Abstract
Progranulin (PGRN), encoded by the GRN gene in humans, was originally isolated as a secreted growth factor that implicates in a multitude of processes ranging from regulation of tumorigenesis, inflammation to neural proliferation. Compelling evidence indicating that GRN mutation can lead to various common neuronal degenerative diseases and rare lysosomal storage diseases. These findings have unveiled a critical role for PGRN as a lysosomal protein in maintaining lysosomal function. The phenotypic spectrum of PGRN imbalance has expanded to encompass a broad spectrum of diseases, including autoimmune diseases, metabolic, musculoskeletal and cardiovascular diseases. These diseases collectively referred to as Progranulinopathy- a term encompasses the wide spectrum of disorders influenced by PGRN imbalance. Unlike its known extracellular function as a growth factor-like molecule associated with multiple membrane receptors, PGRN also serves as an intracellular co-chaperone engaged in the folding and traffic of its associated proteins, particularly the lysosomal hydrolases. This chaperone activity is required for PGRN to exert its diverse functions across a broad range of diseases, encompassing both the central nervous system and peripheral systems. In this comprehensive review, we present an update of the emerging role of PGRN in Progranulinopathy, with special focus on elucidating the intricate interplay between PGRN and a diverse array of proteins at various levels, ranging from extracellular fluids and intracellular components, as well as various pathophysiological processes involved. This review seeks to offer a comprehensive grasp of PGRN’s diverse functions, aiming to unveil intricate mechanisms behind Progranulinopathy and open doors for future research endeavors.
Keywords: Progranulinopathy, progranulin, Neurodegenerative Diseases, Lysosomal Storages, cancer, Autoimmune diseases
1. Introduction
Progranulin (PGRN), also known as granulin–epithelin precursor (GEP), proepithelin, acrogranin, and GP88/PC-cell derived growth factor, is a 593-amino-acid glycoprotein[1]. PGRN contains seven-and-a-half repeats of a cysteine-rich motif in the order, P–G–F–B–A–C–D–E, where A–G are full repeats and P is the half-motif[2]. PGRN is highly expressed in epithelial cells, neurons and macrophages, and is likewise expressed in a broad range of other tissues and cell-types including skeletal muscle, cartilage, adipose tissue, hematopoietic cells, and some immune cells like T cells and dendritic cells[3].
PGRN initially was found as a growth factor that promotes cell proliferation[4]. The discovery of PGRN’s neurotropic function was simultaneously reported by two individual groups who have shown that a PGRN mutation leads to frontotemporal lobar degeneration (FTLD)[5, 6]. Following this study, other groups have also found that PGRN imbalance are associated with Alzheimer’s disease, Parkinson’s disease, Amyotrophic Lateral Sclerosis (ALS), motor neuron disease, multiple sclerosis, and Creutzfeldt-Jakob disease[3]. PGRN has been identified as a crucial molecule in numerous inflammation and developmental disease processes, including autoimmune diseases and musculoskeletal disorders. Additionally, the identification of PGRN as a co-chaperone molecule and an essential lysosomal protein also provides a mechanistic explanation for the association between PGRN insufficiency and a wide range of diseases affecting the central nervous system, peripheral organs, and visceral organs[3]. These disorders are collectively referred to as Progranulinopathy, encompassing the broad spectrum of conditions influenced by PGRN imbalance. Recent Bateman’s[7] and Simon’s[8] reviews nicely center around the role of PGRN in lysosomal functions and its potential implications in the treatment of neurodegenerative diseases. Previously, our team also summarizes available knowledge of PGRN in various kinds of diseases, including common neurological diseases, inflammatory autoimmune diseases, and rare lysosomal storage diseases [3, 9–15]. Understanding the underlying mechanisms of progranulinopathy and the specific pathways and interactions involved in PGRN-related diseases is crucial for the development of targeted therapies and diagnostic approaches. Thus, in this updated prospective review paper, we will discuss how PGRN fits in the pathogenesis puzzle in those diseases, and provide insights into novel therapeutic potential of PGRN or PGRN-derived molecules to treat progranulinopathy. Furthermore, this review will provide updates on recently discovered novel PGRN-associated proteins and explore their potential functions in mediating various PGRN actions.
2. PGRN in progranulinopathy
2.1. PGRN and neurodegenerative diseases
Genetic variation in PGRN has been linked to multiple neurodegenerative conditions, making PGRN an attractive and potentially valuable therapeutic target. It has been believed that GRN mutations frequently serve as a causative factor in the development of FTLD. In other neurodegenerative diseases, they assume a significant role as either an important risk factor or a modifier of disease progression, rather than a direct causal factor. In this section, we summarize the clinical and neuropathological phenotypes of FTLD patients carrying GRN mutations and discuss the evidence supporting a role for PGRN in other neurodegenerative disorders. We then review the role of PGRN in nervous system based on literature from in vitro and in vivo model systems, with a focus on mouse models of PGRN deficiency. Given the biological basis and clinical evidence linking PGRN to diseases in neurological fields (Figure 1), it is well-justified to summarize the participation of PGRN in multiple neurodegenerative conditions.
Figure 1. Cellular pathologies induced by PGRN insufficiency.
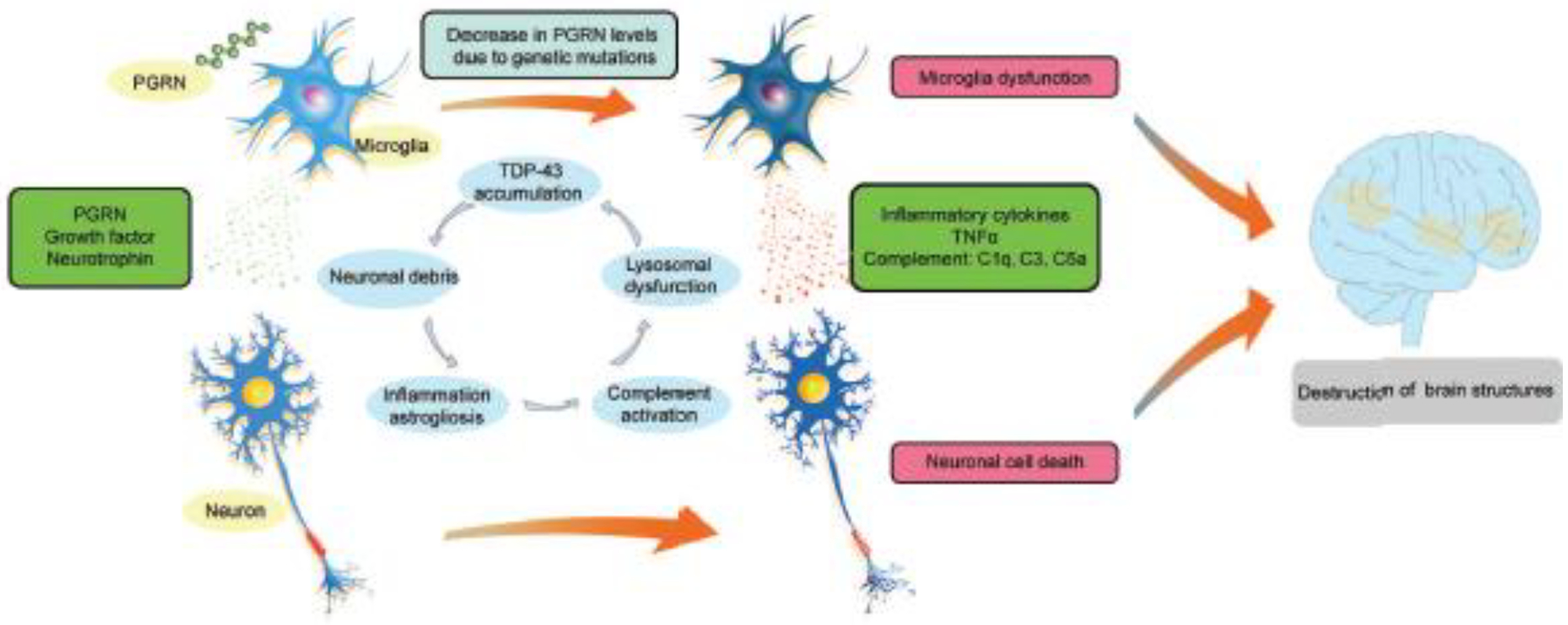
In the brain, PGRN is most highly expressed in microglia and neurons. Lower levels of PGRN cause lysosomal dysfunction, complement activation, inflammation in microglia and nuclear pore defects, TDP-43 cytoplasmic mis-localization, and eventually death in neurons. PGRN, progranulin. TDP-43, TAR DNA-binding protein 43.
2.1.1. Frontotemporal lobe dementia.
Frontotemporal lobe dementia (FTLD), also called Pick’s disease, is a neurodegenerative disorder characterized by prominent behavior and personality changes or aphasia accompanied by degeneration of the frontal and/or temporal lobes. Currently, around 170 GRN gene mutations have been reported in FTLD patients. The neuropathological and clinical presentations are different in PGRN-related FTLD. A retrospective study found that frontotemporal lobar degeneration with ubiquitin-positive inclusions, due to GRN mutations, has several characteristic features including, ubiquitin-immunoreactive neuritic pathology in superficial cortical layers and neuronal intranuclear inclusions[16]. Many studies have focused on understanding the relationship between PGRN deficiency and FTLD. One hypothesis is that PGRN haploinsufficiency may contribute to FTLD through neuroinflammation. It is found that Grn−/− microglia treated with Lipopolysaccharide/Gamma interferon (LPS/IFN-γ) exhibited an amplified inflammatory response, and conditioned media from microglia promoted death of cultured neurons[17]. PGRN deficiency is linked to TDP-43 aggregates, especially in lysosomes[18]. GRN deficiency triggers molecular and cellular changes promoting TDP-43 accumulation, nuclear pore defects, and neuron death[18]. PGRN knockdown alters TDP-43 localization and leads to TDP-43 pathology. PGRN has a direct neurotrophic effect on promoting neurite outgrowth, stimulating synaptic connectivity, and enhancing neuronal survival[19]. This neurotropic function is mainly through granulin E domin, independent of Sortilin, in which deletion of the last three amino acids (Glutamine-Leucine-Leucine) abrogated Sortilin binding to PGRN, while still retaining neurotropic activity[20].
2.1.2. Alzheimer’s disease
Alzheimer disease (AD) is the most common form of senile dementia, with an estimated lifetime risk of nearly one in five for women and one in ten for men. The hallmark neuropathological changes of AD are diffuse and neuritic senile plaques marked by extracellular amyloid beta deposition, and neurofibrillary tangles comprised of intracellular accumulation of hyperphosphorylated tau (p-tau) protein. The pathogenesis of AD is still under investigation, but it’s well established that PGRN plays a crucial role in the development of this disease. Rs5848 variant influences PGRN levels in AD. People with rs5848 variant had higher frequencies of hippocampal sclerosis (a variant of AD) and TDP-43 deposits[21]. In addition to genetic evidence, pathological PGRN was stained positive and formed plaque-like structures in AD patients with Presenilin-1 mutation. The PGRN plaque largely overlapped with beta-amyloid plaques[22]. The close association between PGRN and AD is also confirmed in mice. PGRN was found as one of the top ten up-regulated molecules expressed in two AD animal models[23]. PGRN is up-regulated in microglia, neurons, and neurites around dense-core plaques, but not in astrocytes or oligodendrocytes[23]. Recently, through whole-exome-sequencing, phospholipase D3 (PLD3) was identified accumulating within the neuritic plaques in Alzheimer’s disease brains[24]. Interestingly, PLD3 and PGRN were co-expressed in the AD plaques; furthermore, PGRN was found binding to PLD3[24]. The mechanism by which PGRN impacts AD is complex. While PGRN seems to inhibit Aβ deposition in mouse models and microglial PGRN guards against Aβ toxicity[25], some studies have shown massive lysosome-like organelle accumulations in AD plaques[26], suggesting an axonal, rather than microglial, origin. The precise molecular mechanisms through which PGRN influences AD development remain elusive.
2.1.3. Parkinson’s Disease
Parkinson’s Disease (PD) is characterized by a progressive loss of motor function, co-occurring variability with cognitive deficits at diagnosis, and progressive decline towards dementia. Neuropathologically, PD presents substantive cell death of dopaminergic neurons in the substantia nigra, and the appearance of intraneuronal and intra-axonal/dendritic accumulation of ubiquitin-bound protein aggregates, predominantly consisting of phosphorylated α-synuclein, called Lewy bodies or Lewy neurites, respectively. Brouwers et al. sequenced the GRN gene in 666 Belgian patients with AD and 255 with PD, in which they found two AD patients and one PD patient carrying GRN null mutations[27]. In a separate study sequencing GRN in 63 unrelated French patients with FTLD, one patient expressed a mutation that removed exons 1–11, and the pedigree study of this patient found the sister, who has the same mutation and developed PD[28]. Neudorfer et al. reported six Gaucher diseases (GD) patients that have Parkinsonian symptoms, and they have aggressive progression of the signs and refractoriness of conventional anti-Parkinson therapy[29]. Grn −/− mice developed GD, and lower levels of PGRN and GRN gene variants are found in GD patients[30, 31]. Considering the fact that GRN mutations are also associated with PD, it is speculated that PGRN is involved in the disease development of two distinct diseases, PD and GD. Interestingly the overlap between PD and GD has been reported. GD is caused by a mutation of the GBA1 gene, and these GBA1 mutations are also found frequently in PD patients[32]. Genotype and clinical analyses on the largest homogeneous sample of European PD patients studied to date confirmed that GBA1 mutations are the most common genetic risk factor for PD, particularly in familial forms[33].
2.1.4. Amyotrophic lateral sclerosis
Amyotrophic lateral sclerosis (ALS) is a devastating neurodegenerative condition impacting both upper and lower motor neurons. ALS often presents as asymmetric muscle weakness, progressing to complete paralysis and sometimes involving cognitive and emotional deficits. Genetic studies investigating the association between ALS and the GRN gene have yielded varying results. Some studies have identified PGRN mutations in ALS patients[34], while others have shown no significant link[35]. Interestingly, increased PGRN staining has been detected in motor tracts with degeneration and glial cells within ALS brain and spinal cord sections[36]. Notably, PGRN levels in cerebrospinal fluid (CSF) rise in symptomatic stages of ALS in mouse models[36]. Mechanistically, PGRN appears to influence the cleavage and redistribution of TDP-43[37]. In human ALS patients, serum level of matrix metalloproteinase-9 (MMP-9) is often upregulated and increased CSF MMP-9 has been associated with a more progressive disease course[38]. The relevance of the balance between granulin fragments and a full-length PGRN has not been extensively studied in ALS. Mutations in the ubiquilin 2 gene (UBQLN2) cause ALS and ALS-FTLD. UBQLN2 is also a regulator of PGRN through mediating PGRN’s escape from autophagic degradation and the number of ALS-linked mutations in UBQLN2 results in dose-dependent reduction UBQLN2-mediated upregulation of PGRN[39].
2.2. PGRN and cancer
PGRN exhibits significant expression levels in various types of cancer cells and plays a role in cancer progression. Consequently, it is considered a valuable biomarker for cancer in pathological conditions. The heightened expression of PGRN in cancer cells underscores its potential importance, making it an intriguing target for further investigation and potential therapeutic interventions (Figure 2). In addition, Table 1 lists the PGRN function, molecular targets, and prognostic utility. In this section, we will discuss PGRN function in several illustrative cancers and summarize its shared function and molecular mechanisms in cancer progression.
Figure 2. PGRN and its biological effects in pathogenesis of tumor progress.

PGRN, progranulin; MMP, metalloprotease; ECM, extracellular matrix; VEGF, vascular endothelial growth factor; STAT3, Signal transducers and activators of transcription 3; SORT1, Sortilin 1; CSC, cancer stem cells; ERK, Extracellular signal-regulated kinase; PD-L1, Programmed Cell Death Ligand 1.
Table 1.
