Abstract
Objectives
To examine the associations between changes in cardiorespiratory fitness (CRF) in adulthood and prostate cancer incidence and mortality.
Methods
In this prospective study, men who completed an occupational health profile assessment including at least two valid submaximal CRF tests, performed on a cycle ergometer, were included in the study. Data on prostate cancer incidence and mortality were derived from national registers. HRs and CIs were calculated using Cox proportional hazard regression with inverse probability treatment weights of time-varying covariates.
Results
During a mean follow-up time of 6.7 years (SD 4.9), 592 (1%) of the 57 652 men were diagnosed with prostate cancer, and 46 (0.08%) died with prostate cancer as the primary cause of death. An increase in absolute CRF (as % of L/min) was associated with a reduced risk of prostate cancer incidence (HR 0.98, 95% CI 0.96 to 0.99) but not mortality, in the fully adjusted model. When participants were grouped as having increased (+3%), stable (±3%) or decreased (−3%) CRF, those with increased fitness also had a reduced risk of prostate cancer incidence compared with those with decreased fitness (HR 0.65, 95% CI 0.49 to 0.86), in the fully adjusted model.
Conclusion
In this study of employed Swedish men, change in CRF was inversely associated with risk of prostate cancer incidence, but not mortality. Change in CRF appears to be important for reducing the risk of prostate cancer.
Keywords: Physical fitness
WHAT IS ALREADY KNOWN ON THIS TOPIC
Cardiorespiratory fitness (CRF) is associated with the risk of being diagnosed with, or dying from, certain cancer types.
The associations between CRF measured at one time point and risk of prostate cancer incidence are contradictory, which may be the result of the influence of higher prostate cancer screening rates in men with higher fitness.
WHAT THIS STUDY ADDS
Our results suggest that increases in CRF are inversely associated with risk of prostate cancer incidence, but not mortality.
The results highlight the importance of CRF for prostate cancer risk, which has been challenging to determine with single timepoint studies.
HOW THIS STUDY MIGHT AFFECT RESEARCH, PRACTICE OR POLICY
Improvements in CRF in adult men should be encouraged and may reduce the risk of prostate cancer.
Introduction
Unlike other common cancers such as breast, colon and lung cancer, there are relatively few preventable risk factors with strong evidence for reducing prostate cancer risk. Aside from developmental factors, being diagnosed with overweight or obesity are the main risk factors for developing advanced prostate cancer, but insufficient evidence exists to extend this conclusion to non-advanced prostate cancer.1
While there are well-established relationships between physical activity levels and the incidence and mortality of an increasing number of cancer types, the evidence for prostate cancer is not conclusive.2 Several studies have reported a decreased risk of prostate cancer incidence, while others have reported an increased risk of prostate cancer for those with higher physical activity levels.2 Fewer still have investigated the relationships between cardiorespiratory fitness (CRF) and prostate cancer incidence and mortality risk. Local and systemic inflammation is associated with increased risk of (advanced) prostate cancer; therefore, there is an increasing focus on understanding the role of CRF on cancer risk because of the favourable associations between CRF and systemic inflammation.3 Additionally, CRF has shown to have desirable associations with abdominal obesity,4 dyslipidaemia5 and insulin sensitivity.6 In the studies that have investigated this relationship between CRF and cancer, the findings generally mirror those conflicting findings in studies of physical activity and cancer risk.7–11 The tendency for men with higher CRF to also have higher prostate cancer screening participation rates may be an influencing factor to explain these inconsistent findings.7 10
Examining the association between change in CRF over two time points and prostate cancer incidence and mortality risk could contribute to our understanding of the role of CRF as a risk factor. To date, only four smaller studies have investigated the association between change in CRF or physical activity and cancer incidence or mortality.12–15 Despite the known differences in mechanisms behind both the development of different cancers and the role CRF may play in reducing the risk of cancer, none of these studies examined either incidence or mortality by specific cancer type. To better understand the relationship between CRF and prostate cancer incidence and mortality, and to contribute to cancer-specific prevention recommendations, it is essential to conduct analyses by cancer type. Therefore, the aim of the current study was to determine if there were any associations between changes in CRF and prostate cancer incidence and mortality.
Methods
In this prospective cohort study, data were drawn from the health profile assessment (HPA) database, managed by the HPI Health Profile Institute (HPI, Stockholm Sweden). Participation in the HPA was optional, usually offered to all employees working at an associated company or organisation and conducted by an occupational and health service with no financial cost to the employees. HPI was contracted by the occupational health services and responsible for the development and standardisation of the method, education of data collection staff, and administration of the central database. Each HPA consisted of a questionnaire assessing physical activity, lifestyle, perceived health, measurement of body mass and height, and a submaximal ergometer CRF test.
From October 1982 until December 2019 (month of follow-up of outcome data in national registers) data from a total of 181 673 men (aged 18 years and older) were stored in the central HPA database. People were included if they had data for all covariates included in the full analyses (age, education level, body mass index (BMI) and smoking), and at least two valid CRF tests from two separate health assessments, 11 months or more apart. For participants who had more than two tests, only their first and last tests were included in the analyses. Participants were excluded if they had already been diagnosed with prostate cancer before the second health assessment, and only men were included in the study population.
