Abstract
The lack of clinical progression in some individuals despite prolonged human immunodeficiency virus type 1 (HIV-1) infection may result from infection with less-pathogenic viral strains. To address this question, we examined the HIV-1 envelope protein from a donor with a low viral burden, stable CD4+ T-lymphocyte counts, and little evidence of CD8+ T-cell expansion, activation, or immune activity. To avoid potential changes in envelope function resulting from selection in vitro, envelope clones were constructed by using viral RNA isolated from uncultured peripheral blood mononuclear cells (PBMC). The data showed that recombinant viruses containing envelope sequences derived from RNA isolated from patient PBMC replicated poorly in primary CD4+ T cells but demonstrated efficient growth in macrophages. The unusual phenotype of these viruses could not be explained solely by differential utilization of coreceptors since the chimeric viruses, as well as an uncloned isolate obtained from the same visit date, can utilize CCR5. In addition, the donor’s own cells appeared resistant to infection with chimeric viruses containing autologous envelope sequences. Genotype analysis revealed that the donor was heterozygous for the previously described 32-bp deletion in CCR5 which may be linked with prolonged survival in HIV-1-infected individuals. These data suggest that the changes in envelope sequences confer properties of viral attenuation, which together with the CCR5 +/Δ32 genotype could account for the long-term survival of this patient.
The natural history of human immunodeficiency virus type 1 (HIV-1) infection is variable among individuals and is governed by the link between viral replication and host-specific factors. In fact, several studies have demonstrated that higher levels of HIV-1 replication are associated with more-rapid clinical progression (15, 18, 46, 65, 66, 74, 79, 82, 85). Furthermore, it is well documented that HIV-1 phenotypes associated with enhanced replication have been linked with clinical progression (14, 36, 52, 72, 87, 92, 95).
In contrast, progression rates are slower when there are effective immunologic responses associated with restraint of HIV-1 replication, including production of antiviral cytokines (9, 12, 13, 59), cytotoxic T-lymphocyte responses (6, 43, 96), and antibody-mediated suppression (5, 27, 44, 70, 77). Furthermore, recent use of more-potent and intensive antiretroviral chemotherapy has resulted in reductions in HIV-1 RNA load in plasma and in decreased rates of clinical progression for some patients (24, 31, 32, 40, 42, 51). Interestingly, there are individuals who remain clinically asymptomatic and demonstrate stable immunologic function in the absence of any therapeutic intervention more than 10 years after primary HIV-1 infection. Cohort studies estimate that this group of long-term survivors (LTS) or long-term nonprogressors constitutes approximately 1 to 5% of the infected population (7, 38, 58, 71, 86, 89). There are a number of potential explanations for the phenomenon of slow progression, including infection with attenuated viral strains demonstrating limited capacity for high-level replication in vivo (21, 54, 58, 64).
We report here the functional characteristics of chimeric viruses containing env sequences from an LTS infected with a virus that repeatedly replicates with slower growth kinetics in coculture relative to viral isolates from other LTS and recent seroconverters. This finding suggests that the donor may be infected with a virus which possesses a selective growth disadvantage. Our studies demonstrate both HIV-1 attenuation and an apparently diminished susceptibility to infection in the donor as potential factors for slow clinical progression in this patient.
MATERIALS AND METHODS
Description of donor 6.
The clinical course of this patient, referred to as donor 6 (based on a previous publication [34]), was described. In brief, this individual tested positive for HIV-1 in December 1984 at the time of first presentation to the San Francisco Men’s Health Study Clinic, with a CD4+ T-cell count of 576 cells/mm3 and with a median CD4+ T-cell count of 582 since that time. Viral burden at the time of testing was 6,686 copies/ml.
Viral isolation in CD4+ T lymphocytes.
CD4+ T lymphocytes from seropositive individuals were selected onto commercially available flasks coated with anti-CD4 antibodies (Applied Immune Sciences [AIS]). For this purpose, 30 × 106 peripheral blood mononuclear cells (PBMC) were added to each flask and after a 1-h incubation, the nonadherent cells were removed. Seven million allogeneic CD4+ T lymphocytes previously activated with anti-CD3 (Ortho) antibody (see below for methods) (45) were added to the adherent cell population, and the cultures were maintained in the presence of 5,000 U of interleukin-2 (Chiron) per ml for 21 to 28 days. Culture supernatants were sampled every 7 days, at which time fresh medium was added to the cultures.
Preparation of CD4+ lymphocytes and macrophages for viral replication studies.
CD4+ lymphocytes from a total of six HIV-1-seronegative Red Cross Blood Bank donors were selected onto individual AIS flasks as outlined above. Each flask was treated with anti-CD3 antibody and interleukin-2 for 5 days. Pools of three donors each were prepared at the end of the incubation period and were cryopreserved for future use. Pools of three donors were used to minimize host genetic differences in infectivity potential among different donors. Cells were used for infection within 2 days after thaw. Macrophages (M∅) were prepared as previously described (75). Cells from three donors were cultured for each experiment, and the cells were pooled and plated for infections on day 6. As stated above, pools of cells limited the variability seen with different donors. This was particularly important since the M∅ could not be cryopreserved. Infections were performed on 7-day-old M∅. The M∅ donors were different from the CD4+ T-cell donors.
Amplification of viral RNA sequences.
A blood sample was obtained from donor 6 in December 1994. Cell-associated RNA was isolated from PBMC using TRI-Reagent (Molecular Research Center). Purified RNA was diluted to the limit of detection for env-specific nested reverse transcription-PCR (RT-PCR). The 962-bp amplified fragment included the region encoding V1 to V3 of the HIV-1 envelope. The outer primers used for PCR (Oligos etc) were as follows: sense A2 (HIVNL4-3 [1], nucleotide positions 6320 to 6340) and antisense MFN 20 (HIVNL4-3, nucleotide positions 7352 to 7333). The inner primers were as follows: sense KPN (HIVNL4-3, nucleotide positions 6343 to 6364) and antisense MFN 17 (HIVNL4-3, nucleotide positions 7327 to 7305). Primer KPN includes the KpnI restriction site, and primer MFN 17 includes the MstII restriction site used for subsequent cloning (see below). MFN 20 was used to prime the RT reaction, using Superscript II Moloney murine leukemia virus reverse transcriptase (Gibco) in 50 mM Tris (pH 8.3), 75 mM KCl, and 3 mM MgCl2. PCR samples were gel purified and cloned into the pCRII vector (Invitrogen).
Construction of full-length HIV-1 molecular clones.
Patient-derived envelope sequences were excised from the pCRII vector after digestion with KpnI and MstII. The fragments were gel purified as above, and full-length HIV-1 chimeric clones were constructed. Clones were screened on the basis of restriction digestion.
Sequence analysis.
Plasmid DNA from full-length clones was prepared using an anion-exchange resin membrane (Qiagen, Inc.), and it was subjected to cycle sequencing with an automated DNA sequencer (Applied Biosystems, Inc.). Nucleotide sequences were aligned by Clustal analysis.
Generation and testing of chimeric virus stocks.
Full-length HIV-1 recombinant plasmids (25 μg) were transfected into 293T cells (5 × 106 cells) by standard methods (83). Virus supernatants were collected at 48 h and tested for p24 antigen (Coulter). Alternatively, 25 μg of DNA was electroporated into 5 × 106 phytohemagglutinin-stimulated PBMC, which were immediately cocultured with an additional 5 × 106 PBMC from the same donor. Twenty-four-hour harvests were performed on days 4 to 7, and p24 antigen testing was performed. Viral growth kinetic studies were performed on allogeneic pools of purified monocyte-derived M∅ and anti-CD3-stimulated CD4+ T cells or on autologous M∅ and anti-CD3-stimulated CD4+ T cells. HIV-1 DNA formation was determined by quantitative PCR as previously described (75). The T-cell line-adapted HIVNL4-3 (1) and/or M∅-tropic HIVSX (75), encoding Env from the brain-derived, M∅-tropic HIVJRFL, were used as controls. All PCR assays (Perkin-Elmer) and p24 determinations were made in duplicate. Autologous infectivity assays were performed on M∅ and anti-CD3-stimulated CD4+ T cells isolated from donor 6. Serial half-log dilutions of viral stocks were applied to the cells for 2 h, and supernatants were changed at day 7 and harvested at day 14 to determine the 50% tissue culture infective dose/ml for each cell type.
Coreceptor usage.