PGRN’s function, molecular targets and prognostic utility in cancer
| Functions of PGRN | Molecular targets | Prognostic utility | |
|---|---|---|---|
| Reproductive system cancer | |||
| Breast Cancer | 1. Promotes proliferation, invasion, and migration; 2. CSC maintenance 3. induces angiogenesis and lymphangiogenesis; 4. induces immune escape; 5. Stimulation of fibroblasts and myofibroblasts function 6. induces chemotherapy and endocrine therapy resistance; 7. resulting in decreased CD8+ T cell infiltration and resistance to the αPD-1 antibody therapy. |
VEGF, estrogen receptor, cyclin D1, STAT3 | Patients who were 3 + score in PGRN expression in IHC staining has higher risk for recurrence (5.9-fold) and mortality (2.5-fold) than patient who were < 3+. |
| Cervical Cancer | 1. Induces proliferation (inhibits tumor cell senescence) | PI3K/Akt/mTOR | - |
| Ovarian Cancer | 1. Promotes proliferation and motility of cancer cells (inhibits apoptosis); 2. induces migration and invasion 3. induces chemotherapy resistance; |
PKC, CDK4, cyclin D, MAPK, MMP-2 | Patients with a PGRN concentration at this specified cutoff value achieved a 5-year progression-free survival rate of 60%, in contrast to 0% for those with PGRN levels exceeding 59 ng/ml. |
| Genitourinary cancer | |||
| Kidney cancer | 1. Promotes proliferation | - | - |
| Prostate Cancer | 1. Promotes proliferation, invasion and migration 2. induces radiation therapy resistance; |
Akt, ERK1/2 | Patients with low serum PGRN levels (⩽36.92 ng/ml) exhibit a markedly improved overall survival when compared to individuals with higher concentrations. |
| Bladder Cancer | 1. Stimulates migration and invasion; promotes proliferation and motility of cancer cells; 2. reduces chemosensitivity |
MAPK, Akt | The levels of urinary PGRN were notably elevated in bladder cancer patients in comparison to control subjects, with mean values of 17.5 ± 10 ng/100 mg creatinine and 8.9 ± 3.5 ng/100 mg creatinine, respectively (p < 0.001). This yielded a test sensitivity of 74.6% and a specificity of 85.2% for detecting the presence of bladder cancer. |
| Gastrointestinal cancer | |||
| Colorectal Cancer | 1. Induces cancer growth, migration, and invasion; 2. Tumor angiogenesis and lymphangiogenesis 3. Stimulation of fibroblasts and myofibroblasts function |
TNFR2/Akt, ERK, Wnt | The significant association between PGRN overexpression and a reduced 3-year recurrence-free survival was observed in patients with colorectal cancer who had undergone curative resection, with rates of 66.8% and 92.4%, respectively. |
| Hepatocellular cancer | 1. promotes proliferation, invasion, and migration; 2. Induces angiogenesis; 3. CSC maintenance 4. Tumor immune evasion 5. reduces chemosensitivity |
VEGF, Akt, MAPK, mTOR | 71.8% HCCs demonstrated up-regulation of PGRN compared with their adjacent liver tissues, which was associated with large HCCs, venous infiltration, and early intrahepatic recurrence (P <0.05). High serum PGRN and/or MICA levels were associated with poor recurrence-free survival (P = 0.042). |
| Biliary Tract Cancer | 1. promotes tumor cell proliferation 2. reduces chemosensitivity |
ERK1/2/RSK1/C/EBPβ, Akt | The presence of PGRN-positive tumors emerged as an independent prognostic factor significantly correlated with a diminished progression-free survival (hazard ratio 1.69, 95% CI 1.02–2.81; p = 0.044) |
| Pancreatic cancer | 1. promotes migration 2. Contributes to cytotoxic CD8+ T cell exclusion; 3. evade the immune response 4. Stimulation of fibroblasts and myofibroblasts function |
MHCI; | - |
| Lung cancer | promotes tumor cell proliferation; inhibited cell apoptosis | PI3K/Akt/Bcl-2 | The overall survival was significantly shorter in patients with a progranulin level of ⩾16 ng/ml compared to those with <16 ng/ml (p < 0.001). |
| Neuron system cancer | 1. CSC maintenance 2. Induces chemotherapy resistance; 3. promotes proliferation (inhibits apoptosis) |
AP-1, PI3K/Akt, ERK1/2, CDK4/6/pRb | The levels of PGRN were notably elevated when compared to those in the healthy control group (mean ± SD; 29.6 ± 10.2 and 74.0 ± 34.5 ng/ml, respectively; p < 0.05). Additionally, high total PGRN expression, strong tumor cell PGRN expression, and strong vascular PGRN expression were all linked to a reduced overall survival in glioblastoma patients. |
| Myeloma and Leukemia | 1. Induces an inflammatory response,Promotes cell growth; 2. induces resistance of dexamethasone |
TNF-α,MAPK,PI3K | Patients with PGRN expression levels exceeding the specified cut-off experienced a recurrence-free survival with a median of 5.4 months, whereas those with low PGRN expression (⩽19.95 ng/mL) in the context of acute myeloid leukemia achieved a favorable RFS with a median of 8.9 months. |
2.2.1. Reproductive system cancer
2.2.1.1. Breast Cancer
The PGRN expression level in normal mammary tissue is either zero or weak. In conditions that cause mammary tissue abnormal growth, such as breast cancer, the PGRN has either aberrant expression or overexpression[40]. Antisense cDNA or neutralizing antibody interventions suppressing PGRN curb breast cancer cell proliferation and colony formation[41]. Clinical studies reveal PGRN’s correlation with recurrence in estrogen receptor (ER) positive breast cancer, while ER-negative cases remain unaffected[42]. PGRN thwarts tamoxifen-induced apoptosis, fostering tamoxifen resistance, and hampers the effectiveness of letrozole, an aromatase inhibitor. Notably, PGRN interacts with Her2, driving Herceptin resistance and promoting c-myc expression in Her2-positive cells[43]. PGRN’s impact extends beyond proliferation, exacerbating metastasis. PGRN-overexpressing cells demonstrate heightened migration, produce matrix-degrading MMPs, and elevate VEGF, fostering tumor invasion and neoangiogenesis[44]. Within the immune system and tumor microenvironment, PGRN showcases dual effects. PGRN heightens STAT3 activity, influencing TAM polarization to M2 macrophages and augmenting PD-L1 expression, contributing to immune evasion and T cell exclusion[45]. Inhibiting STAT3 attenuates this effect. Additionally, PGRN triggers iTreg cell formation, curbing T lymphocyte and NK cell proliferation and functionality[46]. This results in an immunosuppressive milieu favoring tumor progression and metastasis.
2.2.1.2. Cervical Cancer
PGRN levels in cervical cancer cells and tissues are also increased. PGRN promotes cervical cancer cell proliferation, partially through activation of AKT and ERK pathway[47]. Feng et al. reported that PGRN promoted the phosphorylation of mTOR and activated mTOR signaling in human cervical mucosa epithelial cells and cervical cancer cells. PGRN triggering mTOR activation dependent on TNFR2, one of the binding receptors for PGRN. Inhibition of mTOR signaling with rapamycin decreased PGRN-stimulated protein synthesis, transformation and proliferation of cervical cells in vitro, as well as decreased tumor formation and growth in vivo[47].
2.2.1.3. Ovarian Cancer
Serum level of PGRN correlated with ovarian cells malignancy. PGRN levels are much higher in malignant ovarian cancer tissues than in normal tissues and benign tumors. By screening gene libraries from low malignant potential and invasive ovarian tumors, PGRN is only expressed in invasive ovarian cancer and absent in low malignant potential cells[48]. PGRN promotes ovarian cancer cells proliferation and density-independent growth potential and increases S-phase fraction. PGRN is expressed in ovarian cancer cells and its level is upregulated by endothelin (ET-1), lysophosphatidic acid (LPA) through activation ERK signaling pathway[49]. Neutralizing PGRN antibody reversed basal as well as LPA, ET-1 and 8-CPT-induced ovarian cancer cell growth and induced apoptosis as demonstrated by caspase-3 and PARP cleavage, DNA fragmentation, and nuclear condensation[49]. Reducing PGRN level by enhancing elastase-mediated cleavage inhibited proliferation in a dose-dependent manner, while secretory leukocyte protease inhibitor (SLPI) protected PRGN from elastase-mediated degradation and restored its survival and proliferative function[50].
2.2.2. Genitourinary cancer
2.2.2.1. Kidney cancer
A few studies have linked PGRN with kidney cancer as well. He et al. reported that both transfection of PGRN gene and addition of recombinant PGRN protein into non-transformed renal epithelial cells resulted in clonogenicity in semisolid agar and increased mitosis in monolayer culture[4]. PGRN levels were low in benign renal tissue and higher in malignant renal tissue. In addition, immunohistochemistry (IHC) revealed that high-grade renal cell carcinoma exhibited higher levels of PGRN than that of low-grade renal cell carcinoma and normal tissue[51].
2.2.2.2. Prostate Cancer
Pan et al. reported that normal prostate epithelial cells have either no expression of PGRN or 10% of cells express very low levels of PGRN, while prostate intraepithelial neoplasia exhibits much higher levels of PGRN[52]. Analyzed in tumors from 442 Prostate Cancer patients, PGRN expression was positively correlated with the Prostate-Specific Antigen (PSA) value at prostatectomy and cytoplasmic cytokeratin 20 (CK20) staining; moreover, increased PGRN in tumor tissue was a prognostic factor because it was correlated with shorter follow-up times for overall, disease-specific and relapse-free survival[53]. PGRN expression is not correlated with age, Gleason score, pathological stage, status of lymph node metastasis, extra-prostatic extension, perineural invasion, surgical margins, and vascular invasion[52]. Tanimoto et al. reported that there is very low expression of Sortilin in prostate cancer cells; enhanced Sortilin expression reduced PGRN levels, and inhibited motility, invasion, proliferation, and anchorage-independent growth[54]. Similar to other types of cancer, PGRN is able to activate AKT and ERK signaling pathways, and promote cell migration and anchorage-independent growth in prostate cancers[55]. Knockdown of GRN in prostate cancer (PC-3) cells exhibited increased sensitivity to ionizing radiation[56].
2.2.2.3. Bladder Cancer
PGRN treatment, although doesn’t affect cell growth, promotes bladder cancer cells migration and stimulates invasion in vitro[57]. This pro-oncogenic function is mediated by activation of MAPK pathway[57]. Additionally, PGRN interacted with Drebrin, an F-actin binding protein, which mediated cell migration and F-actin remodeling in bladder cancer cells[58]. PGRN knockdown inhibits bladder cancer growth and sensitizes cancer cells to cisplatin[59].
2.2.3. Gastrointestinal cancer
2.2.3.1. Colorectal Cancer
Colorectal Cancer (CRC) accounts for the second and third most common cancers in women and men respectively. Overexpression of PGRN was highly observed in CRC, and its level closely related with increased expression of proliferation and angiogenesis markers, advanced stages and poorer prognosis of CRC[60]. An in vitro cell culture model showed that PGRN promoted the expression of Ki67 and VEGF-A as well as growth rate in CRC cells[60]. PGRN acts as a STAT3 cofactor and physically interacts with STAT3 in CRC cells, and its knockdown with a specific antisense oligonucleotide inhibits STAT3 activation and restrains the expression of STAT3-related oncogenic proteins, thus causing cell cycle arrest and apoptosis[61]. However, it’s important to note that this interaction wasn’t confirmed using alternative techniques like Immunofluorescence co-localization within the cell since PGRN is conventionally considered luminal to the ER/GOLGI/secretory system/lysosomes and therefore might not contact STAT3 which is conventionally extraluminal and found in the cytosol. Colorectal cancer stem cells (CSC) and oxaliplatin resistant cells exhibit enriched secretion of an array of soluble factors, including PGRN[62]. In animal models, PGRN introduction of mixed colorectal chemoresistant cells and parental cell populations results in tumor growth significantly [62].
2.2.3.2. Hepatocellular cancer
Hepatocellular carcinoma (HCC) is a prevalent liver cancer linked to risk factors like hepatitis infection, cirrhosis, and parasitic fluke exposure. PGRN has emerged as a significant player in HCC development. Elevated PGRN levels in HCC tissues and serum samples correlate with tumor characteristics and diminished survival[63]. Studies exploring PGRN’s therapeutic potential in HCC have yielded promising outcomes. Knocking down PGRN curbed HCC cell proliferation, invasiveness, and tumor growth in animal models[63]. Anti-PGRN antibodies reduced tumor growth by targeting signaling pathways and angiogenesis[64]. Cheung et al. [65] identified the PGRNhigh HCC subpopulation, characterized by the expression of stemness markers, such as Nanog, Oct4 and Sox2, which displayed an increased ability for colony formation and chemoresistance. They further demonstrated that when co-expressed with ABCB5, this subpopulation’s role in maintenance of CSC was enhanced.
PGRN also influences immune surveillance by NK cells. PGRN overexpression in HCC cells impairs NK cell-mediated cytotoxicity by affecting surface expression of MICA and HLA-E molecules. Inhibition of PGRN restores sensitivity to NK cytotoxicity, suggesting a key role in immunosurveillance evasion. PGRN’s interaction with growth factors like midkine promotes processes underlying proliferation and metastasis, potentially influencing poor prognosis[66]. Interestingly, PGRN’s granulin subunits display distinct effects. Granulin A inhibits cancer cell proliferation, induces apoptosis, and interacts with enolase 1[67].
2.2.3.3. Biliary Tract Cancer
Cholangiocarcinoma is a rare gastrointestinal malignancy that develops from the epithelial lining of intrahepatic or extrahepatic bile ducts. Both PGRN expression and secretion are elevated in vitro cholangiocarcinoma cell lines as well as in vivo patient liver biopsies and sera, at least in part, through IL-6-mediated activation of the ERK1/2/RSK1/C/EBPβ pathway. In cholangiocarcinoma, upregulated IL-6-mediated elevation of PGRN promotes tumor cell proliferation through Akt activation and nuclear extrusion of Forkhead box protein O1 (FOXO1), and in vivo knockdown of PGRN resulted in slowed tumor growth[68]. Further, PGRN expression and secretion are elevated and are stimulatory during cholangiocyte proliferation under normal conditions and in a model of cholestasis through inhibition of deacetylase Sirt1 and consequent acetylation-dependent extrusion of FOXO1[69].
2.2.3.4. Pancreatic cancer
Data from Surveillance, Epidemiology, and End Results Program (SEER)[70] revealed that the global burden of pancreatic cancer has more than doubled over the past 25 years and is expected to continue to represent a leading cause of cancer-related mortality. Although granulin A domain has been demonstrated to inhibit proliferation and induce apoptosis in PANC28 cell lines, tumor-but not macrophage-derived PGRN is associated with poor overall survival in pancreatic ductal adenocarcinoma (PDAC)[71, 72]. PGRN blockade restored MHC-I expression by inhibiting lysosomal activity and degradation of autophagosomes[73]. MHC-I molecules are selectively targeted for lysosomal degradation by an autophagy-dependent mechanism. Inhibition of autophagy restores surface levels of MHC-I reduced immune evasion in pancreatic cancer[74].
2.2.4. Neuron system cancer
Astrocytomas account for 75% of gliomas and are distinguished clinically by pathological grading. Elevated serum levels of PGRN have been associated with tumor recurrence in grade II astrocytoma[75] and expression analyses reveal a positive correlation between astrocytoma pathological grade and PGRN level, where glioblastoma (GBM) cells exhibit the most robust PGRN expression. These high total and vascular PGRN expression levels serve as prognostic markers indicative of poor overall survival [75, 76]. Recent work by Bandey et al has revealed that increased PGRN coincides with enhanced expression of DNA repair components and cancer stemness maintenance, which linked to overactive cFos/JunB through a feedforward loop between PGRN and transcription factor AP-1[77]. Importantly, in H4 glioma cells and S1R1 GBM cells, overexpression of PGRN is associated with reduced sensitivity to Temozolomide (TMZ) while PGRN deficient glioma cell lines demonstrate increased sensitivity to TMZ-induced cell death, which can be attenuated with PGRN expression[77].
2.2.5. Myeloma and Leukemia
In multiple myeloma patients, expression of PGRN in bone marrow correlates with disease state, in which strong expression is associated with active disease state and negative immunohistochemical staining among samples from patients in remission[78]. In vitro and animal models of multiple myeloma further reveal a pro-survival influence of PGRN during dexamethasone treatment[78, 79]. Chronic lymphocytic leukemia (CLL) cells secrete high levels of several angiogenic factors including PGRN[80]. Enhanced PGRN may limit the inflammatory and apoptotic response to increased TNF-α signaling in CLL cells through binding TNFRs, preventing TNF-α-mediated ripoptosome complex formation, and then leading to subsequent cell death[81, 82]. PGRN was also identified among the top 20 distinct genes associated with overexpression in prognosticly unfavorable ZAP-70+ CD38+ CLL[83]. Additional studies have supported the applicability of PGRN plasma levels as a prognostic marker in CLL, in which higher PGRN levels are associated with a shorter time between diagnosis to first treatment as well as reduced overall survival[84]. The molecular mechanism underlying altered PGRN expression observed in CLL is not well understood. However, microRNA(miR)-29b has been shown to bind the 3’ region of PGRN mRNA leading to PGRN downregulation and is associated with aggressive CLL[85, 86].
2.3. PGRN and lysosomal storage diseases
Lysosomal storage diseases (LSDs) encompass a collection of genetic metabolic disorders characterized by the progressive accumulation of undegraded substrates within lysosomes, resulting in aberrant lysosomal activity and homeostasis. PGRN plays a significant role in lysosomal function, highlighted by the fact that loss of PGRN causes GD-like phenotypes[30, 31] and homozygous GRN mutations lead to neuronal ceroid lipofuscinosis (NCL)[87]. Subsequent research has established several connections between its mutations/insufficiency and LSDs.
2.3.1. Neuronal ceroid lipofuscinosis
Neuronal ceroid lipofuscinosis (NCL), a rare type of LSDs that mainly affects the central neuron system, is characterized by the presence of autofluorescent storage material in lysosomes resembling ceroid and lipofuscin. By using exome sequencing in a family with two siblings who had NCL, a homozygous mutation in GRN genes were identified[87]. Neurons from Grn −/− mice show NCL-like lysosome accumulation under an electron microscope[87]. Arrant et al. found that nearly complete PGRN deficiency appears to be required to induce lipofuscinosis and gliosis in mice, while partial PGRN insufficiency is sufficient to produce behavioral deficits[88]. Lipofuscin accumulation in retinal pigment epithelium cells will lead to blindness. PGRN increases phagocytosis and reduces lipofuscin accumulation in retinal pigment epithelium[88].
2.3.2. Gaucher disease
Gaucher disease (GD), a common LSD, is caused by mutations in GBA1 resulting in a defective glucocerebrosidase (GCase) and the consequent accumulation of its substrate glucosylceramide (β-GlcCer) in macrophages and other cell types. GD has been regarded as wholly attributable to GBA1 mutations. However, clinical manifestations may have huge variations among patients carrying the same GBA1 mutations, ranging from very early disease onset to very mild clinical presentations. It has therefore been speculated that additional disease modifiers exist in GD patients. Interestingly, we reported that PGRN is a novel factor for GD[30, 31].
OVA-challenged or aged GRN −/− mice developed GD disease phenotype, which included large numbers of Gaucher cells in the lungs of GRN −/− mice, specific β-GlcCer accumulation, and typical tubular-like lysosomes seen under an electron microscope[31]. In addition, GD patients’ serum levels of PGRN were significantly lower than those of healthy controls, and GRN gene variants were identified in GD patients and at the molecular level, PGRN is required to deliver GCase to the lysosome[30]. PGRN functions as a co-chaperone molecule that recruits HSP70 to GCase to form a ternary complex, and facilitates GCase delivery to the lysosome[30]. Although PGRN binds to and co-localizes with GCase in all traffic compartments, as early as the ER and Golgi apparatus, the interactions of GCase with HSP70 highly depend on OVA challenge, which suggest that recruitment of HSP70 to the GCase complex through PGRN may be stress- or disease-dependent[30, 89]. Recombinant PGRN and its derived Pcgin, which contains the C-terminal 98-aa, which directly binds to GCase and HSP70, show therapeutic effects in treating GD. In order to investigate the impact of PGRN in GBA1 mutation-associated pathologies in vivo, the Grn −/−Gba 9v/9v (termed PG9V) mice were generated, which exhibited neurobehavioral deficits, early onset, and more severe GD phenotypes compared to Grn −/− and Gba 9v/9v mice. Pcgin was found to penetrate the blood-brain barrier and ameliorate the neuronopathic GD manifestations and PD pathology in PG9V mice[90].
