Abstract
We evaluated the diagnostic performance of both gadolinium-ethoxybenzyl-diethylenetriamine pentaacetic acid-enhanced magnetic resonance imaging (Gd-EOB-DTPA-MRI) and contrast-enhanced ultrasound (CEUS) for focal liver lesions, especially for the detection of small (<2 cm) intrahepatic lesions. We retrospectively reviewed patients who underwent Gd-EOB-DTPA-MRI and CEUS before liver resection and compared Gd-EOB-DTPA-MRI and CEUS in the detection of focal liver lesions. A total of 216 patients were included, and 309 lesions were found. The sensitivity values of MRI and CEUS for the main lesion were both more than 95%, and the coincidence rates were both more than 80%. Regarding lesions <2 cm, 135 such lesions were detected by MRI, whereas only 85 were detected by CEUS. For lesions <2 cm, the sensitivity, specificity, and coincidence rates of MRI were significantly better than those of CEUS. Among 27 patients, 50 more lesions were detected by MRI than CEUS, 56% (28/50) of which were malignant. For the large lesion, the diagnostic performance is similar between Gd-EOB-DTPA-MRI and CEUS, and the sensitivity and coincidence rates of both methods are high. Gd-EOB-DTPA-MRI is likely to detect small (<2 cm) focal intrahepatic lesions.
Keywords: contrast-enhanced ultrasound, gadolinium, liver lesions, magnetic resonance imaging
1. Introduction
Liver cancer is the third leading cause of death in the world and the fifth leading cause of death in China.[1] The liver is the first metastatic site of most malignant tumors.[2] Hepatocellular carcinoma (HCC) is a malignant tumor that can be clearly diagnosed through imaging examination without biopsy.[3] As a hepatobiliary-specific contrast enhancer, gadolinium-ethoxybenzyl-diethylenetriamine pentaacetic acid (Gd-EOB-DTPA) has the characteristics of not only an extracellular contrast agent (dynamic phase) but also a hepatocellular-specific contrast agent (static phase). Gd-EOB-DTPA is excreted out of the body through the kidney and biliary system, which is conducive to the detection and characterization of lesions.[4] Gd-EOB-DTPA-enhanced magnetic resonance imaging (Gd-EOB-DTPA-MRI), which is recommended by the American Society for the Study of Liver Diseases as a first-line detection method for diagnosis of HCC,[3] has good performance in the detection and qualitative analysis of focal liver lesions.[5–7]
Contrast-enhanced ultrasound (CEUS) is a useful test for focal liver lesions based on the assessment of hemodynamic changes. Studies have shown that CEUS is highly sensitive to arterial hypervascular lesions,[8] and the specificity of CEUS for nodules with hypervascular lesions is higher than that reported in computed tomography (CT) or MRI studies, especially for small (<2 cm) HCC lesions.[9] CEUS is inherently more sensitive to microbubbles than CT or MRI are to iodization or gadolinium contrast agents.[10] In 2018, major guideline updates were presented by the European Association for the Study of the Liver (EASL) as well as the Korean Association for Liver Cancer and the Korean National Cancer Center (KLCA-NCC). Hepatobiliary contrast agent-enhanced MRI is now included as a first-line diagnostic method in these new guidelines, and CEUS is included as a second-line diagnostic method. Therefore, hepatobiliary contrast agent-enhanced MRI and CEUS will be increasingly used for noninvasive diagnosis and staging of liver cancer.[11]
At present, there are many comparative studies on the sensitivity and specificity of MRI and CEUS for main lesions in the liver but few studies exist on the detection performance of small intrahepatic lesions. The preoperative identification of the presence and absence of metastatic liver lesions in liver cancer patients as well as the determination of the number of metastatic lesions is significantly related to the determination of the Barcelona Clinic Liver Cancer (BCLC) staging of patients with liver cancer in the formulation of surgical procedures and the prognosis of patients.[2,12] Therefore, we conducted this single-center retrospective study to compare and analyze the detection performance of Gd-EOB-DTPA-MRI and CEUS for small intrahepatic lesions, hoping to provide a basis for preoperative detection and postoperative monitoring of liver cancer patients.
2. Patients and Methods
2.1. Study design
This study was approved by the ethics committee of the First Affiliated Hospital of Guangxi Medical University. Informed consent was waived for this retrospective study. The diagnostic performance of each of the 2 imaging methods for the main lesion of the patients was analyzed. In patients with a single focal liver lesion, the sole lesion was the main lesion. In patients with more than 1 focal liver lesion, the largest lesion was identified as the main lesion. The detection and diagnostic performance of each of the 2 methods were analyzed for all focal liver lesions of the patients. The focal liver lesion nature of all patients was confirmed by postoperative pathology.
2.2. Patients
Patients admitted to the First Affiliated Hospital of Guangxi Medical University for liver resection between January 2016 and June 2019 were considered for enrollment. A total of 216 patients were enrolled, including 174 males and 42 females, aged 17 to 88 years old, with an average age of 52 (51 ± 12) years old (Table 1). The average diameter of lesions was 5.3 cm (ranged from 0.7 to 19.5 cm). The mean time interval between MRI and CEUS in the same patient was 5.57 days (ranged from 0 to 20 20 days); CEUS was performed first in 135 patients. The mean time interval between MRI and CEUS was 6.18 days (ranged from 0 to 20 20 days) in 28 patients with different numbers of lesions; CEUS was performed first in 18 patients.
Table 1.
Patient characteristics.
| Parameter | Number |
|---|---|
| Patients | 216 |
| Lesions | 309 |
| Mean age (range) | 52 yr (17–88 yr) |
| Sex (male/female) | 174 (80.6%)/42 (19.4%) |
| Diameter of lesions (range) | 5.3 cm (0.7–19.5 cm) |
| Liver cirrhosis | 182 (84.3%) |
2.3. Inclusion and exclusion criteria
Our inclusion criteria encompassed patients who had both Gd-EOB-DTPA-MRI and CEUS prior to liver resection. The resected liver specimen had to undergo pathological examination. Patients who had an interval of imaging between Gd-EOB-DTPA-MRI and CEUS of more than 3 weeks were excluded. We also excluded cases where the exact number and size of liver lesions in the resected specimen were not reported.
