Abstract
Existing controllers for robotic powered prostheses regulate the prosthesis speed, timing, and energy generation using predefined position or torque trajectories. This approach enables climbing stairs step-over-step. However, it does not provide amputees with direct volitional control of the robotic prosthesis, a functionality necessary to restore full mobility to the user. Here we show that proportional electromyographic (EMG) control of the prosthesis knee torque enables volitional control of a powered knee prosthesis during stair climbing. The proposed EMG controller continuously regulates knee torque based on activation of the residual hamstrings, measured using a single EMG electrode located within the socket. The EMG signal is mapped to a desired knee flexion/extension torque based on the prosthesis knee position, the residual limb position, and the interaction with the ground. As a result, the proposed EMG controller enabled an above-knee amputee to climb stairs at different speeds, while carrying additional loads, and even backwards. By enabling direct, volitional control of powered robotic knee prostheses, the proposed EMG controller has the potential to improve amputee mobility in the real world.
I. Introduction
Most knee prostheses available to individuals with leg amputations are energetically passive devices [1] that cannot generate the net-positive energy required to climb stairs [2]. Due to this limitation, individuals with above-knee amputations must climb stairs one step at a time, leading with their non-amputated leg [3]. This unnatural gait pattern is slow, inefficient, and requires substantial strength and stamina in the non-amputated leg. Robotic powered knee prostheses aim to address this limitation using embedded actuators. During stair climbing, when the foot is in contact with the step, powered knee prostheses can actively generate torque to lift the user’s body mass against gravity [4] [5] [6] [7] [8]. When the foot is off the ground, the embedded actuators can actively control the prosthesis movements to clear the stairs. However, to accomplish this task in the real world, powered prostheses need controllers that can accurately coordinate the action of the prosthesis with the movements of the user.
Most stair controllers rely on information from mechanical sensors embedded in the prosthesis, such as inertial measurement units (IMU) and ground reaction force sensors, to determine the action of the prosthesis. These stair controllers can provide the user with a limited sense of agency over the prosthesis by reacting to the movements of the user’s residual limb. For example, if the user’s residual limb moves faster, the powered knee moves faster. If the user pushes more into the prosthesis, the powered knee generates torque more quickly. This indirect volitional control enables above-knee amputees to climb stairs with different geometry, at their preferred cadence, and using different gait patterns [9] [10]. However, with this control approach, users cannot voluntarily control the movements of the prosthetic joints independently from the movement of their residual limb. Therefore, controllers based only on mechanical sensors cannot provide users with direct volitional control of the prosthesis.
Neural signals encode the intention of movement. Therefore, controllers based on neural signals from the residual-limb muscles can provide users with direct volitional control of their powered prosthesis. Electromyography (EMG) allows for direct control of the prosthesis movements both during non-weight-bearing activities [11] [12] and weight-bearing activities such as walking [13] and stair ascent [14]. However, these EMG controllers require EMG signals from antagonist muscles, which have considerable issues with cross talk and co-activation. As a result, individuals with above-knee amputations struggle to differentiate extension and flexion, which makes the control of a powered knee prosthesis quite challenging even after multiple weeks of training [13].
Limiting EMG-control to regulate the action of powered protheses in stance only can address some of these issues by requiring only one EMG sensor [15]. However, this approach reduces the user’s sense of agency because the users are only in control of the prosthesis when the foot is on the ground. Also, this approach does not allow for stair climbing. Sonomyography can hypothetically address some issues of EMG but has only been used with lower-limb amputees in offline studies [16] [17]. Thus, we do not have a direct volitional controller that enables amputees to climb stairs naturally with a powered knee prosthesis.
Limiting EMG-control to regulate the action of powered protheses in stance only can address some of these issues by requiring only one EMG sensor [15]. However, this approach reduces the user’s sense of agency because the users are only in control of the prosthesis when the foot is on the ground. Also, this approach does not allow for stair climbing. Sonomyography can hypothetically address some issues of EMG but has only been used with lower-limb amputees in offline studies [16] [17]. Thus, we do not have a direct volitional controller that enables amputees to climb stairs naturally with a powered knee prosthesis.
In this paper, we propose a volitional EMG controller for stair ambulation with a powered knee prosthesis. The proposed controller uses a single EMG electrode placed inside of the socket on the back of the residual limb. The EMG electrode records the activation of the residual hamstrings to control the prosthesis torque both in flexion and extension. The EMG signal is translated into torque using a proportional gain that depends on the leg position and the interaction with the ground. Variable damping is used to slow the prosthesis down, depending on the orientation of the residual thigh and the position of the knee. The goal of this paper is to show that the proposed EMG controller can provide an above-knee amputee subject with direct volitional control of a robotic powered knee prosthesis during stair climbing.
II. Methods
A. EMG Control
The proposed controller is inspired by analysis of electromyography and joint biomechanics of nonamputee individuals ascending stairs. The EMG signal from the residual biceps femoris (BF) muscle is mapped to flexion or extension torque depending on the physical interaction with the ground, as measured by an embedded ground reaction force (GRF) sensor. Specifically, the EMG signal is mapped to flexion torque when the prosthesis is off the ground and to extension torque when the prosthesis is on the ground (Supplementary Video). The transition between flexion and extension torque happens continuously as a function of the GRF as shown in Fig. 1(a, c). Moreover, the proportional EMG gain changes continuously based on the knee position () and the global orientation of the residual-limb thigh segment (), which is determined by an onboard IMU. Virtual damping is added to the EMG torque to slow down the knee movements during critical gait phases based on the knee and thigh position as shown in Fig. 1(e, f).
Fig. 1.