2.3.3. Tay-Sachs disease
Tay-Sachs disease (TSD) is a recessively inherited lethal lysosomal storage disease and caused by mutations in the HEXA gene, which, together with HEXB, encodes the heterodimeric β-N-acetylhexosaminidase A (HexA) enzyme, consequently leading to accumulation of its substrate, GM2 ganglioside. A screening to isolate the therapeutic effect of PGRN on lysosomal storage in fibroblasts from 11 patients affected by various LSDs led to the discovery of TSD as another LSD that responded well to PGRN[91]. PGRN deficiency resulted in the manifestation of typical TSD phenotypes, such as the presence of zebra bodies. The application of recombinant PGRN showed a significant reduction in GM2 accumulation and lysosomal storage. Notably, we observed that PGRN deficiency caused the aggregation of HexA (but not HexB) in the cells. Further investigations into the mechanisms involved indicated that PGRN interacted with HexA through its granulin G and E domains. As a result, Pcgin, an engineered derivative of PGRN containing the granulin E domain, effectively increase the activities and lysosomal delivery of HexA and reduced GM2 accumulation[91]. In brief, PGRN holds potential in enhancing HexA activity and as a therapeutic approach for treating TSD.
2.4. PGRN and autoimmune diseases
Autoimmune diseases include a wide spectrum of diseases that are caused by overreacting the immune system or inducing immune responses to self-antigens during an imbalance of immune tolerance. A solid basis of data from in vitro and in vivo models is accumulating to clarify the roles and molecular mechanisms of PGRN in autoimmune disease. PGRN functions as a double-edged sword in playing the pro-inflammatory role and anti-inflammatory role in immune-mediated diseases. The serum PGRN levels increased significantly and positively correlated with serum proinflammatory factor levels like IL-6 and TNF-α compared with healthy controls in a cohort comprising 30 individuals newly diagnosed with severe systemic lupus erythematosus (SLE) and an equal number of healthy control subjects[92]. Serine proteases can digest PGRN into individual granulin units, which are actually pro-inflammatory and can neutralize the anti-inflammatory effects of intact PGRN[93, 94]. Furthermore, PGRN stimulates secretion of interleukin-8 (IL-8) by epithelium cells in multiple sclerosis (MS), a primary chemotactic agent for neutrophils and monocytes, while PGRN is short of this activity[94, 95]. And Progranulin antibodies are present in patients with systemic sclerosis and other autoimmune connective tissue disorders, like primary systemic vasculitides, rheumatoid arthritis (RA) and systemic lupus erythematosus (SLE)[96]. Given the pivotal role of TNF-α in the inflammatory pathways of autoimmune diseases, the presence of progranulin antibodies may potentially exacerbate the proinflammation by counteracting the TNF-blocking capabilities of progranulin.
In contrast to its proinflammatory action, more emphasis has been placed on its anti-inflammatory properties. Centrally, PGRN directly binds to TNFR1 with comparable affinity as to TNF-α, and a higher affinity to TNFR2 than TNF-α[82]. Thus, PGRN directly blocks TNF-mediated inflammatory response through TNFR1, and PGRN also triggers a protective role through activating TNFR2[82]. In addition to being an antagonist of TNF, PGRN acts as the key component for anti-inflammation through other mechanisms. First, PGRN induces CD4+ Foxp3+ regulatory T cells (Treg) in different disease models, including inflammatory arthritis, colitis, and psoriasis vulgaris[82, 97, 98]. Second, PGRN induces IL-10 production in various mouse models, including arthritis, colitis and neuronal injury[82, 97]. IL-10 is a major anti-inflammation cytokine in the immune system to limit immune response against pathogens. PGRN stimulates IL-10 mainly from the Treg population; it depends on TNFR2 and downstream activation of JNK pathway[97]. Third, PGRN inhibits chemokine production from macrophage to limit inflammation. Several chemokines, especially CXCL9 and CXCL10, were upregulated in GRN −/− macrophage, revealing that PGRN protein inhibits CXCL9 and CXCL10, and mainly depends on TFNR1[99].
2.4.1. Rheumatoid Arthritis
Rheumatoid Arthritis (RA) is a systemic autoimmune disorder that primarily affects the joints, in which the wrists and hands are most commonly affected. Although the cause of RA is still unclear, it is believed that TNF-α driven inflammation plays a critical role, and TNF inhibitors are regarded as the most effective treatments for RA. Grn −/− mice have more severe arthritis phenotype in a collagen-induced arthritis (CIA) model[82]. Similarly, deletion of one or both copies of the GRN gene in TNF-transgenic (TNF-Tg) mice significantly accelerates the onset of arthritis[82]. Injection of recombinant PGRN dramatically rescued the RA progression in different inflammatory arthritis models[82]. Atsttrin, composed of half-units of granulins A, C, and F, plus linkers P3, P4, and P5, preserves TNFR-binding domains and PGRN’s anti-inflammatory effects but is devoid of the oncogenic potential with extended half-life (about 120 h) compared with PGRN (about 40 h)[82]. In CIA model, Atsttrin surpasses PGRN in treating rheumatoid arthritis [82]. We also have identified 14-3-3ε as a significant regulator of macrophage plasticity, acting as a crucial cofactor in PGRN/TNFR2 signaling in RA progress. The PGRN/TNFR2/14-3-3ε pathway plays a critical role in determining the balance between proinflammatory and anti-inflammatory responses in macrophages[100].
The serum levels of PGRN were significantly increased in both RA and osteoarthritis (OA) patients, and within synovial fluid the PGRN level was much higher in RA than that of OA patients. Interestingly, the balance between PGRN and TNF is very important for RA progression, as the PGRN/TNF ratio was correlated to the stage of the disease in RA patients[101]. More convincing evidence of PGRN’s implication in autoimmune diseases comes from the discovery of a PGRN auto-antibody in RA patients, as well as patients from other autoimmune diseases[102–104]. It is found that the PGRN auto-antibody recognized hyper-phosphorylated Serine 18, and further interfered with PGRN binding to TNFR1 and TNFR2. It has been observed that hyperphosphorylated progranulin (specifically, at Serine 81, a site within the epitope region recognized by progranulin antibodies) was exclusively identified in progranulin-antibody-positive patients, which impeded the interaction, and consequently, the inhibition of TNF to TNFR, thereby signifying proinflammatory effect [105].
2.4.2. Inflammatory bowel disease
Inflammatory bowel disease (IBD) is a type of autoimmune disease that primarily affects the digestive tract, this type of disease includes ulcerative colitis (UC) and Crohn’s disease (CD). PGRN has been involved in the development of IBD. PGRN autoantibodies were detected in 16.31% of patients with Crohn’s disease and 21.13% of patients with ulcerative colitis. GRN −/− mice have more severe inflammation and are more susceptible in both DSS- and TNBS-induced colitis models and T-cell transplantation-induced colitis model[106]. Administration of recombinant PGRN ameliorated the pathology and reduced the histological score in both DSS and TNBS colitis models[106]. Furthermore, PGRN-mediated protection depends on TNFR2 and IL-10. Therapeutic function of PGRN was dramatically compromised when blocking IL-10 by a specific neutralizing antibody or in TNFR2 −/− mice[106]. Interestingly, PGRN and its derived Atsttrin bind to TNFRSF25 (DR3) in addition to TNFR1 and TNFR2[107]. Both PGRN and Atsttrin directly bound to DR3, thus blocking TL1A, the ligand of DR3[107]. Atsttrin also neutralized TL1A-enhanced osteoclastogenesis in vitro, and exhibited a protective function in the DSS-induced colitis model[107].
2.4.3. Psoriasis
Psoriasis is a chronic inflammatory disease characterized by skin lesions and systemic inflammation. Pathophysiology of psoriasis is a complex immune-mediated inflammation that leads to hyperproliferation of keratinocytes and infiltration of immune cells in skin. Numerous cytokines, including the IL-17 family, TNF, and IFN-α are considered as major cytokines involved in disease development. Serum PGRN levels are dramatically incremented in psoriasis patients and PGRN were also found to be significantly increased in the psoriasis-like lesions of 12-O-tetradecanoylphorbol 13-acetate (TPA)-treated WT mice[108]. In Grn −/− mice, following the application of imiquimod to induce a psoriatic skin lesion model, psoriasis-like damaged appearance and epidermal thickening were aggravated compared with WT mice, meanwhile, proinflammatory cytokine IL-17A levels was increased as well [109]. The paradoxical phenomenon may be attributed to the simultaneous elevation of both anti-inflammatory PGRN and pro-inflammatory cytokines like IL-17 in psoriasis. However, the ratio of PGRN to inflammatory cytokines actually decreases, which may reflect a feedback regulatory mechanism, a phenomenon reported in other inflammatory diseases, including rheumatoid disease.
2.5. PGRN and musculoskeletal disease
PGRN is closely associated with the occurrence and development of various musculoskeletal diseases[10, 11, 110–112]. In addition to promoting inflammation, TNF-α contributes to osteoclastogenesis and inhibit osteoblast differentiation[113]. PGRN binds to TNFR2 and block TNF-α, thus stimulates anabolic and regenerative pathways in musculoskeletal system, particularly in cartilage and bone. Consequently, PGRN plays a crucial role in cartilage homeostasis (Figure 3), probably bone remodeling as well, in conditions such as osteoarthritis, osteoporosis and fracture healing.
Figure 3. A proposed model for explaining the role of PGRN in the cartilage and bone homeostasis.
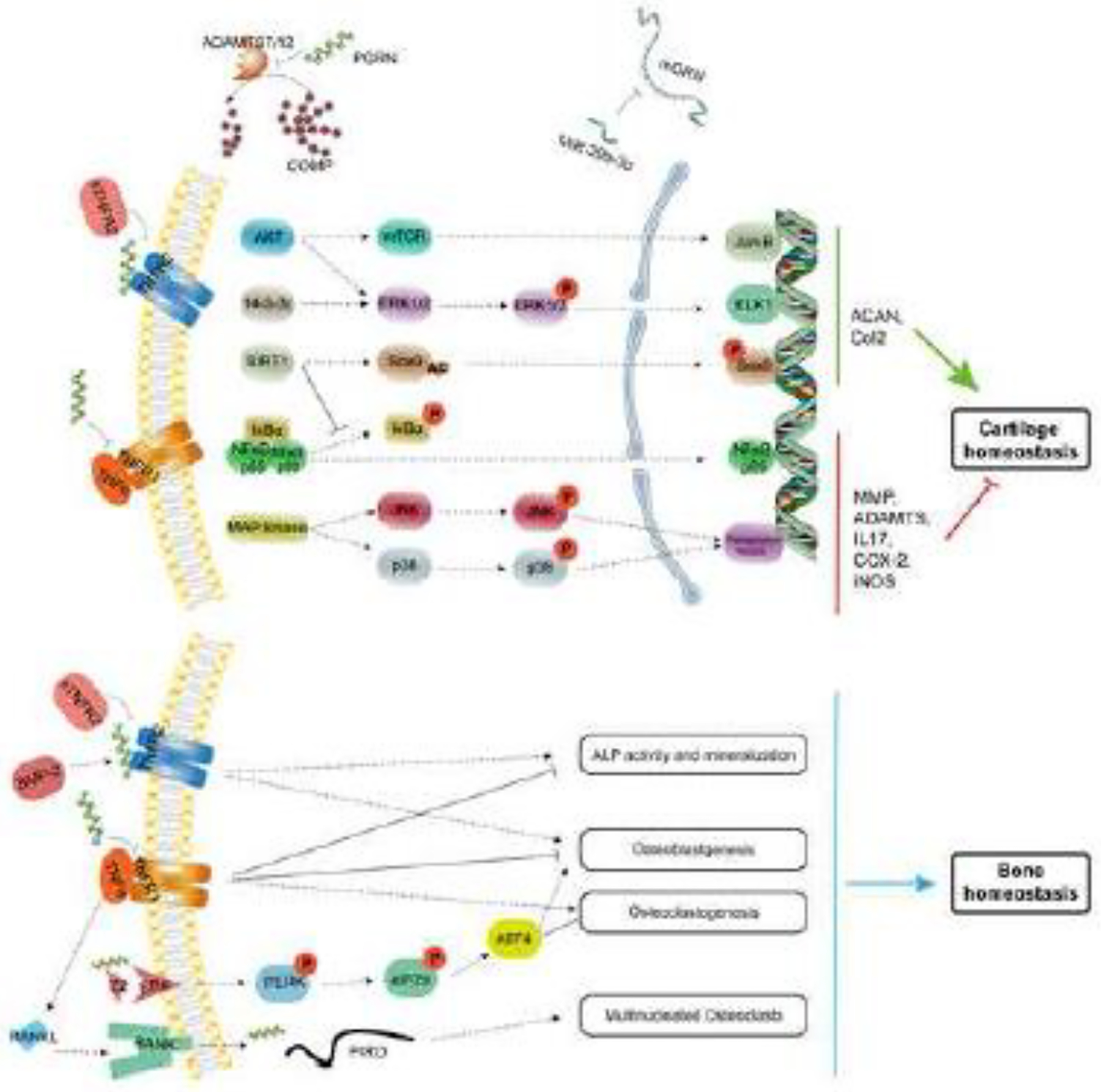
PGRN promotes chondrocyte proliferation, differentiation, and plays a chondroprotective role through activating TNFR2 anti-inflammatory and anabolic pathway. Additionally, PGRN competitively binds to TNFR1 and blocks TNF-α mediated inflammatory and catabolic process. Via suppressing TNFR signaling, PGRN also regulates osteoclastogenesis and osteoblastogenesis, thus maintaining the bone homeostasis
2.5.1. Osteoarthritis
Osteoarthritis (OA) is chronic degenerative joint disease that mainly affects weight-bearing joints, such as hip, knee, and ankles. Growing evidence indicates that OA is a chronic mild inflammatory disease. PGRN is expressed in chondrocytes and it binds to ADAMTS-7 and ADAMTS-12 to inhibit degradation of cartilage oligomeric matrix protein (COMP) [114–116]. Aged Grn −/− develop OA-like phenotype spontaneously, and in a surgical-induced OA model, PGRN-deficient mice showed an exaggerated breakdown of cartilage structure and OA progression. Local injection of recombinant PGRN attenuated degradation of cartilage matrix and protected against OA development. In addition, protective function of PGRN is also shown to be primarily mediated via TNFR2[113]. Further, PGRN/TNFR2/14-3-3ε signal works through activating ERK-dependent Elk-1 in regulating chondrocyte anabolism[117]. PGRN also shows anti-inflammation effects by inhibiting TNF-triggered catabolic pathways. PGRN inhibits TNF-mediated loss of proteoglycan in human cartilage. The catabolic markers induced by TNF such as MMP13, ADAMTS-5, COX-2, and iNOS were dramatically suppressed by PGRN[113].
PGRN and its derived Atsttrin have shown promising therapeutic effects in treating OA in different animal models[118–121]. In a PGRN-releasing hyaline cartilage graft model, PGRN protected cartilage grafts from degradation in an inflammatory environment. Intra-articular injection of MSC-Atsttrin prevented the progression of degenerative changes in the surgically induced OA mouse model[122]. Atsttrin delivered by an injectable hydrogel significantly protected against post-traumatic osteoarthritis (PTOA) onset and progression in a rabbit anterior cruciate ligament transection model[123].
Recently, it was reported that intra-articular injection of etanercept (sTNFR2), known to be effective in treating patients with RA, caused more severe joint destruction in a mouse model of OA[124]. Of note, sTNFR2 is not a TNF-α-specific inhibitor; it is also a PGRN blocker. sTNFR2 likely inhibits PGRN much more efficiently than it inhibits TNF-α, since PGRN exhibits an approximately 600-fold higher binding affinity than does TNF-α to TNFR2[2]. Accordingly, these seemingly paradoxical results may be explained through the importance of PGRN’s protective role in the pathogenesis of OA[124].
2.5.2. Fracture
A fracture refers to a broken bone resulting from physical force or trauma applied to the bone. Bone healing involves a series of coordinated processes, including hematoma formation, cartilage callus formation, calcification, ossification, and bone remodeling. The identification of PGRN as a regulator in cartilage and bone regeneration offers promising prospects for potential therapeutic interventions in clinical bone healing. Furthermore, the discovery that PGRN acts as a regulatory factor by modulating BMP-2 and TNF/TNFR signaling highlights its ability to promote chondrocyte proliferation, differentiation, and endochondral ossification during development[125–127]. PGRN was essential for BMP-2 induction of osteoblastogenesis and ectopic bone formation. Additionally, PGRN promoted bone regeneration in deficiency of TNFR1 but lost such effect in TNFR2 deficient mice, revealing that PGRN’s ability to mediated bone formation is primarily dependent on the presence of TNFR2, but not TNFR1[128]. Patients with diabetes are at a higher risk of bone fractures and experience significant delays in the healing process. Utilizing a closed bone fracture model, local administration of recombinant PGRN was found to effectively promote the healing of bone fractures in diabetic animals. By suppressing TNF-α/TNFR1 inflammatory signaling and activating TNFR2 anti-inflammatory and regenerative pathway, PGRN and its derivatives can potentially improve the healing outcomes for diabetic patients with bone fractures[112, 129], and probably other impaired fracture healing as well.
2.5.3. Osteoporosis
In a cross-sectional study involving 244 obese participants suggested a potential association between central obesity and elevated levels of PGRN. Furthermore, the study indicated a direct correlation between PGRN concentration and bone formation parameters, implying a protective role of PGRN in maintaining bone density[130]. Another study included total of 171 obese participants (BMI ≥30) showed that in osteopenic group, PGRN concentration was significantly decreased[130]. TNF-α induces RANKL production from various cells and contributes to osteoclastogenesis via RANKL/RANK axis. Oh’s research revealed genes named PIRO (PGRN-induced receptor-like gene during osteoclastogenesis), which were significant activated (20-fold) by PGRN. This finding suggests that PIRO may serve as a direct target for the formation of multinucleated osteoclasts under the influence of PGRN[131]. Wang, et al. reported that PGRN blocks TNF-α-mediated inhibition of osteoblast differentiation[132]. PGRN enhanced ALP activity and the expression of osteoblast-related mRNAs (ALP, osteocalcin). Additionally, PGRN stimulated osteoblastic differentiation by inhibiting the phosphorylation of ERK1/2 in mouse calvarial cells[133]. Risedronate, a nitrogen-containing bisphosphonate commonly employed for osteoporosis treatment, has been found to stimulate the expression and secretion of PGRN, which suggested that PGRN is one of the major molecular targets of risedronate, and in turn PGRN inhibits osteoclastic bone resorption. Further study demonstrated that recombinant human PGRN at a concentration of 6nM stimulated the phosphorylation of mitogen-activated protein kinase in both Hobit (a human osteoblast-like cell line) and osteocytic cells and induced cell proliferation and survival[134]. Additionally, PGRN has a regulatory function in the maintenance of bone homeostasis by modulating the estrogen (E2)/ERα signal. This regulation occurs through the activation of PERK/p-eIF2 signaling pathway[135].
