Abstract
The respiratory syncytial virus (RSV) G glycoprotein promotes differentiation of type 2 CD4+ T lymphocytes and induces an eosinophilic response in lungs of RSV-infected mice. A unique feature of G is that a second initiation codon in the transmembrane region of the glycoprotein results in secretion of soluble protein from infected cells. Recombinant vaccinia viruses that express wild-type G (vvWT G), only secreted G (vvM48), or only membrane-anchored G (vvM48I) were used to define the influence of G priming on immunopathogenesis. Mice immunized with vvM48 had more severe illness following RSV challenge than did mice primed with vvWT G or vvM48I. Coadministration of purified G during priming with the construct expressing membrane-anchored G shifted immune responses following RSV challenge to a more Th2-like response. This was characterized by increased interleukin-5 in lung supernatants and an increase in G-specific immunoglobulin G1 antibodies. Eosinophils were present in the infiltrate of all mice primed with G-containing vectors but were greatest in mice primed with regimens including secreted G. These data suggest the form of G protein available for initial antigen processing and presentation is an important factor in promoting Th2-like immune responses, including the induction of lung eosinophilia. The ability of RSV to secrete G protein may therefore represent a viral strategy for immunomodulation and be a key determinant of disease pathogenesis.
Respiratory syncytial virus (RSV) is the leading cause of serious respiratory virus infections in infants and is a high priority for vaccine development. One obstacle to vaccine development is the legacy of vaccine-enhanced disease in children following natural infection (32, 34). Studies of vaccine-enhanced disease in animal models have suggested that the process is related to an altered pattern of CD4+ T-lymphocyte activation and cytokine production (5, 10, 18, 19, 35, 49, 62, 69). The G glycoprotein in particular has been implicated as an RSV antigen that promotes activation of Th2 CD4+ T lymphocytes and induces eosinophilic infiltrates in the lung following RSV challenge (2, 4, 58).
The large glycoprotein G serves as the attachment protein of RSV (40) and is one of the major glycoproteins expressed in the membrane of the virus (67, 68). The protein is expressed on the surface of infected cells and secreted into the extracellular environment (26, 27). Detailed structural studies have demonstrated that G possesses characteristics that are unusual among viral membrane proteins (31, 39, 70). These traits include secretion of the protein, extensive O-glycosylation, and a unique peptide sequence with no significant homology to other viral proteins.
RSV G protein plays a role in both induction of protective immunity and disease pathogenesis. RSV-infected children produce a well-characterized antibody response to G (23, 43, 64, 65). Passive transfer of antibodies to G (63, 66) or active vaccination with recombinant vaccinia viruses expressing RSV G (6, 45) is partially protective against live virus challenge in rodent models of RSV (60, 61, 66). Screening of a panel of monoclonal antibodies raised against RSV G demonstrated that the majority of antibodies were reactive only with the glycosylated form of the protein and did not recognize the unglycosylated protein backbone (47). When protein glycosylation was blocked or altered, production of G protein was inhibited (38). Nevertheless, a peptide fragment, hypothesized to constitute a major part of the protection domain, elicited an antibody response and reduced viral titers in immunized mice (57). While capable of inducing an antibody response, G is not a recognized target of cytotoxic T-lymphocyte-mediated immune responses in humans or mice (2, 4, 7, 8, 48, 59).
Use of recombinant vaccinia virus expressing RSV G (vacG) to prime mice generated a Th2 CD4+ T lymphocyte response, while vaccination with fusion (F) protein-expressing virus (vacF) induced a Th1 CD4+ T-cell response (2, 48). The disease profiles and lung pathology in naive mice injected with T cells from vacG- or vacF-primed mice also differed dramatically (1, 3, 46). Transfer of G-specific CD4+ T-cell clones to naive mice predisposed for more severe lung pathology and disease upon RSV challenge than did 22K- or F-specific clones (1). In vacG-primed RSV-challenged mice, bronchoalveolar lavage (BAL) revealed 14 to 25% of cells in the pulmonary infiltrate to be eosinophils, whereas <3% of cells were eosinophils in mice primed with vacF or vacN (nucleoprotein [N]-expressing vaccinia virus) (46). Attempts to promote Th1-like responses rather than Th2 responses with the adjuvant QS21 minimally decreased eosinophilia and interleukin-5 (IL-5) production induced by G priming, whereas it had a much greater effect on responses to F (25). These data suggest that the primary or secondary antigenic structure of G may influence the composition of subsequent RSV-specific immune responses, but that RSV immunization with secreted glycoproteins can itself predispose to Th2-like immune responses.
Membrane-anchored and secreted forms of G protein are produced from alternative initiation codons (52). Characterization of these two forms of RSV G demonstrate that the two proteins are structurally and antigenically similar, except for lack of the cytoplasmic tail in the secreted form of the protein. The production of two forms of G protein is a trait conserved among all known strains of RSV, suggesting that it provides an advantage for RSV survival. We have addressed the contribution of secreted G glycoprotein to the composition of subsequent RSV-specific immune responses relative to influences from the primary or secondary antigen structure of G alone. Recombinant vaccinia viruses which express the three forms of G, secreted, membrane anchored, and wild type (both secreted and membrane anchored), have been constructed (52). Using these constructs, we examined whether the route of antigen processing of the G antigen, rather than its primary or secondary antigenic structure, was the major determinant in regulating subsequent immune responses.
MATERIALS AND METHODS
Virus stocks and cell lines.
Recombinant vaccinia viruses were obtained that express various RSV proteins (Table 1). Viral stocks were grown and purified by density gradient centrifugation on potassium tartrate gradients, using a modification of the protocol of Hall and Martin (24). Briefly, BSC40 monolayers were infected with vaccinia virus and grown for 2 days. The infected cells and media were then harvested and sonicated, and the material was layered onto a cushion of 1.46 M sucrose–1 mM NaH2PO4 (pH 7.2). After centrifugation for 90 min at 62,000 × g, the supernatant was removed and the pelleted virions were resuspended in phosphate-buffered saline (PBS). The virus was sonicated and layered onto a continuous gradient from 0.85 to 2.13 M potassium tartrate in 0.01 M Tris-HCl (pH 8.5). The gradients were centrifuged 60 min at 62,000 × g; the opaque band of virus was removed from the gradient and sonicated. The isolated virus was washed in cold PBS and pelleted by centrifugation for 90 min at 85,000 × g. The virus pellet was resuspended in Eagle minimal essential medium (EMEM)–10% fetal calf serum (FCS), aliquoted, and stored at −70°C. All steps of vaccinia virus purification process were performed at 4°C. A stock of RSV (A2 strain) was generated in HEp-2 cells and stored at −70°C as previously described (20). All cell lines and viral stocks were determined to be free of mycoplasma contamination by PCR analysis (American Type Culture Collection, Rockville, Md.).
TABLE 1.
Products used for priming immunization
Purification of secreted RSV G protein.
Monolayers of BSC40 cells were infected with vvM48 at a multiplicity of infection of 0.5. After 4 days growth, the culture supernatant was transferred to 50-ml centrifuge tubes and stored at −70°C. To separate cells and intact virions from secreted proteins, the culture supernatant was overlaid on a cushion of 1.46 M sucrose–1 mM NaH2PO4 (pH 7.2) in an SW28 ultracentrifuge tube and centrifuged 90 min at 62,000 × g in a Beckman (Palo Alto, Calif.) L8-80 ultracentrifuge. The supernatant was removed and dialyzed overnight at 4°C against 20 mM Tris-HCl–0.5 M NaCl (pH 7.4) (ConA [concanavalin A] binding buffer) and was then applied to a ConA-Sepharose lectin column (Pharmacia Biotech, Piscataway, N.J.). Unbound proteins were removed from the column by washing with binding buffer until OD280 (optical density at 280 nm) readings were less than 0.02. Bound proteins were eluted by application of a continuous gradient from 0 to 0.5 M methyl-α-mannopyranoside in 20 mM Tris-HCl–0.5 M NaCl (pH 7.4). Three-milliliter fractions were collected throughout the chromatographic run. Fractions were screened by capture enzyme-linked immunosorbent assay (ELISA), and G-containing fractions were pooled and concentrated in Centriprep concentrators (Amicon, Inc., Beverly, Mass.). The purified protein was stored at −70°C.
Mouse priming and challenge.
