Abstract
This investigation evaluated immunity to vaginal herpes simplex virus type 2 (HSV-2) infection after local or parenteral immunization with attenuated HSV-2. Vaginal immunization induced sterilizing immunity against challenge with a high dose of wild-type virus, whereas parenteral immunizations protected against neurologic disease but did not entirely prevent infection of the vagina. Vaginal immunization caused 86- and 31-fold increases in the numbers of immunoglobulin G (IgG) plasma cells in the vagina at 6 weeks and 10 months after immunization, whereas parenteral immunizations did not increase plasma cell numbers in the vagina. Vaginal secretion/serum titer ratios and specific antibody activities in vaginal secretions and serum indicated that IgG viral antibody was produced in the vagina and released into vaginal secretions at 6 weeks and 10 months after vaginal immunization but not after parenteral immunizations. In contrast to the case for plasma cells, the numbers of T and B lymphocytes in the vagina were similar in vaginally and parenterally immunized mice. Also, lymphocyte numbers in the vagina were markedly but similarly increased by vaginal challenge with HSV-2 in both vaginally and parenterally immunized mice. Lymphocyte recruitment to the vagina after virus challenge appeared to involve memory lymphocytes, because it was not observed in nonimmunized mice. Thus, local vaginal immunization with attenuated HSV-2 increased the number of IgG plasma cells in the vagina and increased vaginal secretion/serum titer ratios to 3.0- to 4.7-fold higher than in parenterally immunized groups but caused little if any selective homing of T and B lymphocytes to the vagina.
An understanding of the immune mechanisms that protect the female genital tract against infections in animal models is essential for development of vaccines to protect women against sexually transmitted diseases (35). A mouse model of immunity against vaginal herpes simplex virus type 2 (HSV-2) infection has been described by McDermott and coworkers (22) and modified by Parr et al. (32). In this model, vaginal immunization with attenuated HSV-2 elicits immunity against a subsequent vaginal challenge with wild-type virus. The protective immunity in this model is quite strong (34). Twenty-four hours after immune mice were challenged in the vagina with wild-type virus, infection of the vaginal epithelium ranged from 1.0 to 2.5% of that measured in nonimmune mice, and at 72 h after vaginal challenge, no shed virus protein was detected in the vaginal lumen of immune mice whereas shed virus protein titers of 5,000 to 6,000 were present in nonimmune mice. No immune mice developed neurologic illness, whereas nearly all nonimmune mice died 8 to 14 days after challenge. The dose of challenge virus used in these studies was 1,000-fold higher than the minimum needed to cause lethal illness in nonimmune mice; thus, vigorous immunity was needed to suppress the challenge infections so effectively.
Antibody in vaginal secretions is an important component of immunity to vaginal HSV-2 infection. McDermott et al. (20) and Milligan and Bernstein (24) first demonstrated immunoglobulin G (IgG) antibodies specific for HSV-2 in vaginal secretions of young immune mice; antiviral IgA either was not detected or was detected at very low titers. We subsequently measured IgG viral antibody in vaginal secretions of adult immune mice at a mean enzyme-linked immunosorbent assay (ELISA) titer of 6,200, whereas the mean titer of viral secretory IgA (S-IgA) in the same secretions was 1.9 (30). The protective role of IgG and S-IgA in the vaginal secretions was investigated by neutralization and passive-transfer experiments (29). Affinity-purified IgG from vaginal secretions of adult immune mice, at its concentration in vivo in the vaginal mucus, effectively neutralized HSV-2, whereas S-IgA in the same secretions had little or no effect. Purified IgG from sera of immune mice provided significant protection against epithelial infection after passive transfer to nonimmune mice, even though the mean IgG anti-HSV-2 titers in sera and vaginal secretions of recipient mice at the time of challenge were only 29 and 7%, respectively, of the mean titers in standards prepared from actively immunized mice. The data indicated that IgG viral antibody in vaginal secretions of immune mice provided early protection against challenge infection by neutralizing virus in the vaginal lumen, whereas viral S-IgA contributed relatively little to protection.
A potential involvement of cell-mediated immunity in the mouse vaginal HSV-2 model was first indicated by the observation that adoptive transfer of lymphocytes from the iliac lymph nodes of immune mice protected naive mice against neurologic illness after vaginal challenge with wild-type virus (21). We have further investigated the role of T cells in vaginal immunity by in vivo depletion of these cells in immune mice 1 week before vaginal challenge (34). Depletion of T cells for this short period had no effect on antibody titers in vaginal secretions at the time of challenge. The results showed that immune mice depleted of CD4+ and CD8+ cells, Thy-1+ cells, or CD8+ cells alone had greater viral infection of the vaginal epithelium than did nondepleted immune mice. The T cells of immune mice thus inhibited infection of the vaginal epithelium within 24 h after inoculation of challenge virus.
The results summarized above indicate that vaginal immunization of mice with attenuated HSV-2 elicits a strong protective immune response in the vagina, consisting of T-cell immunity and viral IgG antibody in vaginal secretions. Parenteral immunization with viruses also typically stimulates both vigorous T-cell immunity and high titers of anti-viral IgG. It was thus of interest to ask whether parenteral immunization with attenuated HSV-2 would protect against vaginal challenge infection as effectively as vaginal immunization. Typically, levels of antiviral antibody and resistance to infection are greater at sites of antigenic stimulation than at distant sites; hence, immunity against virus infection is achieved most successfully by immunization at the sites that are directly involved in virus infection (25). However, the basis of enhanced immune protection at sites of mucosal immunization is generally thought to be local production of specific S-IgA antibody, and vaginal immunization with HSV-2 induces mainly IgG viral antibody. Hence, it is unclear at present whether local vaginal immunization with HSV-2 would protect against reinfection any better than parenteral immunization. An answer to this question is critically important for the development of vaccines to protect the female genital tract against infections. The present studies were therefore designed to compare the immunity resulting from local immunization in the vagina to that resulting from immunization at three parenteral sites and in particular to determine whether vaginal immunization caused either local production of IgG viral antibody or selective homing of memory lymphocytes to the vagina.
MATERIALS AND METHODS
Animals and virus.
Female BALB/c mice were purchased from Harlan/Sprague-Dawley, Indianapolis, Ind., and were 10 weeks old at the beginning of treatment. They were housed in compliance with all institutional and federal animal welfare requirements, and all experimental procedures were approved by the institutional Animal Care and Use Committee. Wild-type HSV-2 and attenuated HSV-2, a strain that contains a partial deletion of the thymidine kinase gene, as well as HSV-2-infected Vero cell lysates and uninfected Vero cell lysates, were generously provided by Mark McDermott, McMaster University, Hamilton, Canada (22).
Experimental design.
One hundred seventy-five age-matched mice were allocated to seven groups of 25 mice each. Four groups were used to study immunity at 6 weeks after immunization in the vagina, peritoneal cavity, footpads, or pelvic presacral space (42). The fifth group served as a nonimmunized control for these groups. The sixth group was immunized in the vagina as described above, but its immunity was assessed 10 months later; the seventh group was a nonimmunized control for group 6. The mice were immunized with attenuated HSV-2 as follows: vagina, 20 μl at 1.5 × 106 PFU/ml; peritoneal cavity, 200 μl at 1 × 106 PFU/ml; footpads, 20 μl at 7 × 106 PFU/ml into each hind footpad; and pelvis, 30 μl at 7 × 106 PFU/ml. In preliminary studies these doses of attenuated virus were observed to elicit similar serum antibody titers in all immunized groups. Mice to be immunized in the vagina were pretreated with 0.10 μg of estradiol benzoate in peanut oil subcutaneously, followed 1 day later by 2.0 mg of Depo-Provera (DP) (Upjohn Co., Kalamazoo, Mich.) in phosphate-buffered saline (PBS) subcutaneously. These mice were immunized in the vagina 6 days after treatment with DP (32). Parenterally immunized mice were not pretreated with steroids.
