Abstract
The stool antigen test (SAT) and the serum Helicobacter pylori (H. pylori) IgG antibody assays exhibit significant utility in the clinical diagnosis of H. pylori infection and in distinguishing between acute and chronic infections. The main objective of the current study was to identify the diagnostic value of serum H. pylori IgG antibody and SAT in the detection of H. pylori infections among chronic H. pylori-infected patients residing in Ibb Governorate, Yemen. 200 patients with H. pylori infection, confirmed through positive results in the serum immunochromatographic antibody test, were selected for H. pylori infection confirmation using serum H. pylori IgG antibodies and SAT across diverse hospitals, gastroenterology, and Hepatology clinics in Ibb Governorate. After the selection of patients, blood and stool specimens were obtained from all participants and underwent analysis via the Statistical Package for the Social Sciences (SPSS). The prevalence of H. pylori infection demonstrated variability based on the confirmatory tests, with rates of 54% for SAT and 78.5% for serum H. pylori IgG antibody, contrasting with a 100% prevalence observed in the screening serum immunochromatographic antibody test. Clinically, the study categorized H. pylori infections into four stages, whereby a significant proportion of patients (40.5%) exhibited positivity for both serum H. pylori IgG antibody and SAT, indicative of active chronic infections. The majority of positive cases only manifested serum H. pylori IgG antibody presence (chronic infections) at 38%, whereas 13.5% exclusively tested positive for SAT, corresponding to acute infections. Moreover, 88% of patients did not have either serum H. pylori IgG antibody or SAT (absence of infections) during confirmatory tests. Noteworthy is the study's approach employing multiple tests for H. pylori infection detection, focusing predominantly on chronic infections-prevailing types caused by H. pylori. The results revealed a significant association between serum levels of H. pylori IgG antibody and SAT results with the presence of diverse gastrointestinal symptoms among patients, which increased with long H. pylori infection durations.
Subject terms: Immunological techniques, Microbiology techniques, Health care
Introduction
Helicobacter pylori is a Gram-negative bacterium, measuring 2–4 µm in length and 0.5–1 µm in width usually spiral-shaped but the bacterium can appear as a rod, while coccoid shapes appear after prolonged in vitro culture or antibiotic treatment1,2.
The incidence of H. pylori infection is nearly 50% of the global population, it affected by low hygienic conditions and poor socioeconomic status. In addition, the incidence of H. pylori infections in UK and USA were 13.4% and 27.1%, respectively. While in developing countries, the estimated prevalence was 90% and approximately 4.4 billion individuals worldwide were positive for H. pylori infections (more than half the world population). There are a wide variation in the prevalence of H. pylori infections between different regions in Yemen, which depend on symptomatic patients with chronic dyspepsia the prevalence of H. pylori infections were 75–82.2%3–6.
Gastritis defines as any inflammation of the gastric mucosa. Indeed, worldwide H. pylori infection is the most common cause of chronic gastritis; it is also the most important risk factor for peptic ulcer and its complications as well as for gastric cancer (GC). H. pylori causes progressive damage to the gastric mucosa and plays as a causative role in a number of important diseases, including: duodenal ulcer (DU) disease, gastric ulcer (GU) disease, gastric adenocarcinoma and gastric mucosa associated lymphoid tissue lymphoma. It is estimated that H. pylori positive patients have 10–20% lifetime risk of developing ulcer disease and 1–2% have risk of developing distal GC6–10.
Various diagnostic techniques are employed for the detection of H. pylori infections, including both invasive methods (such as endoscopy for obtaining biopsies for histology, rapid urease test, and culture) and non-invasive methods (such as serology, urea breath test, and SAT). Serological diagnosis of H. pylori used to identify antibodies against Hp. The enzyme immunoassay (EIA) test has been the most prevalently used. Most commercial EIA tests are based on detecting immunoglobulin G (IgG), with sensitivity and specificity values ranging from 60 to 100%. Additionally, SAT is able to diagnose an ongoing infection, while the serological tests are limited to diagnose a contact with the bacterium, which can be current or lifetime. The SAT has many positive aspects: it is noninvasive, quick, has good sensitivity, specificity and reliability. This test can be used both for diagnosis of the infection and for monitoring therapy effectiveness, already 4 weeks after the end of treatment, its low cost, easy use and the possibility to collect samples have increasingly widespread the use of this method11–17. The current study was designed to evaluate of H. pylori infections in Ibb governorate, Yemen using mmunochromatographic antibody testing, serum H. pylori IgG antibody testing via EIA, and the SAT method and determine the value of both serum IgG H. pylori antibody and SAT in detection the chronic H. pylori infections.
Materials and methods
The current research employed a cross-sectional design involving the collection of stool and blood specimens from 200 patients afflicted with H. pylori infections who had tested positive for rapid serum antibody of H. pylori between October 2019 and November 2020. Diagnosis of patients based on the detection of serum H. pylori antibodies via rapid diagnostic testing. Validation of the results was carried out utilizing the serum H. pylori IgG antibody (NovaLisa Helicobacter pylori IgG ELISA (HEL G0220) with 96 wells for determinations, manufactured by NOVA TEC IMMUNODIAGNOSTICA GMBH in Germany, exhibiting a specificity of 93.0% and sensitivity of 98%) for chronic infections, and the serum SAT (developed by Intech corporation, showcasing a sensitivity of 98.4% and a specificity of 98.8%) for acute infections at various hospitals, hepatology, gastrointestinal clinics, and different medical laboratories in Ibb Governorate, Yemen. All patients with confirmed H. pylori infections, irrespective of age or gender, were considered for inclusion in this study. Exclusion criteria comprised malignancies such as ovarian, endometrial, cervical, lung, breast, liver, gastric, pancreatic, and colorectal cancers, individuals with other gastrointestinal infections, and those who provided incomplete responses in the questionnaire.
