, personal communication, Vo Thi Trang Dai). Similarly, the National Pediatric Hospital reported 15 IMD cases with severe complications (2016–2020), all of which were due to serogroup B (personal communication, Do Thien Hai).
Fig. 7.
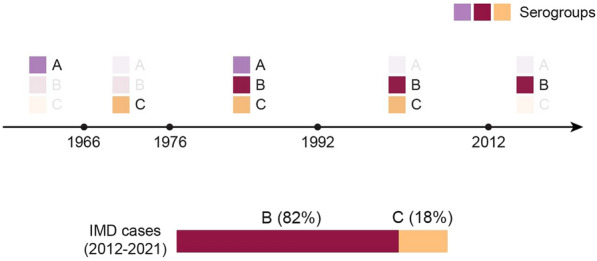
IMD serogroups in Vietnam over time (1966–2012) and serogroup distribution (2012–2021). The prevalent IMD serogroups in Vietnam from 1966 to 2012 and the serogroup distribution of IMD cases from 2012 to 2021 are shown. IMD invasive meningococcal disease
The “108 Military Central Hospital” monitoring healthy carriers found a positive rate for IMD in around 25% of new military recruits aged 18–25 years old (2012–2014), of which most cases (53.19%) were serogroup B [15].
Challenges in the Diagnosis and Treatment of IMD
IMD patients are Often Hospitalized Late, Due to Mild or Nonspecific Early Symptoms and Misdiagnosis
Neisseria meningitidis bacteria usually reside within the nasopharynx with other common bacteria and viruses. In mild cases, it only causes nasopharyngitis. In the case of invasive disease (i.e., IMD), initial symptoms (e.g., fever, vomiting, fatigue, and lethargy) are flu-like and nonspecific, making early diagnosis difficult, especially in nonspecialized hospitals. Meningococcal disease is rare, so many doctors lack experience of early diagnosis. Patients are often admitted to the hospital when they become seriously ill (with meningitis or sepsis). The typical triad of meningitis symptoms, i.e., fever, stiff neck, and impaired consciousness, was only present in around 27% of patients. For severe cases, only central hospitals have the modern treatment facilities and techniques available for treatment (i.e., intubation, mechanical ventilation, dialysis, vasopressors, and doctors with experienced with resuscitation). Road traffic in Vietnam can lead to long hospital transfer times and contribute to an increased risk of sequelae and death (personal communication, Do Thien Hai and Nguyen The Nguyen).
IMD can Progress Extremely Rapidly and has a Severe Clinical Presentation
The three common severe clinical presentations of IMD are meningitis (≥ 50% of cases), meningococcemia (17–37%) and both meningitis and meningococcemia (4–22%) [16]. Patients can progress to shock, weak pulse, and death within 12–24 h of onset (Table 1). Myocardial contractility may be reduced in 50% of cases. Adrenal infarction causes acute adrenal insufficiency which leads to cardiovascular collapse and rapid lowering of blood pressure. Dark purple rash occurs in 42–70% of cases and purpura fulminans in 15–25% of cases, which can progress to bullous or gaseous necrosis of subcutaneous tissue and even bone (personal communication from Phung Nguyen The Nguyen).
Table 1.
Time to onset of IMD symptoms, complications, or death (personal communication, Phung Nguyen The Nguyen)
| Symptoms/complications | Hours | % of patients |
|---|---|---|
| Fever, vomiting, and fatigue | 4–6 | |
| Leg pain | 8 | 31–63 |
| Cold extremities | 8 | 35–47 |
| Abnormal skin color | 8 | 17–21 |
| Meningitis (neck stiffness, sensitivity to light, and bulging fontanel) | 12–15 | 27 |
| Convulsions, delirium, and altered mental status | 15–24 | |
| Dark purple rash | 42–70 | |
| Purpura fulminans | 15–25 | |
| Death | 24 | |
| Average length of hospital stay, days | 19 | – |
IMD invasive meningococcal disease
IMD Patients are at High Risk of Mortality and Sequelae
IMD has a high mortality rate without treatment and up to 10% with treatment; almost 90% of childhood deaths due to IMD occur within 24 h [17]. Survivors may develop severe sequelae including amputations and neurologic effects (e.g., hydrocephalus, sensory deficits, and nerve damage), as well as skin scarring [18].
