BACKGROUND
Introduction
Local tissue rearrangement through the targeted manipulation of local tissue flaps is a fundamental technique in facial reconstruction. A flap is a unit of tissue that maintains its own blood supply while being transferred from a donor site to a recipient site.1 Local flaps are obtained from tissue in the immediate region of the defect to be reconstructed. They can be transferred using a variety of methods including advancement, transposition, rotation, and interpolation. Small and moderate-sized facial cutaneous defects are very often reconstructed with local flaps.
The intrinsic skin vascularity of a local flap is the most critical determinant of successful transfer2 (Figs. 1 and 2). Fortunately, the head and neck cutaneous regions are richly vascularized, with significant redundancy of the vascular system. Perfusion is dictated by major distributing vessels along septocutaneous and musculocutaneous perforators, which lead to a densely interconnected network of dermal and subdermal plexuses3 (Fig. 3). One concept used to understand perfusion is that of the angiosome, proposed by Taylor and Palmer, whereby the body is said to be comprised anatomically of multiple three-dimensional composite tissue blocks, consisting of muscle, fascia, subcutaneous fat, and skin, that are supplied by particular source arteries.4 The head and neck region has 13 angiosomes supplied mostly from branches of the external carotid, internal carotid, and subclavian arteries, which form the theoretic basis for the design of tissue flaps. It is this redundant network that allows for the creation and survival of local flaps. Local flaps are often classified based on their vascularity. McGregor and Morgan classified cutaneous flaps as random flaps, axial pattern or reverse-flow axial pattern flaps, and island flaps.5 Random flaps are based on the subdermal plexus and traditionally limited to 3:1 length-to-width ratios, although this is region dependent and can be theoretically extended to 4:1 in the well-perfused head and neck regions.2 Axial pattern flaps contain a single direct artery within the longitudinal axis of the flap.
Fig. 1.
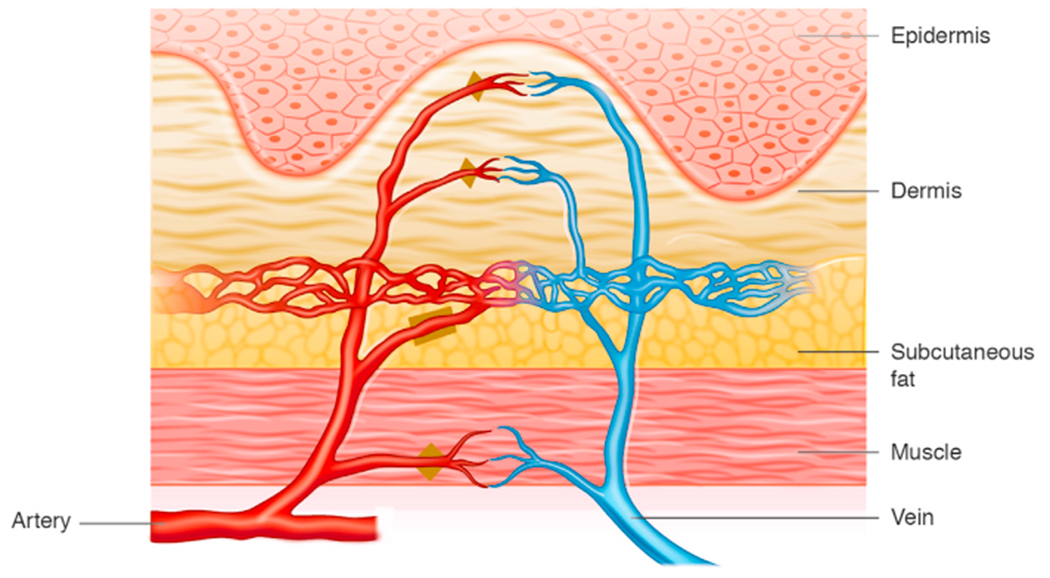
Skin contains precapillary sphincters (designated by triangle), which regulates blood perfusion by local stimuli and preshunt sphincters (designated by square) which regulates thermoregulation. (Borrowed with permission from Local Flaps in Head and Neck Reconstruction, Figure 2.1, page 14.51)
Fig. 2.

Vasculature of facial skin. (Borrowed with permission from Local Flaps in Head and Neck Reconstruction, Figure 26.10, page 700.51)
Fig. 3.

Overlapping vascular skin regions from multiple perforating vessels that interconnect to maximize skin perfusion. (Borrowed with permission from Local Flaps in Head and Neck Reconstruction, Figure 2.4, page 16.51)
Common etiologies of flap failure
Complications are an inherent and inevitable part of surgery, and learning how to prevent them, as well as manage them when they occur, is essential. As the face is a complex region with critical esthetic and functional purposes, complications during reconstruction of this region can result in disfiguring and functional consequences.6,7
Complications from reconstruction with local flaps can take many forms. Etiologies of skin flap failure include tension-related, ischemic, hematologic, and infectious causes.6 Complications may arise intraoperatively or may not occur until the early or late postoperative phases. Flap perfusion is paramount, and strict evaluation of perfusion is essential to prevent, recognize, and treat impending complications.2 Common causes of flap compromise include systemic patient factors such as comorbidities (diabetes, malnutrition, conditions of poor wound healing and immunosuppression), concurrent smoking, recreational drug or medication use, hypovolemia, hypotension, and anemia.1 In addition, poor surgical technique, cool ambient room temperature, kinking of the flap or pedicle, hematoma, and pressure on the flap may also lead to flap failure.8 Therefore, careful preparation and excellent technique are critical for a successful outcome.
SURGICAL TECHNIQUE
Patient Preparation
Having a consistent perioperative routine for skin flap care is the first step in reducing complications. Avoiding complications begins in the preoperative period with a thorough patient history and physical examination. Every patient should be preoperatively optimized and comorbidities should be actively managed. Numerous comorbidities have a significant impact on wound healing. These include age, functional status, diabetes, renal disease, cardiac disease, liver failure, peripheral vascular disease, tobacco use, vasculitis, malnutrition, immunosuppression, obesity, inherited blood dyscrasias, inflammatory skin conditions, history of hypertrophic scarring or keloids, and history of radiation to the site.2,6,7 The overall nutrition of the patient, including the presence of nutritional and vitamin deficiencies (eg, albumin <2.5 mg/dL), can significantly alter the wound healing pathway.9 Although some of these factors may be optimized, others are not modifiable. Nonetheless, knowledge of their presence is helpful and may impact surgical approaches and modify the risk of skin flap failure.
