Abstract
Background
Analysing genomes of animal model organisms is widely used for understanding the genetic basis of complex traits and diseases, such as obesity, for which only a few mouse models exist, however, without their lean counterparts.
Objective
To analyse genetic differences in the unique mouse models of polygenic obesity (Fat line) and leanness (Lean line) originating from the same base population and established by divergent selection over more than 60 generations.
Methods
Genetic variability was analysed using WGS. Variants were identified with GATK and annotated with Ensembl VEP. g.Profiler, WebGestalt, and KEGG were used for GO and pathway enrichment analysis. miRNA seed regions were obtained with miRPathDB 2.0, LncRRIsearch was used to predict targets of identified lncRNAs, and genes influencing adipose tissue amount were searched using the IMPC database.
Results
WGS analysis revealed 6.3 million SNPs, 1.3 million were new. Thousands of potentially impactful SNPs were identified, including within 24 genes related to adipose tissue amount. SNP density was highest in pseudogenes and regulatory RNAs. The Lean line carries SNP rs248726381 in the seed region of mmu-miR-3086-3p, which may affect fatty acid metabolism. KEGG analysis showed deleterious missense variants in immune response and diabetes genes, with food perception pathways being most enriched. Gene prioritisation considering SNP GERP scores, variant consequences, and allele comparison with other mouse lines identified seven novel obesity candidate genes: 4930441H08Rik, Aff3, Fam237b, Gm36633, Pced1a, Tecrl, and Zfp536.
Conclusion
WGS revealed many genetic differences between the lines that accumulated over the selection period, including variants with potential negative impacts on gene function. Given the increasing availability of mouse strains and genetic polymorphism catalogues, the study is a valuable resource for researchers to study obesity.
Supplementary Information
The online version contains supplementary material available at 10.1007/s13258-024-01507-9.
Keywords: Obesity, Mouse models, Whole-genome sequencing, Single-nucleotide polymorphism
Introduction
Genome sequencing has become an important approach for exploring gene functions, phenotypical diversity among individuals and populations, and personalised medicine (Lu et al. 2014; Uffelmann et al. 2021). Genome-wide association studies (GWAS) have identified thousands of genetic variants associated with the increased risk for various human diseases (Sun et al. 2022).
Obesity, considered by many a 21st-century epidemic, has been widely believed to result from the disequilibrium between energy intake and expenditure. However, the aetiology of obesity is more complex, resulting from various factors, including genetic predispositions (González-Muniesa et al. 2017). It significantly increases the risk of various diseases such as type II diabetes mellitus, fatty liver disease, hypertension, myocardial infarction, stroke, dementia, osteoarthritis, and several cancers, thereby declining one’s life quality and life expectancy (Blüher 2019), as well as increasing the economic burden of the country (Pineda et al. 2018).
Studies have estimated that between 40% and 70% of obesity is heritable. Obesity is broadly divided into two categories: the monogenic type, which results from chromosomal deletions or single gene defects with a large effect, and the more common polygenic type, in which hundreds of genetic polymorphisms, each having a small effect, contribute to the obesity phenotype. The latter type follows a similar pattern of heritability as other complex traits and diseases (Loos and Yeo 2022).
Analysing genomes of animal model organisms, such as the mouse (Mus musculus), has been widely used to improve animal production and to understand the genetic basis of complex traits and diseases in both animals and humans (Andersson and Georges 2004; Saul et al. 2019). There are few mouse models for monogenic (for example ob/ob, db/db, and fa/fa), as well as polygenic types of obesity (for example NZO, TSOD, and C3H) (Suleiman et al. 2020). However, the latter strains, have no lean counterparts derived from the same base population. Prolonged selective breeding of inbred mice for desired divergent phenotypes produces novel, polygenic, and reproducible models of disease (Saul et al. 2019; Palma-Vera et al. 2022).
In this study, we present the results of whole-genome sequencing (WGS) analysis focused on single nucleotide polymorphisms (SNPs) in two unique mouse models for polygenic obesity (FLI, Fat line) and leanness (FHI, Lean line) which were established from the same base population using divergent selection on body fat percentage for over 60 generations (Sharp et al. 1984), differing 5-6-fold in fat content at the end of the selection process (Bünger and Hill 1999). Horvat et al. (2000) identified four major quantitative trait loci (QTL) responsible for their divergent phenotype, which is independent of known mutations in the leptin system or individual obesity-associated gene loci such as Cpe and Ay. Subsequent studies led to the identification of the Deptor gene as a novel regulator of adipogenesis (Laplante et al. 2012) and mitochondrial thiosulfate sulfurtransferase (Tst) as the causative gene for resistance to obesity (Morton et al. 2016). The identified polymorphisms and their corresponding genes identified in this study provide novel candidates and represent a valuable resource for researchers to study obesity.
Materials and methods
Mouse selection lines
Starting with a base population of two inbred mouse lines (JU, CBA) and one outbred mouse line (CFLP), two unique mouse models were developed following more than 60 generations of divergent selection for increased (FLI, Fat line) or decreased (FHI, Lean line) body fat percentage (Sharp et al. 1984). At the end of the selection process, the initial value of approximately 10% body fat in generation 0 increased to 22% in the Fat line and decreased to 4% in the Lean line, differing by a factor of 5–6. In the 53rd generation, the divergence between lines was 18% or a factor of 5–6, corresponding to about six standard deviations. This divergence in body fat percentage was shown to be due to the gradual accumulation of “obese” alleles in the Fat line and “lean” alleles in the Lean line (Bünger and Hill 1999). Subsequently, inbred lines were established from both lines by full-sib mating. Mice were maintained at The Centre for Laboratory Animals at the University of Ljubljana, Slovenia, the only research facility maintaining the Fat and Lean mouse lines. Animals were housed in individually ventilated cages (IVC), with tunnels for handling and nesting material as enrichment. They received a regular chow diet and had unlimited access to water under controlled environmental conditions: temperature (21 °C ± 2 °C), humidity (40–70%), and light (12 h day:12 h night cycle).
Whole-genome sequencing to explore genomic variability
Mouse strains are considered inbred after being maintained by successive brother-to-sister matings for at least 20 generations when on average at least 98.6% of the loci in each mouse are homozygous (Beck et al. 2000). In the present study whole genome sequencing (WGS) analysis was performed on spleen DNA samples from the Fat and Lean mouse lines in the 70th and 68th generation of inbreeding, respectively, as described by Mikec et al. (2022). In short, WGS was performed using the Illumina NextSeq 500 platform at the Medical Council Clinical Sciences Centre, University of Edinburgh, Scotland, UK. Reads were preprocessed and then mapped to the mouse reference genome (version GRCm38.86) using the Burrows-Wheeler Aligner (BWA) alignment tool (Li and Durbin 2009). The Genome Analysis Toolkit (GATK) (McKenna et al. 2010) Best Practices recommendations (Depristo et al. 2011; Van der Auwera et al. 2013) were followed for variants calling. Called SNPs were filtered to obtain SNPs with variant confidence standardised by depth above 2 (QD < 2.0; QualByDepth), quality above 30 (QUAL < 30), median mapping quality of reads supporting that site above 40 (MQ < 40.0; RMSMappingQuality), strand odds ratio under 3 (SOR > 3.0; StrandOddsRatio), Fisher’s exact test (FS) for strand bias under 60 (FS > 60.0), rank sum test for mapping qualities of reference vs. alternative reads above − 12.5 (MQRankSum < -12.5), rank sum test for site position within reads above − 8.0 (ReadPosRankSum < -8.0), and coverage depth above 30. Indels were filtered using QD < 2.0, QUAL < 30.0, FS > 200.0, ReadPosRankSum < -20.0, and SOR > 10.0. Finally, the identified variants were annotated using the Ensembl Variant Effect Predictor (VEP) (https://www.ensembl.org/Tools/VEP) (McLaren et al. 2016). Impact of SNPs are according to SnpEff (Cingolani et al. 2012b) and SnpSift (Cingolani et al. 2012a). Only homozygous polymorphisms were included in the analysis. Selected variants were validated by Sanger sequencing. The primer pairs used for PCR are given in Mikec et al. (2022), Šimon et al. (2024) and Supplementary Table S1. Sequence alignment was done using MEGA11: Molecular Evolutionary Genetics Analysis version 11 (Tamura, Stecher, and Kumar 2021).
Bioinformatics analyses
For bioinformatics analyses, we primarelly focused on line-specific SNPs (alleles present in one line but not in the other, compared to the mouse reference genome). The g.Profiler (Raudvere et al. 2019), WebGestalt (Liao et al. 2019), and Kyoto Encyclopedia of Genes and Genomes - KEGG (Kanehisa and Goto 2000) were used for Gene Ontology and pathway enrichment analysis. Cytoscape (Shannon 2003) and Cytoscape plugin ClueGO (Bindea et al. 2009) were used to visualize KEGG pathways and gene biological functions, respectively. The miRPathDB 2.0 (Kehl et al. 2020) was used to obtain the miRNA seed region, which was visualised using the Golden Helix GenomeBrowse® v3.1.0 visualisation tool (http://www.goldenhelix.com) (Golden Helix, Inc, Bozeman, MT), and the miRTargetLink 2.0 (Kern et al. 2021) was used to retrieve validated targets of miRNAs. The LncRRIsearch tool was used to predict targets of identified lncRNAs (Fukunaga et al. 2019). Genes related to obesity were obtained from the International Mouse Phenotyping Consortium - IMPC database (Birling et al. 2021) using the search keyword “abnormal adipose tissue amount”. QTL intervals were obtained from our previous study (Horvat et al. 2000). Annotations for obesity candidate genes were done for SNPs with Genomic Evolutionary Rate Profiling (GERP) score above six located in regulatory regions (promoters, promoter flanking regions, enhancers, CTCF binding sites, transcription factor binding sites, open chromatin regions) or having a potential moderate (inframe insertion, inframe deletion, missense variant, protein altering variant) or high (splice acceptor variant, splice donor variant, stop gained, frameshift variant, stop lost, start lost, transcript amplification, feature elongation, feature truncation) impact (McLaren et al. 2016), knockout effect from IMPC database, and their biological function, effect on phenotype, involvement of orthologous genes in human diseases related to obesity and its comorbidities using the Mouse Genome Database (MGD) (Bult et al. 2019), and differential expression. The expression data are according to our unpublished microarray analysis done on various tissues and RNA-seq on the hypothalamus. Finally, the potential involvement of SNPs within candidate genes in obesity was validated by comparing alleles between our mice lines and obesity-prone NZO/HlLtJ and obesity-resistant A/J strain.
Results
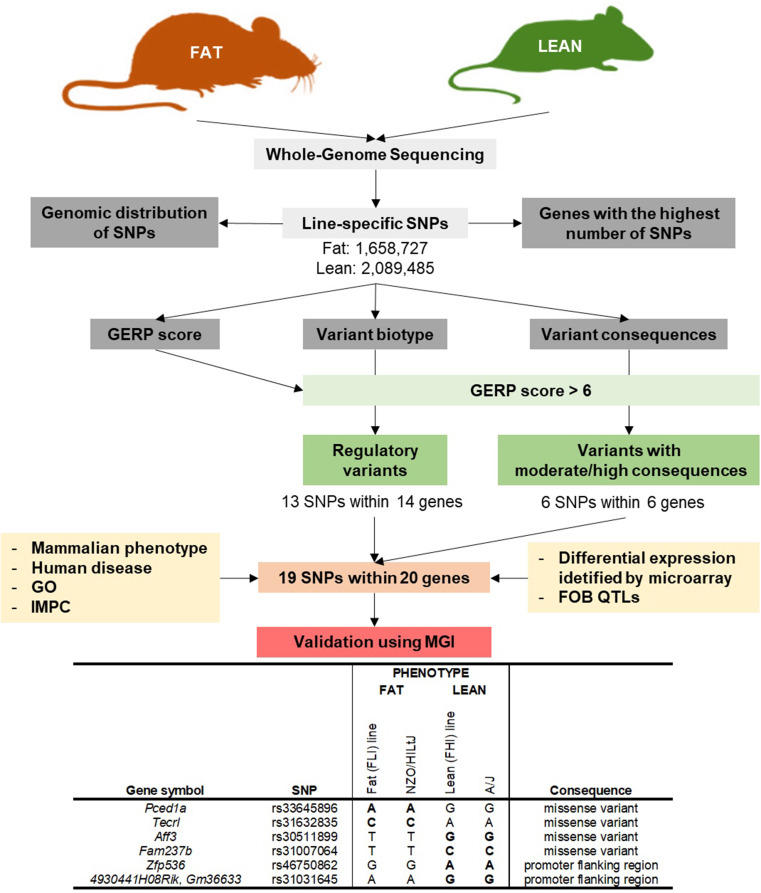
In the present study, we analysed genomes of Fat and Lean mouse lines, divergently selected for body fat percentage, focusing on line-specific SNPs. Firstly, their genomic distribution was analysed, followed by the identification of genes with the highest number and density of SNPs, location of constrained elements (GERP score), and analysis of variant biotypes and consequences. Then, we focused on regulatory variants and variants with predicted moderate or high impacts and GERP score above 6, resulting in 19 SNPs within 20 genes. For these genes, their previous annotations related to obesity were obtained from a public database, and their positions within previously identified obesity QTLs in our mouse models and their expressions in different tissues were examined. Additionally, the potential importance of identified SNP was validated by comparing the presence of alleles in obesity-resistant A/J and obesity-prone NZO/HlLtJ mice lines. The workflow and main results are shown in Fig. 1.
