Abstract
Background
While the 23-valent pneumococcal polysaccharide vaccine (PPV23) has demonstrated its role in preventing severe pneumococcal disease, its impact on more non-specific conditions like acute respiratory tract infection (ARI) and lower respiratory tract infections (LRTI) remains unclear. We aimed to investigate the role of PPV23 in prevention of presentations for ARI and LRTI and related antibiotic prescriptions among older adults in primary care.
Methods
Using a nationwide general practice dataset, we followed a cohort of regularly attending patients aged ≥65 years from 1 January 2014 until 31 December 2018 for presentations for ARI, LRTI, and related antibiotic prescriptions. Associations between PPV23 receipt and each outcome were assessed using a multiple failures survival model to estimate hazard ratios (HR) adjusted for age, sex, socioeconomic status, and various health measures.
Results
A cohort of 75,264 patients aged ≥65 years (mean 75.4, 56% female) in 2014 was followed. The incidence of presentations for ARI, ARI-related antibiotic prescription, LRTI, and LRTI-related antibiotic prescription was 157.6, 76.0, 49.6, and 24.3 per 1000 person-years, respectively. Recent PPV23 vaccine receipt was associated with a small reduction in ARI presentations (adjusted HR vaccinated vs. unvaccinated 0.96; 95%CI 0.94–0.98; p = 0.002); however, there was no reduction in ARI-related antibiotic prescription, LRTI presentation, nor LRTI-related antibiotic prescription (adjusted HR were 0.99[95%CI 0.96–1.03], 1.04[95%CI 0.99–1.09], 1.07[95%CI 1.00–1.14]).
Conclusion
PPV23 vaccination in older adults may result in a small reduction in the incidence of total ARI presentations in primary care. However, the effect is small and residual confounding cannot be excluded.
Introduction
Pneumococcal disease, caused by Streptococcus pneumoniae, can present as a heterogenous medical condition that ranges from mild respiratory tract infections such as bronchitis, rhinitis, acute sinusitis, otitis media, conjunctivitis to severe life-threatening invasive pneumococcal diseases such as meningitis and septicaemia [1]. The burden of pneumococcal disease varies substantially across geographical regions and by different age groups, with higher burden found particularly in young children and the elderly. Data from Australia’s National Notifiable Disease Surveillance System (NNDSS) indicates that in 2016 the crude notification rates for invasive pneumococcal disease were 188.9 per million among infants and 166.9 per million among individuals aged 65 years and above [2]. Systematic reviews suggest that the pneumococcal vaccines, 23-valent pneumococcal polysaccharide vaccine (PPV23), and 13-valent pneumococcal conjugate vaccine (PCV13) can protect against invasive pneumococcal disease and pneumococcal pneumonia in the elderly [3–6]. However, it is uncertain how much pneumococcal vaccines may prevent more nonspecific outcomes such as lower respiratory tract infections (LRTI) [7–9]. A large proportion of acute respiratory tract infections (ARI) lead to antibiotic prescriptions, with ARIs accounting for an estimated 50% of all antibiotic prescriptions in adults [10–12]. As a reduction in antibiotic use is likely to help to curb the global emergence of antimicrobial resistance, it is also important to quantify whether pneumococcal vaccination has any role in reduction of ARI presentations and subsequently ARI-related antibiotic prescriptions, and no previous study has yet investigated this in adults.
In Australia, similar to many high-income countries, pneumococcal vaccines are routinely recommended for specific groups such as young children, older adults, and individuals with certain at-risk conditions to prevent pneumococcal disease [13]. Since the introduction of the universal infant PCV vaccination program in 2005, vaccine coverage in eligible children has been consistently over 90% [14]. A systematic review analysing Australian studies from 1992 to 2013 estimated that pneumococcal vaccination coverage among individuals aged 65 and above ranged from 50.3% to 72.8% [15]. Australia’s pneumococcal vaccination program has evolved significantly over time [15], with the most recent changes implemented on 1 July 2020 [13]. Prior to this, in 1997 adults aged 65 years and above were recommended to receive at least one dose of PPV23 with funding of the single dose available since 2005 [16]. This study aims to estimate the association of PPV23 vaccination with ARI and LRTI presentations and ARI- and LRTI-related antibiotic prescription in primary care settings among older Australian adults.
Materials and methods
Data source and study population
MedicineInsight is a de-identified longitudinal research database derived from national electronic health records for over 3.6 million patients in approximately 600 general practices (GPs) across Australia. A detailed description of MedicineInsight has been published elsewhere [17]. Australians have universal health coverage including access to GPs and patients are free to visit multiple GPs of their choice. The majority of vaccines included in the National Immunisation Programme (NIP) for children and adults aged 65 and above are administered through general practice [18]. We used a 25% random sample of patients in the database (the maximum allowable for external researchers), which represented 573 general practice sites. Information extracted included patients’ demographics, reasons for encounters (free-text), diagnoses (free-text), prescription items, vaccination history, and practices’ geographic information. We only included data from GPs that met practice quality criteria each year from 2012 to 2018 as described previously [19]; this was GPs with annually (a) ≥250 encounter records; (b) ≥300 prescription records; (c) ≥100 unique patients who visited the practice; and (d) <15% of prescription records with missing medicine names. Ethical approval for the study was granted by the University of New South Wales Human Research Ethics Committee (approval no. HC200176). The data were accessed on 23 June 2021 for research purposes.
