Abstract
Fetal restriction (FR) alters insulin sensitivity, but it is unknown how the metabolic profile associated with restriction affects development of the dopamine (DA) system and DA-related behaviors. The Netrin-1/DCC guidance cue system participates in maturation of the mesocorticolimbic DA circuitry. Therefore, our objective was to identify if FR modifies Netrin-1/DCC receptor protein expression in the prefrontal cortex (PFC) at birth and mRNA in adulthood in rodent males. We used cultured HEK293 cells to assess if levels of miR-218, microRNA regulator of DCC, are sensitive to insulin. To assess this, pregnant dams were subjected to a 50% FR diet from gestational day 10 until birth. Medial PFC (mPFC) DCC/Netrin-1 protein expression was measured at P0 at baseline and Dcc/Netrin-1 mRNA levels were quantified in adults 15 min after a saline/insulin injection. miR-218 levels in HEK-293 cells were measured in response to insulin exposure. At P0, Netrin-1 levels are downregulated in FR animals in comparison to controls. In adult rodents, insulin administration results in an increase in Dcc mRNA levels in control but not FR rats. In HEK293 cells, there is a positive correlation between insulin concentration and miR-218 levels. Since miR-218 is a Dcc gene expression regulator and our in vitro results show that insulin regulates miR-218 levels, we suggest that FR-induced changes in insulin sensitivity could be affecting Dcc expression via miR-218, impacting DA system maturation and organization. As fetal adversity is linked to nonadaptive behaviors later in life, this may contribute to early identification of vulnerability to chronic diseases associated with fetal adversity.
Keywords: Prenatal adversity, Intrauterine growth restriction, Food restriction, Insulin, Axonal guidance cues, Mesocorticolimbic dopamine pathway
Introduction
Reduced growth in fetal life has been strongly linked to the development of impaired glucose tolerance and type II diabetes,1 mainly due to altered growth and development of the pancreas.2 Insulin secretion relative to insulin sensitivity is highly impaired in these individuals throughout their lifetime as a consequence of poor growth before birth.3–8 Many models have shown that reduced growth due to poor nutrition increases the risk for adult chronic diseases. One of the most important models being that of caloric restriction1,9,10 because it re-capitulates many features seen in intrauterine growth restriction (IUGR) cases in humans such as catch-up growth,11 hyperinsulinemia,12 type II diabetes,13 and altered behaviors such as increased preference for palatable foods9 and impulsivity.14 Impulsivity includes two dimensions: action and choice.15 Impulsive action refers to the inability in the inhibition of response such as responding without thinking16 while impulsive choices involve a decision-making process and are expressed by the tendency to prefer immediate gains over long-term ones.17,18 Both action and choice impulsivity involve proper function of the mesocorticolimbic dopamine (DA) system.19
It is yet to be fully established how IUGR-induced changes in insulin alter impulsivity and whether regulators of mesocorticolimbic DA development are involved. Tyrosine hydroxylase (TH) is the rate-limiting enzyme which converts L-tyrosine to L-3,4-dihydroxyphenylalanine, which ultimately turns into DA.20 In response to sweet food intake, adult fetal growth restricted (FR) animals, meaning offspring of female rodents that were subjected to caloric restriction during pregnancy, show an increase in the expression of TH in the prefrontal cortex (PFC) in comparison to controls (referred to as AdLib in the manuscript as the dams were fed ad libitum diet).9,21 Since DA in the PFC is involved in attention and impulsivity-related behaviors in rats, increased TH expression in FR animals could be associated to the animals’ reward-associated value of sweet food, as evident by Alves et al. where the mean change in DA signaling in PFC was measured in controls and FR animals in response to standard chow and Froot Loops® consumption.22 They observed that Froot Loops® consumption caused a blunted increase in DA concentration in FR animals when compared to AdLib animals.22 Another study showed that FR animals exhibited higher peripheral insulin levels than AdLib animals23 suggesting that IUGR modifies the modulation of mesolimbic DA neurons by insulin beyond the well-known alteration in peripheral glucose metabolism.22 In controls, the PFC DA levels in response to Froot Loops® consumption were greater than the levels in response to standard chow, while FR animals did not show a difference in DA levels in response to chow and Froot Loops®. The weakened response to sweet food seen in the PFC of IUGR animals may result from decreased activation of striatal areas, leading to a lower valuation of the reward magnitude and therefore, favoring impulsive choices for immediate small rewards.21 Within the PFC, there seems to be a positive correlation between impulsive choice and expression of D1 receptors19 since micro-infusion of D1 receptor antagonist in the area increased impulsive choice. Altered expression of D2 receptors, which are found to be increased in the PFC of IUGR animals at 160 d of life, is also linked with impulsive choice.24 Together, these findings indicate that fetal adversity alters brain sensitivity to insulin and modulates behavioral responses to palatable foods.22,23 Additionally, in rodents exposed to IUGR, there is systemic hyperglycemia/hyperinsulinemia25 and alterations in the expression of genes or proteins associated with DA signaling in the PFC, including TH, pTH, D1, and D2.9,21,23,26 These metabolic and neurochemical effects are accompanied by changes in cognitive flexibility, sensitivity to reward, and poor inhibitory control.9,21,22,26,27 The behavioral and metabolic phenotype observed in animals exposed to FR recapitulates the profile detected in humans born after exposure to prenatal adversity28–48 with striking accuracy. In humans, differences in DA function associate with the increased palatable food consumption in IUGR individuals.49 Furthermore, Silveira et al. discovered, in two independent cohorts, that catch-up growth, an insulin-dependent process, influences decision-making and inhibitory control specifically in IUGR children tested in tasks that did not involve the choice for food rewards.14 Given the involvement of the mesocorticolimbic DA pathway in impulsive behaviors, we are interested in investigating the molecules involved in the maturation of this pathway and characterize the effect of early life adversity in this developmental process by using the FR animal model.