Fig. 1.
Workflow and main results from whole-genome sequencing of the Fat and Lean mice lines
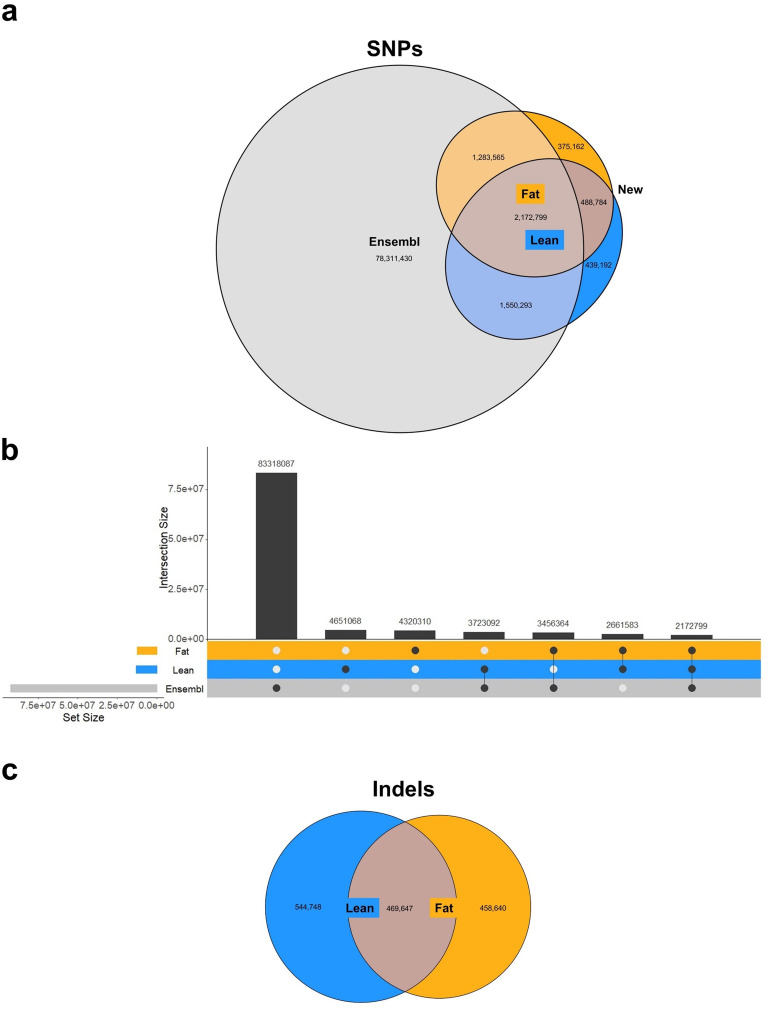
Sequencing genomes of male Lean (FHI line) and Fat (FLI line) mice revealed 4,651,068 and 4,320,310 SNPs in each line respectively among which the majority (2,661,583) are shared between both lines (Fig. 2). Out of 6,309,795 SNPs in total, we identified 1,303,138 SNPs that have not been previously reported (without rs ID). Of those variants, 488,784 are shared by both lines, while 439,192 and 375,162 SNPs are private to the Lean and Fat line, respectively (Fig. 2a, c). Moreover, 1,014,395 and 928,287 insertions and deletions (indels) were identified in the Lean and Fat line, respectively, and 469,647 indels occurred in both (Fig. 2b).
Fig. 2.
Sequence variants (SNPs and indels) identified in the Lean and Fat mouse selection lines. (a) Numbers of private and shared SNPs from the Fat and Lean mouse lines and the Ensembl database. The shape sizes are not proportional to the number of SNPs. (b) In scale comparison between SNPs deposited in the Ensembl and identified in the Lean and Fat lines, (c) number of indels identified in the Lean and Fat lines
Three selected SNPs were validated using Sanger sequencing and are summarized along with SNPs from Mikec et al. (2022) and Šimon et al. (2024) on Supplementary Fig. S1-3. Among these SNPs, it is worth mentioning a SNP rs37739792 within the intron of Hif1a and also overlapping protein-coding gene Gm15283 (Supplementary Fig. S2).
Out of 1,303,138 novel SNPs, 439,192 and 375,162 were private either for the Lean or the Fat line, respectively, while 488,784 were present in both lines. In total, 180 novel variants with the predicted high-impact and 779 deleterious (including low confidence) missense variants were identified (Supplementary Table S2).
From 1,303,138 novel SNPs, 552,539 (Fat: 164,055, Lean: 178,498, Both: 209,986) are located within 20,600 genes involved in biological processes such as localization, response to stimuli, and signalling/signal transduction (Supplementary Fig. S4a). Meanwhile, the predicted 180 high-impact and 779 deleterious missense variants (DMVs) are located within 676 genes primarily involved in immune response (Supplementary Fig. 4b).
Distribution of line-specific SNPs
In the Lean line, most line-specific SNPs were located on chromosome 2 (184,531), followed by chromosomes 7 (162,635) and 15 (154,786). In contrast, in the Fat line, chromosome 6 (154,366) has the most SNPs, followed by chromosomes 1 (140,760) and 13 (136,462) (Supplementary Fig. S5a, b). A total of 36 line-specific and novel SNPs (Fat: 15, Lean: 21) were also identified on chromosome Y, of which one and nine were in the introns of genes Mid1-ps1 and Gm47283 in the Lean line, respectively (Supplementary Fig. S5c).
Genes with the most line-specific SNPs
Sixty-four genes (Lean: 25, Fat: 23) contain at least 3000 SNPs, all within protein coding genes. Two genes with more than 3000 line-specific SNPs were identified in both lines, Macrod2 and Tenm2. In the Lean line genes Csmd3, Erbb4, and Inpp4b had the most SNPs. Meanwhile, in the Fat line, these were Skint5, Exoc4, and Galnt2l. Other genes in the Fat line include Adcy2, Cpq, Ctnna3, Dcc, Fgf14, Gm37013, Hdac9, Lhfpl3, Mast4, Mctp1, Ndst4, Skint6, Slc14a2, Slc9a9, Smyd3, Sntg1, Sox5, and Trpm3, and in the Lean line 5730522E02Rik, Atrnl1, Cdkal1, Diaph3, Edil3, Fstl5, Gabrb3, Gmds, Hcn1, Hs3st4, Immp2l, Oca2, Pak5, Pcsk5, Prkn, Prr16, Slc7a11, Tenm4, and Zfp536 (Supplementary Table S3).
Considering the relative number of SNPs per gene (SNPs/bp), 30 genes (Lean: 12, Fat: 18) had at least four SNPs per 100 bp. Compared to the genes with the highest absolute number of SNPs, the SNP density was highest in pseudogenes and various types of regulatory RNAs. In more detail: gene segment (Ighd2-6), miRNAs (Gm23063, Mir3086, Mir7237), miscellaneous RNA (Gm25403), pseudogenes (AC152418.1, Ear-ps10, Gm15115, Gm34658, Gm37489, Gm43943, Gm47555, Gm4873, Gm49055, Gm9002, Olfr1139-ps1, S100a11-ps), pseudogenic gene segments (Gm43220, Igkv1-136), RNase P RNA gene (Rprl1), rRNAs (Gm23668, n-R5s3), snoRNA gene (Gm24127), and snRNA genes (Gm27385, Gm26449, Gm25785, Gm24725, Gm24582, Gm23511, Gm22828) (Supplementary Table S4).
For two miRNAs, Mir7237 and Mir3086, we explored whether SNPs fall into their seed region, altering their target sequence recognition. Whereas no SNP was within the for Mir7237 seed region, the SNP rs248726381 (T/C) in the Lean line fells into the seed region of mmu-miR-3086-3p (CCAAUGA◊ CCAAUGG) (Fig. S6a). Several biological processes in the Lean line could be affected due to polymorphism in the seed region of this miRNA caused by rs248726381, including histone and protein acetylation (Fig. S6b). Further enrichment analysis of mmu-miR-3086-3p target genes revealed that, in addition to amino acid metabolism and the nervous system, the target genes also participate in fatty acid biosynthesis (Supplementary Fig. S6c).
GERP scores of line-specific SNPs
GERP score of SNPs was then retrieved to reveal potentially functional SNPs. Most SNPs have GERP score between − 1 and 1, however, 276,397 SNPs have GERP score above 2, 1,369 SNPs above 4, and 125 SNPs above 6 (Supplementary Fig. 7). The latter group include 13 SNPs in regulatory elements within or close to 14 genes, and 6 SNPs with predicted moderate or high impact on 6 genes.
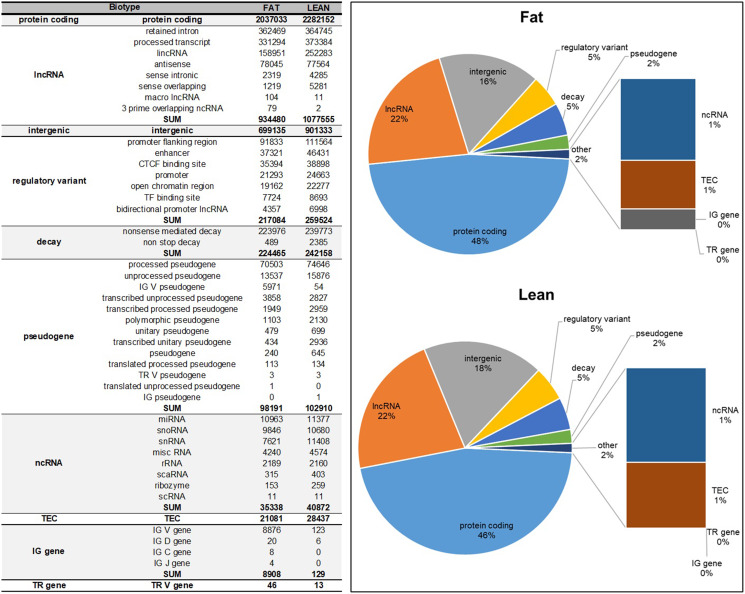
Variant biotype of line-specific SNPs
The most represented line-specific variant biotype was protein coding, followed by lncRNA and intergenic variants. Interestingly, the largest relative difference in the number of SNPs between the lines was IG gene biotype (especially IG V gene) with 8908 SNPs identified in the Fat line and only 129 in the Lean line (Fig. 3).
Fig. 3.
Biotypes and share of line-specific variants identified in the Lean and Fat mouse selection lines
Variant consequences of line-specific SNPs
In both lines, most of the SNPs were intronic variants, followed by intergenic, non-coding transcript, downstream, and upstream variants (Supplementary Fig. S8). In total, 887,371 (Lean) and 778,626 (Fat) line-specific SNPs were located within 13,120 and 13,094 genes, representing 45% and 47% of all the SNPs, respectively.
In the Lean line, synonymous and missense variants accounted for 0.32% and 0.15%, respectively, while in the Fat line they accounted for 0.36% and 0.19% of all SNPs. Among all the missense variants, predicted deleterious variants represented 17.4% and 17.7% in the Lean and Fat lines, respectively. In the Lean line, they were mainly located on chromosomes 2, 7, and 9, while in the Fat line they were found on chromosomes 7, 4, and 6 (Supplementary Fig. S9).
