Abstract
Using a metagenomic sequencing approach on stool samples from children with Acute Flaccid Paralysis (AFP), we describe the genetic diversity of Sapoviruses (SaVs) in children in Nigeria. We identified six complete genome sequences and two partial genome sequences. Several SaV genogroups and genotypes were detected, including GII (GII.4 and GII.8), GIV (GIV.1), and GI (GI.2 and GI.7). To our knowledge, this is the first description of SaV infections and complete genomes from Nigeria. Pairwise identity and phylogenetic analysis showed that the Nigerian SaVs were related to previously documented gastroenteritis outbreaks with associated strains from China and Japan. Minor variations in the functional motifs of the nonstructural proteins NS3 and NS5 were seen in the Nigerian strains. To adequately understand the effect of such amino acid changes, a better understanding of the biological function of these proteins is vital. The identification of distinct SaVs reinforces the need for robust surveillance in acute gastroenteritis (AGE) and non-AGE cohorts to better understand SaVs genotype diversity, evolution, and its role in disease burden in Nigeria. Future studies in different populations are, therefore, recommended.
Keywords: metagenomics, sapovirus, Acute Flaccid Paralysis, Acute Gastroenteritis, Nigeria
1. Introduction
Sapovirus (SaV) infections are a significant public health problem with the virus implicated in acute gastroenteritis (AGE) in humans and animals [1]. The virus has been associated with both outbreaks and isolated cases of AGE among children and adults [2,3,4,5,6,7,8,9,10]. Sapovirus infections frequently cause diarrhea and vomiting, which usually last for about a week [11]. However, people exhibiting symptoms for longer than usual and with greater severity have also been documented, particularly in immune-compromised individuals [12,13]. The asymptomatic circulation of SaVs has also been reported in children without symptoms of AGE [14,15].
The species Sapporo virus belongs to the genus Sapovirus, family Caliciviridae. Sapoviruses of human origin were first discovered in the stools of children with gastroenteritis in 1976 and in fecal samples collected from babies during a stool survey of Glasgow children using electron microscopy (EM). However, the strain Hu/SaV/Sapporo/1982/JPN (thought to have originated from an outbreak in Sapporo, Japan, in 1982) is widely regarded as the Sapovirus genus prototype strain due to its extensive genetic and virologic characterization [1,16]. The virus is non-enveloped, with a positive-sense, single-stranded RNA genome, which is approximately 7.1 to 7.7 kb in length, containing two open reading frames (ORFs). The large ORF1 encodes a polyprotein which is cleaved by a virus-encoded protease into nonstructural proteins (NS1 [p11], NS2 [p28], NS3 [NTpase], NS4 [p32], NS5 [viral genome-linked protein-VPg], and NS6-NS7 [protease–polymerase, which is further cleaved to form an RNA-dependent RNA-polymerase-RdRp]) and VP1 (the major structural protein). The ORF 2 encodes VP2 (the minor structural protein) [17,18,19,20]. A third ORF has been reported, although its function is currently unknown [1,18].
The most variable (both genetically and antigenically) region of SaVs is the VP1 domain, which is important for eliciting immune responses. The classification of SaV is based on complete VP1 amino acid (aa) sequences, with strains exhibiting ≥57% pairwise VP1 aa identity placed in the same genogroup [21]. Currently, SaVs have been classified into 19 genogroups (GI to GXIX), with four genogroups (GI, GII, GIV, and GV) known to infect humans [22,23,24]. SaVs in other genogroups have been identified in minks (GXII), bats (GXIV, GXVI-GXIX), dogs (GXIII), rodents (GXV), swine (GIII and GV-GXI), and sea lions (GV) [25,26,27]. Human SaVs are further classified into 17 different genotypes [1], and a recently detected genotype in Peru has been proposed as GII.8 [11,28].
Metagenomics, an alternate culture- and sequence-independent method, does not require the presence of a specific gene in all subject entities. The original goal for developing this methodology was to enable the sequence-based and functional analysis of collective microbial genomes in environmental samples [29,30]. Viral metagenomics has proven to be an effective technique for discovering new viruses and expanding our understanding of the diversity of viruses found in clinical samples, including the identification of new SaV strains [29,31]. Metagenomic analyses using whole genome sequencing are becoming more common in clinical settings, and they have been used for the in-depth genomic analysis of SaVs in four different countries in the Americas [32] and China [33,34]. The use of whole genome rather than short genome sequences has improved the in-depth analysis of viral genomes, including members in the Caliciviridae family that have the capability to rapidly evolve, recombine, and acquire mutations [32,35]. Moreover, metagenomic shotgun sequencing has enabled researchers to track viral infection transmission and conduct effective epidemiological studies. These advancements have contributed to reducing the burden of treatment for patients by preventing and controlling infections [34].
Different genomic regions, particularly those encoding RdRp and VP1, can cause discrepancies in phylogenetic clustering, resulting in the discovery of intra- and inter-genogroup recombinant strains [36]. Similar to noroviruses, several recombinant SaV strains have been reported [36,37,38]. These strains may have changed virulence as a result of recombination, which, in turn, may enhance and increase disease burden [12]. Recombinant SaV strains have been classified as those with a discordant clustering of the VP1 encoding region and the RNA-dependent RNA-polymerase (RdRp) [37], with the RdRp-VP1 junction and the NS3-NS4 junction found to be the two main recombination hotspots [35,36,38].
In Africa, the landscape of circulating human SaV genogroups in recent years has been dominated by GI and GII SaVs [39,40]. Genogroup V (GV) viruses have been rarely reported in Africa. In contrast, GIV has been reported in Burkina Faso [41] and South Africa [42], where they were identified in up to one-third of infections in patients with gastroenteritis [42,43]. There are currently no published data on SaV infections in Nigeria. In this study, we describe the molecular characterization and genetic diversity of SaV genomes identified in the stool samples of children 15 years and below diagnosed with Acute Flaccid Paralysis (AFP) in Nigeria.
2. Methodology
2.1. Faecal Specimen Collection and Processing
The fecal samples analyzed in this study were collected as part of the National AFP surveillance program in Nigeria. Samples were collected from children aged 15 years and below diagnosed with AFP in Nigeria in 2020 [44]. These stool samples were collected between January and December 2020 following national ethical guidelines and sent to the WHO National Polio Laboratory in Ibadan, Nigeria.
