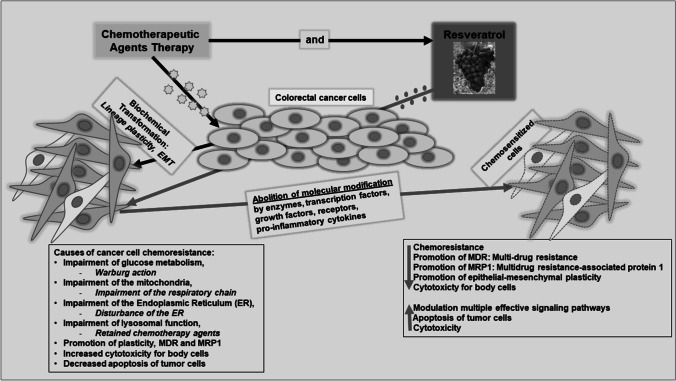
Abstract
Abstract
Despite tremendous medical treatment successes, colorectal cancer (CRC) remains a leading cause of cancer deaths worldwide. Chemotherapy as monotherapy can lead to significant side effects and chemoresistance that can be linked to several resistance-activating biological processes, including an increase in inflammation, cellular plasticity, multidrug resistance (MDR), inhibition of the sentinel gene p53, and apoptosis. As a consequence, tumor cells can escape the effectiveness of chemotherapeutic agents. This underscores the need for cross-target therapeutic approaches that are not only pharmacologically safe but also modulate multiple potent signaling pathways and sensitize cancer cells to overcome resistance to standard drugs. In recent years, scientists have been searching for natural compounds that can be used as chemosensitizers in addition to conventional medications for the synergistic treatment of CRC. Resveratrol, a natural polyphenolic phytoalexin found in various fruits and vegetables such as peanuts, berries, and red grapes, is one of the most effective natural chemopreventive agents. Abundant in vitro and in vivo studies have shown that resveratrol, in interaction with standard drugs, is an effective chemosensitizer for CRC cells to chemotherapeutic agents and thus prevents drug resistance by modulating multiple pathways, including transcription factors, epithelial-to-mesenchymal transition-plasticity, proliferation, metastasis, angiogenesis, cell cycle, and apoptosis. The ability of resveratrol to modify multiple subcellular pathways that may suppress cancer cell plasticity and reversal of chemoresistance are critical parameters for understanding its anti-cancer effects. In this review, we focus on the chemosensitizing properties of resveratrol in CRC and, thus, its potential importance as an additive to ongoing treatments.
Graphical abstract
Keywords: Colorectal cancer, Chemoresistance, Chemosensitization, Multidrug resistance, Resveratrol, Cancer cell plasticity
Introduction
Across populations, physicians are faced with treating colorectal cancer (CRC), ranked third worldwide for cancer incidence and cancer-associated deaths [1], according to a summation of case numbers from 185 countries. Worldwide, more than 1.18 million people were affected by CRC in 2020 [1], and in 2022, there were over 151,000 new diagnoses and approximately 53,000 CRC-related deaths in the USA alone [2]. These collected data include neoplasms of both the colon and rectum. In the search for causes, the age-associated adenoma-carcinoma theory [3], in which malignant degenerations arise from benign precursors during life, is very relevant. Moreover, as a multifactorial process, CRC is often only detected at an advanced stage of the disease, as there are no clear and conspicuous signs. If the cells have already metastasized, the treatment process is highly time-consuming and rarely leads to the expected therapeutic success [2]. Therefore, early detection of a possible CRC in its benign or early-stage CRC should be encouraged, paving the way for appropriate and effective intervention.
At the molecular level, CRC development and spread are initiated and accelerated primarily by pro-inflammatory processes [4]. After developing a primary tumor, cancer cells can also detach from it and spread via lymphogenic or hematogenic routes to other organs. In the case of CRC, this leads primarily to portal vein-type metastases, 30–60% of which often [5] manifest themselves first in the liver. Unimpeded metastatic growth leads to organ failure and is one of the frequent causes of death in cancer patients. To prevent or treat metastasis, CRC patients receive chemotherapy in most cases after colorectal surgery. Due to the aggressiveness of the disease, this usually consists of several components, such as folinic acid and 5-fluorouracil (5-FU) combined with oxaliplatin (FOLFOX) or irinotecan (FOLFIRI). Tumor cells are becoming increasingly chemoresistant to common chemotherapeutic agents used against various cancers, with a high frequency of recurrence due to the modification of multiple metabolic pathways [6]. In this regard, increasing B-cell lymphoma 2 (Bcl-2) expression and thus inhibition of apoptosis, enhancing expression of hypoxia-inducible factor (HIF)-1 and thus tumor cell survival, raising the expression of multidrug resistance (MDR) protein 1, epithelial-mesenchymal-plasticity, sensitizes the cell to trans-differentiation, and hence, the promotion of drug efflux and initiation of medication inhibition are of central importance. Increasing the expression of the pro-inflammatory transcription factor ‘nuclear factor kappa-light-chain-enhancer of activated B-cells’ (NF-κB), which regulates the expression of inflammatory and cytoprotective genes, inhibiting the cellular tumor antigen p53 (p53) and thus inducing cell survival, which is a significant obstacle to the treatment of cancer patients [7–10] are further key examples. Moreover, undesirable side effects, such as the development of acquired resistance of CRC cells to chemotherapeutic agents, lead to a significant decrease in the efficacy of cytostatic drugs; thus, cancer continues to epithelial-mesenchymal-plasticity and spread despite treatment [6, 11]. Indeed, classical drugs with limited efficacy have not been able to solve this dramatic and widespread problem, so scientists are constantly searching for agents without side effects and for innovative solutions to improve cancer treatment.
In addition, the ever-increasing importance of people’s lifestyle habits, especially among modern populations, is now recognized as the most critical cause of CRC, particularly their diet. In this context, regular consumption of fermented foods, cigarette smoking, processed meats, alcohol, increased body mass index, and lack of exercise are considered unhealthy and have a favorable effect on the development of CRC [12]. Indeed, it was shown that a biologically balanced diet, exceptionally high in plant foods and fruits, can significantly reduce the risk of cancer [13, 14]. Therefore, interest is increasingly directed toward natural substances, which have been used for many decades in medical therapy to prevent various diseases, including cancer [15–17].
Whether secondary plant compounds can be used in the management of human diseases has been investigated for many years. These natural substances possess a poly-target action capability and thus have more versatile attack options in parallel than synthetically developed one’s mono-target drugs [18–20]. Against this backdrop, resveratrol is a well-researched plant-derived polyphenol, preventing the onset and advancement of CRC. This phytopharmaceutical occurs naturally in berries, grapes, and nuts [21–23], protecting the fruit from fungal infestation, oxidative processes, aging, and spoilage. It has already shown numerous relevant medical effects in mammalian and human cells. For example, in the field of cardiovascular diseases, a vasodilatory effect on a blood vessel by resveratrol-induced nitric oxide (NO)-mediated mechanisms is known [24, 25] and has the effect as a phytoestrogen [26–30]. In addition, resveratrol has protective and regeneration-promoting effects on nerves after injuries [31] and suppresses inflammatory cytokine storms related to chronic obstructive pulmonary disease [32]. Due to its high overall prevalence, resveratrol is a topic of current cancer research. Its effects on cancer cells and patients are being studied in vitro, in vivo, and clinically [33–35]. Specifically, this phytopharmaceutical has shown significant immunomodulatory potential [36–39] and immune system-balancing effects concerning tumor necrosis factors (TNFs) and interleukins (ILs) in both healthy and lymphoma patients [40]. Moreover, Resveratrol is a poly-target agent capable of modifying several cell signaling cascades, selectively exerting cytotoxicity on cancer cells and able to attenuate cell metastasis by suppressing the epithelial-to-mesenchymal transition (EMT) plasticity signaling [41] and, simultaneously, no toxicity on normal cells [42].
In this review, we address the anti-CRC and chemosensitization mechanisms of resveratrol, focusing on tumor cell plasticity (phenotypic trans-differentiation of cancer cells), which plays an essential role in transformation, progression, malignancy, metastasis, and also therapy resistance of cancer cells to conventional drugs. Consequently, the prevention of this dynamic developmental process is a key prerequisite for the prevention and improvement of clinical treatment success in cancer patients.
Goal of the review
This review aims to provide an overview of the high prevalence of CRC, focusing on the importance and fundamental role of cellular plasticity in frequent metastasis and the consequences of the development of resistance in CRC cells to classical chemotherapeutic agents. In this context, the anti-CRC potential of resveratrol, based on the suppression of EMT-plasticity, is reviewed, providing a pathway for overcoming chemoresistance that may represent a groundbreaking new advance in the treatment of CRC.
Resveratrol, a plant-derived polyphenol
Resveratrol is a plant stilbene with two phenolic rings linked by a double styrene chain, and it exists in two isoforms, cis and trans (Fig. 1). The trans-isoform is the most abundant and best studied and is catalyzed by the enzyme stilbene synthase [43]. Resveratrol was initially detected in the roots of white hellebore (Veratrum grandiflorum) and is now found in over 70 widely distributed plant species, including red wine grapes, cranberries, peanuts, and root extracts of the weed Polygonum cuspidatum [44, 45].
Fig. 1.
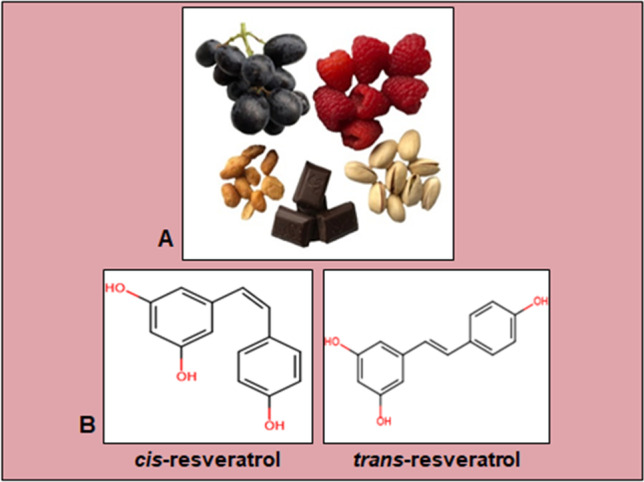
Description of resveratrol. A Exemplary photo of resveratrol-containing food products. B Chemical structure of cis-resveratrol and trans-resveratrol, composed with a creator from Fisher Scientific (Schwerte, Germany)
Resveratrol‘s natural sources and chemical properties
The phytoalexin resveratrol was extracted for the first time from the herbaceous plant Veratrum grandiflorum by the Japanese scientist Takaoka in 1939 [46]. It is a solid, alcohol-soluble ingredient alternatively known as 3,5,4′-trihydroxystilbene [47], with the sum formula being C14H12O3 [48]. Structurally, both cis- and trans-isomer exist, with the trans-form (Fig. 1) being more abundant and convertible to the cis-form [49, 50]. Numerous different targets of resveratrol have been identified, including integrin-receptors, estrogen-receptors, and sirtuins (SIRT) [51–53].
The best-known source of naturally occurring resveratrol is grapes of any color (Fig. 1), and the ingredient has been detected in bilberries, cranberries, and strawberries [54]. Consequently, this secondary phytochemical is also found in juices produced from these fruits and grape-based alcoholic beverages such as wine and champagne [54]. Furthermore, nuts such as peanuts and pistachios contain resveratrol, as does dark chocolate (Fig. 1) [55], which the presence of this plant component in cocoa beans could explain. In addition, some plants like Polygonum cuspidatum are particularly resveratrol-containing, leading to the occurrence of the Itadori tea brewed from it in Asia [56].
The legitimate inferential question is: Why has resveratrol been detected in many natural products? The answer is astonishing: The production of resveratrol is stimulated in plants whenever they are exposed to the stress of different genesis, for example, UV-radiation [22], ozone-exposition, or pathogenic confrontation [57]. Overall, this plant-native polyphenol balances the consequences of harmful environmental influences, protects the vegetables from fungal attack, parasite infestation, over-ripening, and rot [56], and thus represents a natural survival strategy. As humans are also permanently exposed to environmental influences of various kinds and often live a lifestyle that is detrimental to their health, research is constantly being conducted to determine whether and how to use resveratrol’s protective properties for the benefit of humankind.
Bioavailability, absorption, and metabolism of resveratrol
According to previous publications, the absorption of resveratrol by the oral route in man is around 75-80% and presumed to be predominantly by transepithelial diffusion in vitro and in vivo [58, 59]. It has already been reported that resveratrol is distributed in high concentrations in tissues. Indeed, resveratrol accumulated nearly 40-fold in the human CRC cell line Caco-2 in vitro versus medium [60], concluding that intestinal cells are a primary target for this polyphenol. Because of the strong metabolism in the digestive tract and hepatobiliary system, with a short half-life of about 1.5 hours [60], bioavailability by oral ingestion is relatively limited, less than 1%, which does not change with increasing intakes. Biochemical studies have shown that resveratrol’s major degradation products in blood plasma and urine are glucuronides, di-hydro-resveratrol conjugates, as well as sulfates [61, 62] and about 50–60% of the ingested phytopharmaceutical is eliminated from the body in urine [58]. Encouragingly, the ingestion of 500mg resveratrol in tablet form, resulting in plasma concentrations of about 70ng/ml, was reported to be well-tolerated and safe [63]. However, some adverse effects, such as diarrhea, nausea, anemia, vomiting, and flatulence, were reported when resveratrol was administered in high doses [64, 65].
Optimizing this topic, a higher bioavailability of this compound has been achieved by creating more sophisticated preparations, including nanoparticles and nano-constructed lipid vehicles incorporating resveratrol, including verification of the efficacy of the association in recovering chemosensitivity. In addition, the oral bioavailability of resveratrol incorporated in casein nanoparticles is tenfold increased compared to the administration of the polyphenol in the form of an oral suspension [66, 67]. Furthermore, enhancement of resveratrol’s bioavailability in rats has already been targeted by treatment with 3,5,4’-tri-O-acetyl-resveratrol, an acetylated resveratrol precursor that can be hydrolyzed in cells to free trans-resveratrol [68, 69]. Research in this area is still in the preliminary stages, but it is anticipated and promising that these novel and innovative nano-transporters will yield significant benefits.
Moreover, in addition to organs such as the intestine and the hepatic system, the vital colon microflora is suspected of playing a central role in resveratrol metabolism [61]. Considering the significant and indispensable importance of the intestinal microbiota for many essential body functions, and here for the metabolism of resveratrol, it could be of fundamental and crucial relevance, as it has already been demonstrated for other natural products such as soy isoflavone and lignans [70, 71]. Altogether, the high accumulation and concentration of resveratrol in the enterocytes of the colon epithelium suggest that this area of the colon [72], because of the intestinal microflora, probably plays an essential role in the bioactivity of resveratrol metabolism.
Resveratrol’s versatile anti-carcinogenic effects
Resveratrol’s effect on different cancer types
Research into resveratrol’s effects and detailed mechanisms of action on different cancer cell types and lines has been of high scientific interest for decades. To illustrate this with some examples, a treatment with this natural polyphenol disrupts the cell cycle flow of cervical cancer cells [73], reduces the inflammation-related invasiveness of gastric cancer cells [41, 74], and initiates apoptosis in prostate cancer cells [75–77]. Moreover, as a multitargeting agent in CRC cells, this active plant compound prevents invasion, proliferation, and metastasis [52]. It averts EMT-associated plasticity, necessary for metastasis, with a parallel promotion of apoptosis and being a chemosensitizer for treating cancers [78–81]. Resveratrol modulates epigenetic changes in tumor cells, as it can stimulate the ATP2A3 gene, leading to down-regulation of histone deacetylase (HDAC) and thus HDAC2 expression in the nucleus, or through deoxyribonucleic acid (DNA) methylation, histone modification, non-coding messenger ribonucleic acid (mRNA), and telomerase levels, leading to suppression of cancer spread [81, 82]. In line with this, a clinical study showed that a daily oral intake of 0.5–1g resveratrol could have anti-carcinogenic effects in the human gastrointestinal tract and, at the same time, be well-tolerated by cancer patients [35].
Resveratrol has also been shown to inhibit cellular processes associated with the development of tumors through mutation. Indeed, it has been reported that resveratrol possesses several active anti-oxidant capacities [83], acts as an active scavenger of hydroxylenes/superoxides [84] and significantly blocks the formation of free radicals caused, for example, by 12-O-tetradecanoylphorbol-13-acetate (TPA) in cancer cells [85]. Furthermore, resveratrol also contributes to protection against lipid peroxidation in cell membranes and DNA damage caused by the release of reactive oxygen species (ROS) [86, 87]. In addition, the phytopharmaceutical has been found to have mutation-inhibiting and anti-carcinogenic effects, such as preventing the mutagenicity of N-methyl-N’-nitro-N-nitrosoguanidine in Salmonella typhimurium [88].
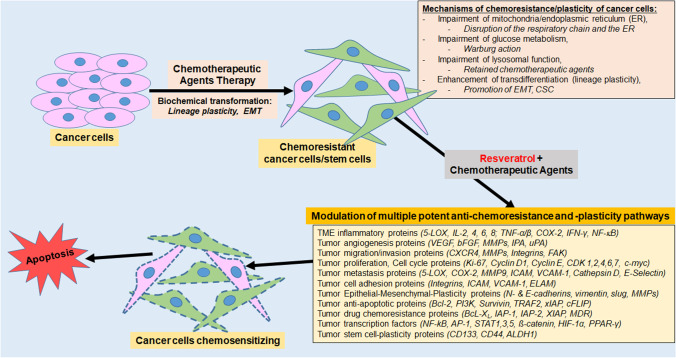
Cancer cells can become chemoresistant to various chemotherapeutic agents due to modifications in diverse biological processes in the subcellular signaling pathways [7, 8]. Resveratrol has great promise for targeting several molecular and cell signaling pathways. It has already been explored in various preclinical and clinical approaches as a chemosensitizer drug for combined therapy with standard drugs for diverse types of cancer [89, 90]. Figure 2 provides an overview of the beneficial properties of the combination treatment of standard chemotherapeutic substances with resveratrol and its various potential biological pathways involved in the chemosensitization of tumor cells. Table 1 lists recent research findings on the different subcellular signaling pathways and their mechanisms of resveratrol having anti-inflammatory, anti-proliferative, anti-metastatic, anti-oxidative, immunomodulatory, and pro- or anti-apoptotic effects in various in vitro or in vivo studies in cancer and healthy cells.
Fig. 2.
Generation of resistance in cancer cells and its recovery by co-treatment with resveratrol. Cancer cells become resistant to several chemotherapeutic medications because of modifications in different regulatory pathways. Co-treatment with resveratrol and ongoing chemotherapeutic agents transforms these biological changes by simultaneously affecting multiple signaling pathways, resulting in the chemosensitization of tumor cells to chemotherapy agents. Abbreviations: 5-LOX, 5-lipoxygenase; ALDH, aldehyde dehydrogenase; AP-1, activator protein 1; Bcl-2, B-cell lymphoma 2; Bcl-xL, B-cell lymphoma extra-large; bFGF, basic fibroblast growth factor; CSC, cancer stem cell; CD, cluster of differentiation; CDK, cyclin-dependent kinase; cFLIP, cellular FLICE-inhibitory protein; cMyc, Cellular myelocytomatosis oncogene; COX, cyclooxygenase; CXCR, C-X-C chemokine receptor; ELAM, Endothelial Leukocyte Adhesion Molecule; EMT, epithelial-to-mesenchymal transition; ER, endoplasmic reticulum; FAK, focal adhesion kinase; HIF, hypoxia-inducible factor; IAP, inhibitor of apoptosis protein; ICAM, intercellular adhesion molecule; IFN, interferone; IL, interleukin; IPA, indolephenoxyacetamide; Ki-67, Kiel-antigen 67; MDR, multidrug resistance; MMP, matrix metalloproteinase; NF-κB, nuclear factor kappa-light-chain-enhancer of activated B-cells; PI-3K, phosphoinositide 3-kinase; PPAR-γ, Peroxisome proliferator-activated receptor gamma; Slug, SNAI2; snail homolog 2; STAT, signal transducer and activator of transcription; TME, tumor microenvironment; TNF, tumor necrosis factor; TRAF, TNF receptor-associated factor; uPA, urokinase-type plasminogen activator; VCAM, vascular cell adhesion molecule; VEGF, vascular endothelial growth factor
Table 1.
Various subcellular signal-modulating networks in cancer and healthy cells as resveratrol targets
| Subcellular signaling / Mechanism | References | |
|---|---|---|
| Down-regulation of | Transcription factor signaling pathway | |
| - NF-κB signaling pathway | [91–102] | |
| - HIF-1α signaling pathway | [93, 103–113] | |
| - MAPK signaling pathway | [114–119] | |
| - AP-1 signaling pathway | [120, 121] | |
| - STAT3 signaling pathway | [122–124] | |
| - β-Catenin signaling pathway | [125–127] | |
| - Cell cycle signaling pathway | [128, 129] | |
| - Growth factor signaling pathway | [92, 130, 131] | |
| - Mitochondrial signaling pathway | [132, 133] | |
| - Inflammation signaling pathway | [134, 135] | |
| - Oxidative signaling pathway | [136, 137] | |
| - Mutagenesis signaling pathway | [138, 139] | |
| - Angiogenesis signaling pathway | [140] | |
| - Plasticity/Migration signaling pathway | [141–145] | |
| - Estrogen signaling pathway | [146, 147] | |
| - RANKL signaling pathway | [148–152] | |
| - Apoptosis signaling pathway | [153–159] | |
| Up-regulation of | Transcription factors signaling pathway | |
| - Sox9 signaling pathway | [154, 155, 160–162] | |
| - Scleraxis signaling pathway | [163] | |
| - PPAR-γ/RUNX2 signaling pathway | [164–170] | |
| - PI3K/Akt/mTOR signaling pathway | [171–173] | |
| - p53 signaling pathway | [174, 175] | |
| - Autophagy signaling pathway | [176, 177] | |
| - Apoptosis signaling pathway | [178, 179] | |
| - Estrogen signaling pathway | [26–30] | |
| - Maintenance of the cellular signaling pathway | [180–184] | |
| - Immunomodulatory signaling pathway | [36–39] | |
Abbreviations: Akt protein kinase B, AP-1 activator protein 1, HIF hypoxia-inducible factor, mTOR mammalian target of Rapamycin, NF-κB nuclear factor kappa-light-chain-enhancer of activated B-cells, PI-3K phosphoinositide 3-kinase, PPAR-γ Peroxisome proliferator-activated receptor gamma, RANKL receptor activator of NF-kappaB ligand, RUNX2 Runt-related transcription factor 2, SOX9 SRY-Box Transcription Factor 9, STAT3 signal transducer and activator of transcription 3, MAPK mitogen-activated protein kinase
Resveratrol modulates inflammation and acts anti-carcinogenic in CRC cells
Acute inflammation is activated by immune-specific cells, especially in infections and allergies. Thus, it is part of the healthy immune system in the body, and it lasts only for a short time. However, if the inflammation lasts longer, it becomes chronic [185]. Key mechanisms such as chronic inflammation and the associated induction of angiogenesis, metabolizing enzymes, oxidation, cell cycling, cell plasticity, and anti-apoptotic proteins are among the most important prerequisites for the development of chronic diseases, including colitis and CRC, making agents to prevent and inhibit inflammation in the tissues, like colon and thus prevent colitis and CRC of particular interest. Well-documented inflammatory signaling pathways associated with the pathogenesis of colitis-related CRC include NF-κB, IL-6/STAT3, cyclooxygenase (COX)-2/prostaglandin E2 (PGE2), and IL-23/T helper 17 cells (Th17) [186]. More specifically, pro-inflammatory chemokines and cytokines, such as IL-1, -6, -8, TNF-α, and TNF-β, are produced very rapidly by injured body tissues and can trigger a variety of inflammatory responses and the expression of pro-inflammatory transcription factors, such as NF-κB, mitogen-activated protein kinase (MAPK), signal transducer and activator of transcription (STAT) 3, HIF-1α, activator protein-1 (AP-1), and nuclear factor erythroid 2–related factor 2 (Nrf2), and their secondary inflammatory substances such as inflammatory mediators, such as matrix metalloproteinases (MMPs), 5-lipoxygenase (5-LOX), COX-2, and also the production of ROS [187–191]. Inflammatory bowel disease (IBD) and Lynche’s syndrome have been shown to contribute to significantly increased development and pathogenesis of CRC, suggesting an intense interaction between inflammation and cancer development. In addition, experimental animal models of IBD have clearly demonstrated that resveratrol is a beneficial agent for the management of IBD [192].