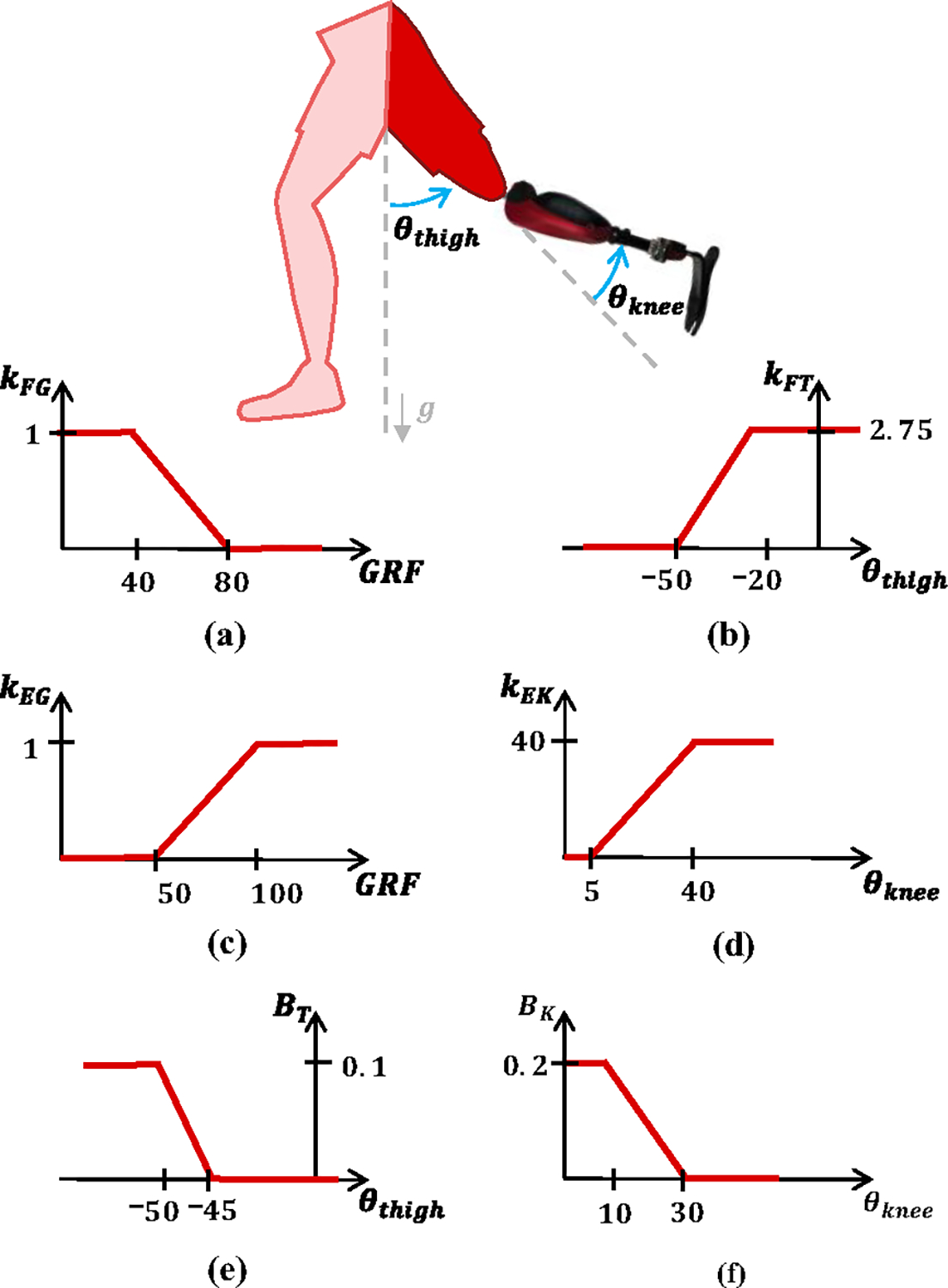
Controller Diagram. (a) , flexion torque gain as a function of GRF, (b) , flexion torque gain as a function of thigh position, (c) , extension torque gain as a function of GRF, (d) , extension torque gain as a function of knee position, (e) , damping gain as a function of thigh position, and (f) , damping gain as a function of knee position.
Combining proportional EMG control and virtual damping, the total desired knee torque is the sum of four different parts: EMG flexion torque (), EMG extension torque , thigh damping () and knee damping , as shown in (1) and Fig. 1.
| (1) |
Knee flexion torque is necessary during stair ascent at the beginning of swing (i.e., foot off the ground) to quickly move the prothesis shank backwards and provide toe clearance between the prosthetic foot and the next stair. This flexion torque is generated voluntarily by the user with proportional EMG control. Specifically, the EMG flexion torque is defined by multiplying the EMG signal by two gains, and , as shown in (2).
| (2) |
ensures that flexion torque is only provided when the prosthetic foot has minimal-to-no contact with the ground, allowing EMG flexion torque during late stance and swing only. To this end, is 1 when the GRF < 40 N, and 0 when GRF > 80 N and increases linearly between 0 and 1 as the GRF decreases from 80 to 40 N (Fig. 1(a)).
changes the sensitivity of the EMG torque as a function of the global orientation of the residual-limb thigh segment . When the residual limb is directly below the subject , the proportional gain is at its maximum, increasing the sensitivity of flexion torque relative to the EMG signal. As the thigh moves in front of the subject , the proportional gain decreases linearly to 0, decreasing the sensitivity of flexion torque at the end of swing phase. As a result, the sensitivity of the EMG torque is high at the beginning of swing, when flexion torque is needed to clear the step, and zero at the end of swing, when the prosthetic foot hovers over the step before the subject places their foot down. To achieve this effect, depends on the global orientation of the residual-limb thigh segment , which is determined by an onboard IMU. As shown in Fig. 1(b), increases linearly from 0 to 1 as increases from −50° to −20°. If , then . If , then .
