Abstract
The tumor microenvironment corresponds to a complex mixture of bioactive products released by local and recruited cells whose normal functions have been “corrupted” by cues originating from the tumor, mostly to favor cancer growth, dissemination and resistance to therapies. While the immune and the mesenchymal cellular components of the tumor microenvironment in colon cancers have been under intense scrutiny over the last two decades, the influence of the resident neural cells of the gut on colon carcinogenesis has only very recently begun to draw attention. The vast majority of the resident neural cells of the gastrointestinal tract belong to the enteric nervous system and correspond to enteric neurons and enteric glial cells, both of which have been understudied in the context of colon cancer development and progression. In this review, we especially discuss available evidence on enteric glia impact on colon carcinogenesis. To highlight “corrupted” functioning in enteric glial cells of the tumor microenvironment and its repercussion on tumorigenesis, we first review the main regulatory effects of enteric glial cells on the intestinal epithelium in homeostatic conditions and we next present current knowledge on enteric glia influence on colon tumorigenesis. We particularly examine how enteric glial cell heterogeneity and plasticity require further appreciation to better understand the distinct regulatory interactions enteric glial cell subtypes engage with the various cell types of the tumor, and to identify novel biological targets to block enteric glia pro-carcinogenic signaling.
Keywords: Enteric glial cell, Colon cancer, Tumor microenvironment, Cancer stem cell, Intestinal stem cell
Introduction
With approximately 107,000 new cases and 52,500 deaths from colon (and rectum) cancer in the US in 2023, colon cancer is the second deadliest cancer after lung (and bronchus) cancer[1]. Major improvements in screening have greatly promoted the detection of early stage and localized tumors, for which surgical resection alone is essentially sufficient to achieve complete remission. However, late stage cancers, which require complementary systemic chemotherapy, are still associated with poor survival outcomes, with the five-year survival rate of patients with stage IV colon cancer remaining a dismal 14%[2]. A substantial body of evidence accumulated over the last two decades[3] suggests that the impetus of colonic tumors corresponds to a specific subset of cancer cells, which, upon stimulation by specific intrinsic or extrinsic cues[4], acquire increased tumor-propagating[5,6], metastasis[7] and chemoresistance[8] abilities. These cells have been broadly termed cancer stem cells and are prime targets for the development of colon cancer therapies. One exciting approach to target cancer stem cells is to block extrinsic molecular signals that trigger cancer stem cell pro-carcinogenic abilities, for instance through the use neutralizing monoclonal antibody-based therapy. Thus, a better understanding of the molecular interactions between cancer (stem) cells and the tumor microenvironment will likely result in the identification of new molecular targets to block cancer stem cells.
The tumor microenvironment corresponds to a complex mixture of bioactive molecules originating from various local and recruited cell types. Current evidence indicates that the vast majority of cells present in the tumor microenvironment are subjected to profound alterations “corrupting” their normal functioning so that they become pro-carcinogenic. In colon cancer, we and others have shown that the two cellular components of the enteric nervous system, i.e., enteric neurons and enteric glial cells, are integral elements of the tumor microenvironment [9–11], yet how the tumor microenvironment alters the cells of the enteric nervous system and how in return the cells of the enteric nervous system influence colon carcinogenesis remains poorly understood. In this review devoted to enteric glia, we will briefly summarize the main regulatory effects of enteric glial cells on the intestinal epithelium in homeostatic conditions, with a specific emphasis on the stem cell compartment. Next, we will review current knowledge on enteric glia influence on colon tumorigenesis and highlight critical unanswered questions.
Enteric glial cells are potent regulators of epithelial cell functions
Enteric glial cells are the most abundant cell type of the enteric nervous system as they outnumber enteric neurons by 4–6 depending on species[12,13]. They express the glial markers GFAP (glial fibrillary acidic protein), S-100β (S100 calcium-binding protein B), Sox10 (SRY-Box Transcription Factor 10) and PLP1 (Proteolipid protein 1). In contrast to neurons whose cell bodies are confined to the ganglia of the submucosal plexus and myenteric plexus, enteric glial cells are found throughout every layer of the gastrointestinal wall where they functionally interact with a wide variety of cell types to regulate gastrointestinal functions, including notably enteric neurons, immune cells and intestinal epithelial cells. In this paragraph we will restrict our focus to the functional interactions enteric glial cells engage with epithelial cells of the intestinal crypts.
While enteric glial cells were long known to be an integral cellular component of the lamina propriae of the intestinal mucosa[14–16], it was only in 1998 that they were shown to markedly affect the intestinal epithelium[17]. In this seminal study, Bush and colleagues engineered the transgenic GFAP-HSV-Tk (herpes simplex virus thymidine kinase) mouse model to assess the impact of GFAP-expressing cell ablation following ganciclovir treatment. In this mouse model, the suicide gene HSV-Tk is expressed in GFAP+ cells where it phosphorylates the non-toxic prodrug ganciclovir into a DNA synthesis inhibitor and a cell cycle blocker resulting in cell death. They found that ganciclovir, delivered continuously subcutaneously at a rate of 100 mg/kg/day, induced the death of GFAP+ cells specifically in the jejunum and ileum, invariably leading to the death of mice within 19 days due to a fulminant jejuno-ileitis. In addition to severe inflammation, hemorrhage and distension, mice given ganciclovir for >11 days showed drastic alterations of the epithelium including pronounced crypt hyperplasia, increased cell sloughing at the villus top and loss of continuity of the epithelium. Subsequent studies using the same mouse model demonstrated that increased crypt proliferation[18] and increased intestinal permeability[19] preceded signs of inflammation, and confirmed in vitro in co-culture studies that enteric glial cells limit intestinal epithelial cell proliferation[18,20] and promote barrier function[19]. While these initial studies set out the basis for a central role of enteric glial cells in the regulation of epithelial cell functions, the development of multiple distinct transgenic mouse models and the refinement of technologies have led to the generation of conflicting results. For instance, while transgenic animals allowing for CD8(+) T cell-mediated autoimmune targeting of GFAP+ glial cells confirmed that loss of enteric glia leads to a fulminant and lethal inflammatory response[21] and an increase in intestinal permeability independent of the inflammation[22], other transgenic models using the diphtheria toxin inducible system to deplete PLP1+ cells[23] or inversely using the DREADD (Designer Receptors Exclusively Activated by Designer Drugs) technology to activate calcium signaling in GFAP+ cells[24] showed no impact of enteric glial cells on intestinal permeability. Similarly, whereas initial studies proposed that loss of GFAP+ cells using the HSV-Tk/ganciclovir system led to crypt hyperplasia and increased 3H-thymidine uptake[18], more recent studies suggested that loss of PLP1 cells using the diphtheria toxin system did not impact crypt proliferation[23]. One likely explanation reconciling these seemingly contradictory findings is that enteric glia, contrary to previous dogma, are a functionally heterogeneous cell population as revealed by elegant studies from Boesmans and colleagues. Using the MADM (mosaic analysis with double markers) mouse model to label individual Sox10+ cells, they confirmed seminal work by Hanani and Reichenbach[25] demonstrating the existence of four distinct enteric glial cell subtypes with unique morphological characteristics based on their localization in the gut wall, and importantly, they showed that these different enteric glial cell subtypes exhibit differential calcium responses to ATP[26]. Consistently, recent work by Touvron et al. indicates that enteric glial cells isolated from the mucosa/submucosa have different calcium responses to ATP as compared to enteric glial cells isolated from the muscle layers and the myenteric plexus[27]. Similarly, recent work from the Gulbransen lab shows that duodenal enteric glial cells and colonic enteric glial cells produce different calcium responses to ADP and cholecystokinin[28]. Differential calcium responses to neuromodulators in discrete enteric glial cell populations strongly suggest that these enteric glial cell subtypes may perform different functions. While thorough functional analysis of single cell RNAseq datasets, such as the ones recently generated[29–33], combined with follow-up functional studies are needed to comprehensively characterize the functional heterogeneity of enteric glia, this body of work also implies that targeting different enteric glial cell populations using distinct promoters in transgenic mice is likely to lead to different functional outcomes. This postulate was recently confirmed by Baghdadi and colleagues who used the diphtheria toxin system to assess the impact of PLP1+ or GFAP+ cell depletion on crypt proliferation and on intestinal stem cell activity[34]. Confirming prior studies[17,18,23] they reported an increase in crypt proliferation in mice depleted for GFAP+ cells, but not PLP1+ cells. They also observed an early increase in intestinal permeability followed at later time points by a collapse of the intestinal epithelium in double transgenic mice lacking both GFAP+ and PLP1+ cells. Importantly, Baghdadi et al. further found that loss of GFAP+ cells, but not PLP1+ cells, led to decreased intestinal stem cell number and organoid-forming ability as well as reduced early epithelial regenerative response driven by intestinal stem cells, demonstrating that discrete enteric glial cell populations engage distinct regulatory interactions with the various cell types within intestinal crypts, including intestinal stem cells.
