Abstract
Engineered cytokine products represent promising agents for the treatment of immunogenic tumors, such as malignant melanoma, in addition to immune checkpoint inhibitors. Here we describe the results of a controlled, randomized phase II clinical trial, aimed at assessing the therapeutic potential of L19IL2, a fully human fusion protein consisting of the L19 antibody specific to the alternatively spliced extra-domain B of fibronectin, fused to human interleukin-2 in advanced metastatic melanoma. In one arm, patients received dacarbazine (DTIC; 1000 mg/m2 of body surface on day 1 of 21-day cycles) as single agent, while in two other arms L19IL2 (22.5 million international units of IL2 equivalents) was added, based on two different schedules of administration. In total, 69 patients with stage IV melanoma were enrolled (24 in the dacarbazine arm, 23 and 22 in the other combination arms, respectively) and 67 received treatment. Analyses of efficacy results show a statistically significant benefit in terms of overall response rate and median progression-free survival for patients receiving L19IL2 in combination with DTIC, compared to DTIC as single agent. In light of these results, further clinical investigations with L19IL2 (alone or in combination with other agents) are warranted.
Electronic supplementary material
The online version of this article (10.1007/s00262-019-02383-z) contains supplementary material, which is available to authorized users.
Keywords: Stage IV melanoma, Immunocytokine, L19IL2, Dacarbazine, Phase II study
Introduction
For over 30 years, therapeutic options for metastatic melanoma have been mostly limited to the chemotherapeutic agent dacarbazine (DTIC), which has been considered standard of care despite the absence of clear evidence of a survival benefit [1, 2]. The introduction of immune checkpoint inhibitors [3–6] and of BRAF/MEK kinase inhibitors [7–9] has revolutionized the management of stage IV melanoma, offering more options to patients and, in many cases, a substantial survival benefit. High-dose interleukin-2 (IL2) has also played a role, having demonstrated the ability to induce long-term remission of metastatic melanoma in a small proportion of patients [10]. This result was achieved, however, at the price of severe side-effects requiring administration in a dedicated setting [10, 11].
There is a growing interest in the clinical use of engineered IL2 derivatives, which may display an increased therapeutic index. For example, a pegylated version of IL2 (NKTR-214) has shown promising initial clinical data in combination with immune checkpoint inhibitors, leading to what has been dubbed as “the largest licensing fee in the history of biotechnology” [12].
A different strategy for the enhancement of the therapeutic index of IL2 consists in its fusion to antibodies, capable of selective localization to the site of disease [13–18]. Based on encouraging preclinical results, a number of antibody-IL2 fusions have been moved to clinical trials, including Hu14.18-IL2 (anti-GD2 fused to IL2) [19, 20], DI-Leu16-IL2 (de-immunized anti-CD20 fused to IL2) [21], F16IL2 [22, 23] and L19IL2 [24–26].
L19IL2 is a recombinant fusion protein, consisting of the L19 antibody in diabody format fused to human IL2 [16]. L19 is specific to the alternatively spliced extra-domain B (EDB) domain of fibronectin, a marker of angiogenesis. The protein was shown to selectively localize to solid tumors in mouse models of cancer [16, 22, 27–33] and to substantially increase leukocyte density and activity at the site of disease [24, 26, 34].
L19IL2 has been administered to over 200 patients, showing excellent tolerability and clinical benefit in a substantial proportion of treated subjects. The immunocytokine has been studied as a monotherapy for patients with renal cell carcinoma in a phase I/II dose finding study (EudraCT no.: 2005-002716-16) [25], and in a phase I/II study in combination with gemcitabine in pancreas cancer patients (EudraCT no.: 2007-001609-81).
The systemic combination of L19IL2 with DTIC in patients with stage IV melanoma has been studied in a phase II clinical trial (EudraCT no.: 2007-005737-11), which was divided in different phases, as follows. An initial dose confirmation study of L19IL2 in combination with 1000 mg/m2 DTIC in melanoma patients (phase IIa) confirmed that the recommended dose (RD) of 22.5 Mio IU per administration was well tolerated when administered on days 1, 3 and 5 of every 21-day cycle. In the following phase IIb Step 1 part of the study, 22 additional metastatic melanoma patients received L19IL2 at the RD (as determined in phase IIa) in combination with DTIC. A proportion of treated patients (eight out of 29, 28%) presented an objective response, including 1/29 patient with a confirmed complete response, still ongoing 21 months after treatment beginning. The results of these two parts of the study have been published already [26].
Subsequently, a controlled part of the study (phase IIb step 2) was carried out in 69 additional patients, which were randomized into three arms in order to compare the combination of L19IL2 and DTIC using two different administration schedules to DTIC monotherapy. The results of this controlled trial are discussed in this article. All data presented in this manuscript exclusively refer to the 69 patients treated in this controlled randomized part of the study except those presented in supplementary Fig. 1, which refer to the whole population of patients who were treated with L19IL2 at the dose of 22.5 Mio IU and DTIC at 1000 mg/m2 in the different parts of the study mentioned above.
Methods
Patient characteristics
Adult (≥ 18 years of age) patients with histologically or cytologically confirmed unresectable metastatic (stage IV) non-uveal melanoma represented the target population of the study. Eligible patients had to have measurable disease, defined as at least one lesion that could be accurately and serially measured as per RECIST criteria v.1.1. Cutaneous lesions measuring at least 1 cm would be considered measurable.
Patient population’s characteristics are summarized in Table 1.
Table 1.
Patients’ demography and characteristics
| Arm 1 | Arm 2 | Arm 3 | |
|---|---|---|---|
| Enrolled (n = 69) | 23 (33.3%) | 22 (31.9%) | 24 (34.8%) |
| Safety evaluable (n = 67) | 22 (32.8%) | 22 (32.8%) | 23 (34.4%) |
| Efficacy evaluable (n = 60) | 17 (28.3%) | 21 (35.0%) | 22 (36.7%) |
| Sexa | |||
| Female | 9 (40.9%) | 14 (63.6%) | 10 (43.5%) |
| Male | 13 (59.1%) | 8 (36.4%) | 13 (56.5%) |
| Patient with baseline LDH,U/la | |||
| 0 < LDH < 250 | 17 (77.3%) | 20 (90.9%) | 20 (87.0%) |
| LDH > 250 | 5 (22.7%) | 2 (9.1%) | 3 (13.0%) |
| ECOGa | |||
| 0 | 19 (86.4%) | 17 (77.3%) | 20 (87.0%) |
| 1 | 3 (13.6%) | 5 (22.7%) | 3 (13.0%) |
| Stage of metastatic disease at baselinea | |||
| IV M1a | 5 (22.7%) | 5 (22.7%) | 6 (26.1%) |
| IV M1b | 9 (40.9%) | 11 (50.0%) | 5 (21.7%) |
| IV M1c | 8 (36.4%) | 6 (27.3%) | 12 (52.2%) |
| Prior systemic antineoplastica | |||
| Chemotherapy | 9 (40.9%) | 11 (50.0%) | 11 (47.8%) |
| Radiotherapy | 4 (18.2%) | 2 (9.1%) | 4 (17.4%) |
aPercent based on safety evaluable population
Study design and treatment
The design of the phase IIa (dose definition) and of the phase IIb step 1 (activity evaluation) of the study have been described elsewhere [26].
The controlled phase II study (phase IIb step 2) consisted of a prospective, open-label, multicenter, randomized study comparing three arms.