A structured and reliable questionnaire was designed and distributed among all participants included in the study. This questionnaire encompassed the following sections: the demographic profiles of patients, including age, gender, Khat chewing (Catha edulis) smoking, educational background, dietary patterns, weight, height, familial medical history including incidences of H. pylori infections and peptic ulcer disease (PUD), duration of H. pylori infection, gastrointestinal symptoms including (nausea, vomiting, heartburn, and abdominal pain, along with the severity of these symptoms and the treatment administered for H. pylori. Blood samples of 5–8 ml were collected from patients, with serum segments isolated and preserved in sterile Eppendorf tubes for immediate usage in both rapid H. pylori antibody testing and serum H. pylori IgG antibody detection via an enzyme immunoassay (ELISA) test. Furthermore, stool specimens were promptly collected from all subjects for the prompt identification of H. pylori antigens in stool. Additionally, the body mass index (BMI) was calculated by dividing the individual's weight by (height)2.
Statistical analysis
A Statistical Package of Social Sciences (SPSS version 21) analyzed the data. The results presented as percentages, means, standard deviations (SD). In addition, Chi-square X2 test was used for categorical variables. Furthermore, comparison was made by independent sample t-test and one-way ANOVA. P values < 0.05 were considered as statistically significant.
Ethical approval
The protocol of this work is approved by the Ethics Committee of Biological Sciences Department, Faculty of Science, Sana’a University, Yemen. This Ethics Committee agreed with the Helsinki Declaration for the Protection of Human Subjects. Informed consent was obtained from all the study participants.
Results
Association of socio-demographic characteristics of study subjects with confirmatory H. pylori tests
Initially, upon examining the demographic profiles of 200 patients diagnosed with H. pylori infections and their correlation with confirmatory diagnostic procedures, a noteworthy rise in the average age of the patients was observed. Out of all patients, a majority of 108 individuals (54%) were female, while 92 (46%) were male. Additionally, 148 patients (74%) reported the consumption of Khat (Catha edulis), 48 (24%) were identified as smokers, 127 (63%) had a familial history of H. pylori infections, and 60 (30%) had familial occurrences of PUD. Notably, the results of the SAT revealed a significant statistical association at (P < 0.05) with various factors including body mass index (BMI), intake of fruits and vegetables, recurrent infections, treatment protocols, repeated infections, and endoscopic assessments. Conversely, no significant correlations were detected with age, gender, residency, Khat consumption, smoking habits, familial predisposition to H. pylori infections and PUD. Moreover, a statistically significant correlation at (P < 0.05) was established between positivity to serum H. pylori IgG antibodies and parameters such as age, BMI, smoking history, familial incidence of H. pylori infections and PUD, recurrent infections, and treatment regimens. However, no significant association were observed concerning place of residence, gender, dietary habits regarding fruits and vegetables, or coffee consumption, as shown in Table 1.
Table 1.
Association of Socio-demographic characteristics of study participants with confirmatory H. pylori tests.
| Variable | SAT | P value | H. pylori IgG | P value | |||
|---|---|---|---|---|---|---|---|
| Positive (108) | Negative (92) | Positive (157) | Negative (43) | ||||
| Age (years) | Mean ± SD | 39.41 ± 16.36 | 33.73 ± 14.22 | 0.744 | 24.37 ± 9.32 | 21.90 ± 8.74 | 0.001* |
| BMI (kg/m2) | Mean ± SD | 33.75 ± 4.24 | 23.95 ± 4.47 | 0.015* | 39.57 ± 6.46 | 26.69 ± 5.21 | < 0.001* |
| No. (%) | No. (%) | No. (%) | No. (%) | No. (%) | |||
|---|---|---|---|---|---|---|---|
| Gender | |||||||
| Male | 92 (46) | 52 (48.1%) | 40 (43.5) | 0.509 | 88 (56.1%) | 21 (48.8%) | 0.095 |
| Female | 108 (54) | 56 (51.9%) | 52 (56.5%) | 69 (43.9%) | 22 (51.2%) | ||
| Residency | |||||||
| Rural | 110 (55) | 63 (58.3%) | 47 (51.1%) | 0.305 | 68 (43.3%) | 24 (55.8%) | 0.145 |
| Urban | 90 (45) | 45 (41.7%) | 46 (48.9%) | 89 (56.7%) | 19 (44.2%) | ||
| Education level | |||||||
| Elementary | 105 (52.5) | 62 (57.4%) | 43 (46.7%) | 0.089 | 87 (55.4%) | 18 (41.9%) | 0.239 |
| High school | 54 (27) | 30 (27.8%) | 24 (26.1%) | 41 (261%) | 13 (30.2%) | ||
| Professional | 41 (20.5) | 16 (14.8%) | 25 (27.2%) | 29 (18.5%) | 12 (27.9%) | ||
| Vegetables and fruit eating | |||||||
| Yes | 172 (86) | 98 (90.7%) | 74 (80.4%) | 0.036* | 131 (83.4%) | 41 (95.3%) | 0.046* |
| No | 28 (14) | 10 (9.3%) | 18 (19.6%) | 26 (16.6%) | 2 (4.7%) | ||
| Khat (Catha edulis) Chewing | |||||||
| Yes | 148 (74) | 81 (75%) | 67 (72.8%) | 0.727 | 121 (77.1%) | 27 (62.8%) | 0.059 |
| No | 52 (26) | 27 (25%) | 25 (27.2%) | 36 (22.9%) | 16 (37.2%) | ||
| Smoking | |||||||
| Yes | 48 (24) | 27 (25) | 21 (22.8%) | 0.720 | 45 (28.7%) | 3 (7%) | 0.003* |
| No | 152 (76) | 81 (75) | 71 (77.2%) | 112 (71.3%) | 40 (93%) | ||
| Family history of H. pylori | |||||||
| Yes | 127 (63.5) | 68 (63%) | 59 (64.1%) | 0.864 | 107 (68.2%) | 20 (46.5%) | 0.009* |
| No | 73 (36.5) | 40 (37%) | 33 (35.9%) | 50 (31.8%) | 23 (53.5%) | ||
| Family history of PUD | |||||||
| Yes | 60 (30) | 33 (30.6%) | 27 (29.3%) | 0.853 | 54 (34.4%) | 6 (14%) | 0.010* |
| No | 140 (70) | 75 (69.4%) | 65 (70.7%) | 103 (65.6%) | 37 (86%) | ||
| Treatment | |||||||
| Yes | 109 (54.5) | 51 (47.2%) | 58 (63%) | 0.025* | 96 (61.1%) | 13 (30.2%) | < 0.001* |
| No | 91 (45.5) | 57 (52.8%) | 34 (37%) | 61 (38.9%) | 30 (69.8%) | ||
| Recurrent infection | |||||||
| Yes | 89 (44.5) | 58 (53.7%) | 31 (33.7%) | 0.005* | 83 (52.9%) | 6 (14%) | < 0.001* |
| No | 111 (55.5) | 50 (46.3%) | 61 (66.3%) | 74 (47.1%) | 37 (86%) | ||
| Endoscopy | |||||||
| Yes | 18 (9%) | 14 (13%) | 4 (4.3%) | 0.034* | 16 (10.2%) | 2 (4.7%) | 0.261 |
| No | 182 (91%) | 94 (87%) | 88 (95.7%) | 141 (89.8%) | 41 (95.3%) | ||
The results represented by frequency and percent %, H. pylori: Helicobacter pylori, PUD peptic ulcer disease, BMI body mass index, P ≤ 0.05 considered as significant.