We next investigated if predicted DMVs were located within genes related to obesity (abnormal adipose tissue amount - IMPC). In both mice, line-specific DMVs within several obesity-related genes were identified (Fat: 11, Lean: 12). In addition, 24 genes had shared DMVs (Supplementary Table S5). Among the DMVs, nine SNPs within nine genes were newly identified: 12_13433013_T/A in Nbas, 6_128327592_C/T in Tulp3, and 7_77124609_C/T in Agbl1 of the Fat line, 15_48791841_G/C in Csmd3, 18_59409565_G/T in Chsy3, and 6_95117339_C/T in Kbtbd8 of the Lean line, and 11_87874953_A/G in Epx, 16_35824901_G/A in Hspbap1, and 8_84872251_G/A in Syce2 of both lines. Other obesity genes in the Fat line include 2210408I21Rik, Cep250, Fam81b, Il6st, Mamld1, Nbas, Pth1r, Sema4d, and Slco1b2. Meanwhile, in the Lean line these are Alg8, Alpk2, Aspm, D430041D05Rik, D630045J12Rik, Dock9, Gpr15, Phldb1, and Zfp462. Worth mentioning is Csmd3, a gene with the highest number of SNPs in the Lean line (shown in Supplementary Table S3).
Enrichment analysis revealed that in both lines the genes with line-specific DMVs are involved in the following pathways: graft-versus-host disease, type I diabetes mellitus, and allograft rejection. Other pathways in the Fat line included antigen processing and presentation, serotonergic synapse, viral myocarditis, and asthma. In contrast, genes with Lean line-specific DMVs were involved in cell adhesion molecules, autoimmune thyroid disease, and inflammatory bowel disease. Interestingly, in both lines, the genes mostly related to pathways potentially involved in food perception attained the lowest p-value; olfactory transduction in the Lean line and taste transduction in the Fat line (Supplementary Fig. S10).
For the taste and olfactory transduction pathways, we then explored whether the genes with DMVs map to previously identified obesity/leanness QTLs. While Plcb2, a gene involved in the taste transduction, locate within the Fob1 QTL, 26 out of 75 genes of olfactory transduction are within Fob1 QTL: Or4b1d, Or5d47, Or5aq7, Or9g4b, Or5m12, Or5m13b, Or8k1, Or8k20, Or8k24, Or8k28, Or8k32, Or8h9, Or8h10, Or5j1, Or5aq6, Or5w10, Or10ag59, Or8w1, Or5w1b, Or5w13, Or5w15, Or5w17, Or4a2, Or4a69, Or4a74, and Or4f57.
We than analyzed the density of line-specific missense variants in protein-coding transcripts to identify proteins exhibiting significant differences between the lines. We found 38 transcripts (Fat line: 28, Lean lin: 10) that had an average density of missense variants at intervals of fewer than 25 amino acids. These transcripts correspond to 32 genes (Fat line: 21, Lean line: 10, and 1 common to both). Notably, the genes associated with the highest density of missense variants in the Fat line include Cx3cl1, Nlrp1b, Tas2r136, Cd22, and Or5p58. Meanwhile, in the Lean line these are Hbb-bh2, Kcnmb2, Ang5, Or5w17, and Cbr1b, as detailed in Supplementary Table S6. Interestingly, Hamp2 appears in the top five for both lines, albeit represented by different transcripts (ENSMUST00000205641 in the Fat line and ENSMUST00000109753 in the Lean line). Additionally, the genes Skint5 and Macrod2 were identified as having a notably high number of variants, as shown in Supplementary Table S3.
Eighteen of these genes are involved in 35 different KEGG pathways (Supplementary Table S7). However, three genes are involved in the majority of these pathways: Cx3cl1 (immune and inflammatory reactions) and H2-Aa and H2-Ab1 (various disease, immune system, and type I diabetes mellitus). Worth mentioning are eight genes potentially involved in food perception, among which Or5w17 and Or8k28 are within FOB QTL Fob1. Another gene in FOB QTL is Tmsb15b1. In addition, Kcnmb2 and Nlrp1b are involved in insulin secretion and NOD-like receptor signalling pathway, respectively.
Annotation of candidate genes
Genes with regulatory variants and variants with moderate or high impact with the GERP score above 6 were further analysed and their annotation related to obesity retrieved from various sources. In total, 14 genes have 13 regulatory SNPs, and 6 genes have 6 SNPs with predicted moderate or high impact. These 20 genes include three lncRNAs (4930441H08Rik, 4930595O18Rik, Gm36633), one polymorphic pseudogene (Or56b2j), 15 protein coding genes (Aff3, Angpt1, Atpsckmt, Cpped1, Erc2, Gfra1, Fam237b, Mast4, Pced1a, Prr5l, Serpine2, Tecrl, Tmem132d, Trim24, Zfp536), and one pseudogene (Gm17131). Two genes are within FOB QTL (Pced1a and Prr5l). Gfra1, Or56b2j, Serpine2, and Tecrl were the only genes previously annotated with obesity-related traits. In addition to the above-mentioned Gfra1, Prr5l, and Serpine2, differential expression was also measured for Angpt1, Tmem132d, and Trim24.
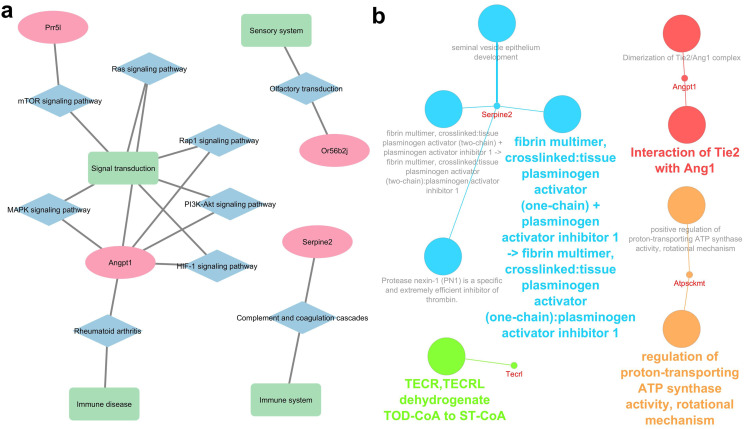
Out of 20 genes only four are annotated in the KEGG database: Angpt1 being involved in various signalling pathways, Prr5l in mTOR signalling pathway, Serpine2 in immune system, and Or56b2j in sensory (olfactory) system (Fig. 4a). The signalling function of ANGPT1 protein might be due to the interaction with TIE2 (TEK receptor tyrosine kinase). ATPSCKMT is a positive regulator v ATP synthase activity, and TECRL catalyses trans-octadec-2-enoyl-CoA to stearyl-CoA (Fig. 4b).
Fig. 4.
Biological function of the genes carrying SNPs with GERP score above 6 located in regulatory regions or having predicted moderate or high impact on protein function identified in the Fat and Lean mouse selection lines. a) KEGG ontology terms, b) biological function of identified genes
Finally, the potential involvement of 19 identified SNPs within 20 genes in obesity was validated by comparing alleles between our mice lines and obesity prone NZO/HlLtJ and obesity resistant A/J strain. The analysis revealed four missense SNPs and 2 regulatory variants within 7 genes (4930441H08Rik, Aff3, Fam237b, Gm36633, Pced1a, Tecrl, Zfp536) (Table 2).
Table 2.
Allele comparison between fat lines (our Fat line and NZO/HlLtJ) and lean lines (our Lean line and A/J)
| Gene symbol | SNP | PHENOTYPE | Consequence | |||
|---|---|---|---|---|---|---|
| FAT | LEAN | |||||
| Fat (FLI) line | NZO/HILtJ | Lean (FHI) line | A/J | |||
| Pced1a | rs33645896 | A | A | G | G | missense variant |
| Tecrl | rs31632835 | C | C | A | A | missense variant |
| Aff3 | rs30511899 | T | T | G | G | missense variant |
| Fam237b | rs31007064 | T | T | C | C | missense variant |
| Zfp536 | rs46750862 | G | G | A | A | promoter flanking region |
| 4930441H08Rik, Gm36633 | rs31031645 | A | A | G | G | promoter flanking region |
Discussion
Obesity has become one of the serious health challenges worldwide. Since 1975, the number of obese individuals has tripled (Vaamonde and Álvarez-Món 2020). With advances in DNA/RNA sequencing technologies, several genes and genetic variants responsible for monogenic and polygenic forms of obesity have been identified, as well as strategies for obesity treatment (Hinney et al. 2010; Ginete et al. 2021; Loos and Yeo 2022). In this study, we delve into this intricate genetic landscape using WGS to explore SNPs in two distinct unique mouse models, representing the polygenic nature of obesity and its counterpart, leanness. The study not only expands our understanding of the genetic basis of obesity and proposes several candidate genes but also paves the way for developing novel or more effective treatments and interventions.
The WGS analysis identified 6,309,795 SNPs (Lean: 4,651,068, Fat: 4,320,310, Both: 2,661,583), of which 1,303,138 were newly identified, primarily involved in localization, response to stimulus, and signalling. The highest number of SNPs was identified within protein-coding genes, two of which had more than 3000 line-specific SNPs in both lines, Macrod2 (mono-ADP ribosylhydrolase 2) and Tenm2 (teneurin transmembrane protein 2). In the Lean line, Csmd3 (CUB and Sushi multiple domains 3), Erbb4 (erb-b2 receptor tyrosine kinase 4), and Inpp4b (inositol polyphosphate-4-phosphatase, type II) genes had the most SNPs, while in the Fat line, these were Skint5 (selection and upkeep of intraepithelial T cells 5), Exoc4 (exocyst complex component 4), and Galnt2l (polypeptide N-acetylgalactosaminyltransferase 2-like). Both Macrod2 and Tenm2 have been implicated in adipogenesis (Tews et al. 2017; Chang et al. 2018). Significant associations were observed between morbid obese Han Chinese and CSMD3 (obesity related, IMPC) and ERBB4 (Chiang et al. 2019). INPP4B protects against metabolic syndrome and associated disorders (Zhang et al. 2021) and EXOC4 is involved in insulin-stimulated glucose transport and is associated with type II diabetes and fasting glucose (Laramie et al. 2008). There is no scientific literature available for the mouse gene Galnt2l and no ortholog has been detected so far in human. However, according to the Ensembl database Galnt2l shares 98.69% similarity with Galnt2. Loss of function of GALNT2 lowers high-density lipoproteins in different species (Khetarpal et al. 2016) and is associated with metabolic diseases, including obesity (Antonucci et al. 2022). Worth mentioning are also two genes that are located within the Fob2 QTL on chromosome 12; Hdac9 (histone deacetylase 9) and Immp2l (IMP2 inner mitochondrial membrane peptidase-like (S. cerevisiae). Global deletion of Hdac9 protects against high-fat diet (HFD) induced obesity and metabolic disease in mice (Chatterjee et al. 2014), and Immp2l is associated with food intake (Han et al. 2013). Another potential candidate gene is Pcsk5 (proprotein convertase subtilisin/kexin type 5). In total, 4587 SNPs were identified in the gene (Lean: 3497, Fat: 617, Both: 473). More importantly, 26 SNPs in the Fat line are shared with other obese mice strains (AKR/J, NZO/HILtJ) but not with the lean lines (A/J, BALB/cJ, FVB/NJ, CAST/EiJ). PCSK5 locus was associated with serum low- and high-density lipoprotein (LDL and HDL) and cholesterol levels (Iatan et al. 2009; De Vries et al. 2019). Moreover, PCSK5 might be a potential target to lower LDL levels (De Vries et al. 2019) and valuable for the ANGPTL4 (angiopoietin like 4)-mediated target therapy of diabetes and metabolic syndrome (Li and Teng 2014). Therefore, the Pcsk5 might be a potential obesity candidate gene to be explored in the future.
Considering the relative number of SNPs per gene (SNPs/bp), the SNP density was the highest in pseudogenes and various types of regulatory RNAs, which is consistent with previous findings (Balasubramanian et al. 2002; Yang et al. 2017b). This could be because pseudogenes and regulatory RNAs are subjected to less selection pressure at the sequence level compared to the protein-coding genes that are evolutionarily more highly conserved (Balasubramanian et al. 2002; Yang et al. 2017b). Although having been labelled as “junk” DNA, the potential functions of pseudogenes in the expression regulation of protein-coding genes have been described, mainly acting as small interfering RNAs or miRNA scavengers (Pink et al. 2011). Among the genes with at least four line-specific SNPs per 100 bp identified in the Lean and Fat lines, three genes were located in the Fob2 QTL interval: snRNA Gm26449, and pseudogenes S100a11-ps (S100 calcium binding protein A11, pseudogene) and Gm47555, and one in the Fob1 QTL interval: pseudogene Olfr1139-ps1 (olfactory receptor 1139, pseudogene 1). Based on the Ensembl database, Gm26449 is part of the spliceosomal small nuclear ribonucleoprotein complexes, and an orthologue of U6, suggesting differential pre-mRNA processing between the two lines. The function of the other three genes is currently unknown.