Knee extension torque is necessary during stair ascent to provide the energy to extend the knee joint and lift the subject up the stair. This extension torque is voluntarily generated by the user using proportional EMG control. Specifically, the EMG extension torque is determined by multiplying the EMG signal by two gains, and , as shown in (3).
| (3) |
ensures that the proportional EMG extension torque is only provided when the foot is in contact with the ground. To achieve this result, increases linearly from 0 to 1 when the GRF increases from 50 to 100 N (Fig. 1(c)). Moreover, is 1 when the GRF > 100 N, and 0 when GRF < 50 N. A threshold of 50 N ensures that the user loads the prosthesis sufficiently before the prosthesis starts extending.
regulates the sensitivity of the EMG torque based on the knee joint position , so that the extension torque is high at the beginning of stance, when high torque is needed, and zero at the end of stance, when no torque is needed. increases linearly as the knee joint position increases between 5° and 40° as shown in (d). If the , then . If the , then .
Virtual damping is added to the proportional EMG torque to slow down and smooth the movements of the powered knee prosthesis during specific phases of stair ascent. Two separate terms determine the virtual damping, the thigh damping torque and the knee damping torque .The thigh damping torque depends on two gains, and as shown in (4).
| (4) |
(Fig. 1(a)) is the same gain used in (2) for the EMG flexion torque and varies between zero and one depending on the GRF so that the thigh damping torque is zero when the prosthetic foot is in contact with the ground. The second gain (), depends on the thigh orientation. increases linearly from 0 to 0.1 as the thigh orientation decreases from −45° to −50° (Fig. 1(e)). Moreover, if , then . If , then . Therefore, the thigh damping is zero at the beginning of swing, when the thigh is vertical and the prosthetic knee needs to flex quickly to clear the step. Moreover, the thigh damping is at its highest value at the end of swing when the thigh is in front of the torso and the subject is ready to place their foot on the next step.
The knee damping torque slows down the knee joint as it gets closer to full extension regardless of the GRF. It only provides damping against knee extension () and depends on only one gain (), as shown in (5).
| (5) |
depends on the knee position . If , then . If , then . When increases linearly from zero to its maximum value (0.2) as the knee position decreases (Fig. 1(f). Essentially, the knee damping torque slows down the knee joint as it gets close to full extension regardless of the ground reaction force.
B. Utah Bionic Leg
For this study, we used the Utah Bionic Knee in combination with a passive foot/ankle (Fig. 2) [18]. The Utah Bionic Knee is a self-contained, battery-operated powered knee prosthesis that can replicate the biomechanical functions of the human knee during ambulation. It is the lightest powered knee in the field. The passive ankle/foot prosthesis is an off-the-shelf prosthesis (Taleo, Ottobock) retrofitted with a custom instrumented pyramid adapter capable of measuring vertical ground reaction forces [19]. The knee and ankle modules are connected using a standard 30-mm pylon, cut to the correct height for the user. The instrumented pyramid adapter communicates with the powered knee using a digital communication line that runs inside the pylon.
Fig. 2.
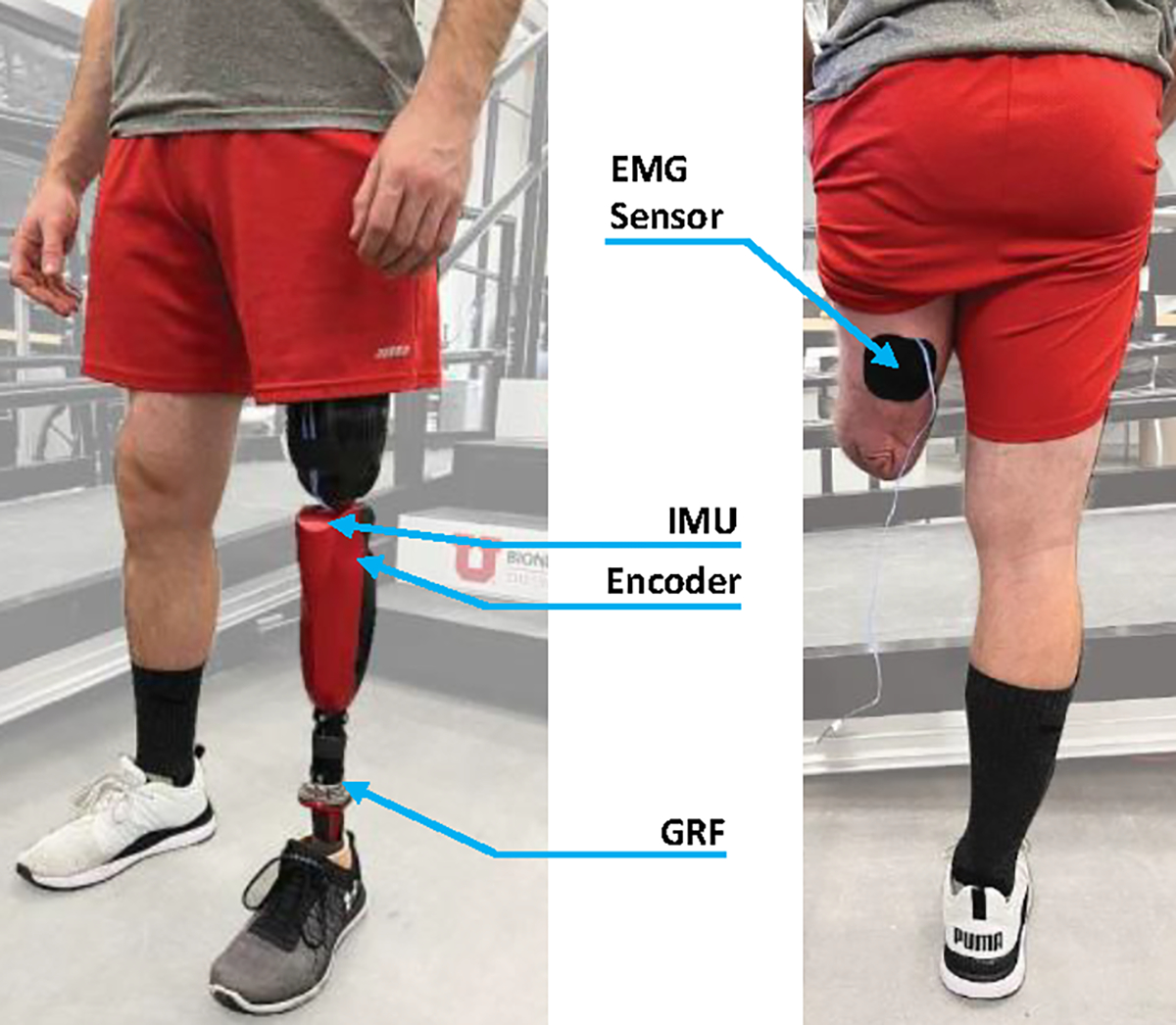
View of subject wearing the Utah Bionic Leg (left). Back view of subject with EMG sensor location on the residual Biceps Femoris muscle (right).
