Abstract
Antibody-mediated neutralization of viruses has been extensively studied in vitro, but the precise mechanisms that account for antibody-mediated protection against viral infection in vivo still remain largely uncharacterized. The two points under discussion are antibodies conferring sterilizing immunity by neutralizing the virus inoculum or protection against the development of disease without complete inhibition of virus replication. For tick-borne encephalitis virus (TBEV), a flavivirus, transfer of neutralizing antibodies specific for envelope glycoprotein E protected mice from subsequent TBEV challenge. Nevertheless, short-term, low-level virus replication was detected in these mice. Furthermore, mice that were exposed to replicating but not to inactivated virus while passively protected developed active immunity to TBEV rechallenge. Despite the priming of TBEV-specific cytotoxic T cells, adoptive transfer of serum but not of T cells conferred immunity upon naive recipient mice. These transferred sera were not neutralizing and were predominantly specific for NS1, a nonstructural TBEV protein which is expressed in and on infected cells and which is also secreted from these cells. Results of these experiments showed that despite passive protection by neutralizing antibodies, limited virus replication occurs, indicating protection from disease rather than sterilizing immunity. The protective immunity induced by replicating virus is surprisingly not T-cell mediated but is due to antibodies against a nonstructural virus protein absent from the virion.
For flaviviruses, envelope glycoprotein E plays a central role in the viral life cycle, and the importance of antibodies to E in antiviral protection has been demonstrated by passive transfer experiments (12, 30). Whereas some studies have shown in vivo protection only by in vitro neutralizing antibodies (13, 23), others have shown antibody-mediated protection irrespective of in vitro neutralization (2). More generally, antibody-mediated neutralization of viruses has been extensively studied in vitro (6), but the precise mechanisms that account for antibody-mediated protection against viral infection in vivo still remain largely uncharacterized. The two points under discussion are antibodies conferring sterilizing immunity by neutralizing the virus inoculum (29) or protection against the development of disease, not essentially synonymous with the complete inhibition of virus replication (15).
Tick-borne encephalitis virus (TBEV), a flavivirus highly pathogenic for humans, is endemic in several European countries, Russia, and China. Transfer of monoclonal antibodies specific for TBEV E (13, 25, 26) or of E-specific antisera as well as of polyclonal immunoglobulin preparations with the same specificities (14, 16, 21) into mice resulted in protection of experimental animals from subsequent TBEV challenge, and in vivo protection by antivirion antibodies correlated with in vitro neutralization (13, 25, 26). In a recent study (21), we confirmed the protection of mice against a lethal intraperitoneal (i.p.) TBEV challenge by E-specific antibodies. Infectious virus could not be detected in the blood or brain of passively protected mice subsequent to TBEV challenge. Local virus replication, however, might have gone undetected in these experiments. To answer the question of whether passive antibody-mediated immunity to TBEV reflects protection against disease or sterilization of the TBEV inoculum, we sought to refine our method for the determination of infectious virus in passively protected animals. Furthermore, because low levels of virus replication are known to induce long-lasting and even lifelong immunity for other viruses, we rechallenged mice exposed to TBEV under passive protection after the passively administered antibodies had disappeared from these animals. Results of these experiments showed that limited virus replication occurs in animals passively protected by neutralizing antibodies and, as a consequence, these animals develop long-lasting immunity to TBEV challenge.
MATERIALS AND METHODS
Animals.
Female BALB/c mice (Charles River Wiga, Sulzfeld, Federal Republic of Germany) were given dry food pellets and water ad libitum and were used for experiments at 15 to 17 g of body weight.
Virus.
TBEV, a flavivirus (37), was kindly donated by P. Noel Barrett (Biomedical Research Center, IMMUNO AG, Orth/Donau, Austria). The virus was titrated by a plaque assay on PS cells as described previously (21), and the concentrations of infectious TBEV are expressed as PFU per milliliter of sample.
To detect TBEV in blood, brain, spleen, or peritoneal exudate cells (PEC) of infected animals with a high sensitivity, specimens obtained from two donor mice after TBEV challenge were prepared as outlined below, and one-third of the resulting sample was transferred i.p. into each of three recipient mice. Blood was aseptically collected by cardiac puncture of ether-anesthetized animals, and the approximately 1-ml sample obtained was diluted with an equal volume of phosphate-buffered saline (PBS) before transfer. Brains or spleens were aseptically removed and, after the addition of an equal volume of PBS, homogenized through steel mesh. PEC were washed from the peritoneal cavity, pelleted by centrifugation, and resuspended in PBS for transfer. After transfer, survival of recipient mice was monitored for 28 days. As determined by spiking of samples with TBEV, the limit of detection of this method is approximately 10 PFU/organ (22).
Inactivation of TBEV.
When needed TBEV was inactivated by treatment with UV-psoralen. As described earlier (20), the infectivity of TBEV is entirely lost after exposure for 10 min to UV A irradiation at 2 mW/cm2 in the presence of 1 μg of 4′-aminomethyl-4, 5′-8-trimethylpsoralen hydrochloride (Lee Biomolecular Research Inc., San Diego, Calif.) per ml.
TBEV exposure and passive protection of mice.