The SNPs in miRNAs may affect the miRNA functions by altering miRNA maturation, structure, expression, and target binding and, consequently, contribute to phenotype and disease susceptibility (Titov and Vorozheykin 2018). In the present study, the SNP rs248726381 identified in the Lean line locates within the seed region of mmu-miR-3086-3p, potentially affecting histone and protein acetylation modifications. Both, histone and protein acetylation regulate obesity (Xu et al. 2019). The genes targeted by mmu-miR-3086-3p are also involved in fatty acid biosynthesis, making mmu-miR-3086-3p and its polymorphism of interest for the future research. In our previous study (Kunej et al. 2010), a SNP within the seed region of obesity-related mmu-miR-717 was identified in the lean mouse strain 129/Sv. The presence of SNP (rs30372501) within the seed region of this miRNA was also identified in the present study, however, it is present in both lines. Future work might focus on the potential polymorphisms within the binding sites on its target genes.
In addition to the above, identified SNPs and indels in the introns of the pseudogene Mid1-ps1 (midline 1, pseudogene 1) and the long non-coding RNA Gm47283 are also worth mentioning. For the Mid1-ps1, one SNP was identified in the Lean line and four were shared by both. For the Gm47283, four SNPs and several indels were identified in both lines, however, nine were specific to the Lean line. Interestingly, all variants in this gene were located within the same intron and overlap various regulatory elements (Supplementary Fig. S11). Introns play an important role in gene regulation and expression, and therefore SNPs in introns can potentially affect the phenotype and cause disease (Salih et al. 2021). The two genes, Mid1-ps1 and Gm47283 (also known as Erdr1 (erythroid differentiation regulator 1) (Merkenschlager et al. 2019), are within the pseudoautosomal region (PAR) of the sex chromosome Y (Yamazaki et al. 2021). Although PAR genes should behave like autosomal genes, the expression of both genes was unexpectedly higher in the XY oocytes than in XX and XO oocytes (Yamazaki et al. 2021). Importantly, the Fat and Lean lines average less than four pups per litter with other reproductive parameters (i.e. time from mate to litter) also being at the lower end compared to other standard inbred strains. Our preliminary analysis (Pirman et al. 2022) suggests that a large negative effect on reproduction may come from the males. Since Erdr1 knockout has been shown to result in the embryonic lethality in mice (Zuo et al. 2017), and Mid1 regulates mTOR (mammalian target of rapamycin) signalling (Liu et al. 2011), which is required for embryonic development (Gangloff et al. 2004), the identified genetic variants on chromosome Y in genes Gm47283 and Mid1-ps1 may be functionally important for the reproductive performance of the two lines and are worth exploring in further functional analyses.
In total, 2826 predicted DMVs have been identified (Lean: 843, Fat: 991, Both: 992), which are also present in several obesity-related genes (IMPC). Among these are nine novel DMVs within nine genes: Agbl1 (ATP/GTP binding protein-like 1), Chsy3 (chondroitin sulfate synthase 3), Csmd3 (the aforementioned gene with the most line-specific SNPs), Nbas (neuroblastoma amplified sequence), Epx (eosinophil peroxidase), Hspbap1 (Hspb associated protein 1), Kbtbd8 (kelch repeat and BTB (POZ) domain containing 8), Syce2 (synaptonemal complex central element protein 2), and Tulp3 (tubby-like protein 3). Moreover, DMVs were also found within the “obesity” genes located in FOB QTLs: D430041D05Rik and Frmd5 (FERM domain containing 5) in Fob1, the aforementioned Nbas in Fob2, and Mamld1 (mastermind-like domain containing 1) in Fob4. Human AGBL1 was associated with levels of circulating adiponectin, a hormone released from adipose tissue that affects insulin sensitivity and inflammatory patterns, and linked to body mass index, visceral fat, and risk of type 2 diabetes mellitus (Hasegawa et al. 2022). EPX has both anti-inflammatory and pro-inflammatory effects and its expression increases in obesity (Yi et al. 2021). A histone demethylase HSPBAP1 may be involved in regulating stress responses in cells by inhibiting HSP27 (Cloos et al. 2008), which has been suggested to prevent obesity-induced insulin resistance (McCarty 2006). KBTBD8, a gene involved in cell mitosis, has been associated with growth, particularly idiopathic short stature in a Korean population (Kim et al. 2010). Importantly, the novel DMV 6_95117339_C/T in the Lean line is also present in other lean mice strains (A/J, BALB/cJ, FVB/NJ, CAST/EiJ) but not in the obese lines (AKR/J, NZO/HILtJ), suggesting this DMV to be functionally important and may contribute to the lean phenotype. Recently, TULP3 has been demonstrated to be required for the transport of G protein-coupled receptors to primary cilia, and its knockdown impaired ciliary FFAR4 (free fatty acid receptor 4) and PTGER4 (prostaglandin e receptor 4) localization and regulated glucagon or insulin secretion (Wu et al. 2021). As for the genes in FOB QTLs, variant rs524908 in human FRMD5 was linked to the serum lipid profile, such as low-density lipoprotein cholesterol and apolipoprotein B levels (Guo et al. 2017). Considering the previous paragraph, it is important to note that Mamld1 mediates testosterone production for male sex development (Nakamura et al. 2011), and pathogenic variants within MAMLD1 are associated with 46,XY differences/disorders of sex development, with genital abnormalities at birth and possibly associated with age-dependent deterioration of testicular function (Miyado et al. 2022). In addition, MAMLD1 is one of the candidate genes for early-onset obesity (Pettersson et al. 2017), linking the genetic basis of obesity with reduced reproductive performance.
KEGG pathway enrichment analysis revealed that genes carrying DMVs are enriched in various pathways. While the pathways graft-versus-host disease, type I diabetes mellitus, and allograft rejection were common to both lines, other, line-specific pathways were also identified, such as taste transduction, antigen processing and presentation, and serotonergic synapse for the Fat line, and olfactory transduction, cell adhesion molecules, autoimmune thyroid disease, and inflammatory bowel disease for the Lean line. These results suggest that the two lines have different taste, olfaction, and immune signalling. For example, graft-versus-host disease and allograft rejection pathways are enhanced by obesity (Molinero et al. 2016; Khuat et al. 2020). Given the available literature (Lei et al. 2016; Zuo and Ng 2018; Khuat et al. 2020), the two pathways and the inflammatory bowel disease pathway could indicate that the Fat and Lean lines have distinct gut microbiomes and environments that may affect nutrient metabolism and energy expenditure (Aoun et al. 2020). Moreover, the enrichment of genes carrying DMVs in the antigen processing and presentation pathway of the Fat line is consistent with the notion that the Fat line has more SNPs in genes encoding different immunoglobulin chains compared to the Lean line. According to Winer et al. (2014) and Majdoubi et al. (2016) (Winer et al. 2014; Majdoubi et al. 2016) these findings may indicate alterations in B cell function, inflammation, and insulin resistance in the Fat line. Interestingly, the pathways involved in food perception were the most enriched in both lines; taste transduction in the Fat line, and olfactory transduction in the Lean line. Recent studies have shown that food perception alone can induce metabolic changes (Brandt et al. 2018; Kaplan et al. 2018). Hypothalamus is at the crossroad of olfactory and gustatory sensing and regulation of food intake and energy homeostasis (Chao et al. 2016; Faour et al. 2022). In our preliminary study on the hypothalamic transcriptome difference of the two mouse lines fed by a normal chow 148 differentially expressed genes (adjusted p < 0.05, fold change ≥ 1.5) were identified, including four involved in olfactory perception: Or10h1b, Or5v1b, Or52n4, and Or5m3b (unpublished data). In this study, 26 out of 75 genes of olfactory transduction carrying DMVs in the Lean line are within Fob1 QTL (Horvat et al. 2000). Moreover, interestingly, the Fat line carries DMVs in 12 receptors for bitter taste and downstream signalling component for bitter/sweet/umami taste, Plcb2 (phospholipase C, beta 2) (Behrens and Meyerhof 2006). Furthermore, a novel DMV 17_37050960_C/T in the Fat line is also within Gabbr1 (gamma-aminobutyric acid (GABA) B receptor, 1), a gene associated with conditioned taste aversion (Jacobson et al. 2006). In addition, our whole transcript termini site sequencing (Mikec et al. 2023) revealed differential usage of polyadenylation sites in the 3’UTR of this gene between the two lines. In short, these results are suggesting that food perception might contribute to the obese/lean phenotype of our mouse models and require further functional examinations to identify the candidate genes.
Thirty-eight transcripts (Fat line: 28, Lean line: 10) have an average density of missense variants at intervals of fewer than 25 amino acids. Among the genes coding for these proteins are also Or5w17, Or8k28, and Tmsb15b1 located within FOB QTLs. The olfactory receptors have already been discussed above. Meanwhile, thymosin beta 15 (Tmsb15b1) plays an important role in maintaining the dynamic balance of actin, angiogenesis, axonal formation, and wound healing. Recent evidence suggests it to participate in the differentiation and function of thymic epithelial cells, which play an important role in in the thymus response to external stimuli and production of mature T cells (Xu et al. 2022). According to Park and Shastri (2022), the potential imbalance of thymocyte differentiation in the Fat line may contribute to its obese phenotype. Another gene also involved in angiogenesis is Ang5 (angiogenin, ribonuclease A family, member 5). Angiogenic factors, including angiogenin, were associated with adipose tissue remodelling and development of obesity (Kurki et al. 2012).
Several other genes with high density of missense variants are involved in inflammation and immune response, such as Cx3cl1, H2-Aa, H2-Ab1, Fkbpl, Mok, Cd22, Nlrp1b and members of selection and upkeep of intraepithelial T cells (Skint1, Skint5, Skint8). For example, microglial CX3CR1 (C-X3-C motif chemokine ligand 1) signalling determine obesity susceptibility in mice (Dorfman et al. 2017) and its deficiency induces inflammation and insulin resistance in adipose tissue (Nagashimada et al. 2021). In addition, this signalling affects hypothalamic POMC neuronal excitability and melanocortin system activity (Banerjee et al. 2022). These results make Cx3cr and its polymorphisms in the Fat line strong obesity candidates. Meanwhile, CD22 (CD22 antigen) was suggested as potential therapeutic target in hypertensive overweight and obese adults (Liao et al. 2022).
WGS revealed high density of missense variants in different transcripts of Hamp2 (hepcidin antimicrobial peptide 2) between the two lines. Hepcidin (a master regulator of iron metabolism) induction by human symbiont Bacteroides thetaiotaomicron was demonstrated to increase fat deposition by supressing the aforementioned lipoprotein lipase inhibitor ANGPTL4 in the small intestine (Cho et al. 2022). Hepcidin also affects iron acquisition for hemoglobin synthesis (González-Domínguez et al. 2020). Interestingly, in the Lean line, the highest density of missense variants was observed in Hbb-bh2 (hemoglobin beta, bh2), suggesting distinct iron metabolism between the two lines may contribute to their divergent phenotypes. In the Fat line, two transcripts of Nlrp1b (NLR family, pyrin domain containing 1B) were among those with the highest density of missense variants. Mice deactivated for NLRP1 inflammasome were shown to spontaneously develop obesity due to the decreased IL-18 production and lipolysis (Murphy et al. 2016). A metabolic enzyme CBR1B (carbonyl reductase 1B) amplifies glucocorticoid action in adipose tissue and impairs glucose tolerance in mice (Bell et al. 2021). Considering missense variants within Cbr1b were identified in the Lean line, Cbr1b deficiency may contribute to the lean phenotype.
The GERP score is defined as the reduction in the number of substitutions in a multi-species sequence alignment compared to what’s expected under neutral evolution. High GERP scores suggest that mutations in these conserved areas are likely harmful, impacting the species’ genetic health, and GERP scores between 4 and 6 were defined to have “large” deleterious effects (Huber et al. 2020). We annotated the genes with regulatory variants and variants with moderate or high impact with the GERP score above six (Table 1). In total, 14 genes have 13 regulatory SNPs, and 6 genes have 6 SNPs with predicted moderate or high impact. These 20 genes are 4930441H08Rik, 4930595O18Rik, Aff3, Angpt1, Atpsckmt, Cpped1, Erc2, Gfra1, Gm17131, Gm36633, Fam237b, Mast4, Or56b2j, Pced1a, Prr5l, Serpine2, Tecrl, Tmem132d, Trim24, and Zfp536. Of these, only four have been annotated with obesity-related ontologies: Gfra1, Or56b2j, Serpine2, and Tecrl. The remaining 16 might represent novel obesity candidates, especially 4930441H08Rik, Aff3, Fam237b, Gm36633, Pced1a, and Zfp536 with alleles differing between fat (our Fat line and NZO/HlLtJ) and lean lines (our Lean line and A/J).