Assessment of CRF
The standardised Åstrand test, a submaximal cycle ergometer test, was used to assess the study exposure variable CRF as estimated VO2max.16–18 Previously, the Åstrand test has been validated against directly measured VO2max during a maximal treadmill test to exhaustion in an adult population. The validation study demonstrated a marginal and insignificant mean difference of −0.07 L/min (95% CI −0.21 to 0.06) in VO2max between the Åstrand test and VO2max measured directly. The SE of estimate was 0.48 L/min and the coefficient of variation was 18.1%, which align with validations of other submaximal VO2max tests.16 Participants were requested to refrain from the following before the test: vigorous activity the day before, consuming a heavy meal 3 hours before, smoking/snuff use 1 hour before and avoiding stress directly before test. Change in CRF was expressed as absolute (L/min) and relative (mL/kg/min) per year and divided into groups based on if their absolute CRF increased by more than 3% (increase), decreased by more than 3% (decrease) or remained within ±3% (stable). In addition, we divided participants into tertials according to their baseline relative CRF, resulting in three equally sized groups: low CRF (<32.4 mL/kg/min), moderate CRF (32.4–40.7 mL/kg/min) and high CRF (>40.7 mL/kg/min). In this stratified analysis the individual change in absolute CRF was standardised to the mean and SD of the baseline relative CRF category group the individual belonged to, in order to minimise the regression toward the mean effect of the groups.
Prostate cancer incidence and mortality
The Swedish National Hospital and National Cause of Death registries19 were used to draw data on prostate cancer diagnosis and mortality. This data was linked to the HPA database by linking each participant’s unique Swedish personal identification number. For the incidence analyses, all participants were followed from the date of the last HPA to the date of their prostate cancer diagnosis, death (of any cause) or until 31 December 2019. For the mortality analyses, all participants were followed from the date of their second HPA to either the date of death from prostate cancer, death from any cause, or until 31 December 2019. The ICD-10 code used to identify prostate cancer was C61.
Assessment of covariates
Covariates were included based on current evidence of their potential to influence the relationship between CRF and prostate cancer incidence or mortality. Highest level of education attained at the time of the HPA (defined as length of education <9 years to postgraduate education) was obtained from Statistics Sweden by linking the participant personal identity number. Smoking at the time of the HPA was self-reported using the following statements: I smoke … with the options: At least 20 cig/day, 11–19 cig/day, 1–10 cig/day, occasionally or never. Body mass was assessed at the HPA with a calibrated scale in light-weight clothing to the nearest 0.5 kg. Height was assessed at the HPA using a wall mounted stadiometer and measured to the nearest 0.5 cm. BMI (kg/m2) was subsequently calculated.
Patient and public involvement
We did not involve patients or the public in the design or while conducting our research. In addition to scientific journal articles, the results of the study will be disseminated to the public through online print media and with the cancer community through the Swedish Cancer Society.
Equity, diversity and inclusion statement
Our study did not purposefully exclude people based on their race, ethnicity or socioeconomic level. Information on race, ethnicity or socioeconomic level was not included in the database. The project group included four women and three men and was led by a woman. Researchers involved are from all career levels from postdoctoral researcher to professor. All researchers reside in Stockholm, Sweden.
Statistical analyses
Outliers that lay −20% and 20% (L/min/year) from the mean were removed (n=1319) to minimise influence of measurement error, as this level of change per year over several years is extremely rare and unlikely. Associations between CRF at baseline and at follow-up and prostate cancer incidence and mortality were analysed using Cox proportional hazard regression models. Associations between changes in CRF and prostate cancer incidence and mortality were analysed using Cox proportional hazard regression models with inverse probability treatment weights of time-varying confounders such as age, BMI and smoking. Non-time varying confounders (education, baseline CRF and year of last HPA test) were adjusted for conventionally. For visualisation purposes, restricted cubic splines with knots at 5th, 50th and 95th percentile were performed for % change in absolute CRF with 0% change as the reference.
Four models were created with an increasing number of variables for each model. Model 1 included only baseline CRF. Model 2 additionally included age, education and year of the last HPA test. Model 3 additionally included BMI, and model 4 further included smoking. Model 5 (online supplemental file) additionally included adjustment for self-reported physical activity. Baseline CRF was not adjusted for in the one timepoint analysis (baseline and follow-up associations) or in the subanalysis stratified by baseline CRF.
bjsports-2023-107007supp001.pdf (401.6KB, pdf)
In the subgroup analyses, Cox proportional hazard modelling with inverse probability treatment weight of time-varying confounders was used to examine the relationships between the groups, stratified by direction of change in absolute fitness (stable, decrease and increase) and baseline relative CRF (low, moderate and high), and prostate cancer incidence and mortality. For all models the proportional hazard assumption was checked using scaled Schoenfeld residuals. To investigate if change in absolute CRF were associated with change in self-reported physical activity, a linear regression weighted for age, and adjusted for education, and the year of the last HPA test was performed.