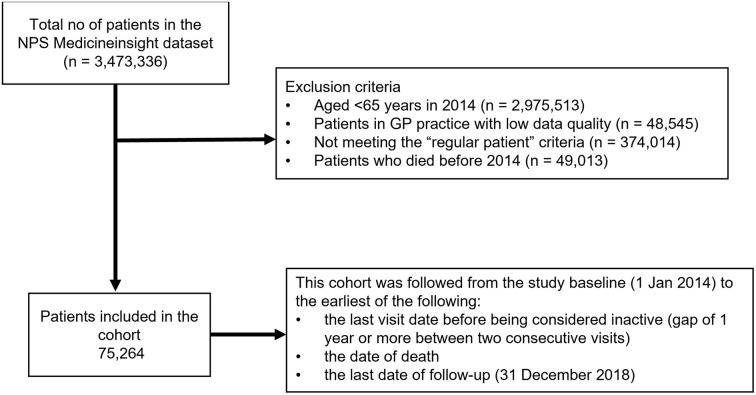
A retrospective cohort study design was used to assess the associations of PPV23 vaccination with ARI, ARI-related antibiotic prescription, LRTI, and LRTI-related antibiotic prescription. We defined a “regular attender” as a patient who attended the same GP at least once in each of the preceding two years (e.g., 2012 and 2013) [19]. We included all patients aged ≥65 years in 2014 who were regular attenders. In Australia, individuals have the flexibility to choose and switch general practitioners at any time, therefore we limited our study to those who consistently attended. Patients recorded as inactive, deceased (before 2014), or with missing data on year of birth and sex were excluded from analyses (see Fig 1 for details). This 2014 cohort was followed through to 2018.
Fig 1. Flow chart of study cohort selection.
Pneumococcal vaccination
Information on pneumococcal vaccination and date of vaccination was retrieved from MedicineInsight’s Immunisation dataset. We used vaccine brand names as search terms (Pneumovax or Prevenar 13) as well as a combination of all plausible terms (pneumo’ and ‘vax’, ‘vac’ or ‘injection’) to capture GP encounters related to pneumococcal vaccination (see S2 Table for details). In our cohort, 99.5% of those who received a pneumococcal vaccine received PPV23, 0.1% received PCV13, and 0.4% received an unspecified pneumococcal vaccine. Therefore, we focussed on quantifying the association of PPV23 with the outcomes and excluded the small number who received PCV13 and unspecified pneumococcal vaccine from analyses.
Outcomes
Four outcomes were assessed—i) acute respiratory tract infection (ARI) events, ii) ARI-related antibiotic prescriptions, iii) lower respiratory tract infection (LRTI), and iv) LRTI-related antibiotic prescriptions.
For ARI and LRTI, we searched the “encounter reason,” “diagnosis reason,” and “prescription reason” data fields in the corresponding datasets from the MedicineInsight database using a combination of relevant medical terminologies and shorthand abbreviations. Search terms were based on and earlier study (19) and are provided in S3 Table. If a patient had multiple records of ARI or LRTI events within a 30-day period, these were considered part of the same event. Conversely, ARI or LRTI events occurring more than 30 days apart in the same patient were treated as recurrent events.
We identified antibiotic prescribing from the “medicine active ingredient” field of the prescription dataset using combinations of search terms for systemic antibiotics [19] commonly prescribed to treat respiratory illnesses (S4 Table). If patients had a record of an antibiotic prescription and a record of ARI or LRTI diagnosis on the same date, then they were considered to have had an ARI- or LRTI-related antibiotic prescription.
Statistical analysis
To describe the cohort characteristics, we used proportions for categorical variables and means and standard deviation (SD) for continuous variables. For the longitudinal analyses, we followed patients from the study baseline (1 Jan 2014) to the earliest of the following: the last visit date before being considered as inactive, the date of death, or the last date of follow-up (31 December 2018). A patient was considered inactive if they had a gap of one year or more between two consecutive visits. We examined the distribution of each outcome as recurrent events and estimated an overall incidence rate with a 95% confidence interval (CI). The incidence rate was calculated by dividing the number of cases over the number of person-years accumulated during the follow-up period.
We treated pneumococcal (PPV23) vaccination status as a time-varying variable. Those with a record of PPV23 receipt between 2000 and 2013 (i.e., before baseline) were defined as vaccinated at baseline. Those with a first record of pneumococcal vaccination after the baseline (i.e., between 2014 and 2018) were initially classified as unvaccinated and then contributed person-time to the vaccinated category from 14 days after their date of vaccine receipt. We used the Prentice, Williams, and Peterson Total Time (PWPTT) multiple failures survival model to estimate hazard ratios (HR) with 95% CI for each of the four outcomes (ARI, ARI-related antibiotic prescriptions, LRTI, and LRTI-related antibiotic prescriptions) comparing vaccinated vs. unvaccinated patients. More details on how we dealt the data for time-varying vaccination status and multiple outcome events are given in S1 Methods in S1 File S1 Fig. Models were adjusted for potential confounders: age at baseline (categorized as 65–69; 70–74; 75–79; 80–84; ≥85 years), sex, socio-economic status of GP in quintiles (Index of Relative Socio-economic Advantage and Disadvantage (IRSAD) [20], a macroeconomic indicator of relative economic and social advantage/disadvantage position within an area/postcode compared to the rest of the country), the total number of GP visits in 2012 and 2013 (categorized as 2–10; 11–20; ≥ 21 visits), remoteness of practice (categorized as major cities; inner regional; outer regional/remote/very remote, based on the Accessibility and Remoteness Index of Australia [21], smoking status (categorized as a smoker; ex-smoker; never smoker), influenza vaccination status during the follow-up period (yes, no), and comorbidity status (i.e. ever recorded asthma, chronic obstructive pulmonary disease (COPD), diabetes, chronic heart disease, chronic kidney disease, chronic liver disease, and haematological malignancy). We used pre-coded conditions information available in the MedicineInsight data to define these comorbidities, except for haematological malignancies which was defined by free-text searching (more details in the S1 Methods in S1 File S1 Table).