2.6. PGRN and cardiovascular and metabolic diseases
PGRN has been found to play a role in adipocyte function, insulin resistance, energy metabolism, and inflammation, all of which are interconnected with the development of metabolic disorders. When discussing the role of PGRN in cardiovascular diseases, highlighting the interaction between PGRN and TNFR2 and underscoring its anti-inflammatory characteristics is crucial. Investigations into the role of PGRN in cardiovascular and metabolic diseases could potentially lead to novel therapeutic strategies and interventions in these areas.
2.6.1. Cardiovascular diseases
Kojima et al. reported that PGRN is expressed in atherosclerotic plaques in human carotid endoatherectomy specimens[136]. IL-8 secretion from human aortic smooth muscle cells (HASMCs) was reduced by forced expression of PGRN and increased by RNAi-mediated knockdown of PGRN[136]. Kawase et al. reported that PGRN level is higher in macrophages than that in smooth muscle cells of atherosclerotic lesions in ApoE −/− mice fed on a high-fat diet[137]. PGRN is protective in atherosclerotic lesions development, as Grn −/− and ApoE −/− mice exhibited severe atherosclerotic lesions, accumulated excessive cholesterol in the macrophages, and altered HDL-associated proteins[137]. Interestingly, PGRN is found to regulate vascular tone. PGRN pre-treatment augmented acetylcholine-induced endothelium-dependent relaxation. This effect is mainly through activation of acetylcholine-induced nitric oxide-mediated relaxation through the increased cGMP production in smooth muscle. Grn −/− mice are susceptible to homocyste-ineinduced renal and cardiac injury, and PGRN’s protective function seems to reply to PGRN-triggered Wnt/β-catenin signaling pathway[138].
2.6.2. Ischaemic stroke
Ischemia is a restriction in blood supply to tissues, causing a shortage of oxygen and glucose needed for cellular metabolism. In acute ischaemic stroke patients, serum PGRN levels were significantly higher and were correlated with body mass index, alcohol consumption, fasting blood glucose, total cholesterol, and high-density lipoprotein cholesterol[139]. Grn −/− mice significantly aggravated renal injury[140]. Recombinant PGRN attenuated hypoxia-induced inflammatory actions and apoptosis in proximal tubule epithelial cells. Moreover, PGRN could effectively ameliorate ischemia-reperfusion injury in the brain and in the heart by suppressing inflammation. PGRN could protect against acute focal cerebral ischaemia by a variety of mechanisms including attenuation of blood-brain barrier (BBB) disruption, neuroinflammation suppression, and neuroprotection. Grn −/− mice have more BBB disruption and massive brain hemorrhages[141]. Egashira et al. also reported that recombinant PGRN protein suppresses neutrophil recruitment during brain ischaemic stroke, reduces TNF-induced inflammation in the brain, and reduces inflammatory cytokine production[142]. PGRN also accelerates ischemic neurogenesis and cell proliferation and thus reduces anxiety and learning/memory spatial disorders post-cerebral ischemia[143]. It’s also important to note that PGRN may have a role in regulating vascular permeability by activating the NF-κB-VEGF pathway in post-brain ischemia[44, 144].
2.6.3. Diabetic Mellitus
Diabetes mellitus (DM) is marked by persistent high blood glucose due to insufficient insulin production (type 1) or insulin insensitivity (type 2). Circulating PGRN correlates positively with diabetes markers like BMI, waist circumference, inflammation, and cholesterol levels. In type 2 DM, PGRN levels rise and are reduced by exercise or dietary changes[145]. PGRN’s impact on DM progression is evident in studies. It contributes to insulin resistance, obesity, and inflammation. In murine models, Grn−/− mice are protected against high-fat diet-induced insulin resistance and obesity. PGRN-induced insulin resistance involves TNFR1-mediated IL-6 production, ER stress, and autophagy disruption[146]. PGRN’s association with microvascular complications in DM is apparent in elevated serum levels among patients with diabetic nephropathy and retinopathy[147], suggesting potential kidney damage correlation.
3. Update of binding partners of PGRN
PGRN is a unique protein with seven and half cysteine rich domains; there is no other homolog protein that shares a similar domain structure. Another typical characteristic is that PGRN has diverse biological functions at different distributions, including extracellular matrix, membrane receptors, intracellular organelles, and nuclear distribution (Figure 4 and Table 2).
Figure 4. Schematic of the binding partners of PGRN.
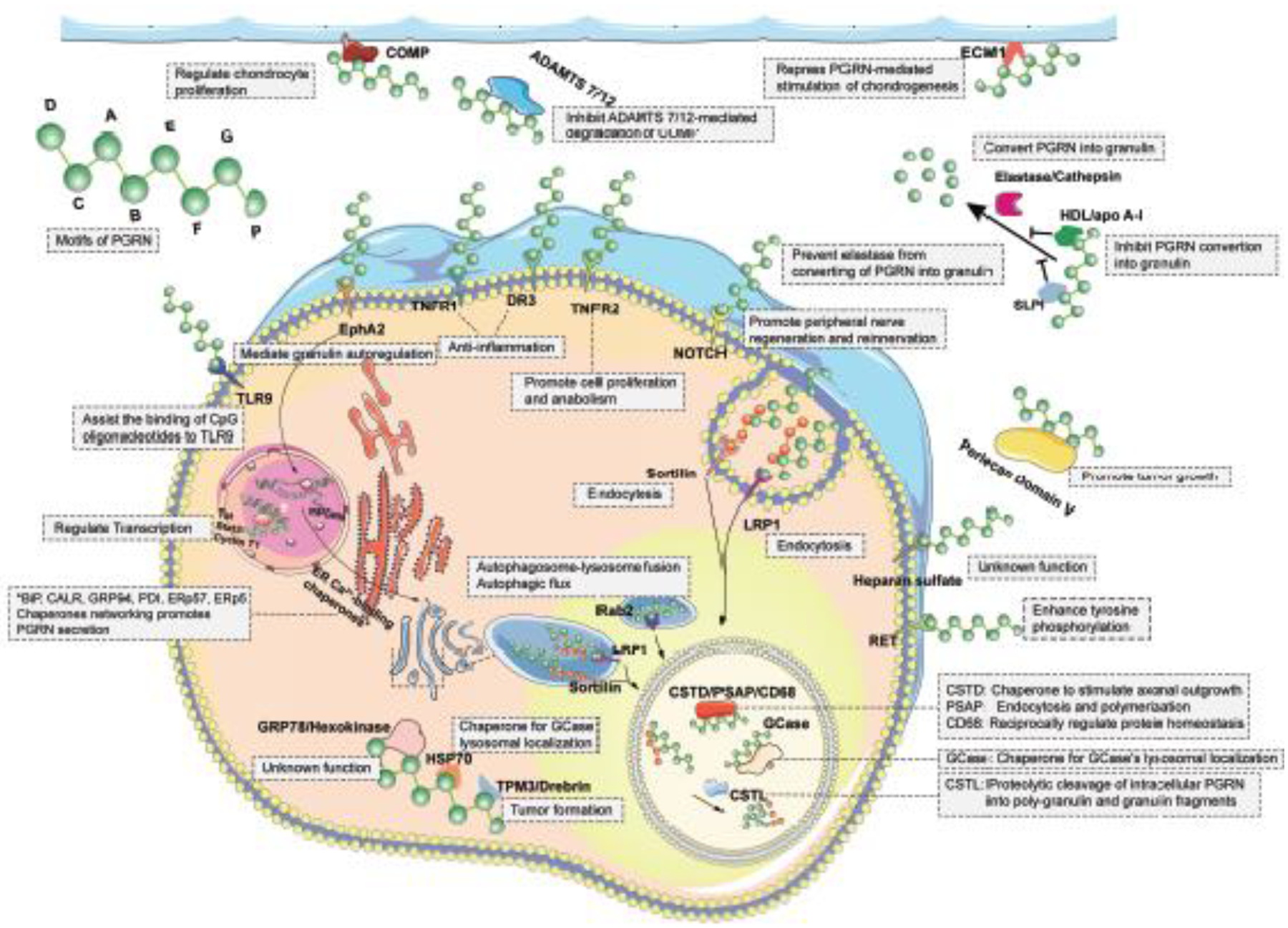
PGRN interacts with its binding proteins at various locations, including extracellular matrix, cell membrane, intracellular organelles, and nuclear. The specific binding partners evoke the indicated pathways leading to multiple biological process.
Table 2.
PGRN-interacting proteins
| PGRN associated proteins | Interaction sites | Interaction domain PGRN | Function |
|---|---|---|---|
| SLPI | Circulation | NA | Prevent elastase from converting of PGRN into GRN |
| HDL/apo A-I | Circulation | NA | HDL inhibits PGRN convertion into GRN |
| ADAMTS7 | Matrix | PGRN A-G | Inhibition of ADAMTS 7 activity |
| ADAMTS12 | Matrix | PGRN A-G | Inhibition of ASAMTS 12 activity |
| COMP | Matrix | PGRN A | COMP enhances PGRN-induced proliferation |
| Perlecan Domain V | Matrix | PGRN F+B | Promotion of tumor growth |
| ECM1 | Matrix | PGRN E+linker | Chondrogenesis |
| TNFR1 | Membrane receptors | PGRN F+A+C | Anti-inflammation |
| TNFR2 | Membrane receptors | PGRN F+A+C | Promote cell proliferation and anabolism |
| DR3 | Membrane receptors | PGRN F+A+C | Anti-inflammation |
| Sortilin | Membrane receptors | C-terminal QLL | Endocytosis |
| Toll-like receptor 9 (TLR9) | Membrane receptors | PGRN ACDE | Assist the binding of CpG oligonucleotides to TLR9 |
| Notch | Membrane receptors | NA | Promote peripheral nerve regeneration and reinnervation |
| EphA2 | Membrane receptors | NA | Mediate GRN autoregulation |
| Heparan sulfate | Membrane receptors | PGRN E | Unknown function |
| RET | Membrane receptors | NA | Enhance tyrosine phosphorylation |
| GRP78/HSPA5 | Cytoplasm | NA | Unknown function |
| GCase | Lysosome | PGRN F E | Chaperone for GCase’s lysosomal localization |
| Hexokinase | Cytoplasm | NA | Unknown function |
| HSP70 | Cytoplasm | PGRN E | Chaperone for GCase lysosomal localization |
| TPM3 | Cytoplasm | NA | Promotion of hepatocarcinogenesis |
| Drebrin | Cytoplasm | NA | Tumor formation |
| Cathpesin D (CSTD) | Lysosome | PGRN E | Chaperone to stimulate axonal outgrowth |
| Cathpesin L (CSTL) | Lysosome | NA | Mediate the proteolytic cleavage of intracellular PGRN into poly-granulin and granulin fragments |
| Prosaposin (PSAP) | Lysosome | PGRN D E | Endocytosis and polymerization |
| Rab2 | Lysosome | PGRN E | Autophagosome-lysosome fusion and autophagic flux |
| CD68 | Lysosome | PGRN E | Reciprocally regulate their protein homeostasis[424] |
| BiP, Calreticulin, GRP94, ERp57, ERp5,PDI | Endoplasmic reticulum | NA | Chaperones networking promote PGRN secretion [401] |
| Cyclin T1 | Nucleus | NA | Transcription regulation[432, 434] |
| Tat | Nucleus | NA | Transcription regulation[433, 434] |
| Stat3 | Nucleus | NA | Transcription regulation[175] |
3.1. Extracellular binding partners
By using a yeast-two-hybrid system, PGRN is found binding to COMP through the interaction between the PGRN A domain and the EGF-like domain of COMP[126]. In a follow-up study, PGRN has been found to bind to matrix metalloproteinase ADAMTS7/12, and PGRN inhibits ADAMTS 7/12-mediated degradation of COMP[114, 125]. Perlecan was found to bind to PGRN through the interaction between Grn F-B domain of PGRN and the first two laminin- and EGF-like repeats of Perlecan[148]. Extracellular matrix protein 1 (ECM1) is a matrix protein that is highly expressed in cartilage and bone, as well as other connective tissues. Kong et al. reported that ECM1 is a regulator of chondrogenesis through the interaction with PGRN. PGRN E domain and both side linker regions are responsible for binding to ECM1[149]. PGRN was also found to bind to heparan sulfate in the liver cell line through its C-terminal PGRN E domain. Suppression of a specific heparan sulfate proteoglycan, glypican-3, also showed a partial reduction of PGRN binding and an inhibition on PGRN-mediated activation of AKT[150]. PGRN is also a growth factor-like molecule that is present in extracellular fluid systems. Secretory Leukocyte Protease Inhibitors (SLPI) have been demonstrated to bind with PGRN, and SLPI inhibits the inflammatory response by protecting PGRN’s cleavage to granulins[151].
3.2. Membrane receptors
3.2.1. TNFR1/2 and DR3
PGRN binds to both TNFR1 and TNFR2[82, 152, 153], providing a clear rationale for PGRN’s anti-inflammatory properties. However, certain studies failed to replicate the interaction between TNFR1/2 with PGRN[154–156]. Subsequent reports have revealed that these discrepancies may be attributed to differences in experimental conditions and/or the source of PGRN used.PGRN binds to TNFR1 and blocks TNF-α/TNFR1-mediated signaling pathways and inflammatory cascades[82], whereas PGRN directly binds to and activates the TNFR2 protective signaling pathway[82, 97, 106]. In addition, Krabbe et al. reported that Grn −/− mice develop obsessive–compulsive behavior, and that it is caused by un-antagonize TNF in Grn −/− mice[157]. Deletion of the IKKβ gene in Grn −/− mice completely blocked TNF-induced obsessive–compulsive behavior. Wang, et al. reported that PGRN blocks TNF-α-mediated inhibition of osteoblast differentiation[132]. The most compelling clinical evidence to support the interaction between PGRN and TNFR is the discovery of PGRN auto-antibodies in patients with autoimmune diseases, such as rheumatoid arthritis, colitis, vasculitis, and psoriatic arthritis[102–104]. Hyperphosphorylated progranulin was exclusively identified in progranulin-antibody-positive patients, which impeded the interaction, and consequently, the inhibition of TNF binding to TNFR, thereby signifying the proinflammatory effect [105].
3.2.2. Sortilin
An unbiased, genome-wide screen of healthy controls and FTLD patients identifies that Sortilin is highly associated with PGRN, suggesting that Sortilin is a PGRN regulator[158, 159]. However, unlike the interaction between PGRN-TNFR, PGRN binding to Sortilin does not require proper conformation, as disrupting PGRN conformation by DTT does not lose, but enhances the binding activity to Sortilin[152, 159]. Additionally, PGRN promotes neurite growth independent of Sortilin[160]. Similar discovery was reported by De Muynck et al. in which PGRN’s neurotropic function depends on PGRN E domain but is independent of Sortilin[20]. Sortilin is an important membrane receptor for PGRN endocytosis, vesicle trafficking and degradation. It is also less likely a functional receptor to PGRN as Soritlin is not responsible for PGRN activities, such as neurotrophic function, anti-inflammation, and cell proliferation.
3.2.3. Notch receptors
Altmann et al. reported that PGRN forms a complex with extracellular domain of Notch 1–4. Both transgenic PGRN and recombinant PGRN protein induce Notch downstream Hey1 gene transcription[161]. Given the importance of the Notch signaling pathway, the discovery of PGRN as a binding protein of Notch receptors is interesting. It opens up our understanding of PGRN function and leads to a new field. However, there are several questions that still remain unclear: 1) the Co-IP assay will pull-down protein complex, it is unclear whether PGRN directly binding to Notch receptors, or whether other co-factor(s) are needed for PGRN/Notch interactions; 2) the binding affinity between Notch receptors and PGRN is unclear, and which granulin is responsible for binding to Notch receptors; 3) what biological function of PGRN is mediated through Notch signaling.
3.2.4. RET receptors
RET (Rearranged during Transfection) was defined as an oncogene activated by DNA rearrangement. RET encodes a transmembrane tyrosine kinase featuring a distinctive extracellular domain. This domain comprises four cadherin-like domains and a cysteine-rich region encompassing 16 cysteine residues within a span of 120 amino acids. RET, a receptor tyrosine kinase (RTK) signaling subunit of the neurotrophic nerve growth factor (GDNF) receptor, was identified as one of the putative cell membrane proteins that binds to PGRN[162]. Interestingly, GFRA2, an accessory subunit of the GDNF receptor that presents GDNF to RET, has been recognized as a significant genetic risk factor for FTD-GRN. The significance of PGRN interaction with RET highlights further investigation.
3.2.5. Eph receptors
Eph receptors form the largest known subfamily of receptor tyrosine kinases, and their ligand is the membrane protein, Ephrin. The interaction between Eph receptors and Ephrin mediated important biological functions, such as embryonic development including axon guidance[163], formation of tissue boundaries[164], and cell migration[165]. By using antibody array screening, Neill, et al. reported that recombinant PGRN protein induces phosphorylation of Eph receptors, including EphA2, EphA4, and EphB2, among which EphA2 gave the most significant signal[166]. Co-IP and solid phase binding assay showed that PGRN binds to EphA2. Neill, et al. concluded that PGRN stimulates MAPK and AKT activation mainly through EphA2 receptor, as blocking EphA2-Ephrin interaction with lithocholic acid (LCA) blocked PGRN-induced activation[166].
3.3. Intracellular PGRN binding partners
3.3.1. Lysosomal proteins
PGRN binds to GCase at two binding sites, one with higher affinity and the other one with lower affinity. The binding affinity changes as the pH reduces from a neutral pH7.4 to more acidic conditions of pH5.5, which mimics the trafficking from ER to lysosome. PGRN is required to deliver GCase to the lysosome[30]. Prosaposin (PSAP) is reported to interact with PGRN and the interaction between them modulate the metabolism of lipids, particularly sphingolipids[167]. PSAP is a cofactor of GCase, and mutation of the PSAP gene also causes GD. PGRN binds to GCase through the Grn F and E domains, and binds to PSAP through GRN D and E domains, while PSAP binds to GCase through saposin C and binds to PGRN through the linker region between Saposin B and C. Therefore, the three proteins bind to each other, and their functions are tightly linked to each other. Deficiency in any of those proteins is expected to affect GCase activity and contribute to GD development.
