Abstract
Chimeric antigen receptor (CAR) T-cell therapy achieves great success for hematological malignancies. However, clinical trials have revealed some limitations in both improving the efficacy and reducing the relapse, which calls for innovative strategies to engineer more powerful CAR-T cells. Promoting the formation of CAR clusters provides an alternative approach and potentially improves current CAR T-cell therapy against cancers. Here, we generated CARCys-T cells using a 4-1BB-derived hinge region including 11 cysteines residues. The cysteines in the hinge were found to facilitate CARCys clustering upon antigen stimulation and promote the antitumor activity of CAR-T cells. Compared with most conventionally used CAR-T cells with CD8α-derived hinge (CARconv-T cells), CARCys-T cells exhibited larger diameter of CAR clusters and enhanced antigen-specific tumor lysis both in vitro and in vivo. In addition, the CARCys-mediated enhancement could be applied to HER2, CD19 as well as GPC3-targeted CAR-T cells. More importantly, CARCys-T cells showed potent antitumor efficacy in clinically relevant patient-derived primary tumor cells and organoids. Thus, the novel hinge containing 11 cysteines provides a promising strategy to facilitate CAR clustering and maximize anti-tumor activity of CAR-T cells, which emphasizes the importance of CAR clustering to improve CAR T-cell therapy in the clinic.
Supplementary Information
The online version contains supplementary material available at 10.1007/s00262-022-03195-4.
Keywords: CAR clustering, Cysteines, Hinge domain, CAR-T cell, Cancer immunotherapy
Introduction
Chimeric antigen receptor (CAR) T-cell therapy has resulted in remarkable achievements in the treatment of cancers [1]. Yet, general unresponsiveness and frequent relapse of patients suffering from other cancers, especially solid tumors, emphasize the unmet need for improved CAR T-cell therapies [2, 3].
CARs are synthetic receptors composed of an extracellular domain, a transmembrane domain and an intracellular signal-transduction domain [4, 5]. The extracellular domain typically consists of an immunoglobulin single-chain fragment variable (scFv) for target antigen recognition, and a hinge region [6, 7]. And the intracellular domain involves CD3ζ alone or in combination with one or more costimulatory domains [8–10]. The modular design of CARs contributes to the rapid response against tumor cells, but it also causes a faster detachment rate from the target antigen, compared with natural T cell receptors (TCRs) [11]. Consequently, both effective antigen detection and sustained signal transduction are impaired in CAR-T cells [12, 13]. A comparative analysis has showed that TCR-mediated antigen sensitivity and signaling output were approximately 1000-fold higher than CAR-mediated ones [14], which could be closely correlated to the limited clinical efficacy of CAR-T cells. Thus, several studies have attempted to develop a high affinity of scFv to enhance the antigen detection capabilities of CARs, or define optimal intracellular signaling modules to strengthen the signal transduction of CAR-T cells [15–18]. Unfortunately, these approaches still remain insufficient for durable tumor eradication in CAR T-cell therapy, providing an impetus to search for alternative ways to optimize the CAR structure.
With respect to the structural basis of TCR triggering, TCR clustering is the key property for T cell activation, which determines the level of antigen discrimination and signal transduction [19–21]. Therefore, it is conceivable that promoting CAR clustering could be a promising strategy to improve the therapeutic efficacy of CAR T-cell therapy. It is known that monomers can interact with each other through two cysteine residues forming a pair of disulfide bonds [22, 23]. Hence, this property could be employed to facilitate CAR clustering formation. The hinge seems to be the best region to modify with cysteines, considering the intrinsic function of antigen recognition or signal transduction would be impaired if introducing cysteines into scFv or intracellular domain, respectively. Also, the routinely used hinge domains in CARs are derived from the immunoglobulin superfamily including CD8α, CD28 and IgG with two or less cysteines [24, 25], which might be unfavorable for CAR clustering via disulfide bond formation.
Here, we designed a CARCys with 11 cysteines in the hinge derived from the human 4-1BB molecule, which belongs to the tumor necrosis factor receptor (TNFR) superfamily, and comprises an elongated repeating cysteine-rich motif [26]. As expected, cysteines in the hinge facilitated CARCys cluster formation, leading to a 1.5-fold larger diameter than CARs with the most extensively used CD8α hinge (CARconv) upon antigen stimulation. More importantly, compared with CARconv-T cells, CARCys-T cells displayed stronger tumor antigen-specific activation, cytokine release and cytotoxicity not only in vitro but also in tumor-bearing mice as well as in patient-derived tumor cells or organoids (PDO). Thus, this research provides an attractive strategy for optimizing CAR T-cell therapy by facilitating CAR clustering.
Materials and methods
Generation of CAR constructs and mRNA transcription
The anti-HER2 scFv was derived from the humanized 4D5 antibody, the anti-GPC3 scFv was from the GC33, and the anti-CD19 scFv was from the FMC63. Hinge domains were derived from human IgG4 (GenBank: AAC82527.1), CD28 (NP_006130), CD8α (NP_001759), or 4-1BB (XP_011539688). The resulting gene was cloned into an expression vector (pGEM4Z/GFP/A64; kindly provided by Dr. Xiao He, University of Utah, USA) by replacing the green fluorescent protein gene. The T7 mScript™ standard mRNA production system (CELLSCRIPT) was used to generate in vitro transcription mRNA.
Cell culture and CAR T-cell generation
Expanded human T cells were mixed with transcribed mRNA using an ECM830 Electro Square Wave Operator (Harvard Apparatus BTX; Harvard Apparatus) for electroporation as previously described [27]. CAR expression of these electroporated T cells was detected by flow cytometry. Cell number, viability, and cell size of these T cells were determined using a Countess™ automated cell counter (Invitrogen).
Flow cytometry analysis
These antibodies used for flow cytometry analysis are listed in Table S1. HER2-CAR expression on T cells was assessed by staining with Alexa Fluor 647-labeled polyclonal anti-human IgG F(ab’)2 specific antibody, and the Alexa Fluor 647-labeled anti-mouse IgG F(ab’)2 specific antibody was applied for GPC3- and CD19-CAR detection. FACS analysis was performed on a BD FACSCanto™ II (BD Biosciences), and data were analyzed using FlowJo (v.10.0).
Super-resolution stochastic optical reconstruction microscopy (STORM) and clusters analysis
CAR-T cells were stimulated with 2 μg/mL recombinant human HER2 protein (10,004-H02H-B, Sino Biological Inc) for 30 min at 37 °C. Cells were fixed with 4% paraformaldehyde (PFA) for 10 min at room temperature and then stained with Alexa Fluor 647-labeled polyclonal anti-human IgG F(ab’)2 specific antibody for 30 min. Antibody-stained cells were plated on a Poly-D-lysine (Gibco) pre-coated glass bottom cell culture dish and left to dry at 37 °C in a constant temperature oven, protected from light. Next, the glass bottom cell culture dish was sealed after adding imaging buffer for STORM (Nikon). The tables containing x × y co-ordinates of each molecule derived from STORM image were generated and used for subsequent cluster analysis. For each cell, a region of interest (5 × 5 μm2) was selected for identifying the clustered distribution of CAR molecules, and detecting individual cluster parameters based on density-based spatial clustering of application with noise (DBSCAN) in MATLAB [28].
RNA-seq and data analysis
CAR-T cells were sorted with a purity of > 95% using a Moflo XDP (Beckman Coulter) and stimulated with antigen for 10 h, followed by mRNA sequencing. The RNA sequencing data are publicly available in National Omics Data Encyclopedia at OEP002935. All statistical analyses were performed within the R/Bioconductor statistical environment (https://www.r-project.org/).
