Abstract
Purpose
Ascariasis caused by the helminth Ascaris suum is the most common parasitosis of swine worldwide and it may involve all age categories of pigs. The present study reports an unusual localization of A. suum worms in the biliary system of a piglet slaughtered for human consumption.
Methods
The liver was subjected to ultrasound scan and pathological examination. The isolated worms were morphologically examined and the DNA was extracted for the molecular identification of the species involved.
Results
A total of 43 preadult nematodes were found within the gallbladder and the bile ducts. Parasites were morphologically identified as belonging to the genus Ascaris and molecularly as A. suum. At gross examination, the liver was moderately enlarged, with the bile ducts severely dilated. A chronic inflammatory infiltrate was noted, often centered around ectatic bile ducts (up to 5 mm in diameter), lined by hyperplastic epithelium and filled with sections of nematodes. The worm sections showed smooth cuticle, coelomyarian musculature, and an intestinal tract lined by columnar, uninucleated cells within a pseudocoelom. The ex vivo ultrasonographic examination of the liver allowed the visualization of several nematodes in the bile duct lumen and could be suggested for in vivo diagnosis. Unfortunately, the absence of the intestine did not allow to define the pathogenesis of the infection.
Conclusion
Although, given the unusual nature of this finding, it is difficult to identify predisposing factors for this A. suum localization, it suggests that ascariasis should be considered in the differential diagnosis of pigs with hepatobiliary disease.
Keywords: Ascaris suum, Pig, Bile ducts, Ultrasound, Parasitology
Introduction
Ascariasis is the most common parasitosis in pigs worldwide and is mainly caused by the helminth Ascaris suum, which belongs to the Family Ascarididae and which can reach a relevant size, of up to 40 cm in females [1]. Ascaris lumbricoides can also grow in pigs and both parasite species can mature in humans [2].
This parasitosis has a global distribution and is detected in both organic [3, 4] and industrial pig farms, especially in fattening pigs and old sows [5–7]. Ascaris suum infection can affect all age categories of pigs and depends on several factors mainly related to the type of rearing and management [8]. High prevalence rates were found in Denmark (25–88%) and Canada (18–82%) [9, 10]. In Spain, the prevalence found in reared pigs varied from 28.7 to 48.7% [11, 12].
The relevant diffusion of the parasitosis is mainly due to the high fecundity of female A. suum worms (up to 200,000 eggs per female per day) and to the strong structure of the eggs, which guarantees their survival in the external environment even up to 4 years [13].
The biological cycle is direct and begins with the ingestion of embryonated eggs containing the infective larva, an early third-stage (L3) larva covered by the cuticle of the second stage larva (L2) [14]. Piglets can become infected shortly after birth by ingesting embrionated eggs smearing the mammary skin of the sow or directly through food and drinking water contaminated with eggs, especially in polluted soil [2]. The eggs hatch in the stomach and small intestine and release the ascarid L3 larvae, which penetrate the intestinal wall and start their hepatic-pulmonary migration, first to the liver via the portal system [15]. In the liver, the larvae lose the cuticle and enter the lungs via the systemic circulation, where they embolize in the pulmonary capillaries, enter the alveoli, are coughed up and then swallowed, reaching the small intestine approximately two weeks after infection [16]. Once back in the small intestine, the larvae moult into the fourth-stage (L4) larva, where most or all of the parasites are expelled from the intestine at 14–21 days post-infection (self-cure). Some of these larvae may remain in the small intestine for the rest of their lives, developing into preadult and then adult worms that reach sexual maturity at 6–10 weeks of age [14].
Earthworms and dung beetles may play a role in the transmission, as they can ingest Ascaris eggs while feeding on soil and feces. The eggs can then hatch and infective larvae can remain in the tissues of earthworms and dung beetles until they are eaten by the pig host, acting as potential paratenic hosts in the life cycle of A. suum [17, 18].