Prevalence of H. pylori infection according to different tests
The findings of the current study indicated that the prevalence of H. pylori infections as detected by rapid anti-H. pylori test encompassed all 200 patients (100%), while 157 individuals (78.5%) yielded positive results for serum H. pylori IgG antibodies, and 108 patients (54%) exhibited positive outcomes with the SAT methodology, as shown in Table 2.
Table 2.
Prevalence of H. pylori infection according to different tests.
| Variable | Number | Percent% |
|---|---|---|
| Rapid anti H. pylori | ||
| Yes | 200 | 100 |
| No | 0 | 0 |
| Total | 200 | 100 |
| H. pylori IgG | ||
| Yes | 157 | 78.5 |
| No | 43 | 21.5 |
| Total | 200 | 100.0 |
| SAT | ||
| Yes | 108 | 54.0 |
| No | 92 | 46.0 |
| Total | 200 | 100.0 |
The results represented by frequency and percent %, SAT Stool antigen test.
Classification of different stages of H. pylori infections
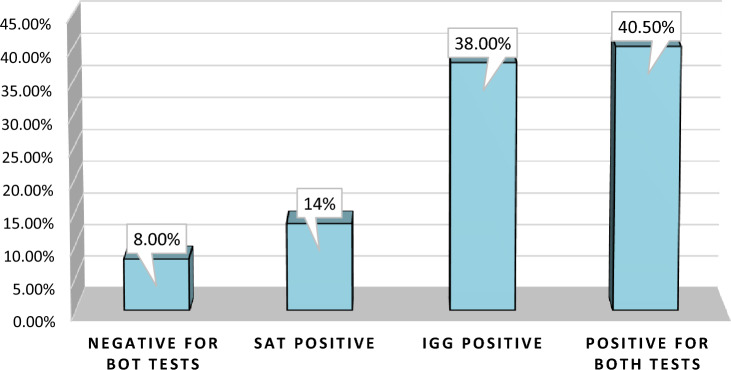
Clinically, the findings of the current study demonstrated the categorization of distinct stages of H. pylori infections as follows: (1) a total of 16 patients (8%) exhibited negative outcomes in both the serum H. pylori IgG antibody and SAT confirmatory tests (indicative of absence of infections). (2) Furthermore, 27 individuals (13.5%) only tested positive in the SAT examination (reflecting acute infections). (3) 76 patients (38%) exclusively demonstrated positive results in the serum H. pylori IgG antibody test (signifying chronic infections). (4) Notably, 81 subjects (40.5%) were positive in both the serum H. pylori IgG antibody and SAT tests (indicative of active chronic infections), as shown in Fig. 1.
Figure 1.
Classification of different stages of H. pylori infections.
The diagnostic value of serum H. pylori IgG antibody and stool antigen test
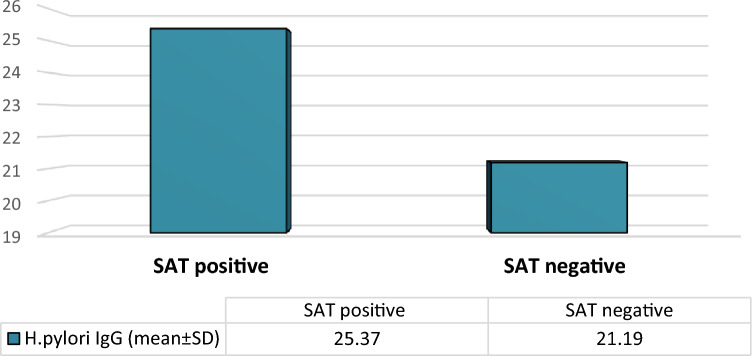
Significantly, the study's findings indicated that the diagnostic value of SAT and serum H. pylori IgG antibody for the diagnosis of H. pylori infections exhibited notably higher values in the SAT-positive groups (25.37 ± 10.47 NTU/ml) compared to those in the SAT-negative groups (21.193 ± 8.22), indicating a statistically significant difference (P < 0.05), as depicted in Fig. 2.
Figure 2.
The diagnostic value of serum H. pylori IgG antibody and stool antigen test.
Association between serum H. pylori IgG and SAT tests of H. pylori patients with gastrointestinal symptoms
Remarkably, the study revealed a significant correlation between SAT outcomes and gastrointestinal symptoms (inclusive of nausea, vomiting, heartburn, and abdominal pain), manifested by an increase in SAT positivity among symptomatic patients, as well as a significantly higher serum level of H. pylori IgG among symptomatic individuals. Additionally, the study indicated a linkage between H. pylori infection and vomiting, as evidenced by higher serum H. pylori IgG levels among patients experiencing vomiting as well as positivity to SAT (P < 0.05), as shown in Table 3.
Table 3.