While juxtracrine communication between enteric glial cells and intestinal epithelial cells has been proposed[35], the vast majority of published studies have reported that enteric glial cells impact intestinal epithelial cell functions via paracrine cues. Overall, early literature suggests that discrete enteric glial cell-derived ligands regulate specific intestinal epithelial cell functions. For instance, enteric glial cells reduce intestinal permeability via the release of GSNO[19] and 15-HETE[36], restrict intestinal epithelial cell proliferation through the secretion of TGF-β1[18] and 15d-PGJ2[37], and promote cell restitution by producing pro-EGF[38], 11βPGF2α[39] and GDNF[40]. Baghdadi and colleagues used single cell RNAseq data to demonstrate that distinct enteric glial cell subpopulations express different levels of Wnt ligands, and confirmed that inhibition of Wnt secretion in GFAP+ cells, but not in PLP1+ cells, leads to defects in intestinal stem cell regenerative abilities[34]. These recent findings reinforce the concept that discrete enteric glial cell subpopulations harbor unique secretomes resulting in distinct regulatory interactions with cells of the intestinal epithelium. Altogether all these exciting findings demonstrate that more work is required to fully characterize the different enteric glial cell subpopulations and better comprehend the complexity of the functional interactions between enteric glial cells and the different types of intestinal epithelial cells. These critical studies will also undoubtedly uncover potential regulatory mechanisms altered in gastrointestinal and extra-digestive pathologies.
Enteric glial cells are an understudied cellular component of the tumor microenvironment
As discussed in recent reviews[41–45], enteric glial cells have been proposed to be involved in virtually all gastrointestinal and extra-digestive pathologies associated with defects in epithelium healing and barrier function. The two seminal studies from 1998[17] and 2001[21] both found that enteric glial cell depletion via cytotoxicity (HSV-Tk/ ganciclovir) or immune targeting (GFAP-HA × CL4-TCR mice) led to a breakdown of the intestinal epithelium, vasculature lesions and massive inflammation, reminiscent of histological features typically observed in inflamed intestinal regions of patients with Crohn’s disease. Since then, numerous studies have been undertaken to characterize enteric glial cell responses to inflammation as well as enteric glial cell network alterations and functional deficiencies in pathologies involving inflammation and barrier defects. For instance, in the case of inflammatory bowel diseases, alterations in levels of expression of enteric glial cell markers (reviewed here[46]) have been consistently reported, and several molecular pathways including purinergic[47,48] and polyunsaturated fatty acid [36,39] signaling have been identified as functionally involved in enteric glial cell-mediated control of barrier function and epithelial healing during colitis. In contrast, except for two recent studies that we will review in depth in the next paragraph, enteric glia’s role in colon carcinogenesis remains to be defined. More broadly, little is known about the enteric nervous system in colon cancer.
Perineural invasion, which, simply stated, corresponds to the tumor invasion of neural structures, is a well-recognized prognostic marker in colon cancer[49,50] associated with decreased 5-year disease-free survival rate[51], increased presence of metastasis at the time of resection[52] and increased recurrence[53]. In addition, although contradictory findings have been reported[54,55], several studies suggest that intrinsic and extrinsic denervation inhibits colon carcinogenesis[56–58]. In turn, a global remodeling of the neural network has been reported as tumor cells invade the gut wall, manifesting as changes in neurochemical coding (summarized here[59]) and an increase in neural element density that positively correlates with tumor grading[60]. Altogether, these data indicate that regulatory interactions between neural and cancer (stem) cells must play a prominent role in colon carcinogenesis. Consistent with this, several studies have demonstrated potent regulatory effects of neurotransmitters and associated signaling pathways on colon carcinogenesis (reviewed here[61–64]). Nevertheless, additional studies harnessing novel technologies, in vitro culture systems and animal models are necessary to fully understand how enteric neurons impact colon cancer development and progression. Along those lines, two recent studies propose that enteric neurons constitute a preferred substrate for tumor epithelial cell adhesion and migration via N-Cadherin and L1CAM[10], and that NDRG4-deficient neurons of the tumor microenvironment release pro-carcinogenic extracellular matrix molecules, nidogen 1 and fibulin 2[65]. Importantly, the translation of findings from preclinical studies to the clinical level will also require further investigation in human cohorts.
Similarly, not enough attention has been paid to determine whether and how enteric glia influence colon cancer development and progression. A few studies have reported alterations of S-100β and GFAP expression in colonic adenocarcinomas[20,66–70], although while some studies suggest a decrease in expression of enteric glial cell markers, others conversely show increased expression levels. These discrepancies might be attributable to the analysis of different regions and anatomical structures of the gut wall, such as enteric glial cells in ganglia versus overall enteric glial network, the relatively low number of patients included in a majority of these studies, and the use of different reagents, staining procedures and imaging systems such as different antibodies and 2D versus 3D imaging. In this regard, recent studies have used the iDISCO+ (immunolabeling-enabled three-dimensional imaging of solvent-cleared organs) staining and imaging procedure to visualize the enteric glial network in 3D in whole tumor specimens using light sheet microscopy[9]. 3D images revealed that colonic tumors were densely infiltrated by enteric glial cell bodies and projections, definitively demonstrating that enteric glia are a major component of the tumor microenvironment [9]. Interestingly, alterations of S-100β expression have been reported in adenomas[71], suggesting that modifications of the enteric glial network occur early during colon tumorigenesis. This was confirmed by more recent functional studies by Yuan and colleagues[67]. Using the HSV-Tk/ ganciclovir mouse model, they showed that depletion of GFAP+ enteric glial cells during early phases of AOM/DSS-induced colon tumorigenesis markedly reduced tumor burden, whereas depletion of GFAP+ enteric glial cells once tumors were established had no effect. These findings further suggest that enteric glia are especially influential on the ability of transformed cells to readily form a tumor, a prerogative of cancer stem cells. A previous study from 2019 by Vales et al. had used xenografts in immunodeficient mice and in vitro 3D co-culture systems of enteric glial cells and cancer stem cells, either from human primary cultures or established human cell lines, to demonstrate that enteric glia promote cancer stem cell tumor-forming abilities[9]. Interestingly, they had found that enteric glial cells must be pre-activated by tumor epithelial cell-derived ligands to release effective quantities of pro-tumorigenic molecule(s) that they identify as prostaglandin E2 (PGE2). Enteric glia acquiring a pro-tumorigenic phenotype upon activation by tumor epithelial cells is reminiscent of various cell types undergoing phenotypic alterations in response to molecular cues present within the tumor microenvironment. For example, cancer-associated fibroblasts are thought to originate from local tissueresident fibroblasts activated to become pro-tumorigenic notably in response to tumor epithelial cellderived ligands such as TGF-β1[72,73] – although other origins have also been proposed[74]. Together this demonstrates that, similar to other well-described cellular components, such as mesenchymal[72,75,76] and immune cells[77], tumor epithelial cells engage in a functional bi-directional molecular conversation with enteric glia, promoting cancer stem cell expansion and tumor formation. Vales and colleagues further demonstrated that, in their experimental settings, in contrast to enteric glial cells, human fibroblasts did not release increased PGE2 in response to tumor epithelial ligands and did not impact cancer stem cell tumor-forming abilities[9], strongly indicating that these bi-directional regulatory molecular interactions between tumor epithelial (stem) cells and cells of the tumor microenvironment are cell-specific. In that same vein, they found that enteric glial cell-derived PGE2 specifically targets the pool of cancer stem cells within tumors, as EP4 (Prostaglandin E2 receptor 4) is significantly enriched in the cancer stem cell fraction of tumor epithelial cells, and only the specific blockade of EP4 abolishes enteric glial cell impact on cancer stem cells[9]. While these data strongly support an exclusive enteric glial cell-PGE2/EP4-cancer stem cell axis and are consistent with prior studies demonstrating the prime role of EP4 in colon cancer stem cell tumor-forming abilities[78], it is very likely that other regulatory pathways are involved in enteric glial cell-cancer stem cell crosstalk and more generally in enteric glial cell-tumor epithelial cell crosstalk.