The migration of larvae within the liver can lead to the onset of multifocal interstitial hepatitis in sensitized animals, characterized by fibrosis appearing as white spots (milk spots) [19, 20], while pulmonary alterations caused by migrating larvae include pneumonia, pleurisy and allergic asthma in infected pigs [21].
Although the course of ascariasis in pigs is usually subclinical, clinical infections can occur in growing pigs [22].
The pathological effects of adult A. suum in the small intestine are less dramatic than those of larval migration, but they can cause a chronic malabsorption syndrome with atrophy of the intestinal villi, resulting in reduced weight gain and poor feed conversion [23, 24].
In rare cases, obstruction of the intestinal lumen may happen in heavily infested animals, as well as obstruction of the bile ducts due to the migratory tendency of ascarids, resulting in jaundice [2].
The present study reports an unusual localization of A. suum worms in the biliary system of a piglet.
Materials and Methods
In February 2022, a 30-days-old piglet slaughtered in Milis (Latitude: 40° 00′ 49″ N Longitude: 8° 35′ 53″ E) in the Oristano province (Sardinia—Italy) presented Ascaris worms in the hepatic biliary tree. The piglet was the seventh of a litter born from a 3-year-old Sarda multiparous sow in which Ascaris eggs were found through the copromicroscopical examination. The piglets were raised indoor, in a box with solid concrete flooring together with his mother and the sow before the parturition had access to the paddock outdoor where she could root.
At the time of the home slaughtering, according to the Legislative Decree 2 February 2021 n.27 which provides the inspection in only 10% of the home slaughtering, the official veterinarian did not attend, and the intestine was discarded. The liver was examined, and transverse cuts were performed to allow the isolation of worms within the bile ducts. The worms were washed with 0.9% NaCl solution, and then examined under a stereo microscope, the measurements were taken and their identification was carried out using the morphometric keys available in literature [25]. The DNA was extracted from the middle portion of a collected worm using a G-spin™ Total DNA Extraction kit (iNtRON Biotechnology, Korea), according to the manufacturer’s instructions. The rDNA region comprising the ITS-1 and the ITS-2 was amplified by PCR using 100 pmol of primers NC5 and NC2 as described by Zhu et al. [26]. PCR products were sent to an external sequencing service (Eurofins Genomics, Ebersberg, Germany) and the sequences obtained were compared with those found in the National Centre for Biotechnology Information (NCBI) database using Basic Local Alignment Search Tool (BLAST) (http:// www. ncbi. nlm. nih. gov/ BLAST/).
Before the parasite removal, the liver was subjected to ex vivo ultrasound examination using a portable ultrasound unit (My Lab Alpha, Esaote, Florence, Italy) equipped with a multifrequency (3–13 MHz) linear probe (SL1543, Esaote, Florence, Italy).
The liver was also processed for the histological examination as follows: tissue samples of the liver were immediately fixed in 10% buffered formalin for 48 h, dehydrated with increasing alcohol concentrations and xylene in an automatic tissue processor and paraffin-embedded. Sections of 3 μm thickness were stained with hematoxylin and eosin (HE) and were observed at light microscopy (Nikon Eclipse 80i). Parasites were categorized according to the available literature [27].
Results
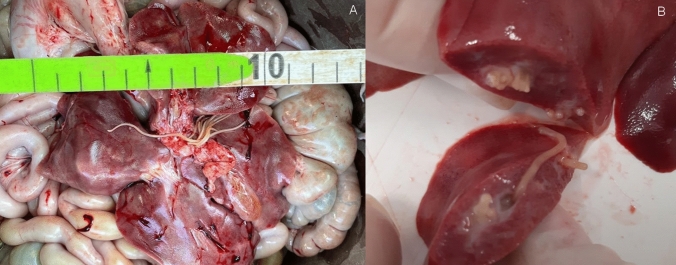
The presence of ascarids in the biliary system was detected only in a single piglet within the whole litter. A total of 43 preadult nematodes, with a mean length of 8 cm and a mean width of 1.7 mm, were found in the gallbladder and biliary tree (Fig. 1).