TMEM106B is another important lysosomal component that is functionally closely related, although no directly binding evidence has been revealed. Emerging evidence revealed that TMEM106B and PGRN share many disease entities, for example Alzheimer’s disease, amyotrophic lateral sclerosis and Parkinson’s disease. Heterozygous loss-of-function mutations in GRN represent ~ 20–25% of cases of FTLD-TDP, yet individuals with GRN mutations who also carry a TMEM106B ‘protective’ haplotype have approximately 50% lower odds of developing FTLD symptoms[168]. Endogenous neuronal TMEM106B co-localizes with PGRN in late endolysosomes[169]. Recently, PGRN was reported to physically interact with Rab2, a critical molecule in autophagosome-lysosome fusion. Additionally, a fragment of PGRN containing the PGRN E domain was required and sufficient for binding to Rab2. Further, this fragment ameliorated PGRN-deficiency associated impairment of autophagosome-lysosome fusion and autophagic flux[170].
3.3.2. Endoplasmic reticulum
By using a co-immunoprecipitation and mass spectrometry, Almeida et al. identified that PGRN binds to several endoplasmic reticulum proteins, including BiP, Calreticulin, GRP94, PDI, ERp57, ERp72, ERp5, and HSP70–1[171]. Based on their report, PGRN is a substrate of the ER chaperone complex that regulates PGRN, as knockdown of ERp57 will reduce PGRN secretion[171]. Recently ERp57 was reported to be as a novel binding partner for PGRN. Notably, when ERp57 was deficient, the therapeutic benefits of both PGRN and its derivative Pcgin were largely abolished. Recombinant ERp57 reinstated the therapeutic effects of PGRN and Pcgin in type 2 GD patient fibroblasts deficient in ERp57[172]. Additionally, PGRN was reported to function as co-chaperone of HSP70[30].
3.3.3. Cytoplasm
Type III hexokinase was identified to bind to PGRN by yeast hybrid screening[173]. Hexokinase is an important enzyme in energy metabolism. PGRN has been found to interact with cytoskeletal proteins such as beta-catenin and tropomyosin[113, 174]. PGRN may play a critical role in maintaining homeostasis of articular cartilage through associating with Wnt/β-catenin signalling pathway in the chondrocyte[113]. Tropomyosin 3 (TPM3) was found to bind to PGRN in human hepatocellular carcinoma according to Co-IP followed with mass spectrometry assay[175]. Although the biological function of PGRN-TPM3 interaction is unclear, it is believed that PGRN contributes to cancer cell proliferation[175].
3.3.4. Nucleus
Hoque et al. reported that PGRN binds with Cyclin T1 in the nucleus[176]. Although Hoque et al. didn’t further investigate which individual granulin domain binds to Cyclin T1, it seems that PGRN binds to Cyclin T1 at two binding sites, similar to the cases in GCase. Interestingly, Yeh, et al. identified that PGRN binds to STAT3 in breast cancer cells[97]. PGRN promotes STAT3 nuclear translocation and enhances constitutive or maximal cytokine-induced STAT3 transcriptional activity. In addition, PGRN promotes Treg and IL-10 production through enhanced activation of STAT3[97].
4. PGRN: a promising therapeutic target
PGRN imbalance is involved in multiple disease contexts – we collectively identify this as Progranulinopathy (listed in Figure 5). Neurodegenerative diseases, including FTLD, Alzheimer’s disease, and Parkinson’s disease, have been linked to a deficiency of PGRN. Although the exact underlying mechanisms are not fully understood, there is growing evidence that boosting PGRN levels in CNS system may be therapeutic to these debilitating conditions. Due to its large molecular size, PGRN faces an obstacle in crossing BBB. Recognizing this challenge, researchers have turned their attention to a promising solution: the fusion of PGRN with a BBB protein transport vehicle like DNL593 in investigational new drug-enabling studies[177]. In addition to the fusion protein approach, other methods to enhance PGRN levels have been proposed, showcasing alternative avenues for therapeutic intervention. The first one is intraventricular injection of a PGRN expressing virus. By directly introducing PGRN into the central nervous system, this method bypasses the challenges posed by the BBB, allowing for targeted delivery to the affected neurons. The in vivo animal experiment provides compelling evidence of the therapeutic efficacy of this approach, as injection of Adeno-associated virus, PGRN corrected lysosomal abnormalities in neurons[178]. To assess the potential of raising PGRN levels, two separate Phase 1/2 studies, namely the PROCLAIM trial (NCT04408625) and the upliFT-D study (NCT04747431) have been undertaken to evaluate the effectiveness of experimental therapies utilizing different AAV vectors (PR006 with the AAV9 vector and PBFT02 with the AAV1 vector, respectively).
Figure 5..

Progranulin deficiency in a wide spectrum of diseases -- from central nervous systems to peripheral systems, named as Progranulinopathy.
Another therapeutic strategy is to find a small compound to enhance endogenous PGRN expression in neurodegenerative diseases. A clinical trial (NCT01835665) was conducted to investigate the potential of nimodipine, an FDA-approved calcium channel blocker, for treating PGRN insufficiency in neurodegenerative diseases. However, no promising results have been released thus far. Furthermore, there is currently no published data supporting the notion that nimodipine induces PGRN expression in cell-based or animal models. Several transcriptional enhancers like suberoylanilide hydroxamic acid (SAHA, Histone deacetylase inhibitor), 5-aza-2’-deoxycytidine (Methyltransferase inhibitor), trehalose (Autophagy regulator), Nor-binaltorphimine dihydrochloride, dibutyryl-cAMP and chloroquine were identified to normalize PGRN-deficient condition. SAHA was further investigated in a phase 2A clinical trial (NCT02149160) and the results of this trial did not demonstrate any clinically significant increase of PGRN levels clinically[179]. The study of AL001, a human Sortilin ectodomain monoclonal antibody that elevate endogenous PGRN by blocking sortilin-mediated degradation, is to evaluate safety and efficacy in FTD-GRN and ALS-C9orf72 (NCT03636204, NCT03987295, NCT04374136 and NCT05053035).
Recent studies have also shed light on possibilities for PGRN’s role in musculoskeletal diseases. These studies have introduced novel concepts that highlight PGRN’s contribution to maintaining bone homeostasis and regulating immunity, particularly within the context of inflammation. PGRN has anti-inflammatory functions through multiple mechanisms, as summarized above. PGRN is a pro-oncogenic protein due to its ability to increase cell proliferation rates, therefore administration of full-length PGRN in treating autoimmune diseases may increase cancer risk. To overcome this problem, a small engineered protein was developed and called Atsttrin, that contains the minimal fragment for binding to TNFR1 and TNFR2, blocks TNF-mediated inflammation, such as in rheumatoid arthritis[82], titanium particle-induced inflammatory osteolysis[180], and dermatitis[181]. Similar to PGRN, Atsttrin protects osteoarthritis mainly through TNFR2[113]. PGRN was also reported to be a co-focal for GD[30]. Recombinant PGRN and its derived Pcgin are therapeutic in treating GD in multiple in vitro and in vivo animal models[30, 31]. The fact that PGRN regulates lysosomal function indicates that lysosomal health may be a common requirement in both disease and aging and points to a unifying underlying mechanism of Progranulinopathy.
In conclusion, PGRN deficiency is causative of NCL and FTD-GRN and is a risk factor for multiple other diseases. The neurotrophic, anti-inflammatory roles of PGRN, along with its roles in lysosomal function, suggest that PGRN elevation may be a promising therapeutic approach to alter the course of Progranulinopathy. Continued research efforts and clinical investigations are necessary to fully elucidate the therapeutic potential of PGRN elevation and to develop targeted interventions that can effectively modulate PGRN levels. By addressing the underlying molecular mechanisms, it is hoped that such interventions will lead to meaningful clinical outcomes and improve the lives of individuals affected by Progranulinopathy and associated conditions.
Highlights.
Progranulin (PGRN), encoded by the GRN gene, functions as a secreted growth factor involved in processes ranging from tumorigenesis and inflammation to neural proliferation.
Mutations in the GRN gene can result in various neuronal degenerative diseases and rare lysosomal storage disorders, emphasizing PGRN’s pivotal role in sustaining lysosomal function.
The range of conditions stemming from PGRN imbalance has expanded to encompass autoimmune diseases, metabolic disorders, musculoskeletal challenges, and cardiovascular issues, collectively termed as Progranulinopathy.
Beyond its recognized role as an extracellular growth factor-like molecule, PGRN also operates as an intracellular co-chaperone, facilitating the folding and trafficking of associated proteins.
This review delves into the complex interplay between PGRN and an assorted array of proteins across different tiers, encompassing extracellular fluids and intracellular components.
Acknowledgments:
We would like to thank Ellen Kim and Aubryanna Hettinghouse for critical reading and editing the manuscript. The Figure was partly generated using Servier Medical Art, provided by Servier, licensed under a Creative Commons Attribution 3.0 unported license (https://smart.servier.com/). This work was supported partly by NIH research grants R01AR062207, R01AR061484, R01AR076900, R01AR078035 and R01NS070328.
Biographies
Guiwu Huang, currently works as a research scientist at Dr. Chuanju Liu’s lab at Yale University School of Medicine. He obtained his Doctor of Medicine (M.D.) degree from Sun Yat-sen University in 2021. From 2022 to 2023, he held the position of a research scientist at the Hospital of Joint Diseases, New York University Grossman School of Medicine. His research interests in musculoskeletal and autoimmune diseases.
Jinlong Jian, currently work as senior research investigator at Gene Therapy Program at University of Pennsylvania from 2018-present. He was a senior research scientist at Dr. Chuanju Liu’s lab at Hospital of Joint Diseases, New York University Grossman School of Medicine from 2011 to 2018. His research interests in gene therapy, lysosomal storage diseases and autoimmune diseases.
Chuan-Ju Liu holds the position of Charles W. Ohse Professor of Orthopaedics at Yale University School of Medicine, with tenure in the Traditional Track, at Yale University School of Medicine. Prior to joining Yale, he worked at New York University Grossman School of Medicine from 2002 to 2023, holding various positions including Assistant Professor, Associate Professor with tenure, and full Professor of Orthopaedic Surgery and Cell Biology. His research primarily focuses on musculoskeletal, autoimmune, and lysosomal storage diseases.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Declaration of Competing Interest
The authors declare no competing interests.
Reference
- [1].Zanocco-Marani T, Bateman A, Romano G, Valentinis B, He ZH, Baserga R, Biological activities and signaling pathways of the granulin/epithelin precursor, Cancer Research 59(20) (1999) 5331–5340. [PubMed] [Google Scholar]
- [2].Hrabal R, Chen Z, James S, Bennett HP, Ni F, The hairpin stack fold, a novel protein architecture for a new family of protein growth factors, Nat Struct Biol 3(9) (1996) 747–752. [DOI] [PubMed] [Google Scholar]
- [3].Cui Y, Hettinghouse A, Liu CJ, Progranulin: A conductor of receptors orchestra, a chaperone of lysosomal enzymes and a therapeutic target for multiple diseases, Cytokine Growth Factor Rev 45 (2019) 53–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].He Z, Bateman A, Progranulin gene expression regulates epithelial cell growth and promotes tumor growth in vivo, Cancer Research 59(13) (1999) 3222–3229. [PubMed] [Google Scholar]
- [5].Baker M, Mackenzie IR, Pickering-Brown SM, Gass J, Rademakers R, Lindholm C, Snowden J, Adamson J, Sadovnick AD, Rollinson S, Cannon A, Dwosh E, Neary D, Melquist S, Richardson A, Dickson D, Berger Z, Eriksen J, Robinson T, Zehr C, Dickey CA, Crook R, McGowan E, Mann D, Boeve B, Feldman H, Hutton M, Mutations in progranulin cause tau-negative frontotemporal dementia linked to chromosome 17, Nature 442(7105) (2006) 916–919. [DOI] [PubMed] [Google Scholar]
- [6].Cruts M, Gijselinck I, van der Zee J, Engelborghs S, Wils H, Pirici D, Rademakers R, Vandenberghe R, Dermaut B, Martin J-J, van Duijn C, Peeters K, Sciot R, Santens P, De Pooter T, Mattheijssens M, Van den Broeck M, Cuijt I, Vennekens K.l., De Deyn PP, Kumar-Singh S, Van Broeckhoven C, Null mutations in progranulin cause ubiquitin-positive frontotemporal dementia linked to chromosome 17q21, Nature 442(7105) (2006) 920–924. [DOI] [PubMed] [Google Scholar]
- [7].Chitramuthu BP, Bennett HPJ, Bateman A, Progranulin: a new avenue towards the understanding and treatment of neurodegenerative disease, Brain 140(12) (2017) 3081–3104. [DOI] [PubMed] [Google Scholar]
- [8].Simon MJ, Logan T, DeVos SL, Di Paolo G, Lysosomal functions of progranulin and implications for treatment of frontotemporal dementia, Trends In Cell Biology 33(4) (2023) 324–339. [DOI] [PubMed] [Google Scholar]
- [9].Jian J, Li G, Hettinghouse A, Liu C, Progranulin: A key player in autoimmune diseases, Cytokine 101 (2018) 48–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Wei J, Hettinghouse A, Liu C, The role of progranulin in arthritis, Ann N Y Acad Sci 1383(1) (2016) 5–20. [DOI] [PubMed] [Google Scholar]
- [11].Konopka J, Richbourgh B, Liu C, The role of PGRN in musculoskeletal development and disease, Front Biosci (Landmark Ed) 19(4) (2014) 662–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Jian J, Konopka J, Liu C, Insights into the role of progranulin in immunity, infection, and inflammation, Journal of Leukocyte Biology 93(2) (2013) 199–208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Liu C.-j., Bosch X, Progranulin: a growth factor, a novel TNFR ligand and a drug target, Pharmacology & Therapeutics 133(1) (2012) 124–132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Liu CJ, Progranulin: a promising therapeutic target for rheumatoid arthritis, FEBS Lett 585(23) (2011) 3675–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Williams A, Wang EC, Thurner L, Liu CJ, Review: Novel Insights Into Tumor Necrosis Factor Receptor, Death Receptor 3, and Progranulin Pathways in Arthritis and Bone Remodeling, Arthritis & rheumatology 68(12) (2016) 2845–2856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Josephs KA, Ahmed Z, Katsuse O, Parisi JF, Boeve BF, Knopman DS, Petersen RC, Davies P, Duara R, Graff-Radford NR, Uitti RJ, Rademakers R, Adamson J, Baker M, Hutton ML, Dickson DW, Neuropathologic features of frontotemporal lobar degeneration with ubiquitin-positive inclusions with progranulin gene (PGRN) mutations, Journal of Neuropathology and Experimental Neurology 66(2) (2007) 142–151. [DOI] [PubMed] [Google Scholar]
- [17].Martens LH, Zhang J, Barmada SJ, Zhou P, Kamiya S, Sun B, Min SW, Gan L, Finkbeiner S, Huang EJ, Jr RVF, Progranulin deficiency promotes neuroinflammation and neuron loss following toxin-induced injury, J Clin Invest 132(1) (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Zhang J, Velmeshev D, Hashimoto K, Huang YH, Hofmann JW, Shi X, Chen J, Leidal AM, Dishart JG, Cahill MK, Kelley KW, Liddelow SA, Seeley WW, Miller BL, Walther TC, Farese RV Jr., Taylor JP, Ullian EM, Huang B, Debnath J, Wittmann T, Kriegstein AR, Huang EJ, Neurotoxic microglia promote TDP-43 proteinopathy in progranulin deficiency, Nature 588(7838) (2020) 459–465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Van Damme P, Van Hoecke A, Lambrechts D, Vanacker P, Bogaert E, van Swieten J, Carmeliet P, Van Den Bosch L, Robberecht W, Progranulin functions as a neurotrophic factor to regulate neurite outgrowth and enhance neuronal survival, The Journal of Cell Biology 181(1) (2008) 37–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].De Muynck L, Herdewyn S, Beel S, Scheveneels W, Van Den Bosch L, Robberecht W, Van Damme P, The neurotrophic properties of progranulin depend on the granulin E domain but do not require sortilin binding, Neurobiol Aging 34(11) (2013) 2541–7. [DOI] [PubMed] [Google Scholar]
