SUMMARY
Background:
CHAPLE is an ultra-rare genetic disorder characterized by intestinal lymphatic damage, lymphangiectasia, and protein-losing enteropathy caused by overactivation of the complement system. We assessed the efficacy and safety of pozelimab, an antibody blocking complement component 5 (C5).
Methods:
This open-label, single-arm, historically-controlled study evaluated ten patients with CHAPLE disease (ClinicalTrials.gov, NCT04209634) treated weekly with subcutaneous pozelimab (following loading). The primary endpoint was proportion of patients achieving serum albumin normalization with improvement in active and no worsening in inactive clinical manifestations (frequency of problematic abdominal pain, bowel movement frequency, facial edema severity, and peripheral edema severity) at week 24. Secondary/exploratory efficacy endpoints and safety are also reported.
Findings:
All ten patients experienced serum albumin normalization and improvement with no worsening in clinical outcomes. Complete inhibition of complement activity (CH50) was achieved. Nine patients experienced adverse events; two were severe, and one experienced adverse events considered related to pozelimab.
Interpretation:
Pozelimab inhibits complement overactivation and resolves the clinical and laboratory manifestations of CHAPLE disease.
INTRODUCTION
CD55 deficiency with hyperactivation of complement, angiopathic thrombosis, and protein-losing enteropathy (CHAPLE) disease is an ultra-rare autosomal recessive disorder caused by loss-of-function variants of the CD55 gene. Heterozygous persons are unaffected (1). Loss of CD55 (also known as decay-accelerating factor), a cell surface regulator of complement components 3 (C3) and 5 (C5), (2) leads to overactivation of the terminal complement system (1, 3). The intestinal lymphatics are damaged by complement deposition, causing lymphangiectasia, gastrointestinal (GI) dysfunction, and protein-losing enteropathy (PLE) (1, 3). To date, <100 patients with CHAPLE disease have been identified (1, 3–8).
CHAPLE disease is associated with severe morbidity and mortality (1). Clinical and laboratory features of CHAPLE disease are often apparent in infancy and early childhood and include hypoalbuminemia and edema; hypogammaglobulinemia leading to infections; and GI symptoms such as abdominal pain, loss of appetite, vomiting, and diarrhea. Due to protein loss and malnutrition, patients present with micronutrient deficiency, anemia, and retardation of growth and maturation (1, 6). CHAPLE disease may progress to GI obstruction and perforation as well as mesenteric, portal, and cardiopulmonary thromboembolic events, which can be fatal (6, 8).
Patients with CHAPLE disease receive supportive interventions such as albumin infusions, immunoglobulin (Ig) replacement therapy, immunosuppressive medications, bowel resection surgery, and vitamin/micronutrient supplements, but these are not curative and do not achieve lasting improvement (6). Given the etiology of CHAPLE disease, anti-complement therapy has been proposed as a treatment (6). In a series of observational case studies, off-label use of eculizumab, a humanized monoclonal anti-C5 antibody, (5) demonstrated consistent results across multiple cohorts of patients with CHAPLE disease (21 patients in total) (3, 6, 9–11). These studies collectively demonstrated a reduction in complement activation, increased albumin and total protein concentrations, restoration of metabolic and immune function, and improvement in signs and symptoms and overall quality of life. These findings were not collected in accordance with the U.S Food and Drug Administration’s (FDA) patient-focused drug development guidance (12), but held promise for future interventional trials evaluating complement inhibitor therapies in CHAPLE disease. Eculizumab necessitates intravenous (IV) infusion every two weeks, posing challenges in regions lacking access to specialized medical facilities. Furthermore, it is not available in many countries. Thus, a comprehensive investigation of a more accessible treatment approach is warranted.
Pozelimab is a subcutaneously (SC)-administered, fully human immunoglobulin G4P (IgG4P; IgG4 with a proline substitution to promote light chain stabilization) antibody directed against C5. A previous study demonstrated that pozelimab inhibits C5 cleavage and membrane attack complex (MAC) formation (13). Here, we conducted an open-label, historically-controlled study to assess the efficacy and safety of pozelimab in patients with CHAPLE disease. Importantly, pozelimab was recently approved by the U.S. FDA as the first treatment for adult and pediatric patients 1 year of age and older with CD55-deficient PLE, also known as CHAPLE disease, based on the findings presented in this report (14).
METHODS
Study design
This ongoing, open-label, single-arm, historically-controlled, multicenter, phase 2/3 study evaluated the efficacy and safety of pozelimab in pediatric and adult patients with CHAPLE disease (ClinicalTrials.gov, NCT04209634). This study comprised a screening period (≤4 weeks; ≤10 weeks for patients with specific circumstances as defined in the protocol), a 144-week treatment period, and a follow-up period (Figure S1). At baseline (day 1), patients received a single IV loading dose of pozelimab 30 mg/kg, followed by SC dosing once weekly over the treatment period based on body weight at a concentration of 200 mg/mL as either a single injection (<40 kg) or two injections (≥40 kg) (Table S1).
The study was initiated on January 27, 2020. The efficacy data presented in this report corresponds to all patients completing the week 48 assessment and having at least 52 weeks of treatment exposure (data cut-off date of May 24, 2022). The safety data in this report includes an additional 90 days and corresponds to all patients having at least 72 weeks of treatment exposure (data cut-off date of November 2, 2022). Additional study information, including amendment history, is described in the protocol.
Trial oversight
This study was conducted at three study sites in Thailand, Türkiye, and the United States. The study protocol was approved by the local institutional review boards and/or ethics committees prior to study initiation. All patients or legally-authorized representatives provided written informed consent prior to study enrollment.
This study was conducted in accordance with the principles of the Declaration of Helsinki and Council for International Organizations of Medical Sciences International Ethical Guidelines, and was consistent with Good Clinical Practices of the International Conference on Harmonisation and applicable regulatory requirements. Monitoring and site supervision were performed with oversight by the sponsor.
Protocol deviations are described in the Supplementary Material.
Patients
Patients aged ≥1 year with a clinical diagnosis of CHAPLE disease and a CD55 loss-of-function variant identified by genetic analysis and confirmed by flow cytometry or Western blot of CD55 from peripheral blood cells were eligible for this study. Qualifying disease was defined as hypoalbuminemia ≤3·2 g/dL during screening and one of the following signs or symptoms attributable to CHAPLE disease within the previous 6 months: diarrhea, vomiting, abdominal pain, peripheral or facial edema, infection with concomitant hypogammaglobulinemia, or a new thromboembolic event. Exclusion criteria included a history of meningococcal infection, concomitant disease causing hypoproteinemia or secondary intestinal lymphangiectasia, or a history of hereditary complement deficiency besides CD55 deficiency. Full inclusion and exclusion criteria are described in the protocol. The sex of each patient was determined at screening by the investigators.
Endpoints
The primary efficacy endpoint was the proportion of patients with active disease at baseline who achieved both the following at week 24: 1) normalization of serum albumin, defined as serum albumin within the normal range for ≥70% of measurements between weeks 12 and 24, and no single albumin measurement of <2·5 g/dL or requirement for albumin infusion between weeks 12 and 24, and 2) improvement in four prespecified clinical outcomes (frequency of problematic abdominal pain, bowel movement frequency, facial edema severity, and peripheral edema severity) that were evaluable for improvement at baseline, with no worsening of the outcome (if not evaluable for improvement at baseline) at week 24. Serum albumin was analyzed at a local laboratory. Additional information on collection and evaluation of the clinical outcomes are presented in the Supplementary Material.
Secondary efficacy endpoints included total complement activity (CH50) over time, frequency of albumin infusions, weight-for-age and stature-for-age, hospitalization days over time (emergency and planned admissions), use and dose frequency of concomitant medications including corticosteroids, improvement in stool consistency (additional information in the Supplementary Material), concentrations of total pozelimab in serum, and the following laboratory measurements: total albumin, protein and immunoglobulin levels over time; alpha-1 antitrypsin levels in blood and stool at weeks 12 and 24; vitamin B12, iron, and iron-binding capacity over time; and plasma lipids (triglycerides/cholesterol) over time. Additional secondary endpoints included improvement in the patient’s most bothersome sign or symptom at week 24 (see the Supplementary Material for details on qualitative interviews conducted with patients/caregivers at screening and week 24), changes in limitations in food and drink, as assessed by the PedsQL™ GI Symptom Scales, and changes in health-related quality of life as measured by the PedsQL™ Generic Core Scales. The secondary endpoint of abdominal ascites is not presented, since only one patient had ascites at baseline.
The incidence and severity of treatment-emergent adverse events and other safety variables (including anti-drug antibodies) over time were also evaluated as secondary endpoints. The following adverse events of special interest were assessed: moderate or severe infusion reactions, confirmed Neisseria infection, and thrombotic or embolic events. Markers of erythropoiesis and platelets were reported in the hematology panel.
Exploratory endpoints are described in the Supplementary Material.
Statistical analysis
Analysis sets are described in the Supplementary Material.