Sensitivity analysis to address potential reverse causality was performed by dropping incident cases that occurred ≤2 years after the last HPA test for the main analysis and the grouped (increase, decrease and stable) analysis.
All statistical analyses were performed using R Studio, V.4.2.1 (2022-06-23).20 The IPW package21 was used to create inverse probability treatment weights and the survival22 package was used to create Cox proportional hazard regression models. The splines,20 ggplot223 and survminer24 package were used to visualise the restricted cubic splines. Conduct and presentation of statistical analyses in this study are in accordance with the CHecklist for statistical Assessment of Medical Papers) statement.25
Results
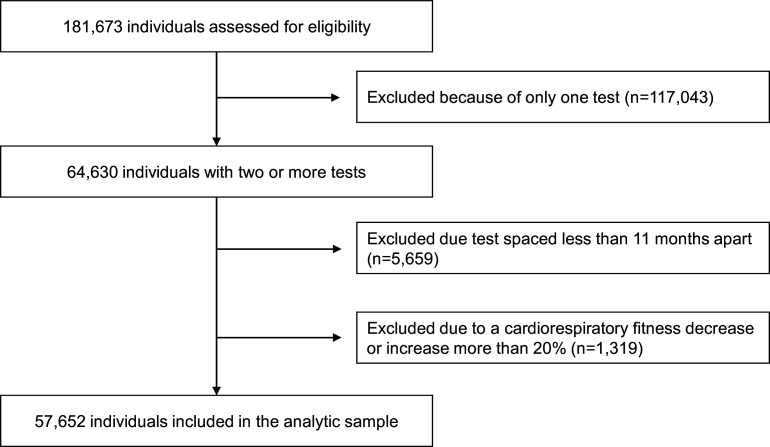
Among the 181 673 men, 58 971 met the criteria of having undertaken a minimum of two tests spaced at least 11 months apart. We excluded 1319 individuals due to a CRF decrease or increase of over 20% per year (figure 1). The analytic sample consisted of 57 652 men, with a mean age of 41.4 years (range 18–79 years) and an average BMI of 26.0 kg/m2 (SD 3.6) at the first HPA (table 1). Of the 57 652 men, 592 (1%) were diagnosed with prostate cancer, while 46 (0.08%) died of prostate cancer as the primary cause of death. At baseline, the sample’s mean relative and absolute CRF were 37.4 mL/kg/min and 3.12 L/min, respectively. From the first to the second HPA relative CRF decreased by 0.27 mL/kg/min (SD 2.2), while absolute CRF decreased by 0.01 L/min (SD 0.17), on average. The time between the tests was on average 4.9 years (SD 3.6). The mean follow-up time for the incidence analyses was 6.7 years (SD 4.9) from the last HPA test.
Figure 1.
Flow chart of participant inclusion.
Table 1.
Participant descriptive characteristics, all, and grouped by changes in and baseline cardiorespiratory fitness
| All | Decrease −3% (L/min) |
Stable ±3% (L/min) |
Increase +3% (L/min) |
Low <32.4 (mL/kg/min) |
Moderate 32.4–40.7 (mL/kg/min) |
High >40.7 (mL/kg/min) |
|
| n | 57 652 | 13 695 | 31 581 | 12 376 | 19 218 | 19 219 | 19 215 |
| Age (years), mean (SD) | 41.36 (10.55) | 42.57 (11.10) | 41.25 (10.27) | 40.83 (11.36) | 46.05 (9.74) | 41.57 (10.11) | 36.47 (9.54) |
| BMI (kg/m2), mean (SD) | 26.03 (3.58) | 26.12 (3.57) | 25.99 (3.54) | 26.15 (3.79) | 27.97 (4.01) | 25.80 (3.04) | 24.32 (2.55) |
| Education, n (%) | |||||||
| College/University degree, n (%) | 21 625 (37.5) | 2858 (20.9) | 15 552 (49.2) | 3215 (26.0) | 5712 (29.7) | 7128 (37.1) | 8785 (45.7) |
| Never smoked, n (%) | 47 979 (83.2) | 6084 (81.6) | 35 283 (83.5) | 6612 (83.0) | 15 596 (81.2) | 15 952 (83.0) | 16 431 (85.5) |
| Exercise >1 time/week, n (%) | 35 161 (61.0) | 8982 (65.6) | 19 027 (60.2) | 7152 (57.8) | 8836 (46.0) | 11 645 (60.6) | 14 680 (76.4) |
