Abstract
Colorectal cancer (CRC) is a type of cancer that develops in the colon or rectum and is the second leading cause of cancer-related death worldwide. Several epidemiology studies have identified a significant sexual dimorphism in CRC, with women exhibiting a lower incidence rate and delayed onset compared to men. This study aims to investigate the sexual dimorphism in the inflammatory response in colitis-associated CRC and its relationship with estrogen and estrogen receptors. An azoxymethane (AOM)/dextran sodium sulfate (DSS) mouse model was used to induce colitis-associated CRC. Five-week-old male and female mice were randomly assigned into either the control group or the AOM/DSS CRC group, with 10 mice in each group. Colitis-associated CRC was induced by injecting AOM (10 mg/kg) and administering two-cycles of DSS treatment in the drinking water. The results revealed a significant decrease in colon length exclusively in the female group, indicating more severe colonic inflammation (P < 0.01). A significant interaction was identified between sex and AOM/DSS treatment in the female AOM/DSS group, with higher visceral fat weight compared to their male counterparts (P < 0.05). The female AOM/DSS group also exhibited elevated production of M1 macrophage-related pro-inflammatory cytokines, suggesting increased tumor-associated macrophage activity. Surprisingly, the male AOM/DSS group showed a marked increase in serum estradiol levels, while the female AOM/DSS group exhibited a decrease compared to the normal control group. Additionally, a notable upregulation of both estrogen receptor α and estrogen receptor β expression was observed in the colon tissues of the AOM/DSS groups compared to the normal control groups, with estrogen receptor β expression being particularly pronounced in females. Taken together, our findings suggest that a decline in endogenous estrogen and increased estrogen receptors potentially contribute to the pro-inflammatory response in early CRC by augmenting cytokine expressions associated with M1 macrophage polarization in females.
Keywords: Colorectal cancer, Colitis, Sexual dimorphism, Macrophage, Estrogen, Estrogen receptor
Abbreviations
- CRC
colorectal cancer
- ER
estrogen receptor
- AOM
azoxymethane
- DSS
dextran sodium sulfate
- IFN-γ
interferon-γ
- IL
interleukin
- TNF-α
tumor necrosis factor-α
- TAM
tumor associated macrophage
- DAI
disease activity index
- Arg1
Arginase 1
- Th
T helper
- PPARγ
peroxisome proliferator-activated receptor γ
- SREBP
sterol regulatory element binding proteins
1. Introduction
Colorectal cancer (CRC) is the third most diagnosed cancer and the second leading cause of cancer-related death worldwide as of 2020 [1]. The high morbidity and mortality rates can be attributed to the heterogeneous nature of CRC, which is characterized by a high potential for relapse, metastasis, and a low response rate to conventional anti-cancer treatments. Notably, there is a strong association between chronic inflammatory conditions in the gastrointestinal tract and inflammatory bowel disease, including ulcerative colitis and Crohn's disease [2].
In 2020, the global incidence of all cancer types in men was 19% higher compared to women [1], and this disparity that also applies to CRC [3]. Furthermore, males exhibit a higher likelihood of developing colorectal polyps, colonic adenomas, and carcinomas, which often appear at an earlier age in males [4]. A meta-analysis of population-based cohort studies consistently found a greater risk of CRC in male ulcerative colitis patients [5]. However, it has been demonstrated that adaptive inflammatory and cytotoxic T cell responses are more pronounced in females than in males, and sex hormones play a critical role in determining the intensity of the immune response [[6], [7], [8]].
Inflammatory processes and imbalances in the intestinal immune system have been identified as key risk factors for the initiation and subsequent progression of CRC. CRC is greatly affected by the function and composition of the tumor microenvironment. Macrophages, which differentiate from peripheral monocytes, can adopt two distinct phenotypes: M1 and M2 [9]. The M1 phenotype, triggered by stimulation from interferon-γ (IFN-γ) alone or in conjunction with lipopolysaccharides, exhibits a pro-inflammatory profile characterized by the release of cytokines such as interleukin (IL)-6, IL-12, and tumor necrosis factor-α (TNF-α). On the other hand, IL-4-induced M2 macrophages contribute to pro-tumor immunity [9,10]. Given the association between macrophage infiltration, metastasis, and a poorer prognosis for CRC, tumor-associated macrophages (TAMs) largely consisting of M2 macrophages with a low abundance of M1 macrophages could represent a promising target for the personalized cancer therapy.
Estrogen, a steroid hormone, has been proposed to have a protective effect against CRC. The functions of estrogen in target tissues are mediated through their binding with specific ERs [11]. Estrogen receptor (ER) subtypes, namely alpha (ER-α) and beta (ER-β), exhibit differential expression across various tissues and organs [12]. ER-α is implicated in the initiation of numerous signaling pathways, including STAT, PI3K, and MAPK, and mediates cell proliferation and carcinogenesis in CRC. ER-β, on the other hand, has been found to inhibit cell proliferation, and its expression appears to diminish as CRC progresses, suggesting its protective role against CRC [13]. The protective mechanisms of ER-β in the colon involve the upregulation of occludin and junctional adhesion molecule-A, which helps maintain intestinal permeability homeostasis [14]. Collectively, these observations underscore the potential utility of ERs as a therapeutic target for CRC management.
Although numerous epidemiological studies have elucidated a higher incidence and mortality rate among men compared to women for advanced CRC, the disparities in CRC progression attributed to sex-based differences in immune response or inflammatory mechanisms during early CRC stages remain unclear. In addition, despite the noticeable sex-based disparity across multiple disease spectra, such differences are often not adequately considered in preclinical studies. Thus, it is crucial to gain insight into the physiological characteristics underpinning these sex differences to advance therapeutic strategies. The present study aimed to investigate the underlying relationship of inflammation with estrogen and ERs in the context of sex dimorphism in colitis-associated CRC.