The proportion of patients achieving normalization of albumin and improvement in the prespecified evaluable clinical outcomes, and Clopper Pearson 90% confidence interval, were reported. Summary statistics were calculated to evaluate change from baseline in CH50 activity, albumin, protein, and immunoglobulin levels; alpha-1 antitrypsin levels; and micronutrients including vitamin B12 and iron. Other clinical laboratory values (markers of erythropoiesis, platelets, and plasma lipid levels) were summarized descriptively including median time to normalization estimated using Kaplan-Meier estimation with 95% confidence intervals. Frequency of albumin infusions, hospitalization days, and number of bowel movements per day were summarized using counts by day or periods of time. Patient/Caregiver Global Impression of Change (GIC) and Patient/Caregiver Global Impression of Severity (GIS) scores were reported overall and stratified by respondent type. Safety events were described descriptively, and treatment-emergent adverse events were recorded using the Medical Dictionary for Regulatory Activities version 25·0. Statistical analyses were performed using SAS version 9·4 or higher according to the statistical analysis plan. Success criteria, sample size, power considerations, and the post-hoc piecewise model for estimating change in albumin levels are presented in the Supplementary Material.
Role of the funding source
The principal investigators and the sponsor designed the trial protocol and selected the participating sites. Additional study contributors are listed in the Supplementary Material. The sponsor participated in the collection, analysis, and interpretation of the data and checked information provided in the manuscript. All authors had access to the data, contributed to the drafting and revision of the manuscript, and agreed with the decision to submit the manuscript for publication. The authors vouch for the accuracy and completeness of the data and for the fidelity of the trial to the protocol.
RESULTS
Baseline demographics and clinical characteristics
Ten patients were enrolled in the study (Figure S2) and included in the analysis populations. Baseline demographics and clinical characteristics are presented in Table 1. Patients were predominantly pediatric with a median (IQR) age of 8·5 (5·0) years. Most patients originated from Türkiye (5/10), with the remainder originating from Syria (2/10), Thailand (2/10), and Bolivia (1/10). Three patients (including siblings from Thailand) were born to non-consanguineous parents and all seven patients from Türkiye and Syria were born to consanguineous parents. Patients had markedly low weight-for-age and stature-for-age, with median (interquartile range [IQR]) growth percentiles of 13·0 (29·1) and 6·9 (18·7), respectively. The median (IQR) age of symptom onset was 2·2 (2·9) years, and median (IQR) duration of symptoms at baseline was 6·0 (5·1) years. Mean albumin at baseline was 2·2 g/dL, which was considerably below the local laboratory reference range. All ten patients had received vaccination for Neisseria meningitidis and took antibacterials to prevent of meningococcal infections while on pozelimab. Five patients had a history of iron deficiency and received iron supplementation prior to and during the study. No patients were receiving Ig replacement therapy at baseline.
Table 1:
Baseline demographics and clinical characteristics
| Pozelimab (N = 10) | |
|---|---|
|
| |
| Age, years, median (IQR) | 8·5 (5·0) |
| Sex, n | |
| Male | 4 |
| Female | 6 |
| Country of origin, n | |
| Türkiye | 5 |
| Syria | 2 |
| Thailand | 2 |
| Bolivia | 1 |
| Race, n | |
| White | 7 |
| Asian | 2 |
| Other | 1 |
| Weight-for-age CDC percentile, median (IQR) | 13·0 (29·1) |
| Stature-for-age CDC percentile, median (IQR) | 6·9 (18·7) |
| Age at symptom onset, years, median (IQR) | 2·2 (2·9) |
| Duration of symptoms at baseline, years, median (IQR) | 6·0 (5·1) |
| Patients with history of albumin transfusions, n | 10 |
| Patients with prior thromboembolic events, n | 1 |
| Albumin, g/dL, mean (SD)a | 2·2 (0·5) |
| Patients evaluable at baseline for each clinical outcome, n | |
| Frequency of problematic abdominal pain | 7 |
| Bowel movements per day | 1 |
| Facial edema by physician assessment | 4 |
| Peripheral edema by physician assessment | 5 |
| Facial edema score by physician assessment, median (IQR)b | 2·0 (1·0) |
| Peripheral edema score by physician assessment, median (IQR)b | 2·5 (1·0) |
| IgG, g/L, mean (SD)c | 2·2 (0·9) |
| Fecal/serum A1AT ratio, mean (SD)d | 3·1 (1·4) |
| Most bothersome sign or symptom, n | |
| Abdominal pain | 9 |
| Facial edema | 1 |
Full Analysis Set presented.
Local clinical laboratory reference range: 3·5–5·0 g/dL.
The edema scoring system employs a Likert scale, which spans from 1 to 5, where a rating of 5 indicates the most severe level of edema.
The LLN for IgG varied by age and sex, with the lowest LLN of 4·13 g/L in males aged 1–3 years and the highest LLN of 8·04 g/L in females aged 16–17 years.
Central clinical laboratory reference range: normal ≤0·28.
A1AT, alpha-1-antitrypsin; CDC, Centers for Disease Control and Prevention; IgG, immunoglobulin G; IQR; interquartile range; LLN, lower limit of normal; N, number of patients in analysis set; n, number of patients within a specified category; SD, standard deviation.
All ten patients had active disease at baseline. Most patients had mild-to-moderate facial and peripheral edema, with median (IQR) scores of 2·0 (1·0) and 2·5 (1·0), respectively (1 indicates no edema, 5 indicates very severe edema). The median (IQR) score for frequency of problematic abdominal pain was 56·3 (62·5) and the median (IQR) number of daily bowel movements was 1.6 (1·9). During the qualitative interviews, 31 different signs and symptoms of CHAPLE disease were reported, and the core signs and symptoms of CHAPLE disease (experienced by ≥90% of patients) were abdominal pain (10/10), diarrhea (10/10), facial edema (10/10), peripheral edema (10/10), vomiting (10/10), and nausea (9/10). The most bothersome sign/symptom of CHAPLE disease was abdominal pain for 9/10 of patients and facial edema for 1/10 of patients. Patient histories revealed that abdominal pain was frequent and substantial, with some needing parenteral analgesics for pain relief. Clinical history of four patients included in this study was previously reported: patients 1 and 2 (8); patient 6 (1); and patient 9 (6). Only patient 9 had prior exposure to eculizumab. Following the discontinuation of eculizumab on day −125, this patient re-developed active disease. Additional patient characteristics are presented in Table S2.
Loss-of-function mutations in the CD55 gene for each patient are presented in Table S2. Eight patients have homozygous mutations and two patients (siblings) have compound heterozygous mutations. Two pairs of patients share identical mutations, one of which is the sibling pair. Three patients had novel CD55 variants, with one previously described as a variant of uncertain significance (15). The remaining variants have been previously reported (1, 3, 6, 8). All patients had deficient CD55 cell surface protein expression.
Complement activity
Complete inhibition of the ex vivo CH50 biomarker of the classical complement pathway (16) was observed in in patients treated with pozelimab (Figure S3A). Furthermore, mean sC5b-9 concentrations in plasma, a biomarker of terminal complement pathway activation, (17) sharply decreased following treatment and reached a plateau after week 2 with continued dosing (Figure S3B).
Albumin
All ten patients reported persistent hypoalbuminemia (Table S3) and had a history of albumin transfusions, with abnormal albumin values at baseline (Table 1). Following pozelimab treatment, all ten patients achieved normalization of serum albumin between weeks 12 and 24 (Table 2). Albumin levels rapidly improved after pozelimab treatment and were sustained at normal levels, with all time points after four weeks above 3·5 g/dL (Figure 1A).
Table 2:
Primary efficacy endpoint
| Pozelimab (N = 10) | |
|---|---|
|
| |
| Patients meeting the primary endpoint – normalization of serum albumin and improvement in clinical symptoms that were evaluable for improvement at baseline, n/N | 10/10 |
| Clopper Pearson 90% confidence interval (%) | 74·1, 100·0 |
| Normalization of serum albumin between week 12 and 24, n/N | 10/10 |
| 70% of serum albumin measurements ≥3·5 g/dL between week 12 and week 24 | 10/10 |
| No single albumin measurement of <2·5 g/dL between week 12 and week 24 | 10/10 |
| No requirement for albumin transfusion between week 12 and week 24 | 10/10 |
| Improvement and no worsening in clinical symptoms at week 24, n/N | 10/10 |
| Improvement and no worsening in frequency of problematic abdominal pain at week 24a | 10/10 |
| Improvementb | 7/7 |
| No worseningc | 3/3 |
| Improvement and no worsening in the number of bowel movements per day at week 24d | 10/10 |
| Improvementb | 1/1 |
| No worseningc | 9/9 |
| Improvement and no worsening in facial edema by physician assessment at week 24e | 10/10 |
| Improvementb | 4/4 |
| No worseningc | 6/6 |
| Improvement and no worsening in peripheral edema by physician assessment at week 24e | 10/10 |
| Improvementb | 5/5 |
| No worseningc | 5/5 |
Full Analysis Set presented.
Patient/caregiver assessment using the Stomach pain and Hurt subscale of the PedsQL™ GI Symptoms Scales.
Evaluable is defined as ≤70 points at baseline; improvement is defined as an increase of ≥25 points; worsening is defined as a decrease of ≥25 points.
In patients evaluable for improvement at baseline.
In patients not evaluable for improvement at baseline.
Patient/caregiver assessment using an e-diary. Evaluable is defined as having ≥3 bowel movements per day at baseline; improvement is defined as a reduction of ≥50% in the one-week average; worsening is defined as an increase of ≥30% in the one-week average.