In the first of the treatment arms (Arm 1) patients received L19IL2 on day 1, 3 and 5 during four 21-day cycles, in combination with DTIC administered on day 1 of each cycle (induction phase A), followed by induction phase B (cycles 5–8) in which L19IL2 was administered on day 1, 8 and 15, in combination with DTIC administered on day 1 of each cycle. These induction phases were followed by a maintenance phase lasting for a maximum of 1 year after start of treatment, with L19IL2 being administered on day 1, 8 and 15, in combination with DTIC administered on day 1 of each cycle.
In the second treatment arm (Arm 2) L19IL2 was administered to patients on day 1, 8 and 15 of each 21-day cycles, for a maximum of 1 year after start of treatment, in combination with DTIC administered on day 1 of each cycle.
Finally, in the control arm (Arm 3) DTIC monotherapy was administered on day 1 of each 21-day cycles, for a maximum of eight cycles (standard of care).
Primary objective of the study was overall response rate (ORR) after 8 cycles of treatment (week 24), defined as the proportion of patients with either complete response (CR) or partial response (PR). The same efficacy for the two arms with different schedule was hypothesized and the hypotheses to test were: “True probability of response did not exceed p0” (Null hypothesis; H0: p ≤ p0) or “True probability of response exceeded p0” (Alternative hypothesis; H1: p > p0) with p: probability of response under L19IL2 + DTIC combination therapy (the hypothesized p was 0.25) and p0: probability of response in the monotherapy arm with DTIC only (p0 set at 0.05).
The null hypothesis was tested with a one-sided type-one error probability of α = 5% at a power of 80%.
The estimated sample size 30 + 30 + 30 for the three arms was also sufficient to detect a 1.3 months improvement in PFS (assuming the median PFS in 1.7 months for DTIC alone group and 3 months for the L19IL2 + DTIC groups) with α = 5% and a power of 80%.
The assessment of response was performed using RECIST v1.1.
Secondary objectives included PFS, OS and ORR defined as the proportion of patients with either CR or PR as best overall response.
Safety and tolerability assessment was performed by AEs, serious adverse events (SAEs) and laboratory toxicities graded according to the Common Terminology Criteria for Adverse Events (CTCAE), version 3.0. Other safety variables were: physical examination, laboratory parameters, vital signs, physical examination, Eastern Cooperative Oncology Group (ECOG) performance status, 12-lead electrocardiography and multigated radionuclide angiography (MUGA).
Inclusion and exclusion criteria
A maximum of one prior therapy line for metastatic melanoma and involvement of up to three organs was allowed. Patients with primary ocular melanoma or evidence of brain metastases were excluded. Not eligible to the study were also patients with known history of allergy to IL2, DTIC, or other intravenously administered human proteins/peptides/antibodies or who had received anti-tumor therapy within 4 weeks prior to administration of study treatment.
Exposure to monoclonal antibodies for biological therapy in the 6 weeks before administration of study treatment, to DTIC treatment in the last 6 months prior to study entry or to growth factors or immunomodulatory agents within 7 days prior to administration of study treatment was not allowed. The complete list of inclusion/exclusion criteria follows.
Inclusion criteria
Adult patients with histologically or cytologically confirmed unresectable metastatic (stage IV) non-uveal melanoma were included if following features applied.
Age ≥ 18 years; measurable disease defined as at least one lesion that could be accurately and serially measured per RECIST, cutaneous lesions measuring at least 1 cm were considered measurable; prior chemotherapy including dacarbazine for metastatic melanoma was allowed if treatment had completed > 6 months prior to study entry; ≤ than three organs involved or cutaneous and/or subcutaneous metastases only; an ECOG performance status of ≤ 2; a life expectancy of at least 12 weeks; absolute neutrophil count > 1500/μL; platelets 100,000/μL; hemoglobin > 9.0 g/dL; total bilirubin < 2.0 mg/dL; alanine aminotransferase or aspartate aminotransferase ≤ 2.5 times the upper limit of normal (ULN), ≤ 5 × ULN for patients with hepatic involvement with tumor; normal LDH; serum creatine < 1.5 × ULN. All acute toxic effects (excluding alopecia) of any prior therapy were to be resolved to National Cancer Institute (NCI) CTCAE v3.0 Grade ≤ 1.
Female patients of childbearing potential had to be negative for the serum pregnancy test at screening visit; moreover, they had to agree to use adequate contraceptive methods beginning at the screening visit and continuing until 3 months following last treatment with study drug.
Exclusion criteria
Primary ocular melanoma; evidence of brain metastasis by computed tomography (CT) scan in the 2 months prior to study entry; previous or concurrent cancer that was distinct in primary site or histology from the cancer being evaluated in the study; history of HIV infection or chronic hepatitis B or C; presence of active infections (e.g., requiring antimicrobial therapy) or other severe concurrent disease; recent history of acute or sub-acute coronary syndromes; heart insufficiency; uncontrolled hypertension; ischemic peripheral vascular disease; severe diabetic retinopathy; active autoimmune disease; history of organ allograft or stem cell transplantation; recovery from major trauma; surgery and anti-tumor therapy within 4 weeks prior to study treatment; known history of allergy to IL2 and DTIC; breast-feeding female; previous in vivo exposure to monoclonal antibodies for biological therapy within 6 weeks prior to study treatment; previous DTIC treatment within 6 months prior to study entry; more than one prior line of treatment; growth factors or immunomodulatory agents within 7 days of the administration of study treatment; corticosteroids or other immunosuppressant drugs that had to be taken on a long-term basis.
Safety and efficacy assessment
After obtaining patients informed consent, the screening procedures, which have already been described in [26], were conducted within 14 days before initiating study drug treatment. Patients returned for the end of treatment visit 30–37 days after the last dose of study drug, unless the objective tumor response was assessed as “progressive disease” in a previous treatment visit or the patient at some point discontinued the treatment. Adverse events and toxicities were graded as per CTCAE version 3.0.
Disease status was assessed at baseline, after every two cycles (i.e., 6 weeks) and at study discontinuation using RECIST v.1.1 [35]. Chest, abdomen, pelvis, and brain scans, either CT (preferred) or MRI (at discretion of the investigator), were conducted at baseline. At the following tumor assessments, scans of chest, abdomen and pelvis were taken; brain scans were conducted only if patients were symptomatic.
Patients remained on study until the occurrence of unacceptable toxicity, disease progression, withdrawal of consent, or until a L19IL2 or DTIC infusion in the first two cycles was missed, except in the case of a missed dose because of toxicity. Best overall responses were defined as the largest shrinkage in the sum of diameters of target lesions at any moment of time, compared with baseline.
L19IL2 production, characterization and pharmacokinetics
Production, characterization, and toxicology testing of L19IL2 were described previously [25, 36]. Determination of L19IL2 concentration in samples of human serum from patients enrolled in the phase IIa of the study has already been described in [26].