Each group of 25 mice was subdivided into two groups of 10 and one group of 5. One group of 10 mice was examined without vaginal challenge by wild-type HSV-2. From these mice we collected vaginal secretions and sera for measurements of IgG concentrations and IgG anti-HSV-2 titers, vaginae for plasma cell and lymphocyte counts, and uteri for plasma cell counts. The second group of 10 mice was examined 24 h after vaginal challenge with wild-type HSV-2; from these mice we collected vaginae for measurements of percent epithelial infection and for plasma cell and lymphocyte counts. The remaining group of five mice was examined for signs of illness 8 to 14 days after vaginal challenge. Mice that were challenged with HSV-2 in the vagina were pretreated 7 and 6 days previously with estradiol and DP as described above and then challenged with 20 μl of wild-type HSV-2 at 107 PFU/ml.
Vaginal secretions and serum.
Vaginal washes were collected once daily on the fifth, sixth, and seventh days after the last DP treatment as previously described (29). Vaginal washes were centrifuged at 12,000 × g for 10 min to separate the mucus from the PBS wash solution shortly after collection. The mucus and supernatant were frozen separately at −20°C. The PBS wash solution contained a cocktail of proteinase inhibitors (28). Blood was collected by cardiac puncture under tribromoethanol anesthesia; serum was obtained by standard methods.
Extraction of immunoglobulin from vaginal mucus.
Mucus samples were thawed, pooled for each mouse, weighed, and extracted twice for 2 h each in 300 μl of PBS with rotation in a 12-ml polystyrene tube at 4°C. The two extracts and the original wash supernatants were pooled, made up to 900 μl, and frozen at −20°C until needed. This method provided essentially complete recovery of S-IgA and presumably also IgG from the mucus (30).
Measurement of IgG concentrations by ELISA.
Capture antibody (goat anti-mouse IgG[γ] [Sigma Chemical Co., St. Louis, Mo.] at 5 μg/ml) was bound to Immulon 1 (Dynatech Laboratories, Alexandria, Va.) microtiter plate wells overnight in 0.10 M carbonate buffer at pH 9.5. After being washed in PBS–0.05% Tween 20, plate wells were blocked for 30 min with 2% normal goat serum in PBS–Tween 20. Serial twofold dilutions of samples and immunoglobulin standards in duplicate in blocking medium were then applied to the wells overnight in a humid chamber, followed the next day by washing in PBS–Tween 20. Bound immunoglobulins were detected with horseradish peroxidase–goat anti-mouse IgG(γ) (Jackson Immunoresearch Laboratories, West Grove, Pa.) in PBS– Tween 20, followed by washing and incubation in tetramethylbenzidine substrate. The IgG standard was purchased from Sigma Chemical Co. Its purity was confirmed by biotinylation of the protein, followed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis under reducing conditions and immunoblotting. The only biotinylated polypeptides detected corresponded to heavy and light chains. The concentration of the standard was determined by absorbance at 280 nm, using E2801% = 14.0.
Specific viral antibodies.
Microtiter plate wells (Falcon Pro-Bind no. 3915; Becton-Dickinson and Co., Lincoln Park, N.J.) were filled with 100 μl of UV-inactivated lysate of HSV-2-infected Vero cells in carbonate buffer at pH 9.5, covered with sealing film, centrifuged at 2,700 rpm for 2 h in a Beckman GS-6R centrifuge, and then incubated overnight at 4°C. The next day, plate wells were washed with PBS–0.05% Tween 20 and blocked for 30 min with 2% normal goat serum in PBS–Tween 20. Serial twofold dilutions of samples in blocking medium were then placed in the wells and incubated overnight in a humid chamber. After being washed in PBS–Tween 20, the wells received horseradish peroxidase–goat anti-mouse IgG(γ) (Jackson Immunoresearch Laboratories) in PBS–Tween 20 for 2 h, followed by washing and addition of tetramethylbenzidine substrate. Reactions were stopped with 1.0 M sulfuric acid, and absorbances were measured at 450 nm. The sample antibody titer was defined as the reciprocal of the sample dilution at which the absorbance declined to 1.0, which was in the central, most linear part of the dilution curve. Control experiments demonstrated that background reaction absorbances were 0.05 or less when immune and nonimmune samples were incubated on lysates of uninfected or infected Vero cells, respectively. Serum or vaginal secretion samples from the vaginal immunization group at 6 weeks were included each time samples from other groups were measured. Titers of other groups were thus always measured in direct comparison to vaginal group samples and are stated as a fraction of the geometric mean titer in the vaginal group. Vaginal secretion/serum titer ratios for each mouse were measured side by side on the same microtiter plate to minimize error in this measurement.
Tissues.
Vaginae and uteri were fixed with 2% paraformaldehyde in 0.1 M phosphate buffer, pH 7.4 (4°C, 2 h), and washed with PBS containing 10% sucrose (4°C, 2 h). Tissues were embedded in O.C.T. (Tissue-Tek; Miles Scientific, Naperville, Ill.), frozen in isopentane cooled with liquid nitrogen, and stored at −70°C until needed. Cryostat sections (5 μm) were mounted on polylysine-coated slides, air dried, and stored with desiccant at −20°C until needed.
Quantitation of epithelial infection.
Cryostat sections of vagina were postfixed in methanol, blocked (30 min) in 2% normal goat serum, and labeled (60 min) with fluorescein isothiocyanate (FITC)–rabbit IgG anti-HSV-2 (Dako Corp., Carpinteria, Calif.). The specificity of labeling was confirmed by labeling normal and infected cell cultures (Ortho Diagnostic Systems, Inc., Raritan, N.J.), by labeling vaginal sections from infected and noninfected mice, and by using FITC-normal rabbit IgG. The percentage of vaginal epithelium infected by HSV-2 was determined with an image analysis system. A high-resolution RGB color camera with integration (AIC-O-VI 470, Hyper HAD CCD; Optronics Engineering, Goleta, Calif.) was attached to the fluorescence microscope. Captured images were analyzed by using a MacIntosh computer (Quadra 840 AV) equipped with the LG-3 frame grabber (Scion Corp., Frederick, Md.) and with National Institutes of Health Image 1.55 (Wayne Rasband, National Institutes of Health). The lengths of HSV-2-labeled segments of vagina were measured in four histological sections, each derived from a different region of the vagina. The total length of vaginal epithelium was measured after staining with hematoxylin and eosin. The mean percentage of HSV-2-infected epithelium in each group was calculated, and the statistical significance of differences was evaluated by Student’s t test.
Quantitation of plasma cells and lymphocytes.
Cryostat sections were postfixed for 10 min in acetone, blocked for 30 min in 2% goat serum, and incubated for 1 h in one of the following primary antibodies: rabbit anti-mouse IgA(α) or rabbit anti-mouse IgG(γ) (Jackson Immunoresearch Laboratories), rat anti-mouse CD4 or CD8 (Becton Dickinson, Mountain View, Calif.), or rat anti-mouse B220 (American Type Culture Collection, Rockville, Md.). The secondary antibody was FITC–goat anti-rabbit IgG (Jackson Immunoresearch Laboratories) or FITC–donkey anti-rat IgG (Chemicon International, El Segundo, Calif.). Specificity of labeling was indicated by the absence of staining when normal rabbit or rat IgG was substituted for the primary antibody.
Plasma cells were counted in one complete cross section from each of four separate regions of the vagina and uterus from each mouse. Lymphocytes (CD4+, CD8+, and B220+) were counted in four randomly selected high-power fields from each of two separate regions of the vagina. The stained cells in captured images were counted with the image analysis system described above. The numbers of CD8+ lymphocytes in measured lengths of the vaginal epithelium were also counted as described above.
Quantitation of major histocompatibility complex (MHC) class II staining in vaginal epithelium.
Cryostat sections of vagina were stained as described above for lymphocytes, using rat anti-mouse Ia (Boehringer-Mannheim, Indianapolis, Ind.). The intensity of staining in coded sections was evaluated and recorded as nil, weak, moderate, or bright, along with the approximate length of epithelium with each kind of staining. All staining was evaluated in captured images. Overall staining of the epithelium was then ranked from 0 to 3 for each mouse, and group means were calculated.
Illness scores.
