Abstract
As important messengers of intercellular communication, exosomes can regulate local and distant cellular communication by transporting specific exosomal contents and can also promote or suppress the development and progression of gastric cancer (GC) by regulating the growth and proliferation of tumor cells, the tumor-related immune response and tumor angiogenesis. Exosomes transport bioactive molecules including DNA, proteins, and RNA (coding and noncoding) from donor cells to recipient cells, causing reprogramming of the target cells. In this review, we will describe how exosomes regulate the cellular immune response, tumor angiogenesis, proliferation and metastasis of GC cells, and the role and mechanism of exosome-based therapy in human cancer. We will also discuss the potential application value of exosomes as biomarkers in the diagnosis and treatment of GC and their relationship with drug resistance.
Keywords: Exosomes, Gastric cancer, Immune regulation, Diagnosis, Cancer therapy
Core Tip: Exosome-mediated transport plays an important role in the invasion and metastasis of gastric cancer (GC). In this review, we focus on the immunoregulatory role of exosomes and the relationship between exosomes and GC, with special attention to their role in the growth, invasion, metastasis, and therapeutic resistance of GC, as well as their potential clinical application value as biomarkers and therapeutic targets, and the mechanism and clinical application prospects of exosomal immunotherapy for human tumors.
INTRODUCTION
Gastric cancer (GC) is the fifth most commonly diagnosed cancer and the fourth leading cause of cancer death worldwide, with an incidence of 5.6%. Although the current level of treatment has been greatly improved, the mortality rate is still as high as 7.7%[1]. Metastasis, as the main cause of death in patients with GC, is caused by a multistep molecular cascade reaction inside and outside tumor cells and stromal cells. Exosome-mediated transport plays an important role in the invasion and metastasis of tumor cells[2].
Exosomes are bilayer lipid vesicles with a diameter of 30-100 nm, which are a subgroup of extracellular vesicles (EVs). These vesicles are released into extracellular space by a large number of cells and play a key role in the initiation and development of intercellular signalling networks. EVs include microvesicles, ectosomes, exosomes, membrane particles, apoptotic vesicles and many other types. Many cell types including tumor cells, epithelial cells, mast cells, fibroblasts, stem cells, and immune cells such as macrophages, monocytes, dendritic cells (DCs), B and T lymphocytes, and natural killer cells (NK), can secrete exosomes[3]. Exosomes are composed of lipid bilayers, and their main components are proteins, nucleic acids [mRNA, microRNAs (miRNAs) and DNA], amino acids, lipids and metabolites[4]. These cargos are selectively packaged in exosomes. Conventional markers of exosomes include CD63, tumor susceptibility gene 101 protein (TSG101), ALG2-interacting protein X (ALIX) and proteasome component HSC10. Exosomes are produced by the endolysosomal pathway[5].
Exosomes can transmit information between tumor cells or to other normal cells, thus participating in complex intercellular communication. They also participate in a variety of cellular processes related to cancer development and drug resistance, showing the dual characteristics of promoting and inhibiting cancer.
In this review, we focus on the immunoregulatory role of exosomes and the relationship between exosomes and GC, with special attention to their role in the growth, invasion, metastasis, and therapeutic resistance of GC, as well as their potential clinical application value as biomarkers and therapeutic targets, and the mechanism and clinical application prospects of exosomal immunotherapy for human tumors.
BIOGENESIS OF EXOSOMES
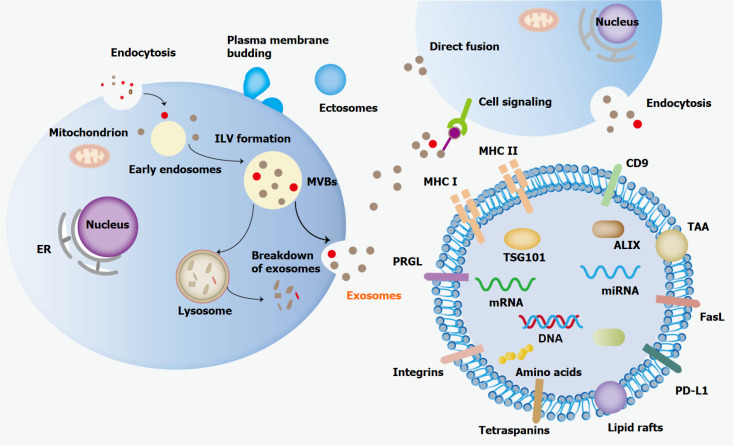
The biogenesis of exosomes includes four main stages: Initiation, endocytosis, formation of multivesicular bodies (MVBs) and exosome secretion[6]. The biogenesis of exosomes begins with the inward budding of the plasma membrane, forming a small intracellular body called endosome[7]. With the maturity of these early endosomes, intraluminal vesicles (ILVs) containing late endosomes will be formed under the control of endosomal sorting complex required for transport (ESCRT). These late endosomes containing ILVs are also called MVBs[8]. MVBs are formed by inward invagination of the endosomal limiting membrane, which causes MVBs to contain multiple ILVs. ILVs are eventually secreted as exosomes with a diameter of approximately 40 to 160 nm through exocytosis and the fusion of MVBs with the plasma membrane[9]. Rab GTPase mediates the intracellular transport of MVBs[6]. Exosomes are absorbed in neighboring cells through direct fusion, endocytosis, or interactions between proteins and recipient cells, and then transmit the information contained to target cells[10] (Figure 1).
Figure 1.
The biogenesis and release of exosomes. Exosomes form early endosomes in the cell membrane and then transform into multivesicular bodies, which are fused with the cell membrane and released into the extracellular matrix. Exosomes enter target cells in three ways: Direct fusion, endocytosis and protein receptor interaction. ER: Endoplasmic reticulum; ILV: Intraluminal vesicle; MVBs: Multivesicular bodies; MHC: Major histocompatibility complex; PRGL: Proteoglycan; TSG101: Tumor susceptibility gene 101; miRNA: MicroRNA; ALIX: ALG2-interacting protein X; TAA: Target-associated antigen; PD-L1: Programmed cell death-ligand 1; FasL: Fas ligand.
The mechanism regulating the formation of MVBs is mainly driven by ESCRT. ESCRT is a complex composed of ESCRT-0, ESCRT-I, ESCRT-II, ESCRT-III and related proteins[11]. Other studies have found the existence of ESCRT independent mechanisms, mainly including syndecan-syntenin-ALIX protein, Rab protein family, the ceramide pathway, p53 state, intracellular Ca2+ level, high level of heparanase and pH[2]. The exosomal protein ALIX promotes endosomal membrane budding and abscission as well as exosomal cargo selection through interaction with syndecan. Ceramide-rich lipid domains and tetraspanin CD63 outside membrane cells are essential for the formation of ILVs[12].
IMMUNOMODULATORY EFFECTS OF EXOSOMES
Exosomes act as transporters in the process of immune cell-cell communication and participate in the regulation of immunity. The exosomes secreted by APCs are rich in major histocompatibility complex class (MHC)-I/II and costimulatory molecules, which directly present peptide antigens to specific T cells and induce their activation[13]. The regulation of immunity by exosomes mainly depends on tetraspanin-related proteins, such as integrins, immunoglobulin superfamily receptors and growth factor receptors, immune-related noncoding RNAs (ncRNAs), and other immune molecules expressed on exosomes, such as MHC and costimulatory molecules[14]. In summary, exosomes from tumor and immune cells can play related immunomodulatory roles by transferring signals related to immune stimulation or immunosuppression (Table 1).
Table 1.
Exosome-mediated regulation of immunity
|
Origin of exosomes
|
Target cells
|
Functional molecules
|
Mechanisms
|
Ref.
|
| DC | CD4 and CD8+ T cells | CD80 and IL-2 | Induce anti-tumor immune response | [18] |
| Treg | T cells and B cells | IL-35 | Induce peripheral tolerance of T cells and B cells by transferring IL-35 | [21] |
| Treg | Th-1 | Let-7d | Inhibition of Th1 cell proliferation and cytokine release | [22] |
| TAM | CD4+ T cells | miR-29a-3p miR-21-5p | Induce Treg/Th17 imbalance | [25] |
| NK | T cells | miR-186 | Inhibition of tumorigenic potential and TGFβ-dependent immune escape | [31] |
| TDE | CD4 and CD8+ T cells | IL-2 | Inhibition of IL-2-mediated proliferation of CD4 and CD8 T cells | [34] |
| TDE | T cells | FasL and CD3-ζ | Induce T cell apoptosis or inhibit T cell receptor signaling | [35] |
| GC-exosomes | T cells | PI3K proteasome and caspases 3, 8, 9 | Mediate degradation of PI3K proteasome and activation of caspases 3, 8 and 9, and induce apoptosis of Jurkat T cells | [36] |
| GC-exosomes | CD8+ T cells | IL-10 | Inducing PD1 signal can effectively enhance the immunosuppressive activity | [37] |
| GC-exosomes | MSCs | NF-κB signaling pathway | Promote macrophage phagocytosis, up-regulate the secretion of proinflammatory factors and promote the activation of CD69 and CD25 | [38] |
| TDE | DC | miR-212-3p | Inhibit MHC II transcription factor RFXAP in dendritic cells | [39] |
DC: Dendritic cell; IL: Interleukin; Treg: Regulatory T cell; Th1: Type 1 T helper; TAM: Tumor-associated macrophage; NK: Natural killer; TGF: Transforming growth factor; TDE: Tumor-derived exosome; GC: Gastric cancer; PD1: Programmed cell death 1; MSC: Mesenchymal stem cell; NF-κB: Nuclear factor kappa-beta; MHC: Major histocompatibility complex; FasL: Fas ligand.
