Abstract
This case describes a unique presentation of a rare malignancy: giant melanoma. Due to the accessibility of healthcare in the United States, it is unusual for melanomas to grow to massive sizes without clinical intervention. In fact, an in-depth literature review elicited only a handful of similar cases. Giant malignant melanomas are typically defined by a cutoff size of no less than 10 cm in diameter. They often present with distant metastases and are highly invasive. Due to limited yet highly variable presentations, there is no standardized approach to treating this class of melanomas. We present a case with unique features not previously documented in similar cases that was ultimately treated with a novel approach.
Keywords: cutaneous malignancy, oncology imaging, cancer immunotherapy, giant melanoma, malignant melanoma metastasis
Introduction
Giant melanomas are a rare subtype of malignant melanomas defined as lesions measuring greater than 10 cm in diameter [1]. These are uncommon not only for their rare incidence in general but also because patients typically seek medical attention prior to these tumors reaching such a large size. With the exception of one case report in which a 14.5 x 10.4 cm mass grew within three months, most of these lesions grow over six months prior to reaching such massive sizes [2]. Giant melanoma patients are typically aware of their disease course and are even able to describe the initial onset and progression of their lesions due to their superficial etiology. Unfortunately, at the time of the patients' presentations, their lesions are often found to have already metastasized, as was the case with our patient. Upon his first encounter for evaluation of his lesion, he was found to have numerous metastases to his lungs as well as a suspicious distant lesion on his left arm.
Case presentation