Table 1.
Prioritization of candidate genes. Genes carrying SNPs with GERP score above 6 located in regulatory regions or having a predicted moderate or high impact on protein function
| Gene symbol | Gene name | Gene biotype | Regulatory SNP | Moderate or high impact SNPs | FOB QTL | IMPC2 | Gene ontology3 | Mammalian phenotype4 | Human disease5 | Expression6 | High number of SNPs7 | Validated by MGI8 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 4930441H08Rik | RIKEN cDNA 4930441H08 gene | lncRNA gene | L:1 PFR, F:0 | - | ✓ | |||||||
| 4930595O18Rik | RIKEN cDNA 4930595O18 gene | lncRNA gene | L:1 PFR, F:0 | - | ||||||||
| Aff3 | AF4/FMR2 family, member 3 | protein coding gene | - | L:1, F:0 | ✓ | |||||||
| Angpt1 | angiopoietin 1 | protein coding gene | L:1 TFBS, F:0 | - | ✓ | |||||||
| Atpsckmt | ATP synthase C subunit lysine N-methyltransferase | protein coding gene | L:0, F:1 TFBS | - | ||||||||
| Cpped1 | calcineurin-like phosphoesterase domain containing 1 | protein coding gene | L:1 PFR, F:0 | - | ||||||||
| Erc2 | ELKS/RAB6-interacting/CAST family member 2 | protein coding gene | L:0, F:1 E | - | ||||||||
| Gfra1 | glial cell line derived neurotrophic factor family receptor alpha 1 | protein coding gene | L:1 E, F:0 | - | ✓ | ✓ | ||||||
| Gm17131 | predicted gene 17,131 | pseudogene | L:0, F:1 PFR and CTCFBS | - | ||||||||
| Gm36633 | predicted gene, 36,633 | lncRNA gene | L:1 PFR, F:0 | - | ✓ | |||||||
| Fam237b | family with sequence similarity 237, member B | protein coding gene | - | L:1, F:0 | ✓ | |||||||
| Mast4 | microtubule associated serine/threonine kinase family member 4 | protein coding gene | L:1 PFR, F:1 PFR | - | ✓ | |||||||
| Or56b2j | olfactory receptor family 56 subfamily B member 2 J | polymorphic pseudogene | - | L:0, F:1 | ✓ | |||||||
| Pced1a | PC-esterase domain containing 1 A | protein coding gene | - | L:0, F:1 | ✓ | ✓ | ||||||
| Prr5l | proline rich 5 like | protein coding gene | L:1 PFR and OCR, F:0 | - | ✓ | ✓ | ||||||
| Serpine2 | serine (or cysteine) peptidase inhibitor, clade E, member 2 | protein coding gene | L:1 P, F:0 | - | ✓ | ✓ | ||||||
| Tecrl | trans-2,3-enoyl-CoA reductase-like | protein coding gene | - | L:0, F:1 | ✓ | ✓ | ||||||
| Tmem132d | transmembrane protein 132D | protein coding gene | L:0, F:1 PFR and CTCFBS | - | ✓ | |||||||
| Trim24 | tripartite motif-containing 24 | protein coding gene | - | L:1, F:0 | ✓ | |||||||
| Zfp536 | zinc finger protein 536 | protein coding gene | L:1 PFR, F:0 | - | ✓ | ✓ |
1 - E: enhancer, CTCFBS: CTCF binding site, P: promoter, PFR: promoter flanking region, TFBS: transcription factor binding site
2 - search term: abnormal adipose tissue amount
3 - search keywords: adipo, cholesterol, digestion, eating, energy, fat, feeding, glucose, glycogen, insulin, lean, leptin, lipid, lipoprotein, obese, olfactory, phagy, smell, storage, taste, thermogenesis
4 - search keywords: adipose, cholesterol, digestion, eating, energy, fat, fatigue, feeding, glucose, glycogen, insulin, lean, leptin, lipid, lipoprotein, obese, olfactory, phagy, smell, storage, taste, thermogenesis
5 - search keywords: cardio, diabetes, dystrophy, eating, feeding, glycemia, glycogen, heart, hypertension, insulin, lean, leptin, lipidemia, lipodystrophy, liver, muscular, obesity, olfactory, osteoarthritis, phagy, smell, taste, thermogenesis
6 - Differential gene expression (Microarray or RNA-seq)
7 - Supplementary Table S3
8 - SNP is present in Fat line and obese mouse NZO/HlLtJ or present in the Lean line and obesity resistant A/J mouse line
GFRA1 receptor (glial cell line derived neurotrophic factor family receptor alpha 1) in human was identified as the mediator for the anorectic effects of the protein GDF15 (growth differentiation factor 15), which has shown potential in decreasing food intake and body weight in obese animal models (Yang et al. 2017a), GDF15–GFRAL system acting independently of the GLP1 (glucagon-like peptide-1) and leptin pathways (Cimino et al. 2017). No relevant scientific data are available for Or56b2j (olfactory receptor family 56 subfamily B member 2 J), however, a study by Choquette et al. (2012) suggests that variations in human olfactory receptor genes can influence eating behaviours and adiposity. In addition, deletion of olfactory receptor-rich region 11q11 was identified as a risk factor for obesity (Diels et al. 2020). SERPINE2 (serine (or cysteine) peptidase inhibitor, clade E, member 2) pathway is evolutionary conserved and involved in tumorigenesis (Shen et al. 2017). Moreover, SERPINE2 was one of the genes most differentially methylated and expressed between non-obese and obese individuals in human adipose tissue (Keller et al. 2017) and recognized as a potential biomarker of obesity-induced alteration in placenta development (Altmäe et al. 2017). TECRL (trans-2,3-enoyl-CoA reductase-like) deficiency is associated with aberrant mitochondrial function in cardiomyocytes (Hou et al. 2022), due to the alterations in fatty acid metabolism and consequent electrophysiological anomalies (Gelinas et al. 2017). TECRL was identified as a new life-threatening inherited arrhythmia gene associated with overlapping clinical features of both long QT syndrome and catecholaminergic polymorphic ventricular tachycardia (Devalla et al. 2016). A missense variant rs31632835 in the Fat line (as well in NZO/HILtJ) may therefore account for its lower physical activity compared to the Lean line observed by Simončič et al. (2008).
In addition to Tecrl, MGI validation uncovered other genes: 4930441H08Rik, Aff3, Fam237b, Gm36633, Pced1a, and Zfp536. No scientific literature is available for 4930441H08Rik. However, among the top ten predicted targets of 4930441H08Rik by LncRRIsearch tool include genes potentially involved in obesity: Hal (histidine ammonia lyase) (Böhm et al. 2014; Ren et al. 2016), H19 (H19, imprinted maternally expressed transcript) (Schmidt et al. 2018; Wang et al. 2021a), Igf2 (insulin-like growth factor 2) (Queiroz et al. 2015; Ács et al. 2017), and lncRNA Nova2os (NOVA alternative splicing regulator 2, opposite strand sequence) (Mikec et al. 2022). AFF3 (AF4/FMR2 family, member 3) was recognized as a master regulator of metabolic inflexibility in type 2 diabetes (Son et al. 2019) and was associated with serum lipid alterations and blood triglyceride levels (Li et al. 2015). Perhaps very interesting is Fam237b (family with sequence similarity 237, member B) that encodes an orexigenic hormone microprotein (Martinez et al. 2023). Methylation of PCED1A (PC-esterase domain containing 1 A) in human was affected by dietary fat quality (Voisin et al. 2015), and PCED1A might affect cellular rearrangement during adipocyte expansion in muscles (Silva-Vignato et al. 2022). Transcription factor ZFP536 (zinc finger protein 536) is involved in neurone differentiation (Qin et al. 2009) and was associated with both Attention-Deficit/Hyperactivity Disorder (ADHD) and excessive body weight (Dmitrzak-Weglarz et al. 2021), linking psychological disorders with obesity.
Other candidate genes include 4930595O18Rik, Angpt1, Atpsckmt, Cpped1, Erc2, Gm17131, Gm36633, Mast4, Prr5l, Tmem132d, and Trim24. As for 4930441H08Rik, no relevant information exists for 4930595O18Rik. Interestingly though, as 4930441H08Rik, it is predicted to target both Hal and not-yet mentioned Fam98b that is involved in liver fibrosis (Pazo et al. 2019), cancer (Akter et al. 2017), and activation of mRNA translation (Pazo et al. 2019). Among the top targets involved in obesity include Pde6h (phosphodiesterase 6 H, cGMP-specific, cone, gamma) (Sosa-Madrid et al. 2020), Lrp5 (low density lipoprotein receptor-related protein 5) (Guo et al. 2006), potentially lncRNA Gm37985 that is predicted to target Hif3a (hypoxia inducible factor 3, alpha subunit) (Mikec et al. 2022), and circRNA Arf3 (ADP-ribosylation factor 3) (Rashad et al. 2023). Interestingly, as hypoxia is associated with obesity, angiogenesis marker ANGPT1 (angiopoietin 1) correlates positively with adipocyte size and body mass index-standard deviation score (BMI-SDS), a measure to define childhood obesity (Gaebler et al. 2019). CPPED1 (calcineurin-like phosphoesterase domain containing 1) expression negatively affect glucose metabolism in human adipocytes (Vaittinen et al. 2013). ERC2 (ELKS/RAB6-interacting/CAST family member 2) is responsible for neurotransmitter release at inhibitory synapses (Kaeser et al. 2009) and was associated with body weight gain and feed conversion ratio in chicken as identified by the GWAS (Marchesi et al. 2021). The lncRNA Gm36633 potentially also targets, among others, the above-mentioned Fam98b and Igf2, suggesting a coordinate role of the identified lncRNAs 4930595O18Rik, 4930441H08Rik, and Gm36633 in obesity.
Moreover, MAST4 (microtubule associated serine/threonine kinase family member 4), involved in neurodevelopment (Zhang et al. 2023), is repressed by glucocorticoids (Nguyen et al. 2022). In addition, a knock-down of numerous microtubule-binding or -associated proteins including MAST4 caused a change in fat accumulation during adipogenesis (Söhle et al. 2012), and higher expression of MAST4 was observed in the fat compared to the lean chicken line (Wang et al. 2021b). PRR5L (proline rich 5 like, also known as Protor-1 or 2) is part of the mTORC2 complex (Pearce et al. 2007), which is involved in various cellular processes including fatty acid and lipid synthesis (Guri et al. 2017). PRR5L expression was found differential in visceral adipose tissue of obesity patients (Guo et al. 2018) and found to be required for efficient mTORC2-mediated activation of SGK1 (serum/glucocorticoid regulated kinase 1) (Pearce et al. 2011). SGK1 is upregulated in obesity (Li et al. 2013; Bapat et al. 2022) and its genetic inhibition prevents obesity-related atrial fibrillation (Bapat et al. 2022). TMEM132D (transmembrane protein 132D) is primarily known for its association with addiction and anxiety (Hodgson et al. 2016). However, more recent studies also linked this gene with metabolic syndrome (Wan et al. 2021) and circadian rhythms (Li and Zhao 2020). Another interesting candidate is Trim24 (tripartite motif-containing 24), an insulin dependent regulator of transcription and ubiquitination-dependent protein degradation (Wei et al. 2022). Wei et al. (2022) showed that TRIM24 interacts with components of processing bodies, which consequently stabilises Pparγ (peroxisome proliferator activated receptor gamma) mRNA (Wei et al. 2022), a master regulator of adipogenesis and obesity (Shao et al. 2016).
Taken together, a significant number of genes carrying potentially impactful variants were identified, including those previously related to obesity, as well as some novel potential candidates. In the present study, we primarily focused on SNPs with GERP score above 6 located in regulatory regions or having potential moderate or high impact on protein function, however, SNPs with lower GERP score and genes with other variant biotypes need to be explored in future studies.
