Abstract
Aging is a gradual and irreversible process that is accompanied by an overall decline in cellular function and a significant increase in the risk of age-associated disorders. Generally, delaying aging is a more effective method than treating diseases associated with aging. Currently, researchers are focused on natural compounds and their therapeutic and health benefits. Curcumin is the main active substance that is present in turmeric, a spice that is made up of the roots and rhizomes of the Curcuma longa plant. Curcumin demonstrated a positive impact on slowing down the aging process by postponing age-related changes. This compound may have anti-aging properties by changing levels of proteins involved in the aging process, such as sirtuins and AMPK, and inhibiting pro-aging proteins, such as NF-κB and mTOR. In clinical research, this herbal compound has been extensively examined in terms of safety, efficacy, and pharmacokinetics. There are numerous effects of curcumin on mechanisms related to aging and human diseases, so we discuss many of them in detail in this review.
Keywords: Curcumin, Aging, Longevity
Introduction
Generally, human life expectancy has increased. In aging biology studies, biochemical and genetic processes that lead to aging are investigated. Now, effective approaches are being examined to combat this. To combat this process, lots of effective approaches are being examined. Aging is an irreversible and natural pathophysiological process associated with the development of many age-related diseases, such as musculoskeletal disorders, neurodegenerative diseases, cardiovascular disease, cancer, and arthritis [1]. The results of recent studies demonstrated that the aging process is manageable and that the rate and quality of aging can be modulated [2]. The biochemical changes that occur in all organisms are hallmarks of aging. These hallmarks are genetic instability, epigenetic changes, telomerase dysfunction, cellular senescence, stem cell exhaustion, and mitochondrial dysfunction [2]. Diets that include anti-oxidants and anti-inflammatory properties have been shown to reduce the risk of age-related cognitive decline and neurodegenerative diseases. In the last few years, the focus of studies has been on curcumin. This natural compound has a positive impact on the treatment of human disorders [3–6].
Curcumin is an orange-yellow pigment that was first isolated from the aromatic rhizomas of turmeric (Curcuma longa L.), a plant from the ginger family (Zingiberaceae) [7]. There are many biological and pharmacological properties of curcumin, including anti-oxidant properties, immunomodulatory properties, anti-inflammatory properties, anti-microbic properties, cardio-protective properties, nephroprotective properties, hepato-protective properties, anti-neoplastic properties, and anti-rheumatic and anti-aging properties [8]. Curcumin modulates a wide variety of signaling molecules at the molecular level. Depending on the target structure, it may increase or decrease their activity. Curcumin binding can be activated in two ways: directly or indirectly. An indirect modulator may be an inflammatory mediator, a transcription factor, a protein that regulates drug resistance, enzymes, a cell adhesion molecule, a kinase, a protein that regulates drug resistance, a growth factor, a protein that controls cell survival, or a protein that regulates the cell cycle. Curcumin’s direct effect is mediated by inflammation molecules, kinases, reductases, histone acetyltransferases, integrins, DNA methyltransferase 1, carriers, and metal ions [9, 10]. The purpose of this review is to summarize the therapeutic potential of curcumin, especially its potential for preventing and delaying aging.
The biology of aging process
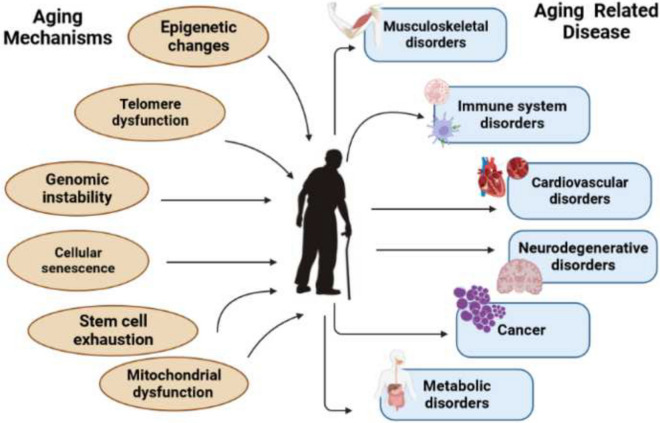
The biological process of aging is intricate and complex. It is generally accepted that aging refers to a physiological decrease in several biological activities in the organ along with a gradual reduction in the capacity of the cells to adjust to external injuries as well as internal ones [11, 12]. Aging has a highly heterogeneous phenotype. There are several factors that contribute to aging, including random, environmental, genetic, and/or epigenetic factors [13, 14]. A number of premature age-associated diseases can be predicted based on the decline in the physiological function of most living organisms, which is characterized by changes in molecular pathways. As a result of aging, all levels of the human body experience multifaceted changes. It includes cardiovascular disorders, neurodegenerative disorders, musculoskeletal disorders, metabolic disorders, immune system disorders, and cancer [1, 13]. Despite aging’s complexity, the basic molecular mechanisms and main pathways are highly conserved and are responsible for maintaining tissue homeostasis. Cellular and molecular changes associated with aging are composed of nine major hallmarks: DNA instability, telomere attrition, epigenetic alterations, loss of proteostasis, deregulated nutrient-sensing, mitochondrial dysfunction, cellular senescence, stem cell exhaustion, and altered intercellular communication [15] (Fig. 1).
Fig. 1.
Cellular and molecular changes associated with aging are composed of nine major hallmarks: DNA instability, telomere attrition, epigenetic alterations, loss of proteostasis, deregulated nutrient sensing, mitochondrial dysfunction, cellular senescence, stem cell exhaustion, and altered intercellular communication
Due to the reduced ability of several stem cells to repair tissues with age, many tissues’ regenerative and repair potential declines [16]. There is no definitive evidence of which biological changes (molecular, cellular, or physiological changes) are most important in the aging process and how they interact with each other, despite available research. Oxidative stress is the result of ROS overproduction or insufficient anti-oxidant defenses in the body. Additionally, ROS are produced as a natural by-product of cellular metabolism and the production of ATP. Oxidative stress plays a key role in the development of age-related conditions. Also, many drugs, alcohol abuse, smoking, radiation, and environmental pollution contribute to the formation of free radicals in the body and oxidative stress [17]. The activation of neutrophils during inflammation increases the production of ROS, an intrinsic immune molecule that modulates the immune response by activating the MAPK pathway [18]. By using physiological signals, dietary ingredients, and drugs, the body can scavenge oxidative stressors, including reactive oxygen species. In the process of aging, diet plays a crucial role. From pharmacological and therapeutic points of view, some compounds have been extracted and examined over the years for their anti-aging and therapeutic properties. several natural bioactive products and plant extracts with protective anti-oxidant properties, such as caffeic acid, ginsenosides, blueberry extract, and chayote [19–22]. The successful aging model developed by Rowe and Kahn utilizes three key components: absence of disease and disease-related disability, maintenance of high mental and physical function, and continued engagement with life. According to the study, age, gender, educational level, economic status, heavy drinking, subjective health status, and health screening were the factors that correlated in the individual system; living arrangement, satisfaction with spouse, and frequency of contacting family, siblings, and relatives; and how often you contact neighbors and friends, how many of your neighbors and friends you know, and whether neighborhood facilities are accessible [23].
Supplements can delay aging
Natural factors that prevent aging have received attention in life span research. Natural compounds that have emerged as anti-aging agents include omega-3 fatty acids (polyunsaturated fatty acid (PUFA)) [24], vitamins (E, D, K, and C) [25, 26], coenzyme Q10 (a small, lipid-soluble anti-oxidant molecule) [27], epigallocatechin gallate (EGCG) (a polyphenol compound concentrated in green tea) [28, 29], gingerol (an active component of ginger) [30], collagen (a protein that helps maintain skin structure and slows down the aging process, preventing wrinkles) [31], and curcumin (derived from turmeric) [32].
These compounds offer improved cardiovascular health, reduced inflammation, and enhanced cognitive function. They may reduce the risk of age-related diseases, such as dementia and cancer. Some foods and dietary supplements have been the subject of scientific studies regarding the presence of certain compounds [33]. We will look at the current research to explore how curcumin, as a natural product, may prevent aging-related changes.
Curcumin
Chemistry
Curcumin was extracted from the rhizomes of C. longa in 1815 by Vogel and Pelletier. After two centuries, more efficient and advanced extraction methods have been reported [34]. The molecule curcumin is symmetrical, also called diferuloyl methane. The chemical formula of curcumin is C21H20O6, and its molecular weight is 368.38 [35]. The most common method for separating curcumin from turmeric is solvent extraction, followed by column chromatography [36].
In addition to its keto form, curcumin also exists in an enol form. While the keto form is predominant, in alkaline conditions, the enol tautomer is exclusively present, which can be rationalized by intramolecular hydrogen bonding in the enol form [37, 38]. Under both neutral and alkaline conditions, curcumin degrades rapidly, but its solubility increases under alkaline conditions. In methanol, curcumin absorbs most efficiently at 430 nm, and in acetone, it absorbs most efficiently at 415–420 nm. The maximum absorbance of curcumin at 467 nm occurs in alkaline conditions [39, 40].
The metabolism
It is important to understand the metabolism of curcumin in order to fully benefit from its bioactive properties as well as the health benefits it provides [41]. It is suggested that curcumin is primarily metabolized in the liver, along with the intestines and gut microbiota [42]. In oral administration, curcumin is primarily metabolized to glucuronates and glucuronate/sulfate conjugates. In particular, the finding indicates that the gastrointestinal tracts play an important role in the glucuronidation of curcuminoids, which may have significant implications for their pharmacokinetics [43]. After administration of curcumin, a small amount of free and intact curcumin can be detected in plasma [44]. The metabolism of curcuminoids, however, mainly results in reductive metabolites, for example, hexahydrocurcuminoids [45]. Since hexahydrocurcuminoids do not contain olefinic double bonds, they are more stable at pH 7.4 than curcumin [46]. Furthermore, secondary biliary metabolism involves dihydroferulic acid and ferulic acid [35, 47]. In addition, microbiota such as Escherichia coli and Blautia sp. also metabolize curcumin alternatively. In a two-step reduction pathway from curcumin to dihydrocurcumin and then to tetrahydrocurcumin, Escherichia coli was found to be active by an NADPH-dependent reductase [48]. In Blautia sp., curcumin is demethylated into demethylcurcumin and bis-demethylcurcumin. Curcumin’s polypharmacology is attributed to its metabolites, which have been identified as anti-oxidants, anti-inflammatory, anti-tumor, cardioprotective, and anti-diabetic [49–52].
Bioavailability and new formulation
Pure curcumin crystals typically have low bioaccessibility due to their high melting point and low water solubility under acidic and neutral conditions [53]. For example, at 37 °C and pH 7.2, curcumin t1/2 was reported as less than 10 min [54]. Since curcumin does not degrade under acidic conditions, it should be stable in the stomach [49]. A number of attempts have been made over the years to improve curcumin’s bioavailability due to its broad spectrum of potential health benefits. Increasing curcumin’s bioaccessibility, retarding its metabolism, and/or enhancing its absorption can enhance its bioavailability. Consequently, by trapping curcumin inside hydrophobic phases, such as micellar, liposomal, microemulsions, emulsions, solid fats, or biopolymers, the metabolism of curcumin can be inhibited [55, 56]. There are three main types of curcumin formulations available, which are either bioavailable or bioenhanced. The first-generation formulation of curcumin included adjuvants that inhibited essential detoxification enzymes like piperine from black pepper or turmeric oils. The absorption of curcumin is enhanced through these adjuvants that delay its metabolism [57–59]. The second-generation curcumin supplements such as BioCurc, Cavacurcmin, CurcuWIN, Hydrocurc, Meriva, Nanocurcumin, Novasol, Theracurmin, and Turmipure Gold use emulsifiers like polysorbates, lipid complexes, and water-dispersible nanopreparations to increase solubility [59]. The conjugated metabolites of curcuminoid (glucuronides and sulfates) are vital in increasing plasma curcuminoid levels. However, according to several studies, these metabolites do not have any significant biological effects due to their large size, quick renal elimination, limited membrane permeability, and limited blood brain barrier (BBB) permeability [59–61]. Therefore, to maximize curcumin’s therapeutic effects, delivering it in its free form (without conjugation) is crucial. Thanks to third-generation curcumin formulations such as Longvida and CurQfen, the issue of “free” curcuminoids is no longer a problem. These formulations have improved bioavailability, membrane permeability, and cellular uptake without artificial emulsifiers like polysorbates [59]. One of the well-studied curcumin formulations is the phytosomal formulation of curcumin (Meriva), which is a complex of curcumin and phosphatidylcholine. Compared to uncomplexed curcumin, Meriva has improved bioavailability and pharmacokinetic profiles [56]. Researchers studied theracurmin, another curcumin formulation consisting of dispersed curcumin and nanoparticles, to determine how well it treats osteoarthritis compared to turmeric powder alone [62, 63]. Compared to turmeric powder, theracurmin has a 40-fold higher bioavailability in rats and a 27-fold higher bioavailability in humans. Nakagawa et al. found that theracurmin was effective at decreasing pain without any adverse effects [64].