2. Materials and methods
2.1. Animal experiment
Five-week-old male and female BALB/c mice (Central Lab Animal Inc., Seoul, Korea) were maintained in pathogen-free environments and provided with unlimited access to food and water. After a seven-day acclimation period, 40 mice were randomly divided into two groups: a control group treated with vehicle (PBS) and an experimental group treated with azoxymethane/dextran sodium sulfate (AOM/DSS) for nine weeks. The resulting four groups were as follows: (1) Male normal control (M-NC; n = 10), (2) Female normal control (F-NC; n = 10), (3) Male AOM/DSS (M-AOM/DSS; n = 10), and (4) Female AOM/DSS (F-AOM/DSS; n = 10). Colitis-associated CRC was induced using AOM (Sigma-Aldrich, St Louis, MO) and DSS (MP Biomedicals, Irvine, CA) administration as previously described [15] with slight modifications. Each mouse in the AOM/DSS group received an intraperitoneal injection of 10 mg/kg-body-weight AOM. Seven days after AOM administration, a 2% DSS solution was provided ad libitum for seven days, followed by a 14-day recovery period with plain water. This cycle was repeated once, with an additional two-week resting period. The disease activity index (DAI) was calculated every other day from the start of the DSS cycle to monitor disease progression, using body weight loss, stool consistency, and fecal bleeding for the DAI score analysis [16]. The scoring was conducted by researchers who were not aware of the treatment groups. Details of the scoring system are provided in Table 1. The sum of the scores was used as the DAI score.
Table 1.
Detailed scoring system for the disease activity index score used in study.
| Score | Weight loss | Stool consistency | Fecal bleeding |
|---|---|---|---|
| 0 | <1% | Normal | Normal |
| 1 | 1–5% | Slightly loose feces | Slightly bloody |
| 2 | 6–10% | Loose | Bloody |
| 3 | 11–15% | Very loose and wet | Blood in whole colon |
| 4 | >15% | Diarrhea | Blood in whole colon |
Upon termination of the experiment, the mice were euthanized, and their colons were dissected and opened longitudinally. The length of the colons was measured using a digital caliper. The colon tissues were divided into proximal, middle, and distal segments, and all samples were preserved at −80 °C. All procedures were conducted in compliance with the guidelines approved by the Animal Care and Use Committee of Ewha Womans University (Approval No.22-006-t).
2.2. RNA extraction and RT-qPCR
Total RNA was isolated from the colonic tissue using TRIzol reagent (Invitrogen, Waltham, MA) following the manufacturer's guidelines. Lithium chloride (Sigma-Aldrich, St Louis, MO) was employed to purify RNA from the colonic tissue of AOM/DSS-treated mice, as DSS can impede complementary DNA synthesis [17]. RevertAid First strand cDNA Synthesis Kit (Thermo Fisher Scientific, Waltham, MA) was used to generate the first strand cDNA. Quantitative PCR (qPCR) was performed with 1 μm total RNA using a Rotor-Gene SYBR Green PCR (Qiagen, Hilden, Germany). The mRNA levels of the target genes were normalized relative to Tbp. The primer sequences used are provided in Supplementary Table 1.
2.3. Enzyme-linked immunosorbent assay (ELISA)
The concentrations of estradiol and TNF-α in mouse serum were quantified using commercially available ELISA kits (R&D Systems, Minneapolis, MN) following the manufacturer's guidelines.
2.4. Western blot analysis
Colonic tissues were lysed using an ice-cold PRO-PREP protein extraction solution (iNtRON Biotechnology, Seongnam, Korea) containing a phosphatase inhibitor cocktail (Sigma-Aldrich). Protein lysate was resolved on SDS-PAGE gels and transferred onto polyvinylidene fluoride membranes (Millipore, Burlington, MA). The membranes were blocked with 5% non-fat milk for 1 h, followed by overnight incubation at 4 °C with primary antibodies for ER-α (Santa Cruz Biotechnology, Dallas, TX), ER-β (GeneTex, Irvine, CA, USA), and GAPDH (Invitrogen). The membranes were then incubated with an anti-mouse secondary antibody (Santa Cruz Biotechnology) for 1 h. GAPDH was employed as a loading control. Enhanced chemiluminescence (Animal Genetics Inc., Suwon, Korea) was used for band visualization. Band densities were assessed using Image J (National Institutes of Health, Bethesda, MD, USA).
2.5. Statistical analysis
All data are presented as the mean ± standard error of the mean (SEM). Data analysis was performed using GraphPad PRISM (GraphPad Software, San Diego, CA). The Student's t-test was used for comparisons between two groups. A two-way analysis of variance (ANOVA) with a Bonferroni post-hoc test was utilized for analyzing the effects of sex and AOM/DSS treatment. Differences with P values below 0.05 were considered statistically significant.
3. Results
3.1. Sex differences in inflammation-related markers in colitis-associated CRC mice
Throughout the duration of the experiment using the colitis-associated cancer model, the DAI scores were notably higher in the M-AOM/DSS group compared to the F-AOM/DSS group. The greatest difference was observed on day 38, where the M-AOM/DSS group exhibited a 75% higher score (4.2 ± 0.57 vs. 2.4 ± 0.30; P < 0.05) (Fig. 1A).
Fig. 1.
Sex differences in DAI score, fat composition, and the colon length in colitis-associated CRC cancer mice model. (A) Changes of DAI scores, (B) Subcutaneous fat weight, (C) Visceral fat weight (sum of mesenteric, perirenal, and gonadal adipose tissues), and (D) Length of colon, and (E) Concentration of serum TNF-α were analyzed. Each set of data was presented as the means ± SEM. *Bonferroni correction for multiple comparisons following two-way ANOVA. *P < 0.05 and **P < 0.01. CRC, colorectal cancer; DAI, disease activity index.