CAR T-cell in vitro function assessment
The degranulation (CD107a) and cytokine release of CAR-T cells were measured after co-cultured with tumor cells as previously described [27]. For cytotoxicity assay, N87 or Huh7 tumor cells used as target cells were plated at 5 × 104 cells/well in a 96-well flat-bottom plate treated with Poly-D-lysine and incubated at 37 °C overnight for well adherence. The target cells were then co-cultured with CAR-T cells at indicated ratios for 6, 12, or 48 h. Then, 10 μL Cell Counting Kit-8 (CCK8; Dojindo) was added after removing dead tumor cells and resuspension of the CAR-T cells by washing with warm PBS for three times. The cytotoxicity of CAR-T cells was calculated by the target cell viability according to the optical density at 450 nm.
Mouse xenograft studies
N87 tumor cells (2 × 106 cells/100 μL PBS) were implanted subcutaneously into anesthetized nude mice. Nine days after tumor implantation, mice were administered cyclophosphamide (100 mg/kg) intravenously (i.v.). Three days later, CAR-T cells (5 × 106) were adoptively transferred i.v. into mice every 4 days for a total five injections. Tumor size was measured using digital calipers and quantified using the following formula: 1/2 × L (length) × W2 (width). The relative tumor volume (RTV) was calculated as the ratio of the volume at the beginning of treatment to that at the last day. After the final treatment, tumor samples obtained from the mice were dissociated into single cells for FACS analysis or immersed in Tissue-Tek O.C.T. Compound for fluorescence microscopy imaging. Images were captured with Olympus FV3000 confocal microscopy at an original magnification of × 20, and analyzed by Fiji software (ImageJ).
Human HCC cells dissociation and PDO establishment
Human HCC samples were dissociated into single-cell suspensions using tumor dissociation kit (Miltenyi Biotec) combined with the gentleMACS™ Dissociators (Miltenyi Biotec) according to the manufacturer’s instructions. Single tumor cell was collected from HCC patient pleural effusion for PDO establishment according to previous studies [29, 30].
Statistical analysis
Data are presented as the mean ± SEM. Analyses were conducted with GraphPad Prism software (v.7.0). The data were analyzed with Student’s t test and one- or two-way ANOVA. The statistical significance levels were set as *P < 0.05, **P < 0.01, and ***P < 0.001.
Results
Cysteines in the hinge facilitate antigen-triggered CAR cluster formation and promote CAR T-cell activity
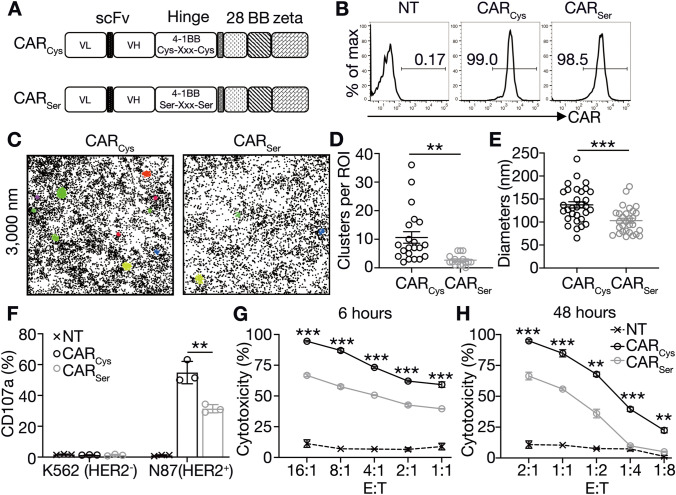
In an attempt to introduce cysteines into CARs, we first identified an extracellular region with 11 cysteines from human 4-1BB as a hinge domain in a third-generation HER2-CAR structure (CARCys, Fig. 1A, upper). Also, we mutated all 11 cysteines in CARCys into serines as a control (CARSer, Fig. 1A, lower). These HER2-CAR-encoding mRNAs were generated based on in vitro transcription system and then introduced into expanded human T cells via electroporation to manufacture mRNA-engineered T cells with transient CAR expression to improve safety in future clinical applications [27]. According to the flow cytometry analysis, the surface expression levels of both CARCys and CARSer exceeded 95% on T cells with non-transduced (NT) T cells serving as the negative control (Fig. 1B). To explore the contribution of cysteine-hinge to CAR clustering formation, STORM was applied to scan the CARs distribution on the T cell plasma membrane at the molecular level and DBSCAN was utilized to analyze the CAR clustering formation (Fig. S1A). After HER2 antigen stimulation, the number of HER2-CARCys clusters per ROI on T cells was fourfold increase compared to HER2-CARSer clusters (10.62 vs. 2.68, respectively), while also the diameter of HER2-CARCys clusters was 1.3-fold larger than those of HER2-CARSer clusters (137.33 nm vs. 102.99 nm, respectively, Fig. 1C–E). But without the HER2 antigen stimulation, the average number of HER2-CAR clusters was just around 2.40 ± 0.63 and 1.75 ± 0.41 per ROI on T cells in both CARCys and CARSer groups, respectively (Fig. S1B). Meanwhile, the expression levels of activation markers (CD25, CD69) in CARCys group were only slightly up-regulated than those in NT group, but similar to those in CARSer group (Fig. S1C). These data indicated that cysteines-mediated CAR clusters did not increase spontaneous CAR signaling. Furthermore, to investigate the role of CAR clustering in CAR T-cell function, we co-cultured NT-, CARCys-, or CARSer-T cells with either HER2+ N87 or HER2− K562 tumor cells. When exposed to HER2+ N87, CARCys-T cells had 1.7-fold upregulated CD107a translocation (cytotoxicity marker), compared with CARSer-T cells (Fig. 1F). This resulted in a superior in vitro cytotoxic activity of the CARCys-T cells towards HER2+ N87 tumor cells relative to CARSer-T cells after 6-h or 48-h co-cultures at all ratios of E: T (Fig. 1G, H). Overall, these observations suggest the cysteines in hinge facilitate the formation of antigen-triggered CAR clusters, and enhance the tumor-specific activity of CAR-T cells.
Fig. 1.