DC-derived exosomes
DC is the primary antigen-presenting cell in the immune system and plays a role in initiating and maintaining T-cell-mediated responses. DC-derived exosomes contain intercellular adhesion molecule 1 (ICAM-1), which can interact with lymphocyte function-associated antigen-1 (LFA-1), bind to lymphocytes, and act as a ligand for ICAM-1 expressed on CD8+ DCs, thereby activating T cells and promoting T lymphocyte proliferation[15]. Due to the mature state of DCs, DCs acquiesce immunostimulatory and inhibitory properties, which depend on the expression levels of costimulatory molecules (CD80 and CD86), MHC and coregulatory molecules [such as programmed cell death-ligand 1 (PD-L1) and PD-L2][16].
Because both mature DCs and immature DCs secrete exosomes, DC-derived exosomes are expected to have two phenotypes. It is known that immature DCs strongly release exosomes, and the number of exosomes gradually decreases with the maturation process. However, the exosomes released by mature DCs seem to have stronger antigen presentation ability to T cells than those released by immature DCs[17]. DCs play an important role in tumor immunity. DC-derived exosomes activate CD4+ and CD8+ T cells and induce an antitumor immune response through endogenous interleukin (IL)-2 and exosomal CD80 in vivo[18].
T lymphocyte derived exosomes
T cells are mainly divided into two types according to phenotype, including CD4+ helper T cells and CD8+ cytotoxic T lymphocytes (CTLs)[19]. Exosomes isolated from CD4+ helper T cells express T-cell markers [CD4, TCR, LFA-1, CD25 and Fas ligand (FasL)] and exosome-related proteins, which are involved in the CTL reaction and antitumor immune response[20].
Regulatory T cells (Tregs)-derived exosomes are thought to contribute to immunosuppression mediated by CD25, CD73 and CTLA-4. On the other hand, the presence of CD39 and CD73 contributes to the inhibition of Tregs through the production of adenosine, an anti-inflammatory mediator[3]. Exosomal IL-35 targets T cells and B cells to induce infectious tolerance[21]. Treg-derived exosome-mediated Let-7d secretion inhibits type 1 T helper (Th1) cell proliferation and cytokine release by inhibiting the production of interferon-γ mediated by cyclooxygenase-2[22].
Macrophage-derived exosomes
Macrophages have dual activities, M1 cells have proinflammatory effects and play an important role in antitumor immunity, and they are tumor-suppressing cells. M2 macrophages have anti-inflammatory and immunosuppressive activities and are tumor-promoting cells. Tumor-associated macrophage (TAM)-derived exosomes usually exhibit an M2-like phenotype. They lack cytotoxic activity, provide growth factors for cancer cells, and have immunosuppressive activity[23]. In the case of Mycobacterium tuberculosis infection, macrophage-derived exosomes both initiate a protective immune response and promote prior BCG immunization[24]. Exosomes released by TAMs can induce an imbalance in Treg/Th17 cells by transferring miR-29a-3p and miR-21-5p to CD4+ T cells, thus directly creating an immunosuppressive microenvironment and promoting cancer progression[25]. Exosomes can also be used as transmitters to deliver antigens to immune cells and enhance the immune response. Exosomes released by macrophages act as transmitters to deliver antigens to DCs in a ceramide-dependent manner, thereby enhancing the CD4+ T-cell immune response[26].
Mast cell-derived exosomes
Mast cell-derived exosomes contain MHC class II, LFA-1, CD86 and ICAM-1. Mast cells can display mitosis activity on B and T lymphocytes in vitro and in vivo by secreting exosomes, and regulate the proliferation of B and T cells[27]. BMC-exosomes partially promoted CD4+ T cells proliferation, and BMC-exosomes promoted the proliferation and differentiation of Th2 cells between exosomes and T cells via ligation of OX40L and OX40[28]. Exosomes mediate the information exchange between mast cells and nerves, and exosomes derived from mast cells can regulate neuroimmunity at the local site of acupuncture[29].
NK cell-derived exosomes
Studies have found that NK-derived exosomes express both NK cell markers (such as NKG2D, CD94, perforin, granzyme, and CD40L) and exosome-specific markers (such as TSG101, CD81, CD63, and CD9), all of which are involved in cytotoxicity and immune responses[30]. Exosomes derived from NK cells contain the tumor suppressor miR-186, which can effectively inhibit tumorigenic potential and transforming growth factor (TGF)β-dependent immune escape mechanisms[31].
Tumor-derived exosomes
Tumor-derived exosomes (TDEs) play a critical role in tumor growth by influencing different immunomodulatory mechanisms such as antigen expression, immune activation, immunosuppression, immune surveillance, and intercellular communication. TDEs can activate the immune response. Exosomes from tumor cells present new antigens and/or MHC- peptide complexes to activate NK cells or macrophages directly, or activate T cells via DCs[32]. TDEs also showed strong immunosuppressive responses. Exosomes polarize TAMs, induce tumor-associated neutrophils, regulate T-cell differentiation and function, inhibit DC maturation, suppress NK cell activity and induce myeloid suppressor cells (MDSCs), thereby exerting immunosuppressive effects[33].
T cells are potential targets for TDEs to exert immunosuppression. Human TDEs can inhibit the IL-2-mediated proliferation of CD4 and CD8 T cells, and exosomes directly inhibit the killing function of NK cells in a manner independent of T cells[34]. TDEs can selectively induce T-cell apoptosis through FasL or inhibit T-cell receptor signalling by decreasing the expression of CD3-ζ[35]. Exosomes derived from GC can induce apoptosis of Jurkat T cells in a time- and dose-dependent manner by mediating the degradation of the PI3K proteasome and the activation of caspases 3, 8 and 9[36].
Exosomes derived from cancer cells contain PD-L1, which can inhibit T-cell function and promote tumor growth. GC-derived exosomes effectively induce the production of PD-L1+ TAMs and impair CD8+ T-cell function via IL-10, and this immunosuppressive activity can be effectively enhanced by inducing programmed cell death 1 (PD-1) signalling[37].
Mesenchymal stem cells (MSCs) treated with exosomes derived from GC cells promote the phagocytosis of macrophages through the nuclear factor kappa-beta (NF-κB) signalling pathway, promote the secretion of proinflammatory factors, and promote the activation of CD69 and CD25 on the surface of T cells, thus enhancing the ability of MSCs to activate immune cells and maintain the inflammatory environment[38].
MDSCs are immature suppressor cells that have the ability to promote tumor progression. Exosomes released by cancer cells can regulate the activation and expansion of MDSCs and enhance their immunosuppressive function[35]. Exosomes may also regulate immunity by influencing gene expression and signalling pathways in recipient cells, mainly through miRNA transfer. Exosomes secreted by cancer cells transfer miR-212-3p to DCs, which inhibits the expression of the MHC II transcription factor RFXAP in DCs, leading to decreased expression of MHC II and promoting the immune escape of cancer cells[39]. Exosomal miR-451 is not only an indicator of poor postoperative prognosis in GC patients, but is also associated with increased Th17 distribution in GC. The redistribution of miR-451 from cancer cells into infiltrating T cells during hypoglycaemic therapy can enhance Th17 differentiation by enhancing mechanistic target of rapamycin (mTOR) activity[40].
Therefore, exosomes play both immune-activating and immunosuppressive roles in cancer. The effect of immune activation mainly depends on the antigen presentation of exosomes, while the immunosuppressive effect of exosomes mainly depends on the ligands, proteins and miRNAs they carry, which inhibit the activity of cytotoxic T cells or increase the number of immunosuppressive cells[41].
EXOSOMES AND GC DEVELOPMENT
Exosomes can influence recipient cells through autocrine and paracrine signalling. First, exosomal proteins can influence the cells that release exosomes through the autocrine pathway. Second, exosomal DNA itself affects cell survival. Exosomes can also regulate the tumor microenvironment through paracrine mechanisms. Exosomes derived from host cancer cells can activate receptors or transfer proteins and RNAs to recipient cells, thereby affecting the tumor microenvironment or altering the biological phenotype of recipient cells[41].