Mice were experimentally inoculated with virus by i.p. injection of 0.2 ml of an appropriately diluted TBEV stock containing 1,000 PFU of TBEV, with approximately 4 PFU comprising a 50% lethal dose (21). When needed, other challenge doses were used or the virus was administered intravenously (i.v.) to experimental animals. Passive protection of mice was achieved by subcutaneous (s.c.) administration of 0.2 ml of either a preparation of human TBEV immunoglobulin (Ig) or mouse TBEV hyperimmune serum, diluted with PBS (for details, see reference 21). The human TBEV Ig preparation is commercially available (FSME BULIN; IMMUNO AG, Vienna, Austria; 90 to 153 mg of gamma globulin per ml; hemagglutination inhibition [HAI] titer [mean ± standard error of the mean {SEM}; 16 determinations], 1:1,100 ± 60; neutralization titer [NT] [mean ± SEM; 9 determinations], 1:4,406 ± 518), and mouse TBEV hyperimmune serum was prepared from blood collected by retro-orbital puncture under light ether anesthesia from mice hyperimmunized with a commercially available whole killed virus vaccine (FSME IMMUN Inject; IMMUNO AG; HAI titer [mean ± SEM; 6 determinations], 1:747 ± 107; NT [mean ± SEM; 6 determinations], 1:983 ± 126). Survival of TBEV-exposed animals was monitored for at least 28 days after infection or until no further deaths occurred for another 7 days.
Transfer of serum, spleen cells, or T-enriched spleen cells.
Serum was prepared from blood collected by retro-orbital puncture from ether-anesthetized mice and was administered i.v. to naive animals. Spleens were aseptically removed, and a spleen single-cell suspension (SPC) was prepared by maceration of spleens through steel mesh. After hemolysis (hypotonic NH4Cl buffer), cells were counted and appropriately diluted in PBS for transfer. T-enriched cells were prepared from crude spleen cells with nylon wool columns (18) and were judged to contain approximately 80% T cells by staining for the Thy-1.2 antigen in a fluorescence-activated cell sorter analysis (data not shown). Both spleen cells and T-enriched spleen cells were administered i.p. to naive animals.
ELISA for TBEV antibodies.
An enzyme-linked immunosorbent assay (ELISA) was performed with formaldehyde-inactivated TBEV as a coating antigen and a goat anti-mouse immunoglobulin G (IgG)-horseradish peroxidase conjugate (Accurate Chemical & Scientific Corp., Westbury, N.Y.) for the detection of bound antibodies. Serial twofold dilutions of samples were tested in duplicate, and titers were the highest dilution yielding greater than twofold the absorption of a control mouse serum, with the cutoff value being set to 1:100.
NT50.
TBEV neutralization was tested as described previously (21). Briefly, serial dilutions of samples were incubated with 100 tissue culture infective doses of TBEV, and then the mixtures were incubated for 7 days on TBEV-susceptible Vero cell (CCL-81; American Type Culture Collection [ATCC]) monolayers. The resulting tissue culture supernatants were tested for the presence of TBEV antigen, indicative of TBEV replication, by an ELISA, and the sample dilution resulting in virus neutralization in 50% of the replicates (NT50) was calculated.
HAI titer.
Serial dilutions of samples were incubated with TBEV, and then triplicates of the resulting samples were incubated with goose erythrocytes to allow antibody-free TBEV to induce hemagglutination. The HAI titer was the reciprocal of the highest sample dilution resulting in complete HAI.
Western blotting.
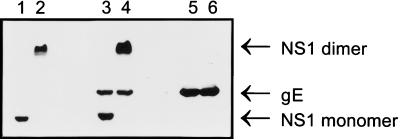
TBEV-infected 3T3 fibroblasts (CCL-92; ATCC) were lysed with NP-40 buffer (20 mM Tris, 150 mM NaCl, 1% Nonidet P-40 [pH 7.5]) containing phenylmethylsulfonyl fluoride (2 mM), aprotinin (10 μg/ml), and leupeptin (10 μg/ml), and the proteins were separated by nonreducing sodium dodecyl sulfate-polyacrylamide gel electrophoresis, either with or without heating of the samples for 2 min to 100°C. For calculation of the apparent molecular weights of proteins, a standard was included with all gels (SeeBlue; NOVEX, San Diego, Calif.). Proteins were transferred to nitrocellulose by electroblotting, the membranes were blocked with skim milk powder (5% in PBS), and the blotted proteins were incubated with antisera that had been preadsorbed to uninfected-cell lysates at various dilutions (see Fig. 2). Bound antibodies were reacted with horseradish peroxidase-coupled anti-mouse IgG (Jackson Immuno Research Laboratories Inc., West Grove, Pa.) as a second step, and bands were visualized by enhanced chemiluminescence (ECL kit; Amersham LIFE SCIENCE, Buckinghamshire, United Kingdom). The identity of TBEV surface protein E or TBEV nonstructural protein NS1 on Western blots was confirmed by staining of sera obtained from mice immunized either with a whole killed TBEV vaccine (FSME IMMUN Inject) or with Escherichia coli-derived recombinant NS1 protein (generously provided by Barbara Plaimauer, IMMUNO AG; 26a).
FIG. 2.
Reactivity of mouse sera with TBEV proteins. A Western blot of a lysate of TBEV-infected cells (5 × 105 cells per lane) was prepared as described in Materials and Methods, either with (lanes 1, 3, and 5) or without (lanes 2, 4, and 6) heating of the samples to 100°C prior to electrophoresis. The membrane was then probed with an NS1 antiserum (lanes 1 and 2), serum obtained from mice 28 days after they were challenged with TBEV while being passively protected (lanes 3 and 4), or an E-monospecific TBEV hyperimmune serum (lanes 5 and 6), all diluted 1:300. Bound antibodies were subsequently reacted with the second-step antibody and visualized by enhanced chemiluminescence. gE, glycoprotein E.
51Cr release assay.
