Abstract
Balo's concentric sclerosis (BCS) is a rare subtype of multiple sclerosis. Advanced MRI metrics, such as magnetization transfer ratio (MTR), fractional anisotropy (FA), mean diffusivity (MD), and the ratio of total N-acetylaspartate concentration/total creatine concentration (tNAA/tCr) using proton magnetic resonance spectroscopy (1H-MRS), are commonly used in research studies to investigate the effect of a disease modifying therapy (DMT). We report a patient diagnosed with BCS, receiving ocrelizumab, and provide a comparison of the lesion volume, T1-gadolinium lesion volume, MTR, FA, MD, and MRS metrics at baseline, 6- and 12-month follow-up. There was a reduction in Balo's lesion volume on fluid-attenuated inversion recovery (FLAIR) imaging observed in our patient from baseline (23.925 mL) to 12-month follow-up (2.391 mL), with the largest decrease from baseline to 6-month follow-up (3.650 mL). There was no T1-gadolinium enhancement seen at month 6 and 12. The MTR of the lesion did not change significantly (baseline = 50.9%, 6-month = 49.9%, 12-month =50.1%) but the FA increased from 0.188 (at baseline) to 0.304 (at 6 months), while the 12-month follow-up FA was 0.297. We also noted a reduction in MD from baseline (1.333 × 10−3 mm2/s) to 6-month follow-up (1.037 × 10−3 mm2/s), while the 12-month follow-up MD was 1.086 × 10−3 mm2/s. There was a 10.3% increase in tNAA/tCr from 1.583 (at month 0) to 1.747 (at month 12). Our results demonstrate for the first time a direct effect of ocrelizumab on BCS lesions. To validate our findings, more observations are needed in a larger group of BCS patients.
Keywords: Balo's concentric sclerosis, Tumefactive lesion, Multiple sclerosis, Ocrelizumab
Introduction
A tumefactive lesion (TL) is a solitary demyelinating tumor-like lesion greater than 2 cm [1]. TLs are common in multiple sclerosis (MS) and can often be challenging to distinguish from brain tumors on neuroimaging because of the similar clinical manifestations [2]. Balo's concentric sclerosis (BCS), also known as Balo's disease, is a TL of the central nervous system (CNS) and a rare subtype of MS in which there are large regions of alternating zones of demyelinated and myelinated white matter [3]. The age of diagnosis is between 20 and 50 years, and patients generally present with headaches, aphasia, cognitive, or behavioral dysfunction, and seizures [4]. Diagnosis is based on clinical presentation, exclusion of other neurologic diseases, and the characteristic concentric rings on neuroimaging [5]. Clinical differential diagnosis includes acute disseminated encephalomyelitis, MS, neoplasms, and abscesses [6].
BCS shares a basic pathologic similarity to MS, except for a lamellar pattern [7]. The remarkable concentric pattern of demyelination, otherwise commonly referred to as the ‘onion peel’ appearance, has produced much speculation about its pathogenesis [8]. The exact reason for BCS lesion formation is not clear. It is hypothesized that the lesions begin at the central core and the primary demyelinating activity spreads from this focus, with a centrifugally spreading band of lymphocytes originating from this initial site [9].
Initially believed to have a fatal outcome, recent studies have reported nonfatal forms of BCS, emphasizing the role of early diagnosis using various magnetic resonance imaging (MRI) metrics. Among the several advanced imaging modalities used in the diagnosis of MS and hence BCS, magnetization transfer imaging (MTI), characterized by magnetization transfer ratio (MTR), diffusion tensor imaging (DTI), among which fractional anisotropy (FA) and mean diffusivity (MD) are the most used parameters, and the ratio of total N-acetylaspartate concentration/total creatine concentration (tNAA/tCr) using proton magnetic resonance spectroscopy (1H-MRS) provide a more distinct multidimensional approach to quantifying these pathological processes in the CNS.
Advances in MRI have led to the precise diagnosis of BCS. Concentric rings on T2-weighted and contrast-enhanced T1-weighted images are seen on MRI in patients with BCS [10]. T2-weighted MRI scans may reveal a unique pattern of hypo-/isointense and hyperintense rings corresponding to bands of preserved and destroyed myelin [11]. High signal intensity is often seen at the edge of the Balo's lesion on diffusion-weighted imaging. Gadolinium enhancement is commonly seen peripherally, however, sometimes multiple layers may show enhancement, corresponding to layers of T2 hyperintensity [12]. These Balo lesions may be either multiple or singular at disease onset.[13].
