Abstract
Lung cancer is the most common and fatal malignant disease worldwide and has the highest mortality rate among tumor-related causes of death. Early diagnosis and precision medicine can significantly improve the survival rate and prognosis of lung cancer patients. At present, the clinical diagnosis of lung cancer is challenging due to a lack of effective non-invasive detection methods and biomarkers, and treatment is primarily hindered by drug resistance and high tumor heterogeneity. Liquid biopsy is a method for detecting circulating biomarkers in the blood and other body fluids containing genetic information from primary tumor tissues. Bronchoalveolar lavage fluid (BALF) is a potential liquid biopsy medium that is rich in a variety of bioactive substances and cell components. BALF contains information on the key characteristics of tumors, including the tumor subtype, gene mutation type, and tumor environment, thus BALF may be used as a diagnostic supplement to lung biopsy. In this review, the current research on BALF in the diagnosis, treatment, and prognosis of lung cancer is summarized. The advantages and disadvantages of different components of BALF, including cells, cell-free DNA, extracellular vesicles, and microRNA are introduced. In particular, the great potential of extracellular vesicles in precision diagnosis and detection of drug-resistant for lung cancer is highlighted. In addition, the performance of liquid biopsies with different body fluid sources in lung cancer detection are compared to facilitate more selective studies involving BALF, thereby promoting the application of BALF for precision medicine in lung cancer patients in the future.
Keywords: Lung cancer, bronchoalveolar lavage fluid, extracellular vesicles, liquid biopsy, precision medicine
Introduction
Lung cancer is one of the most common malignancies and accounts for approximately 21% of cancer-related deaths worldwide1. The incidence of lung cancer ranks first and second among all cancers in China among adult males and females, respectively, representing substantial medical and economic burdens2. Lung cancer is also considered one of the most fatal malignancies, with a 5-year survival rate of only 23%, which is largely attributable to late diagnosis. It has been reported that when lung cancer is detected in a localized stage, the 5-year survival rate can reach 60%, while the 5-year survival rate drops to 6% when detected in a distant stage1. Therefore, an effective screening method to prolong the life of lung cancer patients by early diagnosis has always been highly sought. Currently, low-dose computerized tomography (LDCT) screening is prioritized for the early detection of lung cancer, and its implementation is associated with reduced mortality due to improvements in nodule characterization3. However, LDCT alone lacks specificity for distinguishing between benign and malignant nodules, with a false-positive rate of up to 24%, which may lead to overtreatment and cause unnecessary anxiety3,4. Thus, it is necessary to establish improved detection techniques with high sensitivity and specificity in the early stage of lung cancer.
Lung cancer is predominantly categorized into two subtypes [non-small cell lung cancer (NSCLC) and small cell lung cancer (SCLC)]. Over the past two decades, molecular targeted therapies and immunotherapies have achieved encouraging results in treating NSCLC, with unprecedented improvement in patient survival. Unfortunately, not all patients respond to targeted therapies, and for those who do, resistance typically emerges during subsequent treatment5. The main obstacle for immunotherapy is how to select appropriate responders6. SCLC is characterized by a high proliferative capacity, genomic instability, and early metastasis7. Precision medicine for SCLC involves multiple dimensions, such as genetics, epigenetics, proteomics, and the tumor microenvironment. Drug resistance and high tumor heterogeneity are the primary challenges to effective and accurate treatment8. Therefore, to improve the survival rate and prognosis of lung cancer patients, both early diagnosis and the identification of the key tumor characteristics, including the tumor subtype, gene mutation type, and tumor microenvironment, are important. Dynamic detection of relevant indicators during treatment is conducive to monitoring efficacy, optimizing treatment strategies, and improving drug resistance. Tissue biopsy remains the preferred method for detecting these indicators, but tissue biopsy is impeded by the scarcity of tissue samples, sampling frequency, and incomplete representation of the entire tumor bulk9. It is important to note that liquid biopsy may be able to compensate for these defects.
Liquid biopsy, a method for detecting circulating biomarkers in body fluids with genetic information from primary tumor tissues, is a minimally invasive, repeatable, and comprehensive option that can provide personalized and dynamic diagnostic and prognostic information for patient care10. Circulating tumor cells (CTCs), cell-free DNA (cfDNA), extracellular vesicles (EVs), cfRNA, and microRNA (miRNA) are the most commonly used biologic materials in liquid biopsy. Among these biologic materials, several assays for detecting CTCs and cfDNA have been approved by the FDA9. The clinical use of peripheral blood-derived cfDNA as a supplementary specimen for assessing epidermal growth factor receptor (EGFR) mutation status and detecting the T790M mutation when tumor tissue is not readily available has been approved for NSCLC11. Currently, the concept of liquid biopsy has been expanded from blood to bronchoalveolar lavage fluid (BALF), pleural effusions, and other body fluids.
BALF is obtained from the lung through flexible bronchoscopy. BALF is an important bioactive material that includes cellular and non-cellular components which provides information on biological processes occurring in the alveoli, such as inflammation and infection. Over the past few years, BALF has mainly been used to diagnose interstitial and infectious lung diseases12. At the same time, there have been corollary studies focusing on the role of BALF in lung cancer. A review has summarized studies of BALF for lung cancer in analyzing the tumor immune microenvironment and assessing immune-related adverse events of immunotherapy13. Another review described the cell metabolism changes in BALF at various stages of lung cancer14. With the advent of liquid biopsy technology, liquid biopsy using BALF as the medium is expected to become a diagnostic supplement to lung biopsy, but a comprehensive summary on BALF in liquid biopsy for lung cancer is lacking. To fill this gap, we have provided a comprehensive overview of the current research findings based on BALF in lung cancer according to the different liquid components and addressed the advantages and disadvantages of BALF compared to other liquid biopsy techniques using different body fluids. Finally, we discussed the challenges and highlight future directions for implementing these techniques in lung cancer clinical trials.
Overview of the development of BALF
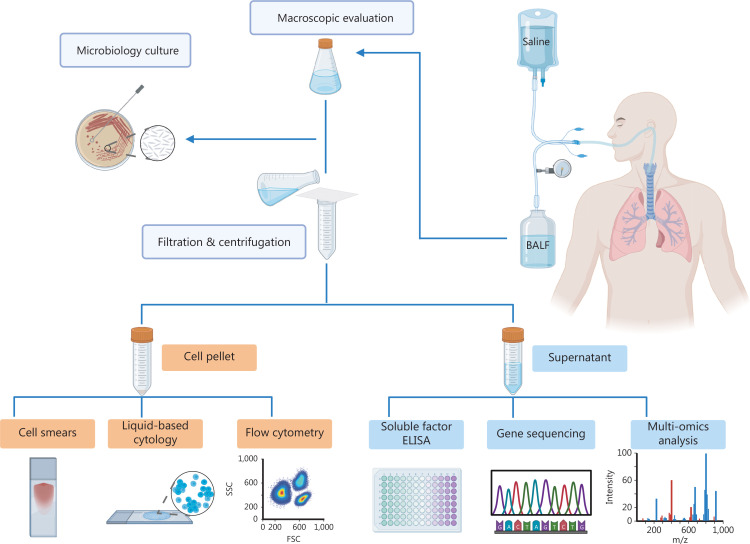
Bronchoalveolar lavage technology has a long history of development worldwide. Initially, bronchoalveolar lavage was used for clinical testing via a rigid bronchoscope, followed by major breakthroughs in the invention of the flexible bronchoscope, which is safer and better tolerated by patients. In 1974, bronchoalveolar lavage was first described as a diagnostic method15. Since then this technique has been progressively utilized to diagnose abnormal lung diseases, including interstitial, infectious, and even malignant diseases12. There are three steps involved in BALF collection. First, the flexible bronchoscope is “wedged” into the target bronchial segment after local anesthesia. Second, 100–200 mL (≤ 300 mL) of normal saline is injected into the alveolar cavity through the bronchoscope, followed by negative pressure suction to recover perfusion. The recollected fluid is termed BALF, and different centers may have personalized modifications for the collection protocol. Third, cells and supernatant are obtained from BALF through pretreatment and centrifugation, which can then be used for further detection (Figure 1).
Figure 1.
Overall workflow of BALF. Normal saline is injected into the alveolar cavity through the flexible bronchoscope, followed by negative pressure suction to recover the perfusate. The recollected fluid is BALF. After pretreatment and centrifugation, BALF is divided into two parts (cells and supernatant). Cells are used for cell smears, liquid-based cytology, and flow cytometry analysis. The supernatant can be subjected to ELISA, gene sequencing, or multi-omics analysis, such as proteomics, metabolomics, and microbiome analysis, allowing in-depth analysis of proteins, nucleic acids, metabolites, and the microbiome. (Created with BioRender.com).
Under normal circumstances, “healthy” BALF is sterile; extra cells, bacteria, or acellular material are aberrant and can be utilized as a diagnostic tool for various lung disorders12. Cellular components of “healthy” BALF include < 0.5% eosinophils, < 5% neutrophils, < 20% lymphocytes, and > 80% macrophages13; however, this pattern is not static. For example, compared to non-smokers, smokers have a significant increase in the absolute cell count, leading to a three-fold higher number of macrophages. Furthermore, smokers have a lower percentage of lymphocytes and the ratio of different subtypes of lymphocytes is also changed.
Application of BALF in the diagnosis and prognosis of lung cancer
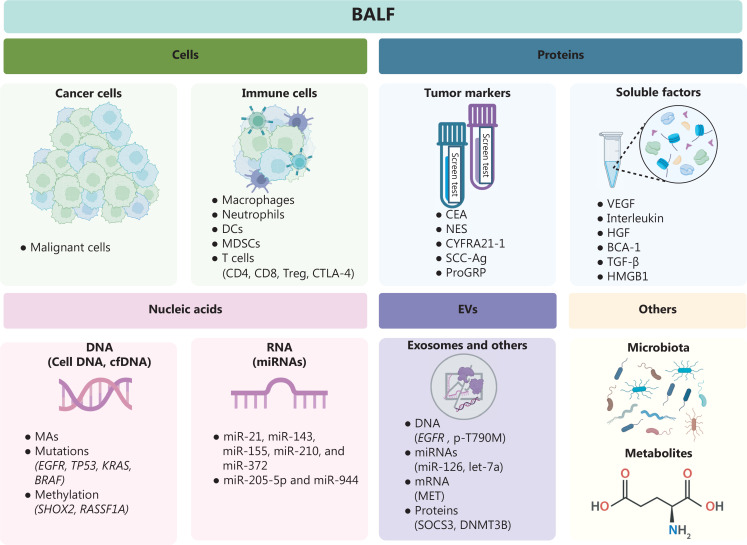
The application of BALF in lung cancer diagnosis and prognosis is still in the research stage. The BALF supernatant obtained by centrifugation is rich in various non-cellular components, including proteins, nucleic acids, and EVs (Figure 2). Because it is possible to distinguish cellular and non-cellular content from proximal airways and lung alveoli, BALF is the ideal “lung liquid biopsy” material for the acquisition of biomarkers pertaining to lung cancer13.
Figure 2.
Application of BALF in lung cancer. BALF contains many different components. The cell components can be divided into cancer and immune cells. The presence of tumor cells reflects the possibility of malignancy. Different proportions of immune cells reflect the local immune microenvironment to some extent. The studies of protein focus on serum tumor markers and some soluble factors. Molecular analysis includes microsatellite alterations, mutations in oncogenes, or tumor suppressors and gene methylation. Multiple miRNAs are specifically expressed in lung cancer. DNA, miRNA, mRNA, and proteins in BALF-EVs also have the potential to serve as biomarkers for lung cancer. Some metabolites or microbiota are also differentially expressed in BALF of lung cancer patients. (Created with BioRender.com).
Cytologic examination
Cancer cells
Cell pellets can be obtained from BALF through centrifugation, allowing for cytologic examination. Conventional methods, such as cell smears, have poor diagnostic performance due to the limitations of smear thickness, overlap, and immune cells. With the advent of liquid-based cytology, the diagnostic rate of lung cancer has improved, but the sensitivity of cytologic examinations based on BALF fluctuates widely in different studies, ranging from 28%–87%14,16–20. The possible reason for such a large fluctuation is that various clinical factors, such as pneumonia, viral infections, and inflammatory changes, lead to severely dysplastic cells, which are difficult to distinguish from malignant cells. Some artificial factors also affect the accuracy of cytological examinations. The effects of different bronchoscopic specimens combined with liquid-based cytology on the early diagnosis of lung cancer have been compared. The sensitivity and specificity of BALF are comparable to bronchoscopic wash cultures and transbronchial lung biopsies; the performance of endobronchial ultrasound-guided transbronchial needle aspiration has the best sensitivity and specificity17. Other studies have shown that the sensitivity of liquid-based cytology of BALF is higher in visible lesions, but can be significantly improved by combining BALF with bronchial brushing when the lesion is not visible16,19. Another study evaluated the performance of peripheral blood-derived CTCs and BALF-derived tumor cells, and found that the sensitivity and specificity of the two samples were similar in distinguishing benign lesions from lung cancer20.