Spleens were obtained from three or more donor mice after cervical dislocation, and an SPC was prepared from the pooled organs. After restimulation of SPC for 5 days with live TBEV at a multiplicity of infection of 0.2 as described by Rothman et al. (31), the SPC was tested for cytotoxic activity. P815 cells (TIB-64; ATCC) were either used as uninfected control target cells or infected with TBEV at a multiplicity of infection of 1 for 7 days, resulting in approximately 90% TBEV-infected cells, as judged by immunofluorescence. The target cells were labeled with Na51CrO4 (400 μCi per 5 × 106 cells), and 104 target cells were incubated with restimulated effector cells at various effector/target cell ratios for 4 h. 51Cr release in supernatants was determined, and specific release was calculated as [(experimental release − spontaneous release)/(maximum release − spontaneous release)] × 100. For the results obtained, the mean ± SEM of virus-specific cytotoxicity (percent release from TBEV-infected target cells − percent release from uninfected control target cells) is reported.
RESULTS
After challenge of passively protected mice, low levels of TBEV can be recovered.
When in a recent study blood and brain samples from mice passively protected with TBEV antibodies (0.2 ml of a human TBEV Ig preparation diluted 1:10 in PBS and delivered s.c.) were obtained at daily intervals after i.p. challenge with 1,000 PFU of TBEV and tested for the presence of infectious TBEV by a plaque assay or by sample transfer into naive recipient mice (a more sensitive assay) (22), no virus was detectable up to the time at which unprotected control mice died (21). To detect the eventual local replication of TBEV, we also tested spleen cells or PEC from passively protected mice for TBEV replication by sample transfer in this study. Organs from antibody-protected mice were thus obtained at daily intervals after i.p. TBEV exposure (1,000 PFU per mouse), the samples from two donor mice were pooled, and equal volumes of the specimens were transferred into each of three naive recipient mice (see Materials and Methods). While the transfer of blood, brain, or spleen samples was never lethal for recipient mice, the transfer of PEC induced lethality in recipient mice. For three experiments, however, TBEV was only detectable on a single day after TBEV exposure, i.e., on day 3, 4, or 6, and resulted in TBEV-induced death in two, two, or one of the three recipients, respectively. These low levels of lethality in recipient mice (67, 67, and 33%) were recently determined to correspond to 1 to 10 PFU of TBEV in the samples (22). Thus, despite passive protection by TBEV antibodies, short-term, low-level virus replication can be detected after virus exposure. Among mice receiving identically prepared tissues from uninfected mice, none died in several consecutive experiments.
TBEV exposure under passive protection results in active immunity.
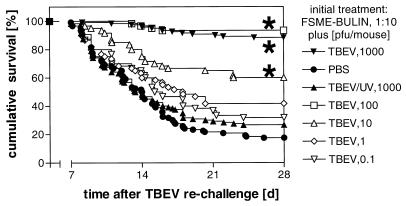
As demonstrated earlier (21), s.c. treatment of mice with a human TBEV Ig preparation (diluted 1:10) or a mouse TBEV hyperimmune serum (diluted 1:3) 2 h prior to a lethal i.p. TBEV challenge (1,000 PFU) resulted in complete protection against the development of tick-borne encephalitis. After 28 or 70 days, i.e., at a time when protection by passively administered human or mouse TBEV antibodies had declined to low residual levels, the animals were rechallenged with TBEV. As a control for the decline of passive protection, groups of mice were initially treated with either human or murine TBEV Ig but were left unchallenged. As antibody-neutralized nonreplicating virus might also induce an immune response, another group of animals was treated with TBEV antibodies and UV-inactivated TBEV corresponding in amount to the challenge dose, i.e., 1,000 PFU. Only a low percentage of mice—or none at all—that received the initial passive protection regimen without TBEV challenge survived when challenged with TBEV 28 or 70 days later (Table 1), indicative of few residual TBEV antibodies present at the time of rechallenge. In contrast, mice that were initially challenged with infectious TBEV while being passively protected by TBEV antibodies were immune to this second TBEV exposure. Active immunity in these mice was not the result of previous exposure to nonreplicating viral antigen, as mice that were injected with the same amount of UV-inactivated TBEV succumbed to this second TBEV exposure. To determine the minimum dose of infectious virus required at the first challenge to induce this immunity, different amounts of TBEV were applied to passively protected animals. As shown in Fig. 1, there was a dose-dependent relationship between the dose of the initial TBEV challenge and the resulting degree of immunity to rechallenge, with as little as 10 PFU at the first challenge significantly increasing survival of rechallenge.
TABLE 1.
Rechallenge after initial TBEV exposure under passive protectiona
| Initial treatment | Survival (%) | MST (days) | No. of animals (no. of exper- iments) |
|---|---|---|---|
| Mouse TBEV antiserum plus | |||
| 1,000 PFU of TBEV | 82 ± 7 | 15.1 ± 3.1 | 38 (4) |
| PBS | 0 ± 0 | 10.7 ± 0.5 | 40 (4) |
| 1,000 PFU of UV-inactivated TBEV | 0 ± 0 | 10.0 ± 0.2 | 40 (4) |
| Human TBEV Ig plus | |||
| 1,000 PFU of TBEV | 81 ± 5 | 14.0 ± 1.2 | 164 (13) |
| PBS | 18 ± 3 | 12.4 ± 0.4 | 168 (13) |
| 1,000 PFU of UV-inactivated TBEV | 22 ± 6 | 12.0 ± 0.7 | 150 (11) |
Mice were injected s.c. with 0.2 ml of either a 1:3-diluted mouse TBEV hyperimmune serum or a 1:10-diluted human TBEV Ig preparation 2 h before they were injected i.p. with 1,000 PFU of TBEV, PBS, or the equivalent of 1,000 PFU of UV-psoralen-inactivated TBEV (initial treatment). After 70 days (recipients of murine TBEV hyperimmune serum) or 28 days (recipients of human TBEV Ig), mice were rechallenged by i.p. injection of 1,000 PFU of TBEV, and their survival was recorded. The results presented are the mean (± SEM) survival percentages and the MST (in days) of mice that died. The data presented were collected from the indicated number of independent experiments, with groups of 8 or more mice each.