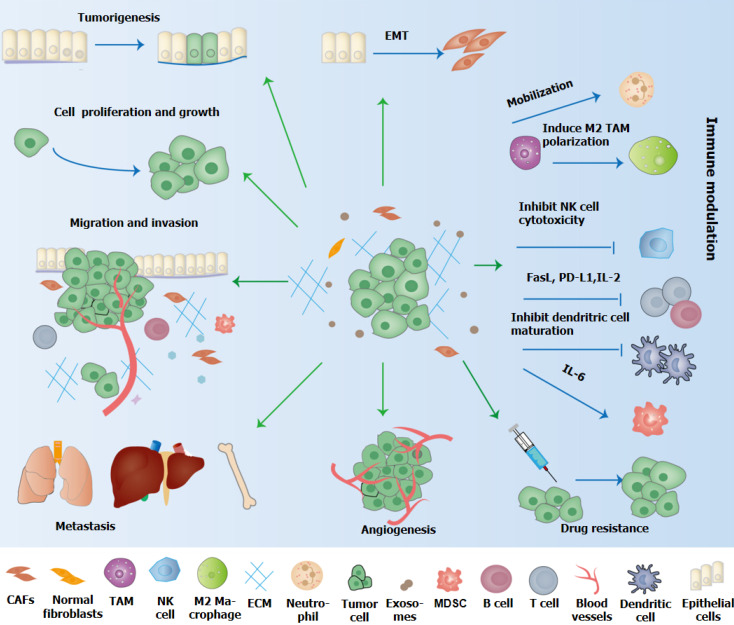
Tumour-derived microvesicles can be transmitted as messengers between GC cells and influence several important processes of malignant progression of GC, including angiogenesis, tumor migration, the establishment of pre-metastatic niche (PMN) and epithelial-mesenchymal transformation[42,43] (Table 2 and Figure 2). GC cells can establish the PMN through various mechanisms, including immunosuppression, matrix remodeling, angiogenesis, mesenchymal transformation and organotropy[33].
Table 2.
Role of exosomes cargo in initiation and progression of gastric cancer
|
Type
|
Contents
|
Mechanisms
|
Function
|
Ref.
|
| Protein | YB-1 | Up-regulation of angiogenic factors in endothelial cells | Promote gastric cancer angiogenesis | [45] |
| miRNA | miR-519a-3p | Causes M2-like polarization of macrophages | Induce angiogenesis and promote pre-metastatic niche formation | [46] |
| Protein | X26nt | Decrease vascular endothelial cadherin | Promote angiogenesis | [47] |
| circRNA | circ29 | Regulates the VEGF pathway in endothelial cells | Promote angiogenesis | [48] |
| Protein | GKN1 | Tumor suppressor protein | Inhibit cell proliferation and induce cell apoptosis | [51] |
| Protein | TRIM3 | TRIM3 knockdown regulates stem cell factors and EMT regulators | Promote the growth and metastasis of gastric cancer | [52] |
| Protein | GC-exosome | Activation of NF-κB activates macrophages in human peripheral blood mononuclear cells | Promote tumor cell proliferation and migration | [53] |
| Protein | GC-exosome | Induce pericytes to transform into CAFs | Induce malignant progression of cancer | [58] |
| Protein | CD97 | Activate the MAPK signaling pathway | Promote cell proliferation and invasion | [54] |
MiRNA: MicroRNA; circRNA: Circular RNA; VEGF: Vascular endothelial growth factor; GC: Gastric cancer; NF-κB: Nuclear factor kappa-beta; EMT: Epithelial-to-mesenchymal transition; CAFs: Cancer-associated fibroblast.
Figure 2.
The role of exosome in the initiation and development of gastric cancer. Exosomes affect tumorigenesis, epithelial-mesenchymal transformation, proliferation, invasion, angiogenesis, metastasis, immune escape and drug resistance of gastric cancer. EMT: Epithelial-to-mesenchymal transition; TAM: Tumor-associated macrophage; NK: Natural killer; FasL: Fas ligand; PD-L1: Programmed cell death-ligand 1; IL: Interleukin; CAFs: Cancer-associated fibroblasts; MDSC: Myeloid suppressor cell.
Exosomes and angiogenesis
Angiogenesis is closely related to the occurrence, invasion and metastasis of tumors by providing oxygen and nutrients, and plays an important role in the progression of GC. Exosomes can regulate the characteristics of endothelial cells to promote angiogenesis. Exosomes derived from GC cells promote tumor angiogenesis by activating endothelial cells[44]. YB-1 plays a key role in exosomes promoting GC angiogenesis by upregulating angiogenic factors in endothelial cells treated with exosomes[45]. GC-derived exosomal miR-519a-3p activates the MAPK/ERK pathway by targeting DUSP2, thereby causing M2-like polarization of macrophages, inducing angiogenesis and promoting the formation of an intrahepatic premetastatic niche to accelerate liver metastasis of GC[46].
It was found that X26nt expression was significantly increased in GC and GC-derived exosomes. Exosomal X26nt reduces vascular endothelial cadherin expression by directly binding to VE-cadherin mRNA in human umbilical vein endothelial cells, thereby increasing vascular permeability. In vivo experiments have shown that X26nt can promote angiogenesis in a mouse subcutaneous tumor model[47]. GC-derived exosomes mediate circular RNAs (circRNAs) delivery, and circ29, as a sponge of miR-29a, promotes angiogenesis by regulating the vascular endothelial growth factor (VEGF) pathway in endothelial cells[48].
Studies have shown that exosomes derived from MSCs can promote or inhibit tumor angiogenesis through the Akt (protein kinase B)/eNOS pathway, the ERK1/2 pathway or miRNA transport[49]. Other scholars have found that exosomes loaded with hepatocyte growth factor (HGF) small interfering RNA can inhibit the growth and angiogenesis of GC cells and the growth rate of blood vessels[50].
Exosomes and GC growth
Cancer cell-derived exosomes and GC growth: The absence of some exosomal molecules may increase cell viability and promote proliferation. Gastrin-1 is a tumor suppressor protein. The deletion of gastrokine 1 in exosomes can promote the proliferation of GC cell lines[51]. TRIM3 knockdown in serum exosomes of GC patients promotes the growth and metastasis of GC in vitro and in vivo by regulating stem cell factors and epithelial-to-mesenchymal transition regulatory factors[52]. GC-derived exosomes significantly increase the phosphorylation level of NF-κB in macrophages, and activate macrophages in human peripheral blood mononuclear cells by activating NF-κB to increase the level of proinflammatory factors, thus promoting the proliferation and migration of tumor cells[53].
The extracellular signal-regulated kinase (MAPK/ERK) signalling pathway is mainly involved in the development and progression of tumors and is regulated by exosomes. Tumor cell-derived exosomes partially activated the MAPK/ERK and PI3K/Akt pathways in SGC7901 and BGC823 GC cells. Exosomes containing CD97 activate the MAPK signalling pathway in SGC-7901 GC cells, which can promote cell proliferation and invasion. Exosomal miRNAs may be involved in the activation of CD97-related pathways[54]. ZFAS1 is a newly identified long ncRNA (lncRNA) that exists in exosomes and can be transmitted through them. Overexpression of ZFAS1 promotes the proliferation and migration of GC cells[55].
MiRNAs are the most abundant and important biomolecules in exosomes, which play a key role in tumor regulation and may be related to tumor development, metastasis and prognosis. In the early stage of tumorigenesis, the downregulation of antitumor miRNA in cancer cells and the insufficient supplementation of exosomal miR-101 in the microenvironment of residential cells will stimulate the development of GC[56]. The level of exosomal miR-423-5p in the serum of GC patients and in the supernatant of GC cell culture was significantly increased, and miR-423-5p inhibited the expression of fusion protein inhibitory factor, promoting the proliferation and migration of GC cells[57]. GC cells induce pericytes to transform into cancer-associated fibroblasts (CAFs) through exosome-mediated BMP transfer and activation of the PI3K/AKT and MEK/ERK pathways, thereby inducing malignant progression of cancer[58]. There are abundant let-7 miRNAs in both the intracellular and extracellular environments of the metastatic GC cell line AZ-P7a, and AZ-P7a cells release let-7 miRNAs into extracellular environments through exosomes to support gastric carcinogenesis[59].
Microenvironment-derived exosomes and GC growth: The tumor microenvironment includes MSCs, endothelial cells, tumor-associated fibroblasts, TAMs, pericytes, extracellular matrix, signalling molecules, etc. Tumor microenvironment-derived exosomes are also important for the progression of GC. Exosomes derived from bone marrow MSCs promote the growth of GC SGC7901 cells by activating Hedgehog signaling pathway[60]. Exosomal miRNA-34 derived from CAFs inhibit the proliferation and invasion of GC cells in vitro and inhibit tumor growth in vivo[61]. M1 macrophage-derived exosomes containing miR-16-5p can trigger T-cell immune responses and inhibit tumor formation in vivo and in vitro by reducing the expression of PD-L1[62]. Macrophage-derived exosomes can serve as carriers to deliver exogenous miR-21 inhibitors to BGC-823 GC cells, promoting cell migration and inhibiting cell apoptosis[63].