Ocrelizumab (Ocrevus) is an anti-CD-20 monoclonal antibody that depletes circulating immature and mature B cells but spares the CD-20 negative plasma cells [14]. Although there are reports of advanced imaging metrics in BCS, there are no reports of the effect of ocrelizumab on MTR and DTI parameters (FA and MD) on Balo's lesions. Here we report a patient diagnosed with BCS, receiving ocrelizumab, and compare the tumor volumes, T1-gadolinium tumor volume, MTR, FA, and MD at baseline, 6- and 12-month follow-up.
Case presentation
A 40-year-old woman presented with left eye visual loss. On neurological examination, she was found to have a left afferent pupillary defect, pain with extraocular movements, and only light perception in her left eye. Four years ago, she experienced slurred speech that persisted for 2 weeks, treated with IV steroids, and recovered. However, no official diagnosis was made and the patient was lost to follow-up. During her current presentation, she received a 5-day course of solumedrol 1000 mg twice daily. A lumbar puncture was performed and showed cerebrospinal fluid (CSF) oligoclonal bands of 12 and serum oligoclonal bands of 2 as well as CSF IgG index level of 0.97. An MRI of her brain showing 3 T2 FLAIR hyperintense, nonenhancing, ovoid lesions, in the supratentorial white matter, the largest on the right frontoparietal lobe measuring 13 mm in length, one in the left frontoparietal corona radiata and one in the selenium of the left corpus collosum. Additional laboratory data including antinuclear antibody (ANA), extractable nuclear antigen antibody (ENA), anti-double stranded deoxyribonucleic acid antibody (dsDNA), neuromyelitis optica (NMO) antibody, serum protein electrophoresis, syphilis enzyme immunoassay (EIA), and Lyme antibody were negative. The patient was diagnosed with relapsing-remitting MS and was started on Glatiramer acetate injections.
Two months later, the patient's vision showed slight improvement from light perception to 20/800. Two years later, her MRI was unchanged and she was switched to dimethylfumarate as she was having injection fatigue. However, during the COVID pandemic, the patient stated that she no longer had insurance and was off disease-modifying therapy at that time.
Three years after her diagnosis, she developed new onset right-hand weakness, slurred speech, urinary frequency, and urgency, and was seen at urgent care where she was given a 5-day course of oral steroids, with no improvement. The patient then resumed care in the clinic and was treated with a 5-day course of intravenous solumedrol with significant improvement in right-hand weakness and dysarthria. A 3T MRI demonstrated a T2 FLAIR enhancing 35.5 x 38.2 mm tumefactive left frontoparietal lesion and redemonstration of 13 mm right frontoparietal lesion. Her baseline MTR was 50.8%, MD was 1.33 × 10-3 mm2/s, and FA was 0.18. A decision to start her on ocrelizumab was made.
The patient received the first split dose and a 6-month maintenance therapy followed by a repeat MRI showing a significant reduction in the size of the right lesion to 12.6 × 7.5 mm and left lesion to 5.7 × 9.1 mm. A repeat MRI was obtained in a year, before the administration of her 12-month infusion; it showed a slight enlargement of the right lesion to 12.1 × 10.2 mm and left lesion to 14 × 9.2 mm. There was a favorable effect of ocrelizumab on the lesion MTR that increased from 49.9% (at 6 months) to 50.1% (at 12 months), as well as FA that increased from 0.18 (at baseline) to 0.30 (at 6 months). There was a 10.3% increase in tNAA/tCr from 1.583 (at month 0) to 1.747 (at month 12). At the 12-month examination, the patient demonstrated a chronic finding of 20/800 vision in her left eye but no other residual neurological symptoms. She reported improved fatigue, no new or worsening symptoms, no relapses, and no infusion side effects.