| Changes in self-reported physical activity, mean (SD) | 0.15 (1.10) | 0.17 (1.11) | −0.09 (1.06) | 0.37 (1.06) | 0.24 (1.13) | 0.17 (1.11) | 0.04 (1.04) |
| Baseline test absolute VO2max
(L/min), mean (SD) |
3.12 (0.74) | 3.38 (0.76) | 3.13 (0.73) | 2.79 (0.65) | 2.47 (0.43) | 3.06 (0.44) | 3.82 (0.60) |
| Baseline test relative VO2max
(mL/kg1/min1), mean (SD) |
37.43 (9.70) | 40.52 (10.14) | 37.65 (9.56) | 33.35 (8.61) | 27.39 (3.63) | 36.48 (2.37) | 48.42 (6.41) |
| Time between tests (years), mean (SD) | 4.92 (3.61) | 3.36 (2.14) | 6.33 (3.96) | 3.02 (2.03) | 4.54 (3.28) | 5.02 (3.64) | 5.20 (3.84) |
| Follow-up (years), mean (SD) | 6.73 (4.85) | 7.22 (4.99) | 6.25 (4.63) | 7.42 (5.09) | 6.66 (4.81) | 6.74 (4.86) | 6.80 (4.87) |
| Changes in absolute VO2max per year (L/min), mean (SD) |
−0.01 (0.17) | −0.29 (0.14) | −0.02 (0.08) | 0.26 (0.12) | 0.03 (0.14) | −0.01 (0.16) | −0.06 (0.20) |
| Change in relative VO2max per year (mL∙kg-1∙min-1), mean (SD) |
−0.27 (2.20) | −3.63 (1.95) | −0.31 (1.10) | 3.09 (1.80) | 0.33 (1.71) | −0.18 (2.03) | −0.96 (2.58) |
| Changes in BMI per year (kg/m2), mean (SD) |
0.08 (0.54) | 0.09 (0.41) | 0.10 (0.61) | 0.05 (0.70) | 0.03 (0.64) | 0.09 (0.49) | 0.12 (0.45) |
BMI, body mass index; VO2max, estimated maximal oxygen consumption.
There was no association between baseline CRF or CRF at last test and the risk of prostate cancer incidence, when the models were adjusted for relevant covariates (online supplemental table s1). Only in the unadjusted model 1 was there an inverse association between CRF both at baseline and at last test and the risk of incidence prostate cancer.
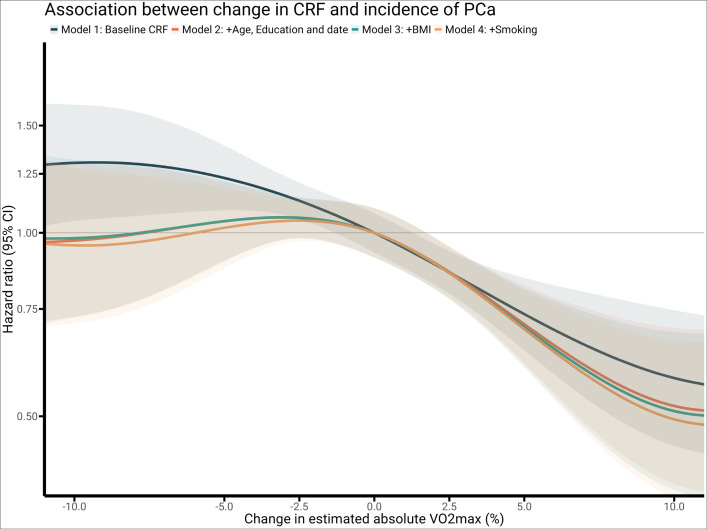
Percentage change in absolute CRF (L/min) was inversely associated with risk of being diagnosed with prostate cancer (figure 2 and table 2) and remained significant after adjusting for all covariates (baseline CRF, age, education level, year of test, BMI, smoking) (table 2) and physical activity (online supplemental table s2). In comparison, per cent change in relative CRF (mL/kg/min) was associated with prostate cancer incidence only for model 1 (adjusted for baseline CRF).
Figure 2.
Restricted cubic splines of the Cox proportional model examining the association between % change in cardiorespiratory fitness and incidence of prostate cancer. Model 1: adjusted for baseline fitness. Model 2: adjusted for baseline fitness, age, education and year of last test. Model 3: adjusted for baseline fitness, age, education, year of last test and body mass index. Model 4: adjusted for baseline fitness, age, education, year of last test, body mass index and smoking. BMI, body mass index; VO2max, estimated maximal oxygen consumption.
Table 2.