Fig.1.
Gross examination of the liver, with several preadult specimens of 7 cm mean length (A), and a detail of nematodes occupying the bile ducts (B). The scale bar in A has centimeter (cm) as unit
Parasites were morphologically identified under the stereomicroscope as belonging to the genus Ascaris, presenting the characteristic three lips, without interlabia and cervical alae, as described in the literature with morphological keys [25].
Molecular biology allowed to identify the specimens as A. suum, and the nucleotide sequence obtained had a 99% similarity with a sequence from Thailand (accession number MF358937.1) deposited by Sadaow et al. [28].
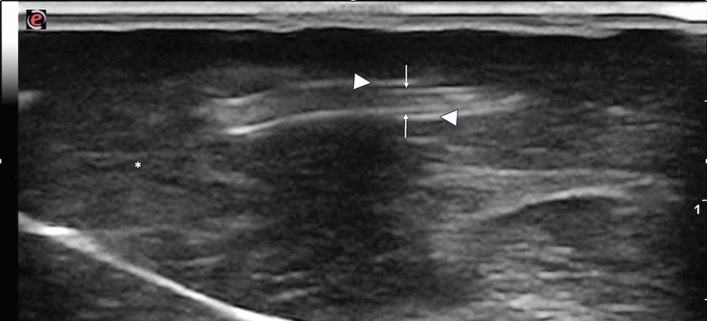
The hepatic ultrasound examination performed before the isolation of the parasites revealed the presence of several nematodes in the lumen of dilated and thick-walled bile ducts (Fig. 2). In longitudinal section, worms appeared as nonshadowing linear, tubular structures, characterized by two parallel hyperechoic lines separated by a hypoechoic zone. In transverse section, the worms had a “doughnut-like” appearance with a circular hyperechoic nonshadowing wall and a hypoechoic center.
Fig.2.
Ex vivo ultrasound image of the liver. The white arrows indicate a longitudinal parasite section, white arrowheads indicate the thickened walls of the bile ducts. *Liver parenchyma
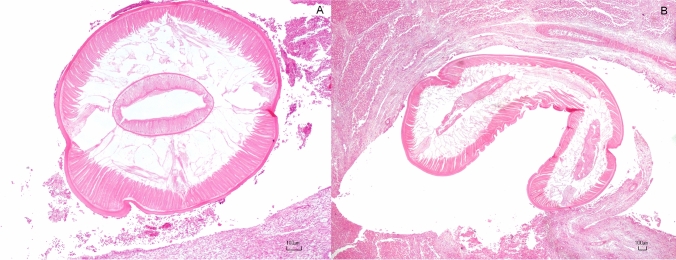
Macroscopically, the liver was moderately enlarged, with accentuated lobular pattern and scattered pales areas admixed with multifocal hemorrhages. Multifocally, the parasitized bile ducts were severely enlarged. The expansion of portal areas determined the compression of adjacent sinusoids and hepatic cords with a severe, chronic inflammatory infiltrate around the bile ducts. The latter were up to 5 mm in diameter, lined by hyperplastic epithelium (biliary epithelial hyperplasia) and filled by sections of nematodes. They were also surrounded by a moderate number of plump reactive fibroblasts and lesser numbers of fibrocytes. The cells were observed embedded in a moderate amount of eosinophilic fibrillary material (collagen) interpreted as fibroplasia, often infiltrated by a high number of eosinophils, fewer lymphocytes, macrophages and plasma cells. Within the lumen of bile ducts, sloughed epithelial cells and cellular debris were admixed with longitudinal up to 4 mm and transverse (up to 1.5 mm) sections of nematodes with a smooth cuticle, coelomyarian musculature, intestinal tract lined by columnar uninucleated cells within a pseudocoelom (Fig. 3).
Fig.3.