Furthermore, one of the most critical pro-inflammatory transcription factors in inflammatory tissues is NF-κB, expressed by multiple cancers such as CRC [193]. This transcription factor induces genes involved in cell survival, cell adhesion, inflammation, differentiation, and growth. NF-κB is activated by various influences such as carcinogens, phorbol ester, pro-inflammatory agents, cigarette smoke, and cytokines such as IL-1β and TNF-α or TNF-β [194]. These factors promote the dissociation of NF-κB inhibitor alpha (IκBα) through phosphorylation, and the activated NF-κB migrates from the cytoplasm to the nucleus, leading to the binding and activation of transcription of target genes essential for the development of aggressive cancers. The expression of proliferation proteins (cyclin D1, Kiel-antigen 67 (Ki-67)), apoptosis suppressor proteins (Bcl-2 and B-cell lymphoma extra-large (Bcl-xL)), and proteins responsible for metastasis (MMPs, COX-2, CXCR4) as well as angiogenesis (vascular endothelial growth factor (VEGF)) are promoted by NF-κB [194]. Similarly, IL-6/JAK/STAT3 signaling pathway potently activates inflammatory response via tumor-infiltrating immune cells in the tumor immune microenvironment in CRC. Moreover, IL-6/JAK/STAT3 signaling up-regulates downstream target genes with anti-apoptotic and proliferative effects, promotes plasticity, invasion, and metastasis of cancer cells and angiogenesis, and induces cancer resistance [195, 196]. The COX-2/PGE2 signaling has been closely associated with all stages of colorectal carcinogenesis. The role of COX-2 and its product PGE2 in the pathogenesis of CRC is based on the function of fibroblasts from the mesenchymal (stromal) layer, which are the main target of cytokines e.g. TNF-α and IL-1β. Fibroblasts from non-neoplastic colorectal tissue are an important source of COX-2 expression that is well-validated as one of the most important risk factors of CRC [197]. Finally, IL-23 receptors play a crucial role in chronic inflammatory diseases due to their function in the processes of differentiation of Th17. IL-23 up-regulates PGE2 levels and Th17 cell function that include expression increase of inflammatory cytokines, such as IL-17A, IL-17F, IL-21, and IL-22. For these reasons, the IL-23/Th17 signaling is strongly included in the pathogenesis of colitis-associated CRC [198].
Chemopreventive phytochemicals such as resveratrol inhibit several pro-inflammatory-related activations of transcription factors, cytokines, chemokines, proteins, and enzymes [199–205]. Much evidence suggests that resveratrol is a multi-factorial bioactive phytochemical with numerous beneficial preventive effects on subcellular biological pathways, especially anti-inflammatory effects by inhibiting pro-inflammatory cytokines (IL-1β, TNF-α, and TNF-β), the pro-inflammatory transcription factor NF-κB and thus NF-κB-promoted end-proteins. A recent molecular docking study documented that resveratrol could be effective against CRC by targeting NF-κB signaling [206], and in this regard, our group described resveratrol’s NF-κB suppression by resveratrol associated with an anti-inflammatory mode of action in CRC [207]. Furthermore, a seven-day treatment with resveratrol (10mg/kg body weight) suppressed the dextran sulfate sodium-induced inflammatory colon injury via down-regulation of NF-κB, STAT3, ERK, and iNOS expressions in IRC mice [208]. Combinational application of resveratrol with 5-FU inhibited Akt/STAT3 signaling, which was associated with pro-apoptotic effects and increased anti-telomerase activity in human CRC cells [209]. Using DLD1 and HCT15 CRC cells, resveratrol inhibited cancer growth by targeting the Akt/STAT3 signaling pathway. These anti-cancer effects of resveratrol correlated with pro-apoptotic effects and blockage of the G1 phase cell cycle in cancer cells [123]. In addition, the resveratrol treatment sensitized HT-29 and SW620 CRC cell lines to 5-FU (via increased oxidative stress) through the down-regulation of Akt and STAT3 signal proteins [210]. Another study showed the inhibitory effect of resveratrol on the COX-2/PGE2 signaling pathway (decreased both miRNA and protein levels) and consequent anti-cancer efficacy in HCT-116 human CRC cell lines [211]. A similar result was found in a Serra et al. study describing the inhibition of COX-2/PGE2 signaling by resveratrol (also isoenzymes of NO synthase (iNOS) expression) in HT-29 colon epithelial cells [212]. The combination of resveratrol and ginkgetin synergistically attenuated the 5-FU-induced inflammation in HT-29 colon cancer xenograft nude mice through decreased expressions of COX-2 and inflammatory cytokines [213]. A micro-immunotherapy sequential medicine including resveratrol showed significant immunomodulatory effects on human macrophages via several cytokine-induced signaling pathways (including IL-23) and consequent tumor-suppressive efficacy using in vitro 2D and 3D spheroid models and animal xenograft colon carcinoma experimental approach [214]. Furthermore, the phytopharmaceutical possesses potential anti-cancer functions, such as CRC cell survival reduction, activation of apoptosis (caspase-3), inhibition of invasion, and preventing of EMT-plasticity in the CRC tumor microenvironment (TME) in vitro and in vivo (Table 2) [215–234].
Table 2.
The modulatory impact of resveratrol on inflammation and progression in CRC cells in vitro and in vivo
| Signal modulating networks | Kind of test, CRC cell- /animal Type | Mode of action | References |
|---|---|---|---|
| Suppression of pro-inflammatory cytokines in CRC |
In vitro, HCT-116, HCT-116R cells |
Modulation of TNF-β signaling pathway, suppression of NF-κB activation | [80] |
|
In vitro and in vivo, LoVo cells and Mice |
Inhibition of inflammation, EMT through TGF-β1/Smads signaling, suppression of metastasis and plasticity in CRC cells | [196] | |
| In vitro, HCT-116/ RKO/SW480 cells | Inhibition of TNF-β/TNF-β-receptor-induced activation of NF-κB, NF-κB-promoted gene products | [199] | |
|
In vivo, Rat |
Inhibition of neutrophil infiltration, cytokines and oxidative stress, reduction of colitis | [215] | |
|
In vivo, Mice |
Modified gut microcosm leads to anti-inflammatory response and alleviation of inflammation-promoted CRC | [216] | |
| Suppression of inflammation, proliferation and tumor formation / induction of apoptosis in CRC |
In vitro, HT-29/WiDr cells |
Inhibition of inflammation and proliferation in CRC cells | [217] |
|
In vitro, HCT-116 cells |
Inhibition of Wnt/survivin signaling, inflammation, activation of p53-independent apoptosis |
[218] | |
|
In vitro, HCT-116/RKO/SW480 cells |
Inhibition of TNF-β/TNF-βR-induced EMT via suppression of NF-κΒ, FAK and plasticity | [196, 207] | |
|
In vitro, HCA17/SW480/HT-29 cells |
Inhibition of inflammation, proliferation and apoptosis in CRC cells | [219] | |
|
In vitro, HCT-116/Caco-2 cells |
Inhibition of proliferation by inducing G1/S-phase cell cycle arrest, apoptosis by caspase/cyclin-CDK pathways | [220] | |
|
In vitro, HCT-116/CO115 cells |
Activation of p53-mediated apoptosis, inhibition of inflammation | [221] | |
|
In vitro, DLD1/HCT-15 cells |
Inhibition of inflammation and proliferation by targeting the Akt/STAT3 signaling pathway | [123] | |
|
In vivo, Rat |
Inhibition of DMH-induced irregular crypt lesion foci, CRC formation, increased expression of anti-oxidant enzymes | [222] | |
|
In vivo, Mice |
Inhibition of CRC formation and inflammation | [89] | |
|
In vivo, Rat |
Inhibition of CRC occurrence through regulation of inflammatory enzymes, growth of aberrant crypt lesions | [223] | |
|
In vivo, Rat |
Inhibition of precancerous lesions in the colon and inflammation | [224] | |
|
In vivo, Mice |
Inhibition of intestinal tumorigenesis by reducing genes involved in cell proliferation and inflammation | [225] | |
|
In vitro and in vivo, HCT-116 cells and Mice |
Resveratrol with curcumin inhibited CRC cell growth more effectively than either agent alone, associated with reduction of inflammation, proliferation, stimulation of apoptosis, together with a decrease in NF-κB activity | [226] | |
|
In vivo, Mice |
Inhibition of PGE2, COX-2 expression and the number of adenomas | [227] | |
|
In vivo, Rat |
Inhibition of CRC, inflammation and aberrant crypt foci by targeting Bax and p21 expression | [228] | |
|
In vivo, IRC mice |
Inhibition of iNOS, NF-κB, STAT3, and ERK expressions | [208] | |
|
In vitro, HCT-116 and DLD1 cells |
Inhibition of Akt/STAT3 and NFκB | [209] | |
|
In vitro, DLD1 and HCT-15 cells |
Inhibition of Akt/STAT3, cyclin D1, cyclin E2, and Bcl-2, increase of p53 | [123] | |
|
In vitro, HT-29 and SW-620 cells |
Inhibition of Akt and STAT3, induction of ROS | [210] | |
|
In vitro, HCT-116 cells |
Inhibition of COX-2 in both miRNA and protein levels | [211] | |
|
In vitro, HT-29 cells |
Inhibition of NO and PGE2 production, iNOS and COX-2 expression and ROS, suppression of JAK/STAT and SAPK/JNK pathways | [212] | |
|
In vivo, HT-29 colon cancer xenograft using nude mice |
Resveratrol+ginkgetin enhanced the anti-tumor effect of 5-FU via decrease in microvessel density of tumors, suppressing expressions of COX-2, TNF-α and IL-6 | [213] | |
|
In vitro and in vivo, 2D and 3D model of HCT-116 cells murine subcutaneous xenograft HCT-116 model |
Micro-immunotherapy medicine + resveratrol induced immunomodulation of macrophages, decrease in the volume of 3D spheroids, decrease in the volume of xenografts |
[214] |
Abbreviations: 5-FU 5-fluorouuracil, Akt protein kinase B, Bax bcl-2-like protein 4, Bcl-2 B-cell lymphoma 2, CDK cyclin-dependent kinase, COX-2 cyclooxygenase 2, CRC colorectal cancer, DMH 1,2-dimethylhydrazine, EMT epithelial-to-mesenchymal transition, ERK extracellular signal-regulated kinases, FAK focal adhesion kinase, iNOS inducible nitric oxide synthase, JAK janus kinase, NF-κB nuclear factor kappa-light-chain-enhancer of activated B-cells, PGE2 prostaglandin E2, IL interleukin, MMP matrix metalloproteinase, NO nitric oxide, ROS reactive oxygen species, SAPK stress-activated protein kinase, STAT3 signal transducer and activator of transcription 3, TGF transforming growth factor, TME tumor microenvironment, TNF tumor necrosis factor
An intense functional collaboration between tumor cells and immune cells is known to exist in the TME, which is essential for the further growth and progressive spread of tumors. Therefore, it is also important that the interaction between both systems is altered in favor of anti-tumor immunity [235]. T-lymphocytes (CD4+), which are among the most important immune cells of the entire defense system, have the ability to differentiate into other T-lymphocytes such as CD8+ and regulatory T-cells (Tregs) in TME in a variable and cytokine-dependent manner, making this interaction a critical factor in slowing tumor growth and providing a good prognosis for patients [236, 237]. Interestingly, resveratrol has been shown to modulate T-cells (CD4+) by excreting interferon (IFN)-γ and thereby up-regulating its biotarget Sirt-1 in CD4+ T-cells [238]. Moreover, these tumor-specific cytotoxic cells (CD8+ T-cells) can attack cancer cells through apoptosis-specific factors (IFN-γ, IL-4, and TNF-α) and mechanisms (perforation of the membrane) and initiate active suicide of tumor cells [239], promoted by resveratrol by participating in anti-tumor immunity, as reported by Choi and colleagues [240].
As previously reported, resveratrol suppresses HDACs, which correlates with the formation of various anti-inflammatory lymphocytes and Treg cells in the gut. In addition, a study in The Cancer Genome Atlas (TCGA) reported that an increase in the activity of the Treg-specific transcription factor FoxP3 or the anti-inflammatory IL-10 contributes to improved survival in patients with CRC. This suggests that changes in the gut microbiome may lead to an anti-inflammatory T-cell response that attenuates inflammation-related CRC [12, 216].
In addition, several investigations showed a significantly enhanced expression and stimulation of NO by iNOS in CRC cells, indicating an essential role of NO in tumorigenesis in the colon cells [241]. Interestingly, resveratrol decreases specific iNOS expression in CRC cells [242]. One of the most essential and primary mechanisms of action of resveratrol is its anti-inflammatory potential, the suppression of the p38-MAPK signaling pathway, which is involved in the production of inflammatory mediators such as cytokines, COX-2, p53, and iNOS [243] and thus is an important requirement for the prevention and inhibition of colitis and CRC (Table 2). For this reason, specific targeting and modulation of pro-inflammatory metabolic pathways, such as NF-κB have significant implications for preventing and eliminating serious diseases [193].
Resveratrol acts as a chemosensitizer in CRC cells
Difficulty of chemoresistance in CRC cells
Surgical intervention and chemotherapeutic drug administration remain the main treatment options for CRC patients. However, the decision to use one or more of these approaches in treating CRC subjects depends on the tumor’s location, the cancer stage at diagnosis, and the case-by-case characteristics of the patient [244]. The challenge often arises when a patient (a) shows inherent resistance to the drug due to intrinsic drug resistance capabilities of the cancer cell or (b) develops resistance to a chemotherapeutic drug through exposure to the drug, called acquired resistance [245]. Chemotherapeutic drug resistance leads to lower sensitivity to the drug, compromised drug inefficacy, cancer cell plasticity, tumor relapse, poor prognosis, and higher mortality rates among CRC patients [246–248].
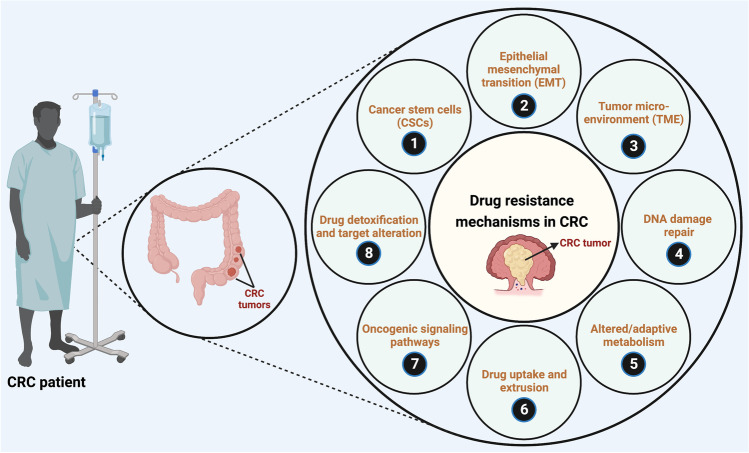
One or more non-cellular (such as limited vascular accessibility and TME) and cellular (such as drug targets, levels and activity of detoxifying enzymes, levels, and activity drug uptake and extrusion transporters) factors play vital roles in CRC drug resistance [244]. Detailed discussions on the various mechanisms of chemotherapeutic drug resistance are beyond the scope of this current review article. Several articles have reported the multiple aspects and mechanisms of chemoresistance in CRC (illustrated in Fig. 3) and novel strategies to reverse resistance extensively and thoroughly [249–251].
Fig. 3.
The different mechanisms of chemoresistance in CRC (Figure and Figure legend adapted from Samuel SM, et al., 2020) [245]. The mechanisms of cancer cell plasticity in therapeutic resistance mainly include; (1) the presence and influence of cancer stem cells (CSCs) that can initiate and re-populate tumors, (2) epithelial–to-mesenchymal transition (EMT), (3) tumor microenvironment (characterized by hypoxia, inflammation, autophagy, and presence of cancer-associated fibroblasts, immune cells such as tumor-associated macrophages, and tumor endothelial cells), (4) active DNA damage repair mechanisms, (5) altered/adaptive/aberrant metabolism (characterized by the Warburg effect, altered amino acid/protein/lipid and nucleotide metabolism, utilization of glutamine, and isoforms of metabolic enzymes that support cancer initiation, progression, and resistance to therapy), (6) variations in drug uptake and active drug extrusion systems (ATP binding cassette; ABC/multidrug transporters), (7) activation of oncogenic, pro-survival and anti-apoptotic signaling pathways (PI3K, phosphatidylinositol-3-kinase; Akt, protein kinase B; mTOR, mammalian target of rapamycin; MAPK, mitogen activated protein kinase; NF-κB, nuclear factor kappa-light-chain-enhancer of activated B-cells; Wnt/β-catenin; JAK, janus kinase; STAT3, signal transducer and activator of transcription 3; HIF-1, hypoxia inducible factor 1 pathways), and (8) active drug detoxification and target alteration systems. Created with BioRender.com
The most widely used chemotherapeutic drugs for treating CRC include 5-FU, oxaliplatin, doxorubicin, cetuximab, irinotecan, and various combinations of the same. Drug resistance has been reported against one or more of these drugs and is summarized in Table 3 [252–276]. In some instances, however, CRC resistance to drugs such as oxaliplatin enhanced the sensitivity to tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) via the up-regulation of death receptor 4 and localization of lipid rafts [277]. Interestingly, a significant reduction in viable circulating tumor cells (CTCs) was observed in metastatic CRC patients treated with TRAIL liposomes [277].
Table 3.
Selected drug resistance in CRC: molecular basis and mechanisms
| Drug | Pathway or related enzyme/protein | Type of study | Effects and possible mechanisms of chemoresistance | References |
|---|---|---|---|---|
| 5-Fluorouracil (5-FU) | Thymidylate synthase (TS) | Meta-analysis, 20 studies, 3497 CRC patients | ↑TS expression leading to; ↑5-FU target inhibition and ↓5-FU sensitivity | [249, 252] |
| In vitro, SNU-C1 parental and SNU-C1 (5-FU resistant) | ↑TS mRNA expression; ↑TS activity | [253] | ||
| Clinical, formalin-fixed paraffin-embedded specimens, 132 CRC patients (Dukes’ B=36 cases, Dukes’ C=60 cases, Dukes’ B=36 cases) | ↑TS expression correlated to 5-FU resistance, shorter recurrence free-interval, and reduced overall survival | [254] | ||
| Clinical, tissue samples (normal, primary, and liver metastases), digital karyotyping | ↑Gene amplification of TS major mechanism of 5-FU resistance | [255] | ||
| Clinical, PCR analysis of genomic DNA, 121 CRC patients | ↑TS expression associated with poor response to 5-FU | [256] | ||
| Inhibitor of DNA binding 1, HLH protein (ID1) | In vitro, CRC-stem-like cells; In vivo, mice xenograft model | ↑ID1 (stemness marker) expression | [257] | |
| p53 related apoptosis | In vitro, HCT-116 | ↓p53 correlated to; ↑5-FU resistance and ↓apoptosis | [258] | |
| RhoGDI2 and apoptosis | In vitro, LoVo | ↑RhoGDI2; ↑CapG; ↓Maspin; ↓Apoptosis | [259] | |
| SHMT2 and autophagy | Clinical, paired-frozen-primary samples (CRC and adjacent normal tissue), 50 patients; clinical, paraffin-embedded samples, 378 patients; In vivo, mouse xenograft model | ↓SHMT2; ↓SHMT2 binding of cytosolic p53; ↓Apoptosis; ↑Pro-survival autophagy, ↑Plasticity | [196, 260] | |
| p38-MAPK, apoptosis, and autophagy | In vitro, RKO, HT-29, LoVo, SW620 and HCT-116 | ↓p38-MAPKα; ↓Apoptosis; ↑Autophagy | [261] | |
| RAC3, apoptosis, and autophagy | In vitro, HT-29, LoVo and HCT-116 | ↑RAC3; ↓Apoptosis; ↓Autophagy | [262] | |
| TGF-β and EMT | In vitro, HCT-116, HCT-116p53KO and HT-29; In vivo, mice xenograft model | ↑TGF-β; ↑Proliferation; ↓Cell death; ↑Plasticity | [263] | |
| OPRT-RR and TP-TK pathways | In vitro, SW48 and LS174T (parental and resistant) |
In SW48-5-FUR cells - ↓OPRT; ↓TP; ↓FdUMP; ↓Drug sensitivity; ↑IC50 In LS174T-5-FUR cells - ↓OPRT; ↓RR; ↑TK; ↑dTMP; ↓Drug sensitivity; ↑IC50 |
[264] | |
| Hedgehog pathway | In vitro, LoVo (parental and resistant) | ↑GLI1; ↑IC50 for 5-FU | [265] | |
| Irinotecan (SN-38; irinotecan metabolite) | ABCC1/MRP1, ABCC2/MRP2, ABCG2/BCRP, multi-drug resistance protein (MDR/MRP) | In vitro, HCT-116 | ↑Expression of MDR/MRP proteins; ↑Drug efflux; ↑Plasticity | [196, 266] |
| ABCG2/BCRP, multi-drug resistance protein (MDR/MRP) | In vitro, HCT-116, S1-IR20 (novel irinotecan resistant CRC cell line) | ↑ABCG2/BCRP; ↑Drug efflux | [267, 268] | |
| ATP7A, copper transporter | Clinical, 50 patients with advanced CRC | ↑ATP7A; ↑Drug efflux; ↑Drug uptake in membrane vesicles | [269] | |
| Oxaliplatin | OCT, drug intake | In vitro, LS180, SW620, DLD, HT20, RKO and HCT-116 | ↓OCT1 (SLC22A1); ↓OCT2 (SLC22A2); ↓Oxaliplatin sensitivity | [270] |
| CHK2, DNA repair | In vitro, HT-29, LoVo, Colo201 and Colo205; In vivo, mice xenograft model | ↑phosphorylated CHK2 (pCHK2T68); ↑Homologous recombination repair pathways; ↑DNA repair; ↑CHK2/PARP1 interaction | [271] | |
| ABGC2, a multi-drug resistance protein (MDR/MRP) | In vitro, LoVo (parental and resistant) | ↑ABGC2; ↓G2 cell-cycle arrest; ↑phosphorylated NF-κB; ↓ER stress; ↓Apoptosis | [272] | |
| Nrf2, oxidative stress | In vitro, HCT-116 and SW620 | ↑Nrf2; ↑Nrf2 regulated gene expression; ↓Proliferation; ↑IC50 for various drugs | [273] | |
| Cetuximab | KRAS WT tumors | Clinical, 220 chemorefractory metastatic CRC (cmCRC) patients | ↑EREG; ↑AREG; ↓Survival | [274] |
| EGFR somatic sequence alterations (G465R, G465E, S468R, S492R) | In vitro, LIM1215 and OXCO-2 | ↓mAb binding; ↓EGFR pathway inhibition | [275] | |
| EGFR mutation (pS492R) | Clinical; plasma samples from 1010 patients with metastatic CRC | ↓mAb binding; ↓EGFR pathway inhibition | [276] |
The upward arrow (↑) indicates an activation/increase/up-regulation and the downward arrow (↓) indicates a decrease/down-regulation/suppression. Abbreviations: 5-FU 5-fluorouracil, 5-FUR 5-FU resistant, ABCC1/2 ATP binding cassette subfamily C member 1 or 2, also known as MRP1/2, ABGC2 ATP binding cassette subfamily G member 2, also known as BCRP, AREG Amphiregulin, ATP7A ATPase copper transporting 7 alpha, BCRP Breast cancer resistance protein, also known as ABGC2, CapG Capping actin protein, gelsolin like, CHK2 Checkpoint kinase 2, EGFR epidermal growth factor receptor, EMT epithelial-tomesenchymal-transition, EREG Epiregulin, FdUMP Fluoro-deoxyuridine monophosphate, GLI1 Glioma-associate oncogene family zinc finger 1, ID1 Inhibitor of DNA binding 1, KRAS Kirsten rat sarcoma, mAb monoclonal antibody, MAPK mitogen activated protein kinase, MDR/MDRP multi drug resistance protein, MRP1/2 multidrug resistance associated protein 1 or 2, also known as ABCC1/2, NF-κB nuclear factor kappa-light-chain-enhancer of activated B-cells, Nrf2 Nuclear factor erythroid 2 related factor 2, OCT1 Organic cation transporter 1, also known as SLC22A1, OCT2, Organic cation transporter 2, also known as SLC22A2, OPRT, Orotate phosphoribosyl transferase, RAC3 Rac family small GTPase 3, RhoGDI2 Rho GDP dissociation inhibitor 2/β, RR Ribonucleotide reductase, SHMT2 Serine hydroxymethyl transferase 2, TGFβ Transforming growth factor beta, TK thymidine kinase, TP Thymidine phosphatase, TS Thymidylate synthase, WT Wild-type
Chemoresistance through tumor cell plasticity in CRC cells
Although chemotherapy is the mainstay of cancer treatment, the lack of efficacy of this treatment is a major concern. For many years, researchers believed that the failure of cancer treatments might be due to intrinsic genomic mechanisms, such as the development of mutations in the drug target that prevent its binding [144, 278]. Nevertheless, the primary cause of the ineffectiveness of chemotherapeutic agents is the acquisition of drug resistance by tumor cells throughout treatment [279]. Therefore, it is vital to understand the mechanisms of drug resistance to develop more efficacious treatments that can reduce the risk of relapse. An important factor that is implicated in drug resistance is the plasticity of tumor cells [143]. Cellular plasticity, concerning tumorigenesis, refers to the capacity of terminally differentiated cancer cells to undergo drastic changes in their cell phenotypes in response to oncogenic drivers or external stimuli [145]. The different types of plastic behaviors that help tumor cells acquire drug resistance are EMT, otherwise called epithelial-mesenchymal-plasticity, attaining properties of cancer stem cells (CSCs), and transdifferentiation into other cell types [143, 144]. A deeper insight into the molecular mechanisms underlying CRC drug resistance has revealed a convincing link between these tumor cell plasticity hallmarks and CRC progression [280, 281].