C. Experimental Protocol
For this study, we recruited one individual with an above knee amputation (32-year-old, male, 77.25 kg, 1.8 m, 6 years post amputation). Prior to the study, the subject gave written informed consent to participate, including consent to use photos and videos. A certified prosthetist fit and aligned the powered prosthesis, which was used in combination with the user’s socket. All protocols for this study were approved by the University of Utah Institutional Review Board.
Before data acquisition, we determined an appropriate location for the EMG sensor (13E202=60, Ottobock) on the user’s residual biceps femoris (hamstring) by palpating the back of the participant’s residual limb while we asked him to contract his residual-limb muscles. Then, we cleaned the skin area and placed the EMG sensor following SENIAM guidelines [20]. After the EMG sensor was placed, we set the manual gain of the sensor by asking the subject to contract and visually inspecting that the output signal peaked between 1 V and 2 V. After that, we secured the electrode to the skin with kinesiology tape and the subject put their socket back on.
After the EMG setup was completed, the subject donned the powered prosthesis, and a certified prosthetist adjusted the alignment as necessary. After the prosthesis was aligned, the subject started familiarizing himself with the EMG controller. During this familiarization phase, we adjusted two parameters of the proposed EMG controller as needed for the subject to perform stair ascent comfortably. Specifically, we adjusted to ensure the user received enough flexion torque to clear the step. Moreover, we adjusted to provide sufficient extension torque to climb the stair. All other parameters were set prior to the experiment using able body biomechanics and were not tuned specifically to the subject. The familiarization lasted about 15 minutes and was performed on a staircase with four steps.
After the familiarization period, we moved to a staircase with 11 steps and started the data acquisition. The protocol for data acquisition included several stair ascent tests. First, we asked the subject to climb stairs as fast and as slow as he could. Next, we asked the subject to climb stairs at his self-selected speed, with and without a 30 lb. backpack (13.61 kg). Finally, we asked the subject to climb stairs backwards. Each test included two flights of stairs with 11 steps in each flight. The subject rested between tests. All data was collected in one experimental session that lasted less than two hours.
D. Data Analysis/Processing
Data were saved by the onboard electronics of the Utah Bionic Leg at 500 Hz and processed offline using MATLAB. First, we filtered the data with a zero-phase fourth order lowpass Butterworth filter with a cutoff frequency of 7 Hz. We then segmented the data from heel strike to heel strike using the GRF signal from the onboard GRF sensor. After segmentation, we resampled and time-normalized the data. Finally, we calculated the average knee position and torque trajectories for each test. For some tests, we also computed the peak torque and velocity during stance and swing phases.
III. Results
A. Stair Ascent at Fast and Slow Speeds
Using the proposed EMG controller, the participant was able to climb stairs step-over-step at fast and slow speeds (Fig. 3, Supplementary Video (b)). The average stride duration was 3.9 ± 0.2 s during slow and 1.6 ± 0.2 s during fast stair climbing. During slow stair climbing, the average knee velocity was 216.3 ± 44.3 deg/s in stance and 272.8 ± 62.0 deg/s in swing (Fig. 3). During fast stair climbing, the average knee velocity was 337.0 ± 37.7 deg/s in stance and 363.8 ± 48.1 deg/s in swing (Fig. 3). Thus, the peak knee velocity increased by 55.8% in stance and 33.4% in swing during fast stair climbing, compared to slow stair climbing.
Fig. 3.
