Abstract
A reverse genetics system for birnavirus, based on synthetic transcripts of the infectious bursal disease virus (IBDV) genome, was recently developed (E. Mundt and V. N. Vakharia, Proc. Natl. Acad. Sci. USA 93:11131–11136, 1996). To study the function of the 17-kDa nonstructural (NS) protein in viral growth and pathogenesis, we constructed a cDNA clone of IBDV segment A in which the first and only initiation codon (ATG) of NS protein was mutated to a stop codon (TAG). Transfection of Vero cells with combined transcripts of either modified or unmodified segment A, and with segment B, generated viable IBDV progeny. When chicken embryo fibroblast cells infected with transfectant viruses were analyzed by immunofluorescence assays using NS-specific antiserum, the mutant virus did not yield a fluorescence signal, indicating a lack of NS protein expression. Furthermore, replication kinetics and cytotoxic effects of the mutant virus were compared with those of the parental attenuated vaccine strain of IBDV (D78) in vitro. The mutant virus grew to slightly lower titers than D78 virus and exhibited decreased cytotoxic and apoptotic effects in cell culture. To evaluate the characteristics of the recovered viruses in vivo, we inoculated 3-week-old chickens with D78 or mutant virus and analyzed their bursa for histopathological lesions. The recovered D78 virus caused microscopic lesions and atrophy of the bursa, while the mutant virus failed to induce any pathological lesions or clinical signs of disease. In both instances, the virus was recovered from the bursa, and the presence or absence of mutation in these viruses was confirmed by nucleotide sequence analysis of NS gene. Although the mutant virus exhibited a delay in replication in vivo, it induced levels of IBDV neutralizing antibodies that were similar to those of D78 virus. In addition, no reversion of mutation was detected in the mutant virus recovered from inoculated chickens. These results demonstrate that NS protein is dispensable for viral replication in vitro and in vivo and that it plays an important role in viral pathogenesis. Thus, generation of such NS protein-deficient virus will facilitate the study of immunosuppression and aid in the development of live-attenuated vaccines for IBDV.
Infectious bursal disease virus (IBDV), a pathogen of economic importance to the poultry industry, causes severe immunodeficiency in young chickens by destroying the precursors of antibody-producing B cells in the bursa of Fabricius (BF). In a fully susceptible flock (between 3 and 6 weeks of age), the clinical disease causes immunosuppression and is responsible for losses due to impaired growth and death. Chickens less than 3 weeks of age do not exhibit outward clinical signs but have a marked infection characterized by microscopic lesions in the bursa. Damage to the bursa ultimately causes immunodeficiency, which then leads to an increased susceptibility to other etiologic agents (9).
IBDV belongs to the genus Avibirnavirus of the Birnaviridae family, and its genome consists of two segments of double-stranded RNA (4). The smaller segment B encodes VP1, a 97-kDa multifunctional protein with polymerase and capping enzyme activities (21, 22). The larger segment A encodes a 110-kDa precursor protein in a single large open reading frame (polyprotein ORF) which is processed into mature VP2, VP3, and VP4 proteins (6). VP2 and VP3 are the major structural proteins of the virion, whereas VP4 is a minor protein involved in the processing of the precursor protein (8). Segment A also encodes VP5, a 17-kDa nonstructural (NS) protein, from a small ORF partly preceding and overlapping the polyprotein ORF. However, this protein is not present in the virion (2), and it is detected only in IBDV-infected cells (12). Therefore, VP5 should be designated an NS protein.
Complete nucleotide sequences of the large segment A of various IBDV strains have been determined (23). In all cases, the small ORF, which codes for the 17-kDa NS protein, is invariably present. In infectious pancreatic necrosis virus (IPNV), a prototype virus of the Birnaviridae family, segment A also encodes a 17-kDa NS protein (from a small ORF), which is found in IPNV-infected cells (10). Although the small ORF coding for NS protein is detected in all birnaviruses studied so far, the function of this protein is still unknown. In IBDV, it was recently shown that NS protein is not required for viral replication in vitro (13).
The NS protein of IBDV is highly basic, cysteine-rich, and conserved among all serotype I IBDV strains. In chicken anemia virus, another virus causing immunosuppression, an analogous basic, cysteine-rich 14-kDa protein was shown to cause apoptosis and was implicated in pathogenesis (16). Since IBDV is also known to induce apoptosis in chicken blood lymphocytes (26), we speculated that NS protein of IBDV may play a similar role in pathogenesis.
Therefore, to study the function of NS protein in viral growth and pathogenesis, we constructed a full-length cDNA clone of segment A of IBDV vaccine strain D78, in which the initiation codon of the NS gene was mutated to a stop codon. We also constructed a full-length cDNA clone of segment B of the homologous IBDV strain. Using the reverse genetics system, we generated the parent IBDV (D78), as well as a mutant IBDV that lacked the expression of the NS protein. In this report, we compare the properties of the recovered D78 IBDV and mutant IBDV in cell culture and evaluate their pathological functions in the natural host.
MATERIALS AND METHODS
Cells and viruses.
Vero cells, used for transfection experiments, were maintained in medium 199 (M199) supplemented with 5% fetal bovine serum at 37°C in a humidified 5% CO2 incubator. Primary chicken embryo fibroblast (CEF) cells were prepared as described previously (14). Secondary CEF cells, used for virus titration, virus neutralization (VN), immunofluorescence, and apoptosis assays, were maintained in growth medium consisting of M199 and F10 (50%-50%, vol/vol) and 5% fetal bovine serum. The D78 vaccine strain of IBDV (Intervet Inc., Millsboro, Del.) and its transcript-derived progeny were plaque purified twice, propagated, and titrated in secondary CEF cells as described previously (14). Virus stocks were established by serial passage of the recovered viruses in CEF cells, at a multiplicity of infection (MOI) of 0.01.