FIG. 1.
Active immunity to TBEV. Female BALB/c mice were administered s.c. 0.2 ml of a 1:10-diluted human TBEV Ig preparation 2 h before they were injected i.p. with different amounts of TBEV (1,000, 100, 10, 1, or 0.1 PFU per animal), with PBS, or with the equivalent of 1,000 PFU of UV-psoralen-inactivated TBEV. After 28 days (d), mice were rechallenged i.p. with 1,000 PFU of TBEV, and their survival was monitored for 28 days or until no further deaths occurred for a consecutive week. The data presented are cumulative survival in percentages from three to eight independent experiments with 10 animals per group in each experiment. Differences in the survival of groups of mice were compared to the survival of mice which received UV-inactivated TBEV instead of the initial TBEV challenge by a two-sided log-rank test, and significances are indicated by asterisks (P < 0.0001).
Active immunity after TBEV exposure under passive protection can be transferred by serum.
To determine the effector mechanisms of the observed TBEV immunity, mice were treated with human TBEV antibodies (0.2 ml, 1:10 in PBS, s.c.) and challenged 2 h later with TBEV (1,000 PFU i.p.). At 28 days after this first TBEV challenge, the mice were used for further experiments or received another i.p. challenge with 1,000 PFU of TBEV and were used 28 days after this second challenge. Titers of TBEV E-specific antibodies in serum were tested by an ELISA (five experiments with 10 mice each; sera from 2 to 10 mice were pooled) after the first or the second challenge. Both, however, were found to be low to negative (mean ± SEM after the first challenge, 1:180 ± 100, 25 determinations; after the second challenge, 1:230 ± 160, 19 determinations). A positive control, i.e., serum from mice immunized with FSME IMMUN Inject, validated the ELISA (mean ± SEM, 1:46,280 ± 11,090, 13 determinations from seven experiments; sera from 2 to 10 mice were pooled). Sera from mice after both single and double challenges were furthermore found to be nonneutralizing in a TBEV NT50 test (i.e., NT, <1:2, six determinations each from six experiments; sera from two mice were pooled).
To identify the mechanism responsible for the observed protective immunity to TBEV, serum, crude spleen cells, or T-enriched spleen cells from 6 to 30 donor animals were pooled after survival of the second challenge and were then transferred into naive recipients. Recipient animals were subsequently challenged i.p. with 1,000 PFU of TBEV per animal. Table 2 shows that i.p. transfer of crude (108) or of T-enriched (4 × 107) spleen cells 2 h prior to challenge exerted no significant influence on the percent survival or mean survival times (MST) of the recipient animals. In contrast, i.v. transfer of serum (0.2 ml each at 2 days and 2 h before challenge) conferred complete protection upon naive recipient animals. When combinations of serum and cells were transferred into naive mice, they did not provide protection in excess of that provided by serum alone. In experiments with donors after a single challenge, serum provided partial protection, whereas crude or T-enriched spleen cells had no effect; transfer of serum or crude or T-enriched spleen cells from naive control animals had no effect (data not shown).
TABLE 2.
Adoptive transfer of serum, spleen cells, or T-enriched spleen cellsa
| Transferred material | Survival (%) | MST (days) | No. of animals (no. of experiments) |
|---|---|---|---|
| Serum | 98 ± 2 | 12 ± 0 | 27 (3) |
| Crude spleen cells | 6.7 ± 6.7 | 10.1 ± 1.7 | 33 (3) |
| T-enriched spleen cells | 0 | 11.9 ± 1.7 | 15 (2) |
Mice that were exposed to 1,000 PFU of TBEV while being passively protected by human TBEV Ig and that survived an identical rechallenge were used as donor animals for serum, crude spleen cells, or T-enriched spleen cells. Naive donor mice received either serum (0.2 ml i.v., 2 days and 2 h prior to challenge), crude spleen cells (108 cells i.p., 3 to 6 h prior to challenge), or T-enriched spleen cells (4 × 107 cells i.p., 3 to 6 h prior to challenge) from pools of 6 to 30 donor animals before they were inoculated i.p. with 1,000 PFU of TBEV. The survival of the mice was monitored for 28 days or until no further deaths occurred for a consecutive week. The results presented are the mean (± SEM) survival percentages and the MST (in days) of mice that died. The data presented were collected from two or three independent experiments with the indicated number of animals.
Humoral immunity is predominantly specific for a nonstructural virus protein.
As demonstrated by the transfer experiments, protective immunity that developed after TBEV challenge under passive protection could be passed to naive recipients only by immune serum. This serum, as well as that obtained from mice after the first challenge, however, had low to negative titers of TBEV E-specific antibodies in an ELISA and negative titers in a TBEV NT50 test. Thus, sera from mice after a single challenge were tested by Western blotting for reactivity with different TBEV proteins; TBEV-infected cells were used as the blotted antigen. Depending on whether samples were boiled or not prior to use in nonreducing sodium dodecyl sulfate-polyacrylamide gel electrophoresis, sera from mice rendered immune by having survived a TBEV challenge under passive protection (Fig. 2, lanes 3 and 4, sera pooled from 10 donor mice) stained proteins with apparent molecular masses of approximately 45 and 88 kDa on Western blots, as well as a heat-stable protein of approximately 51 kDa. The size and behavior upon heating of these proteins were in agreement with those of the NS1 protein monomer (heated) and dimer (unheated) (39) and protein E of TBEV, respectively. The identity of these proteins was subsequently confirmed by use of murine sera specific for either the E protein (Fig. 2, lanes 5 and 6) or the NS1 protein (lanes 1 and 2) of TBEV.
TBEV replication under passive protection induces CTL activity.