The work by Vales et al., by using simplistic co-culture systems, suggests that enteric glial cells directly act upon cancer stem cell abilities, i.e., without the involvement of an additional cell type. This is further supported by the study from Yuan and colleagues which demonstrates that the impact of enteric glia on tumorigenesis is independent of lymphocytes using Rag2−/− γc−/− mice. However, due to their privileged interactions with enteric neurons[79], it is very likely that enteric glial cells may also impact colon carcinogenesis indirectly via neuronal input, or via regulatory interactions with other cell types of the tumor microenvironment. Interestingly Vales and colleagues’ work suggests that enteric glial cells are activated to produce and secrete pro-tumorigenic amounts of PGE2 in response to IL-1 (interleukin-1) released by tumor epithelial cells [9]. Current evidence suggests that enteric glial cells adopt a “reactive phenotype” in response to pro-inflammatory cytokines such as IL-1[80] or IFNγ (interferon gamma)[81]. Such pathological activation of enteric glia, or “enteric gliosis”, is typically associated with increased GFAP expression, cellular shape alterations, increased cellular proliferation and increased release of pro-inflammatory molecules. Notably reactive enteric glia were recently shown to drive intestinal inflammation and tissue repair by activating local and recruited immune cells [80,81]. As stated earlier in this paragraph, the study by Yuan et al. suggests that enteric glia’s pro-carcinogenic effects do not involve lymphocytes as Rag2−/− γc−/− are devoid of T, B, and NK cells[67]. However it remains to be demonstrated whether or not in response to cytokines of the tumor microenvironment, and in particular IL-1, enteric glial cells adopt a reactive phenotype and engage in functional interactions with other immune cells to regulate colon carcinogenesis. Interestingly, Yuan et al. also shows that GFAP+ cell-specific deletion of MyD88 and RelA/p65, which is a component of NF-κB (nuclear factor kappa-light-chain-enhancer of activated B cells), a key downstream mediator of MyD88, did not impact tumor burden in AOM/DSS-treated mice[67]. Together this suggests that IL-1 present in the tumor microenvironment enhances enteric glial cell release of PGE2 via a MyD88- and RelA/p65- independent mechanism. As prior studies in neurons have proposed an alternate pathway by which IL-1 activates PI3K/Akt in the absence of MyD88[82], it is thus tempting to speculate that such a MyD88-independent pathway is responsible for IL-1-induced synthesis of PGE2 in enteric glial cells.
Unanswered questions and future directions
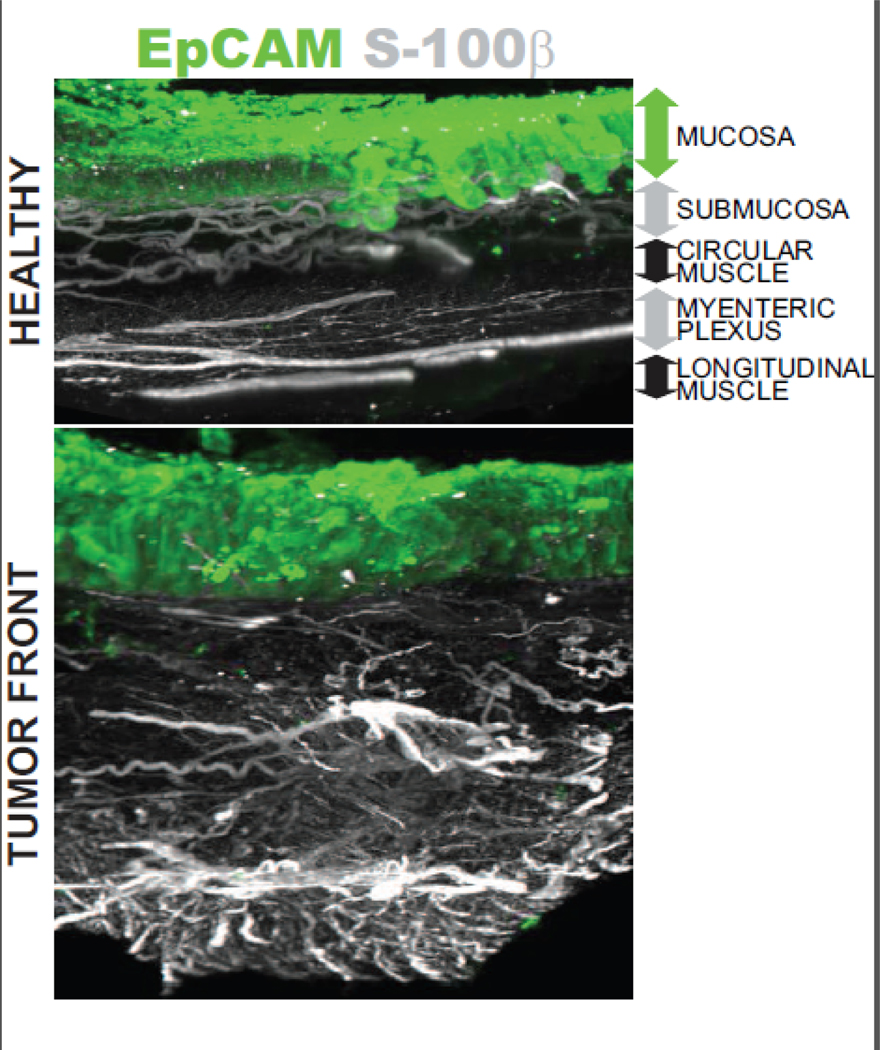
Altogether -the little- available evidence on the influence of enteric glia on colon carcinogenesis indicates that enteric glial cells undergo a phenotypic switch in response to molecular signals present in the tumor microenvironment, leading to changes in their secretome with increased release of pro-tumorigenic factors -in particular PGE2 - as well as in enteric glial cell marker expression and/or cellular density, although the last two are debated. Preliminary work using the iDISCO+ platform demonstrates profound structural changes in the enteric glial network at the tumor front with notable enteric glial cell extension thickening and chaotic trajectories (Figure 1), however the cause of these structural changes and how they correlate with the acquisition of pro-tumorigenic properties remain to be investigated. Interestingly, earlier studies had proposed that enteric glia restrict normal and cancer intestinal epithelial cell proliferation[66], and this has been confirmed in recent studies demonstrating that enteric glial cells exert anti-proliferative effects on crypt progenitors/transit-amplifying cells[34]. However, recent findings also indicate that enteric glial cells favor intestinal stem cells[34] and cancer stem cells[9,67] functions, and more specifically their ability to form (tumor-) organoids in vitro and to give rise to new (tumor) epithelial cells in vivo. This apparent discrepancy is easily explained by the fact that earlier studies were using 2D models, which are not favorable to intestinal stem cells and cancer stem cells, and that enteric glial cells seem to have cell-specific regulatory interactions with the different cells of the crypts and the different cells of the tumor. Therefore, more work is necessary to better understand enteric glial cell regulatory effects on the different epithelial cell types present in crypts and in tumors.