- [21].Vardarajan BN, Reyes-Dumeyer D, Piriz AL, Lantigua RA, Medrano M, Rivera D, Jiménez-Velázquez IZ, Martin E, Pericak-Vance MA, Bush W, Farrer L, Haines JL, Wang LS, Leung YY, Schellenberg G, Kukull W, De Jager P, Bennett DA, Schneider JA, Mayeux R, Progranulin mutations in clinical and neuropathological Alzheimer’s disease, Alzheimers Dement (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Gliebus G, Rosso A, Lippa CF, Progranulin and beta-amyloid distribution: a case report of the brain from preclinical PS-1 mutation carrier, Am J Alzheimers Dis Other Demen 24(6) (2009) 456–460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].Pereson S, Wils H, Kleinberger G, McGowan E, Vandewoestyne M, Van Broeck B, Joris G, Cuijt I, Deforce D, Hutton M, Van Broeckhoven C, Kumar-Singh S, Progranulin expression correlates with dense-core amyloid plaque burden in Alzheimer disease mouse models, J Pathol 219(2) (2009) 173–181. [DOI] [PubMed] [Google Scholar]
- [24].Satoh J-I, Kino Y, Yamamoto Y, Kawana N, Ishida T, Saito Y, Arima K, PLD3 is accumulated on neuritic plaques in Alzheimer’s disease brains, Alzheimer’s Research & Therapy 6(9) (2014) 70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Minami SS, Min S-W, Krabbe G, Wang C, Zhou Y, Asgarov R, Li Y, Martens LH, Elia LP, Ward ME, Mucke L, Farese RV, Gan L, Progranulin protects against amyloid β deposition and toxicity in Alzheimer’s disease mouse models, Nat Med 20(10) (2014) 1157–1164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Gowrishankar S, Yuan P, Wu Y, Schrag M, Paradise S, Grutzendler J, De Camilli P, Ferguson SM, Massive accumulation of luminal protease-deficient axonal lysosomes at Alzheimer’s disease amyloid plaques, Proceedings of the National Academy of Sciences of the United States of America 112(28) (2015) E3699–E3708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].Brouwers N, Nuytemans K, van der Zee J, Gijselinck I, Engelborghs S, Theuns J, Kumar-Singh S, Pickut BA, Pals P, Dermaut B, Bogaerts V, De Pooter T, Serneels S, Van den Broeck M, Cuijt I, Mattheijssens M, Peeters K, Sciot R, Martin J-J, Cras P, Santens P, Vandenberghe R, De Deyn PP, Cruts M, Van Broeckhoven C, Sleegers K, Alzheimer and Parkinson diagnoses in progranulin null mutation carriers in an extended founder family, Arch Neurol 64(10) (2007) 1436–1446. [DOI] [PubMed] [Google Scholar]
- [28].Rovelet-Lecrux A, Deramecourt V, Legallic S, Maurage C-A, Le Ber I, Brice A, Lambert J-C, Frébourg T, Hannequin D, Pasquier F, Campion D, Deletion of the progranulin gene in patients with frontotemporal lobar degeneration or Parkinson disease, Neurobiology of Disease 31(1) (2008) 41–45. [DOI] [PubMed] [Google Scholar]
- [29].Neudorfer O, Giladi N, Elstein D, Abrahamov A, Turezkite T, Aghai E, Reches A, Bembi B, Zimran A, Occurrence of Parkinson’s syndrome in type I Gaucher disease, QJM 89(9) (1996) 691–694. [DOI] [PubMed] [Google Scholar]
- [30].Jian J, Tian QY, Hettinghouse A, Zhao S, Liu H, Wei J, Grunig G, Zhang W, Setchell KDR, Sun Y, Overkleeft HS, Chan GL, Liu CJ, Progranulin Recruits HSP70 to β-Glucocerebrosidase and Is Therapeutic Against Gaucher Disease, EBioMedicine 13 (2016) 212–224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Jian J, Zhao S, Tian QY, Liu H, Zhao Y, Chen WC, Grunig G, Torres PA, Wang BC, Zeng B, Pastores G, Tang W, Sun Y, Grabowski GA, Kong MX, Wang G, Chen Y, Liang F, Overkleeft HS, Saunders-Pullman R, Chan GL, Liu CJ, Association Between Progranulin and Gaucher Disease, EBioMedicine 11 (2016) 127–137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [32].Aharon-Peretz J, Rosenbaum H, Gershoni-Baruch R, Mutations in the glucocerebrosidase gene and Parkinson’s disease in Ashkenazi Jews, The New England Journal of Medicine 351(19) (2004) 1972–1977. [DOI] [PubMed] [Google Scholar]
- [33].Lesage S, Anheim M, Condroyer C, Pollak P, Durif F, Dupuits C, Viallet F, Lohmann E, Corvol J-C, Honoré A, Rivaud S, Vidailhet M, Dürr A, Brice A, Large-scale screening of the Gaucher’s disease-related glucocerebrosidase gene in Europeans with Parkinson’s disease, Human Molecular Genetics 20(1) (2011) 202–210. [DOI] [PubMed] [Google Scholar]
- [34].Sleegers K, Brouwers N, Maurer-Stroh S, van Es MA, Van Damme P, van Vught PWJ, van der Zee J, Serneels S, De Pooter T, Van den Broeck M, Cruts M, Schymkowitz J, De Jonghe P, Rousseau F, van den Berg LH, Robberecht W, Van Broeckhoven C, Progranulin genetic variability contributes to amyotrophic lateral sclerosis, Neurology 71(4) (2008) 253–259. [DOI] [PubMed] [Google Scholar]
- [35].Del Bo R, Corti S, Santoro D, Ghione I, Fenoglio C, Ghezzi S, Ranieri M, Galimberti D, Mancuso M, Siciliano G, Briani C, Murri L, Scarpini E, Schymick JC, Traynor BJ, Bresolin N, Comi GP, No major progranulin genetic variability contribution to disease etiopathogenesis in an ALS Italian cohort, Neurobiology of Aging 32(6) (2011) 1157–1158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Irwin D, Lippa CF, Rosso A, Progranulin (PGRN) expression in ALS: an immunohistochemical study, Journal of the Neurological Sciences 276(1–2) (2009). [DOI] [PubMed] [Google Scholar]
- [37].Zhang YJ, Xu YF, Dickey CA, Buratti E, Baralle F, Bailey R, Pickering-Brown S, Dickson D, Petrucelli L, Progranulin mediates caspase-dependent cleavage of TAR DNA binding protein-43, J Neurosci 27(39) (2007) 10530–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Fang L, Huber-Abel F, Teuchert M, Hendrich C, Dorst J, Schattauer D, Zettlmeissel H, Wlaschek M, Scharffetter-Kochanek K, Tumani H, Ludolph AC, Brettschneider J, Linking neuron and skin: matrix metalloproteinases in amyotrophic lateral sclerosis (ALS), Journal of the Neurological Sciences 285(1–2) (2009) 62–66. [DOI] [PubMed] [Google Scholar]
- [39].Osaka M, Ito D, Yagi T, Nihei Y, Suzuki N, Evidence of a link between ubiquilin 2 and optineurin in amyotrophic lateral sclerosis, Human Molecular Genetics 24(6) (2015) 1617–1629. [DOI] [PubMed] [Google Scholar]
- [40].Li LQ, Huang HL, Ping JL, Wang XH, Zhong J, Dai LC, Clinicopathologic and prognostic implications of progranulin in breast carcinoma, Chinese medical journal 124(13) (2011) 2045–50. [PubMed] [Google Scholar]
- [41].Lu R, Serrero G, Inhibition of PC cell-derived growth factor (PCDGF, epithelin/granulin precursor) expression by antisense PCDGF cDNA transfection inhibits tumorigenicity of the human breast carcinoma cell line MDA-MB-468, Proceedings of the National Academy of Sciences of the United States of America 97(8) (2000) 3993–3998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [42].Serrero G, Hawkins DM, Yue B, Ioffe O, Bejarano P, Phillips JT, Head JF, Elliott RL, Tkaczuk KR, Godwin AK, Weaver J, Kim WE, Progranulin (GP88) tumor tissue expression is associated with increased risk of recurrence in breast cancer patients diagnosed with estrogen receptor positive invasive ductal carcinoma, Breast Cancer Res 14(1) (2012) R26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [43].Kim WE, Yue B, Serrero G, Signaling Pathway of GP88 (Progranulin) in Breast Cancer Cells: Upregulation and Phosphorylation of c-myc by GP88/Progranulin in Her2-Overexpressing Breast Cancer Cells, Breast Cancer : Basic and Clinical Research 9(Suppl 2) (2015) 71–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [44].Tangkeangsirisin W, Serrero G, PC cell-derived growth factor (PCDGF/GP88, progranulin) stimulates migration, invasiveness and VEGF expression in breast cancer cells, Carcinogenesis 25(9) (2004) 1587–92. [DOI] [PubMed] [Google Scholar]
- [45].Fang W, Zhou T, Shi H, Yao M, Zhang D, Qian H, Zeng Q, Wang Y, Jin F, Chai C, Chen T, Progranulin induces immune escape in breast cancer via up-regulating PD-L1 expression on tumor-associated macrophages (TAMs) and promoting CD8(+) T cell exclusion, J Exp Clin Cancer Res 40(1) (2021) 4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [46].Huang A, Shinde PV, Huang J, Senff T, Xu HC, Margotta C, Häussinger D, Willnow TE, Zhang J, Pandyra AA, Timm J, Weggen S, Lang KS, Lang PA, Progranulin prevents regulatory NK cell cytotoxicity against antiviral T cells, JCI Insight 4(17) (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [47].Wei Z, Huang Y, Xie N, Ma Q, Elevated expression of secreted autocrine growth factor progranulin increases cervical cancer growth, Cell Biochem Biophys 71(1) (2015) 189–193. [DOI] [PubMed] [Google Scholar]
- [48].Jones MB, Michener CM, Blanchette JO, Kuznetsov VA, Raffeld M, Serrero G, Emmert-Buck MR, Petricoin EF, Krizman DB, Liotta LA, Kohn EC, The granulin-epithelin precursor/PC-cell-derived growth factor is a growth factor for epithelial ovarian cancer, Clinical Cancer Research : an Official Journal of the American Association For Cancer Research 9(1) (2003) 44–51. [PubMed] [Google Scholar]
- [49].Kamrava M, Simpkins F, Alejandro E, Michener C, Meltzer E, Kohn EC, Lysophosphatidic acid and endothelin-induced proliferation of ovarian cancer cell lines is mitigated by neutralization of granulin-epithelin precursor (GEP), a prosurvival factor for ovarian cancer, Oncogene 24(47) (2005) 7084–7093. [DOI] [PubMed] [Google Scholar]
- [50].Simpkins FA, Devoogdt NM, Rasool N, Tchabo NE, Alejandro EU, Kamrava MMRN, Kohn EC, The alarm anti-protease, secretory leukocyte protease inhibitor, is a proliferation and survival factor for ovarian cancer cells, Carcinogenesis 29(3) (2008) 466–472. [DOI] [PubMed] [Google Scholar]
- [51].Donald CD, Laddu A, Chandham P, Lim SD, Cohen C, Amin M, Gerton GL, Marshall FF, Petros JA, Expression of progranulin and the epithelin/granulin precursor acrogranin correlates with neoplastic state in renal epithelium, Anticancer Res 21(6A) (2001) 3739–3742. [PubMed] [Google Scholar]
- [52].Pan C-X, Kinch MS, Kiener PA, Langermann S, Serrero G, Sun L, Corvera J, Sweeney CJ, Li L, Zhang S, Baldridge LA, Jones TD, Koch MO, Ulbright TM, Eble JN, Cheng L, PC cell-derived growth factor expression in prostatic intraepithelial neoplasia and prostatic adenocarcinoma, Clinical Cancer Research : an Official Journal of the American Association For Cancer Research 10(4) (2004) 1333–1337. [DOI] [PubMed] [Google Scholar]
- [53].Abdulrahman A, Eckstein M, Jung R, Guzman J, Weigelt K, Serrero G, Yue B, Geppert C, Stöhr R, Hartmann A, Wullich B, Wach S, Taubert H, Lieb V, Expression of GP88 (Progranulin) Protein Is an Independent Prognostic Factor in Prostate Cancer Patients, Cancers (Basel) 11(12) (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [54].Tanimoto R, Morcavallo A, Terracciano M, Xu S-Q, Stefanello M, Buraschi S, Lu KG, Bagley DH, Gomella LG, Scotlandi K, Belfiore A, Iozzo RV, Morrione A, Sortilin regulates progranulin action in castration-resistant prostate cancer cells, Endocrinology 156(1) (2015) 58–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [55].Monami G, Emiliozzi V, Bitto A, Lovat F, Xu S-Q, Goldoni S, Fassan M, Serrero G, Gomella LG, Baffa R, Iozzo RV, Morrione A, Proepithelin regulates prostate cancer cell biology by promoting cell growth, migration, and anchorage-independent growth, The American Journal of Pathology 174(3) (2009) 1037–1047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [56].Lo HC, Hsu JH, Lai LC, Tsai MH, Chuang EY, MicroRNA-107 enhances radiosensitivity by suppressing granulin in PC-3 prostate cancer cells, Sci Rep 10(1) (2020) 14584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [57].Monami G, Gonzalez EM, Hellman M, Gomella LG, Baffa R, Iozzo RV, Morrione A, Proepithelin promotes migration and invasion of 5637 bladder cancer cells through the activation of ERK1/2 and the formation of a paxillin/FAK/ERK complex, Cancer Research 66(14) (2006) 7103–7110. [DOI] [PubMed] [Google Scholar]
- [58].Xu S-Q, Buraschi S, Morcavallo A, Genua M, Shirao T, Peiper SC, Gomella LG, Birbe R, Belfiore A, Iozzo RV, Morrione A, A novel role for drebrin in regulating progranulin bioactivity in bladder cancer, Oncotarget 6(13) (2015) 10825–10839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [59].Buraschi S, Xu S-Q, Stefanello M, Moskalev I, Morcavallo A, Genua M, Tanimoto R, Birbe R, Peiper SC, Gomella LG, Belfiore A, Black PC, Iozzo RV, Morrione A, Suppression of progranulin expression inhibits bladder cancer growth and sensitizes cancer cells to cisplatin, Oncotarget 7(26) (2016) 39980–39995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [60].Yang D, Wang L-L, Dong T-T, Shen Y-H, Guo X-S, Liu C-Y, Liu J, Zhang P, Li J, Sun Y-P, Progranulin promotes colorectal cancer proliferation and angiogenesis through TNFR2/Akt and ERK signaling pathways, American Journal of Cancer Research 5(10) (2015) 3085–3097. [PMC free article] [PubMed] [Google Scholar]
- [61].Laudisi F, Cherubini F, Di Grazia A, Dinallo V, Di Fusco D, Franzè E, Ortenzi A, Salvatori I, Scaricamazza S, Monteleone I, Sakamoto N, Monteleone G, Stolfi C, Progranulin sustains STAT3 hyperactivation and oncogenic function in colorectal cancer cells, Mol Oncol 13(10) (2019) 2142–2159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [62].Bose D, Zimmerman LJ, Pierobon M, Petricoin E, Tozzi F, Parikh A, Fan F, Dallas N, Xia L, Gaur P, Samuel S, Liebler DC, Ellis LM, Chemoresistant colorectal cancer cells and cancer stem cells mediate growth and survival of bystander cells, Br J Cancer 105(11) (2011) 1759–1767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [63].Cheung ST, Wong SY, Leung KL, Chen X, So S, Ng IO, Fan ST, Granulin-epithelin precursor overexpression promotes growth and invasion of hepatocellular carcinoma, Clinical Cancer Research : an Official Journal of the American Association For Cancer Research 10(22) (2004) 7629–7636. [DOI] [PubMed] [Google Scholar]
- [64].Ho JC, Ip YC, Cheung ST, Lee YT, Chan KF, Wong SY, Fan ST, Granulin-epithelin precursor as a therapeutic target for hepatocellular carcinoma, Hepatology (Baltimore, Md.) 47(5) (2008) 1524–1532. [DOI] [PubMed] [Google Scholar]
- [65].Cheung PFY, Cheung ST, Methods to Analyze the Role of Progranulin (PGRN/GEP) on Cancer Stem Cell Features, Methods Mol Biol 1806 (2018) 145–153. [DOI] [PubMed] [Google Scholar]
- [66].Huang H, Li J, Lu Y, Min L, Li D, Dai L, Role of midkine-progranulin interaction during angiogenesis of hepatocellular carcinoma, Int J Clin Exp Pathol 8(8) (2015) 8809–8820. [PMC free article] [PubMed] [Google Scholar]
- [67].Chen X, Xu H, Wu N, Liu X, Qiao G, Su S, Tian Y, Yuan R, Li C, Liu X, Lin X, Interaction between granulin A and enolase 1 attenuates the migration and invasion of human hepatoma cells, Oncotarget 8(18) (2017) 30305–30316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [68].Frampton G, Invernizzi P, Bernuzzi F, Pae HY, Quinn M, Horvat D, Galindo C, Huang L, McMillin M, Cooper B, Rimassa L, DeMorrow S, Interleukin-6-driven progranulin expression increases cholangiocarcinoma growth by an Akt-dependent mechanism, Gut 61(2) (2012) 268–277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [69].Frampton G, Ueno Y, Quinn M, McMillin M, Pae HY, Galindo C, Leyva-Illades D, DeMorrow S, The novel growth factor, progranulin, stimulates mouse cholangiocyte proliferation via sirtuin-1-mediated inactivation of FOXO1, American Journal of Physiology. Gastrointestinal and Liver Physiology 303(11) (2012) G1202–G1211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [70].The global, regional, and national burden of pancreatic cancer and its attributable risk factors in 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017, Lancet Gastroenterol Hepatol 4(12) (2019) 934–947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [71].Nielsen SR, Quaranta V, Linford A, Emeagi P, Rainer C, Santos A, Ireland L, Sakai T, Sakai K, Kim Y-S, Engle D, Campbell F, Palmer D, Ko JH, Tuveson DA, Hirsch E, Mielgo A, Schmid MC, Macrophage-secreted granulin supports pancreatic cancer metastasis by inducing liver fibrosis, Nat Cell Biol 18(5) (2016) 549–560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [72].Quaranta V, Rainer C, Nielsen SR, Raymant ML, Ahmed MS, Engle DD, Taylor A, Murray T, Campbell F, Palmer DH, Tuveson DA, Mielgo A, Schmid MC, Macrophage-Derived Granulin Drives Resistance to Immune Checkpoint Inhibition in Metastatic Pancreatic Cancer, Cancer Res 78(15) (2018) 4253–4269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [73].Cheung PF, Yang J, Fang R, Borgers A, Krengel K, Stoffel A, Althoff K, Yip CW, Siu EHL, Ng LWC, Lang KS, Cham LB, Engel DR, Soun C, Cima I, Scheffler B, Striefler JK, Sinn M, Bahra M, Pelzer U, Oettle H, Markus P, Smeets EMM, Aarntzen E, Savvatakis K, Liffers ST, Lueong SS, Neander C, Bazarna A, Zhang X, Paschen A, Crawford HC, Chan AWH, Cheung ST, Siveke JT, Progranulin mediates immune evasion of pancreatic ductal adenocarcinoma through regulation of MHCI expression, Nat Commun 13(1) (2022) 156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [74].Yamamoto K, Venida A, Yano J, Biancur DE, Kakiuchi M, Gupta S, Sohn ASW, Mukhopadhyay S, Lin EY, Parker SJ, Banh RS, Paulo JA, Wen KW, Debnath J, Kim GE, Mancias JD, Fearon DT, Perera RM, Kimmelman AC, Autophagy promotes immune evasion of pancreatic cancer by degrading MHC-I, Nature 581(7806) (2020) 100–105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [75].Wang M, Li G, Yin J, Lin T, Zhang J, Progranulin overexpression predicts overall survival in patients with glioblastoma, Medical Oncology (Northwood, London, England) 29(4) (2012) 2423–2431. [DOI] [PubMed] [Google Scholar]