In this study, 254 archived (−20 °C freezers stored) poliovirus culture-negative samples from five states in Nigeria (Supplementary Figure S1) were combined into 55 pools by the state of collection and the month of sample collection and subsequently analyzed. Briefly, about 0.5 g of stool was dissolved in 4.5 mL of phosphate-buffered saline (PBS) and 0.5 g of glass beads. After 20 min of vortexing, the mixture was subjected to 20 min of centrifugation at 3000 rpm. Subsequently, 2 mL of the supernatant was aliquoted into 1 mL cryovials and stored at −20 °C. Thereafter, the stool suspensions were pooled. To make a pool, 200 uL of fecal suspensions were mixed, with each sample pool containing between 1 and 7 fecal suspensions (Supplementary Table S1). Sample pools were subsequently shipped on ice packs to the University of Leuven, Rega Institute, Laboratory of Clinical and Epidemiological Virology in Belgium. The samples were stored at −80 °C until further processing.
2.2. Sequencing and Read Processing
The NetoVIR protocol was used to purify virus-like particles (VLPs) from the samples, as previously described [45]. Briefly, using a MINILYS homogenizer, fecal suspensions were homogenized for 1 min at 3000 rpm and filtered through a 0.8 μm PES filter. Free-floating nucleic acids were digested via treatment with a mixture of Benzonase (Millipore, Billerica, MA, USA), (Novagen, Madison, WI, USA), and Micrococcal Nuclease (New England Biolabs, Ipswich, MA, USA). Subsequently, nucleic acid was extracted using the QIAamp Viral RNA Mini Kit (Qiagen, Hilden, Germany) according to the manufacturer’s instructions, but without the addition of carrier RNA. A slightly modified Whole Transcriptome Amplification (WTA2) Kit procedure (Sigma-Aldrich, St Louis, MO, USA) was used for the first- and second-strand synthesis, as well as a random PCR amplification over 17 cycles. The WTA2 products were purified using MSB Spin PCRapace spin columns (Stratec Biomedical, Birkenfeld, Germany). The libraries for Illumina sequencing were prepared using the Nextera XT Library Preparation Kit (Illumina, San Diego, CA, USA). After that, samples were paired-end-sequenced (2 × 150 bp) on an Illumina Novaseq 6000 platform.
Raw reads were processed with the Virome Paired-End Reads (ViPER) pipeline (https://github.com/Matthijnssenslab/ViPER, accessed on 14 March 2024). Using Trimmomatic, the reads were trimmed for quality and adapters [46], and reads mapping to the human genome were removed using Bowtie 2 [47]. Subsequently, the trimmed and filtered reads were de novo assembled into contigs using metaSPAdes [48]. The sensitive option in DIAMOND was then used to annotate the contigs [49]. Kronatool files were manually inspected to identify all SaV genomes. To determine the depth of coverage, trimmed reads were mapped against the SaV contigs using Bowtie2 [40].
2.3. Sapovirus Genotyping and Phylogenetic Analyses
A BLASTn search was performed against the GenBank database using the SaV contigs identified in this study as queries. The top five hits (sequences with the highest percentage of identity and query coverage) were downloaded and added to the alignment, along with reference human SaV sequences. The SaV sequences generated in this study were aligned with reference human SaV sequences downloaded from GenBank using the MAFFT online tool [50]. The human calicivirus genotyping tool [51] was used to determine the genogroups and genotypes of each SaV sequence generated in this study. To construct the corresponding maximum-likelihood phylogenetic trees, various genomic regions of interest, including individual genes encoding structural (VP1 and VP2) and nonstructural (NS1-7) proteins, were selected from the alignment. Phylogenetic trees were constructed using MEGA version 11 [52] and the maximum-likelihood method with 1000 bootstrap replications. Subsequently, we aligned each distinct pair of sequences to determine the pairwise identity of the sequences from this study and published reference sequences using the Sequence Demarcation Tool [53]. The conserved amino acid motifs for SaV were identified and analyzed using NCBI’s conserved domain database (CDD) [54]. Sequences from this study were also analyzed for recombination events using the Recombination Detection Program (RDP) 4 [55]. To detect recombination, nine different detection techniques—including RDP, GENECONV, BootScan, MaxChi, Chimaera, 3Seq, PhylPro, LARD, and SiScan—were used with the default parameters. Recombination events were considered reliable if they were predicted by at least six different detection methods in the RDP4 program.
2.4. GenBank Submission
The Sapovirus nucleotide sequences and mapped reads described in this study were submitted to GenBank and the SRA and assigned accession numbers OR837774-OR837781 and PRJNA1043841, respectively.
3. Results
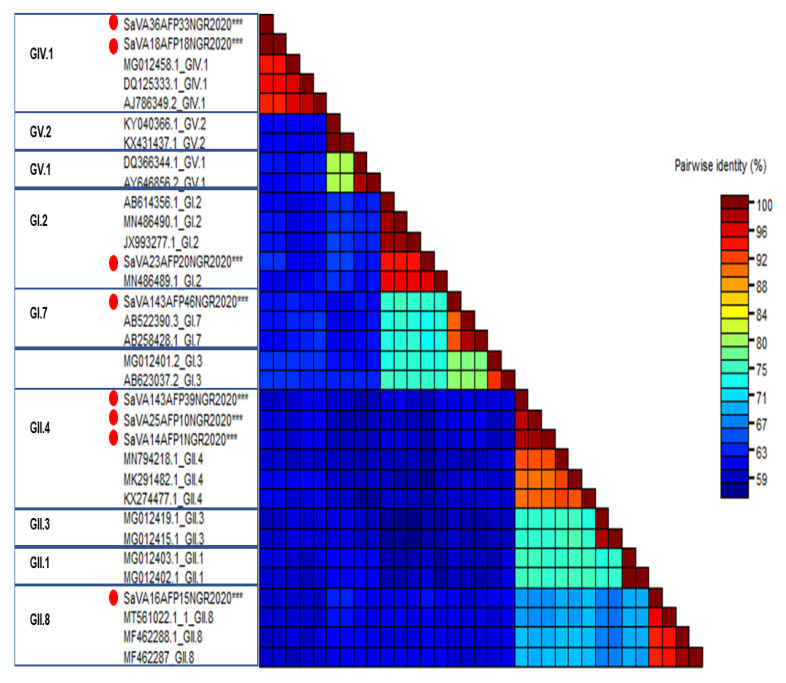
Eight (14.5%) of the fifty-five non-polio AFP sample pools tested using deep sequencing contained SaV reads, corresponding to 0.1% to 2.41% of the generated reads (Table 1). Of the eight samples with SaV reads, two sample pools each from Edo, Abuja, Kaduna, and Lagos states had SaV reads (Supplementary Table S1 and Figure S1). No SaV reads were detected in sample pools from the Anambra state. We obtained six complete genome sequences (SaV-A14-AFP1-NGR-2020, SaV-A16-AFP15-NGR-2020, SaV-A18-AFP18-NGR-2020, SaV-A36-AFP33-NGR-2020, SaV-A143-AFP39-NGR-2020, SaV-A143-AFP46-NGR-2020) having complete coding regions, and two partial genome sequences (SaV-A23-AFP20-NGR-2020 and SaV-A25-AFP10-NGR-2020) having VP1 and VP2 capsid gene but only partial nonstructural genes. Complete VP1-based sequence genotyping using the calicivirus typing tool and Sequence Demarcation Tool showed that the eight contigs belonged to three of the four recognized human SaV genogroups (GI, GII, and GIV). Furthermore, they belonged to genotypes GI.2 [n = 1], GI.7 [n = 1], and GII.4 [n = 3], and the newly discovered genotypes GII.8 [n = 1] and GIV.1 [n = 2] (Table 1, Figure 1).