Transverse (A) and longitudinal (B) histological sections of nematodes with a smooth cuticle, coelomyarian musculature, intestinal tract lined by columnar uninucleated cells within a pseudocoelom. H.E bar 100 µm
Adjacent to bands of fibrosis, there were limitedly shrunken and hypereosinophilic individualized hepatocytes (atrophy). In less affected areas, portal areas were multifocally expanded by fibrous connective tissue that often connects adjacent portal areas (bridging fibrosis), admixed with increased numbers of small bile ducts (ductular reaction) and previously described inflammatory cells. The parasites were histologically identified as belonging to the Family Ascarididae, based on the microstructural features indicated by the literature [27].
Discussion
Here, we present a rare case with preadult worms of A. suum in the biliary system of a piglet. To our knowledge, this is the second literature report of this localization of A. suum in a piglet. In contrast to humans, where hepatobiliary ascariasis is a known complication of A. lumbricoides infection, the only other report of hepatobiliary A. suum infection in pigs on a global scale dates back 70 years [29].
There are only a few reports of ascarid migration into host bile ducts in the literature, one by Toxocara vitulorum in a buffalo [30] and a more recent report of adult Parascaris sp. in the biliary tree of a foal [31].
In addition, there are a few reports of accidental infection of A. suum in hosts other than pigs, such as cattle [32], sheep [33] and a dog [34], but with the exception of the sheep case, only larvae were found in these unusual hosts.
Unfortunately, due to the absence of gut, it was not possible to define the pathogenesis of the infection. However, as has been reported with A. lumbricoides infection in humans, the A. suum worms likely migrated from the intestine through the ductus choledocus to the liver [35]. Biliary ascariasis in humans is a rare disease in nonendemic areas, but in endemic areas it is the cause of a significant proportion of biliary and pancreatic diseases. In particular, in India, in the highly endemic region of Kashmir, ascariasis was found to be the cause in 36.7% of 109 patients with confirmed biliary and pancreatic diseases [35]. In the human host, altered intestinal motility is the usual condition for ascarids to reach the duodenum from their natural habitat, the jejunum. This may be due to a severe worm infection or other intestinal infections of viral, bacterial, or parasitic origin, but the host response to an adult worm may also alter vasomotor reflexes and secretory responses, which in turn affect intestinal tone and motility [36]. In this condition, worms can reach the Vater ampulla from the duodenum and then they can be located: (a) in the ampulla itself, (b) in the common bile duct, or (c) in the hepatic ducts or somewhere in the biliary tree [1].
It has been reported that worms in humans actively migrate in and out of the bile ducts from the duodenum and therefore can lead to even transient biliary tree parasitosis [35].
The number of parasites found in the bile ducts and the nature of the histologic reaction around them, seems to rule out the hypothesis of backward migration into the small intestine. Unfortunately, it was not possible to examine the small intestine of the piglet reported here, so it is impossible to know whether there was a concomitant intestinal infection. It would be interesting to follow the infection pathway to assess whether the parasites could reach sexual maturity, mate and complete the life cycle in the biliary system. Such an experiment could have been performed by in vivo ultrasound examination, which would have made it possible to exclude a possible concomitant intestinal infection and to detect ascarids infection at the level of the bile ducts. The ultrasonographic appearance of nematodes has been previously described in humans and animals [37–39]. In longitudinal section, they appear as linear “train track” structures consisting of two parallel hyperechoic lines representing the cuticle, separated by a narrow hypo/anechoic line representing the alimentary canal. In cross-section, they have a “donut” appearance characterized by a hyperechoic circular wall with a hypoanechoic centre. The sonographic diagnosis of biliary ascariasis has been prevoiusly described in human medicine [37, 40]. However, to our knowledge, this report is the first to describe the ultrasonographic appearance of roundworms in the biliary system of pigs, although the examination was performed on an organ and not on a live animal. Because roundworms are easily detected by ultrasound, this test could provide an in vivo diagnostic method for detecting parasites in the biliary tract of piglets.