The guidance cue Netrin-1 participates in the developmental organization of neural networks by either attracting or repelling extending axons and dendrites and is highly expressed in terminal fields of mesocorticolimbic DA neurons including the PFC.50 Responses to Netrin-1 can be modulated by regulating the availability of DCC (deleted in colorectal cancer) and UNC5 (uncoordinated) receptors. DCC receptors in DA axons promote target recognition events in the NAcc, preventing them from continuing to grow to the PFC,51–55 and thereby organizing PFC local circuits50,56,57 and inhibitory control in adulthood.56,57 DCC receptors are also expressed by PFC local neurons across the lifespan and are involved in susceptibility/resilience to chronic social defeat stress.58 Changes in DCC expression in DA and cortical neurons induced by exposure to drugs of abuse or to stress, respectively, have been shown to be mediated by the microRNA-218 (miR-218).58,59 miR-218 can be detected in blood with circulating levels matching its expression in brain.60,61 Both chronic caloric restriction and chronic high fat diet exposure in adulthood increase the expression of miR-218 in the hypothalamus.62 However, the possible persistent effect of dietary manipulations during fetal development, as well as the effect of differences in insulin sensitivity on the expression of miR-218 and its guidance cue receptor targets in the PFC remain unknown.
Based on these findings, we aimed to investigate if exposure to prenatal dietary restriction alters guidance cue systems known to be intimately involved in DA, PFC, cognitive development, and in vulnerability to chronic stress. Using the FR model of IUGR in male rats, we measured changes in the Netrin-1/DCC guidance cue pathway in the PFC at birth and in adulthood and assessed if alterations in insulin sensitivity and microRNA levels are involved in the process.
Methods
Primiparous Sprague Dawley rats of approximately 70–80 d were maintained in a controlled environment: standard dark/light cycle of 12 h each, ambient temperature of the colony housing and testing rooms was 22 ± 1°C, relative humidity level was approximately 20–50%, cage cleaning once a week, and food and water provided ad libitum. Estrous cycle was determined daily by vaginal smearing and females were time mated. Gestation was confirmed at day 1 by visualizing the presence of sperm cells on the vaginal smear. On gestational day 10, dams were randomly allocated into one of the following dietary groups: control group (AdLib), which received an ad libitum diet of standard laboratory chow (3.1 kcal/g, 24% protein, 18% fat, 58% carbohydrate; Teklad Diets®), or a 50% food restricted group (FR), based on the IUGR model described by Desai et al.,63 which received 50% of the ad libitum-fed dams’ intake (determined by daily quantification of normal intake in a separate cohort of pregnant Sprague Dawley rats). The food restriction occurred from day 10 of pregnancy until the pups were born. The average calories consumption from day 10 to day 22 of pregnancy was 103.4 ± 9.3 kcal/d in AdLib group and 51.7 ± 4.6 kcal/d in FR group. Belkacemi et al.64 showed that this IUGR model increases FR dams’ plasma corticosterone levels compared to AdLib dams, mimicking a stressful environment.
Within 24 h of birth, pups from each litter were individually weighed and standardized to a maximum of eight pups per litter, with four females and four males. Cross-fostering was performed, and all litters were adopted by AdLib dams. Only male offspring were used in this study and only one animal per litter was used in each experiment. Tissues were collected in two separate age groups. The first group’s tissue was collected on the day the animals were born. The second group’s pups were weaned on postnatal day (PND) 21, separated into groups of two same-sex (same litter/group) individuals per cage, and kept in a controlled environment as previously described. Except for the cage cleaning once a week, animals were left undisturbed from PND 21 until tissue collection which occurred at approximately PND 100.
Tissue collection
Tissue collection for protein level analysis took place at two different ages: PND 0 and PND 100. For the PND 0 group, the animals were decapitated, the brains were quickly removed and a series of 0.5 mm brain punches from the PFC were dissected from brain coronal sections corresponding to plates 7–9 of the Brain Rat Atlas.65 Tissue was stored at −80°C until further analysis. For the PND 100 group, the animals received either a peripheral saline injection or insulin injection. Fifteen minutes after the injection, the animals were decapitated, the brains were quickly removed, the PFC was dissected through 1.0 mm thick coronal slices with the aid of an atlas,66 and the tissue was stored at −80°C until further analysis.
Western blot
Total protein fractions were isolated using the mirVana™ PARIS RNA and Native Protein Purification Kit Protocol (Cat#AM1556, Thermo Scientific, Toronto, ON, Canada). Briefly, protein samples (20 μg) were separated on a 10% SDS-PAGE and transferred to a PVDF membrane that was incubated overnight at 4°C with antibodies against DCC (1:1000, Cat#554223, BD Pharmingen), Netrin-1 (1:1000, cat# ab126729, Abcam), and alpha-tubulin (1:2000, Cat #2144S, Cell Signaling). We obtained the optical density for each Netrin-1, DCC, and tubulin band, and the fold change was calculated by normalizing to tubulin’s optical density.
Tissue RNA extraction and quantitative real-time PCR
Total RNA and microRNA fraction were isolated from the frozen tissue using the Norgen Biotek Corp. RNA/DNA/Protein Purification Plus Kit (Cat#47700, Norgen Biotek Corp, Thorold, ON, Canada) as per manufacture’s instruction. All RNA samples were determined to have 260/280 and 260/230 values ≥1.8, using the Nanodrop 1000 system (Thermo Scientific, Toronto, ON, Canada). Reverse transcription for Dcc, Netrin-1 (Ntn-1), and Glyceraldehyde-3-phosphatedehydrogenase (GAPDH) mRNA were performed using a High-Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA) according to manufacturer’s instructions. Real-time PCR, using TaqMan assay (Applied Biosystems, Foster City, CA) was carried out with an Applied Biosystems 7900HT RT PCR system. Data for Dcc and Ntn-1 mRNA expression were analyzed by using the Relative Quantification standard curve method and Gapdh was used as reference gene. In all cases, the real-time PCR was run in technical triplicates.