EMT is a developmental process during embryogenesis, tissue remodeling, and wound healing that allows epithelial cells to attain mesenchymal phenotype to enable its motility and invasiveness [282–284]. However, molecular pathways comparable to development have been observed in cancer cells leading to EMT-plasticity [284]. This transition occurs through transcriptional repression of E-cadherin (cell adhesion molecule) by Snail, Slug, zeb 1/2, smad interacting protein 1 (SIP1) or Twist1, and elevated expression of vimentin and N-cadherin, through a complex network of signaling cascade which facilitates collective cell migration and invasion. Cellular growth factors, including epidermal growth factor (EGF), hepatocyte growth factor (HGF), and transforming growth factor beta (TGF-β), are potent stimulators of EMT, which upon binding to their corresponding receptors, initiate signaling pathways including Notch, β-catenin/Wnt and PI3K/Erk signaling [284]. Further research has demonstrated the link between several EMT markers, apoptosis evasion, and enhanced cancer cell survival [285, 286]. This correlation has stimulated interest among researchers to investigate the connection of these EMT markers with resistance to anti-neoplastic treatment modalities. Apart from initiating tumor and inducing metastasis, EMT also confers resistance to cancer treatment interventions, including radiotherapy [285, 287, 288]. For instance, Liu and co-workers demonstrated that vincristine-resistant colon adenocarcinoma cells showed increased expression of Twist1 and thus exhibited elevated migratory and invasive ability. More importantly, this study showed that up-regulation of Twist1 markedly increased chemoresistance to vincristine by up-regulating the ATP-binding cassette transporters, ABCB1 and ABCC1 [288]. Nonetheless, the mechanism of how EMT-plasticity contributes to drug resistance is not yet fully understood. However, it is believed that the cells become more resistant to pro-apoptotic signals and excessive drug efflux by membrane transporters, leading to cell survival despite treatment [289].
An interesting study identified circulating tumor microemboli (CTMs) and three subpopulations of CTCs, namely, E-CTCs, M-CTCs, and E/M-CTCs based on the expression of epithelial cell adhesion molecule (EpCAM), the mesenchymal cell marker vimentin, or both EpCAM and vimentin respectively from blood samples of 126 CRC patients. However, the results showed that M-CTCs and CTMs were highly detected in patients with lymph node metastasis of CRC [290]. Another investigation explored the active involvement of reactive stroma in the modulation of EMT-plasticity in CRC and TNF-α produced by macrophages accelerated the process of TGF-β-induced EMT. This study also demonstrated an interplay between TNF-α and TGF-β signaling in the morphological conversion of organized colon epithelial cells to scattered mesenchymal cells. Besides, TNF-α stimulated Erk activation, which causes increased production of this cytokine by tumor cells. Hence, the role of stroma in the EMT-plasticity of CRC was further elucidated [291].
Moreover, Bates and colleagues revealed a significant role of the integrin αvβ6 (a receptor for fibronectin and tenascin) in CRC progression and metastasis. Their results showed that αvβ6 activated autocrine TGF-β, which causes EMT-plasticity. Clinical analysis of 488 CRC patient samples exhibited a reduction in survival of patients with increased expression of αvβ6, compared to the patients with low or no β6 expression and suggested that β6 expression could be a potential prognostic variable for CRC [292]. Another study reported that caspase-3 gene knockout caused reduced expression of EMT markers such as N-cadherin, Snail, Slug, and zeb1 and elevated E-cadherin expression and chemosensitivity compared to parental CRC cells. Furthermore, the critical role of caspase-3 in cancer cell invasion and metastasis was indicated [293]. Additionally, another examination demonstrated pre-mRNA processing factor (PRP4) as an essential factor in inducing EMT-plasticity and drug resistance in CRC cells by directly binding to p53 and causing its phosphorylation and up-regulating HIF-1α and miR-210, which activates p53 [294].
Focusing chronic oxaliplatin treatment of CRC cells, this therapy resulted in phenotypic changes associated with cellular plasticity, such as loss of polarity, spindle shape, and increase in mobility of these cells along with a decrease in E-cadherin expression and an increase in the expression of Snail and vimentin. This study showed that chronic oxaliplatin resistance in CRC cells leads them to switch to an invasive phenotype and initiates EMT [295]. Long-term exposure of CRC cells to 5-FU enabled these cells to overcome S-phase arrest, evade apoptosis and activate autophagy which is evident by the up-regulation of LC3B, vimentin, Twist1, Slug, and zeb2 mRNA levels and down-regulation of E-cadherin and Claudin-3 [296]. These results suggested that CRC cells respond to chemotherapy-induced cell stress by undergoing EMT-plasticity as an adaptive mechanism, leading to cell survival, plasticity, and evasion of apoptosis.
Another critical determinant of plasticity in CRC cells is CSC. CSCs are a subset of cells found in tumors that can self-renew, differentiate, and produce all cancer cell types. They are responsible for tumor initiation, maintenance, and recurrence [297]. CSCs are isolated and enriched from different tumors by identifying the CRC-specific expression of cell surface markers, including CD44, CD133, CD166, Lgr5, ALDH1, and EpCAM [298]. Further, they initiate aberrant expression of several cellular signaling pathways to maintain their stemness and self-replenishing properties. For instance, Wnt/β-catenin, Notch, TGF-β, and Hedgehog pathways are well implicated in colon cancer CSC development [299–303].
Colon CSCs are the initiators of tumor cell proliferation, invasion, and metastasis to distinct locations. They are attributed to showing high resistance to chemotherapy and are a fundamental reason for tumor relapse or recurrence [304]. For example, many studies have reported that colon CSCs exhibit increased expression of anti-apoptotic proteins and apoptotic inhibitors, as well as ABC transporter proteins that expel drugs out of cancer cells [305, 306]. Moreover, an interesting study revealed that most circulating or migratory CRC cells are Lgr5- and this plastic behavior has an immense capacity for distant metastasis [307]. Additionally known is that the CD133+ CRC cell population resists anti-angiogenesis therapy, and this resistance occurs through an anti-apoptotic pathway including PP2A, p38MAPK, MAPKAPK2, and Hsp27 [308]. In sum, understanding the biology of CSCs is vital to develop novel therapies that can effectively target this hallmark of tumor plasticity.
Transdifferentiation (lineage plasticity), which refers to the ability of tumor cells and CSCs to switch their phenotypic characteristics into a different cell type, represents a different type of cell plasticity. Accumulating evidence has demonstrated that tumor cells vulnerable to therapeutic drugs transdifferentiate into other specialized cell lineages that are not drug targets [144, 309, 310]. Overall, transdifferentiation is a complex, poorly understood process but is believed to be an essential factor in developing drug resistance to cancer. Therefore, research is needed to understand the differentiation mechanism and its impact on drug resistance in cancer.
Resveratrol’s chemosensitizing effect by modulation of tumor cell plasticity in CRC cells
Data from different studies have shown that resveratrol sensitizes CRC cells toward chemotherapeutic drugs by modulating their plasticity via many signaling pathways and transcription factors. For example, a fascinating study revealed resveratrol’s suppression of CRC cell invasion and migration by inhibiting the TGF-β1/Smads signaling pathway and EMT. Resveratrol elevated the levels of E-cadherin but down-regulated EMT-inducing transcription factors, Snail and vimentin [196].
Even more interesting, a co-treatment with 5-FU and resveratrol in HCT-116 cells significantly lowered the levels of EMT regulatory factors such as Slug and vimentin and the stemness of the treated cells compared to untreated cells [209]. In accordance therewith, Buhrmann et al. reported that resveratrol treatment chemosensitized HCT-116 CRC cells to 5-FU and induced apoptosis while suppressing NF-κB activation, EMT-plasticity (decreased slug and vimentin, increased E-cadherin) and CSC formation (decreased CD133, CD44, and ALDH1) via the modulation of the TNF-β signaling pathway [80]. Resveratrol (5μM) treatment in 5-FU-resistant CRC cell lines HCT-116R and SW480R and their parental forms (HCT-116 and SW480) blocked cell proliferation and synergistically inhibited 5-FU (0.01-1nM) mediated effects on cell invasion [79]. Resveratrol increased cell-cell contact via increased desmosomes, gap- and tight junctions, and increased the expression of E-cadherin cell adhesion protein in both the parental and 5-FU-resistant forms of the HCT-116 CRC cell line [79]. Interestingly, a significant decrease in the vimentin and Slug (plasticity-associated factors) and the down-regulation of the activation and nuclear translocation of NF-κB (abolishing NF-κB driven gene expression of MMP9 and caspase-3) correlated to the ability of resveratrol to attenuate drug resistance in the 5-FU-resistant CRC cells [79]. Beyond that, the role of oxyresveratrol, a natural derivative of resveratrol, in inhibiting EMT-plasticity and metastasis of CRC cells has also been proven [311].
Similarly, many studies focus on targeting aberrant signaling pathways leading to the proliferation and enrichment of colorectal CSCs. It is well established that mutations in the Wnt/β-catenin signaling pathway, which take place in the stem cells of the intestinal crypt, are crucial for the continued proliferation of cancerous cells and stemness activity in colorectal stem cells [312]. For instance, an intriguing study demonstrated that resveratrol potentiated the anti-cancer effects of grape seed extract against colorectal CSCs in a rodent model by suppressing the Wnt/β-catenin pathway. Besides, the compound also induced mitochondrial apoptosis of colon CSCs by up-regulating the Bax/Bcl-2 ratio, p53, and cleaved PARP [313].
Transdifferentiation of CSCs to endothelial cells is an essential event in vascular bed formation for angiogenesis and metastasis of tumor cells [314]. Pouyafar and colleagues reported that resveratrol, in combination with sulindac (a Wnt-3a inhibitor), reduced the clonogenicity potential of colon CSCs and prevented the differentiation of CSCs to endothelial cells. Moreover, resveratrol treatment reduced angiogenesis factor YKL-40 and autophagy-related genes in CRC cells [315].
The pathogenesis of CRC is a complex process that can be distinguished on the basis of three different phenotypes: Chromosomal instability (CIN), Microsatellite instability (MSI), and CpG island methylator (CIMP). Indeed, intracellular biotargets of resveratrol, including a protein with high binding affinity, quinone reductase 2 (QR2), were found to be significantly overexpressed in CRC defined by CIN, particularly in cells harboring a positive KRAS (Kirsten rat sarcoma viral oncogene homolog) mutation, and by the MSI but not the CIMP phenotype. Analysis of data from Oncomine showed very good agreement between mRNA expression of QR2 and specific CRC causes [316]. In addition, several genes involved in the regulation of apoptosis, such as PMAIP1, BID, ZMAT3, CASP3, CASP7, and FAS, have been shown to be novel targets for gene regulatory treatment with resveratrol [317].
Conclusively, it is evident that resveratrol can target different tumor cell plasticity markers, provides a novel approach for treating CRC, and can potentially be a potent therapeutic agent.
Resveratrol’s further chemosensitizing effects on CRC cells
Several articles have extensively documented the synergistic effects of resveratrol and its ability to sensitize drug-resistant cancers, including CRC, to therapeutic intervention [318]. The chemosensitizing effects of this phytopharmaceutical in CRC are summarized in Table 4 [319–324].
Table 4.
Effect of resveratrol in a combinatory therapeutic approach
| Drug | CRC cell line/cancer model | Chemosensitizing/resensitizing effect of resveratrol in a combinatory therapeutic approach | References |
|---|---|---|---|
| 5-Fluorouracil (5-FU) | DLD1 and HCT-116 cells | ↓Akt signaling pathway; ↓Cellular proliferation and migration; ↑S-phase cell-cycle arrest; ↑Apoptosis; ↓Slug and vimentin (EMT signaling factors); ↓Stemness; ↓STAT3 binding to hTERT promoter site, ↓Plasticity | [209] |
| HT-29 and SW620 cells | ↑Mitochondrial oxidative stress; ↓Akt; ↓STAT3 | [210] | |
| HCT-116 and HCT-116R* cells | ↑Apoptosis (caspase-3); ↓Vimentin and slug, while ↑E-cadherin (EMT factors); ↓CSC phenotype (CD133, CD44, ALDH1); ↓TNFβ induced activation of NF-κB, MMP9, CXCR4 | [80] | |
| HCT-116, HCT-116R*; SW480 and SW480R* cells | ↓Cell proliferation; ↓Cell invasion; ↑Cell-cell contact (↑ desmosomes, gap- and tight-junctions); ↑E-cadherin; ↓Vimentin and slug; ↓NF-κB activation and nuclear translocation; ↓NF-κB driven genes (MMP9, caspase-3) | [79] | |
| HCT-116 and HCT-116R* cells | ↓β1-integrin/HIF1α axis B activation; ↓TME promoted viability; ↓Proliferation; ↓Colony formation; ↓Invasion tendency; ↓EMT; ↓NF-κB; ↓VEGF; ↓HIF1α; ↓Stem cell markers (CD44, CD133, ALDH1); ↑Caspase-3; ↑Apoptosis | [319] | |
| Cetuximab | HCT-116 and CT-26 (mouse cell line) cells | ↓Growth; ↑Cx43 expression and phosphorylation; ↑Gap junction function; ↓Akt; ↓NF-κB, ↓Plasticity | [320] |
| Doxorubicin (Adriamycin) | Caco-2 cells | ↓P-gp and MDR1; ↓Drug-efflux/extrusion from cells; ↓ CYP3A4 and GST (drug metabolizing enzymes); ↑Caspases-3, -8 and -9; ↑Apoptosis | [321] |
| HT-29 and HCT-116 cells | ↓IC50 of doxorubicin; ↑Bax; ↑Apoptosis; ↑S-phase cell-cycle arrest; ↓P-gp, ↓Plasticity | [322] | |
| Oxaliplatin | Caco-2 cells | ↓Cell proliferation; ↓Growth; ↓Survivin; ↑PARP cleavage; ↑Caspase-3 activity; ↑Apoptosis, ↓Plasticity | [323] |
| Drug | CRC cell line/cancer model | Anti-chemosensitizing effect of resveratrol in a combinatory therapeutic approach | References |
| Oxaliplatin | HCT-116 cells | ↑Survivin; ↓Apoptosis, ↓Plasticity | [324] |
The upward arrow (↑) indicates an activation/increase/up-regulation and the downward arrow (↓) indicates a decrease/down-regulation/suppression. R* indicates resistant cell line. Abbreviations: Akt protein kinase B, ALDH1 aldehyde dehydrogenase 1, CSC cancer stem cell, Cx43 connexin 43, CXCR4 C-X-C motif chemokine receptor 4, EMT epithelial-to-mesenchymal transition, hTERT telomerase reverse transcriptase (human), MMP9 matrix metalloproteinase 9, NF-κB nuclear factor kappa-light-chain-enhancer of activated B-cells, STAT3 signal transducer and activator of transcription 3, TME tumor microenvironment, TNFβ tumor necrosis factor β
5-FU is one of the most frequently chemotherapeutic drugs in the treatment of CRC, either as a monotherapy or in the drug combinations such as FOLFIRI (Folinic acid + Fluorouracil l + Irinotecan hydrochloride) and FOLFOX (Folinic acid + Fluorouracil + Oxaliplatin) [325]. Due to several mechanisms, drug resistance to 5-FU has been observed in many CRC patients [325]. Resveratrol seems to be able to re-sensitize the 5-FU-resistant CRC cells, rendering them susceptible to 5-FU intervention. Inhibition of the Akt signaling pathway, induction of S-phase cell-cycle arrest, inhibition of cellular proliferation and migration, and activation of programmed cell death occurred when the HCT-116 CRC cells were treated with a combination of 5-FU (10μM) and resveratrol (25μM) [209]. Furthermore, the combination of 5-FU and resveratrol inactivated STAT3 and blocked STAT3 binding to its hTERT promoter site, thereby blocking telomerase activity in HCT-116 cells [209]. Another study, using HT-29 and SW620 CRC cell lines, reported that resveratrol (100μM) exposure synergistically potentiated the 5-FU (10μM) treatment-mediated inhibition of cellular growth via the induction of mitochondrial oxidative stress and created an imbalance in the intracellular anti-oxidant enzymes [210]. The combination (resveratrol and 5-FU) treatment-induced increase in cellular oxidative stress was attributed to the significant inhibition of oncogenic Akt and STAT3 in the treated cells [210].
The epidermal growth factor receptor (EGFR), frequently overexpressed in malignant cells, is a crucial contributor to cancer proliferation, angiogenesis, inhibition of apoptosis, and metastasis [320]. Hence, the EGFR pathway can be targeted to curb tumor growth. Cetuximab, a monoclonal antibody, targets EGFR, hinders endogenous ligand binding to EGFR, and thus suppresses the phosphorylation and activation of EGFR. Subsequently, disruption in the EGFR-related downstream pathways, such as the Ras-Raf-MAPK and PI3K-Akt pathways, have also been reported [320]. However, mutations caused by NRAS, KRAS, BRAF, PI3KCA, and Akt activation in cancers may confer the cancer cells’ therapeutic resistance to cetuximab and render it a less effective anti-cancer agent [320, 326]. Cancer cells develop resistance to cetuximab via the activation of Akt [320]. Wang et al. showed that resveratrol (5μg/ml) treatment in HCT-116 and CT-26 (murine colon adenocarcinoma) cells abolished the resistance to cetuximab and sensitized the cells to cetuximab (10μg/ml) exposure [320]. Resveratrol treatment in cetuximab-exposed cells up-regulated the expression and phosphorylation of connexin 43 with a resultant increase in cell-cell contact via gap junction function. It inhibited the activation of Akt and NF-κB which is related to the increase in cetuximab treatment associated with the suppression of the growth of the cancer cells [320].
Resveratrol (50–100μM) potentiated the oxaliplatin (1μM) mediated inhibition of Caco-2 CRC cell growth [323]. This growth inhibitory effect of the combination of resveratrol and oxaliplatin also increased the apoptotic death of Caco-2 cells [323]. Furthermore, the conditioned media from resveratrol and oxaliplatin-treated Caco-2 cells, when used to grow human-monocyte-derived macrophages, activated the tumoricidal potential of the macrophages while preventing their immunosuppressive characteristics [323].
The efficacy of a chemotherapeutic intervention and/or resistance to the drug quite often depends on levels of expression of drug extrusion membrane transporters (such as P-gp, MDR1, and BCRP) and the levels and activity of intracellular drug-metabolizing enzymes (such as CYP3A4 and GST) [321]. In a doxorubicin-resistant CRC cell line, Caco-2, increasing concentrations of resveratrol (1–500μM) inhibited the drug extrusion capabilities of P-gp and MDR1 proteins in a concentration-dependent manner [321]. The resveratrol (20μM) exposed Caco-2 cells were susceptible to doxorubicin treatment compared to non-resveratrol exposed doxorubicin treated cells [321]. Resveratrol also inhibited the activity of drug-metabolizing enzymes CYP3A4 and GST in these cells and increased the caspases-3, caspases-8, and caspases-9, indicating activation of apoptotic cell death in the treated cells [321]. The mRNA levels of P-gp, MDR1, BCRP, CYP3A4, and GST were significantly reduced upon resveratrol treatment in the drug-resistant Caco-2 cells [321]. In HCT-116 and HT-29 CRC cells, a combination of doxorubicin and resveratrol significantly reduced the IC50 value of the doxorubicin in the cells [322]. The expression of the pro-apoptotic Bax gene and apoptosis significantly increased when the doxorubicin-exposed HCT-116 cells were treated with resveratrol while inducing S-phase arrest in these cells [322]. Resveratrol seems to sensitize the HCT-116 cells to the anti-cancer effects of doxorubicin by blocking the P-gp drug efflux mechanism in these cells [322].
It is, however, noteworthy that certain studies reported contradicting data; that resveratrol (30 or 50μM) reverses the inhibitory effect of oxaliplatin (2 or 5μM) on the mRNA expression and protein levels of anti-apoptotic survivin in HCT-116 CRC cells and thereby abolishes the cytotoxic effects of oxaliplatin [324].
Insights of clinical resveratrol application in CRC patients
Based on the promising preclinical results, resveratrol’s modulatory impact on CRC was clinically tested. Despite the advanced stage of the tumor and the associated vulnerability of CRC patients, resveratrol supplementation was well tolerated in all studies to date (Table 5). Regardless of the dosage form, there was no toxicity or gastrointestinal problems [35, 327, 328].
Table 5.
Proven anti-CRC effects of resveratrol in clinical trials
| # of CRC Patients | Study phase | Resveratrol application | Resveratrol’s effects | Signaling target | Year of publication | Reference |
|---|---|---|---|---|---|---|
| 9 | 1 | 5g SRT501 (microparticular, in sachets)/day, for 10–21 day | Resveratrol was well-tolerated and was detected in blood plasma as well as hepatic tissue affected by metastases. It induced an up-regulation (39%) of apoptosis in malignant tissue. | Apoptosis, caspase-3 | 2011 | [327] |
| 20 | 1 | 0.5g or 1g (caplets)/day, for 8 days | Resveratrol was well-tolerated and reduced (p=0.05) CRC cell proliferation. | Proliferation, Ki-67 | 2010 | [35] |
| 12 selected, 8 completed | 1 | 20mg or 80mg (tablets)/day, for 14 days | Resveratrol was well tolerated and inhibited significantly (p<0.03) the CRC initiation in normal colonic mucosa. | Initiation, Wnt signaling gene | 2009 | [328] |
Abbreviations: CRC colorectal cancer, Ki-67 kiel-antigen 67, Wnt wingless-related integration site
Interestingly, a relatively low dose of the phytopharmaceutical (tablets containing 20mg or 80mg resveratrol) significantly reduced the expression of the Wnt gene in the mucosa of the colon cells [328]. Moreover, as the Wnt pathway represents a key signaling for CRC initiation [328], this finding could be highly relevant to break the cancerous cycle. This approach is supported by another clinical trial where CRC patients received 0.5g or 1g resveratrol as caplets for 8 days [35]. Here, treatment with the natural polyphenol inhibited the detection of cell proliferation parameter Ki-67 [35]. Furthermore, the detection of resveratrol uptake in blood and liver affected by metastases succeeded [327]. In malignant hepatic tissue, the phytopharmaceutical induced apoptosis in metastasized CRC cells [327].
Apart from metastasis, chemoresistance is also a common issue in the advanced stage of the disease, so it is currently being investigated with great interest whether resveratrol increases the susceptibility of CRC patients to classic chemotherapeutic agents.