Association between serum H. pylori IgG and SAT tests of H. pylori patients with gastrointestinal symptoms.
| Variable | H. pylori IgG | P value | SAT | X2 | P value | |
|---|---|---|---|---|---|---|
| Mean ± SD | Positive No. (%) |
Negative No. (%) |
||||
| Nausea | ||||||
| Yes (138) | 25.10 ± 9.53 | ˂ 0.001* | 89 (82.4%) | 49 (53.3%) | 19.73 | ˂ 0.001* |
| No (62) | 19.76 ± 8.59 | 19 (17.6%) | 43 (46.7%) | |||
| Vomiting | ||||||
| Yes (99) | 25.52 ± 10.51 | 0.002* | 71 (65.7%) | 28 (30.4%) | 24.77 | ˂ 0.001* |
| No (101) | 21.41 ± 8.06 | 37 (30.4%) | 64 (69.6%) | |||
| Heartburn | ||||||
| Yes (116) | 26.01 ± 9.73 | ˂ 0.001* | 70 (64.8%) | 46 (50%) | 5.47 | 0.034* |
| No (84) | 19.90 ± 8.11 | 38 (35.2%) | 46 (50%) | |||
| Abdominal pain | ||||||
| Yes (150) | 25.02 ± 9.85 | ˂ 0.001* | 89 (82.4%) | 61 (66.3%) | 8.87 | 0.009* |
| No (50) | 18.73 ± 6.75 | 19 (17.6%) | 31 (33.7%) | |||
The results represented by frequency and percent %, Independent t test, X2: Chi square test *P ≤ 0.05 considered as significant.
Association between serum H. pylori IgG and SAT tests with severity of gastrointestinal symptoms
Clinically, the study's results indicated a statistically significant association between SAT outcomes and the severity of gastrointestinal symptoms, with a heightened rate of SAT positivity observed with increased symptom severity. Moreover, the mean level of serum H. pylori IgG antibodies was significantly higher in the group exhibiting very severe symptoms (P < 0.05), as shown in Table 4.
Table 4.
Association between serum H. pylori IgG and SAT tests with severity of gastrointestinal symptoms.
| Symptoms | SAT | P value | H. pylori IgG (mean ± SD) | P value | |
|---|---|---|---|---|---|
| Positive (108) No. (%) |
Negative (92) No. (%) |
||||
| Without symptoms | 0 | 19 (20.7) | < 0.001* | 14.97 ± 4.14 | < 0.001* |
| Mild symptoms (1 symptom) | 14 (13%) | 16 (17.4%) | 19.22 ± 6.37 | ||
| Moderate symptoms (2 symptoms) | 21 (19.4%) | 19 (20.7%) | 22.79 ± 7.83 | ||
| Severe symptoms (3 symptoms) | 37 (34.3%) | 22 (23.9%) | 25.73 ± 9.61 | ||
| Very Severe symptoms (4 symptoms) | 36 (33.3%) | 16 (17.4%) | 26.89 ± 10.92 | ||
The results represented by frequency and percent %, X2: Chi square test, Independent t test, *P ≤ 0.05 considered as significant.
Association of serum H. pylori IgG and SAT tests with H. pylori infection duration
Lastly, it was observed that the serum H. pylori IgG levels were significantly elevated (P < 0.05) among patients with an infection duration exceeding 3 years. On the other hand, the association between the SAT status of study participants and the duration of H. pylori infection indicated no substantial variance in SAT test outcomes when compared against the duration of H. pylori infection (P > 0.05), as shown in Table 5.
Table 5.
Association between serum H. pylori IgG and SAT tests with H. pylori infection duration.
| Symptoms | SAT | P value | H. pylori IgG (mean ± SD) | P value | |
|---|---|---|---|---|---|
| Positive (108) No. (%) |
Negative (92) No. (%) |
||||
| 1 month ago | 34 (31.5%) | 17 (18.5%) | 0.155 | 17.55 ± 8.36 | < 0.001* |
| 6 months ago | 19 (17.6%) | 22 (23.9%) | 23.70 ± 8.04 | ||
| 1–3 years | 34 (31.5%) | 29 (31.5%) | 24.43 ± 8.38 | ||
| More than 3 years | 21 (19.4%) | 24 (26.1%) | 28.51 ± 10.37 | ||
The results represented by frequency and percent %, X2: Chi square test, independent t test, *P ≤ 0.05 considered as significant.
Discussion
Helicobacter pylori infection remains a significant global public health concern, with an estimated 4.4 billion individuals worldwide testing positive for H. pylori. Chronic H. pylori infections are commonly acquired during childhood (4).
The study results revealed a significant association between SAT outcomes and BMI, consumption of vegetables and fruits, recurrent infections, treatments, as well as endoscopic procedures. Conversely, no significant correlations were found with age, gender, place of residence, consumption of Khat (Catha edulis), smoking habits, familial history of H. pylori infections, or PUD. Notably, there was a significant correlation between serum H. pylori IgG antibody levels and age, BMI, smoking habits, familial history of H. pylori infections and PUD, recurrent infections, and treatments. These results are consistent with previous studies18,19 that indicated the elderly population over 59 years to be most affected by H. pylori infections. A study conducted in Yemen20 revealed a peak SAT positivity of 60% in the age range of 40–80 years and 45.6% in the age group of 19–40 years. Similarly, a study from Saudi Arabia21 reported a higher prevalence of H. pylori infections using SAT in individuals over 50 years of age.
Regarding gender, the present study did not find any significant association between gender and seropositivity to H. pylori detected by serum H. pylori IgG antibody and SAT (P > 0.05), contrasting findings from another study18 that reported a statistically significant link between H. pylori seropositivity and gender. The current study aligns with study by22,23 which also found no significant gender-based differences in H. pylori infection. Furthermore, there was no significant disparity in H. pylori SAT outcomes based on gender (P > 0.05).
Consistent with a study by24, the findings of this study demonstrated a higher H. pylori positivity rate in patients who chewed Khat (Catha edulis). However, another study25 contradicted these results, suggesting no significant difference in H. pylori infection rates based on Khat chewing (Catha edulis). Divergent outcomes have been documented in various studies regarding the correlation between H. pylori infection and smoking habits.
In a case–control study conducted in Nairobi, a higher prevalence of H. pylori infection was observed among among Khat chewers (Catha edulis), indicating a potential link between Khat (Catha edulis) consumption and susceptibility to gastrointestinal disorders26. Another study also suggested a greater occurrence of gastrointestinal disorders among Khat (Catha edulis) chewers, implying that Khat (Catha edulis) consumption might predispose individuals to gastrointestinal issues beyond just H. pylori infection27. Additionally, a study indicated a significant association between Khat (Catha edulis) chewing and duodenal ulcers, potentially attributed to stress related to Khat (Catha edulis) consumption28 Furthermore, a separate study found a higher prevalence of H. pylori infection among individuals who engaged in Khat (Catha edulis) chewing29.
