Abstract
STUDY QUESTION
Do different boys with different types of cryptorchidism exhibit different anogenital distances (AGDs)?
SUMMARY ANSWER
Length of AGD seemed to differ in different groups of patients with cryptorchidism.
WHAT IS KNOWN ALREADY
AGD, which is used as an indicator of prenatal androgen action, tends to be shorter in boys with cryptorchidism compared to unaffected boys. Shorter AGDs have also been reported in boys with hypospadias, in men with poor semen quality, and in men with testicular cancer.
STUDY DESIGN, SIZE, DURATION
A prospective descriptive cohort study was performed using data from consecutively selected boys with cryptorchidism (n = 169) operated in a single center over a period of 3 years (September 2019 to October 2022).
PARTICIPANTS/MATERIALS, SETTING, METHODS
AGD was measured in 169 infant boys, at 3 to 26 months of age, during anesthesia with a vernier caliper measuring the distance from the anus to the base of the scrotum (AGDAS) and from the anus to the anterior base of the penis (AGDAP) in two body positions according to the methods by ‘The Infant Development and the Environment Study’ (TIDES) and ‘Cambridge Baby Growth Study’, resulting in four mean values per patient (TIDES AGDAS/AP and Cambridge AGDAS/AP). Normal values for AGD by age were set by our hospital Department of Growth and Reproduction based on a large cohort of healthy infant boys (n = 1940). Testicular biopsies were performed at orchidopexy as a clinical routine. The germ cell number (G/T) and type Ad spermatogonia number (AdS/T) per cross-sectional tubule of at least 100 and 250 tubules, respectively were measured and related to normal samples. Blood samples were obtained by venipuncture for measuring serum LH, FSH, and inhibin B. They were analyzed in our hospital Department of Growth and Reproduction where the normal reference was also established. Correlations between the four mean AGD measurements for each boy were evaluated by Spearman rank correlation analyses. The AGD measurement of every boy was transferred to the multiple of the median (MoM) of the normal AGD for age and named MoM AGD.
MAIN RESULTS AND THE ROLE OF CHANCE
There were 104 boysoperated for unilateral, and 47 boys operated for bilateral, undescended testes, whereas 18 boys had vanished testis including one boy with bilateral vanished testes. Only 6% of cases with vanished testes had a MoM AGD higher than the normal median compared to 32% with undescended testes (P < 0.05). MoM AGD increased with the age at surgery for boys with vanished testis (Spearman r = 0.44), but not for boys with undescended testes (Spearman r = 0.14). Boys with bilateral cryptorchidism had longer AGDs and more often had hypogonadotropic hypogonadism than boys with unilateral cryptorchidism (P < 0.005) and (P < 0.000001).
LIMITATIONS, REASONS FOR CAUTION
Although being the largest published material of AGD measurements of infant boys with cryptorchidism, one limitation of this study covers the quite small number of patients in the different groups, which may decrease the statistical power. Another limitation involves the sparse normal reference material on G/T and AdS/T. Finally, there are currently no longitudinal studies evaluating AGD from birth to adulthood and evaluating childhood AGD in relation to fertility outcome. Our study is hypothesis generating and therefore the interpretation of the results should be regarded as exploratory rather than reaching definite conclusions.
WIDER IMPLICATIONS OF THE FINDINGS
The study findings are in agreement with literature as the total included group of boys with cryptorchidism exhibited shorter than normal AGDs. However, new insights were demonstrated. Boys with vanished testis had shorter AGDs compared to unaffected boys and to boys with undescended testes. This finding challenges the current concept of AGD being determined in ‘the masculinization programming window’ in Week 8 to 14 of gestation. Furthermore, boys with bilateral cryptorchidism had longer AGDs and more often had hypogonadotropic hypogonadism than boys with unilateral cryptorchidism, suggesting that the lack of fetal androgen in hypogonadotropic hypogonadism is not that significant.
STUDY FUNDING/COMPETING INTEREST(S)
No external funding was used and no competing interests are declared.
TRIAL REGISTRATION NUMBER
The trial was not registered in an ICMJE-recognized trial registry.
Keywords: anogenital distance, testes, cryptorchidism, androgen, gonadotropins, germ cells
Introduction
Normal masculinization is dependent upon androgen secretion by Leydig cells. Based on rodent studies, it has been hypothesized that androgens initially act during a critical ‘masculinization programming window’ most likely between gestational week 8 to 14 in humans, ensuring normal male reproductive development (Larsen, 1997; Welsh et al., 2008; Jain et al., 2018). Among many other targets, androgens mediate the development of the perineal tissues determining the distance between the anus and genital tubercle (Schwartz et al., 2019). Thus, the anogenital distance (AGD), referring to the distance between the anus and external genitals, is used as a measure of androgen action in both experimental animals and humans (Thankamony et al., 2016). In rodents, AGD is one of the most conspicuous sexually dimorphic structures of the external genitals and is used in practice to determine the sex. The distance is approximately twice as long in male as in female rodents, and the same difference in AGD is found in humans (Thankamony et al., 2009; Priskorn et al., 2018). In toxicity rodent studies, AGD serves as a sensitive marker of in utero deficient androgen exposure, suggesting a link between reductions in AGD and androgen deprivation during the ‘masculinization programming window’ in male reproductive abnormalities (Welsh et al., 2008; Dean and Sharpe, 2013). In humans, a shorter AGD has been reported in boys with cryptorchidism or hypospadias (Swan et al., 2005; Hsieh et al., 2008; Thankamony et al., 2009; Jain and Singal, 2013; Thankamony et al., 2014; Abbas and Ali, 2021), in men with poor semen quality (Eisenberg et al., 2011; Mendiola et al., 2011; Priskorn et al., 2019), and in men with testicular cancer (Priskorn et al., 2021).
Cryptorchidism is characterized by a lack of one or both testes, or an incomplete descent of one or both testes, and a risk of abnormal germ cell development (Cortes et al., 1995; Cortes, 1998). Besides a congenital dysgenesis of the testis, there may be an endocrine imbalance or an insufficient hypothalamic–pituitary–gonadal axis, i.e. a hypogonadotropic hypogonadism (a condition with a reduced number of germ cells and/or low inhibin B level despite of that no elevated gonadotropins) (Hadziselimovic, 1997; Hadziselimovic et al., 2004; Hadziselimovic and Hoecht, 2008; Thorup et al., 2012; Hildorf, 2022).
Although androgens are disputably important in inguinoscrotal testicular descent and it has been found that boys with cryptorchidism have reduced AGD, a relationship between AGD and the type of cryptorchidism has not yet been reported. The aim of this study was to assess the relationship of AGD with the type of cryptorchidism (e.g. vanished testis, unilateral or bilateral cryptorchidism) and findings regarding testicular biopsies and serum reproductive hormone levels obtained at orchidopexy. Our study is hypothesis generating and therefore the interpretation of the results should be regarded as exploratory rather than reaching definite conclusions. We seek to find out whether different groups of boys with cryptorchidism exhibit differences in length of AGD.