Cysteines in the hinge facilitate antigen-triggered CAR cluster formation and promote CAR T-cell activity A Gene schematics of CARCys and CARSer targeting HER2. B Human T cells derived from healthy donors were transduced with or without HER2-CARCys or HER2-CARSer mRNA to generate NT or HER2-CAR-T cells. Twelve hours post-transduction, the CAR expression on these T cells was detected by flow cytometry. Data were representative of at least three independent experiments. C–E CARCys and CARSer distribution on these T cells was observed using STORM system after stimulation with HER2 antigen for 30 min. The tables containing x × y co-ordinates of each CAR molecule were obtained from STORM, and DBSCAN implemented in MATLAB was used for CAR clusters identification. For each T cell, a 5 × 5 μm2 ROI in cell center was selected for further analysis of cluster number and diameter by DBSCAN. C The representative cluster maps of CARCys and CARser on primary human T cells. D The number of CAR clusters per ROI. E The diameters of CAR clusters. Data shown as mean ± SEM were from 13 to 16 cells of healthy donors. F CARCys and CARSer-T cells were co-cultured with HER2+N87 or HER2−K562 tumor cells. Four hours later, CD107a translocation of CAR-T cells was detected by flow cytometry. Data shown as mean ± SEM were pooled from three healthy donors. G, H CARCys and CARSer-T cells were co-cultured with HER2+N87 at indicated E: T ratios for 6 or 48 h. The cytotoxicity of CAR-T cells was calculated by the target cells’ viability using CCK8. Data shown as mean ± SEM were from one of two independent experiments using two healthy donors. P values were determined by unpaired Student’s t test in (D, E), and two-way ANOVA in F, G, and H. **P < 0.01, ***P < 0.001
CARCys-T cells exhibit increased antigen-specific activity compared to CARconv-T cells in vitro
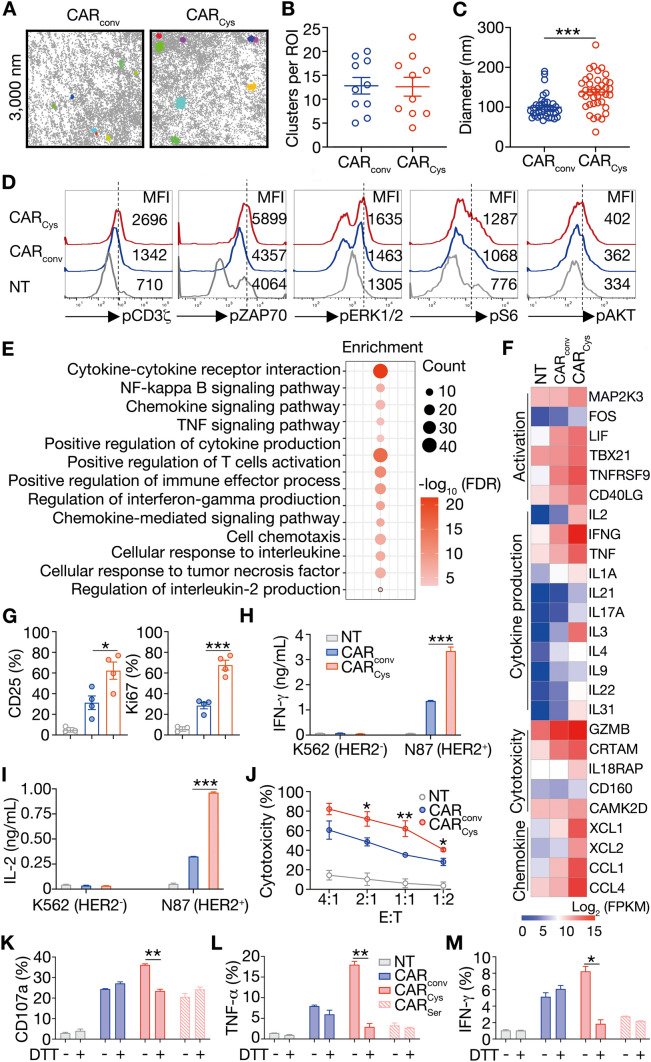
The hinge domain in CARs is usually derived from human CD28, IgG4, or CD8α, among which CD8α hinge is the most routinely used in CARs [31–33]. Thus, we constructed HER2-CARs containing the same scFv, transmembrane, and intracellular domains to CARCys, but with CD8α-derived hinge as a comparison (CARconv, also called CAR8α). According to the STORM and DBSCAN analysis, the number of CARCys and CARconv clusters was similar, 12.60 ± 1.97 and 12.80 ± 1.74 per ROI, respectively, while the diameter of CARCys clusters (~ 140 nm) was much larger than that of CARconv clusters (~ 100 nm) (Fig. 2A–C). To explore whether large-sized CARCys clustering could recruit qualitatively proximal signaling, we compared its signaling strength with CARconv. CARCys-T cells exhibited the stronger phosphorylation of CD3ζ, zeta-chain associated protein kinase (ZAP70), extracellular signal-regulated kinase 1/2 (ERK1/2), S6 kinase and AKT kinase than CARconv-T cells (Fig. 2D). To further evaluate the phenotypical and functional difference between CARconv-T cells and CARCys-T cells, we compared the genome-wide transcriptional profiles of these CAR-T cells after 10-h HER2 antigen stimulation. CARCys-T cells displayed heterogeneous gene expression patterns and significant upregulation of the gene sets associated with T-cell activation (TNFRSF9, TBX21, CD40LG), cytokine production (IFNG, TNF, IL2), cytotoxicity (GZMB, CRTAM, CAMK2D) and chemokine (CCL4, CCL1)-mediated pathways relative to CARconv-T cells (Figs. 2E, F and S2A, B). To confirm the above mRNA sequencing results, we developed in vitro co-culture systems to measure CAR T-cell activity and functionality. After 12-h co-culture with HER2+ N87 cells, the expression level of CD25 (activation) was twofold higher, and the expression level of Ki67 (proliferation) was 2.4-fold higher in CARCys-T cells than CARconv-T cells when co-cultured with N87 for 24 h (Fig. 2G). After 24-h co-culture, the IFN-γ and IL-2 release of CARCys-T cells (3343.0 ± 154.7 pg/mL and 963.3 ± 6.1 pg/mL, respectively) were significantly increased in comparing with CARconv-T cells (1360.0 ± 18.6 pg/mL and 325.5 ± 0.6 pg/mL, respectively) (Fig. 2H, I). Accordingly, the cytotoxicity of CARCys-T cells was greatly enhanced at the E: T ratios of 2: 1, 1: 1, and 1: 2 during a 12-h cytotoxicity assay relative to CARconv-T cells (Fig. 2J). Subsequently, to assert the importance of cysteine-cysteine contact in CARCys T-cell function, dithiothreitol (DTT) was used to break the contact and CAR T-cell function was re-examined. As expected, the CD107a translocation and cytokine production (TNF-α and IFN-γ) were severely impaired in CARCys-T cells but not in CARconv- or CARSer-T cells after DTT treatment (Fig. 2K–M). In addition, we constructed HER2-CARs with a CD28 (CAR28), or IgG4 (CARIgG4) derived hinge region, all of those contained two cysteines but different amino acids composition (Fig. S3A). There was no significant difference among the various CAR-T cells in terms of CAR expression level, cell viability and cell size (Fig. S3B, C). All the four CAR-T cells had almost the same ability of IFN-γ secretion upon PMA stimulation (Fig. S3D), indicating no impairment of T-cell function after CAR electroporation. When exposed to HER2 antigen, CAR8α-T cells had similar levels in IFN-γ secretion and CD107a translocation to CAR28-T cells, but showed a slight increase compared to CARIgG4-T cells (Fig. S3D, E). However, when compared with CARCys-T cells, CAR28-T cells, CARIgG4-T cells and CAR8α-T cells had significantly down-regulated levels of IFN-γ secretion and decreased frequency of CD107a translocation (Fig. S3D, E). Meanwhile, we mutated 0, 1, 3, 5, 7, 9, and all the 11 cysteines in 4-1BB hinge to generate variant CARs: WT (11 Cys, namely CARCys), MT (10 Cys), MT (8 Cys), MT (6 Cys), MT (4 Cys), MT (2 Cys), and MT (0 Cys, namely CARSer). CD107a translocation analysis of these CARs revealed that the general trend was less cysteines with less frequency of CD107a translocation in all the groups, although CD107a translocation levels in MT (8 Cys) groups were lower than MT (6 Cys) group, implying a complicated mechanism in cysteine-cysteine contacts (Fig. S4). These above data suggested an important role of cysteines presented in the hinge in CAR T-cell function. Together, these transcriptional and functional results demonstrate that CARCys-T cells exhibit increased antigen-specific activity than CARconv-T cells.
Fig. 2.