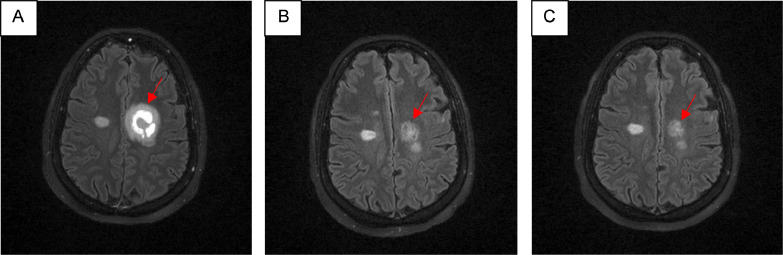
The MRI images and metrics for the patient visits at months 0, 6, and 12 are summarized in Fig. 1 and Table 1 respectively.
Fig. 1.
Axial view of T2 FLAIR sequence with presence of tumefactive lesion (red arrow) at month 0 (Panel A), month 6 (Panel B), month 12 (Panel C).
Table 1.
Tumefactive lesion volume and its associated metrics in the patient
| Patient visit | Lesion Volume on FLAIR (mL) | Lesion Volume on T1-gadolinium (mL) | FA in lesion | MD in lesion (x10−3 mm2/s) | MTR (%) in lesion | Mean NAA + NAAG/Cr +PCr |
|---|---|---|---|---|---|---|
| Month 0 | 23.925 | 8.233 | 0.188 | 1.333 | 50.885 | 1.583 |
| Month 6 | 3.650 | 0.000 | 0.304 | 1.037 | 49.938 | 1.706 |
| Month 12 | 2.391 | 0.000 | 0.297 | 1.086 | 50.130 | 1.747 |
Cr, creatine, FA, fractional anisotropy, FLAIR, fluid-attenuated inversion recovery, MD, mean diffusivity, MTR, magnetization transfer ratio, NAA, N-acetylaspartate, NAAG, N-acetylaspartylglutamate, PCr, phosphocreatine.
Discussion
The findings of this case study align with current literature on the efficacy of ocrelizumab in the treatment of MS. Ocrelizumab, a humanized anti-CD20 monoclonal antibody, has demonstrated significant efficacy in reducing relapse rates, disability progression, and lesion activity in patients with relapsing forms of MS and primary progressive MS.
The pivotal phase III trials, OPERA I and II, demonstrated a substantial reduction in annualized relapse rates and disability progression in patients with relapsing-remitting MS treated with ocrelizumab compared to interferon beta-1a [15,16]. Additionally, the phase III trial ORATORIO showed a significant reduction in disability progression in primary progressive MS patients receiving ocrelizumab compared to placebo [17].
Our patient's clinical stability aligns with the broader literature on the favorable safety and tolerability profile of ocrelizumab.
Lindquist et al reported a marked reduction of the MTR at the lesion core in acute Balo's lesions and surrounding rings followed by improvement again over several months [18]. However, it might be challenging to determine whether the difference in MTR was solely due to the effect of ocrelizumab or to the contribution of the pathological evolution of the lesion overtime. We also noted a notable increase of FA and a reduction in MD from baseline to 6-month follow-up, and a relative stability in M12, a finding not previously described.
The increase in tNAA/tCr observed in our patient is indicative of potential neuroprotective effects associated with ocrelizumab treatment. While the literature on the specific impact of ocrelizumab on spectroscopy outcomes is limited, studies assessing other disease-modifying therapies, such as fingolimod, have reported similar positive effects on neuroaxonal integrity using proton magnetic resonance spectroscopy [19].
The effect of ocrelizumab in reducing the number and volume of gadolinium-enhancing lesions was remarkably fast in a phase 2 study [20]. This result is similar to our case report in which T1-gadolinium lesion volume reduced from 8.23 mL (at baseline) to 0 mL in the subsequent follow-up visits. In accordance with a previously described case of BCS of increased choline from cellular membrane turnover and decreased NAA from neuronal injury in the MRS, our patient had a low baseline NAA+NAAG/Cr +PCr that has gradually and consistently improved at M6 and M12 [21].
Furthermore, there was a reduction in Balo's lesion volume on FLAIR imaging from baseline to 12-month follow-up, with the largest decrease from baseline to 6-month follow-up. Similarly, a previous case report reporting an aggressive case of tumefactive MS, noted an improvement in the patient's condition and a reduction in the size of the pre-existing tumefactive lesions on MRI after receiving ocrelizumab treatment over one year [22].