Results
Study design
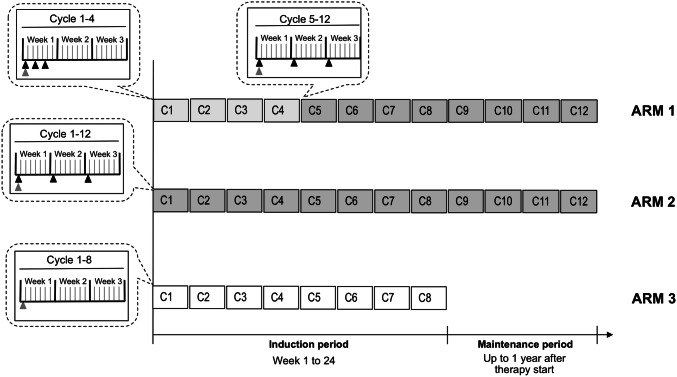
In this trial, patients were randomized to three different study arms, in order to compare the combination of L19IL2 and DTIC using two different administration schedules for L19IL2 to DTIC monotherapy (Fig. 1). The choice of testing two different administration schedules for L19IL2 aimed at comparing data collected during the previous parts of the study [26] (infusions on day 1, 3 and 5 of a 21-day cycle) to the favorable tolerability data recorded in trials run with the similar product F16IL2 in combination with chemotherapeutics [23] and based on weekly administration of the immunocytokine. Moreover, while still providing the patient with the same cumulative amount of drug over the 21-day cycle, the once-weekly schedule presents the advantage that patients do not need to be hospitalized over an entire week.
Fig. 1.
Administration Schedule of L19IL2 and DTIC in the three arms of the study. Schematic representation of the administration schedule of L19IL2 and DTIC in the three arms of the randomized Phase II study. A detailed view of one single cycle is shown in the callouts on the left hand side and on top. Black triangle L19IL2; grey triangle DTIC. Arm 1 used the previously tested schedule (L19IL2 administrations on days 1, 3 and 5 of each 21-day cycle; light grey squares) during the first four cycles, followed by weekly administrations of L19IL2 (dark grey squares). Patients in Arm 2 received directly weekly administration of the immunocytokine (dark squares). DTIC was administered once on day 1 of each cycle (all Arms). In Arm 3 (empty squares), although the protocol foresaw a maximum of eight cycles of DTIC treatment, responders would be allowed to continue with the same treatment until progression
Patient population
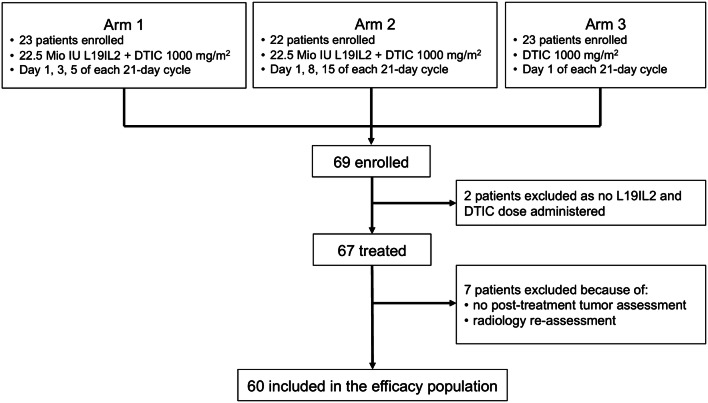
69 patients in total were recruited between October 2010 and March 2012 and randomized to the three study arms (23, 22 and 24 to Arms 1–3, respectively). The safety-evaluable population consisted of 22 patients in Arms 1 and 2 and of 23 patients in Arm 3. Evaluable for efficacy were 17 patients in Arm 1, 21 patients in Arm 2 and 22 patients in Arm 3, respectively (Fig. 2).
Fig. 2.
Flowchart of trial profile and patient allocation to each treatment arm
The median age at the start of the treatment was of 59 ± 12.8 years (range 22–82), patients were fairly equally distributed between sexes, with 33 males (49.2%) and 34 females (50.8%) in the safety-evaluable population. The demography and characteristics of patients enrolled in the study, divided by arm are summarized in Table 1.
Safety data
Analyzing the cumulative data for patients included in the two treatment arms, L19IL2 given in combination with DTIC revealed an acceptable safety profile. The rate and type of AEs was in line with the known toxicity profile of L19IL2 and DTIC [37–39]. Drug-related adverse events by grade, recorded in the three arms of the study are shown in Table 2. Most frequent adverse reactions were classified as grade 1, with fewer grade 2 and grade 3 toxicities. These last only included hypotension and arthralgia (1 event each in both Arms 1 and 2) and abnormalities of laboratory parameters [2 events each of alanine amino transferase (ALT) and gamma glutamyl transpeptidase (GGT) elevation in Arms 1 and 4 events of thrombocytopenia in Arm 2]. No grade 3 events were recorded in Arm 3 (DTIC alone).
Table 2.
Drug-related adverse events according to CTCAE v.3.0
| CTCAE grade | L19IL2/DTIC Arm 1 (n = 22) | L19IL2/DTIC Arm 2 (n = 22) | DTIC Arm 3 (n = 23) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| G1 | G2 | G3 | G4 | G1 | G2 | G3 | G4 | G1 | G2 | G3 | G4 | |
| ALT increaseda | 3 | 3 | 2 | 0 | 0 | 1 | 0 | 0 | 0 | 1 | 0 | 0 |
| Anorexiaa | 7 | 0 | 0 | 0 | 3 | 1 | 0 | 0 | 2 | 0 | 0 | 0 |
| Arthralgia | 4 | 2 | 1 | 0 | 5 | 5 | 2 | 0 | 0 | 0 | 0 | 0 |
| AST increaseda | 1 | 4 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 |
| Chills | 7 | 1 | 0 | 0 | 3 | 2 | 0 | 0 | 0 | 0 | 0 | 0 |
| Diarrhea | 5 | 1 | 0 | 0 | 4 | 0 | 0 | 0 | 0 | 1 | 0 | 0 |
| Fatigue | 6 | 0 | 0 | 0 | 6 | 1 | 0 | 0 | 0 | 0 | 0 | 0 |
| GGT increaseda | 0 | 1 | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 |
| Nausea | 11 | 2 | 0 | 0 | 8 | 6 | 0 | 0 | 4 | 1 | 0 | 0 |
| Neutropenia | 0 | 0 | 0 | 2 | 0 | 0 | 1 | 2 | 0 | 0 | 0 | 1 |
| Edema peripheralb | 3 | 0 | 0 | 0 | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Pyrexia | 14 | 3 | 0 | 0 | 12 | 2 | 0 | 0 | 0 | 0 | 0 | 0 |
| Rashb | 2 | 0 | 0 | 0 | 4 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Thrombocytopeniaa | 1 | 1 | 0 | 0 | 1 | 1 | 3 | 1 | 0 | 0 | 1 | 0 |
| Vomiting | 6 | 0 | 0 | 0 | 5 | 2 | 0 | 0 | 2 | 0 | 0 | 0 |
aIncidence > 15% in at least one arm of the randomized part of the study
bIncidence was also > 15% in the previously published phase IIa + IIb step 1 parts of the study [26]
Safety and tolerability data analysis essentially confirmed that the combination of L19IL2/DTIC was generally well tolerated in both treatment groups, irrespectively of the administration schedule. The most common treatment-related AEs by preferred term (any grade) were nausea, pyrexia, and arthralgia, and were reported in higher proportion of patients in Arm 1 and in Arm 2 than in Arm 3. Moreover, pyrexia, anorexia, chills and ALT increases were more common in Arm 1 than in Arm 2, while arthralgia and thrombocytopenia were more common in Arm 2 than in Arm 1. Regarding deaths, fatal AEs occurred in two patients (aged 76 and 82, respectively), all in Arm 1 (13.6%). The first events consisted in one case of acute myocardial infarction, which occurred between week 1 and 2 of the first treatment cycle and in a patient with pre-existing risk factors for coronary disease including hypertension, hypercholesterolemia and type 2 diabetes. The second event consisted in a case of stroke, which occurred at the end of week 3 (cycle 2) and was followed by death due sepsis with infection of unknown origin. Both events were considered unexpected and possibly related to L19IL2 and not to DTIC (myocardial infarction) or to both L19IL2 and DTIC (stroke).