Exosomes and GC invasion and metastasis
Cancer cell-derived exosomes and GC invasion and metastasis: During tumor cell metastasis, the cells adhere to the stroma and migrate into the blood, reaching the premetastatic niche and then causing secondary tumors. Studies have shown that bioactive substances carrying exosomes, such as proteins, miRNAs, or lncRNAs, may be functional signals to induce tumor growth and metastasis in GC cells.
GC-derived exosomes induce increased neutrophil-autophagy through the HMGB1/TLR4/NF-κB signalling pathway, thus promoting the migration of GC cells[64]. Exosomal circSHKBP1 sponged miR-582-3p increased the expression of HUR, enhanced the stability of VEGF mRNA and inhibited the degradation of heat shock protein 90, thus promoting the progression of GC[65]. The expression of exosomal TGF-β1 and the proportion of Treg cells in the draining lymph nodes were significantly correlated with the pathological stage and lymph node metastasis of GC[66]. Exosomes are specific to the recipient cell type and are prone to organotropic metastasis. Organotropism is caused by primary GC cells actively selecting PMNs in a specific remote microenvironment. Exosomal epidermal growth factor receptor is transmitted from GC cells to Kupffer cells and hepatic stellate cells to effectively activate HGF by inhibiting the expression of miR-26a/b. In addition, HGF binds to c-MET receptors on cancer cells and copromotes liver-specific metastasis[67].
In addition to lymph node metastasis, peritoneal metastasis is also a main type of metastasis in advanced GC patients. Cancer-derived exosomes induce increased expression of adhesion molecules in mesothelial cells, which is a necessary condition for peritoneal metastasis of GC. GC-derived exosomes are internalized into mesothelial cells, which can significantly promote the adhesion between mesothelial cells and GC cells. After internalization into GC cells and malignant pleural effusion, the expression of adhesion-related molecules such as fibronectin 1 and laminin gamma 1 in mesothelial cells increased, which further promotes the migration of GC cells[68].
The intact mesothelium is the protective barrier of the peritoneum. GC-derived exosomes promote peritoneal metastasis through peritoneal fibrosis and mesodermal barrier breakdown. Upregulation of p-ERK in peritoneal mesothelial cells leads to mesothelial-to-mesothelial transition, which in turn leads to GC-derived exosomes damaging peritoneal mesothelial cells and causing peritoneal metastasis of GC[69]. This finding supports that exosomes play a key role in mediating peritoneal metastasis.
Microenvironment-derived exosomes and GC invasion and metastasis: CAFs partially promote the migration of GC cells through matrix metallopeptidase (MMP)11, and the CAFs-derived exosomal miR-139 can inhibit the progression of GC by decreasing the expression of MMP11[70]. MSC-exosomes activate the AKT signalling pathway, which then enhances the migration and invasion of GC cells by inducing epithelial-mesenchymal transformation, increases the expression of mesenchymal markers of GC cells, and enhances the tumorigenicity of GC cells in vitro[71]. M2 macrophage-derived exosomes remodel the cytoskeleton to support gastric cell migration[72]. The lncRNA PCGEM1 is specifically expressed in exosomes derived from hypoxia-cultured GC cells (HGCs). PCGEM1-rich exosomes derived from HGCs can promote the invasion and migration of GC cells in normoxic culture[73]. These studies all suggest that exosomes from the tumor microenvironment are also involved in GC metastasis.
THE APPLICATION OF EXOSOMES IN GC
Applications as biomarkers in GC
The existence and content of exosomes, as well as their detectable characteristics in body fluids, make the diagnosis based on exosomes considered to be the best method for noninvasive diagnosis, and therefore exosomes can be used as biomarkers for disease diagnosis and treatment (Table 3).
Table 3.
Application of exosomes as biomarkers in gastric cancer
|
TDE-molecule
|
TDEs source
|
Application
|
Ref.
|
| miR10b-5p | Serum | Diagnosis and prognosis | [74] |
| miR296-5p | |||
| LINC00152 | Plasma | Diagnosis | [76] |
| TRIM3 | Serum | Diagnosis | [52] |
| lncUEGC1 | Serum | Early diagnosis of gastric cancer | [77] |
| lncUEGC2 | |||
| TGF-β1 | Serum | Pathological stage and lymph node metastasis | [66] |
| miR-21 | Malignant ascites, peritoneal lavage fluid and culture supernatants | Serosal invasion | [78] |
| miR-1225-5p |
TDE: Tumor-derived exosome.
Serum exosomal miRNAs have been identified as potential biomarkers for GC. Studies have found that the expression levels of miR195-5p, miR10b-5p, miR296-5p and miR20a-3p in the exosomes of GC serum samples are significantly increased, and patients with high expression of miR10b-5p or miR296-5p have worse prognosis[74]. These studies suggest that exosomal miRNAs can serve not only as novel noninvasive diagnostic markers, but also as potential GC prognostic biomarkers.
ncRNAs are some of the most abundant RNAs in exosomes. Exosomes contain some ncRNAs with biological functions, and the membrane structure of exosomes significantly improves the stability of exosomal ncRNAs. These characteristics make exosomal ncRNAs suitable as biomarkers for diagnosis and prognosis[75]. The plasma LINC00152 level in patients with GC is significantly higher than that in healthy controls, and one of the possible mechanisms for its stable existence in the blood is the protection of exosomes. Studies have shown that the specificity and sensitivity of plasma LINC00152 in the diagnosis of GC are 85.2% and 48.1%[76].
Exosomal TRIM3 can be used as a biomarker for the diagnosis of GC, and exosome transport of TRIM3 may provide a potential treatment for GC[52]. Studies on biomarkers of early GC have found that two exosomal lncRNAs specific to early GC, lncUEGC1 and lncUEGC2, are significantly upregulated in patients with early GC and in exosomes derived from GC cells. lncUEGC1 is expected to be a stable, highly sensitive and noninvasive biomarker for the diagnosis of early GC[77].
Lymph node and peritoneal metastasis are associated with a poor prognosis in GC patients. Some scholars have found that the expression of exosomal TGF-β1 in GC patients is related to pathological stage and lymph node metastasis[66]. Researchers identified five highly expressed miRNAs (miR-320c, miR-1202, miR-1225-5p, miR-4270, and miR-1207-5p) in malignant ascites, peritoneal lavage fluid and culture supernatants. Among them, miR-21 and miR-1225-5p are related to serosal invasion of GC, and may be used as biomarkers of peritoneal recurrence after GC surgery[78].
Exosomes and cancer therapy
The membrane of exosomes protects their contents from degradation and is very stable. In contrast to nonhost vectors, exosomes are relatively nonimmunogenic and do not induce immune rejection. Exosomes easily cross biological barriers, especially blood-brain barriers. Organophilic factors, such as integrins on the surface of exosomes, can deliver drugs to specific tissues, thus improving the targeting specificity[12,79,80]. Because of these characteristics of exosomes, they are considered as ideal delivery carriers.
As a new drug delivery method, exosomes are used to deliver biomolecules and chemotherapy drugs in cancer therapy. MSCs can incorporate and deliver paclitaxel to recipient cells through exosomes, which have strong antitumor effects[81]. Exosomes act as nanoparticles to deliver anti-miR-214 and reverse the resistance of GC to cisplatin, which may become a potential alternative for cisplatin refractory GC treatment in the future[82]. Exosomes derived from macrophages can be used as carriers to deliver exogenous miR-21 inhibitors to GC cells, which can promote cell migration and inhibit cell apoptosis. Compared with traditional transfection methods, exosome-mediated miR-21 inhibitor delivery has a greater inhibitory effect and less cytotoxicity, demonstrating the potential of miR-21 and exosomes as therapeutic carriers for GC[63].
Exosome vaccines
Because exosomes can deliver tumor-derived antigens that activate CTLs, they are expected to be used for cancer immunotherapy. Tumor cell-derived exosomes may represent a new type of cancer vaccine. Exosomes from heat-treated malignant ascites can promote the maturation of DCs and induce tumorigenic CTL response, suggesting that heat stress can improve the immunogenicity of exosomes from malignant ascites in GC patients[83]. However, TDEs carry many oncogenes, which can induce tumor progression, so the safety of TDEs vaccine is still uncertain. DC vaccines can be quickly eliminated by antigen-specific CTLs. Since DC-derived exosomes express MHC-I and MHC-II molecules, which can promote the T-cell immune response and tumor rejection, DC-derived exosome vaccines have a longer lifespan than DC vaccines, so they are considered as a substitute for DC vaccines[41].