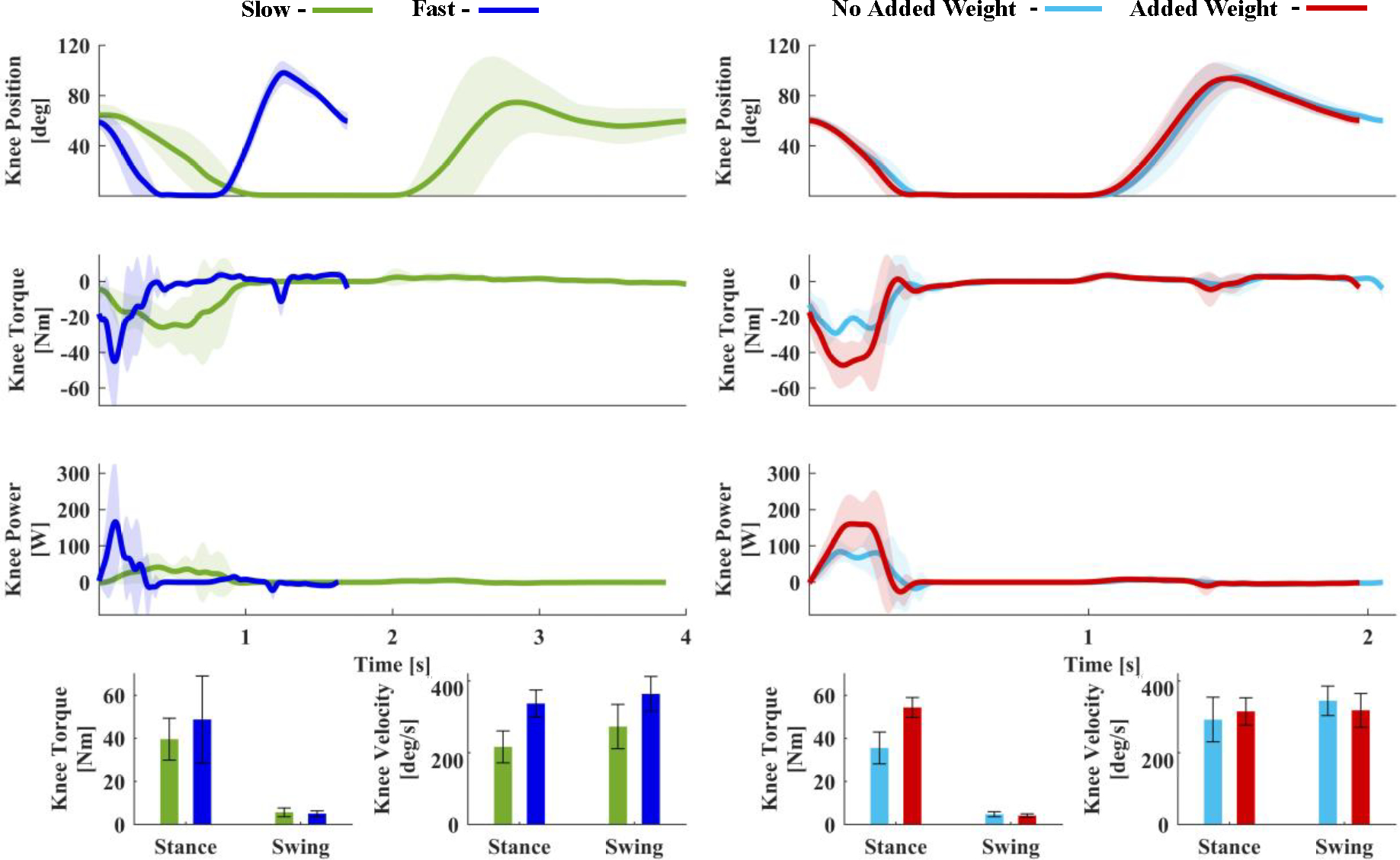
Joint kinematics for two tests. The left column compares stair ascent at slow (green) and fast (blue) speeds with respect to time. The right column compares stair ascent with (red) and without (blue) added weight with respect to time. The first, second, and third rows of both columns show the knee position [deg], knee torque [Nm], and knee power [W], respectively. The left bar graph in each column show maximum extension torque during stance and maximum flexion torque during swing. The right bar graph in each column represents maximum knee velocity during stance and swing. Shaded areas and error bars denote mean ± one standard deviation.
The kinetic analysis also shows substantial differences between climbing at two speeds. When climbing stairs at slow speed, the powered knee generated a peak extension torque of 39.6 ± 9.8 Nm in stance and a peak flexion torque of 5.7 ± 2.0 Nm in swing (Fig. 3). In comparison, when climbing stairs at the fast speed, the powered knee generated 50.4 ± 17.4 Nm of extension torque in stance and 5.5 ± 1.1 Nm of flexion torque in swing. Therefore, the extension torque increased by 27.1% during fast stair climbing compared to slow stair climbing. In contrast, there was only a 4.4% difference in the flexion torque when climbing stairs at a fast and slow speed. The combination of faster knee velocities and higher knee torques resulted in marked difference in peak knee power during stance. The participant received an average of 93.9 ± 32.5 W while climbing slowly, and 204.8 ± 106.6 W while climbing fast. Thus, the peak power increased 118.1% during fast climbing compared to slow stair climbing.
B. Stair Ascent With and Without Added Weight
The individual was able to climb stairs at his self-selected speed without and with a 30-lb. backpack (Fig. 3, Supplementary Video (c)). There were no visible differences in the knee position trajectory between weight conditions. The average knee velocities during stance and swing also had similar values both without and with added weight. During stance, the average knee velocities were 292.1 ± 62.1 deg/s without added weight and 315.1 ± 37.9 deg/s with added weight, respectively. During swing, the average knee velocities were 344.8 ± 41.1 deg/s without and 318.2 ± 47.0 deg/s with added weight (Fig. 3). In contrast, we observed substantial differences in knee torque and speed during stance. Specifically, the powered knee generated 35.6 ± 7.3 Nm of extension torque without added weight and 54.3 ± 4.6 Nm with added weight. The prosthesis knee power also increased in stance from 124.3 ± 48.6 W without added weight to 220.8 ± 25.0 W with added weight. Thus, the torque and power increased in stance by 52.8% and 77.6%, respectively, when the subject carried extra weight, compared to without added weight. Not surprisingly, the powered prosthesis generated similar amounts of torque in swing when the weight of subject is not supported by the prosthesis. Specifically, the flexion torque in swing was 4.8 ± 1.1 Nm without added weight and 4.2 ± 0.8 Nm with added weight.