Data from ClincalTrials.gov (https://clinicaltrials.gov/) showed a list of nineteen (search performed on 28 March 2023; using keywords ‘cancer’ and ‘resveratrol’) resveratrol administration-based clinical trials in different cancers. Out of the nineteen trials, there were only three clinical trials pertaining specifically to colon/colorectal cancer and resveratrol intervention’s therapeutic effect/efficacy in these patients (Table 5). Interestingly none of the studies aimed at studying the ability of resveratrol to overcome chemoresistance. Given the abundance of in vitro/in vivo data pointing towards the therapeutic efficacy of resveratrol and its ability to re-sensitize cancers to a drug intervention in treating CRC, more clinical trials are warranted to determine whether the therapeutic chemosensitizing and anti-plasticity effects of resveratrol can be translated to clinical use.
Outlook and future perspectives
In treating CRC, chemotherapy represents one of the most important and best therapeutic options by present-day standards. This kind of therapy is highly specialized, mono-targeted, and expensive. In recent years, conventional drug strategy has achieved relatively low efficacy with many severe side effects [329], mainly due to the development of chemoresistance, which represents a significant obstacle to their use [330].
This resistance of cancer cells is based on different molecular pathways that give them key advantages in resisting the applied chemotherapeutic drugs, such as impairment of mitochondria and associated disruption of the respiratory chain. Also, an impairment of the endoplasmic reticulum (ER) and associated disruption of ER functions, impairment of glucose metabolism and related activation of Warburg action, impairment of lysosomal function, and associated residues of chemotherapeutic drugs [331–334] are of significance.
In this regard, it is known that fundamental causes contributing to the emergence of resistance include poorer drug accessibility, complete signaling alteration in CRC cells, and activation of pathways that promote metastasis formation [207, 329, 330]. Relating to that, a sensitization of tumor cells to conventional drugs seems to be a possible way to overcome the resistance of CRC cells. For this purpose, certain natural plant-based compounds could be administered to optimize the effect of chemotherapeutics by modulating different resistance mechanisms [335, 336].
The secondary plant compound resveratrol enhances the effect of common chemotherapeutic agents such as 5-FU or oxaliplatin in CRC cell treatment by simultaneously influencing numerous cellular signal transduction processes [79, 337]. Unlike conventional drugs, phytopharmaceuticals use a poly-targeting strategy, thus preventing the development of acquired resistance and blocking intrinsic drug resistance mechanisms in some instances. This article, therefore, discusses the use of resveratrol as an anti-plasticity agent based on its ability to target and regulate multiple pro-oncogenic and tumor-suppressing mechanisms (EMT, CSC) and circumvent and overcome the chemoresistance in CRC. The natural polyphenol is proposed as an alternative co-therapeutic agent in combination with standard chemotherapeutic interventions, and its application would be a novel, innovative way.
Considered as a whole, the use of active plant ingredients in oncology has many advantages. They represent a unique, virtually inexhaustible source in nature with their mechanisms of action based on a millennium of experience, development, and adaptation. Their function as chemosensitizers is mainly performed by enhancing the effects of conventional drugs in cancer cells, inhibiting tumor-promoting inflammation, repressing cancer cell plasticity triggering their cell death by activating pro-apoptotic targets and inhibiting anti-apoptotic targets, and causing DNA damage. Non-physiologic inflammation represents a critical initiator and promotor of carcinogenesis and thus can substantially affect the therapy in cancer patients. Resveratrol demonstrates significant anti-inflammatory characteristics, which have the potential to enhance the therapeutic outcome and prevent the resistance of CRC cells to chemotherapy drugs in oncology practice. Indeed, resveratrol is a chemosensitizing agent in CRC cells that reduces inflammation response via decreased levels of phosphorylated NF-κB, JAK/STAT3, MMPs, and COX-2 signaling, and pro-inflammatory cytokine levels [79, 80, 209, 338]. All these effects can enhance the toxic properties of conventional anti-cancer drugs and create a kind of synergy with the standard chemotherapeutics, thus leading to anti-resistance mechanisms [64, 65].
Moreover, good tolerability in patients with the natural-component material should be emphasized in this context [339]. A sometimes-cited criticism of the use of phytopharmaceuticals, including resveratrol, is the low bioavailability in the human body. It is undisputed but known that continuous oral supplementation can achieve the desired health-promoting quantities [340]. Similarly, the realization of a piperine-coupled multiplication of resveratrol’s bioavailability [341] could be enriching.
Overall, a majority of the work that has been done has shown the chemosensitization effects of herbal agents such as resveratrol. Still, preventive and clinical approaches are needed to establish whether the above combinations have synergistic effects during direct application on CRC patients. Based on the great need for complementary therapeutic strategies and the favorable availability of secondary plant polyphenols, this area of research will expand rapidly in the future to provide effective treatments for CRC chemoresistance.
Conclusion
Based on comprehensive preclinical research, resveratrol exerts chemoprotective and chemosensitizing effects through anti-plasticity, anti-oxidant, anti-inflammatory, and pro-apoptotic modes of action. In addition to its anti-cancer efficacy, resveratrol could work as a mitigating agent in chemotherapy-induced toxicities in normal cells/tissues.
In this article, we have shown that the polyphenol resveratrol could be used not only in the prevention of CRC but also as an anti-cancer agent (targeting multiple mechanisms) in conjunction with conventional chemotherapeutic agents to enhance their therapeutic effects and reduce chemoresistance effects through additive and synergistic effects.
Furthermore, we show here that the molecular targets of chemopreventive resveratrol are similar to those currently used for treating CRC, highlighting the importance of more clinical trials to validate its anti-cancer efficacy and reliability in a clinical setting. Using resveratrol as a cancer chemoprotector or chemosensitizing agent combined with conventional chemotherapeutics in CRC patients needs further in-depth clinical evaluations to elucidate discrepancies between the preclinical cancer research and clinical practice. These studies must (a) include precise analysis of pharmacokinetic parameters of resveratrol in humans; (b) find sufficient and safe dosing of resveratrol when used as a monotherapy and/or in combination with existing chemotherapeutics; (c) find the effective combinations of resveratrol with conventionally used chemotherapeutic drugs to re-sensitize the chemo-/radiotherapy-resistant cancers; (d) provide a detailed description of the cellular targets of resveratrol and its effect on specific problems of oncology such as disease relapse and therapy resistance; (e) provide an understanding on how specific individual characteristics of a cancer patient (which can vary from one patient to another) may affect the therapeutic efficacy, chemosensitizing potential and prognosis of resveratrol formulations among treated CRC patients; and (f) asses advanced drug formulations and improved drug deliver techniques, such as nanotechnology, to specifically target neoplastic cells/tissue and this avoid any possible off-target and side-effects of resveratrol when used as a monotherapy and/or in combination with existing chemotherapeutics.
Overall, this review summarizes, for the first time to our knowledge, the chemosensitization of tumor cells to chemotherapeutic agents by resveratrol from the perspective of cellular plasticity, which is of enormous importance for cellular adaptations to the TME during cancer cell transformation and metastasis. It also provides a new impetus for further research into cancer epigenetics and resveratrol-mediated suppression of plasticity, which is a strong indication that resveratrol, an active phytochemical, will play an influential role in the prevention and treatment of CRC in the future.
Acknowledgements
We thank Andreas Eimannsberger for his reliable technical support.
Abbreviations
- 5-LOX
5-lipoxygenase
- 5-FU
5-fluorouracil
- AP-1
activator protein 1
- Bcl-2
B-cell lymphoma 2
- Bcl-xL
B-cell lymphoma extra-large
- CIMP
CpG island methylator
- CIN
chromosomal instability
- COX
cyclooxygenase
- CRC
colorectal cancer
- CSC
cancer stem cell
- CTC
circulating tumor cell
- CTM
circulating tumor microemboli
- DNA
deoxyribonucleic acid
- EGF
epidermal growth factor
- EGFR
epidermal growth factor receptor
- EMT
epithelial-to-mesenchymal transition
- EpCAM
epithelial cell adhesion molecule
- ER
endoplasmic reticulum
- HDAC
histone deacetylase
- HGF
hepatocyte growth factor
- HIF
hypoxia-inducible factor
- IkBα
NF-κB inhibitor alpha
- IBD
inflammatory bowel disease
- IFN
interferon
- IL
interleukin
- iNOS
isoenzymes of NO synthase
- Ki-67
Kiel-antigen 67
- MAPK
mitogen-activated protein kinase
- MDR
multidrug resistance
- MMP
matrix metalloproteinase
- mRNA
messenger ribonucleic acid
- MRP
MDR-associated protein
- MSI
microsatellite instability
- NF-κB
nuclear factor kappa-light-chain-enhancer of activated B-cells
- NO
nitric oxide
- Nrf2
nuclear factor erythroid 2–related factor 2
- p53
cellular tumor antigen p53
- PGE2
prostaglandin
- P-gp
p-glycoproteins
- PRP4
pre-mRNA processing factor
- QR2
quine reductase 2
- ROS
reactive oxygen species
- SIP1
smad interacting protein 1
- SIRT
sirtuins
- STAT
signal transducer and activator of transcription
- TGF-β
transforming growth factor beta
- Th17
T helper 17 cells
- TME
tumor microenvironment
- TNF
tumor necrosis factor
- TPA
12-o-tetradecanoylphorbol-13-acetate
- TRAIL
tumor necrosis factor-related apoptosis-inducing ligand
- Tregs
regulatory T-cells
- VEGF
vascular endothelial growth factor.
Author contribution
AB and MS prepared the original structure and draft of the manuscript, while all authors wrote, reviewed, and edited the manuscript and prepared the Figures and Tables. MS coordinated project management and correspondence. All authors read and agreed to the published manuscript version.
Funding
Open Access funding enabled and organized by Projekt DEAL. Samson Mathews Samuel was supported by a National Priorities Research Program grant (NPRP 14S-0311-210033; awarded to Prof. Dr. Dietrich Büsselberg, January 2023-Current) from the Qatar National Research Fund (QNRF, a member of Qatar Foundation). The statements made herein are solely the responsibility of the authors.
Data availability
All data are available in the manuscript.
Declarations
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 Countries. CA: a Cancer Journal for Clinicians. 2021;71(3):209–249. doi: 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- 2.Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. CA: a Cancer Journal for Clinicians. 2022;72(1):7–33. doi: 10.3322/caac.21708. [DOI] [PubMed] [Google Scholar]
- 3.Sadahiro S. A study of tumor-associated cellular proteins from carcinoma and adenoma of the human colon. Nihon Geka Gakkai Zasshi. 1987;88(5):551–561. [PubMed] [Google Scholar]
- 4.Welch JP, Donaldson GA. Perforative carcinoma of colon and rectum. Annals of Surgery. 1974;180(5):734–740. doi: 10.1097/00000658-197411000-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Donadon M, Ribero D, Morris-Stiff G, Abdalla EK, Vauthey JN. New paradigm in the management of liver-only metastases from colorectal cancer. Gastrointestinal Cancer Research: GCR. 2007;1(1):20–27. [PMC free article] [PubMed] [Google Scholar]
- 6.Teufel A, Li M, Gerken M, Ebert MP, Schlitt HJ, Evert M, et al. Second cancer after additive chemotherapy in patients with colon cancer. Clinical Colorectal Cancer. 2022;21(4):354–361. doi: 10.1016/j.clcc.2022.07.002. [DOI] [PubMed] [Google Scholar]
- 7.Krishna R, Mayer LD. Multidrug resistance (MDR) in cancer. Mechanisms, reversal using modulators of MDR and the role of MDR modulators in influencing the pharmacokinetics of anticancer drugs. European Journal of Pharmaceutical Sciences. 2000;11(4):265–283. doi: 10.1016/s0928-0987(00)00114-7. [DOI] [PubMed] [Google Scholar]
- 8.Yoganathan, S., Alagaratnam, A., Acharekar, N., & Kong, J. (2021). Ellagic Acid and Schisandrins: Natural Biaryl Polyphenols with Therapeutic Potential to Overcome Multidrug Resistance in Cancer. Cells, 10(2). 10.3390/cells10020458 [DOI] [PMC free article] [PubMed]
- 9.Lv L, Qiu K, Yu X, Chen C, Qin F, Shi Y, et al. Amphiphilic copolymeric micelles for doxorubicin and curcumin co-delivery to reverse multidrug resistance in breast cancer. Journal of Biomedical Nanotechnology. 2016;12(5):973–985. doi: 10.1166/jbn.2016.2231. [DOI] [PubMed] [Google Scholar]
- 10.Duan J, Mansour HM, Zhang Y, Deng X, Chen Y, Wang J, et al. Reversion of multidrug resistance by co-encapsulation of doxorubicin and curcumin in chitosan/poly(butyl cyanoacrylate) nanoparticles. International Journal of Pharmaceutics. 2012;426(1-2):193–201. doi: 10.1016/j.ijpharm.2012.01.020. [DOI] [PubMed] [Google Scholar]
- 11.Krepler C, Xiao M, Sproesser K, Brafford PA, Shannan B, Beqiri M, et al. Personalized preclinical trials in BRAF inhibitor-resistant patient-derived xenograft models identify second-line combination therapies. Clinical Cancer Research. 2016;22(7):1592–1602. doi: 10.1158/1078-0432.Ccr-15-1762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Watson KM, Siemens KN, Anand S, Gardner IH, Sharpton TJ, Dewey EN, et al. Dietary and lifestyle associations with microbiome diversity. Gut Pathogens. 2022;14(1):49. doi: 10.1186/s13099-022-00525-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Johnson IT. Cruciferous Vegetables and Risk of Cancers of the Gastrointestinal Tract. Molecular Nutrition & Food Research. 2018;62(18):e1701000. doi: 10.1002/mnfr.201701000. [DOI] [PubMed] [Google Scholar]
- 14.Metere A, Giacomelli L. Absorption, metabolism and protective role of fruits and vegetables polyphenols against gastric cancer. European Review for Medical and Pharmacological Sciences. 2017;21(24):5850–5858. doi: 10.26355/eurrev_201712_14034. [DOI] [PubMed] [Google Scholar]
- 15.Aggarwal BB, Van Kuiken ME, Iyer LH, Harikumar KB, Sung B. Molecular targets of nutraceuticals derived from dietary spices: potential role in suppression of inflammation and tumorigenesis. Experimental Biology and Medicine (Maywood, N.J.) 2009;234(8):825–849. doi: 10.3181/0902-mr-78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bishayee A, Sethi G. Bioactive natural products in cancer prevention and therapy: Progress and promise. Seminars in Cancer Biology. 2016;40-41:1–3. doi: 10.1016/j.semcancer.2016.08.006. [DOI] [PubMed] [Google Scholar]
- 17.Aggarwal BB, Vijayalekshmi RV, Sung B. Targeting inflammatory pathways for prevention and therapy of cancer: short-term friend, long-term foe. Clinical Cancer Research. 2009;15(2):425–430. doi: 10.1158/1078-0432.Ccr-08-0149. [DOI] [PubMed] [Google Scholar]
- 18.Brockmueller A, Mueller AL, Kunnumakkara AB, Aggarwal BB, Shakibaei M. Multifunctionality of Calebin A in inflammation, chronic diseases and cancer. Frontiers in Oncology. 2022;12:962066. doi: 10.3389/fonc.2022.962066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Muhammad N, Steele R, Isbell TS, Philips N, Ray RB. Bitter melon extract inhibits breast cancer growth in preclinical model by inducing autophagic cell death. Oncotarget. 2017;8(39):66226–66236. doi: 10.18632/oncotarget.19887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bhattacharya S, Muhammad N, Steele R, Peng G, Ray RB. Immunomodulatory role of bitter melon extract in inhibition of head and neck squamous cell carcinoma growth. Oncotarget. 2016;7(22):33202–33209. doi: 10.18632/oncotarget.8898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Celotti E, Ferrarini R, Zironi R, Conte LS. Resveratrol content of some wines obtained from dried Valpolicella grapes: Recioto and Amarone. Journal of Chromatography. A. 1996;730(1-2):47–52. doi: 10.1016/0021-9673(95)00962-0. [DOI] [PubMed] [Google Scholar]
- 22.Rolfs CH, Fritzemeier KH, Kindl H. Cultured cells of Arachis hypogaea susceptible to induction of stilbene synthase (resveratrol-forming) Plant Cell Reports. 1981;1(2):83–85. doi: 10.1007/bf00269281. [DOI] [PubMed] [Google Scholar]
- 23.Lorenz P, Roychowdhury S, Engelmann M, Wolf G, Horn TF. Oxyresveratrol and resveratrol are potent antioxidants and free radical scavengers: Effect on nitrosative and oxidative stress derived from microglial cells. Nitric Oxide. 2003;9(2):64–76. doi: 10.1016/j.niox.2003.09.005. [DOI] [PubMed] [Google Scholar]
- 24.Chen CK, Pace-Asciak CR. Vasorelaxing activity of resveratrol and quercetin in isolated rat aorta. General Pharmacology. 1996;27(2):363–366. doi: 10.1016/0306-3623(95)02001-2. [DOI] [PubMed] [Google Scholar]
- 25.Rakici O, Kiziltepe U, Coskun B, Aslamaci S, Akar F. Effects of resveratrol on vascular tone and endothelial function of human saphenous vein and internal mammary artery. International Journal of Cardiology. 2005;105(2):209–215. doi: 10.1016/j.ijcard.2005.01.013. [DOI] [PubMed] [Google Scholar]
- 26.Cai, X., Liu, M., Zhang, B., Zhao, S. J., & Jiang, S. W. (2021). Phytoestrogens for the management of endometriosis: Findings and issues. Pharmaceuticals (Basel), 14(6). 10.3390/ph14060569 [DOI] [PMC free article] [PubMed]
- 27.Lephart, E. D. (2021). Phytoestrogens (resveratrol and equol) for estrogen-deficient skin-controversies/misinformation versus anti-aging in vitro and clinical evidence via nutraceutical-cosmetics. International Journal of Molecular Sciences, 22(20). 10.3390/ijms222011218 [DOI] [PMC free article] [PubMed]
- 28.Louis XL, Raj P, Chan L, Zieroth S, Netticadan T, Wigle JT. Are the cardioprotective effects of the phytoestrogen resveratrol sex-dependent? Canadian Journal of Physiology and Pharmacology. 2019;97(6):503–514. doi: 10.1139/cjpp-2018-0544. [DOI] [PubMed] [Google Scholar]
- 29.Stahl S, Chun TY, Gray WG. Phytoestrogens act as estrogen agonists in an estrogen-responsive pituitary cell line. Toxicology and Applied Pharmacology. 1998;152(1):41–48. doi: 10.1006/taap.1998.8500. [DOI] [PubMed] [Google Scholar]
- 30.Zhang N, Zhuang L, Gai S, Shan Y, Wang S, Li F, et al. Beneficial phytoestrogenic effects of resveratrol on polycystic ovary syndromein rat model. Gynecological Endocrinology. 2021;37(4):337–341. doi: 10.1080/09513590.2020.1812569. [DOI] [PubMed] [Google Scholar]
- 31.Tanyeri G, Celik O, Erbas O, Oltulu F, Yilmaz Dilsiz O. The effectiveness of different neuroprotective agents in facial nerve injury: An experimental study. Laryngoscope. 2015;125(11):E356–E364. doi: 10.1002/lary.25554. [DOI] [PubMed] [Google Scholar]
- 32.Culpitt SV, Rogers DF, Fenwick PS, Shah P, De Matos C, Russell RE, et al. Inhibition by red wine extract, resveratrol, of cytokine release by alveolar macrophages in COPD. Thorax. 2003;58(11):942–946. doi: 10.1136/thorax.58.11.942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Berman, A. Y., Motechin, R. A., Wiesenfeld, M. Y., & Holz, M. K. (2017). The therapeutic potential of resveratrol: A review of clinical trials. NPJ Precision Oncology, 1. 10.1038/s41698-017-0038-6 [DOI] [PMC free article] [PubMed]
- 34.Magyar K, Halmosi R, Palfi A, Feher G, Czopf L, Fulop A, et al. Cardioprotection by resveratrol: A human clinical trial in patients with stable coronary artery disease. Clinical Hemorheology and Microcirculation. 2012;50(3):179–187. doi: 10.3233/ch-2011-1424. [DOI] [PubMed] [Google Scholar]
- 35.Patel KR, Brown VA, Jones DJ, Britton RG, Hemingway D, Miller AS, et al. Clinical pharmacology of resveratrol and its metabolites in colorectal cancer patients. Cancer Research. 2010;70(19):7392–7399. doi: 10.1158/0008-5472.can-10-2027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chhabra, G., Singh, C. K., Amiri, D., Akula, N., & Ahmad, N. (2021). Recent advancements on immunomodulatory mechanisms of resveratrol in tumor microenvironment. Molecules, 26(5). 10.3390/molecules26051343 [DOI] [PMC free article] [PubMed]
- 37.Fuggetta M, Mattivi F. The immunomodulating activities of resveratrol glucosides in humans. Recent Patents on Food, Nutrition & Agriculture. 2011;3(2):81–90. doi: 10.2174/2212798411103020081. [DOI] [PubMed] [Google Scholar]
- 38.Martin LM, Johnson PJ, Amorim JR, Honaker AR, Donaldson RS, DeClue AE. Investigation of the potential immunomodulatory effects of resveratrol on equine whole blood: An in vitro investigation. Research in Veterinary Science. 2016;106:97–99. doi: 10.1016/j.rvsc.2016.03.015. [DOI] [PubMed] [Google Scholar]
- 39.Sorrenti, V., Benedetti, F., Buriani, A., Fortinguerra, S., Caudullo, G., Davinelli, S., et al. (2022). Immunomodulatory and antiaging mechanisms of resveratrol, rapamycin, and metformin: Focus on mTOR and AMPK signaling networks. Pharmaceuticals, 15(8). 10.3390/ph15080912 [DOI] [PMC free article] [PubMed]