2.4. MRI
A 3.0T MRI system (Siemens Company, Erlangen, Germany) was utilized for the MRI. Gd-EOB-DTPA packaged at 10 mL/bottle with a concentration of 0.25 mol/L was purchased (Bayer Schering Pharma AG, Berlin, Germany).
The inspection method was as follows. Before examination, each patient was trained to breathe evenly and hold their breath properly during various parts of their scan so as to ensure the image quality and similarity of the scan layer. The procedure was performed using a breath gating technique. The scan covered the entire liver from the top of the diaphragm to the lower pole of both kidneys. A routine plain scan and a Gd-EOB-DTPA-enhanced scan were performed in all patients, followed by T2WI, T1-vibe with reverse phase scan, DWI scan, and T1-vibe lipid-suppressing plain scan. Then, on the basis of T1-vibe lipid-suppression scanning, the 3 stages of Gd-EOB-DTPA (hepatic artery, portal vein, and equilibrium stage) were enhanced dynamically, and the delayed liver parenchyma was scanned.
Gd-EOB-DTPA-enhanced scanning was carried out as follows. Gd-EOB-DTPA was injected by peripheral venous mass injection at a dose of 0.025 mmol/kg at an injection rate of 3 mL/s, followed by immediate injection of 20 mL normal saline at the same rate to rinse the catheter. After injection of Gd-EOB-DTPA, a 3D VIBE scan was performed in the arterial stage (18–23 seconds), portal vein stage (45–50 seconds), and equilibrium stage (120 seconds). Then, axial delayed scanning was performed for 5, 10, 20, and 30 minutes, and oblique coronal scanning was added for 20 and 30 minutes.
2.5. CEUS
A LOGIQ E9 color ultrasound diagnostic instrument (General Electric Company, Milwaukee, Wis, USA) was used in CEUS. Sulphur Hexafluoride Microbubble (Bracco Suisse SA, Plan-Les-Ouates, Switzerland) packaged at 59 mg/bottle with a concentration of 8 mul/mL was purchased.
The inspection method was carried out in the following sequence: a routine ultrasound examination was conducted; the imaging examination mode was entered, and the imaging condition was adjusted; the probe section was placed in the area of interest, and the target lesion was as close as possible to the middle of the image. Contrast agent was injected through the anterior cubital vein mass (see general discussion). The conventional recommended dosage of sonnovi® was 2.4 mL, but it was increased to 4.8 mL for patients with obesity, severe cirrhosis, or fatty liver. Meanwhile, the time recorder was opened to observe the enhancement of the lesion and surrounding liver tissue and its dynamic change process for approximately 4–6 minutes. The storage function was activated in the angiography, and dynamic images were stored according to the predetermined scheme corresponding to the examination purpose.
2.6. Image analysis and diagnostic criteria
All images were viewed by an imaging specialist with more than 10 years of experience. The diagnosis of the focal liver lesion was made with CEUS according to the standard protocols[3,13,14] and with Gd-EOB-DTPA-MRI according to published guidelines.[3,15] More details are provided in Tables S1 and S2, Supplemental Digital Contents 1 and 2, http://links.lww.com/MD/H247.
2.7. Stage A in the BCLC staging system
BCLC A subclassification was divided into 4 stages, and their status score was 0. There were only 1 tumor in A1, A2, and A3, and there were 3 tumors in A4; all of which were <3 cm in diameter in A4. The bilirubin of A1 was normal without portal hypertension. The bilirubin of A2 was normal with portal hypertension. Abnormal bilirubin was in A3 with portal hypertension. The Child-Pugh score of A4 was less than or equal to 7.
2.8. Indications and procedures of surgery
Indications for partial hepatectomy are as follows: a WHO performance status of less than or equal to 1; The tumor can be resected; Residual liver volume was > 30% of total liver volume in noncirrhotic patients and > 50% in cirrhotic patients; Child-Pugh score is less than or equal to 7; No distant metastasis. The specific type of hepatectomy is determined by tumor location, degree of cirrhosis, appropriate surgical margin, and residual liver volume. Histopathological studies of resected specimens were performed independently by 3 pathologists, who discussed and reached consensus if there was any dispute.
2.9. Pathological examination
The pathological examination of the excised liver specimen was completed by 3 pathologists independently. If there was any dispute, a consensus would be reached after discussion among the 3 experts. Specific pathological diagnosis of tissue sampling, diagnostic process and standard basis referred to the literature.[16]
2.10. Statistical analysis
First, the lesion assessment was classified as benign, malignant, or indeterminate. Second, the lesion assessment was classified as HCC, non-HCC, or uncertain. The inconclusive classification was defined as a misclassification. In the first case, malignant was defined as positive and benign as negative. In the second case, HCC was defined as positive and non-HCC as negative. The coincidence rate was calculated as the sum of true negative and true positive diagnoses divided by the number of lesions with clear pathological diagnosis. Sensitivity was calculated as the number of lesions that were positive in both tests and pathological diagnosis divided by the number of positive lesions according to the pathological diagnosis. The specificity was calculated as the number of lesions that were negative in both tests and pathological diagnosis divided by the number of lesions that were negative according to the pathological diagnosis. The positive predictive value was calculated as the number of true positive diagnoses divided by the total number of positive diagnoses, and the negative predictive value was calculated as the number of true negative diagnoses divided by the total number of negative diagnoses. The chi-square test was used to compare the rates between the 2 groups. P < .05 was statistically significant. The software R 3.4.0 with RStudio 1.0.136 was used for statistical analysis.