Construction of full-length cDNA clones.
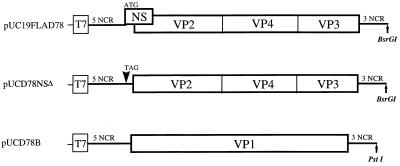
All manipulations of DNAs were performed according to standard protocols (19). Construction of a full-length cDNA clone of IBDV genome segment A of strain D78 has been described previously (14). It encodes all of the structural proteins (VP2, VP4, and VP3) as well as the NS protein (Fig. 1). To construct a mutant cDNA clone of segment A lacking the initiation codon of the NS gene, two primer pairs (see below) were synthesized and used for PCR amplification of the parent plasmid pUC19FLAD78. These primer pairs, pUCNde+ (5′-CCATATGCGGTGTGAAATACCG-3′, nucleotide positions 482 to 503 in pUC19) plus NSΔ− (5′-CTCTACTAACCTACAATGATAGCG-3′, positions 86 to 109 of IBDV segment A) and NSΔ + (5′-CGCTATCATTGTAGGTTAGTAGAG-3′) plus NdeBV (5′-CATACCCAAGATCATATGATGTG-3′, positions 640 to 662 of segment A), yielded DNA fragments of 342 and 576 bp, respectively. These fragments were combined and subsequently amplified by PCR, using the flanking primers (pUCNde+ and NdeBV) to produce a 918-bp fragment. This fragment was cloned into a pCRII vector (Invitrogen Corp.) to obtain plasmid pCRNSΔ. This plasmid was digested with NdeI, and the resulting fragment was cloned into NdeI-cleaved parent plasmid pUC19FLAD78. Finally, a mutant clone of segment A in which positions 97 and 98 of NS gene initiation codon (ATG) were substituted to create a stop codon (TAG) was obtained. A representative mutant clone of segment A was selected and designated pUCD78NSΔ (Fig. 1).
FIG. 1.
Schematic presentation of IBDV cDNA constructs for the generation of plus-sense RNA transcripts with T7 RNA polymerase. Plasmid pUC19FLAD78 encodes the polyprotein (VP2-VP4-VP3) and the NS protein (NS) of D78-IBDV. In plasmid pUCD78NSΔ, the initiation codon of NS gene is mutated to a stop codon. Plasmid pUCD78B encodes the RNA-dependent RNA polymerase (VP1). All plasmids contain a T7 promoter sequence at their 5′-ends. Plasmids were linearized with the appropriate restriction enzymes as indicated (↑).
To construct a cDNA clone of segment B of homologous IBDV strain D78, two primer pairs (B5′-D78 plus B5-IPD78 and B3′-D78 plus B3-IPD78) were synthesized and used for reverse transcription (RT)-PCR amplification. Sequences of the primers were identical to the one used for the construction of segment B cDNA clone of P2 strain (14). With genomic double-stranded RNA as a template, cDNA fragments were synthesized and amplified as specified by the supplier (Perkin-Elmer). Amplified fragments were cloned between the EcoRI site of a pCRII vector to obtain plasmids pCRD78A5′ and pCRD78A3′. To construct a full-length clone of segment B, the 5′-end fragment of IBDV (from plasmid pCRD78B5′) was first subcloned between EcoRI and PstI sites of pUC19 vector to obtain pUCD785′. Then the 3′-end fragment of IBDV (from plasmid pCRD78B3′) was inserted between the unique BglII and PstI sites of plasmid pUCD78B5′ to obtain full-length plasmid pUCD78B, which encodes VP1 protein (Fig. 1).
pUCD78NSΔ and pUCD78B DNA was sequenced by the dideoxy chain termination method (20), using an Applied Biosystems automated DNA sequencer, and the sequence data were analyzed by using PC/Gene (Intelligenetics) software. The integrity of the full-length constructs was tested by an in vitro transcription-translation coupled reticulocyte lysate system using T7 RNA polymerase (Promega Corp.).
Transcription and transfection of synthetic RNAs.
Transcription and transfection assays were performed as described in detail previously (14). Briefly, plasmids pUC19FLAD78, pUCD78NSΔ, and pUCD78B were digested with enzymes BsrGI and PstI (Fig. 1), respectively, and used as templates for in vitro transcription with T7 RNA polymerase (Promega). Vero cells were transfected with combined transcripts of either mutant or wild-type segment A and of segment B, using Lipofectin reagent (GIBCO/BRL). The resulting progeny were designated recombinant D78NSΔ (rD78NSΔ) and rD78, respectively.
Characterization of transcript-derived IBDV in vitro.
Infection of CEF cells by transcript-derived or transfectant viruses was analyzed by immunofluorescence assay (IFA) using rabbit anti-IBDV polyclonal or rabbit anti-NS-specific serum as described before (14). The anti-NS protein serum was kindly provided by Egbert Mundt (Federal Research Center for Virus Diseases of Animals, Insel Riems, Germany). Preparation of this anti-NS protein serum and its specificity have been described previously (12, 13). Briefly, CEF cells were infected with rD78 or rD78NSΔ IBDV at an MOI of 1 and incubated at 37°C for an appropriate time interval. The cells were then washed with phosphate-buffered saline, pH 7.4 (PBS), fixed with ice-cold methanol-acetone (1:1), and treated with either rabbit anti-NS serum or rabbit anti-IBDV serum. After being washed with PBS, the cells were treated with fluorescein-labeled goat anti-rabbit antibody (Kirkegaard & Perry Laboratories) and examined by fluorescence microscopy.