Curcumin analogous
In clinical practice, curcumin is one of the most difficult compounds to administer due to its low solubility in water, low oral bioavailability, and rapid degradation under physiological conditions [65]. A series of curcumin analogs have thus been developed, such as EF24, HO-3867, 2-HBA, and dimethoxy curcumin, in order to improve its biological activity, bioavailability, and therapeutic efficacy. These compounds are more effective than curcumin in preventing and treating a wide range of diseases and reducing age-associated disorders. It is still not clear how these compounds act as anti-aging agents [66–69]. The process of cellular senescence occurs when normal cells stop dividing after they have multiplied extensively or have been exposed to a lot of stress [2]. As a result of senescent cells (SCs) accumulating in various tissues with aging, it can be described as a hallmark of aging. Immune senescence decreases the ability of the body to clear SCs [70]. Despite the fact that cellular senescence is a tumor-suppressing mechanism, SCs can also play a causal role in aging and diseases due to aging by accumulating over time [71–73]. By considering that the pharmacological clearance of SCs may have the capacity to increase healthy life span and postpone the onset of age-related disorders, researchers hypothesized that curcumin and its analogs may also increase health span in part by functioning as novel senolytic agents. EF24, one of the synthetic analogs of curcumin, shows a tenfold greater potential for inducing cancer cell death than curcumin. Furthermore, EF24 is more effective against several cancer cells, and it has less toxicity against normal cells in comparison to cisplatin [74]. In addition, studies have shown that HO-3867, 2-HBA, and DIMC, which are other representative curcumin analogs, have also been reported to have improved anti-cancer activity compared to curcumin itself. Because curcumin has anti-aging activities, curcumin analogs can be used as a rich resource for the discovery of novel senolytic agents [67, 68, 75]. The result from the Wen Li et al. study shows that EF24 selectively induces SC apoptosis via increases in the proteasome degradation of the Bcl-2 anti-apoptotic protein. As a result of this study, curcumin analogs have the potential to function as anti-aging agents and may be useful for treating age-related diseases [76].
Bioactivity
The wide range of beneficial biological functions of curcumin makes it a unique molecule that is able to affect multiple biological targets. It has been shown that this molecule can be effective against several disorders, such as cancer, cardiovascular dysfunction, neurological diseases, and autoimmune diseases [9, 34, 77]. A variety of studies have shown curcumin to have anti-oxidant, anti-inflammatory, and immunomodulatory effects in lung tissue, as well as anti-fibrotic and lung protective properties [78, 79]. Many biological targets, such as transcription factors, inflammatory mediators, growth factors, cytokines, cell cycle proteins, enzymes, protein kinases, and apoptotic proteins, as well as cellular pathways, can be modulated by curcumin [34, 80, 81]. According to research, it could regulate multiple cell signaling pathways, including tumor suppressor, cell survival caspase pathways, protein kinase, and death receptors, to modulate tumor growth [77, 80].
Curcumin and its anti-ageing role
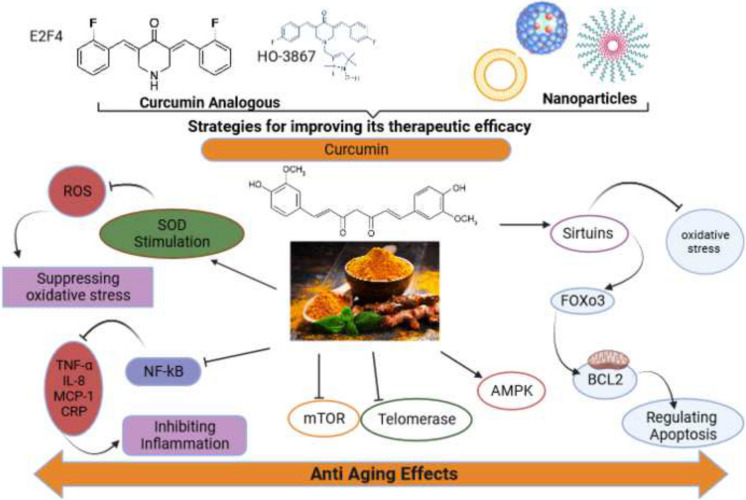
Curcumin has various roles in preventing aging and diseases related to it, among which are its effects on oxidative stress, inflammation, telomere length, signaling pathways, and proteins such as sirtuin. These are discussed in detail below and illustrated in Fig. 2.
Fig. 2.
Curcumin has various roles in preventing aging and diseases related to it, among which are its effects on oxidative stress, inflammation, telomere length, signaling pathways, and proteins such as sirtuin
Curcumin effect on oxidative stress
There is evidence that elevated reactive oxygen species (ROS) levels can cause oxidative stress and severe damage to the organelle membranes, proteins, DNA, and lipids. This damage results in mitochondrial dysfunction, cell death, and an aging phenotype. Aging is associated with a decline in the production of ATP and ROS and a decline in anti-oxidant defense [82]. According to research, curcumin may be an effective anti-oxidant that reduces the effects of oxidative stress. The ability of curcumin to interact with various molecular mechanisms results in a reduction of oxidative stress, which is related to, among other things, its ability to chelate heavy metals and regulate enzyme activity [83]. Potential therapies to promote healthy aging can be developed using this approach. The anti-oxidant activity of curcumin has been demonstrated both in vitro and in vivo. Multiple cellular and animal models have shown that curcumin protects against oxidative and nitrosative stress. Researchers found that curcumin inhibited lipid and protein oxidation by reducing MDA, as well as protein carbonyls, thiols, and nitrotyrosine levels [84]. It has been demonstrated in several studies that heart tolerance to oxidative stress decreases as we age due to a decrease in anti-oxidant enzyme concentrations (GSH-Px and SOD), contributing to CV alterations [85]. Curcumin also stimulates the activity of the key anti-oxidant enzymes (superoxide dismutase (SOD) and catalase) of the defense mechanisms against free radicals produced during metabolic reactions [86]. One of the best strategies to reduce oxidative stress is to increase levels of endogenous and exogenous anti-oxidants. There are two ways to reduce endogenous oxidative stress: to prevent ROS formation or to quench it with anti-oxidants [87]. The chemical structure of curcumin makes it an excellent scavenger of reactive oxygen species (ROS) and reactive nitrogen species (RNS) [88]. Combined, these findings confirm curcumin’s anti-oxidant potential for organ health function as we age. Researchers need to investigate further the molecular targets and signaling pathways responsible for curcumin’s anti-oxidant effects.
Curcumin effect on inflammation
With age, inflammation-regulating mechanisms can become impaired, which may contribute to older people’s susceptibility to infection and chronic diseases. Chronic pro-inflammatory status is a hallmark of aging [89]. The presence of chronic low-grade inflammation in the elderly without overt infection has been termed “inflammaging” and represents a significant health risk [90]. This low-grade, chronic inflammation develops with age and is characterized by high serum levels of mediators and inflammatory cytokines such as C-reactive protein (CRP), IL-8, IL-6, and tumor necrosis factor (TNF). There is an association between this pleiotropic cytokine and atherosclerosis, osteoporosis, and sarcopenia, as well as disability development and all-cause mortality [91]. Improved immune function in older adults is being explored in various ways. One of the low-tech strategies to combat inflammation is dietary intervention, including curcumin supplementation and healthy nutrition. Different cellular and animal models have illustrated curcumin’s age-modulating properties and healthful effects [92]. A recent study demonstrated that curcumin may influence NF-κB (nuclear factor κB) activation due to its anti-oxidant properties. The NF-κB is involved in the response of the cell to the activation of free radicals, heavy metals, radiation, and the oxidation of low-density lipoproteins (LDL) [93]. Inflammation is triggered by all of these factors through the activation of NF-κB. Curcumin, by blocking the most relevant proinflammatory pathway, NF-kB, helps reduce inflammation and cellular damage. Therefore, this effect is useful for preventing aging-related cellular damage. In addition, results from studies have shown treatment with a low dose of curcumin leads to a decrease in the level of secreted pro-inflammatory cytokines such as IL-8 in normal young cells [94]. Inflammaging markers include not only cytokines but also acute-phase proteins such as CRP and mannose-binding lectins [95]. The results of a randomized clinical trial found that 80 mg/day of curcumin nanomicelles resulted in a statistically significant improvement in plasma levels of C-reactive protein [96]. A recent study has shown that the plasma level of circulating monocyte chemoattractant protein-1 (MCP-1) is associated with chronologic age in humans and can be considered a potential age marker [97, 98]. In vitro and in vivo studies demonstrated that curcumin significantly reduced the expression as well as production of inflammatory cytokines, such as monocyte chemoattractant/chemotactic protein-1 (MCP-1), and could be an effective and therapeutic agent for various inflammatory diseases by targeting multiple molecules [99]. Overall, curcumin is an important biochemical compound for reversing inflammation and enhancing immune system performance, both of which are essential factors in improving health and thereby slowing down aging.
Curcumin effect on lengthens telomeres
The telomeres are highly conserved repetitive DNA sequences that undergo shortening with every division of a cell as well as with oxidative stress and aging [100, 101]. The catalytic subunit of hTERT (telomerase, human telomerase reverse transcriptase) functions during chromosomal replication to stabilize telomere length [102]. Numerous studies have shown that curcumin lengthens telomeres by enhancing telomerase activity [103], while curcumin inhibits telomerase activity in tumor cells, where growth is undesirable, supporting the body’s natural anti-tumor response [104].
Curcumin effect on the mTOR—the aging protein
One of the major evolutionary conserved signaling pathways that influences longevity is the mammalian target of rapamycin (mTOR). The mTOR (mammalian target of rapamycin), a phosphoinositide 3-kinase-related protein kinase, acts as a catalytic subunit in two protein complexes, including mTORC1 and mTORC2 (mTOR complex) [105, 106]. The primary evidence that mTOR modulates aging comes from studies conducted in Saccharomyces cerevisiae, where it was shown that deletion of the Sch9 gene (a functional ortholog of the mammalian S6K gene) leads to an increase in the chronological life span of yeast [107]. In another study in the Caenorhabditis elegans model organism, both daf-15 (raptor) and let-363 (Ce TOR) mutants extended adult life spans by shifting metabolism to accumulate fat [108]. In some other aging model organisms, such as fruit flies and yeast, mutations in mTOR and numerous other mTORC1 cascade components are also associated with prolonged life spans [109, 110]. These studies determine that mTORC1 is a major evolutionarily conserved modulator of longevity. Research demonstrated that in RMS cells, curcumin at low concentrations could inhibit the IGF-1-stimulated, mTORC1-mediated phosphorylation of S6K1 and 4E-BP1 and the IGF-1-stimulated, mTORC2-mediated phosphorylation of Akt at higher concentrations [111]. These results were also observed in prostate (DU145), breast (MCF-7), and cervical (HeLa) cancer cell lines [111]. In human colorectal cancer cells (HCT116), curcumin decreased transcriptional and translational expression of mTOR, Raptor, and Rictor [112]. According to Lim et al., curcumin inhibits mTOR signaling by acting as a protonophoric uncoupler and activator of F0F1-ATPase, resulting in AMPK activation and inhibition of mTOR [113].