3. Results
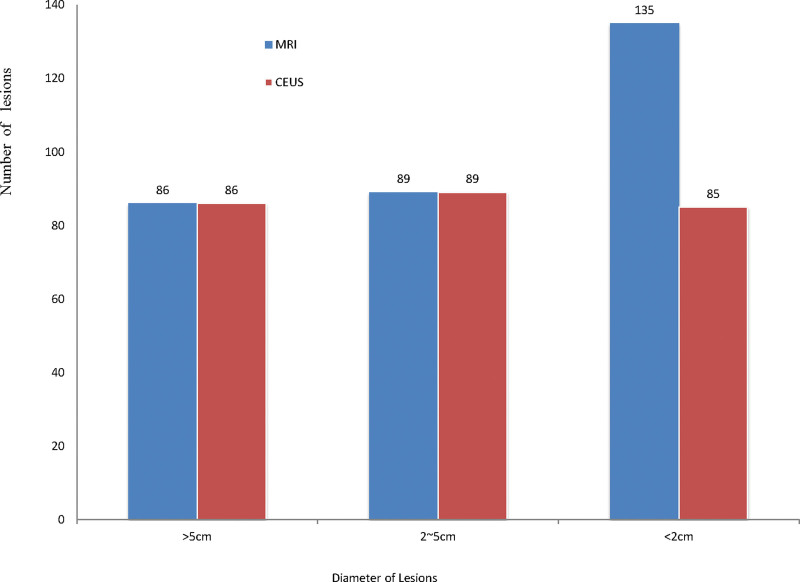
In this study, 80% of patients and nearly 80% of lesions were HCC, whereas 28 patients and 52 lesions were benign, as shown in Table 2. Eleven patients had an indeterminate diagnosis with MRI, and 9 patients had an indeterminate diagnosis with CEUS. Two patients had an indeterminate diagnosis with both MRI and CEUS. The diagnostic performance of MRI and CEUS for benign and malignant lesions was similar to that for HCC and non-HCC. The sensitivity, specificity, and coincidence rates of MRI and CEUS for the main lesion and lesions larger than 2 cm were similar, but those of MRI were significantly better than those of CEUS for lesions smaller than 2 cm among all intrahepatic lesions (P < .01), as shown in Tables 3 and 4. MRI detected 309 lesions, and CEUS detected 259 lesions. Both MRI and CEUS could identify lesions larger than 2 cm. In terms of the detection of lesions smaller than 2 cm, MRI found 135, whereas CEUS only found 85, as shown in Figure 1. MRI detected 50 more lesions than CEUS among the 27 patients. In 21 of these patients, CEUS only found 1 lesion, whereas MRI detected multiple lesions, as shown in Table 5 and Figure 2. Twenty-two of the 50 lesions were benign, and 28 were malignant. Among the 27 patients, 21 patients were patients with HCC, and the preoperative BCLC staging of 14 patients were different according to the 2 different methods, as shown in Table 6.
Table 2.
Postoperative pathology.
| Pathology results | Patients | Lesions |
|---|---|---|
| HCC | 174 | 239 |
| ICC | 11 | 15 |
| Hepatic metastasis | 2 | 2 |
| Sarcoma | 1 | 1 |
| Hemangioma | 19 | 33 |
| FNH | 4 | 5 |
| Inflammatory pseudotumor | 4 | 6 |
| Dysplastic nodule | 1 | 8 |
FNH = focal nodular hyperplasia, HCC = hepatocellular carcinoma, ICC = intrahepatic cholangiocarcinoma.
Table 3.
Comparison of diagnostic performance between MRI and CEUS.
| Lesions | Methods | Sensitivity (95% CI) | Specificity (95% CI) | AUC (95% CI) | PPV (95% CI) | NPV (95% CI) | CR (%) |
|---|---|---|---|---|---|---|---|
| Benign or malignant† | |||||||
| The main lesion (n = 216) | MRI | 96.28 (92.48–98.49) | 57.14 (37.18–75.54) | 0.77 (0.71–0.82) | 93.78 (90.76–95.86) | 69.57 (50.81–83.49) | 91.20 |
| CEUS | 97.87 (94.64–99.42) | 53.57 (33.87–72.49) | 0.76 (0.69–0.81) | 93.40 (90.48–95.47) | 78.95 (57.27–91.30) | 92.13 | |
| MRI/CEUS‡ | 94.68 (90.44–97.42) | 64.29 (44.07–81.36) | 0.79 (0.74–0.85) | 94.68 (91.84–96.69) | 64.29 (48.11–7.75) | 90.74 | |
| Main lesion > 2 cm (n = 164) | MRI | 95.97 (91.44–98.51) | 66.67 (38.38–88.18) | 0.81 (0.75–0.87) | 96.62 (93.32–98.32) | 62.50 (41.32–79.78) | 97.45 |
| CEUS | 97.98 (94.23–99.58) | 60 (32.29–83.66) | 0.79 (0.72–0.85) | 96.05 (92.9–97.83) | 75 (47.62–90.83) | 94.51 | |
| Main lesion < 2 cm (n = 52) | MRI | 97.44 (85.82–99.94) | 46.15 (19.22–74.86) | 0.72 (0.58–0.83) | 84.44 (76.59–90.0) | 85.71 (44.27–97.84) | 84.62 |
| CEUS | 97.44 (85.82–99.94) | 46.15 (19.22–74.86) | 0.72 (0.58–0.83) | 84.44 (76.59–90.0) | 85.71 (44.27–97.84) | 84.62 | |
| All lesions (n = 309) | MRI | 95.33 (91.98–97.56) | 55.77 (41.33–69.53) | 0.76 (0.71–0.80) | 91.42 (88.69–93.54) | 70.73 (56.94–81.54) | 88.67 |
| CEUS | 87.55 (82.88–91.32) | 34.62 (21.97–49.09) | 0.61 (0.55–0.67) | 86.87 (84.38–89.02) | 36.0 (25.54–47.98) | 78.64 | |
| Lesion > 2 cm (n = 174) | MRI | 96.23 (91.97–98.60) | 66.67 (38.38–88.18) | 0.81 (0.75–0.87) | 96.84 (93.73–98.73) | 62.50 (41.29–79.79) | 93.68 |
| CEUS | 98.11 (94.59–99.61) | 60.0 (32.29–83.66) | 0.79 (0.72–0.85) | 96.29 (93.33–97.97) | 75.0 (47.60–90.83) | 94.83 | |
| Lesion < 2 cm (n = 135) | MRI* | 93.88 (87.15–97.72) | 51.35 (34.40–68.08) | 0.73 (0.64–0.78) | 83.64 (78.53–87.72) | 76.0 (57.84–87.96) | 82.22 |
| CEUS | 70.41 (60.34–79.21) | 24.32 (11.77–41.20) | 0.47 (0.39–0.56) | 71.13 (66.34–75.49) | 23.68 (14.0–37.17) | 57.78 | |
AUC = area under the curve, CEUS = contrast-enhanced ultrasound, CI = confidence interval, CR = coincidence rate, MRI = magnetic resonance imaging, NPV = negative predictive value, PPV = positive predictive value.