Conclusion
In conclusion, our sequencing and downstream bioinformatics analyses of two unique mouse models for polygenic obesity and healthy leanness revealed numerous genetic differences between the two lines and identified a plethora of genetic variants with potentially negative effects on gene function, including those previously associated with obesity. Given the growing catalogue of genetic polymorphisms in mice, this study provides a valuable resource of candidate genes for researchers to evaluate causality and function in obesity or leanness.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
The authors also want to thank Dr Tim J. Aitman (Centre for Genomic and Experimental Medicine, Institute of Genetics and Molecular Medicine, University of Edinburgh, Edinburgh, United Kingdom) for the help with WGS.
Author contributions
MŠ: Formal analysis, Writing - original draft preparation. ŠM and SSA: Formal analysis. JK: Resources. NMM: Formal analysis. Writing – review & editing. TK and SH: Conceptualization, Writing – review & editing, Supervision.
Funding
The authors acknowledge the study was financially supported by the Slovenian Research and Innovation Agency under the postgraduate research program Young researchers (ŠM), J4-2548 research project and P4-0220 research program.
Data availability
The data are available on request.
Declarations
Ethics approval and consent to participate
The FLI (Fat) and FHI (Lean) selection lines have been maintained in our laboratory for more than 70 generations. All mice used in this study were maintained according to local ethical and EU regulatory guidelines under the Veterinary Administration of Republic of Slovenia permit No. U34401-23/2020/6.
Conflict of interest
All authors have read and approved the final version of the manuscript for submission and declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Martin Šimon, Email: martin.simon@bf.uni-lj.si.
Tanja Kunej, Email: tanja.kunej@bf.uni-lj.si.
References
- Ács O, Péterfia B, Hollósi P, et al. Methylation status of CYP27B1 and IGF2 correlate to BMI SDS in children with obesity. Obes Facts. 2017;10:353–362. doi: 10.1159/000477462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akter KA, Mansour MA, Hyodo T, Senga T. FAM98A associates with DDX1-C14orf166-FAM98B in a novel complex involved in colorectal cancer progression. Int J Biochem Cell Biol. 2017;84:1–13. doi: 10.1016/j.biocel.2016.12.013. [DOI] [PubMed] [Google Scholar]
- Altmäe S, Segura MT, Esteban FJ et al (2017) Maternal pre-pregnancy obesity is associated with altered placental transcriptome. PLoS ONE 12. 10.1371/journal.pone.0169223 [DOI] [PMC free article] [PubMed]
- Andersson L, Georges M. Domestic-animal genomics: deciphering the genetics of complex traits. Nat Rev Genet. 2004;5:202–212. doi: 10.1038/nrg1294. [DOI] [PubMed] [Google Scholar]
- Antonucci A, Marucci A, Trischitta V, Di Paola R. Role of GALNT2 on insulin sensitivity, lipid metabolism and Fat Homeostasis. Int J Mol Sci. 2022;23:929. doi: 10.3390/ijms23020929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aoun A, Darwish F, Hamod N. The influence of the gut microbiome on obesity in adults and the role of probiotifcs prebiotics and synbiotics for weight loss. Prev Nutr Food Sci. 2020;25:113–123. doi: 10.3746/pnf.2020.25.2.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balasubramanian S, Harrison P, Hegyi H, et al. SNPs on human chromosomes 21 and 22 - analysis in terms of protein features and pseudogenes. Pharmacogenomics. 2002;3:393–402. doi: 10.1517/14622416.3.3.393. [DOI] [PubMed] [Google Scholar]
- Banerjee J, Dorfman MD, Fasnacht R et al (2022) CX3CL1 action on Microglia protects from Diet-Induced obesity by restoring POMC neuronal excitability and Melanocortin System Activity impaired by High-Fat Diet Feeding. Int J Mol Sci 23. 10.3390/ijms23126380 [DOI] [PMC free article] [PubMed]
- Bapat A, Li G, Xiao L et al (2022) Genetic inhibition of serum glucocorticoid kinase 1 prevents obesity-related atrial fibrillation. JCI Insight 7. 10.1172/jci.insight.160885 [DOI] [PMC free article] [PubMed]
- Beck JA, Lloyd S, Hafezparast M, et al. Genealogies of mouse inbred strains. Nat Genet. 2000;24:23–25. doi: 10.1038/71641. [DOI] [PubMed] [Google Scholar]
- Behrens M, Meyerhof W (2006) Bitter taste receptors and human bitter taste perception. In: Cellular and Molecular Life Sciences. pp 1501–1509 [DOI] [PMC free article] [PubMed]
- Bell R, Villalobos E, Nixon M, et al. Carbonyl Reductase 1 overexpression in adipose amplifies local glucocorticoid action and impairs glucose tolerance in lean mice. J Endocr Soc. 2021;5:A806–A806. doi: 10.1210/jendso/bvab048.1639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bindea G, Mlecnik B, Hackl H, et al. ClueGO: a Cytoscape plug-in to decipher functionally grouped gene ontology and pathway annotation networks. Bioinformatics. 2009;25:1091–1093. doi: 10.1093/bioinformatics/btp101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birling MC, Yoshiki A, Adams DJ, et al. A resource of targeted mutant mouse lines for 5,061 genes. Nat Genet. 2021;53:416–419. doi: 10.1038/s41588-021-00825-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blüher M. Obesity: global epidemiology and pathogenesis. Nat Rev Endocrinol. 2019;15:288–298. doi: 10.1038/s41574-019-0176-8. [DOI] [PubMed] [Google Scholar]
- Böhm A, Halama A, Meile T et al (2014) Metabolic signatures of cultured human adipocytes from metabolically healthy versus unhealthy obese individuals. PLoS ONE 9. 10.1371/journal.pone.0093148 [DOI] [PMC free article] [PubMed]
- Brandt C, Nolte H, Henschke S, et al. Food Perception primes hepatic ER homeostasis via melanocortin-dependent control of mTOR activation. Cell. 2018;175:1321–1335e20. doi: 10.1016/j.cell.2018.10.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bult CJ, Blake JA, Smith CL, et al. Mouse Genome Database (MGD) 2019. Nucleic Acids Res. 2019;47:D801–D806. doi: 10.1093/nar/gky1056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bünger L, Hill WG. Inbred lines of mice derived from long-term divergent selection on fat content and body weight. Mamm Genome. 1999;10:645–648. doi: 10.1007/s003359901063. [DOI] [PubMed] [Google Scholar]
- Chang YC, Hee SW, Lee WJ, et al. Genome-wide scan for circulating vascular adhesion protein-1 levels: MACROD2 as a potential transcriptional regulator of adipogenesis. J Diabetes Investig. 2018;9:1067–1074. doi: 10.1111/jdi.12805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chao DHM, Argmann C, Van Eijk M, et al. Impact of obesity on taste receptor expression in extra-oral tissues: emphasis on hypothalamus and brainstem. Sci Rep. 2016;6:29094. doi: 10.1038/srep29094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chatterjee TK, Basford JE, Knoll E, et al. HDAC9 knockout mice are protected from adipose tissue dysfunction and systemic metabolic disease during high-fat feeding. Diabetes. 2014;63:176–187. doi: 10.2337/db13-1148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiang KM, Chang HC, Yang HC, et al. Genome-wide association study of morbid obesity in Han Chinese. BMC Genet. 2019;20:97. doi: 10.1186/s12863-019-0797-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cho SH, Cho YJ, Park JH. The human symbiont Bacteroides thetaiotaomicron promotes diet-induced obesity by regulating host lipid metabolism. J Microbiol. 2022;60:118–127. doi: 10.1007/s12275-022-1614-1. [DOI] [PubMed] [Google Scholar]
- Choquette AC, Bouchard L, Drapeau V, et al. Association between olfactory receptor genes, eating behavior traits and adiposity: results from the Quebec Family Study. Physiol Behav. 2012;105:772–776. doi: 10.1016/j.physbeh.2011.10.015. [DOI] [PubMed] [Google Scholar]
- Cimino I, Coll AP, Yeo GSH. GDF15 and energy balance: homing in on a mechanism. Nat Med. 2017;23:1119–1120. doi: 10.1038/nm.4414. [DOI] [PubMed] [Google Scholar]
- Cingolani P, Patel VM, Coon M et al (2012a) Using Drosophila melanogaster as a model for Genotoxic Chemical Mutational Studies with a New Program, SnpSift. 10.3389/fgene.2012.00035. Front Genet 3: [DOI] [PMC free article] [PubMed]
- Cingolani P, Platts A, Wang LL, et al. A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w1118; iso-2; iso-3. Fly (Austin) 2012;6:80–92. doi: 10.4161/fly.19695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cloos PAC, Christensen J, Agger K, Helin K. Erasing the methyl mark: histone demethylases at the center of cellular differentiation and disease. Genes Dev. 2008;22:1115–1140. doi: 10.1101/gad.1652908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Vries PS, Brown MR, Bentley AR, et al. Multiancestry Genome-Wide Association Study of Lipid Levels Incorporating Gene-Alcohol Interactions. Am J Epidemiol. 2019;188:1033–1054. doi: 10.1093/aje/kwz005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Depristo MA, Banks E, Poplin R, et al. A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat Genet. 2011;43:491–501. doi: 10.1038/ng.806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devalla HD, Gélinas R, Aburawi EH, et al. TECRL, a new life-threatening inherited arrhythmia gene associated with overlapping clinical features of both LQTS and CPVT. EMBO Mol Med. 2016;8:1390–1408. doi: 10.15252/emmm.201505719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diels S, Huybreghts S, Van Hoorenbeeck K, et al. Copy number variant analysis and expression profiling of the olfactory receptor-rich 11q11 region in obesity predisposition. Mol Genet Metab Rep. 2020;25:100656. doi: 10.1016/j.ymgmr.2020.100656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dmitrzak-Weglarz M, Paszynska E, Bilska K, et al. Common and unique genetic background between attention-deficit/hyperactivity disorder and excessive body weight. Genes (Basel) 2021 doi: 10.3390/genes12091407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorfman MD, Krull JE, Douglass JD, et al. Sex differences in microglial CX3CR1 signalling determine obesity susceptibility in mice. Nat Commun. 2017;8:14556. doi: 10.1038/ncomms14556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faour M, Magnan C, Gurden H, Martin C. Olfaction in the context of obesity and diabetes: insights from animal models to humans. Neuropharmacology. 2022;206:108923. doi: 10.1016/j.neuropharm.2021.108923. [DOI] [PubMed] [Google Scholar]
- Fukunaga T, Iwakiri J, Ono Y, Hamada M. LncRRIsearch: a web server for lncRNA-RNA Interaction Prediction Integrated with tissue-specific expression and Subcellular Localization Data. Front Genet. 2019;10:462. doi: 10.3389/fgene.2019.00462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaebler N, Haggenmüller B, Kapapa M, et al. Age- and BMI-Associated expression of angiogenic factors in White Adipose tissue of children. Int J Mol Sci. 2019;20:5204. doi: 10.3390/ijms20205204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gangloff Y-G, Mueller M, Dann SG, et al. Disruption of the mouse mTOR gene leads to early postimplantation lethality and prohibits embryonic stem Cell Development. Mol Cell Biol. 2004;24:9508–9516. doi: 10.1128/mcb.24.21.9508-9516.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gelinas R, Goyette P, Forest A, et al. A severe inherited arrhythmia syndrome highlights the role of fatty acid metabolism in the regulation of Cardiac Electrical Activity. FASEB J. 2017 doi: 10.1096/fasebj.31.1_supplement.782.14. [DOI] [Google Scholar]
- Ginete C, Serrasqueiro B, Silva-Nunes J, et al. Identification of genetic variants in 65 obesity related genes in a cohort of Portuguese obese individuals. Genes (Basel) 2021;12:603. doi: 10.3390/genes12040603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- González-Domínguez Á, Visiedo-García FM, Domínguez-Riscart J, et al. Iron metabolism in obesity and metabolic syndrome. Int J Mol Sci. 2020;21:5529. doi: 10.3390/ijms21155529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- González-Muniesa P, Mártinez-González M-A, Hu FB, et al. Obesity. Nat Rev Dis Prim. 2017;3:17034. doi: 10.1038/nrdp.2017.34. [DOI] [PubMed] [Google Scholar]