To examine viral structural proteins expressed by transfectant viruses, rD78 and rD78NSΔ IBDV were purified by sucrose and CsCl gradient centrifugation as described previously (15) and were adjusted to a protein concentration of 0.1 mg/ml. Equivalent amounts of purified viral samples were fractionated on a sodium dodecyl sulfate–12.5% polyacrylamide gel, transferred onto a nitrocellulose membrane, reacted with rabbit anti-IBDV serum, and detected with streptavidin-alkaline phosphatase and naphthol phosphate fast red color development reagents (24).
Growth curve of IBDV.
To analyze the growth characteristics of IBDV, confluent secondary CEF cells (in T-25 flasks) were infected with the parental D78 or with transcript-derived rD78 or rD78NSΔ virus stock (generated after five passages in CEF cells) at an MOI of 0.1. Infected cell cultures were harvested at different time intervals, and the titer of infectious virus present in the culture was determined by plaque assay on CEF cells as described previously (14).
Assays for cell viability and apoptosis.
Cell viability was measured by the trypan blue exclusion method and by colorimetric MTT (tetrazolium) (see below) assay (11). For the trypan blue exclusion method, secondary CEF cells were grown to 80% confluence in T-25 flask and infected with D78, rD78, or rD78NSΔ IBDV at an MOI of 1. At daily intervals (up to 7 days), the cells in the cultures were harvested, trypsinzed, and assayed for viability by using trypan blue stain (0.4%). For the colorimetric MTT assay, secondary CEF cells were seeded in 96-well plates at a density of 5 × 104 cells/well and mock infected or infected with either rD78 or rD78NSΔ IBDV at an MOI of 1. MTT [3-(4,5-dimethythiazol-2-yl)-2,5 diphenyl tetrazolium bromide; Sigma] was dissolved in serum and phenol-free medium (5 mg/ml) and filtered just prior to use. At daily intervals (up to 8 days), 20 μl of MTT solution was added to each well containing 200 μl of phenol-free medium, and the plate was incubated at 37°C for 2 h in a humidified chamber with 5% CO2. After the medium was removed, 200 μl of dimethy sulfoxide was added to each well, and the plates were read at an optical density (OD) of 540 nm in an enzyme-linked immunosorbent assay (ELISA) reader. The mean of the OD value ≥1.0 unit above that of control wells at each day was defined as 100% cell survival. To study apoptosis, secondary CEF cells (grown on coverslips to 80% confluence) were either mock infected or infected with transfectant viruses at an MOI of 1 and analyzed by terminal deoxynucleotidyl transferase (TdT)-mediated dUTP nick end labeling (TUNEL) assay. Apoptosis was monitored at 24, 48, and 72 h postinfection by using the TdT-based In Situ Cell Death Detection kit (Boehringer Mannheim Corp.), which measures the DNA strand breakage. Briefly, infected CEF cells (on coverslips) were washed with PBS, and fixed with freshly made 4% formaldehyde in PBS for 30 min at room temperature. The fixed cells were rinsed three times with PBS and then placed (upside down) in a dish containing 50 μl of TdT labeling mix (1× TdT reaction buffer, 2.5 mM CoCl2, 0.1 mM dithiothreitol, 0.25 U of TdT per ml, 10 mM fluorescein-16-dUTP). The reaction was carried out at 37°C in a humidified incubator for 60 min. The cells were then rinsed with PBST (PBS with 0.1% Triton X-100 and 0.5% Tween 20) and examined by fluorescence microscopy.
Chicken inoculation and serology.
Three-week-old specific-pathogen-free (SPF) chickens were obtained (SPAFAS, Inc., Storrs, Conn.), and housed in isolators. Prior to inoculation, the chickens were bled and their sera were tested by ELISA to ensure that they were negative for IBDV-specific antibodies. Three groups of SPF chickens (50 in each group) were given one of three treatments consisting of 2 drops of either culture medium (control), rD78 virus (5 × 103 PFU/ml), or rD78NSΔ virus (5 × 103 PFU/ml) into the conjunctival sac. Eight chickens from each group were removed at 2, 4, 6, 9, and 21 days postinfection and humanely killed by cerebrocervical separation. The BF was excised from each chicken and bisected. One BF hemisection was stored at −70°C and used for virus isolation and RT-PCR assay. The other BF hemisection was fixed and sectioned for histopathological examination and indirect IFA as described.
To evaluate the immune response of the mutant virus in vivo, blood samples were collected from each sampled chicken’s ventral medial wing vein at 14 and 21 days postinfection. VN antibody titer was determined by using parental IBDV strain D78 on CEF cells as described previously (25). The VN titer was reported as log2 of the highest reciprocal dilution that was capable of neutralizing 500 PFU of D78 IBDV. In addition, the serum was assayed for reovirus, Newcastle disease virus, infectious laryngotracheitis virus, and infectious bronchitis virus antibodies by using ELISA kits (Kirkegaard & Perry Laboratories).
Characterization of recovered viruses in vivo.
To detect and isolate the viruses from the chickens inoculated with the transfectant viruses, the bursa from each sampled chicken was ground in PBS to make a 10% bursal suspension. One-half milliliter of bursal homogenate was mixed with 4.5 ml of M199 and passed through 0.45-μm-pore-size syringe filter. The filtrate was used to infect confluent monolayers of CEF cells in T-75 flask. The cells were examined daily (up to 5 days) for the presence or absence of IBDV-specific cytopathic effect. In addition, the titer of the virus present in these cultures was determined by plaque assays on CEF cells as described previously (14).
Identification of recovered viruses by RT-PCR.