Table 1.
Summary of SaV reads detected in the pooled AFP samples.
| Sample ID | Deep Sequencing-Derived Reads and Sequences | Calicivirus Genotyping Tool Results | BLASTn Results | |||||
|---|---|---|---|---|---|---|---|---|
| Total Reads | SaV Reads | SaV Reads % | Nucleotide Length | VP1 Length | Type | Accession number of the Closest Hit | Pairwise Identity (%) | |
| AFP1-NGR | 16,751,880 | 51,665 | 0.31% | 7483 | 1677 | GII.4 | MN794218.1 | 91.29 |
| AFP10-NGR | 4,179,088 | 882 | 0.02% | 4554 | 1677 | GII.4 | MN794218.1 | 91.17 |
| AFP15-NGR | 23,759,398 | 24,255 | 0.1% | 7502 | 1667 | GII.8 | MT561022.1 | 95.36 |
| AFP18-NGR | 7,378,192 | 1777 | 0.02% | 7456 | 1656 | GIV.1 | DQ125333.1 | 94.27 |
| AFP20-NGR | 10,079,418 | 53 | 0% | 1819 | 1439 | GI.2 | MN486489.1 | 97.08 |
| AFP33-NGR | 11,500,814 | 83,736 | 0.73% | 7494 | 1656 | GIV.1 | DQ104357.2 | 94.43 |
| AFP39-NGR | 13,103,440 | 41,026 | 0.31% | 7526 | 1676 | GII.4 | AB429084.2 | 91.06 |
| AFP46-NGR | 22,571,328 | 544,044 | 2.41% | 7524 | 1698 | GI.7 | AB258428.1 | 90.76 |
Abbreviations: Nigeria, NGR; Acute Flaccid Paralysis, AFP; Sapovirus, SaV.; pairwise nucleotide identity with the top matching reference sequences for the VP1 region using BLASTn.
Figure 1.
The Sequence Demarcation Tool [53] was used to estimate the pairwise sequence identity between the VP1 of SaV sequences from this study and existing SaV references. The sequences reported in this study are indicated with a red circle and astericks (***).
Several calicivirus proteins possess conserved motifs and domains responsible for their function. Previously described conserved amino acid motifs in caliciviruses include NS3 [NTpase] (GAPGIGKT), NS5 [Viral genome-linked protein (VPg)] (KGKTK and DDEYDE), protease (GDCG), RNA-dependent RNA-polymerase (WKGL, KDEL, DYSKWDST, GLPSG, and YGDD) and VP1(PPG and GWS have been suggested to be vital in stabilizing P-domain formation in the SaV capsid) [1,18,56]. An analysis of the conserved amino acid motifs of SaV sequences in this study showed minor variations in NS3 and NS5 motifs. In the NS3, all the sequences, irrespective of the genogroup, had a GPPGIGKT motif (the A replaced with P), while in the NS5, the KGKTK motif was present in all the sequences except two (SaV-A14-AFP1-NGR-2020 and SaV-A143-AFP46-NGR-2020) that had the KGKNK motif (Table 2).
Table 2.
Typical motifs of functional proteins of SaV detected in the pooled AFP samples.
| Strain | NTpase GAPGIGKT |
VPg (KGKTK And DDEYDE |
Protease (GDCG) |
RdRp (WKGL, KDEL, DYSKWDST, GLPSG and YGDD) | VP1 (PPG and GWS) |
|---|---|---|---|---|---|
| SaV-A14-AFP1-NGR-2020 | 481PPGIGKT |
936KGKK And 953DDEYDE |
1169GDCG | 1213WKGL, 1374KDEL, 1449DYSKWDST, 1504GLPSG and 1552YGDD | 1856PPG and 2000GWS |
| SaV-A16-AFP15-NGR-2020 | 465PPGIGKT |
927KGKTK And 948DDEYDE |
1154GDCG | 1198WKGL, 1359KDEL, 1434DYSKWDST, 1489GLPSG and 1537YGDD | 1841PPG and 1987GWS |
| SaV-A18-AFP18-NGR-2020 | 481PPGIGKT |
942KGKTK And 963DDEYDE |
1169GDCG | 1213WKGL, 1374KDEL, 1449DYSKWDST, 1504GLPSG and 1552YGDD | 1851PPG and 1996GWS |
| SaV-A36-AFP33-NGR-2020 | 481PPGIGKT |
942KGKTK And 963DDEYDE |
1169GDCG | 1213WKGL, 1374KDEL, 1449DYSKWDST, 1504GLPSG and 1552YGDD | 1851PPG and 1996GWS |
| SaV-A143-AFP39-NGR-2020 | 481PPGIGKT |
942KGKTK And 963DDEYDE |
1169GDCG | 1213WKGL, 1374KDEL, 1449DYSKWDST, 1504GLPSG and 1552YGDD | 1856PPG and 2001GWS |
| SaV-A143-AFP46-NGR-2020 | 479PPGIGKT |
940KGKK And 961DDEYDE |
1167GDCG | 1211WKGL, 1373KDEL, 1448DYSKWDST, 1503GLPSG and 1552YGDD | 1855PPG and 2000GWS |
The substitution observed in NS3 and NS5 motifs are highlighted in red box.