Pathogen-free 8-week-old BALB/c mice were obtained from Charles Rivers Laboratories (Raleigh, N.C.) and were housed in a barrier facility. Mice were primed with 5 × 105 PFU (in 0.05 ml) of recombinant vaccinia virus by intradermal inoculation at the base of the tail and were monitored daily for lesion formation. In G reconstitution experiments, 850 ng of purified G (in 25 μl) was injected intradermally at the base of the tail on each of the days 0, 1, 2, and 3 of priming. Formalin-inactivated, alum-precipitated RSV (FI-RSV) was prepared as previously described (19) and administered intramuscularly. Six weeks after immunization (4 weeks after clearance of cutaneous lesions), the mice were anesthetized and intranasally infected with 0.1 ml containing 107 PFU of live RSV. RSV-infected mice were weighed for 12 days following challenge. Illness was graded daily by a blinded observer where clinical features of illness were scored as follows: 0, no apparent illness; 1, slightly ruffled fur; 2, ruffled fur but active; 3, ruffled fur and inactive; 4, ruffled, inactive, hunched posture, and gaunt; 5, dead.
RSV plaque assays.
Four or eight days following RSV challenge, mice were sacrificed by CO2 narcosis and cervical dislocation. The lungs were removed, placed in EMEM–10% FCS, and quick-frozen in an alcohol-dry ice bath. RSV titers in the lungs were measured by standard plaque assays using 80% confluent HEp-2 monolayers. Briefly, lungs were quick-thawed and ground with a mortar and pestle. Serial 10-fold dilutions of lung supernatants were used to infect the monolayers in triplicate, and cultures were grown under 0.75% methylcellulose in EMEM–10% FCS. Cells were formalin fixed 5 days after infection and stained with hematoxylin and eosin, and plaques were counted under a dissecting microscope. Data are represented as the geometric mean log10 PFU per gram of lung tissue ± standard error of the mean (SEM) at the dilution producing more than five plaques per well.
Analysis of vaccinia virus replication in vivo.
Pathogen-free 8-week-old BALB/c mice were infected with 5 × 105 PFU (in 0.05 ml) of recombinant vaccinia virus by intradermal inoculation at the base of the tail and were monitored daily for lesion formation. On days 4, 7, 12, and 17 postinfection, mice were sacrificed. Spleens and inoculation site (10 to 12 mm of tail) were removed, placed in EMEM–10% FCS, and quick-frozen. Vaccinia virus titers in each tissue sample were measured by plaque assay as described above for RSV plaque assay with modification. The modifications included use of 80% confluent BSC40 cells, a 2-day incubation, and staining with crystal violet rather than with hematoxylin and eosin.
Quantitation of IFN-γ and IL-5 in lung tissues.
Levels of gamma interferon (IFN-γ) and IL-5 in lung tissues of primed and challenged mice were measured using commercially available ELISA kits (Endogen, Woburn, Mass.) according to the kit protocols. Briefly, supernatants from ground lungs of primed mice 4 and 8 days following RSV challenge (used for RSV titrations above) were thawed, added to precoated wells, and incubated for 2 h. Dilutions of recombinant cytokine were included for generation of a standard curve. Peroxidase-labeled anticytokine antibody was added to detect bound cytokine, and the plates were developed by the addition of tetramethylbenzidene substrate. Concentrations of cytokine in the lung supernatants were calculated from the standard curve produced.
Histopathology of lungs from RSV-challenged mice.
Mice were sacrificed 8 days after RSV challenge, and their uncut lungs were fixed in phosphate-buffered 10% formalin for histopathological examination. Thin sections of formalin-fixed, paraffin-embedded lung tissue were stained with hematoxylin and eosin. Analysis included grading the degree of cellular infiltration and edema at the level of the alveolus and determination of the types of infiltrating cells. Inflammation was assessed according to the following scale: 0, no infiltrate; 1, mixed generalized increase in interstitial mononuclear cells without widening of alveolar septa; 2, dense septal mononuclear infiltrates with septal thickening and occasional foci of intra-alveolar cells; 3, significant alveolar space consolidation (intra-alveolar edema, inflammation, or hemorrhage) in addition to interstitial inflammation. Eosinophil-specific Luna staining was used to demonstrate tissue eosinophilia.
To quantitatively assess and compare the tissue eosinophilia induced by the various priming regimens, the percentage of eosinophils was determined by counting cells in lung infiltrates. Bronchovascular bundles in which the arteries were cut in cross section (where the length and width of the diameter were <1.5× different in relative lengths) were identified in Luna-stained lung sections. Total cells and the number of eosinophils in the infiltrate around each qualifying vessel were counted. The entire infiltrate around each bronchovascular bundle was counted until the total cell count exceeded 300 cells. An average of three to five bronchovascular bundle infiltrates were counted per lung. The percentage of eosinophils were then calculated as (number of eosinophils/number of total cells) × 100.
BAL of RSV-challenged mice.
Mice were sacrificed 6 days after RSV challenge. The trachea was surgically exposed, and a 19-gauge blunt-end needle was inserted into a small cut made in the trachea. Through this endotracheal tube, 0.5 ml PBS containing 5% FCS was injected into the lungs. After washing for approximately 30 s, the fluid was withdrawn and transferred to a microcentrifuge tube. Smears were made of all BAL samples and were air dried overnight. The dried smears were then stained with Diff-Quick (Fisher Scientific, Pittsburgh, Pa.), and differential counts of standard cell types were performed.
Titers and isotype profiles of G-specific antibodies.
Prechallenge sera were collected from vaccinia virus-primed mice by retro-orbital bleeding the day before RSV challenge. Sera were stored at −70°C until postchallenge samples were collected. Six weeks following RSV challenge, the mice were again bled, and serum was separated and frozen. To measure G-specific antibody, Nunc Immulon II microtiter plates were coated for 1 h with purified G protein (Wyeth-Lederle, Pearl River, N.Y.), the G protein isolated from RSV subtype A virus. Serial two-fold dilutions of pre- and postchallenge sera were added to the coated wells in duplicate and incubated for 1 h. Bound immunoglobulin was detected by the addition of horseradish peroxidase-conjugated rabbit anti-mouse immunoglobulin G1 (IgG1), IgG2a, or IgG/A/M (Zymed Laboratories, San Francisco, Calif.). The plates were developed with tetramethylbenzidene. Data are represented as log2 values of the serum dilution producing 0.100 OD450 unit and greater than twice the antigen-negative well.
Production of neutralizing antibodies to RSV and vaccinia virus.
Titers of neutralizing antibodies to both RSV and vaccinia virus in pre- and postchallenge sera were measured by plaque reduction neutralization assays as previously described (20).
Statistical analysis.
Data from individual experiments were maintained in a Paradox database. Statistical analysis was performed by transferring data from the database into the SAS (Chapel Hill, N.C.) statistical software to perform analysis of variance using Kruskal-Wallis and Wilcoxon rank sum tests. Comparisons were made between individual experiments by using statistical modeling and trend analysis calculated by the general linear model method in the SAS package. P values less than 0.05 were considered statistically significant.
RESULTS
Weight loss and illness following challenge of primed mice.
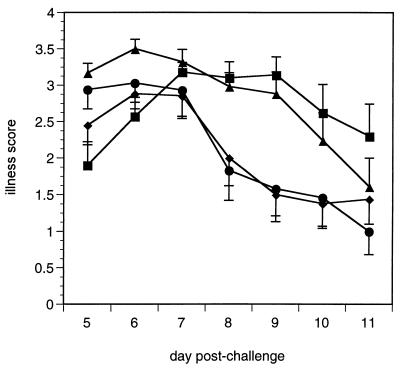
Mice were immunized with vaccinia virus constructs expressing F, wild-type G (vvWT G), secreted G only (vvM48), or membrane-anchored G only (vvM48I) (Table 1). Consistent with previous observations (1, 61), illness in mice vaccinated with vvWT G was more severe and weight loss was significantly greater than in vacF-immunized mice (P < 0.05 at days 4 to 12, Kruskal-Wallis test [data not shown]). Mice primed with vvM48I exhibited patterns of illness and weight loss similar to those of vvWT G-primed mice (Fig. 1; P > 0.05 at all days). However, mice immunized with vvM48 demonstrated illness and weight loss profiles significantly more severe than observed in vvM48I-primed mice (Fig. 1; P < 0.05 at all days except day 7, where P = 0.07). Thus, the presence of secreted G alone during priming, rather than membrane-anchored G, was associated with more severe disease upon subsequent RSV infection. However, the presence of membrane-anchored G can have a modulating effect on the illness profile induced by secreted G, decreasing the severity, as seen in mice immunized with vvWT G.