Various previous studies have highlighted smoking as a risk factor for different diseases, including gastric cancer and H. pylori infection25,30,31. The current study's findings revealed a significantly elevated presence of H. pylori IgG antibodies in smokers compared to non-smokers, with a statistically significant distinction. These findings contrasted with those from32, which indicated no significant difference in H. pylori infection rates between smokers and non-smokers (P > 0.05).
Concerning residency, the results of the current study aligned with those of33, indicating no significant difference in H. pylori infection rates between patients residing in urban and rural areas (P > 0.05).
Significantly, the study's findings highlighted a statistically significant variance in serum levels of H. pylori IgG antibodies and SAT concerning BMI (P < 0.05). These findings were in line with findings of many previous studies34–38.
The mechanisms of association between H. pylori and obesity are varied, involving gastrointestinal hormones such as ghrelin and leptin, pivotal in metabolic regulation and energy balance. Ghrelin, produced in the stomach, stimulates food intake, while leptin functions in the opposite direction. Studies have reported lower serum levels of leptin and ghrelin in patients with H. pylori infections. Moreover, insulin resistance, a significant risk factor for several metabolic disorders including obesity, may be influenced by H. pylori infection. Obesity's interaction with H. pylori infection is being explored, with indications that the immune milieu in obese individuals may support H. pylori survival39,40.
In relation to a family history of PUD, the present study's results concurred with those from41, showing an association between H. pylori infection and a PUD history (P < 0.05). However, these findings contradicted those of42, which indicated no statistically significant difference in H. pylori infection positivity based on a family history of PUD. The potential association of H. pylori infection with a family history of the infection might be attributed to inter-familial transmission, emphasizing close and intimate contact among family members as a crucial mode of H. pylori spread43.
In this study, the consumption of vegetables and fruits showed a significant association with H. pylori infection. This could be attributed to the unhealthy conditions in Yemen, inadequate access to clean water sources, and insufficient awareness regarding the necessity of thoroughly washing fruits and vegetables. Previous studies have suggested that fruits and vegetables could be exposed to contamination through irrigation with polluted water. Moreover, the main sources for H. pylori include soil, water, animal waste, and human feces44–46.
Furthermore, the study findings indicated that the prevalence of H. pylori infections detected through rapid tests stood at 100%, while serum H. pylori IgG positivity was at 78.5%, and the rate of H. pylori infection positivity using SAT among the participants was 54%. These results were consistent with a study of47, which reported IgG seropositivity for H. pylori in 48.3% of the study subjects and a SAT positivity rate of 28.2%. A study conducted in Yemen20, showed a blood antibody test positivity of 72% and SAT positivity of 49%. Other studies19,48 indicated higher serum antibody positivity compared to stool antigen positivity. However49 found a SAT positivity rate of 82%, while50 revealed a lower SAT positivity rate of 32.22% among participants. The detection of H. pylori stool antigen and UBT are reliable indicators of an active infection51. On the other hand, serological tests for H. pylori IgG antibodies denote a past or chronic infection, with a decrease in H. pylori shedding observed with the progression of chronic infections, a pattern often seen in elderly individuals52.
The current study revealed distinct stages of H. pylori infections: 16 (8%) of the patients were negative for both serum H. pylori IgG antibody and SAT (no infections), 27 (13.5%) were only SAT positive (acute infections), 76 (38%) were only serum H. pylori IgG positive for both the serum H. pylori IgG antibody and SAT (chronic infections), and 81 (40.5%) were positive for both (active chronic infections). A study conducted in Ethiopia reported similar findings53.
Importantly, the study demonstrated that SAT and serum H. pylori IgG antibody had higher diagnostic value in SAT-positive groups (25.37 ± 10.47 NTU/ml) compared to SAT-negative groups (21.193 ± 8.22) at (P < 0.05). This aligns with previous studies54,55, reporting a seroprevalence of 78.4% using H. pylori serum IgG ELISA. However, the findings contrasted with a study56 from KSA showing a lower prevalence of 28.3% using ELISA for H. pylori IgG detection.
The results of this study also indicated a significant association between SAT and gastrointestinal symptoms like nausea, vomiting, heartburn, and abdominal pain. Patients with symptoms had higher SAT positivity and serum H. pylori IgG levels. The study also showed that vomiting was particularly linked to H. pylori infection, with higher IgG levels among patients experiencing vomiting and positive SAT results. Similar associations were found in previous studies linking epigastric pain, heartburn, and vomiting to H. pylori infection57–60.
Moreover, the findings of the current study demonstrated a significant correlation between SAT positivity and the severity of gastrointestinal symptoms, with a higher rate of SAT positivity observed in individuals with more severe symptoms. Additionally, there was a statistically significant increase in the mean level of serum H. pylori IgG antibody among participants experiencing very severe symptoms. Another study explored the link between H. pylori infection and symptom severity, noting that moderate to severe symptoms were associated with RUT positivity compared to individuals with mild symptoms61.
Finally, patients with an H. pylori infection duration exceeding 3 years exhibited significantly elevated levels of serum H. pylori IgG. On the other hand, no significant difference was observed in the association between the SAT status of participants and the duration of H. pylori infection when compared. The prevalence of active chronic H. pylori infections was notably high, primarily attributed to key risk factors such as advancing age, smoking, Khat consumption (Catha edulis), family history of H. pylori infection, presence and severity of gastrointestinal symptoms, and prolonged duration of H. pylori infection.
Acknowledgements
We would like to express our deep gratitude to the Biological Sciences Department, Faculty of Science, Sana’a University, Yemen, for providing the necessary facilities to carry out this work. We also extend a great deal of thanks to the specialist doctors and staff in hospitals and clinics in Ibb City, Yemen, for their assistance, and we thank our patients for their acceptance to participate in this study.
Author contributions
M.K.S. prepared and wrote the main manuscript, was responsible for conducting the main analysis and interpretation of the results, B.A.A.O. contributed to the supervision of the study, and M.A.Q. contributed to the supervision. M.A.A.J. and A.M.A. were involved in the conception of the manuscript.