Illness was indicated by ruffled fur, arched backs, feeble movements, paralysis of one or both hind limbs, and a swollen red vulva. Illness usually, but not always, led to death or euthanasia. An illness score of 3.0 was assigned to mice that died or became so ill that euthanasia was desirable by 9 days after inoculation of wild-type virus. Mice that died or required euthanasia from 10 to 14 days after infection were scored 2.0. Mice that developed some sign of illness but survived beyond 14 days were scored 1.0. Mice that never showed signs of illness were scored 0.0.
Statistics.
The statistical significance of the results was evaluated by Student’s t test, the chi-square test, or analysis of variance (ANOVA) as appropriate and is reported in the tables and figure legends.
RESULTS
Antibody responses and protection.
Immunization with attenuated HSV-2 at both vaginal and parenteral sites elicited IgG viral antibodies that were detected 6 weeks later in both sera and vaginal secretions (Table 1). Geometric mean IgG titers were 1.7- to 2.4-fold higher in sera and 4.7- to 18-fold higher in vaginal secretions of vaginally immunized mice than in the parenteral immunization groups. No infection of the vaginal epithelium was detected in any of the mice in the vaginal immunization group 24 h after vaginal challenge with wild-type virus (Table 1). In contrast, epithelial infection was found in all nonimmunized mice and in most parenterally immunized mice after challenge. The percentage of the vaginal epithelium that was infected was 12 ± 2% in the nonimmune group and ranged from 0.5 ± 0.1 to 1.9 ± 0.6% in the parenteral immunization groups. The highest level of epithelial infection in parenterally immunized mice was significantly lower than that in nonimmunized mice, indicating that parenteral immunizations induced immunity against vaginal challenge. The lowest level of epithelial infection in parenterally immunized mice was significantly higher than that in vaginally immunized mice, indicating that vaginal immunization provided the strongest protection against epithelial infection. All immunized mice, those immunized both vaginally and parenterally, were completely protected against the neurologic illness that developed in nonimmune mice after vaginal challenge with the wild-type virus.
TABLE 1.
Immunity to vaginal challenge 6 weeks after vaginal or parenteral immunization with attenuated HSV-2
| Site of immunization | Geometric mean titer of IgG anti-HSV-2 in:
|
Infection of vaginal epithelium
|
Illness score | ||
|---|---|---|---|---|---|
| Serum (104) | Vagina (102) | No. of micea | %b (mean ± SEM) | ||
| None | 10/10 | 12 ± 2 | 3.0 | ||
| Vagina | 20 | 15 | 0/10 | 0.0 | 0.0 |
| Peritoneum | 8.6 | 2.2 | 9/10 | 1.9 ± 0.6 | 0.0 |
| Footpad | 9.4 | 0.84 | 6/7 | 1.5 ± 0.4 | 0.0 |
| Pelvis | 12 | 3.2 | 8/9 | 0.50 ± 0.1 | 0.0 |
Infection was identified by immunostaining of HSV-2 proteins in equivalent total lengths of vaginal epithelium in four independent sections from each mouse 24 h after challenge. The number of infected mice in the vaginal immunization group was significantly lower than those in the other groups (P < 0.0001; chi-square test).
The percentage of vaginal epithelium that was HSV-2 infected at 24 h after challenge was significantly lower in the peritoneally immunized group than in the nonimmunized group (P = 0.0001; one-tailed t test) and was significantly lower in the vaginal immunization group than in the pelvic immunization group (P = 0.0006; one-tailed t test).
Local IgG production.
Vaginal secretion and serum IgG anti-HSV-2 titers in each mouse were measured side by side on the same microtiter plate so that accurate vaginal secretion/serum titer ratios could be obtained. The mean vaginal secretion/serum titer ratio in the vaginal immunization group was 3.0- to 4.7-fold higher than that in the parenteral immunization groups (Table 2). The high titer ratio in vaginally immunized mice could have been due to increased transudation of serum IgG into the vaginal secretions. However, increased transudation would have increased the concentration of IgG in the vaginal secretions. The data in Table 2 indicate that the vaginal IgG concentration in vaginally immunized mice was higher than that in parenterally immunized mice but was not high enough to account for the high titer ratio. The high titer ratio might also be due to local production and secretion of virus-specific IgG in the vagina. In this case, the titer per unit of IgG concentration (specific antibody activity) in vaginal secretions would be higher than that in serum. The vaginal secretion/serum ratios of specific antibody activities were not significantly different from 1.0 in the parenteral immunization groups, indicating that IgG anti-HSV-2 titers per unit of IgG were the same in vaginal secretions and serum and that vaginal IgG was derived mainly from serum (Table 2). In contrast, the specific antibody activity ratio in the vaginal immunization group was 2.0 ± 0.2. This was significantly larger than 1.0 and indicated that specific viral IgG was produced in the vagina and released into vaginal secretions in amounts sufficient to double the virus-specific activity of the IgG in vaginal secretions in comparison to serum.
TABLE 2.
Concentrations of IgG and vaginal secretion/serum IgG anti-HSV-2 titer ratios 6 weeks after immunization with attenuated HSV-2
| Site of immunization | Conc. of IgG (mean ± SEM) in:
|
Vaginal secretion/serum ratio (mean ± SEM)
|
||
|---|---|---|---|---|
| Serum (mg/ml) | Vagina (μg/ml) | Titera (10−3) | Titer per μg of IgG per mlb | |
| None | 0.70 ± 0.11 | 1.1 ± 0.14 | ||
| Vagina | 1.4 ± 0.19 | 5.8 ± 0.75 | 9.9 ± 2 | 2.0 ± 0.2 |
| Peritoneum | 1.3 ± 0.12 | 4.5 ± 0.54 | 3.3 ± 0.7 | 1.1 ± 0.1 |
| Footpad | 1.5 ± 0.13 | 2.6 ± 0.53 | 2.1 ± 0.5 | 1.2 ± 0.07 |
| Pelvis | 1.5 ± 0.13 | 5.6 ± 1.2 | 2.9 ± 0.3 | 0.94 ± 0.07 |
The mean vaginal secretion/serum IgG anti-HSV-2 titer ratio in the vaginal immunization group was significantly higher than that in any other group (P < 0.0001; four-group ANOVA).
The mean vaginal secretion/serum ratio of IgG anti-HSV-2 titer per unit IgG concentration (micrograms per milliliter) was significantly larger than 1.0 in the vaginal immunization group but not in the other groups (two-tailed t tests: vagina, P < 0.0001; peritoneum, P = 0.42; footpad, P = 0.06; pelvis, P = 0.41).
Plasma cells in the vagina.
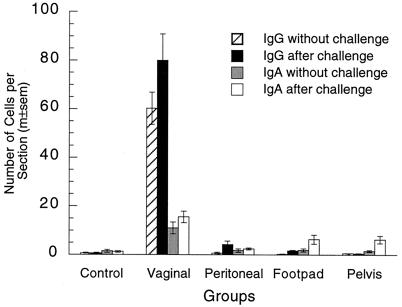
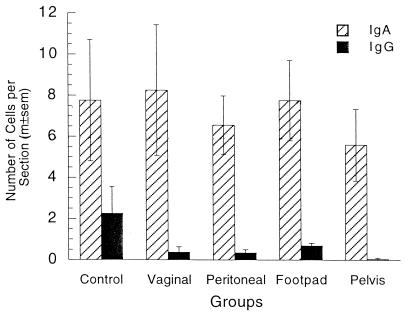
The number of IgG plasma cells in the vagina 6 weeks after vaginal immunization was 86-fold larger than that in the nonimmunized group (Fig. 1 and 2), while the numbers of IgG cells in the parenteral immunization groups were not significantly increased in comparison to those in the nonimmunized mice. Similarly, the number of IgA plasma cells in the vagina was sevenfold larger in vaginally immunized mice than in nonimmunized mice, but there was no increase in that number in parenterally immunized mice. The numbers of both kinds of plasma cells in the vagina 24 h after challenge were also much larger in vaginally immunized mice than in any other groups (Fig. 1). To determine whether the increase in plasma cell numbers was restricted to the site of immunization, we counted such cells in uteri of all groups (Fig. 3). In contrast to the case for the vagina, most uterine plasma cells contained IgA. Vaginal immunization did not significantly increase the number of IgA plasma cells in the uterus, and all immunizations reduced the number of IgG plasma cells in the uterus.