FIG. 1.
Illness in vaccine-primed mice after RSV challenge. Mice were primed with either vvWT G (•), vvM48I (⧫), vvM48 (▴), or VSC8 (▪) intradermally at the base of the tail and 6 weeks later challenged intranasally with live RSV. Illness was monitored for 12 days following challenge. The data represent the mean and SEM for each group from four experiments (n = 20 for all groups). P > 0.05 at days 5 to 11 when VSC8-primed mice are compared to vvM48-primed mice and when vvWT G- and vvM48I-primed mice are compared. When vvM48-primed mice are compared to vvM48I-primed mice, P < 0.05 at each day except day 7 (where P = 0.07). Illness mirrored weight loss measurements (data not shown).
RSV replication following viral challenge.
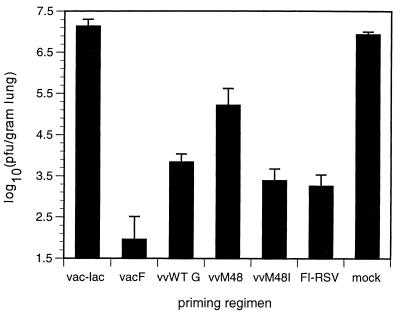
When RSV titers in the lungs were measured 4 days postchallenge, the degree of viral clearance differed among the priming groups (Fig. 2). VacF priming resulted in greater reduction of RSV titers in lung than vvWT G, confirming earlier observations (45). Reduction in RSV titers in lung was similar between vvWT G- and vvM48I-primed mice. In contrast, priming with vvM48 resulted in significantly less reduction in RSV titers than did vvWT G priming (P < 0.05, Tukey’s Studentized range test). Therefore, G is less immunogenic than F and secreted G appears to be less immunogenic than membrane-anchored G.
FIG. 2.
RSV titers in lungs of vaccine-primed mice. Mice were primed and challenged as for Fig. 1. On day 4 postchallenge, viral titers were measured by standard plaque assay on HEp-2 monolayers and are represented as the log10 (PFU/gram of lung) ± SEM. Data from a representative experiment (of four) are shown (n = 6 for FI-RSV and mock-primed groups; n = 5 for all other groups; P < 0.005).
In vivo production of G.
Antigen dose has been shown to influence T-cell differentiation (12, 29, 51). Thus, production of different concentrations of antigen by different virus vectors may potentially result in different profiles of immune responses following challenge. vvWT G, vvM48I, and vvM48 have been shown to produce similar levels of RSV G in vitro in tissue culture systems (52). However, due to immunoregulatory mechanisms, this may not be the case in vivo. Thus, we sought to confirm equal antigen production in vivo. Due to dissemination of vaccinia virus, a direct measure of RSV G concentrations in a given tissue was not considered to be an accurate representation of antigen production. Therefore, we measured the dissemination and titers of vvM48I and vvM48. Similar patterns of dissemination to spleen and equivalent viral titers in tail sections were observed at all time points examined (data not shown). To further evaluate replication of these two vaccinia virus constructs in vivo, neutralizing antibody titers were measured. vvM48I and vvM48 immunizations resulted in levels of neutralizing antibodies similar to those for vaccinia virus (data not shown). Production of equal amounts of G from vvM48I and vvM48 in vitro (52) suggests that in vivo levels of production of G antigen between vvM48I and vvM48 are similar.
Reconstitution with purified G during priming.
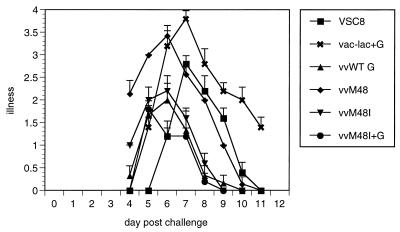
To determine whether enhanced disease and reduced viral clearance produced by vvWT G and vvM48 sensitization were indeed related to the presence of secreted G during priming, immunization protocols in which purified G was coadministered early in the vaccination schedule were designed. Secreted G was purified to homogeneity from vvM48-infected cell culture supernatant by chromatography on a ConA-Sepharose affinity column (data not shown). To simulate the conditions of priming with wild-type G, purified G was administered on days 0 to 3 to vvM48I-primed mice. The immunogenicity of G alone was examined by administration of purified protein during VSC8 priming. All mice were challenged with RSV 6 weeks after immunization, and illness and weight loss were monitored. The addition of purified G during VSC8 priming established immunologic conditions in which enhanced disease occurred following RSV challenge, as evidenced by increased illness and weight loss relative to VSC8-primed mice (Fig. 3 and data not shown). Coadministration of purified G during vvM48I immunization did not result in a significant change in either illness or weight loss compared to mice primed with vvWT G or vvM48I alone.
FIG. 3.
RSV-induced illness in mice primed with RSV G-vaccinia virus constructs and RSV G reconstitution. Mice were primed intradermally at the base of the tail with the indicated constructs and challenged intranasally with live RSV 6 weeks after priming; illness was measured daily. Data from a representative experiment (of three) are shown and represented as mean ± SEM (n = 5; P < 0.05 at days 4 to 12).
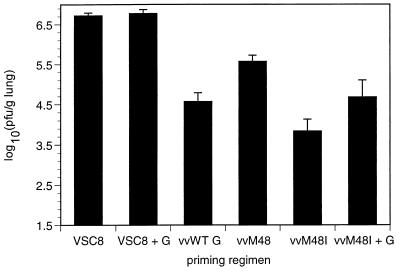
Clearance of RSV was also examined in mice primed with the recombinant vaccinia constructs and purified G. While purified G in combination with VSC8 priming resulted in enhanced disease, it did not reduce RSV titers after challenge compared to mice primed with VSC8 alone (Fig. 4). Mice immunized with vvM48I had significantly lower RSV titers in lung following challenge than mice primed with vvWT G or vvM48 (Fig. 4; P < 0.05, Tukey’s Studentized range test). However, in mice coimmunized with purified G and vvM48I, the RSV titers in lungs 4 days after RSV challenge were not significantly different from those in vvM48- or vvWT G-primed mice (P > 0.05). Thus, addition of purified G during vaccination alters the immunogenicity of vvM48I priming, decreasing viral clearance, suggesting secreted G actively influences the magnitude, specificity, or composition of the immune response.
FIG. 4.
RSV titers in lungs of mice primed with RSV G-vaccinia virus constructs and RSV G reconstitution. Mice were primed and challenged as for Fig. 3. On day 4 postchallenge, viral titers were measured by standard plaque assay on HEp-2 monolayers and are represented as the log10 (PFU/gram of lung) ± SEM. Data from a representative experiment (of three) are shown (n = 5 for all groups; P < 0.0001).
G-specific antibody isotype response.
Antibody titers in prechallenge sera of immunized mice were evaluated in a G capture ELISA. Detectable levels of immunoglobulin were measured in each priming group except mice primed with VSC8 (Fig. 5). Vaccination with regimens containing membrane-anchored G produced higher titers of antibody than did regimens containing only secreted G protein. Subsequent to RSV challenge, antibody titers were increased to similar levels in all groups (data not shown).
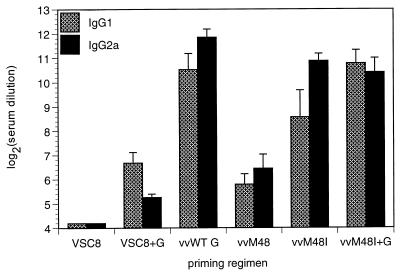
FIG. 5.
G-specific antibody titers and isotype profiles. Mice were primed and challenged as for Fig. 3. Serum samples were collected the day prior to challenge. Titers and isotypes of G-specific antibodies were measured in a capture ELISA by coating wells with purified G protein, adding dilutions of sera, and detecting bound IgG with horseradish peroxidase-conjugated anti-isotype antibodies. Prechallenge IgG1 and IgG2a were measured. Data from a representative experiment (of three) are represented as the mean ± SEM of the log2 (serum dilution) producing 0.1 OD450 unit for each group. When IgG1 and IgG2a titers were compared in each priming group, the following P values were obtained: >0.5 for VSC8, vvWT G, vvM48, and vvM48I plus G; 0.025 for VSC8 plus G; and 0.01 for vvM48I (Student’s t test; n = 5 for all groups but vvWT G and vvM48, where n = 6 and 7, respectively).