Data availability
The data are not publicly available due to privacy or ethical consideration. The data sets generated during and/or analyzed during the current study are available from the corresponding author upon request.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Bashir A. Al Ofairi, Email: b.alafiri@su.edu.ye, Email: bofairy@yahoo.com
Marwan K. Saeed, Email: marwan0yemen0@gmail.com
References
- 1.Chan W-Y, Hui P-K, Leung K-M, Chow J, Kwok F, Ng C-S. Coccoid forms of Helicobacter pylori in the human stomach. Am. J. Clin. Pathol. 1994;102(4):503–507. doi: 10.1093/ajcp/102.4.503. [DOI] [PubMed] [Google Scholar]
- 2.Krzyżek P, Grande R. Transformation of Helicobacter pylori into coccoid forms as a challenge for research determining activity of antimicrobial substances. Pathogens. 2020;9(3):184. doi: 10.3390/pathogens9030184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Archampong TNA, Asmah RH, Wiredu EK, Gyasi RK, Nkrumah KN, Rajakumar K. Epidemiology of Helicobacter pylori infection in dyspeptic Ghanaian patients. Pan Afr. Med. J. 2015;20:1. doi: 10.11604/pamj.2015.20.178.5024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hooi JK, Lai WY, Ng WK, Suen MM, Underwood FE, Tanyingoh D, et al. Global prevalence of Helicobacter pylori infection: Systematic review and meta-analysis. Gastroenterology. 2017;153(2):420–429. doi: 10.1053/j.gastro.2017.04.022. [DOI] [PubMed] [Google Scholar]
- 5.Gunaid AA, Hassan NA, Murray-Lyon I. Prevalence and risk factors for Helicobacter pylori infection among Yemeni dyspeptic patients. Saudi Med. J. 2003;24(5):512–517. [PubMed] [Google Scholar]
- 6.Park JS, Jun JS, Seo J-H, Youn H-S, Rhee K-H. Changing prevalence of Helicobacter pylori infection in children and adolescents. Clin. Exp. Pediatr. 2021;64(1):21. doi: 10.3345/cep.2019.01543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fock KM, Graham DY, Malfertheiner P. Helicobacter pylori research: Historical insights and future directions. Nat. Rev. Gastroenterol. Hepatol. 2013;10(8):495. doi: 10.1038/nrgastro.2013.96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kotilea K, Bontems P, Touati E. Epidemiology, diagnosis and risk factors of Helicobacter pylori infection, Helicobacter pylori in human diseases. Adv. Microbiol. Infect. Dis. Public Health. 2019;11:17–33. doi: 10.1007/5584_2019_357. [DOI] [PubMed] [Google Scholar]
- 9.Graham DY, Rugge M. Clinical practice: Diagnosis and evaluation of dyspepsia. J. Clin. Gastroenterol. 2010;44(3):167. doi: 10.1097/MCG.0b013e3181c64c69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Malfertheiner P, Megraud F, O'Morain CA, Atherton J, Axon AT, Bazzoli F, et al. Management of Helicobacter pylori infection—the Maastricht IV/Florence consensus report. Gut. 2012;61(5):646–664. doi: 10.1136/gutjnl-2012-302084. [DOI] [PubMed] [Google Scholar]
- 11.Garza-González E, Perez-Perez GI, Maldonado-Garza HJ, Bosques-Padilla FJ. A review of Helicobacter pylori diagnosis, treatment, and methods to detect eradication. World J. Gastroenterol. 2014;20(6):1438. doi: 10.3748/wjg.v20.i6.1438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wong, U., & McLean, L. Diagnosis and management of Helicobacter pylori, 1–6 (2016).
- 13.Stefano K, Rosalia A, Chiara B, Federica G, Marco M, Gioacchino L, et al. Non-invasive tests for the diagnosis of Helicobacter pylori: State of the art. Acta Bio Med. Atenei Parmensis. 2018;89(Suppl 8):58. doi: 10.23750/abm.v89i8-S.7910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Opekun AR, Zierold C, Rode A, Blocki FA, Fiorini G, Saracino IM, et al. Clinical Performance of the Automated LIAISON® Meridian H pylori SA Stool Antigen Test. BioMed Research International; 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Han Y, Dai W, Meng F, Gan X, Liu M, Deng X, et al. Diagnosis of Helicobacter pylori infection in the elderly using an immunochromatographic assay-based stool antigen test. MicrobiologyOpen. 2020;9(9):e1102. doi: 10.1002/mbo3.1102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dechant F-X, Dechant R, Kandulski A, Selgrad M, Weber F, Reischl U, et al. Accuracy of different rapid urease tests in comparison with histopathology in patients with endoscopic signs of gastritis. Digestion. 2020;101(2):184–190. doi: 10.1159/000497810. [DOI] [PubMed] [Google Scholar]
- 17.Bordin DS, Voynovan IN, Andreev DN, Maev IV. Current Helicobacter pylori diagnostics. Diagnostics. 2021;11(8):1458. doi: 10.3390/diagnostics11081458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mathewos B, Moges B, Dagnew M. Seroprevalence and trend of Helicobacter pylori infection in Gondar University Hospital among dyspeptic patients, Gondar, North West Ethiopia. BMC Res. Notes. 2013;6(1):1–4. doi: 10.1186/1756-0500-6-346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kouitcheu Mabeku LB, Bello Epesse M, Fotsing S, Kamgang R, Tchidjo M. Stool antigen testing, a reliable noninvasive method of assessment of Helicobacter pylori infection among patients with gastro-duodenal disorders in Cameroon. Digest. Dis. Sci. 2021;66(2):511–520. doi: 10.1007/s10620-020-06219-0. [DOI] [PubMed] [Google Scholar]
- 20.Naji AS, Ameri G, Alkadasi MN, Hanash S, Ali W, Zaid A. Comparison of stool antigen and blood antibody test methods for detection of Helicobacter pylori infection and the risk factors. Int. J. Curr. Microbiol. 2014;3(12):118–127. [Google Scholar]