HEK293 in vitro analysis
HEK293 cells were cultured in 12-well plates with DMEM medium with 10% fetal bovine serum (FBS). Once the cells reached 80% of confluency, they were starved in serum-free medium for 16–18 h. After that period two different treatments were applied: (1) exposure to different concentrations of insulin (50, 100 or 200 nM) or basal medium to measure total micro-RNA levels after 6 h and (2) exposure to 100 mM of insulin or basal medium to measure total micro-RNA levels at different time points (10, 30, 60 or 360 min). In both cases, a control, undisturbed cell culture was kept (UNT). Cell cultures were harvested by trypsinization (0.125 trypsin/1% EDTA in PBS) and homogenized using the miRNeasy Mini Kit (Qiagen, Toronto, ON, Canada). Total RNA and microRNA fraction were isolated according to the manufacturer’s instructions. All RNA samples were determined to have 260/280 and 260/230 values ≥1.8, using the Nanodrop 1000 system (Thermo Fisher Scientific, Toronto, ON, Canada). Reverse transcription for miR-218 was performed using the TaqMan MicroRNA Reverse Transcription Kit and TaqMan probes (Applied Biosystems, QC, Canada). Real-time PCR was run in technical triplicates with an Applied Biosystems QuantStudio RT PCR system (Applied Biosystems, QC, Canada). The small nucleolar RNA (snoRNA) RNU6B was used as an endogenous control of miRNA measures. Expression levels of miR-218 were calculated using the Relative Quantitation standard curve method and normalized by RNU6B. The expression of miR-218 was relativized to UNT group and presented as a fold change.
Statistical analysis
Student’s t-test was used for the Western Blot analysis for PND 0 animal results. All variables were expressed as mean ± standard error of the mean (SEM). One-way ANOVA was used to analyze the Western Blot analysis for PND 100 animal results and the in vitro results from HEK293 cultures followed by Tukey post hoc test when appropriate. Results were considered significant when p ≤ 0.05. Data were analyzed using the RStudio software.67
Results
PND 0 measures
Protein levels of Netrin-1 and DCC receptors in the PFC were measured in AdLib and FR animals of age PND 0 (AdLib weight = 7.55 ± 0.21, FR weight = 6.84 ± 0.11, Student’s t-test, n = 8/group, t(14) = 3.039; p = 0.009) as shown in Fig. 1. Netrin-1 protein levels were found to be downregulated in the PFC of FR animals when compared to AdLib animals (W(8) = 51; p = 0.0499). DCC protein expression was not significantly different in the PFC of FR animals when compared to AdLib animals (t(14) = −1.25; p = 0.231).
Figure 1.

PFC Netrin-1 and DCC expression in PND0 males. Levels of Netrin-1 and DCC receptors in the PFC were measured in AdLib and FR animals of age PND 0. (a) Netrin-1 protein levels were found to be downregulated in the PFC of FR animals when compared to AdLib animals (W(8) = 51; p = 0.0499). (b) DCC protein expression was not significantly different in the PFC of FR animals when compared to AdLib animals (t(14) = −1.25; p = 0.231).
PND 100 measures
Levels of Ntn-1 and Dcc mRNA in the PFC were measured 15 min after an intraperitoneal saline or insulin injection after a 4 h fast (1 IU/kg) in AdLib and FR animals of age PND 100 as shown in Fig. 2. There were no significant differences in Ntn-1 mRNA levels between the four groups: AdLib Saline, AdLib Insulin, FR Saline, and FR Insulin (F(3,33) = 0.066, p = 0.977). There was a significant difference in Dcc levels across the four groups (F(3,33) = 4.407, p = 0.0103). A Tukey post hoc test revealed that there is a significant upregulation of Dcc mRNA in AdLib animals which received the insulin injection when comparing to AdLib animals which were administered a saline injection (p = 0.008). We also found a significant downregulation of Dcc mRNA levels in FR animals which received a saline injection when compared to the AdLib animals treated with insulin (p = 0.032).
Figure 2.

PFC Netrin-1 and Dcc mRNA expression in PND 100 males. Levels of Netrin-1 and Dcc in the PFC were measured 15 min after a peripheral saline or insulin injection in AdLib and FR animals. (a) There were no significant differences in Netrin-1 mRNA levels across the four groups: AdLib Saline, AdLib Insulin, FR Saline, and FR Insulin (F(3,33) = 0.066, p = 0.98). (b) There was a significant difference in Dcc mRNA levels over groups (F(3,33) = 4.41, p = 0.0103). There is a significant upregulation of Dcc mRNA in AdLib animals treated with an insulin injection when comparing to AdLib animals administered a saline injection (p = 0.008) and a significant downregulation in Dcc mRNA levels in FR animals treated with the saline injection when compared to the AdLib animals given the insulin injection (p = 0.032).
miR-218 measures in HEK293 cultures
miR-218 expression was measured in response to four different insulin concentrations, as shown in Fig. 3, in addition to the control group (UNT): 0 nM, 50 nM, 100 nM, 200 nM. There was a significant difference in the fold change of miR-218 between the five groups (F(4,10) = 4.394, p = 0.0262). A Tukey post hoc test revealed that there is a significant upregulation in miR-218 in response to 100 nM of insulin (p = 0.018) when comparing to the control group.
Figure 3.