Medications may impact the outcome of local flap reconstruction. Patients should be questioned about the use of medications and herbal supplements that reduce clotting or contribute to immunosuppression before surgery, with clear instructions regarding reducing these substances if possible, in the perioperative period. These include nonsteroidal anti-inflammatory drugs, commonly used anticoagulants such as warfarin, aspirin, and clopidogrel, and steroids.1 In addition, the use of herbal supplements should be queried. Ginkgo biloba, ginseng, ephedra, kava, St. John’s wort, echinacea, garlic, valerian, and vitamin E are among the more commonly used herbal supplements and can exert substantial effects on coagulation.10
Alcohol and tobacco use both significantly increase the risk of local flap complications. Heavy alcohol use modulates liver function, which affects the clotting cascade and drug metabolism, increasing risks for excessive bleeding or hematoma formation.11 Tobacco use has a well-documented detrimental effect on tissue perfusion.2 Smoking one cigarette reduces tissue perfusion by more than 30% within 45 min in some regions of the body.12 The injurious effects of nicotine seem to increase with prolonged exposure.13 The mechanism whereby tobacco or nicotine lowers flap survival is believed to be direct endothelial damage or vasoconstriction secondary to catecholamine release or local concentrations of prostaglandins. Smoking impairs flap healing by causing vasoconstriction from nicotine and increased levels of carboxyhemoglobin, which limits oxygen delivery. In addition, smoking decreases neutrophilic function and decreases collagen synthesis.14,15 Nonsmokers produce 1.8 times more collagen compared with smokers.15 Interestingly, the role of nicotine and its relationship to tissue hypoxia is not fully known, as nicotine transdermal patch use did not have comparable effects on wound healing compared with tobacco smoking in one clinical trial.14
Smoking has been associated with an increased incidence of flap necrosis in facelift operations.16 Exposure to tobacco smoke resulted in increased flap necrosis of dorsal flaps in rodents.17 In a retrospective study of 91 patients with facial skin defects reconstructed with local flaps, of which 38 (42%) were active smokers, complications occurred in 23 patients (25%; 37% in smokers, 17% in ex-smokers, and 17% in nonsmokers; P < .03), and all full-thickness skin losses and all cellulitis occurred in active smokers. The authors concluded that active smokers are at a higher risk for complications in facial skin flap surgery and that ex-smokers had a complication rate similar to that of nonsmokers.18 In a study examining the association between cigarette smoking and necrosis of flaps and full-thickness grafts in 220 patients, a review of a series of 916 flaps and full-thickness grafts revealed 44 patients in whom some degree of tissue necrosis occurred. Current high-level smokers (those smoking one or more packs per day) had necrosis developed approximately three times more often than never-smokers, low-level smokers (less than one pack per day), or former smokers (95% confidence interval [CI], 1.2 to 8.2). Former smokers (relative risk, 1.4; 95% CI, 0.6 to 3.2) and low-level smokers (relative risk, 1.1; 95% CI, 0.2 to 6.1) were not at increased risk for necrosis compared with never-smokers. Once tissue necrosis developed, the median percent of the visible flap or graft tissue that necrosed was approximately threefold greater among current smokers than never smokers.19 These studies suggest that part of smoking’s adverse effect on skin flaps may be an acute phenomenon, and that smoking cessation for shorter (<1 year) periods of time before surgery may have a beneficial effect.
Smoking also results in a higher incidence of surgical site infection (SSI). This effect is present even for small, clean wounds.14 Preoperative smoking abstinence reduces postoperative wound infections.20 Poor wound healing and infection may be detrimental to local flaps and can lead to flap failure, especially when perfusion to flap tissue is tenuous.
The optimal duration of preoperative abstinence from smoking is debated. In one study, 4 weeks of abstinence reduced the wound infection rate to the level of never-smokers.14 The wound healing process itself may require a longer time of abstinence. Smoking cessation for at least 4 to 6 weeks is recommended before skin flap surgery.1,21 Medications such as varenicline (Chantix) may assist in smoking cessation. Other strategies, such as the electronic cigarette or nicotine replacement therapy (eg, transdermal nicotine patch, nicotine gum) may be helpful in reducing smoking and improving flap outcomes, although do not entirely eliminate nicotine from the body.
Whenever possible, avoid flap transposition in previously irradiated regions due to decreased vascularity. In addition, if the patient has a previous history of surgery at the operative site, one needs to be cognizant as to whether vascularity has already been compromised.
Intraoperative Strategies
Good surgical technique is critical to the success of local flap reconstruction. The reconstructive surgeon should always remember that “blood supply is king.” Techniques that promote maximal preservation of blood supply and the highest levels of perfusion will decrease flap failure. Various fundamental principles and techniques intraoperatively should be used to increase the probability of success.
This starts with an appropriate flap design. Excessive tension on the flap should be avoided, which is dependent first and foremost on selecting an effective flap design. When designing a flap, relaxed skin tension lines and facial subunits should be also taken into consideration. Placement of incisions parallel to relaxed skin-tension lines and along esthetic subunit borders allows for maximal extensibility and camouflage of the resultant scar.6 Tension should not distort surrounding landmarks, especially mobile and functional ones such as the nose, eyelids, and lips. The flap should be designed such that excessive stretch is not required for closure, and the pedicle will not be kinked during inset. It should be noted that increasing the pedicle width does not necessarily ensure adequate perfusion to the distal flap.22 In fact, raising a thicker flap with deeper subcutaneous vessels is more beneficial than making a wider flap base (Fig. 4). The old adage “measure twice, cut once” is essential in flap design.
Fig. 4.