Physician assessment (physician-assessed score). Evaluable is defined as ≥3 of 5 points at baseline; improvement is defined as a reduction of ≥2 points (out of 5); worsening is defined as an increase of ≥2 points (out of 5).
N, number of patients in analysis set; n, number of patients within a specified category.
Figure 1: Serum albumin, albumin transfusions, and hospitalizations over time.
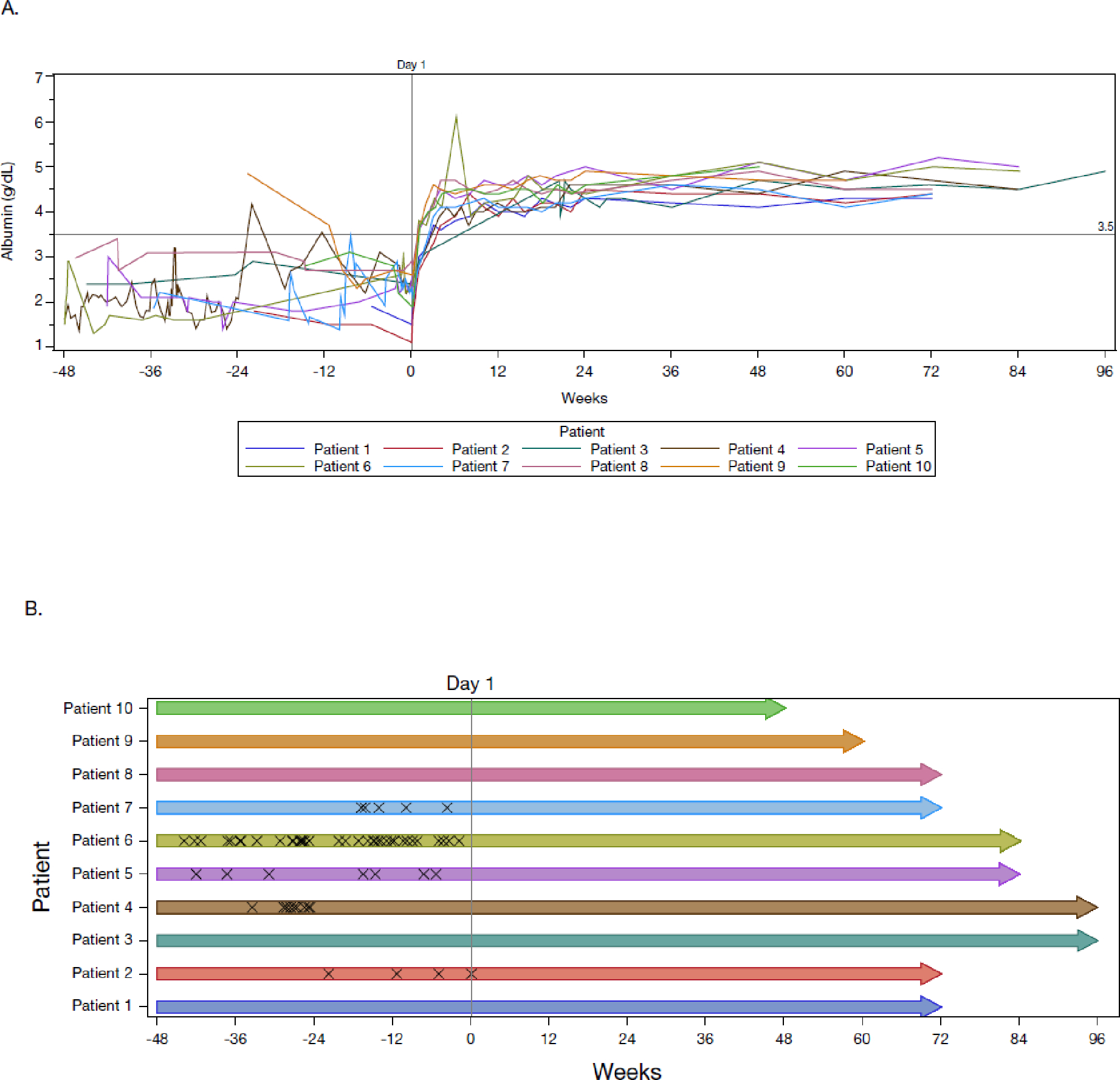
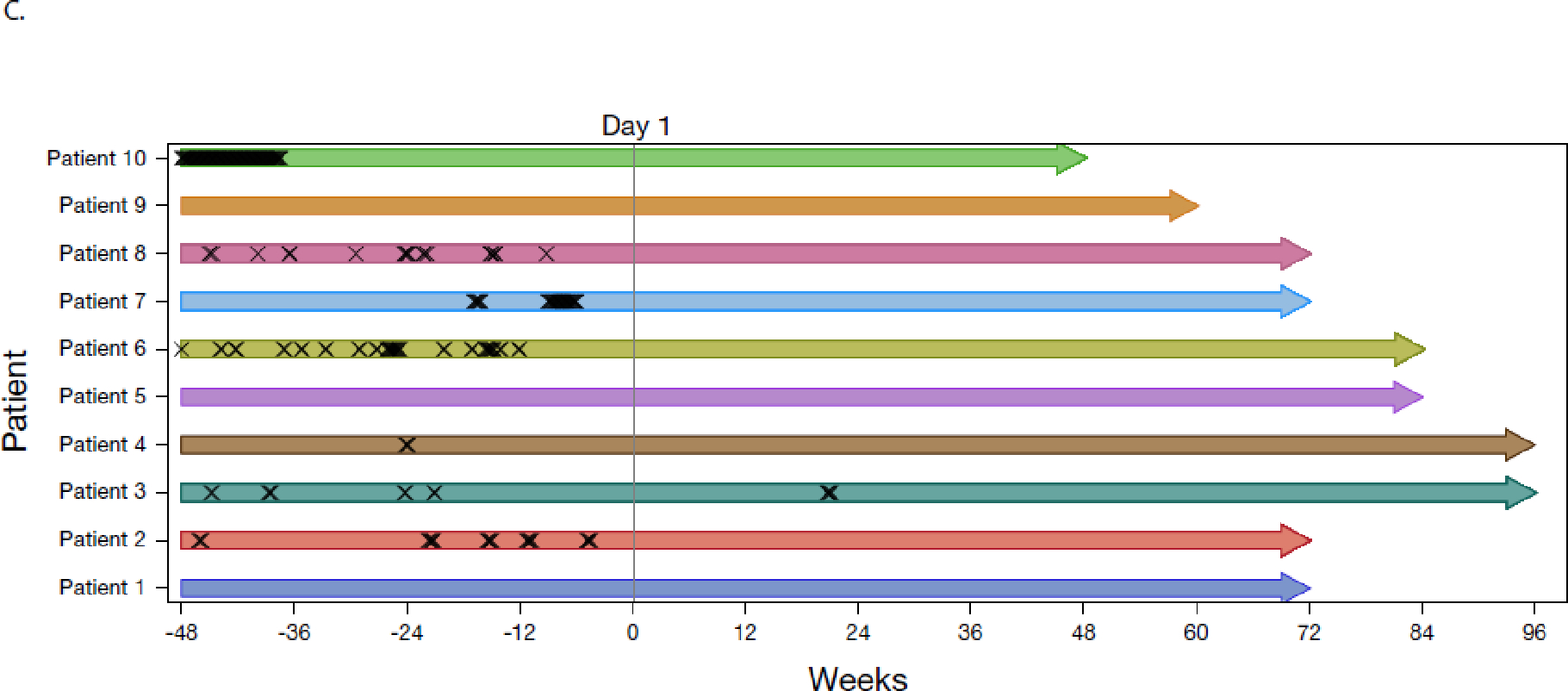
Panel A is a plot of serum albumin concentration as a function of time. Each colored line represents a patient. Horizontal line at 3·5 g/dL is the lower range of the normal per the local clinical laboratory. Panel B is a plot of albumin transfusions (“X” symbols) as a function of time. Pre-study albumin transfusions were collected from medical history and prior/concomitant medications, and on-study albumin transfusions were captured as adverse events. Panel C is a plot of the post hoc analysis of hospitalizations due to either ≥1 sign or symptom of CHAPLE disease or complications of CHAPLE disease (“X” symbols) as a function of time. Pre-study hospitalizations are based on historical medical records of hospitalizations regardless of reason or duration. On-study hospitalizations were defined as admission to a hospital or emergency room for longer than 24 hours. For panels B and C, each bar represents a patient, and the length of the bar represents the time to the last scheduled visit prior to the cut-off date. Data in all panels are plotted from 48 weeks prior to baseline through the data cut-off date of May 24, 2022. Treatment was initiated at study day 1. Full Analysis Set presented.
Following pozelimab treatment, the need for albumin transfusions was eliminated (Figure 1B). Five of ten patients received an albumin transfusion in the 48 weeks prior to treatment, with a pre-treatment mean of 6·0 albumin transfusions across all ten patients (60 transfusions in total). The mean number of albumin transfusions across all ten patients decreased to 0·1 (1 transfusion) by week 24 (Table S4). Only one patient received an albumin transfusion during the study, which occurred on day 1 due to low albumin (1·1 g/dL) at baseline. A post-hoc analysis of pre- and post-treatment albumin levels shows no change in albumin levels during the pre-treatment period, with a sharp increase during weeks 0 to 12 post-treatment and sustained normalization during weeks 12 to 48 (Table S5).