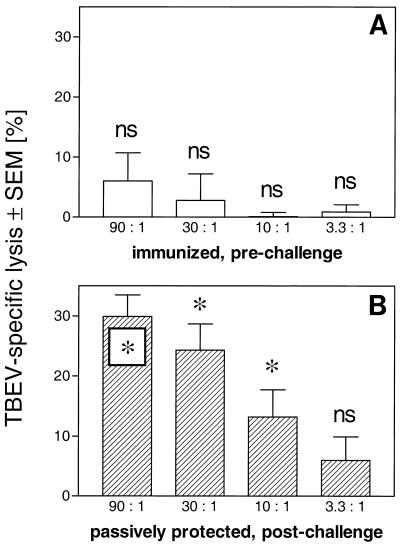
Virus replication in vivo is recognized to induce cytotoxic T-cell (CTL) responses. Although virus replication after TBEV challenge of passively protected mice was demonstrated above, T cells from these mice could not confer protection upon naive recipient mice. To directly determine eventual CTL priming, spleen cells were obtained from mice having survived a TBEV challenge under passive protection. After in vitro restimulation with TBEV for 5 days (31), these cells were tested for TBEV-specific cytolysis in a 51Cr release assay on TBEV-infected or uninfected control P815 target cells. In parallel, spleen cells from mice immunized three times at 3-week intervals with a commercially available whole killed TBEV vaccine (FSME IMMUN Inject, 0.2 ml s.c., diluted 1:10 in PBS) were tested as a control. Hyperimmunization with the whole killed TBEV vaccine did not induce detectable TBEV-specific 51Cr release in our experiments (Fig. 3A). In contrast, however, mice that had been challenged with TBEV while being passively protected by TBEV antibodies manifested pronounced TBEV-specific cytotoxic activity (Fig. 3B).
FIG. 3.
TBEV-specific 51Cr release assay. Spleen cells were obtained from mice vaccinated with a whole killed TBEV vaccine (A) or from mice passively protected by administration of human TBEV antibodies after they had been challenged with TBEV (B). After in vitro restimulation with TBEV, cells were tested for cytotoxicity against TBEV-infected or uninfected control P815 cells in a 51Cr release assay. Percent specific lysis [(experimental release − spontaneous release)/(maximum release − spontaneous release)] × 100, all in counts per minute, at the indicated effector/target cell ratios is given as TBEV-specific lysis (percent lysis of TBEV-infected target cells − percent lysis of uninfected control target cells). The results presented are the means ± SEM for four (A) or five (B) experiments. The significance of the differences between the lysis of uninfected target cells (about 20%) and that of TBEV-infected target cells, i.e., the significance of TBEV-specific lysis, was tested by a paired Student’s t test; the results are indicated as follows: ∗, P < 0.05; ns, not significant.
Neutralization occurs in vitro but cannot be accomplished in vivo.
Results so far indicated short-term, low-level TBEV replication in vivo, despite passive protection by TBEV antibodies with neutralizing capacity in vitro. To determine whether this result was due to insufficient concentrations of TBEV antibodies, the following experiments were performed. Mice were treated s.c. with 1:10-diluted human TBEV Ig as before; after 2 days, i.e., when TBEV antibodies have reached their maximum concentration in serum (21), some animals were challenged i.v. with TBEV, whereas others were bled for serum. Aliquots (0.1 ml) of these sera were then incubated for 1 h with an equivalent of the TBEV challenge dose, i.e., 1,000 or 100 PFU, contained in a volume of 0.1 ml of PBS. These virus-Ig mixtures were further applied to PS cell monolayers for a plaque assay. Sera from mice treated with the 1:10-diluted TBEV Ig showed close to complete neutralization of 1,000 PFU of TBEV (residual virus, 2.3% ± 0.8%; six experiments), while 100 PFU was always entirely neutralized in vitro. The protective immune response with NS1 specificity was nevertheless comparably induced after i.v. challenge with 100 or 1,000 PFU of TBEV (Fig. 1). These results suggest that under conditions that are generally suited for neutralization in vitro, complete neutralization does not necessarily occur in vivo.
DISCUSSION
As demonstrated earlier, the administration of a human TBEV Ig preparation or mouse TBEV hyperimmune serum protected mice against an otherwise highly lethal challenge with TBEV (21). The precise mechanism of protection, however, has remained enigmatic. Here we demonstrate that infectious TBEV can be recovered from passively protected mice, although in very small amounts and for a short period. These results indicate that short-term, low-level virus replication takes place in passively protected animals. Antibody treatment thus provides protection from disease, not protection from infection. For many viruses, low-level replication is known to potently induce long-lasting or even lifelong immunity, a phenomenon that has been successfully exploited by some of the most efficient antiviral vaccines available. In agreement with this notion, only infectious virus induced immunity to rechallenge in our experiments (Table 1), and the resulting degree of immune protection was directly related to the amount of infectious virus initially administered to the experimental animals (Fig. 1). As only replicating virus induced the immunity observed, T-cell-mediated effector mechanisms were considered a likely explanation. To provide direct proof for such effector mechanisms, adoptive-transfer experiments were performed. Surprisingly, however, only the transfer of serum provided some passive protection (donors after a single challenge; data not shown) or almost complete passive protection (donors after a double challenge; Table 2) for naive recipient mice; the transfer of crude or T-enriched spleen cells had no effect (donors after a first challenge; data not shown) (donors after a second challenge; Table 2).