CARCys-T cells exhibit increased antigen-specific activity compared to CARconv-T cells in vitro Human T cells derived from healthy donors were transduced with or without HER2-CARconv or HER2-CARCys mRNA to generate NT or HER2-CAR-T cells. A These CAR-T cells were stimulated with HER2 antigen for 30 min, and identified the antigen-triggered CAR clusters by STROM and DBSCAN analysis. B, C The number of CAR clusters per ROI (5 × 5 mm2) and the diameter of CAR clusters were calculated by DBSCAN analysis. Data shown as mean ± SEM were from 10 CAR-T cells of healthy donors. D Twelve hours post-transduction, NT- or CAR-T cells were cultured in Opti-MEM for 6 h to reduce background. These T cells were incubated with biotinylated HER2 protein (2.5 μg/mL) for 30 min at 4 °C and washed twice with cold PBS. Then, these T cells were cross-linked with streptavidin (5 μg/mL) at 37 °C for 30 s to measure the phosphorylation of CD3ζ and ZAP70, and cross-linked for 2 min to detect the phosphorylation of ERK1/2, S6 and AKT. E The enriched gene sets of CARconv-T cell comparing with CARCys-T cells after 10-h antigen stimulation. F The heat map of selected differentially expressed genes related to T cell activation, cytokine production, cytotoxicity, and chemokine signaling. Differentially expressed genes were selected with an adjusted P value (false discovery rate, FDR) < 0.05 and absolute fold change ≥ 2. G–J NT, CARconv, or CARCys-T cells were co-cultured with HER2−K562 or HER2+N87. (G) CD25 and Ki67 expressions of T cells were detected by flow cytometry 24 and 48 h post-co-culture, respectively. Data shown as mean ± SEM were pooled from four healthy donors. H, I IFN-γ and IL-2 production of T cells were measured by ELISA 24 h post-co-culture. Data shown as mean ± SEM were from one of three independent experiments using three healthy donors. J Cytotoxic activity of T cells was measured by performing CCK8 assay 12 h post-co-culture at indicated E: T ratios; data shown as mean ± SEM were from one of two independent experiments using two healthy donors. K–M Human T cells were transduced with HER2-CARconv, HER2-CARCys or HER2-CARSer mRNA to generate CAR-T cells. Twelve hours post-transduction, these CAR-T cells were treated with 10 mM/mL DTT and co-cultured with HER2+N87 tumor cells at the ratio of 1: 2. Six hours later, the CD107a translocation (K), the TNF-α (L) and IFN-γ (M) production were detected by flow cytometry. Data shown as mean ± SEM were from two independent experiments. P values were determined by unpaired Student’s t-test in (C), one-way ANOVA in (G), and two-way ANOVA in (H–M). *P < 0.05, **P < 0.01, and ***P < 0.001
CARCys-T cells display enhanced anti-tumor efficacy compared to CARconv-T cells in tumor-bearing mice
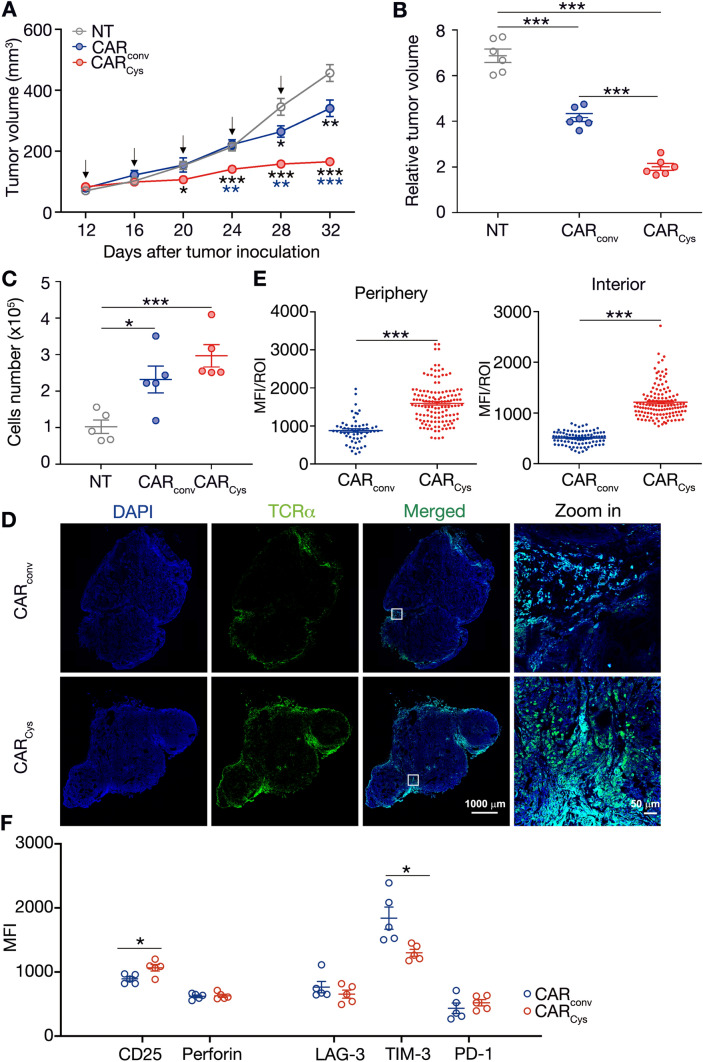
To investigate whether the increased antigen-specific activity of CARCys-T cells could be replicated in vivo, we established a tumor-bearing mouse model in immunocompromised nude mice by subcutaneous implantation of HER2+ N87 tumor cells. We detected CAR expression levels of both CARconv-T cells and CARCys -T cells in vitro each day for 5 days so as to choose a suitable therapeutic cycle of CAR-T cells in vivo. As shown in Fig. S5A and B, both CARconv and CARCys expression levels were decreased greatly from the fourth day; hence, CAR-T cells were re-administered to the mice every four days. When the tumor volumes were around 50 mm3, 9 days post-N87 cells transplantation, mice received an injection of cyclophosphamide via tail vein. Three days later, NT-, CARconv- or CARCys-T cells were adoptively transferred via the tail vein every four days for a total of five times (Fig. 3A). Tumor volumes were measured every four days, and the tumors were collected on day 32. As compared to NT-T cells, CARconv-T cells only modestly delayed tumor growth and decreased tumor volumes, whereas CARCys-T cells markedly inhibited tumor growth and reduced tumor volumes (Fig. 3A, B). Furthermore, the number of adoptively transferred T cells at the tumor site was around 1.3-fold in CARCys group than that in CARconv group (Fig. 3C). According to results of the immunofluorescent staining, these adoptive T cells from CARCys or CARconv groups could be found at the surrounding area (0–200 μm away from the tumor margin) and also the tumor center (Fig. 3D). And the mean fluorescence intensity (MFI) of adoptive T cells in CARCys group was much higher than that in CARconv group both at the surrounding area and the tumor center (Fig. 3E). In addition, these adoptively transferred tumor-infiltrating T cells in CARCys group exhibited a higher level of CD25 (activation marker), lower level of TIM-3 (exhaustion marker), and slightly increased frequency in the CCR7 + CD45RA long-lived central memory subset, compared to those in CARconv group (Fig. 3F and S6A-C). Collectively, CARCys-T cells display enhanced anti-tumor efficacy relative to CARconv-T cells in tumor-bearing mice.
Fig. 3.