- [76].Liau LM, Lallone RL, Seitz RS, Buznikov A, Gregg JP, Kornblum HI, Nelson SF, Bronstein JM, Identification of a human glioma-associated growth factor gene, granulin, using differential immuno-absorption, Cancer Research 60(5) (2000) 1353–1360. [PubMed] [Google Scholar]
- [77].Bandey I, Chiou SH, Huang AP, Tsai JC, Tu P.h., Progranulin promotes Temozolomide resistance of glioblastoma by orchestrating DNA repair and tumor stemness, Oncogene 34(14) (2015) 1853–1864. [DOI] [PubMed] [Google Scholar]
- [78].Wang W, Hayashi J, Kim WE, Serrero G, PC cell-derived growth factor (granulin precursor) expression and action in human multiple myeloma, Clinical Cancer Research : an Official Journal of the American Association For Cancer Research 9(6) (2003) 2221–2228. [PubMed] [Google Scholar]
- [79].Wang W, Hayashi J, Serrero G, PC cell-derived growth factor confers resistance to dexamethasone and promotes tumorigenesis in human multiple myeloma, Clinical Cancer Research : an Official Journal of the American Association For Cancer Research 12(1) (2006) 49–56. [DOI] [PubMed] [Google Scholar]
- [80].Schulze-Edinghausen L, Dürr C, Öztürk S, Zucknick M, Benner A, Kalter V, Ohl S, Close V, Wuchter P, Stilgenbauer S, Lichter P, Seiffert M, Dissecting the Prognostic Significance and Functional Role of Progranulin in Chronic Lymphocytic Leukemia, Cancers (Basel) 11(6) (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [81].Maas C, Tromp JM, van Laar J, Thijssen R, Elias JA, Malara A, Krippner-Heidenreich A, Silke J, van Oers MH, Eldering E, CLL cells are resistant to smac mimetics because of an inability to form a ripoptosome complex, Cell Death & Disease 4(8) (2013) e782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [82].Tang W, Lu Y, Tian Q-Y, Zhang Y, Guo F-J, Liu G-Y, Syed NM, Lai Y, Lin EA, Kong L, Su J, Yin F, Ding A-H, Zanin-Zhorov A, Dustin ML, Tao J, Craft J, Yin Z, Feng JQ, Abramson SB, Yu X-P, Liu C.-j., The growth factor progranulin binds to TNF receptors and is therapeutic against inflammatory arthritis in mice, Science (New York, N.Y.) 332(6028) (2011) 478–484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [83].Hüttmann A, Klein-Hitpass L, Thomale J, Deenen R, Carpinteiro A, Nückel H, Ebeling P, Führer A, Edelmann J, Sellmann L, Dührsen U, Dürig J, Gene expression signatures separate B-cell chronic lymphocytic leukaemia prognostic subgroups defined by ZAP-70 and CD38 expression status, Leukemia 20(10) (2006) 1774–1782. [DOI] [PubMed] [Google Scholar]
- [84].Göbel M, Eisele L, Möllmann M, Hüttmann A, Johansson P, Scholtysik R, Bergmann M, Busch R, Döhner H, Hallek M, Seiler T, Stilgenbauer S, Klein-Hitpass L, Dührsen U, Dürig J, Progranulin is a novel independent predictor of disease progression and overall survival in chronic lymphocytic leukemia, PloS One 8(8) (2013) e72107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [85].Jiao J, Herl LD, Farese RV, Gao F-B, MicroRNA-29b regulates the expression level of human progranulin, a secreted glycoprotein implicated in frontotemporal dementia, PloS One 5(5) (2010) e10551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [86].Ngankeu A, Ranganathan P, Havelange V, Nicolet D, Volinia S, Powell BL, Kolitz JE, Uy GL, Stone RM, Kornblau SM, Andreeff M, Croce CM, Bloomfield CD, Garzon R, Discovery and functional implications of a miR-29b-1/miR-29a cluster polymorphism in acute myeloid leukemia, Oncotarget 9(4) (2018) 4354–4365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [87].Smith KR, Damiano J, Franceschetti S, Carpenter S, Canafoglia L, Morbin M, Rossi G, Pareyson D, Mole SE, Staropoli JF, Sims KB, Lewis J, Lin WL, Dickson DW, Dahl HH, Bahlo M, Berkovic SF, Strikingly different clinicopathological phenotypes determined by progranulin-mutation dosage, Am J Hum Genet 90(6) (2012) 1102–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [88].Arrant AE, Filiano AJ, Patel AR, Hoffmann MQ, Boyle NR, Kashyap SN, Onyilo VC, Young AH, Roberson ED, Reduction of microglial progranulin does not exacerbate pathology or behavioral deficits in neuronal progranulin-insufficient mice, Neurobiol Dis 124 (2019) 152–162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [89].Craig EA, Hsp70 at the membrane: driving protein translocation, BMC Biol 16(1) (2018) 11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [90].Zhao X, Lin Y, Liou B, Fu W, Jian J, Fannie V, Zhang W, Setchell KDR, Grabowski GA, Sun Y, Liu CJ, PGRN deficiency exacerbates, whereas a brain penetrant PGRN derivative protects, GBA1 mutation-associated pathologies and diseases, Proc Natl Acad Sci U S A 120(1) (2023) e2210442120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [91].Chen Y, Jian J, Hettinghouse A, Zhao X, Setchell KDR, Sun Y, Liu CJ, Progranulin associates with hexosaminidase A and ameliorates GM2 ganglioside accumulation and lysosomal storage in Tay-Sachs disease, J Mol Med (Berl) 96(12) (2018) 1359–1373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [92].Qiu F, Song L, Ding F, Liu H, Shu Q, Yang N, Liu W, Li X, Expression level of the growth factor progranulin is related with development of systemic lupus erythematosus, Diagnostic Pathology 8(1) (2013) 88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [93].Kessenbrock K, Fröhlich L, Sixt M, Lämmermann T, Pfister H, Bateman A, Belaaouaj A, Ring J, Ollert M, Fässler R, Jenne DE, Proteinase 3 and neutrophil elastase enhance inflammation in mice by inactivating antiinflammatory progranulin, J Clin Invest 118(7) (2008) 2438–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [94].Zhu J, Nathan C, Jin W, Sim D, Ashcroft GS, Wahl SM, Lacomis L, Erdjument-Bromage H, Tempst P, Wright CD, Ding A, Conversion of proepithelin to epithelins: roles of SLPI and elastase in host defense and wound repair, Cell 111(6) (2002) 867–78. [DOI] [PubMed] [Google Scholar]
- [95].Vercellino M, Grifoni S, Romagnolo A, Masera S, Mattioda A, Trebini C, Chiavazza C, Caligiana L, Capello E, Mancardi GL, Giobbe D, Mutani R, Giordana MT, Cavalla P, Progranulin expression in brain tissue and cerebrospinal fluid levels in multiple sclerosis, Mult Scler 17(10) (2011) 1194–201. [DOI] [PubMed] [Google Scholar]
- [96].Klemm P, Assmann G, Preuss KD, Fadle N, Regitz E, Martin T, Pfreundschuh M, Thurner L, Progranulin autoantibodies in systemic sclerosis and autoimmune connective tissue disorders: A preliminary study, Immun Inflamm Dis 7(4) (2019) 271–275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [97].Fu W, Hu W, Shi L, Mundra JJ, Xiao G, Dustin ML, Liu C-J, Foxo4- and Stat3-dependent IL-10 production by progranulin in regulatory T cells restrains inflammatory arthritis, FASEB J 31(4) (2017) 1354–1367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [98].Wei F, Zhang Y, Zhao W, Yu X, Liu C-J, Progranulin facilitates conversion and function of regulatory T cells under inflammatory conditions, PloS One 9(11) (2014) e112110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [99].Mundra JJ, Jian J, Bhagat P, Liu C-J, Progranulin inhibits expression and release of chemokines CXCL9 and CXCL10 in a TNFR1 dependent manner, Scientific Reports 6 (2016) 21115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [100].Fu W, Hu W, Yi YS, Hettinghouse A, Sun G, Bi Y, He W, Zhang L, Gao G, Liu J, Toyo-Oka K, Xiao G, Solit DB, Loke P, Liu CJ, TNFR2/14-3-3ε signaling complex instructs macrophage plasticity in inflammation and autoimmunity, J Clin Invest 131(16) (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [101].Yamamoto Y, Takemura M, Serrero G, Hayashi J, Yue B, Tsuboi A, Kubo H, Mitsuhashi T, Mannami K, Sato M, Matsunami H, Matuo Y, Saito K, Increased serum GP88 (Progranulin) concentrations in rheumatoid arthritis, Inflammation 37(5) (2014) 1806–1813. [DOI] [PubMed] [Google Scholar]
- [102].Thurner L, Zaks M, Preuss K-D, Fadle N, Regitz E, Ong MF, Pfreundschuh M, Assmann G, Progranulin antibodies entertain a proinflammatory environment in a subgroup of patients with psoriatic arthritis, Arthritis Research & Therapy 15(6) (2013) R211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [103].Thurner L, Stöger E, Fadle N, Klemm P, Regitz E, Kemele M, Bette B, Held G, Dauer M, Lammert F, Preuss K-D, Zimmer V, Pfreundschuh M, Proinflammatory progranulin antibodies in inflammatory bowel diseases, Digestive Diseases and Sciences 59(8) (2014) 1733–1742. [DOI] [PubMed] [Google Scholar]
- [104].Thurner L, Preuss K-D, Fadle N, Regitz E, Klemm P, Zaks M, Kemele M, Hasenfus A, Csernok E, Gross WL, Pasquali J-L, Martin T, Bohle RM, Pfreundschuh M, Progranulin antibodies in autoimmune diseases, J Autoimmun 42 (2013) 29–38. [DOI] [PubMed] [Google Scholar]
- [105].Thurner L, Fadle N, Regitz E, Kemele M, Klemm P, Zaks M, Stöger E, Bette B, Carbon G, Zimmer V, Assmann G, Murawski N, Kubuschok B, Held G, Preuss K-D, Pfreundschuh M, The molecular basis for development of proinflammatory autoantibodies to progranulin, J Autoimmun 61 (2015) 17–28. [DOI] [PubMed] [Google Scholar]
- [106].Wei F, Zhang Y, Jian J, Mundra JJ, Tian Q, Lin J, Lafaille JJ, Tang W, Zhao W, Yu X, Liu C-J, PGRN protects against colitis progression in mice in an IL-10 and TNFR2 dependent manner, Scientific reports 4 (2014) 7023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [107].Liu C, Li X-X, Gao W, Liu W, Liu D-S, Progranulin-derived Atsttrin directly binds to TNFRSF25 (DR3) and inhibits TNF-like ligand 1A (TL1A) activity, PloS One 9(3) (2014) e92743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [108].Farag AGA, Shoaib MA, Samaka RM, Abdou AG, Mandour MM, Ibrahim RAL, Progranulin and beta-catenin in psoriasis: An immunohistochemical study, J Cosmet Dermatol 18(6) (2019) 2019–2026. [DOI] [PubMed] [Google Scholar]
- [109].Zhou T, Zhang S, Zhou Y, Lai S, Chen Y, Geng Y, Wang J, Curcumin alleviates imiquimod-induced psoriasis in progranulin-knockout mice, Eur J Pharmacol 909 (2021) 174431. [DOI] [PubMed] [Google Scholar]
- [110].Zhao YP, Tian QY, Liu B, Cuellar J, Richbourgh B, Jia TH, Liu CJ, Progranulin knockout accelerates intervertebral disc degeneration in aging mice, Scientific reports 5 (2015) 9102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [111].Lata M, Hettinghouse AS, Liu CJ, Targeting tumor necrosis factor receptors in ankylosing spondylitis, Ann N Y Acad Sci 1442(1) (2019) 5–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [112].Ding Y, Wei J, Hettinghouse A, Li G, Li X, Einhorn TA, Liu C-J, Progranulin promotes bone fracture healing via TNFR pathways in mice with type 2 diabetes mellitus, Annals of the New York Academy of Sciences 1490(1) (2021) 77–89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [113].Zhao YP, Liu B, Tian QY, Wei JL, Richbourgh B, Liu CJ, Progranulin protects against osteoarthritis through interacting with TNF-α and β-Catenin signalling, Ann Rheum Dis 74(12) (2015) 2244–2253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [114].Guo F, Lai Y, Tian Q, Lin EA, Kong L, Liu C, Granulin-epithelin precursor binds directly to ADAMTS-7 and ADAMTS-12 and inhibits their degradation of cartilage oligomeric matrix protein, Arthritis and Rheumatism 62(7) (2010) 2023–2036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [115].Lin EA, Liu CJ, The emerging roles of ADAMTS-7 and ADAMTS-12 matrix metalloproteinases, Open Access Rheumatol 1 (2009) 121–131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [116].Liu C-J, The role of ADAMTS-7 and ADAMTS-12 in the pathogenesis of arthritis, Nature Clinical Practice. Rheumatology 5(1) (2009) 38–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [117].Fu W, Hettinghouse A, Chen Y, Hu W, Ding X, Chen M, Ding Y, Mundra J, Song W, Liu R, Yi YS, Attur M, Samuels J, Strauss E, Leucht P, Schwarzkopf R, Liu CJ, 14-3-3 epsilon is an intracellular component of TNFR2 receptor complex and its activation protects against osteoarthritis, Ann Rheum Dis 80(12) (2021) 1615–1627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [118].Wei J, Wang K, Hettinghouse A, Liu C, Atsttrin Promotes Cartilage Repair Primarily Through TNFR2-Akt Pathway, Front Cell Dev Biol 8 (2020) 577572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [119].Hettinghouse A, Fu W, Liu CJ, Monitoring Atsttrin-Mediated Inhibition of TNFα/NF-κβ Activation Through In Vivo Bioluminescence Imaging, Methods Mol Biol 2248 (2021) 201–210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [120].Fu W, Hettinghouse A, Liu CJ, In Vitro Physical and Functional Interaction Assays to Examine the Binding of Progranulin Derivative Atsttrin to TNFR2 and Its Anti-TNFα Activity, Methods Mol Biol 2248 (2021) 109–119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [121].Wei JL, Fu W, Ding YJ, Hettinghouse A, Lendhey M, Schwarzkopf R, Kennedy OD, Liu CJ, Progranulin derivative Atsttrin protects against early osteoarthritis in mouse and rat models, Arthritis Res Ther 19(1) (2017) 280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [122].Xia Q, Zhu S, Wu Y, Wang J, Cai Y, Chen P, Li J, Heng BC, Ouyang HW, Lu P, Intra-articular transplantation of atsttrin-transduced mesenchymal stem cells ameliorate osteoarthritis development, Stem Cells Transl Med 4(5) (2015) 523–531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [123].Katyal P, Hettinghouse A, Meleties M, Hasan S, Chen C, Cui M, Sun G, Menon R, Lin B, Regatte R, Montclare JK, Liu CJ, Injectable recombinant block polymer gel for sustained delivery of therapeutic protein in post traumatic osteoarthritis, Biomaterials 281 (2022) 121370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [124].Wei J-L, Buza J, Liu C-J, Does progranulin account for the opposite effects of etanercept and infliximab/adalimumab in osteoarthritis?: Comment on Olson et al.: “Therapeutic Opportunities to Prevent Post-Traumatic Arthritis: Lessons From the Natural History of Arthritis After Articular Fracture”, J Orthop Res 34(1) (2016) 12–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [125].Bai X-H, Wang D-W, Kong L, Zhang Y, Luan Y, Kobayashi T, Kronenberg HM, Yu X-P, Liu C-J, ADAMTS-7, a direct target of PTHrP, adversely regulates endochondral bone growth by associating with and inactivating GEP growth factor, Molecular and Cellular Biology 29(15) (2009) 4201–4219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [126].Xu K, Zhang Y, Ilalov K, Carlson CS, Feng JQ, Di Cesare PE, Liu C.-j., Cartilage oligomeric matrix protein associates with granulin-epithelin precursor (GEP) and potentiates GEP-stimulated chondrocyte proliferation, The Journal of Biological Chemistry 282(15) (2007) 11347–11355. [DOI] [PubMed] [Google Scholar]
- [127].Feng JQ, Guo F-J, Jiang B-C, Zhang Y, Frenkel S, Wang D-W, Tang W, Xie Y, Liu C-J, Granulin epithelin precursor: a bone morphogenic protein 2-inducible growth factor that activates Erk1/2 signaling and JunB transcription factor in chondrogenesis, FASEB Journal : Official Publication of the Federation of American Societies For Experimental Biology 24(6) (2010) 1879–1892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [128].Zhao Y.-p., Tian Q.-y., Frenkel S, Liu C.-j., The promotion of bone healing by progranulin, a downstream molecule of BMP-2, through interacting with TNF/TNFR signaling, Biomaterials 34(27) (2013) 6412–6421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [129].Wei J, Zhang L, Ding Y, Liu R, Guo Y, Hettinghouse A, Buza J, De La Croix J, Li X, Einhorn TA, Liu C-J, Progranulin promotes diabetic fracture healing in mice with type 1 diabetes, Annals of the New York Academy of Sciences 1460(1) (2020) 43–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [130].Hossein-Nezhad A, Mirzaei K, Ansar H, Khooshechin G, Ahmadivand Z, Keshavarz SA, Mutual role of PGRN/TNF-α on osteopenia developing in obesity’s inflammation state, Minerva Medica 103(3) (2012) 165–175. [PubMed] [Google Scholar]
- [131].Oh J, Kim JY, Kim HS, Oh JC, Cheon YH, Park J, Yoon KH, Lee MS, Youn BS, Progranulin and a five transmembrane domain-containing receptor-like gene are the key components in receptor activator of nuclear factor κB (RANK)-dependent formation of multinucleated osteoclasts, J Biol Chem 290(4) (2015) 2042–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [132].Wang N, Zhang J, Yang JX, Growth factor progranulin blocks tumor necrosis factor-α-mediated inhibition of osteoblast differentiation, Genet Mol Res 15(3) (2016). [DOI] [PubMed] [Google Scholar]