Immune cells and the tumor microenvironment
BALF contains a variety of immune cells that can be analyzed by flow cytometry. Most studies have demonstrated that compared to healthy subjects, lung cancer patients exhibit increased levels of neutrophils and T cells in BALF13,21. Further analysis of T-cell subtypes in BALF revealed an increase in CD8+ T cells, while the CD4:CD8 ratio was decreased13. In addition, multiple studies have indicated that BALF from lung cancer patients has an immunosuppressive microenvironment that is characterized by an elevated proportion of Treg cells and CTLA-4+ Treg cells, as well as polarization of macrophages toward the M2 population13,21–23. Compared to NSCLC patients, the proportion of IL10+CD206+CD14+ M2-like macrophages are elevated in BALF from SCLC patients, positively correlated with tumor stage, and negatively correlated with survival time24.
With the development of immunotherapy, immune checkpoint inhibitors (ICIs) have been used to treat various advanced tumors, including NSCLC. PD-1-PD-L1 pathway blockers have been approved as first-line therapy for advanced NSCLC, with approximately one-half of patients achieving a response to ICIs and experiencing longer survival rates, ranging from 20%–30%13. Therefore, it is essential to analyze the tumor microenvironment to identify which patients will benefit from immunotherapy. The tumor microenvironment includes tumor, stromal, and immune cells, as well as related cytokines. Tumor biopsies available for tumor microenvironment analysis are very limited, so BALF may serve as a superior source of information. Compared to peripheral blood, BALF is more proximal or “vicinal” to the tumor cells in the lung, so BALF can provide more direct information on the local immune status in lung tissue13. The detection of PD-L1 expression by cells derived from BALF is more consistent with paired tissue biopsies, which in turn is better than pleural effusion cells25. A tumor microenvironment analysis based on BALF showed that the expression of CXCL9 in BALF of NSCLC patients who responded to nivolumab is higher than non-responders26. BALF can also be used to evaluate immune-related adverse events of immunotherapy13, which manifests in the lung as pneumonia, and is referred to as checkpoint inhibitor pneumonitis27. The number of central memory T cells is increased in BALF of patients with checkpoint inhibitor pneumonitis and the expression of CTLA-4 and PD-1 is decreased in BALF Tregs compared to patients without this complication28. Therefore, dysregulation of immune cell subsets in BALF may serve as a potential target for the treatment of immune-related adverse events.
There are significant differences in the immune cell profile between BALF and peripheral blood. The same cells account for a high proportion in BALF and may be very low in peripheral blood21,23,29. The consistency between the immune cell profile based on BALF and the tumor-infiltrating lymphocytes detected in tumor resection tissues also differs. A study has shown that a higher proportion of Treg cells is present in the BALF of adenocarcinoma patients22, which is consistent with the results directly obtained from tumor tissues30. However, another study comparing the immune cell subtypes of lymphocytes, dendritic cells, myeloid suppressor cells, and macrophages in BALF and tumor tissues showed that except for myeloid-derived suppressor cells, the proportion of other cells in the two samples had no significant correlation31. The above disparities may be because BALF reflects the overall immune environment of the lung, and various contributing factors, including smoking and chronic obstructive pulmonary disease (COPD), ultimately affect the proportion of immune cells, while tumors principally cause a local immune response.
Tumor-related proteins
Tumor markers
There are some common serum tumor markers that have been used in the clinical diagnosis of lung cancer. Carcinoembryonic antigen (CEA) is a glycoprotein complex expressed on the surface of tumor cells. Compared with a control group, CEA expression in the BALF of lung cancer patients is significantly upregulated32–34. Numerous studies have reported that the level of CEA expression in the BALF of lung cancer patients is higher than serum, so the sensitivity of detecting CEA in BALF is higher35,36. By comparing the expression of CEA in BALF and serum among patients with lung cancer and pneumonia, and healthy individuals, it was found that CEA expression in serum could not discriminate between lung cancer and pneumonia patients, while these levels were significantly higher in BALF from lung cancer patients than BALF from pneumonia patients37. Another study showed that CEA expression in BALF was significantly increased in non-smokers with tumors, while no significant difference was observed in smokers with tumors38. Another study demonstrated non-significant variations in the CEA concentration in BALF from patients with various pathologic types34. Neuron-specific enolase (NSE) is a neuronal form of the glycolytic enzyme, enolase. NSE mainly exists in neural and neuroendocrine tissues, as well as tumors derived from these tissues39. SCLC is classified as a neuroendocrine tumor and serum levels of NSE are elevated in 66%–81% of patients with SCLC40. The NSE content in BALF samples from tumor groups has been shown to be significantly higher than BALF from control groups32,34,41,42. However, unlike NSE in serum, most studies (with the exception of one study34) have indicated no difference in the expression of BALF-derived NSE in SCLC and NSCLC32,41,42. The expression of NSE between BALF and serum was not shown to be significantly different, but simultaneous detection of NSE from two sources could improve the sensitivity42. Cytokeratin 19 fragment antigen 21-1 (CYFRA21-1) is a fragment of cytokeratin 19 and serves as the most sensitive serum marker of NSCLC, especially in patients with lung squamous cell carcinoma (LSCC)43; however, there has been ongoing debate regarding the diagnostic value of CYFRA21-1 in BALF. An early study concluded that CHFRA21-1 had poor diagnostic value44, while subsequent studies reported improved diagnostic performance32,34. In addition to the tumor markers mentioned above, research has been conducted on other markers, such as squamous cell carcinoma antigen and precursor gastrin-releasing peptide33,35. Based on existing reports, however, the detection of tumor markers in BALF is insufficient for the diagnosis of lung cancer, but combined detection can significantly improve the sensitivity of diagnosis32,34.
Soluble factors
In addition to common tumor markers, BALF also contains several soluble factors that can be detected by ELISA. Among the soluble factors, the vascular endothelial growth factor (VEGF) family has been extensively studied. The content of VEGF-A is notably elevated in the BALF of lung cancer patients and higher than plasma45,46. VEGF-C is another member of the VEGF family, the expression of which in the peripheral blood of lung cancer patients is significantly increased47. In the BALF of LSCC patients, VEGF-C expression has been shown to be significantly increased48. VEGF-D expression is also upregulated in the BALF of patients with LSCC49. Indeed, there are several other soluble factors that warrant consideration. Among these findings, transforming growth factor beta (TGF-β) expression has been shown to be upregulated in the BALF of patients with primary lung cancer50. In a subsequent study of NSCLC, researchers also showed that the level of TGF-β in the BALF of cancer patients was higher than healthy controls and positively correlated with the overall survival of patients with stage IIIB NSCLC51. In the same study high mobility group box 1 in BALF was associated with metastasis and overall survival of patients with stage IIIB NSCLC51. A series of studies have indicated that the expression of various soluble factors in BALF, such as B-cell-attracting chemokine 1(BCA-1), hepatocyte growth factor (HGF), interleukin 18 (IL-18), IL-22, IL-27, and IL-29, are abnormal in NSCLC patients, some of which are related to disease progression and response52–55. NSCLC patients with higher sRANKL52 or BCA-153 levels in BALF were associated with poorer survival. Another study also demonstrated a significant rise in the level of IL-27 expression in the BALF of early NSCLC patients56. Researchers have only demonstrated relevant evidence for the above indicators but have not conducted in-depth studies, which is insufficient for clinical practice. In a recent study involving patients with lung nodules < 8 mm in size that were suspicious for cancer, the BALF was assessed for combination indices (VEGF, TGF-β, and HGF) and compared to the histopathologic findings. The accuracy of detecting malignant and benign nodules was 71.4% and 74.5%, respectively57. This finding may be used for preliminary screening of small nodules for which histopathologic specimens could not be obtained.
Proteomics
Proteomics technology has also been applied to BALF research. The relevant clinical studies using this method are summarized in Table 1. Matrix-assisted laser desorption/ionization time of flight (MALDI-TOF) mass spectrometry was first reported for detecting BALF in NSCLC patients, suggesting that BALF is a suitable biofluid for screening biomarkers of NSCLC58. Subsequently, bidimensional polyacrylamide gel electrophoresis (2D-PAGE) and MALDI-TOF/TOF were utilized to analyze the BALF of NSCLC patients; seven specific proteins were identified (CTSD, ALDO A, FBP1, ERZ, AKR1B10, TKT, and SELENBP1). Further validation via western blotting of paired BALF proteins showed that only the AKR1B10 protein was upregulated in the NSCLC group59. The proteome of BALF was analyzed by 2D-PAGE and mass spectrometry (MS). In combination with bead-based immunoassays, a panel of proteins (APOA1, CO4A, CRP, GSTP1, and SAMP) was screened out. The sensitivity and specificity of this panel were 95% and 81%, respectively, indicating an effective assay for lung cancer diagnosis. This study also identified two proteins, STMN1 and GSTP1, which can be used to distinguish SCLC from NSCLC with a sensitivity and specificity of 90% and 57%, respectively61. Proteomics quantification of BALF in patients with primary lung adenocarcinoma was performed by liquid chromatography–mass spectrometry (LC–MS). Thirty-three proteins were identified to be consistently overexpressed in all samples, some of which were shown to be associated with lung cancer progression62. Proteomic analysis was conducted for the first time using sequential windowed acquisition of all theoretical fragment ion mass spectra (SWATH) MS on the BALF of patients with lung adenocarcinoma. A total of 44 specific and highly expressed proteins were screened out, among which HPT, CO4A, and GTSP1 were consistent with previous reports63. Label-free MS was performed to analyze the proteome of BALF in NSCLC patients. Compared to patients without tumors, significant changes in protein abundance and molecular function were found in NSCLC patients. The protein expression profile was also different between adenocarcinoma and LSCC patients. According to proteomic analysis, four proteins (cystatin C, TIMP-1, lipocalin-2, and HSP70/HSPA1A) were significantly elevated in NSCLC patients, and the expression of the first three proteins was further verified in plasma samples64. An in-depth proteomics method for BALF analysis was further developed that effectively detected both high- and low-abundance proteins, enabling the identification of the largest number of BALF proteins67. The control samples for the above studies were all from patients without tumors. In one study, however, BALF was collected from the tumor-bearing and healthy lungs of the same patient. According to tandem mass tag (TMT)-based quantitative MS analysis, four proteins (DSTN, RNPEP, CNDP2, and CA1) were shown to be highly expressed in tumor-bearing lungs, and these proteins were validated by tissue immunohistochemistry66. According to the expression of prominent transcription factors, SCLC can be categorized into four subtypes. ASCL1 and NEUROD1 are associated with neuroendocrine-high (NE-high) markers. All five patients in the study were NE-high, and further bioinformatics analysis revealed that CNDP2 and RNPEP are potential markers for ASCL1-high and NEUROD1-high subtypes of SCLC66.
Table 1.