We also assessed the association of PPV23 with the outcome of interest by time since vaccination (unvaccinated, vaccinated within <5 years of baseline, and vaccinated ≥5 years prior to baseline). We also stratified analyses by the presence of the specified comorbidities (yes, no) and by smoking status (ever smoker, never smoker). To examine the validity of our primary analysis, we undertook analyses with negative control outcomes, including gastroenteritis and urinary tract infection (UTI), using definitions from earlier studies [22, 23]. We also conducted a sensitivity analysis where we restricted follow-up to one year (from Jan 2014- Dec 2014) to reduce any potential confounding due to changes in age over time.
All analyses were performed using Stata V.17.1 (StataCorp, College Station, Texas, USA). All statistical analyses were two-sided, and a p-value <0.05 was considered statistically significant.
Results
A total of 75,264 regular attendees aged a mean of 75.4 years at baseline were included. Over half (56%) of patients were female, and 58% came from practices in major cities. The majority of patients were never smokers (53.3%), and 27% had at least one of the listed comorbidities. Table 1. shows the baseline characteristics according to the record of receipt of at least one dose of PPV23 between 2000 and 2018. A total of 52,277 patients (70%) had received PPV23 vaccine in this time period. Among those who received at least one dose of PPV23, 60.8% had one dose, and 39.2% had two or more doses. Compared to those who did not receive PPV23, those who received the vaccine were more likely to be older, ex-smokers, and have at least one comorbidity. The vaccinated group also had more GP visits in the preceding two years and were more likely to have received an influenza vaccination during follow-up period.
Table 1. Characteristics of included patients aged 65 years or above at study baseline (2014) according to pneumococcal vaccination (PPV23) received during 2000 to 2018, Australia.
| Total | Never received PPV23 | Ever received at least one dose of PPV23 | p-value | |
|---|---|---|---|---|
| N = 75264 | N = 22,987 | N = 52,277 | ||
| Age at baseline (2014) | <0.001 | |||
| 65–69 years | 21,599 (28.7%) | 8,706 (37.9%) | 12,893 (24.7%) | |
| 70–74 years | 17,374 (23.1%) | 4,873 (21.2%) | 12,501 (23.9%) | |
| 75–79 years | 14,382 (19.1%) | 3,552 (15.5%) | 10,830 (20.7%) | |
| 80–84 years | 10,711 (14.2%) | 2,672 (11.6%) | 8,039 (15.4%) | |
| 85+ years | 11,198 (14.9%) | 3,184 (13.9%) | 8,014 (15.3%) | |
| Sex | 0.084 | |||
| Male | 33,222 (44.1%) | 10,255 (44.6%) | 22,967 (43.9%) | |
| Female | 42,042 (55.9%) | 12,732 (55.4%) | 29,310 (56.1%) | |
| Socioeconomic status of GP (quintiles) | <0.001 | |||
| Most disadvantaged | 14,000 (18.6%) | 4,331 (18.8%) | 9,669 (18.5%) | |
| Disadvantaged | 13,505 (17.9%) | 4,464 (19.4%) | 9,041 (17.3%) | |
| Middle | 18,893 (25.1%) | 5,429 (23.6%) | 13,464 (25.8%) | |
| Advantaged | 11,723 (15.6) | 3,650 (15.9%) | 8,073 (15.4%) | |
| Most advantaged | 16,867 (22.4%) | 5,004 (21.8%) | 11,863 (22.7%) | |
| Missing | 276 (0.4%) | 109 (0.5%) | 167 (0.3%) | |
| Remoteness of GP | <0.001 | |||
| Major cities | 43,523 (57.8%) | 13,657 (59.4%) | 29,866 (57.1%) | |
| Inner regional | 22,525 (29.9%) | 6,192 (26.9%) | 16,333 (31.2%) | |
| Outer regional / remote / very remote | 9,216 (12.2%) | 3,138 (13.7%) | 6,078 (11.6%) | |
| Smoking status | <0.001 | |||
| Smoker | 3,690 (4.9%) | 1,357 (5.9%) | 2,333 (4.5%) | |
| Ex-smoker | 27,282 (36.2%) | 7,234 (31.5%) | 20,048 (38.3%) | |
| Never smoker | 40,078 (53.2%) | 11,965 (52.1%) | 28,113 (53.8%) | |
| Missing | 4,214 (5.6%) | 2,431 (10.6%) | 1,783 (3.4%) | |
| No. of GP visits in 2012–13 | <0.001 | |||
| 2–10 visits | 30,717 (40.8%) | 11,825 (51.4%) | 18,892 (36.1%) | |
| 11–20 visits | 23,922 (31.8) | 6,458 (28.1%) | 17,464 (33.4%) | |
| 21 or more visits | 20,625 (27.4%) | 4,704 (20.5%) | 15,921 (30.5%) | |
| Asthma | 4,328 (5.8%) | 1,057 (4.6%) | 3,271 (6.3%) | <0.001 |
| COPD | 4,252 (5.6%) | 890 (3.9%) | 3,362 (6.4%) | <0.001 |
| Heart disease | 7,493 (10.0%) | 2,043 (8.9%) | 5,450 (10.4%) | <0.001 |
| Diabetes | 6,813 (9.1%) | 1,768 (7.7%) | 5,045 (9.7%) | <0.001 |
| Chronic liver disease | 124 (0.2%) | 47 (0.2%) | 77 (0.1%) | 0.075 |
| Chronic kidney disease | 1,698 (2.3%) | 379 (1.6%) | 1,319 (2.5%) | <0.001 |
| Haematological malignancy | 245 (0.3%) | 60 (0.3%) | 185 (0.4%) | 0.039 |
| Any of the above comorbidities | 20,274 (26.9%) | 5,160 (22.4%) | 15,114 (28.9%) | <0.001 |