The lesion assessment was classified as benign, malignant, or indeterminate, and malignancy was identified as positive and benign as negative. The main lesion: in patients with a single lesion, the sole lesion was the main lesion, and in patients with more than 1 intrahepatic lesion, the largest lesion was identified as the main lesion.
As long as 1 of the 2 diagnoses of MRI and CEUS was negative, the result was considered negative.
P < .01.
Table 4.
Comparison of diagnostic performance between MRI and CEUS.
| Lesions | Methods | Sensitivity (95% CI) | Specificity (95% CI) | AUC (95% CI) | PPV (95% CI) | NPV (95% CI) | CR (%) |
|---|---|---|---|---|---|---|---|
| HCC or non-HCC† | |||||||
| The main lesion (n = 216) | MRI | 95.98 (91.88–98.37) | 54.76 (38.67–70.15) | 0.75 (0.69–0.81) | 89.78 (86.93–92.47) | 76.67 (60.19–87.71) | 87.96 |
| CEUS | 97.70 (94.22–99.37) | 59.52 (43.28–74.37) | 0.79 (0.73–0.84) | 90.91 (87.38–93.52) | 86.21 (69.69–94.44) | 90.27 | |
| MRI/CEUS‡ | 94.25 (89.69–97.21) | 64.29 (48.03–78.45) | 0.79 (0.73–0.85) | 91.62 (87.92–94.26) | 72.97 (58.68–83.69) | 88.43 | |
| Main lesion > 2 cm (n = 164) | MRI | 95.59 (90.65–98.36) | 60.71 (40.58–78.49) | 0.78 (0.71–0.84) | 92.19 (88.16–94.94) | 73.91 (55.09–86.75) | 89.63 |
| CEUS | 97.79 (93.69–99.54) | 64.29 (44.07–81.36) | 0.81 (.074–0.87) | 93.01 (88.99–95.63) | 85.71 (65.46–95.0) | 92.70 | |
| Main lesion < 2 cm (n = 52) | MRI | 97.37 (86.19–99.93) | 42.86 (17.66–71.14) | 0.70 (0.56–0.82) | 82.22 (74.55–87.96) | 85.71 (44.46–97.85) | 82.69 |
| CEUS | 97.37 (86.19–99.93) | 50.0 (23.04–76.96) | 0.74 (0.59–0.85) | 74.09 (75.74–89.95) | 87.5 (48.56–98.11) | 84.62 | |
| All lesions (n = 309) | MRI | 94.12 (90.38–96.76) | 52.86 (40.55–6491) | 0.74 (0.68–0.78) | 87.21 (84.15–89.75) | 72.55 (60.29–82.15) | 84.79 |
| CEUS | 86.19 (81.16–90.30) | 40.0 (28.47–52.41) | 0.63 (0.57–0.69) | 83.07 (80.09–85.67) | 45.90 (35.63–56.54) | 75.73 | |
| Lesion > 2 cm (n = 174) | MRI | 95.17 (90.01–98.04) | 58.62 (38.94–76.48) | 0.77 (0.69–0.83) | 92.0 (88.16–94.67) | 70.83 (52.56–84.48) | 89.08 |
| CEUS | 97.93 (94.07–99.57) | 62.07 (42.26–79.31) | 0.80 (0.73–0.86) | 92.81 (89.01–95.37) | 85.71 (65.39–95.01) | 91.95 | |
| Lesion < 2 cm (n = 135) | MRI* | 92.55 (85.26–96.95) | 48.78 (32.88–64.87) | 0.71 (0.62–0.78) | 80.56 (75.35–84.88) | 74.07 (56.74–86.16) | 79.26 |
| CEUS | 67.02 (56.56–76.38) | 24.39 (12.36–40.31) | 0.46 (0.37–0.55) | 67.02 (61.89–71.78) | 24.39 (14.89–37.28) | 54.70 | |
AUC = area under the curve, CEUS = contrast-enhanced ultrasound, CI = confidence interval, CR = coincidence rate, MRI = magnetic resonance imaging, NPV = negative predictive value, PPV = positive predictive value.
The lesion assessment was classified as HCC, non-HCC, or uncertain, and HCC was identified as positive and non-HCC as negative. The main lesion: in patients with a single lesion, the sole lesion was the main lesion. In patients with more than 1 intrahepatic lesion, the largest lesion was identified as the main lesion.
As long as 1 of the 2 diagnoses of MRI and CEUS was negative, the result was considered negative.
P < .01.
Figure 1.