Coreceptor usage was determined after infection of human osteosarcoma cells (HOS) (gift of D. Littman) expressing CD4 and one of the following chemokine receptors: CCR1, CCR2, CCR3, CCR4, CCR5, GPR15 (BONZO), STRL33 (BOB), or CXCR4. In addition, the cells contain the green fluorescence protein (GFP) under the control of the HIV long terminal repeat. Cultures infected by HIV-1 chimeras resulted in the production of GFP at 48 h postinfection. GFP expression was measured in formaldehyde-fixed (2%) samples by flow cytometry (Becton Dickinson; FACScan).
PCR analysis of CCR5 from genomic DNA.
Genomic DNA was isolated from PBMC by using DNAzol according to the manufacturer’s instructions (GIBCO BRL). Primers (CCR5+ and CCR5−) and amplification conditions were as described previously (94). One-tenth of the amplified product was run on a 4% NuSieve GTG agarose (FMC) gel in the presence of 10 μg of ethidium bromide/ml. These primers yield a 172-bp fragment for the CCR5 wild-type allele and a 142-bp fragment for the CCR5 Δ32 allele (94).
RESULTS
Slow viral growth kinetics of a virus isolated from an LTS.
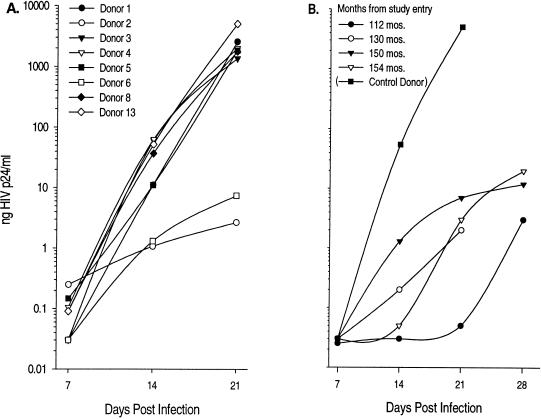
We have previously demonstrated that virus isolated from an LTS displays delayed growth in vitro (donor 6), suggesting infection with a growth-attenuated virus. Moreover, this donor has maintained relatively stable CD4+ T-cell levels since he was first identified in 1984, with little evidence of CD8+ cell activation (as measured by CD38 expression) or immune activity (as measured by cytotoxic T-lymphocyte activity against gag, pol, or env targets) (34). In the current study, we examined the kinetics of virus replication in the context of eight other donors (Fig. 1A) from our first report (34). As expected, virus from donor 6 grew slowly relative to that from five of the other HIV-1-infected LTS (donors 1, 3, 4, 5, and 8) and one recent seroconverter (donor 13). Donor 6 was tested an additional two times for virus growth patterns to confirm this observation. In all instances (Fig. 1B) virus isolation still required 14 to 21 days to reach a concentration of greater than 1 ng of p24/ml, reinforcing the presence of a poorly replicating virus. Donor 2 virus also replicated with significantly diminished growth kinetics relative to that of the other donors, but on two additional occasions we were unable to recover virus and elected not to examine this individual further in our current study.
FIG. 1.
Virus isolated from CD4+ T lymphocytes of donor 6 grows slowly in culture relative to that of other HIV-infected individuals. CD4+ lymphocytes from HIV-infected donors were cocultured with a pool of allogeneic CD4+ T cells and sampled at the times indicated. CD4+ lymphocytes were selected onto AIS flasks coated with anti-CD4 antibodies. Thirty million PBMC were added to each flask, and after a 1-h incubation, the nonadherent cells were removed. Seven million allogeneic CD4+ lymphocytes activated with anti-CD3 antibody (45) were added to the adherent cell population, and the cultures were maintained in the presence of 5,000 U of interleukin-2 (Chiron)/ml for 21 to 28 days. Culture supernatants were sampled every 7 days, at which time fresh medium was added to the cultures. (A) CD4+ lymphocytes from eight HIV-infected individuals. Donors are as indicated on the figure. Donor 13, a rapid progressor to disease, is included for the purposes of comparison. (B) CD4+ lymphocytes from donor 6 were tested on four different occasions.
Recombinant viruses containing envelope sequences from donor 6 demonstrate growth attenuation in primary CD4+ T cells but not in M∅.
We examined whether viral envelope sequences from donor 6 contribute to the slow in vitro growth phenotype we observed with virus isolated from this LTS. For this purpose, the C1 to V3 regions of the envelope were amplified from PBMC-derived RNA. This region of the envelope was chosen for study based on the numerous phenotypic properties which have been mapped to this region, including viral tropism, phenotypic diversity, and in vitro growth characteristics (17, 23, 36, 39, 75, 76, 87). Purified RNA was diluted to the limit of detection for env-specific RT-PCR (envelope region C1 to V3). We chose to use RNA to avoid potential changes in envelope function which may result from passage and selection during the isolation of virus in vitro. The function of the amplified env gene product was evaluated by inserting RT-PCR fragments into the corresponding region of an infectious molecular clone (pNL4-3). The nucleotide analysis of the cloned envelope sequences for V1, V2, and V3 demonstrated that they were closely related and comprised a relatively homogeneous population (Fig. 2).
FIG. 2.
Viral envelope sequences of the clones are homogeneous. Plasmid DNA from full-length clones were prepared by using an anion-exchange resin membrane (Qiagen, Inc.) and were subjected to cycle sequencing with an automated DNA sequencer (Applied Biosystems, Inc.). Potential N-linked glycosylation sites are underlined. The numbering system is based on the sequence of gp120 from HIVJRFL. Dashes indicate that the corresponding sequences of the recombinant clones containing donor 6 envelope sequences are identical to those of HIVJRFL. Amino acid substitutions are indicated, and deletions are identified by a boldface dot. (A) V1 sequences. (B) V2 sequences. (C) V3 sequences.
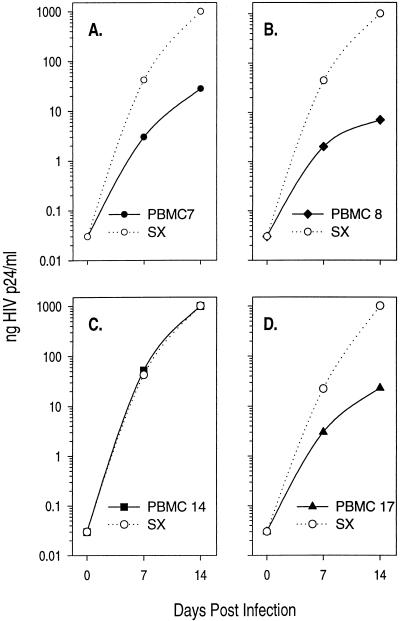
In order to determine whether chimeric viruses containing env sequences from this donor recapitulated the growth attenuation phenotype observed with uncloned virus, virus stocks were generated and infection of allogeneic CD4+ T cells was performed (Fig. 3). As with the uncloned virus shown in Fig. 1B, the recombinant viruses PBMC 7 (Fig. 3A), PBMC 8 (Fig. 3B), and PBMC 17 (Fig. 3D) also demonstrated significantly delayed kinetics of replication relative to that of the control strain HIVSX which contains envelope sequences from the M∅-tropic strain HIVJRFL cloned into HIVNL4-3 (75). This virus strain uses CCR5 and is similar to typical M∅-tropic HIV-1 strains. In contrast, the recombinant virus PBMC 14 mirrored the control virus HIVSX in its growth kinetics (Fig. 3C).
FIG. 3.
Growth kinetics of recombinant viruses on allogeneic CD4+ T cells recapitulate growth patterns of the uncloned virus. Infections were performed in 96-well microtiter plates. CD4+ lymphocytes from three donors were selected onto individual, commercially available AIS flasks coated with anti-CD4 antibodies. Each flask was treated with anti-CD3 antibody and interleukin-2 for 5 days. A pool of the three donors was prepared at the end of the incubation period, and it was cryopreserved for future use. Cells were used for infection within 2 days after thaw. For infections, 2 × 105 cells were incubated with virus made by electroporation of PBMC for 2 h as described in Materials and Methods. Clones are as indicated. Data points represent the averages of two independent experiments.