Adipose tissue, known for its role in the production of steroid hormones and inflammatory mediators, contributes to sustained low-grade intestinal inflammation [18]. Although no statistically significant differences were observed in subcutaneous fat weights across the groups (Fig. 1C), a significant interaction between sex and AOM/DSS treatment was identified in relation to visceral fat weight (P < 0.05). Males tended to have lower levels of visceral fat compared to the NC and AOM/DSS groups, while the females displayed an increased tendency for fat accumulation in the AOM/DSS group. Furthermore, the F-AOM/DSS group exhibited significantly higher visceral fat weight than the M-AOM/DSS group, with 41% higher fat weight (P < 0.05) (Fig. 1B).
Colon length shortening is a hallmark of colon-specific inflammation. After AOM/DSS treatment, a significant reduction (11.6%) in mean colon length was exclusively observed in the female group, compared to the F-NC group (83.57 ± 2.72 mm vs. 73.80 ± 2.31 mm; P < 0.01). The mean colon length in the M-AOM/DSS group was decreased by 8.7% compared to the M − NC group, but this effect was not statistically significant (Fig. 1D). To examine potential sex-based differences in systemic inflammation severity, serum TNF-α levels were analyzed (Fig. 1E). The interaction between sex and AOM/DSS treatment was significant (P < 0.05), with only the female group showing a significant increase in serum TNF-α levels following AOM/DSS treatment compared to the NC group (P < 0.05). Additionally, the serum TNF-α concentration was 45% higher in the F-AOM/DSS group compared to the M-AOM/DSS group (550.6 ± 49.43 pg/mL vs. 378.4 ± 33.34 pg/mL; P < 0.05). These finding highlight the presence of sex-based differences in the inflammatory response.
3.2. Greater macrophage polarization towards M1 macrophages in colitis-associated female CRC mice compared to males
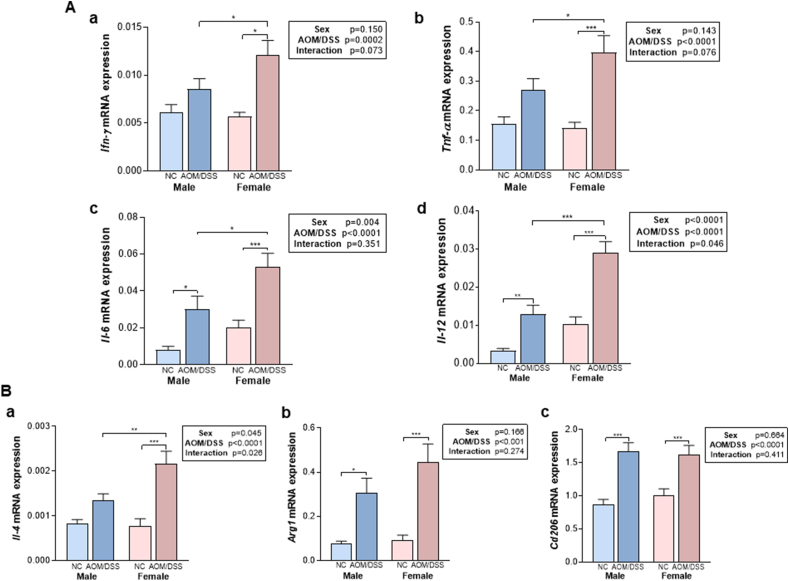
Investigations were carried out to assess the association between macrophage polarization and sexual dimorphism in colitis-associated cancer development. As IFN-γ is known to trigger M1 macrophage polarization [9], the mRNA levels of Ifn-γ, along with M1 markers such as Tnf-α, Il-6, and Il-12 were examined in total colon tissues (Fig. 2A). A significant interaction was observed between sex and AOM/DSS treatment in relation to Il-12 expression (P < 0.05). Expression levels of Ifn-γ (an M1 macrophage stimulator) and M1 markers, Tnf-α, Il-6, and Il-12 were significantly higher in females compared to males after AOM/DSS treatment (P < 0.05 for Ifn-γ, Tnf-α, and Il-6, P < 0.001 for Il-12). AOM/DSS treatment increased mRNA expression of Ifn-γ and three M1 markers in females compared to the normal control, whereas only Il-6 and Il-12 expression levels were elevated in males (Fig. 2Ab and 2Ad).
Fig. 2.
Expression of M1 and M2 macrophage polarization markers in whole colon tissue of colitis-associated CRC cancer mice model. (A) mRNA expressions of (a) Ifn-γ and markers, (b) Tnf-α, (c) Il-6, and (d) Il-12 and (B) The mRNA expressions of (a) Il-4, (b) Arg1, and (c) Cd206 were analyzed by RT-qPCR. Tbp expression served as loading control. Each set of data was presented as the means ± SEM. *Bonferroni correction for multiple comparisons following two-way ANOVA. *P < 0.05, **P < 0.01 and ***P < 0.001. CRC, colorectal cancer.
On the other hand, the expression of Il-4, an M2 macrophage polarization stimulus, showed a significant interaction between sex and AOM/DSS treatment (P < 0.05), with only females demonstrating significantly higher Il-4 expression levels following AOM/DSS treatment (P < 0.001; Fig. 2Ba). In contrast to M1 macrophage markers, no sex-based differences were observed in M2 macrophage markers Arg1 and Cd206, both showing upregulated expressions in the AOM/DSS groups compared to the NC groups (Fig. 2Bb and 2Bc). These results suggest a stronger association between sexual dimorphism in colitis-associated CRC development and M1 macrophage polarization, with AOM/DSS treatment inducing more pronounced polarization towards M1 macrophages in females.