Providing further evidence of virus replication in these animals, serum antibodies from animals actively immune after survival of an initial virus challenge (Fig. 2) as well as after survival of a second virus challenge (data not shown) showed a predominant specificity for NS1, a flavivirus protein which is expressed within and on virus-infected cells (8, 33, 38) and is secreted from these cells (4, 8, 27) but which is not contained within the virion. Protection by antibodies to the NS1 protein has been described for several flaviviruses (9) and for TBEV (17). As the sera were negative in TBEV neutralization assays and furthermore had a borderline TBEV E ELISA titer, the pronounced reactivity with the TBEV NS1 protein likely represents the main effector mechanism of the immunity discussed. Although a contribution to protection by antibodies recognizing the native E protein, as detected in Western blots, cannot be formally ruled out, a comparison of these sera with an E-monospecific TBEV hyperimmune serum (ELISA titer, 1:180 ± 100 versus 1:46,280 ± 11,090; NT, negative versus 1:983 ± 126; see Materials and Methods) which confers complete protection under identical challenge conditions only when administered to mice concentrated or as a 1:3 dilution (21) makes a major role for these E-specific antibodies in protection unlikely. The seeming discrepancy between E staining of the sera on Western blots and only a borderline ELISA titer of these same sera is not considered to arise from differences in the sensitivities of the two methods. Rather it may be due to the different nature of the test antigens, i.e., formaldehyde-inactivated TBEV particles (denatured) in the ELISA versus TBEV-infected cell lysates (native) on Western blots.
The induction of high levels of NS1 antibodies as compared to rather low levels of E antibodies by virus replication in passively protected mice may seem discrepant. It is known, however, that the presence of antibodies may specifically suppress the induction of a primary immune response to the respective antigen (32); this fact has been demonstrated for the TBEV E protein as well (19). This fact is increasingly recognized to be of particular importance for simultaneous passive and active immunization (10, 24, 36), and coligation of the Fc receptor on B cells (Fc gamma RIIB1) with the B-cell antigen receptor, leading to abortive B-cell antigen receptor signaling, has been suggested as a mechanism (5). As human Ig is able to bind to mouse Fc receptors (28), the presence of E antibodies in the course of antigen exposure, i.e., virus challenge in the described model, may thus explain the reduced amounts of E antibodies actively produced. Alternatively, NS1, in contrast to E, is expressed on the surface of infected cells (8, 38) and even secreted from them (4, 27) and may therefore be more readily available for the induction of a humoral immune response.
Clearance from mice of TBEV-infected cells, at least some of which reside in the peritoneal cavity of these animals, would in our model be accomplished by the upcoming antibody response to NS1, a protein which in addition to being secreted is also expressed on the surface of infected cells (8, 38). Consistent with this idea, antibody-dependent cellular cytotoxicity (34) and complement-mediated lysis (33) of flavivirus-infected cells, as mediated by NS1 antibodies, have both been demonstrated. Alternatively, the TBEV-specific CTL activity (Fig. 3B) that we demonstrated in the present study might have served to eliminate virus-infected cells from the challenged mice.
Protection against tick-borne encephalitis by neutralizing antibodies is not brought about by sterilizing immunity, i.e., extensive neutralization of the virus inoculum; low-level virus replication does occur. Failure of in vivo neutralization was observed despite conditions in experimental animals suitable for in vitro neutralization of the amount of virus used for challenge. Such a situation was recently also described for human immunodeficiency virus (HIV) (3), but in contrast to our results, chimpanzees in those experiments were not protected against the development of disease. For HIV, however, follicular dendritic cells were shown earlier to harbor antibody-neutralized virus such that it may subsequently be transmitted to susceptible target cells (11). A similar mechanism allowing for local virus replication despite the presence of neutralizing antibodies may also be operative in our model. Whereas for HIV the ubiquitous presence of target cells did not allow for protection against disease by neutralizing antibodies, TBEV is a neurotropic virus, and the passive-protection regimen used in our study precluded its gaining access to its target organ, the brain. Thus, while local virus replication gave rise to active immunity, disease did not occur. The development of active immunity following virus exposure under passive antibody-mediated protection was observed earlier for hepatitis A virus (40). However, the development of immunity was preceded by abnormal liver functions indicative of subclinical infection (7, 35), so the symptoms resulting from hepatitis A virus infection of its target organ, the liver, were only ameliorated by Ig treatment.
Results from our present study showed that protection against infection by a flavivirus was readily afforded by the administration of TBEV-neutralizing antibodies. Neutralization, however, did not occur in vivo. As recently suggested by others (1), the basis for the widely used correlation between in vitro neutralization and in vivo protection must be reexamined.
ACKNOWLEDGMENTS
We thank Ingrid Burger and Eva Attakpah for skillful help with animal experimentation, Barbara Plaimauer for generously providing TBEV NS1 protein, P. Noel Barrett for constructive advice and for providing TBEV, Aysen Samstag and Katja Olas for TBEV ELISA testing, and Oskar Enzersberger and Friedrich Dorner for providing TBEV neutralization testing (all from IMMUNO AG, Vienna, Austria). Also, we are grateful to Alan L. Rothman (University of Massachusetts, Worcester) for sharing details on the assay of flavivirus-specific CTLs.
We thank IMMUNO AG for financial support.