Materials and methods
Patients
Between September 2019 and October 2022, boys with cryptorchidism were included consecutively by one surgeon performing surgery (J.T.) at the Department of Pediatric Surgery, Copenhagen University Hospital Rigshospitalet, Denmark, in a prospective study. Included were boys who were up to 26 months of age, diagnosed with non-syndromic cryptorchidism (no other diagnoses), scheduled for initial orchidopexy, and had not undergone previous hernia repair nor received hormonal treatment. An ascending testis was characterized as a testis which had been scrotal early in life and later ascended into a cryptorchid position. The diagnosis of vanished testis was verified with laparoscopy with or without surgical exploration. These boys had a remnant (testicular regression syndrome with no presence of Sertoli, germ or Leydigcells) or complete absence of testicular remnants (blind ending vessels and spermatic cord intra-abdominally, near to the closed internal annular ring, or more distally).
AGD measurements
Measurements of AGDs were performed during anesthesia with a vernier caliper measuring the distances from anus to the base of scrotum (AGDAS) and from anus to anterior base of the penis (AGDAP) in two body positions according to the methods by ‘The Infant Development and the Environment Study’ (TIDES) (Barrett et al., 2014) and ‘Cambridge Baby Growth Study’ (Cambridge) (Prentice et al., 2016). The TIDES method places the patient in supine position with the legs held in a frog-like position; the knees flexed, and slightly pushed toward to shoulders, while the Cambridge method places the patient in supine position with the feet on the surface and light pressure exerted onto the thighs. In each body position, every distance was measured three times resulting in four mean values per patient (TIDES AGDAS/AP and Cambridge AGDAS/AP). The aforementioned methods of measurements used in the present study equals the methods of measurements used in the study establishing our normal values (Fischer et al., 2020). Normal values for AGD by age were set by the Department of Growth and Reproduction based on a large cohort of healthy infant boys (n = 1940) and was extensively described by Fischer et al. (2020) including cross reference open access raw data. Inter-observer discrepancy related to AGD measurements between the investigator performing measurements at surgery (J.T.) and the investigator who established the normal reference values (M.B.F.) was <5%.
Histology
Testicular biopsies were performed at orchidopexy as a clinical routine (Hildorf, 2022). No testicular biopsies were taken from the descended testes in boys with unilateral cryptorchidism. Each specimen was fixed in Stieve’s medium, cut into 2 μm sections, and put on coated slides to be stained with hematoxylin and eosin and incubated with primary antibodies for D2-40 (1:25, M3619, Dako, Glostrup, Denmark), CD99 (1:100, 12E7; Dako), and placenta-like-alkaline phosphatase (1:200, PL8-F6; Biogenex, Fremont, USA). First, the histopathology of all specimens was examined by the same pathologist (E.C.-L.). Secondly, the germ cell number (G/T) and type Ad spermatogonia number (AdS/T) per cross-sectional tubule of at least 100 and 250 tubules, respectively, were measured. Values of G/T and AdS/T were measured blindly by three observers (A.E.H., S.H., and D.C.). Inter-observer discrepancy between the investigators was <5%. There were no surgical complications related to performance of the testicular biopsies. Reference values for the median of G/T according to age were defined by a meta-analysis by Masliukaite et al. (2016) including biopsies from normal prepubertal boys (n = 334). AdS/T was defined in accordance with the literature (Hildorf, 2022).
Hormonal assays
Blood samples were obtained by venipuncture between 8:00 and 12:00 am during anesthesia. In brief, after centrifugation, the serum samples were stored at −20°C until analysis. Serum LH and FSH concentrations were analyzed by time-resolved fluoroimmunometric assays (AutoDELFIA, PerkinElmer, Turku, Finland; B031-101 and B017-201, respectively). For both assays, the limits of detection (LOD) were 0.05 IU/l, and the intraassay coefficients of variation (CVs) were <5%.
Inhibin B was measured using a double antibody enzyme-immunometric assay (Inhibin B GenII ELISA, A81303, Beckman Coulter, Brea, CA, USA) with a LOD of 3 pg/ml and a CV of 11%. The blood samples were analyzed by the Department of Growth and Reproduction, Copenhagen University Hospital Rigshospitalet, the same laboratory as that used for the normal reference materials (Johannsen et al., 2018; Busch et al., 2021; Laboratory methods and reference, Department of Growth and Reproduction, Rigshospitalet: https://hormlabvejl.regionh.dk/Metodeliste.asp (access date October 2022)). Inhibin B levels between the normal 2.5 and 97.5 percentile were defined as normal, whereas levels below 2.5 percentile were defined as low. Reference values for LH and FSH according to age were defined by Busch et al. (2021) up to 400 days of age and afterwards by Johannsen et al. (2018). FSH and LH values higher than the 97.5 percentage were defined as high.
Analysis and statistics
Correlations between the four mean AGD measurements for each boy were evaluated by Spearman rank correlation analyses. Afterwards, it was decided to use one of the measurements for all analyses. Because AGD increases during infancy, age-related normal values are important. Therefore, the AGD measurement of every boy was transferred to the multiple of the median (MoM) of the normal AGD for age and named MoM AGD. The ratio between the patients’ result and the median AGD appropriate for the age of the boy was estimated as MoM AGD based on multiple median values from healthy boys. For example, if the AGD of the boy was exactly at the median of healthy boys, at the same age, the MoM AGD was 1, but if the AGD was 90% of normal median the MoM AGD was 0.90.
Equivalently, the ratio between the patient’s inhibin B result and the normal reference median inhibin B level according to age was estimated as MoM Inhibin B. Moreover, MoM G/T was estimated for each boy based on ratio between mean G/T (bilateral cryptorchidism) or G/T (unilateral cryptorchidism) and age-related normal median G/T value.
Based on G/T and hormonal findings at orchidopexy, the boys with undescended testes were subdivided into three groups according to our previously published papers (Thorup et al., 2012; Hildorf, 2022). Group 1 had an elevated FSH level; Group 2 had no compensatory increase in FSH despite reduced G/T and/or impaired inhibin B (bilateral cryptorchidism), or impaired inhibin B (unilateral cryptorchidism); and Group 3 had normal FSH, and normal G/T and inhibin B (bilateral cryptorchidism), or normal inhibin B (unilateral cryptorchidism).
A correlation coefficient (Spearman r) of 0.20 to 0.39 was considered as a weak correlation, 0.40 to 0.59, as a moderate correlation, and 0.60 to 0.79, as a strong correlation. A weak correlation was only reported in cases of a P-value <0.05.
Chi-square tests, Mann–Whitney’s U-test, Kruskal–Wallis test, and Fischer’s exact test were used to assess statistical significance between values between different groups. A two-sided P-value <0.05 was considered statistically significant. All statistical analyses were performed using GraphPad Prism (GraphPad Software, San Diego, CA, USA) or OpenEpi (https://www.openepi.com/Menu/OE_Menu.htm).