FIG. 1.
Plasma cells in vaginae of locally and parenterally immunized mice. The numbers of IgG and IgA plasma cells in the vagina were significantly larger in the vaginal immunization group than in the other four groups with or without vaginal challenge (P < 0.0001 in all four tests; five-group ANOVAs). Plasma cell numbers in the control and parenteral immunization groups were not significantly different without vaginal challenge (IgG, P = 0.49; IgA, P = 0.97; four-group ANOVAs). After vaginal challenge, plasma cell numbers in the parenteral immunization groups were significantly larger than those in the control group in all cases except IgG cells in the pelvic group (IgG, P = 0.0069; IgA, P = 0.0029; four-group ANOVAs), and the numbers of these cells in the parenteral immunization groups after challenge were also significantly larger than the numbers without challenge in the majority of cases (P < 0.05; two-tailed t tests). m±sem, mean ± standard error of the mean.
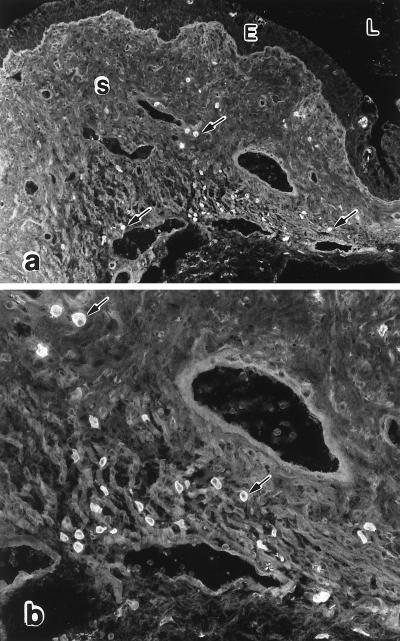
FIG. 2.
IgG plasma cells in vaginae of vaginally immunized mice. Fluorescent staining of IgG plasma cells (arrows) in a vaginal section from a mouse that was immunized in the vagina with attenuated HSV-2 6 weeks previously is shown. The cells tended to be most numerous in the periphery of the vagina and often occurred in groups. All of the cells identified as plasma cells contained cytoplasmic IgG(γ) or IgA(α). Some had the classical appearance of large ovoid cells with an eccentric nucleus, while others appeared to be smaller and to have only a thin rim of cytoplasm. It is not clear whether the latter cells were typical plasma cells in a plane of section through the nuclear side of the cell or whether they were immature plasma cells (plasmablasts). E, epithelium; L, lumen; S, stroma. Magnifications, ×121 (a) and ×315 (b).
FIG. 3.
Plasma cells in uteri of locally and parenterally immunized mice. Most plasma cells in the uterus contained IgA. The numbers of these cells were not significantly different in any of the groups (P = 0.90; five-group ANOVA). The number of IgG plasma cells in the uterus was significantly larger in the control group than in any of the immunized groups (P = 0.0036; five-group ANOVA). m±sem, mean ± standard error of the mean.
Lymphocytes in the vaginal mucosa.
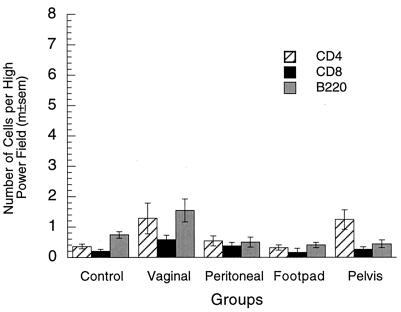
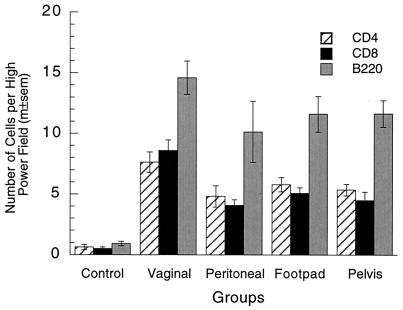
The increased number of plasma cells in the vagina after vaginal immunization suggested that the numbers of other lymphoid cells might also be increased in the vagina by local immunization. Few lymphocytes were present in the vaginal mucosae of immunized or nonimmunized mice without vaginal challenge (Fig. 4). Lymphocyte numbers in the vaginal immunization group tended to be somewhat larger than those in the nonimmunized and parenterally immunized groups, but not all of the differences were statistically significant. After vaginal challenge, lymphocyte numbers in the immunized groups were 5- to 10-fold higher than those before challenge and also 5- to 10-fold higher than those in nonimmunized mice after challenge (Fig. 5). Vaginal challenge did not increase lymphocyte numbers in nonimmunized mice. The observations indicate that T- and B-lymphocyte numbers were rapidly increased in the vaginal mucosae of immunized mice after vaginal challenge, either by stimulation of resident memory lymphocytes or by recruitment of memory lymphocytes to the vagina from the blood. Among the four immunized groups after challenge, lymphocytes were marginally more numerous in vaginally immunized mice than in parenterally immunized mice.
FIG. 4.
Lymphocytes in vaginae of locally and parenterally immunized mice without challenge. Lymphocyte numbers (CD4+, CD8+, and B220+) in the vaginal mucosa without challenge were somewhat larger in vaginally immunized mice than in control or parenterally immunized mice, but the statistical significance of the differences was marginal (CD4, P = 0.035; CD8, P = 0.068; B220, P = 0.0024; five-group ANOVAs). m±sem, mean ± standard error of the mean.
FIG. 5.
Lymphocytes in vaginae of locally and parenterally immunized mice after challenge. Vaginal lymphocyte numbers in immunized groups after vaginal challenge were 5- to 10-fold higher than those before challenge (P <0.0001 in each case; two-tailed t tests) and also 5- to 10-fold higher than those in the nonimmunized group (P < 0.0001 in all three tests; five-group ANOVAs). Lymphocyte numbers in nonimmunized mice were not significantly increased by challenge (P > 0.05 in all three tests; two-tailed t test). Lymphocyte numbers in vaginally immunized mice were somewhat larger than those in parenterally immunized mice, but the differences were not all significant (CD4, P = 0.058; CD8, P < 0.0001; B220, P = 0.29; four-group ANOVAs). m±sem, mean ± standard error of the mean.
Lymphocytes in the vaginal epithelium.
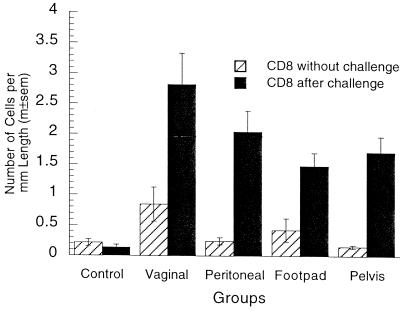
Few CD8+ T cells were present in the vaginal epithelia of immunized or nonimmunized mice without vaginal challenge (Fig. 6). The number of these cells in vaginally immunized mice was modestly but significantly larger than those in the other groups. After vaginal challenge, the numbers of CD8+ cells in the immunized groups were 3- to 10-fold higher than those before challenge and 10- to 20-fold higher than those in the nonimmunized group after challenge. Vaginal challenge did not increase the number of CD8+ cells in the epithelia of the nonimmunized mice. More CD8+ cells were observed in the epithelia of vaginally immunized mice after challenge than in parenterally immunized mice, but the difference was not statistically significant. Collectively, the data indicate that the numbers of CD8+ cells in the vaginal epithelium closely mirrored the numbers of CD4+, CD8+, and B220+ cells in the vaginal mucosa.
FIG. 6.