Proteomic analysis based on BALF for lung cancer
| Study subjects | Proteomic analysis | Validation subjects | Validation method | Specific proteins (numbers/name) | Comments | Ref. |
|---|---|---|---|---|---|---|
| NSCLC (n = 6) and control (n = 16) | MALDI-TOF/TOF MS | NA | NA | 2/HTN3 and S100A12 | HTN3 and S100A12 were significantly upregulated in the BALF of NSCLC patients. | 58 |
| COPD (n = 15), NSCLC (n = 15), NSCLC with COPD (n = 15), and control group with neither NSCLC nor COPD (n = 15) | 2D-PAGE and MALDI-TOF/TOF MS | Paired BALF proteins | Western blot | 1/AKR1B10 | Compared with the control group, the expression of AKR1B10 was increased in the NSCLC group, while no difference was detected in the COPD group. | 59 |
| Lung adenocarcinoma (n = 4) and benign lung diseases (n = 4) | iTRAQ labeling and LC-MS/MS | Lung adenocarcinoma (n = 18), lung SQCC (n = 9), SCLC (n = 6), and benign lung diseases (n = 6) | ELISA in BALF samples | 1/Napsin A | The protein level of Napsin A was significantly increased in BALF of adenocarcinoma. | 60 |
| Lung cancer (n = 139) and benign lung diseases (n = 49) | 2D-PAGE and MS | Lung cancer (n = 204) and benign lung diseases (n = 48) | Bead-based immunoassays in BALF samples. | 5/APOA1, CO4A, CRP, GSTP1, SAMP | The levels of these five proteins led to a lung cancer diagnostic panel with 95% Sn and 81% Sp. | 61 |
| 2/STMN1 and GSTP1 | The quantification of these two proteins differentiated NSCLC and SCLC patients with 90% Sn and 57% Sp. | |||||
| Lung adenocarcinoma (n = 8) and non-tumor patients (n = 8) | LC-MS/MS | NA | NA | 33/S100-A8, TYMP, annexin A1, annexin A2, and TG2 | These five proteins of the DEPs were associated with lung cancer progression in previous studies and may be potential biomarkers for lung adenocarcinoma. | 62 |
| Lung adenocarcinoma (n = 12) and non-tumor patients (n = 10) | SWATH DIA MS† | NA | NA | 44/HPT, CO4A, GTSP1 | Three of the DEPs that we suggest as potential biomarkers were consistent with previous studies using BALF samples. | 63 |
| NSCLC (n = 26) and non-tumor patients (n = 16) | Label-free MS | NSCLC (n = 46) and non-tumor patients (n = 26) | ELISA in plasma | 3/Cystatin-C, Lipocalin-2, TIMP-1 | These three proteins were upregulated in BALF of NSCLC patients, and the results were confirmed in plasma. | 64 |
| Lung adenocarcinoma (n = 5), lung SQCC (n = 4), SCLC (n = 4), and benign lung diseases (n = 3) | MS-based quantitative N-glycoproteomic | Lung adenocarcinoma (n = 18), lung SQCC (n = 9), SCLC (n = 6), and benign lung diseases (n = 6) | ELISA in BALF | 1/Periostin | The expression of periostin was increased in BALF from all three tumor types, which is in agreement with the observation of elevated serum levels of this protein in a previous study. | 65 |
| SCLC (n=5): tumor-bearing lungs and non-tumor lungs | TMT-based quantitative MS | Paired tumor tissues | IHC in SCLC tissues | 4/DSTN, RNPEP, CNDP2, and CA1 | These four proteins were upregulated in BALF from tumor-bearing lungs and tumor tissues. | 66 |
| NA | SCLC-CellMiner resource‡ | 2/CNDP2 and RNPEP | 1. CNDP2 and RNPEP appeared to be potential indicators for ASCL1-high and NEUROD1-high subtypes of SCLC. 2. CNDP2 was found to be positively correlated with responses to etoposide, carboplatin, and irinotecan. |
NSCLC, non-small-cell lung cancer; MALDI-TOF/TOF, matrix-assisted laser desorption/ionization time of flight; MS, mass spectrometry; NA, not available; COPD, chronic obstructive pulmonary disease; 2D-PAGE, two-dimensional polyacrylamide gel electrophoresis; iTRAQ, isobaric tags for relative and absolute quantitation; LC-MS/MS, liquid chromatography tandem mass spectrometry; SQCC, squamous cell carcinoma; SCLC, small cell lung cancer; SWATH, sequential windowed acquisition of all theoretical fragment ion mass spectra; DIA, data-independent acquisition; ELISA, enzyme-linked immunosorbent assay; Sn, sensitivity; Sp, specificity; DEPs, differentially expressed proteins; TMT, tandem mass tags; IHC, immunohistochemistry.
†Indicates the analysis of BALF proteins by LC–MS/MS combining a simple sample pre-treatment and a DIA approach.
‡Indicates a bioinformatic resource for SCLC cell line genomics and pharmacology based on genomic signatures (https://discover.nci.nih.gov/rsconnect/SclcCellMinerCDB/).
Given that the majority of the secreted proteins in BALF are glycoproteins, N-glycoproteins were detected through the use of isobaric tags for relative and absolute quantitation (iTRAQ) labeling and LC–MS. Eight glycoproteins were highly expressed in the BALF of lung adenocarcinoma. Among these glycoproteins, Napsin A has been reported to be highly expressed in lung adenocarcinoma tissues68. Therefore, the expression of Napsin A was further validated in BALF samples by ELISA in another independent cohort, and a significant increase in Napsin A was found in the BALF of lung adenocarcinoma patients60. The MS-based quantitative N-glycoproteomic technique was also used to analyze BALF samples, revealing periostin in all samples from lung cancer patients. Validation of the screened periostin with ELISA in another 39 BALF samples also yielded the same conclusion65.
The above studies collectively indicate that proteomics-based analysis of BALF can be used to screen potential diagnostic markers for lung cancer, especially when combined with other experimental methods for validation to further narrow the range of target proteins. However, owing to the diversity of proteomic techniques, validation populations and sample types, the different selection of control groups, and the limited sample size of the studies, the clinical translation of proteomics technology on BALF analysis has not been established.
Molecular analysis
DNA
Common genetic alterations of tumor-related molecules include microsatellite alterations, mutations in oncogenes or tumor suppressors, and gene hypermethylation. Initially, researchers collected BALF cells for molecular analysis. With the advances in liquid biopsy technology, research on BALF supernatants has gradually increased. The clinical studies pertaining to BALF genetic analysis-based lung cancer detection are listed in Table 2.
Table 2.
Genetic analysis detection based on BALF for lung cancer
| Study population of BALF | Genetic mutations | Source of origin | Methods | Sn/Sp (%) | Comparison (Sn/Sp) (%) | Comments | Ref. |
|---|---|---|---|---|---|---|---|
| Lung adenocarcinoma (n = 33) |
EGFR 33 | Cell pellet |
Sanger sequencing | 9.09/NA | Pleural effusion: 13.3/NA (unpaired) |
BALF cell NGS had higher sensitivity than Sanger sequencing, and therefore could be a diagnostic supplement for better characterization of lung cancer; BALF had higher sensitivity than pleural effusion. | 69 |
| NGS | 78.8/NA | Pleural effusion: 53.3/NA (unpaired) |
|||||
| Lung adenocarcinoma (n = 20) |
EGFR 12 vs. WT 8 | CfDNA | PNA-PCR and PANAMutyper† | 91.7/100 | Bronchial washing samples: 18.2/100 (paired) |
BALF cfDNA was useful in detecting EGFR mutation and identifying T790M mutations; BALF was more useful than bronchial washing samples. | 70 |
| Lung adenocarcinoma (n = 26) |
EGFR 13 vs. WT 13 | Mixture‡ | PCR | 92.3/100 | Peripheral blood: 38.5/100 (paired) |
BALF had much higher diagnostic accuracy than blood for EGFR mutation detection; two additional mutations were identified in BALF compared with tissue biopsy. | 71 |
| Malignant (n = 31) and benign (n = 9) | Mutation profiling | CfDNA | NGS | 67.7/77.8 | Bronchoscopy biopsy: 23.1/100 (unpaired) Bronchial brushing cytology: 9.7/100 (unpaired) Lavage cytology: 3.1/100 (unpaired) Plasma methylation: 66.7/71.4 (unpaired) |
BALF cfDNA methylation analysis had better sensitivity in distinguishing small malignant tumors (≤ 2 cm) from benign nodules than mutation analysis; both were better than conventional methods. |
72
|
| Malignant (n = 27) and benign (n = 21) | Methylation profiling | Bisulfite sequencing and RT-PCR | 81.5/81.0 |
||||
| Lung cancer stage I-II (n = 18) | Mutation profiling | CfDNA | Tumor naïve approach | 66.7/NA | Plasma cfDNA 11.1/NA (paired) |
BALF cfDNA analysis was more sensitive than plasma cfDNA for detecting lung cancer-derived mutations in stage I-II disease. | 73 |
| Lung cancer stage I-II (n = 22) | Tumor-informed analysis | 77.3/NA | Plasma cfDNA: 45.5/NA (paired) |
||||
| Lung cancer (n = 17) and non-cancer (n = 16) | Genomic classifier | 64.7/100 | BAL cytology: 11.8/100 (paired) |
BALF cfDNA analysis was useful for the diagnosis of lung cancer. | |||
| Lung cancer (n = 31) | Methylation of CDH1, APC, RASSF1A, MGMT, P16, GSTP1, ARF, and RAR-β2 | Cell pellet | RT-PCR and RT-MSP | 67.7/NA | NA | Aberrant DNA methylation was detected in the majority of lung cancer patients. | 74 |
| Lung cancer (n = 123); benign lung diseases (n = 112); malignancies in other systems (n = 18) | Methylation of SHOX2 and/or RASSF1A | Cell pellet | RT-PCR and Sanger sequencing |
SHOX2: 64.2/92.3; RASSF1A: 50.4/96.2; SHOX2 and RASSF1A: 71.5/90.0 |
BALF cytology: 5.7/99.2 | BALF cell methylation analysis (SHOX2 and RASSF1A) improved diagnostic accuracy for lung cancer, and was superior to cytology diagnosis. |
75 |
| Lung cancer (n = 284); benign lung diseases (n = 35); malignancies in other systems (n = 3) |
Methylation of SHOX2 and RASSF1A | Cell pellet | RT-PCR and Sanger sequencing | Total: 81/97.4 Stage I: 85.7/NA |
Total Cytology: 68.3/97.4; Serum CEA: 30.6/100 Stage I Cytology: 46.4/NA Serum CEA: 10.7/NA |
BALF cell methylation analysis had relatively satisfactory diagnostic accuracy for lung cancer, especially at early stage, and was complementary to cytology diagnosis. | 76 |
Sn, sensitivity; Sp, specificity; NA, not available; NGS, next-generation sequencing; WT, wild type; PCR, polymerase chain reaction; PNA-PCR, peptide nucleic acid-mediated PCR; RT-PCR, real-time PCR; RT-MSP, real-time methylation-specific polymerase chain reaction.
†Indicates PANAMutyper™ R EGFR kit with PNA clamping-assisted fluorescence melting curve analysis was used for mutation detection and genotyping.
‡Indicates non-centrifuged BALF.
In 1999 researchers used the molecular analysis results of tumor tissues as a reference to detect the genetic alterations in the corresponding BALF cell-derived DNA and found that the detection rates of microsatellite alterations, P53 and KRAS mutations, and P16 methylation were 13.6% (3/22), 39.3% (11/28), 33.3% (4/12), and 63.2% (12/19), respectively77. The detection of EGFR mutations was better than detection of P53 or KRAS mutations 71,78,79. The most common mutation in NSCLC involves the EGFR gene. It has been shown that BALF has higher sensitivity than pleural fluids for detecting EGFR mutations in lung adenocarcinoma patients based on next-generation sequencing (NGS)69. The sensitivity of BALF-derived cfDNA for detecting EGFR mutations was 91.7% in lung adenocarcinoma patients, which was better than paired bronchial washing samples (18.2%)70. In 13 lung adenocarcinoma patients with confirmed EGFR mutations, BALF identified 2 additional mutations compared with tissue biopsy and had higher sensitivity (92.3%) than peripheral blood samples (38.5%) for detecting EGFR mutations71. These results suggest that EGFR mutation detection in BALF may become a convenient and sensitive method for the diagnosis of NSCLC.
In addition to determining the status of EGFR mutations before treatment, monitoring changes in drug resistance gene mutations during treatment is also critical. After EGFR-tyrosine kinase inhibitor (TKI) treatment, most patients with EGFR mutations will develop secondary mutations 9–14 months later, among which the T790M mutation is most common78–80. At present studies involving detection of the T790M mutation in cfDNA mainly focus on serum/plasma81,82, but there are several studies showing that the T790M mutation can also be identified in BALF. A patient with lung adenocarcinoma developed a T790M mutation 3 months after initiation of gefitinib. The detection results of BALF-derived cfDNA were consistent with tumor tissues, but the mutation could not be detected in bronchial washing samples70. Two additional mutations were detected in BALF samples compared to the corresponding lung adenocarcinoma tissue samples, one of which was the T790M mutation79.
In addition to EGFR mutations, TP53, KRAS, and BRAF were also found in BALF cfDNA from lung cancer patients, and the genomic profile had a sensitivity of 67.7%72. Analysis of tumor-associated gene mutation profiles using a tumor-naïve or -informed approach showed that BALF cfDNA exhibited higher sensitivity than plasma cfDNA for detecting lung cancer-derived mutations in stage I-II disease. A diagnostic classifier for lung cancer-derived mutations was then developed with a sensitivity of 64.7%, which was superior to BALF cytology (11.8%)73. The above results indicate that BALF, as a liquid biopsy medium, has good sensitivity in the detection of gene mutations from lung cancer and may be used as a diagnostic supplement for tissue biopsy.