3.3. Greater downregulation of 17β estradiol and upregulation of Esr2 expression in female mice compared to male mice
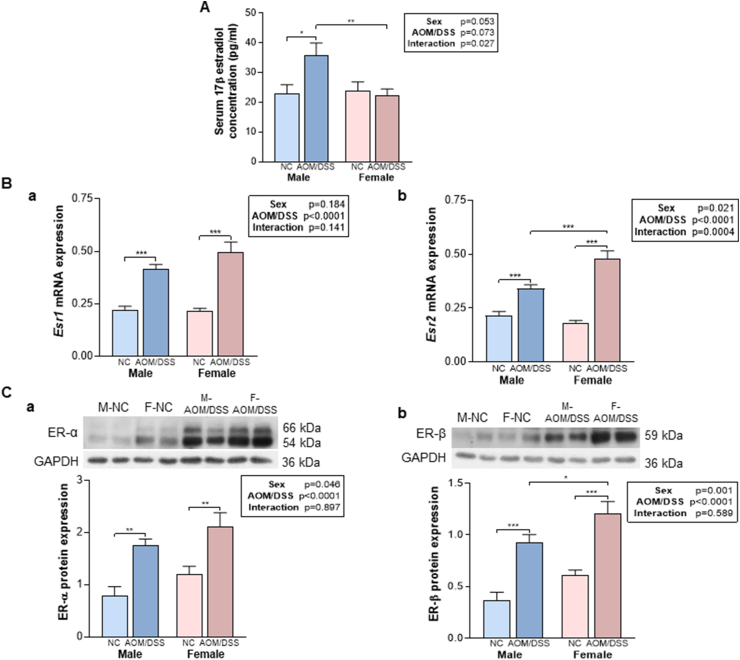
To elucidate the observed difference in inflammatory response between sexes, we investigated the primary biological distinction, sex hormones and their corresponding receptors. A significant interaction between sex and AOM/DSS treatment in serum 17β estradiol levels was detected (P < 0.05). Serum 17β estradiol levels were significantly higher in the male AOM/DSS group compared to the control group (P < 0.05), as well as in the M-AOM/DSS group compared to the F-AOM/DSS group (P < 0.01) (Fig. 3A). The mRNA levels of both Esr1 and Esr2 were significantly upregulated following AOM/DSS treatment in both sexes (P < 0.001). However, there was a sex-based variation, with the F-AOM/DSS group exhibiting higher Esr2 mRNA levels than the M-AOM/DSS group (P < 0.001) (Fig. 3B). These observations in mRNA levels were consistent with the protein levels (Fig. 3C). These findings indicate an association between reduced estradiol levels and enhanced ER-β expression, correlating with more severe inflammation in females in the colitis-associated CRC model.
Fig. 3.
Serum levels of 17β estradiol and expression of ER-α and ER-β in colon tissue of colitis-associated CRC mice. (A) Serum 17β estradiol levels were assessed by ELISA. (B) The mRNA expression of (a) Esr1 and (b) Esr2 was confirmed by RT-qPCR. Tbp expression was served as loading control. (C) Representative blots were shown and (a) ER-α and (b) ER-β expression levels were quantified. GAPDH was used as loading control. Full images of uncropped for (C) are provided in Supplementary Fig. 3C(a) and Supplementary Fig. 3C(b). Each value was presented as the means ± SEM. * Bonferroni correction for multiple comparisons following two-way ANOVA. *P < 0.05, **P < 0.01, ***P < 0.001. CRC, colorectal cancer.
3.4. Upregulated ERs expression induced by AOM/DSS treatment is negatively correlated with colon length
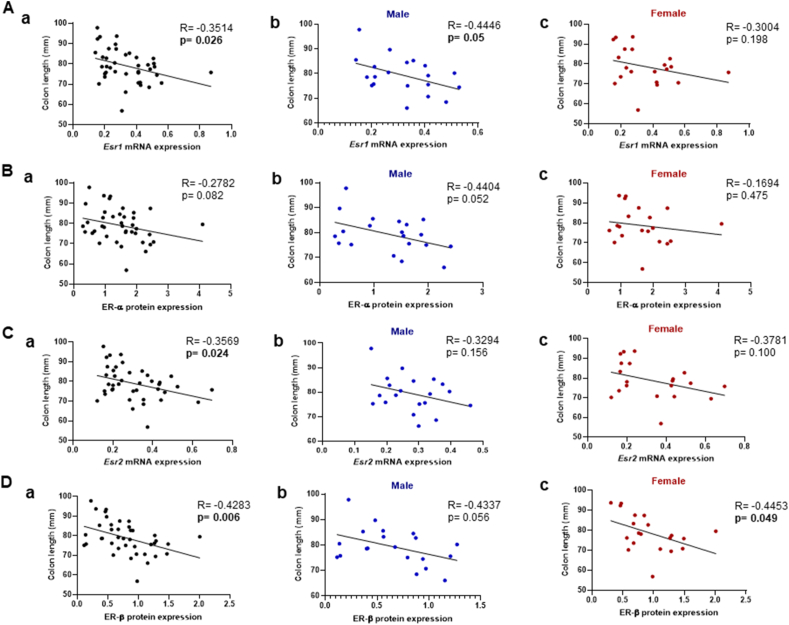
To explore the association between the observed upregulation of ER expression and colonic inflammation, a correlation analysis was conducted between colon length and ER expression (Fig. 4). A significant negative correlation was found between colon length and Esr1 mRNA expression in all mice, when the two sexes were combined (r = −0.3514; P < 0.05) (Fig. 4Aa). The negative correlation between colon length and Esr1 mRNA expression was only observed in male mice (r = −0.444; P = 0.05) (Fig. 4Ab). At the protein levels, no significant correlation was observed between colon length and ER-α expression in all mice (Fig. 4Ba). While a trend toward a negative correlation between colon length and ER-α protein expression was observed in males, no such correlation was found in females (Fig. 4Bb and 4Bc). These findings suggest that the correlation between increased ER-α expression and the severity of colonic inflammation was stronger in males compared to in females.