CARCys-T cells display enhanced anti-tumor efficacy relative to CARconv-T cells in tumor-bearing mice. Mice engrafted with 2 × 106 N87 tumor cells and treated with 5 doses of NT, CARconv, or CARCys-T cells (5 × 106) every four days from day 12. A Tumor growth of N87-bearing mice was measured every 4 days, and the arrow represented a single injection of CAR-T cells. The blue asterisk represented analysis between CARCys and CARconv, and the black one indicated the comparison of CARCys or CARconv with NT. B RTV was calculated by the ratio of the final tumor volume (day 32) to the baseline (day 12). C The number of adoptive tumor-infiltrating T cells was measured using counting beads (APC-labeled) by flow cytometry. D Tumor slices were observed under FV3000 at an original magnification of × 20 (scale bar = 1000 μm). E The images were analyzed using Fiji software. F The expression of functional (CD25, Perforin) and exhaustion markers (LAG3, TIM-3, and PD-1) on adoptive tumor-infiltrating T cells were detected by flow cytometry. Data shown as mean ± SEM were pooled from five or six mice per group. P values were determined by unpaired Student’s t test in (E, F), one-way ANOVA in (B, C) and two-way ANOVA in (A). *P < 0.05, **P < 0.01, and ***P < 0.001
CARCys-T cells with different CAR framework exhibit stronger antigen-specific activity than CARconv-T cells
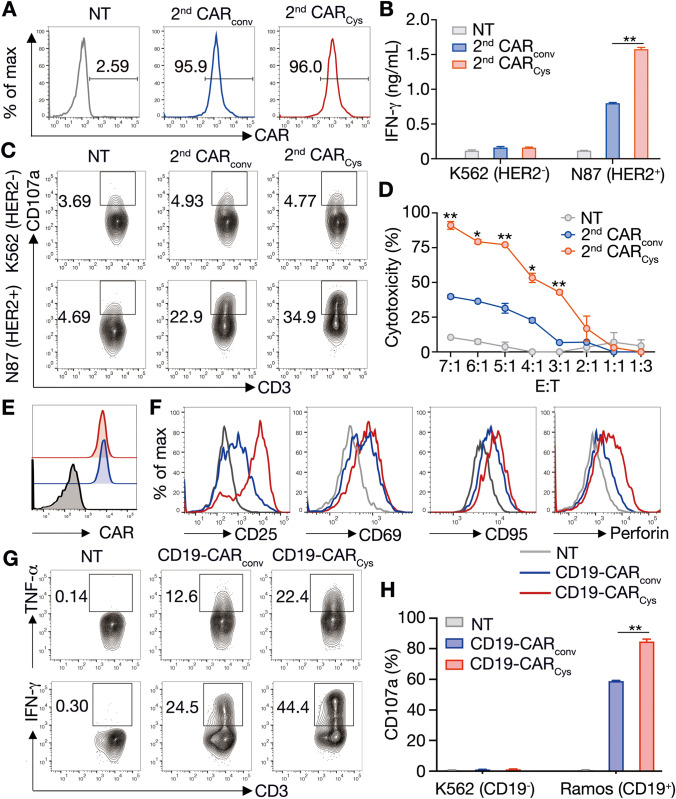
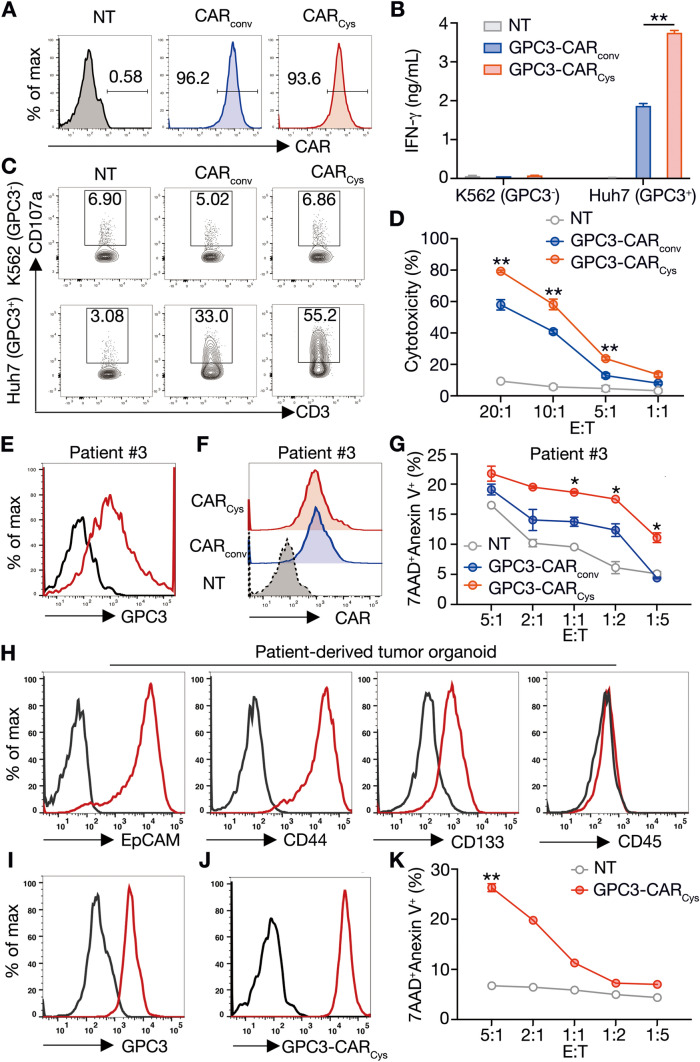
To explore whether the enhanced anti-tumor efficacy of CARCys-T cells could be extrapolated to different CAR frameworks, we constructed second-generation HER2-CARCys containing one 4-1BB costimulatory domain. Both second-generation CARconv and CARCys mRNAs were transduced into primary T cells, and the CAR expressions in CARconv and CARCys-T cells were similar, about 96% (Fig. 4A). In accordance to the third-generation HER2-CARCys-T cells, second-generation CARCys-T cells exhibited increased IFN-γ production and CD107a translocation, when exposed to HER2+N87 tumor cells (Fig. 4B, C). Meanwhile, second-generation CARCys-T cells had stronger cytotoxicity than CARconv-T cells at the ratios from 3: 1 to 7: 1 (Fig. 4D). To further investigate whether the boosted function of CARCys-T cells could be extended to other antigen-targeted CARCys-T cells, we constructed CD19-CARCys and GPC3-CARCys. These CARCys mRNAs were transduced into primary T cells from healthy donors to generate CAR-T cells at similar transduction rate (Figs. 4E and 5A). After co-culture with CD19+ Ramos tumor cells, CD19-CARCys-T cells exhibited great up-regulation of CD25 and perforin, nearly twofold increased IFN-γ and TNF-α production, and significantly enhanced CD107a translocation compared to CD19-CARconv-T cells (Fig. 4F–H). Similarly, when exposed to GPC3+ Huh7 cells, GPC3-CARCys-T cells displayed remarkably up-regulated CD107a translocation, increased IFN-γ production, and enhanced cytotoxicity at E: T ratios from 5: 1 to 20: 1, compared with GPC3-CARconv-T cells (Fig. 5B–D). Therefore, the CARCys-mediated enhancement of anti-tumor activity could be applied in the second-generation CAR-T cells as well as CD19 and GPC3-targeted CAR-T cells.
Fig. 4.
CARCys-T cells with different CAR framework exhibit stronger antigen-specific activity than CARconv-T cells. Human T cells derived from healthy donors were transduced with or without second-generation HER2-CARconv or HER2-CARCys with a 4-1BB costimulatory domain, or third-generation CD19-CARconv or CD19-CARCys mRNA to generate NT- or CAR-T cells. A Twelve hours post-transduction, HER2-CAR expression levels of NT-, HER2-CARconv-, or HER2-CARCys-T cells were detected by flow cytometry. B NT-, HER2-CARconv- or HER2-CARCys-T cells were co-cultured with HER2−K562 or HER2+N87. Twenty-four hours post-co-culture, IFN-γ production of these T cells were measured by ELISA. Data shown as mean ± SEM were from one of two independent experiments using two healthy donors. C CD107a translocation of NT-, HER2-CARconv-, or HER2-CARCys-T cells was detected by flow cytometry four hours post-co-culture. D Twelve hours post-co-culture, the cytotoxicity of NT-, HER2-CARconv-, or HER2-CARCys-T cells was analyzed by performing CCK8 assay after at indicated E: T ratios. Data shown as mean ± SEM were from one of two independent experiments using two healthy donors. E Twelve hours post-transduction, CD19-CAR expression levels of NT-, CD19-CARconv-, or CD19-CARCys-T cells were detected by flow cytometry. F, G NT-, CD19-CARconv-, or CD19-CARCys-T cells were co-cultured with CD19+Ramos for 12 h. CD25, CD69, CD95, and Perforin expression levels and cytokine production (TNF-α, IFN-γ) of these T cells were detected by flow cytometry. H NT-, CD19-CARconv-, or CD19-CARCys-T cells were co-cultured with CD19− K562 or CD19+ Ramos. CD107a translocation of NT, CD19-CARconv, or CD19-CARCys-T cells was detected by flow cytometry four hours post-co-culture. Data shown as mean ± SEM were pooled from two healthy donors. P values were determined by two-way ANOVA in (B, D, H). *P < 0.05, and **P < 0.01
Fig. 5.