Curcumin effect on AMPK
It has been demonstrated in several species that AMPK can exert pro-longevity effects [114]. AMPK is involved in the regulation of cellular metabolism, homeostasis, cell survival and growth, resistance to stress, and cell death, which are all the most important factors in aging and longevity [115]. A variety of indirect AMPK activators are currently being studied in clinical trials for their effects on aging-related characteristics, tissue homeostasis, and metabolic dysfunction in humans [116, 117]. A recent study showed that curcumin enhances AMPK phosphorylation by acting as an AMP-activated protein kinase (AMPK) agonist [118]. In addition, curcumin increased the phosphorylation of AMP-activated protein kinase (AMPK) in H4IIE and Hep3B cells [116].
Curcumin effect on sirtuins
Sirtuins (nicotine adenine dinucleotide (NAD)‐dependent deacetylases) are one of the most promising potential targets for delaying aging and increasing life span. In order to protect cells from DNA damage, efficient repair systems are sufficient to prevent it from accumulating. They play an essential role in maintaining the stability of genomes and the structure of telomeres. In addition, the function of these proteins is to assist in DNA repair as well as in epigenetic modification of histones [119]. In several studies, pretreatment with curcumin significantly increased Sirt1 activation and reduced oxidative stress [120]. Research has shown that curcumin can increase Caenorhabditis elegans’ life span when sirtuin 2 (the homolog of mammalian sirtuin 1) is not mutated [94]. In primary cortical neurons, curcumin’s neuroprotective effects are also mediated by sirtuin-1 induction. Neuronal injuries can result from the accumulation of extracellular glutamate. It is involved in cognitive functions such as synaptic plasticity, learning, and memory. In cortical neurons, curcumin inhibited glutamate excitotoxicity by deacetylating PGC-1α through Sirt1 and preserving mitochondrial functioning [121, 122]. Through synergetic activation of SIRT1 and PI3K/Akt signaling, as a result of enhancing FOXO3 phosphorylation, resveratrol improves the effects of exercise in elderly rat hearts [123]. According to studies, curcumin supplementation has similar effects. A combination of curcumin and physical performance can upregulate SIRT1 more effectively than dietary curcumin alone [124]. In conjunction with exercise, curcumin supplementation may increase AMPK phosphorylation, NAD/NADH ratio, and SIRT1 expression in muscles, as well as affect the time of exhaustion, improve exercise tolerance, and enhance the effect of exercise [125]. Due to increased resistance to stress conditions, improved exercise performance and fatigue prevention were observed in mice [126].
Curcumin protects brain aging
Aging has a significant impact on the nervous system because of oxidative stress, mitochondrial dysfunction, and cell death [127]. In the CNS, microglial cells support the normal functioning of neurons and monitor their health in homeostasis, the resting state. In normal conditions, microglia display protective effects. The activation of microglia plays a pivotal role in neurodegenerative diseases such as Alzheimer’s disease, Parkinson’s disease, ALS, or injuries or infections to the brain by inducing oxidative stress and neuroinflammation in the brain. Various brain diseases and disorders cause microglia to transform into M1 phenotypes, which release cytokines, chemokines, reactive oxygen species (ROS), and reactive nitrogen species (RNS) that promote inflammation. As a result of the overproduction of these inflammatory mediators, neuronal damage and death can occur. In previous studies, microglial activation and neuroinflammation have been shown to reduce the severity of neurodegenerative disorders [128–130]. There is a possibility that curcumin is a therapeutic molecule in the brain, as it inhibits microglial transformation, modulates inflammatory mediators, and counteracts neuroinflammation, which is the first step in neurodegenerative diseases [131]. In microglial cells, curcumin interacts with multiple molecular targets, including NF-κB. This may reduce the levels of pro-inflammatory markers and, therefore, the process of inflammation. The study demonstrated that curcumin blocked the LPS-mediated induction of COX2 by inhibiting NF-κB and activator protein 1 (AP1) in BV2 microglial cells [132]. Further, curcumin also promotes the development of the M2 microglial phenotype in an HO-1-dependent manner, thereby inhibiting the induction of iNOS and therefore reducing oxidative stress in microglia [133]. It has also been shown that curcumin inhibits microglial activation in the retinas of rd1 mice with retinal degeneration [134].
Evidence from clinical trials
According to both in vitro and in vivo pre-clinical studies, curcumin has shown promise in reducing inflammation associated with chronic disease and infection, as well as limiting cancer proliferation [135–138]. There is still a question as to whether these benefits also apply to humans. Curcuminoid bioactivity has been demonstrated in rodent models, but concerns have been raised about curcuminoid bioavailability in humans. Curcuminoids undergo hepatic conjugation and circulate primarily as inactive glucuronides when ingested by humans and rodents [135, 139, 140]. Thus far, curcumin has been shown to be effective against a wide range of human diseases in clinical trials. It has also been shown that it can protect against hepatic conditions, chronic exposure to arsenic, and alcohol poisoning. These clinical trials tested curcumin in combination with quercetin, gemcitabine, piperine, docetaxel, isoflavone forms, bioperine, sulfasalazine, mesalamine, prednisone, lactoferrin, pantoprazole, and N-acetylcysteine [55]. There is no doubt that curcumin is efficacious against a wide range of human ailments [47]. We describe several clinical trials using curcumin in the following part of this article (Table 1).
Table 1.
Several clinical trials of curcumin
| Disease | Dose | Outcome | Ref |
|---|---|---|---|
| Obese and overweight | 500 mg | Curcumin had beneficial effects on body mass index, high-density lipoprotein levels, and triglyceride/high-density lipoprotein ratio | [163] |
| Solid tumors | 180 mg | Curcumin could suppress systemic inflammation by reducing TNF-α, CGRP, substance P, MCP-1, IL-6, and CRP | [164] |
| COVID-19 | 160 mg | Significant decrease in IL-6 and IL-1β gene expression and secretion level in serum and supernatant occurred | [165] |
| Fatty liver | 70 mg | Reduced liver fat content, body mass index, low-density lipoprotein cholesterol, triglycerides, aspartate aminotransferase, alanine aminotransferase, glucose, and glycated hemoglobin levels | [166] |
| Knee osteoarthritis | 500 mg | There was a significant reduction in flatulence episodes and anti-ulcer effect | [167] |
| Diabetes | 2 tablets (each tablet containing 500 mg Meriva® corresponding to 100 mg curcumin) | There was a significant improvement in the venoarteriolar response, as well as a decrease in the score of peripheral edema (a sign associated with failure of the venoarteriolar response) | [168] |
| Chronic kidney | Meriva® 500 mg/tablet | Curcumin caused a significant reduction in plasma proinflammatory mediators including CCL-2, IFN-γ, and IL-4 as well as a reduction in lipid peroxidation | [[169] |
| Diabetes | Two tablets/day (1 g Meriva/day) | Edema score decreased significantly and venoarteriolar response improved correspondingly | [170] |
| Osteoarthritis | 200 mg | CRP levels decreased and osteoarthritis was effectively managed | [171] |
| Lung cancer | 1.5 g | Curcumin caused a significant reduction in the urinary excretion of mutagens in the smokers which are in risk of lung cancer | [172] |
| Inflammatory bowel disease | 550 mg | In all patients with proctitis, symptoms, and inflammatory indices include ESR and CRP decreased significantly | [173] |
| β-Thalassemia | 500 mg | Curcumin causes a reduction in oxidative damage in patients with β-thalassemia | [174] |
| Prostate cancer | 100 mg | The PSA levels of the patients decreased | [175] |
COVID-19 coronavirus disease, TNF-α tumor necrosis factor alpha, CGRP calcitonin gene-related peptide, MCP-1 monocyte chemoattractant protein-1, IL interleukin, CRP C-reactive protein, ESR erythrocyte sedimentation rate, IFN-γ interferon gamma, PSA prostate-specific antigen
Psoriasis
About 3% of the world’s population suffers from psoriasis (PsO), a chronic inflammatory, multifactorial disease. A thick, silvery plaque is the result of the uncontrolled proliferation of keratinocytes. Psoriasis pathogenesis begins with the activation of mature and inflammatory dendritic cells (DC), which leads to the release of cytokines, chemokines, and anti-microbial peptides. This is the first step in the pathogenesis of psoriasis [141]. Studies show that psoriasis patients have an increased number of Th22 cells. It is known that IL-22 is the prototypical cytokine secreted by Th22 cells, a subpopulation of T cells that produce IL-22 exclusively but not IL-17 or IFN-γ [142]. So, IL-22 plays a major role in several steps of the disease’s pathogenesis, including inflammation and keratinocyte proliferation [143]. The clinical trial was conducted by Emiliano Antiga et al. and consisted of a randomized, double-blind, placebo-controlled design in patients with psoriasis vulgaris to assess the effectiveness of a bioavailable oral curcumin. In a study conducted by our team, 63 patients with mild-to-moderate psoriasis vulgaris were randomly divided into two groups. One group received topical steroids and Meriva, a commercially available curcumin delivery system based on lecithin, at a dose of 2 g per day (arm 1), and the other group received topical steroids alone. Clinical assessment and immunoenzymatic analysis show that curcumin has been demonstrated to be effective as an adjuvant therapy for the treatment of psoriasis vulgaris and to significantly reduce serum levels of IL-22. In addition, a study by Bahraini et al. highlighted that, compared to the placebo, turmeric tonic could lead to an improvement in psoriasis symptoms by significantly reducing the erythema, scaling, and induration of lesions (PASI score), as well as improving the patients’ quality of life index (DLQI) [144].
Vitiligo
As a result of vitiligo, the cells responsible for producing pigment (color) in the skin (melanocytes) are destroyed, resulting in white patches on the surface of the skin. It has been suggested that oxidative stress plays an important role in the pathogenesis of this disease [145]. In order to treat vitiligo effectively, narrowband ultraviolet B radiation (NB-UVB) is often prescribed. NB-UVB emits light of wavelengths between 311 and 312 nm, which belongs to the UVB spectrum [146]. In view of the anti-oxidant properties of curcumin, it is a potential treatment option for vitiligo patients. There was one study that examined whether the combination of NB-UVB and tetrarchy drocurcuminoid cream could lead to synergistic therapeutic effects against vitiligo when used together. There were ten patients enrolled in this study who had vitiligo that was focal or generalized. In this study, two similar lesions were treated either with NB-UVB combined with topical tetrahydrocurcuminoid cream or with UVB alone. There was a statistically significant repigmentation in both treatment groups at the end of the study compared with baseline. It should also be noted that the combination group showed a slightly greater degree of repigmentation at 8 and 12 weeks, and the tetrahydrocurcuminoid treatment was well tolerated [147].