Description of a Clinical Case at Children’s Hospital 1 [1, 18]
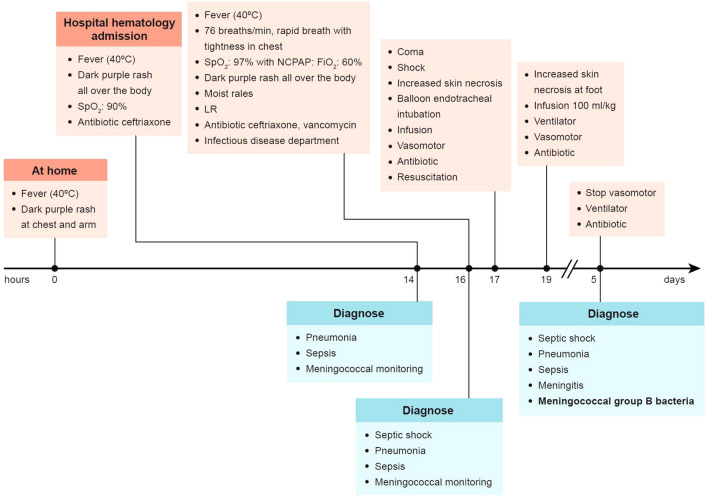
The patient was a 4.5-month-old boy (second child, birth weight 3.1 kg, full-term infant, and vaccinated under the Expanded Immunization Program) admitted with fever with petechiae. Disease progression was rapid (Fig. 8), and he was diagnosed with serogroup B IMD.
Fig. 8.
Case study—serogroup B IMD disease progression. The IMD disease progression in a case study is shown. FiO2 fraction of inspired oxygen; IMD invasive meningococcal disease; LR lactate ringer; min minute; NCPAP nasal continuous positive airway pressure; SpO2 peripheral capillary oxygen saturation
The patient continued treatment in the intensive care unit for 1 week and was discharged after nearly 2 months of hospitalization. The patient suffered amputation, scarring from tissue necrosis, and had to return to the surgical department for excision of overgrown stump every few months.
Expert Discussion of Epidemiology Data for Vietnam: the Number of IMD cases Reported in Vietnam May be Underestimated Due to Diagnostic Problems and Antibiotic (Mis)use
The experts discussed why IMD incidence from passive reports to the National Surveillance System was lower than the IMD incidence reported from the prospective study by Kim et al. (2012) [11] in children < 5 years. Several possible explanations were put forward. Antibiotic misuse or use at hospital admission before laboratory testing may occur, which can lead to incorrect polymerase chain reaction (PCR) and bacterial culture results [19]. Bacterial culture testing tends to be less sensitive than PCR for the diagnosis of IMD [20]. Before PCR test results for meningococcal disease are confirmed, the patient may have been diagnosed with another disease with similar symptoms. The shortage of biological products and testing limits diagnosis capabilities in less specialized hospitals. IMD is rare and experience of diagnosing and treating the disease is limited. The system’s surveillance reports may be missed, necessitating surveillance system improvements. Surveillance system data are limited to invasive disease reports and do not include ear, nose, and throat infections or meningococcal case reports.
The decrease in IMD cases reported in recent years (Fig. 4) may be related to the COVID-19 pandemic affecting IMD detection and case reporting (personal communication, Pham Quang Thai). IMD has flu-like onset symptoms, and patients afraid of COVID-19 may have taken antibiotics immediately, affecting test results and potentially reducing the number of hospitalized patients.
Expert Consensus on IMD Burden, Shift in Disease-Causing Serogroups, and Age Distribution.
IMD has a high burden, especially in children, with severe clinical presentations (sepsis, meningitis or both) and a high risk of mortality and sequelae.
The pediatric and infectious disease experts believe that IMD incidence from epidemiological reports is likely underestimated. Serogroup B has predominated for more than 10 years, and vaccination to prevent meningococcal B IMD is needed.
Guidelines and recommendations from the Ministry of Health and the Vietnam Association of Preventive Medicine are required to improve surveillance of epidemiological characteristics and reporting of the actual number of cases.