CD8+ T cells in the vaginal epithelia of locally and parenterally immunized mice with and without challenge. The number of CD8+ cells in the vaginal epithelia of vaginally immunized mice without challenge was significantly larger than those in the other groups (P = 0.010; five-group ANOVA). After challenge, the numbers of CD8+ cells in the immunized groups were 3- to 10-fold larger than those before challenge (P < 0.0001 in all four tests; two-tailed t tests) and 10- to 20-fold larger than those in the nonimmunized group (P < 0.0001; five-group ANOVA). The numbers of CD8+ cells in nonimmunized mice with and without vaginal challenge were not significantly different (P = 0.30; two-tailed t test). The number of CD8+ cells after challenge was higher in vaginally immunized mice than in parenterally immunized mice, but the difference was not statistically significant (P = 0.074; four-group ANOVA). m±sem, mean ± standard error of the mean.
Expression of MHC class II antigen in vaginal epithelium.
Fluorescent staining of MHC class II antigens was bright on Langerhans cells within the vaginal epithelial layer of nonimmune mice 24 h after vaginal challenge, but the epithelial cells were unstained. In contrast, vaginally immunized mice exhibited maximal (3.0) staining of MHC class II antigens in vaginal epithelial cells 24 h after challenge, at which time the staining of Langerhans cells was entirely obscured by the epithelial staining. Staining of vaginal epithelial cells in parenterally immunized mice after vaginal challenge was intermediate: 1.2, 1.6, and 2.0 in the peritoneal, footpad, and pelvic groups, respectively. Upregulation of MHC class II antigens in the vaginal epithelium thus correlated with the increased numbers of T cells in the vaginae of immunized mice after vaginal challenge.
Duration of immunity.
Immunization with live viruses typically stimulates long-lasting immunity. At 10 months after a single vaginal immunization with attenuated HSV-2, serum and vaginal secretion IgG anti-HSV-2 titers declined to about 20 to 30% of values measured at 6 weeks after immunization (Table 3). Similarly, the vaginal secretion/serum titer ratio, the titer ratio per unit of IgG concentration (micrograms per milliliter), and early protection against epithelial infection were each reduced in comparison to values measured at 6 weeks after immunization. However, the vaginal secretion/serum titer ratio in vaginally immunized mice at 10 months was still significantly higher than that in parenterally immunized mice at 6 weeks, and the vaginal/serum ratio of specific antibody activities was still significantly greater than 1.0, both observations indicating that local secretion of virus-specific IgG still occurred in the vagina 10 months after local immunization. The numbers of IgG and IgA plasma cells in the vagina remained elevated, especially after vaginal challenge (Fig. 7). Moreover, challenge virus increased the numbers of T and B lymphocytes in the vagina (Fig. 8) and upregulated expression of MHC class II antigens to a level of 1.2 in the epithelia of immunized mice but not in nonimmunized mice, indicating that substantial numbers of memory lymphocytes were still present. None of the immunized mice developed neurologic illness after challenge, whereas all nonimmunized mice died within 10 days.
TABLE 3.
Immunity to vaginal challenge 10 months after vaginal immunization with attenuated HSV-2
| Site of immunization | Concn of IgG (mean ± SEM) in:
|
Geometric mean titer of IgG anti-HSV-2 in:
|
Vaginal secretion/serum ratioa (mean ± SEM)
|
Infection of vaginal epithelium
|
||||
|---|---|---|---|---|---|---|---|---|
| Serum (mg/ml) | Vagina (μg/ml) | Serum (104) | Vagina (102) | Titer (10−3) | Sp act | No. of mice | %b (mean ± SEM) | |
| Vagina | 1.8 ± 0.09 | 7.1 ± 0.81 | 6.2 | 3.2 | 6.8 ± 1.5 | 1.3 ± 0.10 | 9/10 | 1.4 ± 0.7 |
| None | 1.1 ± 0.10 | 2.6 ± 0.40 | 10/10 | 6.4 ± 1.2 | ||||
The vaginal secretion/serum titer ratio was significantly larger than the titer ratios for the peritoneal, footpad, and pelvic groups at 6 weeks after immunization (P = 0.023; four-group ANOVA), and the vaginal secretion/serum ratio of specific antibody activities was significantly larger than 1.0 (P = 0.014; one-tailed t test).
The percentage of vaginal epithelium that was infected in the vaginally immunized group was significantly lower than that in the nonimmunized control group (P = 0.0013; one-tailed t test).
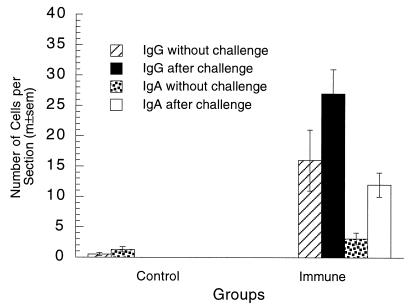
FIG. 7.
Plasma cells in vaginae of immune and control mice 10 months after vaginal immunization. Vaginal IgG and IgA plasma cell numbers 10 months after vaginal immunization were significantly larger than those in nonimmunized control mice (P < 0.0001 in each test; two-tailed t tests). These numbers were further increased after vaginal challenge. Plasma cell counts were not done in control mice after challenge. m±sem, mean ± standard error of the mean.
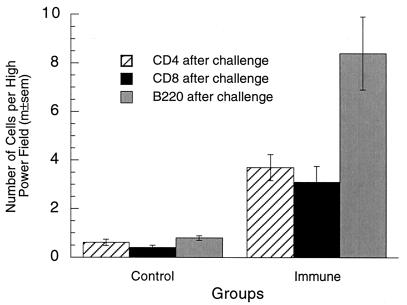
FIG. 8.
Lymphocytes in vaginae of immune and control mice 10 months after vaginal immunization. Vaginal lymphocyte numbers after vaginal challenge were 6- to 10-fold higher in mice that were immunized in the vagina 10 months previously than in nonimmune control mice (P < 0.0001 in all three tests; two-tailed t tests). m±sem, mean ± standard error of the mean.
DISCUSSION
Vaginal immunization of adult mice with attenuated HSV-2 has previously induced strong immunity against vaginal challenge infection with wild-type HSV-2 (32, 34). In the present study this immunization induced sterilizing immunity against a challenge inoculum that was 1,000-fold larger than the minimum needed to cause lethal illness in nonimmune mice. This is arguably the strongest immunity yet observed against an infection of the female genital tract (35), and it suggests that local immunization in the genital tract with attenuated or recombinant live virus vaccines may be an effective means to elicit immunity against sexually transmitted pathogens and sperm.
The effectiveness of vaginal immunization with attenuated HSV-2 derives from a confluence of three key factors. First, highly immunogenic quantities of virus antigen are able to cross the epithelial barrier and reach lymphoid cells and lymphatic vessels in the vaginal stroma, because the virus penetrates the epithelium and replicates throughout the epithelial layer. Penetration of virus into the epithelial layer is facilitated by pretreatment of the mice with a progestin, which thins and transforms the epithelium and increases its permeability to exogenous proteins (32). Similarly, we have found in preliminary studies that equivalent immunization as judged by viral antibody titers in serum can be achieved in estradiol-treated mice by vaginal inoculation of virus in conjunction with scarification of the vaginal epithelium. In contrast, nonreplicating and/or noninvasive antigens in the vagina produce only weak immune responses. This is evidenced by weak responses to nonreplicating protein antigens (12, 26, 27, 43) and weak responses to natural infections by replicating but noninvasive pathogens that colonize the superficial epithelial cells of the reproductive tract for weeks or months before elimination by an immune response (35). The latter organisms include Campylobacter fetus in cows (10); trichomonas species in cows (8, 9), mice (1), and humans (1); Candida albicans in rats and mice (7, 13); and papillomaviruses in animals and humans (6, 11). Chlamydial infection of the female genital tract in mice and guinea pigs also elicits only a modest convalescent immunity (39), which may be due in part to release of progeny organisms mainly into the genital tract lumen rather than the stroma.