Total nucleic acids of uninfected and IBDV-infected CEF cells or bursal homogenates were isolated and analyzed by RT-PCR, performed essentially as described elsewhere (14). Segment A-specific primer NheΔ(−) (5′-CCCATTGTAGCATGCATCTGTCAG-3′, binding to nucleotide positions 536 to 559) was used for RT of genomic RNA. Following RT, the reaction products were amplified by PCR using segment A-specific primer A5′-D78 (5′-GGATACGATCGGTCTGACCCCGGGGGAGTCA-3′, nucleotide positions 1 to 31). The reaction products were separated by agarose gel electrophoresis and purified by using a QIAquick gel extraction kit (Qiagen Inc.). The PCR fragment, comprising the NS gene and the 5′ noncoding region of segment A, was cloned into a pCRII vector and sequenced as described above.
Histopathological studies.
The BF tissues were fixed by immersion in 10% neutral buffered formalin. The ratio of fixative to BF exceeded 10:1. Seven days later, a cross-sectional portion of each BF was processed through paraffin, stained with hematoxylin and eosin, and examined with a light microscope. The severity of the lesion was graded on a scale of + to +++ based on the extent of lymphocyte necrosis, follicular depletion, and atrophy.
RESULTS
Construction of full-length cDNA clones.
To study the role of NS protein in IBDV, we constructed a mutant cDNA clone of segment A in which the first and only initiation codon of the NS gene was mutated to a stop codon (Fig. 1). Thus, plasmid pUCD78NSΔ could encode only the precursor of the structural proteins (VP2, VP4, and VP3). In addition, we constructed a full-length cDNA clone of segment B of the homologous IBDV strain D78 which encoded VP1 protein (Fig. 1). Comparison of the deduced amino acid sequence of D78 segment B with that of the P2 strain showed 99.97% amino acid identity, indicating that these two European strains are closely related. Coupled transcription and translation of the above-specified plasmids in a rabbit reticulocyte system yielded protein products which comigrated with the marker IBDV proteins after fractionation on a sodium dodecyl sulfate-polyacrylamide gel and autoradiography (data not shown).
Characterization of generated viruses in vitro.
To study the function of NS protein in viral replication, Vero cells were transfected with combined transcripts of segments A and B. As expected, RNA transcripts of unmodified segments A and B from strain D78 generated rD78. When Vero cells were transfected with combined transcripts of modified segment A and B, it also generated a viable mutant virus (rD78NSΔ). To verify the mutation in rD78NSΔ, the genomic RNA was isolated and analyzed by RT-PCR using a primer pair specific for segment A. Sequence analysis of the cloned PCR product confirmed the expected nucleotide mutations in the NS gene from the mutant virus (data not shown). To detect the expression of NS protein, CEF cells were infected with the recovered viruses and analyzed by IFA using NS-specific antiserum. Figure 2 shows the results of immunofluorescence staining of IBDV-infected cells. Cells infected with rD78 virus expressed NS protein and gave a positive immunofluorescence signal (Fig. 2c and f). However, cells infected with mutant rD78NSΔ virus failed to give any fluorescence signal, indicating the absence of NS protein expression, even after passage 10 (Fig. 2b and e). No fluorescence was detected in the mock-infected cells (Fig. 2a and d).
FIG. 2.
Immunofluorescence staining of IBDV-infected cells used to detect NS protein expression. CEF cells were infected with passage 1 (b) or passage 10 (e) rD78NSΔ mutant virus stock or with passage 1 (c) or passage 10 (f) rD78 virus stock at an MOI of 1. Uninfected CEF cells were used as negative controls (a and d). After 24 h postinfection, the cells were fixed and analyzed by immunofluorescence staining with rabbit anti-NS protein serum. Magnifications, ×400.
To determine the replication kinetics of D78, rD78, and rD78NSΔ, CEF cells were infected with each virus and the virus titer was determined by plaque assay. Figure 3A depicts the growth curve of each virus (expressed as log PFU/milliliter) at different days postinfection. Our results indicate that the mutant virus (lacking the expression of NS protein) replicated somewhat more slowly and had a titer that was 1 log lower than that of the parent vaccine strain (D78) or recovered (rD78) virus at 6 days post-infection. However, the plaques produced by the mutant virus were similar in size to those produced by the parental virus. Furthermore, the transfectant viruses were purified by CsCl gradient, and their proteins were analyzed by Western blot analysis using IBDV antiserum. Qualitatively, viral structural proteins (VP2, VP4, and VP3) produced by the mutant virus were identical to the proteins synthesized by the parent D78 or recovered rD78 virus (data not shown). These results indicate that NS protein is not required for replication in cell culture, in agreement with the recent report by Mundt et al. (13).
FIG. 3.
Growth curve of IBDV (A) and cytotoxic effect in IBDV-infected cells (B). (A) Monolayers of CEF cells were infected with the indicated viruses at an MOI of 0.1 and harvested at the indicated time points, and infectious titers were determined by plaque assay. (B) Cell cultures were assayed for viability at the indicated times by the MTT assay. The mean of the OD value ≥1.0 unit above that of control wells at each day was defined as 100% cell survival. Each value is the average of two independent experiments.
Cytotoxic and apoptotic effects of recovered viruses.
The cytopathogenicity of rD78NSΔ IBDV was compared with that of rD78 or D78 IBDV in CEF cells by using the MTT assay and trypan blue exclusion method. Figure 3B shows the cytotoxicity results for these viruses at different days postinfection with the MTT assay. CEF cells infected with the parent D78 or rD78 IBDV showed a decrease in cell viability (40 to 45%) compared to cells infected with rD78NSΔ IBDV (about 90%). At 3 days postinfection, CEF cells exhibited considerable cell death due to IBDV replication, which coincided with the virus titer (Fig. 3A). The viability of CEF cells infected with the mutant virus (rD78NSΔ) was almost more than threefold greater than that of cells infected with the parent (D78) or recovered (rD78) virus at 6 days postinfection. Similar results were obtained by the trypan blue exclusion method (data not shown).