Treatment-related SAEs were reported in four patients (18.2%) in Arm 1 (including the two death cases above), in 5 (22.7%) in Arm 2 and in none (0.0%) in Arm 3. Six patients (27.3%) in Arms 1, 3 (13.6%) in Arm 2 and none (0.0%) in Arm 3, discontinued the study due to AEs. Therefore, the results of treatment-related AEs suggest that the addition of L19IL2 to DTIC in the two different regimens may be associated with an increased risk of AE, compared to DTIC monotherapy, in line with the known safety profile of the two investigational products.
Efficacy data
For the primary endpoint analysis, objective responses were recorded in a number of patients of the efficacy-evaluable population.
One patient in Arm 1 (5.9%) and 1 in Arm 2 (4.8%) had CR as best tumor response. One patient (5.9%) in Arm 1, 4 (19.0%) in Arm 2 and 1 (4.5%) in Arm 3 had PR as best tumor response. The number of patients with stable disease (SD) was 4 (23.5%), 3 (14.3%) and 4 (18.2%), in the three arms, respectively. Finally, progressive disease (PD) was recorded in 11 (64.7%), 13 (61.9%) and 17 (77.3%) patients, respectively, in the three groups.
An ORR (CR + PR) of 18.4% (7/38 patients) was cumulatively observed for patients receiving the L19IL2/DTIC combination treatment (2/17 in Arm 1 and 5/21 in Arm 2, respectively). In contrast, in the control arm (DTIC monotherapy) only one partial response (1/22; 4.5%) could be recorded.
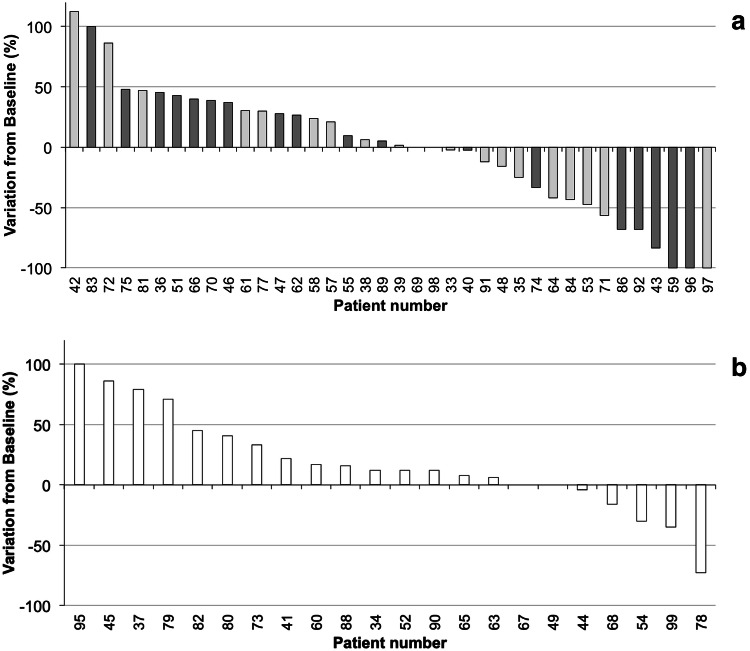
A waterfall plot of the best overall response recorded for all patients enrolled in Arms 1 and 2 of the controlled phase II is shown in Fig. 3 (panel A), whereas panel B shows the best overall response for patients enrolled in Arm 3 (DTIC alone).
Fig. 3.
Waterfall plot of best objective response for target lesions. Waterfall plot of the best response recorded for each patient evaluable for efficacy in the randomized phase II study. a Waterfall plot of the best response for patients who received L19IL2/DTIC; light gray bars = Arm 1 patients, dark gray bars = Arm 2 patients. b Waterfall plot of the best response for patients who received DTIC alone (Arm 3)
The ORR comparison between Arms 1 + 2 vs. Arm 3 was statistically significant at both the first (week 6; p = 0.028) and second tumor assessment (week 12; p = 0.015).
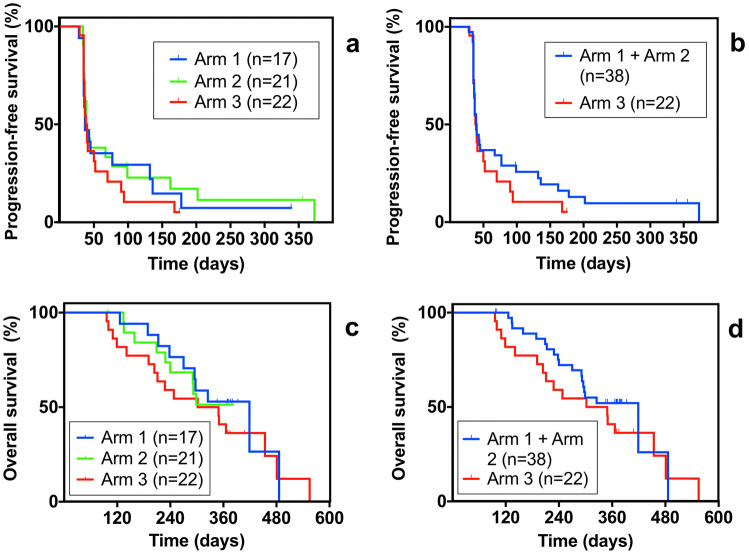
With regards to progression-free survival, a trend towards a small advantage in PFS was recorded for patients who received the weekly administration of L19IL2 in the L19IL2/DTIC treatment Arm 2 (mean 99.0 ± 25.1 days) as compared to the patients who received L19IL2 three times a week in the L19IL2/DTIC treatment Arm 1 (mean 74.0 ± 13.9 days), or to patients treated with DTIC monotherapy in Arm 3 (mean 59.1 ± 9.3 days). Figure 4, panel A shows the results described above (Arm 1, blue line; Arm 2, green line; Arm 3, red line). The analysis of PFS results for all patients who received the L19IL2/DTIC combination (irrespectively of the administration schedule; blue line) vs patients who received DTIC alone (red line) is shown in Fig. 4, panel B. Median PFS was not statistically different (41 vs 39 days, respectively; p value = 0.312; HR = 0.76) between the L19IL2/DTIC group (mean 87.8 ± 14.9 days) and the control group (mean 56.8 ± 8.7 days).,
Fig. 4.