Given the unusual nature of this finding, it is difficult to identify predisposing factors for this A. suum localization, such as factors related to age, breed, concurrent intestinal infections, diet, etc.
Although the presence of the preadult worms was a slaughter finding with no associated pathology, it suggests that ascariasis should be considered in the differential diagnosis of pigs with hepatobiliary disease.
Author Contributions
Claudia Tamponi: Parasitological examination, paper writing, read and approved the final manuscript. Lia Cavallo: Parasitological examination, input for paper writing, read and approved the final manuscript. Giorgia Dessì: Parasitological examination, molecular analysis, input for paper writing, read and approved the final manuscript. Francesco Sardu: Collection of sample, input for paper writing, read and approved the final manuscript. Carlo Carta: Parasitological examination, input for paper writing, read and approved the final manuscript. Andrea Corda: Ultrasound examination, input for paper writing, read and approved the final manuscript. Giovanni Pietro Burrai: Histopathological examination, input for paper writing, read and approved the final manuscript. Antonio Varcasia: Input for paper writing, critical revision of the manuscript, read and approved the final manuscript. Antonio Scala: Experimental conception and design, paper writing, critical revision of the manuscript, read and approved the final manuscript.
Funding
Open access funding provided by Università degli Studi di Sassari within the CRUI-CARE Agreement. The authors did not receive support from any organization for the submitted work.
Data Availability
All the data and materials related to the current study are available from the corresponding author on reasonable request.
Declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Stewart TB, Hoyt PG. Internal parasites. In: Straw BE, Zimmerman JJ, D’Allaire S, Taylor DJ, editors. Taylor disease of swine. Oxoford: Blakwell publishing; 2013. [Google Scholar]
- 2.Bowman DD. Georgis’ parasitology for veterinarians. London: Saunders; 1999. p. 414. [Google Scholar]
- 3.Nansen P, Roepstorff A. Parasitic helminths of the pig: factors influencing transmission and infection levels. Int J Parasitol. 1999;29:877–891. doi: 10.1016/S0020-7519(99)00048-X. [DOI] [PubMed] [Google Scholar]
- 4.Nissen S, Poulsen IH, Nejsum P, Olsen A, Roepstorff A, Rubaire-Akiiki C, Thamsborg SM. Prevalence of gastrointestinal nematodes in growing pigs in Kabale district in Uganda. Trop Anim Health Prod. 2011;43:567–572. doi: 10.1007/s11250-010-9732-x. [DOI] [PubMed] [Google Scholar]
- 5.Roepstorff A. Helminth surveillance as a prerequisite for anthelmintic treatment in intensive sow herds. Vet Parasitol. 1997;73:139–151. doi: 10.1016/S0304-4017(97)00062-9. [DOI] [PubMed] [Google Scholar]
- 6.Roepstorff A, Nilsson O, O’Callaghan CJ, Oksanen A, Gjerde B, Richter SH, et al. Intestinal parasites in swine in the Nordic countries: multilevel modelling of Ascaris suum infections in relation to production factors. Parasitology. 1999;119:521–534. doi: 10.1017/S0031182099004941. [DOI] [PubMed] [Google Scholar]
- 7.Roepstorff A, Mejer H, Nejsum P, Thamsborg SM. Helminth parasites in pigs: new challenges in pig production and current research highlights. Vet Parasitol. 2011;180:72–81. doi: 10.1016/j.vetpar.2011.05.029. [DOI] [PubMed] [Google Scholar]