Arthritis
Arthritis is a chronic disease caused by inflammation of the joints. The disorder usually results from a dysregulation of pro-inflammatory cytokines and pro-inflammatory enzymes that lead to the production of prostaglandins (such as COX-2) and leukotrienes (such as lipoxygenase), as well as matrix metalloproteinases and adhesion molecules. Osteoarthritis is typically treated with exercise, lifestyle changes, and NSAIDs. There are, however, numerous adverse effects associated with the use of NSAIDs. Several studies have shown that curcumin improves the symptoms and delays the progression of rheumatoid arthritis by inhibiting several proteins such as extracellular signal-regulated protein kinase (ERK), mitogen-activated protein kinase family (MAPK or MAP kinase), activator protein-1 (AP-1), and nuclear factor-κB (NF-κB) signaling pathways in rheumatoid arthritis patients. In addition, the use of 250–1500 mg/day of turmeric by rheumatoid arthritis patients over 8–12 weeks can increase the function of dysfunctional immune cells (including TH1, TH17, Treg, and B cells) and decrease clinical symptoms.
Curcumin effect on aging related disease
Alzheimer’s disease
Aging is the most critical risk factor for many noninfectious diseases, including Alzheimer’s disease (AD). Alzheimer’s disease is generally a progressive neurological disease that affects mostly people over the age of 65. There are three phases involved in the pathogenesis of Alzheimer’s disease: fibril formation, amyloid plaque formation, and plaque deposition in the brain. Patients with Alzheimer’s disease are believed to lose cholinergic neurons in the basal forebrain due to these plaques [148]. It is evident that alternative treatments are needed for this disease because the currently available treatments have numerous adverse effects. The study was conducted as a randomized, double-blind, placebo-controlled study by Baum et al. on 34 patients with Alzheimer’s disease. Curcumin was administered at two different doses (1 or 4 g) or placebo (4 g). After curcumin treatment, the Mini-Mental State Examination (MMSE) score did not improve [149]. Curcumin can help slow the progression of disease and modulate cognitive function and biomarker levels. Clinical trials may have failed due to the poor bioavailability of curcumin, the selection of cohorts at an advanced stage of AD, and biological differences between rodent models and AD patients. Despite being successful in animal models, many interventions have failed in clinical trials [150].
Cardiovascular disease
With aging, the cardiovascular system is significantly affected, which can lead to atherosclerosis, hypertension, myocardial infarction, and strokes. A number of pathological changes may occur in aging cardiovascular tissues, including hypertrophy, altered diastolic function of the left ventricular (LV), diminished reverse systolic capacity of the LV, increased arterial stiffness, and impaired endothelial function [151]. There has been much attention focused on unraveling the mechanisms and optimizing treatment regimens for cardiovascular diseases (CVDs), which cause the highest mortality rates worldwide [152]. Numerous studies have demonstrated the cardiovascular-protective properties of curcumin. Randomized clinical trial study results by Jessica R. et al. show that curcumin supplementation improves vascular endothelial function in healthy middle-aged and older adults, corroborated by murine studies [153]. Further, curcumin consumption positively correlates with improved central arterial hemodynamics and reduced endothelial dysfunction in postmenopausal women [154, 155]. An additional randomized placebo-controlled study of 45 postmenopausal women found that endurance exercise in combination with curcumin significantly reduced left ventricular afterload. Those who received endurance exercise and curcumin together also experienced a decrease in systolic blood pressure [154]. Moreover, in another study, after 7 days of curcuminoid supplementation, serum triglyceride concentrations were significantly reduced [156]. According to a randomized controlled trial conducted on 87 non-alcoholic fatty liver disease patients, 1-g curcumin supplements for 8 weeks significantly reduced total cholesterol, non-high-density lipoprotein cholesterol (non-HDL-C), uric acid, and triglycerides [157]. The effects of curcumin on the well-being of 38 healthy volunteers were investigated using a lower dose of curcumin (80 mg/day). According to the results, curcumin significantly lowered the plasma level of triglyceride and some other relevant parameters, such as plasma beta-amyloid protein and salivary amylase [158].
Side effects of curcumin
Several clinical trials have demonstrated curcumin’s efficacy and safety. Despite the safety of this product, some side effects have been reported. Some mild adverse effects have been reported with high doses of curcumin, including diarrhea, headache, rash, and yellow stool [159]. Also, nausea and diarrhea and increased serum alkaline phosphatase and lactate dehydrogenase contents were experienced in patients who took low doses of curcumin [160]. Studies have shown that taking turmeric supplements of up to 8000 mg per day for 8 months is safe and does not harm humans. Up to 8000 mg of curcumin daily has been safely administered for 3 months [161]. When consumed with piperine, curcumin enhances bioavailability, which in turn may impact its safety profile [162]. Unfortunately, curcumin’s inadequate bioavailability and delivery render it ineffective for medicinal purposes. This review presents practical strategies to comprehensively enhance curcumin bioavailability.
Conclusions and future perspective
Modern aging research focuses on discovering new agents and strategies to fight aging and age-related diseases. To current knowledge, curcumin is one of the most promising candidates to achieve this purpose. It has powerful anti-inflammatory effects and is a very strong anti-oxidant with great potential to impact on age-related cellular proteins. Despite this, evidence from clinical studies on curcumin’s long-term effects on age-related pathology remains largely unexplored. Several strategies have been proposed to increase the systemic bioavailability of curcumin, but the long-term effects of such preparations are unknown, and higher concentrations may even have the opposite effect. However, curcumin’s poor bioavailability limits its clinical use against cancer. The list above includes some strategies that can help address the issue of curcumin’s poor bioavailability. Furthermore, any method that enhances curcumin’s bioavailability can boost its efficacy while increasing the risk of side effects. For instance, nanoparticles or carriers used in drug delivery can reduce the required dosage by releasing the drug slowly in tumor sites, thereby minimizing curcumin’s toxicity. Additionally, curcumin should be studied as a synergistic treatment for age-related disorders with other existing drugs. In addition, in the future, further studies are warranted to clarify the mechanisms of curcumin function and determine the possibility of applying it in clinical settings.
Acknowledgements
Our sincere gratitude goes to authors who contributed their time and expertise to accomplish this article.
Author contribution
Mehran Izadi, Nariman Sadri, Amirhossein Abdi, and Mohammad Mahdi Raeis Zadeh contributed to hypothesis, data gathering, and writing the main text of the manuscript. Dorsa Jalali, Mohammad Mahdi Ghazimoradi, and Sara Shouri contributed to hypothesis, data gathering, designing figure and tables, and final editing. Safa Tahmasebi contributed to the writing, scientific and structural editing, hypothesis, correspondence, and verifying the manuscript before submission.
Data availability
Not applicable.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Li Z, Zhang Z, Ren Y, Wang Y, Fang J, Yue H, et al. Aging and age-related diseases: from mechanisms to therapeutic strategies. Biogerontology. 2021;22(2):165–187. doi: 10.1007/s10522-021-09910-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell. 2013;153(6):1194–1217. doi: 10.1016/j.cell.2013.05.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ghasemi F, Shafiee M, Banikazemi Z, Pourhanifeh MH, Khanbabaei H, Shamshirian A, et al. Curcumin inhibits NF-kB and Wnt/β-catenin pathways in cervical cancer cells. Pathol Res Pract. 2019;215(10):152556. doi: 10.1016/j.prp.2019.152556. [DOI] [PubMed] [Google Scholar]
- 4.Mortezaee K, Salehi E, Mirtavoos-Mahyari H, Motevaseli E, Najafi M, Farhood B, et al. Mechanisms of apoptosis modulation by curcumin: implications for cancer therapy. J Cell Physiol. 2019;234(8):12537–12550. doi: 10.1002/jcp.28122. [DOI] [PubMed] [Google Scholar]
- 5.Panahi Y, Khalili N, Sahebi E, Namazi S, Simental-Mendía LE, Majeed M, et al. Effects of curcuminoids plus piperine on glycemic, hepatic and inflammatory biomarkers in patients with type 2 diabetes mellitus: a randomized double-blind placebo-controlled trial. Drug Res. 2018;68(7):403–409. doi: 10.1055/s-0044-101752. [DOI] [PubMed] [Google Scholar]
- 6.Sadeghian M, Rahmani S, Jamialahmadi T, Johnston TP, Sahebkar A. The effect of oral curcumin supplementation on health-related quality of life: a systematic review and meta-analysis of randomized controlled trials. J Affect Disord. 2021;1(278):627–636. doi: 10.1016/j.jad.2020.09.091. [DOI] [PubMed] [Google Scholar]
- 7.Farzaei MH, Zobeiri M, Parvizi F, El-Senduny FF, Marmouzi I, Coy-Barrera E, et al. Curcumin in liver diseases: a systematic review of the cellular mechanisms of oxidative stress and clinical perspective. Nutrients. 2018;10(7):855. 10.3390/nu10070855. [DOI] [PMC free article] [PubMed]
- 8.Vaiserman A, Koliada A, Zayachkivska A, Lushchak O. Curcumin: a therapeutic potential in ageing-related disorders. PharmaNutrition. 2020;14:100226. doi: 10.1016/j.phanu.2020.100226. [DOI] [Google Scholar]
- 9.Gupta SC, Patchva S, Koh W, Aggarwal BB. Discovery of curcumin, a component of golden spice, and its miraculous biological activities. Clin Exp Pharmacol Physiol. 2012;39(3):283–299. doi: 10.1111/j.1440-1681.2011.05648.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Barchitta M, Maugeri A, Favara G, Magnano San Lio R, Evola G, Agodi A, et al. Nutrition and wound healing: an overview focusing on the beneficial effects of curcumin. Int J Mol Sci. 2019;20(5):1119. doi: 10.3390/ijms20051119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tosato M, Zamboni V, Ferrini A, Cesari M. The aging process and potential interventions to extend life expectancy. Clin Interv Aging. 2007;2(3):401–412. [PMC free article] [PubMed] [Google Scholar]