| No. of influenza vaccines in 2014–18 | <0.001 | |||
| 0 | 17,984 (23.9%) | 11,503 (50.0%) | 6,481 (12.4%) | |
| 1–3 | 25,382 (33.7%) | 7,506 (32.7%) | 17,876 (34.2%) | |
| 4 or more | 31,898 (42.4%) | 3,978 (17.3%) | 27,920 (53.4%) | |
| Description of pneumococcal vaccination No. of PPV23 doses received 2000–2018 | ||||
| 1 | 31,777 (60.8%) | |||
| 2+ | 20,500 (39.2%) | |||
| Vaccinated at baseline | 41,203 (78.8%) | |||
| Vaccinated during follow-up (2014–2018) | 11,074 (21.2%) | |||
| The last dose received within 5 years of baseline or during follow-up | 35,292 (67.5%) |
During follow-up, a total of 40,246 ARI events, 19,543 ARI-related antibiotic prescriptions, 12,777 LRTI events, and 6,268 LRTI-related antibiotic prescriptions were recorded in 75,264 individuals over an average of 3.4 person-years of follow-up. The majority of those who had an outcome of interest, only had one event reported during the four-year study period. The overall incidence of ARI, ARI-antibiotic, LRTI, and LRTI-antibiotic was 157.6, 76.0, 49.6, and 24.3 per 1000 person-years, respectively (data not shown). For every outcome, individuals who had ever been vaccinated showed a higher incidence than those who had never been vaccinated. Table 2 shows the follow-up time and incidence of study outcomes, stratified by vaccination status.
Table 2. Incidence of acute respiratory tract infections (ARIs), ARI-related antibiotic prescription, lower respiratory tract infection (LRTIs), and LRTI-related antibiotic prescription during follow-up (2014–2018) among patients aged 65 years or above, Australia.
| Acute respiratory tract infections (ARIs) | ARI-related antibiotic prescription | Lower respiratory tract infection (LRTIs) | LRTI-related antibiotic prescription | |||||
|---|---|---|---|---|---|---|---|---|
| Never vaccinated | Ever vaccinated | Never vaccinated | Ever vaccinated | Never vaccinated | Ever vaccinated | Never vaccinated | Ever vaccinated | |
| Total follow-up time (person-years) | 84396 | 170972 | 84904 | 172097 | 85074 | 172485 | 85220 | 172845 |
| Total no. of events | 12602 | 27644 | 6127 | 13416 | 3455 | 9322 | 1710 | 4558 |
| Incidence per 1000 person-years (95% CI) | 149.3 (146.7–151.9) | 161.7 (159.8–163.6) | 72.2 (70.4–74.0) | 78.0 (76.6–79.3) | 40.6 (39.3–42.0) | 54.0 (53.0–55.2) | 20.1 (19.1–21.0) | 26.4 (25.6–27.1) |
| No. of patients with at least one event (% of total) | 7781 (23.1) | 15989 (30.6) | 4439 (13.2) | 9354 (17.9) | 2744 (8.1) | 6756 (12.9) | 1471 (4.4) | 3680 (7.1) |
| Among patients with at least one event | ||||||||
| Only one event, n (%) | 5133 (15.2) | 9835 (18.8) | 3344 (9.9) | 6808 (13.0) | 2232 (6.6) | 5096 (9.8) | 1277 (3.8) | 3055 (5.9) |
| Two events, n (%) | 1527 (4.5) | 3449 (6.6) | 744 (2.2) | 1685 (3.2) | 381 (1.1) | 1135 (2.2) | 161 (0.5) | 456 (0.9) |
| Three or more events, n (%) | 1121 (3.3) | 2705 (5.2) | 351 (1.0) | 861 (1.7) | 131 (0.4) | 525 (1.0) | 33 (0.1) | 169 (0.3) |
The associations of PPV23 vaccination with each study outcome are shown in Table 3. In the fully adjusted models, we did not find statistically significant associations of PPV23 vaccination with ARI events and ARI-related antibiotic prescriptions. However, for LRTI events and LRTI-related antibiotic prescriptions, we observed a significant positive association with PPV23 vaccination. S6 Table presents the sensitivity analysis with a follow-up period limited to one year. This analysis shows HRs with similar directions and magnitudes for the incidence of ARIs and ARI-related antibiotic prescriptions as the main analysis. However, the association with reduced ARI incidence was borderline statistically significant in the sensitivity analysis, and associations of PPV23 vaccination with LRTI and LRTI-related antibiotic prescriptions were no longer statistically significant. We did find some evidence of a reduction in ARI presentations when time since vaccination was considered (Table 4). Compared to those without pneumococcal vaccination, those who received PPV23 within <5 years of baseline had a slightly reduced incidence of ARI (adjusted HR 0.96; 95% CI 0.94–0.98; p = 0.002), but this was not the case among those who received PPV23 ≥5 years prior to baseline where the risk was marginally increased (adjusted HR 1.04; 95% CI 1.01–1.07; p = 0.013). For other study outcomes, we found no evidence of a reduction in incidence by recency of PPV23 receipt but rather an increased risk associated with PPV23 vaccination more than five years prior to baseline (Table 4).