A long-term plan is needed, with a comprehensive assessment of IMD epidemiology; the vaccine’s efficacy, safety, and cost-effectiveness; and the feasibility of introducing meningococcal B vaccination into the Expanded Immunization Program to benefit children, especially in remote areas with difficult economic conditions.
In Vietnam, children under 2 years of age are the highest risk group and should be given priority to receive meningococcal B vaccine as soon as possible before 6 months of age. If the vaccine is given to a child aged 2 to 6 months, the child will be protected until the age of 5 years. For adolescents, before college or military enlistment, a single booster shot is considered enough to provide protection.
Update on Technological Developments for Meningococcal Vaccines
4CMenB, Developed by Reverse Vaccinology, is a Broad-Spectrum Vaccine Against Meningococcus B with Four Antigenic Components
During the 1960s and 1970s, despite many efforts to develop meningococcal B vaccines, very few were effective against serogroup B IMD [21]. The polysaccharide capsule of serogroup B is structurally similar to polysialic acid units found in the human body, therefore, poorly immunogenic [22]. The outer membrane vesicle (OMV) vaccines were developed based on the common circulating strain of a specific area and the vaccine cannot be widely used in the world, as countries have different B strains and the vaccine is only effective against strains common to specific areas [21].
To overcome these challenges, reverse vaccinology was used to develop the 4CMenB vaccine. The entire genome of 2,158 bacterial genes was screened [23] and three potential protein antigens for vaccine development were identified: factor H binding protein (fHbp), Neisseria adhesin A (NadA), and Neisserial heparin-binding antigen (NHBA). A fourth antigenic component, the OMV protein from the original New Zealand OMV vaccine (NZ PorA P1.4), was added [24]. The fHbp binds factor H (which enables bacterial survival in the blood) [25], NadA promotes adherence to and invasion of human epithelial cells [26], NHBA binds heparin (which may promote bacterial survival in the blood) [23], and NZ PorA P1.4 induces strain-specific bactericidal responses [27].
4CMenB Vaccine Produces a High Immune Response to All Four Antigenic Components, from Infancy to Adolescence
The immunogenicity of 4CMenB in infants < 6 months was similar with a 2 + 1 and a 3 + 1 schedule (i.e., two or three primary doses plus a booster dose, respectively) [28] (Fig. S1) and was unaffected by coadministration of routine vaccines [29] (Fig. S2). Prophylactic paracetamol reduced the incidence of fever when 4CMenB was administered with other routine vaccines [30].
4CMenB is Used Globally, with Infant Vaccination from Two Months Old Using a 2 + 1 Dosing Schedule
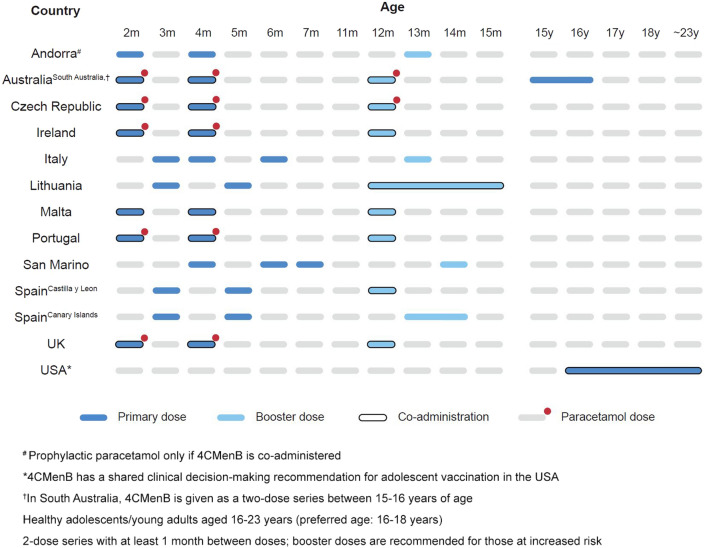
The 4CMenB vaccine was approved in Europe and the USA in 2015 and was registered in 56 countries by 2023, with recommendations in 33 in 2021. It is included in 13 National Immunization Programs (NIPs), and over 52 million doses have been distributed worldwide [31–33]. Most countries (except the USA and Switzerland) recommend vaccination from the age of 2 months, and most NIPs use a 2 + 1 schedule (Fig. 9).