- 8.Boes J, Kanora A, Havn KT, Christiansen S, Vestergaard-Nielsen K, Jacobs J, Alban L. Effect of Ascaris suum infection on performance of fattening pigs. Vet Parasitol. 2010;172:269–276. doi: 10.1016/j.vetpar.2010.05.007. [DOI] [PubMed] [Google Scholar]
- 9.Nejsum P, Parker ED, Frydenberg J, Roepstorff A, Boes J, Haque R, et al. Ascariasis is a zoonosis in Denmark. J Clin Microbiol. 2005;43:1142–1148. doi: 10.1128/jcm.43.3.1142-1148.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Haugegaard J. Prevalence of nematodes in Danish industrialized sow farms with loose housed sows in dynamic groups. Vet Parasitol. 2010;168:156–159. doi: 10.1016/j.vetpar.2009.10.009. [DOI] [PubMed] [Google Scholar]
- 11.García Vallejo TB (1999) Endoparasitosis del porcino ibérico en Extremadura (España): epidemiología y control. Tesis doctorales Universidad de Etremadura. Edita: Universidad de Extremadura Servicio de Publicaciones, Cáceres, Spain. http://www.pcid.es/public.htm
- 12.Frontera-Carrión EMF (2000) Repercusiones orgánicas de la infección experimental por Ascaris suum en el cerdo ibérico. Tesis doctorales Universidad de Etremadura. Edita: Universidad de Extremadura Servicio de Publicaciones, Cáceres, Spain. http://www.pcid.es/public.htm
- 13.Taylor MA, Coop RL, Wall RL. Veterinary Parasitology. 3. Blackwell Publishing; 2007. [Google Scholar]
- 14.Jungersen G. Immunity and Immune Responses to Ascaris suum in Pigs. In: Holland CV, Kennedy MW, editors. The geohelminths: ascaris, trichuris and hookworm. world class parasites. Boston: Springer; 2002. [Google Scholar]
- 15.Kassai T. Veterinary Helmintology. Oxford: Butler worth heinemann; 1999. [Google Scholar]
- 16.Holland C. Ascaris: the neglected parasite. Academic press; 2013. p. 439. [Google Scholar]
- 17.Roepstorff A, Grønvold J, Larsen MN, Kraglund HO, Fagerholm HP. The earthworm Lumbricus terrestris as a possible paratenic or intermediate host of the pig parasite Ascaris suum. Comp Parasitol. 2002;69:206–210. doi: 10.1654/1525-26472002069. [DOI] [Google Scholar]
- 18.Nichols E, Gómez A. Dung beetles and fecal helminth transmission: patterns, mechanisms and questions. Parasitology. 2014;141:614–623. doi: 10.1017/S0031182013002011. [DOI] [PubMed] [Google Scholar]
- 19.Roepstorff A. Ascaris suum in pigs: population biology and epidemiology. D.Sc: Thesis, Royal Veterinary and Agricultural University, Copenhagen; 2003. [Google Scholar]
- 20.Knecht D, Jankowska A, Zalesny G. The impact of gastrointestinal parasites infection on slaughter efficiency in pigs. Vet Parasitol. 2012;184:291–297. doi: 10.1016/j.vetpar.2011.09.006. [DOI] [PubMed] [Google Scholar]
- 21.Vlaminck J, Levecke B, Vercruysse J, Geldhof P. Advances in the diagnosis of Ascaris suum infections in pigs and their possible applications in humans. Parasitology. 2014;141:1904–1911. doi: 10.1017/S0031182014000328. [DOI] [PubMed] [Google Scholar]
- 22.Joachim A, Dülmer N, Daugschies A, Roepstorff A. Occurrence of helminths in pig fattening units with different management systems in Northern Germany. Vet Parasitol. 2001;96:135–146. doi: 10.1016/S0304-4017(00)00431-3. [DOI] [PubMed] [Google Scholar]
- 23.Crompton DWT. Ascaris and ascariasis. Adv Parasitol. 2001;48:285–375. doi: 10.1016/S0065-308X(01)48008-0. [DOI] [PubMed] [Google Scholar]
- 24.Vázquez-Rodríguez J (2015) Evaluación del estado sanitario del jabalí en la Sierra de Aracena (Huelva) y sus factores de riesgo asociados. Doctoral thesis, Universidad de Murcia, Spain. http://hdl.handle.net/10803/336665