In mice primed with vvM48I, IgG2a antibodies were the dominant isotype generated (n = 5, P = 0.01, Student’s t test comparing IgG1 and IgG2a titers in vvM48I-primed mice). In vvWT G- and vvM48-primed mice, IgG2a were higher than IgG1 titers, but the ratio of IgG2a to IgG1 was lower. With the coadministration of secreted G during priming, a shift to IgG1-producing conditions was observed. In VSC8-primed mice which also received purified G, the major antibody response was of an IgG1 isotype (n = 5, P = 0.025 relative to IgG2a titers from the same priming group). Coadministration of secreted G during vvM48I immunization resulted in a shift from an IgG2a-dominated antibody response to a response in which IgG1 and IgG2a antibody titers were about equal. The IgG1/IgG2a ratios in vvM48I-primed mice were significantly different from the IgG1/IgG2a ratios in mice primed with vvM48I plus G (P = 0.04, Student’s t test comparing IgG1/IgG2a ratios [data not shown]). Thus, addition of secreted G to the priming environment can induce a significant shift away from an IgG2a-dominated antibody response. These data suggest that the presence of secreted G during priming reinforces the tendency for G-specific responses to produce IgG1 antibodies.
Cytokine production in the lungs.
The concentrations of IL-5 and IFN-γ in the lung supernatants from mice 4 and 8 days after RSV challenge were measured by ELISA. Both IL-5 and IFN-γ could be measured in day 4 samples and are shown in Table 2. Little IL-5 was detectable in day 8 lung supernatants, and IFN-γ levels were greatly reduced (data not shown). Little IL-5 was measured in the lung supernatants of mice immunized with VSC8 (Table 2). Highest concentrations of IL-5 were found in mice primed with vvM48I plus G and vvM48. Statistically significant differences were found when IL-5 levels in vvWT G-primed mice were compared to those in either vvM48-primed mice or vvM48I-plus-G-primed mice (P > 0.0001, Student’s t test). Similarly, compared to vvM48I-plus-G-primed mice, the IL-5 levels in vvM48I-primed mice (P = 0.01) and in vvM48-primed mice (P = 0.004) were significantly different. Mice primed with vvWT G and VSC8 plus G had intermediate IL-5 levels, and vvM48I-primed mice had the lowest levels, although the differences between the three groups were not statistically significant (P > 0.05, Student’s t test). Thus, increased IL-5 levels were correlated with priming regimens that included secreted G and paralleled the titers of G-specific IgG1.
TABLE 2.
Quantitative measures of lung immunopathology in primed mice after RSV challengea
| Priming regimen | % Eosinophil in BAL fluidb | Concn (mean ± SEM; n = 5)
|
|
|---|---|---|---|
| IL-5 (pg/ml)c | IFN-γ (ng/ml) | ||
| VSC8 | 0.0 ± 0.0 | 10.4 ± 2.1 | 17.8 ± 3.8 |
| VSC8 + G | 8.3 ± 3.7 | 21.9 ± 3.5 | 5.1 ± 0.3 |
| vvWT G | 5.9 ± 2.6 | 22.3 ± 5.1 | 10.5 ± 2.9 |
| vvM48 | 7.6 ± 3.4 | 104.4 ± 5.4 | 27.5 ± 4.4 |
| vvM48I | 1.9 ± 0.9 | 15.1 ± 6.5 | 31.4 ± 2.5 |
| vvM48I + G | 1.3 ± 0.6 | 64.0 ± 3.4 | 15.7 ± 2.7 |
The mice were primed and challenged as for Fig. 3. Percentage of eosinophils in BAL fluid was determined by counting Diff-Quik-stained smears. Concentrations of IL-5 and IFN-γ were measured in day 4 (shown) and day 8 (not shown) lung supernatants by ELISA. Limits of detection are 10 pg/ml for IL-5 and 50 pg/ml for IFN-γ.
Differences between VSC8, vvWT G, and vvM48 were not statistically significant by Student’s t test, nor were the differences between vvM48I and vvM48 plus G. However, the differences between the mice primed with vvM48I (or vvM48I plus G) and the mice primed with VSC8 plus G, vvWT G, or vvM48 were significant (P < 0.05 for all comparisons).
Statistically significant differences were found as follows: vvM48I compared to vvM48I plus G (P = 0.01), and vvM48 compared to all other groups (P < 0.025). Differences between VSC8 and VSC8 plus G, between vvWT G and vvM48I, and between vvWT G and VSC8 plus G were not significant.
Lung pathology following RSV challenge.
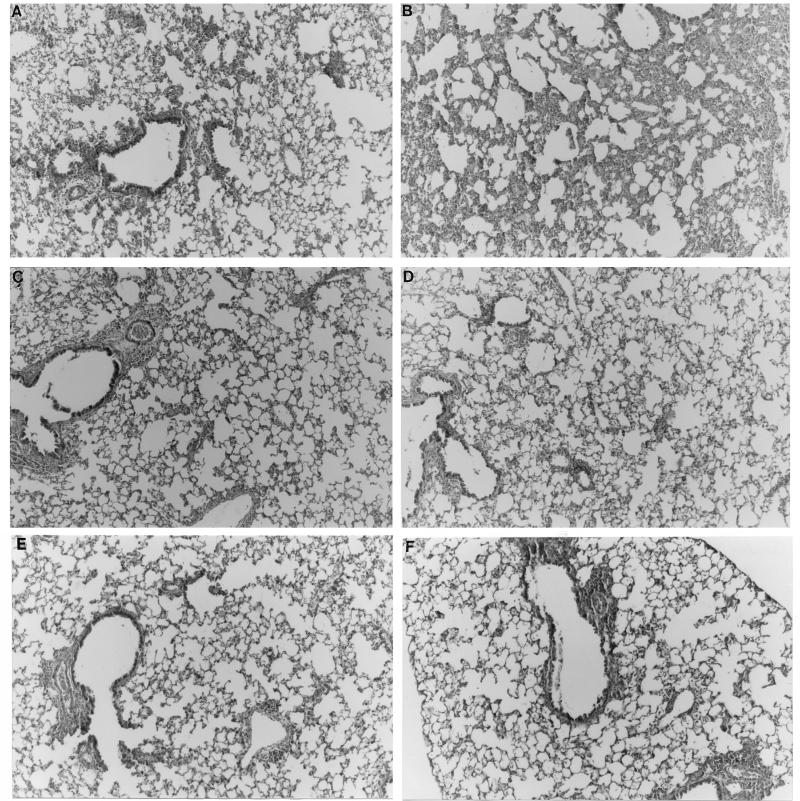
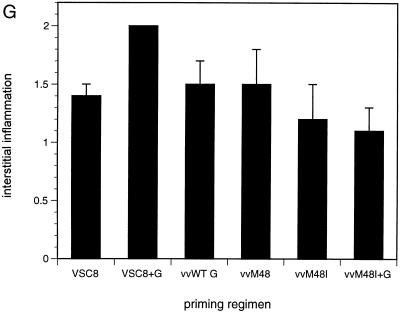
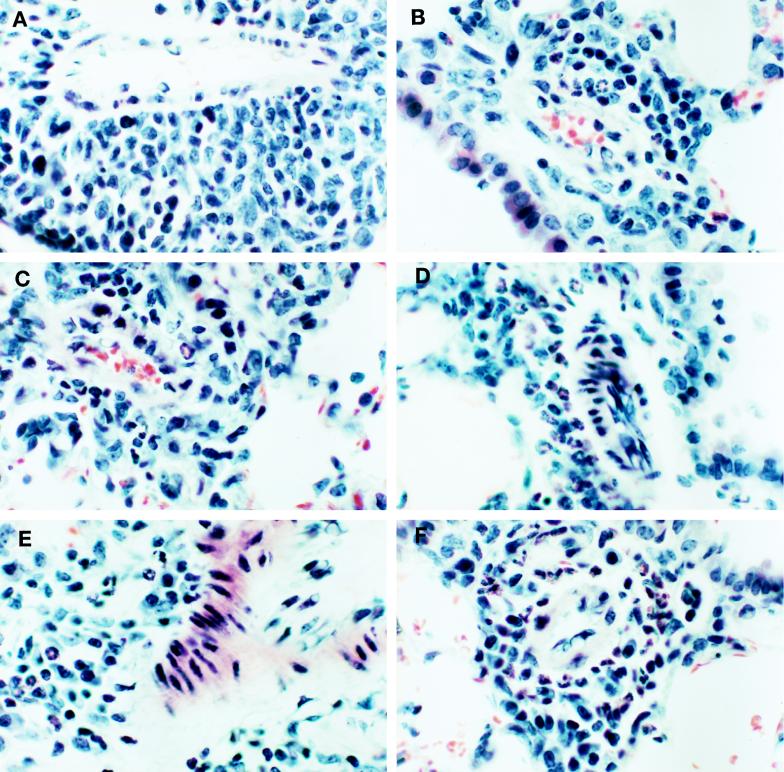
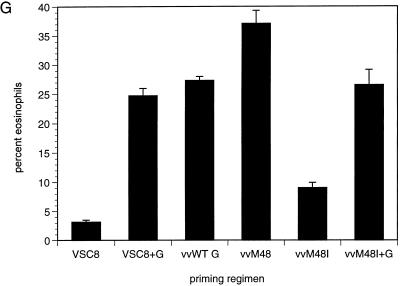
The alveolar inflammation induced by priming with the different forms of G protein was evaluated (Fig. 6 and 7). In mice immunized with VSC8, vvWT G, or vvM48, moderate alveolar mononuclear infiltration occurred upon RSV challenge (Fig. 6A, C, and D, respectively). In mice primed with vvWT G and vvM48, a significant eosinophil component was present in the infiltrates around arteries (Fig. 7C, D, and G). Although vvM48I priming reduced the density of infiltration (Fig. 6E), small numbers of eosinophils were still detected focally (Fig. 7E and G). When G was coadministered during VSC8 priming, the pathology resulting from challenge was more severe (Fig. 6B and G) and induced prominent eosinophilia (Fig. 7F and G), absent in mice immunized with VSC8 alone (Fig. 7A). The addition of secreted G during vvM48I priming did not significantly increase the severity of lymphocytic infiltration (Fig. 6F and G); however, this priming regimen did result in the recruitment of abundant eosinophils into the periarterial infiltrates (Fig. 7F and G). When the degree of eosinophilia was quantitatively examined (Fig. 7G), similar percentages of eosinophils were found in the lungs of mice primed with VSC8 plus G, vvWT G, and vvM48I plus G (P > 0.1, Student’s t test). The addition of secreted G to vvM48I priming resulted in significantly greater percentage of eosinophils infiltrating the lung (P = 0.002, vvM48I compared to vvM48I plus G). Similar increases were found with the addition of secreted G to VSC8 priming (P < 0.0001). Thus, the presence of G-specific IgG1 titers and IL-5 content in lung correlated with the amount of tissue eosinophilia.
FIG. 6.
Lung histopathology. Mice were primed with VSC8 (A), VSC8 plus G (B), vvWT G (C), vvM48 (D), vvM48I (E), or vvM48I plus G (F) and challenged as for Fig. 3. Eight days following challenge, lung histopathology was evaluated by staining formalin-fixed lung sections with hematoxylin and eosin (original magnification, ×25). Day 8 lungs were examined and graded for severity and composition of alveolar inflammation on a semiquantitative scale (G). No statistical difference was found among any group except the VSC8-plus-G group, in which inflammation was significantly higher than in any other group (P < 0.03, Student’s t test, n = 5).