IBDV is known to induce apoptosis in chicken blood lymphocytes. To determine the apoptotic effects of rD78 and rD78NSΔ IBDV in CEF cells, the cells were infected with each virus at an MOI of 1, harvested at different time points, and analyzed by TUNEL assay. The cells infected with rD78 induced apoptosis, as evidenced by a green fluorescence signal (Fig. 4c, f, and i). In contrast, apoptosis induced by the mutant rD78NSΔ virus was significantly lower than that produced by rD78 (Fig. 4b, e, and h). No appreciable level of apoptosis was detected in the mock-infected cells (Fig. 4a, d, and g). Furthermore, the TUNEL-positive cells were also quantified in a blinded fashion, as shown in Table 1. The percentage of apoptotic cells in the group infected with rD78 virus was significantly higher (48 to 69%) than that infected with rD78NSΔ virus (15 to 23%) at 48 h and 72 h postinfection, respectively. These results indicate that IBDV-induced cell death is apparently reduced due to the absence of NS protein expression.
FIG. 4.
Analysis of IBDV-induced apoptosis by TUNEL assay. CEF cells were either mock infected (a, d, and g) or infected with rD78NSΔ (b, e, and h) or rD78 (c, f, and i) IBDV at an MOI of 1. Cells were fixed at 24 h (a to c), 48 h (d to f), and 72 h (g to i) postinfection and assayed by immunofluorescence for DNA breakage (TdT assay; green signal). The TUNEL staining of CEF cells, infected with NS protein-deficient mutant IBDV, show a substantial reduction in IBDV-induced apoptosis. Magnifications, ×100.
TABLE 1.
Quantification of apoptosis induced by rD78 or rD78NSΔ IBDV in vitro
| h postinfection | Group | Apoptotic cells/total cells counteda | Mean % of apoptotic cellsb ± SE |
|---|---|---|---|
| 24 | Control | 31/928 | 3.4 ± 0.9 |
| rD78NSΔ | 86/892 | 9.6 ± 1.1 | |
| rD78 | 109/887 | 12.3 ± 0.7 | |
| 48 | Control | 53/933 | 5.7 ± 1.1 |
| rD78NSΔ | 110/742 | 14.7 ± 1.3 | |
| rD78 | 282/587 | 48.1 ± 0.9 | |
| 72 | Control | 95/837 | 11.4 ± 1.3 |
| rD78NSΔ | 152/667 | 22.9 ± 1.2 | |
| rD78 | 264/381 | 69.4 ± 1.7 |
Determined by TUNEL assay using secondary CEF cells. The data are expressed as the mean of apoptotic cells (green signal) as a fraction of the total numbers of cell counted (mean of six different fields).
Each field for the cells counted was randomly chosen with blinding to the TUNEL results. The data are from two independent experiments.
Histopathological examination of the bursa.
To study the role of NS protein in viral pathogenesis, groups of 3-week-old SPF chickens were mock inoculated or inoculated with either rD78 or rD78NSΔ virus, and their bursa were analyzed for pathological lesions. Table 2 summarizes the results of histopathological examination of bursa obtained from different groups of chickens at selected days postinfection. Chickens inoculated with rD78 virus showed gross bursal lesions (bursal atrophy) at 6 and 9 days postinoculation. In addition, this virus produced microscopic lesions in the bursa at 2, 4, 6, and 9 days postinoculation, as indicated in Table 2. No gross or microscopic lesions were observed in chickens that were inoculated with the control medium or rD78NSΔ virus. Moreover, histopathologic sections of bursa derived from the control or rD78NSΔ groups were similar and had normal follicles and follicular connective tissues (Fig. 5a and b). However, the sections derived from rD78 group showed lymphocytic necrosis and follicular (B-lymphocyte) depletion at 2, 4, 6, and 9 days postinoculation. Subsequently, extensive accumulation of mononuclear cells resulted in the loss of distinction between cortex and medulla of infected bursal section (Fig. 5c). These results clearly indicate that the mutant virus does not cause bursal lesions.
TABLE 2.
Gross and microscopic lesions in the BF from chickens infected with rD78 or rD78NSΔ IBDV
| Day postinfection | Group | Pathology
|
|
|---|---|---|---|
| Grossa | Microscopicb | ||
| Control | − | − | |
| 2 | rD78 | − | ++ (4/8) |
| rD78NSΔ | − | − | |
| Control | − | − | |
| 4 | rD78 | − | +++ (6/8) |
| rD78NSΔ | − | − | |
| Control | − | − | |
| 6 | rD78 | + | +++ (8/8) |
| rD78NSΔ | − | − | |
| Control | − | − | |
| 9 | rD78 | + | +++ (7/8) |
| rD78NSΔ | − | − | |
| Control | − | − | |
| 21 | rD78 | − | − |
| rD78NSΔ | − | − | |
−, no gross lesion; +, small bursa.
−, no histological lesion; + to +++, degree of extent of multifocal purulent necrotizing bursitis with follicular depletion. Numbers in parentheses represent number of bursa with lesions/number of bursa examined. The result is the representative of two independent experiments.
FIG. 5.
Histopathologic appearance of sections (hematoxylin and eosin) of BF derived from mock-infected and infected chickens at day 6 postinfection. (a) Cortical lymphocytes (dark gray cells adjacent to connective tissue that separates follicles) and medullary lymphocytes (light gray cells in follicle centers) in portions of six follicles from an uninfected chicken are normal. In addition, the interfollicular connective tissues are normal. (b) Follicles and interfollicular connective tissues from a chicken infected with the mutant rD78NSΔ virus are normal and cannot be differentiated from their control counterparts. (c) There is lymphocytic necrosis and heterophilic inflammation in six follicles in the BF from a chicken infected with rD78 virus. Notice the loss of distinction between the cortex and the medulla and the bands of interfollicular connective tissue that are infiltrated by myriad heterophils and macrophages. Magnifications, ×100.