Figure 1. Enteric glial network of the tumor front exhibits profound structural alterations.
Light-sheet microscopy imaging of full thickness human colonic wall at distance (>10 cm) from tumor (healthy) or at the tumor front stained against the enteric glial cell marker S-100β (white) and the epithelial cell marker EpCAM (green) using the iDISCO+ method. Tumor shown is a stage II colon adenocarcinoma.
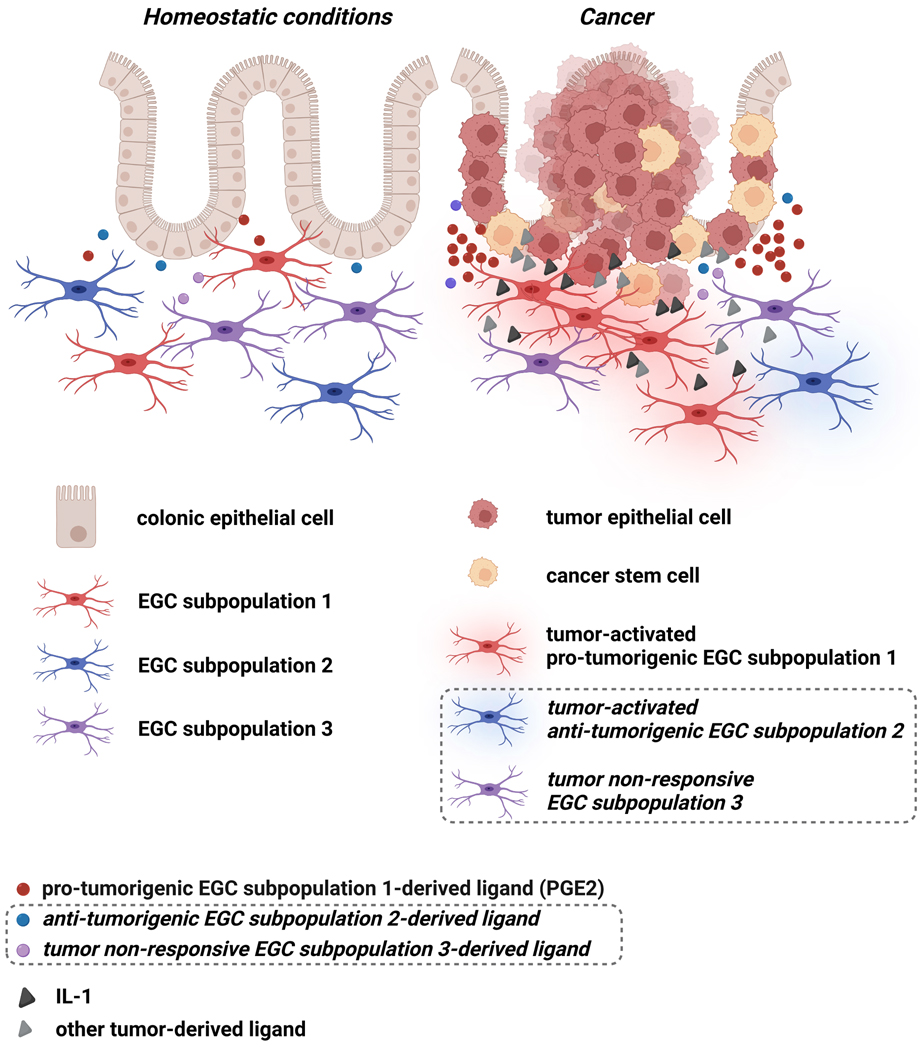
The banalization of the use of transgenic mouse models and the advent of single cell technologies have both contributed to confirm that enteric glia correspond to a heterogeneous pool of cells (Figure 2). However, how this heterogeneity influences the biology of intestinal crypts and colon carcinogenesis remains to be clarified. Interestingly, whereas Baghdadi et al. showed that the loss of GFAP+, but not PLP1+, enteric glial cells was detrimental to intestinal stem cell function, Yuan et al. demonstrated that the loss of GFAP+ and PLP1+ cells decreases tumor formation. This suggests that the different subtype(s) of enteric glial cells that support intestinal stem cell function may not, at least entirely, overlap with the different subtype(s) of enteric glial cells that support cancer stem cell function. While these cancer stem cell-supporting enteric glial cells remained to be characterized, available evidence suggest that they arise from plasticity events triggered by tumor-derived cues, and that they promote cancer stem cell function via the production of PGE2[9] (Figure 2). As it is tempting to speculate that enteric glia heterogeneity persists in the tumor microenvironment (Figure 2), one can wonder if, similar to heterogenous cancer-associated fibroblasts[83], different subsets of enteric glial cells assist different stages of carcinogenesis, i.e., tumor initiation, progression, angiogenesis, immune modulation, dissemination, recurrence and resistance to therapies. In addition, considering the fact that some cancer associated fibroblasts exhibit tumor suppressive functions[84], it is also possible to think that other enteric glial cell subpopulation(s) respond to tumor-derived cues by conversely increasing their production of anti-tumorigenic molecules (Figure 2). However, these enteric glial cell subpopulations seem to be functionally marginal, as co-cultures of primary enteric glial cells and primary cancer stem cells from colon cancer patients consistently lead to increased cancer stem cell tumor-forming abilities[9]. Similarly, it is possible that others may not respond at all to tumor-derived cues (Figure 2). Accordingly, the diversity of enteric glial cells may additionally expand upon external stimuli within the tumor microenvironment, triggering different plasticity mechanisms in discrete enteric glial cell subtypes. While Vales and colleagues have uncovered the influence of IL-1 present in the tumor microenvironment, there is no doubt that many additional signals originating from diverse sources in the tumor microenvironment or from therapies may induce phenotypic changes in specific enteric glial cell subtypes. Furthering the diversity of the glial cell lineage in the colon wall and the tumor microenvironment is the presence of Schwann cells, which have a high affinity for colon cancer cells[85] and were recently shown to be hijacked by cancer cells to promote metastasis[86,87].
Figure 2. Putative heterogeneity and plasticity of enteric glial cells in the TME.
Schematic proposing that, in light of (1) the heterogeneity of enteric glia in homeostatic conditions and (2) the plasticity events triggered by tumor epithelial cell-derived IL-1 in enteric glia, different subtypes of enteric glial cells may subsist in the tumor microenvironment and may be differentially “corrupted” by the tumor, resulting in the existence of pro-tumorigenic (PGE2 producing; predominant) and anti-tumorigenic (marginal) enteric glial cell subpopulations in the tumor microenvironment (represented as red enteric glia and blue enteric glia, respectively). Some other enteric glial cell subpopulation(s) may not undergo any phenotypic changes in response to tumor-derived cues (purple enteric glia). Dotted rectangles indicate items for which there is no direct evidence available in the literature. The figure was created with BioRender.com.
Altogether current evidence strongly indicates that enteric glia are a critical cell type of the crypt niche and the tumor microenvironment. However, much is left to be done to comprehensively understand the respective contribution of discrete enteric glial cell subtypes and their unique regulatory interactions with the different cell types of colonic crypts and tumors. The use of single cell omics data combined with lineage tracing of various enteric glial cell lineages during tumorigenesis, tumor progression and treatment will be invaluable to identify novel biological targets to block the pro-carcinogenic influence of enteric glia.
Highlights.
Enteric glia correspond to a heterogeneous pool of cells that engage in distinct regulatory interactions with the various epithelial cell types of the intestinal epithelium.
Enteric glial cells are a major component of the tumor microenvironment.
Enteric glial cells undergo a phenotypic switch and become pro-carcinogenic in response to molecular signals present in the tumor microenvironment.
Enteric glial cell heterogeneity and plasticity in the tumor microenvironment must be comprehensively characterized to identify novel biological targets to block enteric glial cell procarcinogenic signaling.