- Guo Y-f, Xiong D-h, Shen H, et al. Polymorphisms of the low-density lipoprotein receptor-related protein 5 (LRP5) gene are associated with obesity phenotypes in a large family-based association study. J Med Genet. 2006;43:798–803. doi: 10.1136/jmg.2006.041715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo T, Yin RX, Pan L, et al. Integrative variants, haplotypes and diplotypes of the CAPN3 and FRMD5 genes and several environmental exposures associate with serum lipid variables. Sci Rep. 2017;7:45119. doi: 10.1038/srep45119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo Q, Zheng R, Huang J et al (2018) Using integrative analysis of DNA methylation and gene expression data in multiple tissue types to prioritize candidate genes for Drug Development in obesity. Front Genet 9. 10.3389/fgene.2018.00663 [DOI] [PMC free article] [PubMed]
- Guri Y, Colombi M, Dazert E, et al. mTORC2 promotes tumorigenesis via lipid synthesis. Cancer Cell. 2017;32:807–823. doi: 10.1016/j.ccell.2017.11.011. [DOI] [PubMed] [Google Scholar]
- Han C, Zhao Q, Lu B. The role of nitric oxide signaling in food intake; insights from the inner mitochondrial membrane peptidase 2 mutant mice. Redox Biol. 2013;1:498–507. doi: 10.1016/j.redox.2013.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasegawa M, Taniguchi J, Ueda H, Watanabe M. Twin study: genetic and epigenetic factors affecting circulating adiponectin levels. J Clin Endocrinol Metab. 2022;108:144–154. doi: 10.1210/clinem/dgac532. [DOI] [PubMed] [Google Scholar]
- Hinney A, Vogel CIG, Hebebrand J. From monogenic to polygenic obesity: recent advances. Eur Child Adolesc Psychiatry. 2010;19:297–310. doi: 10.1007/s00787-010-0096-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hodgson K, Almasy L, Knowles EEM, et al. Genome-wide significant loci for addiction and anxiety. Eur Psychiatry. 2016;36:47–54. doi: 10.1016/j.eurpsy.2016.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horvat S, Bünger L, Falconer VM, et al. Mapping of obesity QTLs in a cross between mouse lines divergently selected on fat content. Mamm Genome. 2000;11:2–7. doi: 10.1007/s003350010002. [DOI] [PubMed] [Google Scholar]
- Hou C, Jiang X, Zhang H, et al. TECRL deficiency results in aberrant mitochondrial function in cardiomyocytes. Commun Biol. 2022;5:470. doi: 10.1038/s42003-022-03414-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huber CD, Kim BY, Lohmueller KE. Population genetic models of GERP scores suggest pervasive turnover of constrained sites across mammalian evolution. PLoS Genet. 2020;16:e1008827. doi: 10.1371/journal.pgen.1008827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iatan I, Dastani Z, Do R, et al. Genetic variation at the Proprotein Convertase Subtilisin/Kexin type 5 Gene Modulates High-Density Lipoprotein Cholesterol Levels. Circ Cardiovasc Genet. 2009;2:467–475. doi: 10.1161/CIRCGENETICS.109.877811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobson LH, Kelly PH, Bettler B, et al. GABA B(1) receptor isoforms differentially mediate the Acquisition and extinction of aversive taste Memories. J Neurosci. 2006;26:8800–8803. doi: 10.1523/JNEUROSCI.2076-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaeser PS, Deng L, Chávez AE, et al. ELKS2alpha/CAST deletion selectively increases neurotransmitter release at inhibitory synapses. Neuron. 2009;64:227–239. doi: 10.1016/J.NEURON.2009.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanehisa M, Goto S. KEGG: Kyoto Encyclopedia of genes and genomes. Nucleic Acids Res. 2000;28:27–30. doi: 10.1093/nar/28.1.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaplan REW, Webster AK, Chitrakar R, et al. Food perception without ingestion leads to metabolic changes and irreversible developmental arrest in C. Elegans. BMC Biol. 2018;16:112. doi: 10.1186/s12915-018-0579-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kehl T, Kern F, Backes C, et al. MiRPathDB 2.0: a novel release of the miRNA pathway Dictionary Database. Nucleic Acids Res. 2020;48:D142–D147. doi: 10.1093/nar/gkz1022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keller M, Hopp L, Liu X, et al. Genome-wide DNA promoter methylation and transcriptome analysis in human adipose tissue unravels novel candidate genes for obesity. Mol Metab. 2017;6:86–100. doi: 10.1016/j.molmet.2016.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kern F, Aparicio-Puerta E, Li Y, et al. miRTargetLink 2.0—interactive miRNA target gene and target pathway networks. Nucleic Acids Res. 2021;49:W409–W416. doi: 10.1093/nar/gkab297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khetarpal SA, Schjoldager KT, Christoffersen C, et al. Loss of function of GALNT2 lowers high-density lipoproteins in humans, Nonhuman Primates, and rodents. Cell Metab. 2016;24:234–245. doi: 10.1016/j.cmet.2016.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khuat LT, Le CT, Pai C-CS et al (2020) Obesity induces gut microbiota alterations and augments acute graft-versus-host disease after allogeneic stem cell transplantation. Sci Transl Med 12. 10.1126/scitranslmed.aay7713 [DOI] [PMC free article] [PubMed]
- Kim JJ, Lee HI, Park T, et al. Identification of 15 loci influencing height in a Korean population. J Hum Genet. 2010;55:27–31. doi: 10.1038/jhg.2009.116. [DOI] [PubMed] [Google Scholar]
- Kunej T, Skok DJ, Horvat S, et al. The glypican 3-hosted murine Mir717 gene: sequence conservation, seed region polymorphisms and putative targets. Int J Biol Sci. 2010;6:769–772. doi: 10.7150/ijbs.6.769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurki E, Shi J, Martonen E, et al. Distinct effects of calorie restriction on adipose tissue cytokine and angiogenesis profiles in obese and lean mice. Nutr Metab (Lond) 2012;9:64. doi: 10.1186/1743-7075-9-64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laplante M, Horvat S, Festuccia WT, et al. DEPTOR Cell-autonomously promotes adipogenesis, and its expression is Associated with obesity. Cell Metab. 2012;16:202–212. doi: 10.1016/j.cmet.2012.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laramie JM, Wilk JB, Williamson SL, et al. Polymorphisms near EXOC4 and LRGUK on chromosome 7q32 are associated with type 2 diabetes and fasting glucose; the NHLBI Family Heart Study. BMC Med Genet. 2008;9:46. doi: 10.1186/1471-2350-9-46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lei YM, Chen L, Wang Y, et al. The composition of the Microbiota modulates allograft rejection. J Clin Invest. 2016;126:2736–2744. doi: 10.1172/JCI85295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H, Durbin R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics. 2009;25:1754–1760. doi: 10.1093/bioinformatics/btp324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y, Teng C. Angiopoietin-like proteins 3, 4 and 8: regulating lipid metabolism and providing new hope for metabolic syndrome. J Drug Target. 2014;22:679–687. doi: 10.3109/1061186X.2014.928715. [DOI] [PubMed] [Google Scholar]
- Li X, Zhao H. Automated feature extraction from population wearable device data identified novel loci associated with sleep and circadian rhythms. PLOS Genet. 2020;16:e1009089. doi: 10.1371/journal.pgen.1009089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li P, Pan F, Hao Y, et al. SGK1 is regulated by metabolic-related factors in 3T3-L1 adipocytes and overexpressed in the adipose tissue of subjects with obesity and diabetes. Diabetes Res Clin Pract. 2013;102:35–42. doi: 10.1016/j.diabres.2013.08.009. [DOI] [PubMed] [Google Scholar]
- Li C, Bazzano LAL, Rao DC, et al. Genome-wide linkage and Positional Association Analyses Identify Associations of Novel AFF3 and NTM genes with triglycerides: the GenSalt Study. J Genet Genomics. 2015;42:107–117. doi: 10.1016/j.jgg.2015.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liao Y, Wang J, Jaehnig EJ, et al. WebGestalt 2019: gene set analysis toolkit with revamped UIs and APIs. Nucleic Acids Res. 2019;47:W199–W205. doi: 10.1093/nar/gkz401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liao L, Zhang L, Chen H, et al. Identification of key genes from the visceral adipose tissues of Overweight/Obese adults with hypertension through transcriptome sequencing. Cytogenet Genome Res. 2022;162:541–559. doi: 10.1159/000528702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu E, Knutzen CA, Krauss S, et al. Control of mTORC1 signaling by the Opitz syndrome protein MID1. Proc Natl Acad Sci U S A. 2011;108:8680–8685. doi: 10.1073/pnas.1100131108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loos RJF, Yeo GSH. The genetics of obesity: from discovery to biology. Nat Rev Genet. 2022;23:120–133. doi: 10.1038/s41576-021-00414-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu Y-F, Goldstein DB, Angrist M, Cavalleri G. Personalized Medicine and Human Genetic Diversity. Cold Spring Harb Perspect Med. 2014;4:a008581–a008581. doi: 10.1101/cshperspect.a008581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Majdoubi A, Kishta OA, Thibodeau J. Role of antigen presentation in the production of pro-inflammatory cytokines in obese adipose tissue. Cytokine. 2016;82:112–121. doi: 10.1016/j.cyto.2016.01.023. [DOI] [PubMed] [Google Scholar]
- Marchesi JAP, Ono RK, Cantão ME, et al. Exploring the genetic architecture of feed efficiency traits in chickens. Sci Rep. 2021 doi: 10.1038/s41598-021-84125-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez TF, Lyons-Abbott S, Bookout AL, et al. Profiling mouse brown and white adipocytes to identify metabolically relevant small ORFs and functional microproteins. Cell Metab. 2023;35:166–183e11. doi: 10.1016/j.cmet.2022.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCarty MF. Induction of heat shock proteins may combat insulin resistance. Med Hypotheses. 2006;66:527–534. doi: 10.1016/j.mehy.2004.08.033. [DOI] [PubMed] [Google Scholar]
- McKenna A, Hanna M, Banks E, et al. The genome analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 2010;20:1297–1303. doi: 10.1101/gr.107524.110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLaren W, Gil L, Hunt SE, et al. The Ensembl variant effect predictor. Genome Biol. 2016;17:122. doi: 10.1186/s13059-016-0974-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merkenschlager J, Eksmond U, Danelli L, et al. MHC class II cell-autonomously regulates self-renewal and differentiation of normal and malignant B cells. Blood. 2019;133:1108–1118. doi: 10.1182/blood-2018-11-885467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mikec Š, Šimon M, Morton NM et al (2022) Genetic variants of the hypoxia-inducible factor 3 alpha subunit (Hif3a) gene in the Fat and lean mouse selection lines. Mol Biol Rep 1–13. 10.1007/s11033-022-07309-0 [DOI] [PubMed]
- Mikec Š, Horvat S, Wang H, et al. Differential alternative polyadenylation response to high-fat diet between polygenic obese and healthy lean mice. Biochem Biophys Res Commun. 2023;666:83–91. doi: 10.1016/j.bbrc.2023.05.005. [DOI] [PubMed] [Google Scholar]
- Miyado M, Fukami M, Ogata T. MAMLD1 and Differences/Disorders of Sex Development: an update. Sex Dev. 2022;16:126–137. doi: 10.1159/000519298. [DOI] [PubMed] [Google Scholar]
- Molinero LL, Yin D, Lei YM, et al. High-fat diet-induced obesity enhances allograft rejection. Transplantation. 2016;100:1015–1021. doi: 10.1097/TP.0000000000001141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morton NM, Beltram J, Carter RN, et al. Genetic identification of thiosulfate sulfurtransferase as an adipocyte-expressed antidiabetic target in mice selected for leanness. Nat Med. 2016;22:771–779. doi: 10.1038/nm.4115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murphy AJ, Kraakman MJ, Kammoun HL, et al. IL-18 production from the NLRP1 inflammasome prevents obesity and metabolic syndrome. Cell Metab. 2016;23:155–164. doi: 10.1016/j.cmet.2015.09.024. [DOI] [PubMed] [Google Scholar]
- Nagashimada M, Sawamoto K, Ni Y et al (2021) CX3CL1-CX3CR1 Signaling Deficiency exacerbates obesity-induced inflammation and insulin resistance in male mice. Endocrinology 162. 10.1210/endocr/bqab064 [DOI] [PubMed]