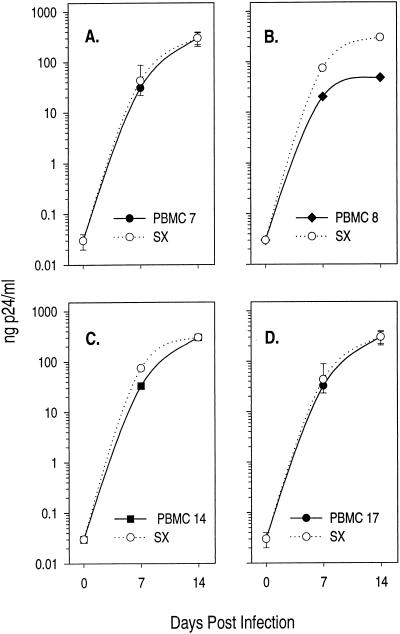
Since three of the four recombinant viruses examined exhibited growth kinetics similar to that of the uncloned virus, we reasoned that circulating virus might be growth attenuated in CD4+ T cells but might replicate efficiently in the other major target cell for HIV-1 infection, the monocyte-derived M∅. In order to examine this hypothesis, replication kinetics were examined on pools of allogeneic M∅ and evaluated in the context of infection in CD4+ T cells (Fig. 4). Most M∅-tropic strains of HIV-1, like HIVSX (76), replicate comparably in both M∅ and CD4+ T cells. In contrast, the chimeric viruses PBMC 7, 8, and 17 demonstrated better replication in M∅ than in CD4+ T cells (compare Fig. 3 with Fig. 4). Since there was no evidence for increased cell death in these cultures relative to the control HIVSX, these differences could not be explained on the basis of decreases in cell viability in the CD4 T-cell cultures. These data are consistent with our hypothesis that donor 6 is infected with a virus that demonstrates growth attenuation in CD4+ T cells.
FIG. 4.
Recombinant viruses are capable of replicating in M∅. Infections were performed in 96-well microtiter plates. For infections, 2 × 105 cells were incubated with virus made by electroporation of PBMC for 2 h as described in Materials and Methods. M∅ from three donors were prepared, and the cells were pooled and plated for infection on day 6. Infections were performed on 7-day-old M∅. The M∅ donors were different from the CD4+ T-cell donors in Fig. 3. Clones are as indicated. Data points represent the averages of at least two independent experiments. If error bars are drawn, data points represent the averages of three independent experiments.
Differences in replication kinetics are linked to the efficiency of viral entry.
The experiments described above revealed an increase in growth kinetics in M∅ versus primary CD4+ T cells. Since these chimeric viruses contained env sequences from donor 6, it appeared likely that the differences we observed were due to restrictions at the level of entry. As such, viral entry was examined by infecting purified CD4+ T cells and M∅ with the recombinant viruses PBMC 7, 14, and 17. The T-cell line-tropic HIVNL4-3 and the M∅-tropic HIVSX were included as controls. Viral entry was examined by quantitative PCR for early viral cDNA synthesis, an early step following viral entry as previously described (75). Whereas levels of viral cDNA for the M∅-tropic virus HIVSX were essentially equivalent in both CD4+ T cells and M∅ (1.4-fold higher in M∅ than in CD4+ T cells), levels of viral cDNA from chimeric viruses containing envelope sequences from donor 6 that demonstrated growth attenuation in CD4+ T cells were 6.5-fold (PBMC 17) and 15-fold (PBMC 7) higher in M∅ than in CD4+ T cells (Table 1). In contrast, PBMC 14 cDNA levels were only 2.5-fold higher in M∅ than in CD4+ T cells and most closely resembled those of the typical M∅-tropic strain HIVSX. This finding is consistent with the growth-attenuation phenotype observed in CD4+ T cells and extends the above findings to demonstrate that differences in infectivity are most likely due to differences in viral entry.
TABLE 1.
Defects in replication of chimeric viruses in CD4+ T lymphocytes are correlated with decreased entry
| Virusb | Copies of HIV-1 DNA/μg of total DNAa
|
|
|---|---|---|
| CD4+ T cellsc | M∅d | |
| NL | 590 | 40 |
| SX | 140 | 200 |
| PBMC 7 | 37 | 585 |
| PBMC 14 | 160 | 400 |
| PBMC 17 | 60 | 385 |
HIV DNA was measured by quantitative PCR 18 h after infection and are expressed as copies of HIV DNA/μg of total DNA. Heat-inactivated controls were negative (data not shown).
Infections were performed in 96-well microtiter plates. Cells (2 × 105) were infected for 2 h with 20 ng of the recombinant viruses generated in 293T cells.
CD4+ T cells were prepared as described in the legend to Fig. 2.
M∅ were prepared as described in the legend to Fig. 3.
LTS envelope-containing viruses use CCR5 for infection.
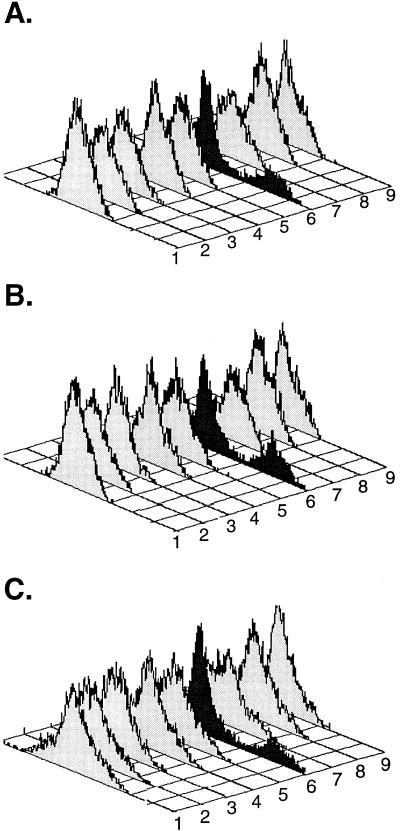
Viral entry is influenced by a number of parameters, including the potential use of a wide spectrum of newly described second receptors (2, 10, 26, 28, 29, 33, 60). Since we had observed a preferential replication in M∅ over primary CD4+ T cells, we hypothesized that this phenotype might be linked to coreceptor use. Therefore, we characterized the use of known coreceptors by our chimeric viruses. For these experiments, we used stable HOS cell transfectants (GHOST cells) which coexpress CD4 and the chemokine receptors CCR1 through CCR5, CXCR4, BOB (STRL33), or BONZO (GPR15). Despite differences in infectivity of M∅ versus CD4+ T cells, the recombinant viruses we tested, as well as the uncloned virus obtained from the same visit date, used only the β-chemokine receptor CCR5 (Fig. 5). These data indicate that differences in infectivity patterns with these viruses are not due to differential use of known coreceptors.
FIG. 5.
Recombinant viruses use CCR5 for entry in a single round of infection. Coreceptor usage was determined after infection of HOS cells (gift of D. Littman) expressing CD4 and one of the following chemokine receptors: CCR1, CCR2, CCR3, CCR4, CCR5, GPR15 (BONZO), STRL33 (BOB), or CXCR4. In addition, the cells contain the GFP under the control of the HIV long terminal repeat. Cultures infected by HIV-1 chimeras resulted in the production of GFP 48 h postinfection. GFP expression was measured in paraformaldehyde-fixed (2%) samples by flow cytometry (Becton Dickinson; FACScan). GHOST cells were infected with 50 to 70 ng of recombinant virus and cultured for 2 days. (A) PBMC 7. (B) PBMC 14. (C) PBMC 17. Rows: 1, GHOST CD4 cells; 2, GHOST CD4 + CCR1 cells; 3, GHOST CD4 + CCR2b; 4, GHOST CD4 + CCR3; 5, GHOST CD4 + CCR4; 6, GHOST CD4 + CCR5; 7, GHOST CD4 + BOB; 8, GHOST CD4 + BONZO; 9, GHOST CD4 + CXCR4. In this assay, approximately 10 to 20% of the cells will be positive in a single round of infection. Infections performed with vesicular stomatitis virus protein-G pseudotyped envelope yielded an average of 10% positive cells in all nine cell lines (data not shown).
Chimeric viruses replicate to lower titers in the cells of donor 6.
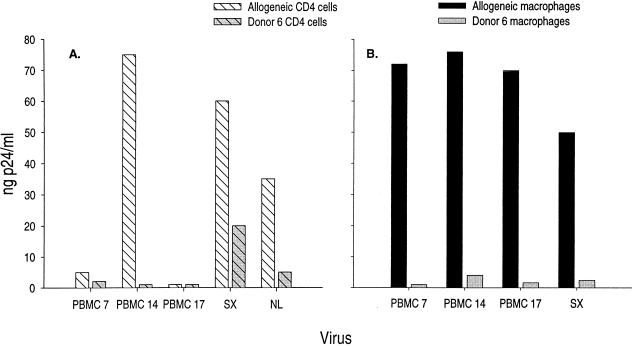
Our data thus far implicated viral envelope sequences in the growth-attenuated phenotype we have observed with a virus from an LTS. It was possible, however, that a host determinant might influence this donor’s long-term survival. In order to address the latter possibility, we examined viral replication in cells from donor 6. We found that chimeric viruses replicated less efficiently than HIVSX in CD4+ T cells from donor 6 (Fig. 6). These data suggest a decreased susceptibility to infection of both CD4+ T cells and M∅ isolated from donor 6 by autologous as well as heterologous (HIVNL4-3 and HIVSX) envelope-containing viruses.