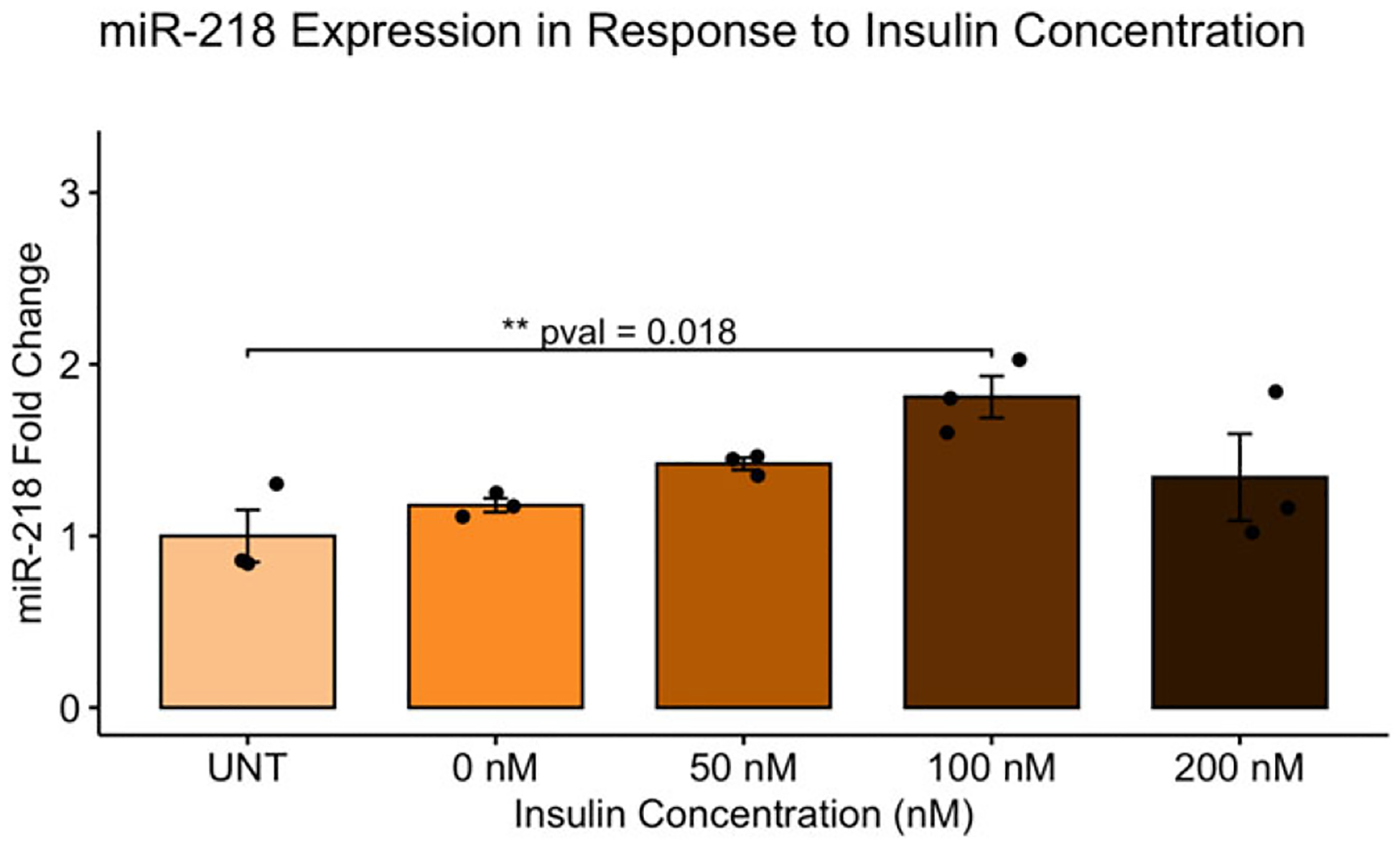
Insulin regulates miR-218 expression in a concentration-dependent manner. miR-218 expression was measured in response to four different insulin concentrations in addition to the control group (UNT): 0 nM, 50 nM, 100 nM, 200 nM. There was a significant difference in miR-218 expression across groups (F(4,10) = 4.394, p = 0.0262). miR-218 levels were upregulated at 100 nM of insulin when compared to the control group.
miR-218 expression was measured over a time curve when exposed to 100 nM of insulin, as shown in Fig. 4, at five different time points in addition to the control group (UNT): 0 min, 10 min, 30 min, 60 min, and 360 min. There was a significant difference in miR-218 expression across the six groups (F(5,12) = 27.26, p < 0.001). A Tukey post hoc test revealed that there is a significant upregulation in miR-218 at 10 min (p = 0.004), 30 min (p < 0.001), 60 min (p < 0.001), and 360 min (p < 0.001) when comparing to the control group, upregulation at 10 min (p = 0.022), 30 min (p < 0.001), 60 min (p < 0.001), and 360 min (p < 0.001) when comparing to 0 min, and upregulation at 60 min (p = 0.043) when comparing to 10 min.
Figure 4.

Effects of insulin signaling on miR-218 levels over time. Fold change in miR-218 were measured in response to 100 nM of insulin over a time curve at five different time points in addition to the control group (UNT): 0 min, 10 min, 30 min, 60 min, and 360 min. There was a significant difference in miR-218 expression across groups (F(5,12) = 27.26, p < 0.001). miR-218 levels were upregulated at 10 min, 30 min, 60 min, and 360 min in comparison to the control group.
Discussion
The food restriction model that we used in this study was based on the successful IUGR model described by Desai et al.25 The 50% food restriction protocol is quite stressful to the dams, increasing corticosterone levels at gestational day 20,64 and impacting offspring birth weight21–23,68 and brain weight at birth.69 This model was established as a useful approach able to mimic many of the adverse outcomes frequently observed in IUGR in humans, including decreased plasma leptin levels, increased food intake, obesity, increased percentage of body fat, insulin resistance, and catch-up growth.70 More recently, this model has been used to investigate alterations in feeding behavior that in some way influence food choices and consumption.25 In this context, interesting changes were found in attentional skills, in physical activity, in reward sensitivity (indicated by a decreased conditioning for palatable food in a place preference test), in hedonic responses to sweet food, and in impulsive behavior. Therefore, this model was best suited for our study where we investigated whether environmental insults early in life, modeled through prenatal dietary restriction, affect the Netrin-1/DCC guidance cue system which is known to be involved in DA and PFC maturation and function.54,59,71,72 We showed that Netrin-1 protein levels are downregulated in the PFC of FR animals in comparison to AdLib animals at age PND 0, indicating that prenatal dietary restriction affects developmental guidance cue systems as early as birth. In the matured cortex, at PND 100, we do not see changes in Ntn-1 mRNA transcript expression at baseline (AdLib Saline group and FR Saline group) suggesting that the changes observed at birth do not persist into adulthood. FR does not alter baseline DCC protein expression in the PFC at birth or in mRNA transcript levels in adulthood. However, at PND 100, AdLib animals showed Dcc mRNA upregulation in response to an insulin challenge when compared to AdLib animals who received saline, but this response is absent in FR animals. This lack of regulation of Dcc by insulin in FR rodents is in line with our previous work showing that FR animals have different sensitivity to insulin23 and suggest that exposure to fetal growth restriction prevents insulin-induced regulation of Dcc expression in the PFC in adulthood. It was vital to test the effects of insulin on the guidance cues involved in the maturation of the DA pathway within the IUGR context because the IUGR model has been shown to affect insulin signaling and sensitivity within the brain. It is important to note that protein levels were measured in PND 0 animals while mRNA levels were measured in PND 100 animals but the positive correlation between mRNA and protein expression levels73 allows us to compare these transcripts between these ages.