To improve local skin perfusion, increasing skin flap thickness is more beneficial than widening its pedicle. (Borrowed with permission from Local Flaps in Head and Neck Reconstruction, Figure 26.11, page 701.51)
When administering subcutaneous local anesthesia, avoid excessive injection of vasoconstrictors such as epinephrine around the vascular pedicle and underneath the flap. During second stage skin flap surgery or delaying a flap, consider using a 1:200,000 epinephrine concentration due to increased sensitivity of the precapillary sphincters. During surgery, gentle flap manipulation, delicate tissue handling, and avoidance of pressure, especially at the distal edge of the flap, will help preserve tissue viability. For example, the surgeon can place a single retraction suture at the distal end of the flap instead of grasping the flap with forceps to minimize pressure on the flap. For axial flaps, the feeding vessel must be preserved, or the flap will not survive. Excessive distal flap thinning should be avoided to maximize perfusion especially if the patient is a smoker.23
Hemostasis can be improved by injection 5 to 10 min before making the incision. Bleeding is most often encountered during the initial incision or while undermining the flap. Meticulous hemostasis and good technique help avoid postoperative hematoma formation. Undermining in a uniform plane at the correct depth minimizes blood loss. Vessel identification with the precise use of bipolar electrocautery is helpful.6 Overly aggressive electrocautery should be avoided, since the dermal and subdermal plexuses contribute to flap perfusion. Larger (>1 mm) arterial vessels should be ligated. Copious irrigation with sterile water and gently blotting with gauze can help define bleeding vessels. If it is not contraindicated by the anesthesia team, maintain systolic blood pressure <105 mm Hg during the flap procedure to minimize flap vascular oozing.
Once adequate hemostasis is achieved, attention is turned to closure. A tension-less closure is important for reducing the risk of flap failure and improving the functional and esthetic result. Tension decreases perfusion and distorts critical landmarks. In one study, there was a statistically significant increase in flap necrosis for those flaps closed with greater than 250 g of tension.24 Extensive surrounding undermining may be necessary to help achieve tension-less closure, with anchor sutures placed more deeply to assist in closure. In some instances, “delaying” a skin flap may be beneficial to prime and lengthen the random portion of the skin flap in preparation before flap transposition. Delaying a skin flap can be especially important in increasing skin flap survival if prior medical co-morbidities exist (such as diabetes or conditions of poor healing). Skin flap “delay” involves making the skin flap peripheral incisions 2 weeks before raising and transposing the flap. In this way, it primes the distal region of the skin for improved tissue perfusion. When an interpolated flap is transposed to a slow healing region, pedicle detachment of the flap may require 6 to 8 weeks for formation of adequate blood supply. To test a flap before detachment in a poor healing bed, a vascular loop tourniquet can be applied for 10 min. If cyanosis is apparent, flap detachment should be delayed.7
MANAGEMENT STRATEGIES WHEN SKIN FLAP COMPROMISE IS RECOGNIZED POSTOPERATIVELY
Attentive postoperative monitoring is critical for identifying flap compromise and recognizing potential complications early. Thus, periodic postoperative flap inspections are important. The sooner a problem is recognized, the higher probability of successful salvage of the skin flap. Flaps can be clinically monitored using a variety of indicators, including color, capillary refill, punctate bleeding, temperature, and firmness. Healthy flaps will be pink in color, have less than 4 s capillary refill after light manual pressure, will bleed bright red blood upon poking the skin with a needle, and will be soft with modest turgor.7 Microvascular free flaps (not covered in this article) can also be monitored using Doppler, pulse oximetry, pH, temperature sensors, and laser angiography with indocyanine green.6,25 However, for local skin flaps, there is no widely accepted device to monitor skin flap perfusion besides clinical inspection.
Once skin flap compromise is recognized, ideally within hours after surgery, the next step is to determine the underlying etiology of compromise. Identifying the underlying cause for skin flap compromise is essential to determine the best clinical management. Common etiologies for flap compromise include ischemic (arterial or venous), hematologic, and infectious causes (Fig. 5).
Fig. 5.

Algorithm for management of an acute failing skin flap.
Ischemic compromise
Ischemia is defined as inadequate perfusion to meet tissue needs. Causes of ischemia may be reversible or irreversible. Fortunately, significant redundancy is present in the perfusion system. It is thought that most tissue can survive on 10% of its average blood flow.7 Most flaps can endure arterial insufficiency for up to 13 h, but venous congestion can kill a flap within 3 h.7 Blood supply is essential for flap survival. Thus, prompt recognition of ischemic compromise is extremely important for preventing flap failure.
Several clinical indicators are helpful to detect skin flap compromise. In an arterial compromise, the flap may exhibit pale color, slow capillary refill (>4 s), slow or absent pinpoint bleeding, cool temperature, and/or low turgor. In a venous compromise, the flap may exhibit a mottled blue to purple cyanotic color, brisk dark pinpoint bleeding, increased temperature, and/or increased turgor (tense or swollen).8 Evidence of arterial or venous insufficiency in the postoperative period requires immediate exploration within 4 h to salvage the local flap.
Arterial compromise
If skin flap ischemia is suspected, immediate methods to increase flap perfusion should be instituted. Arterial ischemia can be attributed to pedicle tension or torsion, vessel plaque, arterial spasm, pressure on the pedicle, technical error with pedicle injury, flap too large for its blood supply, or small-vessel disease (eg, smoking, diabetes).26 Initial immediate responses to improve skin flap perfusion include releasing flap torsion, removing tight sutures, and applying topical heat to help restore arterial perfusion.8
Pharmacologic interventions sympatholytics, corticosteroids, calcium channel blockers, rheological drugs, direct vasodilators, and free radical scavengers (such as dimethyl sulfoxide) to salvage the flap with arterial insufficiency have been attempted, with inconclusive results.27 Arterial ischemia can be caused by vessel occlusion from clotted blood. Given this, anticoagulant and thrombolytic agents have been used in an attempt to minimize or reverse arterial compromise. Heparin acts in conjunction with antithrombin III to inhibit thrombosis by inactivation of factor X, but it has been shown to be more effective at preventing venous thrombosis than arterial thrombosis.2 Tissue plasminogen activator (tPA) and anisoylated plasminogen streptokinase activator complex (APSAC) exert their effects by cleaving fibrin within a thrombus. However, these agents will only improve flap perfusion if an arterial thrombus is the primary etiology of ischemia. Heparin and thrombolytic agents are more often used in free flap salvage, where the microvascular anastomosis is more susceptible to thrombotic ischemia.28 In addition, the use of heparin is not without risk, as heparin can paradoxically lead to thrombosis via heparin-induced thrombocytopenia.