CARCys-T cells have potent antitumor activity in HCC patient-derived primary tumor cells and organoids. A Human T cells derived from healthy donors were transduced with or without GPC3-CARconv or GPC3-CARCys mRNA to generate NT or GPC3-CAR-T cells. Twelve hours post-transduction, GPC3-CAR expression levels of these T cells were detected by flow cytometry. B NT-, GPC3-CARconv-, or GPC3-CARCys-T cells were co-cultured with GPC3− K562 or GPC3+ Huh7. Twenty-four hours post-co-culture, IFN-γ production of these T cells were measured by ELISA. Data shown as mean ± SEM were from one of two independent experiments using two healthy donors. C CD107a translocation of NT-, GPC3-CARconv-, or GPC3-CARCys-T cells was detected by flow cytometry four hours post-co-culture. D Six hours post-co-culture, the cytotoxicity of NT-, GPC3-CARconv-, or GPC3-CARCys-T cells was analyzed by performing CCK8 assay at indicated E: T ratios. Data shown as mean ± SEM were from one of two independent experiments using two healthy donors. E GPC3 expression level of primary tumor cells from HCC patient #3 was detected by flow cytometry. F Human T cells derived from HCC patient #3 were transduced with or without GPC3-CARconv or GPC3-CARCys mRNA to generate NT- or GPC3-CAR-T cells. Twelve hours post-transduction, GPC3-CAR expression levels of these T cells were detected by flow cytometry. G HCC patient #3-derived NT-, GPC3-CARconv-, or GPC3-CARCys-T cells were co-cultured with autologous primary tumor cells. Six hours post-co-culture, the apoptosis (Annexin V+ 7-AAD+) of primary tumor cells was analyzed by flow cytometry. H, I Primary tumor cells from HCC patient #5 were collected from pleural effusion and cultured in vitro to establish PDO. Thirty-three days later, EpCAM, CD44, CD133 and GPC3 expressions on PDO were detected by flow cytometry. J Human T cells derived from HCC patient #5 were transduced with or without GPC3-CARCys mRNA to generate NT- or GPC3-CARCys-T cells. Twelve hours post-transduction, GPC3-CAR expression levels of these T cells were detected by flow cytometry. K HCC patient #5-derived NT-, or GPC3-CARCys-T cells were co-cultured with autologous PDO. Six hours post-co-culture, the apoptosis (Annexin V+ 7-AAD+) of PDO was analyzed by flow cytometry. Data are shown as mean ± SEM; P values were determined by two-way ANOVA in (B, D, G, K). *P < 0.05, and **P < 0.01
CARCys-T cells have potent antitumor activity in HCC patient-derived primary tumor cells and organoids
To further support the clinical use of GPC3-CARCys-T cells in patients with HCC, we isolated primary tumor cells and peripheral blood mononuclear cells from four HCC patients (#1–#4). According to the flow cytometry analysis, only the primary tumor cells from the #3 patient expressed high level of GPC3 compared with those from #1, #2 and #4 patients (Figs. 5E and S7A). Then, T cells were purified and expanded from matched patients’ peripheral blood mononuclear cells, and transduced by CARconv or CARCys mRNAs to generate GPC3-CAR-T cells (Figs. 5F and S7B). After that, CAR-T cells were co-cultured with their autologous primary tumor cells. CD107a translocation of CARconv- and CARCys-T cells derived from #1, #2 and #4 patients was slightly up-regulated compared with NT-T cells (Fig. S7C), indicating both CARCys- and CARconv-T cells were unable to detect and eliminate primary tumor cells with low expression of GPC3. However, the cytotoxicity of CARCys-T cells derived from #3 patient was greatly enhanced at the ratios from 1: 1 to 1: 5, compared with CARconv-T cells (Fig. 5G), suggesting improved GPC3-specific activity of GPC3-CARCys-T cells. To evaluate GPC3-CARCys-T cells against limited surgical tumor samples, we successfully established a HCC PDO from patient’s pleural effusion (Fig. S8A, B), which is a clinically relevant model for predicting therapy response [30]. To verify whether PDOs could retain the phenotypes of the original HCCs, we detected the expression of tumor antigen (GPC3) and stemness markers (EpCAM, CD44 and CD133) on the original HCC tissue slides by immunofluorescence histochemistry. As shown in Fig. S8C and D, the original HCC expressed GPC3 and various stem cell surface markers, which all had been retained in PDOs (Fig. 5H and I). Then, the PDO was isolated from Geltrex and digested into single cells for co-culturing with autologous CARCys-T cells (Fig. 5J). After co-cultured with PDO, GPC3-CARCys-T cells induced around 28% of 7-AAD+ Annexin V+ PDO at the ratio of 5: 1, compared with the control group (Fig. 5K). Taken together, CARCys-T cells have potent anti-tumor activity in HCC patient-derived primary tumor cells and organoids, pointing towards a potential application of CARCys-T cells in the clinic.
Discussion
CAR T-cell therapy has yielded remarkable clinical responses in patients with hematological malignancies [34]. However, the therapeutic efficacy of CAR-T cell in both non-solid and solid tumors still requires improvement particularly with regard to the clinical response rate. In this study, we developed a hinge containing 11 cysteines to facilitate CAR clustering upon antigen stimulation. More importantly, we found that the tumor-specific activity of CARCys-T cells was significantly enhanced compared with CARconv-T cells both in vitro and in vivo. Therefore, our study uncovers the determinant role of CAR clustering in CAR T-cell function, which also provides a promising strategy for the improvement of CAR T-cell efficacy in the clinic.
The formation of TCR clusters upon antigen recognition has been proved to be crucial in amplifying initial T cell activation signals [35, 36]. A previous study revealed that TCRs were organized into clusters with a diameter of ~ 185 nm in antigen-stimulated Jurkat-ILA1 cells, which impacted the signaling competency of T cell activation [37]. In addition, a study based on a synthetic T cell signaling system demonstrated that TCR clustering increased the probability of ZAP70 recruitment, which supported the formation of clusters in T-cell signaling [38]. Although CARs also can rapidly organize into clusters when encountering antigen, these clusters disassociate quickly from the target antigen in comparison to the TCR complex [11]. Thus far, there has been limited study to investigate the parameters of CAR clusters. According to our data in Fig. 2C, the diameter of CARconv clusters is small relative to that of TCR clusters (~ 100 nm vs. ~ 185 nm, respectively) [37]. The small size of CAR clusters could be the reason of fast disassociation of CARs from antigen, which would severely impair the signal transduction and the subsequent function of CAR-T cells. Therefore, we investigated a new 4-1BB-derived hinge (CARCys) containing 11 cysteine residues to augment the formation of CAR clusters,. As expected, we observed that the diameter of CARCys clusters was increased into ~ 140 nm as a consequence of cysteines-mediated cross-linking. According to our data in Fig. 2D, it could be speculated that the larger CARCys clusters would recruit robust intracellular secondary messenger molecules by ζ in CARs for phosphorylation, thus leading to sufficient signaling input. In addition, the magnitude of CAR signaling would affect T cell differentiation as it was observed in Figs. 3F and S6C. It is therefore conceivable that CAR T-cell function can be strengthened by the introduction of cysteine-rich proteins like TNFR superfamily members, or the selection of shorter scFv linker [39] to facilitate CAR clustering.