Fig. 9.
4CMenB dosing regimens in National Immunization Programs [33]. An overview of four-component meningococcal B vaccine (4CMenB) schedules used in National Immunization Programs around the world is shown. 4CMenB four component meningococcal B vaccine; UK United Kingdom; USA United States of America
In addition to efficacy demonstrated in clinical trials, the real-world effectiveness of 4CMenB vaccine has been demonstrated in several countries (Fig. S3) [34–40]. Safety studies found 4CMenB safety to be similar to other vaccines, with no more prominent adverse events identified [37, 41–51].
Experience of IMD Prevention and Meningococcal Awareness Campaigns
Since 2015 in Türkiye, the highest incidence of IMD has been observed in infants [8, 52, 53], and there has been a shift in the predominant disease-causing serogroups [8]. 4CMenB was approved in 2018, first with a 3 + 1 schedule and from 2021, a 2 + 1 schedule for infants from 2 months of age. Vaccination campaigns were launched with plans to manage issues that may be of concern to pediatricians and family doctors regarding the disease (burden and epidemiology) and vaccine. Workshops were organized to share experience on vaccine efficacy, side effects, burden of disease, and the importance of early protection for children.
Expert Recommendations and Discussion of Measures to Improve Mother’s Agreement of 4CMenB Vaccine, if Approved in Vietnam.
Good health communication is important for people and healthcare professionals to understand the disease burden, the shift to serogroup B predominance, and to ensure safe vaccine implementation.
Postinjection fever may arise; however, it should be noted that currently, only around 20% of mothers comply with prophylactic paracetamol for other vaccines. Immunization facilities and hospitals should strengthen communication and advice around postinjection reactions, providing specific instructions on taking antipyretic drugs before, during, and after vaccination with 4CMenB.
The experts recommend using 4CMenB vaccine for infants from 2 months of age and to administer 4CMenB separately from pneumococcal vaccinations to avoid overlap and confusion about postinjection reactions. The 6-in-1 and pneumococcal vaccines are prioritized, and there should be an interval of at least 2 weeks before administering 4CMenB. 4CMenB may be recommended at 2.5 months, 3 months, or 3.5 months of age, as long as the first dose is guaranteed before 6 months of age. Vaccination facilities can decide on the specific vaccination schedule. After gaining experience with 4CMenB, coadministration with other routine vaccines can be considered.
Conclusions
In Vietnam, IMD imposes a significant disease burden. According to pediatric specialists, the reported clinical manifestations of meningitis and meningococcemia are severe, difficult to treat, and associated with numerous sequelae. No data on the cost of illness and economic burden of IMD in Asia or Vietnam were identified.
According to the experts, the incidence rates reported by national disease surveillance data may not reflect the true incidence rates. In the past 10 years, serogroup B IMD has become predominant in Vietnam. This increases the demand for meningococcal B vaccines, particularly for infants younger than 6 months of age.
Reverse vaccinology assisted in the development of the 4CMenB vaccine, which contains four antigen components. Immunogenic efficacy and safety for children > 2 months of age have been demonstrated in clinical trials, and data from real world evidence have demonstrated effectiveness/impact at the country level. The vaccine is being used in many countries.
The experts recommend unequivocally that the Vietnamese Ministry of Health approve 4CMenB for infants from 2 months of age. A reasonable vaccination schedule includes early vaccination, i.e., at least one dose before the age of 6 months, and a 2 + 1 schedule (two primary doses and one booster). According to the current pediatric vaccination schedule, infants could receive 4CMenB at 2.5 and 4.5 months of age, with a supplemental dose at 12 months.
Within the childhood immunization schedule, 4CMenB use could alternate with other routine vaccines at the start of implementation with a specific vaccination schedule, depending on vaccination facilities. After gaining experience, coadministration with routine vaccines could be considered. Providing vaccine counseling to parents is essential.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgments
Medical Writing, Editorial, and Other Assistance
The authors thank Business & Decision Life Sciences Medical Communication Service Center for editorial assistance and manuscript coordination, on behalf of GSK. Kavi Littlewood (Littlewood Writing Solutions) provided writing support, on behalf of GSK. The authors thank Dr Bach Thi Chinh for their participation in the Advisory board.