The expression of the Netrin-1/DCC system in the mesocorticolimbic DA systems, including the PFC, decreases dramatically from early postnatal life to adulthood.60,74,75 This shift in expression levels coincides with the distinct role that Netrin-1 and DCC play across postnatal development – from axonal pathfinding and synaptic pruning/refinement in early postnatal life to synaptic plasticity in adulthood.71,76 It is likely that the early changes in Netrin-1 expression observed in FR animals impact the core organization of developing PFC neuronal networks, including DA axonal growth, while the altered insulin-response in DCC receptor expression in adulthood involve reorganization of matured local synaptic connectivity. In the PFC, Netrin-1 has been shown to be highly expressed by pyramidal and GABA interneurons, while DCC receptors are expressed by pyramidal neurons as well as non-DA axons.50,75 It remains to be determined whether changes in guidance cue expression induced by FR or by insulin administration in adulthood localize to specific PFC neuronal populations and if they are predominately occurring at the pre- or postsynaptic level.
miR-218 levels in the brain have been known to be affected by altered metabolism as shown by Sangiao-Alvarellos et al.62 where caloric restriction and high fat diet in adulthood resulted in an increase in miR-218 expression in the hypothalamus in comparison to the control group. Since miR-218 regulates Dcc mRNA and DCC protein expression,59 it could be involved in the effect of prenatal dietary restriction on this guidance cue system. To assess this hypothesis, we did in vitro analysis involving miR-218 in HEK-293 cells. We showed that there is a time-dependent and dose-dependent increase in miR-218 expression in HEK-293 cells. Whether and how insulin-induced regulation of miR-218 may be involved in the changes in DCC expression observed in the PFC in adulthood remains to be established. Indeed, previous studies have shown that miR-218 can stimulate the transcription of certain transcripts77 and cumulative evidence indicates that miRNAs can upregulate genes and protein expression in specific cell types and under particular conditions.78 Since miR-218 is a regulator of Dcc gene expression and our in vitro results show that insulin regulates miR-218 levels, we suggest that FR-induced changes in insulin sensitivity could be affecting Dcc expression via miR-218.
In conclusion, our work explores how insulin mediates the effects of prenatal dietary restriction on the Netrin-1/DCC guidance cue system which is known to be involved in PFC maturation. Previous animal work from our lab has shown that animals exposed to prenatal dietary restriction showed a pronounced aversion to delayed rewards in addition to an increase in PFC D2 levels.22 Our current findings may partially explain the mechanism involved in this relationship. This guidance cue pathway is affected by prenatal dietary restriction through changes in insulin, as shown by our results, which could ultimately reorganize pre- and postsynaptic networks and impact executive functions in adulthood. As the FR model parallels many of the alterations observed in IUGR in humans, the current findings have the potential to inform future studies to explore these mechanisms in human populations.
Financial support.
We acknowledge the support of the Natural Sciences and Engineering Research Council of Canada (NSERC), RGPIN-2018-05063 (PPS) RGPIN-2020-04703 to C.F.; The National Institute on Drug Abuse at the National Institutes of Health (R01DA037911 to C.F.), the Canadian Institutes of Health Research (FRN: 156272 to C.F.). This research was supported by the Fonds de recherche du Québec - Santé (FRQS to AB and PPS), awarded for the project: Les variations de la function de l’insuline modulent l’adversité au début de la vie sur les compartements liés à la dopamine mésocorticolimbique.
Footnotes
Competing interests. None.
Ethical standard. The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national guides on the care and use of laboratory animals sprague dawley and have been approved by the institutional committees (Research Ethics Committee of Hospital de Clínicas de Porto Alegre (GPPG/HCPA); McGill University; Douglas Mental Health University Institute AUP DOUG-8018).