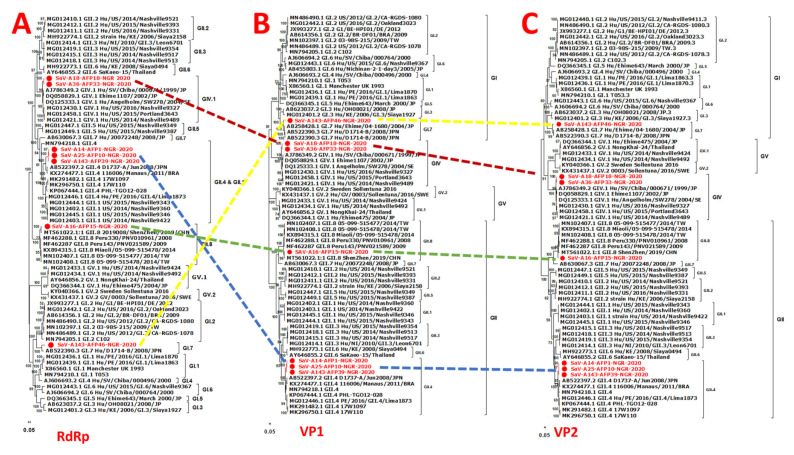
Phylogenetic analysis using the individual genes encoding both the structural (VP1 and VP2) and nonstructural proteins (NS1-7) and reference human SaVs showed topological incongruence. Specifically, all the nonstructural genes (Figure 2 and Figure 3) of genomes reported in this study and previously reported reference sequences, including the RdRp gene, were clustered into three main genogroups (GI, GII, and GV) (Figure 2A–D and Figure 3A–D). In contrast, the structural genes (VP1 and VP2) were grouped into four clusters (GI, GII, GIV, and GV) (Figure 4B,C). All GIV nonstructural genes were consistently found among the GII clusters, while their structural genes were in a group independent of GII. Interestingly, the GIV sequences in this study clustered independently from previously documented strains from Asia and North America. The GII.8 detected in this study clustered with a novel variant of the GII.8 genotype, which was associated with an outbreak of SaV among primary school students in Shenzhen city, China, in 2019 [57].
Figure 2.
Maximum-likelihood trees of the NS1-4 genes. The trees were constructed based on the full-length amino acid sequences of (A) the p11 (NS1) protein, (B) the p28 (NS2) protein, (C) the NTpase (NS3), and (D) the p32 (NS4). Bootstrap support values greater than 50 are shown. Sequences reported in this study are highlighted in red.
Figure 3.
Maximum-likelihood trees of the NS4-7 and RdRp gene. The trees were constructed based on the full-length amino acid sequences of (A) the p32 (NS4) protein, (B) the viral genome-linked protein (NS5), (C) protease–polymerase (NS6-7), and (D) RdRp. Bootstrap support values greater than 50 are shown. Sequences reported in this study are highlighted in red.
Figure 4.
Maximum-likelihood trees of SaV genes. The trees were constructed based on the full-length amino acid sequences of (A) RdRp, (B) the capsid protein (VP1), and (C) the small basic protein (VP2). Bootstrap support values greater than 50 are shown. Sequences reported in this study are highlighted in red.
The human SaV sequences detected in this study did not contain any significant recombination breakpoints according to RDP4 sequence analysis.
4. Discussion
Without a doubt, the global health community has made significant investments and taken targeted actions to address the primary causes of child death through high-impact interventions, such as access to nutrition, safe water, sanitation, and vaccination. Malnutrition and diarrheal diseases, on the other hand, continue to be among the top causes of death among children [58,59]. In Nigeria, there is a dearth of information on SaV’s genetic diversity, epidemiology, and evolution [60]. In the present study, we describe six complete genome sequences (all with complete coding regions) and two partial genome sequences from children with AFP. This is the first detection of human SaVs in Nigeria. Interestingly, multiple genotypes were detected, indicating the circulation of various strains in Nigeria. Specifically, we documented the presence of genogroups GII (GII.4 and GII.8), GIV (GIV.1), and GI (GI.2 and GI.7) in Nigeria.
All the identified human SaVs, irrespective of genotype, had amino acid substitution A482P in the NS3 motifs (Table 2). A similar motif was reported in SaVs from pigs [25]. Since many caliciviruses, including SaVs, are difficult to grow in cell cultures, studying the biological function of their nonstructural proteins remains challenging. However, few studies have elucidated the role and activities of the polymerase and protease (3C-like protease (NS6) and the 3CD-like protease–polymerase (NS6-7) [18,61,62,63]. Interestingly, the mutational analysis of the RdRp-conserved GDD amino acid motif from a calicivirus rabbit hemorrhagic disease virus (RHDV) showed that the substitution of the RHDV 3Dpol 1605 aspartate residue by asparagine, glycine or glutamate residues resulted in a complete loss of enzymatic activity [64]. Understanding the biological functions of various SaV proteins and the role of various amino acid substitutions in the evolution of viruses is needed to understand the potential implications of newly observed mutations.
Regarding seasonality, Nigeria has two seasons in a year as follows: the wet (April to October) and dry (November to March) seasons. In this study, SaVs were detected in sample pools collected in both the wet and dry seasons, with 62.5% (5/8) of the SaVs detected in samples collected during the wet season. Our findings support the widely accepted hypothesis that SaV is primarily found in the winter and during the rainy season [8,36]. Since no other work on SaV’s prevalence on a monthly basis has been reported in Nigeria, it is difficult to determine the true prevalence of SaV in Nigeria using this approach. We are aware that the SaV’s diversity described in this study might not completely capture variants present in this sample, considering the study design. Therefore, future studies aimed at identifying the seasonal nature of SaV transmission patterns could help with infection prevention, control, and diagnosis strategies.
The genetic analysis of currently circulating SaV strains is critical for understanding the cryptic geographic distribution of SaVs in the population, both regionally and globally. Previous studies have revealed that GI is the most common SaV genogroup around the globe and has been increasingly detected in many African countries [8,39,65,66]. In this study, we observed the circulation of a variety of SaV strains throughout the year. Sapovirus GII.4 was the predominant genotype detected and was closely followed by GIV.1. It is important to note that the presence of GII.4 sequences in this study, which have been classified among the rare SaV genotypes [32], may indicate that the SaV landscape in Africa might be changing. Furthermore, the preponderance of GII.4 sequences from this research is contrary to previous SaV studies in Africa, where GI was the most abundant SaV genotype [39,40,65]. In Thailand, a significant proportion of genotype GII.4 SaV was identified [67]. GIV, on the other hand, is a genotype that was frequently detected in developed countries around 2007 [1], as well as in Africa between 2009 and 2013 [41,42].