HRs with 95% CIs for the association between change in cardiorespiratory fitness and prostate cancer incidence and mortality
| Incidence | Model 1 | Model 2 | Model 3 | Model 4 | N | Cases | ||||
| HR | 95% CI | HR | 95% CI | HR | 95% CI | HR | 95% CI | |||
| % change in VO2max (L/min) | 0.97*** | 0.96 to 0.98 | 0.98** | 0.97 to 0.99 | 0.98** | 0.97 to 0.99 | 0.98** | 0.96 to 0.99 | 57 652 | 592 |
| Change in VO2max (L/min) | 0.29*** | 0.24 to 0.51 | 0.45** | 0.26 to 0.77 | 0.44** | 0.25 to 0.77 | 0.46** | 0.27 to 0.80 | 57 652 | 592 |
| Mortality | ||||||||||
| % change in VO2max (L/min) | 0.98 | 0.94 to 1.03 | 0.99 | 0.95 to 1.02 | 0.99 | 0.95 to 1.02 | 0.98 | 0.94 to 1.02 | 57 652 | 46 |
| Change in VO2max (L/min) | 0.48 | 0.08 to 2.86 | 0.55 | 0.12 to 2.53 | 0.52 | 0.11 to 2.37 | 0.38 | 0.07 to 2.11 | 57 652 | 46 |
Model 1: adjusted for baseline fitness. Model 2: adjusted for baseline fitness, age, education and year of last test. Model 3: adjusted for baseline fitness, age, education, year of last test and body mass index. Model 4: adjusted for baseline fitness, age, education, year of last test, body mass index and smoking.
*p<0.05, **p<0.01, ***p<0.001.
VO2max, estimated maximal oxygen consumption.
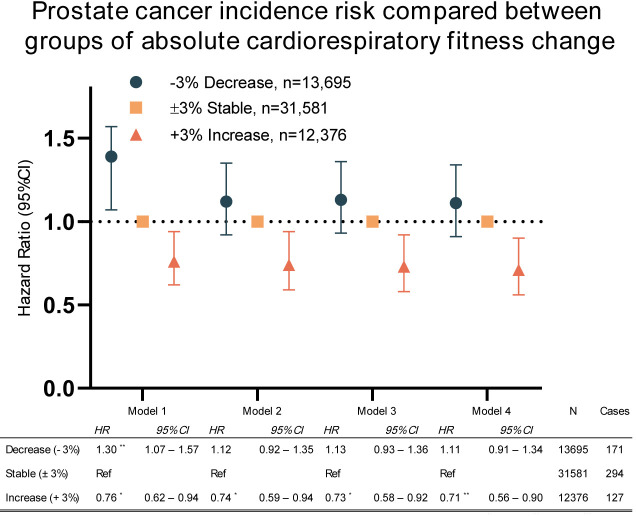
When participants were grouped by those with increased (+3%), stable (±3%) or decreased (−3%) absolute CRF, those with increased CRF had significantly lower cancer incidence risk compared with those who held their CRF stable (figure 3). The associations remained significant after adjusting for the covariates in all models. In a sensitivity analysis, individuals who were diagnosed with prostate cancer in the 2 years following the last HPA were excluded (n=103). The sensitivity analyses showed that the association between absolute CRF and prostate cancer risk remained significant (see online supplemental table s3). However, in the directional analysis, comparing the stable group to the decreased and increased group, models 2 and 3 were no longer significant.
Figure 3.
Cox proportional HRs with 95% CIs showing decrease (−3%) compared with stable (±3%) and increase (+3%). Model 1: adjusted for baseline fitness. Model 2: adjusted for baseline fitness, age, education and year of last test. Model 3: adjusted for baseline fitness, age, education, year of last test and body mass index. Model 4: adjusted for baseline fitness, age, education, year of last test, body mass index and smoking. *p<0.05; **p<0.01; ***p<0.001.
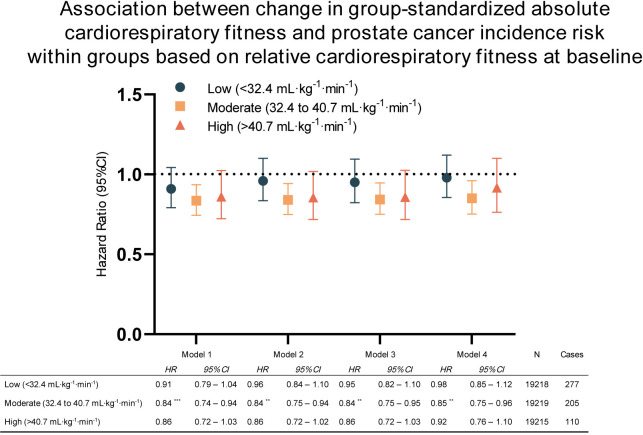
Figure 4 shows the results from subgroup analyses stratified by baseline relative CRF using group-standardised absolute fitness change. For the group with moderate baseline CRF (32.4–40.7 mL/kg/min), every SD increase in absolute CRF (L/min) reduced the risk of prostate cancer incidence by 16% for model 1. Significant associations remained even after adjusting for covariates in models 2 to 4. None of the models yielded significant associations for the low and high fitness groups. Even when using the percentage change in absolute fitness to analyse the data, the results remained consistent with the standardised analysis. However, in the case of the high fitness group, the least adjusted model (model 1) did show a significant inverse association with prostate cancer incidence, but this association was attenuated when adjusting for covariates (see online supplemental table s4).
Figure 4.