- 40.Hasan ZYM, Obed FAA, Jasim AA, Alwan AF. In vitro study of the effect of resveratrol purified from the skin of Iraqi black grape (Vitis vinifera) on lymphocyte cultures isolated from the blood of patients with lymphoma. Journal of Medicine and Life. 2022;15(6):778–783. doi: 10.25122/jml-2022-0038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Rojo, D., Madrid, A., Martín, S. S., Párraga, M., Silva Pinhal, M. A., Villena, J., et al. (2022). Resveratrol decreases the invasion potential of gastric cancer cells. Molecules, 27(10). 10.3390/molecules27103047 [DOI] [PMC free article] [PubMed]
- 42.Aggarwal BB, Bhardwaj A, Aggarwal RS, Seeram NP, Shishodia S, Takada Y. Role of resveratrol in prevention and therapy of cancer: Preclinical and clinical studies. Anticancer Research. 2004;24(5a):2783–2840. [PubMed] [Google Scholar]
- 43.Soleas GJ, Diamandis EP, Goldberg DM. Resveratrol: A molecule whose time has come? And gone? Clinical Biochemistry. 1997;30(2):91–113. doi: 10.1016/s0009-9120(96)00155-5. [DOI] [PubMed] [Google Scholar]
- 44.Khanna D, Sethi G, Ahn KS, Pandey MK, Kunnumakkara AB, Sung B, et al. Natural products as a gold mine for arthritis treatment. Current Opinion in Pharmacology. 2007;7(3):344–351. doi: 10.1016/j.coph.2007.03.002. [DOI] [PubMed] [Google Scholar]
- 45.Shakibaei M, Harikumar KB, Aggarwal BB. Resveratrol addiction: To die or not to die. Molecular Nutrition & Food Research. 2009;53(1):115–128. doi: 10.1002/mnfr.200800148. [DOI] [PubMed] [Google Scholar]
- 46.Takaoka M. Resveratrol, a new phenolic compound, from Veratrum grandiflorum. Nippon Kagaku Kaishi. 1939;60:1090–1100. doi: 10.1246/nikkashi1921.60.1090. [DOI] [Google Scholar]
- 47.Rupprich N, Kindl H. Stilbene synthases and stilbenecarboxylate synthases, I Enzymatic synthesis of 3,5,4-trihydroxystilbene from p-coumaroyl coenzyme A and malonyl coenzyme A. Hoppe-Seyler's Zeitschrift für Physiologische Chemie. 1978;359(2):165–172. [PubMed] [Google Scholar]
- 48.Schöppner A, Kindl H. Purification and properties of a stilbene synthase from induced cell suspension cultures of peanut. The Journal of Biological Chemistry. 1984;259(11):6806–6811. doi: 10.1016/S0021-9258(17)39799-5. [DOI] [PubMed] [Google Scholar]
- 49.Frémont L. Biological effects of resveratrol. Life Sciences. 2000;66(8):663–673. doi: 10.1016/s0024-3205(99)00410-5. [DOI] [PubMed] [Google Scholar]
- 50.Bertelli AA, Giovannini L, Bernini W, Migliori M, Fregoni M, Bavaresco L, et al. Antiplatelet activity of cis-resveratrol. Drugs under Experimental and Clinical Research. 1996;22(2):61–63. [PubMed] [Google Scholar]
- 51.Bhat KP, Pezzuto JM. Resveratrol exhibits cytostatic and antiestrogenic properties with human endometrial adenocarcinoma (Ishikawa) cells. Cancer Research. 2001;61(16):6137–6144. [PubMed] [Google Scholar]
- 52.Brockmueller, A., Shayan, P., & Shakibaei, M. (2022). Evidence That β1-Integrin Is Required for the Anti-Viability and Anti-Proliferative Effect of Resveratrol in CRC Cells. International Journal of Molecular Sciences, 23(9). 10.3390/ijms23094714 [DOI] [PMC free article] [PubMed]
- 53.Buhrmann C, Shayan P, Popper B, Goel A, Shakibaei M. Sirt1 is required for resveratrol-mediated chemopreventive effects in colorectal cancer cells. Nutrients. 2016;8(3):145. doi: 10.3390/nu8030145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ehala S, Vaher M, Kaljurand M. Characterization of phenolic profiles of Northern European berries by capillary electrophoresis and determination of their antioxidant activity. Journal of Agricultural and Food Chemistry. 2005;53(16):6484–6490. doi: 10.1021/jf050397w. [DOI] [PubMed] [Google Scholar]
- 55.Yin TF, Wang M, Qing Y, Lin YM, Wu D. Research progress on chemopreventive effects of phytochemicals on colorectal cancer and their mechanisms. World Journal of Gastroenterology. 2016;22(31):7058–7068. doi: 10.3748/wjg.v22.i31.7058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Burns J, Yokota T, Ashihara H, Lean ME, Crozier A. Plant foods and herbal sources of resveratrol. Journal of Agricultural and Food Chemistry. 2002;50(11):3337–3340. doi: 10.1021/jf0112973. [DOI] [PubMed] [Google Scholar]
- 57.Schubert R, Fischer R, Hain R, Schreier PH, Bahnweg G, Ernst D, et al. An ozone-responsive region of the grapevine resveratrol synthase promoter differs from the basal pathogen-responsive sequence. Plant Molecular Biology. 1997;34(3):417–426. doi: 10.1023/a:1005830714852. [DOI] [PubMed] [Google Scholar]
- 58.Soleas GJ, Yan J, Goldberg DM. Measurement of trans-resveratrol, (+)-catechin, and quercetin in rat and human blood and urine by gas chromatography with mass selective detection. Methods in Enzymology. 2001;335:130–145. doi: 10.1016/s0076-6879(01)35238-2. [DOI] [PubMed] [Google Scholar]
- 59.Artursson P, Karlsson J. Correlation between oral drug absorption in humans and apparent drug permeability coefficients in human intestinal epithelial (Caco-2) cells. Biochemical and Biophysical Research Communications. 1991;175(3):880–885. doi: 10.1016/0006-291x(91)91647-u. [DOI] [PubMed] [Google Scholar]
- 60.Marier JF, Vachon P, Gritsas A, Zhang J, Moreau JP, Ducharme MP. Metabolism and disposition of resveratrol in rats: extent of absorption, glucuronidation, and enterohepatic recirculation evidenced by a linked-rat model. The Journal of Pharmacology and Experimental Therapeutics. 2002;302(1):369–373. doi: 10.1124/jpet.102.033340. [DOI] [PubMed] [Google Scholar]
- 61.Walle T. Bioavailability of resveratrol. Annals of the New York Academy of Sciences. 2011;1215:9–15. doi: 10.1111/j.1749-6632.2010.05842.x. [DOI] [PubMed] [Google Scholar]
- 62.Vang O, Ahmad N, Baile CA, Baur JA, Brown K, Csiszar A, et al. What is new for an old molecule? Systematic review and recommendations on the use of resveratrol. PLoS One. 2011;6(6):e19881. doi: 10.1371/journal.pone.0019881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Sergides C, Chirilă M, Silvestro L, Pitta D, Pittas A. Bioavailability and safety study of resveratrol 500 mg tablets in healthy male and female volunteers. Experimental and Therapeutic Medicine. 2016;11(1):164–170. doi: 10.3892/etm.2015.2895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Alayev A, Salamon RS, Schwartz NS, Berman AY, Wiener SL, Holz MK. Combination of rapamycin and resveratrol for treatment of bladder cancer. Journal of Cellular Physiology. 2017;232(2):436–446. doi: 10.1002/jcp.25443. [DOI] [PubMed] [Google Scholar]
- 65.Novelle MG, Wahl D, Diéguez C, Bernier M, de Cabo R. Resveratrol supplementation: Where are we now and where should we go? Ageing Research Reviews. 2015;21:1–15. doi: 10.1016/j.arr.2015.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Gokce EH, Korkmaz E, Dellera E, Sandri G, Bonferoni MC, Ozer O. Resveratrol-loaded solid lipid nanoparticles versus nanostructured lipid carriers: evaluation of antioxidant potential for dermal applications. International Journal of Nanomedicine. 2012;7:1841–1850. doi: 10.2147/ijn.S29710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Peñalva, R., Morales, J., González-Navarro, C. J., Larrañeta, E., Quincoces, G., Peñuelas, I., et al. (2018). Increased Oral Bioavailability of Resveratrol by Its Encapsulation in Casein Nanoparticles. International Journal of Molecular Sciences, 19(9). 10.3390/ijms19092816 [DOI] [PMC free article] [PubMed]
- 68.Saleem Z, Rehman K, Hamid Akash MS. Role of Drug Delivery System in Improving the Bioavailability of Resveratrol. Current Pharmaceutical Design. 2022;28(20):1632–1642. doi: 10.2174/1381612828666220705113514. [DOI] [PubMed] [Google Scholar]
- 69.de Vries, K., Strydom, M., & Steenkamp, V. (2021). A brief updated review of advances to enhance resveratrol’s bioavailability. Molecules, 26(14). 10.3390/molecules26144367 [DOI] [PMC free article] [PubMed]
- 70.Frankenfeld CL, McTiernan A, Aiello EJ, Thomas WK, LaCroix K, Schramm J, et al. Mammographic density in relation to daidzein-metabolizing phenotypes in overweight, postmenopausal women. Cancer Epidemiology, Biomarkers & Prevention. 2004;13(7):1156–1162. doi: 10.1158/1055-9965.1156.13.7. [DOI] [PubMed] [Google Scholar]
- 71.Woting A, Clavel T, Loh G, Blaut M. Bacterial transformation of dietary lignans in gnotobiotic rats. FEMS Microbiology Ecology. 2010;72(3):507–514. doi: 10.1111/j.1574-6941.2010.00863.x. [DOI] [PubMed] [Google Scholar]
- 72.Kaldas MI, Walle UK, Walle T. Resveratrol transport and metabolism by human intestinal Caco-2 cells. The Journal of Pharmacy and Pharmacology. 2003;55(3):307–312. doi: 10.1211/002235702612. [DOI] [PubMed] [Google Scholar]
- 73.Zoberi I, Bradbury CM, Curry HA, Bisht KS, Goswami PC, Roti Roti JL, et al. Radiosensitizing and anti-proliferative effects of resveratrol in two human cervical tumor cell lines. Cancer Letters. 2002;175(2):165–173. doi: 10.1016/s0304-3835(01)00719-4. [DOI] [PubMed] [Google Scholar]
- 74.Xu J, Liu D, Niu H, Zhu G, Xu Y, Ye D, et al. Resveratrol reverses Doxorubicin resistance by inhibiting epithelial-mesenchymal transition (EMT) through modulating PTEN/Akt signaling pathway in gastric cancer. Journal of Experimental & Clinical Cancer Research. 2017;36(1):19. doi: 10.1186/s13046-016-0487-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Martínez-Martínez D, Soto A, Gil-Araujo B, Gallego B, Chiloeches A, Lasa M. Resveratrol promotes apoptosis through the induction of dual specificity phosphatase 1 and sensitizes prostate cancer cells to cisplatin. Food and Chemical Toxicology. 2019;124:273–279. doi: 10.1016/j.fct.2018.12.014. [DOI] [PubMed] [Google Scholar]
- 76.Benitez DA, Hermoso MA, Pozo-Guisado E, Fernández-Salguero PM, Castellón EA. Regulation of cell survival by resveratrol involves inhibition of NF kappa B-regulated gene expression in prostate cancer cells. Prostate. 2009;69(10):1045–1054. doi: 10.1002/pros.20953. [DOI] [PubMed] [Google Scholar]
- 77.Stewart JR, O’Brian CA. Resveratrol antagonizes EGFR-dependent Erk1/2 activation in human androgen-independent prostate cancer cells with associated isozyme-selective PKC alpha inhibition. Investigational New Drugs. 2004;22(2):107–117. doi: 10.1023/B:DRUG.0000011787.75522.ec. [DOI] [PubMed] [Google Scholar]
- 78.Bouyahya, A., Omari, N. E., Bakrim, S., Hachlafi, N. E., Balahbib, A., Wilairatana, P., et al. (2022). Advances in dietary phenolic compounds to improve chemosensitivity of anticancer drugs. Cancers, 14(19). 10.3390/cancers14194573 [DOI] [PMC free article] [PubMed]
- 79.Buhrmann C, Shayan P, Kraehe P, Popper B, Goel A, Shakibaei M. Resveratrol induces chemosensitization to 5-fluorouracil through up-regulation of intercellular junctions, Epithelial-to-mesenchymal transition and apoptosis in colorectal cancer. Biochemical Pharmacology. 2015;98(1):51–68. doi: 10.1016/j.bcp.2015.08.105. [DOI] [PubMed] [Google Scholar]
- 80.Buhrmann, C., Yazdi, M., Popper, B., Shayan, P., Goel, A., Aggarwal, B. B., et al. (2018). Resveratrol chemosensitizes TNF-β-induced survival of 5-FU-treated colorectal cancer cells. Nutrients, 10(7). 10.3390/nu10070888 [DOI] [PMC free article] [PubMed]
- 81.Izquierdo-Torres E, Hernández-Oliveras A, Meneses-Morales I, Rodríguez G, Fuentes-García G, Zarain-Herzberg Á. Resveratrol up-regulates ATP2A3 gene expression in breast cancer cell lines through epigenetic mechanisms. The International Journal of Biochemistry & Cell Biology. 2019;113:37–47. doi: 10.1016/j.biocel.2019.05.020. [DOI] [PubMed] [Google Scholar]
- 82.Kocabas, Ş., & Sanlier, N. (2021). A comprehensive overview of the complex relationship between epigenetics, bioactive components, cancer, and aging. Critical Reviews in Food Science and Nutrition, 1–13. 10.1080/10408398.2021.1986803 [DOI] [PubMed]
- 83.Martinez J, Moreno JJ. Effect of resveratrol, a natural polyphenolic compound, on reactive oxygen species and prostaglandin production. Biochemical Pharmacology. 2000;59(7):865–870. doi: 10.1016/s0006-2952(99)00380-9. [DOI] [PubMed] [Google Scholar]
- 84.Leonard SS, Xia C, Jiang BH, Stinefelt B, Klandorf H, Harris GK, et al. Resveratrol scavenges reactive oxygen species and effects radical-induced cellular responses. Biochemical and Biophysical Research Communications. 2003;309(4):1017–1026. doi: 10.1016/j.bbrc.2003.08.105. [DOI] [PubMed] [Google Scholar]
- 85.Cho J, Rho O, Junco J, Carbajal S, Siegel D, Slaga TJ, et al. Effect of combined treatment with ursolic acid and resveratrol on skin tumor promotion by 12-O-tetradecanoylphorbol-13-acetate. Cancer Prevention Research. 2015;8(9):817–825. doi: 10.1158/1940-6207.Capr-15-0098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Sengottuvelan M, Deeptha K, Nalini N. Resveratrol ameliorates DNA damage, prooxidant and antioxidant imbalance in 1,2-dimethylhydrazine induced rat colon carcinogenesis. Chemico-Biological Interactions. 2009;181(2):193–201. doi: 10.1016/j.cbi.2009.06.004. [DOI] [PubMed] [Google Scholar]
- 87.Yuan W, Zhang M, Wang C, Li B, Li L, Ye F, et al. Resveratrol Attenuates High-Fat Diet-Induced Hepatic Lipotoxicity by Upregulating Bmi-1 Expression. The Journal of Pharmacology and Experimental Therapeutics. 2022;381(2):96–105. doi: 10.1124/jpet.121.001018. [DOI] [PubMed] [Google Scholar]
- 88.Uenobe F, Nakamura S, Miyazawa M. Antimutagenic effect of resveratrol against Trp-P-1. Mutation Research. 1997;373(2):197–200. doi: 10.1016/s0027-5107(96)00191-1. [DOI] [PubMed] [Google Scholar]
- 89.Huderson AC, Myers JN, Niaz MS, Washington MK, Ramesh A. Chemoprevention of benzo(a)pyrene-induced colon polyps in ApcMin mice by resveratrol. The Journal of Nutritional Biochemistry. 2013;24(4):713–724. doi: 10.1016/j.jnutbio.2012.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Xu S, Sun F, Ren L, Yang H, Tian N, Peng S. Resveratrol controlled the fate of porcine pancreatic stem cells through the Wnt/β-catenin signaling pathway mediated by Sirt1. PLoS One. 2017;12(10):e0187159. doi: 10.1371/journal.pone.0187159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Buhrmann C, Yazdi M, Popper B, Shayan P, Goel A, Aggarwal BB, et al. Evidence that TNF-β induces proliferation in colorectal cancer cells and resveratrol can down-modulate it. Experimental Biology and Medicine. 2019;244(1):1–12. doi: 10.1177/1535370218824538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Chávez E, Reyes-Gordillo K, Segovia J, Shibayama M, Tsutsumi V, Vergara P, et al. Resveratrol prevents fibrosis, NF-kappaB activation and TGF-beta increases induced by chronic CCl4 treatment in rats. Journal of Applied Toxicology. 2008;28(1):35–43. doi: 10.1002/jat.1249. [DOI] [PubMed] [Google Scholar]
- 93.Ebrahim HA, Kamar SS, Haidara MA, Latif NSA, Ellatif MA, ShamsEldeen AM, et al. Association of resveratrol with the suppression of TNF-α/NF-kB/iNOS/HIF-1α axis-mediated fibrosis and systemic hypertension in thioacetamide-induced liver injury. Naunyn-Schmiedeberg's Archives of Pharmacology. 2022;395(9):1087–1095. doi: 10.1007/s00210-022-02264-w. [DOI] [PubMed] [Google Scholar]
- 94.Estrov Z, Shishodia S, Faderl S, Harris D, Van Q, Kantarjian HM, et al. Resveratrol blocks interleukin-1beta-induced activation of the nuclear transcription factor NF-kappaB, inhibits proliferation, causes S-phase arrest, and induces apoptosis of acute myeloid leukemia cells. Blood. 2003;102(3):987–995. doi: 10.1182/blood-2002-11-3550. [DOI] [PubMed] [Google Scholar]
- 95.Feng J, Liu S, Ma S, Zhao J, Zhang W, Qi W, et al. Protective effects of resveratrol on postmenopausal osteoporosis: Regulation of SIRT1-NF-κB signaling pathway. Acta Biochimica et Biophysica Sinica Shanghai. 2014;46(12):1024–1033. doi: 10.1093/abbs/gmu103. [DOI] [PubMed] [Google Scholar]
- 96.Li Y, Feng L, Li G, An J, Zhang S, Li J, et al. Resveratrol prevents ISO-induced myocardial remodeling associated with regulating polarization of macrophages through VEGF-B/AMPK/NF-kB pathway. International Immunopharmacology. 2020;84:106508. doi: 10.1016/j.intimp.2020.106508. [DOI] [PubMed] [Google Scholar]
- 97.Ren Z, Wang L, Cui J, Huoc Z, Xue J, Cui H, et al. Resveratrol inhibits NF-kB signaling through suppression of p65 and IkappaB kinase activities. Pharmazie. 2013;68(8):689–694. [PubMed] [Google Scholar]
- 98.Shang X, Lin K, Yu R, Zhu P, Zhang Y, Wang L, et al. Resveratrol protects the myocardium in sepsis by activating the phosphatidylinositol 3-kinases (PI3K)/AKT/mammalian target of rapamycin (mTOR) pathway and inhibiting the nuclear factor-κB (NF-κB) signaling Pathway. Medical Science Monitor. 2019;25:9290–9298. doi: 10.12659/msm.918369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Tang XL, Wang X, Fang G, Zhao YL, Yan J, Zhou Z, et al. Resveratrol ameliorates sevoflurane-induced cognitive impairment by activating the SIRT1/NF-κB pathway in neonatal mice. The Journal of Nutritional Biochemistry. 2021;90:108579. doi: 10.1016/j.jnutbio.2020.108579. [DOI] [PubMed] [Google Scholar]
- 100.Wang Z, Li X, Chen H, Han L, Ji X, Wang Q, et al. Resveratrol alleviates bleomycin-induced pulmonary fibrosis via suppressing HIF-1α and NF-κB expression. Aging. 2021;13(3):4605–4616. doi: 10.18632/aging.202420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Yi H, Zhang W, Cui ZM, Cui SY, Fan JB, Zhu XH, et al. Resveratrol alleviates the interleukin-1β-induced chondrocytes injury through the NF-κB signaling pathway. Journal of Orthopaedic Surgery and Research. 2020;15(1):424. doi: 10.1186/s13018-020-01944-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Zhao H, Mei X, Yang D, Tu G. Resveratrol inhibits inflammation after spinal cord injury via SIRT-1/NF-κB signaling pathway. Neuroscience Letters. 2021;762:136151. doi: 10.1016/j.neulet.2021.136151. [DOI] [PubMed] [Google Scholar]
- 103.Fei J, Qin X, Ma H, Zhang X, Wang H, Han J, et al. Resveratrol ameliorates deep vein thrombosis-induced inflammatory response through inhibiting HIF-1α/NLRP3 pathway. Inflammation. 2022;45(6):2268–2279. doi: 10.1007/s10753-022-01689-y. [DOI] [PubMed] [Google Scholar]
- 104.Kim DH, Hossain MA, Kim MY, Kim JA, Yoon JH, Suh HS, et al. A novel resveratrol analogue, HS-1793, inhibits hypoxia-induced HIF-1α and VEGF expression, and migration in human prostate cancer cells. International Journal of Oncology. 2013;43(6):1915–1924. doi: 10.3892/ijo.2013.2116. [DOI] [PubMed] [Google Scholar]
- 105.Kim DH, Sung B, Kim JA, Kang YJ, Hwang SY, Hwang NL, et al. HS-1793, a resveratrol analogue, downregulates the expression of hypoxia-induced HIF-1 and VEGF and inhibits tumor growth of human breast cancer cells in a nude mouse xenograft model. International Journal of Oncology. 2017;51(2):715–723. doi: 10.3892/ijo.2017.4058. [DOI] [PubMed] [Google Scholar]
- 106.Li X, Li J, Wang L, Li A, Qiu Z, Qi LW, et al. The role of metformin and resveratrol in the prevention of hypoxia-inducible factor 1α accumulation and fibrosis in hypoxic adipose tissue. British Journal of Pharmacology. 2016;173(12):2001–2015. doi: 10.1111/bph.13493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Ma Z, Zhang Y, Li Q, Xu M, Bai J, Wu S. Resveratrol improves alcoholic fatty liver disease by downregulating HIF-1α expression and mitochondrial ROS production. PLoS One. 2017;12(8):e0183426. doi: 10.1371/journal.pone.0183426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Park SY, Jeong KJ, Lee J, Yoon DS, Choi WS, Kim YK, et al. Hypoxia enhances LPA-induced HIF-1alpha and VEGF expression: their inhibition by resveratrol. Cancer Letters. 2007;258(1):63–69. doi: 10.1016/j.canlet.2007.08.011. [DOI] [PubMed] [Google Scholar]
- 109.Sha W, Liu M, Sun D, Qiu J, Xu B, Chen L, et al. Resveratrol ameliorated endothelial injury of thoracic aorta in diabetic mice and Gly-LDL-induced HUVECs through inhibiting TLR4/HIF-1α. Journal of Cellular and Molecular Medicine. 2021;25(13):6258–6270. doi: 10.1111/jcmm.16584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Srivani G, Behera SK, Dariya B, Aliya S, Alam A, Nagaraju GP. Resveratrol binds and inhibits transcription factor HIF-1α in pancreatic cancer. Experimental Cell Research. 2020;394(1):112126. doi: 10.1016/j.yexcr.2020.112126. [DOI] [PubMed] [Google Scholar]
- 111.Wang H, Jia R, Lv T, Wang M, He S, Zhang X. Resveratrol suppresses tumor progression via inhibiting STAT3/HIF-1α/VEGF pathway in an orthotopic rat model of non-small-cell lung cancer (NSCLC) Oncotargets and Therapy. 2020;13:7057–7063. doi: 10.2147/ott.S259016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Yan, C., Wang, Z., Liu, W., Pu, L., Li, R., Ai, C., et al. (2022). Resveratrol ameliorates high altitude hypoxia-induced osteoporosis by suppressing the ROS/HIF signaling pathway. Molecules, 27(17). 10.3390/molecules27175538 [DOI] [PMC free article] [PubMed]
- 113.Zhang M, Li W, Yu L, Wu S. The suppressive effect of resveratrol on HIF-1α and VEGF expression after warm ischemia and reperfusion in rat liver. PLoS One. 2014;9(10):e109589. doi: 10.1371/journal.pone.0109589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.El-Mowafy AM, White RE. Resveratrol inhibits MAPK activity and nuclear translocation in coronary artery smooth muscle: Reversal of endothelin-1 stimulatory effects. FEBS Letters. 1999;451(1):63–67. doi: 10.1016/s0014-5793(99)00541-4. [DOI] [PubMed] [Google Scholar]
- 115.Shakibaei M, Mobasheri A, Buhrmann C. Curcumin synergizes with resveratrol to stimulate the MAPK signaling pathway in human articular chondrocytes in vitro. Genes & Nutrition. 2011;6(2):171–179. doi: 10.1007/s12263-010-0179-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Shih A, Davis FB, Lin HY, Davis PJ. Resveratrol induces apoptosis in thyroid cancer cell lines via a MAPK- and p53-dependent mechanism. The Journal of Clinical Endocrinology and Metabolism. 2002;87(3):1223–1232. doi: 10.1210/jcem.87.3.8345. [DOI] [PubMed] [Google Scholar]