Detection of virus from the bursa.
To detect the presence of virus in the BF of infected chickens, BF from each group were pooled and homogenized in M199, and the filtered homogenate was analyzed for infectivity in CEF cells. After 2 to 3 days postinfection, the virus was detected (as evidenced by cytopathic effect) in cells infected with the homogenates of rD78 or rD78NSΔ groups at 2, 4, 6, and/or 9 days postinoculation (Table 3). No virus was detected in 21-day bursal homogenate, indicating that the virus was cleared from the bursa. As expected, no virus was detected in the control group. In addition, the presence of viral antigen in the bursal sections of rD78 and rD78NSΔ groups was also confirmed by IFA using IBDV polyclonal antiserum, even on sections that did no show histopathological lesions at 2, 4, 6, and/or 9 days postinfection (data not shown). To verify the presence or absence of mutation in the recovered viruses, whole-cell nucleic acid was isolated from each bursal homogenate and analyzed by RT-PCR as described above. A 555-bp fragment was amplified from the homogenates of rD78 and rD78NSΔ groups at 2, 4, 6, and/or 9 days postinoculation but not from the control group or from the 21-day bursal homogenates (Table 3).
TABLE 3.
Detection of virus in the BF of chickens infected with rD78 or rD78NSΔ IBDV
| Group | Virus isolationa on day:
|
RT-PCRb on day:
|
||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 2 | 4 | 6 | 9 | 21 | 2 | 4 | 6 | 9 | 21 | |
| Control | − | − | − | − | − | − | − | − | − | − |
| rD78 | + | + | + | − | − | + | + | + | − | − |
| rD78NSΔ | + | + | + | + | − | + | + | + | + | − |
BF from each group were pooled, and bursal homogenates were used to inoculate secondary CEF cells. +, presence of cytopathic effect; −, absence of cytopathic effect. The virus titers of each group are shown in Table 4.
+, virus detected by RT-PCR; −, no virus detected by RT-PCR.
Genetic stability of recovered viruses.
To determine the genetic stability of the transfectant viruses in vitro, we propagated the viruses in CEF cells (up to 10 passages), isolated whole-cell nucleic acids, and amplified the NS gene by RT-PCR. Sequence analysis of the cloned PCR product confirmed the expected nucleotide mutations in the NS gene of the mutant virus, whereas no mutation was detected in the PCR product of rD78 virus. Similarly, to determine the genetic stability of these viruses in vivo, chickens were inoculated with transfectant viruses, and their BF were collected at various days postinfection. Total nucleic acid was extracted from bursal tissue, and the NS gene was amplified by RT-PCR using a primer pair specific for segment A. Sequence analysis of the NS gene from the mutant virus revealed the presence of two mutations (97A→T, 98T→A) in bursal samples collected at 2, 4, 6, and 9 days postinfection. As expected, no nucleotide mutation was detected in the NS gene of the bursal materials infected with rD78 virus. These results clearly demonstrate that the mutant virus replicated in the BF of chickens but did not revert to the parent D78 IBDV.
Replication and immunogenicity of recovered viruses.
To compare the replication behaviors of recovered viruses in vivo, chickens were inoculated with equal amounts (5 × 103 PFU) of rD78 and rD78NSΔ IBDV. Virus titers in the bursa from each group at different time points were determined by plaque assay on CEF cells (Table 4). The results indicate that rD78NSΔ virus replicated at a lower rate than rD78 virus and its virus titer reached a peak at day 6 (versus day 2 for rD78 virus), suggesting a possible role of NS protein during in vivo replication. Indirect IFA was performed on bursal sections of chickens infected with rD78 and rD78NSΔ IBDV, using NS-specific antiserum. Bursal sections from chickens infected with rD78 gave a positive immunofluorescence signal at 2 and 4 days postinfection, but the sections from chickens infected with the mutant virus did not give a fluorescence signal, confirming the lack of NS protein expression (data not shown). These results clearly show that the mutant virus, lacking the expression of NS protein, efficiently replicated in the BF of chickens.
TABLE 4.
Virus and VN antibody titers of chickens infected with rD78 or rD78NSΔ IBDV
| Group | Virus titera (PFU/ml) in bursa on day:
|
Mean VN titerb (log2) ± SE on day:
|
|||||
|---|---|---|---|---|---|---|---|
| 2 | 4 | 6 | 9 | 21 | 14 | 21 | |
| Control | 0 | 0 | 0 | 0 | 0 | <2 | <2 |
| rD78 | 7.2 × 106 | 2.3 × 106 | 1.3 × 102 | 0 | 0 | 11.3 ± 1.1 | 12.2 ± 0.4 |
| rD78NSΔ | 3.0 × 105 | 6.8 × 105 | 2.2 × 106 | 1.8 × 102 | 0 | 10.8 ± 0.5 | 12.0 ± 0.1 |
Determined by plaque assays on CEF cells.
VN assays were performed on CEF cells, and the ability to neutralize 500 PFU of D78 virus is represented as log of the 50% endpoint titer.
To compare the immune response induced by rD78 and rD78NSΔ IBDV, chickens were inoculated with the recovered viruses and bled at 14 and 21 days postinoculation, and their sera were analyzed by the VN test. VN antibody titers of the sera are shown in Table 4. Although rD78NSΔ virus replicated at a lower rate than rD78 virus and its titer peaked at day 6, it induced VN titers that were comparable to those induced by the recovered D78 virus at 14 and 21 days postinoculation. These results indicate that the mutant virus, which is deficient in producing NS protein, does not affect the humoral immune response to IBDV in the natural host.