Kaplan–Meier curves for the progression-free survival and overall survival. a Kaplan–Meier curves showing progression-free survival (PFS) recorded in all efficacy-evaluable patients treated in the randomized phase II study (Arm 1 = blue line; Arm 2 = green line; Arm 3 = red line). b Kaplan–Meier curves of PFS for the 38 patients treated with L19IL2/DTIC (Arm 1 + Arm 2; blue line) and for patients treated with DTIC alone (Arm 3; red line). c Kaplan–Meier curves showing overall survival (OS) for patients of Arm 1 (blue line), Arm 2 (green line) and Arm 3 (red line), recorded in the randomized phase II study. d Kaplan–Meier curves showing OS recorded in all 38 efficacy-evaluable patients treated with L19IL2/DTIC in the phase II study (Arm 1 + Arm 2; blue line) and in the 22 efficacy-evaluable patients treated with DTIC alone (Arm 3; red line)
Durable partial or complete responses (lasting for more than 1 year) were recorded in three patients. A selection of CT images from two patients treated in the randomized study, showing long-lasting reduction in size of target lesions is illustrated in Fig. 5.
Fig. 5.
Examples of responses in patients of the controlled phase II. a CT images of four different pulmonary coin lesions (I–IV) of patient No. 43. The patient was randomized to Arm 2 of the study and presented at baseline with four pulmonary coin lesions (RECIST score 35.3). Already at week 6, the patient enjoyed a partial response (decrease of 41% in RECIST score), which improved over time reaching an 82% reduction of RECIST score (5.8) on week 24. This partial response was still continuing when the patient was dismissed at the end of study (week 52; not shown). Lesions are indicated by green arrows. b CT images of a paratracheal lymph node of patient No. 97 (best response 100%). The patient was randomized to Arm 1 of the study and presented at baseline with a paratracheal and a pre-carinal (not shown) lymph node metastasis (RECIST score 35.0). Also in this case, already at week 6 both lymph nodes returned to a size of < 1.0 cm (complete response) and remained of normal size until the patient was withdrawn from the study on investigator’s decision on week 36. The complete response, however, was still ongoing at week 52 from the beginning of treatment (not shown). A green arrow indicates the paratracheal lesion
In terms of overall survival, a median OS of 419 days was recorded for all patients who received L19IL2/DTIC (Arms 1 and 2), as compared to the 325.5 days of median OS recorded for patients who received DTIC monotherapy. This difference appears to be statistically significant, but with a borderline p value (p value = 0.048; hazard ratio HR = 0.56). Results about OS of patients in the three arms of the study are shown in Fig. 4c, d.
Discussion
We have reported the results of a phase II clinical trial in patients with stage IV melanoma, who received DTIC alone or in combination with L19IL2. The ability of the L19 antibody to preferentially localize at the tumor site (including melanoma lesions) has been extensively validated using nuclear medicine procedures with radiolabeled antibody preparations [40, 41].
In general, the tolerability profile of L19IL2 plus DTIC was acceptable. The main toxicities included hypotension, flu-like symptoms, nausea and arthralgia. Most of these side-effects were associated with peak concentrations of the immunocytokine product. In a different clinical trial, we have recently demonstrated that a prolongation of the infusion time (from 1 to 3 h) allowed a threefold increase of the maximal tolerated dose (NCT02076646; unpublished results). In preclinical studies, the anticancer activity of antibody fusions with pro-inflammatory cytokines is clearly dose-dependent [42] and it is possible that even better clinical results may be achieved in the future, using slow administration procedures.
Analysis of the primary efficacy objective ORR revealed a statistically significant superiority of the combination L19IL2/DTIC compared to DTIC alone. A similar trend is also shown by analysis of the secondary objectives PFS and OS. These results confirm those recorded in the previous parts of the study already being published [26].
Indeed, extending the analysis to the whole population of patients, who received L19IL2 at the dose of 22.5 Mio IU and DTIC at 1000 mg/m2, independently of the administration schedule and including those of the dose-escalation and activity evaluation parts of the study (see supplementary Fig. 1), the proportion of patients with partial or complete responses exceeded 34%. Moreover, although about two-thirds of patients did not achieve a reduction of tumor burden of at least 30%, they reported a reduction in symptoms from the combination treatment (data not shown). The ability of melanoma patients to respond to immunotherapy may be related, in part, to the frequent loss of HLA class I on the tumor cells, making them insensitive to the action of cytotoxic T cells [43]. However, in principle, the targeted delivery of IL2 should potentiate also NK cells, which are ideally suited to recognize and counteract MHC downregulation on tumor cells [16, 44, 45].
Four patients who received L19IL2 at the dose of 22.5 Mio IU and DTIC at 1000 mg/m2 in the different parts of the study mentioned above achieved durable complete responses. The observation of a tail in the overall survival curve, which plateaus for ~ 20% of patients over a > 4-year observation period, is reminiscent of similar curves, obtained with high-dose interleukin-2 (HD IL2) treatment. However, while HD IL2 features the administration of as much as 800 million IL2 units in 1 week, the treatment protocol with L19IL2 featured the administration of 67.5 million IL2 units over a period of 3 weeks, with cycles that could be repeated for up to 12 times. The presence of the L19 antibody allows specific accumulation at the site of disease of the payload IL2, which therefore reaches higher concentration in the cancer lesion as compared to other organs, with an overall reduction of the treatment toxicity. As a consequence, the tolerability profile of L19IL2 plus DTIC is clearly more favorable compared to HD IL2.
While the results observed with L19IL2 indicate a clear activity of the product in a proportion of patients, the response rate was lower compared to the one reported for immune check-point inhibitors in comparable patient populations [5, 6, 46]. It is possible that the use of L19IL2 at a higher dose may lead to a further improvement of response rates. A more interesting approach would feature the combination of L19IL2 with immune checkpoint inhibitors. Favorable clinical results have been reported for ipilimumab in combination with HD IL2 [47] and are being explored for anti-PD1 with HD IL2. In addition, an additive therapeutic effect has been observed in some mouse models of cancer for the combination of antibody-IL2 fusions with immune checkpoint inhibitors [18, 48–50].
In summary, L19IL2 may represent a new tool for the management of patients with metastatic disease, including stage IV melanoma. The target antigen (the EDB domain of fibronectin) is expressed in the majority of solid tumors and lymphomas [51–54], while being undetectable in most normal adult tissues. The combination of L19IL2 with immune checkpoint inhibitors or other immunostimulatory agents appears to be the logical next step for the development of this product.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
We thank all patients, families, staff, and investigators of the participating hospitals. We are very grateful to Dr. Jacopo Piazzi, Philogen S.p.A., for his support with data management and statistical analysis.
Abbreviations
- ALT
Alanine amino transferase
- CTCAE
Common Terminology Criteria for Adverse Events
- CR
Complete response
- CT
Computed tomography
- DTIC
Dacarbazine
- ECOG
Eastern Cooperative Oncology Group
- EDB
Extra-domain B
- GGT
Gamma glutamyl transpeptidase
- HD IL2
High-dose interleukin 2
- MUGA
Multigated radionuclide angiography
- NCI
National Cancer Institute
- ORR
Objective response rate
- PR
Partial response
- PD
Progressive disease
- RD
Recommended dose
- SAE
Serious adverse event
- SD
Stable disease
- ULN
Upper limit of normal
Author contributions
Study conception and design: BW, TE, CC, PAA, SC, JCB, AH, AR, RD, RD, UT, DN, CG. Patient recruitment and management: BW, TE, CC, PAA, SC, JCB, AH, AR, RD, RD, UT, CG. Data acquisition, management, analysis, and interpretation: BW, AH, GE, DN, CG. Manuscript preparation: BW, AH, GE, DN, CG.
Funding
This study was funded by Philogen S.p.A. The sponsor was involved in study design, data collection, analysis, and interpretation, writing of the article, and in the decision to submit the article for publication.