- 117.Wang J, He F, Chen L, Li Q, Jin S, Zheng H, et al. Resveratrol inhibits pulmonary fibrosis by regulating miR-21 through MAPK/AP-1 pathways. Biomedicine & Pharmacotherapy. 2018;105:37–44. doi: 10.1016/j.biopha.2018.05.104. [DOI] [PubMed] [Google Scholar]
- 118.Wang Y, Sun X, Han X, Sun J, Li L, Zhang D, et al. Resveratrol improves hepatic ischemia-reperfusion injury by inhibiting neutrophils via the ERK signaling pathway. Biomedicine & Pharmacotherapy. 2023;160:114358. doi: 10.1016/j.biopha.2023.114358. [DOI] [PubMed] [Google Scholar]
- 119.Woo JH, Lim JH, Kim YH, Suh SI, Min DS, Chang JS, et al. Resveratrol inhibits phorbol myristate acetate-induced matrix metalloproteinase-9 expression by inhibiting JNK and PKC delta signal transduction. Oncogene. 2004;23(10):1845–1853. doi: 10.1038/sj.onc.1207307. [DOI] [PubMed] [Google Scholar]
- 120.Manna SK, Mukhopadhyay A, Aggarwal BB. Resveratrol suppresses TNF-induced activation of nuclear transcription factors NF-kappa B, activator protein-1, and apoptosis: potential role of reactive oxygen intermediates and lipid peroxidation. Journal of Immunology. 2000;164(12):6509–6519. doi: 10.4049/jimmunol.164.12.6509. [DOI] [PubMed] [Google Scholar]
- 121.Surh YJ, Hurh YJ, Kang JY, Lee E, Kong G, Lee SJ. Resveratrol, an antioxidant present in red wine, induces apoptosis in human promyelocytic leukemia (HL-60) cells. Cancer Letters. 1999;140(1-2):1–10. doi: 10.1016/s0304-3835(99)00039-7. [DOI] [PubMed] [Google Scholar]
- 122.Baek SH, Ko JH, Lee H, Jung J, Kong M, Lee JW, et al. Resveratrol inhibits STAT3 signaling pathway through the induction of SOCS-1: Role in apoptosis induction and radiosensitization in head and neck tumor cells. Phytomedicine. 2016;23(5):566–577. doi: 10.1016/j.phymed.2016.02.011. [DOI] [PubMed] [Google Scholar]
- 123.Li D, Wang G, Jin G, Yao K, Zhao Z, Bie L, et al. Resveratrol suppresses colon cancer growth by targeting the AKT/STAT3 signaling pathway. International Journal of Molecular Medicine. 2019;43(1):630–640. doi: 10.3892/ijmm.2018.3969. [DOI] [PubMed] [Google Scholar]
- 124.Peng L, Jiang D. Resveratrol eliminates cancer stem cells of osteosarcoma by STAT3 pathway inhibition. PLoS One. 2018;13(10):e0205918. doi: 10.1371/journal.pone.0205918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Dai H, Deng HB, Wang YH, Guo JJ. Resveratrol inhibits the growth of gastric cancer via the Wnt/β-catenin pathway. Oncology Letters. 2018;16(2):1579–1583. doi: 10.3892/ol.2018.8772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Geng W, Guo X, Zhang L, Ma Y, Wang L, Liu Z, et al. Resveratrol inhibits proliferation, migration and invasion of multiple myeloma cells via NEAT1-mediated Wnt/β-catenin signaling pathway. Biomedicine & Pharmacotherapy. 2018;107:484–494. doi: 10.1016/j.biopha.2018.08.003. [DOI] [PubMed] [Google Scholar]
- 127.Genovese, T., Impellizzeri, D., D’Amico, R., Cordaro, M., Peritore, A. F., Crupi, R., et al. (2022). Resveratrol inhibition of the WNT/β-catenin pathway following discogenic low back pain. International Journal of Molecular Sciences, 23(8). 10.3390/ijms23084092 [DOI] [PMC free article] [PubMed]
- 128.Jang, J., Song, J., Lee, J., Moon, S. K., & Moon, B. (2021). Resveratrol attenuates the proliferation of prostatic stromal cells in benign prostatic hyperplasia by regulating cell cycle progression, apoptosis, signaling pathways, BPH markers, and NF- κB Activity. International Journal of Molecular Sciences, 22(11). 10.3390/ijms22115969 [DOI] [PMC free article] [PubMed]
- 129.Qin, S., Geng, H., Wang, G., Chen, L., Xia, C., Yao, J., et al. (2022). Suffruticosol C-mediated autophagy and cell cycle arrest via inhibition of mTORC1 signaling. Nutrients, 14(23). 10.3390/nu14235000 [DOI] [PMC free article] [PubMed]
- 130.Jin Z, Feng W, Ji Y, Jin L. Resveratrol mediates cell cycle arrest and cell death in human esophageal squamous cell carcinoma by directly targeting the EGFR signaling pathway. Oncology Letters. 2017;13(1):347–355. doi: 10.3892/ol.2016.5391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Vanamala J, Reddivari L, Radhakrishnan S, Tarver C. Resveratrol suppresses IGF-1 induced human colon cancer cell proliferation and elevates apoptosis via suppression of IGF-1R/Wnt and activation of p53 signaling pathways. BMC Cancer. 2010;10:238. doi: 10.1186/1471-2407-10-238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.George J, Nihal M, Singh CK, Ahmad N. 4’-Bromo-resveratrol, a dual Sirtuin-1 and Sirtuin-3 inhibitor, inhibits melanoma cell growth through mitochondrial metabolic reprogramming. Molecular Carcinogenesis. 2019;58(10):1876–1885. doi: 10.1002/mc.23080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Li L, Qiu RL, Lin Y, Cai Y, Bian Y, Fan Y, et al. Resveratrol suppresses human cervical carcinoma cell proliferation and elevates apoptosis via the mitochondrial and p53 signaling pathways. Oncology Letters. 2018;15(6):9845–9851. doi: 10.3892/ol.2018.8571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Baek SJ, Wilson LC, Eling TE. Resveratrol enhances the expression of non-steroidal anti-inflammatory drug-activated gene (NAG-1) by increasing the expression of p53. Carcinogenesis. 2002;23(3):425–434. doi: 10.1093/carcin/23.3.425. [DOI] [PubMed] [Google Scholar]
- 135.Li, X., Lin, F., Wu, Y., Liu, N., Wang, J., Chen, R., et al. (2019). Resveratrol attenuates inflammation environment-induced nucleus pulposus cell senescence in vitro. Bioscience Reports, 39(5). 10.1042/bsr20190126 [DOI] [PMC free article] [PubMed] [Retracted]
- 136.Jalili C, Farzaei MH, Roshankhah S, Salahshoor MR. Resveratrol attenuates malathion-induced liver damage by reducing oxidative stress. Journal of Laboratory Physicians. 2019;11(3):212–219. doi: 10.4103/jlp.Jlp_43_19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Ovesná Z, Kozics K, Bader Y, Saiko P, Handler N, Erker T, et al. Antioxidant activity of resveratrol, piceatannol and 3,3’,4,4’,5,5’-hexahydroxy-trans-stilbene in three leukemia cell lines. Oncology Reports. 2006;16(3):617–624. [PubMed] [Google Scholar]
- 138.Fidrus, E., Hegedűs, C., Janka, E. A., Paragh, G., Emri, G., & Remenyik, É. (2021). Inhibitors of nucleotide excision repair decrease UVB-induced mutagenesis-an in vitro study. International Journal of Molecular Sciences, 22(4). 10.3390/ijms22041638 [DOI] [PMC free article] [PubMed]
- 139.Thiel G, Rössler OG. Resveratrol stimulates c-Fos gene transcription via activation of ERK1/2 involving multiple genetic elements. Gene. 2018;658:70–75. doi: 10.1016/j.gene.2018.03.008. [DOI] [PubMed] [Google Scholar]
- 140.Hsieh TC, Juan G, Darzynkiewicz Z, Wu JM. Resveratrol increases nitric oxide synthase, induces accumulation of p53 and p21(WAF1/CIP1), and suppresses cultured bovine pulmonary artery endothelial cell proliferation by perturbing progression through S and G2. Cancer Research. 1999;59(11):2596–2601. [PubMed] [Google Scholar]
- 141.Jang YG, Go RE, Hwang KA, Choi KC. Resveratrol inhibits DHT-induced progression of prostate cancer cell line through interfering with the AR and CXCR4 pathway. The Journal of Steroid Biochemistry and Molecular Biology. 2019;192:105406. doi: 10.1016/j.jsbmb.2019.105406. [DOI] [PubMed] [Google Scholar]
- 142.Li T, Zhang X, Cheng L, Li C, Wu Z, Luo Y, et al. Modulation of lncRNA H19 enhances resveratrol-inhibited cancer cell proliferation and migration by regulating endoplasmic reticulum stress. Journal of Cellular and Molecular Medicine. 2022;26(8):2205–2217. doi: 10.1111/jcmm.17242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Doherty, M. R., Smigiel, J. M., Junk, D. J., & Jackson, M. W. (2016). Cancer Stem Cell Plasticity Drives Therapeutic Resistance. Cancers, 8(1). 10.3390/cancers8010008 [DOI] [PMC free article] [PubMed]
- 144.Qin S, Jiang J, Lu Y, Nice EC, Huang C, Zhang J, et al. Emerging role of tumor cell plasticity in modifying therapeutic response. Signal Transduction and Targeted Therapy. 2020;5(1):228. doi: 10.1038/s41392-020-00313-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Yuan S, Norgard RJ, Stanger BZ. Cellular plasticity in cancer. Cancer Discovery. 2019;9(7):837–851. doi: 10.1158/2159-8290.cd-19-0015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Bhat KP, Lantvit D, Christov K, Mehta RG, Moon RC, Pezzuto JM. Estrogenic and antiestrogenic properties of resveratrol in mammary tumor models. Cancer Research. 2001;61(20):7456–7463. [PubMed] [Google Scholar]
- 147.Lu R, Serrero G. Resveratrol, a natural product derived from grape, exhibits antiestrogenic activity and inhibits the growth of human breast cancer cells. Journal of Cellular Physiology. 1999;179(3):297–304. doi: 10.1002/(SICI)1097-4652(199906)179:3<297::AID-JCP7>3.0.CO;2-P. [DOI] [PubMed] [Google Scholar]
- 148.Khera A, Kanta P, Kalra J, Dumir D, M, T. Resveratrol restores the level of key inflammatory cytokines and RANKL/OPG ratio in the femur of rat osteoporosis model. Journal of Women & Aging. 2019;31(6):540–552. doi: 10.1080/08952841.2018.1522126. [DOI] [PubMed] [Google Scholar]
- 149.Niwano, Y., Kohzaki, H., Shirato, M., Shishido, S., & Nakamura, K. (2022). Anti-osteoporotic mechanisms of polyphenols elucidated based on in vivo studies using ovariectomized animals. Antioxidants, 11(2). 10.3390/antiox11020217 [DOI] [PMC free article] [PubMed]
- 150.Ren Z, Raut NA, Lawal TO, Patel SR, Lee SM, Mahady GB. Peonidin-3-O-glucoside and cyanidin increase osteoblast differentiation and reduce RANKL-induced bone resorption in transgenic medaka. Phytotherapy Research. 2021;35(11):6255–6269. doi: 10.1002/ptr.7271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Shakibaei M, Buhrmann C, Mobasheri A. Resveratrol-mediated SIRT-1 interactions with p300 modulate receptor activator of NF-kappaB ligand (RANKL) activation of NF-kappaB signaling and inhibit osteoclastogenesis in bone-derived cells. The Journal of Biological Chemistry. 2011;286(13):11492–11505. doi: 10.1074/jbc.M110.198713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Voronov I, Heersche JN, Casper RF, Tenenbaum HC, Manolson MF. Inhibition of osteoclast differentiation by polycyclic aryl hydrocarbons is dependent on cell density and RANKL concentration. Biochemical Pharmacology. 2005;70(2):300–307. doi: 10.1016/j.bcp.2005.04.028. [DOI] [PubMed] [Google Scholar]
- 153.Baarine M, Thandapilly SJ, Louis XL, Mazué F, Yu L, Delmas D, et al. Pro-apoptotic versus anti-apoptotic properties of dietary resveratrol on tumoral and normal cardiac cells. Genes & Nutrition. 2011;6(2):161–169. doi: 10.1007/s12263-011-0232-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Buhrmann C, Busch F, Shayan P, Shakibaei M. Sirtuin-1 (SIRT1) is required for promoting chondrogenic differentiation of mesenchymal stem cells. The Journal of Biological Chemistry. 2014;289(32):22048–22062. doi: 10.1074/jbc.M114.568790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Buhrmann C, Popper B, Aggarwal BB, Shakibaei M. Resveratrol downregulates inflammatory pathway activated by lymphotoxin α (TNF-β) in articular chondrocytes: Comparison with TNF-α. PLoS One. 2017;12(11):e0186993. doi: 10.1371/journal.pone.0186993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Busch F, Mobasheri A, Shayan P, Stahlmann R, Shakibaei M. Sirt-1 is required for the inhibition of apoptosis and inflammatory responses in human tenocytes. The Journal of Biological Chemistry. 2012;287(31):25770–25781. doi: 10.1074/jbc.M112.355420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Ma P, Yue L, Yang H, Fan Y, Bai J, Li S, et al. Chondroprotective and anti-inflammatory effects of amurensin H by regulating TLR4/Syk/NF-κB signals. Journal of Cellular and Molecular Medicine. 2020;24(2):1958–1968. doi: 10.1111/jcmm.14893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Wu CC, Huang YS, Chen JS, Huang CF, Su SL, Lu KC, et al. Resveratrol ameliorates renal damage, increases expression of heme oxygenase-1, and has anti-complement, anti-oxidative, and anti-apoptotic effects in a murine model of membranous nephropathy. PLoS One. 2015;10(5):e0125726. doi: 10.1371/journal.pone.0125726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Zhang L, Guo X, Xie W, Li Y, Ma M, Yuan T, et al. Resveratrol exerts an anti-apoptotic effect on human bronchial epithelial cells undergoing cigarette smoke exposure. Molecular Medicine Reports. 2015;11(3):1752–1758. doi: 10.3892/mmr.2014.2925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Csaki C, Mobasheri A, Shakibaei M. Synergistic chondroprotective effects of curcumin and resveratrol in human articular chondrocytes: Inhibition of IL-1beta-induced NF-kappaB-mediated inflammation and apoptosis. Arthritis Research & Therapy. 2009;11(6):R165. doi: 10.1186/ar2850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Liang C, Xing H, Wang C, Xu X, Hao Y, Qiu B. Resveratrol protection against IL-1β-induced chondrocyte damage via the SIRT1/FOXO1 signaling pathway. Journal of Orthopaedic Surgery and Research. 2022;17(1):406. doi: 10.1186/s13018-022-03306-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Wu G, Wang L, Li H, Ke Y, Yao Y. Function of sustained released resveratrol on IL-1β-induced hBMSC MMP13 secretion inhibition and chondrogenic differentiation promotion. Journal of Biomaterials Applications. 2016;30(7):930–939. doi: 10.1177/0885328215614425. [DOI] [PubMed] [Google Scholar]
- 163.Busch F, Mobasheri A, Shayan P, Lueders C, Stahlmann R, Shakibaei M. Resveratrol modulates interleukin-1β-induced phosphatidylinositol 3-kinase and nuclear factor κB signaling pathways in human tenocytes. The Journal of Biological Chemistry. 2012;287(45):38050–38063. doi: 10.1074/jbc.M112.377028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Constanze B, Popper B, Aggarwal BB, Shakibaei M. Evidence that TNF-β suppresses osteoblast differentiation of mesenchymal stem cells and resveratrol reverses it through modulation of NF-κB, Sirt1 and Runx2. Cell and Tissue Research. 2020;381(1):83–98. doi: 10.1007/s00441-020-03188-8. [DOI] [PubMed] [Google Scholar]
- 165.Fang WJ, Li XM, Zhou XK, Xiong Y. Resveratrol improves diabetic cardiomyopathy by preventing asymmetric dimethylarginine-caused peroxisome proliferator-activated receptor-γ coactivator-1α acetylation. European Journal of Pharmacology. 2022;936:175342. doi: 10.1016/j.ejphar.2022.175342. [DOI] [PubMed] [Google Scholar]
- 166.Huang X, Wang Y, Qiu Y, Shi Q, Sun D, Yang J, et al. Resveratrol ameliorates high-phosphate-induced VSMCs to osteoblast-like cells transdifferentiation and arterial medial calcification in CKD through regulating Wnt/β-catenin signaling. European Journal of Pharmacology. 2022;925:174953. doi: 10.1016/j.ejphar.2022.174953. [DOI] [PubMed] [Google Scholar]
- 167.Kim HJ, Braun HJ, Dragoo JL. The effect of resveratrol on normal and osteoarthritic chondrocyte metabolism. Bone & Joint Research. 2014;3(3):51–59. doi: 10.1302/2046-3758.33.2000226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Shakibaei M, Shayan P, Busch F, Aldinger C, Buhrmann C, Lueders C, et al. Resveratrol mediated modulation of Sirt-1/Runx2 promotes osteogenic differentiation of mesenchymal stem cells: Potential role of Runx2 deacetylation. PLoS One. 2012;7(4):e35712. doi: 10.1371/journal.pone.0035712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Tseng PC, Hou SM, Chen RJ, Peng HW, Hsieh CF, Kuo ML, et al. Resveratrol promotes osteogenesis of human mesenchymal stem cells by upregulating RUNX2 gene expression via the SIRT1/FOXO3A axis. Journal of Bone and Mineral Research. 2011;26(10):2552–2563. doi: 10.1002/jbmr.460. [DOI] [PubMed] [Google Scholar]
- 170.Zou J, Du J, Tu H, Chen H, Cong K, Bi Z, et al. Resveratrol benefits the lineage commitment of bone marrow mesenchymal stem cells into osteoblasts via miR-320c by targeting Runx2. Journal of Tissue Engineering and Regenerative Medicine. 2021;15(4):347–360. doi: 10.1002/term.3176. [DOI] [PubMed] [Google Scholar]
- 171.Huang N, Zhang Y, Chen M, Jin H, Nie J, Luo Y, et al. Resveratrol delays 6-hydroxydopamine-induced apoptosis by activating the PI3K/Akt signaling pathway. Experimental Gerontology. 2019;124:110653. doi: 10.1016/j.exger.2019.110653. [DOI] [PubMed] [Google Scholar]
- 172.Liu Y, Wu H, Zhang F, Yang J, He J. Resveratrol upregulates miR-455-5p to antagonize cisplatin ototoxicity via modulating the PTEN-PI3K-AKT axis. Biochemistry and Cell Biology. 2021;99(3):385–395. doi: 10.1139/bcb-2020-0459. [DOI] [PubMed] [Google Scholar]
- 173.Xu, W., Zheng, H., Fu, Y., Gu, Y., Zou, H., Yuan, Y., et al. (2022). Role of PI3K/Akt-mediated Nrf2/HO-1 signaling pathway in resveratrol alleviation of zearalenone-induced oxidative stress and apoptosis in TM4 Cells. Toxins, 14(11). 10.3390/toxins14110733 [DOI] [PMC free article] [PubMed]
- 174.Lu J, Ho CH, Ghai G, Chen KY. Resveratrol analog, 3,4,5,4’-tetrahydroxystilbene, differentially induces pro-apoptotic p53/Bax gene expression and inhibits the growth of transformed cells but not their normal counterparts. Carcinogenesis. 2001;22(2):321–328. doi: 10.1093/carcin/22.2.321. [DOI] [PubMed] [Google Scholar]
- 175.Singh SK, Banerjee S, Acosta EP, Lillard JW, Singh R. Resveratrol induces cell cycle arrest and apoptosis with docetaxel in prostate cancer cells via a p53/ p21WAF1/CIP1 and p27KIP1 pathway. Oncotarget. 2017;8(10):17216–17228. doi: 10.18632/oncotarget.15303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Fan Y, Chiu JF, Liu J, Deng Y, Xu C, Zhang J, et al. Resveratrol induces autophagy-dependent apoptosis in HL-60 cells. BMC Cancer. 2018;18(1):581. doi: 10.1186/s12885-018-4504-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Selvaraj S, Sun Y, Sukumaran P, Singh BB. Resveratrol activates autophagic cell death in prostate cancer cells via downregulation of STIM1 and the mTOR pathway. Molecular Carcinogenesis. 2016;55(5):818–831. doi: 10.1002/mc.22324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Kotha A, Sekharam M, Cilenti L, Siddiquee K, Khaled A, Zervos AS, et al. Resveratrol inhibits Src and Stat3 signaling and induces the apoptosis of malignant cells containing activated Stat3 protein. Molecular Cancer Therapeutics. 2006;5(3):621–629. doi: 10.1158/1535-7163.Mct-05-0268. [DOI] [PubMed] [Google Scholar]
- 179.Vanamala J, Radhakrishnan S, Reddivari L, Bhat VB, Ptitsyn A. Resveratrol suppresses human colon cancer cell proliferation and induces apoptosis via targeting the pentose phosphate and the talin-FAK signaling pathways-A proteomic approach. Proteome Science. 2011;9(1):49. doi: 10.1186/1477-5956-9-49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Ali D, Chen L, Kowal JM, Okla M, Manikandan M, AlShehri M, et al. Resveratrol inhibits adipocyte differentiation and cellular senescence of human bone marrow stromal stem cells. Bone. 2020;133:115252. doi: 10.1016/j.bone.2020.115252. [DOI] [PubMed] [Google Scholar]
- 181.Ding H, Xu X, Qin X, Yang C, Feng Q. Resveratrol promotes differentiation of mouse embryonic stem cells to cardiomyocytes. Cardiovascular Therapeutics. 2016;34(4):283–289. doi: 10.1111/1755-5922.12200. [DOI] [PubMed] [Google Scholar]
- 182.Jiang Y, Luo W, Wang B, Wang X, Gong P, Xiong Y. Resveratrol promotes osteogenesis via activating SIRT1/FoxO1 pathway in osteoporosis mice. Life Sciences. 2020;246:117422. doi: 10.1016/j.lfs.2020.117422. [DOI] [PubMed] [Google Scholar]
- 183.Moon DK, Kim BG, Lee AR, In Choe Y, Khan I, Moon KM, et al. Resveratrol can enhance osteogenic differentiation and mitochondrial biogenesis from human periosteum-derived mesenchymal stem cells. Journal of Orthopaedic Surgery and Research. 2020;15(1):203. doi: 10.1186/s13018-020-01684-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Yao Y, Zhou R, Bai R, Wang J, Tu M, Shi J, et al. Resveratrol promotes the survival and neuronal differentiation of hypoxia-conditioned neuronal progenitor cells in rats with cerebral ischemia. Frontiers in Medicine. 2021;15(3):472–485. doi: 10.1007/s11684-021-0832-y. [DOI] [PubMed] [Google Scholar]
- 185.Reuter S, Gupta SC, Chaturvedi MM, Aggarwal BB. Oxidative stress, inflammation, and cancer: how are they linked? Free Radical Biology & Medicine. 2010;49(11):1603–1616. doi: 10.1016/j.freeradbiomed.2010.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Khalyfa, A. A., Punatar, S., Aslam, R., & Yarbrough, A. (2021). Exploring the Inflammatory Pathogenesis of Colorectal Cancer. Diseases, 9(4). 10.3390/diseases9040079 [DOI] [PMC free article] [PubMed]
- 187.Beck PL, Wallace JL. Cytokines in inflammatory bowel disease. Mediators of Inflammation. 1997;6(2):95–103. doi: 10.1080/09629359791785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Feghali CA, Wright TM. Cytokines in acute and chronic inflammation. Frontiers in Bioscience. 1997;2:d12–d26. doi: 10.2741/a171. [DOI] [PubMed] [Google Scholar]
- 189.Kolios G, Brown Z, Robson RL, Robertson DA, Westwick J. Inducible nitric oxide synthase activity and expression in a human colonic epithelial cell line, HT-29. British Journal of Pharmacology. 1995;116(7):2866–2872. doi: 10.1111/j.1476-5381.1995.tb15938.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Liang YC, Huang YT, Tsai SH, Lin-Shiau SY, Chen CF, Lin JK. Suppression of inducible cyclooxygenase and inducible nitric oxide synthase by apigenin and related flavonoids in mouse macrophages. Carcinogenesis. 1999;20(10):1945–1952. doi: 10.1093/carcin/20.10.1945. [DOI] [PubMed] [Google Scholar]
- 191.Zhang M, Xue Y, Chen H, Meng L, Chen B, Gong H, et al. Resveratrol inhibits MMP3 and MMP9 expression and secretion by suppressing TLR4/NF-κB/STAT3 activation in Ox-LDL-treated HUVECs. Oxidative Medicine and Cellular Longevity. 2019;2019:9013169. doi: 10.1155/2019/9013169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Sánchez-Fidalgo S, Cárdeno A, Villegas I, Talero E, de la Lastra CA. Dietary supplementation of resveratrol attenuates chronic colonic inflammation in mice. European Journal of Pharmacology. 2010;633(1-3):78–84. doi: 10.1016/j.ejphar.2010.01.025. [DOI] [PubMed] [Google Scholar]