Growth
Over the relatively short 48-week treatment period, most patients experienced remarkable catch-up growth (Figure 2, Table S6, Table S7). Weight-for-age and stature-for-age percentiles increased dramatically, with a mean change from baseline in growth percentiles of 15·5 and 11·2, respectively. Corresponding increases in BMI were modest because of parallel increases in height and weight.
Figure 2: Body weight-for-age and stature-for-age based on the CDC growth charts.
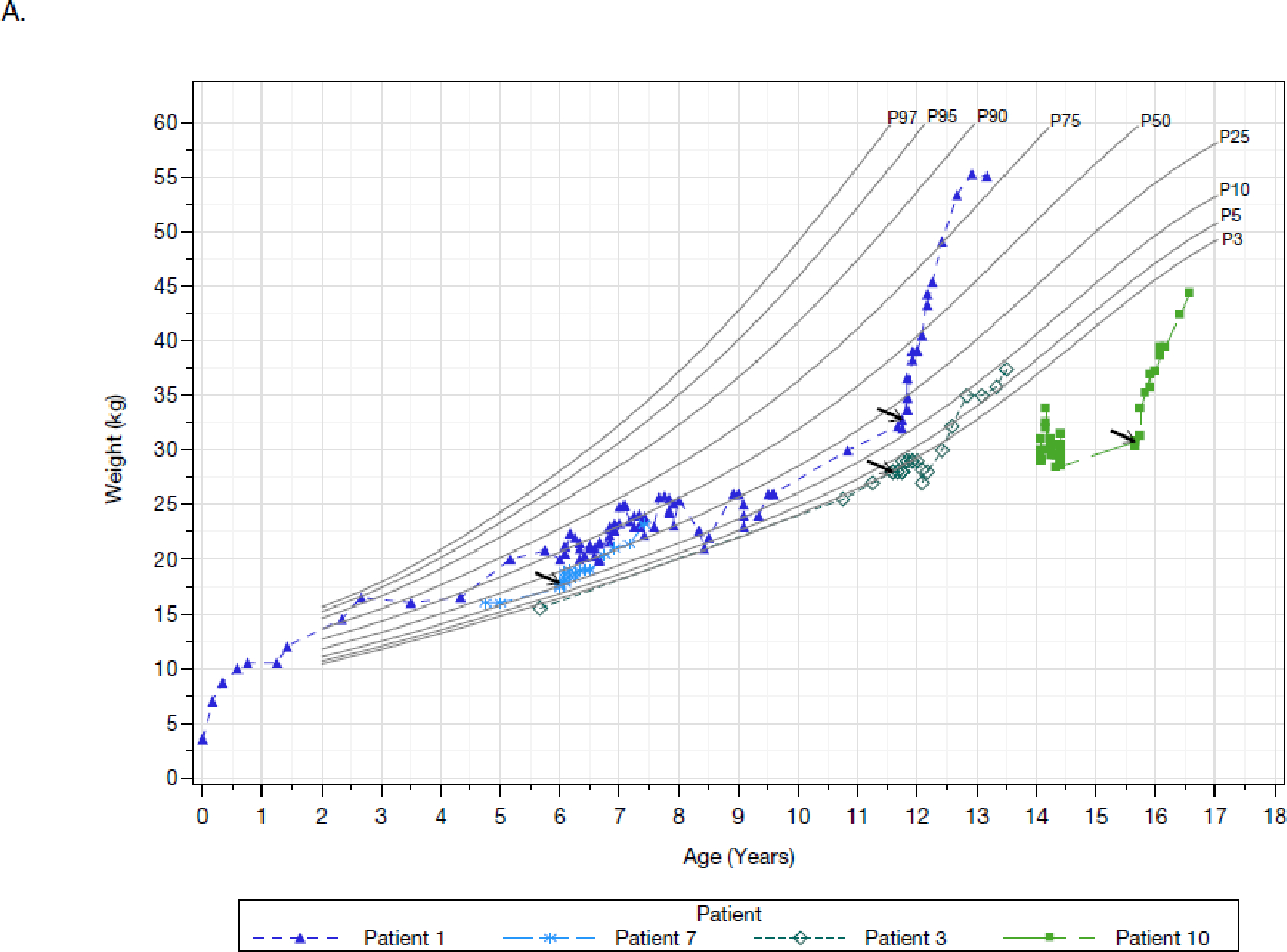
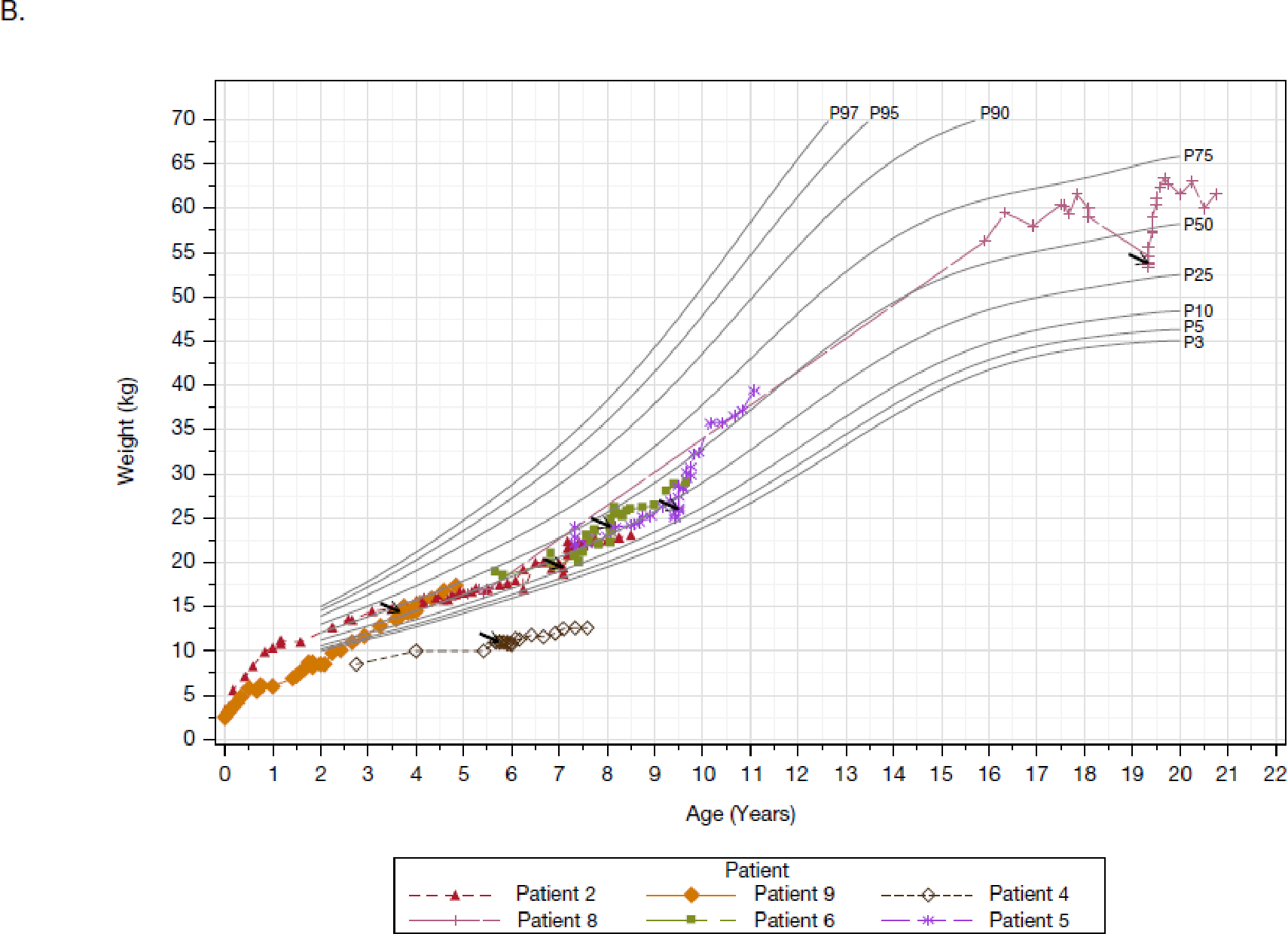
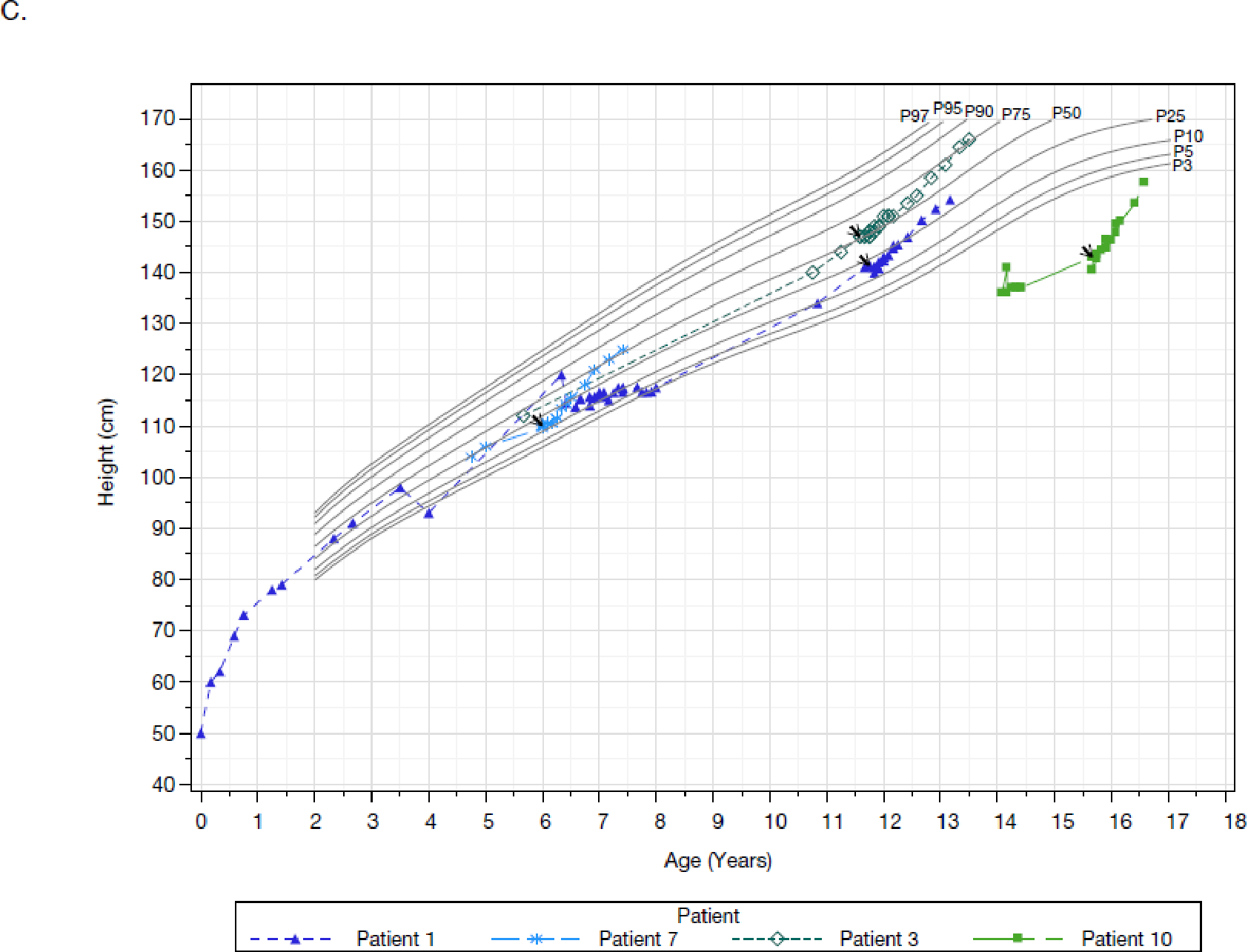
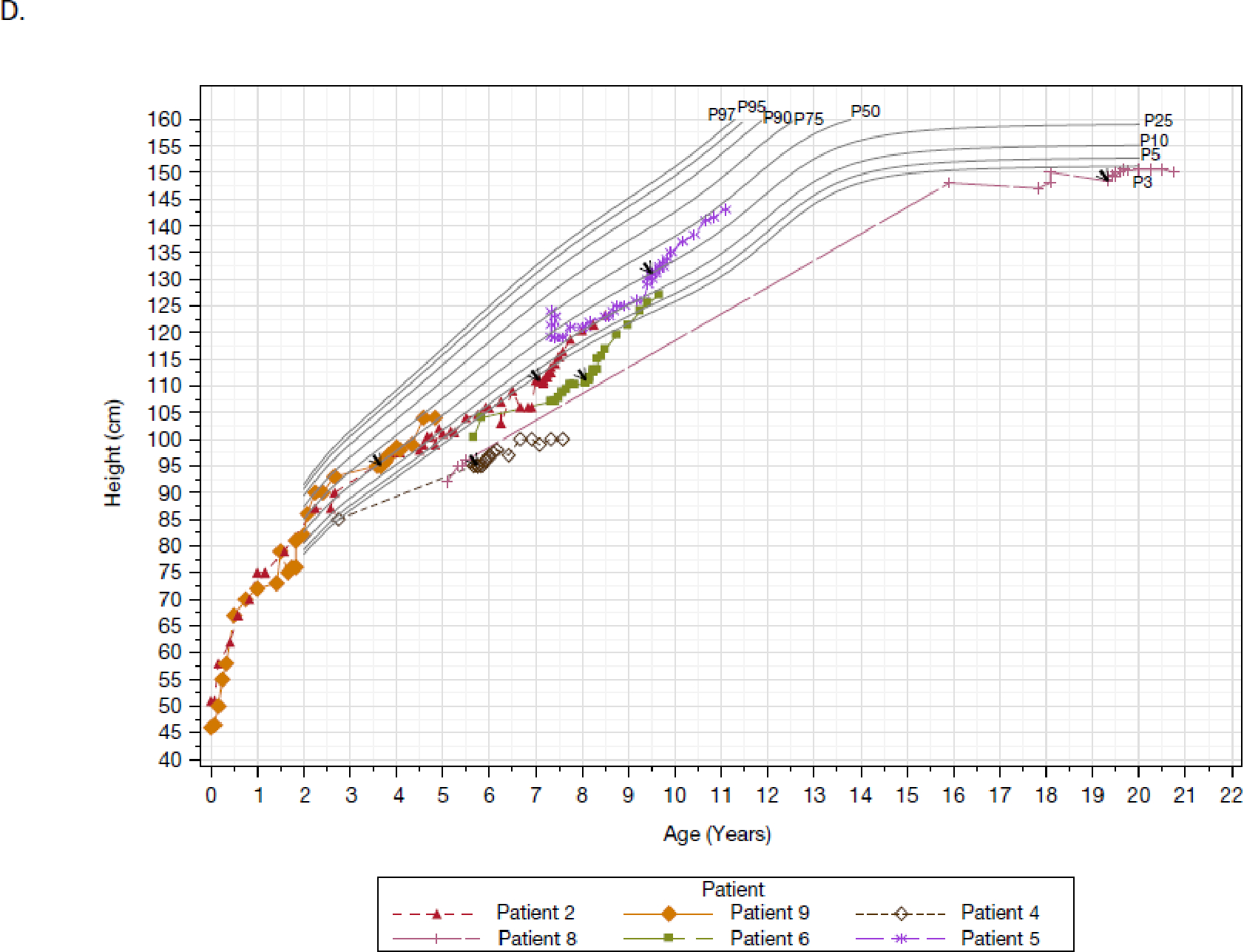
Panels A and B are the weight-for-age plots for males (four patients) and females (six patients), respectively. Panels C and D are the stature-for-age plots for males (four patients) and females (six patients), respectively. Each line and corresponding colored symbol represents a patient. Arrows indicate the first administration of pozelimab. Percentiles are sourced from the CDC (https://www.cdc.gov/growthcharts/percentile_data_files.htm). Full Analysis Set presented.
Morbidity
Prior to treatment, patients experienced substantial morbidity. Patient histories indicate persistent symptoms with various life-threatening episodes that affected patients’ daily functioning and activities. All ten patients had a history of abdominal imaging, endoscopies, and/or surgeries, with two patients having multiple bowel resections. Additionally, all ten patients experienced multiple hospitalizations since disease onset, with some requiring admission to the intensive care unit. In a post-hoc analysis accounting only for hospitalizations with ≥1 