Table 3. Associations of PPV23 vaccination and incidence of acute respiratory tract infections (ARIs), ARI-related antibiotic prescription, lower respiratory tract infection (LRTIs), and LRTI-related antibiotic prescription among patients aged 65 years or above, Australia 2014–2018.
| Outcomes | Age & sex-adjusted model | Fully adjusted model* | ||
|---|---|---|---|---|
| HR (95% CI) | P value | HR (95% CI) | P value | |
| Ever received PPV23 | ||||
| ARIs | 1.06 (1.04–1.08) | <0.001 | 0.98 (0.95–1.00) | 0.066 |
| ARI-related antibiotic | 1.08 (1.05–1.11) | <0.001 | 1.01 (0.97–1.04) | 0.743 |
| LRTIs | 1.17 (1.12–1.22) | <0.001 | 1.08 (1.03–1.13) | <0.001 |
| LRTI-related antibiotic | 1.19 (1.12–1.26) | <0.001 | 1.09 (1.03–1.16) | 0.004 |
*Hazard ratios (HRs) with 95% confidence intervals (Cis) were estimated by adjusting for age group, sex, remoteness of practice, socio-economic status, number of GP visits in 2012 & 2013, smoking status, flu vaccination status during the follow-up period, asthma, Chronic obstructive pulmonary disease (COPD), heart disease, chronic kidney disease, chronic liver disease, diabetes, and haematological malignancy
Table 4. Associations of time since PPV23 vaccination and incidence of acute respiratory tract infections (ARIs), ARI-related antibiotic prescription, lower respiratory tract infection (LRTIs), and LRTI-related antibiotic prescription among patients aged 65 years or above, Australia 2014–2018.
| Time since receipt of PPV23 | Fully adjusted* HR with 95% CI | P value |
|---|---|---|
| ARIs | ||
| Unvaccinated | 1.00 (Ref) | |
| Vaccinated <5 years | 0.96 (0.94–0.98) | 0.002 |
| Vaccinated ≥5 years | 1.04 (1.01–1.07) | 0.013 |
| ARI-related antibiotic | ||
| Unvaccinated | 1.00 (Ref) | |
| Vaccinated <5 years | 0.99 (0.96–1.03) | 0.648 |
| Vaccinated ≥5 years | 1.09 (1.04–1.14) | <0.001 |
| LRTI | ||
| Unvaccinated | 1.00 (Ref) | |
| Vaccinated <5 years | 1.04 (0.99–1.09) | 0.142 |
| Vaccinated ≥5 years | 1.15 (1.09–1.21) | <0.001 |
| LRTI-related antibiotic | ||
| Unvaccinated | 1.00 (Ref) | |
| Vaccinated <5 years | 1.07 (1.00–1.14) | 0.058 |
| Vaccinated ≥5 years | 1.20 (1.11–1.30) | <0.001 |
* Hazard ratios (HRs) with 95% confidence intervals (Cis) were estimated by adjusting for age group, sex, remoteness of practice, socio-economic status, number of GP visits in 2012 & 2013, smoking status, flu vaccination status during the follow-up period, asthma, COPD, heart disease, chronic kidney disease, chronic liver disease, diabetes, and haematological malignancy
In stratified analyses, we found that PPV23 was associated with a reduced likelihood of ARI events among those who did not have comorbidities (HR 0.96, 95% CI 0.94–0.99) but not among those with comorbidities (HR 1.02, 95% CI 0.98–1.07) (Table 5). We found no evidence of a difference in the association of PPV23 with the outcomes by smoking status. Sensitivity analyses with negative control outcomes showed that PPV23 vaccination was not associated with UTI (adjusted HR 1.01, 95% CI 0.96–1.06), but there was a small yet statistically significant positive association with gastroenteritis (adjusted HR 1.09, 95% CI 1.00–1.19) (S5 Table).
Table 5. Associations of PPV23 vaccination and incidence of acute respiratory tract infections (ARIs), ARI-related antibiotic prescription, lower respiratory tract infection (LRTIs), and LRTI-related antibiotic prescription among patients aged 65 years or above according to comorbidity status and smoking status, Australia 2014–2018.
| Outcomes | Adjusted* HR (95% CI) | |||
|---|---|---|---|---|
| No comorbidity | Comorbidity** | Never smoker | Ever smoker | |
| ARIs | 0.96 (0.94–0.99) | 1.02 (0.98–1.07) | 1.00 (0.97–1.04) | 0.97 (0.93–1.00) |
| ARI-related antibiotic | 0.99 (0.95–1.03) | 1.06 (0.99–1.13) | 1.04 (0.99–1.09) | 0.98 (0.93–1.04) |
| LRTIs | 1.05 (0.99–1.11) | 1.13 (1.06–1.22) | 1.11 (1.05–1.18) | 1.09 (1.02–1.16) |
| LRTI-related antibiotic | 1.07 (0.99–1.16) | 1.14 (1.03–1.26) | 1.13 (1.04–1.23) | 1.10 (1.01–1.21) |
* Hazard ratios (HRs) with 95% confidence intervals (CIs) were estimated by adjusting for age group, sex, remoteness of practice, socio-economic status, number of GP visits in 2012 & 2013, smoking status, and flu vaccination status during the follow-up period, as appropriate.