- 193.Prasad S, Sung B, Aggarwal BB. Age-associated chronic diseases require age-old medicine: Role of chronic inflammation. Preventive Medicine. 2012;54(Supplement):S29–S37. doi: 10.1016/j.ypmed.2011.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Garg A, Aggarwal BB. Nuclear transcription factor-kappaB as a target for cancer drug development. Leukemia. 2002;16(6):1053–1068. doi: 10.1038/sj.leu.2402482. [DOI] [PubMed] [Google Scholar]
- 195.Lin Y, He Z, Ye J, Liu Z, She X, Gao X, et al. Progress in Understanding the IL-6/STAT3 Pathway in Colorectal Cancer. Oncotargets and Therapy. 2020;13:13023–13032. doi: 10.2147/ott.S278013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Ji Q, Liu X, Han Z, Zhou L, Sui H, Yan L, et al. Resveratrol suppresses epithelial-to-mesenchymal transition in colorectal cancer through TGF-β1/Smads signaling pathway mediated Snail/E-cadherin expression. BMC Cancer. 2015;15:97. doi: 10.1186/s12885-015-1119-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Sheng J, Sun H, Yu FB, Li B, Zhang Y, Zhu YT. The role of cyclooxygenase-2 in colorectal cancer. International Journal of Medical Sciences. 2020;17(8):1095–1101. doi: 10.7150/ijms.44439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Hirano, T., Hirayama, D., Wagatsuma, K., Yamakawa, T., Yokoyama, Y., & Nakase, H. (2020). Immunological Mechanisms in Inflammation-Associated Colon Carcinogenesis. International Journal of Molecular Sciences, 21(9). 10.3390/ijms21093062 [DOI] [PMC free article] [PubMed]
- 199.Buhrmann, C., Popper, B., Kunnumakkara, A. B., Aggarwal, B. B., & Shakibaei, M. (2019). Evidence that calebin A, a component of curcuma longa suppresses NF-B mediated proliferation, invasion and metastasis of human colorectal cancer induced by TNF-β (lymphotoxin). Nutrients, 11(12). 10.3390/nu11122904 [DOI] [PMC free article] [PubMed]
- 200.Chaturvedi MM, Kumar A, Darnay BG, Chainy GB, Agarwal S, Aggarwal BB. Sanguinarine (pseudochelerythrine) is a potent inhibitor of NF-kappaB activation, IkappaBalpha phosphorylation, and degradation. The Journal of Biological Chemistry. 1997;272(48):30129–30134. doi: 10.1074/jbc.272.48.30129. [DOI] [PubMed] [Google Scholar]
- 201.Kumar A, Dhawan S, Aggarwal BB. Emodin (3-methyl-1,6,8-trihydroxyanthraquinone) inhibits TNF-induced NF-kappaB activation, IkappaB degradation, and expression of cell surface adhesion proteins in human vascular endothelial cells. Oncogene. 1998;17(7):913–918. doi: 10.1038/sj.onc.1201998. [DOI] [PubMed] [Google Scholar]
- 202.Manna SK, Sah NK, Newman RA, Cisneros A, Aggarwal BB. Oleandrin suppresses activation of nuclear transcription factor-kappaB, activator protein-1, and c-Jun NH2-terminal kinase. Cancer Research. 2000;60(14):3838–3847. [PubMed] [Google Scholar]
- 203.Mueller, A. L., Brockmueller, A., Kunnumakkara, A. B., & Shakibaei, M. (2022). Calebin A, a compound of turmeric, down-regulates inflammation in tenocytes by NF-κB/scleraxis signaling. International Journal of Molecular Sciences, 23(3). 10.3390/ijms23031695 [DOI] [PMC free article] [PubMed]
- 204.Singh S, Aggarwal BB. Activation of transcription factor NF-kappa B is suppressed by curcumin (diferuloylmethane) [corrected] The Journal of Biological Chemistry. 1995;270(42):24995–25000. doi: 10.1074/jbc.270.42.24995. [DOI] [PubMed] [Google Scholar]
- 205.Surh YJ. Cancer chemoprevention with dietary phytochemicals. Nature Reviews Cancer. 2003;3(10):768–780. doi: 10.1038/nrc1189. [DOI] [PubMed] [Google Scholar]
- 206.Dariya B, Behera SK, Srivani G, Farran B, Alam A, Nagaraju GP. Computational analysis of nuclear factor-κB and resveratrol in colorectal cancer. Journal of Biomolecular Structure & Dynamics. 2021;39(8):2914–2922. doi: 10.1080/07391102.2020.1757511. [DOI] [PubMed] [Google Scholar]
- 207.Buhrmann, C., Yazdi, M., Popper, B., Kunnumakkara, A. B., Aggarwal, B. B., & Shakibaei, M. (2019). Induction of the epithelial-to-mesenchymal transition of human colorectal cancer by human TNF-β (lymphotoxin) and its reversal by resveratrol. Nutrients, 11(3). 10.3390/nu11030704 [DOI] [PMC free article] [PubMed]
- 208.Youn J, Lee JS, Na HK, Kundu JK, Surh YJ. Resveratrol and piceatannol inhibit iNOS expression and NF-kappaB activation in dextran sulfate sodium-induced mouse colitis. Nutrition and Cancer. 2009;61(6):847–854. doi: 10.1080/01635580903285072. [DOI] [PubMed] [Google Scholar]
- 209.Chung SS, Dutta P, Austin D, Wang P, Awad A, Vadgama JV. Combination of resveratrol and 5-flurouracil enhanced anti-telomerase activity and apoptosis by inhibiting STAT3 and Akt signaling pathways in human colorectal cancer cells. Oncotarget. 2018;9(68):32943–32957. doi: 10.18632/oncotarget.25993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Santandreu FM, Valle A, Oliver J, Roca P. Resveratrol potentiates the cytotoxic oxidative stress induced by chemotherapy in human colon cancer cells. Cellular Physiology and Biochemistry. 2011;28(2):219–228. doi: 10.1159/000331733. [DOI] [PubMed] [Google Scholar]
- 211.Gong WH, Zhao N, Zhang ZM, Zhang YX, Yan L, Li JB. The inhibitory effect of resveratrol on COX-2 expression in human colorectal cancer: A promising therapeutic strategy. European Review for Medical and Pharmacological Sciences. 2017;21(5):1136–1143. [PubMed] [Google Scholar]
- 212.Serra D, Rufino AT, Mendes AF, Almeida LM, Dinis TC. Resveratrol modulates cytokine-induced Jak/STAT activation more efficiently than 5-aminosalicylic acid: An in vitro approach. PLoS One. 2014;9(10):e109048. doi: 10.1371/journal.pone.0109048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Hu, W. H., Chan, G. K., Duan, R., Wang, H. Y., Kong, X. P., Dong, T. T., et al. (2019). Synergy of Ginkgetin and Resveratrol in Suppressing VEGF-Induced Angiogenesis: A Therapy in Treating Colorectal Cancer. Cancers, 11(12). 10.3390/cancers11121828 [DOI] [PMC free article] [PubMed]
- 214.Jacques, C., Marchesi, I., Fiorentino, F. P., Chatelais, M., Lilli, N. L., Appel, K., et al. (2022). A micro-immunotherapy sequential medicine MIM-seq displays immunomodulatory effects on human macrophages and anti-tumor properties towards in vitro 2D and 3D models of colon carcinoma and in an in vivo subcutaneous xenograft colon carcinoma model. International Journal of Molecular Sciences, 23(11). 10.3390/ijms23116059 [DOI] [PMC free article] [PubMed]
- 215.Martín AR, Villegas I, La Casa C, de la Lastra CA. Resveratrol, a polyphenol found in grapes, suppresses oxidative damage and stimulates apoptosis during early colonic inflammation in rats. Biochemical Pharmacology. 2004;67(7):1399–1410. doi: 10.1016/j.bcp.2003.12.024. [DOI] [PubMed] [Google Scholar]
- 216.Alrafas, H. R., Busbee, P. B., Chitrala, K. N., Nagarkatti, M., & Nagarkatti, P. (2020). Alterations in the gut microbiome and suppression of histone deacetylases by resveratrol are associated with attenuation of colonic inflammation and protection against colorectal cancer. Journal of Clinical Medicine, 9(6). 10.3390/jcm9061796 [DOI] [PMC free article] [PubMed]
- 217.Fuggetta MP, Lanzilli G, Tricarico M, Cottarelli A, Falchetti R, Ravagnan G, et al. Effect of resveratrol on proliferation and telomerase activity of human colon cancer cells in vitro. Journal of Experimental & Clinical Cancer Research. 2006;25(2):189–193. [PubMed] [Google Scholar]
- 218.Mahyar-Roemer M, Katsen A, Mestres P, Roemer K. Resveratrol induces colon tumor cell apoptosis independently of p53 and precede by epithelial differentiation, mitochondrial proliferation and membrane potential collapse. International Journal of Cancer. 2001;94(5):615–622. doi: 10.1002/ijc.1516. [DOI] [PubMed] [Google Scholar]
- 219.Feng M, Zhong LX, Zhan ZY, Huang ZH, Xiong JP. Resveratrol treatment inhibits proliferation of and induces apoptosis in human colon cancer cells. Medical Science Monitor. 2016;22:1101–1108. doi: 10.12659/msm.897905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Liu B, Zhou Z, Zhou W, Liu J, Zhang Q, Xia J, et al. Resveratrol inhibits proliferation in human colorectal carcinoma cells by inducing G1/S-phase cell cycle arrest and apoptosis through caspase/cyclin-CDK pathways. Molecular Medicine Reports. 2014;10(4):1697–1702. doi: 10.3892/mmr.2014.2406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Liu Z, Wu X, Lv J, Sun H, Zhou F. Resveratrol induces p53 in colorectal cancer through SET7/9. Oncology Letters. 2019;17(4):3783–3789. doi: 10.3892/ol.2019.10034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Sengottuvelan M, Senthilkumar R, Nalini N. Modulatory influence of dietary resveratrol during different phases of 1,2-dimethylhydrazine induced mucosal lipid-peroxidation, antioxidant status and aberrant crypt foci development in rat colon carcinogenesis. Biochimica et Biophysica Acta. 2006;1760(8):1175–1183. doi: 10.1016/j.bbagen.2006.03.008. [DOI] [PubMed] [Google Scholar]
- 223.Sengottuvelan M, Nalini N. Dietary supplementation of resveratrol suppresses colonic tumour incidence in 1,2-dimethylhydrazine-treated rats by modulating biotransforming enzymes and aberrant crypt foci development. The British Journal of Nutrition. 2006;96(1):145–153. doi: 10.1079/bjn20061789. [DOI] [PubMed] [Google Scholar]
- 224.Alfaras I, Juan ME, Planas JM. trans-Resveratrol reduces precancerous colonic lesions in dimethylhydrazine-treated rats. Journal of Agricultural and Food Chemistry. 2010;58(13):8104–8110. doi: 10.1021/jf100702x. [DOI] [PubMed] [Google Scholar]
- 225.Schneider Y, Duranton B, Gossé F, Schleiffer R, Seiler N, Raul F. Resveratrol inhibits intestinal tumorigenesis and modulates host-defense-related gene expression in an animal model of human familial adenomatous polyposis. Nutrition and Cancer. 2001;39(1):102–107. doi: 10.1207/S15327914nc391_14. [DOI] [PubMed] [Google Scholar]
- 226.Majumdar AP, Banerjee S, Nautiyal J, Patel BB, Patel V, Du J, et al. Curcumin synergizes with resveratrol to inhibit colon cancer. Nutrition and Cancer. 2009;61(4):544–553. doi: 10.1080/01635580902752262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Sale S, Tunstall RG, Ruparelia KC, Potter GA, Steward WP, Gescher AJ. Comparison of the effects of the chemopreventive agent resveratrol and its synthetic analog trans 3,4,5,4’-tetramethoxystilbene (DMU-212) on adenoma development in the Apc(Min+) mouse and cyclooxygenase-2 in human-derived colon cancer cells. International Journal of Cancer. 2005;115(2):194–201. doi: 10.1002/ijc.20884. [DOI] [PubMed] [Google Scholar]
- 228.Tessitore L, Davit A, Sarotto I, Caderni G. Resveratrol depresses the growth of colorectal aberrant crypt foci by affecting bax and p21(CIP) expression. Carcinogenesis. 2000;21(8):1619–1622. doi: 10.1093/carcin/21.5.619. [DOI] [PubMed] [Google Scholar]
- 229.Baur JA, Sinclair DA. Therapeutic potential of resveratrol: the in vivo evidence. Nature Reviews Drug Discovery. 2006;5(6):493–506. doi: 10.1038/nrd2060. [DOI] [PubMed] [Google Scholar]
- 230.Brockmueller, A., Sameri, S., Liskova, A., Zhai, K., Varghese, E., Samuel, S. M., et al. (2021). Resveratrol’s anti-cancer effects through the modulation of tumor glucose metabolism. Cancers, 13(2). 10.3390/cancers13020188 [DOI] [PMC free article] [PubMed]
- 231.Buhrmann, C., Shayan, P., Brockmueller, A., & Shakibaei, M. (2020). Resveratrol suppresses cross-talk between colorectal cancer cells and stromal cells in multicellular tumor microenvironment: A bridge between in vitro and in vivo tumor microenvironment study. Molecules, 25(18). 10.3390/molecules25184292 [DOI] [PMC free article] [PubMed]
- 232.Chan JY, Phoo MS, Clement MV, Pervaiz S, Lee SC. Resveratrol displays converse dose-related effects on 5-fluorouracil-evoked colon cancer cell apoptosis: the roles of caspase-6 and p53. Cancer Biology & Therapy. 2008;7(8):1305–1312. doi: 10.4161/cbt.7.8.6302. [DOI] [PubMed] [Google Scholar]
- 233.Gescher AJ, Steward WP. Relationship between mechanisms, bioavailibility, and preclinical chemopreventive efficacy of resveratrol: A conundrum. Cancer Epidemiology, Biomarkers & Prevention. 2003;12(10):953–957. [PubMed] [Google Scholar]
- 234.Temraz S, Mukherji D, Shamseddine A. Potential targets for colorectal cancer prevention. International Journal of Molecular Sciences. 2013;14(9):17279–17303. doi: 10.3390/ijms140917279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Chen L, Musa AE. Boosting immune system against cancer by resveratrol. Phytotherapy Research. 2021;35(10):5514–5526. doi: 10.1002/ptr.7189. [DOI] [PubMed] [Google Scholar]
- 236.Ashrafizadeh M, Farhood B, Eleojo Musa A, Taeb S, Najafi M. The interactions and communications in tumor resistance to radiotherapy: Therapy perspectives. International Immunopharmacology. 2020;87:106807. doi: 10.1016/j.intimp.2020.106807. [DOI] [PubMed] [Google Scholar]
- 237.Matkowski R, Gisterek I, Halon A, Lacko A, Szewczyk K, Staszek U, et al. The prognostic role of tumor-infiltrating CD4 and CD8 T lymphocytes in breast cancer. Anticancer Research. 2009;29(7):2445–2451. [PubMed] [Google Scholar]
- 238.Craveiro, M., Cretenet, G., Mongellaz, C., Matias, M. I., Caron, O., de Lima, M. C. P., et al. (2017). Resveratrol stimulates the metabolic reprogramming of human CD4+ T cells to enhance effector function. Science Signaling, 10(501). 10.1126/scisignal.aal3024 [DOI] [PubMed]
- 239.He X, Xu C. Immune checkpoint signaling and cancer immunotherapy. Cell Research. 2020;30(8):660–669. doi: 10.1038/s41422-020-0343-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Choi YJ, Yang KM, Kim SD, Yoo YH, Lee SW, Seo SY, et al. Resveratrol analogue HS-1793 induces the modulation of tumor-derived T cells. Experimental and Therapeutic Medicine. 2012;3(4):592–598. doi: 10.3892/etm.2012.472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Pandurangan AK, Esa NM. Dietary non-nutritive factors in targeting of regulatory molecules in colorectal cancer: an update. Asian Pacific Journal of Cancer Prevention. 2013;14(10):5543–5552. doi: 10.7314/apjcp.2013.14.10.5543. [DOI] [PubMed] [Google Scholar]
- 242.Panaro MA, Carofiglio V, Acquafredda A, Cavallo P, Cianciulli A. Anti-inflammatory effects of resveratrol occur via inhibition of lipopolysaccharide-induced NF-κB activation in Caco-2 and SW480 human colon cancer cells. The British Journal of Nutrition. 2012;108(9):1623–1632. doi: 10.1017/s0007114511007227. [DOI] [PubMed] [Google Scholar]
- 243.Cui X, Jin Y, Hofseth AB, Pena E, Habiger J, Chumanevich A, et al. Resveratrol suppresses colitis and colon cancer associated with colitis. Cancer Prevention Research. 2010;3(4):549–559. doi: 10.1158/1940-6207.Capr-09-0117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Hu T, Li Z, Gao CY, Cho CH. Mechanisms of drug resistance in colon cancer and its therapeutic strategies. World Journal of Gastroenterology. 2016;22(30):6876–6889. doi: 10.3748/wjg.v22.i30.6876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Samuel, S. M., Varghese, E., Koklesová, L., Líšková, A., Kubatka, P., & Büsselberg, D. (2020). Counteracting chemoresistance with metformin in breast cancers: Targeting cancer stem cells. Cancers, 12(9). 10.3390/cancers12092482 [DOI] [PMC free article] [PubMed]
- 246.Ma, S. C., Zhang, J. Q., Yan, T. H., Miao, M. X., Cao, Y. M., Cao, Y. B., et al. (2023). Novel strategies to reverse chemoresistance in colorectal cancer. Cancer Medicine. 10.1002/cam4.5594 [DOI] [PMC free article] [PubMed]
- 247.Wang, Q., Shen, X., Chen, G., & Du, J. (2022). Drug resistance in colorectal cancer: From mechanism to clinic. Cancers, 14(12). 10.3390/cancers14122928 [DOI] [PMC free article] [PubMed]
- 248.Chen L, Yang F, Chen S, Tai J. Mechanisms on chemotherapy resistance of colorectal cancer stem cells and research progress of reverse transformation: A mini-review. Frontiers in Medicine. 2022;9:995882. doi: 10.3389/fmed.2022.995882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.Hammond WA, Swaika A, Mody K. Pharmacologic resistance in colorectal cancer: a review. Therapeutic Advances in Medical Oncology. 2016;8(1):57–84. doi: 10.1177/1758834015614530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Skarkova, V., Kralova, V., Vitovcova, B., & Rudolf, E. (2019). Selected aspects of chemoresistance mechanisms in colorectal carcinoma-A focus on epithelial-to-mesenchymal transition, autophagy, and apoptosis. Cells, 8(3). 10.3390/cells8030234 [DOI] [PMC free article] [PubMed]
- 251.Van der Jeught K, Xu HC, Li YJ, Lu XB, Ji G. Drug resistance and new therapies in colorectal cancer. World Journal of Gastroenterology. 2018;24(34):3834–3848. doi: 10.3748/wjg.v24.i34.3834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252.Popat S, Matakidou A, Houlston RS. Thymidylate synthase expression and prognosis in colorectal cancer: A systematic review and meta-analysis. Journal of Clinical Oncology. 2004;22(3):529–536. doi: 10.1200/jco.2004.05.064. [DOI] [PubMed] [Google Scholar]
- 253.Shibata J, Aiba K, Shibata H, Minowa S, Horikoshi N. Detection and quantitation of thymidylate synthase mRNA in human colon adenocarcinoma cell line resistant to 5-fluorouracil by competitive PCR. Anticancer Research. 1998;18(3a):1457–1463. [PubMed] [Google Scholar]
- 254.Kamoshida S, Matsuoka H, Ishikawa T, Maeda K, Shimomura R, Inada K, et al. Immunohistochemical evaluation of thymidylate synthase (TS) and p16INK4a in advanced colorectal cancer: Implication of TS expression in 5-FU-based adjuvant chemotherapy. Japanese Journal of Clinical Oncology. 2004;34(10):594–601. doi: 10.1093/jjco/hyh113. [DOI] [PubMed] [Google Scholar]
- 255.Wang TL, Diaz LA, Jr, Romans K, Bardelli A, Saha S, Galizia G, et al. Digital karyotyping identifies thymidylate synthase amplification as a mechanism of resistance to 5-fluorouracil in metastatic colorectal cancer patients. Proceedings of the National Academy of Sciences of the United States of America. 2004;101(9):3089–3094. doi: 10.1073/pnas.0308716101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Marsh S, McKay JA, Cassidy J, McLeod HL. Polymorphism in the thymidylate synthase promoter enhancer region in colorectal cancer. International Journal of Oncology. 2001;19(2):383–386. doi: 10.3892/ijo.19.2.383. [DOI] [PubMed] [Google Scholar]
- 257.Francipane, M. G., Bulanin, D., & Lagasse, E. (2019). Establishment and characterization of 5-fluorouracil-resistant human colorectal cancer stem-like cells: Tumor dynamics under selection pressure. International Journal of Molecular Sciences, 20(8). 10.3390/ijms20081817 [DOI] [PMC free article] [PubMed]
- 258.Boyer J, McLean EG, Aroori S, Wilson P, McCulla A, Carey PD, et al. Characterization of p53 wild-type and null isogenic colorectal cancer cell lines resistant to 5-fluorouracil, oxaliplatin, and irinotecan. Clinical Cancer Research. 2004;10(6):2158–2167. doi: 10.1158/1078-0432.ccr-03-0362. [DOI] [PubMed] [Google Scholar]
- 259.Zheng Z, Li J, He X, Chen X, Yu B, Ji J, et al. Involvement of RhoGDI2 in the resistance of colon cancer cells to 5-fluorouracil. Hepatogastroenterology. 2010;57(102-103):1106–1112. [PubMed] [Google Scholar]
- 260.Chen X, Sun S, Geng T, Fan X, Zhang S, Zhao S, et al. Resveratrol reduces the progression of titanium particle-induced osteolysis via the Wnt/β-catenin signaling pathway in vivo and in vitro. Experimental and Therapeutic Medicine. 2021;22(4):1119. doi: 10.3892/etm.2021.10553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 261.de la Cruz-Morcillo MA, Valero ML, Callejas-Valera JL, Arias-González L, Melgar-Rojas P, Galán-Moya EM, et al. P38MAPK is a major determinant of the balance between apoptosis and autophagy triggered by 5-fluorouracil: Implication in resistance. Oncogene. 2012;31(9):1073–1085. doi: 10.1038/onc.2011.321. [DOI] [PubMed] [Google Scholar]
- 262.Rubio MF, Lira MC, Rosa FD, Sambresqui AD, Salazar Güemes MC, Costas MA. RAC3 influences the chemoresistance of colon cancer cells through autophagy and apoptosis inhibition. Cancer Cell International. 2017;17:111. doi: 10.1186/s12935-017-0483-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Romano G, Santi L, Bianco MR, Giuffrè MR, Pettinato M, Bugarin C, et al. The TGF-β pathway is activated by 5-fluorouracil treatment in drug resistant colorectal carcinoma cells. Oncotarget. 2016;7(16):22077–22091. doi: 10.18632/oncotarget.7895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Suetsugu, T., Mori, R., Futamura, M., Fukada, M., Tanaka, H., Yasufuku, I., et al. (2021). Mechanism of acquired 5FU resistance and strategy for overcoming 5FU resistance focusing on 5FU metabolism in colon cancer cell lines. Oncology Reports, 45(4). 10.3892/or.2021.7978 [DOI] [PMC free article] [PubMed]
- 265.Zhang L, Song R, Gu D, Zhang X, Yu B, Liu B, et al. The role of GLI1 for 5-Fu resistance in colorectal cancer. Cell & Bioscience. 2017;7:17. doi: 10.1186/s13578-017-0145-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Zhao J, Li W, Zhu D, Yu Q, Zhang Z, Sun M, et al. Association of single nucleotide polymorphisms in MTHFR and ABCG2 with the different efficacy of first-line chemotherapy in metastatic colorectal cancer. Medical Oncology. 2014;31(1):802. doi: 10.1007/s12032-013-0802-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Candeil L, Gourdier I, Peyron D, Vezzio N, Copois V, Bibeau F, et al. ABCG2 overexpression in colon cancer cells resistant to SN38 and in irinotecan-treated metastases. International Journal of Cancer. 2004;109(6):848–854. doi: 10.1002/ijc.20032. [DOI] [PubMed] [Google Scholar]