Cancer immunotherapy based on exosomes
Exosomes have potential applications in cancer immunotherapy. The purpose of tumor immunotherapy is to establish durable and effective anticancer immunity. Tumor-specific CTLs play important roles in this process[84]. PD-L1 inhibits T-cell activation by binding to its receptor PD-1, thereby maintaining immune homeostasis. TDEs carry PD-L1 and thus resist immune checkpoint therapy. Exosomal PD-L1 may have the potential to become a target to overcome therapeutic resistance to anti-PD-1/PD-L1 antibodies[85]. Some studies have proposed that exosomes derived from CAR-T cells can replace CAR-T cells in anticancer immunity[86].
EXOSOMES AND DRUG RESISTANCE IN GC
Drug resistance of GC to conventional treatment is one of the factors causing its poor prognosis. In recent years, the following findings have illustrated the mechanisms of exosomes in the chemotherapy resistance of patients with GC[87]: (1) Exosomes can excrete cytotoxic drugs to mediate chemotherapy resistance; (2) Exosomes can deliver anti-apoptotic agents in cancer cells to interfere with drug metabolism, thereby promoting chemotherapy resistance; (3) Stromal exosomes induce drug resistance in cancer cells; and (4) Drug-sensitive cells become drug-resistant after treatment with exosomes from drug-resistant cells. The natural structure of exosomes makes them good carriers for chemotherapy drugs and good candidates for restoring therapeutic activity. Exosomes loaded with miRNA, mRNA, and other ncRNAs can mediate resistance. In recent years, the targeted delivery of miRNAs in vitro to alter the metabolism of receptor cells has become a rapidly developing new method.
Cancer cell-derived exosomes and therapeutic resistance
Cisplatin is one of the most effective and commonly used basic chemotherapy drugs in the treatment of advanced GC. Exosomes from cisplatin-resistant GC cells deliver miR-500a-3p by targeting FBXW7 in vitro and in vivo, thus enhancing DDP resistance and stemness of GC cells[88]. However, some studies have found that exogenous anti-214 can reverse the resistance of GC cells to cisplatin and inhibit tumor growth[82]. miR-107 secreted by exosomes significantly enhances the sensitivity of drug-resistant GC cells to chemotherapy drugs through the HMGA2/mTOR/P-gp pathway[89].
Microenvironment-derived exosomes and therapeutic resistance
CAFs promote tumor progression and drug resistance by secreting various bioactive substances, including exosomes. MSCs have also been linked to drug resistance in GC. MSC-exosomes suppress 5-fluorouracil-induced apoptosis in GC cells, and enhance the expression of multidrug resistance-related proteins such as multidrug resistance-related protein, multiple drug resistance and lung resistance protein, thus inducing resistance of GC cells to 5-fluorouracil[90]. TAMs are the most abundant immune cells in the tumor microenvironment. Exosomal miR-21 derived from TAMs can be transferred directly to GC cells, inhibit apoptosis by downregulating PTEN, enhance the activation of PI3K/AKT signalling pathway, and promote cisplatin resistance in GC cells[91].
CONCLUSION
Exosomes not only participate in the regulation of immunity, but also participate in various tumor processes, including growth, invasion, metastasis, angiogenesis and therapeutic resistance, and have value in disease diagnosis and prognosis evaluation. Due to the related biological properties, exosome-based GC therapy shows great promise. Using exosomes as carriers to deliver tumor suppressor molecules and drugs alone or in combination with traditional therapies, or blocking the release of GC cell-derived exosomes, all of these methods provide new strategies for the treatment of GC. Although exosomes are a relatively new area of research, in the field of cancer therapy, there has been widespread interest in their potential applications as cancer markers, effective and safe anti-cancer drug delivery methods or as new inhibitors in immunotherapy.
Footnotes
Conflict-of-interest statement: All the authors report no relevant conflicts of interest for this article.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Peer-review started: October 14, 2023
First decision: December 15, 2023
Article in press: January 15, 2024
Specialty type: Oncology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): 0
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Zhang Q, China S-Editor: Wang JJ L-Editor: A P-Editor: Xu ZH
Contributor Information
Xiao-Li Guan, Department of General Medicine, The Second Hospital of Lanzhou University, Lanzhou 730030, Gansu Province, China.
Xiao-Ying Guan, Department of Pathology, The Second Hospital of Lanzhou University, Lanzhou 730030, Gansu Province, China.
Zheng-Yi Zhang, Department of General Medicine, The Second Hospital of Lanzhou University, Lanzhou 730030, Gansu Province, China. zhangzhengyi11@lzu.edu.cn.
References
- 1.Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209–249. doi: 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- 2.Cordonnier M, Chanteloup G, Isambert N, Seigneuric R, Fumoleau P, Garrido C, Gobbo J. Exosomes in cancer theranostic: Diamonds in the rough. Cell Adh Migr. 2017;11:151–163. doi: 10.1080/19336918.2016.1250999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Baghaei K, Tokhanbigli S, Asadzadeh H, Nmaki S, Reza Zali M, Hashemi SM. Exosomes as a novel cell-free therapeutic approach in gastrointestinal diseases. J Cell Physiol. 2019;234:9910–9926. doi: 10.1002/jcp.27934. [DOI] [PubMed] [Google Scholar]
- 4.Zhang B, Yin Y, Lai RC, Lim SK. Immunotherapeutic potential of extracellular vesicles. Front Immunol. 2014;5:518. doi: 10.3389/fimmu.2014.00518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.EL Andaloussi S, Mäger I, Breakefield XO, Wood MJ. Extracellular vesicles: biology and emerging therapeutic opportunities. Nat Rev Drug Discov. 2013;12:347–357. doi: 10.1038/nrd3978. [DOI] [PubMed] [Google Scholar]
- 6.Xie QH, Zheng JQ, Ding JY, Wu YF, Liu L, Yu ZL, Chen G. Exosome-Mediated Immunosuppression in Tumor Microenvironments. Cells. 2022;11 doi: 10.3390/cells11121946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tran TH, Mattheolabakis G, Aldawsari H, Amiji M. Exosomes as nanocarriers for immunotherapy of cancer and inflammatory diseases. Clin Immunol. 2015;160:46–58. doi: 10.1016/j.clim.2015.03.021. [DOI] [PubMed] [Google Scholar]
- 8.Yu S, Cao H, Shen B, Feng J. Tumor-derived exosomes in cancer progression and treatment failure. Oncotarget. 2015;6:37151–37168. doi: 10.18632/oncotarget.6022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yáñez-Mó M, Siljander PR, Andreu Z, Zavec AB, Borràs FE, Buzas EI, Buzas K, Casal E, Cappello F, Carvalho J, Colás E, Cordeiro-da Silva A, Fais S, Falcon-Perez JM, Ghobrial IM, Giebel B, Gimona M, Graner M, Gursel I, Gursel M, Heegaard NH, Hendrix A, Kierulf P, Kokubun K, Kosanovic M, Kralj-Iglic V, Krämer-Albers EM, Laitinen S, Lässer C, Lener T, Ligeti E, Linē A, Lipps G, Llorente A, Lötvall J, Manček-Keber M, Marcilla A, Mittelbrunn M, Nazarenko I, Nolte-'t Hoen EN, Nyman TA, O'Driscoll L, Olivan M, Oliveira C, Pállinger É, Del Portillo HA, Reventós J, Rigau M, Rohde E, Sammar M, Sánchez-Madrid F, Santarém N, Schallmoser K, Ostenfeld MS, Stoorvogel W, Stukelj R, Van der Grein SG, Vasconcelos MH, Wauben MH, De Wever O. Biological properties of extracellular vesicles and their physiological functions. J Extracell Vesicles. 