**Comorbidity: patients were classified as having at least one comorbid condition if there was a corresponding record in the two years prior (2012 and 2013) to the follow-up period.
Discussion
In this population-based cohort of Australian adults aged ≥65 years attending primary care, we observed a small reduction in the incidence of ARI associated with PPV23 vaccination, but this was only significant among those who were vaccinated within five years of study baseline (~4% reduction, p = 0.002). However, PPV23 vaccination was not found to reduce in the incidence of ARI-related antibiotic prescription, LRTI, and LRTI-related antibiotic prescription. Stratified analysis also suggested that this reduction in incidence of ARI with PPV23 vaccination only occurred among those without comorbidities.
We found that 70% of all patients aged ≥65 years had at least one dose of PPV23 between 2000 and the end of the study period (i.e., 2018). A recent study based on the MedicineInsight data showed that 69% of those aged 60–65 years in 2010 had a recorded pneumococcal vaccination by 2017, with a peak age of vaccination at 66 years [18]. Similar to our study, they also reported that pneumococcal vaccination was more likely among those with comorbidities, ex-smokers, and frequent GP attendees. It is important to understand that recommendations for pneumococcal vaccination among adults have changed significantly over time in Australia, which in turn can impact vaccination coverage [13]. For example, a systematic review of Australian studies from 1992–2013 reported that pneumococcal vaccination coverage in people ≥65 years increased from 35.4% prior to 2005 to 56.0% from 2005 onwards, after the introduction of the free vaccine in the National Immunisation Programme (15). Pneumococcal vaccination uptake in our study cohort was similar to that found among patients aged 65 years or over in England (70%) [24].
In our study, recent vaccination with PPV23, that is within 5 years of study baseline, was associated with a significant reduction in ARI incidence (HR 0.96, 95% CI 0.94–0.98) but that was not observed when vaccination was received over 5 years ago. While we could not find any earlier studies of PPV23 vaccine on ARI incidence, previous studies have shown that the effectiveness of PPV23 vaccine against other outcomes, for example, invasive pneumococcal disease, also declined with time since vaccination [3, 5, 6]. We also found that this association between PPV23 and ARI incidence was significant only among those without comorbidities (HR 0.96, 95% CI 0.94–0.99). A potential explanation for this finding might be that comorbid conditions in older adults can be associated with poorer immune response to vaccine [25]. The small reduction in ARI incidence that we found might be expected as PPV23 is specifically targeted for the prevention of infections caused by Streptococcus pneumoniae but not infections caused by other microorganisms. Even though the estimated reduction in ARI incidence in our study was small, the high incidence of ARI in primary care in older adults (157.6 cases per 1000 person-year) suggests vaccination could still lead to a substantial reduction in GP presentations at the population level.
We found PPV23 receipt was associated with LRTI presentation in primary care settings rather than a reduction in risk, although this was predominantly found among those who received their vaccine more than five years prior to baseline. Previous studies examining the effectiveness of PPV23 given in the last five years against LRTI conditions in hospital settings showed no significant reduction in the incidence of all-cause pneumonia [26] and community-acquired pneumonia [27], and a Belgian study based on GP registry data did not find any protective effect of either PPV23 or PCV13 against LRTI [8]. However, this study did find individuals who had received both vaccines had reduced risk. Our findings, when confined to recent receipt of PPV23, are consistent with these earlier studies. As higher-risk individuals were more likely to receive PPV23 vaccine, the association of PPV23 given more than five years earlier with LRTI compared to unvaccinated populations could reflect this propensity to vaccine higher-risk populations.
In our study population, we found that nearly half of all ARI and LRTI presentations were associated with antibiotic prescribing. Previous Australian studies reported that antibiotics are prescribed in 32% and 70% of GP consultations for upper respiratory tract infection (URTI) and “other” respiratory infection, respectively [28, 29]. Studies from other countries also reported high use of antibiotics for ARIs and LRTIs in GP settings [30–32]. However, we found no evidence of association between PPV23 vaccination and ARI-related antibiotic prescriptions, LRTI, and LRTI-related antibiotic prescriptions. An analysis based on data from the Community-Acquired Pneumonia immunization Trial in Adults (CAPiTA) also did not find any association between PCV13 and LRTI-related antibiotic use in primary care [9].
The strengths of our study include the use of electronic health records from a longitudinal general practice database, which includes detailed vaccination, ARI events, and antibiotic prescription records. We also had information on various clinical and patient characteristics to adjust for potential confounders in our analysis and included negative control outcomes. Our study has several limitations. First, we ascertained the diagnosis of ARI and LRTI outcomes from GP electronic records and did not have radiography or laboratory reports to confirm the diagnosis. Additionally, because we relied on primary care records, we could not capture all ARI and LRTI outcomes, particularly severe cases that required hospitalisation. This could lead to misclassification of the outcomes. Third, we defined comorbid conditions based on a pre-coded conditions flag in the MedicineInsight dataset which did not specify the timing of diagnosis. Therefore, we could not properly identify someone’s risk of pneumococcal disease prior to age 65, which may have increased their likelihood of being vaccinated prior to age 65. Fourth, some patients might be vaccinated or diagnosed in primary care services not included in the MedicineInsight data network. Finally, residual confounding may still exist due to insufficient adjustment or unmeasured variables, as evidenced by our analyses. Differences in outcomes between the main analysis, averaging 3.4 years of follow-up, and the sensitivity analysis, limited to 1 year, could be explained by residual confounding by age which was not time-updated in the main analyses. The older adults were more likely to have ever received PPV23 than younger adults and age will affect outcomes. Other explanations for the differences could be related to the smaller numbers in the sensitivity analyses that could explain the lack of significant effect for PPV23 and LRTI and LRTI-related antibiotic prescriptions.