Cox proportional HRs with 95% CIs for the association between change in cardiorespiratory fitness and incidence of prostate cancer within low, moderate and high baseline cardiorespiratory fitness groups. Model 1: adjusted for baseline fitness. Model 2: adjusted for baseline fitness, age, education and year of last test. Model 3: adjusted for baseline fitness, age, education, year of last test, and body mass index. Model 4: adjusted for baseline fitness, age, education, year of last test, body mass index and smoking. *p<0.05; **p<0.01; ***p<0.001.
Change in absolute CRF was positively associated with changes in self-reported physical activity (see online supplemental table s5). Only 46 prostate cancer deaths were observed and changes in CRF were not associated with prostate cancer mortality (table 2).
Discussion
This is the largest study to examine the relationships between change in CRF and cancer incidence and mortality, and the first study to examine change in CRF specifically on prostate cancer incidence and mortality. The main finding of this study of over 57 000 men were that change in CRF (% L/min) was inversely associated with the risk of prostate cancer incidence and remained significant despite adjusting for relevant covariates. Subsequently, individuals with an annual increase in absolute CRF by 3% or more had a significantly lower risk of being diagnosed with cancer compared with individuals whose CRF was stable. When the participants were grouped by baseline CRF, the association between change in absolute CRF and prostate cancer incidence was only significant for the group with moderate baseline CRF. Changes in both absolute and relative CRF were not associated with prostate cancer mortality.
The findings in the present study contribute significantly to our knowledge of the relationship between CRF and prostate cancer as it is the first study to investigate change in CRF rather than CRF at a single time point, and to focus on prostate cancer specifically. Contrary to other cancer types, in the studies that have examined the relationship between CRF at one time point and prostate cancer incidence, the majority have reported an increased risk of prostate cancer incidence with increasing CRF, however there are studies that have reported the opposite.2 7–10 While the reason for these positive associations may not be due to a ‘true’ increased risk for those with higher CRF, but in part due to the influence of higher prostate cancer screening rates in men with higher CRF. This cannot be confirmed in the current analyses due to lack of information on screening rates.2
Analyses showed a reduced risk for prostate cancer incidence for those who increased their absolute CRF by more than 3%. In context, meta-analyses in adults have shown a person’s CRF can be improved by up to 16% in exercise interventions shorter than 1 year in duration.26 27 In the only other study to investigate change in CRF and cancer incidence, Robsahm et al 14 reported similar findings to the current study, that in comparison to a decrease in CRF (>5%), an increase in CRF (>5%) was associated with 19% lower cancer incidence.14
When the full group was stratified by baseline CRF, a reduced risk for prostate cancer incidence (−15%) was seen only in those with moderate CRF. While we can only speculate on the reason for this, it may be that improving one’s CRF from an already high baseline does not confer any discernible additional benefits. It may also be that the increases in CRF in people with low CRF were not sufficient to reach a potential threshold needed to contribute to lowering their risk of prostate cancer. In addition, residual confounding may account for the lack of associations. There is a possibility that the observed change in both the high and low groups may, in part, be a result of a regression to the mean. This means that individuals with initially extreme values, whether high or low, are more likely to have less extreme values on subsequent tests, due to random variation, rather than a change in their underlying fitness level. Consequently, it is plausible to suggest that only individuals in the moderate fitness group have the potential to show changes in both directions. This group’s fitness level may change, in a manner that is more reflective of actual change in their fitness, without the influence of regression to the mean.
Change in CRF was not associated with prostate cancer mortality in the current study. While no study has investigated the association between change in CRF and prostate cancer mortality, three studies have investigated the associations between CRF and cancer mortality in general.13–15 These studies in men found that higher CRF was associated with a 16%–30% lower risk of cancer mortality. Only 46 prostate cancer-related deaths were observed in the present study, and therefore the analyses may not have been sufficiently powered to detect any associations. There is also considerable competing mortality risk from other chronic diseases, such as cardiovascular and metabolic disease, associated with prostate cancer treatment side effects.28 It would therefore be of interest to examine the associations between CRF and mortality from these diseases in men diagnosed with prostate cancer.
The strengths of this study are the large sample size, the possibility to adjust for relevant covariates and the access to the national registry. Importantly, the study focused on a specific cancer type ensuring prostate specific conclusions. A novelty of the study was the objective measurement of CRF at two timepoints using standardised methodology.
Clinical implications
Where previous cancer prevention recommendations have focused on physical activity, the findings of this study provide further clarity on the associations between CRF and cancer incidence in an area of research with conflicting results. Although highly complex in nature, these investigations that aim to understand potential mechanisms behind the beneficial role of physical activity for preventing cancer will lead to more targeted prevention recommendations. The results of this study highlight the important role of supporting the general public to increase their CRF or aim to reach moderate fitness levels.