The hinge domain is located between the scFv and transmembrane domains, and was thought to merely link the two regions. Until recently, several studies have proved that the length and composition of hinge domain would affect CAR T-cell cytotoxicity. CD19-CAR-T cells no matter with a shorter hinge domain derived from CD8α or a longer hinge domain derived from the IgG4-Fc spacers exhibited potent antitumor activity in mice and/or patients [40, 41]. The mutation of IgG-derived hinge domain to abrogate FcγR recognition could improve the persistence and antitumor efficacy of CAR-T cells [41, 42]. Additionally, the hinge domain has been found to play a role in the induction of tonic signaling, which is responsible for CAR T-cell senescence [43]. However, without the HER2 antigen stimulation, there was no obvious difference in the number of CAR clusters and the expression levels of activation markers between CARCys and CARSer groups (Fig. S1B), implying that the tonic signaling wouldn’t be exacerbated in CARCys-T cells.
In summary, we provided a strategy to facilitate CAR clustering using a hinge domain containing 11 cysteines. Our investigation demonstrated that the CARCys-T cells displayed enhanced cytotoxic efficiency both in vitro and in vivo. Thus, by following the basic principles and structure of TCR-induced T cell activation, our study offers a new building block for the next generation of CAR designs.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
The authors thank National Center for Protein Science Shanghai (NCPSS) for STORM system, Prof. Yanhui Xu (Fudan University), Prof. Chenqi Xu (University of Chinese Academy of Sciences) and Ms. Yang Yu (NCPSS) for valuable suggestions, Dr. Yufei Zhang of Olympus (Beijing) sales & service co., Ms. Yao Li of NCPSS, Ms. Shenglan Gu of International Peace Maternity and Child Health Hospital for excellent technical assistance.
Author contributions
YC, FL, and YW designed the experiments. YW and FL wrote the manuscript. YC, FL, JL, SA, GR, and JQ revised the manuscript. YW, YG, CN, SZ, GR, LC, and CY performed the experiments and acquired the data. YW, FL, BW, JQ, and LC analyzed the data and prepared the figures. All authors provided intellectual input and read the manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (82121002, 82130050, 81870375), Shanghai Rising-Star Program (18QA1401000), Open Research Fund of State Key Laboratory of Genetic Engineering, Fudan University (SKLGE1911), and Shenzhen Institute of Synthetic Biology Scientific Research Program (ZTXM20214002). SA is a Senior Clinical Investigator of the Research Fund-Flanders (FWO) (Belgium). GR is supported by a Doctoral Grant Strategic Basic Research of the FWO (Grant 1S72821N) and the MeToYou Foundation (Belgium).
Declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval and ethical standards.
Human N87, Huh7, Ramos and K562 cell lines were purchased from the Institute of Biochemistry and Cell Biology of the Chinese Academy of Science (Shanghai, China). Human peripheral blood mononuclear cells isolated from healthy donors or patients with hepatocellular carcinoma (HCC) were purchased from the Shanghai Blood Center (Shanghai, China), or obtained from Huashan hospital (Shanghai, China), respectively. Fresh human HCC samples were obtained from surgery performed at the Huashan hospital. Informed consent was obtained from HCC patients involved, and the project was approved by the Ethical Committee of Medical Research, Huashan Hospital of Fudan University (No. 2013-005). Six- to eight-week-old female nude mice (BALB/c) were purchased from SLAC (Shanghai, China) and kept under specific pathogen-free conditions in animal facility of Fudan University. The animal experimental protocol was approved by the Ethics Committee of Fudan University (201901003Z) following the Guidelines for the Care and Use of Laboratory Animals (No. 55 issued by Ministry of Health, China on January 25th, 1998).
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Jie Liu, Email: jieliu@fudan.edu.cn.
Feifei Luo, Email: feifeiluo@fudan.edu.cn.
Yiwei Chu, Email: yiweichu@fudan.edu.cn.
References
- 1.June CH, Sadelain M. Chimeric antigen receptor therapy. N Engl J Med. 2018;379:64–73. doi: 10.1056/NEJMra1706169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jackson HJ, Rafiq S, Brentjens RJ. Driving CAR T-cells forward. Nat Rev Clin Oncol. 2016;13:370–383. doi: 10.1038/nrclinonc.2016.36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Majzner RG, Mackall CL. Clinical lessons learned from the first leg of the CAR T cell journey. Nat Med. 2019;25:1341–1355. doi: 10.1038/s41591-019-0564-6. [DOI] [PubMed] [Google Scholar]
- 4.Hong M, Clubb JD, Chen YY. Engineering CAR-T Cells for Next-Generation Cancer Therapy. Cancer Cell. 2020;38:473–488. doi: 10.1016/j.ccell.2020.07.005. [DOI] [PubMed] [Google Scholar]
- 5.Sadelain M, Brentjens R, Riviere I. The basic principles of chimeric antigen receptor design. Cancer Discov. 2013;3:388–398. doi: 10.1158/2159-8290.CD-12-0548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Moritz D, Wels W, Mattern J, Groner B. Cytotoxic T lymphocytes with a grafted recognition specificity for ERBB2-expressing tumor cells. Proc Natl Acad Sci USA. 1994;91:4318–4322. doi: 10.1073/pnas.91.10.4318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Arakawa F, Shibaguchi H, Xu Z, Kuroki M. Targeting of T cells to CEA-expressing tumor cells by chimeric immune receptors with a highly specific single-chain anti-CEA activity. Anticancer Res. 2002;22:4285–4289. [PubMed] [Google Scholar]
- 8.Eshhar Z, Waks T, Gross G, Schindler DG. Specific activation and targeting of cytotoxic lymphocytes through chimeric single chains consisting of antibody-binding domains and the gamma or zeta subunits of the immunoglobulin and T-cell receptors. Proc Natl Acad Sci USA. 1993;90:720–724. doi: 10.1073/pnas.90.2.720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ramos CA, Heslop HE, Brenner MK. CAR-T cell therapy for lymphoma. Annu Rev Med. 2016;67:165–183. doi: 10.1146/annurev-med-051914-021702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Srivastava S, Riddell SR. Engineering CAR-T cells: design concepts. Trends Immunol. 2015;36:494–502. doi: 10.1016/j.it.2015.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Davenport AJ, Cross RS, Watson KA, et al. Chimeric antigen receptor T cells form nonclassical and potent immune synapses driving rapid cytotoxicity. Proc Natl Acad Sci USA. 2018;115:E2068–E2076. doi: 10.1073/pnas.1716266115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Li R, Ma C, Cai H, Chen W. The CAR T-Cell mechanoimmunology at a Glance. Adv Sci. 2020;7:2002628. doi: 10.1002/advs.202002628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wu L, Wei Q, Brzostek J, Gascoigne NRJ. Signaling from T cell receptors (TCRs) and chimeric antigen receptors (CARs) on T cells. Cell Mol Immunol. 2020;17:600–612. doi: 10.1038/s41423-020-0470-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gudipati V, Rydzek J, Doel-Perez I, et al. Inefficient CAR-proximal signaling blunts antigen sensitivity. Nat Immunol. 2020;21:848–856. doi: 10.1038/s41590-020-0719-0. [DOI] [PubMed] [Google Scholar]