- 12.Kiss HJ, Mihalik A, Nánási T, Ory B, Spiró Z, Soti C, et al. Ageing as a price of cooperation and complexity: self-organization of complex systems causes the gradual deterioration of constituent networks. BioEssays : News Rev Mol, Cell Dev Biol. 2009;31(6):651–664. doi: 10.1002/bies.200800224. [DOI] [PubMed] [Google Scholar]
- 13.Xia X, Chen W, McDermott J, Han JJ. Molecular and phenotypic biomarkers of aging. F1000Research. 2017;6:860. doi: 10.12688/f1000research.10692.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cevenini E, Invidia L, Lescai F, Salvioli S, Tieri P, Castellani G, et al. Human models of aging and longevity. Expert Opin Biol Ther. 2008;8(9):1393–1405. doi: 10.1517/14712598.8.9.1393. [DOI] [PubMed] [Google Scholar]
- 15.López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. Hallmarks of aging: an expanding universe. Cell. 2023;186(2):243–78. doi: 10.1016/j.cell.2022.11.001. [DOI] [PubMed] [Google Scholar]
- 16.Shinde P, Kuhikar R, Kulkarni R, Khan N, Limaye L, Kale V. Curcumin restores the engraftment capacity of aged hematopoietic stem cells and also reduces PD-1 expression on cytotoxic T cells. J Tissue Eng Regen Med. 2021;15(4):388–400. doi: 10.1002/term.3180. [DOI] [PubMed] [Google Scholar]
- 17.Brieger K, Schiavone S, Miller FJ, Jr, Krause KH. Reactive oxygen species: from health to disease. Swiss Med Wkly. 2012;142:w13659. doi: 10.4414/smw.2012.13659. [DOI] [PubMed] [Google Scholar]
- 18.Fan J, Ren D, Wang J, Liu X, Zhang H, Wu M, et al. Bruceine D induces lung cancer cell apoptosis and autophagy via the ROS/MAPK signaling pathway in vitro and in vivo. Cell Death Dis. 2020;11(2):126. doi: 10.1038/s41419-020-2317-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Li H, Yu X, Li C, Ma L, Zhao Z, Guan S, et al. Caffeic acid protects against Aβ toxicity and prolongs lifespan in Caenorhabditis elegans models. Food Funct. 2021;12(3):1219–1231. doi: 10.1039/D0FO02784G. [DOI] [PubMed] [Google Scholar]
- 20.Wijaya YT, Setiawan T, Sari IN, Park K, Lee CH, Cho KW, et al. Ginsenoside Rd ameliorates muscle wasting by suppressing the signal transducer and activator of transcription 3 pathway. J Cachexia Sarcopenia Muscle. 2022;13(6):3149–3162. doi: 10.1002/jcsm.13084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Albrahim T, Alonazi M. Effect of blueberry extract on liver in aged rats. Oxid Med Cell Longev. 2022;2022:3490776. doi: 10.1155/2022/3490776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gavia-García G, Rosado-Pérez J, Arista-Ugalde TL, Aguiñiga-Sánchez I, Santiago-Osorio E, Mendoza-Núñez VM. The consumption of Sechium edule (chayote) has antioxidant effect and prevents telomere attrition in older adults with metabolic syndrome. Redox Rep: Commun Free Radic Res. 2023;28(1):2207323. doi: 10.1080/13510002.2023.2207323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Jang HY. Factors associated with successful aging among community-dwelling older adults based on ecological system model. Int J Environ Res Public Health. 2020;17(9):3220. 10.3390/ijerph17093220. [DOI] [PMC free article] [PubMed]
- 24.Troesch B, Eggersdorfer M, Laviano A, Rolland Y, Smith AD, Warnke I, et al. Expert opinion on benefits of long-chain omega-3 fatty acids (DHA and EPA) in aging and clinical nutrition. Nutrients. 2020;12(9):2555. 10.3390/nu12092555. [DOI] [PMC free article] [PubMed]
- 25.Thomas DR. Vitamins in aging, health, and longevity. Clin Interv Aging. 2006;1(1):81–91. doi: 10.2147/ciia.2006.1.1.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gao L, Liu X, Luo X, Lou X, Li P, Li X, et al. Antiaging effects of dietary supplements and natural products. Front Pharmacol. 2023;14:1192714. doi: 10.3389/fphar.2023.1192714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Barcelos IP, Haas RH. CoQ10 and Aging. Biology (Basel). 2019;8(2):28. 10.3390/biology8020028. [DOI] [PMC free article] [PubMed]
- 28.Chen J, Li Y, Zhu Q, Li T, Lu H, Wei N, et al. Anti-skin-aging effect of epigallocatechin gallate by regulating epidermal growth factor receptor pathway on aging mouse model induced by D-galactose. Mech Ageing Dev. 2017;164:1–7. doi: 10.1016/j.mad.2017.03.007. [DOI] [PubMed] [Google Scholar]
- 29.Payne A, Nahashon S, Taka E, Adinew GM, Soliman KFA. Epigallocatechin-3-gallate (EGCG): new therapeutic perspectives for neuroprotection, aging, and neuroinflammation for the modern age. Biomolecules. 2022;12(3):371. doi: 10.3390/biom12030371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ozkur M, Benlier N, Takan I, Vasileiou C, Georgakilas AG, Pavlopoulou A, et al. Ginger for healthy ageing: a systematic review on current evidence of its antioxidant, anti-inflammatory, and anticancer properties. Oxid Med Cell Longev. 2022;2022:4748447. doi: 10.1155/2022/4748447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Al-Atif H. Collagen supplements for aging and wrinkles: a paradigm shift in the fields of dermatology and cosmetics. Dermatol Pract Conceptual. 2022;12(1):e2022018. doi: 10.5826/dpc.1201a18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Benameur T, Soleti R, Panaro MA, La Torre ME, Monda V, Messina G, et al. Curcumin as prospective anti-aging natural compound: focus on brain. Molecules. 2021;26(16):4794. 10.3390/molecules26164794. [DOI] [PMC free article] [PubMed]
- 33.Bjørklund G, Shanaida M, Lysiuk R, Butnariu M, Peana M, Sarac I, et al. Natural compounds and products from an anti-aging perspective. Molecules. 2022;27(20):7084. 10.3390/molecules27207084. [DOI] [PMC free article] [PubMed]
- 34.Prasad S, Gupta SC, Tyagi AK, Aggarwal BB. Curcumin, a component of golden spice: from bedside to bench and back. Biotechnol Adv. 2014;32(6):1053–1064. doi: 10.1016/j.biotechadv.2014.04.004. [DOI] [PubMed] [Google Scholar]
- 35.Esatbeyoglu T, Huebbe P, Ernst IM, Chin D, Wagner AE, Rimbach G. Curcumin—from molecule to biological function. Angew Chem Int Ed Engl. 2012;51(22):5308–5332. doi: 10.1002/anie.201107724. [DOI] [PubMed] [Google Scholar]
- 36.Paulucci VP, Couto RO, Teixeira CCC, Freitas LAP. Optimization of the extraction of curcumin from Curcuma longa rhizomes. Rev Bras Farmacognosia. 2013;23(1):94–100. doi: 10.1590/S0102-695X2012005000117. [DOI] [Google Scholar]
- 37.Payton F, Sandusky P, Alworth WL. NMR study of the solution structure of curcumin. J Nat Prod. 2007;70(2):143–146. doi: 10.1021/np060263s. [DOI] [PubMed] [Google Scholar]
- 38.Priyadarsini KI. Photophysics, photochemistry and photobiology of curcumin: studies from organic solutions, bio-mimetics and living cells. J Photochem Photobiol C: Photochem Rev. 2009;10(2):81–95. doi: 10.1016/j.jphotochemrev.2009.05.001. [DOI] [Google Scholar]
- 39.Goel A, Kunnumakkara AB, Aggarwal BB. Curcumin as “curecumin”: from kitchen to clinic. Biochem Pharmacol. 2008;75(4):787–809. doi: 10.1016/j.bcp.2007.08.016. [DOI] [PubMed] [Google Scholar]
- 40.Aggarwal BB, Kumar A, Bharti AC. Anticancer potential of curcumin: preclinical and clinical studies. Anticancer Res. 2003;23(1A):363–98. [PubMed] [Google Scholar]
- 41.Pulido-Moran M, Moreno-Fernandez J, Ramirez-Tortosa C, Ramirez-Tortosa M. Curcumin and health. Molecules (Basel, Switzerland) 2016;21(3):264. doi: 10.3390/molecules21030264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Luca SV, Macovei I, Bujor A, Miron A, Skalicka-Woźniak K, Aprotosoaie AC, et al. Bioactivity of dietary polyphenols: the role of metabolites. Crit Rev Food Sci Nutr. 2020;60(4):626–659. doi: 10.1080/10408398.2018.1546669. [DOI] [PubMed] [Google Scholar]
- 43.Hoehle SI, Pfeiffer E, Metzler M. Glucuronidation of curcuminoids by human microsomal and recombinant UDP-glucuronosyltransferases. Mol Nutr Food Res. 2007;51(8):932–938. doi: 10.1002/mnfr.200600283. [DOI] [PubMed] [Google Scholar]
- 44.Tsuda T. Curcumin as a functional food-derived factor: degradation products, metabolites, bioactivity, and future perspectives. Food Funct. 2018;9(2):705–714. doi: 10.1039/C7FO01242J. [DOI] [PubMed] [Google Scholar]
- 45.Hoehle SI, Pfeiffer E, Sólyom AM, Metzler M. Metabolism of curcuminoids in tissue slices and subcellular fractions from rat liver. J Agric Food Chem. 2006;54(3):756–764. doi: 10.1021/jf058146a. [DOI] [PubMed] [Google Scholar]
- 46.Huang Y, Cao S, Zhang Q, Zhang H, Fan Y, Qiu F, et al. Biological and pharmacological effects of hexahydrocurcumin, a metabolite of curcumin. Arch Biochem Biophys. 2018;15(646):31–37. doi: 10.1016/j.abb.2018.03.030. [DOI] [PubMed] [Google Scholar]
- 47.Anand P, Kunnumakkara AB, Newman RA, Aggarwal BB. Bioavailability of curcumin: problems and promises. Mol Pharm. 2007;4(6):807–18. doi: 10.1021/mp700113r. [DOI] [PubMed] [Google Scholar]
- 48.Hassaninasab A, Hashimoto Y, Tomita-Yokotani K, Kobayashi M. Discovery of the curcumin metabolic pathway involving a unique enzyme in an intestinal microorganism. Proc Natl Acad Sci USA. 2011;108(16):6615–6620. doi: 10.1073/pnas.1016217108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Heger M, van Golen RF, Broekgaarden M, Michel MC. The molecular basis for the pharmacokinetics and pharmacodynamics of curcumin and its metabolites in relation to cancer. Pharmacol Rev. 2014;66(1):222–307. doi: 10.1124/pr.110.004044. [DOI] [PubMed] [Google Scholar]
- 50.Prasad S, Tyagi AK, Aggarwal BB. Recent developments in delivery, bioavailability, absorption and metabolism of curcumin: the golden pigment from golden spice. Cancer Res Treat. 2014;46(1):2–18. doi: 10.4143/crt.2014.46.1.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ireson CR, Jones DJ, Orr S, Coughtrie MW, Boocock DJ, Williams ML, et al. Metabolism of the cancer chemopreventive agent curcumin in human and rat intestine. Cancer Epidemiol, Biomark Prev: Publ Am Assoc Cancer Res, Cosponsored Am Soc Prev Oncol. 2002;11(1):105–111. [PubMed] [Google Scholar]
- 52.Aggarwal BB, Deb L, Prasad S. Curcumin differs from tetrahydrocurcumin for molecular targets, signaling pathways and cellular responses. Molecules (Basel, Switzerland) 2014;20(1):185–205. doi: 10.3390/molecules20010185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Tomeh MA, Hadianamrei R, Zhao X. A review of curcumin and its derivatives as anticancer agents. Int J Mol Sci. 2019;20(5):1033. 10.3390/ijms20051033. [DOI] [PMC free article] [PubMed]