Aspirin has been used in reconstructive surgery as well. Aspirin acetylates the enzyme cyclooxygenase, thereby decreasing the synthesis of thromboxane A2, a potent vasoconstrictor in platelets, and prostacyclin, a potent vasodilator in vessel walls.2 At low doses (eg, aspirin 81 mg) the effect of aspirin is selective, with inhibition of cyclooxygenase, reduction in the formation of thromboxane, and less vasoconstriction. Despite these effects, there is no empiric evidence in the literature for using aspirin postoperatively.29 Although aspirin is used empirically by some microvascular surgeons after free tissue transfer, in local tissue rearrangement the risks of bleeding from aspirin use outweigh any theoretic benefit on perfusion.
Avoidance of local hypothermia is an important adjunct to promote arterial profusion. Cold environments can provoke arterial ischemia. Temperature modulates blood vessel patency and resultant blood flow. In the range of 22° to 38° Celsius, the relationship between flap blood flow and the temperature was linear, with an increase in blood flow by 3.41% per degree Celsius.30 All patients should be instructed to avoid ice or excessive surface pressure on the flap. If anything, light warm compresses may be used to increase vessel patency.
If arterial compromise is persistent despite efforts to resolve it, hyperbaric oxygen therapy (HBOT) can be considered as an adjunctive modality in a patient with an ischemic compromised skin flap. HBOT delivers oxygen concentrations at pressures higher than atmospheric pressures. With a “dive” of 2.4 atm, tissue wound partial pressure of oxygen will be 800 to 1100 mm Hg. This exposure induces increased partial pressures of oxygen in the blood, which subsequently stimulates angiogenesis and fibroblast proliferation.31 Proponents state that HBOT may reduce hypoxic injury, stimulate angiogenesis, and improve fibroblast function for collagen formation in an injured flap. Some animal studies show that hyperbaric oxygen can improve skin flap survival.32 Multiple studies have shown promise that HBOT may improve ischemic skin flap survival, but robust consistent randomized studies are lacking.1 Unfortunately, there is no standard HBOT protocol, and it remains unclear which skin flap patients should undergo HBOT, the optimal dive session number, session duration, and atmospheres delivered. In addition, HBOT may induce adverse effects such barotrauma and exacerbation of congestive heart failure.2 Despite this, reconstructive surgeons should be aware of this treatment modality as it may be helpful in select cases. If HBOT is considered an adjuvant therapy to salvage an arterial compromised skin flap, it should be instituted within 48 h of the flap insult.33
In regards to perioperative steroid administration to manage an ischemic flap, several animal studies have shown promise to increase flap viability34,35 However, no large human study has been conducted.
Venous Compromise
Venous insufficiency can rapidly compromise flap viability. It can occur secondary to pedicle tension or torsion, flap edema, hematoma, or in the case of an island flap, tension within the tunnel.27 Once venous outflow obstruction has developed, it can lead to erythrocyte extravasation, fibrin deposits in the perivascular regions, endothelial breakdown, microvascular collapse, arterial compromise, arteriovenous shunting away from the capillary bed, ischemia, thrombosis within the microcirculation, and ultimately flap death, which may occur within 8 h.36
Similar methods used in arterial compromise may be applied to suspected venous compromise in some scenarios. If venous congestion develops due to tension on the flap, torsion, or pressure on the venous outflow, appropriate measures to quickly restore the venous flow should be instituted. Pedicle or inset revision may be necessary in the case of axial or island flaps. Tight sutures should be cut, external pressure on the flap removed, hematoma evacuated, and/or tension or kinking on the pedicle corrected.
Medications can occasionally be useful in cases of venous compromises. Vasodilation agents may increase venous outflow in tenuous cases and improve flap survival. Topical nitroglycerin is a potent vasodilator with selective effects on the venous circulation. There is evidence to suggest improved survival of axial flaps using treatment with transdermal nitroglycerin.37
In cases where initial measures to relieve venous congestion are not immediately effective, hirudotherapy, also known as leech therapy, may be considered. Leeches have been used for health purposes for millennial, with reports of bloodletting dating as far back as the Stone Age.26 More recently, since around 1960, the medical leech Hirudo medicinalis has been used clinically to help salvage venous congested skin.38 Leeches exert their effect by injecting hirudin (an anticoagulant) at the site of their bite, reducing tissue necrosis by removing engorged blood from tissues otherwise lacking adequate venous drainage.39 Hirudin is a naturally occurring anticoagulant that inhibits the conversion of fibrin to fibrinogen, but it does not require antithrombin III for activation, unlike heparin.2 Leeches also secrete several additional compounds that may promote flap survival, such as hyaluronidase and vasodilators. Substantial evidence supports the use of leeches in skin flaps with venous congestion (Fig. 6).38,40
Fig. 6.

(A–C) Example of a patient undergoing leech therapy salvage for venous congestion of a paramedian forehead flap to the left nose.
Importantly, hirudotherapy is considered a temporary modality to decrease skin flap venous congestion until further revascularization occurs for venous outflow.38 It is not effective in the setting of arterial flow compromise or other nonvenous causes of flap failure.
There is no standard protocol for administering leech therapy in cases of venous compromise. One author recommends initiation of intervention within 2 to 3 h of the onset of venous congestion, as flap survival decreases precipitously within 8 h after onset.26 Therapy is then continued until further angiogenesis has occurred and sufficient venous outflow is established, usually for 2 to 6 days, and guided by clinical improvement in flap metrics such as color, appearance, and capillary refill.38 Hemoglobin should be monitored at regular intervals throughout therapy due to blood loss from the leeches.