2015;4:27066. doi: 10.3402/jev.v4.27066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mittelbrunn M, Sánchez-Madrid F. Intercellular communication: diverse structures for exchange of genetic information. Nat Rev Mol Cell Biol. 2012;13:328–335. doi: 10.1038/nrm3335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Villarroya-Beltri C, Baixauli F, Gutiérrez-Vázquez C, Sánchez-Madrid F, Mittelbrunn M. Sorting it out: regulation of exosome loading. Semin Cancer Biol. 2014;28:3–13. doi: 10.1016/j.semcancer.2014.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zhu L, Sun HT, Wang S, Huang SL, Zheng Y, Wang CQ, Hu BY, Qin W, Zou TT, Fu Y, Shen XT, Zhu WW, Geng Y, Lu L, Jia HL, Qin LX, Dong QZ. Isolation and characterization of exosomes for cancer research. J Hematol Oncol. 2020;13:152. doi: 10.1186/s13045-020-00987-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Raposo G, Nijman HW, Stoorvogel W, Liejendekker R, Harding CV, Melief CJ, Geuze HJ. B lymphocytes secrete antigen-presenting vesicles. J Exp Med. 1996;183:1161–1172. doi: 10.1084/jem.183.3.1161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chairoungdua A, Smith DL, Pochard P, Hull M, Caplan MJ. Exosome release of β-catenin: a novel mechanism that antagonizes Wnt signaling. J Cell Biol. 2010;190:1079–1091. doi: 10.1083/jcb.201002049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Segura E, Guérin C, Hogg N, Amigorena S, Théry C. CD8+ dendritic cells use LFA-1 to capture MHC-peptide complexes from exosomes in vivo. J Immunol. 2007;179:1489–1496. doi: 10.4049/jimmunol.179.3.1489. [DOI] [PubMed] [Google Scholar]
- 16.Thomson AW, Robbins PD. Tolerogenic dendritic cells for autoimmune disease and transplantation. Ann Rheum Dis. 2008;67 Suppl 3:iii90–iii96. doi: 10.1136/ard.2008.099176. [DOI] [PubMed] [Google Scholar]
- 17.Quah BJ, O'Neill HC. Maturation of function in dendritic cells for tolerance and immunity. J Cell Mol Med. 2005;9:643–654. doi: 10.1111/j.1582-4934.2005.tb00494.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tian H, Li W. Dendritic cell-derived exosomes for cancer immunotherapy: hope and challenges. Ann Transl Med. 2017;5:221. doi: 10.21037/atm.2017.02.23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Geltink RIK, Kyle RL, Pearce EL. Unraveling the Complex Interplay Between T Cell Metabolism and Function. Annu Rev Immunol. 2018;36:461–488. doi: 10.1146/annurev-immunol-042617-053019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhang H, Xie Y, Li W, Chibbar R, Xiong S, Xiang J. CD4(+) T cell-released exosomes inhibit CD8(+) cytotoxic T-lymphocyte responses and antitumor immunity. Cell Mol Immunol. 2011;8:23–30. doi: 10.1038/cmi.2010.59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sullivan JA, Tomita Y, Jankowska-Gan E, Lema DA, Arvedson MP, Nair A, Bracamonte-Baran W, Zhou Y, Meyer KK, Zhong W, Sawant DV, Szymczak-Workman AL, Zhang Q, Workman CJ, Hong S, Vignali DAA, Burlingham WJ. Treg-Cell-Derived IL-35-Coated Extracellular Vesicles Promote Infectious Tolerance. Cell Rep. 2020;30:1039–1051.e5. doi: 10.1016/j.celrep.2019.12.081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Okoye IS, Coomes SM, Pelly VS, Czieso S, Papayannopoulos V, Tolmachova T, Seabra MC, Wilson MS. MicroRNA-containing T-regulatory-cell-derived exosomes suppress pathogenic T helper 1 cells. Immunity. 2014;41:89–103. doi: 10.1016/j.immuni.2014.05.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Shapouri-Moghaddam A, Mohammadian S, Vazini H, Taghadosi M, Esmaeili SA, Mardani F, Seifi B, Mohammadi A, Afshari JT, Sahebkar A. Macrophage plasticity, polarization, and function in health and disease. J Cell Physiol. 2018;233:6425–6440. doi: 10.1002/jcp.26429. [DOI] [PubMed] [Google Scholar]
- 24.Cheng Y, Schorey JS. Exosomes carrying mycobacterial antigens can protect mice against Mycobacterium tuberculosis infection. Eur J Immunol. 2013;43:3279–3290. doi: 10.1002/eji.201343727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zhou J, Li X, Wu X, Zhang T, Zhu Q, Wang X, Wang H, Wang K, Lin Y. Exosomes Released from Tumor-Associated Macrophages Transfer miRNAs That Induce a Treg/Th17 Cell Imbalance in Epithelial Ovarian Cancer. Cancer Immunol Res. 2018;6:1578–1592. doi: 10.1158/2326-6066.CIR-17-0479. [DOI] [PubMed] [Google Scholar]
- 26.Xu Y, Liu Y, Yang C, Kang L, Wang M, Hu J, He H, Song W, Tang H. Macrophages transfer antigens to dendritic cells by releasing exosomes containing dead-cell-associated antigens partially through a ceramide-dependent pathway to enhance CD4(+) T-cell responses. Immunology. 2016;149:157–171. doi: 10.1111/imm.12630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Skokos D, Le Panse S, Villa I, Rousselle JC, Peronet R, David B, Namane A, Mécheri S. Mast cell-dependent B and T lymphocyte activation is mediated by the secretion of immunologically active exosomes. J Immunol. 2001;166:868–876. doi: 10.4049/jimmunol.166.2.868. [DOI] [PubMed] [Google Scholar]
- 28.Li F, Wang Y, Lin L, Wang J, Xiao H, Li J, Peng X, Dai H, Li L. Mast Cell-Derived Exosomes Promote Th2 Cell Differentiation via OX40L-OX40 Ligation. J Immunol Res. 2016;2016:3623898. doi: 10.1155/2016/3623898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chen B, Li MY, Guo Y, Zhao X, Lim HC. Mast cell-derived exosomes at the stimulated acupoints activating the neuro-immune regulation. Chin J Integr Med. 2017;23:878–880. doi: 10.1007/s11655-016-2269-8. [DOI] [PubMed] [Google Scholar]
- 30.Xu Z, Zeng S, Gong Z, Yan Y. Exosome-based immunotherapy: a promising approach for cancer treatment. Mol Cancer. 2020;19:160. doi: 10.1186/s12943-020-01278-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Neviani P, Wise PM, Murtadha M, Liu CW, Wu CH, Jong AY, Seeger RC, Fabbri M. Natural Killer-Derived Exosomal miR-186 Inhibits Neuroblastoma Growth and Immune Escape Mechanisms. Cancer Res. 2019;79:1151–1164. doi: 10.1158/0008-5472.CAN-18-0779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhang HG, Zhuang X, Sun D, Liu Y, Xiang X, Grizzle WE. Exosomes and immune surveillance of neoplastic lesions: a review. Biotech Histochem. 2012;87:161–168. doi: 10.3109/10520291003659042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gao J, Li S, Xu Q, Zhang X, Huang M, Dai X, Liu L. Exosomes Promote Pre-Metastatic Niche Formation in Gastric Cancer. Front Oncol. 2021;11:652378. doi: 10.3389/fonc.2021.652378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Clayton A, Mitchell JP, Court J, Mason MD, Tabi Z. Human tumor-derived exosomes selectively impair lymphocyte responses to interleukin-2. Cancer Res. 2007;67:7458–7466. doi: 10.1158/0008-5472.CAN-06-3456. [DOI] [PubMed] [Google Scholar]
- 35.Chen W, Jiang J, Xia W, Huang J. Tumor-Related Exosomes Contribute to Tumor-Promoting Microenvironment: An Immunological Perspective. J Immunol Res. 2017;2017:1073947. doi: 10.1155/2017/1073947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Qu JL, Qu XJ, Qu JL, Zhao MF, Teng YE, Zhang Y, Hou KZ, Jiang YH, Yang XH, Liu YP. The role of cbl family of ubiquitin ligases in gastric cancer exosome-induced apoptosis of Jurkat T cells. Acta Oncol. 2009;48:1173–1180. doi: 10.3109/02841860903032817. [DOI] [PubMed] [Google Scholar]
- 37.Wang F, Li B, Wei Y, Zhao Y, Wang L, Zhang P, Yang J, He W, Chen H, Jiao Z, Li Y. Tumor-derived exosomes induce PD1(+) macrophage population in human gastric cancer that promotes disease progression. Oncogenesis. 2018;7:41. doi: 10.1038/s41389-018-0049-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Shen Y, Xue C, Li X, Ba L, Gu J, Sun Z, Han Q, Zhao RC. Effects of Gastric Cancer Cell-Derived Exosomes on the Immune Regulation of Mesenchymal Stem Cells by the NF-kB Signaling Pathway. Stem Cells Dev. 2019;28:464–476. doi: 10.1089/scd.2018.0125. [DOI] [PubMed] [Google Scholar]