References
- 1.Barker DJ, Hales CN, Fall C, Osmond C, Phipps K, Clark P. Type 2 (non-insulin-dependent) diabetes mellitus, hypertension and hyperlipidaemia (syndrome X): relation to reduced fetal growth. Diabetologia. 1993; 36(1), 62–67. [DOI] [PubMed] [Google Scholar]
- 2.Hales CN, Barker DJ. Type 2 (non-insulin-dependent) diabetes mellitus: the thrifty phenotype hypothesis. Diabetologia. 1992; 35(7), 595–601. [DOI] [PubMed] [Google Scholar]
- 3.Mericq V, Ong K, Bazaes R, et al. Longitudinal changes in insulin sensitivity and secretion from birth to age three years in small-and appropriate-for-gestational-age children. Diabetologia. 2005; 48(12), 2609–2614. [DOI] [PubMed] [Google Scholar]
- 4.Jensen CB, Storgaard H, Dela F, Holst JJ, Madsbad S, Vaag AA. Early differential defects of insulin secretion and action in 19-year-old Caucasian men who had low birth weight. Diabetes. 2002; 51(4), 1271–1280. [DOI] [PubMed] [Google Scholar]
- 5.Gatford K, Simmons R, De Blasio M, Robinson J, Owens J. Placental programming of postnatal diabetes and impaired insulin action after IUGR. Placenta. 2010; 31, S60–S65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yan J, Yang H. Gestational diabetes mellitus, programing and epigenetics. J Matern Fetal Neonatal Med. 2014; 27(12), 1266–1269. [DOI] [PubMed] [Google Scholar]
- 7.Ruchat S-M, Hivert M-F, Bouchard L Epigenetic programming of obesity and diabetes by in utero exposure to gestational diabetes mellitus. Nutr Rev. 2013; 71(suppl_1), S88–S94. [DOI] [PubMed] [Google Scholar]
- 8.Nielsen JH, Haase TN, Jaksch C, et al. Impact of fetal and neonatal environment on beta cell function and development of diabetes. Acta Obstet Gynecol Scand. 2014; 93(11), 1109–1122. [DOI] [PubMed] [Google Scholar]
- 9.Dalle Molle R, Laureano DP, Alves MB, et al. Intrauterine growth restriction increases the preference for palatable foods and affects sensitivity to food rewards in male and female adult rats. Brain Res. 2015; 1618, 41–49. [DOI] [PubMed] [Google Scholar]
- 10.Gatford KL, Simmons RA. Prenatal programming of insulin secretion in intrauterine growth restriction. Clin Obstet Gynecol. 2013; 56(3), 520–528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Cianfarani S, Germani D, Branca F. Low birthweight and adult insulin resistance: the “catch-up growth” hypothesis. Arch Dis Child Fetal Neonatal Ed. 1999; 81(1), F71–F73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hakan N, Aydin M, Dilli D, Zenciroglu A, Okumus N. Transient hyperinsulinemia may be responsible from electrolyte abnormalities of refeeding syndrome seen in very low birth weight infants with intrauterine growth-restriction. J Perinatol. 2014; 34(3), 247–247. [DOI] [PubMed] [Google Scholar]
- 13.Simmons RA, Templeton LJ, Gertz SJ. Intrauterine growth retardation leads to the development of type 2 diabetes in the rat. Diabetes. 2001; 50(10), 2279–2286. [DOI] [PubMed] [Google Scholar]
- 14.Silveira PP, Pokhvisneva I, Gaudreau H, et al. Birth weight and catch up growth are associated with childhood impulsivity in two independent cohorts. Sci Rep. 2018; 8(1), 1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Winstanley CA, Theobald DE, Dalley JW, Cardinal RN, Robbins TW. Double dissociation between serotonergic and dopaminergic modulation of medial prefrontal and orbitofrontal cortex during a test of impulsive choice. Cereb Cortex. 2006; 16(1), 106–114. [DOI] [PubMed] [Google Scholar]
- 16.van der Laan LN, Barendse ME, Viergever MA, Smeets PA. Subtypes of trait impulsivity differentially correlate with neural responses to food choices. Behav Brain Res. 2016; 296, 442–450. [DOI] [PubMed] [Google Scholar]
- 17.Pardey MC, Kumar NN, Goodchild AK, Cornish JL. Catecholamine receptors differentially mediate impulsive choice in the medial prefrontal and orbitofrontal cortex. J Psychopharmacol. 2013; 27(2), 203–212. [DOI] [PubMed] [Google Scholar]
- 18.Tedford SE, Persons AL, Napier TC. Dopaminergic lesions of the dorsolateral striatum in rats increase delay discounting in an impulsive choice task. PloS One. 2015; 10(4), e0122063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Loos M, Pattij T, Janssen MC, et al. Dopamine receptor D1/D5 gene expression in the medial prefrontal cortex predicts impulsive choice in rats. Cereb Cortex. 2010; 20(5), 1064–1070. [DOI] [PubMed] [Google Scholar]
- 20.Kobayashi K, Nagatsu T. Tyrosine hydroxylase. In Primer on the Autonomic Nervous System, 2012; pp. 45–47. Elsevier, New York. [Google Scholar]
- 21.Alves MB, Dalle Molle R, Desai M, Ross MG, Silveira PP. Increased palatable food intake and response to food cues in intrauterine growth-restricted rats are related to tyrosine hydroxylase content in the orbitofrontal cortex and nucleus accumbens. Behav Brain Res. 2015; 287, 73–81. [DOI] [PubMed] [Google Scholar]
- 22.Alves MB, Laureano DP, Dalle Molle R, et al. Intrauterine growth restriction increases impulsive behavior and is associated with altered dopamine transmission in both medial prefrontal and orbitofrontal cortex in female rats. Physiol Behav. 2019; 204, 336–346. [DOI] [PubMed] [Google Scholar]
- 23.Laureano D, Alves M, Miguel P, et al. Intrauterine growth restriction modifies the accumbal dopaminergic response to palatable food intake. Neuroscience. 2019; 400, 184–195. [DOI] [PubMed] [Google Scholar]