Funding:
This review was supported by the National Cancer Institute of the National Institutes of Health under Award Number R01CA270462, the Lineberger Comprehensive Cancer Center at the University of North Carolina at Chapel Hill and the Center for Gastrointestinal Biology & Disease at the University of North Carolina at Chapel Hill (NIH P30 DK034987) and the College of Veterinary Medicine at North Carolina State University.
Abbreviations:
- EP4
Prostaglandin E2 receptor 4
- GFAP
glial fibrillary acidic protein
- HSV-Tk
herpes simplex virus thymidine kinase
- IL-1
interleukin-1
- PGE2
prostaglandin E2
- PLP1
Proteolipid protein 1
- S-100β
S100 calcium-binding protein B, Sox10: SRY-Box Transcription Factor 10
Footnotes
Declaration of Interest: None.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- [1].Siegel RL, Miller KD, Wagle NS, Jemal A, Cancer statistics, 2023, CA. Cancer J. Clin 73 (2023) 17–48. 10.3322/caac.21763. [DOI] [PubMed] [Google Scholar]
- [2].Siegel RL, Miller KD, Goding Sauer A, Fedewa SA, Butterly LF, Anderson JC, Cercek A, Smith RA, Jemal A, Colorectal cancer statistics, 2020, CA. Cancer J. Clin 70 (2020) 145–164. 10.3322/caac.21601. [DOI] [PubMed] [Google Scholar]
- [3].Lapidot T, Sirard C, Vormoor J, Murdoch B, Hoang T, Caceres-Cortes J, Minden M, Paterson B, Caligiuri MA, Dick JE, A cell initiating human acute myeloid leukaemia after transplantation into SCID mice, Nature. 367 (1994) 645–648. 10.1038/367645a0. [DOI] [PubMed] [Google Scholar]
- [4].Vessoni AT, Filippi-Chiela EC, Lenz G, Batista LFZ, Tumor propagating cells: drivers of tumor plasticity, heterogeneity, and recurrence, Oncogene. 39 (2020) 2055–2068. 10.1038/s41388-019-1128-4. [DOI] [PubMed] [Google Scholar]
- [5].Ricci-Vitiani L, Lombardi DG, Pilozzi E, Biffoni M, Todaro M, Peschle C, De Maria R, Identification and expansion of human colon-cancer-initiating cells, Nature. 445 (2007) 111–115. 10.1038/nature05384. [DOI] [PubMed] [Google Scholar]
- [6].O’Brien CA, Pollett A, Gallinger S, Dick JE, A human colon cancer cell capable of initiating tumour growth in immunodeficient mice, Nature. 445 (2007) 106–110. 10.1038/nature05372. [DOI] [PubMed] [Google Scholar]
- [7].Pang R, Law WL, Chu ACY, Poon JT, Lam CSC, Chow AKM, Ng L, Cheung LWH, Lan XR, Lan HY, Tan VPY, Yau TC, Poon RT, Wong BCY, A Subpopulation of CD26+ Cancer Stem Cells with Metastatic Capacity in Human Colorectal Cancer, Cell Stem Cell. 6 (2010) 603–615. 10.1016/j.stem.2010.04.001. [DOI] [PubMed] [Google Scholar]
- [8].Todaro M, Alea MP, Di Stefano AB, Cammareri P, Vermeulen L, Iovino F, Tripodo C, Russo A, Gulotta G, Medema JP, Stassi G, Colon Cancer Stem Cells Dictate Tumor Growth and Resist Cell Death by Production of Interleukin-4, Cell Stem Cell. 1 (2007) 389–402. 10.1016/j.stem.2007.08.001. [DOI] [PubMed] [Google Scholar]
- [9].Valès S, Bacola G, Biraud M, Touvron M, Bessard A, Geraldo F, Dougherty KA, Lashani S, Bossard C, Flamant M, Duchalais E, Marionneau-Lambot S, Oullier T, Oliver L, Neunlist M, Vallette FM, Van Landeghem L, Tumor cells hijack enteric glia to activate colon cancer stem cells and stimulate tumorigenesis, EBioMedicine. 49 (2019) 172–188. 10.1016/j.ebiom.2019.09.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Duchalais E, Guilluy C, Nedellec S, Touvron M, Bessard A, Touchefeu Y, Bossard C, Boudin H, Louarn G, Neunlist M, Van Landeghem L, Colorectal Cancer Cells Adhere to and Migrate Along the Neurons of the Enteric Nervous System, Cell. Mol. Gastroenterol. Hepatol 5 (2018) 31–49. 10.1016/j.jcmgh.2017.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].Albo D, Akay CL, Marshall CL, Wilks JA, Verstovsek G, Liu H, Agarwal N, Berger DH, Ayala GE, Neurogenesis in colorectal cancer is a marker of aggressive tumor behavior and poor outcomes, Cancer. 117 (2011) 4834–4845. 10.1002/cncr.26117. [DOI] [PubMed] [Google Scholar]
- [12].Gabella G, Trigg P, Size of neurons and glial cells in the enteric ganglia of mice, guinea-pigs, rabbits and sheep, J. Neurocytol 13 (1984) 49–71. 10.1007/BF01148318. [DOI] [PubMed] [Google Scholar]
- [13].Hoff S, Zeller F, von Weyhern CWH, Wegner M, Schemann M, Michel K, Rühl A, Quantitative assessment of glial cells in the human and guinea pig enteric nervous system with an anti-Sox8/9/10 antibody, J. Comp. Neurol 509 (2008) 356–371. 10.1002/cne.21769. [DOI] [PubMed] [Google Scholar]
- [14].Bernstein CN, Vidrich A, Isolation, identification, and culture of normal mouse colonic glia, Glia. 12 (1994) 108–116. 10.1002/glia.440120204. [DOI] [PubMed] [Google Scholar]
- [15].Mestres P, Diener M, Rummel W, Electron microscopy of the mucosal plexus of the rat colon, Acta Anat. (Basel) 143 (1992) 275–282. 10.1159/000147262. [DOI] [PubMed] [Google Scholar]
- [16].Tam PK, An immunohistological study of the human enteric nervous system with microtubule-associated proteins, Gastroenterology. 99 (1990) 1841–1844. 10.1016/00165085(90)90499-q. [DOI] [PubMed] [Google Scholar]
- [17].Bush TG, Savidge TC, Freeman TC, Cox HJ, Campbell EA, Mucke L, Johnson MH, Sofroniew MV, Fulminant Jejuno-Ileitis following Ablation of Enteric Glia in Adult Transgenic Mice, Cell. 93 (1998) 189–201. 10.1016/S0092-8674(00)81571-8. [DOI] [PubMed] [Google Scholar]
- [18].Neunlist M, Aubert P, Bonnaud S, Van Landeghem L, Coron E, Wedel T, Naveilhan P, Ruhl A, Lardeux B, Savidge T, Paris F, Galmiche JP, Enteric glia inhibit intestinal epithelial cell proliferation partly through a TGF-β1-dependent pathway, Am. J. Physiol.-Gastrointest. Liver Physiol 292 (2007) G231–G241. 10.1152/ajpgi.00276.2005. [DOI] [PubMed] [Google Scholar]
- [19].Savidge TC, Newman P, Pothoulakis C, Ruhl A, Neunlist M, Bourreille A, Hurst R, Sofroniew MV, Enteric Glia Regulate Intestinal Barrier Function and Inflammation Via Release of S-Nitrosoglutathione, Gastroenterology. 132 (2007) 1344–1358. 10.1053/j.gastro.2007.01.051. [DOI] [PubMed] [Google Scholar]
- [20].Neunlist M, Van Landeghem L, Bourreille A, Savidge T, Neuro-glial crosstalk in inflammatory bowel disease, J. Intern. Med 263 (2008) 577–583. 10.1111/j.1365-2796.2008.01963.x. [DOI] [PubMed] [Google Scholar]