Despite the possibility of flap salvage with leech therapy, its use is not without risks. Frequently cited disadvantages include the risk of infection, local reactions of pain and itching, the need for serial blood transfusions, anaphylaxis, bleeding, and scarring.2,38 Hirudo medicinalis leeches are frequently associated with bacterial infection with Aeromonas hydrophila, thus antibiotic prophylaxis, commonly with fluoroquinolones, is always necessary.38,41
Hematologic Compromise
Hematologic compromise of flaps is related to excessive bleeding, which may cause hematoma, pressure, or poor perfusion. This is most often related to drug-induced coagulopathy and poor intraoperative hemostasis.6,7 Uncontrolled hypertension, renal or hepatic failure, collagen vascular disorders, hematopoietic system malignancies, and excessive valsalva activity from vomiting or strenuous exercise in the immediate postoperative period are linked to bleeding complications.7 Antiemetics can be prescribed to prevent postoperative nausea and vomiting, thereby decreasing forceful valsalva straining.
Aspirin, warfarin, and clopidogrel are the most implicated anticoagulants. In a retrospective review of patients taking clopidogrel and undergoing cutaneous surgery, severe complications were 6 times more likely with clopidogrel-containing anticoagulation and 8 times more likely with combined clopidogrel and aspirin than with aspirin monotherapy.42 In a meta-analysis of complications attributed to anticoagulation following cutaneous surgery, patients taking warfarin were nearly seven times as likely to have a moderate-to-severe complication compared with controls (odds ratio [OR], 6.69; 95% CI, 3.03 to 14.7, P < .001).43 However, cessation of anticoagulation may lead to serious thrombotic complications such as deep venous thrombosis, pulmonary embolism, myocardial infarction, or stroke. As such, the risks and benefits of cessation of anticoagulation must be carefully weighed, with the help of primary care or prescribing providers.
Hematologic compromise is frequently related to hematoma formation. Hematoma reduces perfusion and can lead to ischemia or necrosis by inducing vasospasm, stretching the subdermal plexus, or separating the flap from its recipient bed. Iron compounds from a hematoma may also promote local free radical production which can contribute to flap necrosis.44 Early recognition of hematoma formation is imperative. Increasing flap tension, a soft expanding mass, excessive bleeding, and an increase in pain may all serve as indicators of hematoma. Aspiration of hematoma and pressure dressings may help control a small hematoma. However, pressure dressings can be difficult to place on the face and too much pressure can lead to ischemia. Wound exploration with hematoma evacuation and identification of the bleeding source may be necessary if conservative measures fail.44 Within the first 48 h, hematomas are in liquid form. Over several days, as the clot becomes more solidified, it becomes more difficult to aspirate. If the hematoma is diagnosed more than 24 h after surgery, exploration may or may not be necessary, depending on the size of the hematoma and the status of the flap.44 Hematomas may again liquefy from fibrinolysis in approximately 14 days.
Aside from deliberate and judicious intraoperative hemostasis, several postoperative measures can be used. These include elevating the head of the bed and administering antiemetics to prevent excessive nausea and vomiting (which can induce excessive hypertension leading to bleeding) and pain control to reduce elevated blood pressure. Patients should be instructed to avoid strenuous activity for at least 1 week after surgery to reduce the risk of bleeding.
Infectious Compromise
Infection is a risk with any procedure, and local flaps are no exception.7 Fortunately, postoperative infections after clean cutaneous facial surgery are rare (<3% in one study).45 However, reconstruction is not always performed on the same day as resection, which may result in an initially clean wound becoming a clean-contaminated one, thereby increasing the risk of infection. Moreover, facial wounds of local flaps are more prone to infections than wounds closed primarily because local flaps are relatively ischemic. Ensuring adequate tissue oxygenation by way of good vascular perfusion is one of the most important factors for infection prevention.7,45,46
Certain anatomic areas of the face have high bacterial colonization, such as the nose, ears, and lips, which predisposes wounds to higher bacterial loads.45 Good aseptic and antiseptic techniques are fundamental preventive strategies, and prevention is the most effective strategy.7 Importantly, antibiotic treatment is not directly correlated with infection eradication. Perioperative antibiotics are not required in clean wounds. For clean contaminated wounds, such as those that involve the oral cavity or intranasal mucosa, prophylactic antibiotics are at the discretion of the provider, as their utility in preventing postsurgical infection remains controversial.6 Most surgeons will administer a perioperative antimicrobial prophylaxis dose for clean-contaminated cases or in revision cases, typically with a first-generation cephalosporin such as cefazolin. Systemic antibiotics are ideally administered 1 h before incision time to obtain adequate blood concentration at the time of surgery.7
Infection typically manifests four to 8 days following surgery and can occur despite every preventive effort. If an infection does develop, as evidenced by induration, erythema, pain, wound warmth, and/or purulence, wound drainage should be sent for culture and sensitivities, to determine which antibiotics should be initiated. Antistaphylococcal antibiotics, with consideration for methicillin-resistant strains, when applicable, are first line. The infection should be fully cleared before any attempts at revision reconstruction are done to prevent further tissue loss. Application of an antibiotic ointment over incision lines may be beneficial to prevent infection, however, keeping the incision covered with any moisture-retentive ointment is important to assist in the incision closure. Bacitracin/polymyxin B ointment is associated with less allergic reactions than neomycin-containing ointments and is therefore preferred.7 Hydrogen peroxide application can be used at wound edges to reduce heavy crusting and local bacterial wound load.