- 15.Guedan S, Chen X, Madar A, et al. ICOS-based chimeric antigen receptors program bipolar TH17/TH1 cells. Blood. 2014;124:1070–1080. doi: 10.1182/blood-2013-10-535245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Johnson LA, Scholler J, Ohkuri T, et al. Rational development and characterization of humanized anti-EGFR variant III chimeric antigen receptor T cells for glioblastoma. Sci Transl Med. 2015 doi: 10.1126/scitranslmed.aaa4963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Long AH, Haso WM, Shern JF, et al. 4–1BB costimulation ameliorates T cell exhaustion induced by tonic signaling of chimeric antigen receptors. Nat Med. 2015;21:581–590. doi: 10.1038/nm.3838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Maher J, Wilkie S, Davies DM, Arif S, Picco G, Julien S, Foster J, Burchell J, Taylor-Papadimitriou J. Targeting of tumor-associated glycoforms of MUC1 with CAR T cells. Immunity. 2016;45:945–946. doi: 10.1016/j.immuni.2016.10.014. [DOI] [PubMed] [Google Scholar]
- 19.Goyette J, Nieves DJ, Ma Y, Gaus K. How does T cell receptor clustering impact on signal transduction? J Cell Sci. 2019 doi: 10.1242/jcs.226423. [DOI] [PubMed] [Google Scholar]
- 20.Ma Y, Lim YJ, Benda A, Lou J, Goyette J, Gaus K. Clustering of the zeta-Chain can initiate T cell receptor signaling. Int J Mol Sci. 2020 doi: 10.3390/ijms21103498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ma Y, Pandzic E, Nicovich PR, Yamamoto Y, Kwiatek J, Pageon SV, Benda A, Rossy J, Gaus K. An intermolecular FRET sensor detects the dynamics of T cell receptor clustering. Nat Commun. 2017;8:15100. doi: 10.1038/ncomms15100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fass D, Thorpe C. Chemistry and enzymology of disulfide cross-linking in proteins. Chem Rev. 2018;118:1169–1198. doi: 10.1021/acs.chemrev.7b00123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hogg PJ. Multiple disulfide-bonded states of native proteins: estimate of number using probabilities of disulfide bond formation. Molecules. 2020 doi: 10.3390/molecules25235729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Guest RD, Hawkins RE, Kirillova N, et al. The role of extracellular spacer regions in the optimal design of chimeric immune receptors: evaluation of four different scFvs and antigens. J Immunother. 2005;28:203–211. doi: 10.1097/01.cji.0000161397.96582.59. [DOI] [PubMed] [Google Scholar]
- 25.Zhang X, Calvert RA, Sutton BJ, Dore KA. IgY: a key isotype in antibody evolution. Biol Rev Camb Philos Soc. 2017;92:2144–2156. doi: 10.1111/brv.12325. [DOI] [PubMed] [Google Scholar]
- 26.Bitra A, Doukov T, Wang J, Picarda G, Benedict CA, Croft M, Zajonc DM. Crystal structure of murine 4–1BB and its interaction with 4–1BBL support a role for galectin-9 in 4–1BB signaling. J Biol Chem. 2018;293:1317–1329. doi: 10.1074/jbc.M117.814905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Luo F, Qian J, Yang J, Deng Y, Zheng X, Liu J, Chu Y. Bifunctional alphaHER2/CD3 RNA-engineered CART-like human T cells specifically eliminate HER2(+) gastric cancer. Cell Res. 2016;26:850–853. doi: 10.1038/cr.2016.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ester M, Kriegel HP, Sander J, Xu X (1996) A Density-Based Algorithm for Discovering Clusters in Large Spatial Databases with Noise. AAAI Press. In: Proceedings of the 2nd International Conference on Knowledge Discovery and Data Mining, pp 226–231
- 29.Broutier L, Mastrogiovanni G, Verstegen MM, et al. Human primary liver cancer-derived organoid cultures for disease modeling and drug screening. Nat Med. 2017;23:1424–1435. doi: 10.1038/nm.4438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cattaneo CM, Dijkstra KK, Fanchi LF, Kelderman S, Kaing S, van Rooij N, van den Brink S, Schumacher TN, Voest EE. Tumor organoid-T-cell coculture systems. Nat Protoc. 2020;15:15–39. doi: 10.1038/s41596-019-0232-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wang LC, Lo A, Scholler J, et al. Targeting fibroblast activation protein in tumor stroma with chimeric antigen receptor T cells can inhibit tumor growth and augment host immunity without severe toxicity. Cancer Immunol Res. 2014;2:154–166. doi: 10.1158/2326-6066.CIR-13-0027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Qin L, Lai Y, Zhao R, et al. Incorporation of a hinge domain improves the expansion of chimeric antigen receptor T cells. J Hematol Oncol. 2017;10:68. doi: 10.1186/s13045-017-0437-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Majzner RG, Rietberg SP, Sotillo E, et al. Tuning the antigen density requirement for CAR T-cell activity. Cancer Discov. 2020;10:702–723. doi: 10.1158/2159-8290.CD-19-0945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Brudno JN, Kochenderfer JN. Chimeric antigen receptor T-cell therapies for lymphoma. Nat Rev Clin Oncol. 2018;15:31–46. doi: 10.1038/nrclinonc.2017.128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Saito T, Yokosuka T. Immunological synapse and microclusters: the site for recognition and activation of T cells. Curr Opin Immunol. 2006;18:305–313. doi: 10.1016/j.coi.2006.03.014. [DOI] [PubMed] [Google Scholar]
- 36.Gao Y, Wang Y, Luo F, Chu Y. Optimization of T Cell redirecting strategies: obtaining inspirations from natural process of T cell activation. Front Immunol. 2021;12:664329. doi: 10.3389/fimmu.2021.664329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Pageon SV, Tabarin T, Yamamoto Y, et al. Functional role of T-cell receptor nanoclusters in signal initiation and antigen discrimination. Proc Natl Acad Sci USA. 2016;113:E5454–E5463. doi: 10.1073/pnas.1607436113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Taylor MJ, Husain K, Gartner ZJ, Mayor S, Vale RD. A DNA-based T cell receptor reveals a role for receptor clustering in ligand discrimination. Cell. 2017;169:108–119. doi: 10.1016/j.cell.2017.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Singh N, Frey NV, Engels B, et al. Antigen-independent activation enhances the efficacy of 4–1BB-costimulated CD22 CAR T cells. Nat Med. 2021;27:842–850. doi: 10.1038/s41591-021-01326-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Alabanza L, Pegues M, Geldres C, Shi V, Wiltzius JJW, Sievers SA, Yang S, Kochenderfer JN. Function of novel Anti-CD19 chimeric antigen receptors with human variable regions is affected by Hinge and transmembrane domains. Mol Ther J American Soc Gene Ther. 2017;25:2452–2465. doi: 10.1016/j.ymthe.2017.07.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Hudecek M, Sommermeyer D, Kosasih PL, Silva-Benedict A, Liu L, Rader C, Jensen MC, Riddell SR. The nonsignaling extracellular spacer domain of chimeric antigen receptors is decisive for in vivo antitumor activity. Cancer Immunol Res. 2015;3:125–135. doi: 10.1158/2326-6066.CIR-14-0127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Jonnalagadda M, Mardiros A, Urak R, et al. Chimeric antigen receptors with mutated IgG4 Fc spacer avoid fc receptor binding and improve T cell persistence and antitumor efficacy. Mol Ther J American Soc Gene Ther. 2015;23:757–768. doi: 10.1038/mt.2014.208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Watanabe N, Bajgain P, Sukumaran S, Ansari S, Heslop HE, Rooney CM, Brenner MK, Leen AM, Vera JF. Fine-tuning the CAR spacer improves T-cell potency. Oncoimmunology. 2016;5:e1253656. doi: 10.1080/2162402X.2016.1253656. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.