Second, the effectiveness of vaginal immunization with attenuated HSV-2 is typical of the strong immunity that is induced by live-virus vaccination in general (2). Live viruses preserve conformation-dependent antigenic epitopes that are frequently the primary targets of neutralizing antibodies, and they selectively induce IgG antibodies of the IgG2a subclass in mice and of the IgG1 and IgG3 subclasses in humans (40). These complement-binding subclasses can be particularly effective in neutralization. For example, complement greatly increased neutralization of HSV-2 by virus-specific IgG2a monoclonal antibodies, and passive administration of IgG2a monoclonal antibodies to mice was much more protective against HSV-2 challenge than equal amounts of IgG1 monoclonal antibodies (15). The mouse IgG2a and human IgG1 and IgG3 subclasses also bind maximally to the high-affinity FcR type 1 on neutrophils and macrophages (40), making these subclasses the most effective opsonizing antibodies for phagocytosis. Live-virus antigens combine effectively with MHC molecules to elicit vigorous T-cell immunity, and they present sufficiently diverse B- and T-cell epitopes to overcome the genetic diversity of immune responses among individuals.
Third, the present study revealed that vaginal immunization with attenuated virus caused an 86-fold increase in the number of IgG plasma cells in the vaginal mucosa, correlated with a 3.0- to 4.5-fold elevation in the secretion/serum ratio of IgG anti-HSV-2 titers in comparison to that in parenterally immunized mice, whose vaginal plasma cell numbers were not increased. The main neutralizing antibody in the vaginal secretions of immune mice has previously been shown to be IgG (29). While most of the IgG plasma cells were localized in the periphery of the mucosa rather than near the epithelium, and while we do not have direct evidence of virus-specific cells among the plasma cells, it is likely that specific cells were present and that they account for the observed increase in specific viral antibody in the vaginal secretions. We assume that the IgG reached the vaginal lumen by transudation, which accounts for the presence of IgG in most mucosal secretions, although the cell biological details of this process have never been clarified (31). It is unlikely that the uterus was the source of increased specific IgG in the vagina, because vaginal immunization actually decreased the number of IgG plasma cells in the uterus.
As mentioned above, studies of the immune mechanisms that protect against vaginal HSV-2 infection in this mouse model have indicated that secretory immunity is mediated mainly by IgG antibody rather than IgA. The ELISA titer of IgG viral antibody in vaginal secretions of immune mice was much higher than that of IgA (30) and this IgG antibody neutralized HSV-2 far more effectively than the IgA (29). Vaginal immunization with nonreplicating protein antigens may also elicit mainly IgG rather than IgA antibodies in vaginal secretions (26, 43), as does vaginal infection with simian immunodeficiency virus in rhesus monkeys (23) and cervicovaginal infection with HSV-2 in humans (4). The predominance of IgG antibodies in immune responses after vaginal immunization may be due to the fact that the cervix and vagina are relatively deficient in mucosal lymphoid nodules, where IgA responses are initiated (31). The available evidence also indicates that IgG antibodies in genital tract secretions are mainly responsible for immune protection against genital tract pathogens. In addition to HSV-2, this has been shown for chlamydiae in guinea pigs (38), C. fetus in cows (8), and trichomonas species in cows (8, 9) and mice (1). The evidence of a primary role of secreted IgG antibodies in protection against genital tract infections in females is contrary to the widely accepted paradigm that IgA is the main protective antibody at mucosal surfaces, a discrepancy that has been noted previously by Patton and Rank (36). While the available evidence indicates that IgG is mainly responsible for secretory immunity in the female genital tract, this may result in part from the relative ease of inducing specific IgG in genital secretions, either by local or parenteral immunization. In contrast, a practical vaccination procedure that elicits a long-lasting and predominantly IgA response in the genital tract continues to be elusive. Interestingly, several observations suggest that prolonged or repeated antigenic stimulation in the female genital tract or its draining lymph nodes is associated with increased IgA/IgG antibody ratios (3, 17, 24, 41).
The main protective antibody in intestinal and upper respiratory tract secretions is IgA, and there is currently much interest in immunization at IgA-inductive sites such as the intestine, nasopharynx, and genital lymph nodes to protect the female genital tract against infections (33). These studies have shown that immunization at IgA-inductive sites can induce IgA responses in the female genital tract and can induce some degree of protection against genital tract infections, but at present there is no direct comparative evidence that such immunization is more protective than alternate routes of vaccination (35). Based on the results of the present study, it is unlikely that immunization at IgA-inductive sites would cause the development of IgG plasma cells in the genital tract or the local production of specific IgG antibody that we observed after vaginal immunization. Thus, while immunization at IgA-inductive sites may induce higher IgA titers in genital tract secretions than local immunization, it remains unclear which route of immunization would provide better protection against genital tract pathogens. This question can be answered only by assessments of protective immunity to each genital tract pathogen after local immunization in the genital tract in direct comparison to immunization at IgA-inductive sites. Such comparisons will often be complicated by differences in both the vaccines and the vaccination sites, since an effective vaccine formulation for one site will often not be effective at another site. In the case of HSV-2, the “gold standard” performance of vaginal immunization with attenuated virus in the mouse model should serve as a useful benchmark against which to compare other vaccine formulations and sites of immunization.
The increased numbers of plasma cells in the vagina after vaginal immunization could result from stimulation of resident precursor B cells, but this is unlikely because secondary lymphoid nodules, where B-cell stimulation and early differentiation normally occur, were not present in the vaginal mucosa either before or after immunization (32). Instead, secondary lymphoid follicles appeared in the iliac lymph nodes shortly after vaginal immunization with HSV-2 (30a), and this is the probable source of the B cells that later appeared as plasma cells in the vagina. The migration or homing of plasma cell precursors from the iliac nodes to the vagina was not an inherent property of iliac lymph node cells, since the pelvic immunization used in the present study targets the iliac lymph nodes (42) and induces secondary follicles in them (30a) but did not result in increased numbers of plasma cells in the vagina. Thus, while Lehner et al. (19) have suggested that T cells, B cells, and macrophages from the iliac lymph nodes of rhesus macaques preferentially home to the rectum and ascending colon, we conclude that there was no preferential homing of iliac lymph node plasmablasts to the mouse vagina. It thus appears that plasma cell precursors were recruited to the vagina from the blood by a factor(s) that was present in the vagina after vaginal immunization. The factor(s) might be antigen or a virus-induced chemokine, although no chemokine that attracts plasma cell precursors to tissues is currently recognized (5). If plasma cells are short-lived, the factor(s) must remain active in the vagina and continue recruiting plasmablasts for at least 10 months. Influenza virus infection increased the number of plasma cells in mouse lungs for at least 11 months (16), but plasma cell numbers were not increased in the vagina by vaginal infection with simian immunodeficiency virus (23). The latter observation has implications for vaccination to prevent heterosexual transmission of human immunodeficiency virus type 1 (HIV-1) in humans (35), since it suggests that vaginal immunization with attenuated HIV-1 would not cause local IgG production. Instead, vaginal immunization with a recombinant, epitheliotrophic virus such as HSV-2 expressing HIV-1 antigenic determinants might increase vaginal plasma cell numbers and elicit local production and secretion of neutralizing IgG antibody and thereby provide enhanced protection.
In view of the apparent recruitment of plasma cell precursors to the vagina by local immunization with HSV-2, it was important to investigate whether vaginal immunization also selectively increased the numbers of T and B lymphocytes in the vagina. Lymphocyte numbers in the vaginal mucosa were 1.2- to 3.0-fold larger in vaginally immunized mice than in nonimmunized and parenterally immunized mice. These results are consistent with data from a previous study utilizing vital dye tracing, in which the number of lymphocytes that migrated from the vaginal epithelium to the iliac lymph nodes was 3.5-fold higher in vaginally immunized mice than in nonimmunized mice (18). It is doubtful, however, that the larger number of vaginal lymphocytes in vaginally immunized mice was due to selective lymphocyte homing or stimulation of resident cells. Vaginal immunization induced 1.7- to 2.4-fold-higher viral antibody titers in serum than the three parenteral immunizations and may have caused greater lymphocyte proliferation as well. A larger number of lymphocytes in the mice might account at least in part for larger lymphocyte numbers in the vagina. If vaginal immunization caused selective and long-lasting recruitment of lymphocytes to the vagina before antigen challenge, the effect was small and difficult to distinguish from effects on total lymphocyte numbers in the animal.