Wound Care (Partial Thickness Versus Full Thickness Loss)
Flap complications can range from superficial epidermolysis to full-thickness necrosis. This necessitates effective wound care to promote good healing and minimize the consequences from flap loss. Management of tissue loss from ischemic changes is conservative. Early debridement should be avoided until the tissue has declared its long-term viability. After a demarcation line of viability is formed, necrotic tissue can be debrided. The wound can be left to heal by secondary intention, or another reconstructive surgery (skin flap or skin graft) can be planned, depending on the size, location, and functional importance of the defect and the patient’s expectations.6,7
Wound care with moist ointments, antimicrobial prophylaxis, and vacuum-assisted closure (VAC) may expedite the process for open wounds to granulate from secondary intention. Maintenance of a moist environment along flap edges promotes epithelialization and serves a fundamental strategy for minimizing scarring.9,47 Silicone gels can begin 1 month after the flap incision to minimize flap incision scars. Excessive scarring 1 month postoperatively may be managed with intralesional 0.5 to 1 mL triamcinolone acetonide (10 to 40 mg/mL) injections every 6 weeks until a desired effect is reached. Verapamil, bleomycin, and 5-fluoriuracil have also been used in more severe cases of hypertrophic scarring. Postoperative application of pressure dressings, silicone sheeting, cryotherapy, and laser therapy may be used as adjuncts.48,49 Surgical scar revision should not be performed until after 6 months to allow for full healing to take place, unless the developing scar is causing a functional deformity.
For poor healing open wounds and multiple skin flap failures, VAC is an option to increase the granulation of an open wound bed for skin grafting or skin flaps. The VAC is an open-cell polyurethane foam dressing sealed with an adherent dressing subjected to sub-atmospheric pressure by a connected vacuum pump. The device functions by removing unwanted wound fluid, decreasing edema, increasing the production of granulation tissue and decreasing bacterial counts in wounds.50 Negative pressure is applied continuously for 48 h and then intermittently. The VAC should not be used in contact with vessels or in the nasal and oral airways and should be used very carefully when in proximity to the eyes and ears. Mobile or highly contoured skin areas, such as the neck, may make the vacuum placement more challenging to achieve a seal. Other experimental modalities are currently being investigated to enhance skin flap viability (Fig. 7).
Fig. 7.

Research modalities to try to improve skin flap survival. SOD, superoxide dismutase. (Borrowed with permission from Local Flaps in Head and Neck Reconstruction, Figure 2.11, page 14.51)
SUMMARY
Avoiding skin flap complications begins in the preoperative period with a thorough patient history, including relevant medications, and physical examination. Every patient should be preoperatively optimized and comorbidities should be actively managed. Intraoperatively, the good surgical technique will reduce the risk of failure. This includes optimal flap design, delicate tissue handling, judicious hemostasis, and tension-less closure, with the aim to promote maximal preservation of blood supply. Postoperatively, careful monitoring of the flap is paramount. Recognizing the etiology of flap compromise is the first step in salvaging a compromised flap. Common causes of flap compromise include systemic patient factors such as concurrent drug or medication use (eg, anticoagulants), hypovolemia, hypotension, and anemia. In addition, poor surgical technique, cool ambient room temperature, kinking of the flap or pedicle, hematoma, and pressure on the flap may also lead to flap failure. Etiologies of flap failure can be broadly classified as tension-related, ischemic, hematologic, and infectious. In the case of ischemic arterial compromise, performing appropriate responses and rapidly restoring perfusion are essential. Commonly used interventions include adjusting the tension on the flap or pedicle and applying heat. HBOT is an adjunctive modality that may promote the healing of an ischemic flap. When venous compromise is identified, swift action to restore venous outflow is required. Additional measures such as administering leech therapy and possible vasodilators can help reduce venous congestion. Hematologic complications are most often related to drug-induced coagulopathy and poor intraoperative hemostasis. If a hematoma is identified postoperatively, it should typically be evacuated to relieve pressure on the flap. Although infection after facial reconstruction is uncommon, good aseptic techniques are important. Standard wound care and antimicrobial therapy may be used if an infection does develop.
KEY POINTS.
Good surgical technique, careful monitoring, and numerous strategies throughout the care pathway can be used to reduce the risk of skin flap failure and ensure favorable outcomes.
The intrinsic vascularity and perfusion of a flap are the most critical determinants of successful transfer. Anything that impacts perfusion will affect the overall outcome of the flap.
Recognizing the etiology of flap compromise is the first step in salvaging a failing flap. Careful monitoring with attention to color, capillary refill, bleeding, flap temperature, and firmness will help identify issues promptly.
Etiologies of flap failure can be broadly classified as venous, ischemic, tension-related, hematologic, and infectious. Common contributory causes of flap compromise include kinking of the flap or pedicle, hematoma, pressure or tension on the flap, systemic patient factors, poor surgical technique, or cool topical temperature.
Ischemic compromise can be either primarily arterial or venous in nature. Swift action to restore perfusion or venous outflow, such as relieving tension, repositioning the pedicle, or applying heat, may be helpful. For arterial comprise, hyperbaric oxygen therapy is a consideration. For venous congestion, leeches may be helpful. Vasodilating agents and aggressive wound care can be used as adjuncts in specific cases.
CLINICS CARE POINTS.
Although flap compromise can occur, good surgical technique, careful monitoring, and numerous strategies throughout the care pathway can be used to reduce risk and ensure positive outcomes.
The intrinsic vascularity and perfusion of a flap are the most critical factors for successful skin flap transfer. Anything that impacts perfusion will affect the overall outcome of the flap.
Avoiding complications begins in the preoperative period with a thorough patient history, including relevant medications and herbal supplement use, as well as physical examination. Every patient should be preoperatively optimized and comorbidities should be actively managed.
Recognizing the etiology of flap compromise is the first step in salvaging a failing flap. Careful monitoring with attention to flap color, temperature, capillary refill, bleeding, and firmness will help identify issues promptly.
In an arterial compromise, the flap may exhibit cool temperature, pale color, slow capillary refill (>4 s), slow or absent pinpoint bleeding, and/or low turgor. In a venous compromise, the flap may exhibit increased temperature, blue to purple cyanotic color, brisk capillary refill (>2 s), brisk dark pinpoint bleeding, and/or increased turgor (tense or swollen).
Etiologies of flap failure can be broadly classified as tension-related, ischemic, hematologic, and infectious. Common causes of flap compromise include systemic patient factors such as comorbidities that reduce perfusion or healing, concurrent drug or medication use, hypovolemia, hypotension, and anemia. In addition, poor surgical technique, cool ambient room temperature, kinking of the flap or pedicle, hematoma, and pressure on the flap can also lead to flap failure.