Compliance with ethical standards
Conflict of interest
Benjamin Weide has received speaker or advisory board honoraria from Amgen, CureVac, Philogen, Novartis as well as research funding from Bristol-Myers Squibb (BMS), Merck, Sharp & Dohme (MSD) and Philogen. Paolo Ascierto reports grants and personal fees from BMS, grants and personal fees from Roche-Genentech, personal fees from MSD, grants and personal fees from Array, personal fees from Novartis, personal fees from Merck Serono, personal fees from Pierre Fabre, personal fees from Incyte, personal fees from Genmab, personal fees from NewLink Genetics, personal fees from Medimmune, personal fees from AstraZeneca, personal fees from Syndax, personal fees from Sun Pharma, personal fees from Sanofi, personal fees from Idera, personal fees from Ultimovacs, personal fees from Sandoz, personal fees from Immunocore, personal fees from 4SC, outside the submitted work; Jürgen C. Becker has received speaker honoraria from Amgen, Merck Serono, and Pfizer, advisory board honoraria from Amgen, CureVac, eTheRNA, Lytix, Merck Serono, Novartis, Rigontec, and Takeda as well as research funding from Alcedis, Boehringer Ingelheim, BMS and Merck Serono; he also received travel support from 4SC and Incyte; Axel Hauschild received clinical trial support, speaker´s honoraria, or consultancy fees from the following companies: Amgen, BMS, Merck Serono, MSD, Novartis, Philogen, Pierre Fabre, Provectus, Regeneron, Roche, OncoSec, Sanofi-Genzyme, and Sun Pharma; Reinhard Dummer reports intermittent, project focused consulting and/or advisory relationships with Novartis, MSD, BMS, Roche, Amgen, Takeda, Pierre Fabre, Sun Pharma, Sanofi, Catalym, Second Genome outside the submitted work; Giuliano Elia is an employee of Philochem AG, a company of the Philogen group; Dario Neri is shareholder and Board Member of Philogen S.p.A.; Claus Garbe reports personal fees from Philogen, during the conduct of the study; personal fees from Amgen, personal fees from MSD, grants and personal fees from Novartis, personal fees from NeraCare, grants and personal fees from BMS, personal fees from Philogen, grants and personal fees from Roche, grants and personal fees from Sanofi, outside the submitted work. All other authors declare no conflict of interest.
Ethical approval and ethical standards
The protocol was approved by the regulatory authorities Paul-Ehrlich-Institut in Germany (authorization no. 593/01 of 01/07/2008), ISS in Italy (authorization of 21/02/2008), Swissmedic in Switzerland (authorization no. 2011DR2225 of 30/12/2011) and BASG in Austria (authorizations nos. 717349/0001 and 717350/0001 of 06/04/2011). The study was also approved by the ethics committee of the coordinating center for the 3 participating centers in Germany (Ethikkommission an der Medizinischen Fakultät der Eberhard-Karls-Universitäts und am Universitätsklinikum Tübingen, Tübingen, Germany—approval number: 33/2008AMG1 of 01/09/2010 for Tübingen; and of 25/01/2011 for Kiel and Berlin); by the central ethic committee at IEO in Milan, Italy (authorization no. R363-IEO S387/607 of 21/02/2008) and four local ethics committees for the other four participating centers in Italy (Ancona, approval number 210520 of 02/12/2010; Naples, approval number CEI/538/10 of 25/11/2010; Pisa, approval number 2497/2008 of 01/12/2010; and Siena, approval number 82/2009 of 21/10/2010); by the local ethics committee for the participating center in Switzerland (Kantonale Ethikkommission Zürich (KEK), Zürich, Switzerland—approval number: KEK-ZH-Nr. 2011-0362 of 15/11/2011); and by the local ethics committee for the participating center in Austria (Ethikkommission Medizinischer Universitäts Graz, Graz, Austria—approval number 23-176 ex 10/11 of 06/04/2011). The study was conducted in accordance with Good Clinical Practice guidelines (EudraCT No.: 2007-005737-11; ClinicalTrials.gov Identifier: NCT01055522). All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent on being treated as well as on the use of generated data for research purposes and publication was obtained from all individual participants included in the study.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Giuliano Elia, Phone: +41 43 544 8803, Email: giuliano.elia@philogen.com.
Dario Neri, Phone: +41 44 633 7401, Email: dario.neri@pharma.ethz.ch.
Claus Garbe, Phone: +49 7071 298 7110, Email: claus.garbe@med.uni-tuebingen.de.
References
- 1.Korn EL, Liu PY, Lee SJ, et al. Meta-analysis of phase II cooperative group trials in metastatic stage IV melanoma to determine progression-free and overall survival benchmarks for future phase II trials. J Clin Oncol. 2008;26(4):527–534. doi: 10.1200/JCO.2007.12.7837. [DOI] [PubMed] [Google Scholar]
- 2.Garbe C, Peris K, Hauschild A, et al. Diagnosis and treatment of melanoma: European consensus-based interdisciplinary guideline. Eur J Cancer. 2010;46(2):270–283. doi: 10.1016/j.ejca.2009.10.032. [DOI] [PubMed] [Google Scholar]
- 3.Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363(8):711–723. doi: 10.1056/NEJMoa1003466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Robert C, Ribas A, Wolchok JD, et al. Anti-programmed-death-receptor-1 treatment with pembrolizumab in ipilimumab-refractory advanced melanoma: a randomised dose-comparison cohort of a phase 1 trial. Lancet. 2014;384(9948):1109–1117. doi: 10.1016/S0140-6736(14)60958-2. [DOI] [PubMed] [Google Scholar]
- 5.Robert C, Thomas L, Bondarenko I, et al. Ipilimumab plus dacarbazine for previously untreated metastatic melanoma. N Engl J Med. 2011;364(26):2517–2526. doi: 10.1056/NEJMoa1104621. [DOI] [PubMed] [Google Scholar]
- 6.Schachter J, Ribas A, Long GV, et al. Pembrolizumab versus ipilimumab for advanced melanoma: final overall survival results of a multicentre, randomised, open-label phase 3 study (KEYNOTE-006) Lancet. 2017;390(10105):1853–1862. doi: 10.1016/S0140-6736(17)31601-X. [DOI] [PubMed] [Google Scholar]