The 2 methods detected the distribution of lesions with different diameters in 216 patients. For lesions <2 cm, MRI detected significantly more lesions than CEUS (P < .001). CEUS = contrast-enhanced ultrasound, MRI = magnetic resonance imaging.
Table 5.
Distribution of different numbers of intrahepatic lesions.
| MRI | CEUS | Total | |||
|---|---|---|---|---|---|
| 1* | 2 | 3 | 4 | ||
| 1 | 160† | 160 | |||
| 2 | 11 | 26 | 37 | ||
| 3 | 5 | 4 | 2 | 11 | |
| 4 | 1 | 1 | 1 | 3 | |
| 5 | 1 | 1 | 1 | 3 | |
| 6 | 2 | 2 | |||
| Total | 180 | 31 | 3 | 2 | 216 |
CEUS = contrast-enhanced ultrasound, MRI = magnetic resonance imaging.
The number of lesions detected by MRI or CEUS in a patient.
The number of patients with the same number of lesions detected by MRI and CEUS.
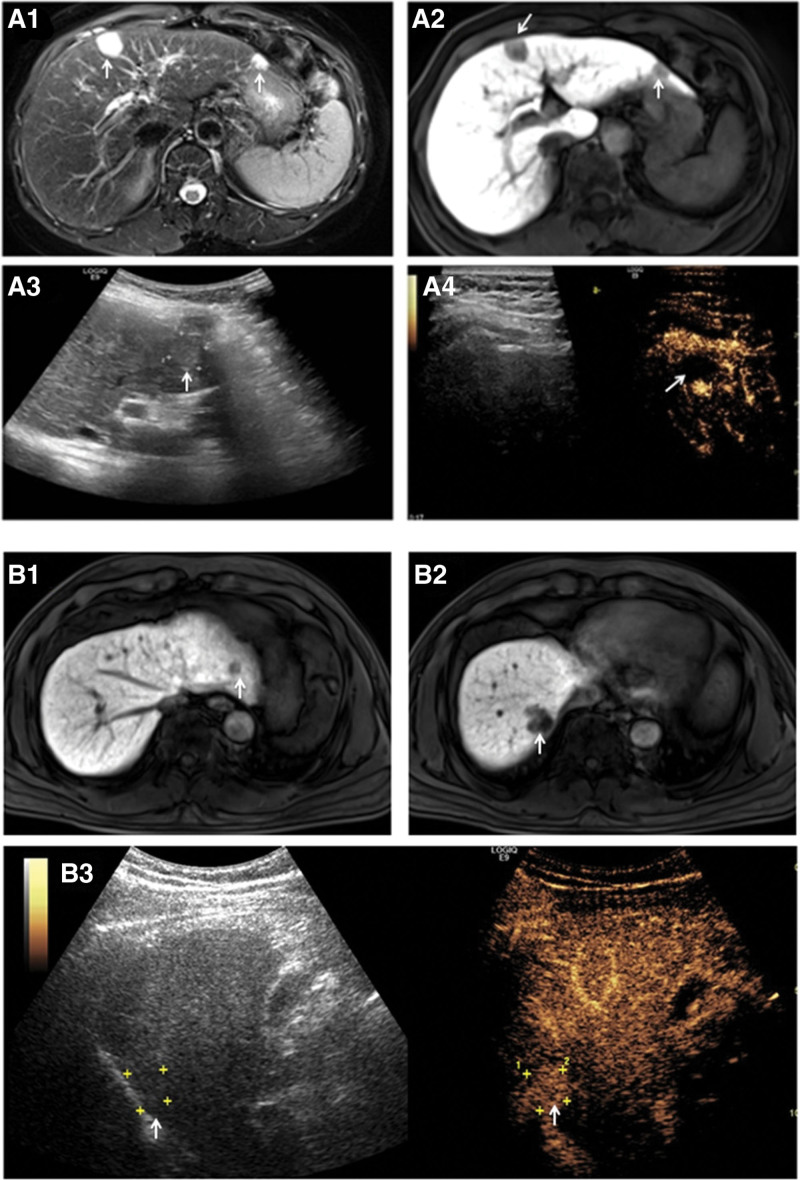
Figure 2.
Two methods identified different numbers of lesions in the same patient. The patient in image A had hemangioma. The patient in image B had hepatocellular carcinoma. For these patients, CEUS detected only 1 lesion, whereas MRI detected 2 lesions. CEUS was performed 2 days before MRI in the patient with hemangioma and 1 day after MRI in the patient with hepatocellular carcinoma. CEUS = contrast-enhanced ultrasound, MRI = magnetic resonance imaging.
Table 6.
Preoperative BCLC staging in 21 patients.
| Methods | BCLC staging | |||||
|---|---|---|---|---|---|---|
| A1 | A2 | A3 | A4 | B | C | |
| CEUS | 7 | 3 | 2 | 2 | 5 | 2 |
| MRI | 0 | 0 | 0 | 6 | 13 | 2 |
BCLC = Barcelona Clinic Liver Cancer, CEUS = contrast-enhanced ultrasound, MRI = magnetic resonance imaging.
4. Discussion
CEUS and MRI can provide standardized noninvasive diagnosis for high-risk HCC patients.[17,18] Some studies suggest that CEUS can be used as a first-line detection method for liver lesions.[19] However, many studies have included cases not confirmed by pathological tests after liver resection. All lesions in our study were confirmed by pathology after hepatectomy. In this way, false negative lesions in biopsy and imaging follow-ups were excluded to the maximum extent.