DISCUSSION
As a first application of IBDV reverse genetics, and to study the function of NS protein in vivo, we generated an NS protein-deficient virus and demonstrated that the mutant virus can replicate in the bursa of inoculated chickens but will not induce lesions. This finding implies that NS protein is directly involved in viral pathogenesis since the parent IBDV, expressing the NS protein, was able to elicit pathological response in the natural host. However, the mechanism by which the NS protein would exert its function remains to be seen.
The NS protein is highly conserved in all serotype I IBDV strains studied to date (greater than 95% identity). However, the NS protein of serotype II IBDV strains appears to be different and shows 73% sequence identity. Since serotype II viruses are naturally avirulent and do not cause any pathological lesions in chickens (7), these residues may play a role in the pathogenicity of the virus. Moreover, in another birnavirus, IPNV, segment A also encodes a 17-kDa NS protein (from a small ORF), which is found in IPNV-infected cells (10). Although the reverse genetics system for IPNV is not available as yet, we would predict a similar function for its NS protein.
The NS proteins of animal viruses have been shown to play an important role in viral replication and pathogenesis (1, 3, 16, 17). For example, the rotavirus NS glycoprotein NSP4 was shown to induce age-dependent diarrhea in mice (1). In foot-and-mouth disease virus (a picornavirus), the 16-kDa NS protein (leader protease) was shown to be dispensable for viral replication in vitro and in vivo (17). Furthermore, this leaderless mutant virus did not cause lesions in the lung when infected by aerosol and was found to be nonpathogenic for cattle (3). In chicken anemia virus (another immunosuppressive virus), a cysteine- and proline-rich, 14-kDa NS protein (VP3) was shown to cause apoptosis in lymphoblastoid T cells and was implicated in pathogenesis (16). However, this protein was found to be essential for viral replication. In our studies, we showed that NS protein of IBDV is not required for viral replication in vitro and in vivo, and NS protein-deficient virus does not cause bursal lesions in vivo. Our results also indicated that IBDV-induced cell death was significantly reduced due to the absence of NS protein expression. It would be interesting to know whether the expression of NS protein alone can induce apoptosis in vitro.
The parental IBDV strain D78 is an attenuated (intermediate) vaccine strain that is widely used in Europe. This vaccine strain can break through waning levels of maternal antibodies and elicit a proper immune response in chickens. However, when chickens are inoculated with an excessive dose of this virus, it can cause bursal lesions. Our studies show that the absence of NS protein expression can further attenuate the virus, and the resultant mutant virus will not induce bursal lesions, even with a higher inoculation dose (104 PFU/ml). The NS protein-deficient virus replicated nearly as well as the parental rD78 virus and was easily recovered from the bursa. Virus recovered from infected chickens retained the mutation that ablated NS protein expression, indicating that NS protein is nonessential for virus replication. It should be noted that NS protein-deficient virus exhibited a slight delay in replication, whereas the rD78 virus replicated quickly in chickens and its titer rapidly declined at 6 days postinfection. In contrast, the mutant virus replicated efficiently in chickens and its titer reached a peak at 6 days postinfection. It is evident that infected chickens were able to clear rD78 virus, but not the mutant virus, at 9 days postinfection. At present, we do not know why NS protein-deficient virus is able to persist longer than rD78 virus in the natural host. It is known that the major histocompatibility complex class I-restricted immune response is important for host against viral infection. It may be possible that the rD78 complex induces a stronger cytotoxic T-lymphocyte response than the NS protein-deficient virus. Although the mutant virus replicated at a slightly lower rate than rD78 virus, it induced VN titers that were comparable to those induced by D78 virus. This indicates that the absence of NS protein expression in the mutant virus will not affect the humoral immune response to IBDV in the natural host. Therefore, it may be feasible to prepare novel, live-attenuated vaccines for IBDV that are nonpathogenic to chickens.
Infection of chickens with IBDV results in lymphocyte depletion of the bursa follicles and bursal necrosis with subsequent immunosuppression. Since immunoglobulin M-bearing B lymphocytes are the major target cells for the virus, the proportion of these cells is significantly reduced after IBDV infection (5, 18). Whether the use of such NS protein-deficient viruses can cause immunosuppression in chickens remains to be seen.
ACKNOWLEDGMENTS
We thank Donald L. Nuss for reviewing the manuscript, and we thank Gerard H. Edwards, E. Craig Player, and Debrah W. Fuchs for technical assistance.
This work was supported in part by a grant from the Maryland Agricultural Experiment Station and U.S. Department of Agriculture (NRICGP grant 97-02492) to V.N.V.