Phylogenetically, the Nigerian SaVs were related to previously reported SaV reference strains. While GIV sequences in this study formed small sub-clusters independent from previously documented strains from Asia and North America, the GII.8 in this study was 95.4% similar and clustered with a novel variant of the GII.8 genotype, which was associated with an outbreak of SaV among primary school students in Shenzhen city, China, in 2019 [57]. The position and length of the ORFs, VP1, and VP2, of the Nigerian GII.8 strains were identical to those of the Shenzhen strain. Remarkably, the GI.7 strain from this study was more than 90% similar to the GI.7 strains from Japan that were associated with gastroenteritis outbreaks linked to the consumption of contaminated shellfish [68]. Notably, GII.4 strains were found in samples collected in Lagos (January), Kaduna (February), and Abuja (September), whereas GIV strains were found in samples collected in the Edo state (May and August 2020). These results imply that these strains may be locally circulating in Nigeria and/or that an outbreak that was not discovered may have occurred there. The robust surveillance of SaV among AGE and non-AGE cohorts in Nigeria is needed to better understand the genotype diversity, evolution, and probable disease association of this virus in the country.
Of note, the RDP4 findings and the phylogenetic tree structure did not provide adequate support to classify any of the Nigerian SaVs as recombinant strains. However, sequences from this study and other reference sequences (including the four genogroups known to infect humans) included in the alignment all showed a phylogenetic pattern in which all nonstructural genes clustered into three major genogroups (GI, GII, and GIV). The structural genes (VP1 and VP2) were divided into the following four clusters: GI, GII, GIV, and GV. A similar topology incongruence has been reported [32], which may indicate an ancient recombination event.
Some of the limitations of our study include the fact that samples suffered more than one round of freezing and thawing, which might have affected the quantity and quality of the genomes recovered in this study. We were also unable to determine the true prevalence of SaV due to our purposive sampling strategy, which included only children with AFP.
In conclusion, we describe six complete and two partial SaV genome sequences. This is the first report on human SaVs in Nigeria. Hence, the data described here can serve as references to help develop tools to enhance the surveillance of and improve epidemiological information on SaVs in Nigeria and Africa at large, where only short-genome regions have been reported. Further, understanding the evolutionary dynamics of SaV, especially the nonstructural proteins, is vital to fully delineate the role of amino acid substitutions in SaV’s evolution and genetic diversity. This would make their nonstructural proteins desirable targets for developing therapeutics to treat human calicivirus infections.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/pathogens13030264/s1, Table S1: Summary of samples analysed in this study including the number of samples per pool, location where samples were collected and month of sample collection; Figure S1: Map of Nigeria indicating the states (highlighted in light blue) spread across five geopolitical zones from where samples analyzed in this study were collected and the number of samples with SaV reads detected (highlighted in red diamond) from each state.
Author Contributions
Conceptualization, U.E.G., T.O.C.F., A.J.A., J.M. and M.O.A.; Data curation, U.E.G.; Formal analysis, U.E.G., T.O.C.F. and L.D.C.; Investigation, U.E.G., L.D.C., S.T.A., E.I.O., A.O.O. (Adebowale Oluseyi Olayinka), T.G.A., T.E.A., B.O.P., O.T.O. and O.A.G.; Methodology, U.E.G., L.D.C., J.M. and M.O.A.; Project administration, U.E.G., T.O.C.F., A.J.A., J.M. and M.O.A.; Resources, A.J.A., J.M. and M.O.A.; Supervision, I.K., A.J.A., J.M., and M.O.A.; Validation, U.E.G.; Visualization, U.E.G., T.O.C.F., S.T.A., I.M.I., B.A.O., E.I.O., A.O.O. (Adebowale Oluseyi Olayinka), T.G.A., A.O.O. (Arthur Obinna Oragwa), T.E.A., B.O.P., O.G.O. and A.I.M.; Writing—original draft, U.E.G.; Writing—review and editing, U.E.G., T.O.C.F., L.D.C., S.T.A., I.M.I., B.A.O., E.I.O., A.O.O. (Adebowale Oluseyi Olayinka), T.G.A., A.O.O. (Arthur Obinna Oragwa), T.E.A., B.O.P., O.G.O., O.T.O., O.A.G., A.I.M., I.K., A.J.A., J.M. and M.O.A. All the authors have read and approved the final version of the manuscript before submission. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
This manuscript does not contain any research involving human participants performed by any of the authors. Specifically, the anonymized and pooled poliovirus-negative stool samples analyzed in this study were collected following national ethical guidelines and sent to the WHO National Polio Laboratory in Ibadan, Nigeria, as part of the National AFP surveillance program in Nigeria. The samples were screened to determine if poliovirus was the etiologic agent of the clinical manifestation (AFP). After confirmation that no poliovirus was present in these samples, they were anonymized for further exploration before use in this study. Please note that no identifying information that can link any individual to the pooled samples analyzed in this study is present in this article.
Informed Consent Statement
Not applicable.
Data Availability Statement
The original data presented in the study including the Sapovirus nucleotide sequences are openly available in GenBank under the accession number OR837774-OR837781. Raw sequence data from this study have also been uploaded the NCBI database under the Bioproject number PRJNA1043841.
Conflicts of Interest
The authors declare that no conflicts of interest exist.