- 7.Falchook GS, Lewis KD, Infante JR, et al. Activity of the oral MEK inhibitor trametinib in patients with advanced melanoma: a phase 1 dose-escalation trial. Lancet Oncol. 2012;13(8):782–789. doi: 10.1016/S1470-2045(12)70269-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Falchook GS, Long GV, Kurzrock R, et al. Dabrafenib in patients with melanoma, untreated brain metastases, and other solid tumours: a phase 1 dose-escalation trial. Lancet. 2012;379(9829):1893–1901. doi: 10.1016/S0140-6736(12)60398-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sosman JA, Kim KB, Schuchter L, et al. Survival in BRAF V600-mutant advanced melanoma treated with vemurafenib. N Engl J Med. 2012;366(8):707–714. doi: 10.1056/NEJMoa1112302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Smith FO, Downey SG, Klapper JA, et al. Treatment of metastatic melanoma using interleukin-2 alone or in conjunction with vaccines. Clin Cancer Res. 2008;14(17):5610–5618. doi: 10.1158/1078-0432.CCR-08-0116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Atkins MB. Treatment selection for patients with metastatic renal cell carcinoma: identification of features favoring upfront IL-2-based immunotherapy. Med Oncol. 2009;26(Suppl 1):18–22. doi: 10.1007/s12032-008-9148-x. [DOI] [PubMed] [Google Scholar]
- 12.Herper M (2018) Bristol-Myers to pay largest fee in biotech history for Nektar cancer drug. Forbes. Retrieved from https://www.forbes.com/sites/matthewherper/2018/02/14/bristol-myers-to-paylargest-licensing-fee-in-biotech-history-for-cancer-drug/#5e90fc0a6d29
- 13.Penichet ML, Dela Cruz JS, Shin SU, Morrison SL. A recombinant IgG3-(IL-2) fusion protein for the treatment of human HER2/neu expressing tumors. Hum Antibodies. 2001;10(1):43–49. doi: 10.3233/HAB-2000-10107. [DOI] [PubMed] [Google Scholar]
- 14.Yang RK, Kalogriopoulos NA, Rakhmilevich AL, et al. Intratumoral hu14.18-IL-2 (IC) induces local and systemic antitumor effects that involve both activated T and NK cells as well as enhanced IC retention. J Immunol. 2012;189(5):2656–2664. doi: 10.4049/jimmunol.1200934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Neri D, Sondel PM. Immunocytokines for cancer treatment: past, present and future. Curr Opin Immunol. 2016;40:96–102. doi: 10.1016/j.coi.2016.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Carnemolla B, Borsi L, Balza E, et al. Enhancement of the antitumor properties of interleukin-2 by its targeted delivery to the tumor blood vessel extracellular matrix. Blood. 2002;99(5):1659–1665. doi: 10.1182/blood.V99.5.1659. [DOI] [PubMed] [Google Scholar]
- 17.Schliemann C, Palumbo A, Zuberbuhler K, Villa A, Kaspar M, Trachsel E, Klapper W, Menssen HD, Neri D. Complete eradication of human B-cell lymphoma xenografts using rituximab in combination with the immunocytokine L19-IL2. Blood. 2009;113(10):2275–2283. doi: 10.1182/blood-2008-05-160747. [DOI] [PubMed] [Google Scholar]
- 18.Schwager K, Hemmerle T, Aebischer D, Neri D. The immunocytokine L19-IL2 eradicates cancer when used in combination with CTLA-4 blockade or with L19-TNF. J Invest Dermatol. 2013;133(3):751–758. doi: 10.1038/jid.2012.376. [DOI] [PubMed] [Google Scholar]
- 19.Ribas A, Kirkwood JM, Atkins MB, Whiteside TL, Gooding W, Kovar A, Gillies SD, Kashala O, Morse MA. Phase I/II open-label study of the biologic effects of the interleukin-2 immunocytokine EMD 273063 (hu14.18-IL2) in patients with metastatic malignant melanoma. J Transl Med. 2009;7:68. doi: 10.1186/1479-5876-7-68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Shusterman S, London WB, Gillies SD, et al. Antitumor activity of hu14.18-IL2 in patients with relapsed/refractory neuroblastoma: a Children’s Oncology Group (COG) phase II study. J Clin Oncol. 2010;28(33):4969–4975. doi: 10.1200/jco.2009.27.8861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lansigan F, Nakamura R, Quick D, Vlock D, Raubitschek AA, Gillies SD, Bachanova V. Phase I/II study of an anti-CD20-interleukin-2 immunocytokine DI-Leu16-IL2 in patients with relapsed b-cell lymphoma (NHL) J Clin Oncol. 2016;34(15_suppl):e19046. doi: 10.1200/jco.2016.34.15_suppl.e19046. [DOI] [Google Scholar]
- 22.Gutbrodt KL, Schliemann C, Giovannoni L, Frey K, Pabst T, Klapper W, Berdel WE, Neri D. Antibody-based delivery of interleukin-2 to neovasculature has potent activity against acute myeloid leukemia. Sci Transl Med. 2013;5(201):201ra118. doi: 10.1126/scitranslmed.3006221. [DOI] [PubMed] [Google Scholar]
- 23.Catania C, Maur M, Berardi R, et al. The tumor-targeting immunocytokine F16-IL2 in combination with doxorubicin: dose escalation in patients with advanced solid tumors and expansion into patients with metastatic breast cancer. Cell Adhes Migr. 2015;9(1–2):14–21. doi: 10.4161/19336918.2014.983785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Weide B, Eigentler TK, Pflugfelder A, et al. Intralesional treatment of stage III metastatic melanoma patients with L19-IL2 results in sustained clinical and systemic immunologic responses. Cancer Immunol Res. 2014;2(7):668–678. doi: 10.1158/2326-6066.CIR-13-0206. [DOI] [PubMed] [Google Scholar]
- 25.Johannsen M, Spitaleri G, Curigliano G, et al. The tumour-targeting human L19-IL2 immunocytokine: preclinical safety studies, phase I clinical trial in patients with solid tumours and expansion into patients with advanced renal cell carcinoma. Eur J Cancer. 2010;46(16):2926–2935. doi: 10.1016/j.ejca.2010.07.033. [DOI] [PubMed] [Google Scholar]
- 26.Eigentler TK, Weide B, de Braud F, et al. A dose-escalation and signal-generating study of the immunocytokine L19-IL2 in combination with dacarbazine for the therapy of patients with metastatic melanoma. Clin Cancer Res. 2011;17(24):7732–7742. doi: 10.1158/1078-0432.CCR-11-1203. [DOI] [PubMed] [Google Scholar]
- 27.Balza E, Carnemolla B, Mortara L, Castellani P, Soncini D, Accolla RS, Borsi L. Therapy-induced antitumor vaccination in neuroblastomas by the combined targeting of IL-2 and TNFalpha. Int J Cancer. 2010;127(1):101–110. doi: 10.1002/ijc.25018. [DOI] [PubMed] [Google Scholar]
- 28.Balza E, Mortara L, Sassi F, et al. Targeted delivery of tumor necrosis factor-alpha to tumor vessels induces a therapeutic T cell-mediated immune response that protects the host against syngeneic tumors of different histologic origin. Clin Cancer Res. 2006;12(8):2575–2582. doi: 10.1158/1078-0432.CCR-05-2448. [DOI] [PubMed] [Google Scholar]
- 29.Gutbrodt KL, Casi G, Neri D. Antibody-based delivery of IL2 and cytotoxics eradicates tumors in immunocompetent mice. Mol Cancer Ther. 2014;13(7):1772–1776. doi: 10.1158/1535-7163.MCT-14-0105. [DOI] [PubMed] [Google Scholar]
- 30.Marlind J, Kaspar M, Trachsel E, Sommavilla R, Hindle S, Bacci C, Giovannoni L, Neri D. Antibody-mediated delivery of interleukin-2 to the stroma of breast cancer strongly enhances the potency of chemotherapy. Clin Cancer Res. 2008;14(20):6515–6524. doi: 10.1158/1078-0432.CCR-07-5041. [DOI] [PubMed] [Google Scholar]