- 54.Nelson KM, Dahlin JL, Bisson J, Graham J, Pauli GF, Walters MA. The essential medicinal chemistry of curcumin. J Med Chem. 2017;60(5):1620–1637. doi: 10.1021/acs.jmedchem.6b00975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Gupta SC, Patchva S, Aggarwal BB. Therapeutic roles of curcumin: lessons learned from clinical trials. AAPS J. 2013;15(1):195–218. doi: 10.1208/s12248-012-9432-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Mirzaei H, Shakeri A, Rashidi B, Jalili A, Banikazemi Z, Sahebkar A. Phytosomal curcumin: a review of pharmacokinetic, experimental and clinical studies. Biomed Pharm= Biomedecine & pharmacotherapie. 2017;85:102–12. doi: 10.1016/j.biopha.2016.11.098. [DOI] [PubMed] [Google Scholar]
- 57.Db M, Sreedharan S, Mahadik K. Role of piperine as an effective bioenhancer in drug absorption. Pharm Anal Acta. 2018;9(7):1–4. doi: 10.4172/2153-2435.1000591. [DOI] [Google Scholar]
- 58.Teiten M-H, Dicato M, Diederich M. Hybrid curcumin compounds: a new strategy for cancer treatment. Molecules (Basel, Switzerland) 2014;19(12):20839–20863. doi: 10.3390/molecules191220839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Pancholi V, Smina TP, Kunnumakkara AB, Maliakel B, Krishnakumar IM. Safety assessment of a highly bioavailable curcumin-galactomannoside complex (CurQfen) in healthy volunteers, with a special reference to the recent hepatotoxic reports of curcumin supplements: a 90-days prospective study. Toxicol Rep. 2021;8:1255–1264. doi: 10.1016/j.toxrep.2021.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Pal A, Sung B, Bhanu Prasad BA, Schuber PT, Jr, Prasad S, Aggarwal BB, et al. Curcumin glucuronides: assessing the proliferative activity against human cell lines. Bioorg Med Chem. 2014;22(1):435–439. doi: 10.1016/j.bmc.2013.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Stohs S, Ray S. Issues with human bioavailability determinations of bioactive curcumin. Biomed J Sci Tech Res. 2019;12(4):9417–9419. [Google Scholar]
- 62.Sasaki H, Sunagawa Y, Takahashi K, Imaizumi A, Fukuda H, Hashimoto T, et al. Innovative preparation of curcumin for improved oral bioavailability. Biol Pharm Bull. 2011;34(5):660–665. doi: 10.1248/bpb.34.660. [DOI] [PubMed] [Google Scholar]
- 63.Kanai M, Imaizumi A, Otsuka Y, Sasaki H, Hashiguchi M, Tsujiko K, et al. Dose-escalation and pharmacokinetic study of nanoparticle curcumin, a potential anticancer agent with improved bioavailability, in healthy human volunteers. Cancer Chemother Pharmacol. 2012;69(1):65–70. doi: 10.1007/s00280-011-1673-1. [DOI] [PubMed] [Google Scholar]
- 64.Nakagawa Y, Mukai S, Yamada S, Matsuoka M, Tarumi E, Hashimoto T, et al. Short-term effects of highly-bioavailable curcumin for treating knee osteoarthritis: a randomized, double-blind, placebo-controlled prospective study. J Orthop Sci: Off J Japan Orthop Assoc. 2014;19(6):933–939. doi: 10.1007/s00776-014-0633-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Purpura M, Lowery RP, Wilson JM, Mannan H, Münch G, Razmovski-Naumovski V. Analysis of different innovative formulations of curcumin for improved relative oral bioavailability in human subjects. Eur J Nutr. 2018;57(3):929–938. doi: 10.1007/s00394-016-1376-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Selvendiran K, Kuppusamy ML, Bratasz A, Tong L, Rivera BK, Rink C, et al. Inhibition of vascular smooth-muscle cell proliferation and arterial restenosis by HO-3867, a novel synthetic curcuminoid, through up-regulation of PTEN expression. J Pharmacol Exp Ther. 2009;329(3):959–966. doi: 10.1124/jpet.108.150367. [DOI] [PubMed] [Google Scholar]
- 67.Dinkova-Kostova AT, Cory AH, Bozak RE, Hicks RJ, Cory JG. Bis(2-hydroxybenzylidene)acetone, a potent inducer of the phase 2 response, causes apoptosis in mouse leukemia cells through a p53-independent, caspase-mediated pathway. Cancer Lett. 2007;245(1–2):341–349. doi: 10.1016/j.canlet.2006.01.024. [DOI] [PubMed] [Google Scholar]
- 68.Tamvakopoulos C, Dimas K, Sofianos ZD, Hatziantoniou S, Han Z, Liu ZL, et al. Metabolism and anticancer activity of the curcumin analogue, dimethoxycurcumin. Clin Cancer Res: Off J Am Assoc Cancer Res. 2007;13(4):1269–1277. doi: 10.1158/1078-0432.CCR-06-1839. [DOI] [PubMed] [Google Scholar]
- 69.He Y, Li W, Hu G, Sun H, Kong Q. Bioactivities of EF24, a novel curcumin analog: a review. Front Oncol. 2018;8:614. doi: 10.3389/fonc.2018.00614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Jeon OH, Kim C, Laberge RM, Demaria M, Rathod S, Vasserot AP, et al. Local clearance of senescent cells attenuates the development of post-traumatic osteoarthritis and creates a pro-regenerative environment. Nat Med. 2017;23(6):775–781. doi: 10.1038/nm.4324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Baker DJ, Wijshake T, Tchkonia T, LeBrasseur NK, Childs BG, van de Sluis B, et al. Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature. 2011;479(7372):232–236. doi: 10.1038/nature10600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Hall BM, Balan V, Gleiberman AS, Strom E, Krasnov P, Virtuoso LP, et al. Aging of mice is associated with p16(Ink4a)- and β-galactosidase-positive macrophage accumulation that can be induced in young mice by senescent cells. Aging. 2016;8(7):1294–1315. doi: 10.18632/aging.100991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Naylor RM, Baker DJ, van Deursen JM. Senescent cells: a novel therapeutic target for aging and age-related diseases. Clin Pharmacol Ther. 2013;93(1):105–116. doi: 10.1038/clpt.2012.193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Adams BK, Ferstl EM, Davis MC, Herold M, Kurtkaya S, Camalier RF, et al. Synthesis and biological evaluation of novel curcumin analogs as anti-cancer and anti-angiogenesis agents. Bioorg Med Chem. 2004;12(14):3871–3883. doi: 10.1016/j.bmc.2004.05.006. [DOI] [PubMed] [Google Scholar]
- 75.Rath KS, Naidu SK, Lata P, Bid HK, Rivera BK, McCann GA, et al. HO-3867, a safe STAT3 inhibitor, is selectively cytotoxic to ovarian cancer. Cancer Res. 2014;74(8):2316–2327. doi: 10.1158/0008-5472.CAN-13-2433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Li W, He Y, Zhang R, Zheng G, Zhou D. The curcumin analog EF24 is a novel senolytic agent. Aging. 2019;11(2):771–782. doi: 10.18632/aging.101787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Kunnumakkara AB, Bordoloi D, Padmavathi G, Monisha J, Roy NK, Prasad S, et al. Curcumin, the golden nutraceutical: multitargeting for multiple chronic diseases. Br J Pharmacol. 2017;174(11):1325–1348. doi: 10.1111/bph.13621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Yadav VS, Mishra KP, Singh DP, Mehrotra S, Singh VK. Immunomodulatory effects of curcumin. Immunopharmacol Immunotoxicol. 2005;27(3):485–97. doi: 10.1080/08923970500242244. [DOI] [PubMed] [Google Scholar]
- 79.Gorabi AM, Hajighasemi S, Kiaie N, Rosano GMC, Sathyapalan T, Al-Rasadi K, et al. Anti-fibrotic effects of curcumin and some of its analogues in the heart. Heart Fail Rev. 2020;25(5):731–43. doi: 10.1007/s10741-019-09854-6. [DOI] [PubMed] [Google Scholar]
- 80.Carolina Alves R, PerosaFernandes R, Fonseca-Santos B, DamianiVictorelli F, Chorilli M. A critical review of the properties and analytical methods for the determination of curcumin in biological and pharmaceutical matrices. Crit Rev Anal Chem. 2019;49(2):138–49. doi: 10.1080/10408347.2018.1489216. [DOI] [PubMed] [Google Scholar]
- 81.Leiherer A, Mündlein A, Drexel H. Phytochemicals and their impact on adipose tissue inflammation and diabetes. Vasc Pharmacol. 2013;58(1):3–20. doi: 10.1016/j.vph.2012.09.002. [DOI] [PubMed] [Google Scholar]
- 82.Maldonado E, Morales-Pison S, Urbina F, Solari A. Aging hallmarks and the role of oxidative stress. Antioxidants. 2023;12(3):651. doi: 10.3390/antiox12030651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Alizadeh M, Kheirouri S. Curcumin reduces malondialdehyde and improves antioxidants in humans with diseased conditions: a comprehensive meta-analysis of randomized controlled trials. Biomedicine. 2019;9(4):23. doi: 10.1051/bmdcn/2019090423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Abrahams S, Haylett WL, Johnson G, Carr JA, Bardien S. Antioxidant effects of curcumin in models of neurodegeneration, aging, oxidative and nitrosative stress: a review. Neuroscience. 2019;15(406):1–21. doi: 10.1016/j.neuroscience.2019.02.020. [DOI] [PubMed] [Google Scholar]
- 85.Abete P, Napoli C, Santoro G, Ferrara N, Tritto I, Chiariello M, et al. Age-related decrease in cardiac tolerance to oxidative stress. J Mol Cell Cardiol. 1999;31(1):227–236. doi: 10.1006/jmcc.1998.0862. [DOI] [PubMed] [Google Scholar]
- 86.Jeeva JS, Sunitha J, Ananthalakshmi R, Rajkumari S, Ramesh M, Krishnan R. Enzymatic antioxidants and its role in oral diseases. J Pharm Bioallied Sci. 2015;7(Suppl 2):S331–S333. doi: 10.4103/0975-7406.163438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Poljsak B. Strategies for reducing or preventing the generation of oxidative stress. Oxid Med Cell Longev. 2011;2011:194586. doi: 10.1155/2011/194586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Chilelli NC, Ragazzi E, Valentini R, Cosma C, Ferraresso S, Lapolla A, et al. Curcumin and Boswellia serrata modulate the glyco-oxidative status and lipo-oxidation in master athletes. Nutrients. 2016;8(11):745. 10.3390/nu8110745. [DOI] [PMC free article] [PubMed]
- 89.Pawelec G, Goldeck D, Derhovanessian E. Inflammation, ageing and chronic disease. Curr Opin Immunol. 2014;29:23–8. doi: 10.1016/j.coi.2014.03.007. [DOI] [PubMed] [Google Scholar]
- 90.Sanada F, Taniyama Y, Muratsu J, Otsu R, Shimizu H, Rakugi H, et al. Source of chronic inflammation in aging. Front Cardiovasc Med. 2018;5:12. doi: 10.3389/fcvm.2018.00012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Fulop T, McElhaney J, Pawelec G, Cohen AA, Morais JA, Dupuis G, et al. Frailty, inflammation and immunosenescence. Interdisc Topics Gerontol Geriatr. 2015;41:26–40. doi: 10.1159/000381134. [DOI] [PubMed] [Google Scholar]
- 92.Peng Y, Ao M, Dong B, Jiang Y, Yu L, Chen Z, et al. Anti-inflammatory effects of curcumin in the inflammatory diseases: status, limitations and countermeasures. Drug Des Dev Ther. 2021;15:4503–4525. doi: 10.2147/DDDT.S327378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Lingappan K. NF-κB in oxidative stress. Curr Opin Toxicol. 2018;7:81–86. doi: 10.1016/j.cotox.2017.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Grabowska W, Suszek M, Wnuk M, Lewinska A, Wasiak E, Sikora E, et al. Curcumin elevates sirtuin level but does not postpone in vitro senescence of human cells building the vasculature. Oncotarget. 2016;7(15):19201–19213. doi: 10.18632/oncotarget.8450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Frasca D, Diaz A, Romero M, Garcia D, Blomberg BB. B cell immunosenescence. Annu Rev Cell Dev Biol. 2020;6(36):551–574. doi: 10.1146/annurev-cellbio-011620-034148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Alizadeh F, Javadi M, Karami AA, Gholaminejad F, Kavianpour M, Haghighian HK. Curcumin nanomicelle improves semen parameters, oxidative stress, inflammatory biomarkers, and reproductive hormones in infertile men: a randomized clinical trial. Phytother Res: PTR. 2018;32(3):514–521. doi: 10.1002/ptr.5998. [DOI] [PubMed] [Google Scholar]