Author Contribution
All authors participated in the discussion and the development of this manuscript, reviewed, and approved the final manuscript.
Funding
GlaxoSmithKline Biologicals SA funded all costs associated with the development and publication of this manuscript, including the journal’s Rapid Service fee.
Data Availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Conflict of Interest
Gaurav Mathur, Selim Badur, Doan Minh Truyen, Nguyen Hoan Le Minh are employed by GSK. Nguyen Huy Luan declares honoraria for presentations in other events and Do Thien Hai reports payment or honoraria for lectures, presentations, speakers bureaus, manuscript events, or educational events, support for attending meetings and/or travel, participation to an advisory board. These authors declare no other financial and nonfinancial relationships and activities. Phung Nguyen The Nguyen, Pham Quang Thai, Tran Minh Dien, Vo Thi Trang Dai declare no financial and nonfinancial relationship and activities.
Ethical Approval
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.The Nguyen PN, Hung NT, Mathur G, Pinto TJP, Minh NHL. Review of the epidemiology, diagnosis and management of invasive meningococcal disease in Vietnam. Hum Vaccin Immunother. 2023;19(1):2172922. doi: 10.1080/21645515.2023.2172922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jafri RZ, Ali A, Messonnier NE, et al. Global epidemiology of invasive meningococcal disease. Popul Health Metr. 2013;11(1):17. doi: 10.1186/1478-7954-11-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Centers for Disease Control and Prevention (CDC) Enhanced meningococcal disease surveillance report, 2020. 2023. https://www.cdc.gov/meningococcal/downloads/NCIRD-EMS-Report-2020.pdf. Accessed 06 March 2023.
- 4.Centers for Disease Control and Prevention (CDC) Enhanced meningococcal disease surveillance report, 2018. 2023. https://www.cdc.gov/meningococcal/downloads/NCIRD-EMS-Report-2018.pdf. Accessed 06 March 2023.
- 5.European Centre for Disease Prevention and Control (ECDC) Invasive meningococcal disease. Annual epidemiological report for 2018. 2023. https://www.ecdc.europa.eu/en/publications-data/invasive-meningococcal-disease-annual-epidemiological-report-2018. Accessed 06 March 2023.
- 6.Alderson MR, Arkwright PD, Bai X, et al. Surveillance and control of meningococcal disease in the COVID-19 era: a global meningococcal initiative review. J Infect. 2022;84(3):289–296. doi: 10.1016/j.jinf.2021.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.European Centre for Disease Prevention and Control (ECDC) Surveillance Atlas of Infectious Diseases. Invasive meningococcal disease 2018–2021. 2023. https://www.ecdc.europa.eu/en/surveillance-atlas-infectious-diseases. Accessed 08 March 2023.
- 8.Ceyhan M, Ozsurekci Y, Basaranoglu ST, et al. Multicenter hospital-based prospective surveillance study of bacterial agents causing meningitis and seroprevalence of different serogroups of Neisseria meningitidis, Haemophilus influenzae Type b, and Streptococcus pneumoniae during 2015 to 2018 in Turkey. mSphere. 2020;5(2):e00060-20. doi: 10.1128/mSphere.00060-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Aye AMM, Bai X, Borrow R, et al. Meningococcal disease surveillance in the Asia-Pacific region (2020): the global meningococcal initiative. J Infect. 2020;81(5):698–711. doi: 10.1016/j.jinf.2020.07.025. [DOI] [PubMed] [Google Scholar]
- 10.Thisyakorn U, Carlos J, Chotpitayasunondh T, et al. Invasive meningococcal disease in Malaysia, Philippines, Thailand, and Vietnam: an Asia-Pacific expert group perspective on current epidemiology and vaccination policies. Hum Vaccin Immunother. 2022;18(6):2110759. doi: 10.1080/21645515.2022.2110759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kim SA, Kim DW, Dong BQ, Kim JS, Anh DD, Kilgore PE. An expanded age range for meningococcal meningitis: molecular diagnostic evidence from population-based surveillance in Asia. BMC Infect Dis. 2012;12:310. doi: 10.1186/1471-2334-12-310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Public Health Agency of Canada Vaccine preventable disease - Surveillance report to December 31, 2019. 2022. https://www.canada.ca/en/public-health/services/publications/healthy-living/vaccine-preventable-disease-surveillance-report-2019.html. Accessed 08 March 2023.