- 31.Mortara L, Balza E, Sassi F, et al. Therapy-induced antitumor vaccination by targeting tumor necrosis factor alpha to tumor vessels in combination with melphalan. Eur J Immunol. 2007;37(12):3381–3392. doi: 10.1002/eji.200737450. [DOI] [PubMed] [Google Scholar]
- 32.Rekers NH, Zegers CM, Yaromina A, et al. Combination of radiotherapy with the immunocytokine L19-IL2: additive effect in a NK cell dependent tumour model. Radiother Oncol. 2015;116(3):438–442. doi: 10.1016/j.radonc.2015.06.019. [DOI] [PubMed] [Google Scholar]
- 33.Zegers CM, Rekers NH, Quaden DH, et al. Radiotherapy combined with the immunocytokine L19-IL2 provides long-lasting antitumor effects. Clin Cancer Res. 2015;21(5):1151–1160. doi: 10.1158/1078-0432.CCR-14-2676. [DOI] [PubMed] [Google Scholar]
- 34.Spitaleri G, Berardi R, Pierantoni C, et al. Phase I/II study of the tumour-targeting human monoclonal antibody-cytokine fusion protein L19-TNF in patients with advanced solid tumours. J Cancer Res Clin Oncol. 2013;139(3):447–455. doi: 10.1007/s00432-012-1327-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1) Eur J Cancer. 2009;45(2):228–247. doi: 10.1016/j.ejca.2008.10.026. [DOI] [PubMed] [Google Scholar]
- 36.Frey K, Fiechter M, Schwager K, Belloni B, Barysch MJ, Neri D, Dummer R. Different patterns of fibronectin and tenascin-C splice variants expression in primary and metastatic melanoma lesions. Exp Dermatol. 2011;20(8):685–688. doi: 10.1111/j.1600-0625.2011.01314.x. [DOI] [PubMed] [Google Scholar]
- 37.Schwartzentruber DJ. Guidelines for the safe administration of high-dose interleukin-2. J Immunother. 2001;24(4):287–293. doi: 10.1097/00002371-200107000-00004. [DOI] [PubMed] [Google Scholar]
- 38.Ma C, Armstrong AW. Severe adverse events from the treatment of advanced melanoma: a systematic review of severe side effects associated with ipilimumab, vemurafenib, interferon alfa-2b, dacarbazine and interleukin-2. J Dermatolog Treat. 2014;25(5):401–408. doi: 10.3109/09546634.2013.813897. [DOI] [PubMed] [Google Scholar]
- 39.Wu CF, Wang HM, Huang WK, Chang JW. Efficacy and safety of bio-chemotherapy with dacarbazine plus interleukin-2 in patients with unresectable malignant melanoma. Asia Pac J Clin Oncol. 2015;11(4):314–318. doi: 10.1111/ajco.12404. [DOI] [PubMed] [Google Scholar]
- 40.Erba PA, Sollini M, Orciuolo E, et al. Radioimmunotherapy with radretumab in patients with relapsed hematologic malignancies. J. Nuclear Med. 2012;53(6):922–927. doi: 10.2967/jnumed.111.101006. [DOI] [PubMed] [Google Scholar]
- 41.Poli GL, Bianchi C, Virotta G, et al. Radretumab radioimmunotherapy in patients with brain metastasis: a 124I-L19SIP dosimetric PET study. Cancer Immunol Res. 2013;1(2):134–143. doi: 10.1158/2326-6066. [DOI] [PubMed] [Google Scholar]
- 42.Halin C, Rondini S, Nilsson F, Berndt A, Kosmehl H, Zardi L, Neri D. Enhancement of the antitumor activity of interleukin-12 by targeted delivery to neovasculature. Nat Biotechnol. 2002;20(3):264–269. doi: 10.1038/nbt0302-264. [DOI] [PubMed] [Google Scholar]
- 43.Rodig SJ, Gusenleitner D, Jackson DG, et al. MHC proteins confer differential sensitivity to CTLA-4 and PD-1 blockade in untreated metastatic melanoma. Sci Transl Med. 2018 doi: 10.1126/scitranslmed.aar3342. [DOI] [PubMed] [Google Scholar]
- 44.Schliemann C, Gutbrodt KL, Kerkhoff A, et al. Targeting interleukin-2 to the bone marrow stroma for therapy of acute myeloid leukemia relapsing after allogeneic hematopoietic stem cell transplantation. Cancer Immunol Res. 2015;3(5):547–556. doi: 10.1158/2326-6066.CIR-14-0179. [DOI] [PubMed] [Google Scholar]
- 45.Murphy K, Weaver C. Janeway's immunobiology. 9. New York: Garland Science, Taylor & Francis Group, LLC; 2017. p. 823. [Google Scholar]
- 46.Robert C, Schachter J, Long GV, et al. Pembrolizumab versus ipilimumab in advanced melanoma. N Engl J Med. 2015;372(26):2521–2532. doi: 10.1056/NEJMoa1503093. [DOI] [PubMed] [Google Scholar]
- 47.Patel SP, Milton D, Milhem MM, et al. Sequential administration of high-dose interleukin-2 and ipilimumab in patients with metastatic melanoma. J Clin Oncol. 2016;34(15_suppl):e21041–e21041. doi: 10.1200/jco.2016.34.15_suppl.e21041. [DOI] [Google Scholar]
- 48.Klein C, Waldhauer I, Nicolini VG, et al. Cergutuzumab amunaleukin (CEA-IL2v), a CEA-targeted IL-2 variant-based immunocytokine for combination cancer immunotherapy: overcoming limitations of aldesleukin and conventional IL-2-based immunocytokines. Oncoimmunology. 2017;6(3):e1277306. doi: 10.1080/2162402X.2016.1277306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Hutmacher C, Gonzalo Nunez N, Liuzzi AR, Becher B, Neri D. Targeted DELIVERY of IL2 to the tumor stroma potentiates the action of immune checkpoint inhibitors by preferential activation of NK and CD8(+) T cells. Cancer Immunol Res. 2019;7(4):572–583. doi: 10.1158/2326-6066.CIR-18-0566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.De Luca R, Neri D. Potentiation of PD-L1 blockade with a potency-matched dual cytokine-antibody fusion protein leads to cancer eradication in BALB/c-derived tumors but not in other mouse strains. Cancer Immunol Immunother. 2018;67(9):1381–1391. doi: 10.1007/s00262-018-2194-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Castellani P, Viale G, Dorcaratto A, Nicolo G, Kaczmarek J, Querze G, Zardi L. The fibronectin isoform containing the ED-B oncofetal domain: a marker of angiogenesis. Int J Cancer. 1994;59(5):612–618. doi: 10.1002/ijc.2910590507. [DOI] [PubMed] [Google Scholar]
- 52.Schliemann C, Wiedmer A, Pedretti M, Szczepanowski M, Klapper W, Neri D. Three clinical-stage tumor targeting antibodies reveal differential expression of oncofetal fibronectin and tenascin-C isoforms in human lymphoma. Leuk Res. 2009;33(12):1718–1722. doi: 10.1016/j.leukres.2009.06.025. [DOI] [PubMed] [Google Scholar]
- 53.Sauer S, Erba PA, Petrini M, et al. Expression of the oncofetal ED-B-containing fibronectin isoform in hematologic tumors enables ED-B-targeted 131I-L19SIP radioimmunotherapy in Hodgkin lymphoma patients. Blood. 2009;113(10):2265–2274. doi: 10.1182/blood-2008-06-160416. [DOI] [PubMed] [Google Scholar]
- 54.Kaspar M, Zardi L, Neri D. Fibronectin as target for tumor therapy. Int J Cancer. 2006;118(6):1331–1339. doi: 10.1002/ijc.21677. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.