Funding Statement
This research received no external funding.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Oka T., Wang Q., Katayama K., Saif L.J. Comprehensive review of human sapoviruses. Clin. Microbiol. Rev. 2015;28:32–53. doi: 10.1128/CMR.00011-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dey S.K., Phan T.G., Nguyen T.A., Nishio O., Salim A.F., Yagyu F., Okitsu S., Ushijima H. Prevalence of sapovirus infection among infants and children with acute gastroenteritis in Dhaka City, Bangladesh during 2004–2005. J. Med. Virol. 2007;79:633–638. doi: 10.1002/jmv.20859. [DOI] [PubMed] [Google Scholar]
- 3.Phan T.G., Trinh Q.D., Yagyu F., Okitsu S., Ushijima H. Emergence of rare sapovirus genotype among infants and children with acute gastroenteritis in Japan. Eur. J. Clin. Microbiol. Infect. Dis. Off. Publ. Eur. Soc. Clin. Microbiol. 2007;26:21–27. doi: 10.1007/s10096-006-0235-7. [DOI] [PubMed] [Google Scholar]
- 4.Romani S., Azimzadeh P., Mohebbi S.R., Bozorgi S.M., Zali N., Jadali F. Prevalence of sapovirus infection among infant and adult patients with acute gastroenteritis in Tehran, Iran. Gastroenterol. Hepatol. Bed Bench. 2012;5:43–48. [PMC free article] [PubMed] [Google Scholar]
- 5.Dey S.K., Sumiya M.K., Shaha M., Haque R., Okitsu S., Ushijima H. Intragenogroup Recombination in the Complete Genome Sequence of Human Sapovirus Circulating in Bangladesh. Genome Announc. 2018;6:e00388-18. doi: 10.1128/genomeA.00388-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sánchez G.J., Mayta H., Pajuelo M.J., Neira K., Xiaofang L., Cabrera L., Ballard S.B., Crabtree J.E., Kelleher D., Cama V., et al. Epidemiology of Sapovirus Infections in a Birth Cohort in Peru. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2018;66:1858–1863. doi: 10.1093/cid/cix1103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Becker-Dreps S., Bucardo F., Vinjé J. Sapovirus: An important cause of acute gastroenteritis in children. Lancet Child Adolesc. Health. 2019;3:758–759. doi: 10.1016/S2352-4642(19)30270-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Varela M.F., Rivadulla E., Lema A., Romalde J.L. Human Sapovirus among Outpatients with Acute Gastroenteritis in Spain: A One-Year Study. Viruses. 2019;11:144. doi: 10.3390/v11020144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Becker-Dreps S., González F., Bucardo F. Sapovirus: An emerging cause of childhood diarrhea. Curr. Opin. Infect. Dis. 2020;33:388–397. doi: 10.1097/QCO.0000000000000671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.De Oliveira-Tozetto S., Santiso-Bellón C., Ferrer-Chirivella J.M., Navarro-Lleó N., Vila-Vicent S., Rodríguez-Díaz J., Buesa J. Epidemiological and Genetic Characterization of Sapovirus in Patients with Acute Gastroenteritis in Valencia (Spain) Viruses. 2021;13:184. doi: 10.3390/v13020184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Liu X., Jahuira H., Gilman R.H., Alva A., Cabrera L., Okamoto M., Xu H., Windle H.J., Kelleher D., Varela M., et al. Etiological Role and Repeated Infections of Sapovirus among Children Aged Less than 2 Years in a Cohort Study in a Peri-urban Community of Peru. J. Clin. Microbiol. 2016;54:1598–1604. doi: 10.1128/JCM.03133-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kumthip K., Khamrin P., Ushijima H., Chen L., Li S., Maneekarn N. Genetic recombination and diversity of sapovirus in pediatric patients with acute gastroenteritis in Thailand, 2010–2018. PeerJ. 2020;8:e8520. doi: 10.7717/peerj.8520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Pietsch C., Liebert U.G. Intrahost viral evolution during chronic sapovirus infections. J. Clin. Virol. Off. Publ. Pan Am. Soc. Clin. Virol. 2019;113:1–7. doi: 10.1016/j.jcv.2019.02.001. [DOI] [PubMed] [Google Scholar]
- 14.Rivera-Gutiérrez X., Morán P., Taboada B., Serrano-Vázquez A., Isa P., Rojas-Velázquez L., Pérez-Juárez H., López S., Torres J., Ximénez C., et al. The fecal and oropharyngeal eukaryotic viromes of healthy infants during the first year of life are personal. Sci. Rep. 2023;13:938. doi: 10.1038/s41598-022-26707-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pitkänen O., Markkula J., Hemming-Harlo M. Sapovirus, Norovirus and Rotavirus Detections in Stool Samples of Hospitalized Finnish Children with and without Acute Gastroenteritis. Pediatr. Infect. Dis. J. 2022;41:e203–e207. doi: 10.1097/INF.0000000000003493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Madeley C.R., Cosgrove B.P. Letter: Caliciviruses in man. Lancet. 1976;1:199–200. doi: 10.1016/S0140-6736(76)91309-X. [DOI] [PubMed] [Google Scholar]
- 17.Robinson S., Clarke I.N., Vipond I.B., Caul E.O., Lambden P.R. Epidemiology of human sapporo-like caliciviruses in the South West of England: Molecular characterisation of a genetically distinct isolate*. J. Med. Virol. 2002;67:282–288. doi: 10.1002/jmv.2219. [DOI] [PubMed] [Google Scholar]
- 18.Oka T., Katayama K., Ogawa S., Hansman G.S., Kageyama T., Ushijima H., Miyamura T., Takeda N. Proteolytic processing of sapovirus ORF1 polyprotein. J. Virol. 2005;79:7283–7290. doi: 10.1128/JVI.79.12.7283-7290.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Smertina E., Hall R.N., Urakova N., Strive T., Frese M. Calicivirus Non-structural Proteins: Potential Functions in Replication and Host Cell Manipulation. Front. Microbiol. 2021;12:712710. doi: 10.3389/fmicb.2021.712710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Li T.-C., Kataoka M., Doan Y.H., Saito H., Takagi H., Muramatsu M., Oka T. Characterization of a Human Sapovirus Genotype GII.3 Strain Generated by a Reverse Genetics System: VP2 Is a Minor Structural Protein of the Virion. Viruses. 2022;14:1649. doi: 10.3390/v14081649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Oka T., Lu Z., Phan T., Delwart E.L., Saif L.J., Wang Q. Genetic Characterization and Classification of Human and Animal Sapoviruses. PLoS ONE. 2016;11:e0156373. doi: 10.1371/journal.pone.0156373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hansman G.S., Natori K., Oka T., Ogawa S., Tanaka K., Nagata N., Ushijima H., Takeda N., Katayama K. Cross-reactivity among sapovirus recombinant capsid proteins. Arch. Virol. 2005;150:21–36. doi: 10.1007/s00705-004-0406-8. [DOI] [PubMed] [Google Scholar]