sign or symptom consistent with CHAPLE disease, 7/10 patients were hospitalized for a mean of 19·6 days across all ten patients (196 hospitalization days) in the 48 weeks prior to treatment. Similarly, in the prespecified analysis of all-cause hospitalization, 9/10 patients were hospitalized for a mean number of 26·8 days across all ten patients (268 hospitalization days) in the 48 weeks prior to treatment.
Seven patients received corticosteroids prior to study enrollment. Pre-treatment clinical data showed corticosteroid effect was partial, dose-dependent, heterogeneous, and not considered a lasting solution because of its limited efficacy on overall disease manifestations (Figure S4). Five patients initiated corticosteroids in the 48 weeks prior to treatment, three of these for the first time; four of these patients were still receiving corticosteroids at baseline. One patient had their corticosteroid doses tapered and withdrawn in the 48 weeks prior to treatment.
Following pozelimab treatment, patients had reduced hospitalization days due to ≥1 sign or symptom consistent with CHAPLE disease (Figure 1C; post-hoc analysis). The mean number of hospitalization days across all ten patients decreased to 0·4 days (4 hospitalization days) by week 24 following treatment, with no hospitalizations after week 24. One patient was hospitalized during the study for vomiting and dehydration due to suspected food poisoning, neither of which were deemed by the investigator as related to pozelimab treatment or the underlying condition. The prespecified analysis of all-cause hospitalization also showed reduced hospitalization days following pozelimab treatment (Table S8).