Limitations
While measuring VO2max directly during a maximal protocol with spirometry is the gold standard, it was not possible in this study. The submaximal protocol used in this study has however been reported to produce valid and reliable estimations compared with directly measured VO2max.16 Furthermore, the submaximal test used has not been validated in change studies, and this must be taken into consideration when translating the findings from this study. It is also important to note that there is a significant genetic component to both an individual’s CRF levels and cancer risk, which must be considered when interpreting the results. Furthermore, the recruited group were employed and therefore findings may not be generalisable to populations outside of this demographic, particularly people who are unemployed. Unfortunately, information on race or ethnicity was not recorded in the current data set, therefore it was not possible to conduct analyses among the different groups. In the present study, two time points were used to capture changes in CRF. It has been put forward that at least three time points are needed to establish the true trajectory of change.29 However, as relatively few individuals in the current study had data for three or more time points, we decided to include those with at least two time points. Finally, while there is an established link between systemic inflammation and advanced prostate cancer, the mechanism between less aggressive or localised prostate tumours is not fully understood and warrants future research.
Conclusions
In this large study of Swedish men, change in CRF was inversely associated with risk of prostate cancer incidence, but not prostate cancer mortality. Participants whose CRF increased by more than 3% had a significantly lower risk of prostate cancer incidence.
Footnotes
Twitter: @katebolam
Contributors: All authors have contributed with the conception and design of the work. KAB and EB-M contributed equally to this paper and share joint first authorship. SP contributed to the acquisition of the data. EB-M contributed by conducting the analyses and he and KAB interpreted the data. KAB and EB-M drafted the work, while all authors have revised the work and contributed with intellectual content. EE-B and EB-M monitored adherence to the design and statistical analyses. All authors had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analyses. All authors have approved the final version of the paper to be published and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work were appropriately investigated and resolved. E-EB was responsible for the overall content of the paper as the guarantor.
Funding: This study was funded by the Swedish Cancer Society, ref. 21 1837 Pj.
Competing interests: None declared.
Patient and public involvement: We did not involve patients or the public in the design or while conducting our research. In addition to scientific journal articles, the results of the study will be disseminated to the public through online print media and with the cancer community through the Swedish Cancer Society.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data may be obtained from a third party and are not publicly available. The data underlying the findings in this study are currently not publicly available as the original ethical approval application and the informed consent form did not include such direct, free access to the data. Data are stored by and can be requested from the HPI Health Profile Institute at support@hpi.se.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
The study was approved by the Stockholm Ethics Review Board (Dnr 2015/1864-31/2, 2016/9-32 and 2019-05711), and adhered to the Declaration of Helsinki.
References
- 1. McMichael AJ. Food, nutrition, physical activity and cancer prevention. Authoritative report from world cancer research fund provides global update. Public Health Nutr 2008;11:762–3. 10.1017/S1368980008002358 [DOI] [PubMed] [Google Scholar]
- 2. Shephard RJ. Physical activity and prostate cancer: an updated review. Sports Med 2017;47:1055–73. 10.1007/s40279-016-0648-0 [DOI] [PubMed] [Google Scholar]
- 3. Lavie CJ, Church TS, Milani RV, et al. Impact of physical activity, cardiorespiratory fitness, and exercise training on markers of inflammation. J Cardiopulm Rehabil Prev 2011;31:137–45. 10.1097/HCR.0b013e3182122827 [DOI] [PubMed] [Google Scholar]
- 4. Ortega R, Grandes G, Sanchez A, et al. Cardiorespiratory fitness and development of abdominal obesity. Prev Med 2019;118:232–7. 10.1016/j.ypmed.2018.10.020 [DOI] [PubMed] [Google Scholar]
- 5. Sui X, Sarzynski MA, Lee D-C, et al. Impact of changes in cardiorespiratory fitness on hypertension, dyslipidemia and survival: an overview of the epidemiological evidence. Prog Cardiovasc Dis 2017;60:56–66. 10.1016/j.pcad.2017.02.006 [DOI] [PubMed] [Google Scholar]
- 6. Solomon TPJ, Malin SK, Karstoft K, et al. Association between cardiorespiratory fitness and the determinants of glycemic control across the entire glucose tolerance continuum. Diabetes Care 2015;38:921–9. 10.2337/dc14-2813 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Byun W, Sui X, Hébert JR, et al. Cardiorespiratory fitness and risk of prostate cancer: findings from the aerobics center longitudinal study. Cancer Epidemiol 2011;35:59–65. 10.1016/j.canep.2010.07.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Crump C, Stattin P, Brooks JD, et al. Early-life cardiorespiratory fitness and long-term risk of prostate cancer. Cancer Epidemiol Biomarkers Prev 2020;29:2187–94. 10.1158/1055-9965.EPI-20-0535 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Kunutsor SK, Voutilainen A, Laukkanen JA. Cardiorespiratory fitness is not associated with reduced risk of prostate cancer: a cohort study and review of the literature. Eur J Clin Invest 2021;51:e13545. 10.1111/eci.13545 [DOI] [PubMed] [Google Scholar]