- 24.Simon NW, Beas BS, Montgomery KS, Haberman RP, Bizon JL, Setlow B. Prefrontal cortical-striatal dopamine receptor m RNA expression predicts distinct forms of impulsivity. Eur J Neurosci. 2013; 37(11), 1779–1788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Desai M, Gayle D, Babu J, Ross MG. The timing of nutrient restriction during rat pregnancy/lactation alters metabolic syndrome phenotype. Am J Obstet Gynecol. 2007; 196(6), 555.e551–555.e557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Vucetic Z, Totoki K, Schoch H, et al. Early life protein restriction alters dopamine circuitry. Neuroscience. 2010; 168(2), 359–370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.da Silva AAM, Borba TKF, de Almeida Lira L, et al. Perinatal undernutrition stimulates seeking food reward. Int J Dev Neurosci. 2013; 31(5), 334–341. [DOI] [PubMed] [Google Scholar]
- 28.Barbieri MA, Portella AK, Silveira PP, et al. Severe intrauterine growth restriction is associated with higher spontaneous carbohydrate intake in young women. Pediatr Res. 2009; 65(2), 215–220. [DOI] [PubMed] [Google Scholar]
- 29.Reis RS, Dalle Molle R, Machado TD, et al. Impulsivity-based thrifty eating phenotype and the protective role of n-3 PUFAs intake in adolescents. Transl Psychiatry. 2016; 6(3), e755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Silveira PP, Agranonik M, Faras H, et al. Preliminary evidence for an impulsivity-based thrifty eating phenotype. Pediatr Res. 2012; 71(3), 293–298. [DOI] [PubMed] [Google Scholar]
- 31.Ayres C, Agranonik M, Portella AK, Filion F, Johnston CC, Silveira PP. Intrauterine growth restriction and the fetal programming of the hedonic response to sweet taste in newborn infants. Int J Pediatr. 2012; 2012, 657379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Silveira PP. Playing with food: the fetal programming of food preferences. Obesity (Silver Spring). 2014; 22(5), 1210. [DOI] [PubMed] [Google Scholar]
- 33.Bischoff AR, Portella AK, Paquet C, et al. Low birth weight is associated with increased fat intake in school-aged boys. Br J Nutr. 2018; 119(11), 1295–1302. [DOI] [PubMed] [Google Scholar]
- 34.Silveira PP, Pokhvisneva I, Gaudreau H, et al. Birth weight and catch up growth are associated with childhood impulsivity in two independent cohorts. Sci Rep. 2018; 8(1), 13705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Abstracts of the 4th World Congress on Developmental Origins of Health and Disease (DOHaD), 13–16 September 2006, Educatorium, University of Utrecht, The Netherlands. Early Hum Dev. 2006; 82(8), 493–565. [Google Scholar]
- 36.Fernandes FS, Portella AK, Barbieri MA, et al. Risk factors for sedentary behavior in young adults: similarities in the inequalities. J Dev Orig Health Dis. 2010; 1(4), 255–261. [DOI] [PubMed] [Google Scholar]
- 37.Portella AK, Kajantie E, Hovi P, et al. Effects of in utero conditions on adult feeding preferences. J Dev Orig Health Dis. 2012; 3(3), 140–152. [DOI] [PubMed] [Google Scholar]
- 38.Portella AK, Silveira PP. Neurobehavioral determinants of nutritional security in fetal growth-restricted individuals. Ann N Y Acad Sci. 2014; 1331(1), 15–33. [DOI] [PubMed] [Google Scholar]
- 39.Dalle Molle R, Silveira PP. Small for gestational age children have specific food preferences. J Pediatr. 2015; 166(6), 1547. [DOI] [PubMed] [Google Scholar]
- 40.Laureano DP, Molle RD, Portella AK, Silveira PP. Facial expressions in small for gestational age newborns. J Child Neurol. 2016; 31(3), 398–399. [DOI] [PubMed] [Google Scholar]
- 41.Agranonik M, Portella AK, Hamilton J, et al. Breastfeeding in the 21st century. Lancet. 2016; 387(10033), 2088–2089. [DOI] [PubMed] [Google Scholar]
- 42.Escobar RS, O’Donnell KA, Colalillo S, et al. Better quality of mother-child interaction at 4 years of age decreases emotional overeating in IUGR girls. Appetite. 2014; 81, 337–342. [DOI] [PubMed] [Google Scholar]
- 43.Reis RS, Bernardi JR, Steiner M, et al. Poor infant inhibitory control predicts food fussiness in childhood - a possible protective role of n-3 PUFAs for vulnerable children. Prostaglandins Leukot Essent Fatty Acids. 2015; 97, 21–25. [DOI] [PubMed] [Google Scholar]
- 44.Dalle Molle R, Bischoff AR, Portella AK, Silveira PP. The fetal programming of food preferences: current clinical and experimental evidence. J Dev Orig Health Dis. 2015; 7(3), 1–9. DOI: 10.1017/S2040174415007187. [DOI] [PubMed] [Google Scholar]
- 45.Portella AK, Silveira PP. Parenting: roots of the sweet tooth. Science. 2014; 345(6204), 1571–1572. [DOI] [PubMed] [Google Scholar]
- 46.Silveira PP, Portella AK, Goldani MZ. Obesity in Latin America: similarity in the inequalities. Lancet. 2005; 366(9484), 451–452. [DOI] [PubMed] [Google Scholar]
- 47.Mucellini AB, Manfro GG, Silveira PP. Tackling obesity: challenges ahead. Lancet. 2015; 386(9995), 1. [DOI] [PubMed] [Google Scholar]
- 48.Mucellini AB, Miguel PM, Dalle Molle R, et al. Diminished insulin sensitivity is associated with altered brain activation to food cues and with risk for obesity - implications for individuals born small for gestational age. Appetite. 2021; 169, 105799. DOI: 10.1016/j.appet.2021.105799. [DOI] [PubMed] [Google Scholar]
- 49.Silveira PP, Pokhvisneva I, Gaudreau H, et al. Fetal growth interacts with multilocus genetic score reflecting dopamine signaling capacity to predict spontaneous sugar intake in children. Appetite. 2018; 120, 596–601. [DOI] [PubMed] [Google Scholar]
- 50.Manitt C, Mimee A, Eng C, et al. The netrin receptor DCC is required in the pubertal organization of mesocortical dopamine circuitry. J Neurosci. 2011; 31(23), 8381–8394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Manitt C, Labelle-Dumais C, Eng C, et al. Peri-pubertal emergence of UNC-5 homologue expression by dopamine neurons in rodents. PloS One. 2010; 5(7), e11463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hong K, Hinck L, Nishiyama M, Poo M-M, Tessier-Lavigne M, Stein E. A ligand-gated association between cytoplasmic domains of UNC5 and DCC family receptors converts netrin-induced growth cone attraction to repulsion. Cell. 1999; 97(7), 927–941. [DOI] [PubMed] [Google Scholar]