Epigenetic studies have revealed a close association between DNA methylation and the occurrence and progression of lung cancer. Aberrant methylation of gene promoters has been used as a biomarker for diagnosing lung cancer83. Methylation typically occurs in the early stage of lung cancer and has been extensively studied in liquid biopsy84. Methylation of 8 gene promoters (CDH1, APC, MGMT, RASSF1A, GSTP1, P16, RAR-β2, and ARF) in BALF cell-derived DNA from 31 patients with primary lung cancer was examined and abnormal methylation of these genes was shown in 67.7% of the samples74. Aberrant methylation of SHOX2 and RASSF1A genes has also been widely reported in various studies on lung cancer. The clinical specimens include tumor tissues, plasma, pleural effusions, and bronchial aspirates83,85–87. In recent years studies based on BALF samples have also begun to increase. The combined detection of SHOX2 and RASSF1A methylation improves the detection rate of lung cancer compared with individual assessment or cytologic examination of BALF. Interestingly, SHOX2 methylation has a histologic subtype preference, with optimal results in SCLC, followed by LSCC and adenocarcinoma75. This finding was consistent with previous studies carried out on plasma and bronchial aspirates83,85. Another study yielded similar results. At all stages of lung cancer, the sensitivity of the BALF-derived SHOX2 and RASSF1A methylation assay was higher than cytology and serum CEA, especially in stage I, with a sensitivity of 85.7%. Therefore, BALF-based methylation analysis of SHOX2 and RASSF1A can be used as an effective complement to cytological examination, especially in the early stage of lung cancer88. The results of this study also showed that the SHOX2 and RASSF1A methylation assay has a preference for histological subtypes, and the conclusion is consistent with previous findings that the sensitivity is highest in SCLC76. Another study investigated the correlation between gene methylation in BALF and patient prognosis and found that the methylation of RASSF1A was associated with shorter progression-free survival in NSCLC patients treated with pemetrexed88. The above methylation detection was based on BALF cell-derived DNA. Another study performed methylation profiling of cfDNA from BALF. The findings indicated that the methylation analysis of BALF-cfDNA possessed high sensitivity in distinguishing small malignant tumors (≤ 2 cm) from benign nodules72.
In summary, BALF-based genetic analysis exhibits good sensitivity with specific methods for the identification of lung cancer. For precision treatment, BALF also has an auxiliary role in the detection of secondary drug-resistant mutations.
RNA
MicroRNAs, small non-coding RNA composed of 20–24 nucleotides, are an important part of liquid biopsy. In fact, miRNAs have been widely reported in blood and other body fluids but are less studied in BALF. In one study researchers analyzed the expression of a specific miRNA panel (miR-21, miR-143, miR-155, miR-210, and miR-372) in BALF from NSCLC patients and found that the diagnostic sensitivity was 85.7%, which was higher than sputum (67.8%)89. In another study, miR-205-5p and miR-944 were used as a prediction model to distinguish LSCC and adenocarcinoma. The accuracy of miR-205-5p and miR-944 in BALF samples was 95.7%, which approximated frozen tissue (96.6%) and paraffin-embedded formalin-fixed tissue (96.4%) and better than cytology (68.1%)90. The above results indicate that this prediction model may have clinical value in differentiating LSCC from adenocarcinoma in tumor tissues and BALF liquid biopsies. Specific miRNAs (miRNA-34a, miRNA-141, miRNA-143, and miRNA-217) that regulate tumor suppressor genes (RARβ and FHIT) were subsequently detected in the BALF of patients with LSCC, but the results showed that the expression of these miRNAs may not have diagnostic value for LSCC91. Based on the above findings, the detection of BALF-derived miRNAs may be a potential means for lung cancer diagnosis in some cases; however, few studies have explored this possibility. Among the studies that have studied BALF-derived miRNAs for lung cancer diagnosis, the miRNAs screened have been different and may require a larger sample size, further screening, and verification.
EVs
EVs are vesicles with lipid bilayer membrane structures that are released by cells into the extracellular matrix and can be divided into exosomes, microvesicles and apoptotic bodies according to the origin and size. A variety of cells secrete EVs into BALF, including bronchial epithelial cells, alveolar macrophages, and endothelial cells. EVs are rich in various components, including DNA, RNA, and proteins. Tumor cells also secrete a large number of EVs. The cargos in EVs reflect information from parental tumor cells, so EVs can be used as biomarkers to assist in the diagnosis of some tumors92. To avoid interference from viruses, proteins, or other aggregates, high-purity EVs are a prerequisite for further analysis. Common methods for isolating EVs include differential centrifugation, density gradient centrifugation, size-exclusive chromatography, and commercial kits. BALF contains many soluble proteins, and the methods for EV isolation from BALF are still in the early stage of development93. The current studies of lung cancer based on BALF-EVs are shown in Table 3.
Table 3.
BALF-derived EVs for lung cancer
| Study population | Type | Methods of isolation | Cargos | Targets | Sn/Sp (%) | Comparison (Sn/Sp) (%) | Comments | Ref. |
|---|---|---|---|---|---|---|---|---|
| NSCLC (n = 23): EGFR mutation 14 vs. WT 9 | EVs | Differential centrifugation† 1. 1000 g × 15 min 2. 200,000 g × 1 h |
DNA |
EGFR mutation |
100/ 100 |
BALF cfDNA: 71.4/100. Plasma EV DNA: 55.0/NA (unpaired) Plasma cfDNA: 30.0/NA (unpaired) |
1. Liquid biopsy with EVs DNA was more sensitive compared to using cfDNA. 2. Sensitivities of BALF samples were significantly higher compared to plasma samples for both cfDNA and EVs DNA. | 94 |
| Patients resistant to EGFR-TKIs (n = 9) |
T790M mutation | 55.5/NA | BALF cfDNA: 33.3/NA Re-biopsy tissue: 22.2/NA (paired) |
BALF-EVs DNA had higher efficiency for detecting T790M mutation compared to tissue re-biopsy. | ||||
| Lung cancer (n = 137): EGFR mutation 54 vs. WT 83 | EVs | Differential centrifugation 1. 1000 g × 10 min 2. 200,000 g × 1 h |
DNA | EGFR mutation | 75.9/86.7 |
T1: 40/NA; T4: 100/NA N0: 63.3/NA; N3: 100/NA M0: 43.5/NA; M1: 100/NA |
BALF EVs-based EGFR genotyping had increased sensitivity with escalating TMN stage and even more useful when biopsy was not feasible. | 95 |
| NSCLC III-IV (n = 224): EGFR mutation 93 vs. WT 131 | EVs | Differential centrifugation 1. 1000 g × 10 min 2. 200,000 g × 1 h |
DNA | EGFR mutation | 97.8/96.9 | Plasma cfDNA: 48.5/86.3 (unpaired) |
BALF-derived EVs testing in advanced NSCLC was rapid with high accuracy, serving as an alternative for tissue biopsy for guiding prompt EGFR-TKI treatment. | 96 |
| NSCLC (n = 30) and non-tumor patients (n = 75) | Exosomes | Differential centrifugation 1. 500 g × 10 min 2. 17,000 g × 20 min 3. 120,000 g × 90 min |
miRNA | miRNA profiles | NA | NA | 1. Exosome levels were significantly higher in plasma than in BALF samples in patients with and without cancer. 2. The concentrations of plasma miRNAs was higher in tumor patients when comparing non-tumors but were similar for BALF miRNA. |
97 |
| Lung adenocarcinoma (n = 13) and benign lung diseases (n = 15) | Exosomes | Commercial kit (ExoQuick Exosome Precipitation Solution) | miRNA | miR-126/ Let-7a |
NA | NA | Exosomal miRNA-126 and Let-7a levels were higher in the BALF of adenocarcinoma than benign BALF. | 98 |
| NSCLC patients with EGFR 19del mutation (n = 10) | Exosomes | Differential centrifugation 1. 300 g×10 min 2. 16,500 g × 20 min 3. 120,000 g × 70 min |
mRNA | MET | NA | NA | MET was detected in plasma- and BALF‡-derived exosomes in metastasis without a significant difference between the two body fluids. |
99 |
| NSCLC (n = 22) and healthy volunteers (n = 12) | EVs | Differential centrifugation 1. 500 g × 10 min 2. 2,500 g × 12 min 3. 17,000 g × 160 min |
Protein | SOCS3 | NA | NA | The protein level of SOCS3 in the BALF was significantly reduced, independent of histopathologic type and smoking status. | 100 |
| Lung cancer (n = 12) and non-lung cancer (n = 12) | EVs | Differential centrifugation 1. 3000 g × 20 min 2. 12,000 g × 1 h 3. 100,000 g × 2 h |
Proteome | DNMT3B | NA | NA | 1. Proteome complexity of BALF-EVs correlated with lung cancer stage IV and mortality within 2 years of follow-up. 2. DNMT3B complex was significantly up-regulated in tumor tissue and BALF-EVs. |
101 |
Sn, sensitivity; Sp, specificity; NSCLC, non-small-cell lung cancer; WT, wild type; EVs, extracellular vesicles; NA, not available; TKIs, tyrosine kinase inhibitors.
†Indicates a separation technique separating and removing components other than EVs from BALF in a stepwise manner, and all the centrifugation steps are performed at 4°C.
‡Plasma and BALF were collected at the time of primary diagnosis and after the treatment with icotinib/gefitnib within a follow-up period of 3–6 months.
BALF-derived EVs are a reliable source for identifying gene mutations. Researchers compared BALF-EV DNA and tissue DNA based on targeted NGS and found that although the yield and coverage depth of BALF-EV DNA were not as good as tissue DNA, the mutation concordance between the two samples could reach 81%102. Compared with tissue biopsy, EGFR genotyping based on BALF-EVs may be more sensitive. BALF-EV DNA was able to detect not only all of the EGFR mutations detected in tissue samples but also six additional cases95. BALF-EV DNA can also be used to detect the T790M mutation in patients with secondary resistance to EGFR-TKIs, even more efficiently and conveniently than tissue re-biopsy94. In addition, the sensitivity of EGFR genotyping based on BALF-EVs was increased with TMN grading and even reached 100%95. This is contrary to the detection results of BALF cfDNA, which is more sensitive in the early stage of lung cancer. For liquid biopsy, DNA derived from EVs is better than cfDNA. EGFR genotyping of EV-DNA and cfDNA from BALF and plasma samples was conducted for NSCLC diagnosis. The results showed that the sensitivity of EV-DNA was better than cfDNA from both sources, which may be due to the protective effect of the EV bilayer membrane on internal DNA94. Furthermore, BALF samples exhibited significantly higher sensitivities for cfDNA and EV DNA than plasma samples94. Another study of patients with stage III-IV NSCLC also showed that EGFR genotyping based on BALF-EVs had higher sensitivity and specificity than EGFR genotyping based on plasma cfDNA96. The above results indicate that EGFR genotyping with BALF-EVs has satisfactory accuracy and is more convenient than solid tissue biopsy and cfDNA- or plasma-based liquid biopsy. Moreover, EGFR genotyping based on BALF-EVs has higher sensitivity in advanced tumors and can detect the T790M mutation. Therefore, EGFR genotyping based on BALF-EVs can be utilized as a substitute for tissue biopsy to guide the prompt treatment of EGFR-TKIs, especially for patients with an advanced disease stage or from whom sample collection is difficult.
For RNA in EVs, current studies primarily focus on detecting the expression of tumor-related gene mRNA and miRNA. Exosomes are one type of EV with a diameter of 30–100 nm. Compared to plasma, BALF contains fewer exosomes. Moreover, the miRNA content in BALF exosomes showed no difference between patients with and without tumors, while the miRNA content in plasma exosomes of tumor patients was higher than patients without tumors. This finding suggested that more tumor-derived miRNAs are released into plasma than into BALF97. MiR 126 and Let-7a concentrations are elevated in the BALF of lung adenocarcinoma patients compared with to control subjects. However, subsequent validation utilizing an independent set of matched adenocarcinoma and normal tissue samples (n = 4) demonstrated that only miR-126 expression is increased in cancer tissues compared to healthy tissues98. In a study related to drug resistance, plasma and BALF samples were collected from NSCLC patients before and after icotinib/gefitinib treatment. Exosomes were isolated and mRNA expression of 10 metastasis-related genes was detected. It was found that MET expression in exosomes was associated with lung cancer metastasis, but there was no significant difference between BALF and plasma99. These results indicated that BALF may not offer benefits in identifying RNA related to lung cancer. The miRNAs screened in the previous study need to be fully validated before clinical use.
In addition to nucleic acids, EVs contain numerous protein components. Exosome proteins make up 35% of the differentially expressed proteins in the BALF of lung cancer patients103. Protein detection in BALF-EVs can be used to further study the therapeutic mechanism underlying lung cancer. Suppressors of cytokine signaling 3 (SOCS3) vesicles from alveolar macrophages are secreted into BALF. The level of SOCS3, as detected by ELISA, is significantly downregulated in NSCLC patients, independent of adenocarcinoma, LSCC, and smoking status100. In another study, proteomic analysis of BALF-EVs was performed with LC–MS. The results showed that the DNMT3B complex is greatly augmented in tumor tissues and BALF-EVs, and protein markers associated with innate immune cells and fibroblasts in BALF-EVs are associated with a poorer prognosis (4 of 5 patients died during the 2-year follow-up)101.