- 13.Ladhani SN, Flood JS, Ramsay ME, et al. Invasive meningococcal disease in England and Wales: implications for the introduction of new vaccines. Vaccine. 2012;30(24):3710–3716. doi: 10.1016/j.vaccine.2012.03.011. [DOI] [PubMed] [Google Scholar]
- 14.Marshall HS. Meningococcal surveillance in Southeast Asia and the Pacific. Microbiol Aust. 2021;42(4):178–181. doi: 10.1071/MA21050. [DOI] [Google Scholar]
- 15.Pham TH, Vo TTD, Phan VT, Uong NDN, Nguyen TKH. Serogroup and antibiotic resistance of Neisseria meningitidis on recruits in some military barracks in the northern region men, 2012–2014. J Prev Med. 2014;55(11):160. [Google Scholar]
- 16.Martinón-Torres F. Deciphering the burden of meningococcal disease: conventional and under-recognized elements. J Adolesc Health. 2016;59(2 Suppl):S12–20. doi: 10.1016/j.jadohealth.2016.03.041. [DOI] [PubMed] [Google Scholar]
- 17.Meningitis Research Foundation 90% of children and teenagers who die of meningococcal meningitis die within 24 hours. 2020. https://www.meningitis.org/research-into-how-fast-meningitis-kills. Accessed 10 March 2023.
- 18.The Nguyen PN. A case report on fulminant meningicoccal infection in infant. Tien Giang Hospital Medicine Conference, Vietnam, 2021.
- 19.Vyse A, Wolter JM, Chen J, Ng T, Soriano-Gabarro M. Meningococcal disease in Asia: an under-recognized public health burden. Epidemiol Infect. 2011;139(7):967–985. doi: 10.1017/S0950268811000574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Guiducci S, Moriondo M, Nieddu F, et al. Culture and real-time polymerase chain reaction sensitivity in the diagnosis of invasive meningococcal disease: does culture miss less severe cases? PLoS ONE. 2019;14(3):e0212922. doi: 10.1371/journal.pone.0212922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sadarangani M, Pollard AJ. Serogroup B meningococcal vaccines-an unfinished story. Lancet Infect Dis. 2010;10(2):112–124. doi: 10.1016/s1473-3099(09)70324-x. [DOI] [PubMed] [Google Scholar]
- 22.Finne J, Bitter-Suermann D, Goridis C, Finne U. An IgG monoclonal antibody to group B meningococci cross-reacts with developmentally regulated polysialic acid units of glycoproteins in neural and extraneural tissues. J Immunol. 1987;138(12):4402–4407. doi: 10.4049/jimmunol.138.12.4402. [DOI] [PubMed] [Google Scholar]
- 23.Tettelin H, Saunders NJ, Heidelberg J, et al. Complete genome sequence of Neisseria meningitidis serogroup B strain MC58. Science. 2000;287(5459):1809–1815. doi: 10.1126/science.287.5459.1809. [DOI] [PubMed] [Google Scholar]
- 24.European Medicines Agency (EMA) Bexsero European public assessment report, Annex I: summary of product characteristics. 2017. http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/002333/WC500137881.pdf. Accessed 10 March 2023.