- 53.Keleman K, Dickson BJ. Short-and long-range repulsion by the Drosophila Unc5 netrin receptor. Neuron. 2001; 32(4), 605–617. [DOI] [PubMed] [Google Scholar]
- 54.Flores C. Role of netrin-1 in the organization and function of the mesocorticolimbic dopamine system. J Psychiatry Neurosci. 2011; 36(5), 296–310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Liu C, Ke X, Wang Y, et al. The level of netrin-1 is decreased in newly diagnosed type 2 diabetes mellitus patients. BMC Endocr Disord. 2016; 16(1), 33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Manitt C, Eng C, Pokinko M, et al. DCC orchestrates the development of the prefrontal cortex during adolescence and is altered in psychiatric patients. Transl Psychiatry. 2013; 3(12), e338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Grant A, Hoops D, Labelle-Dumais C, et al. Netrin-1 receptor-deficient mice show enhanced mesocortical dopamine transmission and blunted behavioural responses to amphetamine. Eur J Neurosci. 2007; 26(11), 3215–3228. [DOI] [PubMed] [Google Scholar]
- 58.Torres-Berrío A, Lopez JP, Bagot RC, et al. DCC confers susceptibility to depression-like behaviors in humans and mice and is regulated by miR-218. Biol Psychiatry. 2017; 81(4), 306–315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Cuesta S, Restrepo-Lozano JM, Silvestrin S, et al. Non-contingent exposure to amphetamine in adolescence recruits miR-218 to regulate Dcc expression in the VTA. Neuropsychopharmacology. 2018; 43(4), 900–911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Torres-Berrío A, Morgunova A, Giroux M, Cuesta S, Nestler EJ, Flores C. miR-218 in adolescence predicts and mediates vulnerability to stress. Biol Psychiatry. 2021; 89(9), 911–919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.van der Zee YY, Eijssen LM, Mews P, et al. Blood miR-144–3p: a novel diagnostic and therapeutic tool for depression. Mol Psychiatry. 2022; 27(11), 4536–4549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Sangiao-Alvarellos S, Pena-Bello L, Manfredi-Lozano M, Tena-Sempere M, Cordido F. Perturbation of hypothalamic microRNA expression patterns in male rats after metabolic distress: impact of obesity and conditions of negative energy balance. Endocrinology. 2014; 155(5), 1838–1850. [DOI] [PubMed] [Google Scholar]
- 63.Desai M, Gayle D, Babu J, Ross MG. Programmed obesity in intrauterine growth-restricted newborns: modulation by newborn nutrition. Am J Physiol. 2005; 288(1), R91–R96. [DOI] [PubMed] [Google Scholar]
- 64.Belkacemi L, Jelks A, Chen C-H, Ross MG, Desai M. Altered placental development in undernourished rats: role of maternal glucocorticoids. Reprod Biol Endocrinol. 2011; 9(1), 1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Paxinos G, Watson C. The Rat Brain in Stereotaxic Coordinates, 1998. Academic Press, Burlington, MA. [Google Scholar]
- 66.Paxinos G, Watson C. The Rat Brain in Stereotaxic Coordinates, 6th ed., 2007. Elsevier Academic Press, San Diego. [Google Scholar]
- 67.Allaire J RStudio: Integrated Development Environment for R, 2012. RStudio, Boston, MA. [Google Scholar]
- 68.Cunha FS, Dalle Molle R, Portella AK, et al. Both food restriction and high-fat diet during gestation induce low birth weight and altered physical activity in adult rat offspring: the “Similarities in the Inequalities” model. PLoS One. 2015; 10(3), e0118586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Potiris A, Manousopoulou A, Zouridis A, et al. The effect of prenatal food restriction on brain proteome in appropriately grown and growth restricted male wistar rats. Front Neurosci. 2021; 15, 665354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Desai M, Gayle D, Babu J, Ross MG. Programmed obesity in intrauterine growth-restricted newborns: modulation by newborn nutrition. Am J Physiol Regul Integr Comp Physiol. 2005; 288(1), R91–R96. [DOI] [PubMed] [Google Scholar]
- 71.Torres-Berrío A, Hernandez G, Nestler EJ, Flores C. The netrin-1/DCC guidance cue pathway as a molecular target in depression: translational evidence. Biol Psychiatry. 2020; 88(8), 611–624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Vosberg DE, Leyton M, Flores C. The Netrin-1/DCC guidance system: dopamine pathway maturation and psychiatric disorders emerging in adolescence. Mol Psychiatry. 2020; 25(2), 297–307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Guo Y, Xiao P, Lei S, et al. How is mRNA expression predictive for protein expression? A correlation study on human circulating monocytes. Acta Biochim Biophys Sin. 2008; 40(5), 426–436. [DOI] [PubMed] [Google Scholar]
- 74.Cuesta S, Nouel D, Reynolds LM, et al. Dopamine axon targeting in the nucleus accumbens in adolescence requires netrin-1. Front Cell Dev Biol. 2020; 8, 487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Goldman JS, Ashour MA, Magdesian MH, et al. Netrin-1 promotes excitatory synaptogenesis between cortical neurons by initiating synapse assembly. J Neurosci. 2013; 33(44), 17278–17289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Glasgow SD, Ruthazer ES, Kennedy TE. Guiding synaptic plasticity: novel roles for netrin-1 in synaptic plasticity and memory formation in the adult brain. J Physiol. 2021; 599(2), 493–505. [DOI] [PubMed] [Google Scholar]
- 77.Rocchi A, Moretti D, Lignani G, et al. Neurite-enriched microRNA-218 stimulates translation of the GluA2 subunit and increases excitatory synaptic strength. Mol Neurobiol. 2019; 56(8), 5701–5714. [DOI] [PubMed] [Google Scholar]
- 78.Vasudevan S, Tong Y, Steitz JA. Switching from repression to activation: microRNAs can up-regulate translation. Science. 2007; 318(5858), 1931–1934. [DOI] [PubMed] [Google Scholar]