In summary, in this large population-based study, we found that recent PPV23 vaccination may lead to a small reduction in the incidence of ARI presentations in older adults, but PPV23 vaccination was not associated with clinically relevant reductions in LRTIs and ARI and LRTI-related antibiotic prescriptions. These findings help us to understand the potential wider benefits of PPV23 vaccination and can inform relevant policy regarding pneumococcal vaccination programs in older adults.
Supporting information
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
Acknowledgments
We would like to thank NPS MedicineWise for providing the data and all the participating general practices for contributing primary health records of MedicineInsight, and patients who allowed the use of their de-identified information for this study.
Data Availability
The MedicineInsight data underlying the results in this study are now held by the Australian Commission on Safety and Quality in Healthcare (contactable at QUMprogram@safetyandquality.gov.au). Ethical approval was required to be obtained prior to use. Data should be accessible in the same manner as the authors and the authors did not have any special access privileges that other would not have.
Funding Statement
Fariha Binte Hossain was supported by the Australian Government Research Training Program Scholarship. She received a University Postgraduate Award (UPA). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Henriques-Normark B, Tuomanen EI. The pneumococcus: epidemiology, microbiology, and pathogenesis. Cold Spring Harbor perspectives in medicine. 2013;3(7):a010215. doi: 10.1101/cshperspect.a010215 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Australian Institute of Health and Welfare. Pneumococcal disease in Australia. 2018.
- 3.Berild JD, Winje BA, Vestrheim DF, Slotved H-C, Valentiner-Branth P, Roth A, et al. A systematic review of studies published between 2016 and 2019 on the effectiveness and efficacy of pneumococcal vaccination on pneumonia and invasive pneumococcal disease in an elderly population. Pathogens. 2020;9(4):259. doi: 10.3390/pathogens9040259 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Blommaert A, Hanquet G, Willem L, Theeten H, Thiry N, Bilcke J, et al. Use of pneumococcal vaccines in the elderly: an economic evaluation. 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Falkenhorst G, Remschmidt C, Harder T, Hummers-Pradier E, Wichmann O, Bogdan C. Effectiveness of the 23-valent pneumococcal polysaccharide vaccine (PPV23) against pneumococcal disease in the elderly: systematic review and meta-analysis. PLoS One. 2017;12(1):e0169368. doi: 10.1371/journal.pone.0169368 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kraicer-Melamed H, O’Donnell S, Quach C. The effectiveness of pneumococcal polysaccharide vaccine 23 (PPV23) in the general population of 50 years of age and older: a systematic review and meta-analysis. Vaccine. 2016;34(13):1540–50. doi: 10.1016/j.vaccine.2016.02.024 [DOI] [PubMed] [Google Scholar]
- 7.Lewnard JA, Bruxvoort KJ, Hong VX, Grant LR, Jódar L, Cané A, et al. Effectiveness of pneumococcal conjugate vaccination against virus-associated lower respiratory tract infection among adults: a case-control study. The Journal of Infectious Diseases. 2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mamouris P, Henrard S, Molenberghs G, Verhaegen J, Lin G, Vaes B. Pneumococcal vaccination prevented severe LRTIs in adults: a causal inference framework applied in registry data. Journal of clinical epidemiology. 2022;143:118–27. doi: 10.1016/j.jclinepi.2021.12.008 [DOI] [PubMed] [Google Scholar]
- 9.van Werkhoven CH, Bolkenbaas M, Huijts SM, Verheij TJ, Bonten MJ. Effects of 13-valent pneumococcal conjugate vaccination of adults on lower respiratory tract infections and antibiotic use in primary care: secondary analysis of a double-blind randomized placebo-controlled study. Clinical Microbiology and Infection. 2021;27(7):995–9. doi: 10.1016/j.cmi.2020.09.011 [DOI] [PubMed] [Google Scholar]
- 10.Akhtar A, Hassali MAA, Zainal H, Ali I, Iqbal MS, Khan AH. Respiratory-tract infections among geriatrics: prevalence and factors associated with the treatment outcomes. Therapeutic Advances in Respiratory Disease. 2021;15:1753466620971141. doi: 10.1177/1753466620971141 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.O’Connor R, O’Doherty J, O’Regan A, O’Neill A, McMahon C, Dunne C. Medical management of acute upper respiratory infections in an urban primary care out-of-hours facility: Cross-sectional study of patient presentations and expectations. BMJ Open. 2019;9:e025396. doi: 10.1136/bmjopen-2018-025396 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Renati S, Linder JA. Necessity of office visits for acute respiratory infections in primary care. Family practice. 2016;33(3):312–7. doi: 10.1093/fampra/cmw019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Australian Technical Advisory Group on Immunisation AGDoH. Public consultation on changes to the recommended use of pneumococcal vaccines. 2020.