A 63-year-old Caucasian male presented with an enlarging, painful left lower extremity mass of one-year duration. His late presentation was secondary to apprehension surrounding the COVID-19 pandemic. Associated symptoms included night sweats, fatigue, palpitations, anorexia, abdominal distention, and lower extremity edema. He also noted another mass on his left upper extremity that has been progressively growing in a similar fashion to the original mass, however, was much smaller and only became noticeable a few months ago.
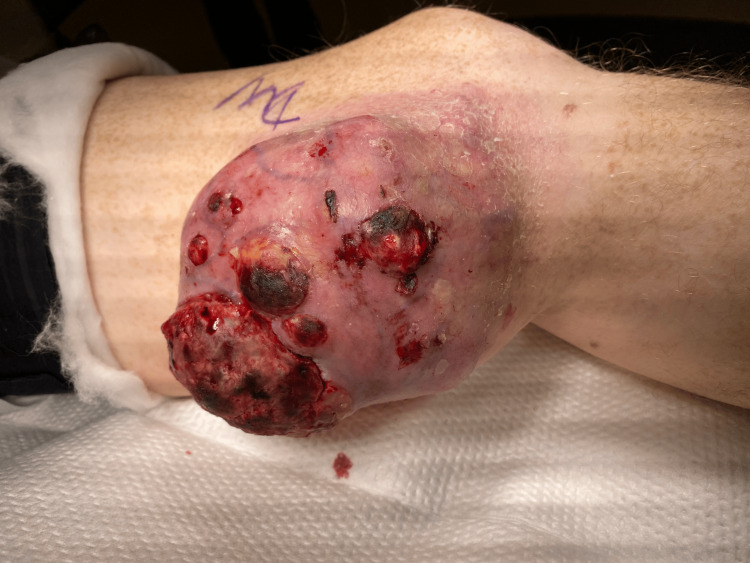
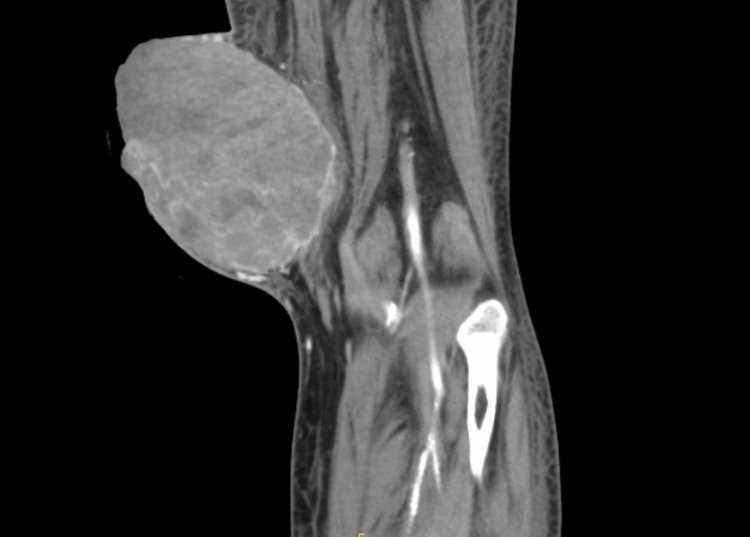
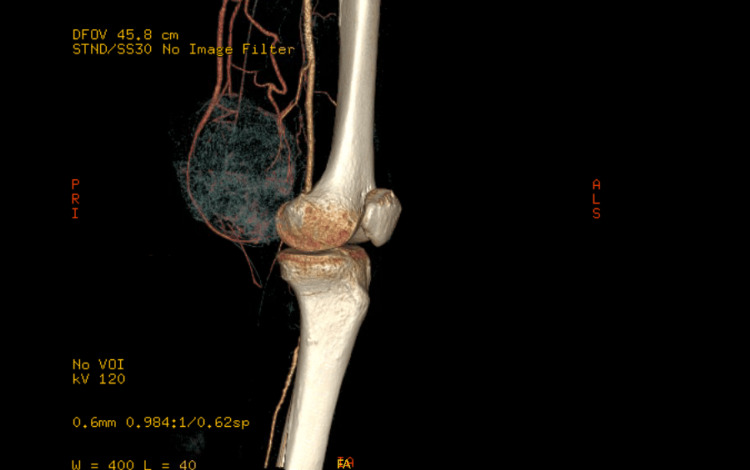
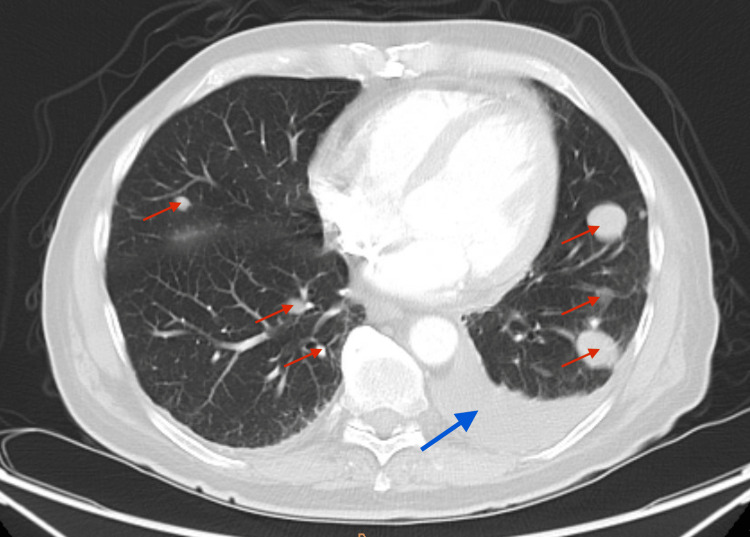
On physical examination, there was a large, pigmented, friable, ulcerated, vegetative mass approximately 10 cm in diameter on the left distal medial thigh with multiple ulcerations (Figure 1). A left lower extremity ultrasound revealed a large irregular-echogenicity and multilobulated mass measuring 9.6 x 10.2 x 10.8 cm as well as thrombi of left superficial femoral, popliteal, and posterior tibial veins. CT with contrast of the left leg demonstrated a large mass with no evidence of invasion of nearby bones (Figures 2, 3). The chest x-ray revealed multiple lung nodules consistent with metastatic disease, and a computed tomography angiography (CTA) of the chest demonstrated a left pleural effusion as well as innumerable well-circumscribed lung nodules (Figure 4). The CTA of the chest was repeated and demonstrated thrombi in the distal arterial branches of the left lower lobe with multiple pulmonary metastases measuring up to 2.5 cm on the right and 2.69 cm on the left. CT abdomen and pelvis with contrast showed splenomegaly and mild ascites. An excisional biopsy was performed, which revealed BRAF V600E+ malignant melanoma on pathology (Appendix). The patient was treated medically and was discharged with close outpatient follow-up with oncology.
Figure 1. Giant melanoma at the time of presentation.
Figure 2. Computed tomography angiography (CTA) of the left lower leg.
Figure 3. Computed tomography angiography (CTA) of the left leg with 3D reconstruction.