The slight lesion enlargement observed at the 12-month mark in this case is noteworthy and warrants consideration in the context of existing literature. One possibility is that at the time of M12 imaging the patient had not received her third dose of Ocrevus, which was received a week after her MRI scans took place. Moreover, long-term data from clinical trials and real-world studies have shown that disease activity can vary over time, and factors influencing treatment response and lesion evolution are complex [23,24]. The potential for lesion enlargement, as observed in this case, highlights the need for ongoing monitoring and assessment to better understand the durability of ocrelizumab's effects.
Conclusion
In conclusion, the findings of this case study contribute to the growing body of evidence supporting the efficacy of ocrelizumab in the treatment of BCS. The observed reduction in lesion size, improvement in imaging metrics, and positive impact on clinical outcomes align with results from pivotal clinical trials. However, the observed lesion enlargement underscores the need for continued research to elucidate factors influencing treatment response and the long-term stability of therapeutic effects.
Patient consent
Written consent for publication has been obtained from the patient(s), their next of kin, their legal representative(s), or, where none of these parties is traceable, that approval to publish has been given by my organization/institution, its legal representative and an ethics committee, and in accordance with local legislation.
Footnotes
Competing Interests: The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- 1.Given CA, Stevens BS, Lee C. The MRI appearance of tumefactive demyelinating lesions. AJR Am J Roentgenol. 2004;182:195–199. doi: 10.2214/ajr.182.1.1820195. [DOI] [PubMed] [Google Scholar]
- 2.Saindane AM, Cha S, Law M, Xue X, Knopp EA, Zagzag D. Proton MR spectroscopy of tumefactive demyelinating lesions. AJNR Am J Neuroradiol. 2002;23:1378–1386. [PMC free article] [PubMed] [Google Scholar]
- 3.Stadelmann C, Ludwin S, Tabira T, Guseo A, Lucchinetti CF, Leel-Ossy L, et al. Tissue preconditioning may explain concentric lesions in Balo's type of multiple sclerosis. Brain. 2005;128(Pt 5):979–987. doi: 10.1093/brain/awh457. [DOI] [PubMed] [Google Scholar]
- 4.Tersegno MM, Reich DR. Balo's concentric sclerosis: a rare form of multiple sclerosis manifested as a dominant cerebral mass without other white matter lesions on MR. AJR Am J Roentgenol. 1993;160:901. doi: 10.2214/ajr.160.4.8456698. [DOI] [PubMed] [Google Scholar]
- 5.Kreft KL, Mellema SJ, Hintzen RQ. Spinal cord involvement in Balo's concentric sclerosis. J Neurol Sci. 2009;279(1-2):114–117. doi: 10.1016/j.jns.2008.12.030. [DOI] [PubMed] [Google Scholar]
- 6.Chen CJ, Ro LS, Chang CN, Ho YS, Lu CS. Serial MRI studies in pathologically verified Balo's concentric sclerosis. J Comput Assist Tomogr. 1996;20:732–735. doi: 10.1097/00004728-199609000-00008. [DOI] [PubMed] [Google Scholar]
- 7.Weinshenker BG, Miller D. In: Frontiers in multiple sclerosis. vol 2. Siva A, Kesselring J, Thompson AJ, editors. Martin Dunitz Ltd; London: 1999. Multiple sclerosis: one disease or many? pp. 37–46. [Google Scholar]
- 8.Arif S, Wali MW, Slehria AU, Khalid H, Malik H. Onion peel appearance in Balos concentric sclerosis–a variant of multiple sclerosis. J Ayub Med Coll Abbottabad. 2015;27(1):236–238. [PubMed] [Google Scholar]
- 9.Moore GR, Neumann PE, Suzuki K, Lijtmaer HN, Traugott U, Raine CS. Balo's concentric sclerosis: new observations on lesion development. Ann Neurol. 1985;17:604–611. doi: 10.1002/ana.410170614. [DOI] [PubMed] [Google Scholar]