FIG. 6.
Cells from donor 6 are less susceptible to infection by virus containing autologous envelope sequences as well as heterologous envelope sequences. Infections were performed in 96-well microtiter plates. For infections, 2 × 105 cells were incubated with 50 pg of virus for 2 h. Allogeneic M∅ (B) and CD4+ T cells (A) were used as pools as described in the legends to Fig. 3 and 4. Infections were performed on 7-day-old M∅. The M∅ donors were different from the CD4+ T-cell donors. M∅ and CD4+ T cells from donor 6 were prepared according to the same methods. Clones are as indicated. Samples were tested for p24 production at day 14.
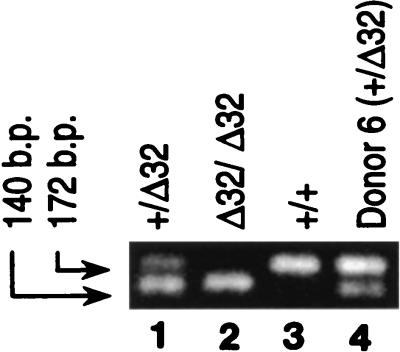
Previous reports have indicated that 20% of the Caucasian population are heterozygous for a 32-bp deletion in the CCR5 allele (+/Δ32) (22, 61, 84). Whereas this mutation has been demonstrated to confer protection from infection on individuals who are Δ32/Δ32 (61, 84), the heterozygous condition (+/Δ32) has been reported to correlate with delayed progression to disease (22, 47, 69). Since the data described above suggested a potential for decreased susceptibility of the donor’s own cells to infection with HIV-1 containing autologous envelope sequences, the CCR5 allele was examined. As shown in Fig. 7, donor 6 was heterozygous (lane 4) for the CCR5 mutant allele. This finding is consistent with the decreased susceptibility to infection with the recombinant CCR5-tropic viruses reported above. It is interesting to note that CD4+ T cells from donor 6 were also more resistant to infection with a CXCR4-tropic virus (HIVNL4-3) relative to allogeneic CD4+ T cells. This finding raises the possibility that there are other blocks to infection in the CD4+ T cells of donor 6 not identified by these studies.
FIG. 7.
Donor 6 is CCR5 +/Δ32. Genomic DNA was extracted as described in Materials and Methods. One-tenth of the amplified product was run on a 4% NuSieve agarose gel in the presence of ethidium bromide. Lanes 1 to 3 contain control samples and are labeled accordingly. Lane 4 contains the sample from donor 6. A 172-bp band is indicative of the +/+ condition (lane 3), a 140-bp band is indicative of the Δ32/Δ32 condition (lane 2), and both bands indicate the heterozygous condition +/Δ32 (lanes 1 and 4).
DISCUSSION
Despite extensive study, much remains to be learned about the virologic determinants of HIV pathogenesis. We describe here the infection of an LTS with a CD4+ T-cell growth-attenuated virus. Although it has been reported that viral isolates from LTS generally replicate poorly in culture, the kinetics of replication were much slower than those seen for other LTS we studied as controls (Fig. 1A). Furthermore, it may be important that viral growth attenuation was observed in CD4+ T cells, a major target for HIV infection in vivo (20, 63). This phenotype was reproduced with chimeric viruses containing RNA-derived env sequences between C1 and V3. Close examination revealed a diminished susceptibility to replicate in CD4+ T cells in three of four recombinant viruses, although infection did occur in M∅. Entry was also less efficient in CD4+ T cells than in M∅. Finally, we observed a decrease in the ability to infect cells isolated from donor 6 by viruses containing envelope sequences derived from his own PBMC RNA as well as those from other unrelated HIV-1 strains. This observation may be related to the heterozygous +/Δ32 genotype.
LTS almost uniformly demonstrate a low viral burden (8, 34), but factors in addition to production rate may also influence the speed of clinical progression. These include infection with attenuated strains (8, 21, 54, 64, 67, 68, 78), the extent of viral diversity (25, 41, 62, 73, 81, 92), and the relative cytopathicity (14, 19, 52, 72, 88, 95) of the infecting and/or evolving virus. Among all these factors, the potential for infection with an attenuated virus has received the most attention. Experimental evidence for HIV attenuation has been described in vitro, following alteration of the auxiliary genes of HIV-1, including nef (11, 50), vif (35, 37, 91), vpr (4, 80), or vpu (30, 55). Furthermore, viruses containing deletions in the nef (53) or vpr (57) gene of simian immunodeficiency virus SIVmaq have been shown to protect adult rhesus macaques against challenge with wild-type SIV, although neonatal macaques were not similarly protected (3). In addition, a lack of intact nef sequences in HIV-1 infection in vivo has been described in some (21, 54, 64, 67, 68), but not all (48), LTS. There is also a report of an attenuated Rev variant in an LTS, but this mutation was also detected in two of four progressors (49). Finally, Connor et al. observed defects in either synthesis or processing of envelope precursor protein (gp160) in clones derived from four of six LTS (16).
We report here on viral env sequences spanning C1 through V3 which confer growth attenuation onto primary CD4+ T cells but not onto primary M∅. Furthermore, we demonstrate that these sequences are responsible for differences in viral entry. We tested the hypothesis that the block to highly productive infection in CD4+ T cells occurs at the level of interaction between viral gp120 and the coreceptor molecule at the cell surface. All chimeric viruses tested, as well as an uncloned culture isolate from this individual, used CCR5 as a coreceptor for infection. It is of interest that CCR5 was used by the viral isolates examined in this study, since this molecule is expressed on both activated CD4+ T cells and M∅ (94). Consequently, our studies cannot exclude the possibility that viruses encoding envelope proteins from donor 6 may use a novel receptor that is present on M∅ but absent on activated CD4+ T cells. In this model, circulating virus may be able to use CCR5 but might preferentially utilize an additional, undefined receptor on M∅. As such, these viruses would have a distinct growth advantage in M∅, since they could enter these cells via two different pathways.
Interestingly, we found that donor 6 was heterozygous (+/Δ32) for the 32-bp deletion in CCR5. This genetic marker has been implicated as an HIV-1 survival factor in some studies (22, 47). Individuals with the homozygous Δ32/Δ32 genotype are resistant to infection with CCR5-tropic viruses due to defects in entry (16, 47). Therefore, it follows that heterozygotes might demonstrate diminished susceptibility to infection with these same viruses. Other viral strains use different chemokine receptors for entry, which may also have an impact on clinical progression. For instance, a point mutation in CCR2 was linked to long-term survival (90); however, recent data indicate that this phenomenon may be a surrogate marker for a mutation in the CCR5 regulatory region (56). Finally, it has been reported that a polymorphism in the 3′ untranslated region of the gene for the ligand (SDF-1) for the coreceptor molecule CXCR4 correlates with survival (93). Although a consensus regarding these analyses is still evolving, they clearly demonstrate that host genetics are likely to play a major role in the rate of disease progression by impacting the interaction of HIV-1 and its cognate receptors at the level of viral entry.
The data presented here support the hypothesis that viral entry may be a critical determinant of long-term survival. Our results indicate that genetic differences in viral envelope sequences which result in inefficient entry into cells may be important determinants of long-term survival. While the current study focused on envelope sequences, it is also possible that other viral genes may be involved in the growth attenuation phenotype of uncloned virus from this individual. Given the impact of host genetic factors, it appears that both viral and genetic influences are at play in this particular individual’s delayed clinical progression. Taken together, studies of delayed progressors to AIDS will continue to offer insights into the mechanisms of viral persistence in the absence of clinical progression.
ACKNOWLEDGMENTS
We express our sincere thanks to the volunteer who donated specimens for this study. We thank Stephanie Lu, Christine Yeramian, and Mary Ann Hausner for expert technical assistance; the staff of the UCLA Multicenter AIDS Cohort Study, especially Roger Detels, Denis Miles, and Martin Majchrowicz; and the Medical Research Service of the Department of Veterans Affairs. We thank Betty Poon and Sheila Stewart for valuable reagents, helpful discussions, and critical reading of the manuscript.
This work was supported by grants UO1-AI-35040, UO1-AI-37613, UO1-AI-28697, VA103, and T32-AI-07988 (K.G.-F.).