Figure 4. Computed tomography angiography (CTA) of the chest. Multiple bilateral pulmonary nodules (red arrows) and large left pleural effusion (blue arrow).
In the outpatient setting, he underwent a whole-body fluorodeoxyglucose-18 positron emission tomography (FDG-PET) scan, which revealed numerous bilateral pulmonary nodules that had increased in size compared to the prior CT of the chest, as well as new multiple hepatic metastases in both lobes of the liver. The PET scan also demonstrated an interval increase in the size of the left external iliac lymph node and left inguinal lymph nodes compared to prior CT scans. There were new osseous metastases within the proximal left femur and T2 vertebral body. The previously documented left upper extremity mass showed increased FDG activity as well. Splenomegaly, ascites, and pleural effusions mildly improved; however, a small pericardial effusion had worsened in the imaging studies. Brain MRI, bone marrow biopsy, and peripheral blood smear were unremarkable.
He was started on pembrolizumab and tolerated three courses of the immunotherapy drug. A repeat CT of the abdomen and pelvis at that time showed an increase in the size of the hepatic mass along the anterolateral aspect of the dome of the liver, along with a new adjacent necrotic peritoneal metastasis in the right paracolic gutter. The scan also showed a new subcutaneous nodule in the left anterior pelvic wall; however, the osseous metastases remained stable. There was also improvement in the remaining adenopathy and metastases described in the initial PET scan. The patient then received two additional courses of pembrolizumab for a total of five at the time of submission for publication. Given the patient’s worsening functional status (the Eastern Cooperative Oncology Group (ECOG) score declined from one to four since initiating therapy) and mixed results with pembrolizumab, there was consideration of halting treatment and deferring to hospice. At the time of submission, a repeat PET scan was ordered prior to making a decision to give a sixth course of the immunotherapy drug.
Discussion
An in-depth review of similar cases was done upon initial diagnosis of this patient revealing multiple unique factors related to this case not previously documented (Table 1).
Table 1. Review of similar cases.
LN, lymph node; TT, targeted therapy with dabrafenib (BRAF-i) and trametinib (MEK-i); CMO, comfort measures only; CT, chemotherapy.
| Location | Size | IHC | Staging | Metastases/positive lymph node | Tx | Result |
| Right forearm [3] | 14 × 7 × 12 cm | S100 (+) Melan-A (+) | T4bN0M0 Clark 5 Breslow 70 mm | NA | Surgery | Remission 4 years later |
| Antero-medial aspect of left arm [4] | 9 × 10 × 15 cm | S100 (+) Melan-A BRAF (+) | Stage IV Clark 5 Breslow >10 mm | Lung/left axillary LN | Surgery, TT | Death: brain metastasis |
| Lateral left arm [5] | 19 × 16 cm | S100 (+) Vimentin (+) SOX10 (+) V600E BRAF (+) | Stage IV | Left upper lung, left iliac wing | Surgery, TT | Remission 1 year later |
| Back [6] | 8 × 6 cm | N/A | T4N2M1c Breslow 48 mm | Back x2 (in-transit metastases)/bilateral axillary LN | Surgery | Death |
| Left upper arm [7] | 10 × 8 × 3 cm | N/A | Breslow 31 mm Clark 5 | Left axillary LN | Surgery | New metastases on vertebral column and ribs |
| Right upper arm [7] | 23 × 21 × 6 cm | S100 (+) HMB45 (+) tyrosinase (+) | Breslow 75 mm Clark 5 | Brain, right upper arm (in-transit)/right axillary LN | Surgery | Lost to follow-up |
| Anterior chest wall [8] | 15 × 13 × 2.5 cm | BRAF (–) V600E (–) | Stage IV | Brain, lungs, liver, L4/retroperitoneal, and perisplenic LN | CMO | Death 15 months later |
| Left gluteal region [9] | 18 × 16 × 5 cm | S100 (+) HMB45 (+) Ki67 (+) | Stage IV Clark 5 Breslow 48 mm | Lung/left inguinal LN | Surgery, declined further treatment | ICU 2 months later |
| Scalp [10] | 12 × 10 cm | HMB45 (+) | Stage IV | Brain, spleen, liver/hilar LN | Plan for: surgery, CT, possible TT | N/A |
| Left shoulder [11] | 20 × 20 cm | NRAS Q61L (+) S100 (+) Cyclin D1 (+) SOX 10 (+) MTF (+) HMB45 (+) MART1 (+) | T4N0M0 Clark 5 Breslow 70 mm | None | Surgery | Death from unknown cause; however, no recurrence 6 months later |