Ethics
Ethics approval was obtained from the institutional ethics committee from the Capital Region (KF-01299830 and H-18063061) and patient consent was obtained in all instances.
Results
A total of 169 boys with cryptorchidism were included. Of these, 104 boys were treated for unilateral, and 47 boys were treated for bilateral, undescended testes. There were 17 boys who were diagnosed with one vanished testis and one boy was diagnosed with bilateral vanished testes (Table 1).
Table 1.
Boys with cryptorchidism who underwent measurement of the ano-scrotal distance (AGD) at time of surgery classified in 18 with vanished testes and 151 with undescended testes.
| Boys with vanished testis | Boys with undescended testis | P-value | |
|---|---|---|---|
| Number | 18# | 151§ | |
| Bilateral cryptorchidism | 6% (1/18) | 31% (47/151) | P = 0.023 |
| Age at surgery (months)* | 14 (4–24) | 11 (3–26) | P = 0.118 |
| Birth weight (g)* | 3405 (3051–3940) | 3540 (640–4910) | P = 0.476 |
| MoM AGD* | 0.89 (0.74–1.05) | 0.90 (0.52–1.35) | P = 0.102 |
| MoM AGD higher than normal median (=1) | 6% (1/18) | 32% (49/151) | P = 0.018 |
| Inhibin B (MoM)* | 0.64 (0.01–1.39) | 0.70 (0.17–1.50) | P = 0.848 |
| Inhibin B low (<2.5th percentile) | 12% (2/17) | 15% (22/151) | P = 0.784 |
| FSH* | 0.75 (0.41–142) | 0.79 (0.08–3.2) | P = 0.305 |
| FSH high (>97.5th percentile) | 41% (7/17) | 23% (34/150) | P = 0.092 |
| LH* | 0.15 (0.05–56.8) | 0.23 (0.05–4.82) | P = 0.466 |
| LH high (>97.5th percentile) | 12% (2/17) | 5% (7/150) | P = 0.220 |
MoM, multiple of the median; MoM AGD, MoM ano-scrotal distance.
Median (range).
One without hormonal values.
One without FSH and two without birth weight.
We observed strong correlations between TIDES AGDAS and TIDES AGDAP (Spearman r = 0.68) and between TIDES AGDAS and Cambridge AGDAS/AP (0.87 and 0.71, respectively). Afterwards, TIDES AGDAS was selected for the further analyses using the term AGD.
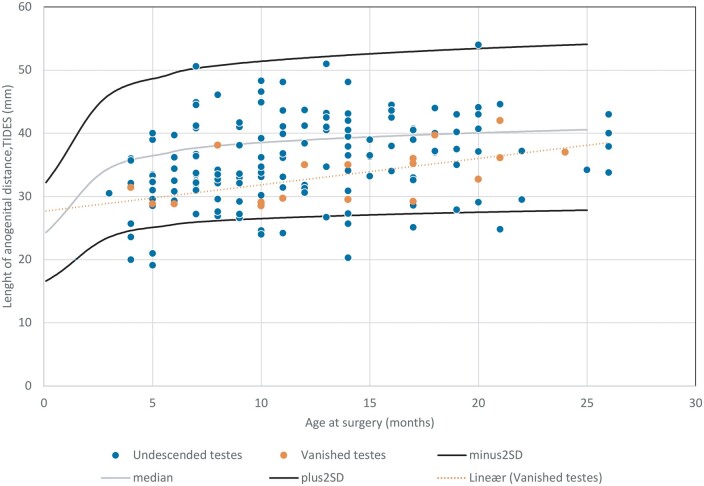
Based on the statistical parameter MoM AGD, the 169 boys with cryptorchidism had shorter AGDs than normal (MoM AGD median 0.90 (range: 0.52–1.35)), and only 30% (50/169) had MoM AGDs higher than the normal median. Figure 1 shows that 12 patients had MoM AGD below −2 SDs of normal median (Fischer et al., 2020 (CrossRef: 10.6084/m9.figshare.12179190)). None of these 12 patients had vanished testis.
Figure 1.
Ano-scrotal distance, anogenital distance (AGD) of 169 boys with cryptorchidism (including 18 boys with vanishing testes) and normal ranges in relation to age.
No boys with vanished testis had a birth weight below 3000 g compared with 14% of boys with undescended testes (P < 0.05). Moreover, the frequency of bilateral cases was lowest in boys with vanished testes (6% (1/18) versus 31% (47/151), P < 0.05) (Table 1).
Only 6% (1/18) of cases with vanished testes had an MoM AGD higher than normal median compared to 32% (49/151) of cases with undescended testes (P < 0.05) (Table 1 and Fig. 1). MoM AGD increased with age at surgery for boys with vanished testis (Spearman r = 0.44), but not for boys with undescended testes (Spearman r = 0.14) (Fig. 1).
For boys with vanished testis and for boys with undescended testes, respectively, there were no significant correlations between MoM AGD and birth weight, MoM Inhibin B, FSH, or LH, respectively.
For boys with undescended testes, MoM AGD correlate with neither MoM G/T nor Spermatogonia A dark/T. However, MoM G/T correlated with MoM Inhibin B and with Spermatogonia Adark/T (Spearman r = 0.26, P < 0.005 and Spearman r = 0.59), respectively.
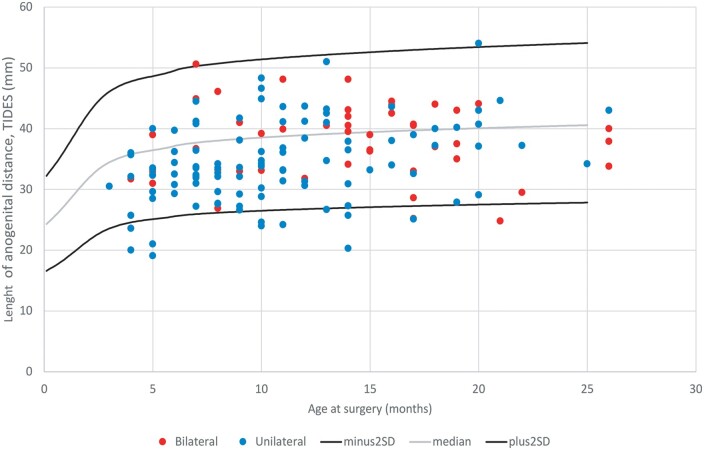
MoM AGD was longer for the 47 boys with bilateral undescended testes (0.99 (0.62–1.34)) than for the 104 boys with unilateral undescended testes (0.89 (0.52–1.35)) (P < 0.005) (Table 2, and Fig. 2).
Table 2.