REFERENCES
- 1.Adachi A, Gendelman E, Koenig S, Folks T, Willey R, Rabson A, Martin M A. Production of acquired immunodeficiency syndrome-associated retrovirus in human and nonhuman cells transfected with an infectious molecular clone. J Virol. 1986;59:284–291. doi: 10.1128/jvi.59.2.284-291.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Alkhatib G, Combadiere C, Broder C C, Feng Y, Kennedy P E, Murphy P M, Berger E A. CC CKR5: a RANTES, MIP-1α, MIP-1β receptor as a fusion cofactor for macrophage-tropic HIV-1. Science. 1996;272:1955–1958. doi: 10.1126/science.272.5270.1955. [DOI] [PubMed] [Google Scholar]
- 3.Baba T W, Jeong Y S, Pennick D, Bronson R, Greene M F, Ruprecht R M. Pathogenicity of live, attenuated SIV after mucosal infection of neonatal macaques. Science. 1995;267:1820–1825. doi: 10.1126/science.7892606. [DOI] [PubMed] [Google Scholar]
- 4.Balliet J W, Kolson D L, Eiger G, Kim F M, McGann K A, Srinivasan A, Collman R. Distinct effects in primary macrophages and lymphocytes of the human immunodeficiency virus type 1 accessory genes vpr, vpu, and nef: mutational analysis of a primary HIV-1 isolate. Virology. 1994;200:623–631. doi: 10.1006/viro.1994.1225. [DOI] [PubMed] [Google Scholar]
- 5.Binley J M, Klasse P J, Cao Y, Jones I, Markowitz M, Ho D D, Moore J P. Differential regulation of the antibody responses to Gag and Env proteins of human immunodeficiency virus type 1. J Virol. 1997;71:2799–2809. doi: 10.1128/jvi.71.4.2799-2809.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Borrow P, Lewicki H, Wei X, Horwitz M S, Peffer N, Meyers H, Nelson J A, Gairin J E, Hahn B H, Oldstone M B A, Shaw G M. Antiviral pressure exerted by HIV-1-specific cytotoxic T lymphocytes (CTLs) during primary infection demonstrated by rapid selection of CTL escape virus. Nat Med. 1997;3:205–211. doi: 10.1038/nm0297-205. [DOI] [PubMed] [Google Scholar]
- 7.Buchbinder S P, Katz M H, Hessol N A, O’Malley P M, Holmberg S D. Long-term HIV-1 infection without immunologic progression. AIDS. 1994;8:1123–1128. doi: 10.1097/00002030-199408000-00014. [DOI] [PubMed] [Google Scholar]
- 8.Cao Y, Qin L, Linqi Z, Safrit J, Ho D D. Virologic and immunologic characterization of long-term survivors of human immunodeficiency virus type 1 infection. N Engl J Med. 1995;332:201–208. doi: 10.1056/NEJM199501263320401. [DOI] [PubMed] [Google Scholar]
- 9.Chen Y, Gupta P. CD8+ T-cell mediated suppression of HIV-1 infection may not be due to chemokines RANTES, MIP-1α and MIP-1β. AIDS. 1997;10:1434–1435. doi: 10.1097/00002030-199610000-00020. [DOI] [PubMed] [Google Scholar]
- 10.Choe H, Farzan M, Sun Y, Sullivan N, Rollins B, Ponath P D, Wu L, Mackay C R, LaRosa G, Newman W, Gerard N, Gerard C, Sodroski J. The β-chemokine receptors CCR3 and CCR5 facilitate infection by primary HIV-1 isolates. Cell. 1996;85:1135–1148. doi: 10.1016/s0092-8674(00)81313-6. [DOI] [PubMed] [Google Scholar]
- 11.Chowers M Y, Spina C A, Kwoh T J, Fitch N J, Richman D D, Guatelli J C. Optimal infectivity in vitro of human immunodeficiency virus type 1 requires an intact nef gene. J Virol. 1994;68:2906–2914. doi: 10.1128/jvi.68.5.2906-2914.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Clerici M, Balotta C, Trabattoni D, Papango L, Ruzzante S, Rusconi S, Fusi M L, Colombo M C, Galli M. Chemokine production in HIV-seropositive long-term asymptomatic individuals. AIDS. 1997;10:1432–1433. doi: 10.1097/00002030-199610000-00019. [DOI] [PubMed] [Google Scholar]
- 13.Cocchi F, DeVico A L, Garzino-Demo A, Arya S K, Gallo R C, Lusso P. Identification of RANTES, MIP-1α, and MIP-1β as the major HIV-suppressive factors produced by CD8+ T cells. Science. 1995;270:1811–1815. doi: 10.1126/science.270.5243.1811. [DOI] [PubMed] [Google Scholar]
- 14.Connor R I, Ho D D. Human immunodeficiency virus type 1 variants with increased replicative capacity develop during the asymptomatic stage before disease progression. J Virol. 1994;68:4400–4408. doi: 10.1128/jvi.68.7.4400-4408.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Connor R I, Mohri H, Cao Y, Ho D D. Increased viral burden and cytopathicity correlate temporally with CD4+ T-lymphocyte decline and clinical progression in human immunodeficiency virus type 1-infected individuals. J Virol. 1993;67:1772–1777. doi: 10.1128/jvi.67.4.1772-1777.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Connor R I, Paxton W A, Sheridan K E, Koup R A. Macrophages and CD4+ T lymphocytes from two multiply exposed, uninfected individuals resist infection with primary non-syncytium-inducing isolates of human immunodeficiency virus type 1. J Virol. 1996;70:8758–8764. doi: 10.1128/jvi.70.12.8758-8764.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Connor R I, Sheridan K E, Ceradini D, Choe S, Landau N R. Change in coreceptor usage correlates with disease progression in HIV-1 infected individuals. J Exp Med. 1997;185:621–628. doi: 10.1084/jem.185.4.621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Coombs R W, Collier A C, Allain J P, Nikora B, Leuther M, Gjerset G F, Corey L. Plasma viremia in human immunodeficiency virus infection. N Engl J Med. 1989;321:1626–1631. doi: 10.1056/NEJM198912143212402. [DOI] [PubMed] [Google Scholar]
- 19.Cornelissen M, Mulder-Kampinga G, Veenstra J, Zorgdrager F, Kuiken C, Hartman S, Dekker J, van der Hoek L, Sol C, Coutinho R. Syncytium-inducing (SI) phenotype suppression at seroconversion after intramuscular inoculation of a non-syncytium-inducing/SI phenotypically mixed human immunodeficiency virus population. J Virol. 1995;69:1810–1818. doi: 10.1128/jvi.69.3.1810-1818.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Dalgleish A G, Beverley P C L, Clapham P R, Crawford D H, Greaves M F, Weiss R A. The CD4 (T4) antigen is an essential component of the receptor for the AIDS retrovirus. Nature. 1984;312:763–767. doi: 10.1038/312763a0. [DOI] [PubMed] [Google Scholar]
- 21.Deacon N J, Tsykin A, Solomon A, Smith K, Ludford-Menting M, Hooker D J, McPhee D A, Greenway A L, Ellett A, Chatfield C. Genomic structure of an attenuated quasi species of HIV-1 from a blood transfusion donor and recipients. Science. 1995;270:988–991. doi: 10.1126/science.270.5238.988. [DOI] [PubMed] [Google Scholar]
- 22.Dean M, Carrington M, Winkler C, Huttley G A, Smith M W, Allikmets R, Goedert J J, Buchbinder S P, Vittinghoff E, Gomperts E, Donfield S, Vlahov D, Kaslow R, Saah A, Rinaldo C, Detels R, O’Brien S J. Genetic restriction of HIV-1 infection and progression to AIDS by a deletion allele of the CKR5 structural gene. Science. 1996;273:1856–1862. doi: 10.1126/science.273.5283.1856. [DOI] [PubMed] [Google Scholar]
- 23.DeJong J-J, Goudsmit J, Keulen W, Klaver B, Krone W, Tersmette M, deRonde A. Human immunodeficiency virus type 1 clones chimeric for the envelope V3 domain differ in syncytium formation and replication capacity. J Virol. 1992;66:757–765. doi: 10.1128/jvi.66.2.757-765.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Delta Coordinating Committee. Delta: a randomised double-blind controlled trial comparing combinations of zidovudine plus didanosine or zalcitabine with zidovudine alone in HIV-infected individuals. Lancet. 1996;348:283–291. [PubMed] [Google Scholar]