REFERENCES
- 1.Bachmann M F, Kalinke U, Althage A, Freer G, Burkhart C, Roost H, Aguet M, Hengartner H, Zinkernagel R M. The role of antibody concentration and avidity in antiviral protection. Science. 1997;276:2024–2027. doi: 10.1126/science.276.5321.2024. [DOI] [PubMed] [Google Scholar]
- 2.Brandriss M W, Schlesinger J J, Walsh E E, Briselli M. Lethal 17D yellow fever encephalitis in mice. I. Passive protection by monoclonal antibodies to the envelope proteins of 17D yellow fever and dengue 2 viruses. J Gen Virol. 1986;67:229–234. doi: 10.1099/0022-1317-67-2-229. [DOI] [PubMed] [Google Scholar]
- 3.Conley A J, Kessler J A, Boots L J, Mckenna P M, Schleif W A, Emini E A, Mark G E, Katinger H, Cobb E K, Lunceford S M, Rouse S R, Murthy K K. The consequence of passive administration of an anti-human immunodeficiency virus type 1 neutralizing monoclonal antibody before challenge of chimpanzees with a primary virus isolate. J Virol. 1996;70:6751–6758. doi: 10.1128/jvi.70.10.6751-6758.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Crooks A J, Lee J M, Easterbrook L M, Timofeev A V, Stephenson J R. The NS1 protein of tick-borne encephalitis virus forms multimeric species upon secretion from the host cell. J Gen Virol. 1994;75:3453–3460. doi: 10.1099/0022-1317-75-12-3453. [DOI] [PubMed] [Google Scholar]
- 5.D’Ambrosio D, Hippen K L, Minskoff S A, Mellman I, Pani G, Siminovitch K A, Cambier J C. Recruitment and activation of PTP1C in negative regulation of antigen receptor signaling by Fc gamma RIIB1. Science. 1995;268:293–297. doi: 10.1126/science.7716523. [DOI] [PubMed] [Google Scholar]
- 6.Dimmock N J. Update on the neutralisation of animal viruses. Rev Med Virol. 1995;5:165–179. [Google Scholar]
- 7.Drake M E, Ming C. Gamma globulin in epidemic hepatitis: comparative value of two dosage levels, apparently near the minimal effective level. JAMA. 1954;155:1302. doi: 10.1001/jama.1954.03690330006002. [DOI] [PubMed] [Google Scholar]
- 8.Fan W F, Mason P W. Membrane association and secretion of the Japanese encephalitis virus NS1 protein from cells expressing NS1 cDNA. Virology. 1990;177:470–476. doi: 10.1016/0042-6822(90)90511-o. [DOI] [PubMed] [Google Scholar]
- 9.Gibson C A, Schlesinger J J, Barrett A D. Prospects for a virus non-structural protein as a subunit vaccine. Vaccine. 1988;6:7–9. doi: 10.1016/0264-410x(88)90004-7. [DOI] [PubMed] [Google Scholar]
- 10.Green M S, Cohen D, Lerman Y, Sjogren M, Binn L N, Zur S, Slepon R, Robin G, Hoke C, Bancroft W, et al. Depression of the immune response to an inactivated hepatitis A vaccine administered concomitantly with immune globulin. J Infect Dis. 1993;168:740–743. doi: 10.1093/infdis/168.3.740. [DOI] [PubMed] [Google Scholar]
- 11.Heath S L, Tew J G, Szakal A K, Burton G F. Follicular dendritic cells and human immunodeficiency virus infectivity. Nature. 1995;377:740–744. doi: 10.1038/377740a0. . (Comments.) [DOI] [PubMed] [Google Scholar]
- 12.Heinz F X. Epitope mapping of flavivirus glycoproteins. Adv Virus Res. 1986;31:103–168. doi: 10.1016/s0065-3527(08)60263-8. [DOI] [PubMed] [Google Scholar]
- 13.Heinz F X, Berger R, Tuma W, Kunz C. A topological and functional model of epitopes on the structural glycoprotein of tick-borne encephalitis virus defined by monoclonal antibodies. Virology. 1983;126:525–537. doi: 10.1016/s0042-6822(83)80010-5. [DOI] [PubMed] [Google Scholar]
- 14.Heinz F X, Tuma W, Kunz C. Antigenic and immunogenic properties of defined physical forms of tick-borne encephalitis virus structural proteins. Infect Immun. 1981;33:250–257. doi: 10.1128/iai.33.1.250-257.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hemming V G, Prince G A, Groothuis J R, Siber G R. Hyperimmune globulins in prevention and treatment of respiratory syncytial virus infections. Clin Microbiol Rev. 1995;8:22–33. doi: 10.1128/cmr.8.1.22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hofmann H, Frisch-Niggemeyer W, Kunz C. Protection of mice against tick-borne encephalitis by different classes of immunoglobulins. Infection. 1978;6:154–157. doi: 10.1007/BF01641903. [DOI] [PubMed] [Google Scholar]
- 17.Jacobs S C, Stephenson J R, Wilkinson G W. High-level expression of the tick-borne encephalitis virus NS1 protein by using an adenovirus-based vector: protection elicited in a murine model. J Virol. 1992;66:2086–2095. doi: 10.1128/jvi.66.4.2086-2095.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Julius M H, Simpson E, Herzenberg L A. A rapid method for the isolation of functional thymus-derived murine lymphocytes. Eur J Immunol. 1973;3:645–649. doi: 10.1002/eji.1830031011. [DOI] [PubMed] [Google Scholar]
- 19.Kreil, T. R., I. Burger, E. Attakpah, K. Olas, and M. M. Eibl. Passive protection reduces immunity resulting from simultaneous immunization against tick-borne encephalitis virus. Vaccine, in press. [DOI] [PubMed]
- 20.Kreil T R, Eibl M M. Viral infection of macrophages profoundly alters requirements for induction of nitric oxide synthesis. Virology. 1995;212:174–178. doi: 10.1006/viro.1995.1465. [DOI] [PubMed] [Google Scholar]
- 21.Kreil T R, Eibl M M. Pre- and postexposure protection by passive immunoglobulin but no enhancement of infection with a flavivirus in a mouse model. J Virol. 1997;71:2921–2927. doi: 10.1128/jvi.71.4.2921-2927.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kreil T R, Zimmermann K, Burger I, Attakpah E, Mannhalter J W, Eibl M M. Detection of tick-borne encephalitis virus by sample transfer, plaque assay and strand-specific reverse transcriptase polymerase chain reaction: what do we detect? J Virol Methods. 1997;68:1–8. doi: 10.1016/s0166-0934(97)00100-6. [DOI] [PubMed] [Google Scholar]
- 23.Mathews J H, Roehrig J T. Elucidation of the topography and determination of the protective epitopes on the E glycoprotein of Saint Louis encephalitis virus by passive transfer with monoclonal antibodies. J Immunol. 1984;132:1533–1537. [PubMed] [Google Scholar]
- 24.Murphy B R, Prince G A, Collins P L, Hildreth S W, Paradiso P R. Effect of passive antibody on the immune response of cotton rats to purified F and G glycoproteins of respiratory syncytial virus (RSV) Vaccine. 1991;9:185–189. doi: 10.1016/0264-410x(91)90151-u. [DOI] [PubMed] [Google Scholar]
- 25.Niedrig M, Klockmann U, Lang W, Roeder J, Burk S, Modrow S, Pauli G. Monoclonal antibodies directed against tick-borne encephalitis virus with neutralizing activity in vivo. Acta Virol. 1994;38:141–149. [PubMed] [Google Scholar]
- 26.Phillpotts R J, Stephenson J R, Porterfield J S. Passive immunization of mice with monoclonal antibodies raised against tick-borne encephalitis virus. Brief report. Arch Virol. 1987;93:295–301. doi: 10.1007/BF01310983. [DOI] [PubMed] [Google Scholar]
- 26a.Plaimauer, B. Unpublished data.