Others
Metabolomics
In addition to the above common components, some studies have analyzed other components in BALF. Metabolomics is a systems biology method that emerged after genomics and proteomics. The non-targeted qualitative and quantitative analysis of metabolites is expected to provide a new perspective for the study of pathologic processes and biomarkers for diseases104. There have been numerous studies on metabolomics analysis of serum, plasma, urine, and sputum105–108 of lung cancer patients, but there are few metabolomics studies involving BALF. Two complementary metabolomics techniques were performed to detect lung cancer-related metabolites in BALF samples (24 cases of lung cancer and 31 cases of non-tumor lung disease) for the first time and identified 42 different metabolites. Glutamic acid and glutamine metabolic pathways are the main changed pathways. Glycerol and phosphate have promising potential as biomarkers that are both sensitive and specific for diagnosing and predicting lung cancer outcomes109. Subsequently, a combined metabolomics analysis was performed on serum, urine, and BALF from patients with lung cancer. Compared to non-tumor lung disease, researchers identified 16 metabolites that are altered in the BALF of lung cancer patients, 6 of which were detected in serum and urine. Among these metabolites, phosphoric acid was the most sensitive and specific110. The aforementioned studies indicate that phosphoric acid/phosphate may have a significant involvement in the pathologic process underlying lung cancer.
Microbiome
The pulmonary microbiome has a vital role in respiratory diseases, but the significance in lung cancer has not been established. Some studies have reported that dysbiosis of the microbiota during the disease process mainly includes the following: enrichment of potential pathogenic bacteria; loss of probiotics; and change in microbial composition111. The BALF microbiota of patients with lung cancer (20 cases) and benign lung masses (8 cases) were assessed via 16S rRNA-based NGS. The findings demonstrated that two phyla (Firmicutes and TM7) were increased and two genera (Veillonella and Megasphaera) were enriched in lung cancer112, among which Veillonella has also been reported to be enriched in the saliva of patients with lung cancer113. Another study showed that Veillonella is dominant in BALF of NSCLC patients in high-PD-L1 and immunotherapy responder groups114. Our group collected BALF samples for the purpose of investigating the microbiome of the lower respiratory tract (LRT) in individuals with lung cancer. Compared to healthy subjects (30 cases), LRT microbiome richness was decreased in lung cancer patients (91 cases). However, there was no significant distinction between the lung cancer and non-malignant lung disease groups (29 cases). We further established a diagnostic model of lung cancer (including age, smoking history, and 11 types of bacteria), with areas under the curve (AUCs) of 0.882 and 0.796 in the training and validation sets (57 lung cancer and 28 non-malignant lung disease patients), respectively, confirming that biomarkers based on the BALF microbiome may be effective in identifying lung cancer115. Similarly, another study discovered that there was no significant distinction in the main microbial composition in BALF between NSCLC (n = 32) and non-tumor patients (n = 15)116. The BALF microbiome of patients with stage I NSCLC (n = 48) before resection was analyzed. It was shown that the BALF microbiome differs between patients with and without a tumor recurrence after surgery during the 32-month follow-up117. A distinctive microbiome signature was also identified, which was associated with shorter recurrence-free survival117. Despite several differentially expressed genera being identified in the above studies, the particular function of these genera in the pathogenesis of lung cancer needs further investigation due to the dynamics and diversity of the microbial composition and the complexity of patient diets, regions, and lifestyles. In addition, a study analyzing the microbiomes of the saliva samples, BALF samples, and non-malignant, peritumoral, and tumor tissue samples from 18 NSCLC patients revealed that BALF has a unique microbial diversity compared to the other three types of lung tissues118.
BALF and liquid biopsy
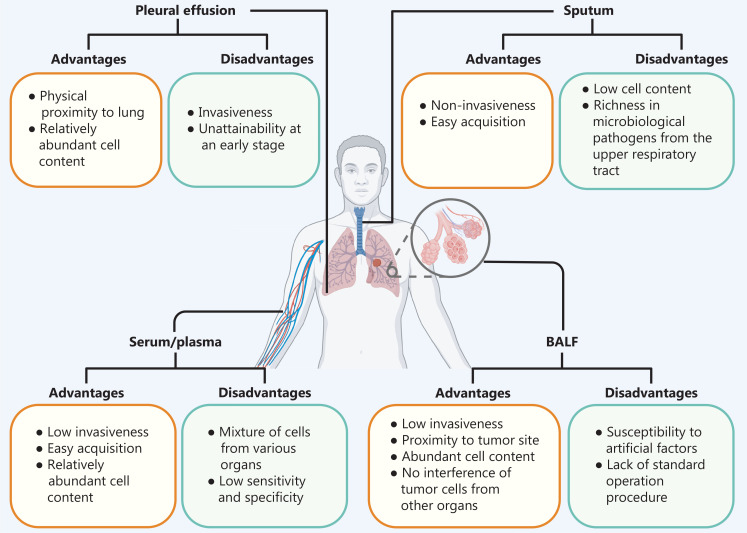
Tissue biopsy is the gold standard for the clinical diagnosis of lung cancer. Percutaneous lung aspiration and transbronchial lung biopsies are invasive diagnostic procedures with restrictions on sampling tumors that are undetectable by bronchoscopy. Liquid biopsy, in contrast, offers many advantages, including simple sampling, low invasiveness, and relative safety. In addition, traditional surgical biopsy tends to be a single-site sample that cannot be used to obtain comprehensive information on the disease. Liquid biopsy contains more representative information and provides a more accurate assessment of the disease because liquid biopsy is used to detect molecules shed/released from different parts of the tumor. Furthermore, liquid biopsy can be used to conduct real-time detection during treatment process and provide objective evidence for clinicians to adjust treatment plans and appraise prognosis.
In the liquid biopsy of lung cancer, distinct characteristics of various body fluids are summarized (Figure 3). Conventional serum markers are metabolic byproducts frequently generated and released by tumor cells. However, false-positives and -negatives are common since these markers can also be present in some benign lesions119. With respect to molecular analysis, the components in peripheral blood are relatively complex and cfDNA may come from any part of the body, so cfDNA lacks specificity. Other body fluids also have certain limitations. Sputum contains few cells, making sputum difficult to meet detection requirements. Moreover, it is also rich in pathogens from the upper respiratory tract, which easily causes interference. Pleural effusions, which are usually produced after lung cancer or lung infection, are invasive and challenging to sample early. In addition, the environment of these body fluids is intricate and affects the integrity of DNA sequences to some extent. Compared with these body fluids, BALF has advantages. BALF is in proximity to the tumor site, contains a larger amount of lung cancer cells, and is less likely to be contaminated with cancer cells from other organs, thus has a relatively higher sensitivity and specificity in early diagnosis. Furthermore, the low invasiveness of BALF is appropriate for advanced patients who are difficult to sample and holds the possibility for serial sampling during treatment.
Figure 3.
Characteristics of different body fluids. (Created with BioRender.com).
Conclusions and future directions
Precision medicine involves accurate diagnosis and classification of disease, accurate administration of drugs, accurate evaluation of curative effects, and accurate prediction of prognosis. Precision medicine is not only a public requirement, but also a clinical necessity for advancement. At present, BALF has been utilized in the accurate diagnosis of several diseases. For example, a lymphocytic-predominant BALF is sufficient for diagnosing lung sarcoidosis or upper respiratory infection with matching medical symptoms and imaging findings120. In patients with intense alveolar phacoemulsification on chest imaging, the presence of significant eosinophils in BALF accurately predicts eosinophilic pneumonia121. For pulmonary infectious diseases, assessment of BALF can be used to effectively identify etiologic pathogens, including bacteria, fungi, parasites, and viruses, to provide focused antimicrobial therapy, reduce the use of unnecessary antibiotics, and slow the rise of antimicrobial drug resistance. With respect to pulmonary alveolar proteinosis (PAP), researchers showed that serum levels of α- and β-defensins cannot differentiate PAP from COPD and sarcoidosis patients, while the BALF of patients with PAP has extremely high levels of these defensins122. For critically ill patients with COVID-19 pneumonia, analysis of cellular composition in BALF as biomarkers is reliable for disease management and prediction123.
The high heterogeneity of lung cancer determines the necessity for precision medicine. EGFR genotyping based on BALF has a satisfactory detection rate, which is expected to achieve accurate diagnosis of lung cancer69–71,94–96. The prediction model established by proteomics or microbiomics exhibits good sensitivity and specificity and may serve as an effective indicator for the precise prediction of lung cancer61,115. Some special proteins or miRNAs have been identified from BALF that can be used to distinguish SCLC from NSCLC or LSCC from adenocarcinoma, which may make precise typing of lung cancer possible61,90. The analysis of BALF components is also helpful to determine whether a patient is a candidate for immunotherapy of NSCLC with nivolumab26. Additionally, some soluble factors51–53, methylation of genes88, and specific cell subsets24 from BALF may also have been shown to be associated with the prognosis of lung cancer. Thus, the assessment of BALF exhibits promising potential in advancing precision medicine for lung cancer.
Although ongoing research into the clinical utility of the various components of BALF is still evolving and is far from being as comprehensive as similar studies conducted on blood, there is no doubt that BALF has great potential for lung cancer detection, especially using the EV DNA in BALF. Given the protective nature of the EV bilayer membrane, EV DNA exhibits heightened stability and is less susceptible to degradation by external factors compared with cfDNA96. In the detection of oncogene mutation, it is noteworthy that BALF-EV DNA exhibits superior sensitivity and specificity compared to both BALF cfDNA and plasma-derived EV DNA. Remarkably, the mutation detection rate of BALF-EV DNA even exceeds that of tissue biopsy, indicating that gene mutation detection based on BALF-EV holds significant promise as the optimal choice for lung cancer prediction. In addition, the sensitivity of BALF-EV DNA detection escalates with the TMN stage, and it is also a valuable tool for detecting the T790M mutation in patients with acquired drug resistance. These features enhance the advantages of BALF, especially in advanced lung cancer patients or in patients in whom sample collection is difficult, and can replace re-biopsy technique in guiding prompt treatment. It is worth noting that two clinical trials (NCT05469022 and NCT03228277) based on BALF-EVs in the context of lung cancer have advanced to phase II, further underlining the reliability and efficacy of BALF-EVs as a potential detection tool.
Nevertheless, the translation of BALF-EV detection into clinical applications faces several substantial challenges. Interference from viruses, proteins, and other aggregates, coupled with the inherently viscous nature of BALF, limits the availability of valuable specimens. In addition, the relatively low concentration of EVs in BALF along with the presence of a myriad of EVs from various cellular sources may often result in limited acquisition of the desired tumor-related EVs. Consequently, there is an urgent need to develop a standardized, high-yield and effective protocol for BALF-EV isolation. Such efforts are crucial to ensure the comparability and reproducibility of the research findings across different laboratories124,125 as well as to develop specialized and commercialized kits for BALF EV.
Currently, the assessment of BALF from lung cancer patients is evolving in various new directions, especially involving the immune response to lung cancer and immune checkpoint inhibitors for precision treatment. Elucidating the clinical utility of BALF as a complementary liquid biopsy for the examination of tumor-related markers will be of paramount importance in advancing lung cancer diagnostics and personalized treatment strategies. Additionally, there is a need to promptly commence large-scale, multicenter trials in pulmonology units with the involvement of oncologists to further advance the research and application of BALF in clinical practice for lung cancer.
Funding Statement
This work was supported by grants from the National Natural Science Foundation of China (Grant No. 82173182), the Sichuan Science and Technology Program (Grant No. 2021YJ0117 to Weiya Wang; Grant No. 2023NSFSC1939 to Dan Liu), and the 1•3•5 project for Disciplines of Excellence–Clinical Research Incubation Project, West China Hospital, Sichuan University (Grant Nos. 2019HXFH034 and ZYJC21074).
Conflict of interest statement
No potential conflicts of interest are disclosed.
Author contributions
Conceived and designed the analysis: Weiya Wang, Dan Liu, Wei Huang.
Collected the data: Hantao Zhang, Dan Deng, Shujun Li, Jing Ren.
Contributed data or analysis tools: Hantao Zhang, Weiya Wang.
Performed the analysis: Hantao Zhang, Wei Huang.
Wrote the paper: Hantao Zhang, Dan Deng.