REFERENCES
- 1.Ball J M, Tian P, Zeng C Q-Y, Morris A P, Estes M K. Age-dependent diarrhea induced by a rotaviral nonstructural glycoprotein. Science. 1996;272:101–104. doi: 10.1126/science.272.5258.101. [DOI] [PubMed] [Google Scholar]
- 2.Böttcher B, Kiselev N A, Stel’Mashchuk V Y, Perevozchikova N A, Borisov A V, Crowther R A. Three-dimensional structure of infectious bursal disease virus determined by electron microscopy. J Virol. 1997;71:325–330. doi: 10.1128/jvi.71.1.325-330.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Brown C C, Piccone M E, Mason P W, McKenna T S-C, Grubman M J. Pathogenesis of wild-type and leaderless foot-and-mouth disease virus in cattle. J Virol. 1996;70:5638–5641. doi: 10.1128/jvi.70.8.5638-5641.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dobos P, Hill B J, Hallet R, Kells D T C, Becht H, Teninges D. Biophysical and biochemical characterization of five animal viruses with bisegmented double-stranded genomes. J Virol. 1979;32:593–605. doi: 10.1128/jvi.32.2.593-605.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hirai K, Calnek B W. In vitro replication of infectious bursal disease virus in established cell lines and chicken B lymphocytes. Infect Immun. 1979;25:964–970. doi: 10.1128/iai.25.3.964-970.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hudson P J, McKern N M, Power B E, Azad A A. Genomic structure of the large RNA segment of infectious bursal disease virus. Nucleic Acids Res. 1986;14:5001–5012. doi: 10.1093/nar/14.12.5001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ismail N M, Saif Y M, Moorhead P D. Lack of pathogenicity of five serotype 2 infectious bursal disease viruses. Avian Dis. 1988;32:757–759. [PubMed] [Google Scholar]
- 8.Jagadish M N, Staton V J, Hudson P L, Azad A A. Birnavirus precursor polyprotein is processed in Escherichia coli by its own virus-encoded polypeptide. J Virol. 1988;62:1084–1087. doi: 10.1128/jvi.62.3.1084-1087.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kibenge F S B, Dhillon A S, Russell R G. Biochemistry and immunology of infectious bursal disease virus. J Gen Virol. 1988;69:1757–1775. doi: 10.1099/0022-1317-69-8-1757. [DOI] [PubMed] [Google Scholar]
- 10.Magyar G, Dobos P. Evidence for the detection of the infectious pancreatic necrosis virus polyprotein and the 17-kDa polypeptide in infected cells and of the NS protease in purified virus. Virology. 1994;204:580–589. doi: 10.1006/viro.1994.1572. [DOI] [PubMed] [Google Scholar]
- 11.Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assay. J Immunol Methods. 1983;65:1170–1174. doi: 10.1016/0022-1759(83)90303-4. [DOI] [PubMed] [Google Scholar]
- 12.Mundt E, Beyer J, Müller H. Identification of a novel viral protein in infectious bursal disease virus-infected cells. J Gen Virol. 1995;76:437–443. doi: 10.1099/0022-1317-76-2-437. [DOI] [PubMed] [Google Scholar]
- 13.Mundt E, Köllner B, Kretzschmar D. VP5 of infectious bursal disease virus is not essential for viral replication in cell culture. J Virol. 1997;71:5647–5651. doi: 10.1128/jvi.71.7.5647-5651.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mundt E, Vakharia V N. Synthetic transcripts of double-stranded birnavirus genome are infectious. Proc Natl Acad Sci USA. 1996;93:11131–11136. doi: 10.1073/pnas.93.20.11131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nick H, Müller H, Becht H. Structural and growth characteristic of infectious bursal disease virus. J Virol. 1976;18:227–234. doi: 10.1128/jvi.18.1.227-234.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Noteborn M H M, Todd D, Verschueren C A J, De Gauw H W F M, Curran W L, Veldkamp S, Douglas A J, McNulty M S, van der Eb A J, Koch G. A single chicken anemia virus protein induces apoptosis. J Virol. 1994;68:346–351. doi: 10.1128/jvi.68.1.346-351.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Piccone M E, Rieder E, Mason P W, Grubman M J. The foot-and-mouth disease virus leader proteinase gene is not required for viral replication. J Virol. 1995;69:5376–5382. doi: 10.1128/jvi.69.9.5376-5382.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rodenberg J, Sharma J M, Belzer S W, Nordgren R M, Naqi S. Flow cytometric analysis of B cell and T cell subpopulations in specific-pathogen-free chicken infected with bursal disease virus. Avian Dis. 1994;38:16–21. [PubMed] [Google Scholar]
- 19.Sambrook J, Fritsch E F, Maniatis T. Molecular cloning: a laboratory manual. 2nd ed. Cold Spring Harbor, N.Y: Cold Spring Harbor Laboratory; 1989. [Google Scholar]
- 20.Sanger F, Nicklen S, Coulson A R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA. 1977;74:5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Spies U, Müller H. Demonstration of enzyme activities required for the cap structure formation in infectious bursal disease virus, a member of the birnavirus group. J Gen Virol. 1990;17:977–981. doi: 10.1099/0022-1317-71-4-977. [DOI] [PubMed] [Google Scholar]
- 22.Spies U, Müller H, Becht H. Properties of RNA polymerase activity associated with infectious bursal disease virus and characterization of its reaction products. Virus Res. 1987;8:127–140. doi: 10.1016/0168-1702(87)90024-4. [DOI] [PubMed] [Google Scholar]
- 23.Vakharia V N, He J, Ahamed B, Snyder D B. Molecular basis of antigenic variation in infectious bursal disease virus. Virus Res. 1994;31:265–273. doi: 10.1016/0168-1702(94)90009-4. [DOI] [PubMed] [Google Scholar]
- 24.Vakharia V N, Snyder D B, He J, Edwards G H, Savage P K, Mengel-Whereat S A. Infectious bursal disease virus structural proteins expressed in a baculovirus recombinant confer protection in chickens. J Gen Virol. 1993;74:1201–1206. doi: 10.1099/0022-1317-74-6-1201. [DOI] [PubMed] [Google Scholar]
- 25.Vakharia V N, Snyder D B, Lütticken D, Mengel-Whereat S A, Savage P K, Edwards G H, Goodwin M A. Active and passive protection against variant and classic infectious bursal disease virus induced by baculovirus expressed structural proteins. Vaccine. 1994;12:452–456. doi: 10.1016/0264-410x(94)90124-4. [DOI] [PubMed] [Google Scholar]
- 26.Vasconcelos A C, Lam K M. Apoptosis induced by infectious bursal disease virus. J Gen Virol. 1994;75:1803–1806. doi: 10.1099/0022-1317-75-7-1803. [DOI] [PubMed] [Google Scholar]