C. Backward Stair Ascent
With the proposed EMG controller, the participant was able to climb stairs backwards (Fig. 4, Supplementary Video (d)). The comparison of forward and backward stair climbing shows substantial differences in the knee kinematics during both stance and swing, although the biggest difference was in swing. Specifically, in swing, the peak of the knee position was 98.1 ± 7.8° during forward stair climbing and 71.0 ± 4.4° during backward stair climbing. Interestingly, forward strides showed a peak swing torque of 4.3 ± 1.5 Nm, whereas backward strides showed 8.6 ± 1.4 Nm of peak swing torque. Thus, in swing, the flexion torque was double during backward strides compared to forward strides. The observed increase in swing torque corresponded directly with an increase in the EMG activation when the user flexed their muscles to get enough clearance to lift the prosthesis into the correct position to climb stairs backward. Specifically, the peak EMG was 1.6 ± 0.6 V for forward strides and 3.7 ± 0.3 V for backward strides (Fig. 4).
Fig. 4.
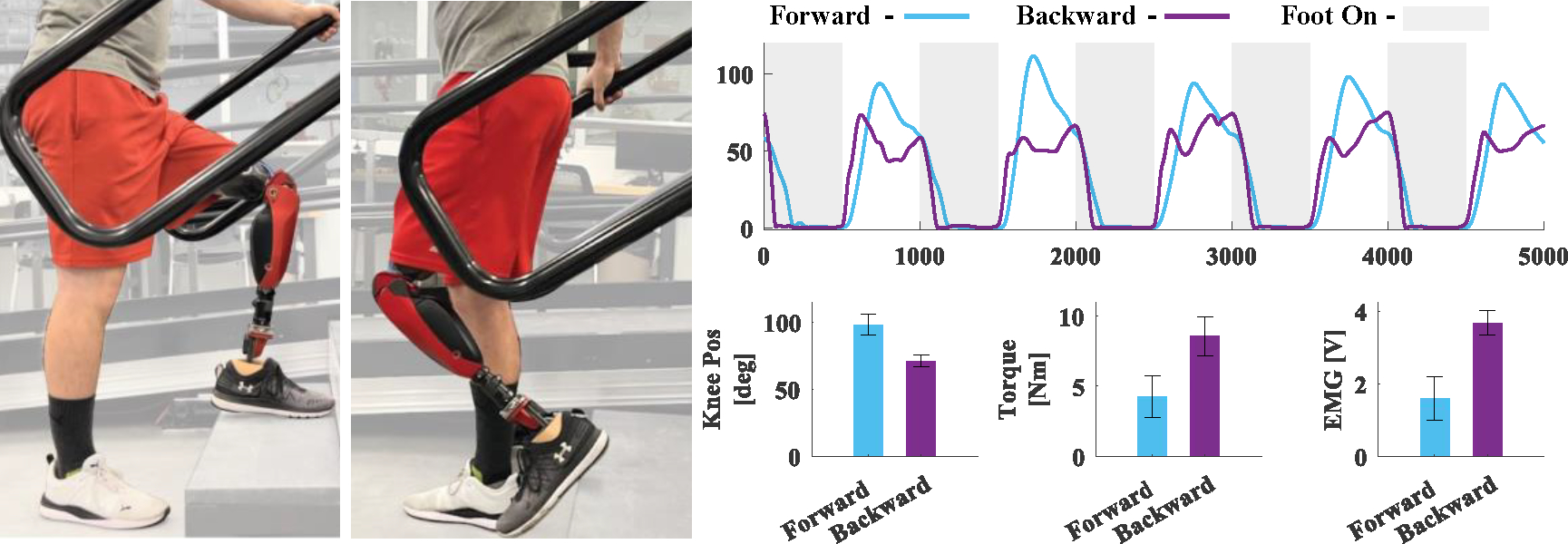
Knee position kinematics averaged across 5 consecutive stair ascent strides. Blue represents forward stair ascent, purple represents backward stair ascent. Grey shaded regions represent when the foot is in contact with the ground and white shaded regions represent when the foot is not in contact with the ground. The bottom three graphs represent, from left to right, maximum Knee Position [deg], Flexion Torque [Nm], EMG [V]. Bar height and error bars show mean ± one standard deviation. The bargraphs are calculated only when the user’s foot was not in contact with the ground.
IV. Discussion
A. Significance
Robotic powered prosthesis controllers need to coordinate the movements of the prosthesis with the movements of the user to properly function in the real world. Most prosthesis controllers attempt to coordinate with the user by reacting to the movements of the user’s residual limb, which are detected by embedded mechanical sensors like GRF sensors and IMUs. This approach is functional but cannot give users direct volitional control of the prosthesis, which is necessary to replicate the function of the healthy human body.
Volitional EMG control has the potential to address this issue. However, crosstalk and co-activation of antagonist muscles reduces the functionality and intuitiveness of existing antagonistic EMG controllers, making them less functional and intuitive than controllers based solely on mechanical sensors. This study suggests that amputees can learn how to regulate their muscle activation patterns to the extent necessary for stairs ambulation [21], but more work is necessary to specifically address learning. Other studies have shown that volitional EMG controllers worked during non-weight-bearing activities but did not result in viable gait controllers [12].
In this paper we show that the proposed EMG controller can provide an above-knee amputee with direct volitional control of a powered knee prosthesis during stair climbing using a single EMG sensor. The proposed controller enabled an above-knee amputee to directly modulate the knee torque during both stance and swing in order to climb stairs faster and slower, and with or without additional weight. Moreover, EMG torque control during swing enabled the subject to correctly place the foot on the step when ascending stairs both forwards and backwards. Thus, this study provides the first demonstration that EMG control can enable voluntary actuation of a powered prosthesis during stair ascent.