| Scalp [12] | 14.5 × 10.4 cm | N/A | T4bN3M1 Clark 5 Breslow 18 mm | Left parotid gland, left ear cartilage/left axillary LN | Surgery | Poor prognosis |
| Lower back [13] | 22 × 25 × 7 cm | S-100 (+) MART-1 (–) HMB-45 (rarely +) | Stage IV | None/left inguinal LN | Palliative surgery, CT | Disease regression |
Unique to our patient is the presentation of a lesion on the lower extremity. During our research into prior similar cases, we were unable to find any cases that presented lower than the buttocks. Also unique to this case was the finding of a positive BRAF V600E mutation. BRAF mutations, while common in melanomas, were only found in two other cases of giant melanomas [4,5]. This genetic mutation, in a patient naive to systemic therapy, the patient’s functional status, and the metastatic nature of presentation, made treating with pembrolizumab an appropriate initial therapeutic option [14-16]. The use of this immunotherapy was not seen in other cases of similar melanomas. After multiple courses of pembrolizumab, a variable response was achieved throughout the different lesions. These results warrant further investigation into pembrolizumab in the treatment of similar malignancies in the future.
Another unique aspect of this presentation was the presence of a second, presumably soft tissue malignancy on the left upper arm. It is unclear at this time whether this lesion is a metastasis of the primary lesion on the thigh or a second independent primary cancer. While it is less likely for a patient to have two primary tumors, giant melanomas have not previously been documented to spread distally to soft tissue. Melanomas, in general, have a known history of spreading to soft tissue through various methods. These metastases can be local (satellite lesions), regional (in-transit), or distant lesions typically seen on imaging as they are easily differentiated from local subcutaneous adipose tissue [2]. Satellite lesions are defined as secondary lesions in the skin or subcutaneous tissue within 2 cm of the primary tumor [17]. Satellite lesions are clearly separated by the normal dermis and are considered intralymphatic extensions of the primary mass. In-transit lesions are more distant, however still within the regional nodal basin. Any soft tissue lesions beyond these limitations are defined as distant metastasis. In the case of our patient, given the lesions in question are on a lower and upper extremity, it is safe to say that it can be classified as a distant metastasis if not a second primary lesion. Only two other cases of giant melanoma in our literature review demonstrated soft tissue metastasis. The first patient presented with a giant melanoma on the back with two lesions later described as in-transit lesions [6]. The second patient had a primary tumor in the right upper extremity that was surgically resected with a split-thickness skin graft [7]. Two months later, he was found to have in-transit metastases at the graft site and upper arm. Whether our patient’s second lesion turns out to be a distant metastasis or a second primary tumor, it remains a previously unseen feature of giant melanomas.
Conclusions
After appropriate work-up and treatment of our patient, as well as completing a comprehensive literature review, we conclude that the vast array of presentations of giant melanomas makes it challenging to devise a standardized treatment approach. Treatment modalities utilized to treat these patients varied, and while not all were successful, they were not incorrect. It is important to treat these patients uniquely in order to target therapy to their individual presentations. All aspects of the disease, including pathology, staging, sites of metastases, comorbidities, and functional status, should be taken into consideration when devising a multimodal approach to treat patients with giant malignant melanomas. Further investigation into similar cases is necessary to gain deeper knowledge of this rare and unique cancer.
Acknowledgments
This research was supported (in whole or in part) by HCA Healthcare and/or an HCA healthcare-affiliated entity. The views expressed in this publication represent those of the author(s) and do not necessarily represent the official views of HCA Healthcare or any of its affiliated entities.