No patients took immunomodulators or anticoagulants after starting pozelimab, and no small bowel resection surgeries were performed. Seven patients who underwent standard of care abdominal CT or MRI imaging before and during the treatment period showed resolution of small bowel wall thickening/enhancement and mesenteric lymphadenopathy. Remarkably, all four patients had been withdrawn from corticosteroids as of the data cut-off for this report (Table S9). There was a decrease in the mean (SD) cumulative corticosteroid use, expressed in mg of hydrocortisone equivalents, across all ten patients from 898·7 (1402·2) in the 24 weeks prior to baseline (weeks −24 to 0) to 589·2 (1016·9) in the 24 weeks after dosing (weeks 0 to 24), and further decrease to 126·0 (398·5) in the subsequent 24 weeks after dosing (weeks 24 to 48). The relationship of between albumin concentration, corticosteroid use, and albumin transfusions in several patients are shown in Figure S4.
Other laboratory measures
Total protein and immunoglobulins
Total protein and total Ig levels rapidly improved with pozelimab treatment (Figure S5, Table S10). Patients experienced rapid and sustained normalization in other serum protein levels, including IgG, IgM, IgA, and total protein (Table S10).
Alpha-1-antitrypsin
We evaluated fecal:serum alpha-1-antitrypsin ratio as a measure of enteric protein loss. Nine patients evaluated at baseline had elevated levels ratios, with a mean (SD) value of 3·1 (1·4), reflective of active disease. Following pozelimab treatment, ratios rapidly normalized, with mean (SD) values of 0·1 (0·0) and 0·1 (0·0) by weeks 12 (n = 4) and 24 (n = 8), respectively (Figure S6).
Erythropoiesis
We observed clinically meaningful improvements in markers of erythropoiesis following pozelimab treatment, with increases in hemoglobin level (Table S11), erythrocyte mean corpuscular hemoglobin, and erythrocyte mean corpuscular volume; and a concomitant decrease from baseline in absolute reticulocytes along with hematologic recovery (Table S12). Levels of vitamin B12, hemoglobin, unsaturated iron binding capacity, and ferritin also increased markedly over time (Table S11).
Other laboratory indicators of protein-losing enteropathy
Elevated platelets are a distinct but consistent feature of CHAPLE disease (compared to other acquired or genetic PLEs). Additionally, high triglycerides are a feature of PLE, possibly related to catabolism of fat as an energy source or dysfunctional chylomicron transport from gut. For both triglycerides and platelets, there was a consistent trend towards a decrease (improvement) in levels over time following pozelimab treatment (Table S13).
Clinical outcomes assessments and qualitative interview results
At baseline, seven patients were evaluable for improvement in frequency of problematic abdominal pain (Figure S7), one patient was evaluable for improvement in the number of bowel movements per day (Figure S8), four patients were evaluable for improvement in facial edema severity (Figure 3A), and five patients were evaluable for improvement in peripheral edema severity (Figure 3B). Following 24 weeks of pozelimab treatment, all patients that were evaluable at baseline met the criteria for improvement in these four prespecified clinical outcomes, with other patients experiencing no worsening (Table 2).
Figure 3: Physician assessments of facial and peripheral edema over time.
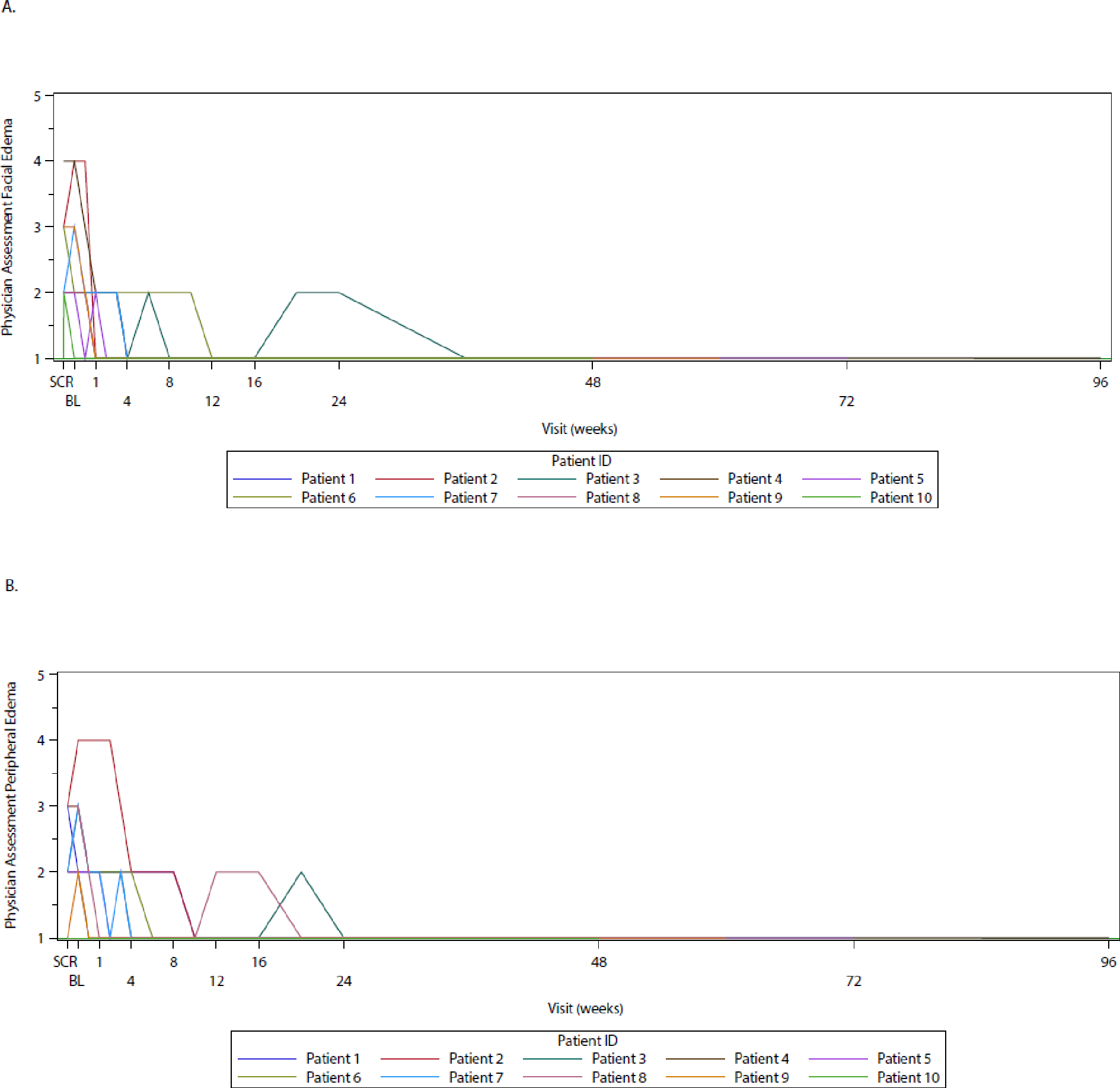
Panels A and B show data for facial and peripheral edema, respectively. Data are plotted from baseline through the data cut-off date of May 24, 2022. Each line represents a patient. Severity assessed with response levels of: 1 = no edema, 2 = mild edema, 3 = moderate edema, 4 = severe edema, 5 = very severe edema. Full Analysis Set presented. BL, baseline; WK, week.
As assessed by qualitative interviews, all ten patients also experienced complete resolution of the core signs and symptoms of CHAPLE disease at week 24, including those that were reported as most bothersome. Nine patients experienced improvement in all disease-related signs and symptoms, and no patients reported worsening of any signs and symptoms.
Patient/caregiver-reported outcomes were collected on stool consistency and quality of life. Patients/caregivers and clinicians also answered questions regarding their global impression of disease severity and change. The median (IQR) number of days per week with loose/watery stool consistency was 2·0 (2·3) at baseline and 0 (2·0) by week 24. Furthermore, consistent with the complete resolution of the core signs and symptoms of disease described above, we observed sustained improvements from baseline in GIC and GIS questions from the perspective of the patient/caregiver and clinician (Table S14, Table S15, Table S16, Table S17). Additionally, other patient/caregiver-reported outcome measures demonstrated substantial and sustained improvements in quality of life and alleviation of signs and symptoms of disease (Figure S9, Figure S10, Figure S11, Table S18). Treatment also had a positive effect on caregiver well-being (Table S19).
Safety
Nine patients experienced 47 treatment-emergent adverse events during the study, two of which were considered severe (COVID-19 and traumatic fracture; Table S20). One patient experienced six adverse events that were considered by the investigator as related to pozelimab treatment, including three events of metabolic acidosis, one event of alopecia, one event of contact dermatitis, and one event of injection-site erythema; all events were mild. The most common adverse events were rhinitis, abdominal pain, vomiting, iron deficiency, upper limb fracture, urticaria, and pyrexia (Table 3); each of these occurred in two patients, and none were considered related to pozelimab treatment. No adverse events were considered to be worsening of CHAPLE disease.