Voluntary modulation of knee extension torque is necessary to climb stairs under different conditions. This study shows that by voluntarily increasing the knee extension torque generated by the prosthesis, an above-knee amputee can climb stairs at substantially different speeds (31 to 75 steps/min) and carry a large additional weight (30 lbs.).
The knee extension torque generated by the powered prosthesis is voluntarily controlled by the user through the activation of their biceps femoris. The biceps femoris is a biarticular hamstring muscle in nonamputee individuals. However, after above-knee amputation, the biceps femoris loses its ability to flex the knee and becomes a monoarticular hip extensor [22]. Most importantly, the biceps femoris is naturally active during stance in stair climbing for both non-amputees and amputees [23], when both hip extension torque and knee extension torque are required to counteract gravity. Therefore, users may not need to learn a new muscle activation pattern to use the proposed EMG controller.
Voluntary modulation of knee flexion is also necessary to climb stairs under different conditions. By controlling the amount of flexion torque as a function of EMG signal from the biceps femoris, the proposed EMG controller enables the subject to control the position of their prosthesis in swing, correctly placing their foot on the next step. As anticipated, we see a large difference in EMG activations and resulting flexion torques when comparing forward and backward climbing, with the latter being substantially larger. Interestingly, when the subject climbed stairs forwards at different speeds (Fig. 3), we did not observe a meaningful difference in swing knee flexion torque despite the large difference in knee peak velocities. This result could be explained by the passive dynamics of the prosthesis dominating this movement. In other terms, due to the combined effect of gravity and inertial torque, climbing stairs backwards requires higher flexion torque and the subject can voluntarily generate that torque with the proposed controller.
B. Limitations
This study shows encouraging outcomes but there are notable limitations. This study only included one subject. Due to the large variability in residual limb length and muscle strength after an amputation, we cannot draw conclusions about the proposed controller’s performance with the broader amputee population. This study also lacks a biomechanical analysis of the contralateral leg and upper body, which limits our understanding of the effects of the proposed EMG controller on full-body motion during gait. Because this study did not compare the proposed EMG controller to non-EMG controllers, we cannot fully assess the benefits and drawbacks of volitional EMG control. Finally, the proposed EMG controller can only regulate stair ascent movements. It is not clear whether other ambulation activities can be achieved using one EMG sensor as in the proposed controller.
V. Conclusions
This study presents a volitional EMG controller for stair ascent with a powered knee prosthesis. Experiments with one individual with an above-knee amputation show that the proposed volitional EMG controller enables climbing stairs at different speeds, while carrying additional loads, and even climbing stairs backward. Future work should focus on testing with a broader population, as well as extending this direct volitional controller to more ambulation activities.
Supplementary Material
Acknowledgments
*Research supported by the National Institutes of Health under grant R01 HD098154/HD/NICHD, the National Institute for Occupational Safety and Health under grant T42/CCT810426, the US Department of Defense under Grant W81XWH2110037 and the National Science Foundation under grant 1925371.
Contributor Information
Suzi Creveling, Department of Mechanical Engineering and the Robotics Center at the University of Utah..
Marissa Cowan, Department of Mechanical Engineering and the Robotics Center at the University of Utah..
Liam M. Sullivan, Department of Mechanical Engineering and the Robotics Center at the University of Utah.
Lukas Gabert, Department of Mechanical Engineering and the Robotics Center at the University of Utah.; Rocky Mountain Center for Occupational and Environmental Health.
Tommaso Lenzi, Department of Mechanical Engineering and the Robotics Center at the University of Utah.; Rocky Mountain Center for Occupational and Environmental Health. Department of Biomedical Engineering at the University of Utah
References
- [1].Liang W et al. , “Mechanisms and component design of prosthetic knees: A review from a biomechanical function perspective,” Front Bioeng Biotechnol, vol. 10, Sep. 2022, doi: 10.3389/fbioe.2022.950110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [2].McFadyen BJ and Winter DA, “An integrated biomechanical analysis of normal stair ascent and descent,” J Biomech, vol. 21, no. 9, pp. 733–744, Jan. 1988, doi: 10.1016/0021-9290(88)90282-5. [DOI] [PubMed] [Google Scholar]
- [3].Aldridge Whitehead JM, Wolf EJ, Scoville CR, and Wilken JM, “Does a Microprocessor-controlled Prosthetic Knee Affect Stair Ascent Strategies in Persons With Transfemoral Amputation?,” Clin Orthop Relat Res, vol. 472, no. 10, pp. 3093–3101, Oct. 2014, doi: 10.1007/s11999-014-3484-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Lawson BE, Varol HA, Huff A, Erdemir E, and Goldfarb M, “Control of Stair Ascent and Descent With a Powered Transfemoral Prosthesis,” IEEE Transactions on Neural Systems and Rehabilitation Engineering, vol. 21, no. 3, pp. 466–473, May 2013, doi: 10.1109/TNSRE.2012.2225640. [DOI] [PubMed] [Google Scholar]