- 25.Madico G, Welsch JA, Lewis LA, et al. The meningococcal vaccine candidate GNA1870 binds the complement regulatory protein factor H and enhances serum resistance. J Immunol. 2006;177(1):501–510. doi: 10.4049/jimmunol.177.1.501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Comanducci M, Bambini S, Brunelli B, et al. NadA, a novel vaccine candidate of Neisseria meningitidis. J Exp Med. 2002;195(11):1445–1454. doi: 10.1084/jem.20020407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Martin DR, Ruijne N, McCallum L, O'Hallahan J, Oster P. The VR2 epitope on the PorA P1.7–2,4 protein is the major target for the immune response elicited by the strain-specific group B meningococcal vaccine MeNZB. Clin Vaccine Immunol. 2006;13(4):486–491. doi: 10.1128/cvi.13.4.486-491.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Martinón-Torres F, Carmona Martinez A, Simkó R, et al. Antibody persistence and booster responses 24–36 months after different 4CMenB vaccination schedules in infants and children: a randomised trial. J Infect. 2018;76(3):258–269. doi: 10.1016/j.jinf.2017.12.005. [DOI] [PubMed] [Google Scholar]
- 29.Vesikari T, Esposito S, Prymula R, et al. Immunogenicity and safety of an investigational multicomponent, recombinant, meningococcal serogroup B vaccine (4CMenB) administered concomitantly with routine infant and child vaccinations: results of two randomised trials. Lancet. 2013;381(9869):825–835. doi: 10.1016/s0140-6736(12)61961-8. [DOI] [PubMed] [Google Scholar]
- 30.Prymula R, Esposito S, Zuccotti GV, et al. A phase 2 randomized controlled trial of a multicomponent meningococcal serogroup B vaccine (I) Hum Vaccin Immunother. 2014;10(7):1993–2004. doi: 10.4161/hv.28666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sohn W, Tahrat H, Abitbol V, Bekkat-Berkani R. Review of the four-component meningococcal serogroup b vaccine registrations, recommendations, and use in regional and national immunization programs. Geneva: European Society for Paediatric Infectious Diseases (ESPID); 2021. [Google Scholar]
- 32.European Centre for Disease Prevention and Control (ECDC) Meningococcal Disease: Recommended vaccinations. 2023. https://vaccine-schedule.ecdc.europa.eu/Scheduler/ByDisease?SelectedDiseaseId=48&SelectedCountryIdByDisease=-1. Accessed 14 July 2023.
- 33.Sohn W-Y, Tahrat H, Novy P, Bekkat-Berkani R. Real-world implementation of 4-component meningococcal serogroup B vaccine (4CMenB): implications for clinical practices. Expert Rev Vaccines. 2022;21(3):325–335. doi: 10.1080/14760584.2022.2021881. [DOI] [PubMed] [Google Scholar]
- 34.Deceuninck G, Lefebvre B, Tsang R, Betala-Belinga JF, De Serres G, De Wals P. Impact of a mass vaccination campaign against serogroup B meningococcal disease in the Saguenay-Lac-Saint-Jean region of Quebec four years after its launch. Vaccine. 2019;37(31):4243–4245. doi: 10.1016/j.vaccine.2019.06.021. [DOI] [PubMed] [Google Scholar]
- 35.Ladhani SN, Andrews N, Parikh SR, et al. Vaccination of infants with meningococcal group B vaccine (4CMenB) in England. N Engl J Med. 2020;382(4):309–317. doi: 10.1056/NEJMoa1901229. [DOI] [PubMed] [Google Scholar]
- 36.Rodrigues FMP, Marlow R, Simões MJ, Danon L, Ladhani S, Finn A. Association of use of a meningococcus group B vaccine with group B invasive meningococcal disease among children in Portugal. JAMA. 2020;324(21):2187–2194. doi: 10.1001/jama.2020.20449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Watson PS, Turner DP. Clinical experience with the meningococcal B vaccine, Bexsero(®): prospects for reducing the burden of meningococcal serogroup B disease. Vaccine. 2016;34(7):875–880. doi: 10.1016/j.vaccine.2015.11.057. [DOI] [PubMed] [Google Scholar]
- 38.Biswas HH, Han GS, Wendorf K, et al. Notes from the field: outbreak of serogroup b meningococcal disease at a University California, 2016. MMWR Morb Mortal Wkly Rep. 2016;65(20):520–521. doi: 10.15585/mmwr.mm6520a3. [DOI] [PubMed] [Google Scholar]
- 39.Azzari C, Moriondo M, Nieddu F, et al. Effectiveness and Impact of the 4CMenB vaccine against group b meningococcal disease in two italian regions using different vaccination schedules: a five-year retrospective observational study (2014–2018) Vaccines (Basel) 2020 doi: 10.3390/vaccines8030469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.McMillan M, Wang B, Koehler AP, Sullivan TR, Marshall HS. Impact of meningococcal B vaccine on invasive meningococcal disease in adolescents. Clin Infect Dis. 2021;73(1):e233–e237. doi: 10.1093/cid/ciaa1636. [DOI] [PubMed] [Google Scholar]
- 41.Mentzer D, Oberle D, Keller-Stanislawski B. Adverse events following immunisation with a meningococcal serogroup B vaccine: report from post-marketing surveillance, Germany, 2013 to 2016. Eurosurveillance. 2018;23(17):17–00468. doi: 10.2807/1560-7917.ES.2018.23.17.17-00468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Agenzia Italiana del Farmaco (AIFA) Rapporto Vaccini. 2017. https://www.aifa.gov.it/sites/default/files/Rapp_Vaccini_2017_0.pdf. Accessed 10 March 2023.