FIG. 7.
Tissue eosinophilia. Mice were primed with VSC8 (A), VSC8 plus G (B), vvWT G (C), vvM48 (D), vvM48I (E), or vvM48I plus G (F) and challenged as for Fig. 3. Eight days following challenge, lung eosinophilia was evaluated by staining tissue sections with Luna’s stain (original magnification, ×250). Tissue eosinophilia was quantitatively measured by counting the percentages of eosinophils in Luna-stained formalin-fixed day 8 lungs (G; n = 5). Differences between VSC8 plus G, vvWT G, and vvM48I plus G were not statistically significant. vvM48-primed mice were significantly different from all other groups (P < 0.05). P = 0.002 for vvM48I compared to vvM48I plus G; P < 0.0001 for VSC8 compared to VSC8 plus G and for vvWT G compared to vvM48I.
Cellular composition of BAL infiltrate after RSV challenge.
To determine whether infiltration of cells into the air space differed from that seen in the periarterial regions and to quantitate the cellular components of the infiltrate, BAL was performed on day 6 following challenge (Table 2). The greatest number of eosinophils occurred in mice primed with VSC8 plus purified G and with vvM48, and slightly fewer eosinophils were obtained in BAL of vvWT G-primed mice, although no statistically significant difference was detected between these groups. Significantly fewer eosinophils were recruited into the bronchial airways of mice immunized with vvM48I with or without purified G (P < 0.05 relative to vvWT G or vvM48), contrasting with the increased interstitial eosinophilia observed with coadministration of secreted G during vvM48I priming (Table 2 and Fig. 7F). Therefore, groups with a high degree of BAL eosinophilia also had increased illness and weight loss. Although the degree of BAL eosinophilia generally corresponded to tissue eosinophilia, mice primed with vvM48I plus G had a higher frequency of tissue eosinophilia yet few eosinophils in BAL.
DISCUSSION
These data suggest that the presence of the secreted form of RSV G during initial antigen presentation (and not solely the primary or secondary antigenic structure) affects the composition of immune responses to RSV infection. Previous work has shown that immunization with RSV G results in more severe disease and lung pathology following RSV challenge (25, 61). The molecular basis for this alteration in the immune response has not been defined. The unique nature of G provides several attributes which may generate the altered immune response to RSV challenge. First, G is extensively glycosylated: >50% of the weight of the mature protein is contributed by carbohydrate residues, attached predominantly via O-glycosidic bonds (70). Also, G contains a high proportion (30.6%) of serine and threonine residues which serve as acceptors for the O-linked oligosaccharides. Third, G protein lacks homology to any other known paramyxovirus protein. The high proline content of 10.1% has more in common with mucinous glycoproteins than viral glycoproteins (31, 39, 70). G exhibits neither hemagglutinating nor neuraminidase activities (22) and is also unusual in that it lacks both a hydrophobic NH2-terminal signal sequence and a hydrophobic COOH-terminal domain, features present in RSV F and other paramyxovirus proteins (70).
In contrast to previously published studies using recombinant vaccinia virus expressing wild-type RSV G (25, 60, 61), mice primed with recombinant vaccinia virus expressing only membrane-anchored G are partially protected during RSV challenge, with reduced viral titers observed, although illness patterns are similar. However, in the absence of membrane-anchored G, secreted G increases illness and decreases viral clearance. We have shown that priming with secreted G (for both vvWT G and vvM48) is associated with increased IL-5 production and more severe immunopathology, including increased tissue eosinophilia. However, illness correlates with alveolar infiltration of eosinophils, as determined by their presence in BAL, and suggests that secreted G induces factors other than IL-5 which may be responsible for a second step in the activation and migration of eosinophils. These data suggest the secreted form of RSV G modulates the composition of the immune response by inducing IL-5 production and altering patterns of leukocyte trafficking, while the membrane-anchored form of G may have a separate and dominant immunoregulatory effect on factors producing illness.
Several immunoregulatory mechanisms have been described for soluble antigen. Soluble antigen has been shown to regulate the development of germinal center B cells by selectively inducing apoptosis of high-affinity antigen-specific B lymphocytes (50, 56). This ability to direct B-cell differentiation is hypothesized to indicate clonal deletion of self-reactive B cells and, thus, be a mechanism of avoiding autoimmune disease. Soluble antigen will be processed and presented in the context of major histocompatibility (MHC) class II molecules. MHC context of antigen presentation (41), the type of antigen-presenting cell used (11, 54), and the presence of different costimulatory molecules (13, 55) have been shown to regulate the development and maturation of T-cell subsets. Antigen dose may also be a determinant in T-cell differentiation. In most systems, immunization with large doses of antigen induces effector cells exhibiting a type 1 cytokine profile, whereas lower concentrations of priming antigen tend to generate type 2 effector cells (12, 29). However, some antigens have the reversed profile, with high doses favoring development of Th2 cells and low doses inducing Th1 cells (51). Thus, the induction of severe disease by the presence of soluble G during priming suggests that either the concentration or processing of G or its subsequent recognition by selected T-cell subsets may be a key determinant regulating immune responses following subsequent RSV challenge.