- 25.Delwart E L, Sheppard H W, Walker B D, Goudsmit J, Mullins J I. Human immunodeficiency virus type 1 evolution in vivo tracked by DNA heteroduplex mobility assays. J Virol. 1994;68:6672–6683. doi: 10.1128/jvi.68.10.6672-6683.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Deng H, Liu R, Ellmeier W, Choe S, Unutmaz D, Burkhart M, Di Marzio P, Marmon S, Sutton R E, Hill C M, Davis C B, Peiper S C, Schall T J, Littman D R, Landau N R. Identification of a major co-receptor for primary isolates of HIV-1. Nature. 1996;381:661–666. doi: 10.1038/381661a0. [DOI] [PubMed] [Google Scholar]
- 27.Devash Y, Calvelli T A, Wood D G, Reagan K J, Rubinstein A. Vertical transmission of human immunodeficiency virus is correlated with the absence of high-affinity/avidity maternal antibodies to the gp120 principal neutralizing domain. Proc Natl Acad Sci USA. 1990;87:3445–3449. doi: 10.1073/pnas.87.9.3445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Doranz B J, Berson J F, Rucker J, Doms R W. Chemokine receptors as fusion cofactors for human immunodeficiency virus type 1 (HIV-1) Immunol Res. 1997;16:15–28. doi: 10.1007/BF02786321. [DOI] [PubMed] [Google Scholar]
- 29.Dragic T, Litwin V, Allaway G P, Martin S R, Huang Y, Nagashima K A, Cayanan C, Maddon P J, Koup R A, Moore J P, Paxton W A. HIV-1 entry into CD4+ cells is mediated by the chemokine receptor CC-CKR-5. Nature. 1996;381:667–673. doi: 10.1038/381667a0. [DOI] [PubMed] [Google Scholar]
- 30.Du B, Wolf A, Lee S, Terwilliger E. Changes in the host range and growth potential of an HIV-1 clone are conferred by the vpu gene. Virology. 1993;195:260–264. doi: 10.1006/viro.1993.1370. [DOI] [PubMed] [Google Scholar]
- 31.Enger C, Graham N, Peng Y, Chmiel J S, Kingsley L A, Detels R, Munoz A. Survival from early, intermediate, and late stages of HIV infection. JAMA. 1996;275:1329–1334. [PubMed] [Google Scholar]
- 32.Eron J J, Benoit S L, Jemsek J, MacArthur R D, Santana J, Quinn J B, Kuritzkes D R, Fallon M A, Rubin M. Treatment with lamivudine, zidovudine, or both in HIV-positive patients with 200 to 500 CD4+ cells per cubic millimeter. N Engl J Med. 1995;333:1662–1669. doi: 10.1056/NEJM199512213332502. [DOI] [PubMed] [Google Scholar]
- 33.Feng Y, Broder C C, Kennedy P E, Berger E A. HIV-1 entry cofactor: functional cDNA cloning of a seven-transmembrane, G protein-coupled receptor. Science. 1996;272:872–877. doi: 10.1126/science.272.5263.872. [DOI] [PubMed] [Google Scholar]
- 34.Ferbas J, Kaplan A H, Hausner M A, Hultin L E, Matud J L, Liu Z, Panicali D L, Nerng-Ho H, Detels R, Giorgi J V. Virus burden in long-term survivors of human immunodeficiency virus (HIV) infection is a determinant of anti-HIV CD8+ lymphocyte activity. J Infect Dis. 1995;172:329–339. doi: 10.1093/infdis/172.2.329. [DOI] [PubMed] [Google Scholar]
- 35.Fisher A G, Ensoli B, Ivanoff L, Chamberlain M, Petteway S, Ratner L, Ballo R C, Wong S F. The sor gene of HIV-1 is required for efficient virus transmission in vitro. Science. 1987;237:888–893. doi: 10.1126/science.3497453. [DOI] [PubMed] [Google Scholar]
- 36.Fouchier R A M, Groenink M, Kootstra N A, Tersmette M, Huisman H G, Miedema F, Schuitemaker H. Phenotype-associated sequence variation in the third variable domain of the human immunodeficiency virus type 1 gp120 molecule. J Virol. 1992;66:3183–3187. doi: 10.1128/jvi.66.5.3183-3187.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Gabuzda D H, Lawrence K, Langhoff E, Terwilliger E, Dorfman T, Haseltine W, Sodroski J. Role of Vif in replication of human immunodeficiency virus type 1 in CD4+ T lymphocytes. J Virol. 1992;66:6489–6495. doi: 10.1128/jvi.66.11.6489-6495.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Gange S J, Mun̈oz A, Schrager L K, Margolick J B, Giorgi J V, Saah A J, Rinaldo C R, Detels R, Phair J P. Design of nested studies to identify factors related to late progression of HIV infection. J Acquired Immune Defic Syndr Hum Retrovirol. 1997;15:S5–S9. [Google Scholar]
- 39.Groenick M, Fouchier R A, Broersen S, Baker C H, Koot M, van’t Wout A B, Huisman H G, Miedema F, Tersmette M, Schuitemaker H. Relation of phenotype evolution of HIV-1 to envelope configuration. Science. 1993;260:1513–1516. doi: 10.1126/science.8502996. [DOI] [PubMed] [Google Scholar]
- 40.Gulick R M, Mellors J W, Havlir D, Eron J J, Gonzalez C, McMahon D, Richman D D, Valentine F T, Rooney J, Jonas L, Meibohm A, Emini E A, Chodakewitz J, et al. Treatment with indinavir, zidovudine and lamivudine in adults with HIV infection and prior antiretroviral therapy. N Engl J Med. 1997;337:734–739. doi: 10.1056/NEJM199709113371102. [DOI] [PubMed] [Google Scholar]
- 41.Hahn B H, Shaw G M, Taylor M E, Redfield R R, Markham P D, Salahudin S Z, Wong-Staal F, Gallo R C, Parks E S, Parks W P. Genetic variation in HTLV-III/LAV over time in patients with AIDS or at risk for AIDS. Science. 1986;232:1548–1553. doi: 10.1126/science.3012778. [DOI] [PubMed] [Google Scholar]
- 42.Hammer S M, Katzenstein D A, Hughes M D, Gundacker H, Schooley R T, Haubrich R H, Henry W K, Lederman M M, Phair J P, Niu M, Hirsch M S, Merigan T C. A trial comparing nucleoside monotherapy with combination therapy in HIV-infected adults with CD4 counts from 200–500 per cubic millimeter. N Engl J Med. 1996;335:1081–1090. doi: 10.1056/NEJM199610103351501. [DOI] [PubMed] [Google Scholar]
- 43.Harrer, E., T. Harrer, S. Buchbinder, D. L. Mann, M. Feinberg, T. Yilma, R. P. Johnson, and B. D. Walker. 1994. HIV-1-specific cytotoxic T lymphocyte response in healthy, long-term. AIDS Res. Hum. Retroviruses 10(Suppl 2):S77–S78. [PubMed]
- 44.Harrer T, Harrer E, Kalams S A, Elbeik T, Staprans S I, Feinberg M B, Cao Y, Ho D D, Yilma T, Caliendo A M, Johnson R P, Buchbinder S P, Walker B D. Strong cytotoxic T cell and weak neutralizing antibody responses in a subset of persons with stable nonprogressing HIV type 1 infection. AIDS Res Hum Retroviruses. 1996;12:585–592. doi: 10.1089/aid.1996.12.585. [DOI] [PubMed] [Google Scholar]
- 45.Hausner M A, Giorgi J V, Plaeger-Marshall S. A reproducible method to detect CD8 T cell mediated inhibition of HIV production from naturally infected CD4 cells. J Immunol Methods. 1993;157:181–187. doi: 10.1016/0022-1759(93)90085-l. [DOI] [PubMed] [Google Scholar]
- 46.Hogervorst E, Jurriaans S, de Wolf F, van Wijk A, Wiersma A, Valk M, Roos M, van Gemen B, Coutinho R, Miedema F. Predictors for non- and slow progression in human immunodeficiency virus (HIV) type 1 infection: low viral RNA copy numbers in serum and maintenance of high HIV-1 p24-specific but not V3-specific antibody levels. J Infect Dis. 1995;171:811–821. doi: 10.1093/infdis/171.4.811. [DOI] [PubMed] [Google Scholar]
- 47.Huang Y, Paxton W A, Wolinsky S M, Neumann A U, Zhang L, He T, Kang S, Ceradini D, Jin Z, Yazdanbakhsh K, Kunstman K, Erickson D, Dragon E, Landau N R, Phair J, Ho D D, Koup R A. The role of a mutant CCR5 allele in HIV-1 transmission and disease progression. Nat Med. 1996;2:1240–1243. doi: 10.1038/nm1196-1240. [DOI] [PubMed] [Google Scholar]