- 23.Hansman G.S., Oka T., Sakon N., Takeda N. Antigenic diversity of human sapoviruses. Emerg. Infect. Dis. 2007;13:1519–1525. doi: 10.3201/eid1310.070402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Oka T., Mori K., Iritani N., Harada S., Ueki Y., Iizuka S., Mise K., Murakami K., Wakita T., Katayama K. Human sapovirus classification based on complete capsid nucleotide sequences. Arch. Virol. 2012;157:349–352. doi: 10.1007/s00705-011-1161-2. [DOI] [PubMed] [Google Scholar]
- 25.Li J., Zhang W., Cui L., Shen Q., Hua X. Metagenomic identification, genetic characterization and genotyping of porcine sapoviruses. Infect. Genet. Evol. J. Mol. Epidemiol. Evol. Genet. Infect. Dis. 2018;62:244–252. doi: 10.1016/j.meegid.2018.04.034. [DOI] [PubMed] [Google Scholar]
- 26.Tse H., Chan W.M., Li K.S., Lau S.K., Woo P.C., Yuen K.Y. Discovery and genomic characterization of a novel bat sapovirus with unusual genomic features and phylogenetic position. PLoS ONE. 2012;7:e34987. doi: 10.1371/journal.pone.0034987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Firth C., Bhat M., Firth M.A., Williams S.H., Frye M.J., Simmonds P., Conte J.M., Ng J., Garcia J., Bhuva N.P., et al. Detection of zoonotic pathogens and characterization of novel viruses carried by commensal rattus Norvegicus in New York City. mBio. 2014;5:e01933-14. doi: 10.1128/mBio.01933-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Tsinda E.K., Malasao R., Furuse Y., Gilman R.H., Liu X., Apaza S., Espetia S., Cama V., Oshitani H., Saito M. Complete Coding Genome Sequences of Uncommon GII.8 Sapovirus Strains Identified in Diarrhea Samples Collected from Peruvian Children. Genome Announc. 2017;5:e01137-17. doi: 10.1128/genomea.01137-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mokili J.L., Rohwer F., Dutilh B.E. Metagenomics and future perspectives in virus discovery. Curr. Opin. Virol. 2012;2:63–77. doi: 10.1016/j.coviro.2011.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Escobar-Zepeda A., Vera-Ponce de León A., Sanchez-Flores A. The Road to Metagenomics: From Microbiology to DNA Sequencing Technologies and Bioinformatics. Front. Genet. 2015;6:348. doi: 10.3389/fgene.2015.00348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yinda C.K., Conceição-Neto N., Zeller M., Heylen E., Maes P., Ghogomu S.M., Van Ranst M., Matthijnssens J. Novel highly divergent sapoviruses detected by metagenomics analysis in straw-colored fruit bats in Cameroon. Emerg. Microbes Infect. 2017;6:e38. doi: 10.1038/emi.2017.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Diez-Valcarce M., Castro C.J., Marine R.L., Halasa N., Mayta H., Saito M., Tsaknaridis L., Pan C.Y., Bucardo F., Becker-Dreps S., et al. Genetic diversity of human sapovirus across the Americas. J. Clin. Virol. 2018;104:65–72. doi: 10.1016/j.jcv.2018.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Li W., Dong S., Xu J., Zhou X., Han J., Xie Z., Gong Q., Peng H., Zhou C., Lin M. Viral metagenomics reveals sapoviruses of different genogroups in stool samples from children with acute gastroenteritis in Jiangsu, China. Arch. Virol. 2020;165:955–958. doi: 10.1007/s00705-020-04549-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Yang S., He Y., Zhang J., Zhang D., Wang Y., Lu X., Wang X., Shen Q., Ji L., Lu H., et al. Viral metagenomics reveals diverse viruses in the fecal samples of children with diarrhea. Virol. Sin. 2022;37:82–93. doi: 10.1016/j.virs.2022.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Tohma K., Kulka M., Coughlan S., Green K.Y., Parra G.I. Genomic Analyses of Human Sapoviruses Detected over a 40-Year Period Reveal Disparate Patterns of Evolution among Genotypes and Genome Regions. Viruses. 2020;12:516. doi: 10.3390/v12050516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Dos Anjos K., Lima L.M., Silva P.A., Inoue-Nagata A.K., Nagata T. The possible molecular evolution of sapoviruses by inter- and intra-genogroup recombination. Arch. Virol. 2011;156:1953–1959. doi: 10.1007/s00705-011-1079-8. [DOI] [PubMed] [Google Scholar]
- 37.Hansman G.S., Oka T., Katayama K., Takeda N. Human sapoviruses: Genetic diversity, recombination, and classification. Rev. Med. Virol. 2007;17:133–141. doi: 10.1002/rmv.533. [DOI] [PubMed] [Google Scholar]
- 38.Hansman G.S., Takeda N., Oka T., Oseto M., Hedlund K.O., Katayama K. Intergenogroup recombination in sapoviruses. Emerg. Infect. Dis. 2005;11:1916–1920. doi: 10.3201/eid1112.050722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Makhaola K., Moyo S., Kebaabetswe L.P. Distribution and Genetic Variability of Sapoviruses in Africa. Viruses. 2020;12:490. doi: 10.3390/v12050490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Traoré A.S., Pothier P., Barro N., Ambert-Balay K. Prevalence and Genetic Diversity of Enteric Viruses in Children with Diarrhea in Ouagadougou, Burkina Faso. PLoS ONE. 2016;11:e0153652. doi: 10.1371/journal.pone.0153652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Matussek A., Dienus O., Djeneba O., Simpore J., Nitiema L., Nordgren J. Molecular characterization and genetic susceptibility of sapovirus in children with diarrhea in Burkina Faso. Infect. Genet. Evol. J. Mol. Epidemiol. Evol. Genet. Infect. Dis. 2015;32:396–400. doi: 10.1016/j.meegid.2015.03.039. [DOI] [PubMed] [Google Scholar]
- 42.Murray T.Y., Nadan S., Page N.A., Taylor M.B. Diverse sapovirus genotypes identified in children hospitalised with gastroenteritis in selected regions of South Africa. J. Clin. Virol. Off. Publ. Pan Am. Soc. Clin. Virol. 2016;76:24–29. doi: 10.1016/j.jcv.2016.01.003. [DOI] [PubMed] [Google Scholar]
- 43.Harada S., Okada M., Yahiro S., Nishimura K., Matsuo S., Miyasaka J., Nakashima R., Shimada Y., Ueno T., Ikezawa S., et al. Surveillance of pathogens in outpatients with gastroenteritis and characterization of sapovirus strains between 2002 and 2007 in Kumamoto Prefecture, Japan. J. Med. Virol. 2009;81:1117–1127. doi: 10.1002/jmv.21454. [DOI] [PubMed] [Google Scholar]
- 44.Tuma J.N., Wilkinson A.L., Diop O.M., Jorba J., Gardner T., Snider C.J., Anand A., Ahmed J. Surveillance to Track Progress Toward Polio Eradication—Worldwide, 2019–2020. MMWR. Morb. Mortal. Wkly. Rep. 2021;70:667–673. doi: 10.15585/mmwr.mm7018a2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Conceição-Neto N., Yinda K.C., Van Ranst M., Matthijnssens J. NetoVIR: Modular Approach to Customize Sample Preparation Procedures for Viral Metagenomics. Methods Mol. Biol. 2018;1838:85–95. doi: 10.1007/978-1-4939-8682-8_7. [DOI] [PubMed] [Google Scholar]