Infectious organisms ensure their survival by developing mechanisms to avoid detection or subvert immune responses. Some strategies include production of cytokine receptors by the organism (30) or blocking induction of the immune response at the level of antigen presentation (14, 28). A body of evidence which suggests that induction of Th2-mediated immune responses provides a survival advantage for many intracellular pathogens, including viruses, is also accumulating (13, 21). For example, measles virus suppresses cell-mediated immunity by down-regulation of IL-12 expression through signals mediated by measles virus binding CD46 (33). Also, disease progression and infection with human immunodeficiency virus have been associated with Th2-dominated responses (16, 37). Human immunodeficiency virus infection has been shown to suppress IL-2, IL-12, and IFN-γ, but not tumor necrosis factor alpha or IL-6, induction by Toxoplasma gondii (15). Thus, the induction of a type 2 T-cell response by secreted RSV G may serve as a defense mechanism the virus has evolved to favorably modulate immune responses.
Previous work has established priming or infection with G-expressing vectors results in increased illness and pathology (61). The ability of RSV G to produce cytokines associated with a Th2 response has been clearly demonstrated (2, 4, 25). In this and other studies, FI-RSV priming has also been shown to induce Th2-type cytokine profiles in mice (19, 62, 69), thereby associating this type of response with the RSV vaccine-enhanced illness seen in children in FI-RSV vaccine studies in the 1960s (32, 34). In this study, we have addressed whether it is the primary or secondary antigen structure of G, or the presence of secreted G or antigens with obligate processing through the endocytic pathway and MHC class II expression, which increases disease severity. The IgG2a isotype antibodies and disease protection observed in mice primed with membrane-anchored G and the IgG1 isotype antibodies and more severe illness induced by immunization with secreted G imply that different Th1- and Th2-associated cytokine profiles may be induced by priming without and with secreted G, respectively. We therefore propose that the link between RSV G immunization, FI-RSV-induced vaccine-enhanced illness, and induction of type 2 cytokine profiles is not induced by G itself but rather results from the initial presentation of G to the immune system in secreted form. This might then promote the induction of Th2 CD4+ T-cell differentiation in part by precluding intracellular antigen processing, presentation of antigen by MHC class I molecules, and loss of the early contribution of CD8+ T cells to the cytokine milieu (17, 59).
Evidence suggests that full activation of eosinophils requires a two-step process involving IL-5, eotaxin, or other cofactors (42, 53). Extracellular matrix proteins (36) and VLA-4 on eosinophils (44) may also be involved in eosinophil recruitment and infiltration. These observations may be invoked to explain the differential patterns of eosinophilia and disease severity induced by priming with membrane-anchored and secreted G. Our data suggest the hypothesis that immunization with any form of G (membrane anchored or secreted) may recruit eosinophils upon challenge via the induction of IL-5, but the presence of secreted G is required for inducing the activation associated with movement of eosinophils into alveolar spaces to produce more severe disease.
The data presented in this paper suggest that priming with secreted RSV G promotes eosinophil recruitment associated with IL-5 production and that illness is associated with the movement of eosinophils from the interstitial compartment in the lung to the alveoli. We propose the secretion of G from RSV-infected cells represents a strategy to modulate and evade effective immune responses. The presence of a small number of alveolar eosinophils in mice primed with membrane-anchored G suggests a direct effect of the primary or secondary antigenic structure of G on the induction of type 2 immune responses that is compounded by its secretion as a soluble protein, resulting in increased eosinophilia. Understanding how secreted G interacts with different elements of the innate and adaptive immune system might provide insight into basic mechanisms of clearance for RSV and other viruses.
ACKNOWLEDGMENTS
This work was supported by National Institutes of Health grants RO1-AI-37216 (B.S.G.) and RO1-AI-20181 (G.W.W.).
We thank Rauf Kuli-Zade and Peter Veldkamp for technical assistance and James E. Crowe, Jr., for editorial comments.
REFERENCES
- 1.Alwan W, Kozlowska W, Openshaw P. Distinct types of lung disease caused by functional subsets of antiviral T cells. J Exp Med. 1994;179:81–89. doi: 10.1084/jem.179.1.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Alwan W, Openshaw P. Distinct patterns of T- and B-cell immunity to respiratory syncytial virus induced by individual viral proteins. Vaccine. 1993;11:431–437. doi: 10.1016/0264-410x(93)90284-5. [DOI] [PubMed] [Google Scholar]
- 3.Alwan W, Record F, Openshaw P. CD4+ T cells clear virus but augment disease in mice infected with respiratory syncytial virus: comparison with the effects of CD8+ T cells. Clin Exp Immunol. 1992;88:527–536. doi: 10.1111/j.1365-2249.1992.tb06482.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Alwan W, Record F, Openshaw P. Phenotypic and functional characterization of T cell lines specific for individual respiratory syncytial virus proteins. J Immunol. 1993;150:5211–5218. [PubMed] [Google Scholar]
- 5.Anderson L, Heilman C. Protective and disease-enhancing immune response to respiratory syncytial virus. J Infect Dis. 1995;171:1–7. doi: 10.1093/infdis/171.1.1. [DOI] [PubMed] [Google Scholar]
- 6.Ball L, Young K, Anderson K, Collins P, Wertz G. Expression of the major glycoprotein G of human respiratory syncytial virus from recombinant vaccinia virus vectors. Proc Natl Acad Sci USA. 1986;83:246–250. doi: 10.1073/pnas.83.2.246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bangham C, Cannon M, Karzon D, Askonas B. Cytotoxic T-cell response to respiratory syncytial virus in mice. J Virol. 1985;56:55–59. doi: 10.1128/jvi.56.1.55-59.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bangham C, Openshaw P, Ball A, King A, Wertz G, Askonas B. Human and murine cytotoxic T cells specific to respiratory syncytial virus recognize the viral nucleoprotein (N), but not the major glycoprotein (G), expressed by vaccinia virus recombinants. J Immunol. 1986;137:3973–3977. [PubMed] [Google Scholar]
- 9.Chakrabarti S, Brechling K, Moss B. Vaccinia virus expression vector: coexpression of β-galactosidase provides visual screening of recombinant virus plaques. Mol Cell Biol. 1985;5:3403–3409. doi: 10.1128/mcb.5.12.3403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Connors M, Collins P, Firestone C, Sotnikov A, Waitze A, Davis A, Hung P, Chanock R, Murphy B. Cotton rats previously immunized with a chimeric RSV FG glycoprotein develop enhanced pulmonary pathology when infected with RSV, a phenomenon not encountered following immunization with vaccinia-RSV recombinants or RSV. Vaccine. 1992;10:475–484. doi: 10.1016/0264-410x(92)90397-3. [DOI] [PubMed] [Google Scholar]
- 11.Conradt P, Kaufmann S. Impact of antigen-presenting cells on cytokine profiles of human Th clones established after stimulation with Mycobacterium tuberculosis antigens. Infect Immun. 1995;63:2079–2081. doi: 10.1128/iai.63.5.2079-2081.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Constant S, Pfeiffer C, Woodard A, Pasqualini T, Bottomly K. Extent of T cell receptor ligation can determine the functional differentiation of naive CD4+ T cells. J Exp Med. 1995;182:1591–1596. doi: 10.1084/jem.182.5.1591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Corry D, Reiner S, Linsley P, Locksley R. Differential effects of blockade of CD28-B7 on the development of Th1 or Th2 effector cells in experimental leishmaniasis. J Immunol. 1994;153:4142–4148. [PubMed] [Google Scholar]