Fig. 4.
Correlation between colon length and ER expression in colitis-associated CRC mice. Correlation of colon length and (A) Esr1 mRNA expression, (B) ER-α protein expression, (C) Esr2 mRNA expression, and (D) ER-β protein expression. (a) whole, (b) male, and (c) female mice. Pearson's correlation coefficient was used for the statistical analyses. The solid line represents the linear function for each mouse and correlation of whole, male, or female mice are shown respectively. P < 0.05 was considered statistically significant. The 95% confidence intervals for each correlation analysis were derived as follows. (A) (a) [-0.5975 to −0.04479], (b) [-0.7413 to −0.002578], (c) [-0.6557 to 0.1639]. (B) (a) [-0.6526 to −0.1345], (b) [-0.7522 to −0.02738], (c) [-0.7254 to 0.03186]. (C) (a) [-0.6015 to −0.05107], (b) [-0.6737 to 0.1324], (c) [-0.7030 to 0.07741]. (D) (a) [-0.6376 to −0.1093], (b) [-0.6522 to 0.1700], (c) [-0.7623 to −0.05082]. CRC, colorectal cancer.
In the case of ER-β, colon length was negatively correlated with mRNA levels in all mice (r = −0.3569, P < 0.05; Fig. 4Ca). No statistically significant correlation was found between colon length and Esr2 mRNA expression in either males or females (Fig. 4Cb and 4Cc). At the protein level, however, a negative correlation between colon length and ER-β expression was observed in all mice (r = −0.4283; P < 0.01; Fig. 4Da) and in females (r = −0.4453; P < 0.05; Fig. 4Dc). In cases of reduced colon length, males showed a stronger correlation between elevated ER-α and colonic inflammation, while females exhibited a stronger correlation with ER-β expression. Overall, the upregulation of ER expression induced by AOM/DSS treatment corresponded with the severity of colonic inflammation, and the relationship between ER subtype and inflammation was found to be sex-dependent. The pronounced pro-inflammatory response in females may be attributed to elevated ER-β expression.
4. Discussion
This study aimed to investigate the molecular mechanisms behind the observed sexual dimorphism in a mouse model of colitis-associated CRC, with a particular focus on the roles of estrogen and ERs in relation to colonic inflammation. AOM/DSS treatment induced a heightened inflammatory response in female mice compared to their male counterparts. This was accompanied by an enhanced polarization towards M1 macrophages and a significantly higher expression of M1 markers in female CRC mice compared to male CRC mice. Furthermore, female mice showed lower serum estradiol levels and higher ER expression compared to male CRC mice. These findings provide evidence that the observed downregulation of estrogen and upregulation of ERs in females exert pro-inflammatory effects in CRC, specifically by increasing the expression of cytokines associated with M1 macrophage polarization.
To investigate the effects of sex on colonic inflammation and early CRC development, we utilized the AOM/DSS-induced CRC mouse model [19]. This model is well-established for determining the multi-step progress of CRC, based on the development of aberrant crypt foci (ACF)-adenoma-carcinoma. It allows for the assessment of biomolecular alterations and tumor progression at specific stages of carcinogenesis. In addition, the fact that the predominant location of tumors is the distal colon reflects the characteristics of human CRC patients [20]. Since most clinical studies have focused on patients with CRC after the early stages, the AOM/DSS model can provide a meaningful basis for a studying the sequential onset of CRC from colitis. However, it is important to consider the differences in the ovulation cycle between humans and mice when analyzing the mechanisms of the endocrine system in future studies.
Throughout the DSS treatment period, male mice exhibited higher DAI scores than female mice within the AOM/DSS groups, which is consistent with a previous study showing a trend of lower DAI scores in female CRC mice [21]. This difference may be attributed to the degree of weight loss included in the DAI score. However, a reduction in colon length was exclusively observed in the F-AOM/DSS group compared to the NC group, indicating a higher level of colonic inflammation induced by AOM/DSS treatment in females. Overall, the severity of colon-specific inflammation was more pronounced in female mice compared to male mice.
The proliferation and progression of CRC can be attributed to the utilization of TAMs. However, there is a lack of evidence regarding the sexual difference in the polarization of macrophages under CRC. In our data, we observed higher expression of M1 polarization stimuli and markers in females. This higher expression of phospholipase D4 (PLD4) and the IL12/GM-CSF pathway promotes polarization towards M1 macrophages, resulting in an anti-tumor effect in CRC [22,23]. Therefore, enhanced activation of M1 macrophages could be intricately linked to improved prognosis in females. Previous studies have shown that a T helper (Th) cell 2-oriented response, including IL-4+CD4+ T cells, is more prevalent within the tumor microenvironment of females, potentially improving the survival of a metastatic CRC model [24]. Therefore, the type of Th cell that is remarkably activated may vary depending on the progressive stage of the CRC model. As IFN-γ and IL-4 function as chief cytokines for the differentiation of Th1 and Th2 cells, and they serve as M1 and M2 macrophage polarization stimuli [25], further investigation is required to understand the molecular mechanisms associated with various macrophage phenotypes and T cell activity in relation to sex disparities.