- 43.Australian Government Department of Health and Aged Care Bexsero meningococcal B vaccine. Final update monitoring finds no new or unexpected safety issues. 2016. https://www.tga.gov.au/news/safety-alerts/bexsero-meningococcal-b-vaccine. Accessed 10 March 2023.
- 44.Bryan P, Seabroke S, Wong J, et al. Safety of multicomponent meningococcal group B vaccine (4CMenB) in routine infant immunisation in the UK: a prospective surveillance study. Lancet Child Adolesc Health. 2018;2(6):395–403. doi: 10.1016/s2352-4642(18)30103-2. [DOI] [PubMed] [Google Scholar]
- 45.Andrews N, Stowe J, Miller E. Nephrotic syndrome in infants and toddlers before and after introduction of the meningococcal B vaccine programme in England: an ecological study. Vaccine. 2020;38(31):4816–4819. doi: 10.1016/j.vaccine.2020.05.055. [DOI] [PubMed] [Google Scholar]
- 46.Stowe J, Andrews NJ, Turner PJ, Miller E. The risk of Kawasaki disease after pneumococcal conjugate & meningococcal B vaccine in England: a self-controlled case-series analysis. Vaccine. 2020;38(32):4935–4939. doi: 10.1016/j.vaccine.2020.05.089. [DOI] [PubMed] [Google Scholar]
- 47.Hall GC, Douglas I, Heath PT, et al. Post-licensure observational safety study after meningococcal B vaccine 4CMenB (Bexsero) vaccination within the routine UK immunisation program. Vaccine. 2021;39(24):3296–3303. doi: 10.1016/j.vaccine.2021.02.065. [DOI] [PubMed] [Google Scholar]
- 48.De Serres G, Billard MN, Gariépy MC, et al. Short-term safety of 4CMenB vaccine during a mass meningococcal B vaccination campaign in Quebec, Canada. Vaccine. 2018;36(52):8039–8046. doi: 10.1016/j.vaccine.2018.10.095. [DOI] [PubMed] [Google Scholar]
- 49.Marshall HS, Koehler AP, Wang B, et al. Safety of meningococcal B vaccine (4CMenB) in adolescents in Australia. Vaccine. 2020;38(37):5914–5922. doi: 10.1016/j.vaccine.2020.07.009. [DOI] [PubMed] [Google Scholar]
- 50.Marshall HS, McMillan M, Koehler AP, et al. Meningococcal B vaccine and meningococcal carriage in adolescents in Australia. N Engl J Med. 2020;382(4):318–327. doi: 10.1056/NEJMoa1900236. [DOI] [PubMed] [Google Scholar]
- 51.Thabuis A, Tararbit K, Taha MK, et al. Community outbreak of serogroup B invasive meningococcal disease in Beaujolais, France, February to June 2016: from alert to targeted vaccination. Eurosurveillance. 2018 doi: 10.2807/1560-7917.Es.2018.23.28.1700590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ceyhan M, Gürler N, Ozsurekci Y, et al. Meningitis caused by Neisseria meningitidis, Hemophilus influenzae Type B and Streptococcus pneumoniae during 2005–2012 in Turkey. A multicenter prospective surveillance study. Hum Vaccin Immunother. 2014;10(9):2706–2712. doi: 10.4161/hv.29678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Ceyhan M, Ozsurekci Y, Gürler N, et al. Bacterial agents causing meningitis during 2013–2014 in Turkey: a multi-center hospital-based prospective surveillance study. Hum Vaccin Immunother. 2016;12(11):2940–2945. doi: 10.1080/21645515.2016.1209278. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.