- 14.Brynley Hull AH, Aditi Dey, Julia Brotherton, Kristine Macartney, Frank Beard. NCIRS Annual Immunisation Coverage Report 2021. 2021. [DOI] [PubMed]
- 15.Dyda A, Karki S, Hayen A, MacIntyre CR, Menzies R, Banks E, et al. Influenza and pneumococcal vaccination in Australian adults: a systematic review of coverage and factors associated with uptake. BMC Infectious Diseases. 2016;16(1):1–15. doi: 10.1186/s12879-016-1820-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Menzies R, Stein A, Booy R, Van Buynder P, Litt J, Cripps A. The impact of the changing pneumococcal national immunisation program among older Australians. Vaccine. 2021;39(4):720–8. doi: 10.1016/j.vaccine.2020.12.025 [DOI] [PubMed] [Google Scholar]
- 17.Busingye D, Gianacas C, Pollack A, Chidwick K, Merrifield A, Norman S, et al. Data Resource Profile: MedicineInsight, an Australian national primary health care database. International journal of epidemiology. 2019;48(6):1741–h. doi: 10.1093/ije/dyz147 [DOI] [PubMed] [Google Scholar]
- 18.Frank O, De Oliveira Bernardo C, González-Chica DA, Macartney K, Menzies R, Stocks N. Pneumococcal vaccination uptake among patients aged 65 years or over in Australian general practice. Human vaccines & immunotherapeutics. 2020;16(4):965–71. doi: 10.1080/21645515.2019.1682844 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Peng Z, Hayen A, Liu B. Practice-and individual-level antibiotic prescribing associated with antibiotic treatment non-response in respiratory tract infections: a national retrospective observational study. Journal of Antimicrobial Chemotherapy. 2021;76(3):804–12. doi: 10.1093/jac/dkaa509 [DOI] [PubMed] [Google Scholar]
- 20.Statistics ABO. Socio-economic indexes for areas (SEIFA). Canberra: Australian Bureau of Statistics. 2011. [Google Scholar]
- 21.Pink B. Australian statistical geography standard (ASGS): volume 5—remoteness structure. Canberra: Australian Bureau of Statistics. 2011. [Google Scholar]
- 22.He W-Q, Kirk M, Hall J, Liu B. 264 Antibiotic use associated with acute gastroenteritis in Australian primary care. International Journal of Epidemiology. 2021;50(Supplement_1):dyab168. 272. [Google Scholar]
- 23.Peng Z, Hayen A, Hall J, Liu B. Microbiology testing and antibiotic treatment for urinary tract infections in general practice: a nationwide observational study. Infection. 2021;49(2):249–55. doi: 10.1007/s15010-020-01512-6 [DOI] [PubMed] [Google Scholar]
- 24.Public Health England D. Pneumococcal polysaccharide vaccine (PPV) coverage report, England, April 2014 to March 2015. Health Prot Rep. 2019;13(39):1–11. [Google Scholar]
- 25.Kwetkat A, Heppner HJ. Comorbidities in the elderly and their possible influence on vaccine response. Vaccines for Older Adults: Current Practices and Future Opportunities. 2020;43:73–85. doi: 10.1159/000504491 [DOI] [PubMed] [Google Scholar]
- 26.Kolditz M, Schmitt J, Pletz MW, Tesch F. Impact of pneumococcal polysaccharide vaccine on incidence and mortality after pneumonia in adults aged≥ 60 years—a population-based retrospective cohort study. Clinical Microbiology and Infection. 2018;24(5):500–4. [DOI] [PubMed] [Google Scholar]
- 27.Domínguez À, Soldevila N, Toledo D, Torner N, Force L, Pérez MJ, et al. Effectiveness of 23-valent pneumococcal polysaccharide vaccination in preventing community-acquired pneumonia hospitalization and severe outcomes in the elderly in Spain. PLoS One. 2017;12(2):e0171943. doi: 10.1371/journal.pone.0171943 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Fletcher-Lartey S, Khan R. Perceptions of barriers to the management of respiratory tract infections in general practice settings in Australia. Australian Journal of Primary Health. 2017;23(5):471–5. doi: 10.1071/PY17017 [DOI] [PubMed] [Google Scholar]
- 29.Sargent L, McCullough A, Del Mar C, Lowe J. Is Australia ready to implement delayed prescribing in primary care?: a review of the evidence. Australian family physician. 2016;45(9):688–90. [PubMed] [Google Scholar]
- 30.Baillie EJ, Merlo G, Magin P, Tapley A, Mulquiney KJ, Davis JS, et al. Antibiotic prescribing for upper respiratory tract infections and acute bronchitis: a longitudinal analysis of general practitioner trainees. Family Practice. 2022. doi: 10.1093/fampra/cmac052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Little P, Francis NA, Stuart B, O’Reilly G, Thompson N, Becque T, et al. Antibiotics for lower respiratory tract infection in children presenting in primary care in England (ARTIC PC): a double-blind, randomised, placebo-controlled trial. The Lancet. 2021;398(10309):1417–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhuo C, Wei X, Zhang Z, Hicks JP, Zheng J, Chen Z, et al. An antibiotic stewardship programme to reduce inappropriate antibiotic prescribing for acute respiratory infections in rural Chinese primary care facilities: study protocol for a clustered randomised controlled trial. Trials. 2020;21(1):1–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
Data Availability Statement
The MedicineInsight data underlying the results in this study are now held by the Australian Commission on Safety and Quality in Healthcare (contactable at QUMprogram@safetyandquality.gov.au). Ethical approval was required to be obtained prior to use. Data should be accessible in the same manner as the authors and the authors did not have any special access privileges that other would not have.