We found that MRI detected some additional small lesions that were not detected by CEUS (50/309) in 12.5% (27/216) of patients, and more than 50% of these additional lesions (28/50) were diagnosed as malignant by pathology. The number of lesions detected by MRI was significantly greater than that of CEUS, and the sensitivity of MRI to all small lesions was significantly higher than that of CEUS (P < .001). Our results are similar to previous studies.[18,20–22] The BCLC staging and the volume of liver resection were increased in 14 of the patients with liver cancer because additional lesions were shown in MRI. The preoperative identification of the presence and absence of metastatic liver lesions in patients with liver cancer and the determination of the number of metastatic lesions are significantly related to the determination of the BCLC staging, the formulation of surgical procedures, and the prognosis of patients.[2,12] However, some studies have shown that the sensitivity of CEUS to the focal lesions of arterial high-vascular liver disease is better than that of MRI.[8,10] Zhang et al[23] studied 127 nodules in 69 patients and demonstrated that CEUS can improve the detection of small liver lesions before surgery. However, just under a third of the patients in the study (21/69) had MRI with an unknown contrast enhancement, and the diagnostic performance of the MRI was not listed separately in the study. In this study, a small number of patients with enhanced MRI were included in the CECT/CEMRI group, which reduced the diagnostic performance of the MRI. Therefore, this result could not indicate that CEUS is superior to MRI regarding the diagnosis of small lesions. In our study, for lesions smaller than 2 cm, MRI could detect all lesions detected by CEUS, but CEUS could not detect all lesions detected by MRI. Therefore, our study shows that MRI can detect small intrahepatic lesions that cannot be detected by CEUS.
In this study, the sensitivity of MRI for lesions was >90%, and the positive predictive value was >80%. The diagnostic coincidence rate of MRI was 88.67% (274/309) for all lesions and 82.22% for lesions smaller than 2 cm. These results are similar to those in previous studies.[24–27] Two cases of metastatic liver cancer were detected and clearly diagnosed by Gd-EOB-DTPA-MRI, and the detection rate of metastatic lesions was up to 100%, the same as previous studies.[28]
Our study found that the diagnostic coincidence rate of CEUS was 94.83% for lesions larger than 2 cm (165/174) and 57.78% for lesions smaller than 2 cm (78/135). Strobel et al[8] showed that the overall diagnostic accuracy of CEUS for focal liver lesions (FLL) ≤ 20 mm was 83.8%, while Kim et al[26] found that the diagnostic coincidence rate of CEUS for lesions smaller than 2 cm was 70%. These 2 results are much higher than our results. This may be due to the fact that MRI found many small lesions not found by CEUS in this study. The number of lesions clearly diagnosed by postoperative pathology was significantly greater than the number of lesions diagnosed by CEUS before surgery; thus, the diagnostic accuracy of CEUS was low.
In this study, the sensitivity of CEUS to the main lesion was >95%, the positive predictive value was >80%, and the negative predictive value was close to 80%, which is similar to previous studies.[8,14,25]
In this study, the specificity of both MRI and CEUS was <70%, which is significantly lower than that of previous studies.[8,14,25,27,29,30] This may be due to the fact that the vast majority of cases included in this study were preoperatively considered malignant or ambiguous. Many benign cases without surgical treatment were not included. In other words, few lesions diagnosed as benign by imaging were included. There were also many preoperatively undiagnosed lesions, so the specificity was relatively low.
In this study, some patients received an indeterminate diagnosis by 1 of the 2 methods, and 2 patients could not be clearly diagnosed by either method. The majority of lesions with an indeterminate diagnosis were inflammatory pseudotumor or hyperplasia. Liver inflammatory pseudotumor is often difficult to differentiate from malignant liver tumors because of the lack of specific symptoms and imaging findings.[31] Inoue et al[32] found that the first phase in liver MRI had a certain value for diagnosing HCC lesions < 2 cm and atypical hyperplasia nodules, but further research was still needed for differential diagnosis. Strobel et al[8] reported that CEUS misclassified 6.2% of cases (15/241; 12 cases of benign FLLs and 3 cases of malignant FLLs), and Seitz et al[33] reported that CEUS returned an uncertain diagnosis for 9.2% of cases (24/262).
The sensitivity of both MRI and CEUS for the main lesion was more than 95%, and the coincidence rates of the 2 methods were both more than 80%. In our study, the specificity of the combination of the 2 methods was higher than that of either of the 2 methods alone, but there was no significant difference. CEUS and MRI each had high reliability in the identification of benign and malignant lesions, the diagnosis of hepatic hemangioma and focal nodular hyperplasia (FNH), and the characteristics of metastatic tumors and liver cancer, and they offered equal value in the differential diagnosis of newly discovered liver tumors.[21] CEUS and MRI with liver-specific contrast media are very reliable and of equal informative value in the characterization of focal liver lesions.[25]
Our study had some limitations. First, this study was a single-center retrospective study, so there is a possibility of selection bias. Second, CEUS images could not fully reflect all sites of the liver at the time of imaging. Third, CEUS had its own internal limitations, such as not being able to observe all lesions in the whole liver at the same time after each injection of contrast agent. Fourth, CEUS must be administered by specially trained physicians, and the detection results of lesions are subjective. Thus, there may have been visible lesions in CEUS scans that were missed by US operators at the time of examination. In addition, there is no way to evaluate the pathology of the entire liver, which may have led to missed diagnoses of some lesions. A prospective multi-center study with a large sample size is needed in the future.
5. Conclusion
For large lesions, the diagnostic performance is similar between Gd-EOB-DTPA-MRI and CEUS, and the sensitivity and coincidence rates of both methods are comparable. However, Gd-EOB-DTPA-MRI is likely to detect more small intrahepatic lesions (<2 cm) than CEUS.
Author contributions
JL and SH made substantial contributions to conception and design. JL, XY, and JW acquisition of data. JL, XY, and GY analysis and interpretation of data. JL, XY, and SH participated in drafting the article or revising it critically for important intellectual content. All listed authors had given final approval of the version to be published.
Conceptualization: Jiangfa Li, Songqing He.
Data curation: Jiangfa Li, Xiaofei Ye, Jiming Wang, Songqing He.
Formal analysis: Jiangfa Li, Xiaofei Ye, Jiming Wang, Guandou Yuan.