Boys with undescended testes who underwent measurement of the ano-scrotal distance (AGD), classified into 104 with unilateral and 47 with bilateral undescended testes.
| Unilateral undescended testis | Bilateral undescended testes | P-value | |
|---|---|---|---|
| Number | 104¤ | 47§ | |
| Age at surgery (months)* | 10 (3–26) | 14 (4–26) | P = 0.001 |
| Birth weight (g)* | 3540 (1200–4910) | 3500 (640–4910) | P = 0.303 |
| MoM AGD* | 0.89 (0.52–1.35) | 0.99 (0.62–1.34) | P = 0.004 |
| MoM AGD higher than median | 25% (26/104) | 49% (23/47) | P = 0.004 |
| Position of undescended testes, boys$ | |||
| Abdominal | 8% (8/104) | 11% (5/47) | P = 0.550 |
| Inguinal | 38% (39/104) | 23% (11/47) | P = 0.087 |
| At external inguinal ring | 41% (43/104) | 11% (5/47) | P = 0.0002 |
| Suprascrotal position | 10% (10/104) | 28% (13/47) | P = 0.004 |
| Ascending | 4% (4/104) | 28% (13/47) | P = 0.0002 |
| MoM G/T* | 0.33 (0.01–1.59) | 0.52 (0.00–2.10) | P = 0.153 |
| Spermatogonia A dark/T* | 0.019 (0–0.178) | 0.029 (0.0–0.233) | P = 0.127 |
| MoM Inhibin B* | 0.70 (0.17–1.24) | 0.71 (0.21–1.50) | P = 0.568 |
| Inhibin B low (<2.5th percentile) | 13% (13/104) | 19% (9/47) | P = 0.285 |
| FSH* | 0.81 (0.08–2.75) | 0.73 (0.16–3.20) | P = 0.992 |
| FSH high (>97.5th percentile) | 22% (23/103) | 23% (11/47) | P = 0.884 |
| LH* | 0.26 (0.05–4.82) | 0.15 (0.05–2.66) | P = 0.352 |
| LH high (>97.5th percentile) | 5% (5/103) | 4% (2/47) | P = 0.872 |
| Groups based on hormonal and histological analyses at surgery | |||
| Group 1 | 23% (23/102) | 23% (11/47) | P = 0.908 |
| Group 2 | 10% (10/102) | 62% (29/47) | P < 0.0000001 |
| Group 3 | 68% (69/102) | 15% (7/47) | P < 0.0000001 |
MoM, multiple of the median; MoM AGD, MoM ano-scrotal distance; G, germ cell number; T, per cross-sectional tubule.
Median (range).
One without FSH, one without histological analysis, and totally four without G/T and Spermatogonia A dark/T.
Two without birth weight, and one without G/T and Spermatogonia A dark/T.
For the four boys with different position of the two undescended testes, the classification was based on the highest testicular position.
Figure 2.
Ano-scrotal distance, anogenital distance (AGD) of 104 boys with unilateral, and 47 boys with bilateral, cryptorchidism, and a normal ranges in relation to age.
Boys who had surgery for bilateral cryptorchidism were older at surgery than those who had surgery for unilateral cryptorchidism (P < 0.005) (Table 2).
Boys with bilateral cryptorchidism most often had testes located suprascrotally or had ascending testes, 28% and 28% compared with 10% and 4% in boys with unilateral cryptorchidism (P < 0.005 and P < 0.0005). In contrast, boys unilateral cryptorchidism more often had testes in the inguinal canal or at the external inguinal ring, 38% and 41%, compared with 23% and 11% for boys with bilateral cryptorchidism (P = 0.44 and P < 0.0005) (Table 2).
Boys with bilateral cryptorchidism were more often suspected of hypogonadotropic hypogonadism (Group 2 based on hormonal and histological analyses) than boys with unilateral cryptorchidism, 62% versus 10% (P < 0.0000001). In contrast, more boys with unilateral cryptorchidism had normal hormonal values at surgery (Group 3 based on hormonal and histological analyses) compared to boys with bilateral cryptorchidism, 68% versus 15% (P < 0.0000001).
The boys with undescended testes were also classified based on their MoM AGD (Table 3). The group of boys with the shortest MoM AGD, up to 0.75, more often had a mother who had smoked at least five cigarettes every day during pregnancy (P < 0.05).
Table 3.
Data for 151 Boys with undescended testes, classified by length of the ano-scrotal distance (AGD).
| MoM AGD up to and including 0.75 | MoM AGD from 0. 76 up to and including median (=1) | MoM AGD higher than median (>1) | P-value | |
|---|---|---|---|---|
| Number | 26 | 76§ | 49¤ | |
| Bilateral cryptorchidism | 19% (5/26) | 25% (19/76) | 47% (23/49) | P = 0.013 |
| Age at surgery (months)* | 10 (4–22) | 10 (3–26) | 13 (4–26) | P = 0.083 |
| Birth weight (g)* | 3405 (640–4800) | 3605 (1200–4910) | 3500 (2200–4910) | P = 0.521 |
| Smoked at least five cigarettes/day during pregnancy | 16% (4/25) | 1% (1/75) | 7% (3/45) | P = 0.029 |
| Position of undescended testes, boys$ | ||||
| Abdominal | 4% (1/26) | 7% (5/76) | 14% (7/49) | P = 0.207 |
| Inguinal | 42% (11/26) | 33% (25/76) | 24% (12/49) | P = 0.276 |
| At external inguinal ring | 27% (7/26) | 37% (28/76) | 27% (13/49) | P = 0.406 |
| Suprascrotal position | 15% (4/26) | 12% (9/76) | 20% (10/49) | P = 0.429 |
| Ascending testes | 12% (3/26) | 11% (8/76) | 12% (6/49) | P = 0.956 |
| MoM Inhibin B* | 0.80 (0.36–1.50) | 0.66 (0.17–1.24) | 0.79 (0.28–1.13) | P = 0.020 |
| Inhibin B low (<2.5th percentile) | 4% (1/26) | 21% (16/76) | 10% (5/49) | P = 0.057 |
| MoM G/T* | 0.53 (0.01–1.69) | 0.33 (0–1.40) | 0.45 (0.03–2.1) | P = 0.520 |
| Spermatogonia A dark/T* | 0.024 (0–0.178) | 0.018 (0–0.158) | 0.020 (0–0.233) | P = 0.651 |
| FSH* | 0.88 (0.21–2.75) | 0.84 (0.18–3.20) | 0.67 (0.08–2.05) | P = 0.204 |
| FSH high (>97.5th percentile) | 27% (7/26) | 21% (16/75) | 22% (11/49) | P = 0.841 |
| LH* | 0.22 (0.05–4.82) | 0.24 (0.05–3.20) | 0.22 (0.05–2.89) | P = 0.715 |
| LH high (>97.5th percentile) | 12% (3/26) | 1% (1/76) | 4% (2/49) | P = 0.085 |
| Groups based on hormonal and histological analyses at surgery | ||||
| Group 1 | 27% (7/26) | 21% (16/75) | 23% (11/48) | P = 0.842 |
| Group 2 | 8% (2/26) | 27% (20/75) | 35% (17/48) | P = 0.034 |
| Group 3 | 65% (17/26) | 52% (39/75) | 42% (20/48) | P = 0.145 |
MoM, multiple of the median; MoM AGD, MoM ano-scrotal distance; G, germ cell number; T, per cross-sectional tubule.