Appendices
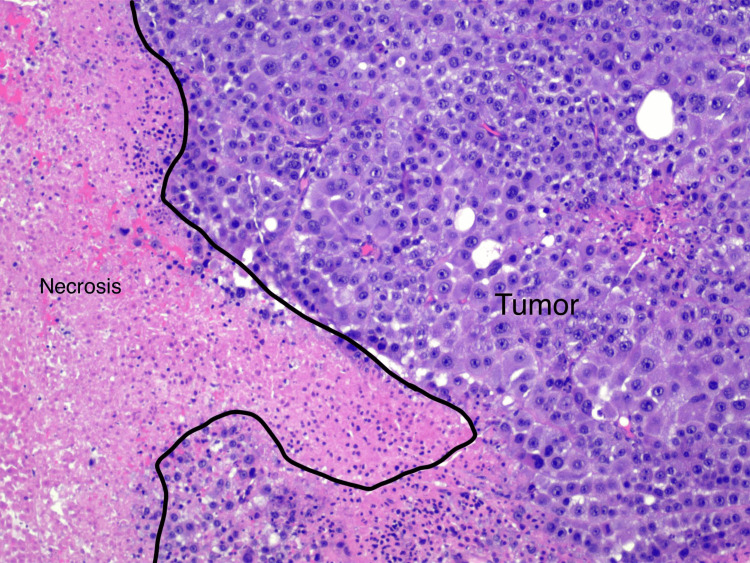
Figure 5. Hematoxylin and eosin stain (H&E), 10x. Sheets of large pleomorphic cells and necrosis. Cells have abundant cytoplasm, large nuclei with prominent nucleoli.
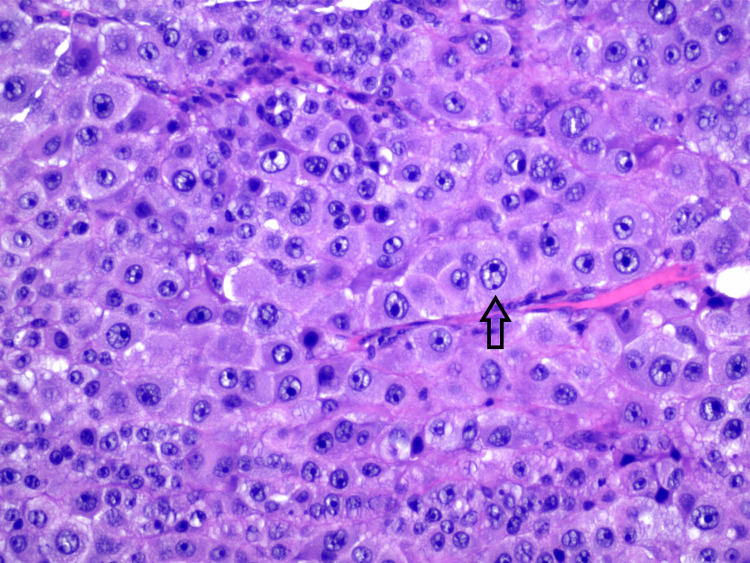
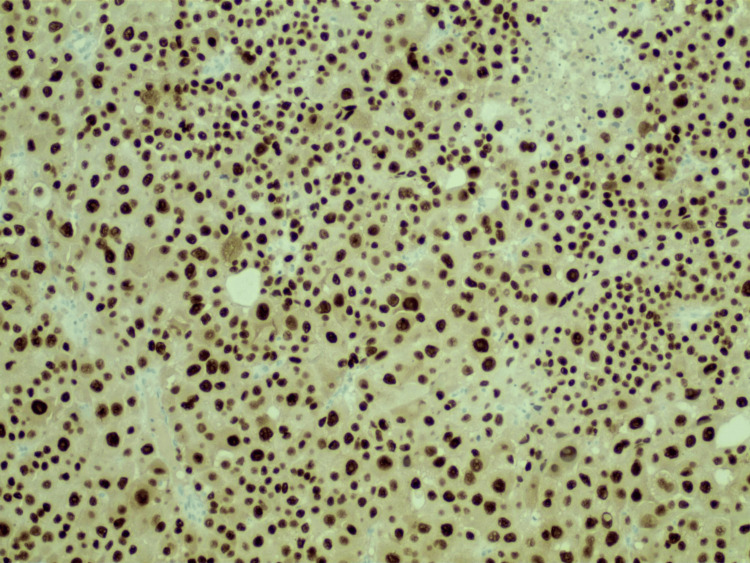
Figure 6. Hematoxylin and eosin stain (H&E), 40x. Cells with large pleomorphic nuclei, prominent nucleoli, and abundant amphophilic cytoplasm (arrow). No pigment, no squamous differentiation, and no glandular differentiation.