References
- 1.Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA Cancer J Clin. 2023;73:17–48. doi: 10.3322/caac.21763. [DOI] [PubMed] [Google Scholar]
- 2.Maomao C, He L, Dianqin S, Siyi H, Xinxin Y, Fan Y, et al. Current cancer burden in China: epidemiology, etiology, and prevention. Cancer Biol Med. 2022;19:1121–38. doi: 10.20892/j.issn.2095-3941.2022.0231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Oudkerk M, Liu S, Heuvelmans MA, Walter JE, Field JK. Lung cancer LDCT screening and mortality reduction - evidence, pitfalls and future perspectives. Nat Rev Clin Oncol. 2021;18:135–51. doi: 10.1038/s41571-020-00432-6. [DOI] [PubMed] [Google Scholar]
- 4.Li C, Wang H, Jiang Y, Fu W, Liu X, Zhong R, et al. Advances in lung cancer screening and early detection. Cancer Biol Med. 2022;19:591–608. doi: 10.20892/j.issn.2095-3941.2021.0690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Boumahdi S, de Sauvage FJ. The great escape: tumour cell plasticity in resistance to targeted therapy. Nat Rev Drug Discov. 2020;19:39–56. doi: 10.1038/s41573-019-0044-1. [DOI] [PubMed] [Google Scholar]
- 6.Wang M, Herbst RS, Boshoff C. Toward personalized treatment approaches for non-small-cell lung cancer. Nat Med. 2021;27:1345–56. doi: 10.1038/s41591-021-01450-2. [DOI] [PubMed] [Google Scholar]
- 7.Rudin CM, Brambilla E, Faivre-Finn C, Sage J. Small-cell lung cancer. Nat Rev Dis Primers. 2021;7:3. doi: 10.1038/s41572-020-00235-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zhang C, Wang H. Accurate treatment of small cell lung cancer: current progress, new challenges and expectations. Biochim Biophys Acta Rev Cancer. 2022;1877:188798. doi: 10.1016/j.bbcan.2022.188798. [DOI] [PubMed] [Google Scholar]
- 9.Heitzer E, Haque IS, Roberts CES, Speicher MR. Current and future perspectives of liquid biopsies in genomics-driven oncology. Nat Rev Genet. 2019;20:71–88. doi: 10.1038/s41576-018-0071-5. [DOI] [PubMed] [Google Scholar]
- 10.Ignatiadis M, Sledge GW, Jeffrey SS. Liquid biopsy enters the clinic - implementation issues and future challenges. Nat Rev Clin Oncol. 2021;18:297–312. doi: 10.1038/s41571-020-00457-x. [DOI] [PubMed] [Google Scholar]
- 11.Rolfo C, Mack P, Scagliotti GV, Aggarwal C, Arcila ME, Barlesi F, et al. Liquid biopsy for advanced NSCLC: a consensus statement from the international association for the study of lung cancer. J Thorac Oncol. 2021;16:1647–62. doi: 10.1016/j.jtho.2021.06.017. [DOI] [PubMed] [Google Scholar]
- 12.Davidson KR, Ha DM, Schwarz MI, Chan ED. Bronchoalveolar lavage as a diagnostic procedure: a review of known cellular and molecular findings in various lung diseases. J Thorac Dis. 2020;12:4991–5019. doi: 10.21037/jtd-20-651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Domagala-Kulawik J. The relevance of bronchoalveolar lavage fluid analysis for lung cancer patients. Expert Rev Respir Med. 2020;14:329–37. doi: 10.1080/17476348.2020.1708720. [DOI] [PubMed] [Google Scholar]
- 14.Kalkanis A, Papadopoulos D, Testelmans D, Kopitopoulou A, Boeykens E, Wauters E. Bronchoalveolar lavage fluid-isolated biomarkers for the diagnostic and prognostic assessment of lung cancer. Diagnostics (Basel) 2022;12:2949. doi: 10.3390/diagnostics12122949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Reynolds HY, Newball HH. Analysis of proteins and respiratory cells obtained from human lungs by bronchial lavage. J Lab Clin Med. 1974;84:559–73. [PubMed] [Google Scholar]
- 16.Cao C, Yu X, Zhu T, Jiang Q, Li Y, Li X. Diagnostic role of liquid-based cytology of bronchial lavage fluid in addition to bronchial brushing specimens in lung cancer. Tumori. 2021;107:325–28. doi: 10.1177/0300891620960218. [DOI] [PubMed] [Google Scholar]
- 17.Han S, Yang W, Li H. A study of the application of fiberoptic bronchoscopy combined with liquid-based cytology test in the early diagnosis of lung cancer. Oncol Lett. 2018;16:5807–12. doi: 10.3892/ol.2018.9372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Debeljak A, Mermolja M, Sorli J, Zupancic M, Zorman M, Remskar J. Bronchoalveolar lavage in the diagnosis of peripheral primary and secondary malignant lung tumors. Respiration. 1994;61:226–30. doi: 10.1159/000196342. [DOI] [PubMed] [Google Scholar]
- 19.Ma S, Yu X, Jin X, Qiu F, Chen X, Wang R, et al. The usefulness of liquid-based cytology of bronchoalveolar lavage fluid combined with bronchial brush specimens in lung cancer diagnosis. J Int Med Res. 2022;50:3000605221132708. doi: 10.1177/03000605221132708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhong CH, Tong D, Zhou ZQ, Su ZQ, Luo YL, Xing J, et al. Performance evaluation of detecting circulating tumor cells and tumor cells in bronchoalveolar lavage fluid in diagnosis of peripheral lung cancer. J Thorac Dis. 2018;10:S830–7. doi: 10.21037/jtd.2017.12.125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Domagala-Kulawik J, Raniszewska A. How to evaluate the immune status of lung cancer patients before immunotherapy. Breathe (Sheff) 2017;13:291–6. doi: 10.1183/20734735.001917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Osińska I, Stelmaszczyk-Emmel A, Polubiec-Kownacka M, Dziedzic D, Domagała-Kulawik J. CD4+/CD25(high)/FoxP3+/CD127-regulatory T cells in bronchoalveolar lavage fluid of lung cancer patients. Hum Immunol. 2016;77:912–5. doi: 10.1016/j.humimm.2016.07.235. [DOI] [PubMed] [Google Scholar]
- 23.Kwiecien I, Stelmaszczyk-Emmel A, Polubiec-Kownacka M, Dziedzic D, Domagala-Kulawik J. Elevated regulatory T cells, surface and intracellular CTLA-4 expression and interleukin-17 in the lung cancer microenvironment in humans. Cancer Immunol Immunother. 2017;66:161–70. doi: 10.1007/s00262-016-1930-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hu X, Gu Y, Zhao S, Hua S, Jiang Y. Increased IL-10+CD206+CD14+M2-like macrophages in alveolar lavage fluid of patients with small cell lung cancer. Cancer Immunol Immunother. 2020;69:2547–60. doi: 10.1007/s00262-020-02639-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mansour MSI, Hejny K, Johansson F, Mufti J, Vidis A, Mager U, et al. Factors influencing concordance of PD-L1 expression between biopsies and cytological specimens in non-small cell lung cancer. Diagnostics (Basel) 2021;11:1927. doi: 10.3390/diagnostics11101927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Masuhiro K, Tamiya M, Fujimoto K, Koyama S, Naito Y, Osa A, et al. Bronchoalveolar lavage fluid reveals factors contributing to the efficacy of PD-1 blockade in lung cancer. JCI Insight. 2022;7:e157915. doi: 10.1172/jci.insight.157915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Suresh K, Naidoo J, Lin CT, Danoff S. Immune checkpoint immunotherapy for non-small cell lung cancer: benefits and pulmonary toxicities. Chest. 2018;154:1416–23. doi: 10.1016/j.chest.2018.08.1048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Suresh K, Naidoo J, Zhong Q, Xiong Y, Mammen J, de Flores MV, et al. The alveolar immune cell landscape is dysregulated in checkpoint inhibitor pneumonitis. J Clin Invest. 2019;129:4305–15. doi: 10.1172/JCI128654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kwiecien I, Skirecki T, Polubiec-Kownacka M, Raniszewska A, Domagala-Kulawik J. Immunophenotype of T cells expressing programmed death-1 and cytotoxic T cell antigen-4 in early lung cancer: local vs. systemic immune response. Cancers (Basel) 2019;11:567. doi: 10.3390/cancers11040567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Schneider T, Kimpfler S, Warth A, Schnabel PA, Dienemann H, Schadendorf D, et al. Foxp3(+) regulatory T cells and natural killer cells distinctly infiltrate primary tumors and draining lymph nodes in pulmonary adenocarcinoma. J Thorac Oncol. 2011;6:432–8. doi: 10.1097/JTO.0b013e31820b80ca. [DOI] [PubMed] [Google Scholar]
- 31.Brcic L, Stanzer S, Krenbek D, Gruber-Moesenbacher U, Absenger G, Quehenberger F, et al. Immune cell landscape in therapy-naïve squamous cell and adenocarcinomas of the lung. Virchows Arch. 2018;472:589–98. doi: 10.1007/s00428-018-2326-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Li J, Chen P, Mao CM, Tang XP, Zhu LR. Evaluation of diagnostic value of four tumor markers in bronchoalveolar lavage fluid of peripheral lung cancer. Asia Pac J Clin Oncol. 2014;10:141–8. doi: 10.1111/ajco.12066. [DOI] [PubMed] [Google Scholar]
- 33.Tian K, Li Z, Qin L. Detection of CEA and ProGRP levels in BALF of patients with peripheral lung cancer and their relationship with CT signs. Biomed Res Int. 2022;2022:4119912. doi: 10.1155/2022/4119912. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 34.Wang H, Zhang X, Liu X, Liu K, Li Y, Xu H. Diagnostic value of bronchoalveolar lavage fluid and serum tumor markers for lung cancer. J Cancer Res Ther. 2016;12:355–8. doi: 10.4103/0973-1482.162111. [DOI] [PubMed] [Google Scholar]
- 35.Nikliński J, Chyczewska E, Furman M, Kowal E, Kozłowski M. Value of CEA and SCC-Ag in bronchoalveolar lavage (BAL) and serum of patients with lung carcinoma. Neoplasma. 1992;39:283–5. [PubMed] [Google Scholar]
- 36.Pina TC, Zapata IT, Hernández FC, López JB, Paricio PP, Hernández PM. Tumour markers in serum, bronchoalveolar lavage and biopsy cytosol in lung carcinoma: what environment lends the optimum diagnostic yield? Clin Chim Acta. 2001;305:27–34. doi: 10.1016/s0009-8981(00)00410-1. [DOI] [PubMed] [Google Scholar]
- 37.de Diego A, Compte L, Sanchis J, Enguidanos MJ, Marco V. Usefulness of carcinoembryonic antigen determination in bronchoalveolar lavage fluid. A comparative study among patients with peripheral lung cancer, pneumonia, and healthy individuals. Chest. 1991;100:1060–3. doi: 10.1378/chest.100.4.1060. [DOI] [PubMed] [Google Scholar]
- 38.Pothal S, Patil KP, Manjhi R, Dutta P. Diagnostic efficacy of broncho-alveolar lavage carcino-embronic antigen in carcinoma of lung. J Family Med Prim Care. 2019;8:1725–9. doi: 10.4103/jfmpc.jfmpc_119_19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Duffy MJ, O’Byrne K. Tissue and blood biomarkers in lung cancer: a review. Adv Clin Chem. 2018;86:1–21. doi: 10.1016/bs.acc.2018.05.001. [DOI] [PubMed] [Google Scholar]
- 40.Ledermann JA. Serum neurone-specific enolase and other neuroendocrine markers in lung cancer. Eur J Cancer. 1994;30a:574–6. doi: 10.1016/0959-8049(94)90519-3. [DOI] [PubMed] [Google Scholar]
- 41.Prados MC, Alvarez-Sala R, Blasco R, Chivato T, García Satué JL, García Río FJ, et al. The clinical value of neuron-specific enolase as a tumor marker in bronchoalveolar lavage. Cancer. 1994;74:1552–5. doi: 10.1002/1097-0142(19940901)74:5<1552::aid-cncr2820740510>3.0.co;2-f. [DOI] [PubMed] [Google Scholar]
- 42.Dowlati A, Bury T, Corhay JL, Weber T, Mendes P, Radermecker M. High neuron specific enolase levels in bronchoalveolar lavage fluid of patients with lung carcinoma: diagnostic value, relation to serum neuron specific enolase, and staging. Cancer. 1996;77:2039–43. doi: 10.1002/(SICI)1097-0142(19960515)77:10<2039::AID-CNCR11>3.0.CO;2-W. [DOI] [PubMed] [Google Scholar]
- 43.Wieskopf B, Demangeat C, Purohit A, Stenger R, Gries P, Kreisman H, et al. Cyfra 21-1 as a biologic marker of non-small cell lung cancer. Evaluation of sensitivity, specificity, and prognostic role. Chest. 1995;108:163–9. doi: 10.1378/chest.108.1.163. [DOI] [PubMed] [Google Scholar]