Adipose tissues secrete various adipokines, such as leptin, TNF-α, and IL-6, which serve as pro-inflammatory cytokines and contribute to cancer progression [26]. Estrogen impacts adiposity by regulating genes associated with lipogenesis and lipolysis, such as SREBP-1c and PPARγ [27]. In addition, there is an inverse correlation between estrogen levels and visceral fat accumulation [28]. Our data revealed that the levels of 17β-estradiol in the female CRC group were lower compared to the male CRC group. Furthermore, the female CRC group exhibited an increase in visceral fat distribution compared to the normal control group, while no corresponding difference was observed in male mice. Hence, consistent with previous findings, these results indicate that the elevated visceral fat accumulation and pro-inflammatory responses in female CRC mice may be due to a reduction in estrogen activity. It is possible that changes in sex steroid levels resulting from CRC induction may affect the composition of visceral adipose tissue, promoting pro-inflammatory response.
In the present study, the upregulation of ER-β expression in the AOM/DSS group compared to the NC group was more notable in females. This upregulated ER-β expression may be linked to a stronger inflammatory response in the female CRC model. ER-β plays a protective role in tumorigenesis by inhibiting the proliferation of cancer cells and promoting cell cycle arrest and differentiation [29,30]. The persistent elevation of ER-β expression in the female AOM/DSS group compared to the male AOM/DSS group supports the anti-tumor role of ER-β in CRC. The functions of inflammatory cells include neutralizing pathogens, fostering the repair of damaged tissues, and obstructing tumor growth. A previous study revealed that TNF-α enhances ER-β transactivation, thereby increasing the anti-tumor efficacy of ER-β in CRC. ER-β achieves this by directly binding to the chromatin regions of NFκB regulators and suppressing the activator genes of NFκB [21]. Consequently, the enhanced ER-β expression and intense inflammatory response in females during early CRC might lead to a reduced risk of cancer progression and mortality. Further exploration of the interplay between estrogen, ER, and the inflammatory response in tumorigenesis is warranted to optimize anti-cancer treatment strategies for CRC.
The strength of the present study is that all groups were statistically analyzed simultaneously, allowing for the identification of the main effects of sex and AOM/DSS treatment, as well as the interaction between these main effects. In addition, as we stated above, the present study contributes to the understanding of fat distribution changes during the CRC development. Previous studies have found that the accumulation of visceral adipose tissue is associated with an increased risk of CRC in postmenopausal women [31,32] and men [33]. While numerous studies have investigated the relationship between obesity and the risk and onset of CRC, there is a lack of evidence regarding the distribution of adipose tissues after the onset of CRC. Therefore, the results of the present study provide valuable insights into the potential correlation between adipose tissue, estrogen, and inflammation in CRC for the first time.
One limitation of this study is that the sex differences in the anatomical location of tumors was not considered. Anatomical location is an important factor that affects the molecular, pathological, and clinical development, as well as the mortality of CRC [34]. Estrogen plays a critical role in CRC development, and its effects vary depending on the part of the colon [35]. There is a growing need for screening CRC in sex-specific sites. In addition, the features of tumor were not investigated sufficiently because the early colitis-associated CRC model was used, and the tumor development was not significant enough to show notable differences between sexes.
5. Conclusion
In the colitis-associated CRC model, inflammatory responses were more pronounced in female mice compared to male mice. Endogenous estrogen, the estrogen receptor, and inflammation contribute to sex-specific variations in early CRC. Furthermore, considering the diverse stages of CRC could enhance our understanding of prognostic outcomes and mortality rate, ultimately leading to improved efficacy of targeted cancer therapies for both sexes.
Funding
This work was supported by Basic Science Research Program through the National Research Foundation of Korea funded by the Minister of the Ministry of Science and ICT [Grant No. 2022R1A2C1009512] and was also supported by the BK21 FOUR (Fostering Outstanding Universities for Research) funded by the 501100002701 Ministry of Education (MOE, Korea) and National Research Foundation of Korea (NRF-5199990614253, Education Research Center for 4IR-Based Health Care).
Data availability statement
The datasets are available from the corresponding author on reasonable request and with permission.
CRediT authorship contribution statement
Seoyoung Jang: Writing – original draft, Validation, Methodology, Data curation. Hyejin Han: Methodology. Yeonsoo Oh: Methodology. Yuri Kim: Writing – review & editing, Supervision, Project administration, Investigation, Funding acquisition, Conceptualization.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.heliyon.2024.e28121.
Appendix A. Supplementary data
The following are the Supplementary data to this article.
Uncropped scans of the Western blots presented in Fig. 3C(a). The first blot of each row is the representative blot shown in the figure. The red boxes mark the borders of the detected band of each antibody. The molecular weight markers are shown on the blots. No 1, 2: Normal control male, No 3, 4: Normal control female, No. 5, 6: AOM/DSS male, 7, 8: AOM/DSS female.
Uncropped scans of the Western blots presented in Fig. 3C(b). The first blot of each row is the representative blot shown in the figure. The red arrows and boxes mark the borders of the detected band of each antibody. The molecular weight markers are shown on the blots. No 1, 2: Normal control male, No 3, 4: Normal control female, No. 5, 6: AOM/DSS male, 7, 8: AOM/DSS female.