- 25.Yamaguti S (1961) Systema Helminthum. Vol. III. The Nematodes of Vertebrates, Pt. II. Interscience Publishers, New York & London
- 26.Zhu X, Chilton NB, Jacobs DE, Boes J, Gasser RB. Characterisation of Ascaris from human and pig hosts by nuclear ribosomal DNA sequences. Int J Parasitol. 1999;29:469–478. doi: 10.1016/s0020-7519(98)00226-4. [DOI] [PubMed] [Google Scholar]
- 27.Gardiner CH, Poynton SL. American registry of pathology (eds.) Washington: DC; 1999. An atlas of metazoan parasites in animal tissues. [Google Scholar]
- 28.Sadaow L, Sanpool O, Phosuk I, Rodpai R, Thanchomnang T, Wijit A, et al. Molecular identification of Ascaris lumbricoides and Ascaris suum recovered from humans and pigs in Thailand, Lao PDR, and Myanmar. Parasitol Res. 2018;117:2427–2436. doi: 10.1007/s00436-018-5931-6. [DOI] [PubMed] [Google Scholar]
- 29.Prchal C. Postmortem condemnations resulting from migratory ascarids in hogs. J Am Vet Med Assoc. 1953;122:42–44. [PubMed] [Google Scholar]
- 30.Srivastava A, Sharma D. Studies on the occurrence, clinical features and pathomorphological aspects of ascariasis in buffalo calves. Vet Res J. 1981;4:160–162. [Google Scholar]
- 31.Sazmand A, Yavari M, Babaei M, Nourian A, Otranto D. Biliary parascarosis in a foal. Vet Med Sci. 2023;9:1149–1153. doi: 10.1002/vms3.1087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Taylor HL, Spagnoli ST, Calcutt MJ, Kim DY. Aberrant Ascaris suum nematode infection in cattle, Missouri, USA. Emerg Infect Dis. 2016;22:339–340. doi: 10.3201/eid2202.150686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Jansen J, Kouwenberg L, Peregrine AS (2010) Sheep livers condemned due to Ascaris suum. Ontario sheep news 29:27. https://www.ontariosheep.org/uploads/userfiles/files/Sheep-Livers.pdf
- 34.Xie Y, Liu Y, Gu X, Zhou X, Peng X, He R, Guo H, Zuo Z, Yang G. First report on aberrant Ascaris suum infection in a dog. China Parasit Vectors. 2020;13:86. doi: 10.1186/s13071-020-3963-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Das AK. Hepatic and biliary ascariasis. J Glob Infect Dis. 2014;6:65–72. doi: 10.4103/0974-777X.132042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Pawlowski ZS. Ascariasis. Clin Gastroenterol. 1978;7:157–178. doi: 10.1016/S0300-5089(21)00201-7. [DOI] [PubMed] [Google Scholar]
- 37.Wu S. Sonographic findings of ascaris lumbricoides in the gastrointestinal and biliary tracts. Ultrasound Q. 2009;25:207–209. doi: 10.1097/RUQ.0b013e3181c47a2d. [DOI] [PubMed] [Google Scholar]
- 38.Corda A, Tamponi C, Meloni R, Varcasia A, Pinna Parpaglia ML, Gomez-Ochoa P, Scala A. Ultrasonography for early diagnosis of Toxocara canis infection in puppies. Parasitol Res. 2019;118:873–880. doi: 10.1007/s00436-019-06239-4. [DOI] [PubMed] [Google Scholar]
- 39.Corda A, Corda F, Secchi V, Pentcheva P, Tamponi C, Tilocca L, Varcasia A, Scala A. Ultrasonography of parasitic diseases in domestic animals: a systematic review. Animals. 2022;12:1252. doi: 10.3390/ani12101252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Khuroo MS, Zargar SA, Mahajan R, et al. Sonographic appearances in biliary ascariasis. Gastroenterology. 1987;93:267–272. doi: 10.1016/0016-5085(87)91013-4. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All the data and materials related to the current study are available from the corresponding author on reasonable request.