Funding acquisition: Jiangfa Li, Songqing He.
Methodology: Jiangfa Li, Xiaofei Ye.
Writing – original draft: Jiangfa Li, Xiaofei Ye.
Writing – review & editing: Jiangfa Li, Guandou Yuan, Songqing He.
Supplementary Material
Abbreviations:
- BCLC =
- Barcelona Clinic Liver Cancer
- CEUS =
- contrast-enhanced ultrasound
- CT =
- computed tomography
- EASL =
- the European Association for the Study of the Liver
- FLL =
- focal liver lesion
- FNH =
- focal nodular hyperplasia
- Gd-EOB-DTPA =
- gadolinium-ethoxybenzyl-diethylene triamine pentaacetic acid
- HCC =
- hepatocellular carcinoma
- MRI =
- magnetic resonance imaging
- KLCA-NCC =
- the Korean Association for Liver Cancer and the Korean National Cancer Center
JL and XY contributed equally to this work.
This study was supported by the National Natural Science Foundation of China (81771674), the 111 Project (D17011), Self-funded Project of Guangxi Zhuang Autonomous Region Health Commission (nos. Z20200013 and Z20200261), and Tianqing Liver Disease Research Fund of China Hepatitis Prevention and Control Foundation (TQGB20210052), Openin Project of Key laboratory of High-Incidence-Tumor Prevention & Treatment (Guangxi Medical University), Ministry of Education; Guangxi Key Laboratory of Early Prevention and Treatment for Regional High Frequency.
The authors have no conflicts of interest to disclose.
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Digital Content is available for this article.
How to cite this article: Li J, Ye X, Wang J, Yuan G, He S. A comparative study of gadolinium-ethoxybenzyl-diethylenetriamine pentaacetic acid-enhanced magnetic resonance imaging and contrast-enhanced ultrasound in the detection of intrahepatic lesion. Medicine 2022;101:36(e30482).
Contributor Information
Xiaofei Ye, Email: 1037645410@qq.com.
Jiming Wang, Email: 361821518@qq.com.
Guandou Yuan, Email: 510149375@qq.com.
References
- [1].Zhou M, Wang H, Zeng X, et al. Mortality, morbidity, and risk factors in China and its provinces, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2019;394:1145–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [2].Battaglia V, Cervelli R. Liver investigations: updating on US technique and contrast-enhanced ultrasound (CEUS). Eur J Radiol. 2017;96:65–73. [DOI] [PubMed] [Google Scholar]
- [3].Heimbach JK, Kulik LM, Finn RS, et al. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology. 2018;67:358–80. [DOI] [PubMed] [Google Scholar]
- [4].Pascolo L, Cupelli F, Anelli PL, et al. Molecular mechanisms for the hepatic uptake of magnetic resonance imaging contrast agents. Biochem Biophys Res Commun. 1999;257:746–52. [DOI] [PubMed] [Google Scholar]
- [5].Son J, Hwang SH, Park S, et al. Imaging features of hepatocellular carcinoma: quantitative and qualitative comparison between MRI-enhanced with Gd-EOB-DTPA and Gd-DTPA. Invest Radiol. 2019;54:494–9. [DOI] [PubMed] [Google Scholar]
- [6].Li J, Li X, Weng J, et al. Gd-EOB-DTPA dynamic contrast-enhanced magnetic resonance imaging is more effective than enhanced 64-slice CT for the detection of small lesions in patients with hepatocellular carcinoma. Medicine (Baltim). 2018;97:e13964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Li J, Wang J, Lei L, et al. The diagnostic performance of gadoxetic acid disodium-enhanced magnetic resonance imaging and contrast-enhanced multi-detector computed tomography in detecting hepatocellular carcinoma: a meta-analysis of eight prospective studies. Eur Radiol. 2019;29:6519–28. [DOI] [PubMed] [Google Scholar]
- [8].Strobel D, Bernatik T, Blank W, et al. Diagnostic accuracy of CEUS in the differential diagnosis of small (</= 20 mm) and subcentimetric (</= 10 mm) focal liver lesions in comparison with histology. Results of the DEGUM multicenter trial. Ultraschall Med. 2011;32:593–7. [DOI] [PubMed] [Google Scholar]
- [9].Wilson SR, Kim TK, Jang HJ, et al. Enhancement patterns of focal liver masses: discordance between contrast-enhanced sonography and contrast-enhanced CT and MRI. AJR Am J Roentgenol. 2007;189:W7–W12. [DOI] [PubMed] [Google Scholar]
- [10].Kim TK, Noh SY, Wilson SR, et al. Contrast-enhanced ultrasound (CEUS) liver imaging reporting and data system (LI-RADS) 2017 - a review of important differences compared to the CT/MRI system. Clin Mol Hepatol. 2017;23:280–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].Kim TH, Yoon JH, Lee JM. Emerging role of hepatobiliary magnetic resonance contrast media and contrast-enhanced ultrasound for noninvasive diagnosis of hepatocellular carcinoma: emphasis on recent updates in major guidelines. Korean J Radiol. 2019;20:863–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Kim H-D, Lim Y-S, Han S, et al. Evaluation of early-stage hepatocellular carcinoma by magnetic resonance imaging with gadoxetic acid detects additional lesions and increases overall survival. Gastroenterology. 2015;148:1371–82. [DOI] [PubMed] [Google Scholar]
- [13].Claudon M, Dietrich CF, Choi BI, et al. Guidelines and good clinical practice recommendations for contrast enhanced ultrasound (CEUS) in the liver--update 2012: a WFUMB-EFSUMB initiative in cooperation with representatives of AFSUMB, AIUM, ASUM, FLAUS and ICUS. Ultraschall Med. 2013;34:11–29. [DOI] [PubMed] [Google Scholar]