- 21.Lim SH, Kim N, Kwon JW, Kim SE, Baik GH, Lee JY, et al. Trends in the seroprevalence of Helicobacter pylori infection and its putative eradication rate over 18 years in Korea: A cross-sectional nationwide multicenter study. PLoS One. 2018;13(10):e0204762. doi: 10.1371/journal.pone.0204762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Al-Ardawi EJR, Al-Hussaini RM, Al-asady HMK, Sahib AA, Tizkam HH. Prevalence and association of Helicobacter pylori infection with gastritis and its age and sex distribution in a population of Karbala. Drug Invent. Today. 2019;12:11. [Google Scholar]
- 23.Moon H-W, Lee S-Y, Hur M, Yun Y-M. Characteristics of Helicobacter pylori-seropositive subjects according to the stool antigen test findings: A prospective study. Korean J. Internal Med. 2018;33(5):893. doi: 10.3904/kjim.2016.353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Al-Awadhi E, Bahaj S, Al-Oferi B. Helicobacter pylori infection among patients with type II diabetes mellitus. J. Diab. Metab. Disord. Control. 2020;7(2):53–56. doi: 10.15406/jdmdc.2020.07.00200. [DOI] [Google Scholar]
- 25.Abdu A, Cheneke W, Adem M, Belete R, Getachew A. Dyslipidemia and associated factors among patients suspected to have Helicobacter pylori infection at Jimma University Medical Center, Jimma, Ethiopia. Int. J. Gener. Med. 2020;13:311. doi: 10.2147/IJGM.S243848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hassan M, Mohamed K, Zipporah N, Hudson LD. Association between Khat (Catha edulis) chewing and infection with Helicobacter pylori: A case control study in Nairobi County. East Afr. Med. J. 2015;92:112–119. [Google Scholar]
- 27.Alyahawi A, Alkaf A, Alzaghrori S. Prevalence of Helicobacter pylori among asymptomatic populations in Sana'a, Yemen. Univ. J. Pharm. Res. 2018;3(3):31–35. doi: 10.22270/ujpr.v3i3.163. [DOI] [Google Scholar]
- 28.Raja'a Y, Noman T, Al Warafi A, Al Mashraki N, Al Yosofi A. Khat chewing is a risk factor of duodenal ulcer. Eastern Mediterr. Health J. 2001;7(3):568–570. doi: 10.26719/2001.7.3.568. [DOI] [PubMed] [Google Scholar]
- 29.Saeed MK, Al-Ofairi BA, Hassan MA, Al-Jahrani MA, Abdulkareem AM. The clinical significance of some serum tumor markers among chronic patients with Helicobacter pylori infections in Ibb Governorate, Yemen. Infect. Agents Cancer. 2023;18(1):60. doi: 10.1186/s13027-023-00542-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Jiang C, Chen Q, Xie M. Smoking increases the risk of infectious diseases: A narrative review. Tobacco Induced Dis. 2020;18:60. doi: 10.18332/tid/123845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Butt J, Varga MG, Wang T, Tsugane S, Shimazu T, Zheng W, et al. Smoking, Helicobacter pylori serology, and gastric cancer risk in prospective studies from China, Japan, and Korea. Cancer Prev. Res. 2019;12(10):667–674. doi: 10.1158/1940-6207.CAPR-19-0238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Odigie A, Adewole A, Ekunwe A. Prevalence and factors associated with Helicobacter pylori infection among treatment naïve dyspeptic adults in University of Benin Teaching Hospital, Benin City, Nigeria. Afr. J. Clin. Exp. Microbiol. 2020;21(2):97–105. doi: 10.4314/ajcem.v21i2.3. [DOI] [Google Scholar]
- 33.Maleki I, Mohammadpour M, Zarrinpour N, Khabazi M, Mohammadpour RA. Prevalence of Helicobacter pylori infection in Sari Northern Iran; a population based study. Gastroenterol. Hepatol. Bed Bench. 2019;12(1):31. [PMC free article] [PubMed] [Google Scholar]
- 34.Al-Akwaa AM. Prevalence of Helicobacter pylori infection in a group of morbidly obese Saudi patients undergoing bariatric surgery: A preliminary report. Saudi J. Gastroenterol. 2010;16(4):264. doi: 10.4103/1319-3767.70610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zhang Y, Du T, Chen X, Yu X, Tu L, Zhang C. Association between Helicobacter pylori infection and overweight or obesity in a Chinese population. J. Infect. Dev. Count. 2015;9(09):945–953. doi: 10.3855/jidc.6035. [DOI] [PubMed] [Google Scholar]
- 36.Lee SP, Lee S-Y, Kim JH, Sung I-K, Park HS, Shim CS. Factors related to upper gastrointestinal symptom generation in 2275 Helicobacter pylori seroprevalent adults. Digest. Dis. Sci. 2017;62(6):1561–1570. doi: 10.1007/s10620-017-4529-7. [DOI] [PubMed] [Google Scholar]
- 37.Jiang T, Chen X, Xia C, Liu H, Yan H, Wang G, et al. Association between Helicobacter pylori infection and non-alcoholic fatty liver disease in North Chinese: A cross-sectional study. Sci. Rep. 2019;9(1):1–6. doi: 10.1038/s41598-019-41371-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Furuto Y, Kawamura M, Yamashita J, Yoshikawa T, Namikawa A, Isshiki R, et al. Relationship between Helicobacter pylori infection and arteriosclerosis. Int. J. Gener. Med. 2021;14:1533. doi: 10.2147/IJGM.S303071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Roper J, Francois F, Shue PL, Mourad MS, Pei Z, Olivares de Perez AZ, et al. Leptin and ghrelin in relation to Helicobacter pylori status in adult males. J. Clin. Endocrinol. Metab. 2008;93(6):2350–2357. doi: 10.1210/jc.2007-2057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Xu X, Li W, Qin L, Yang W, Yu G, Wei Q. Relationship between Helicobacter pylori infection and obesity in Chinese adults: A systematic review with meta-analysis. PLoS One. 2019;14(9):e0221076. doi: 10.1371/journal.pone.0221076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Alzoubi H, Al-Mnayyis A, Aqel A, Abu-Lubad M, Hamdan O, Jaber K. The use of 13C-urea breath test for non-invasive diagnosis of Helicobacter pylori infection in comparison to endoscopy and stool antigen test. Diagnostics. 2020;10(7):448. doi: 10.3390/diagnostics10070448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Aitila P, Mutyaba M, Okeny S, Ndawula Kasule M, Kasule R, Ssedyabane F, et al. Prevalence and risk factors of Helicobacter pylori infection among children aged 1 to 15 years at holy innocents children’s hospital, Mbarara, South Western Uganda. J. Trop. Med. 2019;20:19. doi: 10.1155/2019/9303072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Manfredi M, Maffini V, de'Angelis G. Epidemiology, Transmission Routes and Recurrence of Infection Helicobacter pylori: Detection Methods, Diseases and Health Implications. Nova Science Publishers; 2013. pp. 101–112. [Google Scholar]