- 97.Inadera H, Egashira K, Takemoto M, Ouchi Y, Matsushima K. Increase in circulating levels of monocyte chemoattractant protein-1 with aging. J Interferon Cytokine Res: Off J Int Soc Interferon Cytokine Res. 1999;19(10):1179–1182. doi: 10.1089/107999099313127. [DOI] [PubMed] [Google Scholar]
- 98.Yousefzadeh MJ, Schafer MJ, Noren Hooten N, Atkinson EJ, Evans MK, Baker DJ, et al. Circulating levels of monocyte chemoattractant protein-1 as a potential measure of biological age in mice and frailty in humans. Aging Cell. 2018;17(2):e12706. 10.1111/acel.12706. [DOI] [PMC free article] [PubMed]
- 99.Karimian MS, Pirro M, Majeed M, Sahebkar A. Curcumin as a natural regulator of monocyte chemoattractant protein-1. Cytokine Growth Factor Rev. 2017;33:55–63. doi: 10.1016/j.cytogfr.2016.10.001. [DOI] [PubMed] [Google Scholar]
- 100.Franco S, Blasco MA, Siedlak SL, Harris PL, Moreira PI, Perry G, et al. Telomeres and telomerase in Alzheimer’s disease: epiphenomena or a new focus for therapeutic strategy? Alzheimer's Dement. 2006;2(3):164–168. doi: 10.1016/j.jalz.2006.03.001. [DOI] [PubMed] [Google Scholar]
- 101.Satyanarayana A, Greenberg RA, Schaetzlein S, Buer J, Masutomi K, Hahn WC, et al. Mitogen stimulation cooperates with telomere shortening to activate DNA damage responses and senescence signaling. Mol Cell Biol. 2004;24(12):5459–5474. doi: 10.1128/MCB.24.12.5459-5474.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Xiao Z, Zhang A, Lin J, Zheng Z, Shi X, Di W, et al. Telomerase: a target for therapeutic effects of curcumin and a curcumin derivative in Aβ1-42 insult in vitro. PLoS One. 2014;9(7):e101251. doi: 10.1371/journal.pone.0101251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Taka T, Changtam C, Thaichana P, Kaewtunjai N, Suksamrarn A, Lee TR, et al. Curcuminoid derivatives enhance telomerase activity in an in vitro TRAP assay. Bioorg Med Chem Lett. 2014;24(22):5242–5246. doi: 10.1016/j.bmcl.2014.09.059. [DOI] [PubMed] [Google Scholar]
- 104.Forouzanfar F, Majeed M, Jamialahmadi T, Sahebkar A. Telomerase: a target for therapeutic effects of curcumin in cancer. Adv Exp Med Biol. 2021;1286:135–143. doi: 10.1007/978-3-030-55035-6_10. [DOI] [PubMed] [Google Scholar]
- 105.Johnson SC. Nutrient sensing, signaling and ageing: the role of IGF-1 and mTOR in ageing and age-related disease. Subcell Biochem. 2018;90:49–97. doi: 10.1007/978-981-13-2835-0_3. [DOI] [PubMed] [Google Scholar]
- 106.Weichhart T. Mammalian target of rapamycin: a signaling kinase for every aspect of cellular life. Methods Mol Biol (Clifton, NJ) 2012;821:1–14. doi: 10.1007/978-1-61779-430-8_1. [DOI] [PubMed] [Google Scholar]
- 107.Fabrizio P, Pozza F, Pletcher SD, Gendron CM, Longo VD. Regulation of longevity and stress resistance by Sch9 in yeast. Science (New York, NY) 2001;292(5515):288–290. doi: 10.1126/science.1059497. [DOI] [PubMed] [Google Scholar]
- 108.Vellai T, Takacs-Vellai K, Zhang Y, Kovacs AL, Orosz L, Müller F. Genetics: influence of TOR kinase on lifespan in C. elegans. Nature. 2003;426(6967):620. doi: 10.1038/426620a. [DOI] [PubMed] [Google Scholar]
- 109.Kapahi P, Zid BM, Harper T, Koslover D, Sapin V, Benzer S. Regulation of lifespan in Drosophila by modulation of genes in the TOR signaling pathway. Curr Biol: CB. 2004;14(10):885–890. doi: 10.1016/j.cub.2004.03.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Kaeberlein M, Powers RW, 3rd, Steffen KK, Westman EA, Hu D, Dang N, et al. Regulation of yeast replicative life span by TOR and Sch9 in response to nutrients. Science (New York, NY) 2005;310(5751):1193–1196. doi: 10.1126/science.1115535. [DOI] [PubMed] [Google Scholar]
- 111.Beevers CS, Li F, Liu L, Huang S. Curcumin inhibits the mammalian target of rapamycin-mediated signaling pathways in cancer cells. Int J Cancer. 2006;119(4):757–764. doi: 10.1002/ijc.21932. [DOI] [PubMed] [Google Scholar]
- 112.Johnson SM, Gulhati P, Arrieta I, Wang X, Uchida T, Gao T, et al. Curcumin inhibits proliferation of colorectal carcinoma by modulating Akt/mTOR signaling. Anticancer Res. 2009;29(8):3185–3190. [PMC free article] [PubMed] [Google Scholar]
- 113.Lim HW, Lim HY, Wong KP. Uncoupling of oxidative phosphorylation by curcumin: implication of its cellular mechanism of action. Biochem Biophys Res Commun. 2009;389(1):187–192. doi: 10.1016/j.bbrc.2009.08.121. [DOI] [PubMed] [Google Scholar]
- 114.Ulgherait M, Rana A, Rera M, Graniel J, Walker DW. AMPK modulates tissue and organismal aging in a non-cell-autonomous manner. Cell Rep. 2014;8(6):1767–1780. doi: 10.1016/j.celrep.2014.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Dumitrascu GR, Bucur O. Critical physiological and pathological functions of Forkhead Box O tumor suppressors. Discoveries (Craiova, Romania) 2013;1(1):e5. doi: 10.15190/d.2013.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Kim T, Davis J, Zhang AJ, He X, Mathews ST. Curcumin activates AMPK and suppresses gluconeogenic gene expression in hepatoma cells. Biochem Biophys Res Commun. 2009;388(2):377–382. doi: 10.1016/j.bbrc.2009.08.018. [DOI] [PubMed] [Google Scholar]
- 117.Stancu AL. AMPK activation can delay aging. Discoveries (Craiova, Romania) 2015;3(4):e53. doi: 10.15190/d.2015.45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Liu Z, Cui C, Xu P, Dang R, Cai H, Liao D, et al. Curcumin activates AMPK pathway and regulates lipid metabolism in rats following prolonged clozapine exposure. Front Neurosci. 2017;11:558. doi: 10.3389/fnins.2017.00558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Morris BJ. Seven sirtuins for seven deadly diseases of aging. Free Radical Biol Med. 2013;56:133–171. doi: 10.1016/j.freeradbiomed.2012.10.525. [DOI] [PubMed] [Google Scholar]
- 120.Yang Y, Duan W, Lin Y, Yi W, Liang Z, Yan J, et al. SIRT1 activation by curcumin pretreatment attenuates mitochondrial oxidative damage induced by myocardial ischemia reperfusion injury. Free Radic Biol Med. 2013;65:667–79. doi: 10.1016/j.freeradbiomed.2013.07.007. [DOI] [PubMed] [Google Scholar]
- 121.Rodgers JT, Lerin C, Haas W, Gygi SP, Spiegelman BM, Puigserver P. Nutrient control of glucose homeostasis through a complex of PGC-1alpha and SIRT1. Nature. 2005;434(7029):113–118. doi: 10.1038/nature03354. [DOI] [PubMed] [Google Scholar]
- 122.Chen K, An Y, Tie L, Pan Y, Li X. Curcumin protects neurons from glutamate-induced excitotoxicity by membrane anchored AKAP79-PKA interaction network. Evid-Based Complement Altern Med : eCAM. 2015;2015:706207. doi: 10.1155/2015/706207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Lin CH, Lin CC, Ting WJ, Pai PY, Kuo CH, Ho TJ, et al. Resveratrol enhanced FOXO3 phosphorylation via synergetic activation of SIRT1 and PI3K/Akt signaling to improve the effects of exercise in elderly rat hearts. Age (Dordr) 2014;36(5):9705. doi: 10.1007/s11357-014-9705-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Sahin K, Pala R, Tuzcu M, Ozdemir O, Orhan C, Sahin N, et al. Curcumin prevents muscle damage by regulating NF-κB and Nrf2 pathways and improves performance: an in vivo model. J Inflamm Res. 2016;9:147–154. doi: 10.2147/JIR.S110873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Ray Hamidie RD, Yamada T, Ishizawa R, Saito Y, Masuda K. Curcumin treatment enhances the effect of exercise on mitochondrial biogenesis in skeletal muscle by incrseasing camp levels. Metab: Clin Exp. 2015;64(10):1334–47. doi: 10.1016/j.metabol.2015.07.010. [DOI] [PubMed] [Google Scholar]
- 126.Huang WC, Chiu WC, Chuang HL, Tang DW, Lee ZM, Wei L, et al. Effect of curcumin supplementation on physiological fatigue and physical performance in mice. Nutrients. 2015;7(2):905–921. doi: 10.3390/nu7020905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Ionescu-Tucker A, Cotman CW. Emerging roles of oxidative stress in brain aging and Alzheimer’s disease. Neurobiol Aging. 2021;107:86–95. doi: 10.1016/j.neurobiolaging.2021.07.014. [DOI] [PubMed] [Google Scholar]
- 128.Fu Y, Yang J, Wang X, Yang P, Zhao Y, Li K, et al. Herbal compounds play a role in neuroprotection through the inhibition of microglial activation. J Immunol Res. 2018;2018:9348046. doi: 10.1155/2018/9348046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Liu Z, Ran Y, Huang S, Wen S, Zhang W, Liu X, et al. Curcumin protects against ischemic stroke by titrating microglia/macrophage polarization. Front Aging Neurosci. 2017;9:233. doi: 10.3389/fnagi.2017.00233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Yu Y, Shen Q, Lai Y, Park SY, Ou X, Lin D, et al. Anti-inflammatory effects of curcumin in microglial cells. Front Pharmacol. 2018;9:386. doi: 10.3389/fphar.2018.00386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Hatami M, Abdolahi M, Soveyd N, Djalali M, Togha M, Honarvar NM. Molecular mechanisms of curcumin in neuroinflammatory disorders: a mini review of current evidences. Endocr Metab Immune Disord Drug Targets. 2019;19(3):247–258. doi: 10.2174/1871530319666181129103056. [DOI] [PubMed] [Google Scholar]
- 132.Kang G, Kong PJ, Yuh YJ, Lim SY, Yim SV, Chun W, et al. Curcumin suppresses lipopolysaccharide-induced cyclooxygenase-2 expression by inhibiting activator protein 1 and nuclear factor kappab bindings in BV2 microglial cells. J Pharmacol Sci. 2004;94(3):325–328. doi: 10.1254/jphs.94.325. [DOI] [PubMed] [Google Scholar]
- 133.Parada E, Buendia I, Navarro E, Avendaño C, Egea J, López MG. Microglial HO-1 induction by curcumin provides antioxidant, antineuroinflammatory, and glioprotective effects. Mol Nutr Food Res. 2015;59(9):1690–1700. doi: 10.1002/mnfr.201500279. [DOI] [PubMed] [Google Scholar]
- 134.Wang Y, Yin Z, Gao L, Sun D, Hu X, Xue L, et al. Curcumin delays retinal degeneration by regulating microglia activation in the retina of rd1 mice. Cell Physiol Biochem: Int J Exp Cell Physiol, Biochem, Pharmacol. 2017;44(2):479–493. doi: 10.1159/000485085. [DOI] [PubMed] [Google Scholar]
- 135.Funk JL, Schneider C. Perspective on improving the relevance, rigor, and reproducibility of botanical clinical trials: lessons learned from turmeric trials. Front Nutr. 2021;8:782912. doi: 10.3389/fnut.2021.782912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Giordano A, Tommonaro G. Curcumin and cancer. Nutrients. 2019;11(10):2376. 10.3390/nu11102376. [DOI] [PMC free article] [PubMed]