Good surgical technique is critical to reducing the risk of flap compromise. Fundamental principles include good aseptic and antiseptic techniques, optimal flap design, delicate tissue handling, judicious hemostasis, and tension-less closure
Ischemic compromise can be either arterial or venous in nature. Swift action to restore perfusion or venous outflow, such as relieving tension, revising the pedicle, or applying heat, may be helpful. Hyperbaric oxygen therapy, vasodilating agents, leech therapy, and aggressive wound care can be used as adjuncts in specific cases.
Footnotes
DISCLOSURE
The authors have no financial or commercial conflicts of interest to disclose. No funding sources were used to complete this work.
REFERENCES
- 1.Baker SR. Local Flaps in Facial Reconstruction. Netherlands: Elsevier; 2021. [Google Scholar]
- 2.Janis JE. Essentials of Plastic Surgery. United States. Thieme Medical Publishers, Incorporated; 2014. [Google Scholar]
- 3.Honrado CP, Murakami CS. Wound healing and physiology of skin flaps. Facial Plast Surg Clin North Am 2005;13(2):203–14. [DOI] [PubMed] [Google Scholar]
- 4.Taylor GI, Palmer JH. The vascular territories (angiosomes) of the body: experimental study and clinical applications. Br J Plast Surg 1987;40(2):113–41. [DOI] [PubMed] [Google Scholar]
- 5.McGregor IA, Morgan G. Axial and random pattern flaps. Br J Plast Surg 1973;26(3):202–13. [DOI] [PubMed] [Google Scholar]
- 6.Woodard CR. Complications in facial flap surgery. Facial Plast Surg Clin North Am 2013;21(4):599–604. [DOI] [PubMed] [Google Scholar]
- 7.Vural E, Key JM. Complications, salvage, and enhancement of local flaps in facial reconstruction. Otolaryngol Clin North Am 2001;34(4):739–51, vi. [DOI] [PubMed] [Google Scholar]
- 8.Brown DL, Brown DL, Borschel GH, et al. In: Michigan manual of plastic surgery, In: Lippincott manual series. 2nd edition. United States: Wolters Kluwer Health; 2014. p. 45–6. [Google Scholar]
- 9.Ondrey FG, Hom DB. Effects of Nutrition on Wound Healing. Otolaryngology-Head Neck Surg (Tokyo) 1994;110(6):557–9. [DOI] [PubMed] [Google Scholar]
- 10.Collins SC, Dufresne RG Jr. Dietary supplements in the setting of mohs surgery. Dermatol Surg 2002; 28(6):447–52. [DOI] [PubMed] [Google Scholar]
- 11.Salem RO, Laposata M. Effects of alcohol on hemostasis. Am J Clin Pathol 2005;123(Suppl):S96–105. [DOI] [PubMed] [Google Scholar]
- 12.Jensen JA, Goodson WH, Hopf HW, et al. Cigarette Smoking Decreases Tissue Oxygen. Arch Surg 1991;126(9):1131–4. [DOI] [PubMed] [Google Scholar]
- 13.Forrest CR, Pang CY, Lindsay WK. Dose and time effects of nicotine treatment on the capillary blood flow and viability of random pattern skin flaps in the rat. Br J Plast Surg 1987;40(3):295–9. [DOI] [PubMed] [Google Scholar]
- 14.Sorensen LT, Karlsmark T, Gottrup F. Abstinence from smoking reduces incisional wound infection: a randomized controlled trial. Ann Surg 2003;238(1):1–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Jorgensen LN, Kallehave F, Christensen E, et al. Less collagen production in smokers. Surgery 1998;123(4):450–5. [PubMed] [Google Scholar]
- 16.Rees TD, Liverett DM, Guy CL. The Effect of Cigarette Smoking on Skin-Flap Survival in the Face Lift Patient. Plast Reconstr Surg 1984;73(6):911–5. [DOI] [PubMed] [Google Scholar]
- 17.Craig S, Rees TD. The Effects of Smoking on Experimental Skin Flaps in Hamsters. Plast Reconstr Surg 1985;75(6):842–6. [DOI] [PubMed] [Google Scholar]
- 18.Kinsella JB, Rassekh CH, Wassmuth ZD, et al. Smoking increases facial skin flap complications. Ann Otol Rhinol Laryngol 1999;108(2):139–42. [DOI] [PubMed] [Google Scholar]
- 19.Goldminz D, Bennett RG. Cigarette smoking and flap and full-thickness graft necrosis. Arch Dermatol 1991;127(7):1012–5. [PubMed] [Google Scholar]
- 20.Møller AM, Villebro N, Pedersen T, et al. Effect of preoperative smoking intervention on postoperative complications: a randomised clinical trial. Lancet 2002;359(9301):114–7. [DOI] [PubMed] [Google Scholar]
- 21.Gottrup F. Trends in surgical wound healing. Scand J Surg 2008;97(3):220–5. ; discussion 225-6. [DOI] [PubMed] [Google Scholar]
- 22.Milton SH. Pedicled skin-flaps: the fallacy of the length: width ratio. Br J Surg 1970;57(7):502–8. [DOI] [PubMed] [Google Scholar]
- 23.Koopmann CF Jr. Cutaneous wound healing. An overview. Otolaryngol Clin North Am 1995;28(5):835–45. [PubMed] [Google Scholar]
- 24.Larrabee WF Jr, Holloway GA Jr, Sutton D. Wound tension and blood flow in skin flaps. Ann Otol Rhinol Laryngol 1984;93(2 Pt 1):112–5. [DOI] [PubMed] [Google Scholar]
- 25.Woodard CR, Most SP. Intraoperative angiography using laser-assisted indocyanine green imaging to map perfusion of forehead flaps. Arch Facial Plast Surg 2012;14(4):263–9. [DOI] [PubMed] [Google Scholar]