- 10. Reiter-Brennan C, Dzaye O, Al-Mallah MH, et al. Fitness and prostate cancer screening, incidence, and mortality: results from the henry ford exercise testing (FIT) project. Cancer 2021;127:1864–70. 10.1002/cncr.33426 [DOI] [PubMed] [Google Scholar]
- 11. Wang J, Mai XM, Sun YQ. Estimated cardiorespiratory fitness in relation to overall, breast and prostate cancer incidence: the Norwegian HUNT study. 2023. Available: https://reader.elsevier.com/reader/sd/pii/S1047279722003015?token=7DEAF20236961879873AC787B01645099018FF7FBC7D0DB98578B232ED7CADB2663B22E330ABF93A0D1BD78473274BE4&originRegion=eu-west-1&originCreation=20230126095058 [Accessed 26 Jan 2023]. [DOI] [PubMed]
- 12. Ahmadi MN, Lee I-M, Hamer M, et al. Changes in physical activity and adiposity with all-cause, cardiovascular disease, and cancer mortality. Int J Obes 2022;46:1849–58. 10.1038/s41366-022-01195-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Imboden MT, Harber MP, Whaley MH, et al. The association between the change in directly measured cardiorespiratory fitness across time and mortality risk. Prog Cardiovasc Dis 2019;62:157–62. 10.1016/j.pcad.2018.12.003 [DOI] [PubMed] [Google Scholar]
- 14. Robsahm TE, Heir T, Sandvik L, et al. Changes in midlife fitness, body mass index, and smoking influence cancer incidence and mortality: a prospective cohort study in men. Cancer Med 2019;8:4875–82. 10.1002/cam4.2383 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Zhang P, Sui X, Hand GA, et al. Association of changes in fitness and body composition with cancer mortality in men. Med Sci Sports Exerc 2014;46:1366–74. 10.1249/MSS.0000000000000225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Björkman F, Ekblom-Bak E, Ekblom Ö, et al. Validity of the revised Ekblom bak cycle ergometer test in adults. Eur J Appl Physiol 2016;116:1627–38. 10.1007/s00421-016-3412-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Ekblom-Bak E, Björkman F, Hellenius M-L, et al. A new submaximal cycle ergometer test for prediction of Vo2max. Scand J Med Sci Sports 2014;24:319–26. 10.1111/sms.12014 [DOI] [PubMed] [Google Scholar]
- 18. ASTRAND I. Aerobic work capacity in men and women with special reference to age. Acta Physiol Scand Suppl 1960;49:1–92. [PubMed] [Google Scholar]
- 19. Brooke HL, Talbäck M, Hörnblad J, et al. The Swedish cause of death register. Eur J Epidemiol 2017;32:765–73. 10.1007/s10654-017-0316-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. R Core Team . A language and environment for statistical computing; R foundation for statistical computing. Vienna, Austria; 2013. [Google Scholar]
- 21. van der Wal WM, Geskus RB. Ipw: an R package for inverse probability weighting. J Stat Softw 2011;43:1–23. 10.18637/jss.v043.i13 [DOI] [Google Scholar]
- 22. Therneau TM. TL (original S >R port and R Maintainer until 2009), Elizabeth A, Cynthia C [survival: survival analysis]. 2023. Available: https://CRAN.R-project.org/package=survival [Accessed 22 Feb 2023].
- 23. Wickham H, Chang W, Henry L, et al. ggplot2: create elegant data visualisations using the grammar of graphics. 2023. Available: https://CRAN.R-project.org/package=ggplot2 [Accessed 22 Feb 2023].
- 24. Kassambara A, Kosinski M, Biecek P, et al. Survminer: drawing survival curves using “ggplot2” 2021. Available: https://CRAN.R-project.org/package=survminer [Accessed 22 Feb 2023].
- 25. Mansournia MA, Collins GS, Nielsen RO, et al. A checklist for statistical assessment of medical papers (the CHAMP statement): explanation and elaboration. Br J Sports Med 2021;55:1009–17. 10.1136/bjsports-2020-103652 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Huang G, Gibson CA, Tran ZV, et al. Effect of controlled endurance exercise training on Vo2max changes in older adults: a meta-analysis. Med Sci Sports Exerc 2004;36:S142. 10.1249/00005768-200405001-00676 [DOI] [PubMed] [Google Scholar]
- 27. Scribbans TD, Vecsey S, Hankinson PB, et al. The effect of training intensity on Vo2max in young healthy adults: a meta-regression and meta-analysis. Int J Exerc Sci 2016;9:230–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Riihimäki M, Thomsen H, Brandt A, et al. What do prostate cancer patients die of Oncologist 2011;16:175–81. 10.1634/theoncologist.2010-0338 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Curran PJ, Bauer DJ. The disaggregation of within-person and between-person effects in longitudinal models of change. Annu Rev Psychol 2011;62:583–619. 10.1146/annurev.psych.093008.100356 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bjsports-2023-107007supp001.pdf (401.6KB, pdf)
Data Availability Statement
Data may be obtained from a third party and are not publicly available. The data underlying the findings in this study are currently not publicly available as the original ethical approval application and the informed consent form did not include such direct, free access to the data. Data are stored by and can be requested from the HPI Health Profile Institute at support@hpi.se.