- 14.Fruh K, Ahn K, Djaballah H, Sempe P, van Endert P M, Tampe R, Peterson P A, Yang Y. A viral inhibitor of peptide transporters for antigen presentation. Nature. 1995;375:415–418. doi: 10.1038/375415a0. [DOI] [PubMed] [Google Scholar]
- 15.Gazzinelli R T, Baia S, Stevens R, Baseler M, Wahl L, Kovacs J, Sher A. HIV infection suppresses type 1 lymphokine and IL-12 responses to Toxoplasma gondii but fails to inhibit the synthesis of other parasite-induced monokines. J Immunol. 1995;155:1565–1574. [PubMed] [Google Scholar]
- 16.Gazzinelli R T, Makino M, Chattopadhyay S K, Snapper C M, Sher A, Hugin A W, Morse H C. CD4+ subset regulation in viral infection. Preferential activation of Th2 cells during progression of retrovirus-induced immunodeficiency in mice. J Immunol. 1992;148:182–188. [PubMed] [Google Scholar]
- 17.Graham B S. Immunological determinants of disease caused by respiratory syncytial virus. Trends Microbiol. 1996;4:290–294. doi: 10.1016/0966-842x(96)10032-9. [DOI] [PubMed] [Google Scholar]
- 18.Graham B. Pathogenesis of respiratory syncytial virus vaccine-augmented pathology. Am J Respir Crit Care Med. 1995;152:563–566. doi: 10.1164/ajrccm/152.4_Pt_2.S63. [DOI] [PubMed] [Google Scholar]
- 19.Graham B, Henderson G, Tang Y, Lu X, Neuzil K, Colley D. Priming immunization determines T helper cytokine mRNA expression patterns in lungs of mice challenged with respiratory syncytial virus. J Immunol. 1993;151:2032–2040. [PubMed] [Google Scholar]
- 20.Graham B, Perkins M, Wright P, Karzon D. Primary respiratory syncytial virus infection in mice. J Med Virol. 1988;26:153–162. doi: 10.1002/jmv.1890260207. [DOI] [PubMed] [Google Scholar]
- 21.Griffin D E, Ward B J. Differential CD4 T cell activation in measles. J Infect Dis. 1993;168:275–281. doi: 10.1093/infdis/168.2.275. [DOI] [PubMed] [Google Scholar]
- 22.Gruber C, Levine S. Respiratory syncytial virus polypeptides. III. The envelope-associated proteins. J Gen Virol. 1983;64:825–832. doi: 10.1099/0022-1317-64-4-825. [DOI] [PubMed] [Google Scholar]
- 23.Hall C, Walsh E, Long C, Schnabel K. Immunity to and frequency of reinfection with respiratory syncytial virus. J Infect Dis. 1991;163:693–698. doi: 10.1093/infdis/163.4.693. [DOI] [PubMed] [Google Scholar]
- 24.Hall W, Martin S. Purification and characterization of measles virus. J Gen Virol. 1973;19:175–188. doi: 10.1099/0022-1317-19-2-175. [DOI] [PubMed] [Google Scholar]
- 25.Hancock G, Speelman D, Heers K, Bortell E, Smith J, Cosco C. Generation of atypical pulmonary inflammatory responses in BALB/c mice after immunization with the native attachment (G) glycoprotein of respiratory syncytial virus. J Virol. 1996;70:7783–7791. doi: 10.1128/jvi.70.11.7783-7791.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hendricks D, Baradaran K, McIntosh K, Patterson J. Appearance of a soluble form of the G protein of respiratory syncytial virus in fluids of infected cells. J Gen Virol. 1987;68:1705–1714. doi: 10.1099/0022-1317-68-6-1705. [DOI] [PubMed] [Google Scholar]
- 27.Hendricks D, McIntosh K, Patterson J. Further characterization of the soluble form of the G glycoprotein of respiratory syncytial virus. J Virol. 1988;62:2228–2233. doi: 10.1128/jvi.62.7.2228-2233.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hill A, Jugovic P, York I, Russ G, Bennicnk J, Yewdell J, Ploegh H, Johnson D. Herpes simplex virus turns off the TAP to evade host immunity. Nature. 1995;375:411–415. doi: 10.1038/375411a0. [DOI] [PubMed] [Google Scholar]
- 29.Hosken N A, Shibuya K, Heath A W, Murphy K M, O’Garra A. The effect of antigen dose on CD4+ T helper cell phenotype development in a T cell receptor-alpha beta-transgenic model. J Exp Med. 1995;182:1579–1584. doi: 10.1084/jem.182.5.1579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hu F Q, Smith C A, Pickup D J. Cowpox virus contains two copies of an early gene encoding a soluble secreted form of the type II TNF receptor. Virology. 1994;204:343–356. doi: 10.1006/viro.1994.1539. [DOI] [PubMed] [Google Scholar]
- 31.Johnson P, Spriggs M, Olmstead R, Collins P. The G glycoprotein of human respiratory syncytial viruses of subgroups A and B: extensive sequence divergence between antigenically related proteins. Proc Natl Acad Sci USA. 1987;84:5625–5629. doi: 10.1073/pnas.84.16.5625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kapikian A, Mitchell R, Chanock R, Shvedoff R, Stewart C. An epidemiologic study of altered clinical reactivity to respiratory syncytial (RS) virus infection in children previously vaccinated with an inactivated RS virus vaccine. Am J Epidemiol. 1969;89:405–421. doi: 10.1093/oxfordjournals.aje.a120954. [DOI] [PubMed] [Google Scholar]
- 33.Karp C L, Wysocka M, Wahl L M, Ahearn J M, Cuomo P J, Sherry B, Trinchieri G, Griffin D E. Mechanism of suppression of cell-mediated immunity by measles virus. Science. 1996;273:228–231. doi: 10.1126/science.273.5272.228. [DOI] [PubMed] [Google Scholar]
- 34.Kim H W, Canchola J G, Brandt C D, Pyles G, Chanock R M, Jensen K, Parrott R H. Respiratory syncytial virus disease in infants despite prior administration of antigenic inactivated vaccine. Am J Epidemiol. 1969;89:422–434. doi: 10.1093/oxfordjournals.aje.a120955. [DOI] [PubMed] [Google Scholar]
- 35.Kim H, Leikin S, Arrobio J, Brandt C, Chanock R, Parrott R. Cell-mediated immunity to respiratory syncytial virus induced by inactivated vaccine or by infection. Pediatr Res. 1976;10:75–78. doi: 10.1203/00006450-197601000-00015. [DOI] [PubMed] [Google Scholar]
- 36.Kita H, Horie S, Gleich G J. Extracellular matrix proteins attenuate activation and degranulation of stimulated eosinophils. J Immunol. 1996;156:1174–1181. [PubMed] [Google Scholar]
- 37.Kurup V P, Seymour B W, Choi H, Coffman R L. Particulate Aspergillus fumigatus antigens elicit a TH2 response in BALB/c mice. J Allergy Clin Immunol. 1994;93:1013–1020. doi: 10.1016/s0091-6749(94)70050-8. [DOI] [PubMed] [Google Scholar]
- 38.Lambert D. Role of oligosaccharides in the structure and function of respiratory syncytial virus glycoproteins. Virology. 1988;16:458–466. doi: 10.1016/0042-6822(88)90560-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Langedijk J, Schaaper W, Meloen R, van Oirschot J. Proposed three-dimensional model for the attachment protein G of respiratory syncytial virus. J Gen Virol. 1996;77:1249–1257. doi: 10.1099/0022-1317-77-6-1249. [DOI] [PubMed] [Google Scholar]
- 40.Levine S, Franco-Klaiber R, Paradiso P. Demonstration that glycoprotein G is the attachment protein of respiratory syncytial virus. J Gen Virol. 1987;68:2521–2524. doi: 10.1099/0022-1317-68-9-2521. [DOI] [PubMed] [Google Scholar]
- 41.Milich D, Peterson D, Schodel F, Jones J, Hughes J. Preferential recognition of hepatitis B nucleocapsid antigens by Th1 or Th2 cells is epitope and major histocompatibility complex dependent. J Virol. 1995;69:2776–2785. doi: 10.1128/jvi.69.5.2776-2785.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Mould A W, Matthaei K I, Young I G, Foster P S. Relationship between interleukin-5 and eotaxin in regulating blood and tissue eosinophilia in mice. J Clin Invest. 1997;99:1064–1071. doi: 10.1172/JCI119234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Murphy B, Graham B, Prince G, Walsh E, Chanock R, Karzon D, Wright P. Serum and nasal-wash immunoglobulin G and A antibody response of infants and children to respiratory syncytial virus F and G glycoproteins following primary infection. J Clin Microbiol. 1986;23:1009–1014. doi: 10.1128/jcm.23.6.1009-1014.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Nakajima H, Sano H, Nishimura T, Yoshida S, Iwamoto I. Role of vascular cell adhesion molecule 1/very late activation antigen 4 and intercellular adhesion molecule 1/lymphocyte function-associated antigen 1 interactions in antigen-induced eosinophil and T cell recruitment into the tissue. J Exp Med. 1994;179:1145–1154. doi: 10.1084/jem.179.4.1145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Olmsted R, Elango N, Prince G, Murphy B, Johnson P, Moss B, Chanock R, Collins P. Expression of the F glycoprotein of respiratory syncytial virus by a recombinant vaccinia virus: comparison of the individual contributions of the F and G glycoproteins to host immunity. Proc Natl Acad Sci USA. 1986;83:7462–7466. doi: 10.1073/pnas.83.19.7462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Openshaw P, Clarke S, Record F. Pulmonary eosinophilic response to respiratory syncytial virus infection in mice sensitized to the major surface glycoprotein G. Int Immunol. 1992;4:493–500. doi: 10.1093/intimm/4.4.493. [DOI] [PubMed] [Google Scholar]