- 39.Ding G, Zhou L, Qian Y, Fu M, Chen J, Xiang J, Wu Z, Jiang G, Cao L. Pancreatic cancer-derived exosomes transfer miRNAs to dendritic cells and inhibit RFXAP expression via miR-212-3p. Oncotarget. 2015;6:29877–29888. doi: 10.18632/oncotarget.4924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Liu F, Bu Z, Zhao F, Xiao D. Increased T-helper 17 cell differentiation mediated by exosome-mediated microRNA-451 redistribution in gastric cancer infiltrated T cells. Cancer Sci. 2018;109:65–73. doi: 10.1111/cas.13429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zhang L, Yu D. Exosomes in cancer development, metastasis, and immunity. Biochim Biophys Acta Rev Cancer. 2019;1871:455–468. doi: 10.1016/j.bbcan.2019.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Stec M, Szatanek R, Baj-Krzyworzeka M, Baran J, Zembala M, Barbasz J, Waligórska A, Dobrucki JW, Mytar B, Szczepanik A, Siedlar M, Drabik G, Urbanowicz B. Interactions of tumour-derived micro(nano)vesicles with human gastric cancer cells. J Transl Med. 2015;13:376. doi: 10.1186/s12967-015-0737-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wee I, Syn N, Sethi G, Goh BC, Wang L. Role of tumor-derived exosomes in cancer metastasis. Biochim Biophys Acta Rev Cancer. 2019;1871:12–19. doi: 10.1016/j.bbcan.2018.10.004. [DOI] [PubMed] [Google Scholar]
- 44.Fu M, Gu J, Jiang P, Qian H, Xu W, Zhang X. Exosomes in gastric cancer: roles, mechanisms, and applications. Mol Cancer. 2019;18:41. doi: 10.1186/s12943-019-1001-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Xue X, Huang J, Yu K, Chen X, He Y, Qi D, Wu Y. YB-1 transferred by gastric cancer exosomes promotes angiogenesis via enhancing the expression of angiogenic factors in vascular endothelial cells. BMC Cancer. 2020;20:996. doi: 10.1186/s12885-020-07509-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Qiu S, Xie L, Lu C, Gu C, Xia Y, Lv J, Xuan Z, Fang L, Yang J, Zhang L, Li Z, Wang W, Xu H, Li B, Xu Z. Gastric cancer-derived exosomal miR-519a-3p promotes liver metastasis by inducing intrahepatic M2-like macrophage-mediated angiogenesis. J Exp Clin Cancer Res. 2022;41:296. doi: 10.1186/s13046-022-02499-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Chen X, Zhang S, Du K, Zheng N, Liu Y, Chen H, Xie G, Ma Y, Zhou Y, Zheng Y, Zeng L, Yang J, Shen L. Gastric cancer-secreted exosomal X26nt increases angiogenesis and vascular permeability by targeting VE-cadherin. Cancer Sci. 2021;112:1839–1852. doi: 10.1111/cas.14740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Li S, Li J, Zhang H, Zhang Y, Wang X, Yang H, Zhou Z, Hao X, Ying G, Ba Y. Gastric cancer derived exosomes mediate the delivery of circRNA to promote angiogenesis by targeting miR-29a/VEGF axis in endothelial cells. Biochem Biophys Res Commun. 2021;560:37–44. doi: 10.1016/j.bbrc.2021.04.099. [DOI] [PubMed] [Google Scholar]
- 49.Akad F, Mocanu V, Peiu SN, Scripcariu V, Filip B, Timofte D, Zugun-Eloae F, Cuciureanu M, Hancianu M, Oboroceanu T, Condur L, Popa RF. Mesenchymal Stem Cell-Derived Exosomes Modulate Angiogenesis in Gastric Cancer. Biomedicines. 2023;11 doi: 10.3390/biomedicines11041031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhang H, Wang Y, Bai M, Wang J, Zhu K, Liu R, Ge S, Li J, Ning T, Deng T, Fan Q, Li H, Sun W, Ying G, Ba Y. Exosomes serve as nanoparticles to suppress tumor growth and angiogenesis in gastric cancer by delivering hepatocyte growth factor siRNA. Cancer Sci. 2018;109:629–641. doi: 10.1111/cas.13488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Yoon JH, Ham IH, Kim O, Ashktorab H, Smoot DT, Nam SW, Lee JY, Hur H, Park WS. Gastrokine 1 protein is a potential theragnostic target for gastric cancer. Gastric Cancer. 2018;21:956–967. doi: 10.1007/s10120-018-0828-8. [DOI] [PubMed] [Google Scholar]
- 52.Fu H, Yang H, Zhang X, Wang B, Mao J, Li X, Wang M, Zhang B, Sun Z, Qian H, Xu W. Exosomal TRIM3 is a novel marker and therapy target for gastric cancer. J Exp Clin Cancer Res. 2018;37:162. doi: 10.1186/s13046-018-0825-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Wu L, Zhang X, Zhang B, Shi H, Yuan X, Sun Y, Pan Z, Qian H, Xu W. Exosomes derived from gastric cancer cells activate NF-κB pathway in macrophages to promote cancer progression. Tumour Biol. 2016;37:12169–12180. doi: 10.1007/s13277-016-5071-5. [DOI] [PubMed] [Google Scholar]
- 54.Li C, Liu DR, Li GG, Wang HH, Li XW, Zhang W, Wu YL, Chen L. CD97 promotes gastric cancer cell proliferation and invasion through exosome-mediated MAPK signaling pathway. World J Gastroenterol. 2015;21:6215–6228. doi: 10.3748/wjg.v21.i20.6215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Pan L, Liang W, Fu M, Huang ZH, Li X, Zhang W, Zhang P, Qian H, Jiang PC, Xu WR, Zhang X. Exosomes-mediated transfer of long noncoding RNA ZFAS1 promotes gastric cancer progression. J Cancer Res Clin Oncol. 2017;143:991–1004. doi: 10.1007/s00432-017-2361-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Imamura T, Komatsu S, Ichikawa D, Miyamae M, Okajima W, Ohashi T, Kiuchi J, Nishibeppu K, Kosuga T, Konishi H, Shiozaki A, Okamoto K, Fujiwara H, Otsuji E. Low plasma levels of miR-101 are associated with tumor progression in gastric cancer. Oncotarget. 2017;8:106538–106550. doi: 10.18632/oncotarget.20860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Yang H, Fu H, Wang B, Zhang X, Mao J, Li X, Wang M, Sun Z, Qian H, Xu W. Exosomal miR-423-5p targets SUFU to promote cancer growth and metastasis and serves as a novel marker for gastric cancer. Mol Carcinog. 2018;57:1223–1236. doi: 10.1002/mc.22838. [DOI] [PubMed] [Google Scholar]
- 58.Ning X, Zhang H, Wang C, Song X. Exosomes Released by Gastric Cancer Cells Induce Transition of Pericytes Into Cancer-Associated Fibroblasts. Med Sci Monit. 2018;24:2350–2359. doi: 10.12659/MSM.906641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Ohshima K, Inoue K, Fujiwara A, Hatakeyama K, Kanto K, Watanabe Y, Muramatsu K, Fukuda Y, Ogura S, Yamaguchi K, Mochizuki T. Let-7 microRNA family is selectively secreted into the extracellular environment via exosomes in a metastatic gastric cancer cell line. PLoS One. 2010;5:e13247. doi: 10.1371/journal.pone.0013247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Qi J, Zhou Y, Jiao Z, Wang X, Zhao Y, Li Y, Chen H, Yang L, Zhu H. Exosomes Derived from Human Bone Marrow Mesenchymal Stem Cells Promote Tumor Growth Through Hedgehog Signaling Pathway. Cell Physiol Biochem. 2017;42:2242–2254. doi: 10.1159/000479998. [DOI] [PubMed] [Google Scholar]
- 61.Shi L, Wang Z, Geng X, Zhang Y, Xue Z. Exosomal miRNA-34 from cancer-associated fibroblasts inhibits growth and invasion of gastric cancer cells in vitro and in vivo. Aging (Albany NY) 2020;12:8549–8564. doi: 10.18632/aging.103157. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 62.Li Z, Suo B, Long G, Gao Y, Song J, Zhang M, Feng B, Shang C, Wang D. Exosomal miRNA-16-5p Derived From M1 Macrophages Enhances T Cell-Dependent Immune Response by Regulating PD-L1 in Gastric Cancer. Front Cell Dev Biol. 2020;8:572689. doi: 10.3389/fcell.2020.572689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wang JJ, Wang ZY, Chen R, Xiong J, Yao YL, Wu JH, Li GX. Macrophage-secreted Exosomes Delivering miRNA-21 Inhibitor can Regulate BGC-823 Cell Proliferation. Asian Pac J Cancer Prev. 2015;16:4203–4209. doi: 10.7314/apjcp.2015.16.10.4203. [DOI] [PubMed] [Google Scholar]