- 26.Utley DS, Koch RJ, Goode RL. The failing flap in facial plastic and reconstructive surgery: Role of the medicinal leech. Laryngoscope 1998;108(8):1129–35. [DOI] [PubMed] [Google Scholar]
- 27.Pang CY, Forrest CR, Morris SF. Pharmacological augmentation of skin flap viability: a hypothesis to mimic the surgical delay phenomenon or a wishful thought. Ann Plast Surg 1989;22(4):293–306. [DOI] [PubMed] [Google Scholar]
- 28.Barhoum F, Tschaikowsky K, Koch M, et al. Successful free flap salvage surgery with off-label use of Alteplase: A case report, review of the literature and our free flap salvage algorithm. Int J Surg Case Rep 2020;75:398–402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Carroll WR, Esclamado RM. Ischemia/reperfusion injury in microvascular surgery. Head Neck 2000;22(7):700–13. [DOI] [PubMed] [Google Scholar]
- 30.Awwad AM, White RJ, Webster MH, et al. The effect of temperature on blood flow in island and free skin flaps: an experimental study. Br J Plast Surg 1983;36(3):373–82. [DOI] [PubMed] [Google Scholar]
- 31.Tibbles PM, Edelsberg JS. Hyperbaric-Oxygen Therapy. N Engl J Med 1996;334(25):1642–8. [DOI] [PubMed] [Google Scholar]
- 32.Friedman HIF, Fitzmaurice M, Lefaivre JF, et al. An Evidence-Based Appraisal of the Use of Hyperbaric Oxygen on Flaps and Grafts. Plast Reconstr Surg 2006;117(7S):175S–90S. [DOI] [PubMed] [Google Scholar]
- 33.Zhou YY, Liu W, Yang YJ, et al. Use of hyperbaric oxygen on flaps and grafts in China: Analysis of studies in the past 20 years. Undersea Hyperbar M 2014;41(3):209–16. [PubMed] [Google Scholar]
- 34.Eclamado RM, Larrabee WF, Zel GE. Efficacy of steroids and hyperbaric oxygen on survival of dorsal skin flaps in rats. Otolaryngol Head Neck Surg 1990;102(1):41–4. [DOI] [PubMed] [Google Scholar]
- 35.Kuru B, Dinç S, Çamlibel M, et al. Efficacy of postoperative steroids on ischemic skin flap survival in rats. Eur J Plast Surg 2003;26(2):79–81. [Google Scholar]
- 36.Kerrigan CL, Wizman P, Hjortdal VE, et al. Global flap ischemia: a comparison of arterial versus venous etiology. Plast Reconstr Surg 1994;93(7):1485–95. ; discussion 1496-7. [PubMed] [Google Scholar]
- 37.Rohrich RJ, Cherry GW, Spira M. Enhancement of skin-flap survival using nitroglycerin ointment. Plast Reconstr Surg 1984;73(6):943–8. [DOI] [PubMed] [Google Scholar]
- 38.Welshhans JL, Hom DB. Are leeches effective in local/regional skin flap salvage? Laryngoscope 2016;126(6):1271–2. [DOI] [PubMed] [Google Scholar]
- 39.Dabb RW, Malone JM, Leverett LC. The use of medicinal leeches in the salvage of flaps with venous congestion. Ann Plast Surg 1992;29(3):250–6. [DOI] [PubMed] [Google Scholar]
- 40.Yousef A, Solomon I, Hom DB. Can Hyperbaric Oxygen Salvage a Compromised Local/Regional Skin Flap? Laryngoscope 2022. 10.1002/lary.30160. [DOI] [PubMed] [Google Scholar]
- 41.Nguyen MQ, Crosby MA, Skoracki RJ, et al. Outcomes of flap salvage with medicinal leech therapy. Microsurgery 2012;32(5):351–7. [DOI] [PubMed] [Google Scholar]
- 42.Cook-Norris RH, Michaels JD, Weaver AL, et al. Complications of cutaneous surgery in patients taking clopidogrel-containing anticoagulation. J Am Acad Dermatol 2011;65(3):584–91. [DOI] [PubMed] [Google Scholar]
- 43.Lewis KG, Dufresne RG Jr. A meta-analysis of complications attributed to anticoagulation among patients following cutaneous surgery. Dermatol Surg 2008;34(2):160–4. ; discussion 164-5. [DOI] [PubMed] [Google Scholar]
- 44.Diaz DD, Freeman SB, Wilson JF, et al. Hematoma-Induced Flap Necrosis and Free Radical Scavengers. Arch Otolaryngol Head Neck Surg 1992;118(5):516–8. [DOI] [PubMed] [Google Scholar]
- 45.Sylaidis P, Wood S, Murray DS. Postoperative infection following clean facial surgery. Ann Plast Surg 1997;39(4):342–6. [DOI] [PubMed] [Google Scholar]
- 46.Pearl RM, Arnstein D. A vascular approach to the prevention of infection. Ann Plast Surg 1985;14(5):443–50. [DOI] [PubMed] [Google Scholar]
- 47.Sasaki A, Fukuda O, Soeda S. Attempts to increase the surviving length in skin flaps by a moist environment. Plast Reconstr Surg 1979;64(4):526–31. [DOI] [PubMed] [Google Scholar]
- 48.Ledon JA, Savas J, Franca K, et al. Intralesional Treatment for Keloids and Hypertrophic Scars: A Review. Dermatol Surg 2013;39(12):1745–57. [DOI] [PubMed] [Google Scholar]
- 49.Sherris DA, Larrabee WF, Murakami CS. Management of Scar Contractures, Hypertrophic Scars, and Keloids. Otolaryng Clin N Am 1995;28(5):1057–68. [PubMed] [Google Scholar]
- 50.Hom DB. New Developments in Wound Healing Relevant to Facial Plastic Surgery. Arch Facial Plast Surg 2008;10(6):402–6. [DOI] [PubMed] [Google Scholar]
- 51.Hom DB, Goding G. Chapter 2: Skin Flap Physiology. In: Baker SR, editor. Local flaps in head and neck reconstruction. 4th edition. Mosby; 2022. [Google Scholar]