- 48.Huang Y, Zhang L, Ho D D. Characterization of nef sequences in long-term survivors of human immunodeficiency virus type 1 infection. J Virol. 1995;69:93–100. doi: 10.1128/jvi.69.1.93-100.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Iversen A K, Shpaer E G, Rodrigo A G, Hirsch M S, Walker B D, Sheppard H W, Merigan T C, Mullins J I. Persistence of attenuated rev genes in a human immunodeficiency virus type 1-infected asymptomatic individual. J Virol. 1995;69:5743–5753. doi: 10.1128/jvi.69.9.5743-5753.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Jamieson B D, Aldrovandi G M, Planelles V, Jowett J B M, Gao L, Bloch L M, Chen I S Y, Zack J A. Requirement of human immunodeficiency virus type 1 nef for in vivo replication and pathogenicity. J Virol. 1994;68:3478–3485. doi: 10.1128/jvi.68.6.3478-3485.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Katzenstein D A, Hammer S M, Hughes M D, Gundacker H, Jackson J B, Fiscus S, Rasheed S, Elbeik T, Reichman R, Japour A, Merigan T C, Hirsch M S. The relation of virologic and immunologic markers to clinical outcomes after nucleoside therapy in HIV-infected adults with 200 to 500 CD4 cells per cubic millimeter. New Engl J Med. 1996;335:1091–1098. doi: 10.1056/NEJM199610103351502. [DOI] [PubMed] [Google Scholar]
- 52.Keet I P M, Krijnen P, Koot M, Lange J M A, Miedema F, Goudsmit J, Coutinho R A. Predictors of rapid progression to AIDS in HIV-1 seroconverters. AIDS. 1993;7:51–57. doi: 10.1097/00002030-199301000-00008. [DOI] [PubMed] [Google Scholar]
- 53.Kestler H W, Ringler D J, Mori K, Panicali D L, Sehgal P K, Daniel M D, Desrosiers R C. Importance of the nef gene for maintenance of high virus loads and for development of AIDS. Cell. 1991;65:651–662. doi: 10.1016/0092-8674(91)90097-i. [DOI] [PubMed] [Google Scholar]
- 54.Kirchhoff F, Greenough T C, Brettler D B, Sullivan J L, Desrosiers R C. Brief report: absence of intact nef sequences in a long-term survivor with nonprogressive HIV-1 infection. N Engl J Med. 1995;332:228–232. doi: 10.1056/NEJM199501263320405. [DOI] [PubMed] [Google Scholar]
- 55.Klimkait T, Strebel K, Hoggan M D, Martin M A, Orenstein J M. The human immunodeficiency virus type-specific protein Vpu is required for efficient virus maturation and release. J Virol. 1990;64:621–629. doi: 10.1128/jvi.64.2.621-629.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kostrikis L G, Huang Y, Moore J, Wolinsky S M, Zhang L, Gou Y, Deutsch L, Phair J P, Neumann A U, Ho D D. A chemokine receptor CCR2 allele delays HIV-1 disease progression and is associated with a CCR5 promoter mutation. Nat Med. 1998;4:350–353. doi: 10.1038/nm0398-350. [DOI] [PubMed] [Google Scholar]
- 57.Lang S M, Weeger M, Stahl-Hennig C, Coulibaly C, Hunsmann G, Muller J, Muller-Hermelink H, Fuchs D, Wachter H, Daniel M, Desrosiers R, Fleckenstein B. Importance of vpr for infection of rhesus monkeys with simian immunodeficiency virus. J Virol. 1993;67:902–912. doi: 10.1128/jvi.67.2.902-912.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Learmont J, Tindall B, Evans L, Cunningham A, Cunningham P, Wells J, Penny R, Kaldor J, Cooper D A. Long-term symptomless HIV-1 infection in recipients of blood products. Lancet. 1992;340:863–867. doi: 10.1016/0140-6736(92)93281-q. [DOI] [PubMed] [Google Scholar]
- 59.Levy J A, Mackewicz C E, Barker E. Controlling HIV pathogenesis: the role of the noncytotoxic anti-HIV response of CD8+ T cells. Immunol Today. 1996;17:217–224. doi: 10.1016/0167-5699(96)10011-6. [DOI] [PubMed] [Google Scholar]
- 60.Liao F, Alkhatib G, Peden K W, Sharma G, Berger E A, Farber J M. STRL33, a novel chemokine receptor-like protein, functions as a fusion cofactor for both macrophage-tropic and T cell line-tropic HIV-1. J Exp Med. 1997;185:2015–2023. doi: 10.1084/jem.185.11.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Liu R, Paxton W A, Choe S, Ceradini D, Martin S R, Horuk R, MacDonald M E, Stuhlmann H, Koup R A, Landau N R. Homozygous defect in HIV-1 coreceptor accounts for resistance of some multiply-exposed individuals to HIV-1 infection. Cell. 1996;86:367–377. doi: 10.1016/s0092-8674(00)80110-5. [DOI] [PubMed] [Google Scholar]
- 62.Liu S L, Schacker T, Musey L, Shriner D, McElrath M J, Corey L, Mullins J I. Divergent patterns of progression to AIDS after infection from the same source: human immunodeficiency virus type 1 evolution and antiviral responses. J Virol. 1997;71:4284–4295. doi: 10.1128/jvi.71.6.4284-4295.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Maddon P J, Dalgleish A G, McDougal J S, Clapham P R, Weiss R A, Axel R. The T4 gene encodes the AIDS virus receptor and is expressed in the immune system and the brain. Cell. 1986;47:333–348. doi: 10.1016/0092-8674(86)90590-8. [DOI] [PubMed] [Google Scholar]
- 64.Mariani R, Kirchhoff F, Greenough T C, Sullivan J L, Desrosiers R C, Skowronski J. High frequency of defective nef alleles in a long-term survivor with nonprogressive human immunodeficiency virus type 1 infection. J Virol. 1996;70:7752–7764. doi: 10.1128/jvi.70.11.7752-7764.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Mellors J W, Kingsley L A, Rinaldo C R J, Todd J A, Hoo B S, Kokka R P, Gupta P. Quantitation of HIV-1 RNA in plasma predicts outcome after seroconversion. Ann Intern Med. 1995;122:573–579. doi: 10.7326/0003-4819-122-8-199504150-00003. [DOI] [PubMed] [Google Scholar]
- 66.Mellors J W, Munoz A, Giorgi J V, Margolick J B, Tassoni C J, Gupta P, Kingsley L A, Todd J A, Saah A J, Detels R, Phair J P, Rinaldo C R. Plasma viral load and CD4+ lymphocytes as prognostic markers of HIV-1 infection. Ann Intern Med. 1997;126:946–954. doi: 10.7326/0003-4819-126-12-199706150-00003. [DOI] [PubMed] [Google Scholar]
- 67.Michael N L, Chang G, d’Arcy L A, Ehrenberg P K, Mariani R, Busch M P, Birx D L, Schwartz D H. Defective accessory genes in a human immunodeficiency virus type 1-infected long-term survivor lacking recoverable virus. J Virol. 1995;69:4228–4236. doi: 10.1128/jvi.69.7.4228-4236.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Michael N L, Chang G, d’Arcy L A, Tseng C J, Birx D L, Sheppard H W. Functional characterization of human immunodeficiency virus type 1 nef genes in patients with divergent rates of disease progression. J Virol. 1995;69:6758–6769. doi: 10.1128/jvi.69.11.6758-6769.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Michael N L, Chang G, Louie L G, Mascola J R, Dondero D, Birx D L, Sheppard H W. The role of viral phenotype and CCR-5 gene defects in HIV-1 transmission and disease progression. Nat Med. 1997;3:338–340. doi: 10.1038/nm0397-338. [DOI] [PubMed] [Google Scholar]
- 70.Moore J P, Cao Y, Ho D D, Koup R A. Development of the anti-gp120 antibody response during seroconversion to human immunodeficiency virus type 1. J Virol. 1994;68:5142–5155. doi: 10.1128/jvi.68.8.5142-5155.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Munoz A, Kirby A J, He Y D, Margolick J B, Visscher B R, Rinaldo C R, Kaslow R A, Phair J P. Long-term survivors with HIV-1 infection: incubation period and longitudinal patterns of CD4+ lymphocytes. J Acquired Immune Defic Syndr Hum Retrovirol. 1995;8:496–505. doi: 10.1097/00042560-199504120-00010. [DOI] [PubMed] [Google Scholar]
- 72.Nielsen C, Pedersen C, Lundgren J D, Gerstoft J. Biological properties of HIV isolates in primary HIV infection. AIDS. 1993;7:1035–1040. doi: 10.1097/00002030-199308000-00002. [DOI] [PubMed] [Google Scholar]
- 73.Nowak M A, Bangham C R. Population dynamics of immune responses to persistent viruses. Science. 1996;272:74–79. doi: 10.1126/science.272.5258.74. [DOI] [PubMed] [Google Scholar]