Vaginal inoculation of challenge virus causes a rapid accumulation of memory lymphocytes in the vaginal mucosae of immune mice. In the present study the numbers of CD4+, CD8+, and B220+ lymphocytes in the vagina increased 5- to 10-fold within 24 h after vaginally immunized mice were challenged but remained unchanged when nonimmunized mice were challenged. In the vaginal epithelia of these mice, the number of CD8+ cells per unit length increased 3.5-fold in vaginally immunized mice but did not increase in nonimmunized mice. Thus, vaginal inoculation of wild-type HSV-2 increased vaginal lymphocyte numbers within 24 h only when specific memory lymphocytes were present. Previously, we observed that MHC class II antigens in the vaginal epithelium were upregulated within 24 h after vaginally immunized mice were challenged in the vagina with HSV-2 but not when nonimmune mice were similarly challenged (32). This was presumably due to secretion of gamma interferon by memory T cells at the site of their reexposure to antigen in the vagina. Also, studies utilizing vital dye tracing revealed that the numbers of CD4+, CD8+, and B220+ lymphocytes migrating from the vaginal epithelium to the iliac lymph nodes increased 5- to 10-fold during the 24 h after inoculation of challenge virus into the vaginae of vaginally immunized mice (18). Many, if not most, of the migrating T cells expressed the CD44 hi phenotype of mouse memory T cells. The 5- to 10-fold increase in the numbers of migrating cells observed by vital dye tracing corresponds well to the increased vaginal lymphocyte numbers observed in histological sections in the present study. Additionally, depletion of T cells from vaginally immunized mice in vivo by monoclonal antibodies 1 week before vaginal challenge significantly increased infection of the vaginal epithelium 24 h after challenge while having no effect on IgG viral antibody titers in vaginal secretions (34). This suggests that the memory T cells that accumulate in the vaginae of vaginally immunized mice during the 24 h after vaginal challenge are functionally important in immune protection of the epithelium.
The apparent recruitment of memory lymphocytes to the vagina by vaginal challenge in locally immunized mice raises the question whether such recruitment would occur after parenteral immunization. In the present study we found that vaginal lymphocyte numbers were markedly increased within 24 h after vaginal challenge in both vaginally and parenterally immunized mice but not in nonimmunized mice, indicating that memory lymphocytes were rapidly recruited to the vagina after challenge in immune mice irrespective of the site of immunization. Lymphocyte numbers were larger in the vaginally immunized mice but only by twofold or less. As in the case of immune mice without challenge, the larger numbers of vaginal lymphocytes in vaginally immunized mice may be due in part to larger total numbers of lymphocytes in the mice. Thus, vaginal immunization appeared to have little if any selective effect on lymphocyte homing to the vagina either before or after vaginal challenge. These results are in good agreement with recent studies reporting that mucosal immunization did not selectively increase the numbers of specific T cells in the lymph nodes draining mucosal tissues at 5 to 8 weeks after immunization in mice (14) and sheep (37). However, in contrast to the case for short times after immunization, Gallichan and Rosenthal (14) reported that memory T cells were selectively increased in lymph nodes draining mucosal sites at 5 to 19 months after mucosal immunization. This important observation suggests that local immunization at a mucosal site may elicit a more vigorous long-term cellular immunity against mucosal challenge than parenteral immunization.
ACKNOWLEDGMENTS
This work was supported by grant HD17337 from the National Institute of Child Health and Human Development.
We thank Sheila Scillufo, Li Ying, and Maureen Doran for excellent technical assistance.
REFERENCES
- 1.Abraham M C, Desjardins M, Filion L G, Garber G E. Inducible immunity to Trichomonas vaginalis in a mouse model of vaginal infection. Infect Immun. 1996;64:3571–3575. doi: 10.1128/iai.64.9.3571-3575.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ada G L. Vaccines. In: Paul W E, editor. Fundamental immunology. New York, N.Y: Raven Press; 1993. pp. 1309–1352. [Google Scholar]
- 3.Anderson M L, BonDurant R H, Corbeil R R, Corbeil L B. Immune and inflammatory responses to reproductive tract infection with Tritrichomonas foetus in immunized and control heifers. J Parisitol. 1996;82:594–600. [PubMed] [Google Scholar]
- 4.Ashley R L, Corey L, Dalessio J, Wilson P, Remington M, Barnum G, Trethewey P. Protein-specific cervical antibody responses to primary genital herpes simplex virus type 2 infections. J Infect Dis. 1994;170:20–26. doi: 10.1093/infdis/170.1.20. [DOI] [PubMed] [Google Scholar]
- 5.Baggiolini M, Dewald B, Moser B. Interleukin-8 and related chemotactic cytokines—CXC and CC chemokines. Adv Immunol. 1994;55:97–180. [PubMed] [Google Scholar]
- 6.Campo M S. Infection by bovine papillomavirus and prospects for vaccination. Trends Microbiol. 1995;3:92–97. doi: 10.1016/s0966-842x(00)88889-7. [DOI] [PubMed] [Google Scholar]
- 7.Cassone A, Boccanera M, Adriani D, Santoni G, DeBernardis F. Rats clearing a vaginal infection by Candida albicans acquire specific, antibody-mediated resistance to vaginal reinfection. Infect Immun. 1995;63:2619–2624. doi: 10.1128/iai.63.7.2619-2624.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Corbeil L B. Vaccination strategies against Tritrichomonas foetus. Parisitol Today. 1994;10:103–106. doi: 10.1016/0169-4758(94)90009-4. [DOI] [PubMed] [Google Scholar]
- 9.Corbeil L B. Use of an animal model of trichomoniasis as a basis for understanding this disease in women. Clin Infect Dis. 1995;21:S158–S161. doi: 10.1093/clinids/21.supplement_2.s158. [DOI] [PubMed] [Google Scholar]
- 10.Corbeil L B, Winter A J. Animal model for the study of genital secretory immune mechanisms: venereal vibriosis in cattle. In: Brooks G F, Gotschlich E C, Holmes K K, Sawyer W D, Young F E, editors. Immunobiology of Neisseria gonorrhoeae. Washington, D.C: American Society for Microbiology; 1978. pp. 293–299. [Google Scholar]
- 11.Crum C P, Roche J K. Papillomavirus infection and intraepithelial neoplasia of the cervix. In: Bronson R A, Alexander N J, Anderson D, Branch D W, Kutteh W H, editors. Reproductive immunology. Cambridge, United Kingdom: Blackwell Science; 1996. pp. 540–559. [Google Scholar]
- 12.DiTommaso A, Saletti G, Pizza M, Rappuoli R, Dougan G, Abrignani S, Douce G, DeMagistris M T. Induction of antigen-specific antibodies in vaginal secretions by using a nontoxic mutant of heat-labile enterotoxin as a mucosal adjuvant. Infect Immun. 1996;64:974–979. doi: 10.1128/iai.64.3.974-979.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fidel P L, Lynch M E, Conaway D H, Tait L, Sobel J D. Mice immunized by primary vaginal Candida albicans infection develop acquired vaginal mucosal immunity. Infect Immun. 1995;63:547–553. doi: 10.1128/iai.63.2.547-553.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gallichan W S, Rosenthal K L. Long-lived cytotoxic T lymphocyte memory in mucosal tissues after mucosal but not systemic immunization. J Exp Med. 1996;184:1879–1890. doi: 10.1084/jem.184.5.1879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ishizaka S T, Piacente P, Silva J, Mishkin E M. IgG subtype is correlated with efficiency of passive protection and effector function of anti-herpes simplex virus glycoprotein D monoclonal antibodies. J Infect Dis. 1995;172:1108–1111. doi: 10.1093/infdis/172.4.1108. [DOI] [PubMed] [Google Scholar]