- [21].Cornet A, Savidge TC, Cabarrocas J, Deng WL, Colombel JF, Lassmann H, Desreumaux P, Liblau RS, Enterocolitis induced by autoimmune targeting of enteric glial cells: a possible mechanism in Crohn’s disease?, Proc. Natl. Acad. Sci. U. S. A 98 (2001) 13306–13311. 10.1073/pnas.231474098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Aube A-C, Changes in enteric neurone phenotype and intestinal functions in a transgenic mouse model of enteric glia disruption, Gut. 55 (2006) 630–637. 10.1136/gut.2005.067595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].Rao M, Rastelli D, Dong L, Chiu S, Setlik W, Gershon MD, Corfas G, Enteric Glia Regulate Gastrointestinal Motility but are not Required for Maintenance of the Epithelium in Mice, Gastroenterology. 153 (2017) 1068–1081.e7. 10.1053/j.gastro.2017.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Grubišić V, Gulbransen BD, Enteric glial activity regulates secretomotor function in the mouse colon but does not acutely affect gut permeability, J. Physiol 595 (2017) 3409–3424. 10.1113/JP273492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Hanani M, Reichenbach A, Morphology of horseradish peroxidase (HRP)-injected glial cells in the myenteric plexus of the guinea-pig, Cell Tissue Res. 278 (1994) 153–160. 10.1007/BF00305787. [DOI] [PubMed] [Google Scholar]
- [26].Boesmans W, Lasrado R, Vanden Berghe P, Pachnis V, Heterogeneity and phenotypic plasticity of glial cells in the mammalian enteric nervous system, Glia. 63 (2015) 229–241. 10.1002/glia.22746. [DOI] [PubMed] [Google Scholar]
- [27].Touvron M, Wieland BA, Mariant CL, Hattenhauer AR, Landeghem LV, Enteric glial cells of the two plexi of the enteric nervous system exhibit phenotypic and functional inter-and intra-heterogeneity, (2022) 2022.06.28.497986. 10.1101/2022.06.28.497986. [DOI] [Google Scholar]
- [28].Seguella L, McClain JL, Esposito G, Gulbransen BD, Functional Intraregional and Interregional Heterogeneity between Myenteric Glial Cells of the Colon and Duodenum in Mice, J. Neurosci. Off. J. Soc. Neurosci 42 (2022) 8694–8708. 10.1523/JNEUROSCI.2379-20.2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Drokhlyansky E, Smillie CS, Van Wittenberghe N, Ericsson M, Griffin GK, Eraslan G, Dionne D, Cuoco MS, Goder-Reiser MN, Sharova T, Kuksenko O, Aguirre AJ, Boland GM, Graham D, Rozenblatt-Rosen O, Xavier RJ, Regev A, The human and mouse enteric nervous system at single cell resolution, Cell. 182 (2020) 1606–1622.e23. 10.1016/j.cell.2020.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].May-Zhang AA, Tycksen E, Southard-Smith AN, Deal KK, Benthal JT, Buehler DP, Adam M, Simmons AJ, Monaghan JR, Matlock BK, Flaherty DK, Potter SS, Lau KS, Southard-Smith EM, Combinatorial Transcriptional Profiling of Mouse and Human Enteric Neurons Identifies Shared and Disparate Subtypes In Situ, Gastroenterology. 160 (2021) 755–770.e26. 10.1053/j.gastro.2020.09.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Morarach K, Mikhailova A, Knoflach V, Memic F, Kumar R, Li W, Ernfors P, Marklund U, Diversification of molecularly defined myenteric neuron classes revealed by single-cell RNA sequencing, Nat. Neurosci 24 (2021) 34–46. 10.1038/s41593-020-00736-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [32].Wright CM, Schneider S, Smith-Edwards KM, Mafra F, Leembruggen AJL, Gonzalez MV, Kothakapa DR, Anderson JB, Maguire BA, Gao T, Missall TA, Howard MJ, Bornstein JC, Davis BM, Heuckeroth RO, scRNA-Seq Reveals New Enteric Nervous System Roles for GDNF, NRTN, and TBX3, Cell. Mol. Gastroenterol. Hepatol 11 (2021) 1548–1592.e1. 10.1016/j.jcmgh.2020.12.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].Zeisel A, Hochgerner H, Lönnerberg P, Johnsson A, Memic F, van der Zwan J, Häring M, Braun E, Borm LE, La Manno G, Codeluppi S, Furlan A, Lee K, Skene N, Harris KD, Hjerling-Leffler J, Arenas E, Ernfors P, Marklund U, Linnarsson S, Molecular Architecture of the Mouse Nervous System, Cell. 174 (2018) 999–1014.e22. 10.1016/j.cell.2018.06.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Baghdadi MB, Ayyaz A, Coquenlorge S, Chu B, Kumar S, Streutker C, Wrana JL, Kim T-H, Enteric glial cell heterogeneity regulates intestinal stem cell niches, Cell Stem Cell. 29 (2022) 86–100.e6. 10.1016/j.stem.2021.10.004. [DOI] [PubMed] [Google Scholar]
- [35].Bohórquez DV, Samsa LA, Roholt A, Medicetty S, Chandra R, Liddle RA, An enteroendocrine cell-enteric glia connection revealed by 3D electron microscopy, PloS One. 9 (2014) e89881. 10.1371/journal.pone.0089881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Pochard C, Coquenlorge S, Jaulin J, Cenac N, Vergnolle N, Meurette G, Freyssinet M, Neunlist M, Rolli-Derkinderen M, Defects in 15-HETE Production and Control of Epithelial Permeability by Human Enteric Glial Cells From Patients With Crohn’s Disease, Gastroenterology. 150 (2016) 168–180. 10.1053/j.gastro.2015.09.038. [DOI] [PubMed] [Google Scholar]
- [37].Bach-Ngohou K, Mahé MM, Aubert P, Abdo H, Boni S, Bourreille A, Denis MG, Lardeux B, Neunlist M, Masson D, Enteric glia modulate epithelial cell proliferation and differentiation through 15-deoxy-Δ12,14-prostaglandin J2, J. Physiol 588 (2010) 2533–2544. 10.1113/jphysiol.2010.188409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Van Landeghem L, Chevalier J, Mahé MM, Wedel T, Urvil P, Derkinderen P, Savidge T, Neunlist M, Enteric glia promote intestinal mucosal healing via activation of focal adhesion kinase and release of proEGF, Am. J. Physiol. Gastrointest. Liver Physiol 300 (2011) G976–987. 10.1152/ajpgi.00427.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [39].Coquenlorge S, Van Landeghem L, Jaulin J, Cenac N, Vergnolle N, Duchalais E, Neunlist M, Rolli-Derkinderen M, The arachidonic acid metabolite 11β-ProstaglandinF2α controls intestinal epithelial healing: deficiency in patients with Crohn’s disease, Sci. Rep 6 (2016) 25203. 10.1038/srep25203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [40].Meir M, Flemming S, Burkard N, Bergauer L, Metzger M, Germer C-T, Schlegel N, Glial cell line-derived neurotrophic factor promotes barrier maturation and wound healing in intestinal epithelial cells in vitro, Am. J. Physiol. Gastrointest. Liver Physiol 309 (2015) G613–624. 10.1152/ajpgi.00357.2014. [DOI] [PubMed] [Google Scholar]
- [41].Pellegrini C, Fornai M, D’Antongiovanni V, Antonioli L, Bernardini N, Derkinderen P, The intestinal barrier in disorders of the central nervous system, Lancet Gastroenterol. Hepatol 8 (2023) 66–80. 10.1016/S2468-1253(22)00241-2. [DOI] [PubMed] [Google Scholar]
- [42].Seguella L, Gulbransen BD, Enteric glial biology, intercellular signalling and roles in gastrointestinal disease, Nat. Rev. Gastroenterol. Hepatol 18 (2021) 571–587. 10.1038/s41575-021-00423-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [43].Schneider KM, Kim J, Bahnsen K, Heuckeroth RO, Thaiss CA, Environmental perception and control of gastrointestinal immunity by the enteric nervous system, Trends Mol. Med 28 (2022) 989–1005. 10.1016/j.molmed.2022.09.005. [DOI] [PubMed] [Google Scholar]