- 268.Wu ZX, Yang Y, Zeng L, Patel H, Bo L, Lin L, et al. Establishment and characterization of an irinotecan-resistant human colon cancer cell line. Frontiers in Oncology. 2020;10:624954. doi: 10.3389/fonc.2020.624954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Owatari S, Akune S, Komatsu M, Ikeda R, Firth SD, Che XF, et al. Copper-transporting P-type ATPase, ATP7A, confers multidrug resistance and its expression is related to resistance to SN-38 in clinical colon cancer. Cancer Research. 2007;67(10):4860–4868. doi: 10.1158/0008-5472.Can-06-3096. [DOI] [PubMed] [Google Scholar]
- 270.Zhang S, Lovejoy KS, Shima JE, Lagpacan LL, Shu Y, Lapuk A, et al. Organic cation transporters are determinants of oxaliplatin cytotoxicity. Cancer Research. 2006;66(17):8847–8857. doi: 10.1158/0008-5472.Can-06-0769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Hsieh CC, Hsu SH, Lin CY, Liaw HJ, Li TW, Jiang KY, et al. CHK2 activation contributes to the development of oxaliplatin resistance in colorectal cancer. British Journal of Cancer. 2022;127(9):1615–1628. doi: 10.1038/s41416-022-01946-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Hsu HH, Chen MC, Baskaran R, Lin YM, Day CH, Lin YJ, et al. Oxaliplatin resistance in colorectal cancer cells is mediated via activation of ABCG2 to alleviate ER stress induced apoptosis. Journal of Cellular Physiology. 2018;233(7):5458–5467. doi: 10.1002/jcp.26406. [DOI] [PubMed] [Google Scholar]
- 273.Chian S, Li YY, Wang XJ, Tang XW. Luteolin sensitizes two oxaliplatin-resistant colorectal cancer cell lines to chemotherapeutic drugs via inhibition of the Nrf2 pathway. Asian Pacific Journal of Cancer Prevention. 2014;15(6):2911–2916. doi: 10.7314/apjcp.2014.15.6.2911. [DOI] [PubMed] [Google Scholar]
- 274.Jacobs B, De Roock W, Piessevaux H, Van Oirbeek R, Biesmans B, De Schutter J, et al. Amphiregulin and epiregulin mRNA expression in primary tumors predicts outcome in metastatic colorectal cancer treated with cetuximab. Journal of Clinical Oncology. 2009;27(30):5068–5074. doi: 10.1200/jco.2008.21.3744. [DOI] [PubMed] [Google Scholar]
- 275.Van Emburgh BO, Arena S, Siravegna G, Lazzari L, Crisafulli G, Corti G, et al. Acquired RAS or EGFR mutations and duration of response to EGFR blockade in colorectal cancer. Nature Communications. 2016;7:13665. doi: 10.1038/ncomms13665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Price T, Ang A, Boedigheimer M, Kim TW, Li J, Cascinu S, et al. Frequency of S492R mutations in the epidermal growth factor receptor: analysis of plasma DNA from patients with metastatic colorectal cancer treated with panitumumab or cetuximab monotherapy. Cancer Biology & Therapy. 2020;21(10):891–898. doi: 10.1080/15384047.2020.1798695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Greenlee, J. D., Lopez-Cavestany, M., Ortiz-Otero, N., Liu, K., Subramanian, T., Cagir, B., et al. (2021). Oxaliplatin resistance in colorectal cancer enhances TRAIL sensitivity via death receptor 4 upregulation and lipid raft localization. Elife, 10. 10.7554/eLife.67750 [DOI] [PMC free article] [PubMed]
- 278.Fojo T. Multiple paths to a drug resistance phenotype: Mutations, translocations, deletions and amplification of coding genes or promoter regions, epigenetic changes and microRNAs. Drug Resistance Updates. 2007;10(1-2):59–67. doi: 10.1016/j.drup.2007.02.002. [DOI] [PubMed] [Google Scholar]
- 279.Alfarouk KO, Stock CM, Taylor S, Walsh M, Muddathir AK, Verduzco D, et al. Resistance to cancer chemotherapy: Failure in drug response from ADME to P-gp. Cancer Cell International. 2015;15:71. doi: 10.1186/s12935-015-0221-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Schmidt EM, Lamprecht S, Blaj C, Schaaf C, Krebs S, Blum H, et al. Targeting tumor cell plasticity by combined inhibition of NOTCH and MAPK signaling in colon cancer. The Journal of Experimental Medicine. 2018;215(6):1693–1708. doi: 10.1084/jem.20171455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Shi ZD, Pang K, Wu ZX, Dong Y, Hao L, Qin JX, et al. Tumor cell plasticity in targeted therapy-induced resistance: mechanisms and new strategies. Signal Transduction and Targeted Therapy. 2023;8(1):113. doi: 10.1038/s41392-023-01383-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Hay ED. An overview of epithelio-mesenchymal transformation. Acta Anatomica (Basel) 1995;154(1):8–20. doi: 10.1159/000147748. [DOI] [PubMed] [Google Scholar]
- 283.Thiery JP. Epithelial-mesenchymal transitions in development and pathologies. Current Opinion in Cell Biology. 2003;15(6):740–746. doi: 10.1016/j.ceb.2003.10.006. [DOI] [PubMed] [Google Scholar]
- 284.Thiery JP, Sleeman JP. Complex networks orchestrate epithelial-mesenchymal transitions. Nature Reviews. Molecular Cell Biology. 2006;7(2):131–142. doi: 10.1038/nrm1835. [DOI] [PubMed] [Google Scholar]
- 285.Vega S, Morales AV, Ocaña OH, Valdés F, Fabregat I, Nieto MA. Snail blocks the cell cycle and confers resistance to cell death. Genes & Development. 2004;18(10):1131–1143. doi: 10.1101/gad.294104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 286.Wu YH, Huang YF, Chang TH, Chou CY. Activation of TWIST1 by COL11A1 promotes chemoresistance and inhibits apoptosis in ovarian cancer cells by modulating NF-κB-mediated IKKβ expression. International Journal of Cancer. 2017;141(11):2305–2317. doi: 10.1002/ijc.30932. [DOI] [PubMed] [Google Scholar]
- 287.Inoue A, Seidel MG, Wu W, Kamizono S, Ferrando AA, Bronson RT, et al. Slug, a highly conserved zinc finger transcriptional repressor, protects hematopoietic progenitor cells from radiation-induced apoptosis in vivo. Cancer Cell. 2002;2(4):279–288. doi: 10.1016/s1535-6108(02)00155-1. [DOI] [PubMed] [Google Scholar]
- 288.Liu YR, Liang L, Zhao JM, Zhang Y, Zhang M, Zhong WL, et al. Twist1 confers multidrug resistance in colon cancer through upregulation of ATP-binding cassette transporters. Oncotarget. 2017;8(32):52901–52912. doi: 10.18632/oncotarget.17548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Du, B., & Shim, J. S. (2016). Targeting Epithelial-Mesenchymal Transition (EMT) to Overcome Drug Resistance in Cancer. Molecules, 21(7). 10.3390/molecules21070965 [DOI] [PMC free article] [PubMed]
- 290.Wu F, Zhu J, Mao Y, Li X, Hu B, Zhang D. Associations between the epithelial-mesenchymal transition phenotypes of circulating tumor cells and the clinicopathological features of patients with colorectal cancer. Disease Markers. 2017;2017:9474532. doi: 10.1155/2017/9474532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 291.Bates RC, Mercurio AM. Tumor necrosis factor-alpha stimulates the epithelial-to-mesenchymal transition of human colonic organoids. Molecular Biology of the Cell. 2003;14(5):1790–1800. doi: 10.1091/mbc.e02-09-0583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 292.Bates RC, Bellovin DI, Brown C, Maynard E, Wu B, Kawakatsu H, et al. Transcriptional activation of integrin beta6 during the epithelial-mesenchymal transition defines a novel prognostic indicator of aggressive colon carcinoma. The Journal of Clinical Investigation. 2005;115(2):339–347. doi: 10.1172/jci23183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 293.Zhou M, Liu X, Li Z, Huang Q, Li F, Li CY. Caspase-3 regulates the migration, invasion and metastasis of colon cancer cells. International Journal of Cancer. 2018;143(4):921–930. doi: 10.1002/ijc.31374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 294.Islam, S. U., Ahmed, M. B., Sonn, J. K., Jin, E. J., & Lee, Y. S. (2022). PRP4 induces epithelial-mesenchymal transition and drug resistance in colon cancer cells via activation of p53. International Journal of Molecular Sciences, 23(6). 10.3390/ijms23063092 [DOI] [PMC free article] [PubMed]
- 295.Yang AD, Fan F, Camp ER, van Buren G, Liu W, Somcio R, et al. Chronic oxaliplatin resistance induces epithelial-to-mesenchymal transition in colorectal cancer cell lines. Clinical Cancer Research. 2006;12(14 Pt 1):4147–4153. doi: 10.1158/1078-0432.ccr-06-0038. [DOI] [PubMed] [Google Scholar]
- 296.Sousa-Squiavinato ACM, Arregui Ramos DA, Wagner MS, Tessmann JW, de-Freitas-Junior JCM, Morgado-Díaz JA. Long-term resistance to 5-fluorouracil promotes epithelial-mesenchymal transition, apoptosis evasion, autophagy, and reduced proliferation rate in colon cancer cells. European Journal of Pharmacology. 2022;933:175253. doi: 10.1016/j.ejphar.2022.175253. [DOI] [PubMed] [Google Scholar]
- 297.Yu Z, Pestell TG, Lisanti MP, Pestell RG. Cancer stem cells. The International Journal of Biochemistry & Cell Biology. 2012;44(12):2144–2151. doi: 10.1016/j.biocel.2012.08.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 298.Dalerba P, Dylla SJ, Park IK, Liu R, Wang X, Cho RW, et al. Phenotypic characterization of human colorectal cancer stem cells. Proceedings of the National Academy of Sciences of the United States of America. 2007;104(24):10158–10163. doi: 10.1073/pnas.0703478104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 299.Cho YH, Ro EJ, Yoon JS, Mizutani T, Kang DW, Park JC, et al. 5-FU promotes stemness of colorectal cancer via p53-mediated WNT/β-catenin pathway activation. Nature Communications. 2020;11(1):5321. doi: 10.1038/s41467-020-19173-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 300.Jo MK, Moon CM, Kim EJ, Kwon JH, Fei X, Kim SE, et al. Suppressive effect of α-mangostin for cancer stem cells in colorectal cancer via the Notch pathway. BMC Cancer. 2022;22(1):341. doi: 10.1186/s12885-022-09414-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 301.Kim, H., Yu, Y., Choi, S., Lee, H., Yu, J., Lee, J. H., et al. (2019). Evodiamine eliminates colon cancer stem cells via suppressing notch and Wnt signaling. Molecules, 24(24). 10.3390/molecules24244520 [DOI] [PMC free article] [PubMed]
- 302.Regan JL, Schumacher D, Staudte S, Steffen A, Haybaeck J, Keilholz U, et al. Non-canonical hedgehog signaling is a positive regulator of the WNT pathway and is required for the survival of colon cancer stem cells. Cell Reports. 2017;21(10):2813–2828. doi: 10.1016/j.celrep.2017.11.025. [DOI] [PubMed] [Google Scholar]
- 303.Zhou H, Xiong Y, Peng L, Wang R, Zhang H, Fu Z. LncRNA-cCSC1 modulates cancer stem cell properties in colorectal cancer via activation of the Hedgehog signaling pathway. Journal of Cellular Biochemistry. 2020;121(3):2510–2524. doi: 10.1002/jcb.29473. [DOI] [PubMed] [Google Scholar]
- 304.Zhou Y, Xia L, Wang H, Oyang L, Su M, Liu Q, et al. Cancer stem cells in progression of colorectal cancer. Oncotarget. 2018;9(70):33403–33415. doi: 10.18632/oncotarget.23607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 305.Butler SJ, Richardson L, Farias N, Morrison J, Coomber BL. Characterization of cancer stem cell drug resistance in the human colorectal cancer cell lines HCT116 and SW480. Biochemical and Biophysical Research Communications. 2017;490(1):29–35. doi: 10.1016/j.bbrc.2017.05.176. [DOI] [PubMed] [Google Scholar]
- 306.Kangwan N, Kim YJ, Han YM, Jeong M, Park JM, Go EJ, et al. Sonic hedgehog inhibitors prevent colitis-associated cancer via orchestrated mechanisms of IL-6/gp130 inhibition, 15-PGDH induction, Bcl-2 abrogation, and tumorsphere inhibition. Oncotarget. 2016;7(7):7667–7682. doi: 10.18632/oncotarget.6765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 307.Fumagalli A, Oost KC, Kester L, Morgner J, Bornes L, Bruens L, et al. Plasticity of Lgr5-negative cancer cells drives metastasis in colorectal cancer. Cell Stem Cell. 2020;26(4):569–578.e567. doi: 10.1016/j.stem.2020.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 308.Lin SP, Lee YT, Yang SH, Miller SA, Chiou SH, Hung MC, et al. Colon cancer stem cells resist antiangiogenesis therapy-induced apoptosis. Cancer Letters. 2013;328(2):226–234. doi: 10.1016/j.canlet.2012.08.036. [DOI] [PubMed] [Google Scholar]
- 309.de Thé H. Differentiation therapy revisited. Nature Reviews. Cancer. 2018;18(2):117–127. doi: 10.1038/nrc.2017.103. [DOI] [PubMed] [Google Scholar]
- 310.Beltran H, Hruszkewycz A, Scher HI, Hildesheim J, Isaacs J, Yu EY, et al. The role of lineage plasticity in prostate cancer therapy resistance. Clinical Cancer Research. 2019;25(23):6916–6924. doi: 10.1158/1078-0432.ccr-19-1423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 311.Lin TA, Lin WS, Chou YC, Nagabhushanam K, Ho CT, Pan MH. Oxyresveratrol inhibits human colon cancer cell migration through regulating epithelial-mesenchymal transition and microRNA. Food & Function. 2021;12(20):9658–9668. doi: 10.1039/d1fo01920a. [DOI] [PubMed] [Google Scholar]
- 312.Markowitz SD, Bertagnolli MM. Molecular origins of cancer: Molecular basis of colorectal cancer. The New England Journal of Medicine. 2009;361(25):2449–2460. doi: 10.1056/NEJMra0804588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 313.Reddivari L, Charepalli V, Radhakrishnan S, Vadde R, Elias RJ, Lambert JD, et al. Grape compounds suppress colon cancer stem cells in vitro and in a rodent model of colon carcinogenesis. BMC Complementary and Alternative Medicine. 2016;16:278. doi: 10.1186/s12906-016-1254-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 314.Fan F, Samuel S, Evans KW, Lu J, Xia L, Zhou Y, et al. Overexpression of snail induces epithelial-mesenchymal transition and a cancer stem cell-like phenotype in human colorectal cancer cells. Cancer Medicine. 2012;1(1):5–16. doi: 10.1002/cam4.4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 315.Pouyafar A, Rezabakhsh A, Rahbarghazi R, Heydarabad MZ, Shokrollahi E, Sokullu E, et al. Treatment of cancer stem cells from human colon adenocarcinoma cell line HT-29 with resveratrol and sulindac induced mesenchymal-endothelial transition rate. Cell and Tissue Research. 2019;376(3):377–388. doi: 10.1007/s00441-019-02998-9. [DOI] [PubMed] [Google Scholar]
- 316.Doonan BB, Schaafsma E, Pinto JT, Wu JM, Hsieh TC. Application of open-access databases to determine functional connectivity between resveratrol-binding protein QR2 and colorectal carcinoma. In Vitro Cellular & Developmental Biology. Animal. 2017;53(7):575–578. doi: 10.1007/s11626-017-0174-x. [DOI] [PubMed] [Google Scholar]
- 317.Gavrilas LI, Cruceriu D, Ionescu C, Miere D, Balacescu O. Pro-apoptotic genes as new targets for single and combinatorial treatments with resveratrol and curcumin in colorectal cancer. Food & Function. 2019;10(6):3717–3726. doi: 10.1039/c9fo01014a. [DOI] [PubMed] [Google Scholar]
- 318.Cocetta, V., Quagliariello, V., Fiorica, F., Berretta, M., & Montopoli, M. (2021). Resveratrol as chemosensitizer agent: State of art and future perspectives. International Journal of Molecular Sciences, 22(4). 10.3390/ijms22042049 [DOI] [PMC free article] [PubMed]
- 319.Brockmueller, A., Girisa, S., Kunnumakkara, A. B., & Shakibaei, M. (2023). Resveratrol modulates chemosensitisation to 5-FU via β1-Integrin/HIF-1α axis in CRC tumor microenvironment. International Journal of Molecular Sciences, 24(5). 10.3390/ijms24054988 [DOI] [PMC free article] [PubMed]
- 320.Wang Y, Wang W, Wu X, Li C, Huang Y, Zhou H, et al. Resveratrol sensitizes colorectal cancer cells to cetuximab by connexin 43 upregulation-induced akt inhibition. Frontiers in Oncology. 2020;10:383. doi: 10.3389/fonc.2020.00383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 321.El-Readi MZ, Eid S, Abdelghany AA, Al-Amoudi HS, Efferth T, Wink M. Resveratrol mediated cancer cell apoptosis, and modulation of multidrug resistance proteins and metabolic enzymes. Phytomedicine. 2019;55:269–281. doi: 10.1016/j.phymed.2018.06.046. [DOI] [PubMed] [Google Scholar]
- 322.Khaleel SA, Al-Abd AM, Ali AA, Abdel-Naim AB. Didox and resveratrol sensitize colorectal cancer cells to doxorubicin via activating apoptosis and ameliorating P-glycoprotein activity. Scientific Reports. 2016;6:36855. doi: 10.1038/srep36855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 323.Kaminski BM, Weigert A, Scherzberg MC, Ley S, Gilbert B, Brecht K, et al. Resveratrol-induced potentiation of the antitumor effects of oxaliplatin is accompanied by an altered cytokine profile of human monocyte-derived macrophages. Apoptosis. 2014;19(7):1136–1147. doi: 10.1007/s10495-014-0988-x. [DOI] [PubMed] [Google Scholar]
- 324.Park D-G. Antichemosensitizing effect of resveratrol in cotreatment with oxaliplatin in HCT116 colon cancer cell. Annals of Surgical Treatment and Research. 2014;86(2):68–75. doi: 10.4174/astr.2014.86.2.68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 325.Blondy S, David V, Verdier M, Mathonnet M, Perraud A, Christou N. 5-Fluorouracil resistance mechanisms in colorectal cancer: From classical pathways to promising processes. Cancer Science. 2020;111(9):3142–3154. doi: 10.1111/cas.14532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 326.Takata M, Chikumi H, Miyake N, Adachi K, Kanamori Y, Yamasaki A, et al. Lack of AKT activation in lung cancer cells with EGFR mutation is a novel marker of cetuximab sensitivity. Cancer Biology & Therapy. 2012;13(6):369–378. doi: 10.4161/cbt.19238. [DOI] [PubMed] [Google Scholar]
- 327.Howells LM, Berry DP, Elliott PJ, Jacobson EW, Hoffmann E, Hegarty B, et al. Phase I randomized, double-blind pilot study of micronized resveratrol (SRT501) in patients with hepatic metastases--safety, pharmacokinetics, and pharmacodynamics. Cancer Prevention Research. 2011;4(9):1419–1425. doi: 10.1158/1940-6207.capr-11-0148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 328.Nguyen AV, Martinez M, Stamos MJ, Moyer MP, Planutis K, Hope C, et al. Results of a phase I pilot clinical trial examining the effect of plant-derived resveratrol and grape powder on Wnt pathway target gene expression in colonic mucosa and colon cancer. Cancer Management and Research. 2009;1:25–37. doi: 10.2147/CMAR.S4544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 329.Van Cutsem E, Borràs JM, Castells A, Ciardiello F, Ducreux M, Haq A, et al. Improving outcomes in colorectal cancer: Where do we go from here? European Journal of Cancer. 2013;49(11):2476–2485. doi: 10.1016/j.ejca.2013.03.026. [DOI] [PubMed] [Google Scholar]
- 330.Gao D, Li C, Xie X, Zhao P, Wei X, Sun W, et al. Autologous tumor lysate-pulsed dendritic cell immunotherapy with cytokine-induced killer cells improves survival in gastric and colorectal cancer patients. PLoS One. 2014;9(4):e93886. doi: 10.1371/journal.pone.0093886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 331.Moon JY, Ediriweera MK, Ryu JY, Kim HY, Cho SK. Catechol enhances chemo- and radio-sensitivity by targeting AMPK/Hippo signaling in pancreatic cancer cells. Oncology Reports. 2021;45(3):1133–1141. doi: 10.3892/or.2021.7924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 332.Mottaghi S, Abbaszadeh H. Natural lignans honokiol and magnolol as potential anticarcinogenic and anticancer agents. A comprehensive mechanistic review. Nutrition and Cancer. 2022;74(3):761–778. doi: 10.1080/01635581.2021.1931364. [DOI] [PubMed] [Google Scholar]
- 333.Muthusamy G, Balupillai A, Ramasamy K, Shanmugam M, Gunaseelan S, Mary B, et al. Ferulic acid reverses ABCB1-mediated paclitaxel resistance in MDR cell lines. European Journal of Pharmacology. 2016;786:194–203. doi: 10.1016/j.ejphar.2016.05.023. [DOI] [PubMed] [Google Scholar]
- 334.Muthusamy G, Gunaseelan S, Prasad NR. Ferulic acid reverses P-glycoprotein-mediated multidrug resistance via inhibition of PI3K/Akt/NF-κB signaling pathway. The Journal of Nutritional Biochemistry. 2019;63:62–71. doi: 10.1016/j.jnutbio.2018.09.022. [DOI] [PubMed] [Google Scholar]
- 335.Buhrmann, C., Kunnumakkara, A. B., Popper, B., Majeed, M., Aggarwal, B. B., & Shakibaei, M. (2020). Calebin A potentiates the Effect of 5-FU and TNF-β (Lymphotoxin α) against Human Colorectal Cancer Cells: Potential Role of NF-κB. International Journal of Molecular Sciences, 21(7). 10.3390/ijms21072393 [DOI] [PMC free article] [PubMed]
- 336.Shakibaei M, Kraehe P, Popper B, Shayan P, Goel A, Buhrmann C. Curcumin potentiates antitumor activity of 5-fluorouracil in a 3D alginate tumor microenvironment of colorectal cancer. BMC Cancer. 2015;15:250. doi: 10.1186/s12885-015-1291-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 337.Wang Y, Ma J, Qiu T, Tang M, Zhang X, Dong W. In vitro and in vivo combinatorial anticancer effects of oxaliplatin- and resveratrol-loaded N,O-carboxymethyl chitosan nanoparticles against colorectal cancer. European Journal of Pharmaceutical Sciences. 2021;163:105864. doi: 10.1016/j.ejps.2021.105864. [DOI] [PubMed] [Google Scholar]
- 338.Moutabian H, Majdaeen M, Ghahramani-Asl R, Yadollahi M, Gharepapagh E, Ataei G, et al. A systematic review of the therapeutic effects of resveratrol in combination with 5-fluorouracil during colorectal cancer treatment: with a special focus on the oxidant, apoptotic, and anti-inflammatory activities. Cancer Cell International. 2022;22(1):142. doi: 10.1186/s12935-022-02561-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 339.Almeida L, Vaz-da-Silva M, Falcão A, Soares E, Costa R, Loureiro AI, et al. Pharmacokinetic and safety profile of trans-resveratrol in a rising multiple-dose study in healthy volunteers. Molecular Nutrition & Food Research. 2009;53(Suppl 1):S7–S15. doi: 10.1002/mnfr.200800177. [DOI] [PubMed] [Google Scholar]
- 340.Walle T, Hsieh F, DeLegge MH, Oatis JE, Jr, Walle UK. High absorption but very low bioavailability of oral resveratrol in humans. Drug Metabolism and Disposition. 2004;32(12):1377–1382. doi: 10.1124/dmd.104.000885. [DOI] [PubMed] [Google Scholar]
- 341.Johnson JJ, Nihal M, Siddiqui IA, Scarlett CO, Bailey HH, Mukhtar H, et al. Enhancing the bioavailability of resveratrol by combining it with piperine. Molecular Nutrition & Food Research. 2011;55(8):1169–1176. doi: 10.1002/mnfr.201100117. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data are available in the manuscript.