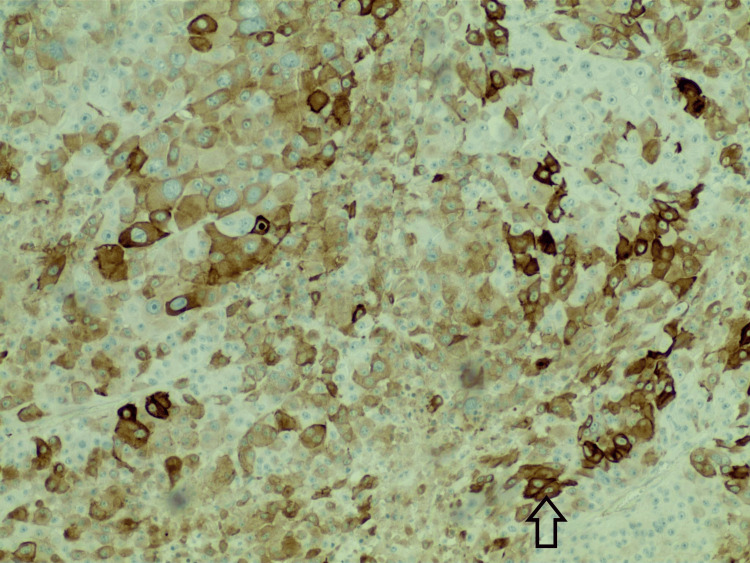
Figure 7. MART1 immunohistochemical stain. 10x. Cytoplasmic staining (arrow). Supports diagnosis of melanoma.
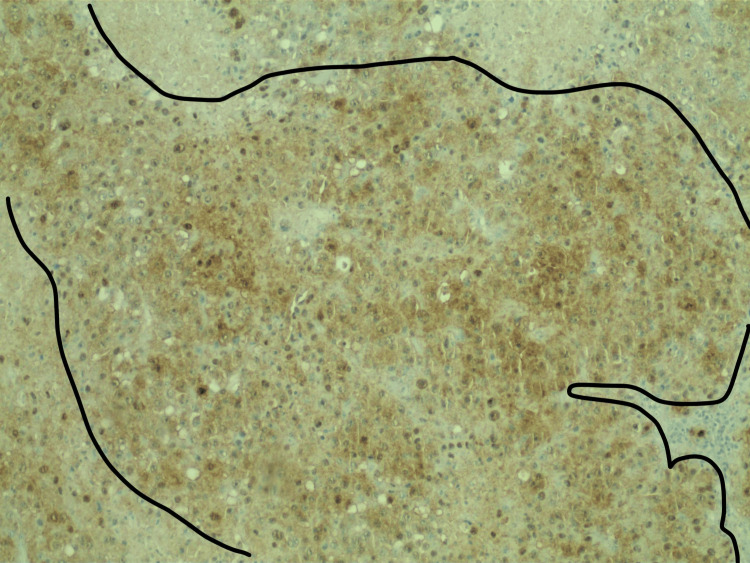
Figure 8. S100 immunohistochemical stain, 10x. Predominantly cytoplasmic staining, but some nuclear staining (within marked borders). Supports diagnosis of melanoma.
Figure 9. SOX10 immunohistochemical stain, 10x. Nuclear stain. Supports diagnosis of melanoma.
The authors have declared that no competing interests exist.