- [133].Noguchi T, Ebina K, Hirao M, Kawase R, Ohama T, Yamashita S, Morimoto T, Koizumi K, Kitaguchi K, Matsuoka H, Kaneshiro S, Yoshikawa H, Progranulin plays crucial roles in preserving bone mass by inhibiting TNF-α-induced osteoclastogenesis and promoting osteoblastic differentiation in mice, Biochem Biophys Res Commun 465(3) (2015) 638–43. [DOI] [PubMed] [Google Scholar]
- [134].Romanello M, Piatkowska E, Antoniali G, Cesaratto L, Vascotto C, Iozzo RV, Delneri D, Brancia FL, Osteoblastic cell secretome: a novel role for progranulin during risedronate treatment, Bone 58 (2014) 81–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [135].Yang Y, Feng N, Liang L, Jiang R, Pan Y, Geng N, Fan M, Li X, Guo F, Progranulin, a moderator of estrogen/estrogen receptor α binding, regulates bone homeostasis through PERK/p-eIF2 signaling pathway, J Mol Med (Berl) 100(8) (2022) 1191–1207. [DOI] [PubMed] [Google Scholar]
- [136].Kojima Y, Ono K, Inoue K, Takagi Y, Kikuta K.-i., Nishimura M, Yoshida Y, Nakashima Y, Matsumae H, Furukawa Y, Mikuni N, Nobuyoshi M, Kimura T, Kita T, Tanaka M, Progranulin expression in advanced human atherosclerotic plaque, Atherosclerosis 206(1) (2009) 102–108. [DOI] [PubMed] [Google Scholar]
- [137].Kawase R, Ohama T, Matsuyama A, Matsuwaki T, Okada T, Yamashita T, Yuasa-Kawase M, Nakaoka H, Nakatani K, Inagaki M, Tsubakio-Yamamoto K, Masuda D, Nakagawa-Toyama Y, Nishida M, Ohmoto Y, Nishihara M, Komuro I, Yamashita S, Deletion of progranulin exacerbates atherosclerosis in ApoE knockout mice, Cardiovasc Res 100(1) (2013) 125–133. [DOI] [PubMed] [Google Scholar]
- [138].Fu Y, Sun Y, Zhou M, Wang X, Wang Z, Wei X, Zhang Y, Su Z, Liang K, Tang W, Yi F, Therapeutic Potential of Progranulin in Hyperhomocysteinemia-Induced Cardiorenal Dysfunction, Hypertension 69(2) (2017) 259–266. [DOI] [PubMed] [Google Scholar]
- [139].Xie S, Lu L, Liu L, Bi G, Zheng L, Progranulin and short-term outcome in patients with acute ischaemic stroke, European Journal of Neurology 23(3) (2016) 648–655. [DOI] [PubMed] [Google Scholar]
- [140].Zhou M, Tang W, Fu Y, Xu X, Wang Z, Lu Y, Liu F, Yang X, Wei X, Zhang Y, Liu J, Geng X, Zhang C, Wan Q, Li N, Yi F, Progranulin protects against renal ischemia/reperfusion injury in mice, Kidney Int 87(5) (2015) 918–929. [DOI] [PubMed] [Google Scholar]
- [141].Jackman K, Kahles T, Lane D, Garcia-Bonilla L, Abe T, Capone C, Hochrainer K, Voss H, Zhou P, Ding A, Anrather J, Iadecola C, Progranulin deficiency promotes post-ischemic blood-brain barrier disruption, The Journal of Neuroscience : the Official Journal of the Society For Neuroscience 33(50) (2013) 19579–19589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [142].Egashira Y, Suzuki Y, Azuma Y, Takagi T, Mishiro K, Sugitani S, Tsuruma K, Shimazawa M, Yoshimura S, Kashimata M, Iwama T, Hara H, The growth factor progranulin attenuates neuronal injury induced by cerebral ischemia-reperfusion through the suppression of neutrophil recruitment, J Neuroinflammation 10 (2013) 105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [143].Liu Y, Ren J, Kang M, Zhai C, Cheng Q, Li J, Wu Y, Ruan X, Zhou J, Fan J, Tian Y, Progranulin promotes functional recovery and neurogenesis in the subventricular zone of adult mice after cerebral ischemia, Brain Res 1757 (2021) 147312. [DOI] [PubMed] [Google Scholar]
- [144].Jackman K, Kahles T, Lane D, Garcia-Bonilla L, Abe T, Capone C, Hochrainer K, Voss H, Zhou P, Ding A, Anrather J, Iadecola C, Progranulin deficiency promotes post-ischemic blood-brain barrier disruption, J Neurosci 33(50) (2013) 19579–89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [145].Youn BS, Bang SI, Kloting N, Park JW, Lee N, Oh JE, Pi KB, Lee TH, Ruschke K, Fasshauer M, Stumvoll M, Bluher M, Serum progranulin concentrations may be associated with macrophage infiltration into omental adipose tissue, Diabetes 58(3) (2009) 627–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [146].Liu J, Li H, Zhou B, Xu L, Kang X, Yang W, Wu S, Sun H, PGRN induces impaired insulin sensitivity and defective autophagy in hepatic insulin resistance, Mol Endocrinol 29(4) (2015) 528–541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [147].Xu L, Zhou B, Li H, Liu J, Du J, Zang W, Wu S, Sun H, Serum Levels of Progranulin Are Closely Associated with Microvascular Complication in Type 2 Diabetes, Disease Markers 2015 (2015) 357279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [148].Gonzalez EM, Mongiat M, Slater SJ, Baffa R, Iozzo RV, A novel interaction between perlecan protein core and progranulin: potential effects on tumor growth, The Journal of Biological Chemistry 278(40) (2003) 38113–38116. [DOI] [PubMed] [Google Scholar]
- [149].Kong L, Zhao Y-P, Tian Q-Y, Feng J-Q, Kobayashi T, Merregaert J, Liu C-J, Extracellular matrix protein 1, a direct targeting molecule of parathyroid hormone-related peptide, negatively regulates chondrogenesis and endochondral ossification via associating with progranulin growth factor, FASEB J 30(8) (2016) 2741–2754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [150].Yip CW, Cheung PFY, Leung ICY, Wong NCL, Cheng CKC, Fan ST, Cheung ST, Granulin-epithelin precursor interacts with heparan sulfate on liver cancer cells, Carcinogenesis 35(11) (2014) 2485–2494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [151].Zhu J, Nathan C, Jin W, Sim D, Ashcroft GS, Wahl SM, Lacomis L, Erdjument-Bromage H, Tempst P, Wright CD, Ding A, Conversion of proepithelin to epithelins: roles of SLPI and elastase in host defense and wound repair, Cell 111(6) (2002) 867–878. [DOI] [PubMed] [Google Scholar]
- [152].Jian J, Zhao S, Tian Q, Gonzalez-Gugel E, Mundra JJ, Uddin SMZ, Liu B, Richbourgh B, Brunetti R, Liu C.-j., Progranulin directly binds to the CRD2 and CRD3 of TNFR extracellular domains, FEBS Letters 587(21) (2013) 3428–3436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [153].Tian Q, Zhao Y, Mundra JJ, Gonzalez-Gugel E, Jian J, Uddin SMZ, Liu C, Three TNFR-binding domains of PGRN act independently in inhibition of TNF-alpha binding and activity, Front Biosci (Landmark Ed) 19(7) (2014) 1176–1185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [154].Chen X, Chang J, Deng Q, Xu J, Nguyen TA, Martens LH, Cenik B, Taylor G, Hudson KF, Chung J, Yu K, Yu P, Herz J, Farese RV Jr., Kukar T, Tansey MG, Progranulin does not bind tumor necrosis factor (TNF) receptors and is not a direct regulator of TNF-dependent signaling or bioactivity in immune or neuronal cells, J Neurosci 33(21) (2013) 9202–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [155].Etemadi N, Webb A, Bankovacki A, Silke J, Nachbur U, Progranulin does not inhibit TNF and lymphotoxin-α signalling through TNF receptor 1, Immunol Cell Biol 91(10) (2013) 661–4. [DOI] [PubMed] [Google Scholar]
- [156].Lang I, Füllsack S, Wajant H, Lack of Evidence for a Direct Interaction of Progranulin and Tumor Necrosis Factor Receptor-1 and Tumor Necrosis Factor Receptor-2 From Cellular Binding Studies, Front Immunol 9 (2018) 793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [157].Krabbe G, Minami SS, Etchegaray JI, Taneja P, Djukic B, Davalos D, Le D, Lo I, Zhan L, Reichert MC, Sayed F, Merlini M, Ward ME, Perry DC, Lee SE, Sias A, Parkhurst CN, Gan W-B, Akassoglou K, Miller BL, Farese RV, Gan L, Microglial NFκB-TNFα hyperactivation induces obsessive-compulsive behavior in mouse models of progranulin-deficient frontotemporal dementia, Proceedings of the National Academy of Sciences of the United States of America 114(19) (2017) 5029–5034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [158].Carrasquillo MM, Nicholson AM, Finch N, Gibbs JR, Baker M, Rutherford NJ, Hunter TA, DeJesus-Hernandez M, Bisceglio GD, Mackenzie IR, Singleton A, Cookson MR, Crook JE, Dillman A, Hernandez D, Petersen RC, Graff-Radford NR, Younkin SG, Rademakers R, Genome-wide screen identifies rs646776 near sortilin as a regulator of progranulin levels in human plasma, Am J Hum Genet 87(6) (2010) 890–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [159].Hu F, Padukkavidana T, Vægter CB, Brady OA, Zheng Y, Mackenzie IR, Feldman HH, Nykjaer A, Strittmatter SM, Sortilin-mediated endocytosis determines levels of the frontotemporal dementia protein, progranulin, Neuron 68(4) (2010) 654–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [160].Gass J, Lee WC, Cook C, Finch N, Stetler C, Jansen-West K, Lewis J, Link CD, Rademakers R, Nykjær A, Petrucelli L, Progranulin regulates neuronal outgrowth independent of sortilin, Molecular Neurodegeneration 7 (2012) 33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [161].Altmann C, Vasic V, Hardt S, Heidler J, Häussler A, Wittig I, Schmidt MHH, Tegeder I, Progranulin promotes peripheral nerve regeneration and reinnervation: role of notch signaling, Molecular Neurodegeneration 11(1) (2016) 69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [162].Chitramuthu BP, Campos-García VR, Bateman A, Multiple Molecular Pathways Are Influenced by Progranulin in a Neuronal Cell Model-A Parallel Omics Approach, Front Neurosci 15 (2021) 775391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [163].Egea J, Klein R, Bidirectional Eph-ephrin signaling during axon guidance, Trends Cell Biol 17(5) (2007) 230–238. [DOI] [PubMed] [Google Scholar]
- [164].Rohani N, Canty L, Luu O, Fagotto F, Winklbauer R, EphrinB/EphB signaling controls embryonic germ layer separation by contact-induced cell detachment, PLoS Biol 9(3) (2011) e1000597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [165].Davy A, Soriano P, Ephrin signaling in vivo: look both ways, Dev Dyn 232(1) (2005). [DOI] [PubMed] [Google Scholar]
- [166].Neill T, Buraschi S, Goyal A, Sharpe C, Natkanski E, Schaefer L, Morrione A, Iozzo RV, EphA2 is a functional receptor for the growth factor progranulin, The Journal of Cell Biology 215(5) (2016) 687–703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [167].He Y, Zhang X, Flais I, Svenningsson P, Decreased Prosaposin and Progranulin in the Cingulate Cortex Are Associated with Schizophrenia Pathophysiology, Int J Mol Sci 23(19) (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [168].Pottier C, Zhou X, Perkerson RB 3rd, Baker M, Jenkins GD, Serie DJ, Ghidoni R, Benussi L, Binetti G, López de Munain A, Zulaica M, Moreno F, Le Ber I, Pasquier F, Hannequin D, Sánchez-Valle R, Antonell A, Lladó A, Parsons TM, Finch NA, Finger EC, Lippa CF, Huey ED, Neumann M, Heutink P, Synofzik M, Wilke C, Rissman RA, Slawek J, Sitek E, Johannsen P, Nielsen JE, Ren Y, van Blitterswijk M, DeJesus-Hernandez M, Christopher E, Murray ME, Bieniek KF, Evers BM, Ferrari C, Rollinson S, Richardson A, Scarpini E, Fumagalli GG, Padovani A, Hardy J, Momeni P, Ferrari R, Frangipane F, Maletta R, Anfossi M, Gallo M, Petrucelli L, Suh E, Lopez OL, Wong TH, van Rooij JGJ, Seelaar H, Mead S, Caselli RJ, Reiman EM, Noel Sabbagh M, Kjolby M, Nykjaer A, Karydas AM, Boxer AL, Grinberg LT, Grafman J, Spina S, Oblak A, Mesulam MM, Weintraub S, Geula C, Hodges JR, Piguet O, Brooks WS, Irwin DJ, Trojanowski JQ, Lee EB, Josephs KA, Parisi JE, Ertekin-Taner N, Knopman DS, Nacmias B, Piaceri I, Bagnoli S, Sorbi S, Gearing M, Glass J, Beach TG, Black SE, Masellis M, Rogaeva E, Vonsattel JP, Honig LS, Kofler J, Bruni AC, Snowden J, Mann D, Pickering-Brown S, Diehl-Schmid J, Winkelmann J, Galimberti D, Graff C, Öijerstedt L, Troakes C, Al-Sarraj S, Cruchaga C, Cairns NJ, Rohrer JD, Halliday GM, Kwok JB, van Swieten JC, White CL 3rd, Ghetti B, Murell JR, Mackenzie IRA, Hsiung GR, Borroni B, Rossi G, Tagliavini F, Wszolek ZK, Petersen RC, Bigio EH, Grossman M, Van Deerlin VM, Seeley WW, Miller BL, Graff-Radford NR, Boeve BF, Dickson DW, Biernacka JM, Rademakers R, Potential genetic modifiers of disease risk and age at onset in patients with frontotemporal lobar degeneration and GRN mutations: a genome-wide association study, Lancet Neurol 17(6) (2018) 548–558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [169].Chen-Plotkin AS, Unger TL, Gallagher MD, Bill E, Kwong LK, Volpicelli-Daley L, Busch JI, Akle S, Grossman M, Van Deerlin V, Trojanowski JQ, Lee VMY, TMEM106B, the risk gene for frontotemporal dementia, is regulated by the microRNA-132/212 cluster and affects progranulin pathways, The Journal of Neuroscience : the Official Journal of the Society For Neuroscience 32(33) (2012) 11213–11227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [170].Zhao X, Liberti R, Jian J, Fu W, Hettinghouse A, Sun Y, Liu CJ, Progranulin associates with Rab2 and is involved in autophagosome-lysosome fusion in Gaucher disease, J Mol Med (Berl) 99(11) (2021) 1639–1654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [171].Almeida S, Zhou L, Gao F-B, Progranulin, a glycoprotein deficient in frontotemporal dementia, is a novel substrate of several protein disulfide isomerase family proteins, PloS One 6(10) (2011) e26454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [172].Liu Y, Zhao X, Jian J, Hasan S, Liu C, Interaction with ERp57 is required for progranulin protection against Type 2 Gaucher disease, Biosci Trends 17(2) (2023) 126–135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [173].Sui D, Wilson JE, Interaction of insulin-like growth factor binding protein-4, Miz-1, leptin, lipocalin-type prostaglandin D synthase, and granulin precursor with the N-terminal half of type III hexokinase, Arch Biochem Biophys 382(2) (2000) 262–274. [DOI] [PubMed] [Google Scholar]
- [174].Cheung PF, Cheung TT, Yip CW, Ng LW, Fung SW, Lo CM, Fan ST, Cheung ST, Hepatic cancer stem cell marker granulin-epithelin precursor and β-catenin expression associate with recurrence in hepatocellular carcinoma, Oncotarget 7(16) (2016) 21644–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [175].Lam CY, Yip CW, Poon TCW, Cheng CKC, Ng EWY, Wong NCL, Cheung PFY, Lai PBS, Ng IOL, Fan ST, Cheung ST, Identification and characterization of tropomyosin 3 associated with granulin-epithelin precursor in human hepatocellular carcinoma, PloS One 7(7) (2012) e40324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [176].Hoque M, Young TM, Lee C-G, Serrero G, Mathews MB, Pe’ery T, The growth factor granulin interacts with cyclin T1 and modulates P-TEFb-dependent transcription, Mol Cell Biol 23(5) (2003) 1688–1702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [177].Logan T, Simon MJ, Rana A, Cherf GM, Srivastava A, Davis SS, Low RLY, Chiu CL, Fang M, Huang F, Bhalla A, Llapashtica C, Prorok R, Pizzo ME, Calvert MEK, Sun EW, Hsiao-Nakamoto J, Rajendra Y, Lexa KW, Srivastava DB, van Lengerich B, Wang J, Robles-Colmenares Y, Kim DJ, Duque J, Lenser M, Earr TK, Nguyen H, Chau R, Tsogtbaatar B, Ravi R, Skuja LL, Solanoy H, Rosen HJ, Boeve BF, Boxer AL, Heuer HW, Dennis MS, Kariolis MS, Monroe KM, Przybyla L, Sanchez PE, Meisner R, Diaz D, Henne KR, Watts RJ, Henry AG, Gunasekaran K, Astarita G, Suh JH, Lewcock JW, DeVos SL, Di Paolo G, Rescue of a lysosomal storage disorder caused by Grn loss of function with a brain penetrant progranulin biologic, Cell 184(18) (2021) 4651–4668.e25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [178].Arrant AE, Filiano AJ, Unger DE, Young AH, Roberson ED, Restoring neuronal progranulin reverses deficits in a mouse model of frontotemporal dementia, Brain : a Journal of Neurology 140(5) (2017) 1447–1465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [179].Ljubenkov PA, Edwards L, Iaccarino L, La Joie R, Rojas JC, Koestler M, Harris B, Boeve BF, Borroni B, van Swieten JC, Grossman M, Pasquier F, Frisoni GB, Mummery CJ, Vandenberghe R, Le Ber I, Hannequin D, McGinnis SM, Auriacombe S, Onofrj M, Goodman IJ, Riordan HJ, Wisniewski G, Hesterman J, Marek K, Haynes BA, Patzke H, Koenig G, Hilt D, Moebius H, Boxer AL, Effect of the Histone Deacetylase Inhibitor FRM-0334 on Progranulin Levels in Patients With Progranulin Gene Haploinsufficiency: A Randomized Clinical Trial, JAMA Netw Open 4(9) (2021) e2125584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [180].Zhao Y.-p., Wei J.-l., Tian Q.-y., Liu AT, Yi Y.-s., Einhorn TA, Liu C.-j., Progranulin suppresses titanium particle induced inflammatory osteolysis by targeting TNFα signaling, Scientific Reports 6 (2016) 20909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [181].Zhao Y-P, Tian Q-Y, Liu C-J, Progranulin deficiency exaggerates, whereas progranulin-derived Atsttrin attenuates, severity of dermatitis in mice, FEBS Letters 587(12) (2013) 1805–1810. [DOI] [PMC free article] [PubMed] [Google Scholar]