References
- 1.Sung H., Ferlay J., Siegel R.L., Laversanne M., Soerjomataram I., Jemal A., Bray F., Global Cancer Statistics 2020: GLOBOCAN Estimates of incidence and mortality worldwide for 36 cancers in 185 Countries. CA. Cancer J. Clin. 2021;71(3):209–249. doi: 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- 2.Ng S.C., Shi H.Y., Hamidi N., Underwood F.E., Tang W., Benchimol E.I., Panaccione R., Ghosh S., Wu J.C.Y., Chan F.K.L., Sung J.J.Y., Kaplan G.G. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: a systematic review of population-based studies. Lancet. 2017;390(10114):2769–2778. doi: 10.1016/S0140-6736(17)32448-0. [DOI] [PubMed] [Google Scholar]
- 3.Kang M.J., Jung K.W., Bang S.H., Choi S.H., Park E.H., Yun E.H., Kim H.J., Kong H.J., Im J.S., Seo H.G. Cancer Statistics in Korea: incidence, mortality, survival, and prevalence in 2020. Cancer Res. Treat. 2023;55(2):385–399. doi: 10.4143/crt.2023.447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.McCashland T.M., Brand R., Lyden E., de Garmo P. Gender differences in colorectal polyps and tumors. Am. J. Gastroenterol. 2001;96(3):882–886. doi: 10.1111/j.1572-0241.2001.3638_a.x. [DOI] [PubMed] [Google Scholar]
- 5.Jess T., Rungoe C., Peyrin–Biroulet L. Risk of colorectal cancer in patients with ulcerative colitis: a meta-analysis of population-based cohort studies. Clin. Gastroenterol. Hepatol. 2012;10(6):639–645. doi: 10.1016/j.cgh.2012.01.010. [DOI] [PubMed] [Google Scholar]
- 6.Butterworth M., McClellan B., Allansmith M. Influence of sex in immunoglobulin levels. Nature. 1967;214(5094):1224–1225. doi: 10.1038/2141224a0. [DOI] [PubMed] [Google Scholar]
- 7.Gilliver S.C. Sex steroids as inflammatory regulators. J. Steroid Biochem. Mol. Biol. 2010;120(2–3):105–115. doi: 10.1016/j.jsbmb.2009.12.015. [DOI] [PubMed] [Google Scholar]
- 8.Hewagama A., Patel D., Yarlagadda S., Strickland F.M., Richardson B.C. Stronger inflammatory/cytotoxic T-cell response in women identified by microarray analysis. Genes Immun. 2009;10(5):509–516. doi: 10.1038/gene.2009.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hao N.B., Lü M.H., Fan Y.H., Cao Y.L., Zhang Z.R., Yang S.M. Macrophages in tumor microenvironments and the progression of tumors. Clin. Dev. Immunol. 2012 doi: 10.1155/2012/948098. 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sica A., Bronte V. Altered macrophage differentiation and immune dysfunction in tumor development. J. Clin. Invest. 2007;117(5):1155–1166. doi: 10.1172/JCI31422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Nilsson S., Gustafsson J. Estrogen receptors: therapies targeted to receptor subtypes. Clin. Pharmacol. Ther. 2011;89(1):44–55. doi: 10.1038/clpt.2010.226. [DOI] [PubMed] [Google Scholar]
- 12.Kuiper G.G.J.M., Carlsson B., Grandien K., Enmark E., Häggblad J., Nilsson S., Gustafsson J-Ak. Comparison of the Ligand binding Specificity and transcript tissue distribution of estrogen receptors α and β. Endocrinology. 1997;138(3):863–870. doi: 10.1210/endo.138.3.4979. [DOI] [PubMed] [Google Scholar]
- 13.Chen J., Iverson D. Estrogen in obesity-associated colon cancer: friend or foe? Protecting postmenopausal women but promoting late-stage colon cancer. Cancer Causes Control. 2012;23(11):1767–1773. doi: 10.1007/s10552-012-0066-z. [DOI] [PubMed] [Google Scholar]
- 14.Braniste V., Leveque M., Buisson-Brenac C., Bueno L., Fioramonti J., Houdeau E. Oestradiol decreases colonic permeability through oestrogen receptor beta-mediated up-regulation of occludin and junctional adhesion molecule-A in epithelial cells. J. Physiol. 2009;587(pt 13):3317–3328. doi: 10.1113/jphysiol.2009.169300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Jeon H.J., Yeom Y., Kim Y.S., Kim E., Shin J.H., Seok P.R., Woo M.J., Kim Y. Effect of vitamin C on azoxymethane (AOM)/dextran sulfate sodium (DSS)-induced colitis-associated early colon cancer in mice. Nutr. Res. Pract. 2018;12(2):101–109. doi: 10.4162/nrp.2018.12.2.101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Okayasu I., Hatakeyama S., Yamada M., Ohkusa T., Inagaki Y., Nakaya R. A novel method in the induction of reliable experimental acute and chronic ulcerative colitis in mice. Gastroenterology. 1990;98(3):694–702. doi: 10.1016/0016-5085(90)90290-h. [DOI] [PubMed] [Google Scholar]
- 17.Viennois E., Chen F., Laroui H., Baker M.T., Merlin D. Dextran sodium sulfate inhibits the activities of both polymerase and reverse transcriptase: lithium chloride purification, a rapid and efficient technique to purify RNA. BMC Res. Notess. 2013;6:360. doi: 10.1186/1756-0500-6-360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Balistreri C.R., Caruso C., Candore G. The role of adipose tissue and adipokines in obesity-related inflammatory diseases. Mediators Inflamm. 2010;2010 doi: 10.1155/2010/802078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tanaka T. Development of an inflammation-associated colorectal cancer model and its Application for Research on carcinogenesis and Chemoprevention. Int. J. Inflamm. 2012;2012 doi: 10.1155/2012/658786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.De Robertis M., Massi E., Poeta M.S., Carotti S., Morini S., Cecchetelli L., Signori E., Fazio V.M. The AOM/DSS murine model for the study of colon carcinogenesis: from pathways to diagnosis and therapy studies. J. Carcinog. 2011;10:9. doi: 10.4103/1477-3163.78279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hases L., Indukuri R., Birgersson M., Nguyen-Vu T., Lozano R., Saxena A., Hartman J., Frasor J., Gustafsson J.-Å., Katajisto P., Archer A., Williams C. Intestinal estrogen receptor beta suppresses colon inflammation and tumorigenesis in both sexes. Cancer Lett. 2020;491:54–62. doi: 10.1016/j.canlet.2020.06.021. [DOI] [PubMed] [Google Scholar]