Median (range).
Two without birthweight, two without G/T and Spermatogonia Adark/T, and one without FSH.
One without histology, totally three without G/T and Spermatogonia Adark/T.
For the four boys with different position of the two undescended testes, the classification was based on the highest testicular position.
In the higher MoM AGD groups, there were higher frequencies of bilateral cryptorchidism (P < 0.05) and the higher frequencies of boys suspected of having hypogonadotropic hypogonadism (Group 2 based on hormonal and histological analyses) (P < 0.05).
Finally, the boys with undescended testes were classified based on the position of the undescended testes (Table 4). More boys with testes in a suprascrotal position or with ascending testes could be suspected of hypogonadotropic hypogonadism (Group 2 based on hormonal and histological analyses) when compared to the other types (P < 0.00005).
Table 4.
Data for 151 boys with cryptorchidism who underwent measurement of the ano-scrotal distance (AGD), classified by the position of the undescended testis.
| Boys with abdominal testis | Boys with inguinal testis or testis at the external inguinal ring | Boys with testis in a suprascrotal position or with ascending testis | P-value | |
|---|---|---|---|---|
| Number | 13§ | 98# | 40 | |
| Bilateral | 38% (5/13) | 16% (16/98) | 65% (26/40) | P < 0.0001 |
| Age at time of surgery, months* | 9 (4–20) | 15.5 (3–26) | 15.5 (5–26) | P < 0.0001 |
| MoM AGD* | 1.01 (0.64–1.35) | 0.89 (0.52–1.26) | 0.93 (0.58–1.30) | P = 0.083 |
| Groups based on hormonal and histological analyses at surgery | ||||
| Group 1 | 15% (2/13) | 25% (24/96) | 20% (8/40) | P = 0.655 |
| Group 2 | 38% (5/13) | 15% (14/96) | 50% (20/40) | P = 0.0001 |
| Group 3 | 46% (6/13) | 60% (58/96) | 30% (12/40) | P = 0.0002 |
| MoM Inhibin B* | 0.57 (0.29–1.12) | 0.70 (0.17–1.30) | 0.79 (0.21–1.50) | P = 0.058 |
| Inhibin B low (<2.5th percentile) | 23% (3/13) | 15% (15/98) | 10% (4/40) | P = 0.480 |
| MoM G/T* | 0.50 (0.04–2.10) | 0.34 (0–1.69) | 0.49 (0.03–1.38) | P = 0.462 |
| Spermatogonia A dark/T* | 0.009 (0–0.165) | 0.017 (0–0.178) | 0.042 (0.01–0.233) | P < 0.0001 |
| FSH* | 0.99 (0.21–1.67) | 0.85 (0.08–3.20) | 0.53 (0.16–1.92) | P = 0.022 |
| High FSH (>97.5th percentile) | 15% (2/13) | 22% (21/97) | 15% (6/40) | P = 0.632 |
| LH* | 0.37 (0.50–2.89) | 0.35 (0.05–4.82) | 0.13(0.05–2.04) | P = 0.012 |
| High LH (>97.5th percentile) | 23% (3/13) | 4% (4/98) | 0% (0/40) | P = 0.002 |
MoM, multiple of the median; MoM AGD, MoM ano-scrotal distance; G, germ cell number; T, per cross-sectional tubule.
Median (range).
One without G/T and Spermatogonia A dark/T.
One without histological analysis, totally four without G/T and Spermatogonia A dark/T, and one without FSH.
Discussion
To our knowledge, this prospective study represents results of the first AGD measurements in infant boys with cryptorchidism in relation to type, serum reproductive hormone levels, and histopathology of the undescended testes.
We found that significantly fewer boys with vanished testis had MoM AGD higher than the normal median value compared to those with undescended testis. To the best of our knowledge, this has not been published before.
Vanished testis is hypothesized to result from a traumatic torsion in the third trimester with testicular demolishment and consequently only half the normal testosterone production (Bader et al., 2011; Gerbo et al., 2021; Hildorf et al., 2022). Therefore, our findings show that such a drastic androgen reduction during third trimester results in a shorter AGD, which supports the importance of androgens in normal male development. This traumatic third trimester event challenges the current concept of AGD being determined in ‘the masculinization programming window’ in Week 8 to 14 of gestation.
Moreover, for boys with vanished testes we found that MoM AGD increased significantly with increasing age at surgery. Thereby, the remaining scrotal testis can undergo compensatory hypertrophy allowing a catch-up in AGD. In boys with vanished testis, compensatory hypertrophy is well-documented concerning testicular volume and inhibin B (Koff, 1991; Huff et al., 1992; Hildorf et al., 2022). Our findings indicate that AGD is influenced by androgens over a longer period in utero and that AGD is more dynamic in humans than previously described (Welsh et al., 2008; Jain et al., 2018). Rodent studies have indicated plasticity in AGD and have demonstrated the need foradequate androgen levels, not only prenatally but also postnatally, in order to achieve or maintain the AGD as programmed in utero (Mitchell et al., 2015; Priskorn et al., 2018, Sharpe 2020). Thus, deficiencies in androgen exposure during the masculinization window (as seen in testicular dysgenesis) or in the third trimester (as seen with vanished testis) result in reduced AGD. However, in cases of a lack of androgen in the third trimester, the AGD may increase during infancy when androgens normalize, as seen with the longer AGDs at increasing ages in the cases of vanished testis.
We also found that maternal gestational smoking, i.e. self-reporting of smoking at least five cigarettes every day during pregnancy, was associated with shorter AGD in the infant. This is in agreement with the hypothesis of AGD being determined in gestational week 8 to 14 and with testicular dysgenesis (Larsen, 1997; Welsh et al., 2008).
During first trimester, fetal androgen production is initiated and maintained by hCG produced by trophoblast cells in the placenta (Scott et al., 2009). A major constituent of cigarette smoke is the group of polycyclic aromatic hydrocarbons, which can affect the placental hCG secretion (Barnea and Shurtz-Swirski, 1992). In addition, an in vitro study applying cigarette smoke extract on human placental cells demonstrated that smoking exposure decreased hCG-β production (Kim et al., 2017). Consistent with this, lower maternal hCG levels have been demonstrated in smoking pregnant women compared to non-smokering women (Biggs et al., 2002).
Since the association between a shorter AGD and cryptorchidism was first reported by Swan et al. (2005), other studies, besides ours, have reproduced this finding (Hsieh et al., 2008; Thankamony et al., 2009; Jain and Singal, 2013; Thankamony et al., 2014; Abbas and Ali, 2021). However, our findings on longer AGD in bilateral versus unilateral cases have not previously been reported (Jain and Singal, 2013; Thankamony et al., 2014). A birth cohort study including 624 male newborns out of which 71 had congenital cryptorchidism, analyzed AGD in newborn boys with respect to type of cryptorchidism as unilateral or bilateral (46%) and further subgroups according to testicular position (Jain and Singal, 2013). They found no difference in AGD between these groups. However, a trend was noted among those boys with higher testicular position displaying shorter AGD. Only 33 cases were bilateral, and the AGD measurement took place within 48 h after birth, thereby a significant risk of later spontaneous descent was present. So, their findings may not differ from ours. Likewise, another study also distinguished between bilateral and unilateral cases and did not find any association with AGD (Thankamony et al., 2014). That study included also a very small study population, so it is not surprising that no differences between unilateral and bilateral cases could be demonstrated, and the data were not shown.