- 74.O’Brien W A, Hartigan P M, Martin D, Esinhart J, Hill A, Benoit S, Rubin M, Simberkoff M S, Hamilton J D. Changes in plasma HIV-1 RNA and CD4+ lymphocyte counts and the risk of progression to AIDS. N Engl J Med. 1996;334:426–431. doi: 10.1056/NEJM199602153340703. [DOI] [PubMed] [Google Scholar]
- 75.O’Brien W A, Koyanagi Y, Namazie A, Zhao J-Q, Diagne A, Idler K, Zack J A, Chen I S Y. HIV-1 tropism for mononuclear phagocytes can be determined by regions of gp120 outside the CD4-binding domain. Nature. 1990;348:69–73. doi: 10.1038/348069a0. [DOI] [PubMed] [Google Scholar]
- 76.O’Brien W A, Mao S-H, Cao Y, Moore J P. Macrophage-tropic and T-cell line-adapted chimeric strains of human immunodeficiency virus type 1 differ in their susceptibilities to neutralization by soluble CD4 at different temperatures. J Virol. 1994;68:5264–5269. doi: 10.1128/jvi.68.8.5264-5269.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Ojo-Amaize E A, Nishanian P, Keith D E, Jr, Houghton R L, Heitjan D F, Fahey J L, Giorgi J V. Antibodies to human immunodeficiency virus in human sera induce cell-mediated lysis of human immunodeficiency virus-infected cells. J Immunol. 1987;139:2458–2463. [PubMed] [Google Scholar]
- 78.Pantaleo G, Menzo S, Vaccarezza M, Graziosi C, Cohen O J, Demarest J F, Montefiori D, Orenstein J M, Fox C, Schrager L K, Margolick J B, Buchbinder S, Giorgi J V, Fauci A S. Studies in subjects with long-term nonprogressive human immunodeficiency virus infection. New Engl J Med. 1995;332:209–216. doi: 10.1056/NEJM199501263320402. [DOI] [PubMed] [Google Scholar]
- 79.Piatak M, Saag M S, Yang L C, Clark S J, Kappes J C, Luk K-C, Hahn B H, Shaw G M, Lifson J D. High levels of HIV-1 in plasma during all stages of infection determined by competitive PCR. Science. 1993;259:1749–1754. doi: 10.1126/science.8096089. [DOI] [PubMed] [Google Scholar]
- 80.Planelles V, Haislip A, Withers-Ward E S, Stewart S A, Xie Y, Shah N P, Chen I S Y. A new reporter system for detection of retroviral infection. Gene Therapy. 1995;2:1–6. [PubMed] [Google Scholar]
- 81.Richman D D, Bozzett S A. The impact of syncytium-inducing phenotype of human immunodeficiency virus on disease progression. J Infect Dis. 1993;169:968–974. doi: 10.1093/infdis/169.5.968. [DOI] [PubMed] [Google Scholar]
- 82.Saag M S, Crain M J, Decker W D, Campbell-Hill S, Robinson S, Brown W E, Leuther M, Whitley R J, Hahn B H, Shaw G M. High-level viremia in adults and children infected with human immunodeficiency virus: relation to disease stage and CD4+ lymphocyte levels. J Infect Dis. 1991;164:72–80. doi: 10.1093/infdis/164.1.72. [DOI] [PubMed] [Google Scholar]
- 83.Sambrook J, Fritsch E F, Maniatis T. Molecular cloning: a laboratory manual. 2nd ed. Cold Spring Harbor, N.Y: Cold Spring Harbor Laboratory; 1989. Calcium phosphate transfection of mammalian cells; pp. 16.30–16.40. [Google Scholar]
- 84.Samson M, Libert F, Doranz B J, Rucker J, Liesnard C, Farber C M, Saragosti S, Lapoumeroulie C, Cognaux J, Forceille C, Muyldermans G, Verhofstede C, Burtonboy G, Georges M, Imai T, Rana S, Yi Y, Smyth R J, Collman R G, Doms R W, Vassart G, Parmentier M. Resistance to HIV-1 infection in caucasian individuals bearing mutant alleles of the CCR-5 chemokine receptor gene. Nature. 1996;382:722–725. doi: 10.1038/382722a0. [DOI] [PubMed] [Google Scholar]
- 85.Schnittman S M, Greenhouse J J, Psallidopoulos M C, Baseler M, Salzman N P, Fauci A S, Lane H C. Increasing viral burden in CD4+ T cells from patients with human immunodeficiency virus (HIV) infection reflects rapidly progressive immunosuppression and clinical disease. Ann Intern Med. 1990;113:438–443. doi: 10.7326/0003-4819-113-6-438. [DOI] [PubMed] [Google Scholar]
- 86.Schrager L K, Young J, Fowler M G, Mathieson B, Vermund S H. Long-term survivors of HIV infection: definitions and research challenges. AIDS. 1994;8:S95–S108. [Google Scholar]
- 87.Schuitemaker H, Koot M, Kootstra N A, Dercksen M W, DeGoede R E Y, Van Steenwijk R P, Lange J M A, Eeftink S J K M, Miedema F, Tersmette M. Biological phenotype of human immunodeficiency virus type 1 clones at different stages of infection: progression of disease is associated with a shift from monocytotropic to T-cell-tropic virus populations. J Virol. 1992;66:1354–1360. doi: 10.1128/jvi.66.3.1354-1360.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Schuitemaker H, Kootstra N A, de Goede R, de Wolf F, Miedema F, Tersmette M. Monocytotropic human immunodeficiency virus type 1 (HIV-1) variants detectable in all stages of HIV-1 infection lack T-cell line tropism and syncytium-inducing ability in primary T-cell culture. J Virol. 1991;65:356–363. doi: 10.1128/jvi.65.1.356-363.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Sheppard H W, Lang W, Ascher M S, Vittinghoff E, Winkelstein W. The characterization of non-progressors: long-term HIV-1 infection with stable CD4+ T-cell levels. AIDS. 1993;7:1159–1166. [PubMed] [Google Scholar]
- 90.Smith M W, Dean M, Carrington M, Winkler C, Huttley G A, Lomb D A, Goedert J J, O’Brien T R, Jacobson L P, Kaslow R, Buchbinder S, Vittinghoff E, Vlahov D, Hoots K, Hilgartner M W, O’Brien S J. Contrasting genetic influence of CCR2 and CCR5 variants on HIV-1 infection and disease progression. Hemophilia Growth and Development Study (HGDS), Multicenter AIDS Cohort Study (MACS), Multicenter Hemophilia Cohort Study (MHCS), San Francisco City Cohort (SFCC), ALIVE Study. Science. 1997;277:959–965. doi: 10.1126/science.277.5328.959. [DOI] [PubMed] [Google Scholar]
- 91.Strebel K, Daugherty D, Clouse K, Cohen D, Folks T, Martin M A. The HIV ’A’ (sor) gene product is essential for virus infectivity. Nature. 1987;328:728–731. doi: 10.1038/328728a0. [DOI] [PubMed] [Google Scholar]
- 92.Tersmette M, de Goede R E Y, Al B J M, Winkel I N, Gruters R A, Cuypers H T, Huisman H G, Miedema F. Differential syncytium-inducing capacity of human immunodeficiency virus isolates: frequent detection of syncytium-inducing isolates in patients with acquired immunodeficiency syndrome (AIDS) and AIDS-related complex. J Virol. 1988;62:2026–2032. doi: 10.1128/jvi.62.6.2026-2032.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Winkler C, Modi W, Smith M W, Nelson G W, Wu X, Carrington M, Dean M, Honjo T, Tashiro K, Yabe D, Buchbinder S, Vittinghoff E, Goedert J J, O’Brien T R, Jacobson L P, Detels R, Donfield S, Willoughby A, Gomperts E, Vlahov D, Phair J, O’Brien S J. Genetic restriction of AIDS pathogenesis by an SDF-1 chemokine gene variant. Science. 1998;279:389–393. doi: 10.1126/science.279.5349.389. [DOI] [PubMed] [Google Scholar]
- 94.Wu L, Paxton W A, Kassam N, Ruffing N, Rottman J B, Sullivan N, Choe H, Sodroski J, Newman W, Koup R A, et al. CCR5 levels and expression pattern correlate with infectability by macrophage-tropic HIV-1, in vitro. J Exp Med. 1997;185:1681–1691. doi: 10.1084/jem.185.9.1681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Zhu T, Mo H, Wang N, Nam D, Cao Y, Koup R, Ho D. Genotypic and phenotypic characterization of HIV-1 in patients with primary infection. Science. 1993;261:1179–1181. doi: 10.1126/science.8356453. [DOI] [PubMed] [Google Scholar]
- 96.Zinkernagel R M. Are HIV-specific CTL responses salutary or pathogenic? Curr Opin Immunol. 1995;7:462–470. doi: 10.1016/0952-7915(95)80089-1. [DOI] [PubMed] [Google Scholar]