Table 3:
Summary of treatment-emergent adverse events
| System Organ Class Preferred Term | Pozelimab (N = 10) |
|---|---|
|
| |
| Number of patients with at least one TEAE, n | 9 |
| Infections and infestations, n | 6 |
| Rhinitis | 2 |
| Acarodermatitis | 1 |
| COVID-19 | 1 |
| Nasopharyngitis | 1 |
| Tonsillitis | 1 |
| Gastrointestinal disorders, n | 5 |
| Abdominal pain | 2 |
| Vomiting | 2 |
| Constipation | 1 |
| Gingival bleeding | 1 |
| Metabolism and nutrition disorders, n | 5 |
| Iron deficiency | 2 |
| Dehydration | 1 |
| Hypokalemia | 1 |
| Metabolic acidosis | 1 |
| Vitamin B12 deficiency | 1 |
| Injury, poisoning and procedural complications, n | 4 |
| Upper limb fracture | 2 |
| Contusion | 1 |
| Fall | 1 |
| Traumatic fracture | 1 |
| Skin and subcutaneous tissue disorders, n | 4 |
| Alopecia | 1 |
| Alopecia areata | 1 |
| Chronic spontaneous urticaria | 1 |
| Dermatitis contact | 1 |
| Urticaria | 1 |
| General disorders and administration site conditions, n | 3 |
| Pyrexia | 2 |
| Injection site erythema | 1 |
| Investigations, n | 2 |
| Blood glucose increased | 1 |
| Blood uric acid increased | 1 |
| Hepatic enzyme increased | 1 |
| Blood and lymphatic system disorders, n | 1 |
| Anemia folate deficiency | 1 |
| Cardiac disorders, n | 1 |
| Tachycardia | 1 |
| Immune system disorders, n | 1 |
| Immunization reactiona | 1 |
| Nervous system disorders, n | 1 |
| Headache | 1 |
| Renal and urinary disorders, n | 1 |
| Hematuria | 1 |
| Proteinuria | 1 |
Safety Analysis Set presented.
Verbatim term is Fever secondary to COVID-19 vaccine.
COVID-19, coronavirus disease 2019; N, number of patients in analysis set; n, number of patients within a specified category; TEAE, treatment-emergent adverse event.
Two patients experienced three serious adverse events. One patient experienced two treatment-emergent serious adverse events of vomiting and diarrhea leading to hospitalization, neither of which were considered related to pozelimab treatment. One patient experienced a treatment-emergent serious adverse event of traumatic limb fracture requiring internal fixation, which was not considered related to pozelimab treatment. There were no deaths and no adverse events that led to withdrawal from the study or permanent discontinuation of study drug.
There were no thrombotic or embolic events, consistent with decreased platelet levels following pozelimab treatment (Table S13). No patients receiving pozelimab had an anti-drug antibody response. No moderate or severe infusion reactions or Neisseria infections were observed.
Pharmacokinetics
The median concentration–time profile of total pozelimab was characterized by a rapid rise in total pozelimab concentrations following the initial 30 mg/kg IV dose, with continued accumulation until achieving steady state by approximately week 20 (Figure S12). The median steady-state pozelimab trough concentration was approximately 426 mg/L (IQR, 160 mg/L).
DISCUSSION
We hypothesized that complement pathway overactivation in CHAPLE disease (18) may be ameliorated by pozelimab treatment, a monoclonal antibody directed against the terminal complement protein C5. Patients with CHAPLE disease showed complement inhibition and a complete and sustained resolution in signs and symptoms of disease following pozelimab treatment. Patients exhibited rapid catch-up growth and substantial improvements in disease morbidity, including reduced hospitalization and use of concomitant therapies such as corticosteroids. Serum albumin levels normalized and the need for albumin transfusions was eliminated. Furthermore, we observed clinically meaningful improvements in other laboratory measures of disease, including serum proteins, fecal alpha-1-antitrypsin, markers of erythropoiesis, micronutrient status, and thrombocythemia.
Signs and symptoms of CHAPLE disease were promptly and reproducibly reversed with pozelimab treatment in all patients. All patients experienced improvement in or no worsening of the prespecified clinical outcomes (frequency of problematic abdominal pain, bowel movement frequency, facial edema severity, and peripheral edema severity) at week 24. Additionally, after 24 weeks of pozelimab treatment, all patients experienced a complete resolution of their core signs and symptoms of CHAPLE disease, including their most bothersome symptom, which had a substantial impact on how the participants felt and functioned.
Pozelimab was generally well tolerated in this study, with few treatment-related side effects, none of which indicated worsening of CHAPLE disease. There were no deaths, no meningococcal infections, and no thrombotic or embolic events. The totality of efficacy and safety data demonstrate an overall encouraging benefit–risk profile.
The design of the current study surmounted multiple challenges in measuring the clinical effects of a therapeutic in a recently identified (1), ultra-rare disorder. The sample size is appropriate for a disease with a worldwide prevalence of <100 patients. Given the rarity of CHAPLE disease in outbred populations, we identified patients from previously identified areas with high consanguinity (6). Importantly, albeit in a very small dataset, we did not observe a difference in response based on genotype, region, age, or disease duration. The rationale for an open-label, single-arm design was that the disease is too severe for patients to be allocated to placebo. Moreover, at the time of study design, eculizumab had not been rigorously studied to be suitable as an active comparator, with its effects described in case series and not yet in a clinical trial. Furthermore, because each patient served as their own control, we utilized a historical control period of at least two years prior to enrollment in the study, including inspection of full records of clinical documentation, to ensure sufficient characterization of the pre-treatment clinical spectrum of disease. This allowed us to select objective endpoints for efficacy measurements, such as hospitalizations and albumin transfusions. Additionally, some of the patient/caregiver- and physician-reported outcome measures used to capture clinical improvement in this study were implemented without prior experience in this disease. It was considered impractical to develop such experience in the context of an ultra-rare disorder, as a validation study would exhaust the potential pool of study patients and potentially delay access to treatment. Therefore, to evaluate the clinical outcome assessment strategy and incorporate the patient perspective, we conducted semi-structured interviews at screening and week 24. The interviews elicited the core signs and symptoms of disease, as well as the most bothersome symptom, and assessment of pozelimab treatment on patients’ signs/symptoms of disease and quality of life. The strategy was to pursue a ‘totality of the data’ approach, with an emphasis on objective endpoints using pre-enrollment data as controls supplemented by qualitative data obtained during the interviews to capture the patient experience.
The apparent disease mechanism is that lack of functioning CD55 on GI lymphatic endothelial cell membranes contributes to constitutive production of MACs, leading to loss of membrane integrity and cellular dysfunction, lymphangiectasia, intestinal inflammation, and lymph-wasting GI disease (19, 20). Furthermore, CD59, a membrane-bound inhibitor that functions downstream of CD55 in the complement activation cascade, is not expressed in lymphatic endothelial cells (21). We hypothesize that the manifestations of CHAPLE disease, a genetic form of primary intestinal lymphangiectasia, may be due to loss of function in CD55 and the absence of complement regulation by CD59 in GI lymphatics. It appears that lymphatic endothelial cells are either not destroyed by MAC or can be regenerated by repair processes, since PLE rapidly and completely resolved upon complement C5 inhibition. Considering the genetic cause of the disease, pharmacotherapy cannot cure it. Whether treatment throughout adulthood will be necessary remains uncertain.
This study confirms and extends the current information on the natural history of CHAPLE disease (1, 3, 4, 6, 8, 9, 18). Historical and baseline data demonstrate the clinical and laboratory manifestations of severe enteric protein loss in children, and the conduct of within-trial semi-structured interviews was used to confirm the appropriateness of the clinical outcome assessment strategy. For children suffering from CHAPLE disease, pozelimab treatment achieves a clear, dramatic, and sustained improvement in disease parameters. Compared to the eculizumab case series (6), our study presents more rigorous evidence for safety and efficacy of pozelimab in CHAPLE disease.
Importantly, this is an interim report of an ongoing study. Additional two- and three-year treatment data will provide further information on efficacy, puberty, persistence of growth, and safety.
In conclusion, we demonstrate that single-agent, targeted therapy with pozelimab inhibits complement overactivation and reverses the manifestations of CHAPLE disease. Pozelimab addresses an unmet medical need, and is the only currently-approved therapeutic, for patients with CHAPLE disease, a life-threatening, ultra-rare condition.
Supplementary Material
RESEARCH IN CONTEXT.
Evidence before the study
CD55 deficiency with hyperactivation of complement, angiopathic thrombosis, and protein-losing enteropathy (CHAPLE) disease is a life-threatening, ultra-rare autosomal recessive disorder caused by loss-of-function variants of the CD55 gene. CHAPLE disease is apparent in infancy and early childhood and is associated with severe morbidity and mortality. Patients present with hypoalbuminemia and edema, severe and debilitating gastrointestinal symptoms, and chronic malabsorption. CHAPLE disease may progress to gastrointestinal obstruction and perforation as well as thromboembolic events, which can be fatal.
Given the etiology of CHAPLE disease, anti-complement therapy has been proposed as a treatment. Off-label use of eculizumab, a humanized monoclonal anti-C5 antibody, improved the clinical and laboratory features of CHAPLE disease. Eculizumab requires intravenous infusion every two weeks. Therefore, a rigorous study of a subcutaneously administered anti-C5 therapy in CHAPLE disease is warranted.