In the present study, at time of surgery, 56% of boys with bilateral cryptorchidism had ascending testes or testes suprascrotally placed in contrast to 14% of boys with unilateral cryptorchidism. Therefore, at the time of birth, only about half of the boys with later surgery for bilateral cryptorchidism had with certainty congenital cryptorchidism in contrast to about 90% of the boys with later surgery for unilateral cryptorchidism and a possible dysgenesis testis. In cases of scrotal testes at time of birth, a normal AGD is supposed. This difference may explain much of the difference in AGD in boys with bilateral and unilateral cryptorchidism at time of surgery. In fact, with a MoM AGD for 47 boys with bilateral undescended testes at 0.99 (0.62–1.34), a normal distribution for this group is indicated.
Moreover, at time of surgery, 62% of the boys with bilateral cryptorchidism had indications of hypogonadotropic hypogonadism (Group 2; having a reduced G/T and/or low inhibin B but despite that no compensatory FSH elevation). This observation fits well with the finding that up to 62% of boys with bilateral cryptorchidism have been reported to have hypogonadotropic hypogonadism (Hadziselimovic, 1997; Hadziselimovic et al., 2004; Hadziselimovic and Hoecht, 2008; Thorup et al., 2012; Hildorf, 2022).
Fetal hypothalamic control of gonadotropic function is not operative until after the first trimester (Huhtaniemi, 1977; Choi and Smitz, 2014). An impaired androgen stimulation is related to incomplete inguinoscrotal descent of the testes that begins at about 25 weeks of gestation (Backhouse, 1982; Hutson et al., 2016). One may speculate that, in cases of hypogonadotropic hypogonadism at time of early surgery for cryptorchidism, the second and third trimester androgen stimulation of the anogenital region had not necessarily been significantly impaired. The fetal levels of androgen may be lower in the majority of boys with unilateral cryptorchidism and, in the third trimester, in all boys with vanished testis.
In our cases of unilateral cryptorchidism, we observed more inguinal testes or testes placed at the external inguinal ring. To understand why our boys with unilateral cryptorchidism overall had shorter AGD than bilateral cases, the spectrum of the abnormality must be taken into consideration. The MoM G/T of the affected testes was reduced in these unilateral cases, which may be due to a dysgenesis of the undescended testis. Unilateral cryptorchidism may be an isolated unilateral expression of a caudal developmental field defect (Cortes et al., 1998). So, fetal Leydig cell androgen production in the later undescended testis might be hampered during fetal life leading to unilateral incomplete inguinoscrotal descent as well as incomplete development of the anogenital region through insufficient androgen stimulation of the dorsal root ganglion to the genitofemoral nerve (Hutson et al., 2016).
Around 10% of boys with unilateral cryptorchidism have a bilateral testicular affect due to hypogonadotropic hypogonadism being diagnosed (Group 2). So, different types of pathogenesis related to unilateral cryptorchidism may also affect the AGD in different ways.
We found no correlations between MoM AGD and MoM G/T, Spermatogonia Adark/T, MoM Inhibin B, FSH, or LH. Therefore, we consider these parameters may not relate to fetal life but reflect testicular conditions postnatally. No other study has previously focused on these factors. However, AGD was not associated with androgen levels in cord blood collected at delivery or with reproductive hormones in young men, and there are conflicting results of the association between AGD and sperm concentrations (Jain et al., 2018; Priskorn et al., 2019). We found no association between AGD and birthweight, consistent with previous reports (Jain and Singal, 2013; Thankamony et al., 2014; Abbas and Ali, 2021).
AGD is a relatively new marker in human research, which means that the anthropometry and method is not fully standardized. Our observations are based on a large cohort of boys with cryptorchidism, using a large in-house study as comparative normative data (Fischer et al., 2020).
We estimated MoM values and used normal AGD according to age, as AGD is age-dependent and dynamic during the first two years of life (Thankamony et al., 2014; Fischer et al., 2020). Furthermore, the strengths of our study include the prospective, single-center design and the comprehensive range of different parameters including both reproductive hormones and histology. In addition, AGD were measured by the four standardized methods with different body positionings, by the same investigator (J.T.), who was trained by M.B.F., who established the normal reference values. We used TIDES AGDAS, as it correlated with the three other AGD-measurements and was the most commonly used in the previous studies (Thankamony et al., 2009; Jain and Singal, 2013; Thankamony et al., 2014; Abbas and Ali, 2021), and replicability and stability during childhood has been reported to be better for AGDAS than for AGDAP (Fischer et al., 2020).
One limitation covers the quite small number of patients in the different groups, which may decrease the statistical power. Another limitation involves the sparse normal reference material on G/T and AdS/T. Finally, there are currently no longitudinal studies evaluating AGD from birth to adulthood and evaluating childhood AGD in relation to fertility outcome.
Conclusion
Significantly fewer boys with vanished testis had MoM AGDs higher than 1 compared to boys with undescended testis. Moreover, only for boys with vanished testes, the AGD increased with age. To the best of our knowledge, such information is new.
This is a hypothesis generating article, and we suggest that in humans, normal exposure to androgens in the third trimester is also important to achieve a normal AGD at time of birth. Thus, androgen deficiencies during the masculinization window likely results in reduced AGD (as seen in testicular dysgenesis), similar to the lack of third trimester exposure to androgens as seen with vanished testis. The latter is likely to be corrected when androgen secretion normalize, as seen with the longer AGD with increasing age in boys with vanished testis.
Moreover, at time of surgery, AGD was longer for boys with bilateral, than those with unilateral, cryptorchidism. At surgery for bilateral cryptorchidism, more than half of boys had ascending testes or testes suprascrotally placed, and consequently many had scrotal testes at time of birth with supposed normal AGD. At time of surgery, hypogonadotropic hypogonadism was present for about 60% of boys with bilateral cryptorchidism, suggesting that the lack of fetal androgen in hypogonadotropic hypogonadism is not that significant. Our observations provide more evidence for the current theory on the determination of AGD in humans.
Contributor Information
Dina Cortes, Department of Paediatric Surgery, Copenhagen University Hospital Rigshospitalet, Copenhagen, Denmark; Department of Clinical Medicine, University of Copenhagen, Copenhagen, Denmark; Department of Paediatrics and Adolescent Medicine, Copenhagen University Hospital Hvidovre, Hvidovre, Denmark.
Margit Bistrup Fischer, Department of Growth and Reproduction and EDMaRC, Copenhagen University Hospital-Rigshospitalet, Copenhagen, Denmark.