- 44.Cremades MJ, Menéndez R, Pastor A, Llopis R, Aznar J. Diagnostic value of cytokeratin fragment 19 (CYFRA 21-1) in bronchoalveolar lavage fluid in lung cancer. Respir Med. 1998;92:766–71. doi: 10.1016/s0954-6111(98)90010-5. [DOI] [PubMed] [Google Scholar]
- 45.Cao C, Sun SF, Lv D, Chen ZB, Ding QL, Deng ZC. Utility of VEGF and sVEGFR-1 in bronchoalveolar lavage fluid for differential diagnosis of primary lung cancer. Asian Pac J Cancer Prev. 2013;14:2443–6. doi: 10.7314/apjcp.2013.14.4.2443. [DOI] [PubMed] [Google Scholar]
- 46.Ohta Y, Ohta N, Tamura M, Wu J, Tsunezuka Y, Oda M, et al. Vascular endothelial growth factor expression in airways of patients with lung cancer: a possible diagnostic tool of responsive angiogenic status on the host side. Chest. 2002;121:1624–7. doi: 10.1378/chest.121.5.1624. [DOI] [PubMed] [Google Scholar]
- 47.Zhao X, Sun X, Li XL. Expression and clinical significance of STAT3, P-STAT3, and VEGF-C in small cell lung cancer. Asian Pac J Cancer Prev. 2012;13:2873–7. doi: 10.7314/apjcp.2012.13.6.2873. [DOI] [PubMed] [Google Scholar]
- 48.Cao C, Chen ZB, Sun SF, Yu YM, Ding QL, Deng ZC. Evaluation of VEGF-C and tumor markers in bronchoalveolar lavage fluid for lung cancer diagnosis. Sci Rep. 2013;3:3473. doi: 10.1038/srep03473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lv D, Tan L, Zhang Q, Ma H, Zhang Y, Zhang Q, et al. Vascular endothelial growth factor-D (VEGF-D) is elevated in bronchoalveolar lavage fluid of patients with lung squamous carcinoma. Clin Lab. 2019;65:125–30. doi: 10.7754/Clin.Lab.2018.180712. [DOI] [PubMed] [Google Scholar]
- 50.Domagała-Kulawik J, Hoser G, Safianowska A, Grubek-Jaworska H, Chazan R. Elevated TGF-beta1 concentration in bronchoalveolar lavage fluid from patients with primary lung cancer. Arch Immunol Ther Exp (Warsz) 2006;54:143–7. doi: 10.1007/s00005-006-0016-0. [DOI] [PubMed] [Google Scholar]
- 51.Jakubowska K, Naumnik W, Niklińska W, Chyczewska E. Clinical significance of HMGB-1 and TGF-β level in serum and BALF of advanced non-small cell lung cancer. Adv Exp Med Biol. 2015;852:49–58. doi: 10.1007/5584_2015_115. [DOI] [PubMed] [Google Scholar]
- 52.Naumnik W, Płońska I, Ossolińska M, Nikliński J, Naumnik B. Prognostic value of osteoprotegerin and sRANKL in bronchoalveolar lavage fluid of patients with advanced non-small cell lung cancer. Adv Exp Med Biol. 2018;1047:1–6. doi: 10.1007/5584_2017_111. [DOI] [PubMed] [Google Scholar]
- 53.Naumnik W, Panek B, Ossolińska M, Naumnik B. B cell-attracting chemokine-1 and progranulin in bronchoalveolar lavage fluid of patients with advanced non-small cell lung cancer: new prognostic factors. Adv Exp Med Biol. 2019;1150:11–6. doi: 10.1007/5584_2018_285. [DOI] [PubMed] [Google Scholar]
- 54.Naumnik W, Naumnik B, Niewiarowska K, Ossolinska M, Chyczewska E. Novel cytokines: IL-27, IL-29, IL-31 and IL-33. Can they be useful in clinical practice at the time diagnosis of lung cancer? Exp Oncol. 2012;34:348–53. [PubMed] [Google Scholar]
- 55.Naumnik W, Naumnik B, Niewiarowska K, Ossolinska M, Chyczewska E. Angiogenic axis angiopoietin-1 and angiopoietin-2/Tie-2 in non-small cell lung cancer: a bronchoalveolar lavage and serum study. Adv Exp Med Biol. 2013;788:341–8. doi: 10.1007/978-94-007-6627-3_46. [DOI] [PubMed] [Google Scholar]
- 56.Kopiński P, Wandtke T, Dyczek A, Wędrowska E, Roży A, Senderek T, et al. Increased levels of interleukin 27 in patients with early clinical stages of non-small cell lung cancer. Pol Arch Intern Med. 2018;128:105–14. doi: 10.20452/pamw.4156. [DOI] [PubMed] [Google Scholar]
- 57.Xiong W, Ding W, Xu M, Pudasaini B, Sun J, Zhao Y. The screening role of a biomarker panel in BALF among patients with cancer-suspected pulmonary nodules less than 8 mm. Clin Respir J. 2020;14:829–38. doi: 10.1111/crj.13215. [DOI] [PubMed] [Google Scholar]
- 58.Oumeraci T, Schmidt B, Wolf T, Zapatka M, Pich A, Brors B, et al. Bronchoalveolar lavage fluid of lung cancer patients: mapping the uncharted waters using proteomics technology. Lung Cancer. 2011;72:136–8. doi: 10.1016/j.lungcan.2011.01.015. [DOI] [PubMed] [Google Scholar]
- 59.Pastor MD, Nogal A, Molina-Pinelo S, Meléndez R, Salinas A, González De la Peña M, et al. Identification of proteomic signatures associated with lung cancer and COPD. J Proteomics. 2013;89:227–37. doi: 10.1016/j.jprot.2013.04.037. [DOI] [PubMed] [Google Scholar]
- 60.Li QK, Shah P, Li Y, Aiyetan PO, Chen J, Yung R, et al. Glycoproteomic analysis of bronchoalveolar lavage (BAL) fluid identifies tumor-associated glycoproteins from lung adenocarcinoma. J Proteome Res. 2013;12:3689–96. doi: 10.1021/pr400274w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Uribarri M, Hormaeche I, Zalacain R, Lopez-Vivanco G, Martinez A, Nagore D, et al. A new biomarker panel in bronchoalveolar lavage for an improved lung cancer diagnosis. J Thorac Oncol. 2014;9:1504–12. doi: 10.1097/JTO.0000000000000282. [DOI] [PubMed] [Google Scholar]
- 62.Almatroodi SA, McDonald CF, Collins AL, Darby IA, Pouniotis DS. Quantitative proteomics of bronchoalveolar lavage fluid in lung adenocarcinoma. Cancer Genomics Proteomics. 2015;12:39–48. [PubMed] [Google Scholar]
- 63.Ortea I, Rodríguez-Ariza A, Chicano-Gálvez E, Arenas Vacas MS, Jurado Gámez B. Discovery of potential protein biomarkers of lung adenocarcinoma in bronchoalveolar lavage fluid by SWATH MS data-independent acquisition and targeted data extraction. J Proteomics. 2016;138:106–14. doi: 10.1016/j.jprot.2016.02.010. [DOI] [PubMed] [Google Scholar]
- 64.Hmmier A, O’Brien ME, Lynch V, Clynes M, Morgan R, Dowling P. Proteomic analysis of bronchoalveolar lavage fluid (BALF) from lung cancer patients using label-free mass spectrometry. BBA Clin. 2017;7:97–104. doi: 10.1016/j.bbacli.2017.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Zhou Y, Yang W, Ao M, Höti N, Gabrielson E, Chan DW, et al. Proteomic analysis of the air-way fluid in lung cancer. Detection of periostin in bronchoalveolar lavage (BAL) Front Oncol. 2020;10:1072. doi: 10.3389/fonc.2020.01072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Vu HM, Mohammad HB, Nguyen TNC, Lee JH, Do Y, Sung JY, et al. Quantitative proteomic analysis of bronchoalveolar lavage fluids from patients with small cell lung cancers. Proteomics Clin Appl. 2023;17:e2300011. doi: 10.1002/prca.202300011. [DOI] [PubMed] [Google Scholar]
- 67.Sim SY, Choi YR, Lee JH, Lim JM, Lee SE, Kim KP, et al. In-depth proteomic analysis of human bronchoalveolar lavage fluid toward the biomarker iscovery for lung cancers. Proteomics Clin Appl. 2019;13:e1900028. doi: 10.1002/prca.201900028. [DOI] [PubMed] [Google Scholar]
- 68.Travis WD, Brambilla E, Noguchi M, Nicholson AG, Geisinger KR, Yatabe Y, et al. International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society International Multidisciplinary Classification of Lung Adenocarcinoma. J Thorac Oncol. 2011;6:244–85. doi: 10.1097/JTO.0b013e318206a221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Buttitta F, Felicioni L, Del Grammastro M, Filice G, Di Lorito A, Malatesta S, et al. Effective assessment of EGFR mutation status in bronchoalveolar lavage and pleural fluids by next-generation sequencing. Clin Cancer Res. 2013;19:691–8. doi: 10.1158/1078-0432.CCR-12-1958. [DOI] [PubMed] [Google Scholar]
- 70.Park S, Hur JY, Lee KY, Lee JC, Rho JK, Shin SH, et al. Assessment of EGFR mutation status using cell-free DNA from bronchoalveolar lavage fluid. Clin Chem Lab Med. 2017;55:1489–95. doi: 10.1515/cclm-2016-0302. [DOI] [PubMed] [Google Scholar]
- 71.Yanev N, Mekov E, Valev D, Yankov G, Milanov V, Bichev S, et al. EGFR mutation status yield from bronchoalveolar lavage in patients with primary pulmonary adenocarcinoma compared to a venous blood sample and tissue biopsy. PeerJ. 2021;9:e11448. doi: 10.7717/peerj.11448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Zeng D, Wang C, Mu C, Su M, Mao J, Huang J, et al. Cell-free DNA from bronchoalveolar lavage fluid (BALF): a new liquid biopsy medium for identifying lung cancer. Ann Transl Med. 2021;9:1080. doi: 10.21037/atm-21-2579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Nair VS, Hui AB, Chabon JJ, Esfahani MS, Stehr H, Nabet BY, et al. Genomic profiling of bronchoalveolar lavage fluid in lung cancer. Cancer Res. 2022;82:2838–47. doi: 10.1158/0008-5472.CAN-22-0554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Topaloglu O, Hoque MO, Tokumaru Y, Lee J, Ratovitski E, Sidransky D, et al. Detection of promoter hypermethylation of multiple genes in the tumor and bronchoalveolar lavage of patients with lung cancer. Clin Cancer Res. 2004;10:2284–8. doi: 10.1158/1078-0432.ccr-1111-3. [DOI] [PubMed] [Google Scholar]
- 75.Ren M, Wang C, Sheng D, Shi Y, Jin M, Xu S. Methylation analysis of SHOX2 and RASSF1A in bronchoalveolar lavage fluid for early lung cancer diagnosis. Ann Diagn Pathol. 2017;27:57–61. doi: 10.1016/j.anndiagpath.2017.01.007. [DOI] [PubMed] [Google Scholar]
- 76.Zhang C, Yu W, Wang L, Zhao M, Guo Q, Lv S, et al. DNA methylation analysis of the SHOX2 and RASSF1A panel in bronchoalveolar lavage fluid for lung cancer diagnosis. J Cancer. 2017;8:3585–91. doi: 10.7150/jca.21368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Ahrendt SA, Chow JT, Xu LH, Yang SC, Eisenberger CF, Esteller M, et al. Molecular detection of tumor cells in bronchoalveolar lavage fluid from patients with early stage lung cancer. J Natl Cancer Inst. 1999;91:332–9. doi: 10.1093/jnci/91.4.332. [DOI] [PubMed] [Google Scholar]
- 78.Mok TS, Wu YL, Thongprasert S, Yang CH, Chu DT, Saijo N, et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med. 2009;361:947–57. doi: 10.1056/NEJMoa0810699. [DOI] [PubMed] [Google Scholar]
- 79.Zhou C, Wu YL, Chen G, Feng J, Liu XQ, Wang C, et al. Erlotinib versus chemotherapy as first-line treatment for patients with advanced EGFR mutation-positive non-small-cell lung cancer (OPTIMAL, CTONG-0802): a multicentre, open-label, randomised, phase 3 study. Lancet Oncol. 2011;12:735–42. doi: 10.1016/S1470-2045(11)70184-X. [DOI] [PubMed] [Google Scholar]
- 80.Arcila ME, Oxnard GR, Nafa K, Riely GJ, Solomon SB, Zakowski MF, et al. Rebiopsy of lung cancer patients with acquired resistance to EGFR inhibitors and enhanced detection of the T790M mutation using a locked nucleic acid-based assay. Clin Cancer Res. 2011;17:1169–80. doi: 10.1158/1078-0432.CCR-10-2277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Pérez-Barrios C, Sánchez-Herrero E, Garcia-Simón N, Barquín M, Clemente MB, Provencio M, et al. ctDNA from body fluids is an adequate source for EGFR biomarker testing in advanced lung adenocarcinoma. Clin Chem Lab Med. 2021;59:1221–9. doi: 10.1515/cclm-2020-1465. [DOI] [PubMed] [Google Scholar]
- 82.Macías M, Alegre E, Alkorta-Aranburu G, Patiño-García A, Mateos B, Andueza MP, et al. The dynamic use of EGFR mutation analysis in cell-free DNA as a follow-up biomarker during different treatment lines in non-small-cell lung cancer patients. Dis Markers. 2019;2019:7954921. doi: 10.1155/2019/7954921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Kneip C, Schmidt B, Seegebarth A, Weickmann S, Fleischhacker M, Liebenberg V, et al. SHOX2 DNA methylation is a biomarker for the diagnosis of lung cancer in plasma. J Thorac Oncol. 2011;6:1632–8. doi: 10.1097/JTO.0b013e318220ef9a. [DOI] [PubMed] [Google Scholar]