- 137.Singh L, Sharma S, Xu S, Tewari D, Fang J. Curcumin as a natural remedy for atherosclerosis: a pharmacological review. Molecules. 2021;26(13):4036. 10.3390/molecules26134036. [DOI] [PMC free article] [PubMed]
- 138.Šudomová M, Hassan STS. Nutraceutical curcumin with promising protection against herpesvirus infections and their associated inflammation: mechanisms and pathways. Microorganisms. 2021;9(2):292. 10.3390/microorganisms9020292. [DOI] [PMC free article] [PubMed]
- 139.Kunihiro AG, Luis PB, Frye JB, Chew W, Chow HH, Schneider C, et al. Bone-specific metabolism of dietary polyphenols in resorptive bone diseases. Mol Nutr Food Res. 2020;64(14):e2000072. doi: 10.1002/mnfr.202000072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Kunihiro AG, Luis PB, Brickey JA, Frye JB, Chow HS, Schneider C, et al. Beta-glucuronidase catalyzes deconjugation and activation of curcumin-glucuronide in bone. J Nat Prod. 2019;82(3):500–509. doi: 10.1021/acs.jnatprod.8b00873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Krueger JG, Brunner PM. Interleukin-17 alters the biology of many cell types involved in the genesis of psoriasis, systemic inflammation and associated comorbidities. Exp Dermatol. 2018;27(2):115–123. doi: 10.1111/exd.13467. [DOI] [PubMed] [Google Scholar]
- 142.Benham H, Norris P, Goodall J, Wechalekar MD, FitzGerald O, Szentpetery A, et al. Th17 and Th22 cells in psoriatic arthritis and psoriasis. Arthritis Res Ther. 2013;15(5):R136. doi: 10.1186/ar4317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Zheng Y, Danilenko DM, Valdez P, Kasman I, Eastham-Anderson J, Wu J, et al. Interleukin-22, a TH17 cytokine, mediates IL-23-induced dermal inflammation and acanthosis. Nature. 2007;445(7128):648–51. doi: 10.1038/nature05505. [DOI] [PubMed] [Google Scholar]
- 144.Bahraini P, Rajabi M, Mansouri P, Sarafian G, Chalangari R, Azizian Z. Turmeric tonic as a treatment in scalp psoriasis: a randomized placebo-control clinical trial. J Cosmet Dermatol. 2018;17(3):461–466. doi: 10.1111/jocd.12513. [DOI] [PubMed] [Google Scholar]
- 145.Schallreuter KU, Wood JM, Pittelkow MR, Buttner G, Swanson N, Korner C, et al. Increased monoamine oxidase A activity in the epidermis of patients with vitiligo. Arch Dermatol Res. 1996;288(1):14–18. doi: 10.1007/BF02505037. [DOI] [PubMed] [Google Scholar]
- 146.Arca E, Taştan HB, Erbil AH, Sezer E, Koç E, Kurumlu Z. Narrow-band ultraviolet B as monotherapy and in combination with topical calcipotriol in the treatment of vitiligo. J Dermatol. 2006;33(5):338–343. doi: 10.1111/j.1346-8138.2006.00079.x. [DOI] [PubMed] [Google Scholar]
- 147.Asawanonda P, Klahan SO. Tetrahydrocurcuminoid cream plus targeted narrowband UVB phototherapy for vitiligo: a preliminary randomized controlled study. Photomed Laser Surg. 2010;28(5):679–684. doi: 10.1089/pho.2009.2637. [DOI] [PubMed] [Google Scholar]
- 148.Mattson MP, Rydel RE. Alzheimer’s disease. Amyloid ox-tox transducers. Nature. 1996;382(6593):674–675. doi: 10.1038/382674a0. [DOI] [PubMed] [Google Scholar]
- 149.Baum L, Lam CW, Cheung SK, Kwok T, Lui V, Tsoh J, et al. Six-month randomized, placebo-controlled, double-blind, pilot clinical trial of curcumin in patients with Alzheimer disease. J Clin Psychopharmacol. 2008;28(1):110–113. doi: 10.1097/jcp.0b013e318160862c. [DOI] [PubMed] [Google Scholar]
- 150.Chen M, Du ZY, Zheng X, Li DL, Zhou RP, Zhang K. Use of curcumin in diagnosis, prevention, and treatment of Alzheimer’s disease. Neural Regen Res. 2018;13(4):742–752. doi: 10.4103/1673-5374.230303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.North BJ, Sinclair DA. The intersection between aging and cardiovascular disease. Circ Res. 2012;110(8):1097–1108. doi: 10.1161/CIRCRESAHA.111.246876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Li H, Sureda A, Devkota HP, Pittalà V, Barreca D, Silva AS, et al. Curcumin, the golden spice in treating cardiovascular diseases. Biotechnol Adv. 2020;38:107343. doi: 10.1016/j.biotechadv.2019.01.010. [DOI] [PubMed] [Google Scholar]
- 153.Santos-Parker JR, Strahler TR, Bassett CJ, Bispham NZ, Chonchol MB, Seals DR. Curcumin supplementation improves vascular endothelial function in healthy middle-aged and older adults by increasing nitric oxide bioavailability and reducing oxidative stress. Aging. 2017;9(1):187–208. doi: 10.18632/aging.101149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Sugawara J, Akazawa N, Miyaki A, Choi Y, Tanabe Y, Imai T, et al. Effect of endurance exercise training and curcumin intake on central arterial hemodynamics in postmenopausal women: pilot study. Am J Hypertens. 2012;25(6):651–656. doi: 10.1038/ajh.2012.24. [DOI] [PubMed] [Google Scholar]
- 155.Akazawa N, Choi Y, Miyaki A, Tanabe Y, Sugawara J, Ajisaka R, et al. Curcumin ingestion and exercise training improve vascular endothelial function in postmenopausal women. Nutr Res (New York, NY) 2012;32(10):795–799. doi: 10.1016/j.nutres.2012.09.002. [DOI] [PubMed] [Google Scholar]
- 156.Pungcharoenkul K, Thongnopnua P. Effect of different curcuminoid supplement dosages on total in vivo antioxidant capacity and cholesterol levels of healthy human subjects. Phytother Res: PTR. 2011;25(11):1721–1726. doi: 10.1002/ptr.3608. [DOI] [PubMed] [Google Scholar]
- 157.Panahi Y, Ahmadi Y, Teymouri M, Johnston TP, Sahebkar A. Curcumin as a potential candidate for treating hyperlipidemia: a review of cellular and metabolic mechanisms. J Cell Physiol. 2018;233(1):141–152. doi: 10.1002/jcp.25756. [DOI] [PubMed] [Google Scholar]
- 158.DiSilvestro RA, Joseph E, Zhao S, Bomser J. Diverse effects of a low dose supplement of lipidated curcumin in healthy middle aged people. Nutr J. 2012;26(11):79. doi: 10.1186/1475-2891-11-79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Lao CD, Ruffin MTT, Normolle D, Heath DD, Murray SI, Bailey JM, et al. Dose escalation of a curcuminoid formulation. BMC Complement Alternat Med. 2006;6:10. doi: 10.1186/1472-6882-6-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Sharma RA, Euden SA, Platton SL, Cooke DN, Shafayat A, Hewitt HR, et al. Phase I clinical trial of oral curcumin: biomarkers of systemic activity and compliance. Clin Cancer Res: Off J Am Assoc Cancer Res. 2004;10(20):6847–6854. doi: 10.1158/1078-0432.CCR-04-0744. [DOI] [PubMed] [Google Scholar]
- 161.Chainani-Wu N. Safety and anti-inflammatory activity of curcumin: a component of tumeric (Curcuma longa) J Alternat Complement Med (New York, NY) 2003;9(1):161–168. doi: 10.1089/107555303321223035. [DOI] [PubMed] [Google Scholar]
- 162.Hegde M, Girisa S, BharathwajChetty B, Vishwa R, Kunnumakkara AB. Curcumin formulations for better bioavailability: what we learned from clinical trials thus far? ACS Omega. 2023;8(12):10713–46. doi: 10.1021/acsomega.2c07326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Saraf-Bank S, Ahmadi A, Paknahad Z, Maracy M, Nourian M. Effects of curcumin on cardiovascular risk factors in obese and overweight adolescent girls: a randomized clinical trial. Sao Paulo Med J= Revista paulista de medicina. 2019;137(5):414–22. doi: 10.1590/1516-3180.2018.0454120419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Panahi Y, Saadat A, Beiraghdar F, Sahebkar A. Adjuvant therapy with bioavailability-boosted curcuminoids suppresses systemic inflammation and improves quality of life in patients with solid tumors: a randomized double-blind placebo-controlled trial. Phytother Res: PTR. 2014;28(10):1461–1467. doi: 10.1002/ptr.5149. [DOI] [PubMed] [Google Scholar]
- 165.Valizadeh H, Abdolmohammadi-Vahid S, Danshina S, Ziya Gencer M, Ammari A, Sadeghi A, et al. Nano-curcumin therapy, a promising method in modulating inflammatory cytokines in COVID-19 patients. Int Immunopharmacol. 2020;89(Pt B):107088. doi: 10.1016/j.intimp.2020.107088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Rahmani S, Asgary S, Askari G, Keshvari M, Hatamipour M, Feizi A, et al. Treatment of non-alcoholic fatty liver disease with curcumin: a randomized placebo-controlled trial. Phytother Res: PTR. 2016;30(9):1540–1548. doi: 10.1002/ptr.5659. [DOI] [PubMed] [Google Scholar]
- 167.Shep D, Khanwelkar C, Gade P, Karad S. Safety and efficacy of curcumin versus diclofenac in knee osteoarthritis: a randomized open-label parallel-arm study. Trials. 2019;20(1):214. doi: 10.1186/s13063-019-3327-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Steigerwalt R, Nebbioso M, Appendino G, Belcaro G, Ciammaichella G, Cornelli U, et al. Meriva®, a lecithinized curcumin delivery system, in diabetic microangiopathy and retinopathy. Panminerva Med. 2012;54(1 Suppl 4):11–16. [PubMed] [Google Scholar]
- 169.Pivari F, Mingione A, Piazzini G, Ceccarani C, Ottaviano E, Brasacchio C, et al. Curcumin supplementation (Meriva(®)) modulates inflammation, lipid peroxidation and gut microbiota composition in chronic kidney disease. Nutrients. 2022;14(1). [DOI] [PMC free article] [PubMed]
- 170.Appendino G, Belcaro G, Cornelli U, Luzzi R, Togni S, Dugall M, et al. Potential role of curcumin phytosome (Meriva) in controlling the evolution of diabetic microangiopathy. A pilot study. Panminerva Med. 2011;53(3 Suppl 1):43–49. [PubMed] [Google Scholar]
- 171.Belcaro G, Cesarone MR, Dugall M, Pellegrini L, Ledda A, Grossi MG, et al. Product-evaluation registry of Meriva®, a curcumin-phosphatidylcholine complex, for the complementary management of osteoarthritis. Panminerva Med. 2010;52(2 Suppl 1):55–62. [PubMed] [Google Scholar]
- 172.Polasa K, Raghuram TC, Krishna TP, Krishnaswamy K. Effect of turmeric on urinary mutagens in smokers. Mutagenesis. 1992;7(2):107–109. doi: 10.1093/mutage/7.2.107. [DOI] [PubMed] [Google Scholar]
- 173.Holt PR, Katz S, Kirshoff R. Curcumin therapy in inflammatory bowel disease: a pilot study. Dig Dis Sci. 2005;50(11):2191–2193. doi: 10.1007/s10620-005-3032-8. [DOI] [PubMed] [Google Scholar]
- 174.Kalpravidh RW, Siritanaratkul N, Insain P, Charoensakdi R, Panichkul N, Hatairaktham S, et al. Improvement in oxidative stress and antioxidant parameters in beta-thalassemia/Hb E patients treated with curcuminoids. Clin Biochem. 2010;43(4–5):424–429. doi: 10.1016/j.clinbiochem.2009.10.057. [DOI] [PubMed] [Google Scholar]
- 175.Ide H, Tokiwa S, Sakamaki K, Nishio K, Isotani S, Muto S, et al. Combined inhibitory effects of soy isoflavones and curcumin on the production of prostate-specific antigen. Prostate. 2010;70(10):1127–1133. doi: 10.1002/pros.21147. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.