- 27.Pryor M J, Wright P J. The effects of site-directed mutagenesis on the dimerization and secretion of the NS1 protein specified by dengue virus. Virology. 1993;194:769–780. doi: 10.1006/viro.1993.1318. [DOI] [PubMed] [Google Scholar]
- 28.Ravetch J V, Kinet J P. Fc receptors. Annu Rev Immunol. 1991;9:457–492. doi: 10.1146/annurev.iy.09.040191.002325. [DOI] [PubMed] [Google Scholar]
- 29.Robbins J B, Schneerson R, Szu S C. Perspective: hypothesis: serum IgG antibody is sufficient to confer protection against infectious diseases by inactivating the inoculum. J Infect Dis. 1995;171:1387–1398. doi: 10.1093/infdis/171.6.1387. [DOI] [PubMed] [Google Scholar]
- 30.Roehrig J T. The use of monoclonal antibodies in studies of the structural proteins of togaviruses and flaviviruses. In: Schlesinger S, Schlesinger M J, editors. The Togaviridae and Flaviviridae. New York, N.Y: Plenum Press; 1986. pp. 251–278. [Google Scholar]
- 31.Rothman A L, Kurane I, Lai C J, Bray M, Falgout B, Men R, Ennis F A. Dengue virus protein recognition by virus-specific murine CD8+ cytotoxic T lymphocytes. J Virol. 1993;67:801–806. doi: 10.1128/jvi.67.2.801-806.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rowley D A, Fitch F W, Stuart F P, Kohler H, Cosenza H. Specific suppression of immune responses. Science. 1973;181:1133–1141. doi: 10.1126/science.181.4105.1133. [DOI] [PubMed] [Google Scholar]
- 33.Schlesinger J J, Brandriss M W, Putnak J R, Walsh E E. Cell surface expression of yellow fever virus non-structural glycoprotein NS1: consequences of interaction with antibody. J Gen Virol. 1990;71:593–599. doi: 10.1099/0022-1317-71-3-593. [DOI] [PubMed] [Google Scholar]
- 34.Schlesinger J J, Foltzer M, Chapman S. The Fc portion of antibody to yellow fever virus NS1 is a determinant of protection against YF encephalitis in mice. Virology. 1993;192:132–141. doi: 10.1006/viro.1993.1015. [DOI] [PubMed] [Google Scholar]
- 35.Schneider A J, Mosley J W. Studies of variations of glutamic-oxaloacetic transaminase in the serum in infectious hepatitis. Pediatrics. 1959;24:367–377. [PubMed] [Google Scholar]
- 36.Schumacher C L, Ertl H C, Koprowski H, Dietzschold B. Inhibition of immune responses against rabies virus by monoclonal antibodies directed against rabies virus antigens. Vaccine. 1992;10:754–760. doi: 10.1016/0264-410x(92)90510-q. [DOI] [PubMed] [Google Scholar]
- 37.Wengler G, Bradley D W, Collett M S, Heinz F X, Schlesinger R W, Strauss J H. Flaviviridae. In: Murphy F A, Fauquet C M, Bishop D H L, Ghabrial S A, Jarvis A W, Martelli G P, Mayo M A, Summers M D, editors. Virus taxonomy. Classification and nomenclature of viruses. Sixth report of the International Committee on Taxonomy of Viruses. New York, N.Y: Springer-Verlag; 1995. pp. 415–427. [Google Scholar]
- 38.Winkler G, Maxwell S E, Ruemmler C, Stollar V. Newly synthesized dengue-2 virus nonstructural protein NS1 is a soluble protein but becomes partially hydrophobic and membrane-associated after dimerization. Virology. 1989;171:302–305. doi: 10.1016/0042-6822(89)90544-8. [DOI] [PubMed] [Google Scholar]
- 39.Winkler G, Randolph V B, Cleaves G R, Ryan T E, Stollar V. Evidence that the mature form of the flavivirus nonstructural protein NS1 is a dimer. Virology. 1988;162:187–196. doi: 10.1016/0042-6822(88)90408-4. [DOI] [PubMed] [Google Scholar]
- 40.Winokur P L, Stapleton J T. Immunoglobulin prophylaxis for hepatitis A. Clin Infect Dis. 1992;14:580–586. doi: 10.1093/clinids/14.2.580. [DOI] [PubMed] [Google Scholar]