- 44.Yahaghi E, Khamesipour F, Mashayekhi F, Safarpoor Dehkordi F, Sakhaei MH, Masoudimanesh M, et al. Helicobacter pylori in vegetables and salads: Genotyping and antimicrobial resistance properties. Biomed. Res. Int. 2014;2014:757941. doi: 10.1155/2014/757941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Tabatabaei M. Application of molecular and cultural methods for identification of Helicobacter SPP. In different animal sources. Glob. Vet. 2012;8(3):292–297. [Google Scholar]
- 46.Pérez LM, Codony F, Leyton DL, Fittipaldi M, Adrados B, Morató J. Quantification of Helicobacter pylori levels in soil samples from public playgrounds in Spain. J. Zhejiang Univ. Sci. B. 2010;11:27–29. doi: 10.1631/jzus.B0900238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Omosor K, Omosor O, Adejumo B, Ibeh I, Dimkpa U. Comparative evaluation of stool antigen immunoassay and blood antibody test methods for the screening of Helicobacter pylori infection in asymptomatic adult population in Delta State, Nigeria. J. Mol. Microbiol. 2018;2(1):3–9. [Google Scholar]
- 48.Kazemi S, Tavakkoli H, Habizadeh MR, Emami MH. Diagnostic values of Helicobacter pylori diagnostic tests: Stool antigen test, urea breath test, rapid urease test, serology and histology. J. Res. Med. Sci. 2011;16(9):1097. [PMC free article] [PubMed] [Google Scholar]
- 49.Talebi Bezmin Abadi A. Diagnosis of Helicobacter pylori using invasive and noninvasive approaches. J. Pathog. 2018;20:18. doi: 10.1155/2018/9064952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Prell C, Osterrieder S, Lottspeich C, Schwarzer A, Rüssmann H, Ossiander G, et al. Improved performance of a rapid office-based stool test for detection of Helicobacter pylori in children before and after therapy. J. Clin. Microbiol. 2009;47(12):3980–3984. doi: 10.1128/JCM.01204-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Cardos AI, Maghiar A, Zaha DC, Pop O, Fritea L, Miere F, et al. Evolution of diagnostic methods for Helicobacter pylori infections: From traditional tests to high technology, advanced sensitivity and discrimination tools. Diagnostics. 2022;12(2):508. doi: 10.3390/diagnostics12020508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zaman A, Shamsuzzaman S, Bhuiyan F, Hasan MR, Saito T. Observation of changes in Helicobacter pylori antigen and antibody positivity according to non-invasive tests before and after Helicobacter pylori eradication therapy in symptomatic patients. Int. J. Gener. Med. 2020;13:1093. doi: 10.2147/IJGM.S273368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Seid A, Demsiss W. Feco-prevalence and risk factors of Helicobacter pylori infection among symptomatic patients at Dessie Referral Hospital, Ethiopia. BMC Infect. Dis. 2018;18(1):1–9. doi: 10.1186/s12879-018-3179-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Bener A, Uduman SA, Ameen A, Alwash R, Pasha MA, Usmani MA, et al. Prevalence of Helicobacter pylori infection among low socio-economic workers. J. Commun. Dis. 2002;34(3):179–184. [PubMed] [Google Scholar]
- 55.Memon IA, Kumar M, Kheemchand FN, Talpur SK, Advani D. Diagnostic accuracy of serologic (IGG) in diagnosis of Helicobacter pylori among patients of dyspepsia by taking stool antigen (HPSA) as gold standard. Pak. J. Med. Health Sci. 2022;16(02):791. [Google Scholar]
- 56.Hanafi MI, Mohamed AM. Helicobacter pylori infection: Seroprevalence and predictors among healthy individuals in Al Madinah, Saudi Arabia. J. Egypt. Public Health Assoc. 2013;88(1):40–45. doi: 10.1097/01.EPX.0000427043.99834.a4. [DOI] [PubMed] [Google Scholar]
- 57.Abdallah TM, Mohammed HB, Mohammed MH, Ali AAA. Sero-prevalence and factors associated with Helicobacter pylori infection in Eastern Sudan. Asian Pac. J. Trop. Dis. 2014;4(2):115–119. doi: 10.1016/S2222-1808(14)60326-1. [DOI] [Google Scholar]
- 58.Hamrah MH, Hamrah MS, Hamrah MH, Kanda M, Hamrah AE, Dahi AE, et al. Prevalence of Helicobacter pylori infection in dyspeptic patients in Andkhoy Afghanistan. Asian Pac. J. Cancer Prev. 2017;18(11):3123. doi: 10.22034/APJCP.2017.18.11.3123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Raj P, Thompson JF, Pan DH. Helicobacter pylori serology testing is a useful diagnostic screening tool for symptomatic inner city children. Acta Paediatr. 2017;106(3):470–477. doi: 10.1111/apa.13724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Galal YS, Ghobrial CM, Labib JR, Abou-Zekri ME. Helicobacter pylori among symptomatic Egyptian children: Prevalence, risk factors, and effect on growth. J. Egypt. Public Health Assoc. 2019;94(1):1–8. doi: 10.1186/s42506-019-0017-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Adu-Aryee NA, Aabakken L, Dedey F, Nsaful J, Kudzi W. Comparison of endoscopic based diagnosis with Helicobacter urease test for Helicobacter pylori infection. BMC Res. Notes. 2016;9(1):421. doi: 10.1186/s13104-016-2237-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data are not publicly available due to privacy or ethical consideration. The data sets generated during and/or analyzed during the current study are available from the corresponding author upon request.