Andrea E Hildorf, Department of Paediatric Surgery, Copenhagen University Hospital Rigshospitalet, Copenhagen, Denmark.
Erik Clasen-Linde, Department of Pathology, Copenhagen University Hospital, Rigshospitalet, Copenhagen, Denmark.
Simone Hildorf, Department of Paediatric Surgery, Copenhagen University Hospital Rigshospitalet, Copenhagen, Denmark.
Anders Juul, Department of Clinical Medicine, University of Copenhagen, Copenhagen, Denmark; Department of Growth and Reproduction and EDMaRC, Copenhagen University Hospital-Rigshospitalet, Copenhagen, Denmark.
Katharina M Main, Department of Clinical Medicine, University of Copenhagen, Copenhagen, Denmark; Department of Growth and Reproduction and EDMaRC, Copenhagen University Hospital-Rigshospitalet, Copenhagen, Denmark.
Jorgen Thorup, Department of Paediatric Surgery, Copenhagen University Hospital Rigshospitalet, Copenhagen, Denmark; Department of Clinical Medicine, University of Copenhagen, Copenhagen, Denmark.
Data availability
All the data underlying this article are available in text, figures, tables and references (CrossRef). If further break down is requested, such data will be shared on reasonable request to the corresponding author.
Authors’ roles
D.C. was responsible for substantial contributions to the study concept and design, analysis and interpretation of data, drafting the article, and final approval of the version to be published. M.B.F. was responsible for acquisition of data and analysis and interpretation of data, revising the article critically for important intellectual content, and final approval of the version to be published. A.E.H. was responsible for acquisition of data, analysis and interpretation of data, drafting part of the article, and final approval of the version to be published. E.C.-L. was responsible for acquisition of data, analysis and interpretation of data, revising the article critically for important intellectual content, and final approval of the version to be published. S.H. was responsible for substantial contributions to acquisition of data, analysis and interpretation of data, revising the article critically for important intellectual content, and final approval of the version to be published. A.J. was responsible for substantial contributions to the study concept and design, interpretation of data, revising the article critically for important intellectual content, and final approval of the version to be published. K.M.M. was responsible for substantial contributions to the study and design and interpretation of data, revising the article critically for important intellectual content, and final approval of the version to be published. J.T. was responsible for substantial contributions to the study concept and design, acquisition and interpretation of data, revising the article critically for important intellectual content, and final approval of the version to be published.
Funding
No external funding was used.
Conflict of interest
None declared.
References
- Abbas TO, Ali M.. Scrotal base distance: a new key genital measurement in males with hypospadias and cryptorchidism. Curr Urol 2021;15:214–218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Backhouse KM. Development and descent of the testis. Eur J Pediatr 1982;139:249–252. [DOI] [PubMed] [Google Scholar]
- Bader MI, Peeraully R, Ba'ath M, McPartland J, Baillie C.. The testicular regression syndrome—do remnants require routine excision? J Pediatr Surg 2011;46:384–386. [DOI] [PubMed] [Google Scholar]
- Barnea ER, Shurtz-Swirski R.. Modification of pulsatile human chorionic gonadotrophin secretion in first trimester placental explants induced by polycyclic aromatic hydrocarbons. Hum Reprod 1992;7:305–310. [DOI] [PubMed] [Google Scholar]
- Barrett ES, Sathyanarayana S, Janssen S, Redmon JB, Nguyen RH, Kobrosly R, Swan SH; TIDES Study Team. Environmental health attitudes and behaviors: findings from a large pregnancy cohort study. Eur J Obstet Gynecol Reprod Biol 2014;176:119–125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biggs ML, Baer A, Critchlow CW.. Maternal, delivery, and perinatal characteristics associated with cryptorchidism: a population-based case-control study among births in Washington State. Epidemiology 2002;13:197–204. [DOI] [PubMed] [Google Scholar]
- Busch AS, Ljubicic ML, Upners EN, Fischer MB, Kolby N, Eckert-Lind C, Jespersen K, Andersson A-M, Frederiksen H, Johannsen TH et al Cohort profile: the Copenhagen Minipuberty Study—a longitudinal prospective cohort of healthy full-term infants and their parents. Paediatr Perinat Epidemiol 2021;35:601–611. [DOI] [PubMed] [Google Scholar]
- Choi J, Smitz J.. Luteinizing hormone and human chorionic gonadotropin: distinguishing unique physiologic roles. Gynecol Endocrinol 2014;30:174–181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cortes D. Cryptorchidism-aspects of pathogenesis, histology and treatment. Scand J Urol Nephrol Suppl 1998;196:1–54. [PubMed] [Google Scholar]
- Cortes D, Thorup JM, Beck BL.. Quantitative histology of germ cells in the undescended testes of human fetuses, neonates and infants. J Urol 1995;154:1188–1192. [PubMed] [Google Scholar]
- Cortes D, Thorup JM, Beck BL, Visfeldt J.. Cryptorchidism as a caudal developmental field defect. APMIS 1998;106:953–958. [PubMed] [Google Scholar]
- Dean A, Sharpe RM.. Anogenital distance or digit length ratio as measures of fetal androgen exposure: relationship to male reproductive development and its disorders. J Clin Endocrinol Metab 2013;98:2230–2238. [DOI] [PubMed] [Google Scholar]
- Eisenberg ML, Hsieh MH, Walters RC, Krasnow R, Lipshultz LI.. The relationship between anogenital distance, fatherhood, and fertility in adult men. PLoS One 2011;6:e18973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fischer MB, Ljubicic ML, Hagen CP, Thankamony A, Ong K, Hughes I, Jensen TK, Main KM, Petersen JH, Busch AS. et al. Anogenital distance in healthy infants: method-, age- and sex-related reference ranges. J Clin Endocrinol Metab 2020;105:2996–3004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerbo M, Crigger C, Samadi Y, Ost MC, Al-Omar O.. Prenatally diagnosed testicular torsion: a rare condition that causes dilemma in management. Case Rep Pediatr 2021;2021:8825763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hadziselimovic F. Cryptorchidism—Ultrastructure of Normal and Cryptorchid Testis Development. Berlin, Heidelberg, Germany: Springer-Verlag, 1997. [PubMed] [Google Scholar]
- Hadziselimovic F, Hoecht B.. Testicular histology related to fertility outcome and postpubertal hormone status in cryptorchidism. Klin Padiatr 2008;220:302–307. [DOI] [PubMed] [Google Scholar]
- Hadziselimovic F, Emmons LR, Buser MW.. A diminished postnatal surge of Ad spermatogonia in cryptorchid infants is additional evidence for hypogonadotropic hypogonadism. Swiss Med Wkly 2004;134:381–384. [DOI] [PubMed] [Google Scholar]