- [44].Liu C, Yang J, Enteric Glial Cells in Immunological Disorders of the Gut, Front. Cell. Neurosci 16 (2022) 895871. 10.3389/fncel.2022.895871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [45].Progatzky F, Pachnis V, The role of enteric glia in intestinal immunity, Curr. Opin. Immunol 77 (2022) 102183. 10.1016/j.coi.2022.102183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [46].Pochard C, Coquenlorge S, Freyssinet M, Naveilhan P, Bourreille A, Neunlist M, Rolli-Derkinderen M, The multiple faces of inflammatory enteric glial cells: is Crohn’s disease a gliopathy?, Am. J. Physiol. Gastrointest. Liver Physiol 315 (2018) G1–G11. 10.1152/ajpgi.00016.2018. [DOI] [PubMed] [Google Scholar]
- [47].Grubišić V, Bali V, Fried DE, Eltzschig HK, Robson SC, Mazei-Robison MS, Gulbransen BD, Enteric glial adenosine 2B receptor signaling mediates persistent epithelial barrier dysfunction following acute DSS colitis, Mucosal Immunol. 15 (2022) 964–976. 10.1038/s41385-022-00550-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [48].Grubišić V, Perez-Medina AL, Fried DE, Sévigny J, Robson SC, Galligan JJ, Gulbransen BD, NTPDase1 and −2 are expressed by distinct cellular compartments in the mouse colon and differentially impact colonic physiology and function after DSS colitis, Am. J. Physiol. Gastrointest. Liver Physiol 317 (2019) G314–G332. 10.1152/ajpgi.00104.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [49].Liebig C, Ayala G, Wilks JA, Berger DH, Albo D, Perineural invasion in cancer, Cancer. 115 (2009) 3379–3391. 10.1002/cncr.24396. [DOI] [PubMed] [Google Scholar]
- [50].Knijn N, Mogk SC, Teerenstra S, Simmer F, Nagtegaal ID, Perineural Invasion is a Strong Prognostic Factor in Colorectal Cancer: A Systematic Review, Am. J. Surg. Pathol 40 (2016) 103–112. 10.1097/PAS.0000000000000518. [DOI] [PubMed] [Google Scholar]
- [51].Liebig C, Ayala G, Wilks J, Verstovsek G, Liu H, Agarwal N, Berger DH, Albo D, Perineural Invasion Is an Independent Predictor of Outcome in Colorectal Cancer, J. Clin. Oncol 27 (2009) 5131–5137. 10.1200/JCO.2009.22.4949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [52].Krasna MJ, Flancbaum L, Cody RP, Shneibaum S, Ari GB, Vascular and neural invasion in colorectal carcinoma. Incidence and prognostic significance, Cancer. 61 (1988) 1018–1023. . [DOI] [PubMed] [Google Scholar]
- [53].Horn A, Dahl O, Morild I, Venous and neural invasion as predictors of recurrence in rectal adenocarcinoma, Dis. Colon Rectum 34 (1991) 798–804. 10.1007/BF02051074. [DOI] [PubMed] [Google Scholar]
- [54].Nelson RL, Briley S, Vaz OP, Abcarian H, The effect of vagotomy and pyloroplasty on colorectal tumor induction in the rat, J. Surg. Oncol 51 (1992) 281–286. 10.1002/jso.2930510416. [DOI] [PubMed] [Google Scholar]
- [55].Watt PC, Patterson CC, Kennedy TL, Late mortality after vagotomy and drainage for duodenal ulcer, Br. Med. J. Clin. Res Ed. 288 (1984) 1335–1338. 10.1136/bmj.288.6427.1335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [56].Vespúcio MVO, Turatti A, Modiano P, de Oliveira EC, Chicote SRM, Pinto AMP, Garcia SB, Intrinsic denervation of the colon is associated with a decrease of some colonic preneoplastic markers in rats treated with a chemical carcinogen, Braz. J. Med. Biol. Res. Rev. Bras. Pesqui. Medicas E Biol 41 (2008) 311–317. . [DOI] [PubMed] [Google Scholar]
- [57].Tatsuta M, Iishi H, Baba M, Taniguchi H, Inhibition of azoxymethane-induced experimental colon carcinogenesis in Wistar rats by 6-hydroxydopamine, Int. J. Cancer 50 (1992) 298–301. 10.1002/ijc.2910500221. [DOI] [PubMed] [Google Scholar]
- [58].Liu V, Dietrich A, Kasparek MS, Benhaqi P, Schneider MR, Schemann M, Seeliger H, Kreis ME, Extrinsic intestinal denervation modulates tumor development in the small intestine of Apc(Min/+) mice, J. Exp. Clin. Cancer Res. CR 34 (2015) 39. 10.1186/s13046-015-0159-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [59].Godlewski J, Kmiec Z, Colorectal Cancer Invasion and Atrophy of the Enteric Nervous System: Potential Feedback and Impact on Cancer Progression, Int. J. Mol. Sci 21 (2020) 3391. 10.3390/ijms21093391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [60].Ciurea RN, Rogoveanu I, Pirici D, Târtea G-C, Streba CT, Florescu C, Cătălin B, Puiu I, Târtea E-A, Vere CC, B2 adrenergic receptors and morphological changes of the enteric nervous system in colorectal adenocarcinoma, World J. Gastroenterol 23 (2017) 1250–1261. 10.3748/wjg.v23.i7.1250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [61].Schledwitz A, Xie G, Raufman J-P, Exploiting unique features of the gut-brain interface to combat gastrointestinal cancer, J. Clin. Invest 131 (2021) e143776, 143776. 10.1172/JCI143776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [62].Schonkeren SL, Thijssen MS, Vaes N, Boesmans W, Melotte V, The Emerging Role of Nerves and Glia in Colorectal Cancer, Cancers. 13 (2021) 152. 10.3390/cancers13010152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [63].Rademakers G, Vaes N, Schonkeren S, Koch A, Sharkey KA, Melotte V, The role of enteric neurons in the development and progression of colorectal cancer, Biochim. Biophys. Acta BBA - Rev. Cancer 1868 (2017) 420–434. 10.1016/j.bbcan.2017.08.003. [DOI] [PubMed] [Google Scholar]
- [64].Vaes N, Idris M, Boesmans W, Alves MM, Melotte V, Nerves in gastrointestinal cancer: from mechanism to modulations, Nat. Rev. Gastroenterol. Hepatol 19 (2022) 768–784. 10.1038/s41575-022-00669-9. [DOI] [PubMed] [Google Scholar]
- [65].Vaes N, Schonkeren SL, Rademakers G, Holland AM, Koch A, Gijbels MJ, Keulers TG, de Wit M, Moonen L, Van der Meer JRM, van den Boezem E, Wolfs TGAM, Threadgill DW, Demmers J, Fijneman RJA, Jimenez CR, Vanden Berghe P, Smits KM, Rouschop KMA, Boesmans W, Hofstra RMW, Melotte V, Loss of enteric neuronal Ndrg4 promotes colorectal cancer via increased release of Nid1 and Fbln2, EMBO Rep. 22 (2021) e51913. 10.15252/embr.202051913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [66].Neunlist M, Aubert P, Bonnaud S, Van Landeghem L, Coron E, Wedel T, Naveilhan P, Ruhl A, Lardeux B, Savidge T, Paris F, Galmiche JP, Enteric glia inhibit intestinal epithelial cell proliferation partly through a TGF-β1-dependent pathway, Am. J. Physiol.-Gastrointest. Liver Physiol 292 (2007) G231–G241. 10.1152/ajpgi.00276.2005. [DOI] [PubMed] [Google Scholar]