- 10.Li Y, Xie P, Fan X, Tang H. Balo's concentric sclerosis presenting with benign clinical course and multiple sclerosis-like lesions on magnetic resonance images. Neurol India. 2009;57(1):66–68. doi: 10.4103/0028-3886.48815. [DOI] [PubMed] [Google Scholar]
- 11.Canellas AR, Gols AR, Izquierdo JR, Subirana MT, Gairin XM. Idiopathic inflammatory-demyelinating diseases of the central nervous system. Neuroradiology. 2007;49(5):393–409. doi: 10.1007/s00234-007-0216-2. [DOI] [PubMed] [Google Scholar]
- 12.Chen C-J, Chu N-S, Lu C-S, Sung C-Y. Serial magnetic resonance imaging in patients with Balò's concentric sclerosis: natural history of lesion development. Ann Neurol. 1999;46:651–656. doi: 10.1002/1531-8249(199910)46:4<651::aid-ana15>3.0.co;2-y. [DOI] [PubMed] [Google Scholar]
- 13.Kastrup O, Stude P, Limmroth V. Balo's concentric sclerosis. Evolution of active demyelination demonstrated by serial contrast-enhanced MRI. J Neurol. 2002;249:811–814. doi: 10.1007/s00415-002-0718-0. [DOI] [PubMed] [Google Scholar]
- 14.Brown J, Coles A, Jones J. First use of alemtuzumab in Balo's concentric sclerosis: a case report. Mult Scler. 2013;19:1673–1675. doi: 10.1177/1352458513498129. [DOI] [PubMed] [Google Scholar]
- 15.Hauser SL, Bar-Or A, Comi G, Giovannoni G, Hartung HP, Hemmer B, et al. Ocrelizumab versus interferon Beta-1a in relapsing multiple sclerosis. N Engl J Med. 2017;376(3):221–234. doi: 10.1056/NEJMoa1601277. [DOI] [PubMed] [Google Scholar]
- 16.Montalban X, Hauser SL, Kappos L, Arnold DL, Bar-Or A, Comi G, et al. Ocrelizumab versus placebo in primary progressive multiple sclerosis. N Engl J Med. 2017;376(3):209–220. doi: 10.1056/NEJMoa1606468. [DOI] [PubMed] [Google Scholar]
- 17.Comi G, Bar-Or A, Lassmann H. Role of ocrelizumab in treating multiple sclerosis, including acute severe relapses. Neurotherapeutics. 2017;14(4):978–985. [Google Scholar]
- 18.Lindquist S, Bodammer N, Kaufmann J. Histopathology and serial, multimodal magnetic resonance imaging in a multiple sclerosis variant. Mult Scler. 2007;13:471–482. doi: 10.1177/1352458506071329. [DOI] [PubMed] [Google Scholar]
- 19.Pardini M, Cutter GR, Sormani MP. Effect of fingolimod on diffusion measures in multiple sclerosis: a two-year study. J Neuroimaging. 2017;27(3):300–306. [Google Scholar]
- 20.Kappos L, Li D, Calabresi PA, O'Connor P, Bar-Or A, Barkhof F, et al. Ocrelizumab in relapsing-remitting multiple sclerosis: a phase 2, randomised, placebo-controlled, multicentre trial. Lancet North Am Ed. 2011;378(9805):1779–1787. doi: 10.1016/S0140-6736(11)61649-8. [DOI] [PubMed] [Google Scholar]
- 21.Yeo CJ, Hutton GJ, Fung SH. Advanced neuroimaging in Balo's concentric sclerosis: MRI, MRS, DTI, and ASL perfusion imaging over 1 year. Radiol Case Rep. 2018;13(5):1030–1035. doi: 10.1016/j.radcr.2018.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bazzurri V, Curti E, Tsantes E, Fiore A, Franceschini A, Granella F. Ocrelizumab in tumefactive multiple sclerosis onset. A case report. Neuroimmunol Rep. 2022;2 [Google Scholar]
- 23.Giovannoni G, Tomic D, Bright JR, Havrdova E. No evident disease activity": the use of combined assessments in the management of patients with multiple sclerosis. Mult Scler. 2017;23(9):1179–1187. doi: 10.1177/1352458517703193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rotstein DL, Healy BC, Malik MT, Chitnis T, Weiner HL. Evaluation of no evidence of disease activity in a 7-year longitudinal multiple sclerosis cohort. JAMA Neurol. 2015;72(2):152–158. doi: 10.1001/jamaneurol.2014.3537. [DOI] [PubMed] [Google Scholar]