- Nakamura M, Fukami M, Sugawa F, et al. Mamld1 knockdown reduces testosterone production and Cyp17a1 expression in mouse leydig tumor cells. PLoS ONE. 2011;6:e19123. doi: 10.1371/journal.pone.0019123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nguyen TTL, Gao H, Liu D, et al. Glucocorticoids unmask silent non-coding genetic risk variants for common diseases. Nucleic Acids Res. 2022;50:11635–11653. doi: 10.1093/nar/gkac1045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palma-Vera SE, Reyer H, Langhammer M, et al. Genomic characterization of the world’s longest selection experiment in mouse reveals the complexity of polygenic traits. BMC Biol. 2022;20:52. doi: 10.1186/s12915-022-01248-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park C-S, Shastri N (2022) The role of T cells in obesity-Associated inflammation and metabolic disease. 10.4110/in.2022.22.e13. Immune Netw 22: [DOI] [PMC free article] [PubMed]
- Pazo A, Pérez-González A, Oliveros JC et al (2019) hCLE/RTRAF-HSPC117-DDX1-FAM98B: a New Cap-binding complex that activates mRNA translation. 10.3389/fphys.2019.00092. Front Physiol 10: [DOI] [PMC free article] [PubMed]
- Pearce LR, Huang X, Boudeau J, et al. Identification of Protor as a novel Rictor-binding component of mTOR complex-2. Biochem J. 2007;405:513–522. doi: 10.1042/BJ20070540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearce LR, Sommer EM, Sakamoto K, et al. Protor-1 is required for efficient mTORC2-mediated activation of SGK1 in the kidney. Biochem J. 2011;436:169–179. doi: 10.1042/BJ20102103. [DOI] [PubMed] [Google Scholar]
- Pettersson M, Viljakainen H, Loid P, et al. Copy number variants are enriched in individuals with early-onset obesity and highlight novel pathogenic pathways. J Clin Endocrinol Metab. 2017;102:3029–3039. doi: 10.1210/jc.2017-00565. [DOI] [PubMed] [Google Scholar]
- Pineda E, Sanchez-Romero LM, Brown M, et al. Forecasting Future trends in obesity across Europe: the value of improving Surveillance. Obes Facts. 2018;11:360–371. doi: 10.1159/000492115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pink RC, Wicks K, Caley DP, et al. Pseudogenes: pseudo-functional or key regulators in health and diseasě. RNA. 2011;17:792–798. doi: 10.1261/rna.2658311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qin Z, Ren F, Xu X, et al. ZNF536, a Novel Zinc Finger protein specifically expressed in the brain, negatively regulates Neuron differentiation by repressing Retinoic Acid-Induced Gene transcription. Mol Cell Biol. 2009;29:3633–3643. doi: 10.1128/MCB.00362-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Queiroz EM, Cândido APC, Castro IM, et al. IGF2, LEPR, POMC, PPARG, and PPARGC1 gene variants are associated with obesity-related risk phenotypes in Brazilian children and adolescents. Brazilian J Med Biol Res. 2015;48:595–602. doi: 10.1590/1414-431x20154155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rashad N, Nawara A, Ahmed S, et al. Alternation in circARF3 (ADP-ribosylation factor 3) and its target gene miR-103 activity promotes Hepatocellular Carcinoma in obese patients with metabolic-Associated fatty liver disease. Egypt J Hosp Med. 2023;90:113–120. doi: 10.21608/ejhm.2023.279213. [DOI] [Google Scholar]
- Raudvere U, Kolberg L, Kuzmin I, et al. G:profiler: a web server for functional enrichment analysis and conversions of gene lists (2019 update) Nucleic Acids Res. 2019;47:W191–W198. doi: 10.1093/nar/gkz369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ren W, Hou X, Wang Y, et al. Overgrazing induces alterations in the hepatic proteome of sheep (Ovis aries): an iTRAQ-based quantitative proteomic analysis. Proteome Sci. 2016;15:2. doi: 10.1186/s12953-016-0111-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salih MH, Adnan F, Al-Azzawie A, Hussain Ali Al-Assie Intronic SNPs and genetic diseases: a review. Int J Res Appl Sci Biotechnol. 2021;8:267–274. doi: 10.31033/ijrasb.8.2.36. [DOI] [Google Scholar]
- Saul MC, Philip VM, Reinholdt LG, Chesler EJ. High-diversity mouse populations for Complex traits. Trends Genet. 2019;35:501–514. doi: 10.1016/j.tig.2019.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt E, Dhaouadi I, Gaziano I, et al. LincRNA H19 protects from dietary obesity by constraining expression of monoallelic genes in brown fat. Nat Commun. 2018;9:3622. doi: 10.1038/s41467-018-05933-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shannon P. Cytoscape: a Software Environment for Integrated Models of Biomolecular Interaction Networks. Genome Res. 2003;13:2498–2504. doi: 10.1101/gr.1239303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shao X, Wang M, Wei X, et al. Peroxisome proliferator-activated Receptor-γ: Master Regulator of adipogenesis and obesity. Curr Stem Cell Res Ther. 2016;11:282–289. doi: 10.2174/1574888X10666150528144905. [DOI] [PubMed] [Google Scholar]
- Sharp GL, Hill WG, Robertson A. Effects of selection on growth, body composition and food intake in mice I. responses in selected traits. Genet Res. 1984;43:75–92. doi: 10.1017/S0016672300025738. [DOI] [PubMed] [Google Scholar]
- Shen Y, Wang X, Xu J, Lu L. SerpinE2, a poor biomarker of endometrial cancer, promotes the proliferation and mobility of EC cells. Cancer Biomarkers. 2017;19:271–278. doi: 10.3233/CBM-160442. [DOI] [PubMed] [Google Scholar]
- Silva-Vignato B, Cesar ASM, Afonso J et al (2022) Integrative Analysis between Genome-Wide Association Study and Expression Quantitative Trait Loci Reveals Bovine Muscle Gene Expression Regulatory Polymorphisms Associated with Intramuscular Fat and Backfat Thickness. Front Genet 13. 10.3389/fgene.2022.935238 [DOI] [PMC free article] [PubMed]
- Šimon M, Mikec Š, Morton NM, Atanur SS, Horvat S, Kunej T (2024) Integration of genomic and transcriptomic data of inbred mouse models for polygenic obesity and leanness revealed “obese” and “lean” candidate alleles in polyadenylation signal. Gene Rep 34:12–31. 10.1016/j.genrep.2024.101903
- Simončič M, Horvat S, Stevenson PL, et al. Divergent physical activity and novel alternative responses to high Fat Feeding in Polygenic Fat and lean mice. Behav Genet. 2008;38:292–300. doi: 10.1007/s10519-008-9199-y. [DOI] [PubMed] [Google Scholar]
- Söhle J, Machuy N, Smailbegovic E, et al. Identification of new genes involved in human adipogenesis and Fat Storage. PLoS ONE. 2012;7:e31193. doi: 10.1371/journal.pone.0031193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Son J, Ding H, Accii D, Califano A. AFF3 and BACH2 are master regulators of metabolic inflexibility, β/α-cell transition, and dedifferentiation in type 2 diabetes. bioRxiv. 2019;768135:768135. doi: 10.1101/768135. [DOI] [Google Scholar]
- Sosa-Madrid BS, Hernández P, Blasco A, et al. Genomic regions influencing intramuscular fat in divergently selected rabbit lines. Anim Genet. 2020;51:58–69. doi: 10.1111/age.12873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suleiman JB, Mohamed M, Bakar ABA. A systematic review on different models of inducing obesity in animals: advantages and limitations. J Adv Vet Anim Res. 2020;7:103–114. doi: 10.5455/JAVAR.2020.G399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun BB, Kurki MI, Foley CN, et al. Genetic associations of protein-coding variants in human disease. Nature. 2022;603:95–102. doi: 10.1038/s41586-022-04394-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tews D, Fromme T, Keuper M, et al. Teneurin-2 (TENM2) deficiency induces UCP1 expression in differentiating human fat cells. Mol Cell Endocrinol. 2017;443:106–113. doi: 10.1016/j.mce.2017.01.015. [DOI] [PubMed] [Google Scholar]
- Titov II, Vorozheykin PS. Comparing miRNA structure of mirtrons and non-mirtrons. BMC Genomics. 2018;19:114. doi: 10.1186/s12864-018-4473-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uffelmann E, Huang QQ, Munung NS, et al. Genome-wide association studies. Nat Rev Methods Prim. 2021;1:59. doi: 10.1038/s43586-021-00056-9. [DOI] [Google Scholar]
- Vaamonde JG, Álvarez-Món MA (2020) Obesity and overweight. In: Med. https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight. Accessed 5 Sep 2022
- Vaittinen M, Kaminska D, Käkelä P, et al. Downregulation of CPPED1 expression improves glucose metabolism in Vitro in Adipocytes. Diabetes. 2013;62:3747–3750. doi: 10.2337/db13-0830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van der Auwera GA, Carneiro MO, Hartl C, et al. From fastQ data to high-confidence variant calls: the genome analysis toolkit best practices pipeline. Curr Protoc Bioinforma. 2013;43:483–492. doi: 10.1002/0471250953.bi1110s43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voisin S, Almén MS, Moschonis G, et al. Dietary fat quality impacts genome-wide DNA methylation patterns in a cross-sectional study of Greek preadolescents. Eur J Hum Genet. 2015;23:654–662. doi: 10.1038/ejhg.2014.139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wan JY, Goodman DL, Willems EL, et al. Genome-wide association analysis of metabolic syndrome quantitative traits in the GENNID multiethnic family study. Diabetol Metab Syndr. 2021;13:59. doi: 10.1186/s13098-021-00670-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang S-H, Zhu X-L, Wang F, et al. LncRNA H19 governs mitophagy and restores mitochondrial respiration in the heart through Pink1/Parkin signaling during obesity. Cell Death Dis. 2021;12:557. doi: 10.1038/s41419-021-03821-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang T, Zhou M, Guo J, et al. Analysis of selection signatures on the Z chromosome of bidirectional selection broiler lines for the assessment of abdominal fat content. BMC Genomic Data. 2021;22:18. doi: 10.1186/s12863-021-00971-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei W, Chen Q, Liu M, et al. TRIM24 is an insulin-responsive regulator of P-bodies. Nat Commun. 2022;13:3972. doi: 10.1038/s41467-022-31735-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winer DA, Winer S, Chng MHY, et al. B lymphocytes in obesity-related adipose tissue inflammation and insulin resistance. Cell Mol Life Sci. 2014;71:1033–1043. doi: 10.1007/s00018-013-1486-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu CT, Hilgendorf KI, Bevacqua RJ, et al. Discovery of ciliary G protein-coupled receptors regulating pancreatic islet insulin and glucagon secretion. Genes Dev. 2021;35:1243–1255. doi: 10.1101/GAD.348261.121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu L, Yeung MHY, Yau MYC, et al. Role of histone acetylation and methylation in obesity. Curr Pharmacol Rep. 2019;5:196–203. doi: 10.1007/s40495-019-00176-7. [DOI] [Google Scholar]
- Xu X, He K, Hoffman RD, et al. Thymosin Beta 15 alters the spatial development of thymic epithelial cells. Cells. 2022;11:3679. doi: 10.3390/cells11223679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamazaki W, Badescu D, Tan SL, et al. Effects of the sex chromosome complement, XX, XO, or XY, on the Transcriptome and Development of Mouse oocytes during Follicular Growth. Front Genet. 2021;12:2569. doi: 10.3389/fgene.2021.792604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang L, Chang CC, Sun Z, et al. GFRAL is the receptor for GDF15 and is required for the anti-obesity effects of the ligand. Nat Med. 2017;23:1158–1166. doi: 10.1038/nm.4394. [DOI] [PubMed] [Google Scholar]
- Yang Y, Zhou R, Zhu S, et al. Systematic identification and molecular characteristics of long noncoding RNAs in Pig tissues. Biomed Res Int. 2017;2017:1–9. doi: 10.1155/2017/6152582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yi SH, Jeong YJ, Jeon H, et al. Anti-obesity effects of Galla Rhois via genetic regulation of adipogenesis. Biomed Pharmacother. 2021;142:112063. doi: 10.1016/j.biopha.2021.112063. [DOI] [PubMed] [Google Scholar]
- Zhang M, Ceyhan Y, Kaftanovskaya EM, et al. INPP4B protects from metabolic syndrome and associated disorders. Commun Biol. 2021;4:416. doi: 10.1038/s42003-021-01940-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang X, Xiao N, Cao Y et al (2023) De novo variants in MAST4 related to neurodevelopmental disorders with developmental delay and infantile spasms: genotype-phenotype association. Front Mol Neurosci 16. 10.3389/fnmol.2023.1097553 [DOI] [PMC free article] [PubMed]
- Zuo T, Ng SC. The gut microbiota in the Pathogenesis and therapeutics of inflammatory bowel disease. Front Microbiol. 2018;9:2247. doi: 10.3389/fmicb.2018.02247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zuo E, Cai YJ, Li K, et al. One-step generation of complete gene knockout mice and monkeys by CRISPR/Cas9-mediated gene editing with multiple sgRNAs. Cell Res. 2017;27:933–945. doi: 10.1038/cr.2017.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data are available on request.