- [5].Azocar AF, Mooney LM, Duval J-F, Simon AM, Hargrove LJ, and Rouse EJ, “Design and clinical implementation of an open-source bionic leg,” Nat Biomed Eng, vol. 4, no. 10, pp. 941–953, Oct. 2020, doi: 10.1038/s41551-020-00619-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Lenzi T, Cempini M, Hargrove L, and Kuiken T, “Design, development, and testing of a lightweight hybrid robotic knee prosthesis,” Int J Rob Res, vol. 37, no. 8, pp. 953–976, Jul. 2018, doi: 10.1177/0278364918785993. [DOI] [Google Scholar]
- [7].Tran M, Gabert L, Cempini M, and Lenzi T, “A Lightweight, Efficient Fully Powered Knee Prosthesis With Actively Variable Transmission,” IEEE Robot Autom Lett, vol. 4, no. 2, pp. 1186–1193, Apr. 2019, doi: 10.1109/LRA.2019.2892204. [DOI] [Google Scholar]
- [8].Wolf EJ, Everding VQ, Linberg AL, Schnall BL, Czerniecki JM, and Gambel JM, “Assessment of transfemoral amputees using C-Leg and Power Knee for ascending and descending inclines and steps,” The Journal of Rehabilitation Research and Development, vol. 49, no. 6, p. 831, 2012, doi: 10.1682/JRRD.2010.12.0234. [DOI] [PubMed] [Google Scholar]
- [9].Hood S, Gabert L, and Lenzi T, “Powered Knee and Ankle Prosthesis With Adaptive Control Enables Climbing Stairs With Different Stair Heights, Cadences, and Gait Patterns,” IEEE Transactions on Robotics, vol. 38, no. 3, pp. 1430–1441, Jun. 2022, doi: 10.1109/TRO.2022.3152134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Hood S, Creveling S, Gabert L, Tran M, and Lenzi T, “Powered knee and ankle prostheses enable natural ambulation on level ground and stairs for individuals with bilateral above-knee amputation: a case study,” Sci Rep, vol. 12, no. 1, p. 15465, Sep. 2022, doi: 10.1038/s41598-022-19701-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].Hargrove LJ, Simon AM, Lipschutz R, Finucane SB, and Kuiken TA, “Non-weight-bearing neural control of a powered transfemoral prosthesis,” J Neuroeng Rehabil, vol. 10, no. 1, p. 62, 2013, doi: 10.1186/1743-0003-10-62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Ha KH, Varol HA, and Goldfarb M, “Volitional Control of a Prosthetic Knee Using Surface Electromyography,” IEEE Trans Biomed Eng, vol. 58, no. 1, pp. 144–151, Jan. 2011, doi: 10.1109/TBME.2010.2070840. [DOI] [PubMed] [Google Scholar]
- [13].Hoover CD, Fulk GD, and Fite KB, “The Design and Initial Experimental Validation of an Active Myoelectric Transfemoral Prosthesis,” J Med Device, vol. 6, no. 1, Mar. 2012, doi: 10.1115/1.4005784. [DOI] [Google Scholar]
- [14].Hoover CD, Fulk GD, and Fite KB, “Stair Ascent With a Powered Transfemoral Prosthesis Under Direct Myoelectric Control,” IEEE/ASME Transactions on Mechatronics, vol. 18, no. 3, pp. 1191–1200, Jun. 2013, doi: 10.1109/TMECH.2012.2200498. [DOI] [Google Scholar]
- [15].Hunt G, Hood S, and Lenzi T, “Stand-Up, Squat, Lunge, and Walk With a Robotic Knee and Ankle Prosthesis Under Shared Neural Control,” IEEE Open J Eng Med Biol, vol. 2, pp. 267–277, 2021, doi: 10.1109/OJEMB.2021.3104261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Murray R, Mendez J, Gabert L, Fey NP, Liu H, and Lenzi T, “Ambulation Mode Classification of Individuals with Transfemoral Amputation through A-Mode Sonomyography and Convolutional Neural Networks,” Sensors, vol. 22, no. 23, p. 9350, Dec. 2022, doi: 10.3390/s22239350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].Mendez J, Murray R, Gabert L, Fey NP, Liu H, and Lenzi T, “A-Mode Ultrasound-Based Prediction of Transfemoral Amputee Prosthesis Walking Kinematics Via an Artificial Neural Network,” IEEE Transactions on Neural Systems and Rehabilitation Engineering, pp. 1–1, 2023, doi: 10.1109/TNSRE.2023.3248647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Tran M, Gabert L, Hood S, and Lenzi T, “A lightweight robotic leg prosthesis replicating the biomechanics of the knee, ankle, and toe joint,” Sci Robot, vol. 7, no. 72, Nov. 2022, doi: 10.1126/scirobotics.abo3996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Gabert L and Lenzi T, “Instrumented Pyramid Adapter for Amputee Gait Analysis and Powered Prosthesis Control,” IEEE Sens J, vol. 19, no. 18, pp. 8272–8282, Sep. 2019, doi: 10.1109/JSEN.2019.2920179. [DOI] [Google Scholar]
- [20].Hermens HJ, Freriks B, Disselhorst-Klug C, and Rau G, “Development of recommendations for SEMG sensors and sensor placement procedures,” Journal of Electromyography and Kinesiology, vol. 10, no. 5, pp. 361–374, Oct. 2000, doi: 10.1016/S1050-6411(00)00027-4. [DOI] [PubMed] [Google Scholar]
- [21].Fleming A, Stafford N, Huang S, Hu X, Ferris DP, and Huang H. (Helen), “Myoelectric control of robotic lower limb prostheses: a review of electromyography interfaces, control paradigms, challenges and future directions,” J Neural Eng, vol. 18, no. 4, p. 041004, Aug. 2021, doi: 10.1088/1741-2552/ac1176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Ranz EC, Wilken JM, Gajewski DA, and Neptune RR, “The influence of limb alignment and transfemoral amputation technique on muscle capacity during gait,” Comput Methods Biomech Biomed Engin, vol. 20, no. 11, pp. 1167–1174, Aug. 2017, doi: 10.1080/10255842.2017.1340461. [DOI] [PubMed] [Google Scholar]
- [23].Geiger DE, Behrendt F, and Schuster-Amft C, “EMG Muscle Activation Pattern of Four Lower Extremity Muscles during Stair Climbing, Motor Imagery, and Robot-Assisted Stepping: A Cross-Sectional Study in Healthy Individuals,” Biomed Res Int, vol. 2019, pp. 1–8, Mar. 2019, doi: 10.1155/2019/9351689. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