- 46.Bolger A.M., Lohse M., Usadel B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics. 2014;30:2114–2120. doi: 10.1093/bioinformatics/btu170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Langmead B., Salzberg S.L. Fast gapped-read alignment with Bowtie 2. Nat. Methods. 2012;9:357–359. doi: 10.1038/nmeth.1923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Nurk S., Meleshko D., Korobeynikov A., Pevzner P.A. metaSPAdes: A new versatile metagenomic assembler. Genome Res. 2017;27:824–834. doi: 10.1101/gr.213959.116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Buchfink B., Xie C., Huson D.H. Fast and sensitive protein alignment using DIAMOND. Nat. Methods. 2015;12:59–60. doi: 10.1038/nmeth.3176. [DOI] [PubMed] [Google Scholar]
- 50.Katoh K., Rozewicki J., Yamada K.D. MAFFT online service: Multiple sequence alignment, interactive sequence choice and visualization. Briefings Bioinform. 2019;20:1160–1166. doi: 10.1093/bib/bbx108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Tatusov R.L., Chhabra P., Diez-Valcarce M., Barclay L., Cannon J.L., Vinjé J. Human Calicivirus Typing tool: A web-based tool for genotyping human norovirus and sapovirus sequences. J. Clin. Virol. Off. Publ. Pan Am. Soc. Clin. Virol. 2021;134:104718. doi: 10.1016/j.jcv.2020.104718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Tamura K., Stecher G., Kumar S. MEGA11: Molecular Evolutionary Genetics Analysis Version 11. Mol. Biol. Evol. 2021;38:3022–3027. doi: 10.1093/molbev/msab120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Muhire B.M., Varsani A., Martin D.P. SDT: A virus classification tool based on pairwise sequence alignment and identity calculation. PLoS ONE. 2014;9:e108277. doi: 10.1371/journal.pone.0108277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Marchler-Bauer A., Derbyshire M.K., Gonzales N.R., Lu S., Chitsaz F., Geer L.Y., Geer R.C., He J., Gwadz M., Hurwitz D.I., et al. CDD: NCBI’s conserved domain database. Nucleic Acids Res. 2015;43:D222–D226. doi: 10.1093/nar/gku1221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Martin D.P., Murrell B., Khoosal A., Muhire B. Detecting and Analyzing Genetic Recombination Using RDP4. Methods Mol. Biol. 2017;1525:433–460. doi: 10.1007/978-1-4939-6622-6_17. [DOI] [PubMed] [Google Scholar]
- 56.Miyazaki N., Song C., Oka T., Miki M., Murakami K., Iwasaki K., Katayama K., Murata K. Atomic Structure of the Human Sapovirus Capsid Reveals a Unique Capsid Protein Conformation in Caliciviruses. J. Virol. 2022;96:e0029822. doi: 10.1128/jvi.00298-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Yan Y., Li Y., Shi W., Kong X., Li H., Zhang Q., Pang L., Jiang L., Liu J., Jin M., et al. An outbreak of gastroenteritis associated with a novel GII.8 sapovirus variant-transmitted by vomit in Shenzhen, China, 2019. BMC Infect. Dis. 2020;20:911. doi: 10.1186/s12879-020-05643-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Bassat Q., Blau D.M., Ogbuanu I.U., Samura S., Kaluma E., Bassey I.A., Sow S., Keita A.M., Tapia M.D., Mehta A., et al. Causes of Death Among Infants and Children in the Child Health and Mortality Prevention Surveillance (CHAMPS) Network. JAMA Netw. Open. 2023;6:e2322494. doi: 10.1001/jamanetworkopen.2023.22494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Sharrow D., Hug L., You D., Alkema L., Black R., Cousens S., Croft T., Gaigbe-Togbe V., Gerland P., Guillot M., et al. Global, regional, and national trends in under-5 mortality between 1990 and 2019 with scenario-based projections until 2030: A systematic analysis by the UN Inter-agency Group for Child Mortality Estimation. Lancet Glob. Health. 2022;10:e195–e206. doi: 10.1016/S2214-109X(21)00515-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Japhet M.O., Famurewa O., Adesina O.A., Opaleye O.O., Wang B., Höhne M., Bock C.T., Marques A.M., Niendorf S. Viral gastroenteritis among children of 0-5 years in Nigeria: Characterization of the first Nigerian aichivirus, recombinant noroviruses and detection of a zoonotic astrovirus. J. Clin. Virol. Off. Publ. Pan Am. Soc. Clin. Virol. 2019;111:4–11. doi: 10.1016/j.jcv.2018.12.004. [DOI] [PubMed] [Google Scholar]
- 61.Fullerton S.W., Blaschke M., Coutard B., Gebhardt J., Gorbalenya A., Canard B., Tucker P.A., Rohayem J. Structural and functional characterization of sapovirus RNA-dependent RNA polymerase. J. Virol. 2007;81:1858–1871. doi: 10.1128/JVI.01462-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Robel I., Gebhardt J., Mesters J.R., Gorbalenya A., Coutard B., Canard B., Hilgenfeld R., Rohayem J. Functional characterization of the cleavage specificity of the sapovirus chymotrypsin-like protease. J. Virol. 2008;82:8085–8093. doi: 10.1128/JVI.00693-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Yokoyama M., Oka T., Kojima H., Nagano T., Okabe T., Katayama K., Wakita T., Kanda T., Sato H. Structural basis for specific recognition of substrates by sapovirus protease. Front. Microbiol. 2012;3:312. doi: 10.3389/fmicb.2012.00312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Vázquez A.L., Alonso J.M., Parra F. Mutation analysis of the GDD sequence motif of a calicivirus RNA-dependent RNA polymerase. J. Virol. 2000;74:3888–3891. doi: 10.1128/JVI.74.8.3888-3891.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Varela M.F., Ouardani I., Kato T., Kadoya S., Aouni M., Sano D., Romalde J.L. Sapovirus in Wastewater Treatment Plants in Tunisia: Prevalence, Removal, and Genetic Characterization. Appl. Environ. Microbiol. 2018;84:e02093-17. doi: 10.1128/AEM.02093-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Su L., Mao H., Sun Y., Yan H., Ge Q., Gong L., Zhang Y. The analysis of the genotype of Sapovirus outbreaks in Zhejiang Province. Virol. J. 2023;20:268. doi: 10.1186/s12985-023-02202-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Kumthip K., Khamrin P., Maneekarn N. Molecular epidemiology and genotype distributions of noroviruses and sapoviruses in Thailand 2000–2016: A review. J. Med. Virol. 2018;90:617–624. doi: 10.1002/jmv.25019. [DOI] [PubMed] [Google Scholar]
- 68.Iizuka S., Oka T., Tabara K., Omura T., Katayama K., Takeda N., Noda M. Detection of sapoviruses and noroviruses in an outbreak of gastroenteritis linked genetically to shellfish. J. Med. Virol. 2010;82:1247–1254. doi: 10.1002/jmv.21791. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The original data presented in the study including the Sapovirus nucleotide sequences are openly available in GenBank under the accession number OR837774-OR837781. Raw sequence data from this study have also been uploaded the NCBI database under the Bioproject number PRJNA1043841.