- Hildorf S. Clinical aspects of histological and hormonal parameters in boys with cryptorchidism. APMIS 2022;130(Suppl 143):1–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hildorf S, Clasen-Linde E, Cortes D, Fossum M, Thorup J.. Serial inhibin B measurements in boys with congenital monorchism indicate compensatory testicular hypertrophy in early infancy. Eur J Pediatr Surg 2022;32:34–41. [DOI] [PubMed] [Google Scholar]
- Hsieh MH, Breyer BN, Eisenberg ML, Baskin LS.. Associations among hypospadias, cryptorchidism, anogenital distance, and endocrine disruption. Curr Urol Rep 2008;9:137–142. [DOI] [PubMed] [Google Scholar]
- Huff DS, Snyder HMI, Hadziselimovic F, Blyth B, Duckett JW.. An absent testis is associated with contralateral testicular hypertrophy. J Urol 1992;148:627–628. [DOI] [PubMed] [Google Scholar]
- Huhtaniemi I. Studies on steroidogenesis and its regulation in human fetal adrenal and testis. J Steroid Biochem 1977;8:491–497. [DOI] [PubMed] [Google Scholar]
- Hutson J, Thorup J, Beasley S.. Descent of the Testis. Switzerland: Springer International Publishing, 2016. [Google Scholar]
- Jain VG, Singal AK.. Shorter anogenital distance correlates with undescended testis: a detailed genital anthropometric analysis in human newborns. Hum Reprod 2013;28:2343–2349. [DOI] [PubMed] [Google Scholar]
- Jain VG, Goyal V, Chowdhary V, Swarup N, Singh RJ, Singal A, Shekhawat P.. Anogenital distance is determined during early gestation in humans. Hum Reprod 2018;33:1619–1627. [DOI] [PubMed] [Google Scholar]
- Johannsen TH, Main KM, Ljubicic ML, Jensen TK, Andersen HR, Andersen MS, Petersen JH, Andersson AM, Juul A.. Sex differences in reproductive hormones during mini-puberty in infants with normal and disordered sex development. J Clin Endocrinol Metab 2018;103:3028–3037. [DOI] [PubMed] [Google Scholar]
- Kim CW, Lee HM, Lee K, Kim B, Lee MY, Choi KC.. Effects of cigarette smoke extracts on cell cycle, cell migration and endocrine activity in human placental cells. Reprod Toxicol 2017;73:8–19. [DOI] [PubMed] [Google Scholar]
- Koff SA. Does compensatory testicular enlargement predict monorchism? J Urol 1991;146:632–633. [DOI] [PubMed] [Google Scholar]
- Laboratory methods and reference, Department of Growth and Reproduction, Copenhagen University Hospital Rigshospitalet. https://hormlabvejl.regionh.dk/Metodeliste.asp (20 October 2022, date last accessed).
- Larsen WJ. Human Embryology. New York, NY, USA: Churchill Livingstone, 1997, 261–309. [Google Scholar]
- Masliukaite I, Hagen JM, Jahnukainen K, Stukenborg JB, Repping S, Van der Veen F, van Wely M, van Pelt AMM.. Establishing reference values for age-related spermatogonial quantity in prepubertal human testes: a systematic review and meta-analysis. Fertil Steril 2016;106:1652–1657.e2. [DOI] [PubMed] [Google Scholar]
- Mendiola J, Stahlhut RW, Jørgensen N, Liu F, Swan SH.. Shorter anogenital distance predicts poorer semen quality in young men in Rochester, New York. Environ Health Perspect 2011;119:958–963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitchell RT, Mungall W, McKinnell C, Sharpe RM, Cruickshanks L, Milne L, Smith LB.. Anogenital distance plasticity in adulthood: implications for its use as a biomarker of fetal androgen action. Endocrinology 2015;156:24–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prentice P, Acerini CL, Eleftheriou A, Hughes IA, Ong KK, Dunger DB.. Cohort Profile: the Cambridge Baby Growth Study (CBGS). Int J Epidemiol 2016;45:35.a–g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Priskorn L, Bang AK, Nordkap L, Krause M, Mendiola J, Jensen TK, Juul A, Skakkebaek NE, Swan SH, Jørgensen N.. Anogenital distance is associated with semen quality but not reproductive hormones in 1106 young men from the general population. Hum Reprod 2019;34:12–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Priskorn L, Kreiberg M, Bandak M, Lauritsen J, Daugaard G, Petersen JH, Aksglaede L, Juul A, Jørgensen N.. Testicular cancer survivors have shorter anogenital distance that is not increased by 1 year of testosterone replacement therapy. Hum Reprod 2021;36:2443–2451. [DOI] [PubMed] [Google Scholar]
- Priskorn L, Petersen JH, Jørgensen N, Kyhl HB, Andersen MS, Main KM, Andersson AM, Skakkebaek NE, Jensen TK.. Anogenital distance as a phenotypic signature through infancy. Pediatr Res 2018;83:573–579. [DOI] [PubMed] [Google Scholar]
- Schwartz CL, Christiansen S, Vinggaard AM, Axelstad M, Hass U, Svingen T.. Anogenital distance as a toxicological or clinical marker for fetal androgen action and risk for reproductive disorders. Arch Toxicol 2019;93:253–272. [DOI] [PubMed] [Google Scholar]
- Scott HM, Mason JI, Sharpe RM.. Steroidogenesis in the fetal testis and its susceptibility to disruption by exogenous compounds. Endocr Rev 2009;30:883–925. [DOI] [PubMed] [Google Scholar]
- Sharpe RM. Androgens and the masculinization programming window: human-rodent differences. Biochem Soc Trans 2020;48:1725–1735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swan SH, Main KM, Liu F, Stewart SL, Kruse RL, Calafat AM, Mao CS, Redmon JB, Ternand CL, Sullivan S. et al. ; Study for Future Families Research Team. Decrease in anogenital distance among male infants with prenatal phthalate exposure. Environ Health Perspect 2005;113:1056–1061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thankamony A, Lek N, Carroll D, Williams M, Dunger DB, Acerini CL, Ong KK, Hughes IA.. Anogenital distance and penile length in infants with hypospadias or cryptorchidism: comparison with normative data. Environ Health Perspect 2014;122:207–211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thankamony A, Ong KK, Dunger DB, Acerini CL, Hughes IA.. Anogenital distance from birth to 2 years: a population study. Environ Health Perspect 2009;117:1786–1790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thankamony A, Pasterski V, Ong KK, Acerini CL, Hughes IA.. Anogenital distance as a marker of androgen exposure in humans. Andrology 2016;4:616–625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorup J, Petersen BL, Kvist K, Cortes D.. Bilateral undescended testes classified according to preoperative and postoperative status of gonadotropins and inhibin B in relation to testicular histopathology at bilateral orchiopexy in infant boys. J Urol 2012;188:1436–1442. [DOI] [PubMed] [Google Scholar]
- Welsh M, Saunders PT, Fisken M, Scott HM, Hutchison GR, Smith LB, Sharpe RM.. Identification in rats of a programming window for reproductive tract masculinization, disruption of which leads to hypospadias and cryptorchidism. J Clin Invest 2008;118:1479–1490. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All the data underlying this article are available in text, figures, tables and references (CrossRef). If further break down is requested, such data will be shared on reasonable request to the corresponding author.