Author Contributions
Concept and design: Omar Al Zarkali, Ana VanDerWall
Acquisition, analysis, or interpretation of data: Omar Al Zarkali, Lakhvir Kaur, Amanda Marker
Drafting of the manuscript: Omar Al Zarkali
Critical review of the manuscript for important intellectual content: Omar Al Zarkali, Lakhvir Kaur, Amanda Marker, Ana VanDerWall
Supervision: Omar Al Zarkali, Ana VanDerWall
Human Ethics
Consent was obtained or waived by all participants in this study
References
- 1.Giant melanoma: novel problem, same approach. Grisham AD. South Med J. 2010;103:1161–1162. doi: 10.1097/SMJ.0b013e3181f3d756. [DOI] [PubMed] [Google Scholar]
- 2.Subcutaneous metastases from malignant melanoma: prevalence and findings on CT. Patten RM, Shuman WP, Teefey S. AJR Am J Roentgenol. 1989;152:1009–1012. doi: 10.2214/ajr.152.5.1009. [DOI] [PubMed] [Google Scholar]
- 3.Patient with giant upper limb melanoma presenting to a UK plastic surgery unit: differentials and experience of management. Honeyman CS, Wilson P. BMJ Case Rep. 2016;2016 doi: 10.1136/bcr-2015-212253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Unusual presentation of giant upper limb malignant melanoma. Khim OG, Leong JF, Sani MH, Mohamed Haflah NH. Cureus. 2021;13:0. doi: 10.7759/cureus.16996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.A case report of metastatic giant sarcomatoid melanoma with BRAF V600E mutation: a complete response to targeted therapy. Torresetti M, Brancorsini D, Morgese F, Cognigni V, Scalise A, Berardi R, Di Benedetto G. Oncotarget. 2020;11:3256–3262. doi: 10.18632/oncotarget.27701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Salvage surgery for a giant melanoma on the back. Kruijff S, Vink R, Klaase J. Rare Tumors. 2011;3:0. doi: 10.4081/rt.2011.e28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Giant cutaneous melanomas: evidence for primary tumour induced dormancy in metastatic sites? Tseng WW, Doyle JA, Maguiness S, Horvai AE, Kashani-Sabet M, Leong SP. BMJ Case Rep. 2009;2009 doi: 10.1136/bcr.07.2009.2073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Giant malignant melanoma of the anterior chest wall with widespread metastasis. Duma N, Khan AM, Kasimis B, Chang V. http://10.5348/Z09-2015-1-CR-1 J Case Rep Images Med. 2015;1:1–4. [Google Scholar]
- 9.Giant malignant melanoma: a case report. Kiyak MV, Yesilada AK, Sevim KZ, Usta U. https://www.prolekare.cz/en/journals/acta-chirurgiae-plasticae/2012-2-1/giant-malignant-melanoma-a-case-report-40119. Acta Chir Plast. 2012;54:59–61. [PubMed] [Google Scholar]
- 10.Giant malignant melanoma of scalp in a 15 year old boy with widespread metastases at presentation. Sarkar S, Maiti M, Barman NM, et al. IOSR J Dent Med Sci. 2016;15:73–75. [Google Scholar]
- 11.Giant malignant melanomas and their clinical implications: review of literature and case report. Gishen K, Maria DJP, Khanlari M, et al. https://www.clinicsinsurgery.com/open-access/giant-malignant-melanomas-and-their-clinical-implications-review-of-literature-and-case-report-2540.pdf Clin Surg. 2016;1:1096. [Google Scholar]
- 12.Giant scalp melanoma: a case report and review of the literature. Ching JA, Gould L. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3501267/ Eplasty. 2012;12:0. [PMC free article] [PubMed] [Google Scholar]
- 13.Massive nodular melanoma: a case report. Harting M, Tarrant W, Kovitz CA, Rosen T, Harting MT, Souchon E. Dermatol Online J. 2007;13:7. [PubMed] [Google Scholar]
- 14.Combination dabrafenib and trametinib versus combination nivolumab and ipilimumab for patients with advanced BRAF-mutant melanoma: the DREAMseq Trial-ECOG-ACRIN EA6134. Atkins MB, Lee SJ, Chmielowski B, et al. J Clin Oncol. 2023;41:186–197. doi: 10.1200/JCO.22.01763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Association of BRAF V600E/K mutation status and prior BRAF/MEK inhibition with pembrolizumab outcomes in advanced melanoma: pooled analysis of 3 clinical trials. Puzanov I, Ribas A, Robert C, et al. JAMA Oncol. 2020;6:1256–1264. doi: 10.1001/jamaoncol.2020.2288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pembrolizumab versus ipilimumab in advanced melanoma (KEYNOTE-006): post-hoc 5-year results from an open-label, multicentre, randomised, controlled, phase 3 study. Robert C, Ribas A, Schachter J, et al. Lancet Oncol. 2019;20:1239–1251. doi: 10.1016/S1470-2045(19)30388-2. [DOI] [PubMed] [Google Scholar]
- 17.Final version of 2009 AJCC melanoma staging and classification. Balch CM, Gershenwald JE, Soong SJ, et al. J Clin Oncol. 2009;27:6199–6206. doi: 10.1200/JCO.2009.23.4799. [DOI] [PMC free article] [PubMed] [Google Scholar]