Added value of this study
Pozelimab is a subcutaneously administered, investigational monoclonal antibody directed against the terminal complement protein C5. We conducted an open-label, historically-controlled study to assess the efficacy and safety of pozelimab in patients with CHAPLE disease. Following pozelimab treatment, we observed complement inhibition and improvements in overall health. Patients experienced a complete and sustained resolution in signs and symptoms of CHAPLE disease. Furthermore, patients exhibited rapid catch-up growth as well as substantial improvements in disease morbidity, including reduced hospitalization and use of corticosteroids. Serum albumin levels normalized and the need for albumin transfusions was eliminated.
Implications of all the available evidence
This study demonstrates that single-agent, targeted therapy with pozelimab inhibits complement overactivation and reverses the manifestations of CHAPLE disease. Pozelimab treatment addresses an unmet medical need for patients with CHAPLE disease, a life-threatening, ultra-rare condition. Subcutaneous administration potentially allows greater access to treatment for this severe illness in low-resource settings. Importantly, pozelimab was recently approved by the U.S Food and Drug Administration as the first treatment for adult and pediatric patients 1 year of age and older with CD55-deficient protein-losing enteropathy (PLE), also known as CHAPLE disease, based on the findings presented in this report.
ACKNOWLEGEMENTS
This study was supported by Regeneron Pharmaceuticals, Inc. This work was supported by the Division of Intramural Research, National Institute of Allergy and Infectious Diseases, National Institutes of Health.
Funding:
Regeneron Pharmaceuticals, Inc. and the Division of Intramural Research, National Institute of Allergy and Infectious Diseases, National Institutes of Health.
Footnotes
DECLARATION OF INTERESTS
AO is a consultant and steering committee member for Regeneron, Pharmaceuticals, Inc.; received sample analysis support for a prior collaborative study (https://doi.org/10.1038/s41590-020-00830-z) from Regeneron, Pharmaceuticals, Inc.; and has a pending patent on C5 inhibitor treatment in CHAPLE disease. VC, NS, and KS received support to conduct the study and provision of the investigational product from Regeneron Pharmaceuticals, Inc. APS, BK, SB, EK-A, RE, and H Moorman have nothing to disclose. JJJ, TB, and LP are Regeneron Pharmaceuticals, Inc. employees/stockholders. KAM is a Regeneron Pharmaceuticals, Inc. employee/stockholder and has both pending and issued patents with Regeneron Pharmaceuticals, Inc. HBF is a consultant and advisory board member for Regeneron, Pharmaceuticals, Inc. IJF is an associate on a cooperative research and development agreement (CRADA) between Merck Pharmaceuticals and the NIH. OAH and GDY are Regeneron Pharmaceuticals, Inc. employees/stockholders and have a pending patent on C5 inhibitor treatment in CHAPLE disease. MJL received support for a federally-approved CRADA to support the clinical trial and has a pending patent on C5 inhibitor treatment in CHAPLE disease.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
DATA SHARING
Qualified researchers may request access to study documents (including the clinical study report, study protocol with any amendments, blank case report form, and statistical analysis plan) that support the methods and findings reported in this manuscript. Individual anonymized participant data will be considered for sharing once the product and indication has been approved by major health authorities (e.g., U.S. Food and Drug Administration, European Medicines Agency, Pharmaceuticals and Medical Devices Agency, etc.), if there is legal authority to share the data and there is not a reasonable likelihood of participant re-identification. Requests should be submitted to https://vivli.org/.
REFERENCES
- 1.Ozen A, Comrie WA, Ardy RC, Dominguez Conde C, Dalgic B, Beser OF, et al. CD55 Deficiency, Early-Onset Protein-Losing Enteropathy, and Thrombosis. N Engl J Med. 2017;377(1):52–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lublin DM, Atkinson JP. Decay-accelerating factor: biochemistry, molecular biology, and function. Annu Rev Immunol. 1989;7:35–58. [DOI] [PubMed] [Google Scholar]
- 3.Kurolap A, Eshach-Adiv O, Hershkovitz T, Paperna T, Mory A, Oz-Levi D, et al. Loss of CD55 in Eculizumab-Responsive Protein-Losing Enteropathy. N Engl J Med. 2017;377(1):87–9. [DOI] [PubMed] [Google Scholar]
- 4.Hagin D, Lahav D, Freund T, Shamai S, Brazowski E, Fishman S, et al. Eculizumab-Responsive Adult Onset Protein Losing Enteropathy, Caused by Germline CD55-Deficiency and Complicated by Aggressive Angiosarcoma. J Clin Immunol. 2021;41(2):477–81. [DOI] [PubMed] [Google Scholar]
- 5.Soliris. Soliris [package insert]. Boston, MA: Alexion Pharmaceuticals, Inc.; 2020. 2020. [Google Scholar]
- 6.Ozen A, Kasap N, Vujkovic-Cvijin I, Apps R, Cheung F, Karakoc-Aydiner E, et al. Broadly effective metabolic and immune recovery with C5 inhibition in CHAPLE disease. Nat Immunol. 2021;22(2):128–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Shani M, Theodor E, Frand M, Goldman B. A family with protein-losing enteropathy. Gastroenterology. 1974;66(3):433–45. [PubMed] [Google Scholar]
- 8.Chongsrisawat V, Suratannon N, Chatchatee P, Ittiwut R, Ittiwut C, Weerapakorn W, et al. Novel CD55 Mutation Associated With Severe Small Bowel Ulceration Mimicking Inflammatory Bowel Disease in a Pair of Siblings. Inflamm Bowel Dis. 2022;28(9):1458–61. [DOI] [PubMed] [Google Scholar]
- 9.Kurolap A, Eshach Adiv O, Hershkovitz T, Tabib A, Karbian N, Paperna T, et al. Eculizumab Is Safe and Effective as a Long-term Treatment for Protein-losing Enteropathy Due to CD55 Deficiency. J Pediatr Gastroenterol Nutr 2019;68(3):325–33. [DOI] [PubMed] [Google Scholar]
- 10.Kaya Z, Derinöz O. Polycythemia as an Uncommon Finding in 2 Children, One With Systemic Capillary Leak Syndrome and the Other With Protein-losing Enteropathy Caused by CD55 Deficiency. Journal of Pediatric Hematology/Oncology. 9900: 10.1097/MPH.0000000000002729. [DOI] [PubMed] [Google Scholar]
- 11.Hagin D, Lahav D, Freund T, Shamai S, Brazowski E, Fishman S, et al. Eculizumab-Responsive Adult Onset Protein Losing Enteropathy, Caused by Germline CD55-Deficiency and Complicated by Aggressive Angiosarcoma. Journal of Clinical Immunology. 2021;41(2):477–81. [DOI] [PubMed] [Google Scholar]
- 12.U.S. Food and Drug Administration. FDA Patient-Focused Drug Development Guidance Series for Enhancing the Incorporation of the Patient’s Voice in Medical Product Development and Regulatory Decision Making 2022. [Available from: https://www.fda.gov/drugs/development-approval-process-drugs/fda-patient-focused-drug-development-guidance-series-enhancing-incorporation-patients-voice-medical.
- 13.Latuszek A, Liu Y, Olsen O, Foster R, Cao M, Lovric I, et al. Inhibition of complement pathway activation with Pozelimab, a fully human antibody to complement component C5. PLoS One. 2020;15(5):e0231892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.FDA approves first treatment for CD55-deficient protein-losing enteropathy (CHAPLE disease) [press release]. U.S. Food and Drug Administration, August 18, 2023. [Google Scholar]
- 15.Landrum MJ, Lee JM, Benson M, Brown GR, Chao C, Chitipiralla S, et al. ClinVar: improving access to variant interpretations and supporting evidence. Nucleic Acids Res. 2018;46(D1):D1062–D7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Costabile M Measuring the 50% haemolytic complement (CH50) activity of serum. J Vis Exp. 2010(37). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bhakdi S, Muhly M. A simple immunoradiometric assay for the terminal SC5b-9 complex of human complement. J Immunol Methods. 1983;57(1–3):283–9. [DOI] [PubMed] [Google Scholar]
- 18.Ozen A CHAPLE syndrome uncovers the primary role of complement in a familial form of Waldmann’s disease. Immunol Rev. 2019;287(1):20–32. [DOI] [PubMed] [Google Scholar]
- 19.Yu CY, Ardoin SP. Complement inhibitor for therapy of CHAPLE. Nat Immunol. 2021;22(2):106–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ozen A, Lenardo MJ. Protein-Losing Enteropathy. New England Journal of Medicine. 2023;389(8):733–48. [DOI] [PubMed] [Google Scholar]
- 21.Park SM, Angel CE, McIntosh JD, Mansell CM, Chen C-JJ, Cebon J, et al. Mapping the Distinctive Populations of Lymphatic Endothelial Cells in Different Zones of Human Lymph Nodes. PLOS ONE. 2014;9(4):e94781. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Qualified researchers may request access to study documents (including the clinical study report, study protocol with any amendments, blank case report form, and statistical analysis plan) that support the methods and findings reported in this manuscript. Individual anonymized participant data will be considered for sharing once the product and indication has been approved by major health authorities (e.g., U.S. Food and Drug Administration, European Medicines Agency, Pharmaceuticals and Medical Devices Agency, etc.), if there is legal authority to share the data and there is not a reasonable likelihood of participant re-identification. Requests should be submitted to https://vivli.org/.