- [67].Yuan R, Bhattacharya N, Kenkel JA, Shen J, DiMaio MA, Bagchi S, Prestwood TR, Habtezion A, Engleman EG, Enteric Glia Play a Critical Role in Promoting the Development of Colorectal Cancer, Front. Oncol 10 (2020). 10.3389/fonc.2020.595892 (accessed December 1, 2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [68].Jaiswal M, Ganapathy A, Singh S, Sarwar S, Quadri JA, Rani N, Das P, Pal S, Shariff A, Morphology of enteric glia in colorectal carcinoma: A comparative study of tumor site and its proximal normal margin, Morphologie. 105 (2021) 267–274. 10.1016/j.morpho.2020.11.003. [DOI] [PubMed] [Google Scholar]
- [69].Târtea EA, Florescu C, Donoiu I, Pirici D, Mihailovici AR, Albu VC, Bălşeanu TA, Iancău M, Badea CD, Vere CC, Sfredel V, Implications of inflammation and remodeling of the enteric glial cells in colorectal adenocarcinoma, Romanian J. Morphol. Embryol. Rev. Roum. Morphol. Embryol 58 (2017) 473–480. [PubMed] [Google Scholar]
- [70].Seguella L, Rinaldi F, Marianecci C, Capuano R, Pesce M, Annunziata G, Casano F, Bassotti G, Sidoni A, Milone M, Aprea G, de Palma GD, Carafa M, Pesce M, Esposito G, Sarnelli G, Pentamidine niosomes thwart S100B effects in human colon carcinoma biopsies favouring wtp53 rescue, J. Cell. Mol. Med 24 (2020) 3053–3063. 10.1111/jcmm.14943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [71].Liu YA, Chung YC, Pan ST, Shen MY, Hou YC, Peng SJ, Pasricha PJ, Tang SC, 3-D imaging, illustration, and quantitation of enteric glial network in transparent human colon mucosa, Neurogastroenterol. Motil 25 (2013) e324–e338. 10.1111/nmo.12115. [DOI] [PubMed] [Google Scholar]
- [72].Sahai E, Astsaturov I, Cukierman E, DeNardo DG, Egeblad M, Evans RM, Fearon D, Greten FR, Hingorani SR, Hunter T, Hynes RO, Jain RK, Janowitz T, Jorgensen C, Kimmelman AC, Kolonin MG, Maki RG, Powers RS, Puré E, Ramirez DC, Scherz-Shouval R, Sherman MH, Stewart S, Tlsty TD, Tuveson DA, Watt FM, Weaver V, Weeraratna AT, Werb Z, A framework for advancing our understanding of cancer-associated fibroblasts, Nat. Rev. Cancer 20 (2020) 174–186. 10.1038/s41568-019-0238-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [73].Hawinkels L.J. a. C., Paauwe M, Verspaget HW, Wiercinska E, van der Zon JM, van der Ploeg K, Koelink PJ, Lindeman JHN, Mesker W, ten Dijke P, Sier CFM, Interaction with colon cancer cells hyperactivates TGF-β signaling in cancer-associated fibroblasts, Oncogene. 33 (2014) 97–107. 10.1038/onc.2012.536. [DOI] [PubMed] [Google Scholar]
- [74].Bartoschek M, Oskolkov N, Bocci M, Lövrot J, Larsson C, Sommarin M, Madsen CD, Lindgren D, Pekar G, Karlsson G, Ringnér M, Bergh J, Björklund Å, Pietras K, Spatially and functionally distinct subclasses of breast cancer-associated fibroblasts revealed by single cell RNA sequencing, Nat. Commun 9 (2018) 5150. 10.1038/s41467-018-07582-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [75].Frisbie L, Buckanovich RJ, Coffman L, Carcinoma-Associated Mesenchymal Stem/Stromal Cells: Architects of the Pro-tumorigenic Tumor Microenvironment, Stem Cells Dayt. Ohio 40 (2022) 705–715. 10.1093/stmcls/sxac036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [76].Kobayashi H, Gieniec KA, Lannagan TRM, Wang T, Asai N, Mizutani Y, Iida T, Ando R, Thomas EM, Sakai A, Suzuki N, Ichinose M, Wright JA, Vrbanac L, Ng JQ, Goyne J, Radford G, Lawrence MJ, Sammour T, Hayakawa Y, Klebe S, Shin AE, Asfaha S, Bettington ML, Rieder F, Arpaia N, Danino T, Butler LM, Burt AD, Leedham SJ, Rustgi AK, Mukherjee S, Takahashi M, Wang TC, Enomoto A, Woods SL, Worthley DL, The Origin and Contribution of Cancer-Associated Fibroblasts in Colorectal Carcinogenesis, Gastroenterology. 162 (2022) 890–906. 10.1053/j.gastro.2021.11.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [77].Hinshaw DC, Shevde LA, The Tumor Microenvironment Innately Modulates Cancer Progression, Cancer Res. 79 (2019) 4557–4566. 10.1158/0008-5472.CAN-18-3962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [78].Wang D, Fu L, Sun H, Guo L, DuBois RN, Prostaglandin E2 Promotes Colorectal Cancer Stem Cell Expansion and Metastasis in Mice, Gastroenterology. 149 (2015) 1884–1895.e4. 10.1053/j.gastro.2015.07.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [79].Sharkey KA, Mawe GM, The enteric nervous system, Physiol. Rev (2022). 10.1152/physrev.00018.2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [80].Schneider R, Leven P, Mallesh S, Breßer M, Schneider L, Mazzotta E, Fadda P, Glowka T, Vilz TO, Lingohr P, Kalff JC, Christofi FL, Wehner S, IL-1-dependent enteric gliosis guides intestinal inflammation and dysmotility and modulates macrophage function, Commun. Biol 5 (2022) 1–16. 10.1038/s42003-022-03772-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [81].Progatzky F, Shapiro M, Chng SH, Garcia-Cassani B, Classon CH, Sevgi S, Laddach A, Bon-Frauches AC, Lasrado R, Rahim M, Amaniti E-M, Boeing S, Shah K, Entwistle LJ, Suárez-Bonnet A, Wilson MS, Stockinger B, Pachnis V, Regulation of intestinal immunity and tissue repair by enteric glia, Nature. 599 (2021) 125–130. 10.1038/s41586-021-04006-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [82].Davis CN, Mann E, Behrens MM, Gaidarova S, Rebek M, Rebek J, Bartfai T, MyD88-dependent and -independent signaling by IL-1 in neurons probed by bifunctional Toll/IL-1 receptor domain/BB-loop mimetics, Proc. Natl. Acad. Sci 103 (2006) 2953–2958. 10.1073/pnas.0510802103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [83].Chhabra Y, Weeraratna AT, Fibroblasts in cancer: Unity in heterogeneity, Cell. 186 (2023) 1580–1609. 10.1016/j.cell.2023.03.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [84].Pallangyo CK, Ziegler PK, Greten FR, IKKβ acts as a tumor suppressor in cancer-associated fibroblasts during intestinal tumorigenesis, J. Exp. Med 212 (2015) 2253–2266. 10.1084/jem.20150576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [85].Demir IE, Boldis A, Pfitzinger PL, Teller S, Brunner E, Klose N, Kehl T, Maak M, Lesina M, Laschinger M, Janssen K-P, Algül H, Friess H, Ceyhan GO, Investigation of Schwann cells at neoplastic cell sites before the onset of cancer invasion, J. Natl. Cancer Inst 106 (2014) dju184. 10.1093/jnci/dju184. [DOI] [PubMed] [Google Scholar]
- [86].Han S, Wang D, Huang Y, Zeng Z, Xu P, Xiong H, Ke Z, Zhang Y, Hu Y, Wang F, Wang J, Zhao Y, Zhuo W, Zhao G, A reciprocal feedback between colon cancer cells and Schwann cells promotes the proliferation and metastasis of colon cancer, J. Exp. Clin. Cancer Res. CR 41 (2022) 348. 10.1186/s13046-022-02556-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [87].Chen S, Chen M, Schwann cells promote the migration and invasion of colorectal cancer cells via the activated NF-κB/IL-8 axis in the tumor microenvironment, Front. Oncol 12 (2022) 1026670. 10.3389/fonc.2022.1026670. [DOI] [PMC free article] [PubMed] [Google Scholar]