- 47.Palomo C, Garcia-Barreno B, Penas C, Melero J. The G protein of human respiratory syncytial virus: significance of carbohydrate side-chains and the C-terminal end to its antigenicity. J Gen Virol. 1991;72:669–675. doi: 10.1099/0022-1317-72-3-669. [DOI] [PubMed] [Google Scholar]
- 48.Pemberton R, Cannon M, Openshaw P, Ball L, Wertz G, Askonas B. Cytotoxic T cell specificity for respiratory syncytial virus proteins: fusion protein is an important target antigen. J Gen Virol. 1987;68:2177–2182. doi: 10.1099/0022-1317-68-8-2177. [DOI] [PubMed] [Google Scholar]
- 49.Prince G, Jenson A, Hemming V, Murphy B, Walsh E, Horswood R, Chanock R. Enhancement of respiratory syncytial virus pulmonary pathology in cotton rats by prior intramuscular inoculation of formalin-inactivated virus. J Virol. 1986;57:721–728. doi: 10.1128/jvi.57.3.721-728.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Pulendran B, Kannourakis G, Nouri S, Smith K, Nossal G. Soluble antigen can cause enhanced apoptosis of germinal-centre B cells. Nature. 1995;375:331–334. doi: 10.1038/375331a0. [DOI] [PubMed] [Google Scholar]
- 51.Rizzo L V, DeKruyff R H, Umetsu D T, Caspi R R. Regulation of the interaction between Th1 and Th2 T cell clones to provide help for antibody production in vivo. Eur J Immunol. 1995;25:708–716. doi: 10.1002/eji.1830250312. [DOI] [PubMed] [Google Scholar]
- 52.Roberts S, Lichtenstein D, Ball L, Wertz G. The membrane-associated and secreted forms of the respiratory syncytial virus attachment glycoprotein G are synthesized from alternative initiation codons. J Virol. 1994;68:4538–4546. doi: 10.1128/jvi.68.7.4538-4546.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Rothenberg M E, Ownbey R, Mehlhop P D, Loiselle P M, van de Rijn M, Bonventre J V, Oettgen H C, Leder P, Luster A D. Eotaxin triggers eosinophil-selective chemotaxis and calcium flux via a distinct receptor and induces pulmonary eosinophilia in the presence of interleukin 5 in mice. Mol Med. 1996;2:334–348. [PMC free article] [PubMed] [Google Scholar]
- 54.Schmitz J, Assenmacher M, Radbruch A. Regulation of T helper cell cytokine expression: functional dichotomy of antigen-presenting cells. Eur J Immunol. 1993;23:191–199. doi: 10.1002/eji.1830230130. [DOI] [PubMed] [Google Scholar]
- 55.Shanafelt M, Soderberg C, Allsup A, Adelman D, Peltz G, Lahesmaa R. Costimulatory signals can selectively modulate cytokine production by subsets of CD4+ T cells. J Immunol. 1995;154:1684–1690. [PubMed] [Google Scholar]
- 56.Shokat K, Goodnow C. Antigen-induced B-cell death and elimination during germinal-centre immune responses. Nature. 1995;375:334–338. doi: 10.1038/375334a0. [DOI] [PubMed] [Google Scholar]
- 57.Simard C, Nadon F, Seguin C, Trudel M. Evidence that the amino acid region 124-203 of glycoprotein G from the respiratory syncytial virus (RSV) constitutes a major part of the polypeptide domain that is involved in the protection against RSV infection. Antiviral Res. 1995;28:303–315. doi: 10.1016/0166-3542(95)00053-4. [DOI] [PubMed] [Google Scholar]
- 58.Srikiatkhachorn A, Braciale T. Virus-specific memory and effector T lymphocytes exhibit different cytokine responses to antigens during experimental murine respiratory syncytial virus infection. J Virol. 1997;71:678–685. doi: 10.1128/jvi.71.1.678-685.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Stott E, Ball L, Young K, Furze J, Wertz G. Human respiratory syncytial virus glycoprotein G expressed from a recombinant vaccinia virus vector protects mice against live-virus challenge. J Virol. 1986;60:607–613. doi: 10.1128/jvi.60.2.607-613.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Stott E, Taylor G, Ball L, Anderson K, Young K, King A, Wertz G. Immune and histopathological responses in animals vaccinated with recombinant vaccinia viruses that express individual genes of human respiratory syncytial virus. J Virol. 1987;61:3855–3861. doi: 10.1128/jvi.61.12.3855-3861.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Tang Y, Graham B. Anti-IL-4 treatment at immunization modulates cytokine expression, reduces illness, and increases cytotoxic T lymphocyte activity in mice challenged with respiratory syncytial virus. J Clin Invest. 1994;94:1953–1958. doi: 10.1172/JCI117546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Taylor G, Stott E, Bew M, Fernie B, Cote P, Collins A, Hughes M, Jebbett J. Monoclonal antibodies protect against respiratory syncytial virus infection in mice. Immunology. 1984;52:137–142. [PMC free article] [PubMed] [Google Scholar]
- 63.Wagner D, Muelenaer P, Henderson F, Snyder M, Reimer C, Walsh E, Anderson L, Nelson D, Murphy B. Serum immunoglobulin G antibody subclass response to respiratory syncytial virus F and G glycoproteins after first, second, and third infections. J Clin Microbiol. 1989;27:589–592. doi: 10.1128/jcm.27.3.589-592.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Wagner D, Graham B, Wright P, Walsh E, Kim H, Reimer C, Nelson D, Chanock R, Murphy B. Serum immunoglobulin G antibody subclass responses to respiratory syncytial virus F and G glycoproteins after primary infection. J Clin Microbiol. 1986;24:304–306. doi: 10.1128/jcm.24.2.304-306.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Walsh E, Hall C, Briselli M, Brandiss M, Schlesinger J. Immunization with glycoprotein subunits of respiratory syncytial virus to protect cotton rats against viral infection. J Infect Dis. 1987;155:1198–1204. doi: 10.1093/infdis/155.6.1198. [DOI] [PubMed] [Google Scholar]
- 66.Walsh E, Hall C, Briselli M, Brandiss M, Schlesinger J. Immunization with glycoprotein subunits of respiratory syncytial virus protect cotton rats against viral infection. J Infect Dis. 1987;155:1198–1204. doi: 10.1093/infdis/155.6.1198. [DOI] [PubMed] [Google Scholar]
- 67.Walsh E, Schlesinger J, Brandiss M. Purification and characterization of GP90, one of the envelope glycoproteins of respiratory syncytial virus. J Gen Virol. 1984;65:761–767. doi: 10.1099/0022-1317-65-4-761. [DOI] [PubMed] [Google Scholar]
- 68.Ward K, Lambden P, Oglivie M, Watt P J. Antibodies to respiratory syncytial virus polypeptides and their significance in human infection. J Gen Virol. 1983;64:1867–1876. doi: 10.1099/0022-1317-64-9-1867. [DOI] [PubMed] [Google Scholar]
- 69.Waris M, Tsou C, Erdman D, Zaki S, Anderson L. Respiratory syncytial virus infection in BALB/c mice previously immunized with formalin-inactivated virus induces enhanced pulmonary inflammatory response with a predominant Th2-like cytokine pattern. J Virol. 1996;70:2852–2860. doi: 10.1128/jvi.70.5.2852-2860.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Wertz G, Collins P, Huang Y, Gruber C, Levine S. Nucleotide sequence of the G protein gene of human respiratory syncytial virus reveals an unusual type of viral membrane protein. Proc Natl Acad Sci USA. 1985;82:4075–4079. doi: 10.1073/pnas.82.12.4075. [DOI] [PMC free article] [PubMed] [Google Scholar]