- 84.Li P, Liu S, Du L, Mohseni G, Zhang Y, Wang C. Liquid biopsies based on DNA methylation as biomarkers for the detection and prognosis of lung cancer. Clin Epigenetics. 2022;14:118. doi: 10.1186/s13148-022-01337-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Dietrich D, Kneip C, Raji O, Liloglou T, Seegebarth A, Schlegel T, et al. Performance evaluation of the DNA methylation biomarker SHOX2 for the aid in diagnosis of lung cancer based on the analysis of bronchial aspirates. Int J Oncol. 2012;40:825–32. doi: 10.3892/ijo.2011.1264. [DOI] [PubMed] [Google Scholar]
- 86.Schneider KU, Dietrich D, Fleischhacker M, Leschber G, Merk J, Schäper F, et al. Correlation of SHOX2 gene amplification and DNA methylation in lung cancer tumors. BMC Cancer. 2011;11:102. doi: 10.1186/1471-2407-11-102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Ilse P, Biesterfeld S, Pomjanski N, Fink C, Schramm M. SHOX2 DNA methylation is a tumour marker in pleural effusions. Cancer Genomics Proteomics. 2013;10:217–23. [PubMed] [Google Scholar]
- 88.Deng Q, Su B, Ji X, Fang Q, Zhou S, Zhou C. Predictive value of unmethylated RASSF1A on disease progression in non-small cell lung cancer patients receiving pemetrexed-based chemotherapy. Cancer Biomark. 2020;27:313–23. doi: 10.3233/CBM-190258. [DOI] [PubMed] [Google Scholar]
- 89.Kim JO, Gazala S, Razzak R, Guo L, Ghosh S, Roa WH, et al. Non-small cell lung cancer detection using microRNA expression profiling of bronchoalveolar lavage fluid and sputum. Anticancer Res. 2015;35:1873–80. [PubMed] [Google Scholar]
- 90.Li H, Jiang Z, Leng Q, Bai F, Wang J, Ding X, et al. A prediction model for distinguishing lung squamous cell carcinoma from adenocarcinoma. Oncotarget. 2017;8:50704–14. doi: 10.18632/oncotarget.17038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Dutkowska A, Antczak A, Domańska-Senderowska D, Brzeziańska-Lasota E. Expression of selected miRNA, RARβ and FHIT genes in BALf of squamous cell lung cancer (squamous-cell carcinoma, SCC) patients: a pilot study. Mol Biol Rep. 2019;46:6593–7. doi: 10.1007/s11033-019-05057-2. [DOI] [PubMed] [Google Scholar]
- 92.Rahbarghazi R, Jabbari N, Sani NA, Asghari R, Salimi L, Kalashani SA, et al. Tumor-derived extracellular vesicles: reliable tools for Cancer diagnosis and clinical applications. Cell Commun Signal. 2019;17:73. doi: 10.1186/s12964-019-0390-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Carnino JM, Lee H, Jin Y. Isolation and characterization of extracellular vesicles from Broncho-alveolar lavage fluid: a review and comparison of different methods. Respir Res. 2019;20:240. doi: 10.1186/s12931-019-1210-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Hur JY, Kim HJ, Lee JS, Choi CM, Lee JC, Jung MK, et al. Extracellular vesicle-derived DNA for performing EGFR genotyping of NSCLC patients. Mol Cancer. 2018;17:15. doi: 10.1186/s12943-018-0772-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Hur JY, Lee JS, Kim IA, Kim HJ, Kim WS, Lee KY. Extracellular vesicle-based EGFR genotyping in bronchoalveolar lavage fluid from treatment-naive non-small cell lung cancer patients. Transl Lung Cancer Res. 2019;8:1051–60. doi: 10.21037/tlcr.2019.12.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Kim IA, Hur JY, Kim HJ, Kim WS, Lee KY. Extracellular vesicle-based bronchoalveolar lavage fluid liquid biopsy for EGFR mutation testing in advanced non-squamous NSCLC. Cancers (Basel) 2022;14:2744. doi: 10.3390/cancers14112744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Rodríguez M, Silva J, López-Alfonso A, López-Muñiz MB, Peña C, Domínguez G, et al. Different exosome cargo from plasma/bronchoalveolar lavage in non-small-cell lung cancer. Genes Chromosomes Cancer. 2014;53:713–24. doi: 10.1002/gcc.22181. [DOI] [PubMed] [Google Scholar]
- 98.Kim JE, Eom JS, Kim WY, Jo EJ, Mok J, Lee K, et al. Diagnostic value of microRNAs derived from exosomes in bronchoalveolar lavage fluid of early-stage lung adenocarcinoma: a pilot study. Thorac Cancer. 2018;9:911–5. doi: 10.1111/1759-7714.12756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Yu Y, Abudula M, Li C, Chen Z, Zhang Y, Chen Y. Icotinib-resistant HCC827 cells produce exosomes with mRNA MET oncogenes and mediate the migration and invasion of NSCLC. Respir Res. 2019;20:217. doi: 10.1186/s12931-019-1202-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Speth JM, Penke LR, Bazzill JD, Park KS, de Rubio RG, Schneider DJ, et al. Alveolar macrophage secretion of vesicular SOCS3 represents a platform for lung cancer therapeutics. JCI Insight. 2019;4:e131340. doi: 10.1172/jci.insight.131340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Carvalho AS, Moraes MCS, Hyun Na C, Fierro-Monti I, Henriques A, Zahedi S, et al. Is the proteome of bronchoalveolar lavage extracellular vesicles a marker of advanced lung cancer? Cancers (Basel) 2020;12:3450. doi: 10.3390/cancers12113450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Lee SE, Park HY, Hur JY, Kim HJ, Kim IA, Kim WS, et al. Genomic profiling of extracellular vesicle-derived DNA from bronchoalveolar lavage fluid of patients with lung adenocarcinoma. Transl Lung Cancer Res. 2021;10:104–16. doi: 10.21037/tlcr-20-888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Carvalho AS, Cuco CM, Lavareda C, Miguel F, Ventura M, Almeida S, et al. Bronchoalveolar lavage proteomics in patients with suspected lung cancer. Sci Rep. 2017;7:42190. doi: 10.1038/srep42190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Lindon JC, Holmes E, Nicholson JK. Metabonomics and its role in drug development and disease diagnosis. Expert Rev Mol Diagn. 2004;4:189–99. doi: 10.1586/14737159.4.2.189. [DOI] [PubMed] [Google Scholar]
- 105.Chen Y, Ma Z, Li A, Li H, Wang B, Zhong J, et al. Metabolomic profiling of human serum in lung cancer patients using liquid chromatography/hybrid quadrupole time-of-flight mass spectrometry and gas chromatography/mass spectrometry. J Cancer Res Clin Oncol. 2015;141:705–18. doi: 10.1007/s00432-014-1846-5. [DOI] [PubMed] [Google Scholar]
- 106.Maeda J, Higashiyama M, Imaizumi A, Nakayama T, Yamamoto H, Daimon T, et al. Possibility of multivariate function composed of plasma amino acid profiles as a novel screening index for non-small cell lung cancer: a case control study. BMC Cancer. 2010;10:690. doi: 10.1186/1471-2407-10-690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Carrola J, Rocha CM, Barros AS, Gil AM, Goodfellow BJ, Carreira IM, et al. Metabolic signatures of lung cancer in biofluids: NMR-based metabonomics of urine. J Proteome Res. 2011;10:221–30. doi: 10.1021/pr100899x. [DOI] [PubMed] [Google Scholar]
- 108.O’Shea K, Cameron SJ, Lewis KE, Lu C, Mur LA. Metabolomic-based biomarker discovery for non-invasive lung cancer screening: a case study. Biochim Biophys Acta. 2016;1860:2682–7. doi: 10.1016/j.bbagen.2016.07.007. [DOI] [PubMed] [Google Scholar]
- 109.Callejón-Leblic B, García-Barrera T, Grávalos-Guzmán J, Pereira-Vega A, Gómez-Ariza JL. Metabolic profiling of potential lung cancer biomarkers using bronchoalveolar lavage fluid and the integrated direct infusion/gas chromatography mass spectrometry platform. J Proteomics. 2016;145:197–206. doi: 10.1016/j.jprot.2016.05.030. [DOI] [PubMed] [Google Scholar]
- 110.Callejón-Leblic B, García-Barrera T, Pereira-Vega A, Gómez-Ariza JL. Metabolomic study of serum, urine and bronchoalveolar lavage fluid based on gas chromatography mass spectrometry to delve into the pathology of lung cancer. J Pharm Biomed Anal. 2019;163:122–9. doi: 10.1016/j.jpba.2018.09.055. [DOI] [PubMed] [Google Scholar]
- 111.Brüssow H. Problems with the concept of gut microbiota dysbiosis. Microb Biotechnol. 2020;13:423–34. doi: 10.1111/1751-7915.13479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Lee SH, Sung JY, Yong D, Chun J, Kim SY, Song JH, et al. Characterization of microbiome in bronchoalveolar lavage fluid of patients with lung cancer comparing with benign mass like lesions. Lung Cancer. 2016;102:89–95. doi: 10.1016/j.lungcan.2016.10.016. [DOI] [PubMed] [Google Scholar]
- 113.Yan X, Yang M, Liu J, Gao R, Hu J, Li J, et al. Discovery and validation of potential bacterial biomarkers for lung cancer. Am J Cancer Res. 2015;5:3111–22. [PMC free article] [PubMed] [Google Scholar]
- 114.Jang HJ, Choi JY, Kim K, Yong SH, Kim YW, Kim SY, et al. Relationship of the lung microbiome with PD-L1 expression and immunotherapy response in lung cancer. Respir Res. 2021;22:322. doi: 10.1186/s12931-021-01919-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Jin J, Gan Y, Liu H, Wang Z, Yuan J, Deng T, et al. Diminishing microbiome richness and distinction in the lower respiratory tract of lung cancer patients: a multiple comparative study design with independent validation. Lung Cancer. 2019;136:129–35. doi: 10.1016/j.lungcan.2019.08.022. [DOI] [PubMed] [Google Scholar]
- 116.Zheng L, Sun R, Zhu Y, Li Z, She X, Jian X, et al. Lung microbiome alterations in NSCLC patients. Sci Rep. 2021;11:11736. doi: 10.1038/s41598-021-91195-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Patnaik SK, Cortes EG, Kannisto ED, Punnanitinont A, Dhillon SS, Liu S, et al. Lower airway bacterial microbiome may influence recurrence after resection of early-stage non-small cell lung cancer. J Thorac Cardiovasc Surg. 2021;161:419–29.e16. doi: 10.1016/j.jtcvs.2020.01.104. [DOI] [PubMed] [Google Scholar]
- 118.Bingula R, Filaire E, Molnar I, Delmas E, Berthon JY, Vasson MP, et al. Characterisation of microbiota in saliva, bronchoalveolar lavage fluid, non-malignant, peritumoural and tumour tissue in non-small cell lung cancer patients: a cross-sectional clinical trial. Respir Res. 2020;21:129. doi: 10.1186/s12931-020-01392-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Lee JH, Chang JH. Diagnostic utility of serum and pleural fluid carcinoembryonic antigen, neuron-specific enolase, and cytokeratin 19 fragments in patients with effusions from primary lung cancer. Chest. 2005;128:2298–303. doi: 10.1378/chest.128.4.2298. [DOI] [PubMed] [Google Scholar]
- 120.Darlington P, Kullberg S, Eklund A, Grunewald J. Lung CD4+ Vα2.3+ T-cells in sarcoidosis cohorts with Löfgren’s syndrome. Respir Res. 2020;21:61. doi: 10.1186/s12931-020-1327-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Sohn JW. Acute eosinophilic pneumonia. Tuberc Respir Dis (Seoul) 2013;74:51–5. doi: 10.4046/trd.2013.74.2.51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Mukae H, Ishimoto H, Yanagi S, Ishii H, Nakayama S, Ashitani J, et al. Elevated BALF concentrations of alpha- and beta-defensins in patients with pulmonary alveolar proteinosis. Respir Med. 2007;101:715–21. doi: 10.1016/j.rmed.2006.08.018. [DOI] [PubMed] [Google Scholar]
- 123.Singh G, Martin Rumende C, Sharma SK, Rengganis I, Amin Z, Loho T, et al. Low BALF CD4 T cells count is associated with extubation failure and mortality in critically ill covid-19 pneumonia. Ann Med. 2022;54:1894–905. doi: 10.1080/07853890.2022.2095012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Zareba L, Szymanski J, Homoncik Z, Czystowska-Kuzmicz M. EVs from BALF-mediators of inflammation and potential biomarkers in lung diseases. Int J Mol Sci. 2021;22:3651. doi: 10.3390/ijms22073651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Yu W, Hurley J, Roberts D, Chakrabortty SK, Enderle D, Noerholm M, et al. Exosome-based liquid biopsies in cancer: opportunities and challenges. Ann Oncol. 2021;32:466–77. doi: 10.1016/j.annonc.2021.01.074. [DOI] [PMC free article] [PubMed] [Google Scholar]