- 22.Gao L., Zhou Y., Zhou S.X., Yu X.J., Xu J.M., Zuo L., Luo Y.H., Li X.A. PLD4 promotes M1 macrophages to perform antitumor effects in colon cancer cells. Oncol. Rep. 2017;37(1):408–416. doi: 10.3892/or.2016.5216. [DOI] [PubMed] [Google Scholar]
- 23.Cheng Y., Zhu Y., Xu W., Xu J., Yang M., Chen P., Zhao J., Geng L., Gong S. PKCα in colon cancer cells promotes M1 macrophage polarization via MKK3/6-P38 MAPK pathway. Mol. Carcinog. 2018;57(8):1017–1029. doi: 10.1002/mc.22822. [DOI] [PubMed] [Google Scholar]
- 24.Ray A.L., Nofchissey R.A., Khan M.A., Reidy M.A., Lerner M.R., Wu X., Guo S., Hill S.L., Weygant N., Adams S.F., Castillo E.F., Berry W.L., Stout M.B., Morris K.T. The role of sex in the innate and adaptive immune environment of metastatic colorectal cancer. Br. J. Cancer. 2020;123(4):624–632. doi: 10.1038/s41416-020-0913-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zissler U.M., Chaker A.M., Effner R., Ulrich M., Guerth F., Piontek G., Dietz K., Regn M., Knapp B., Theis F.J., Heine H., Suttner K., Schmidt-Weber C.B. Interleukin-4 and interferon-γ orchestrate an epithelial polarization in the airways. Mucosal Immunol. 2016;9(4):917–926. doi: 10.1038/mi.2015.110. [DOI] [PubMed] [Google Scholar]
- 26.Cheung O.K., Cheng A.S. Gender differences in Adipocyte Metabolism and Liver cancer progression. Front. Genet. 2016;7:168. doi: 10.3389/fgene.2016.00168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.D'Eon T.M., Souza S.C., Aronovitz M., Obin M.S., Fried S.K., Greenberg A.S. Estrogen regulation of adiposity and fuel partitioning. Evidence of genomic and non-genomic regulation of lipogenic and oxidative pathways. J. Biol. Chem. 2005;280(43):35983–35991. doi: 10.1074/jbc.M507339200. [DOI] [PubMed] [Google Scholar]
- 28.Brown L.M., Clegg D.J. Central effects of estradiol in the regulation of food intake, body weight, and adiposity. J. Steroid Biochem. Mol. Biol. 2010;122(1–3):65–73. doi: 10.1016/j.jsbmb.2009.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wei Y., Huang C., Wu H., Huang J. Estrogen receptor beta (ERβ) mediated-CyclinD1 Degradation via Autophagy plays an anti-proliferation role in colon cells. Int. J. Biol. Sci. 2019;15(5):942–952. doi: 10.7150/ijbs.30930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Konstantinopoulos P.A., Kominea A., Vandoros G., Sykiotis G.P., Andricopoulos P., Varakis I., Sotiropoulou-Bonikou G., Papavassiliou A.G. Oestrogen receptor beta (ERbeta) is abundantly expressed in normal colonic mucosa, but declines in colon adenocarcinoma paralleling the tumour's dedifferentiation. Eur. J. Cancer. 2003;39(9):1251–1258. doi: 10.1016/s0959-8049(03)00239-9. [DOI] [PubMed] [Google Scholar]
- 31.Kabat G.C., Kim M.Y., Stefanick M., Ho G.Y.F., Lane D.S., Odegaard A.O., Simon M.S., Bea J.W., Luo J., Wassertheil-Smoller S., Rohan T.E. Metabolic obesity phenotypes and risk of colorectal cancer in postmenopausal women. Int. J. Cancer. 2018;143(3):543–551. doi: 10.1002/ijc.31345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lee J.Y., Lee H.S., Lee D.C., Chu S.H., Jeon J.Y., Kim N.K., Lee J.W. Visceral fat accumulation is associated with colorectal cancer in postmenopausal women. PLoS One. 2014;9(11) doi: 10.1371/journal.pone.0110587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Seo I.K., Kim B.J., Kim B., Choi C.H., Kim J.W., Kim J.G., Chang S.K., Kang H. Abdominal fat distribution measured using computed tomography is associated with an increased risk of colorectal adenoma in men. Medicine. 2017;96(37) doi: 10.1097/MD.0000000000008051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Missiaglia E., Jacobs B., D'Ario G., Di Narzo A.F., Soneson C., Budinska E., Popovici V., Vecchione L., Gerster S., Yan P., Roth A.D., Klingbiel D., Bosman F.T., Delorenzi M., Tejpar S. Distal and proximal colon cancers differ in terms of molecular, pathological, and clinical features. Ann. Oncol. 2014;25(10):1995–2001. doi: 10.1093/annonc/mdu275. [DOI] [PubMed] [Google Scholar]
- 35.Song C.H., Kim N., Lee S.M., Nam R.H., Choi S.I., Kang S.R., Shin E., Lee D.H., Lee H.N., Surh Y.J. Effects of 17β-estradiol on colorectal cancer development after azoxymethane/dextran sulfate sodium treatment of ovariectomized mice. Biochem. Pharmacol. 2019;164:139–151. doi: 10.1016/j.bcp.2019.04.011. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets are available from the corresponding author on reasonable request and with permission.