- 16.Jones P D, Ada G L. Influenza virus-specific antibody-secreting cells in the murine lung during primary influenza virus infection. J Virol. 1986;60:614–619. doi: 10.1128/jvi.60.2.614-619.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kawabata S, Miller C J, Lehner T, Fujihashi K, Kubota M, MGhee J R, Imaoka K, Hiroi T, Kiyono H. Induction of Th2 cytokine expression for p27-specific IgA B cell responses after targeted lymph node immunization with simian immunodeficiency virus antigens in rhesus macaques. J Infect Dis. 1998;177:26–33. doi: 10.1086/513811. [DOI] [PubMed] [Google Scholar]
- 18.King N J C, Parr E L, Parr M B. Migration of lymphoid cells from vaginal epithelium to the iliac lymph nodes in relation to vaginal infection by herpes simplex virus type 2. J Immunol. 1998;160:1173–1180. [PubMed] [Google Scholar]
- 19.Lehner T, Wang Y, Cranage M, Bergmeier L A, Mitchell E, Tao L, Hall G, Dennis M, Cook N, Brookes R, Klavinskis L, Jones I, Doyle C, Ward R. Protective mucosal immunity elicited by targeted iliac lymph node immunization with a subunit SIV envelope and core vaccine in macaques. Nat Med. 1996;2:767–775. doi: 10.1038/nm0796-767. [DOI] [PubMed] [Google Scholar]
- 20.McDermott M R, Brais L J, Evelegh M J. Mucosal and systemic antiviral antibodies in mice inoculated intravaginally with herpes simplex virus type 2. J Gen Virol. 1990;71:1497–1504. doi: 10.1099/0022-1317-71-7-1497. [DOI] [PubMed] [Google Scholar]
- 21.McDermott M R, Brais L J, Goettsche G C, Evelegh M J, Goldsmith C H. Expression of immunity to intravaginal herpes simplex virus type 2 infection in the genital tract and associated lymph nodes. Arch Virol. 1987;93:51–68. doi: 10.1007/BF01313893. [DOI] [PubMed] [Google Scholar]
- 22.McDermott M R, Smiley B J, Brais P L J, Rudzroga H, Bienenstock J. Immunity in the female genital tract after intravaginal vaccination of mice with an attenuated strain of herpes simplex virus type 2. J Virol. 1984;51:247–253. doi: 10.1128/jvi.51.3.747-753.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Miller C J, Kang D W, Marthas M, Moldoveanu Z, Kiyono H, Marx P, Eldridge J H, Mestecky J, McGhee J R. Genital secretory immune response to chronic simian immunodeficiency virus (SIV) infection: a comparison between intravenously and genitally inoculated rhesus macaques. Clin Exp Immunol. 1992;88:520–526. doi: 10.1111/j.1365-2249.1992.tb06481.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Milligan G N, Bernstein D I. Generation of humoral immune responses against herpes simplex virus type 2 in the murine female genital tract. Virology. 1995;206:234–241. doi: 10.1016/s0042-6822(95)80038-7. [DOI] [PubMed] [Google Scholar]
- 25.Murphy B R. Mucosal immunity to viruses. In: Ogra P L, Mestecky J, Lamm M E, Strober W, McGhee J R, Bienenstock J, editors. Handbook of mucosal immunology. San Diego, Calif: Academic Press, Inc.; 1994. pp. 333–343. [Google Scholar]
- 26.O’Hagan D T, Rafferty D, McKeating J A, Illum L. Vaginal immunization of rats with a synthetic peptide from human immunodeficiency virus envelope glycoprotein. J Gen Virol. 1992;73:2141–2145. doi: 10.1099/0022-1317-73-8-2141. [DOI] [PubMed] [Google Scholar]
- 27.Parr E L, Parr M B. Local immunization for antifertility immunity. In: Griffin P D, Johnson P M, editors. Local immunity in reproductive tract tissues. Oxford, United Kingdom: Oxford University Press; 1993. pp. 441–458. [Google Scholar]
- 28.Parr E L, Bozzola J J, Parr M B. Purification and measurement of secretory IgA in mouse milk. J Immunol Methods. 1995;180:147–157. doi: 10.1016/0022-1759(94)00310-s. [DOI] [PubMed] [Google Scholar]
- 29.Parr E L, Parr M B. Immunoglobulin G is the main protective antibody in mouse vaginal secretions after vaginal immunization with attenuated herpes simplex virus type 2. J Virol. 1997;71:8109–8115. doi: 10.1128/jvi.71.11.8109-8115.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Parr, E. L., J. J. Bozzola, and M. B. Parr. Immunity to vaginal infection by herpes simplex virus type 2 in adult mice: characterization of the immunoglobulins in vaginal mucus. J. Reprod. Immunol., in press. [DOI] [PubMed]
- 30a.Parr, E. L., and M. B. Parr. Unpublished data.
- 31.Parr M B, Parr E L. Mucosal immunity in the female and male reproductive tracts. In: Ogra P L, Mestecky J, Lamm M E, Strober W, McGhee J R, Bienenstock J, editors. Handbook of mucosal immunology. San Diego, Calif: Adademic Press, Inc.; 1994. pp. 677–689. [Google Scholar]
- 32.Parr M B, Kepple L, McDermott M R, Drew M D, Bozzola J J, Parr E L. A mouse model for studies of mucosal immunity to vaginal infection by herpes simplex virus type 2. Lab Invest. 1994;70:369–380. [PubMed] [Google Scholar]
- 33.Parr M B, Parr E L. Protective immunity against HSV-2 in the mouse vagina. J Reprod Immunol. 1997;36:77–92. doi: 10.1016/s0165-0378(97)00055-7. [DOI] [PubMed] [Google Scholar]
- 34.Parr M B, Parr E L. Mucosal immunity to herpes simplex virus type 2 infection in the mouse vagina is impaired by in vivo depletion of T lymphocytes. J Virol. 1998;72:2677–2685. doi: 10.1128/jvi.72.4.2677-2685.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Parr, M. B., and E. L. Parr. Female genital tract immunity in animal models. In P. L. Ogra, J. Mestecky, M. E. Lamm, W. Strober, J. R. McGhee, and J. Bienenstock (ed.), Handbook of mucosal immunology, 2nd ed., in press. Academic Press, Inc., San Diego, Calif.
- 36.Patton D L, Rank R G. Animal models for the study of pelvic inflammatory disease. In: Quinn T C, editor. Sexually transmitted diseases. New York, N.Y: Raven Press; 1992. pp. 85–111. [Google Scholar]
- 37.Premier R R, Jacobs H J, Brandon M R, Meeusen E N. Distribution of antigen-specific memory T cells in lymph nodes after immunization at peripheral or mucosal sites. Immunol Cell Biol. 1996;74:265–273. doi: 10.1038/icb.1996.47. [DOI] [PubMed] [Google Scholar]
- 38.Rank R G, Batteiger B E. Protective role of serum antibody in immunity to chlamydial genital infection. Infect Immun. 1989;57:299–301. doi: 10.1128/iai.57.1.299-301.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rank R G, Batteiger B E, Soderberg L S F. Immunization against chlamydial genital infections in guinea pigs with UV-inactivated and viable chlamydiae administered by different routes. Infect Immun. 1990;58:2599–2605. doi: 10.1128/iai.58.8.2599-2605.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Snapper C M, Finkelman F D. Immunoglobulin class switching. In: Paul W E, editor. Fundamental immunology. New York, N.Y: Raven Press; 1993. pp. 837–864. [Google Scholar]
- 41.Su H, Feilzer K, Caldwell H D, Morrison R P. Chlamydia trachomatis genital tract infection of antibody-deficient gene knockout mice. Infect Immun. 1997;65:1993–1999. doi: 10.1128/iai.65.6.1993-1999.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Thapar M, Parr E L, Parr M B. Secretory immune responses in mouse vaginal fluid after pelvic, parenteral, or vaginal immunization. Immunology. 1990;70:121–125. [PMC free article] [PubMed] [Google Scholar]
- 43.Yang S-L, Schumacher G F B. Immune response after vaginal application of antigens in the rhesus monkey. Fertil Steril. 1979;32:588–598. doi: 10.1016/s0015-0282(16)44365-7. [DOI] [PubMed] [Google Scholar]