- [14].Sporea I, Badea R, Popescu A, et al. Contrast-enhanced ultrasound (CEUS) for the evaluation of focal liver lesions – a prospective multicenter study of its usefulness in clinical practice. Ultraschall Med. 2014;35:259–66. [DOI] [PubMed] [Google Scholar]
- [15].Albiin N. MRI of focal liver lesions. Curr Med Imag Rev. 2012;8:107–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Cong WM, Bu H, Chen J, et al. Practice guidelines for the pathological diagnosis of primary liver cancer: 2015 update. World J Gastroenterol. 2016;22:9279–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].Schellhaas B, Hammon M, Strobel D, et al. Interobserver and intermodality agreement of standardized algorithms for non-invasive diagnosis of hepatocellular carcinoma in high-risk patients: CEUS-LI-RADS versus MRI-LI-RADS. Eur Radiol. 2018;28:4254–64. [DOI] [PubMed] [Google Scholar]
- [18].Alaboudy A, Inoue T, Hatanaka K, et al. Usefulness of combination of imaging modalities in the diagnosis of hepatocellular carcinoma using Sonazoid(R)-enhanced ultrasound, gadolinium diethylene-triamine-pentaacetic acid-enhanced magnetic resonance imaging, and contrast-enhanced computed tomography. Oncology. 2011;81(suppl 1):66–72. [DOI] [PubMed] [Google Scholar]
- [19].Sporea I, Sirli R, Martie A, et al. How useful is contrast enhanced ultrasonography for the characterization of focal liver lesions? J Gastrointes Liver Dis. 2010;19:393–8. [PubMed] [Google Scholar]
- [20].Kunishi Y, Numata K, Morimoto M, et al. Efficacy of fusion imaging combining sonography and hepatobiliary phase MRI with Gd-EOB-DTPA to detect small hepatocellular carcinoma. Am J Roentgenol. 2012;198:106–14. [DOI] [PubMed] [Google Scholar]
- [21].Kobayashi T, Aikata H, Hatooka M, et al. Usefulness of combining gadolinium-ethoxybenzyl-diethylenetriamine pentaacetic acid-enhanced magnetic resonance imaging and contrast-enhanced ultrasound for diagnosing the macroscopic classification of small hepatocellular carcinoma. Eur Radiol. 2015;25:3272–81. [DOI] [PubMed] [Google Scholar]
- [22].Iwamoto T, Imai Y, Kogita S, et al. Comparison of contrast-enhanced ultrasound and gadolinium-ethoxybenzyl-diethylenetriamine pentaacetic acid-enhanced MRI for the diagnosis of macroscopic type of hepatocellular carcinoma. Dig Dis. 2016;34:679–86. [DOI] [PubMed] [Google Scholar]
- [23].Zhang XY, Luo Y, Wen TF, et al. Contrast-enhanced ultrasound: Improving the preoperative staging of hepatocellular carcinoma and guiding individual treatment. World J Gastroenterol. 2014;20:12628–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Tang W, Qin J, Hu B, et al. Comparison of gadoxetic acid disodium-enhanced MRI and biphasic spiral CT in detection of hepatocellular carcinoma in patients meeting the Milan criteria. Int J Clin Exp Med. 2018;11:2551–8. [Google Scholar]
- [25].Beyer LP, Wassermann F, Pregler B, et al. Characterization of focal liver lesions using CEUS and MRI with liver-specific contrast media: experience of a single radiologic center. Ultraschall Med. 2017;38:619–25. [DOI] [PubMed] [Google Scholar]
- [26].Kim SR, Mita K, Maekawa Y, et al. Comparison of diagnostic capability of contrast-enhanced CT, sonazoid contrast-enhanced us, Gd-EOB-DTPA MRI and CT arterioportal angiography in detecting histologically proven HCC nodules smaller than 2 cm. J Hepatol. 2010;52(suppl 1):S221–2. [Google Scholar]
- [27].Yoon JH, Lee JM, Lee YJ, et al. Added value of sequentially performed gadoxetic acid-enhanced liver MRI for the diagnosis of small (10-19 mm) or atypical hepatic observations at contrast-enhanced CT: a prospective comparison. J Magn Res Imag. 2019;49:574–87. [DOI] [PubMed] [Google Scholar]
- [28].Lowenthal D, Zeile M, Lim WY, et al. Detection and characterisation of focal liver lesions in colorectal carcinoma patients: comparison of diffusion-weighted and Gd-EOB-DTPA enhanced MR imaging. Eur Radiol. 2011;21:832–40. [DOI] [PubMed] [Google Scholar]
- [29].Seitz K, Bernatik T, Strobel D, et al. Telaprevir-benefit assessment of the Institute for Quality and Efficiency in Health Care (IQWiG) in accordance with section sign 35a SGB V (dosing evaluation). Z Gastroenterol. 2012;50:271–2. [DOI] [PubMed] [Google Scholar]
- [30].Nowicki TK, Markiet K, Izycka-Swieszewska E, et al. Efficacy comparison of multi-phase CT and hepatotropic contrast-enhanced MRI in the differential diagnosis of focal nodular hyperplasia: a prospective cohort study. BMC Gastroenterol. 2018;18:10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Al-Hussaini H, Azouz H, Abu-Zaid A. Hepatic inflammatory pseudotumor presenting in an 8-year-old boy: a case report and review of literature. World J Gastroenterol. 2015;21:8730–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [32].Inoue T, Kudo M, Komuta M, et al. Assessment of Gd-EOB-DTPA-enhanced MRI for HCC and dysplastic nodules and comparison of detection sensitivity versus MDCT. J Gastroenterol. 2012;47:1036–47. [DOI] [PubMed] [Google Scholar]
- [33].Seitz K, Bernatik T, Strobel D, et al. Contrast-enhanced ultrasound (CEUS) for the characterization of focal liver lesions in clinical practice (DEGUM Multicenter Trial): CEUS vs. MRI--a prospective comparison in 269 patients. Ultraschall Med. 2010;31:492–499. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.