- 64.Zhang X, Shi H, Yuan X, Jiang P, Qian H, Xu W. Tumor-derived exosomes induce N2 polarization of neutrophils to promote gastric cancer cell migration. Mol Cancer. 2018;17:146. doi: 10.1186/s12943-018-0898-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Xie M, Yu T, Jing X, Ma L, Fan Y, Yang F, Ma P, Jiang H, Wu X, Shu Y, Xu T. Exosomal circSHKBP1 promotes gastric cancer progression via regulating the miR-582-3p/HUR/VEGF axis and suppressing HSP90 degradation. Mol Cancer. 2020;19:112. doi: 10.1186/s12943-020-01208-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Yen EY, Miaw SC, Yu JS, Lai IR. Exosomal TGF-β1 is correlated with lymphatic metastasis of gastric cancers. Am J Cancer Res. 2017;7:2199–2208. [PMC free article] [PubMed] [Google Scholar]
- 67.Zhang H, Deng T, Liu R, Bai M, Zhou L, Wang X, Li S, Yang H, Li J, Ning T, Huang D, Li H, Zhang L, Ying G, Ba Y. Exosome-delivered EGFR regulates liver microenvironment to promote gastric cancer liver metastasis. Nat Commun. 2017;8:15016. doi: 10.1038/ncomms15016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Huang T, Song C, Zheng L, Xia L, Li Y, Zhou Y. The roles of extracellular vesicles in gastric cancer development, microenvironment, anti-cancer drug resistance, and therapy. Mol Cancer. 2019;18:62. doi: 10.1186/s12943-019-0967-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Deng G, Qu J, Zhang Y, Che X, Cheng Y, Fan Y, Zhang S, Na D, Liu Y, Qu X. Gastric cancer-derived exosomes promote peritoneal metastasis by destroying the mesothelial barrier. FEBS Lett. 2017;591:2167–2179. doi: 10.1002/1873-3468.12722. [DOI] [PubMed] [Google Scholar]
- 70.Xu G, Zhang B, Ye J, Cao S, Shi J, Zhao Y, Wang Y, Sang J, Yao Y, Guan W, Tao J, Feng M, Zhang W. Exosomal miRNA-139 in cancer-associated fibroblasts inhibits gastric cancer progression by repressing MMP11 expression. Int J Biol Sci. 2019;15:2320–2329. doi: 10.7150/ijbs.33750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Gu H, Ji R, Zhang X, Wang M, Zhu W, Qian H, Chen Y, Jiang P, Xu W. Exosomes derived from human mesenchymal stem cells promote gastric cancer cell growth and migration via the activation of the Akt pathway. Mol Med Rep. 2016;14:3452–3458. doi: 10.3892/mmr.2016.5625. [DOI] [PubMed] [Google Scholar]
- 72.Zheng P, Luo Q, Wang W, Li J, Wang T, Wang P, Chen L, Zhang P, Chen H, Liu Y, Dong P, Xie G, Ma Y, Jiang L, Yuan X, Shen L. Tumor-associated macrophages-derived exosomes promote the migration of gastric cancer cells by transfer of functional Apolipoprotein E. Cell Death Dis. 2018;9:434. doi: 10.1038/s41419-018-0465-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Piao HY, Guo S, Wang Y, Zhang J. Exosome-transmitted lncRNA PCGEM1 promotes invasive and metastasis in gastric cancer by maintaining the stability of SNAI1. Clin Transl Oncol. 2021;23:246–256. doi: 10.1007/s12094-020-02412-9. [DOI] [PubMed] [Google Scholar]
- 74.Huang Z, Zhu D, Wu L, He M, Zhou X, Zhang L, Zhang H, Wang W, Zhu J, Cheng W, Chen Y, Fan Y, Qi L, Yin Y, Zhu W, Shu Y, Liu P. Six Serum-Based miRNAs as Potential Diagnostic Biomarkers for Gastric Cancer. Cancer Epidemiol Biomarkers Prev. 2017;26:188–196. doi: 10.1158/1055-9965.EPI-16-0607. [DOI] [PubMed] [Google Scholar]
- 75.Tang XH, Guo T, Gao XY, Wu XL, Xing XF, Ji JF, Li ZY. Exosome-derived noncoding RNAs in gastric cancer: functions and clinical applications. Mol Cancer. 2021;20:99. doi: 10.1186/s12943-021-01396-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Li Q, Shao Y, Zhang X, Zheng T, Miao M, Qin L, Wang B, Ye G, Xiao B, Guo J. Plasma long noncoding RNA protected by exosomes as a potential stable biomarker for gastric cancer. Tumour Biol. 2015;36:2007–2012. doi: 10.1007/s13277-014-2807-y. [DOI] [PubMed] [Google Scholar]
- 77.Lin LY, Yang L, Zeng Q, Wang L, Chen ML, Zhao ZH, Ye GD, Luo QC, Lv PY, Guo QW, Li BA, Cai JC, Cai WY. Tumor-originated exosomal lncUEGC1 as a circulating biomarker for early-stage gastric cancer. Mol Cancer. 2018;17:84. doi: 10.1186/s12943-018-0834-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Tokuhisa M, Ichikawa Y, Kosaka N, Ochiya T, Yashiro M, Hirakawa K, Kosaka T, Makino H, Akiyama H, Kunisaki C, Endo I. Exosomal miRNAs from Peritoneum Lavage Fluid as Potential Prognostic Biomarkers of Peritoneal Metastasis in Gastric Cancer. PLoS One. 2015;10:e0130472. doi: 10.1371/journal.pone.0130472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Yang T, Martin P, Fogarty B, Brown A, Schurman K, Phipps R, Yin VP, Lockman P, Bai S. Exosome delivered anticancer drugs across the blood-brain barrier for brain cancer therapy in Danio rerio. Pharm Res. 2015;32:2003–2014. doi: 10.1007/s11095-014-1593-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Jiang K, Dong C, Yin Z, Li R, Wang Q, Wang L. The critical role of exosomes in tumor biology. J Cell Biochem. 2019;120:6820–6832. doi: 10.1002/jcb.27813. [DOI] [PubMed] [Google Scholar]
- 81.Pascucci L, Coccè V, Bonomi A, Ami D, Ceccarelli P, Ciusani E, Viganò L, Locatelli A, Sisto F, Doglia SM, Parati E, Bernardo ME, Muraca M, Alessandri G, Bondiolotti G, Pessina A. Paclitaxel is incorporated by mesenchymal stromal cells and released in exosomes that inhibit in vitro tumor growth: a new approach for drug delivery. J Control Release. 2014;192:262–270. doi: 10.1016/j.jconrel.2014.07.042. [DOI] [PubMed] [Google Scholar]
- 82.Wang X, Zhang H, Bai M, Ning T, Ge S, Deng T, Liu R, Zhang L, Ying G, Ba Y. Exosomes Serve as Nanoparticles to Deliver Anti-miR-214 to Reverse Chemoresistance to Cisplatin in Gastric Cancer. Mol Ther. 2018;26:774–783. doi: 10.1016/j.ymthe.2018.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Zhong H, Yang Y, Ma S, Xiu F, Cai Z, Zhao H, Du L. Induction of a tumour-specific CTL response by exosomes isolated from heat-treated malignant ascites of gastric cancer patients. Int J Hyperthermia. 2011;27:604–611. doi: 10.3109/02656736.2011.564598. [DOI] [PubMed] [Google Scholar]
- 84.Borst J, Ahrends T, Bąbała N, Melief CJM, Kastenmüller W. CD4(+) T cell help in cancer immunology and immunotherapy. Nat Rev Immunol. 2018;18:635–647. doi: 10.1038/s41577-018-0044-0. [DOI] [PubMed] [Google Scholar]
- 85.Xie F, Xu M, Lu J, Mao L, Wang S. The role of exosomal PD-L1 in tumor progression and immunotherapy. Mol Cancer. 2019;18:146. doi: 10.1186/s12943-019-1074-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Tang XJ, Sun XY, Huang KM, Zhang L, Yang ZS, Zou DD, Wang B, Warnock GL, Dai LJ, Luo J. Therapeutic potential of CAR-T cell-derived exosomes: a cell-free modality for targeted cancer therapy. Oncotarget. 2015;6:44179–44190. doi: 10.18632/oncotarget.6175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Kahroba H, Hejazi MS, Samadi N. Exosomes: from carcinogenesis and metastasis to diagnosis and treatment of gastric cancer. Cell Mol Life Sci. 2019;76:1747–1758. doi: 10.1007/s00018-019-03035-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Lin H, Zhang L, Zhang C, Liu P. Exosomal MiR-500a-3p promotes cisplatin resistance and stemness via negatively regulating FBXW7 in gastric cancer. J Cell Mol Med. 2020;24:8930–8941. doi: 10.1111/jcmm.15524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Jiang L, Zhang Y, Guo L, Liu C, Wang P, Ren W. Exosomal microRNA-107 reverses chemotherapeutic drug resistance of gastric cancer cells through HMGA2/mTOR/P-gp pathway. BMC Cancer. 2021;21:1290. doi: 10.1186/s12885-021-09020-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Ji R, Zhang B, Zhang X, Xue J, Yuan X, Yan Y, Wang M, Zhu W, Qian H, Xu W. Exosomes derived from human mesenchymal stem cells confer drug resistance in gastric cancer. Cell Cycle. 2015;14:2473–2483. doi: 10.1080/15384101.2015.1005530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Zheng P, Chen L, Yuan X, Luo Q, Liu Y, Xie G, Ma Y, Shen L. Exosomal transfer of tumor-associated macrophage-derived miR-21 confers cisplatin resistance in gastric cancer cells. J Exp Clin Cancer Res. 2017;36:53. doi: 10.1186/s13046-017-0528-y. [DOI] [PMC free article] [PubMed] [Google Scholar]